Intestinal Epithelial STAT6 Activation Rescues the Defective Anti-Helminth Responses Caused by Ogt Deletion
Abstract
1. Introduction
2. Results
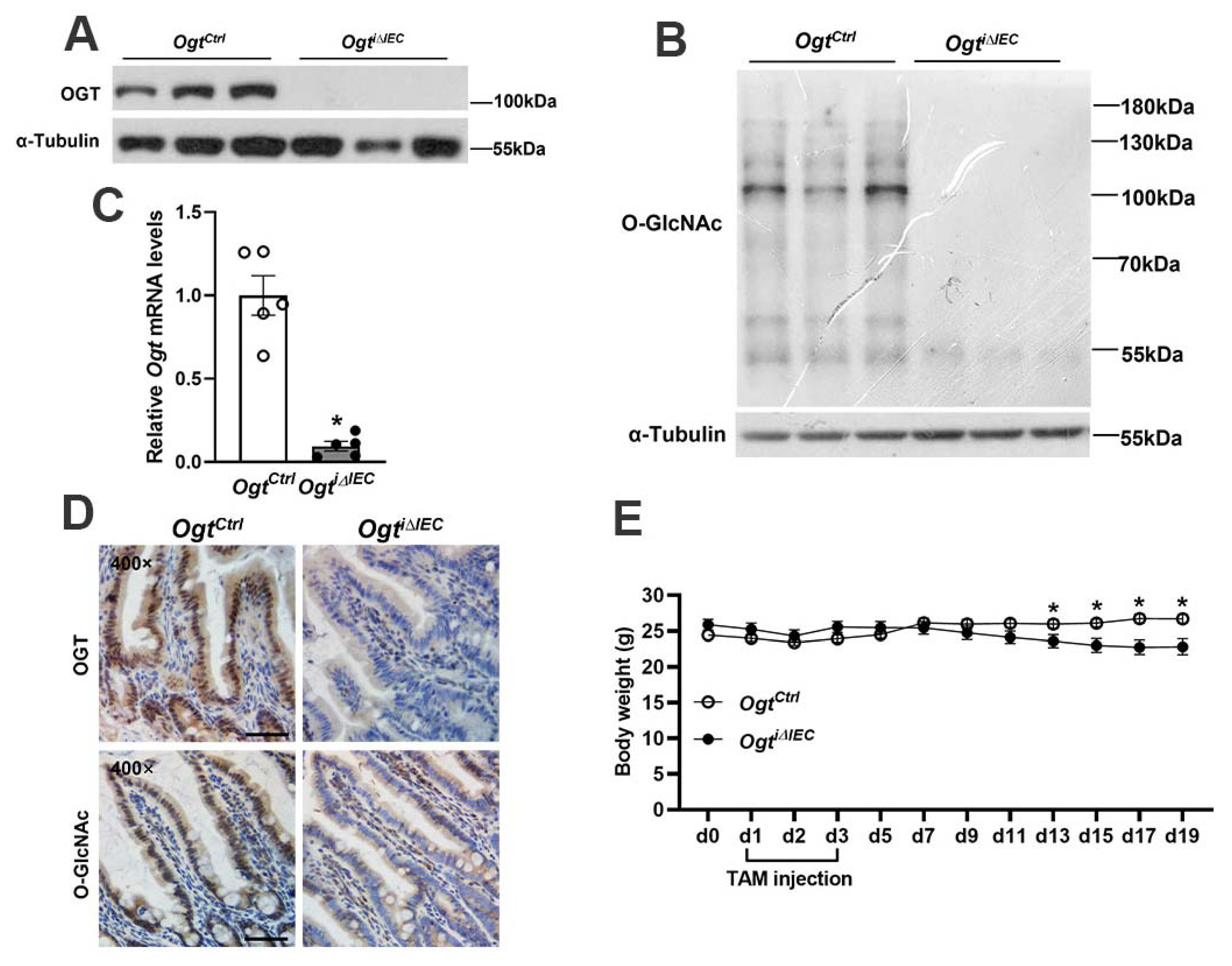
2.1. Generation of Inducible IEC-Specific Ogt Knockout Mice
2.2. Deletion of Ogt in IECs of Adult Mice Leads to Abnormal Epithelial Architecture
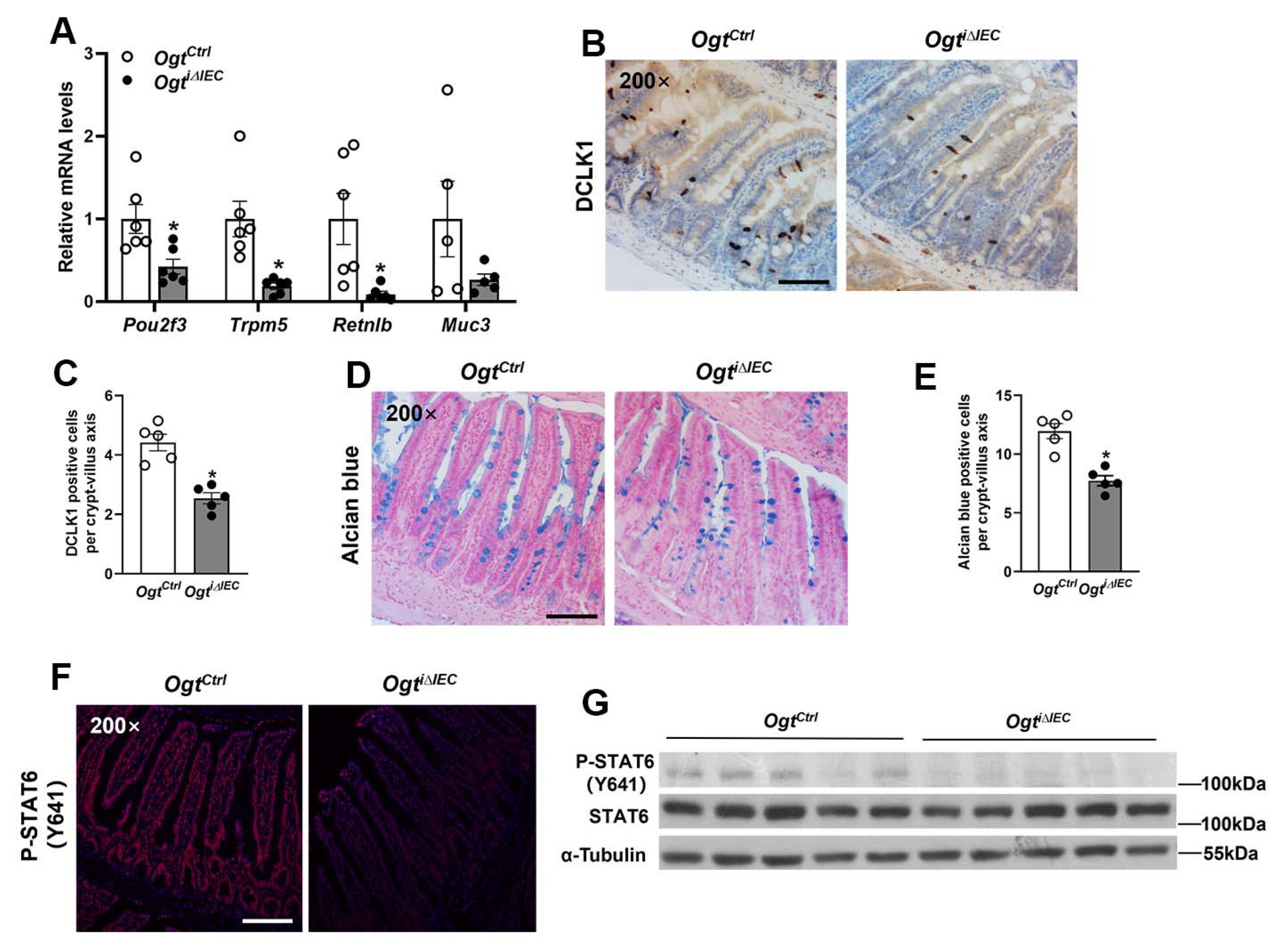
2.3. Epithelial OGT Is Required for Tuft and Goblet Cell Differentiation in Adult Mice
2.4. OGT Regulates Intestinal Crypt Cell Proliferation and Paneth Cell Differentiation in a STAT6-Independent Manner
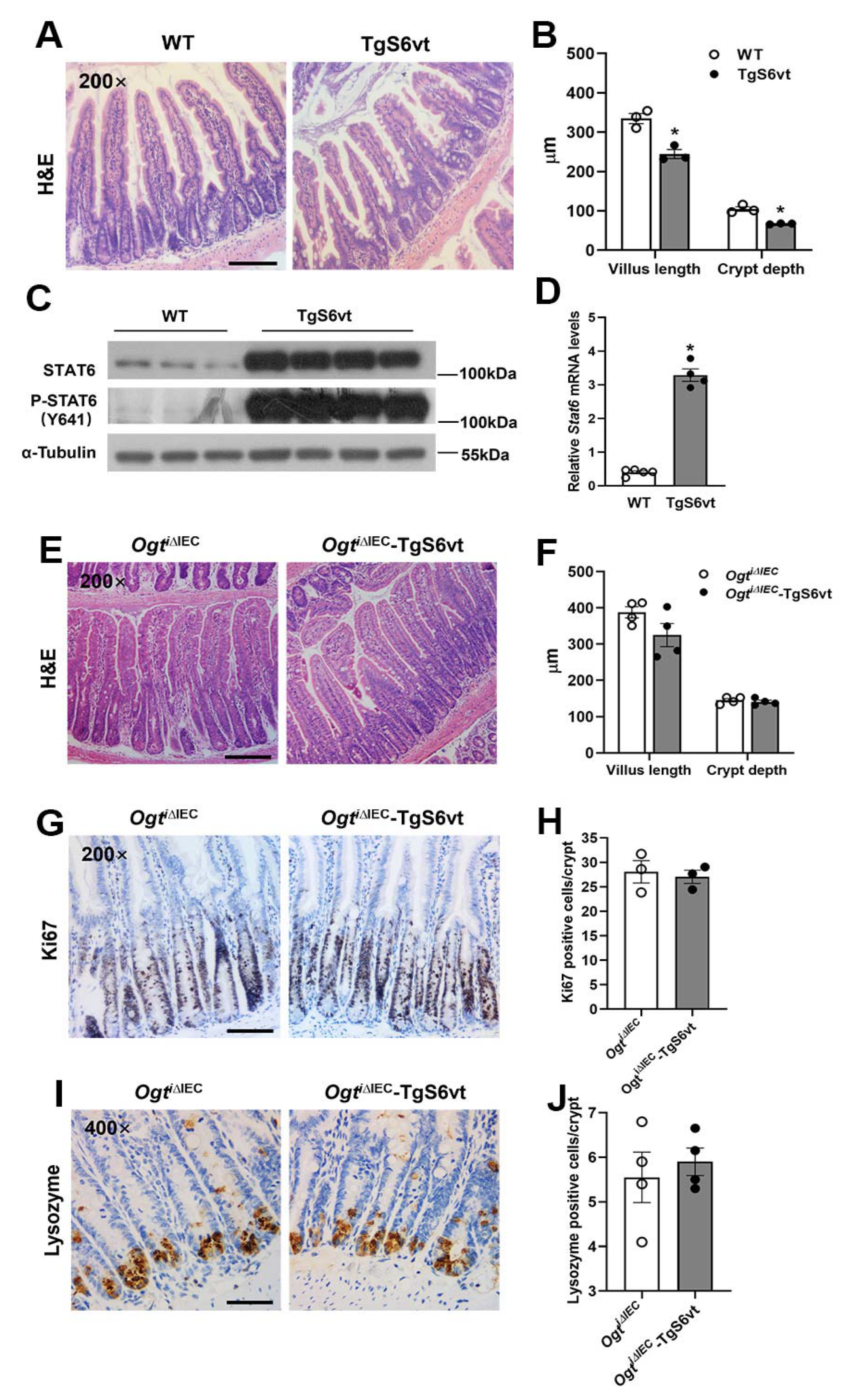
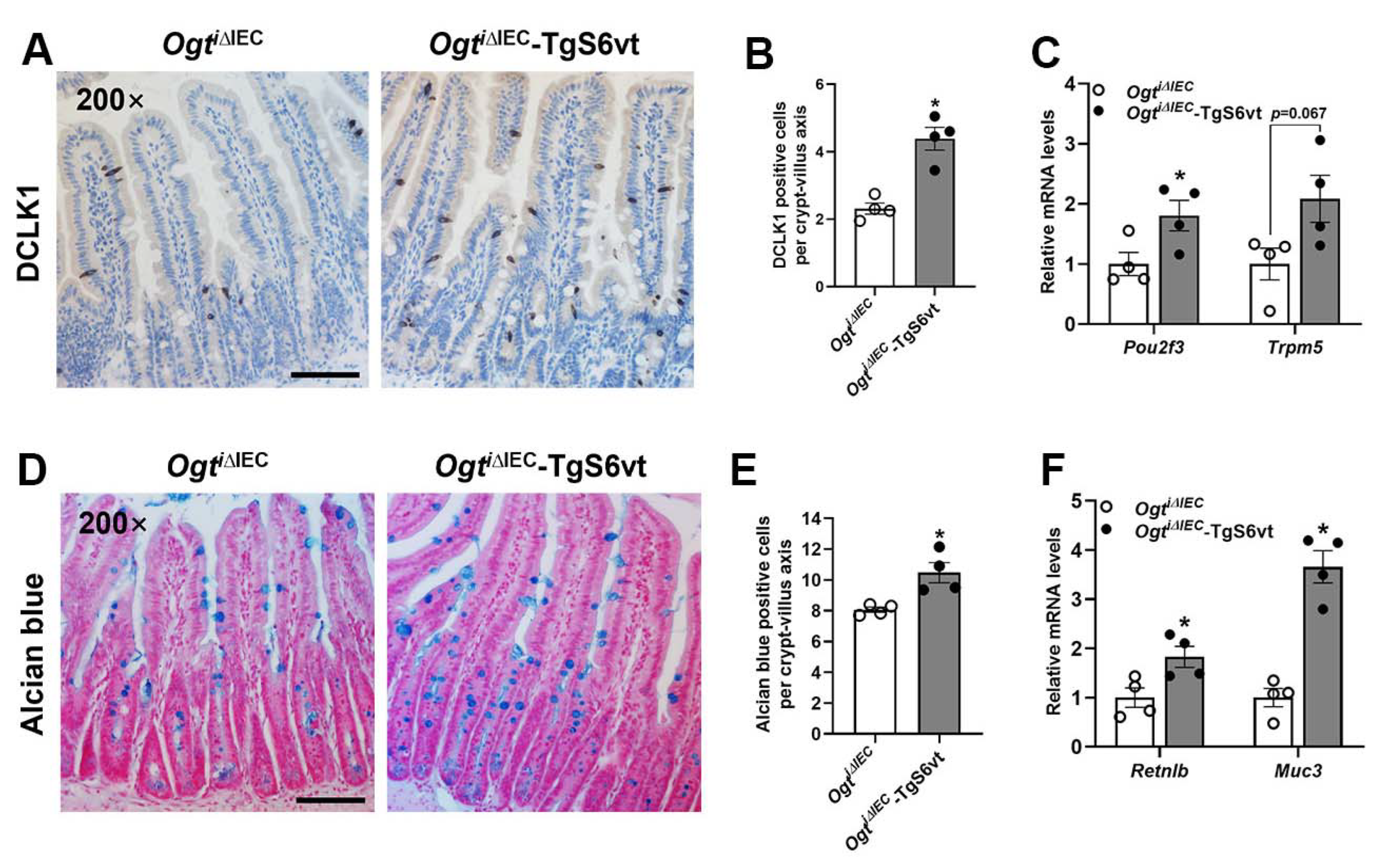
2.5. STAT6 Activation Promotes Intestinal Tuft and Goblet Cell Differentiation in Ogti∆IEC Mice
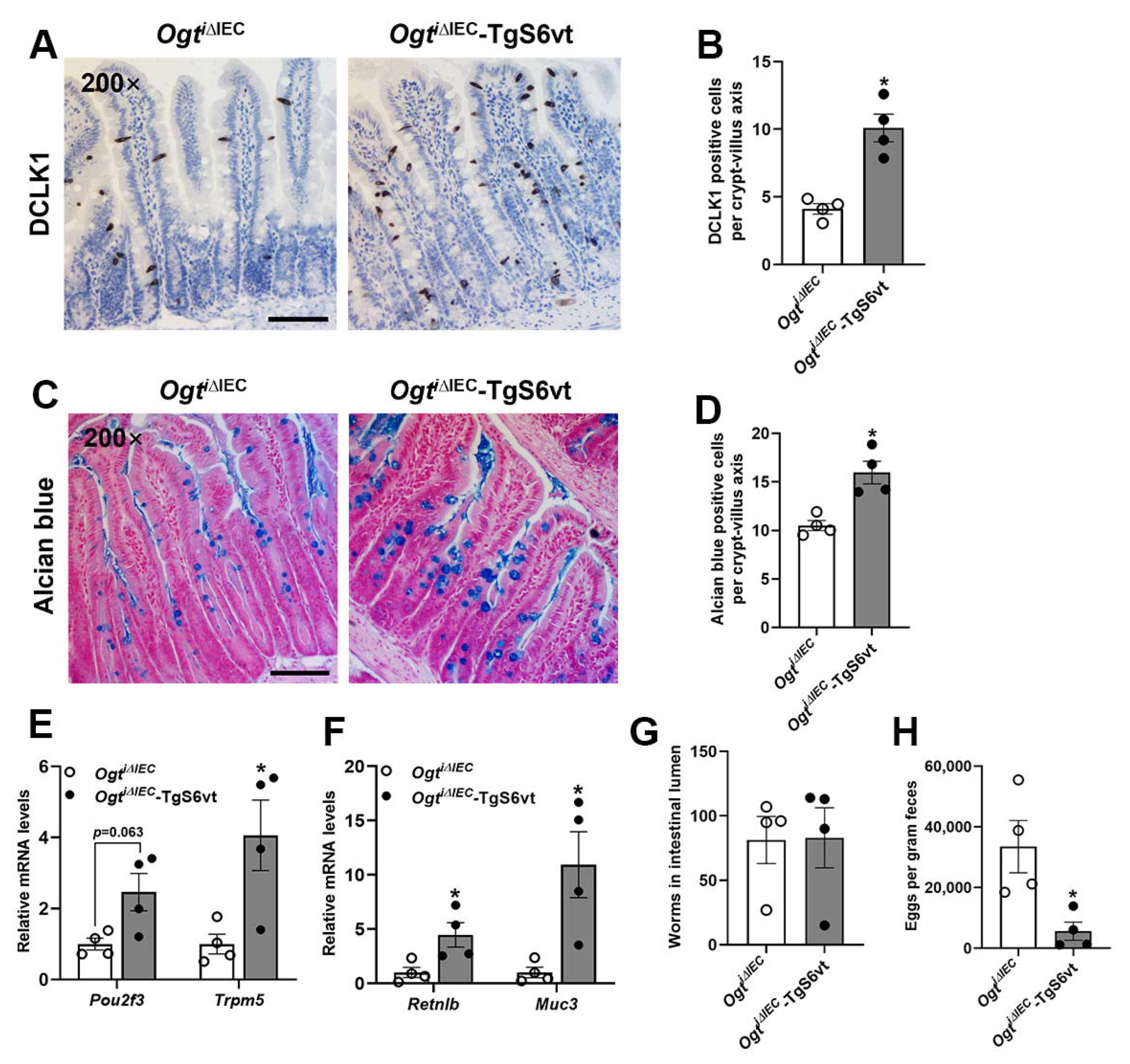
2.6. STAT6 Overexpression Rescues the Defective Anti-Helminth Responses Caused by Epithelial Ogt Ablation
3. Discussion
4. Materials and Methods
4.1. Mice
4.2. Intestinal Epithelium (IECs) Isolation
4.3. RT-qPCR
4.4. Western Blot
4.5. Histological Analysis
4.6. Helminth Infection
