Iron Source and Medium pH Affect Nutrient Uptake and Pigment Content in Petunia hybrida ‘Madness Red’ Cultured In Vitro
Abstract
1. Introduction
2. Results
2.1. Plant Morphological and Growth Parameters
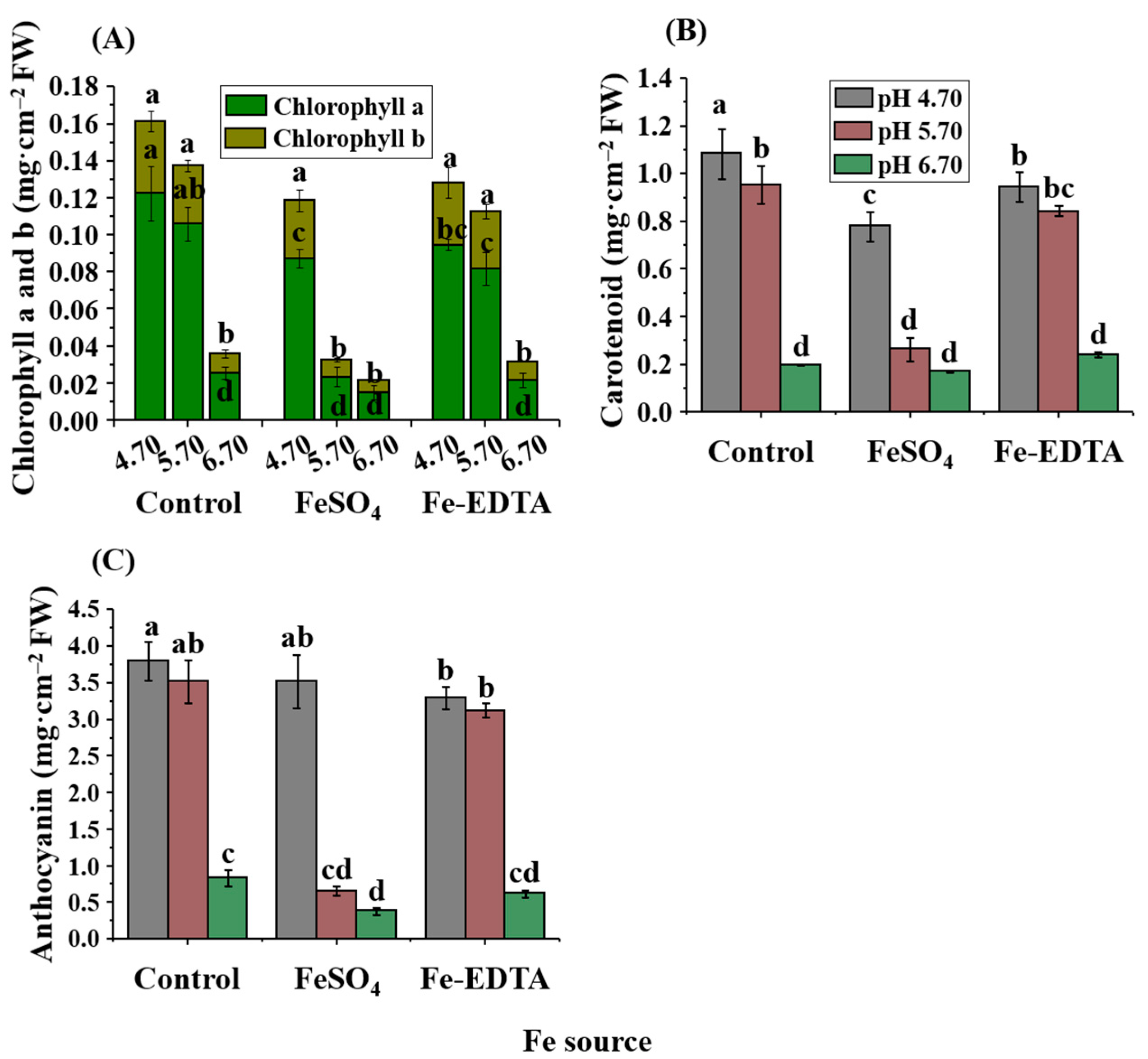
2.2. Leaf Color and Pigment Contents
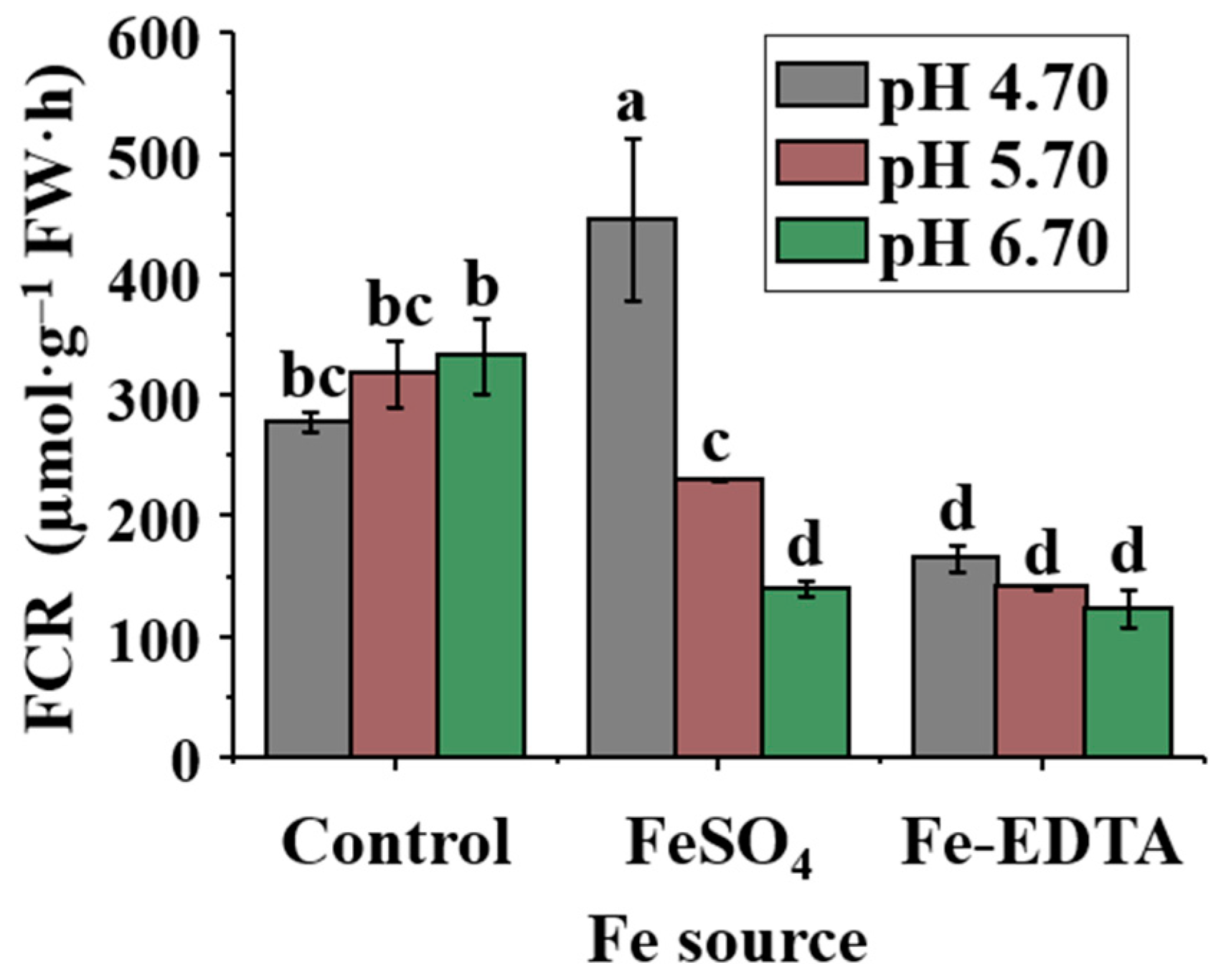
2.3. FCR Activity in Roots
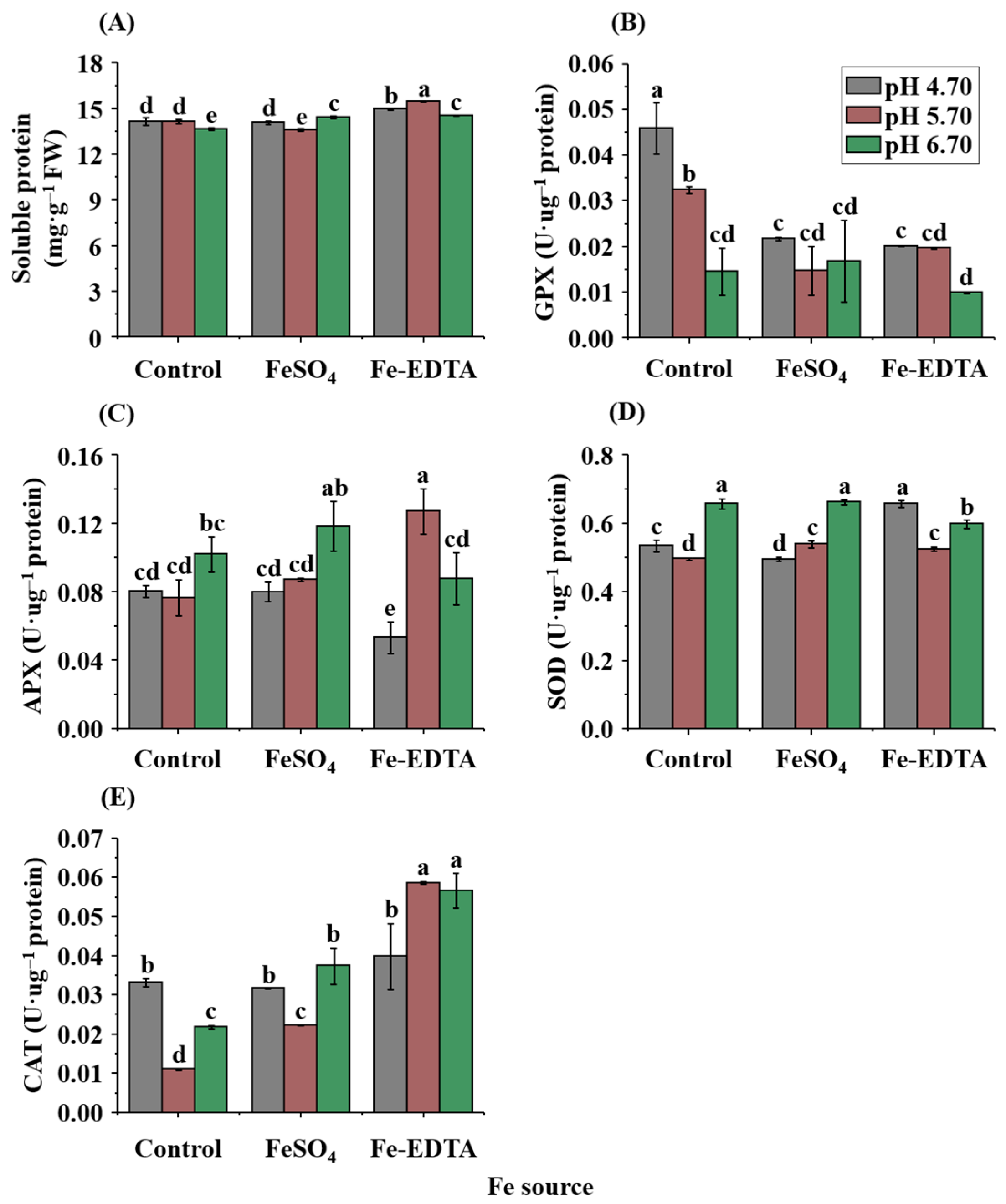
2.4. Soluble Protein Contents and Antioxidant Enzyme Activities
2.5. NH4+ and NO3− Contents
2.6. Contents of Macronutrients and Micronutrients in Leaves
2.7. Correlation Analysis of Pigments with Fe Content, Growth Parameters, and Nutrient Contents in Leaves
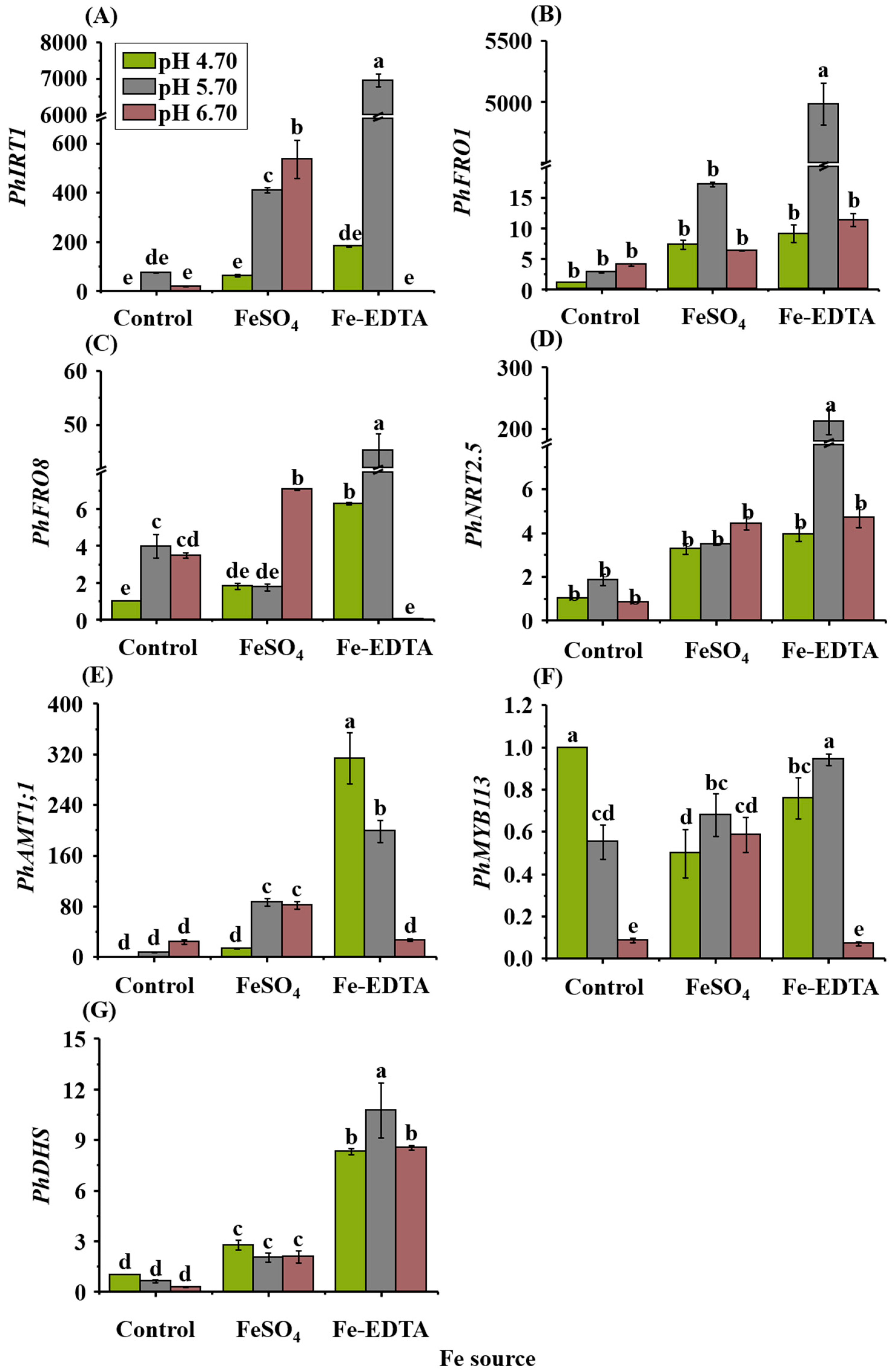
2.8. Expressions of Fe, Pigments, and N Transporter Genes
3. Discussion
4. Materials and Methods
4.1. Seed Materials and Sterilization
4.2. Seed Germination and Culture Conditions
4.3. Experimental Design of the Fe and pH Treatments
4.4. Measurements of the Growth Parameters
4.5. Contents of Chlorophyll, Carotenoid, and Anthocyanin
4.6. Determination of the FCR Activity
4.7. Soluble Protein Content and Antioxidant Enzyme Activity Assays
4.8. Contents of NH4+ and NO3−
4.9. Macronutrient and Micronutrient Contents
4.10. Quantitative RT-PCR Analysis
4.11. Data Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Miller, G.; Pushnik, J.; Welkie, G. Iron chlorosis, a world wide problem, the relation of chlorophyll biosynthesis to iron. J. Plant Nutr. 1984, 7, 1–22. [Google Scholar] [CrossRef]
- Abadia, J. Leaf responses to Fe deficiency: A review. J. Plant Nutr. 1992, 15, 1699–1713. [Google Scholar] [CrossRef]
- Sági-Kazár, M.; Solymosi, K.; Solti, Á. Iron in leaves: Chemical forms, signalling, and in-cell distribution. J. Exp. Bot. 2022, 73, 1717–1734. [Google Scholar] [CrossRef]
- Hell, R.; Stephan, U.W. Iron uptake, trafficking and homeostasis in plants. Planta 2003, 216, 541–551. [Google Scholar] [CrossRef]
- Schmidt, W. Mechanisms and regulation of reduction-based iron uptake in plants. New Phytol. 1999, 141, 1–26. [Google Scholar] [CrossRef]
- Seregin, I.V.; Kozhevnikova, A.D. Low-molecular-weight ligands in plants: Role in metal homeostasis and hyperaccumulation. Photosynth. Res. 2021, 150, 51–96. [Google Scholar] [CrossRef]
- Ricachenevsky, F.K.; Sperotto, R. There and back again, or always there? The evolution of rice combined strategy for Fe uptake. Front. Plant Sci. 2014, 5, 189. [Google Scholar] [CrossRef]
- Hindt, M.N.; Guerinot, M.L. Getting a sense for signals: Regulation of the plant iron deficiency response. Biochim. Biophys. Acta-Mol. Cell Res. 2012, 1823, 1521–1530. [Google Scholar] [CrossRef]
- Takahashi, M.; Yamaguchi, H.; Nakanishi, H.; Shioiri, T.; Nishizawa, N.-K.; Mori, S. Cloning Two Genes for Nicotianamine Aminotransferase, a Critical Enzyme in Iron Acquisition (Strategy II) in Graminaceous Plants. Plant Physiol. 1999, 121, 947–956. [Google Scholar] [CrossRef]
- Higuchi, K.; Suzuki, K.; Nakanishi, H.; Yamaguchi, H.; Nishizawa, N.-K.; Mori, S. Cloning of Nicotianamine Synthase Genes, Novel Genes Involved in the Biosynthesis of Phytosiderophores. Plant Physiol. 1999, 119, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Bashir, K.; Inoue, H.; Nagasaka, S.; Takahashi, M.; Nakanishi, H.; Mori, S.; Nishizawa, N.K. Cloning and Characterization of Deoxymugineic Acid Synthase Genes from Graminaceous Plants. J. Biol. Chem. 2006, 281, 32395–32402. [Google Scholar] [CrossRef]
- Chen, W.W.; Yang, J.L.; Qin, C.; Jin, C.W.; Mo, J.H.; Ye, T.; Zheng, S.J. Nitric Oxide Acts Downstream of Auxin to Trigger Root Ferric-Chelate Reductase Activity in Response to Iron Deficiency in Arabidopsis. Plant Physiol. 2010, 154, 810–819. [Google Scholar] [CrossRef]
