Phosphorylation Signals Downstream of Dopamine Receptors in Emotional Behaviors: Association with Preference and Avoidance
Abstract
1. Introduction
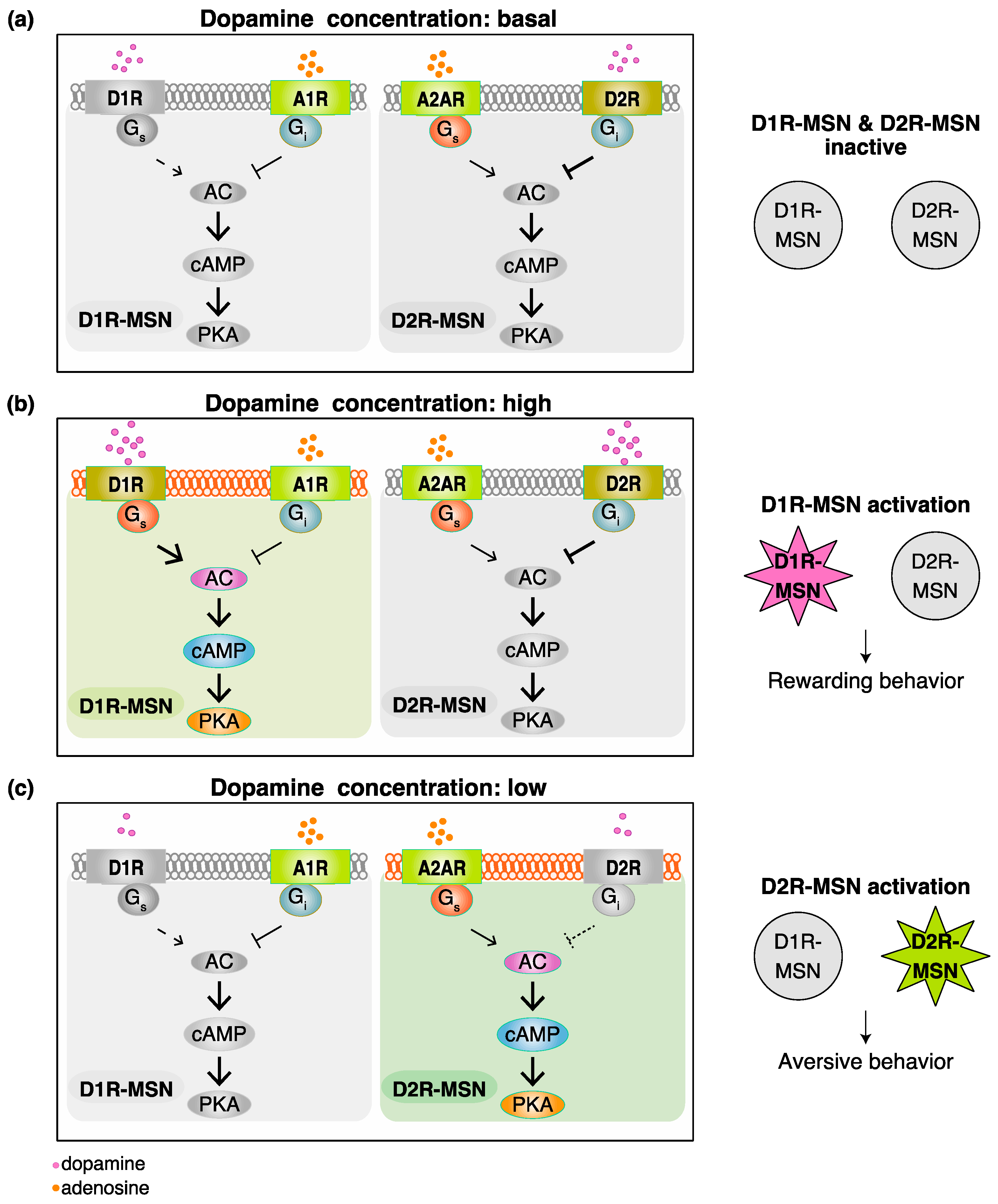
2. Functional Model for Dopamine D1 Receptors Expressing Medium Spiny Neurons (D1R-MSN) and Dopamine D2 Receptors Expressing Medium Spiny Neurons (D2R-MSN) in Rewarding and Aversive Behaviors
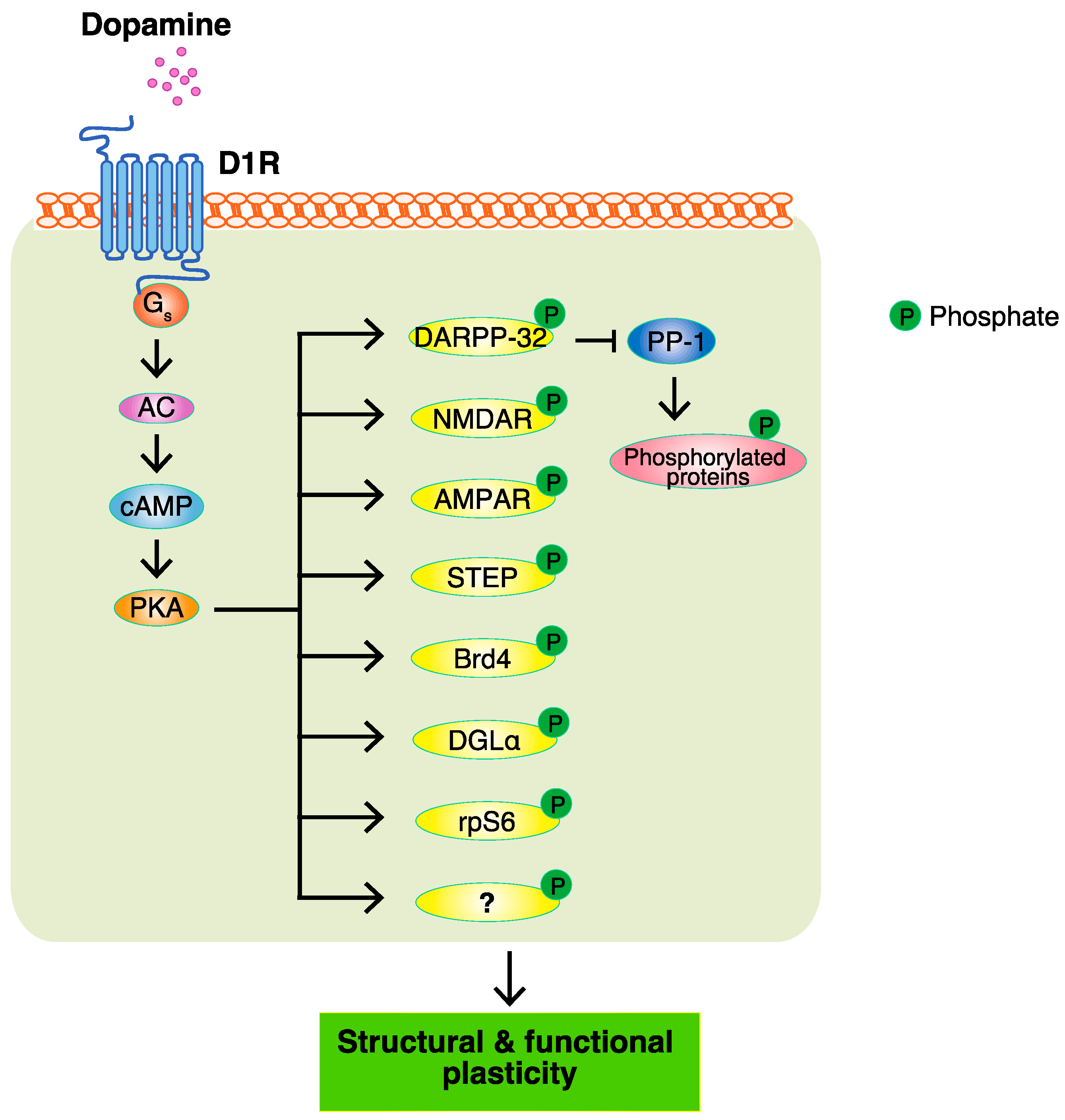
3. PKA and PKA Substrates
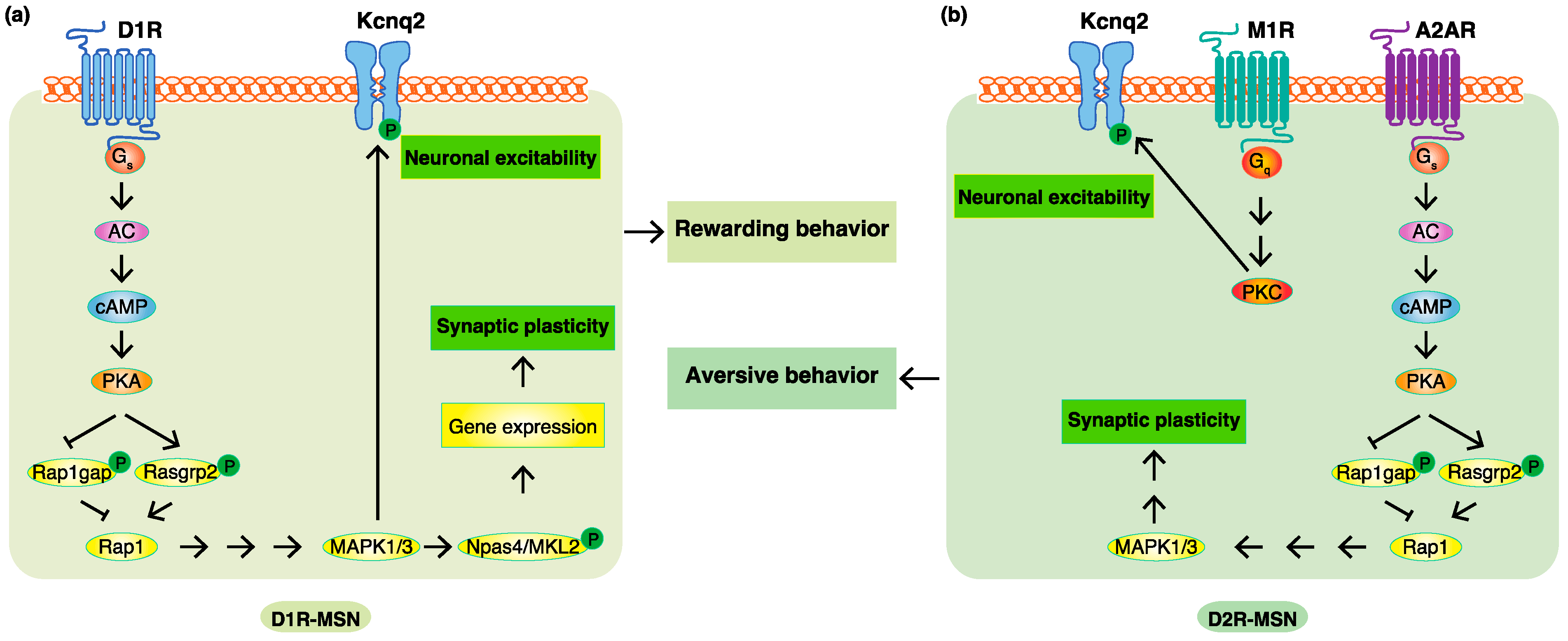
4. Phosphoproteomic Analyses Revealed New Phosphorylation Signals Downstream of Dopamine Receptors in Emotional Behaviors
5. Novel Database of Kinase-Associated Neural Protein Phosphorylation in the Brain: Kinase-Associated Neural PHOspho-Signaling (KANPHOS)
6. Actions of Dopaminergic and Adenosinergic Drugs in Neuropsychiatric Disorders
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yeragani, V.K.; Tancer, M.; Chokka, P.; Baker, G.B. Arvid Carlsson, and the story of dopamine. Indian J. Psychiatry 2010, 52, 87. [Google Scholar] [CrossRef] [PubMed]
- Perez-Fernandez, J.; Barandela, M.; Jimenez-Lopez, C. The Dopaminergic Control of Movement-Evolutionary Considerations. Int. J. Mol. Sci. 2021, 22, 11284. [Google Scholar] [CrossRef] [PubMed]
- Castillo Diaz, F.; Caffino, L.; Fumagalli, F. Bidirectional role of dopamine in learning and memory-active forgetting. Neurosci. Biobehav. Rev. 2021, 131, 953–963. [Google Scholar] [CrossRef] [PubMed]
- Watabe-Uchida, M.; Eshel, N.; Uchida, N. Neural Circuitry of Reward Prediction Error. Annu. Rev. Neurosci. 2017, 40, 373–394. [Google Scholar] [CrossRef]
- Engelhard, B.; Finkelstein, J.; Cox, J.; Fleming, W.; Jang, H.J.; Ornelas, S.; Koay, S.A.; Thiberge, S.Y.; Daw, N.D.; Tank, D.W. Specialized coding of sensory, motor and cognitive variables in VTA dopamine neurons. Nature 2019, 570, 509–513. [Google Scholar] [CrossRef]
- Latif, S.; Jahangeer, M.; Maknoon Razia, D.; Ashiq, M.; Ghaffar, A.; Akram, M.; El Allam, A.; Bouyahya, A.; Garipova, L.; Ali Shariati, M.; et al. Dopamine in Parkinson’s disease. Clin. Chim. Acta 2021, 522, 114–126. [Google Scholar] [CrossRef]
- Weinstein, J.J.; Chohan, M.O.; Slifstein, M.; Kegeles, L.S.; Moore, H.; Abi-Dargham, A. Pathway-Specific Dopamine Ab-normalities in Schizophrenia. Biol. Psychiatry 2017, 81, 31–42. [Google Scholar] [CrossRef]
- Wise, R.A.; Robble, M.A. Dopamine and Addiction. Annu. Rev. Psychol. 2020, 71, 79–106. [Google Scholar] [CrossRef] [PubMed]
- Wise, R.A.; Jordan, C.J. Dopamine, behavior, and addiction. J. Biomed. Sci. 2021, 28, 83. [Google Scholar] [CrossRef]
- Klein, M.O.; Battagello, D.S.; Cardoso, A.R.; Hauser, D.N.; Bittencourt, J.C.; Correa, R.G. Dopamine: Functions, Signaling, and Association with Neurological Diseases. Cell. Mol. Neurobiol. 2019, 39, 31–59. [Google Scholar] [CrossRef] [PubMed]
- Belujon, P.; Grace, A.A. Dopamine System Dysregulation in Major Depressive Disorders. Int. J. Neuropsychopharmacol. 2017, 20, 1036–1046. [Google Scholar] [CrossRef] [PubMed]
- Mitchell, U.H.; Obray, J.D.; Hunsaker, E.; Garcia, B.T.; Clarke, T.J.; Hope, S.; Steffensen, S.C. Peripheral dopamine in rest-less legs syndrome. Front. Neurol. 2018, 9, 155. [Google Scholar] [CrossRef]
- Torrisi, S.A.; Leggio, G.M.; Drago, F.; Salomone, S. Therapeutic challenges of post-traumatic stress disorder: Focus on the dopaminergic system. Front. Pharmacol. 2019, 10, 404. [Google Scholar] [CrossRef]
- Lammel, S.; Lim, B.K.; Ran, C.; Huang, K.W.; Betley, M.J.; Tye, K.M.; Deisseroth, K.; Malenka, R.C. Input-specific control of reward and aversion in the ventral tegmental area. Nature 2012, 491, 212–217. [Google Scholar] [CrossRef] [PubMed]