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Allaire, J.M.; Crowley, S.M.; Law, H.T.; Chang, S.Y.; Ko, H.J.; Vallance, B.A. The Intestinal Epithelium: Central Coordinator of Mucosal Immunity. Trends Immunol. 2018, 39, 677–696. [Google Scholar] [CrossRef]
- Coakley, G.; Harris, N.L. The Intestinal Epithelium at the Forefront of Host-Helminth Interactions. Trends Parasitol. 2020, 36, 761–772. [Google Scholar] [CrossRef]
- Peterson, L.W.; Artis, D. Intestinal epithelial cells: Regulators of barrier function and immune homeostasis. Nat. Rev. Immunol. 2014, 14, 141–153. [Google Scholar] [CrossRef]
- Beumer, J.; Clevers, H. Cell fate specification and differentiation in the adult mammalian intestine. Nat. Rev. Mol. Cell Biol. 2021, 22, 39–53. [Google Scholar] [CrossRef]
- Faniyi, A.A.; Wijanarko, K.J.; Tollitt, J.; Worthington, J.J. Helminth Sensing at the Intestinal Epithelial Barrier-A Taste of Things to Come. Front. Immunol. 2020, 11, 1489. [Google Scholar] [CrossRef] [PubMed]
- Howitt, M.R.; Lavoie, S.; Michaud, M.; Blum, A.M.; Tran, S.V.; Weinstock, J.V.; Gallini, C.A.; Redding, K.; Margolskee, R.F.; Osborne, L.C.; et al. Tuft cells, taste-chemosensory cells, orchestrate parasite type 2 immunity in the gut. Science 2016, 351, 1329–1333. [Google Scholar] [CrossRef] [PubMed]
