Transcription Factor MAFB as a Prognostic Biomarker for the Lung Adenocarcinoma
Abstract
1. Introduction
2. Results
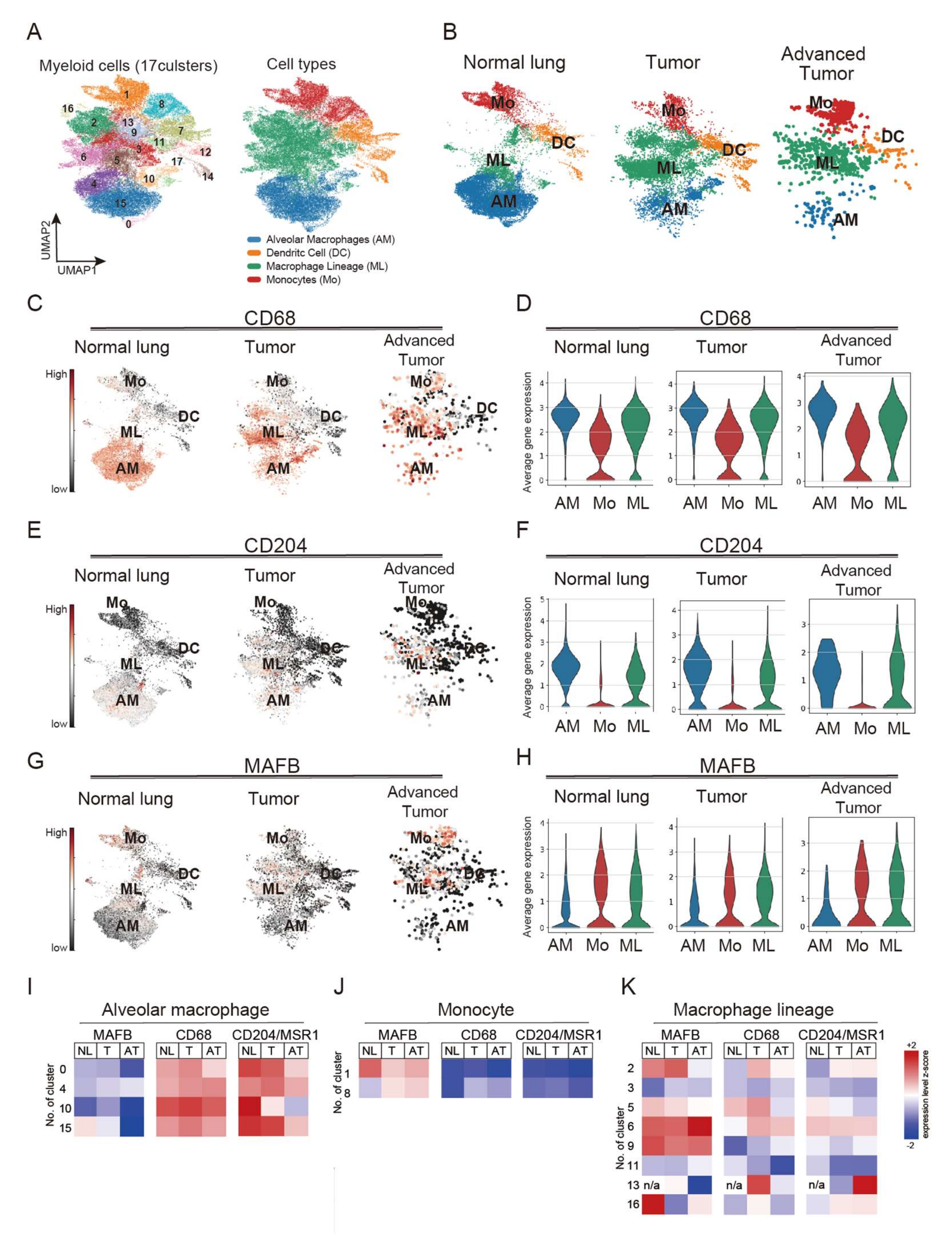
2.1. MAFB Is Specifically Expressed in Monocytes/Macrophages but Not Alveolar Macrophages in Both Normal and Cancerous Tissue
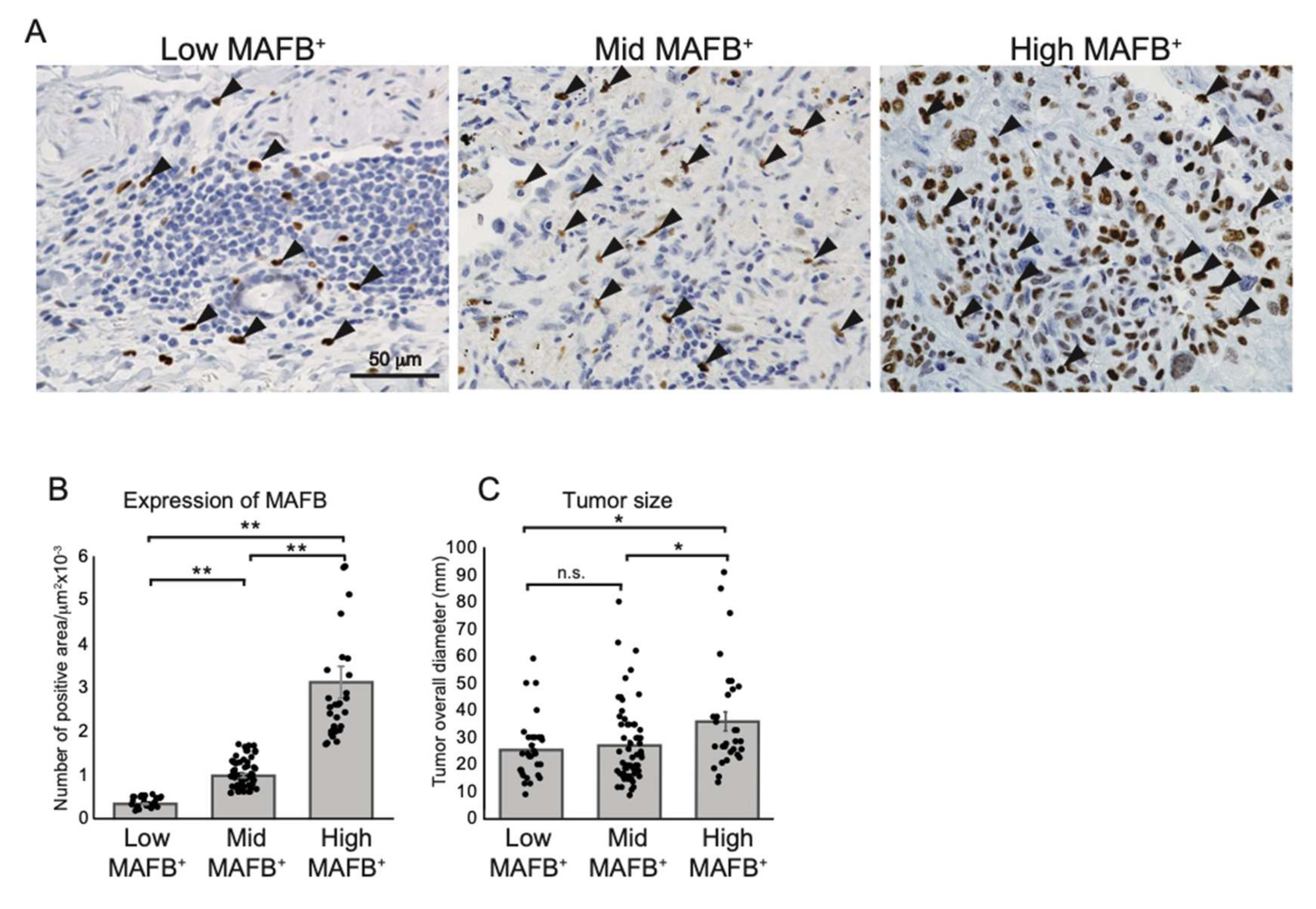
2.2. Higher MAFB+ Cell Density May Be Associated with Poor Clinical Prognosis among Lung Cancer Patients
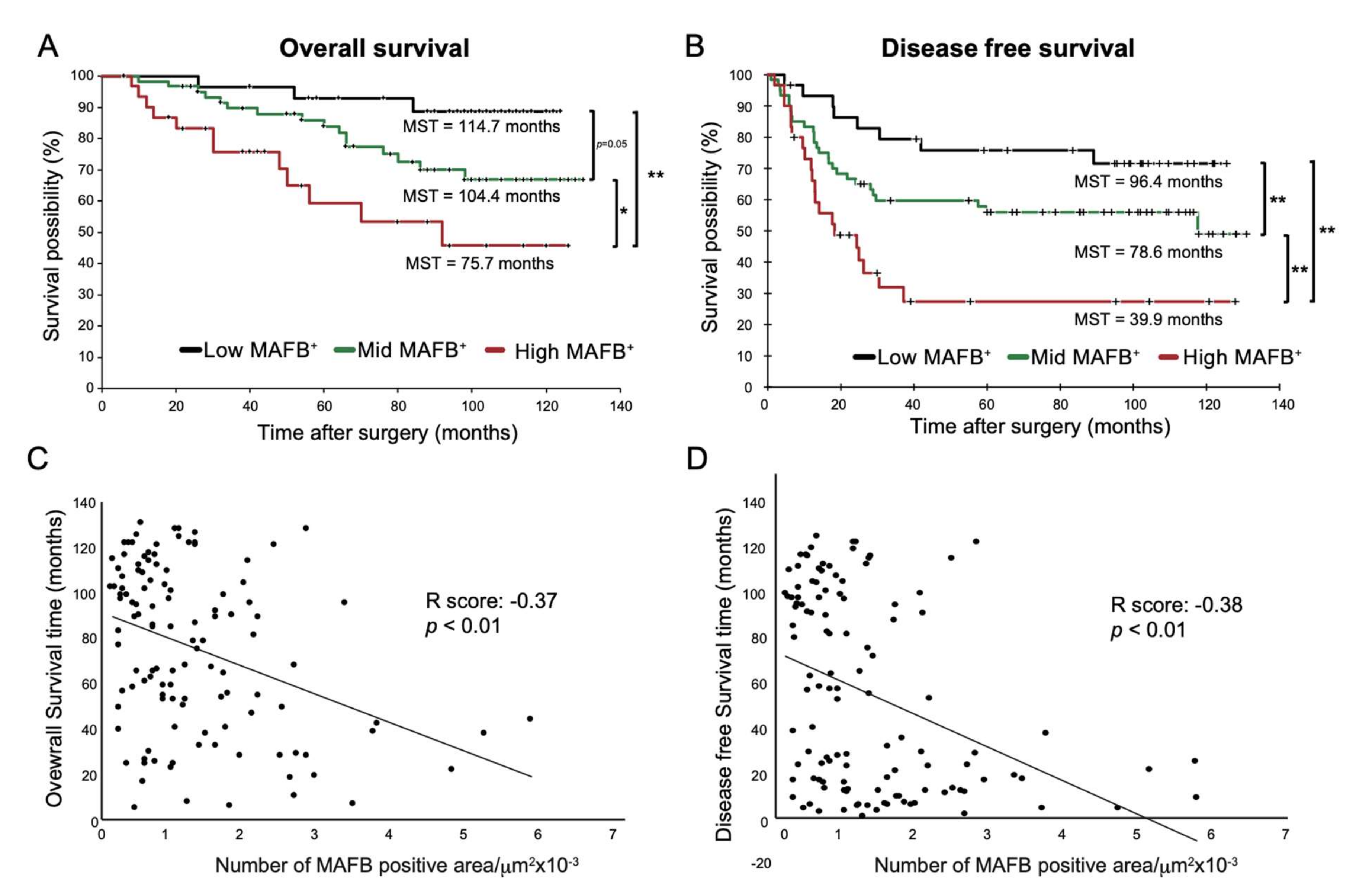
2.3. High-MAFB+ Cell Density Indicated a Higher Mortality Risk
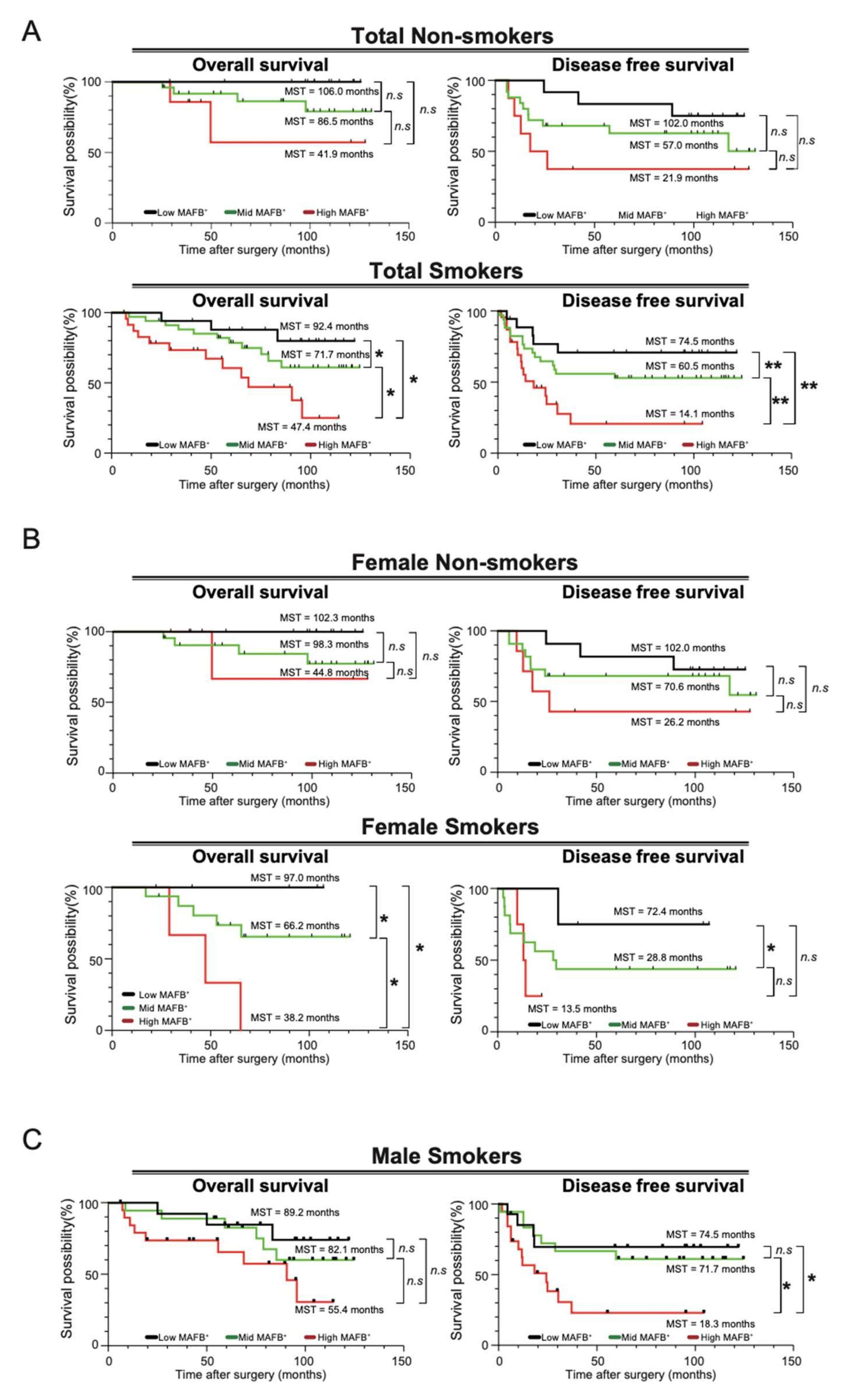
2.4. MAFB Could Be a Prognostic TAM Marker for Patients with Smoking Habits with Lung Adenocarcinoma
3. Discussion
4. Material and Methods
4.1. Single-Cell RNA Sequencing (scRNA-seq) Analysis
4.2. Immunostaining of Human Cancer Tissues
4.3. Evaluation of Clinicopathological Features
4.4. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| AM | Alveolar macrophages |
| bZIP | The basic leucine zipper |
| CD204 | Cluster of differentiation 204 |
| CD68 | Cluster of differentiation 68 |
| DC | Dendritic cells |
| DFS | Disease-free survival |
| FCGR3A | Fc gamma receptor IIIa gene |
| GEO | Gene expression omnibus |
| JSPS | Japan Society for the Promotion of Science |
| LY2 | Lymphocyte antigen 2 gene |
| Maf | Musculoaponeurotic fibrosarcoma |
| MAFB | V-maf musculoaponeurotic fibrosarcoma oncogene homolog B |
| MARCO | Macrophage receptor with collagenous structure gene |
| MEXT | The Ministry of Education, Culture, Sports, Science, and Technology |
| ML | Macrophage lineage |
| Mo | Monocytes |
| MSR1 | Macrophage scavenger receptor 1 |
| Mφs | Macrophages |
| OS | Overall survival |
| PCA | Principal component analysis |
| PCs | Principal components |
| scRNA-seq | Single-cell RNA sequencing |
| SEM | Standard error of mean |
| SNN | Shared nearest neighbor |
| T-CReDO | Tsukuba clinical research and development organization |