- Sun, F.; Zeng, J.; Jing, M.; Zhou, J.; Feng, J.; Owen, S.F.; Luo, Y.; Li, F.; Wang, H.; Yamaguchi, T.; et al. A Genetically Encoded Fluorescent Sensor Enables Rapid and Specific Detection of Dopamine in Flies, Fish, and Mice. Cell 2018, 174, 481–496.e419. [Google Scholar] [CrossRef] [PubMed]
- Rehman, I.; Mahabadi, N.; Sanvictores, T.; Rehman, C.I. Classical conditioning. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2017. [Google Scholar]
- Flagel, S.B.; Clark, J.J.; Robinson, T.E.; Mayo, L.; Czuj, A.; Willuhn, I.; Akers, C.A.; Clinton, S.M.; Phillips, P.E.; Akil, H. A selective role for dopamine in stimulus-reward learning. Nature 2011, 469, 53–57. [Google Scholar] [CrossRef] [PubMed]
- Van Zessen, R.; Flores-Dourojeanni, J.P.; Eekel, T.; van den Reijen, S.; Lodder, B.; Omrani, A.; Smidt, M.P.; Ramakers, G.M.J.; van der Plasse, G.; Stuber, G.D.; et al. Cue and Reward Evoked Dopamine Activity Is Necessary for Maintaining Learned Pavlovian Associations. J. Neurosci. 2021, 41, 5004–5014. [Google Scholar] [CrossRef]
- Morrens, J.; Aydin, C.; Janse van Rensburg, A.; Esquivelzeta Rabell, J.; Haesler, S. Cue-Evoked Dopamine Promotes Con-ditioned Responding during Learning. Neuron 2020, 106, 142–153.e147. [Google Scholar] [CrossRef] [PubMed]
- Danjo, T.; Yoshimi, K.; Funabiki, K.; Yawata, S.; Nakanishi, S. Aversive behavior induced by optogenetic inactivation of ventral tegmental area dopamine neurons is mediated by dopamine D2 receptors in the nucleus accumbens. Proc. Natl. Acad. Sci. USA 2014, 111, 6455–6460. [Google Scholar] [CrossRef]
- Tan, K.R.; Yvon, C.; Turiault, M.; Mirzabekov, J.J.; Doehner, J.; Labouebe, G.; Deisseroth, K.; Tye, K.M.; Luscher, C. GABA neurons of the VTA drive conditioned place aversion. Neuron 2012, 73, 1173–1183. [Google Scholar] [CrossRef] [PubMed]
- Bhatia, A.; Saadabadi, A. Biochemistry, Dopamine Receptors. In StatPearls [Internet]; StatPearls Publishing: Treasure Island, FL, USA, 2019. [Google Scholar]
- Lefkowitz, R.J.; Shenoy, S.K. Transduction of receptor signals by β-arrestins. Science 2005, 308, 512–517. [Google Scholar] [CrossRef]
- Allen, J.A.; Yost, J.M.; Setola, V.; Chen, X.; Sassano, M.F.; Chen, M.; Peterson, S.; Yadav, P.N.; Huang, X.P.; Feng, B.; et al. Discovery of beta-arrestin-biased dopamine D2 ligands for probing signal transduction pathways essential for antipsychotic efficacy. Proc. Natl. Acad. Sci. USA 2011, 108, 18488–18493. [Google Scholar] [CrossRef] [PubMed]
- Donthamsetti, P.; Gallo, E.F.; Buck, D.C.; Stahl, E.L.; Zhu, Y.; Lane, J.R.; Bohn, L.M.; Neve, K.A.; Kellendonk, C.; Javitch, J.A. Arrestin recruitment to dopamine D2 receptor mediates locomotion but not incentive motivation. Mol. Psychiatry 2020, 25, 2086–2100. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Nan, J.; Lan, Y. The nucleus accumbens: A common target in the comorbidity of depression and addiction. Front. Neural Circuits 2020, 14, 37. [Google Scholar] [CrossRef] [PubMed]
- Castro, D.C.; Bruchas, M.R. A motivational and neuropeptidergic hub: Anatomical and functional diversity within the nucleus accumbens shell. Neuron 2019, 102, 529–552. [Google Scholar] [CrossRef] [PubMed]
- Hikida, T.; Kimura, K.; Wada, N.; Funabiki, K.; Nakanishi, S. Distinct roles of synaptic transmission in direct and indi-rect striatal pathways to reward and aversive behavior. Neuron 2010, 66, 896–907. [Google Scholar] [CrossRef] [PubMed]
- Hart, A.S.; Rutledge, R.B.; Glimcher, P.W.; Phillips, P.E. Phasic dopamine release in the rat nucleus accumbens symmetri-cally encodes a reward prediction error term. J. Neurosci. 2014, 34, 698–704. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Kershberg, L.; Wang, J.; Schneeberger, S.; Kaeser, P.S. Dopamine Secretion Is Mediated by Sparse Active Zone-like Release Sites. Cell 2018, 172, 706–718.e715. [Google Scholar] [CrossRef]
- Liu, C.; Kaeser, P.S. Mechanisms and regulation of dopamine release. Curr. Opin. Neurobiol. 2019, 57, 46–53. [Google Scholar] [CrossRef]