- Chen, L.; Zhao, X.; Ding, C.; Wang, S.; Ding, Y. Physiological and Molecular Responses Under Fe Deficiency in Two Rice (Oryza sativa) Genotypes Differing in Iron Accumulation Ability in Seeds. J. Plant Growth Regul. 2014, 33, 769–777. [Google Scholar] [CrossRef]
- Chen, J.; Wu, F.H.; Shang, Y.T.; Wang, W.H.; Hu, W.J.; Simon, M.; Liu, X.; Shangguan, Z.P.; Zheng, H.L. Hydrogen sulphide improves adaptation of Zea mays seedlings to iron deficiency. J. Exp. Bot. 2015, 66, 6605–6622. [Google Scholar] [CrossRef]
- Zhu, C.Q.; Zhang, J.H.; Zhu, L.F.; Abliz, B.; Zhong, C.; Bai, Z.G.; Hu, W.J.; Sajid, H.; James, A.B.; Cao, X.C.; et al. NH4+ facilitates iron reutilization in the cell walls of rice (Oryza sativa) roots under iron-deficiency conditions. Environ. Exp. Bot. 2018, 151, 21–31. [Google Scholar] [CrossRef]
- Sattelmacher, B. The apoplast and its significance for plant mineral nutrition. New Phytol. 2001, 149, 167–192. [Google Scholar] [CrossRef]
- Stephan, U.; Schmidke, I.; Stephan, V. The nicotianamine molecule is made-to-measure for complexation of metal micronutrients in plants. BioMetals 1996, 9, 84–90. [Google Scholar] [CrossRef]
- Tiffin, L.O. Iron Translocation I. Plant Culture, Exudate Sampling, Iron-Citrate Analysis. Plant Physiol. 1966, 41, 510–514. [Google Scholar] [CrossRef]
- Bienfait, H.F.; Scheffers, M.R. Some properties of ferric citrate relevant to the iron nutrition of plants. Plant Soil 1992, 143, 141–144. [Google Scholar] [CrossRef]
- Klatte, M.; Schuler, M.; Wirtz, M.; Fink-Straube, C.; Hell, R.; Bauer, P. The Analysis of Arabidopsis Nicotianamine Synthase Mutants Reveals Functions for Nicotianamine in Seed Iron Loading and Iron Deficiency Responses. Plant Physiol. 2009, 150, 257–271. [Google Scholar] [CrossRef]
- Schuler, M.; Rellán-Álvarez, R.; Fink-Straube, C.; Abadía, J.; Bauer, P. Nicotianamine Functions in the Phloem-Based Transport of Iron to Sink Organs, in Pollen Development and Pollen Tube Growth in Arabidopsis. Plant Cell 2012, 24, 2380–2400. [Google Scholar] [CrossRef]
- Lascelles, J. Adaptation to form bacteriochlorophyll in Rhodopseudomonas spheroides: Changes in activity of enzymes con-cerned in pyrrole synthesis. Biochem. J. 1959, 72, 508. [Google Scholar] [CrossRef] [PubMed]
- Abadia, J.; Morales, F.; Abadía, A. Photosystem II efficiency in low chlorophyll, iron-deficient leaves. Plant Soil 1999, 215, 183–192. [Google Scholar] [CrossRef]
- Mishra, S.N.; Srivastava, H.S. Stimulation of nitrate reductase activity by delta amino levulinic acid in excised maize leaves. Experientia 1983, 39, 1118–1120. [Google Scholar] [CrossRef]
- Liu, J.; Chang, X.; Ding, B.; Zhong, S.; Peng, L.; Wei, Q.; Meng, J.; Yu, Y. PhDHS Is Involved in Chloroplast Development in Petunia. Front. Plant Sci. 2019, 10, 284. [Google Scholar] [CrossRef] [PubMed]
- Sims, D.A.; Gamon, J.A. Relationships between leaf pigment content and spectral reflectance across a wide range of species, leaf structures and developmental stages. Remote Sens. Environ. 2002, 81, 337–354. [Google Scholar] [CrossRef]
- Ambawat, S.; Sharma, P.; Yadav, N.R.; Yadav, R.C. MYB transcription factor genes as regulators for plant responses: An overview. Physiol. Mol. Biol. Plants 2013, 19, 307–321. [Google Scholar] [CrossRef]
- Docimo, T.; Francese, G.; Ruggiero, A.; Batelli, G.; De Palma, M.; Bassolino, L.; Toppino, L.; Rotino, G.L.; Mennella, G.; Tucci, M. Phenylpropanoids Accumulation in Eggplant Fruit: Characterization of Biosynthetic Genes and Regulation by a MYB Transcription Factor. Front. Plant Sci. 2016, 6, 1233. [Google Scholar] [CrossRef]