| TAM | Tumor-associated macrophages |
| THP | Tsukuba Human Tissue Biobank Center |
| TME | Tumor microenvironment |
| TNM | Staging system based on tumor size, nodal involvement, and metastasis |
| UICC | The Union for International Cancer Control |
| UMAP | Uniform Manifold Approximation and Projection |
| UMIs | Unique molecular identifiers |
References
- Kataoka, K.; Fujiwara, K.T.; Noda, M.; Nishizawa, M. MafB, a new Maf family transcription activator that can associate with Maf and Fos but not with Jun. Mol. Cell. Biol. 1994, 14, 7581–7591. [Google Scholar] [CrossRef] [PubMed]
- Sadl, V.S.; Jin, F.; Yu, J.; Cui, S.; Holmyard, D.; Quaggin, S.E.; Barsh, G.S.; Cordes, S.P. The Mouse Kreisler (Krml1/MafB) Segmentation Gene Is Required for Differentiation of Glomerular Visceral Epithelial Cells. Dev. Biol. 2002, 249, 16–29. [Google Scholar] [CrossRef] [PubMed]
- Hang, Y.; Stein, R. MafA and MafB activity in pancreatic β cells. Trends Endocrinol. Metab. 2011, 22, 364–373. [Google Scholar] [CrossRef] [PubMed]
- Nishimura, W.; Kondo, T.; Salameh, T.; El Khattabi, I.; Dodge, R.; Bonner-Weir, S.; Sharma, A. A switch from MafB to MafA expression accompanies differentiation to pancreatic β-cells. Dev. Biol. 2006, 293, 526–539. [Google Scholar] [CrossRef] [PubMed]
- Gautier, E.L.; Shay, T.; Miller, J.; Greter, M.; Jakubzick, C.; Ivanov, S.; Helft, J.; Chow, A.; Elpek, K.G.; Gordonov, S.; et al. Gene-expression profiles and transcriptional regulatory pathways that underlie the identity and diversity of mouse tissue macrophages. Nat. Immunol. 2012, 13, 1118–1128. [Google Scholar] [CrossRef] [PubMed]
- Daassi, D.; Hamada, M.; Jeon, H.; Imamura, Y.; Tran, M.T.N.; Takahashi, S. Differential expression patterns of MafB and c-Maf in macrophages in vivo and in vitro. Biochem. Biophys. Res. Commun. 2016, 473, 118–124. [Google Scholar] [CrossRef]