- Martel, J.C.; Gatti McArthur, S. Dopamine Receptor Subtypes, Physiology and Pharmacology: New Ligands and Con-cepts in Schizophrenia. Front. Pharmacol. 2020, 11, 1003. [Google Scholar] [CrossRef] [PubMed]
- Marcellino, D.; Kehr, J.; Agnati, L.F.; Fuxe, K. Increased affinity of dopamine for D(2) -like versus D(1) -like receptors. Relevance for volume transmission in interpreting PET findings. Synapse 2012, 66, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Cadoni, C.; Solinas, M.; Di Chiara, G. Psychostimulant sensitization: Differential changes in accumbal shell and core dopamine. Eur. J. Pharmacol. 2000, 388, 69–76. [Google Scholar] [CrossRef]
- Yuen, J.; Goyal, A.; Rusheen, A.E.; Kouzani, A.Z.; Berk, M.; Kim, J.H.; Tye, S.J.; Blaha, C.D.; Bennet, K.E.; Jang, D.P.; et al. Cocaine-Induced Changes in Tonic Dopamine Concentrations Measured Using Multiple-Cyclic Square Wave Voltammetry in vivo. Front. Pharmacol. 2021, 12, 705254. [Google Scholar] [CrossRef]
- Oleson, E.B.; Gentry, R.N.; Chioma, V.C.; Cheer, J.F. Subsecond dopamine release in the nucleus accumbens predicts con-ditioned punishment and its successful avoidance. J. Neurosci. 2012, 32, 14804–14808. [Google Scholar] [CrossRef] [PubMed]
- Borea, P.A.; Gessi, S.; Merighi, S.; Vincenzi, F.; Varani, K. Pharmacology of adenosine receptors: The state of the art. Physiol. Rev. 2018, 98, 1591–1625. [Google Scholar] [CrossRef]
- Dunwiddie, T.V.; Masino, S.A. The role and regulation of adenosine in the central nervous system. Annu. Rev. Neurosci. 2001, 24, 31–55. [Google Scholar] [CrossRef]
- Zhang, X.; Nagai, T.; Ahammad, R.U.; Kuroda, K.; Nakamuta, S.; Nakano, T.; Yukinawa, N.; Funahashi, Y.; Yamahashi, Y.; Amano, M.; et al. Balance between dopamine and adenosine signals regulates the PKA/Rap1 pathway in striatal me-dium spiny neurons. Neurochem. Int. 2019, 122, 8–18. [Google Scholar] [CrossRef]
- De Jong, J.W.; Afjei, S.A.; Pollak Dorocic, I.; Peck, J.R.; Liu, C.; Kim, C.K.; Tian, L.; Deisseroth, K.; Lammel, S. A Neural Cir-cuit Mechanism for Encoding Aversive Stimuli in the Mesolimbic Dopamine System. Neuron 2019, 101, 133–151.e137. [Google Scholar] [CrossRef]
- Beninger, R.J.; Nakonechny, P.L.; Savina, I. cAMP-dependent protein kinase and reward-related learning: In-tra-accumbens Rp-cAMPS blocks amphetamine-produced place conditioning in rats. Psychopharmacology 2003, 170, 23–32. [Google Scholar] [CrossRef]
- Nagai, T.; Nakamuta, S.; Kuroda, K.; Nakauchi, S.; Nishioka, T.; Takano, T.; Zhang, X.; Tsuboi, D.; Funahashi, Y.; Nakano, T.; et al. Phosphoproteomics of the Dopamine Pathway Enables Discovery of Rap1 Activation as a Reward Signal In Vivo. Neuron 2016, 89, 550–565. [Google Scholar] [CrossRef]
- Lin, Y.H.; Yamahashi, Y.; Kuroda, K.; Faruk, M.O.; Zhang, X.; Yamada, K.; Yamanaka, A.; Nagai, T.; Kaibuchi, K. Ac-cumbal D2R-medium spiny neurons regulate aversive behaviors through PKA-Rap1 pathway. Neurochem. Int. 2021, 143, 104935. [Google Scholar] [CrossRef] [PubMed]
- Greengard, P. The neurobiology of slow synaptic transmission. Science 2001, 294, 1024–1030. [Google Scholar] [CrossRef]
- Girault, J.A.; Nairn, A.C. DARPP-32 40 years later. Adv. Pharmacol. 2021, 90, 67–87. [Google Scholar] [CrossRef] [PubMed]
- Zachariou, V.; Benoit-Marand, M.; Allen, P.B.; Ingrassia, P.; Fienberg, A.A.; Gonon, F.; Greengard, P.; Picciotto, M.R. Re-duction of cocaine place preference in mice lacking the protein phosphatase 1 inhibitors DARPP 32 or Inhibitor 1. Biol. Psychiatry 2002, 51, 612–620. [Google Scholar] [CrossRef]
- Diering, G.H.; Huganir, R.L. The AMPA receptor code of synaptic plasticity. Neuron 2018, 100, 314–329. [Google Scholar] [CrossRef] [PubMed]
- Stroebel, D.; Paoletti, P. Architecture and function of NMDA receptors: An evolutionary perspective. J. Physiol. 2021, 599, 2615–2638. [Google Scholar] [CrossRef] [PubMed]