- Liu, Y.; Lin-Wang, K.; Espley, R.V.; Wang, L.; Yang, H.; Yu, B.; Dare, A.; Gasic, E.V.; Wang, J.; Zhang, J.; et al. Functional diversification of the potato R2R3 MYB anthocyanin activators AN1, MYBA1, and MYB113 and their interaction with basic helix-loop-helix cofactors. J. Exp. Bot. 2016, 67, 2159–2176. [Google Scholar] [CrossRef]
- Gonzalez, A.; Zhao, M.; Leavitt, J.M.; Lloyd, A.M. Regulation of the anthocyanin biosynthetic pathway by the TTG1/bHLH/Myb transcriptional complex in Arabidopsis seedlings. Plant J. 2008, 53, 814–827. [Google Scholar] [CrossRef]
- Zhou, X.; Chen, R.; Fan, Y.I. Molecular mechanism of iron uptake, translocation and storage in plants. Crop Res. 2012, 26, 605–610. [Google Scholar]
- Van Beusichem, M.L.; Kirkby, E.A.; Baas, R. Influence of Nitrate and Ammonium Nutrition on the Uptake, Assimilation, and Distribution of Nutrients in Ricinus communis. Plant Physiol. 1988, 86, 914–921. [Google Scholar] [CrossRef]
- Thomson, C.J.; Marschner, H.; Römheld, V. Effect of nitrogen fertilizer form on pH of the bulk soil and rhizosphere, and on the growth, phosphorus, and micronutrient uptake of bean. J. Plant Nutr. 1993, 16, 493–506. [Google Scholar] [CrossRef]
- Szczerba, M.W.; Britto, D.T.; Balkos, K.D.; Kronzucker, H.J. Alleviation of rapid, futile ammonium cycling at the plasma membrane by potassium reveals K+-sensitive and -insensitive components of NH4+ transport. J. Exp. Bot. 2008, 59, 303–313. [Google Scholar] [CrossRef]
- Price, J.; Laxmi, A.; Martin, S.K.S.; Jang, J.-C. Global Transcription Profiling Reveals Multiple Sugar Signal Transduction Mechanisms in Arabidopsis. Plant Cell 2004, 16, 2128–2150. [Google Scholar] [CrossRef]
- Wang, R.; Guegler, K.; LaBrie, S.T.; Crawford, N.M. Genomic analysis of a nutrient response in Arabidopsis reveals diverse expression patterns and novel metabolic and potential regulatory genes induced by nitrate. Plant Cell 2000, 12, 1491–1509. [Google Scholar] [CrossRef]
- Murata, Y.; Itoh, Y.; Iwashita, T.; Namba, K. Transgenic petunia with the iron (III)-phytosiderophore transporter gene acquires tolerance to iron deficiency in alkaline environments. PLoS ONE 2015, 10, e0120227. [Google Scholar] [CrossRef]
- Smith, B.R.; Fisher, P.R.; Argo, W.R. Growth and Pigment Content of Container-grown Impatiens and Petunia in Relation to Root Substrate pH and Applied Micronutrient Concentration. HortScience 2004, 39, 1421–1425. [Google Scholar] [CrossRef]
- Smith, B.R.; Fisher, P.R.; Argo, W.R. Nutrient uptake in container-grown impatiens and petunia in response to root substrate pH and applied micronutrient concentration. HortScience 2004, 39, 1426–1431. [Google Scholar] [CrossRef]
- Nelson, P.V.; Niedziela, C.E., Jr.; Pitchay, D.S.; Mingis, N.C. Effectiveness, ammonium impact and potassium adequacy of soybean-base liquid fertilizer on bedding plants. J. Plant Nutr. 2010, 33, 724–735. [Google Scholar] [CrossRef]
- Ramos, L.; Bettin, A.; Herrada, B.M.P.; Arenas, T.L.; Becker, S.J. Effects of Nitrogen Form and Application Rates on the Growth of Petunia and Nitrogen Content in the Substrate. Commun. Soil Sci. Plant Anal. 2013, 44, 473–479. [Google Scholar] [CrossRef]
- Xiao, J.; Park, Y.G.; Guo, G.; Jeong, B.R. Effect of iron source and medium pH on growth and development of Sorbus commixta in vitro. Int. J. Mol. Sci. 2020, 22, 133. [Google Scholar] [CrossRef] [PubMed]