- Tran, M.T.N.; Hamada, M.; Jeon, H.; Shiraishi, R.; Asano, K.; Hattori, M.; Nakamura, M.; Imamura, Y.; Tsunakawa, Y.; Fujii, R.; et al. MafB is a critical regulator of complement component C1q. Nat. Commun. 2017, 8, 1700. [Google Scholar] [CrossRef]
- Shichita, T.; Ito, M.; Morita, R.; Komai, K.; Noguchi, Y.; Ooboshi, H.; Koshida, R.; Takahashi, R.K.S.; Kodama, T.; Yoshimura, A. MAFB prevents excess inflammation after ischemic stroke by accelerating clearance of damage signals through MSR1. Nat. Med. 2017, 23, 723–732. [Google Scholar] [CrossRef]
- Hamada, M.; Nakamura, M.; Tran, M.T.N.; Moriguchi, T.; Hong, C.; Ohsumi, T.; Dinh, T.T.H.; Kusakabe, M.; Hattori, M.; Katsumata, T.; et al. MafB promotes atherosclerosis by inhibiting foam-cell apoptosis. Nat. Commun. 2014, 5, 3147. [Google Scholar] [CrossRef]
- Machiya, J.-I.; Shibata, Y.; Yamauchi, K.; Hirama, N.; Wada, T.; Inoue, S.; Abe, S.; Takabatake, N.; Sata, M.; Kubota, I. Enhanced Expression of MafB Inhibits Macrophage Apoptosis Induced by Cigarette Smoke Exposure. Am. J. Respir. Cell Mol. Biol. 2007, 36, 418–426. [Google Scholar] [CrossRef]
- Sato-Nishiwaki, M.; Aida, Y.; Abe, S.; Shibata, Y.; Kimura, T.; Yamauchi, K.; Kishi, H.; Igarashi, A.; Inoue, S.; Sato, M.; et al. Reduced Number and Morphofunctional Change of Alveolar Macrophages in MafB Gene-Targeted Mice. PLoS ONE 2013, 8, e73963. [Google Scholar] [CrossRef]
- Mantovani, A.; Marchesi, F.; Malesci, A.; Laghi, L.; Allavena, P. Tumour-associated macrophages as treatment targets in oncology. Nat. Rev. Clin. Oncol. 2017, 14, 399–416. [Google Scholar] [CrossRef] [PubMed]
- Mehraj, U.; Qayoom, H.; Mir, M.A. Prognostic significance and targeting tumor-associated macrophages in cancer: New insights and future perspectives. Breast Cancer 2021, 28, 539–555. [Google Scholar] [CrossRef] [PubMed]
- Ma, J.; Liu, L.; Che, G.; Yu, N.; Dai, F.; You, Z. The M1 form of tumor-associated macrophages in non-small cell lung cancer is positively associated with survival time. BMC Cancer 2010, 10, 112. [Google Scholar] [CrossRef] [PubMed]