- Gerbe, F.; Sidot, E.; Smyth, D.J.; Ohmoto, M.; Matsumoto, I.; Dardalhon, V.; Cesses, P.; Garnier, L.; Pouzolles, M.; Brulin, B.; et al. Intestinal epithelial tuft cells initiate type 2 mucosal immunity to helminth parasites. Nature 2016, 529, 226–230. [Google Scholar] [CrossRef]
- Von Moltke, J.; Ji, M.; Liang, H.E.; Locksley, R.M. Tuft-cell-derived IL-25 regulates an intestinal ILC2-epithelial response circuit. Nature 2016, 529, 221–225. [Google Scholar] [CrossRef] [PubMed]
- Goenka, S.; Kaplan, M.H. Transcriptional regulation by STAT6. Immunol. Res. 2011, 50, 87–96. [Google Scholar] [CrossRef]
- Anthony, R.M.; Rutitzky, L.I.; Urban, J.F., Jr.; Stadecker, M.J.; Gause, W.C. Protective immune mechanisms in helminth infection. Nat. Rev. Immunol. 2007, 7, 975–987. [Google Scholar] [CrossRef] [PubMed]
- Urban, J.F., Jr.; Noben-Trauth, N.; Donaldson, D.D.; Madden, K.B.; Morris, S.C.; Collins, M.; Finkelman, F.D. IL-13, IL-4Ralpha, and Stat6 are required for the expulsion of the gastrointestinal nematode parasite Nippostrongylus brasiliensis. Immunity 1998, 8, 255–264. [Google Scholar] [CrossRef]