- Xiao, J.; Guo, G.; Jeong, B.R. Iron Supplement-Enhanced Growth and Development of Hydrangea macrophylla In Vitro under Normal and High pH. Cells 2021, 10, 3151. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Sokhansanj, S.; Wu, S.; Fang, R.; Yang, W.; Winter, P. A transformation technique from RGB signals to the Munsell system for color analysis of tobacco leaves. Comput. Electron. Agric. 1998, 19, 155–166. [Google Scholar] [CrossRef]
- Fisher, P.R.; Wik, R.M.; Smith, B.R.; Pasian, C.C.; Kmetz-González, M.; Argo, W.R. Correcting Iron Deficiency in Calibrachoa Grown in a Container Medium at High pH. HortTechnology 2003, 13, 308–313. [Google Scholar] [CrossRef]
- Wik, R.M.; Fisher, P.R.; Kopsell, D.A.; Argo, W.R. Iron form and concentration affect nutrition of container-grown Pelargonium and Calibrachoa. HortScience 2006, 41, 244–251. [Google Scholar] [CrossRef]
- Laulhere, J.P.; Briat, J.F. Iron release and uptake by plant ferritin: Effects of pH, reduction and chelation. Biochem. J. 1993, 290, 693–699. [Google Scholar] [CrossRef]
- Šrámek, F.; Dubsky, M. Occurrence and correction of chlorosis in young petunia plants. Hortic. Sci. 2009, 36, 147–153. [Google Scholar] [CrossRef]
- Boxma, R. Effect of pH on the behaviour of various iron chelates in sphagnum (moss) peat. Commun. Soil Sci. Plant Anal. 1981, 12, 755–763. [Google Scholar] [CrossRef]
- Poonnachit, U.; Darnell, R. Effect of ammonium and nitrate on ferric chelate reductase and nitrate reductase in Vaccinium species. Ann. Bot. 2004, 93, 399–405. [Google Scholar] [CrossRef]
- Fox, T.C.; Shaff, J.E.; Grusak, M.A.; Norvell, W.A.; Chen, Y.; Chaney, R.L.; Kochian, L. Direct Measurement of 59Fe-Labeled Fe2+ Influx in Roots of Pea Using a Chelator Buffer System to Control Free Fe2+ in Solution. Plant Physiol. 1996, 111, 93–100. [Google Scholar] [CrossRef][Green Version]
- Moog, P.R.; Brüggemann, W. Iron reductase systems on the plant plasma membrane—A review. Plant Soil 1994, 165, 241–260. [Google Scholar] [CrossRef]
- Romera, F.J.; Alcántara, E.; De La Guardia, M.D. The induction of the “turbo reductase” is inhibited by cycloheximide, cordycepin and ethylene inhibitors in Fe-deficient cucumber (Cucumis sativus L.) plants. Protoplasma 1998, 205, 156–162. [Google Scholar] [CrossRef]
- Gogorcena, Y.; Abadía, J.; Abadía, A. Induction of in vivo root ferric chelate reductase activity in fruit tree rootstock. J. Plant Nutr. 2000, 23, 9–21. [Google Scholar] [CrossRef]
- Lindsay, W. Chemical reactions in soils that affect iron availability to plants. A quantative approach. In Iron Nutrition in Soils and Plants; Abadia, J., Ed.; Springer: Dordrecht, The Netherlands, 1995; pp. 7–14. [Google Scholar] [CrossRef]
- Crichton, R.R.; Wilmet, S.; Legssyer, R.; Ward, R.J. Molecular and cellular mechanisms of iron homeostasis and toxicity in mammalian cells. J. Inorg. Biochem. 2002, 91, 9–18. [Google Scholar] [CrossRef]
- Lee, S.-R.; Oh, M.-M.; Park, S.-A. Ferric-chelate reductase activity is a limiting factor in iron uptake in spinach and kale roots. Hortic. Environ. Biotechnol. 2016, 57, 462–469. [Google Scholar] [CrossRef]
- Sunda, W.; Huntsman, S. Effect of pH, light, and temperature on Fe-EDTA chelation and Fe hydrolysis in seawater. Mar. Chem. 2003, 84, 35–47. [Google Scholar] [CrossRef]
- Abbaszadeh-Dahaji, P.; Masalehi, F.; Akhgar, A. Improved Growth and Nutrition of Sorghum (Sorghum bicolor) Plants in a Low-Fertility Calcareous Soil Treated with Plant Growth—Promoting Rhizobacteria and Fe-EDTA. J. Soil Sci. Plant Nutr. 2020, 20, 31–42. [Google Scholar] [CrossRef]