- Man, H.Y.; Sekine-Aizawa, Y.; Huganir, R.L. Regulation of α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic acid receptor trafficking through PKA phosphorylation of the Glu receptor 1 subunit. Proc. Natl. Acad. Sci. USA 2007, 104, 3579–3584. [Google Scholar] [CrossRef] [PubMed]
- Mead, A.N.; Stephens, D.N. Selective disruption of stimulus-reward learning in glutamate receptor gria1 knock-out mice. J. Neurosci. 2003, 23, 1041–1048. [Google Scholar] [CrossRef]
- Mead, A.N.; Brown, G.; Le Merrer, J.; Stephens, D.N. Effects of deletion of gria1 or gria2 genes encoding glutamatergic AMPA-receptor subunits on place preference conditioning in mice. Psychopharmacology 2005, 179, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Snyder, G.L.; Yokakura, H.; Picciotto, M.R.; Nairn, A.C.; Lombroso, P.J. The Dopamine/D1 receptor mediates the phosphorylation and inactivation of the protein tyrosine phosphatase STEP via a PKA-dependent pathway. J. Neurosci. 2000, 20, 5630–5638. [Google Scholar] [CrossRef] [PubMed]
- Snyder, E.M.; Nong, Y.; Almeida, C.G.; Paul, S.; Moran, T.; Choi, E.Y.; Nairn, A.C.; Salter, M.W.; Lombroso, P.J.; Gouras, G.K.; et al. Regulation of NMDA receptor trafficking by amyloid-beta. Nat. Neurosci. 2005, 8, 1051–1058. [Google Scholar] [CrossRef]
- Sikora, M.; Tokarski, K.; Bobula, B.; Zajdel, J.; Jastrzebska, K.; Cieslak, P.E.; Zygmunt, M.; Sowa, J.; Smutek, M.; Kaminska, K.; et al. NMDA Receptors on Dopaminoceptive Neurons Are Essential for Drug-Induced Conditioned Place Preference. eNeuro 2016, 3, e0084-15. [Google Scholar] [CrossRef] [PubMed]
- Nestler, E.J. Molecular basis of long-term plasticity underlying addiction. Nat. Rev. Neurosci. 2001, 2, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Hyman, S.E.; Malenka, R.C. Addiction and the brain: The neurobiology of compulsion and its persistence. Nat. Rev. Neurosci. 2001, 2, 695–703. [Google Scholar] [CrossRef]
- Robinson, T.E.; Kolb, B. Alterations in the morphology of dendrites and dendritic spines in the nucleus accumbens and prefrontal cortex following repeated treatment with amphetamine or cocaine. Eur. J. Neurosci. 1999, 11, 1598–1604. [Google Scholar] [CrossRef] [PubMed]
- Dash, P.; Karl, K.; Colicos, M.; Prywes, R.; Kandel, E. cAMP response element-binding protein is activated by Ca2+/calmodulin-as well as cAMP-dependent protein kinase. Proc. Natl. Acad. Sci. USA 1991, 88, 5061–5065. [Google Scholar] [CrossRef]
- Dudman, J.T.; Eaton, M.E.; Rajadhyaksha, A.; Macías, W.; Taher, M.; Barczak, A.; Kameyama, K.; Huganir, R.; Konradi, C. Dopamine D1 receptors mediate CREB phosphorylation via phosphorylation of the NMDA receptor at Ser897–NR1. J. Neurochem. 2003, 87, 922–934. [Google Scholar] [CrossRef]
- Bilbao, A.; Rieker, C.; Cannella, N.; Parlato, R.; Golda, S.; Piechota, M.; Korostynski, M.; Engblom, D.; Przewlocki, R.; Schutz, G.; et al. CREB activity in dopamine D1 receptor expressing neurons regulates cocaine-induced behavioral effects. Front. Behav. Neurosci. 2014, 8, 212. [Google Scholar] [CrossRef]
- Savell, K.E.; Tuscher, J.J.; Zipperly, M.E.; Duke, C.G.; Phillips, R.A., III; Bauman, A.J.; Thukral, S.; Sultan, F.A.; Goska, N.A.; Ianov, L. A dopamine-induced gene expression signature regulates neuronal function and cocaine response. Sci. Adv. 2020, 6, eaba4221. [Google Scholar] [CrossRef]
- Pulipparacharuvil, S.; Renthal, W.; Hale, C.F.; Taniguchi, M.; Xiao, G.; Kumar, A.; Russo, S.J.; Sikder, D.; Dewey, C.M.; Davis, M.M. Cocaine regulates MEF2 to control synaptic and behavioral plasticity. Neuron 2008, 59, 621–633. [Google Scholar] [CrossRef]