- Murray, P.J.; Allen, J.E.; Biswas, S.K.; Fisher, E.A.; Gilroy, D.W.; Goerdt, S.; Gordon, S.; Hamilton, J.A.; Ivashkiv, L.B.; Lawrence, T.; et al. Macrophage Activation and Polarization: Nomenclature and Experimental Guidelines. Immunity 2014, 41, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Cavnar, M.J.; Zeng, S.; Kim, T.; Sorenson, E.C.; Ocuin, L.M.; Balachandran, V.P.; Seifert, A.M.; Greer, J.B.; Popow, R.; Crawley, M.H.; et al. KIT oncogene inhibition drives intratumoral macrophage M2 polarization. J. Exp. Med. 2013, 210, 2873–2886. [Google Scholar] [CrossRef]
- Segura, E.; Touzot, M.; Bohineust, A.; Cappuccio, A.; Chiocchia, G.; Hosmalin, A.; Dalod, M.; Soumelis, V.; Amigorena, S. Human Inflammatory Dendritic Cells Induce Th17 Cell Differentiation. Immunity 2013, 38, 336–348. [Google Scholar] [CrossRef]
- Yi, H.; Yu, X.; Gao, P.; Wang, Y.; Baek, S.-H.; Chen, X.; Kim, H.L.; Subjeck, J.R.; Wang, X.-Y. Pattern recognition scavenger receptor SRA/CD204 down-regulates Toll-like receptor 4 signaling–dependent CD8 T-cell activation. Blood 2009, 113, 5819–5828. [Google Scholar] [CrossRef]
- Rusmaully, J.; Tvardik, N.; Martin, D.; Billmann, R.; Cénée, S.; Antoine, M.; Blons, H.; Laurent-Puig, P.; Trédaniel, J.; Wislez, M.; et al. Risk of lung cancer among women in relation to lifetime history of tobacco smoking: A population-based case-control study in France (the WELCA study). BMC Cancer 2021, 21, 711. [Google Scholar] [CrossRef]
- Mei, J.; Xiao, Z.; Guo, C.; Pu, Q.; Ma, L.; Liu, C.; Lin, F.; Liao, H.; You, Z.; Liu, L. Prognostic impact of tumor-associated macrophage infiltration in non-small cell lung cancer: A systemic review and meta-analysis. Oncotarget 2016, 7, 34217–34228. [Google Scholar] [CrossRef]