- Curie, C.; Alonso, J.M.; Jean, M.L.; Ecker, J.R.; Briat, J.F. Involvement of NRAMP1 from Arabidopsis thaliana in iron transport. Biochem. J. 2000, 347, 749–755. [Google Scholar] [CrossRef]
- Vert, G.; Briat, J.-F.; Curie, C. Arabidopsis IRT2 gene encodes a root-periphery iron transporter. Plant J. 2001, 26, 181–189. [Google Scholar] [CrossRef]
- Rogers, E.E.; Eide, D.J.; Guerinot, M.L. Altered selectivity in an Arabidopsis metal transporter. Proc. Natl. Acad. Sci. USA 2000, 97, 12356–12360. [Google Scholar] [CrossRef] [PubMed]
- Korshunova, Y.O.; Eide, D.; Clark, W.G.; Guerinot, M.L.; Pakrasi, H.B. The IRT1 protein from Arabidopsis thaliana is a metal transporter with a broad substrate range. Plant Mol. Biol. 1999, 40, 37–44. [Google Scholar] [CrossRef] [PubMed]
- Vert, G.; Grotz, N.; Dédaldéchamp, F.; Gaymard, F.; Guerinot, M.L.; Briat, J.F.; Curie, C. IRT1, an Arabidopsis transporter essential for iron uptake from the soil and for plant growth. Plant Cell 2002, 14, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Bennett, S.A.; Hansman, R.; Sessions, A.; Nakamura, K.-I.; Edwards, K.J. Tracing iron-fueled microbial carbon production within the hydrothermal plume at the Loihi seamount. Geochim. Cosmochim. Acta 2011, 75, 5526–5539. [Google Scholar] [CrossRef]
- Ferrandon, M.; Chamel, A.R. Cuticular retention, foliar absorption and translocation of Fe, Mn and Zn supplied in organic and inorganic form. J. Plant Nutr. 1988, 11, 247–263. [Google Scholar] [CrossRef]
- Waters, B.M.; Blevins, D.G.; Eide, D.J. Characterization of FRO1, a pea ferric-chelate reductase involved in root iron acquisi-tion. Plant Physiol. 2002, 129, 85–94. [Google Scholar] [CrossRef]
- Li, L.; Ye, L.; Kong, Q.; Shou, H. A Vacuolar Membrane Ferric-Chelate Reductase, OsFRO1, Alleviates Fe Toxicity in Rice (Oryza sativa L.). Front. Plant Sci. 2019, 10, 700. [Google Scholar] [CrossRef]
- Bughio, N.; Takahashi, M.; Yoshimura, E.; Nishizawa, N.; Mori, S. Characteristics of light-regulated iron transport system in barley chloroplasts. In Plant Nutrition for Sustainable Food Production and Environment; Ando, T., Fujita, K., Mae, T., Matsumoto, H., Mori, S., Sekiya, J., Eds.; Springer: Dordrecht, The Netherlands, 1997; pp. 157–161. [Google Scholar]
- Jeong, J.; Connolly, E.L. Iron uptake mechanisms in plants: Functions of the FRO family of ferric reductases. Plant Sci. 2009, 176, 709–714. [Google Scholar] [CrossRef]
- Terry, N.; Abadia, J. Function of iron in chloroplasts. J. Plant Nutr. 1986, 9, 609–646. [Google Scholar] [CrossRef]
- Briat, J.-F.; Curie, C.; Gaymard, F. Iron utilization and metabolism in plants. Curr. Opin. Plant Biol. 2007, 10, 276–282. [Google Scholar] [CrossRef]
- Brzezowski, P.; Richter, A.S.; Grimm, B. Regulation and function of tetrapyrrole biosynthesis in plants and algae. Biochim. Biophys. Acta-Bioenerg. 2015, 1847, 968–985. [Google Scholar] [CrossRef]
- Næsted, H.; Holm, A.; Jenkins, T.; Nielsen, H.B.; Harris, C.A.; Beale, M.H.; Andersen, M.; Mant, A.; Scheller, H.; Camara, B. Arabidopsis variegated 3 encodes a chloroplast-targeted, zinc-finger protein required for chloroplast and palisade cell development. J. Cell Sci. 2004, 117, 4807–4818. [Google Scholar] [CrossRef]
- Lv, X.-G.; Shi, Y.-F.; Xu, X.; Wei, Y.-L.; Wang, H.-M.; Zhang, X.-B.; Wu, J.-L. Oryza sativa Chloroplast Signal Recognition Particle 43 (OscpSRP43) Is Required for Chloroplast Development and Photosynthesis. PLoS ONE 2015, 10, e0143249. [Google Scholar] [CrossRef]