- Russo, S.J.; Wilkinson, M.B.; Mazei-Robison, M.S.; Dietz, D.M.; Maze, I.; Krishnan, V.; Renthal, W.; Graham, A.; Birnbaum, S.G.; Green, T.A. Nuclear factor κB signaling regulates neuronal morphology and cocaine reward. J. Neurosci. 2009, 29, 3529–3537. [Google Scholar] [CrossRef]
- Kennedy, P.J.; Feng, J.; Robison, A.; Maze, I.; Badimon, A.; Mouzon, E.; Chaudhury, D.; Damez-Werno, D.M.; Haggarty, S.J.; Han, M.-H. Class I HDAC inhibition blocks cocaine-induced plasticity by targeted changes in histone methylation. Nat. Neurosci. 2013, 16, 434–440. [Google Scholar] [CrossRef]
- Levine, A.A.; Guan, Z.; Barco, A.; Xu, S.; Kandel, E.R.; Schwartz, J.H. CREB-binding protein controls response to cocaine by acetylating histones at the fosB promoter in the mouse striatum. Proc. Natl. Acad. Sci. USA 2005, 102, 19186–19191. [Google Scholar] [CrossRef]
- Maze, I.; Covington III, H.E.; Dietz, D.M.; LaPlant, Q.; Renthal, W.; Russo, S.J.; Mechanic, M.; Mouzon, E.; Neve, R.L.; Haggarty, S.J. Essential role of the histone methyltransferase G9a in cocaine-induced plasticity. Science 2010, 327, 213–216. [Google Scholar] [CrossRef]
- Park, H.-Y.; Ryu, Y.-K.; Kim, Y.-H.; Park, T.-S.; Go, J.; Hwang, J.H.; Choi, D.-H.; Rhee, M.; Lee, C.-H.; Kim, K.-S. Gadd45β ameliorates L-DOPA-induced dyskinesia in a Parkinson’s disease mouse model. Neurobiol. Dis. 2016, 89, 169–179. [Google Scholar] [CrossRef]
- Schroeder, F.A.; Penta, K.L.; Matevossian, A.; Jones, S.R.; Konradi, C.; Tapper, A.R.; Akbarian, S. Drug-induced activation of dopamine D1 receptor signaling and inhibition of class I/II histone deacetylase induce chromatin remodeling in re-ward circuitry and modulate cocaine-related behaviors. Neuropsychopharmacology 2008, 33, 2981–2992. [Google Scholar] [CrossRef]
- Zipperly, M.E.; Sultan, F.A.; Graham, G.-E.; Brane, A.C.; Simpkins, N.A.; Carullo, N.V.; Ianov, L.; Day, J.J. Regulation of dopamine-dependent transcription and cocaine action by Gadd45b. Neuropsychopharmacology 2021, 46, 709–720. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.Y.; Lee, A.Y.; Lai, H.T.; Zhang, H.; Chiang, C.M. Phospho switch triggers Brd4 chromatin binding and activator recruitment for gene-specific targeting. Mol. Cell 2013, 49, 843–857. [Google Scholar] [CrossRef] [PubMed]
- Martin, R.D.; Sun, Y.; MacKinnon, S.; Cuccia, L.; Pagé, V.; Hébert, T.E.; Tanny, J.C. Differential activation of P-TEFb com-plexes in the development of cardiomyocyte hypertrophy following activation of distinct G protein-coupled receptors. Mol. Cell. Biol. 2020, 40, e00048-20. [Google Scholar] [CrossRef]
- Van de Velde, S.; Wiater, E.; Tran, M.; Hwang, Y.; Cole, P.A.; Montminy, M. CREB promotes beta cell gene expression by targeting its coactivators to tissue-specific enhancers. Mol. Cell. Biol. 2019, 39, e00200-19. [Google Scholar] [CrossRef]
- Jones-Tabah, J.; Martin, R.D.; Chen, J.J.; Tanny, J.C.; Clarke, P.B.S.; Hebert, T.E. A role for BET proteins in regulating basal, dopamine-induced and cAMP/PKA-dependent transcription in rat striatal neurons. Cell. Signal. 2022, 91, 110226. [Google Scholar] [CrossRef]
- Babigian, C.J.; Wiedner, H.J.; Wahlestedt, C.; Sartor, G.C. JQ1 attenuates psychostimulant- but not opioid-induced conditioned place preference. Behav. Brain Res. 2022, 418, 113644. [Google Scholar] [CrossRef]
- Sartor, G.C.; Powell, S.K.; Brothers, S.P.; Wahlestedt, C. Epigenetic Readers of Lysine Acetylation Regulate Co-caine-Induced Plasticity. J. Neurosci. 2015, 35, 15062–15072. [Google Scholar] [CrossRef]
- Bilbao, A.; Neuhofer, D.; Sepers, M.; Wei, S.P.; Eisenhardt, M.; Hertle, S.; Lassalle, O.; Ramos-Uriarte, A.; Puente, N.; Ler-ner, R.; et al. Endocannabinoid LTD in Accumbal D1 Neurons Mediates Reward-Seeking Behavior. iScience 2020, 23, 100951. [Google Scholar] [CrossRef]