- Yadav, M.K.; Inoue, Y.; Nakane-Otani, A.; Tsunakawa, Y.; Jeon, H.; Samir, O.; Teramoto, A.; Kulathunga, K.; Kusakabe, M.; Nakamura, M.; et al. Transcription factor MafB is a marker of tumor-associated macrophages in both mouse and humans. Biochem. Biophys. Res. Commun. 2019, 521, 590–595. [Google Scholar] [CrossRef] [PubMed]
- Kim, N.; Kim, H.K.; Lee, K.; Hong, Y.; Cho, J.H.; Choi, J.W.; Lee, J.-I.; Suh, Y.-L.; Ku, B.M.; Eum, H.H.; et al. Single-cell RNA sequencing demonstrates the molecular and cellular reprogramming of metastatic lung adenocarcinoma. Nat. Commun. 2020, 11, 2285. [Google Scholar] [CrossRef] [PubMed]
- Schmall, A.; Al-Tamari, H.M.; Herold, S.; Kampschulte, M.; Weigert, A.; Wietelmann, A.; Vipotnik, N.; Grimminger, F.; Seeger, W.; Pullamsetti, S.S.; et al. Macrophage and Cancer Cell Cross-talk via CCR2 and CX3CR1 Is a Fundamental Mechanism Driving Lung Cancer. Am. J. Respir. Crit. Care Med. 2015, 191, 437–447. [Google Scholar] [CrossRef] [PubMed]
- Cuevas, V.D.; Anta, L.; Samaniego, R.; Orta-Zavalza, E.; De La Rosa, J.V.; Baujat, G.; Domínguez-Soto, Á.; Sánchez-Mateos, P.; Escribese, M.M.; Castrillo, A.; et al. MAFB Determines Human Macrophage Anti-Inflammatory Polarization: Relevance for the Pathogenic Mechanisms Operating in Multicentric Carpotarsal Osteolysis. J. Immunol. 2017, 198, 2070–2081. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Maeda, D.; Yoshida, M.; Umakoshi, M.; Nanjo, H.; Shiraishi, K.; Saito, M.; Kohno, T.; Konno, H.; Saito, H.; et al. The intratumoral distribution influences the prognostic impact of CD68- and CD204-positive macrophages in non-small cell lung cancer. Lung Cancer 2018, 123, 127–135. [Google Scholar] [CrossRef]
- Fu, F.; Zhang, Y.; Gao, Z.; Zhao, Y.; Wen, Z.; Han, H.; Li, Y.; Hu, H.; Chen, H. Combination of CD47 and CD68 expression predicts survival in eastern-Asian patients with non-small cell lung cancer. J. Cancer Res. Clin. Oncol. 2021, 147, 739–747. [Google Scholar] [CrossRef]