- Zhao, M.; Ren, K.; Xiong, X.; Xin, Y.; Zou, Y.; Maynard, J.C.; Kim, A.; Battist, A.P.; Koneripalli, N.; Wang, Y.; et al. Epithelial STAT6 O-GlcNAcylation drives a concerted anti-helminth alarmin response dependent on tuft cell hyperplasia and Gasdermin C. Immunity 2022, 55, 623–638.e5. [Google Scholar] [CrossRef] [PubMed]
- Schubart, C.; Krljanac, B.; Otte, M.; Symowski, C.; Martini, E.; Gunther, C.; Becker, C.; Daniel, C.; Voehringer, D. Selective expression of constitutively activated STAT6 in intestinal epithelial cells promotes differentiation of secretory cells and protection against helminths. Mucosal Immunol. 2019, 12, 413–424. [Google Scholar] [CrossRef]
- Chang, Y.H.; Weng, C.L.; Lin, K.I. O-GlcNAcylation and its role in the immune system. J. Biomed. Sci. 2020, 27, 57. [Google Scholar] [CrossRef]
- Yang, X.; Qian, K. Protein O-GlcNAcylation: Emerging mechanisms and functions. Nat. Rev. Mol. Cell Biol. 2017, 18, 452–465. [Google Scholar] [CrossRef] [PubMed]