- Wang, T.W.; Lu, L.; Wang, D.; Thompson, J.E. Isolation and characterization of senescence-induced cDNAs encoding deoxyhypusine synthase and eucaryotic translation initiation factor 5A from tomato. J. Biol. Chem. 2001, 276, 17541–17549. [Google Scholar] [CrossRef]
- Rout, G.R.; Sahoo, S. Role of iron in plant growth and metabolism. Rev. Agric. Sci. 2015, 3, 1–24. [Google Scholar] [CrossRef]
- Kannan, S. Correlative influence of pH reduction on recovery from iron chlorosis in sorghum varieties. J. Plant Nutr. 1980, 2, 507–516. [Google Scholar] [CrossRef]
- Romero-Puertas, M.C.; Corpas, F.J.; Sandalio, L.M.; Leterrier, M.; Rodríguez-Serrano, M.; Del Río, L.A.; Palma, J.M. Glutathione reductase from pea leaves: Response to abiotic stress and characterization of the peroxisomal isozyme. New Phytol. 2006, 170, 43–52. [Google Scholar] [CrossRef]
- Gamon, J.A.; Surfus, J.S. Assessing leaf pigment content and activity with a reflectometer. New Phytol. 1999, 143, 105–117. [Google Scholar] [CrossRef]
- Koo, Y.; Poethig, R.S. Expression pattern analysis of three R2R3-MYB transcription factors for the production of anthocyanin in different vegetative stages of Arabidopsis leaves. Appl. Biol. Chem. 2021, 64, 5. [Google Scholar] [CrossRef]
- Stracke, R.; Werber, M.; Weisshaar, B. The R2R3-MYB gene family in Arabidopsis thaliana. Curr. Opin. Plant Biol. 2001, 4, 447–456. [Google Scholar] [CrossRef]
- Steyn, W.J.; Wand, S.; Holcroft, D.; Jacobs, G. Anthocyanins in vegetative tissues: A proposed unified function in photopro-tection. New Phytol. 2002, 155, 349–361. [Google Scholar] [CrossRef] [PubMed]
- Gould, K.S. Nature’s Swiss army knife: The diverse protective roles of anthocyanins in leaves. J. Biomed. Biotechnol. 2004, 2004, 314–320. [Google Scholar] [CrossRef] [PubMed]
- Albert, N.W.; Lewis, D.H.; Zhang, H.; Schwinn, K.E.; Jameson, P.E.; Davies, K.M. Members of an R2R3-MYB transcription factor family in Petunia are developmentally and environmentally regulated to control complex floral and vegetative pigmentation patterning. Plant J. 2011, 65, 771–784. [Google Scholar] [CrossRef] [PubMed]
- Khan, A.; Jalil, S.; Cao, H.; Tsago, Y.; Sunusi, M.; Chen, Z.; Shi, C.; Jin, X. The Purple Leaf (pl6) Mutation Regulates Leaf Color by Altering the Anthocyanin and Chlorophyll Contents in Rice. Plants 2020, 9, 1477. [Google Scholar] [CrossRef] [PubMed]
- Maruta, T.; Noshi, M.; Tanouchi, A.; Tamoi, M.; Yabuta, Y.; Yoshimura, K.; Ishikawa, T.; Shigeoka, S. H2O2-triggered retrograde signaling from chloroplasts to nucleus plays specific role in response to stress. J. Biol. Chem. 2012, 287, 11717–11729. [Google Scholar] [CrossRef] [PubMed]
- Bishko, A.J.; Fisher, P.R.; Argo, W.R. The pH-response of a peat-based medium to application of acid-reaction chemicals. HortScience 2003, 38, 26–31. [Google Scholar] [CrossRef]
- Pestana, M.; De Varennes, A.; Faria, E.A. Diagnosis and correction of iron chlorosis in fruit trees: A review. J. Food Agric. Environ. 2003, 1, 46–51. [Google Scholar]
- Belkhodja, R.; Morales, F.; Sanz, M.; Abadía, A.; Abadia, J. Iron deficiency in peach trees: Effects on leaf chlorophyll and nutrient concentrations in flowers and leaves. Plant Soil 1998, 203, 257–268. [Google Scholar] [CrossRef]
- Ghasemi-Fasaei, R.; Ronaghi, A.; Maftoun, M.; Karimian, N.; Soltanpour, P.N. Influence of FeEDDHA on Iron-Manganese Interaction in Soybean Genotypes in a Calcareous Soil. J. Plant Nutr. 2002, 26, 1815–1823. [Google Scholar] [CrossRef]
- Abadía, J.; Nishio, J.; Monge, E.; Montañés, L.; Heras, L. Mineral composition of peach leaves affected by iron chlorosis. J. Plant Nutr. 1985, 8, 965–975. [Google Scholar] [CrossRef]