- Zhang, B.; Yao, G.; Zhang, Y.; Gao, J.; Yang, B.; Rao, Z.; Gao, J. M2-Polarized tumor-associated macrophages are associated with poor prognoses resulting from accelerated lymphangiogenesis in lung adenocarcinoma. Clinics 2011, 66, 1879–1886. [Google Scholar] [CrossRef]
- Jung, K.Y.; Cho, S.W.; A Kim, Y.; Kim, D.; Oh, B.-C.; Park, D.J.; Park, Y.J. Cancers with Higher Density of Tumor-Associated Macrophages Were Associated with Poor Survival Rates. J. Pathol. Transl. Med. 2015, 49, 318–324. [Google Scholar] [CrossRef]
- Ao, L.; Che, X.; Qiu, X.; Li, Z.; Yang, B.; Wang, S.; Hou, K.; Fan, Y.; Qu, X.; Liu, Y. M2 macrophage infiltration into tumor islets leads to poor prognosis in non-small-cell lung cancer. Cancer Manag. Res. 2019, 11, 6125–6138. [Google Scholar] [CrossRef]
- Kreuzer, M.; Boffetta, P.; Whitley, E.; Ahrens, W.; Gaborieau, V.; Heinrich, J.; Jöckel, K.H.; Kreienbrock, L.; Mallone, S.; Merletti, F.; et al. Gender differences in lung cancer risk by smoking: A multicentre case–control study in Germany and Italy. Br. J. Cancer 2000, 82, 227–233. [Google Scholar] [CrossRef]
- Ohtaki, Y.; Ishii, G.; Nagai, K.; Ashimine, S.; Kuwata, T.; Hishida, T.; Nishimura, M.; Yoshida, J.; Takeyoshi, I.; Ochiai, A. Stromal Macrophage Expressing CD204 is Associated with Tumor Aggressiveness in Lung Adenocarcinoma. J. Thorac. Oncol. 2010, 5, 1507–1515. [Google Scholar] [CrossRef] [PubMed]
| Low-MAFB+ (≤0.005) | Mid-MAFB+ (0.006–0.016) | High-MAFB+ (≥0.017) | p-Value of Fisher’s Exact Test | |
|---|---|---|---|---|
| Variables | No of Case n = 30 (25%) | No of Case n = 59 (49%) | No of Case n = 31 (26%) | |
| Age (y) | ||||
| <70 | 20 (67%) | 39 (66%) | 15 (48%) | 0.2249 |
| ≥70 | 10 (33%) | 20 (34%) | 16 (52%) | |
| Gender | ||||
| Male | 15 (50%) | 21 (36%) * | 20 (65%) * | 0.0302 |
| Female | 15 (50%) | 38 (64%) * | 11 (35%) * | |
| Smoking history | ||||
| Never | 12 (40%) | 25 (42%) | 8 (26%) | 0.3046 |