- Shonesy, B.C.; Stephenson, J.R.; Marks, C.R.; Colbran, R.J. Cyclic AMP-dependent protein kinase and D1 dopamine recep-tors regulate diacylglycerol lipase-alpha and synaptic 2-arachidonoyl glycerol signaling. J. Neurochem. 2020, 153, 334–345. [Google Scholar] [CrossRef]
- Ruvinsky, I.; Meyuhas, O. Ribosomal protein S6 phosphorylation: From protein synthesis to cell size. Trends Biochem. Sci. 2006, 31, 342–348. [Google Scholar] [CrossRef] [PubMed]
- Meyuhas, O. Physiological roles of ribosomal protein S6: One of its kind. Int. Rev. Cell Mol. Biol. 2008, 268, 1–37. [Google Scholar]
- Roux, P.P.; Shahbazian, D.; Vu, H.; Holz, M.K.; Cohen, M.S.; Taunton, J.; Sonenberg, N.; Blenis, J. RAS/ERK signaling promotes site-specific ribosomal protein S6 phosphorylation via RSK and stimulates cap-dependent translation. J. Biol. Chem. 2007, 282, 14056–14064. [Google Scholar] [CrossRef]
- Yano, T.; Ferlito, M.; Aponte, A.; Kuno, A.; Miura, T.; Murphy, E.; Steenbergen, C. Pivotal role of mTORC2 and involve-ment of ribosomal protein S6 in cardioprotective signaling. Circ. Res. 2014, 114, 1268–1280. [Google Scholar] [CrossRef]
- Moore, C.E.; Xie, J.; Gomez, E.; Herbert, T.P. Identification of cAMP-dependent kinase as a third in vivo ribosomal pro-tein S6 kinase in pancreatic β-cells. J. Mol. Biol. 2009, 389, 480–494. [Google Scholar] [CrossRef]
- Valjent, E.; Bertran-Gonzalez, J.; Bowling, H.; Lopez, S.; Santini, E.; Matamales, M.; Bonito-Oliva, A.; Hervé, D.; Hoeffer, C.; Klann, E. Haloperidol regulates the state of phosphorylation of ribosomal protein S6 via activation of PKA and phosphorylation of DARPP-32. Neuropsychopharmacology 2011, 36, 2561–2570. [Google Scholar] [CrossRef]
- Biever, A.; Puighermanal, E.; Nishi, A.; David, A.; Panciatici, C.; Longueville, S.; Xirodimas, D.; Gangarossa, G.; Meyuhas, O.; Herve, D.; et al. PKA-dependent phosphorylation of ribosomal protein S6 does not correlate with translation efficiency in striatonigral and striatopallidal medium-sized spiny neurons. J. Neurosci. 2015, 35, 4113–4130. [Google Scholar] [CrossRef]
- Nagai, T.; Yoshimoto, J.; Kannon, T.; Kuroda, K.; Kaibuchi, K. Phosphorylation Signals in Striatal Medium Spiny Neurons. Trends Pharmacol. Sci. 2016, 37, 858–871. [Google Scholar] [CrossRef]
- McAvoy, T.; Zhou, M.M.; Greengard, P.; Nairn, A.C. Phosphorylation of Rap1GAP, a striatally enriched protein, by pro-tein kinase A controls Rap1 activity and dendritic spine morphology. Proc. Natl. Acad. Sci. USA 2009, 106, 3531–3536. [Google Scholar] [CrossRef]
- Sweatt, J.D. The neuronal MAP kinase cascade: A biochemical signal integration system subserving synaptic plasticity and memory. J. Neurochem. 2001, 76, 1–10. [Google Scholar] [CrossRef]
- Delmas, P.; Brown, D.A. Pathways modulating neural KCNQ/M (Kv7) potassium channels. Nat. Rev. Neurosci. 2005, 6, 850–862. [Google Scholar] [CrossRef]
- Tsuboi, D.; Otsuka, T.; Shimomura, T.; Faruk, M.O.; Yamahashi, Y.; Amano, M.; Funahashi, Y.; Kuroda, K.; Nishioka, T.; Kobayashi, K.; et al. Dopamine drives neuronal excitability via KCNQ channel phosphorylation for reward behavior. Cell Rep. 2022, 40, 111309. [Google Scholar] [CrossRef]
- Faruk, M.O.; Tsuboi, D.; Yamahashi, Y.; Funahashi, Y.; Lin, Y.H.; Ahammad, R.U.; Hossen, E.; Amano, M.; Nishioka, T.; Tzingounis, A.V.; et al. Muscarinic signaling regulates voltage-gated potassium channel KCNQ2 phosphorylation in the nucleus accumbens via protein kinase C for aversive learning. J. Neurochem. 2022, 160, 325–341. [Google Scholar] [CrossRef]
- Funahashi, Y.; Ariza, A.; Emi, R.; Xu, Y.; Shan, W.; Suzuki, K.; Kozawa, S.; Ahammad, R.U.; Wu, M.; Takano, T.; et al. Phosphorylation of Npas4 by MAPK Regulates Reward-Related Gene Expression and Behaviors. Cell Rep. 2019, 29, 3235–3252.e9. [Google Scholar] [CrossRef]
- Fu, J.; Guo, O.; Zhen, Z.; Zhen, J. Essential Functions of the Transcription Factor Npas4 in Neural Circuit Development, Plasticity, and Diseases. Front. Neurosci. 2020, 14, 603373. [Google Scholar] [CrossRef]