- Iqbal, A.; Dong, Q.; Madeeha, A.; Wang, X.; Gui, H.; Zhang, H.; Pang, N.; Zhang, X.; Song, M. Untangling the molecular mechanisms and functions of nitrate to improve nitrogen use efficiency. J. Sci. Food Agric. 2020, 100, 904–914. [Google Scholar] [CrossRef]
- Xu, G.; Fan, X.; Miller, A.J. Plant Nitrogen Assimilation and Use Efficiency. Annu. Rev. Plant Biol. 2012, 63, 153–182. [Google Scholar] [CrossRef]
- Huang, L.; Li, M.; Shao, Y.; Sun, T.; Li, C.; Ma, F. Ammonium uptake increases in response to PEG-induced drought stress in Malus hupehensis Rehd. Environ. Exp. Bot. 2018, 151, 32–42. [Google Scholar] [CrossRef]
- Zou, C.; Shen, J.; Zhang, F.; Guo, S.; Rengel, Z.; Tang, C. Impact of nitrogen form on iron uptake and distribution in maize seedlings in solution culture. Plant Soil 2001, 235, 143–149. [Google Scholar] [CrossRef]
- Kosegarten, H.; Schwed, U.; Wilson, G.; Mengel, K. Comparative investigation on the susceptibility of faba bean (Vicia faba L.) and sunflower (Helianthus annuus L.) to iron chlorosis. J. Plant Nutr. 1998, 21, 1511–1528. [Google Scholar] [CrossRef]
- Kosegarten, H.U.; Hoffmann, B.; Mengel, K. Apoplastic pH and Fe3+ Reduction in Intact Sunflower Leaves. Plant Physiol. 1999, 121, 1069–1079. [Google Scholar] [CrossRef]
- Wang, Y.-Y.; Hsu, P.-K.; Tsay, Y.-F. Uptake, allocation and signaling of nitrate. Trends Plant Sci. 2012, 17, 458–467. [Google Scholar] [CrossRef]
- Feng, H.; Yan, M.; Fan, X.; Li, B.; Shen, Q.; Miller, A.J.; Xu, G. Spatial expression and regulation of rice high-affinity nitrate transporters by nitrogen and carbon status. J. Exp. Bot. 2011, 62, 2319–2332. [Google Scholar] [CrossRef]
- Krouk, G.; Lacombe, B.; Bielach, A.; Perrine-Walker, F.; Malinska, K.; Mounier, E.; Hoyerova, K.; Tillard, P.; Leon, S.; Ljung, K.; et al. Nitrate-Regulated Auxin Transport by NRT1.1 Defines a Mechanism for Nutrient Sensing in Plants. Dev. Cell 2010, 18, 927–937. [Google Scholar] [CrossRef]
- Zhu, X.F.; Dong, X.Y.; Wu, Q.; Shen, R.F. Ammonium regulates Fe deficiency responses by enhancing nitric oxide signaling in Arabidopsis thaliana. Planta 2019, 250, 1089–1102. [Google Scholar] [CrossRef]
- Wei, H.; Manivannan, A.; Chen, Y.; Jeong, B.R. Effect of Different Cultivation Systems on the Accumulation of Nutrients and Phytochemicals in Ligularia fischeri. Hortic. Plant J. 2018, 4, 24–29. [Google Scholar] [CrossRef]
- Dere, Ş.; Güneş, T.; Sivaci, R. Spectrophotometric determination of chlorophyll-A, B and total carotenoid contents of some algae species using different solvents. Turk. J. Bot. 1998, 22, 13–18. [Google Scholar]
- Pirie, A.; Mullins, M.G. Changes in anthocyanin and phenolics content of grapevine leaf and fruit tissues treated with sucrose, nitrate, and abscisic acid. Plant Physiol. 1976, 58, 468–472. [Google Scholar] [CrossRef] [PubMed]
- Bienfait, H.F.; Van den Briel, W.; Mesland-Mul, N.T. Free space iron pools in roots: Generation and mobilization. Plant Physiol. 1985, 78, 596–600. [Google Scholar] [CrossRef]
- Chaney, R.L.; Brown, J.C.; Tiffin, L.O. Obligatory Reduction of Ferric Chelates in Iron Uptake by Soybeans. Plant Physiol. 1972, 50, 208–213. [Google Scholar] [CrossRef]
- Manivannan, A.; Soundararajan, P.; Halimah, N.; Ko, C.H.; Jeong, B.R. Blue LED light enhances growth, phytochemical contents, and antioxidant enzyme activities of Rehmannia glutinosa cultured in vitro. Hortic. Environ. Biotechnol. 2015, 56, 105–113. [Google Scholar] [CrossRef]
- Bräutigam, A.; Gagneul, D.; Weber, A.P. High-throughput colorimetric method for the parallel assay of glyoxylic acid and ammonium in a single extract. Anal. Biochem. 2007, 362, 151–153. [Google Scholar] [CrossRef]