| Former or current | 18 (60%) | 34 (58%) | 23 (74%) | |
| Clinical Stage | ||||
| I (n = 57) | 24 (42%) *** | 30 (53%) | 3 (5%) *** | <0.001 |
| II + III (n = 63) | 6 (10%) *** | 29 (46%) | 28 (44%) *** | |
| Cancer Recurrence | ||||
| Negative | 22 (73%) * | 33 (56%) | 10 (32%) * | 0.006 |
| Positive | 8 (27%) * | 26 (44%) | 21 (68%) * | |
| Nodal involvement | ||||
| Negative (N−) | 24 (80%) *** | 32 (54%) | 4 (13%) *** | <0.0001 |
| Positive (N+) | 6 (20%) *** | 27 (46%) | 27 (87%) *** | |
| Lymphatic permeation | ||||
| Ly(–) | 26 (87%) *** | 34 (58%) | 12 (39%) ** | <0.001 |
| Ly(+) | 4 (13%) *** | 25 (42%) | 19 (61%) ** | |
| Vessel invasion | ||||
| V(–) | 23 (77%) *** | 26 (44%) | 5 (16%) *** | <0.0001 |
| V(+) | 7 (23%) *** | 33 (56%) | 26 (84%) *** | |
| Pleural infiltration | ||||
| PL(–) | 23 (77%) * | 31 (53%) | 13 (42%) | 0.019 |
| PL(+) | 7 (23%) * | 28 (47%) | 18 (58%) | |
| Univariate Analysis | Multivariate Analysis | |||||||
|---|---|---|---|---|---|---|---|---|
| Disease-Free Survival | Overall Survival | Disease-Free Survival | Overall Survival | |||||
| Hr (95% Ci) | p Value | Hr (95% Ci) | p Value | Hr (95% Ci) | p Value | Hr (95% Ci) | p Value | |
| MAFB (low versus mid) | 2.998 (0.9882–12.95) | 0.0828 | 3.039 (1.001–13.13) | 0.079 | 1.665 (0.5008–7.565) | 0.4468 | 1.966 (0.5985–8.859) | 0.3086 |
| MAFB (low versus high) | 7.423 (2.393–32.44) | 0.0018 | 8.105 (2.620–35.36) | 0.0011 | 1.773 (0.4873–8.669) | 0.4230 | 2.001 (0.5587–9.646) | 0.3263 |
| Gender (male versus female) | 1.856 (0.9236–3.847) | 0.0861 | 1.826 (0.9081–3.790) | 0.095 | ||||
| Smoking (Yes versus No) | 3.115 (1.369–8.376) | 0.0122 | 0.3208 3.117 (1.368–8.390) | 0.0123 | 3.106 (1290–8.724) | 0.0182 | 2.989 (1.231–8.437) | 0.0235 |
| Age (<70 versus ≥70) | 1.010 (0.9697–1.058) | 0.6397 | 1.013 (0.9715–1.062) | 0.5578 | ||||
| Pstage (I versus II + III) | 7.500 (3.194–20.72) | <0.0001 | 8.464 (3.614–23.24) | <0.0001 | ||||