- Kalita, K.; Kuzniewska, B.; Kaczmarek, L. MKLs: Co-factors of serum response factor (SRF) in neuronal responses. Int. J. Biochem. Cell Biol. 2012, 44, 1444–1447. [Google Scholar] [CrossRef]
- Cahill, M.E.; Walker, D.M.; Gancarz, A.M.; Wang, Z.J.; Lardner, C.K.; Bagot, R.C.; Neve, R.L.; Dietz, D.M.; Nestler, E.J. The dendritic spine morphogenic effects of repeated cocaine use occur through the regulation of serum response factor signaling. Mol. Psychiatry 2018, 23, 1474–1486. [Google Scholar] [CrossRef]
- Vialou, V.; Feng, J.; Robison, A.J.; Ku, S.M.; Ferguson, D.; Scobie, K.N.; Mazei-Robison, M.S.; Mouzon, E.; Nestler, E.J. Se-rum response factor and cAMP response element binding protein are both required for cocaine induction of DeltaFosB. J. Neurosci. 2012, 32, 7577–7584. [Google Scholar] [CrossRef]
- Ariza, A.; Funahashi, Y.; Kozawa, S.; Omar Faruk, M.; Nagai, T.; Amano, M.; Kaibuchi, K. Dynamic subcellular locali-zation and transcription activity of the SRF cofactor MKL2 in the striatum are regulated by MAPK. J. Neurochem. 2021, 157, 1774–1788. [Google Scholar] [CrossRef] [PubMed]
- Amano, M.; Hamaguchi, T.; Shohag, M.H.; Kozawa, K.; Kato, K.; Zhang, X.; Yura, Y.; Matsuura, Y.; Kataoka, C.; Nishioka, T.; et al. Kinase-interacting substrate screening is a novel method to identify kinase substrates. J. Cell Biol. 2015, 209, 895–912. [Google Scholar] [CrossRef] [PubMed]
- Nishioka, T.; Amano, M.; Funahashi, Y.; Tsuboi, D.; Yamahashi, Y.; Kaibuchi, K. In Vivo Identification of Protein Kinase Substrates by Kinase-Oriented Substrate Screening (KIOSS). Curr. Protoc. Chem. Biol. 2019, 11, e60. [Google Scholar] [CrossRef] [PubMed]
- Ahammad, R.U.; Nishioka, T.; Yoshimoto, J.; Kannon, T.; Amano, M.; Funahashi, Y.; Tsuboi, D.; Faruk, M.O.; Yamahashi, Y.; Yamada, K.; et al. KANPHOS: A Database of Kinase-Associated Neural Protein Phosphorylation in the Brain. Cells 2021, 11, 47. [Google Scholar] [CrossRef] [PubMed]
- Connolly, B.S.; Lang, A.E. Pharmacological treatment of Parkinson disease: A review. JAMA 2014, 311, 1670–1683. [Google Scholar] [CrossRef]
- Ellis, J.M.; Fell, M.J. Current approaches to the treatment of Parkinson’s Disease. Bioorg. Med. Chem. Lett. 2017, 27, 4247–4255. [Google Scholar] [CrossRef]
- Zheng, J.; Zhang, X.; Zhen, X. Development of adenosine A2A receptor antagonists for the treatment of Parkinson’s disease: A recent update and challenge. ACS Chem. Neurosci. 2018, 10, 783–791. [Google Scholar] [CrossRef]
- Jenner, P.; Mori, A.; Aradi, S.D.; Hauser, R.A. Istradefylline–a first generation adenosine A2A antagonist for the treat-ment of Parkinson’s disease. Expert Rev. Neurother. 2021, 21, 317–333. [Google Scholar] [CrossRef]
- Pinna, A. Adenosine A2A receptor antagonists in Parkinson’s disease: Progress in clinical trials from the newly ap-proved istradefylline to drugs in early development and those already discontinued. CNS Drugs 2014, 28, 455–474. [Google Scholar] [CrossRef] [PubMed]
- Faraone, S.V. The pharmacology of amphetamine and methylphenidate: Relevance to the neurobiology of atten-tion-deficit/hyperactivity disorder and other psychiatric comorbidities. Neurosci. Biobehav. Rev. 2018, 87, 255–270. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Xiao, H.; Sun, H.; Zou, L.; Zhu, L.Q. Role of dopamine receptors in ADHD: A systematic meta-analysis. Mol. Neurobiol. 2012, 45, 605–620. [Google Scholar] [CrossRef]
- Kapur, S.; Remington, G. Dopamine D2 receptors and their role in atypical antipsychotic action: Still necessary and may even be sufficient. Biol. Psychiatry 2001, 50, 873–883. [Google Scholar] [CrossRef]