- Ong, Q.; Han, W.; Yang, X. O-GlcNAc as an Integrator of Signaling Pathways. Front. Endocrinol. 2018, 9, 599. [Google Scholar] [CrossRef]
- El Marjou, F.; Janssen, K.P.; Chang, B.H.; Li, M.; Hindie, V.; Chan, L.; Louvard, D.; Chambon, P.; Metzger, D.; Robine, S. Tissue-specific and inducible Cre-mediated recombination in the gut epithelium. Genesis 2004, 39, 186–193. [Google Scholar] [CrossRef]
- Zhao, M.; Xiong, X.; Ren, K.; Xu, B.; Cheng, M.; Sahu, C.; Wu, K.; Nie, Y.; Huang, Z.; Blumberg, R.S.; et al. Deficiency in intestinal epithelial O-GlcNAcylation predisposes to gut inflammation. EMBO Mol. Med. 2018, 10, 14255–14259. [Google Scholar] [CrossRef]
- Daniel, C.; Salvekar, A.; Schindler, U. A gain-of-function mutation in STAT6. J. Biol. Chem. 2000, 275, 14255–14259. [Google Scholar] [CrossRef]
- Xiong, X.; Yang, C.; He, W.Q.; Yu, J.; Xin, Y.; Zhang, X.; Huang, R.; Ma, H.; Xu, S.; Li, Z.; et al. Sirtuin 6 maintains epithelial STAT6 activity to support intestinal tuft cell development and type 2 immunity. Nat. Commun. 2022, 13, 5192. [Google Scholar] [CrossRef]