| Nodal involvement (Yes versus No) | 6.497 (2.880–16.71) | <0.0001 | 7.723 (3.411–19.85) | <0.0001 | 4.382 (1.555–13.88) | 0.0078 | 5.304 (1.845–17.12) | 0.0032 |
| Lymphatic invasion (Yes versus No) | 3.072 (1.522–6.398) | 0.002 | 3.294 (1.626–6.887) | 0.0011 | 1.049 (0.4812–2.384) | 0.9064 | 1.094 (0.4918–2.522) | 0.8285 |
| Vessel invasion (Yes versus No) | 5.444 (2.377–14.71) | 0.0002 | 4.875 (2.136–13.14) | 0.0005 | 1.843 (0.6468–5.939) | 0.2753 | 1.370 (0.4730–4.458) | 0.5786 |
| Pleural infiltration (Yes versus No) | 2.740 (1.350–5.803) | 0.0062 | 2.644 (1.306–5.585) | 0.008 | 1.228 (0.5665–2.793) | 0.6108 | 1.216 (0.5498–2.805) | 0.6363 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Samir, O.; Kobayashi, N.; Nishino, T.; Siyam, M.; Yadav, M.K.; Inoue, Y.; Takahashi, S.; Hamada, M. Transcription Factor MAFB as a Prognostic Biomarker for the Lung Adenocarcinoma. Int. J. Mol. Sci. 2022, 23, 9945. https://doi.org/10.3390/ijms23179945
Samir O, Kobayashi N, Nishino T, Siyam M, Yadav MK, Inoue Y, Takahashi S, Hamada M. Transcription Factor MAFB as a Prognostic Biomarker for the Lung Adenocarcinoma. International Journal of Molecular Sciences. 2022; 23(17):9945. https://doi.org/10.3390/ijms23179945
Chicago/Turabian StyleSamir, Omar, Naohiro Kobayashi, Teppei Nishino, Mennatullah Siyam, Manoj Kumar Yadav, Yuri Inoue, Satoru Takahashi, and Michito Hamada. 2022. "Transcription Factor MAFB as a Prognostic Biomarker for the Lung Adenocarcinoma" International Journal of Molecular Sciences 23, no. 17: 9945. https://doi.org/10.3390/ijms23179945
APA StyleSamir, O., Kobayashi, N., Nishino, T., Siyam, M., Yadav, M. K., Inoue, Y., Takahashi, S., & Hamada, M. (2022). Transcription Factor MAFB as a Prognostic Biomarker for the Lung Adenocarcinoma. International Journal of Molecular Sciences, 23(17), 9945. https://doi.org/10.3390/ijms23179945