- Bellon, A.; Feuillet, V.; Cortez-Resendiz, A.; Mouaffak, F.; Kong, L.; Hong, L.E.; de Godoy, L.; Jay, T.M.; Hosmalin, A.; Krebs, M.O. Dopamine-induced pruning in monocyte-derived-neuronal-like cells (MDNCs) from patients with schizophrenia. Mol. Psychiatry 2022, 27, 2787–2802. [Google Scholar] [CrossRef]
- Ceraso, A.; Lin, J.J.; Schneider-Thoma, J.; Siafis, S.; Tardy, M.; Komossa, K.; Heres, S.; Kissling, W.; Davis, J.M.; Leucht, S. Maintenance treatment with antipsychotic drugs for schizophrenia. Cochrane Database Syst. Rev. 2020, 8, CD008016. [Google Scholar]
- Brancato, A.; Cavallaro, A.; Lavanco, G.; Plescia, F.; Cannizzaro, C. Reward-related limbic memory and stimulation of the cannabinoid system: An upgrade in value attribution? J. Psychopharmacol. 2018, 32, 204–214. [Google Scholar] [CrossRef] [PubMed]
- Brancato, A.; Castelli, V.; Lavanco, G.; Tringali, G.; Micale, V.; Kuchar, M.; D’Amico, C.; Pizzolanti, G.; Feo, S.; Canniz-zaro, C. Binge-like Alcohol Exposure in Adolescence: Behavioural, Neuroendocrine and Molecular Evidence of Abnormal Neuroplasticity... and Return. Biomedicines 2021, 9, 1161. [Google Scholar] [CrossRef]
- Plescia, F.; Brancato, A.; Marino, R.A.; Cannizzaro, C. Acetaldehyde as a drug of abuse: Insight into AM281 administra-tion on operant-conflict paradigm in rats. Front. Behav. Neurosci. 2013, 7, 64. [Google Scholar] [CrossRef]
- Brancato, A.; Castelli, V.; Lavanco, G.; D’Amico, C.; Feo, S.; Pizzolanti, G.; Kuchar, M.; Cannizzaro, C. Social stress under binge-like alcohol withdrawal in adolescence: Evidence of cannabidiol effect on maladaptive plasticity in rats. In Psychological Medicine; Cambridge University Press: Cambridge, UK, 2022; pp. 1–13. [Google Scholar]
- Cannizzaro, C.; Talani, G.; Brancato, A.; Mulas, G.; Spiga, S.; de Luca, M.A.; Sanna, A.; Marino, R.A.M.; Biggio, G.; Sanna, E.; et al. Dopamine Restores Limbic Memory Loss, Dendritic Spine Structure, and NMDAR-Dependent LTD in the Nucleus Accumbens of Alcohol-Withdrawn Rats. J. Neurosci. 2019, 39, 929–943. [Google Scholar] [CrossRef]
- Brancato, A.; Lavanco, G.; Cavallaro, A.; Plescia, F.; Cannizzaro, C. The use of the Emotional-Object Recognition as an assay to assess learning and memory associated to an aversive stimulus in rodents. J. Neurosci. Methods 2016, 274, 106–115. [Google Scholar] [CrossRef]
- Brancato, A.; Castelli, V.; Lavanco, G.; Marino, R.A.M.; Cannizzaro, C. In utero Δ9-tetrahydrocannabinol exposure con-fers vulnerability towards cognitive impairments and alcohol drinking in the adolescent offspring: Is there a role for neuro-peptide Y? J. Psychopharmacol. 2020, 34, 663–679. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zhang, X.; Tsuboi, D.; Funahashi, Y.; Yamahashi, Y.; Kaibuchi, K.; Nagai, T. Phosphorylation Signals Downstream of Dopamine Receptors in Emotional Behaviors: Association with Preference and Avoidance. Int. J. Mol. Sci. 2022, 23, 11643. https://doi.org/10.3390/ijms231911643
Zhang X, Tsuboi D, Funahashi Y, Yamahashi Y, Kaibuchi K, Nagai T. Phosphorylation Signals Downstream of Dopamine Receptors in Emotional Behaviors: Association with Preference and Avoidance. International Journal of Molecular Sciences. 2022; 23(19):11643. https://doi.org/10.3390/ijms231911643
Chicago/Turabian StyleZhang, Xinjian, Daisuke Tsuboi, Yasuhiro Funahashi, Yukie Yamahashi, Kozo Kaibuchi, and Taku Nagai. 2022. "Phosphorylation Signals Downstream of Dopamine Receptors in Emotional Behaviors: Association with Preference and Avoidance" International Journal of Molecular Sciences 23, no. 19: 11643. https://doi.org/10.3390/ijms231911643
APA StyleZhang, X., Tsuboi, D., Funahashi, Y., Yamahashi, Y., Kaibuchi, K., & Nagai, T. (2022). Phosphorylation Signals Downstream of Dopamine Receptors in Emotional Behaviors: Association with Preference and Avoidance. International Journal of Molecular Sciences, 23(19), 11643. https://doi.org/10.3390/ijms231911643