- Reynolds, L.A.; Filbey, K.J.; Maizels, R.M. Immunity to the model intestinal helminth parasite Heligmosomoides polygyrus. Semin. Immunopathol. 2012, 34, 829–846. [Google Scholar] [CrossRef]
- Zhao, M.; Ren, K.; Xiong, X.; Cheng, M.; Zhang, Z.; Huang, Z.; Han, X.; Yang, X.; Alejandro, E.U.; Ruan, H.B. Protein O-GlcNAc Modification Links Dietary and Gut Microbial Cues to the Differentiation of Enteroendocrine L. Cells. Cell Rep. 2020, 32, 108013. [Google Scholar] [CrossRef] [PubMed]
- Shafi, R.; Iyer, S.P.; Ellies, L.G.; O’Donnell, N.; Marek, K.W.; Chui, D.; Hart, G.W.; Marth, J.D. The O-GlcNAc transferase gene resides on the X chromosome and is essential for embryonic stem cell viability and mouse ontogeny. Proc. Natl. Acad. Sci. USA 2000, 97, 5735–5739. [Google Scholar] [CrossRef] [PubMed]
- Maizels, R.M.; Yazdanbakhsh, M. Immune regulation by helminth parasites: Cellular and molecular mechanisms. Nat. Rev. Immunol. 2003, 3, 733–744. [Google Scholar] [CrossRef]
- Harris, N.L.; Loke, P. Recent Advances in Type-2-Cell-Mediated Immunity: Insights from Helminth Infection. Immunity 2017, 47, 1024–1036. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Xiong, X.; Huang, R.; Li, Z.; Yang, C.; Wang, Q.; Ruan, H.-B.; Xu, L. Intestinal Epithelial STAT6 Activation Rescues the Defective Anti-Helminth Responses Caused by Ogt Deletion. Int. J. Mol. Sci. 2022, 23, 11137. https://doi.org/10.3390/ijms231911137
Xiong X, Huang R, Li Z, Yang C, Wang Q, Ruan H-B, Xu L. Intestinal Epithelial STAT6 Activation Rescues the Defective Anti-Helminth Responses Caused by Ogt Deletion. International Journal of Molecular Sciences. 2022; 23(19):11137. https://doi.org/10.3390/ijms231911137
Chicago/Turabian StyleXiong, Xiwen, Rong Huang, Zun Li, Chenyan Yang, Qingzhi Wang, Hai-Bin Ruan, and Lin Xu. 2022. "Intestinal Epithelial STAT6 Activation Rescues the Defective Anti-Helminth Responses Caused by Ogt Deletion" International Journal of Molecular Sciences 23, no. 19: 11137. https://doi.org/10.3390/ijms231911137
APA StyleXiong, X., Huang, R., Li, Z., Yang, C., Wang, Q., Ruan, H.-B., & Xu, L. (2022). Intestinal Epithelial STAT6 Activation Rescues the Defective Anti-Helminth Responses Caused by Ogt Deletion. International Journal of Molecular Sciences, 23(19), 11137. https://doi.org/10.3390/ijms231911137