- Huang, L.; Li, M.; Zhou, K.; Sun, T.; Hu, L.; Li, C.; Ma, F. Uptake and metabolism of ammonium and nitrate in response to drought stress in Malus prunifolia. Plant Physiol. Biochem. 2018, 127, 185–193. [Google Scholar] [CrossRef]
- Zhang, H.; Dotson, P. The use of microwave muffle furnace for dry ashing plant tissue samples. Commun. Soil Sci. Plant Anal. 1994, 25, 1321–1327. [Google Scholar] [CrossRef]
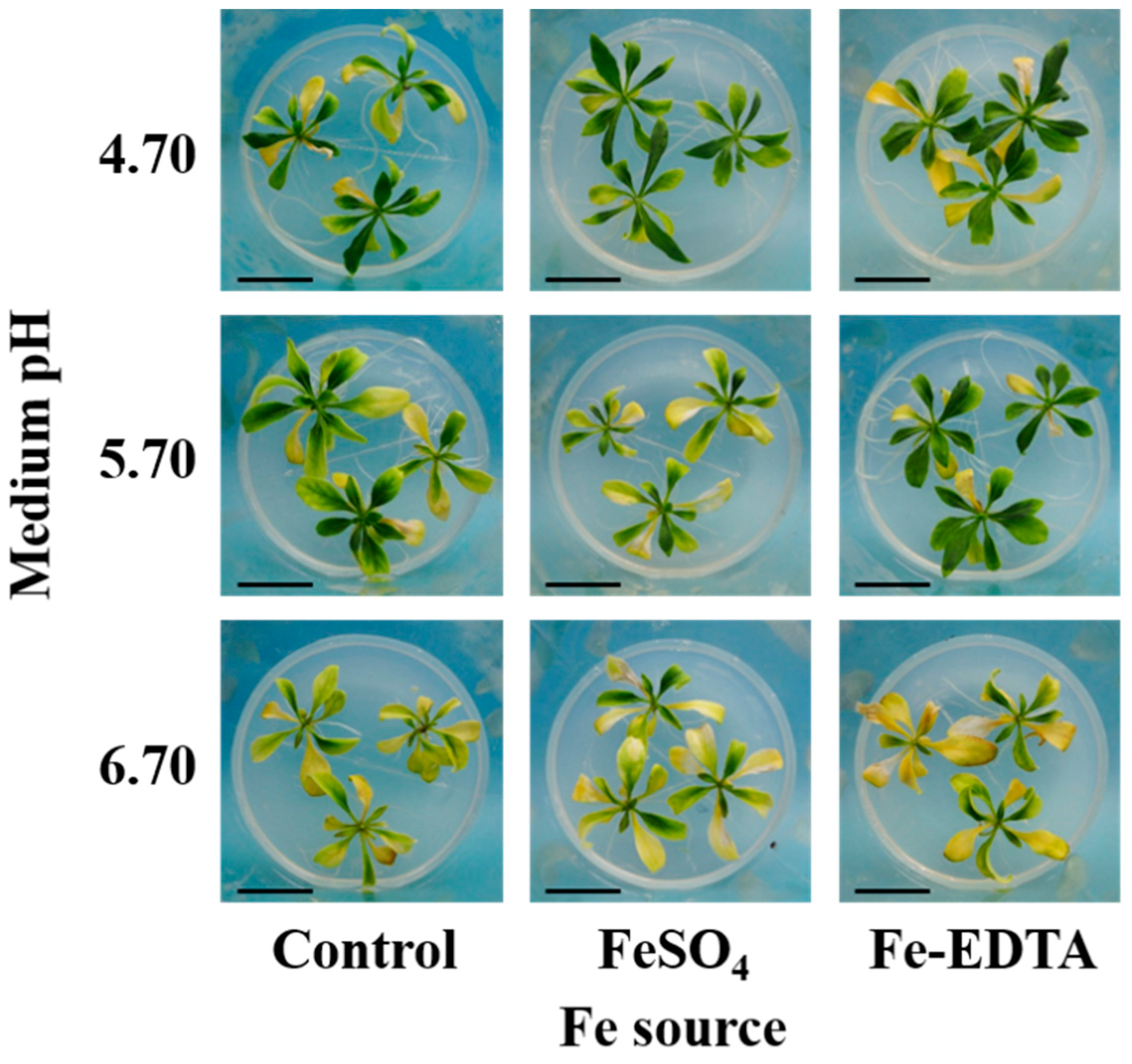

| Fe Source (A) | pH (B) | Leaf | Shoot | Root | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Number | Length (mm) | Width (mm) | Length (mm) | Fresh Weight (mg) | Dry Weight (mg) | Length (mm) | Fresh Weight (mg) | Dry Weight (mg) | ||
| Control | 4.70 | 9.3 z a y | 30.0 ab | 6.0 c | 4.0 bc | 141.0 cd | 13.7 f | 49.0 b | 57.5 c | 2.3 c |
| 5.70 | 8.7 ab | 21.3 c | 7.8 a | 6.3 a | 236.7 a | 17.1 de | 49.5 b | 70.0 b | 3.0 c | |
| 6.70 | 5.7 de | 26.0 bc | 7.7 ab | 3.8 cd | 128.5 d | 17.3 de | 21.5 e | 18.3 e | 2.6 c | |
| FeSO4 | 4.70 | 9.0 ab | 25.7 bc | 5.7 c | 4.7 bc | 255.0 a | 18.3 cd | 42.7 c | 55.3 c | 4.2 b |
| 5.70 | 7.7 c | 25.0 bc | 8.7 a | 3.7 cd | 165.5 cd | 20.5 bc | 29.0 d | 65.5 b | 2.3 c | |
| 6.70 | 6.3 cd | 25.3 bc | 7.8 a | 3.5 cde | 232.0 a | 34.1 a | 32.0 d | 21.8 e | 1.3 d | |
| Fe-EDTA | 4.70 | 8.7 ab | 25.5 bc | 7.3 abc | 3.3 cde | 253.0 a | 21.4 bc | 44.0 c | 69.5 b | 3.8 b |
| 5.70 | 8.7 ab | 32.0 a | 5.7 c | 3.2 de | 200.5 b | 14.8 ef | 55.0 a | 118.6 a | 6.1 a | |
| 6.70 | 4.3 e | 25.0 bc | 8.5 a | 2.8 e | 130.0 d | 17.2 de | 32.5 d | 41.0 d | 2.5 c | |
| F-test x | A | NS | NS | NS | *** | *** | *** | *** | *** | *** |
| B | *** | NS | ** | *** | *** | *** | *** | *** | *** | |
| A × B | NS | * | ** | *** | *** | *** | *** | *** | *** | |
| Fe Source (A) | pH (B) | Red Attribute | Green Attribute | Blue Attribute |
|---|---|---|---|---|
| Control | 4.70 | 115.0 z d y | 126.5 cd | 79.5 d |
| 5.70 | 153.0 c | 165.0 b | 132.5 ab | |
| 6.70 | 209.5 a | 210.7 a | 136.5 a | |
| FeSO4 | 4.70 | 123.5 d | 178.7 b | 113.7 bc |
| 5.70 | 172.0 b | 139.5 c | 112.5 bc | |
| 6.70 | 221.0 a | 210.3 a | 130.5 abc | |
| Fe-EDTA | 4.70 | 115.0 d | 114.0 d | 76.5 d |
| 5.70 | 128.0 d | 126.5 cd | 85.5 d | |
| 6.70 | 159.0 b | 165.0 b | 110.5 c | |
| F-test x | A | *** | *** | *** |
| B | *** | *** | *** | |
| A × B | *** | *** | ** |
| Correlation | Red Attribute | Green Attribute | Blue Attribute | Total Chlorophyll | Carotenoid |
|---|---|---|---|---|---|
| Green attribute | 0.944 ** z | - | - | - | - |
| Blue attribute | 0.735 ** | 0.819 ** | - | - | - |
| Total chlorophyll | −0.717 ** | −0.689 ** | −0.405 * | - | - |
| Carotenoid | −0.790 ** | −0.748 ** | −0.483 * | 0.974 ** | - |
| Anthocyanin | −0.781 ** | −0.737 ** | −0.441 * | 0.957 ** | 0.967 ** |
| Fe Source (A) | pH (B) | Macronutrients (mg·g−1 DW) | Micronutrients (mg·g−1 DW) | |||||||
|---|---|---|---|---|---|---|---|---|---|---|
| P | K | S | Ca | Mg | Cu | Mn | Zn | Fe | ||
| Control | 4.70 | 102.7 z b y | 2029.2 b | 103.3 c | 459.6 b | 109.0 c | 5.93 a | 0.83 b | 2.01 a | 7.00 b |
| 5.70 | 86.4 d | 1500.9 d | 567.1 a | 295.7 de | 88.0 e | 3.25 cd | 0.66 c | 1.34 cd | 5.93 cd | |
| 6.70 | 99.8 bc | 1000.5 e | 306.9 b | 309.8 d | 118.7 b | 5.02 ab | 0.53 d | 1.81 ab | 3.56 e | |
| FeSO4 | 4.70 | 91.8 cd | 1834.4 c | 117.5 c | 362.8 c | 92.4 de | 4.12 bc | 0.80 b | 1.27 de | 6.36 bc |
| 5.70 | 104.3 b | 1003.0 e | 98.7 c | 282.9 de | 70.3 f | 3.91 bcd | 0.64 c | 0.97 f | 3.68 e | |
| 6.70 | 73.5 e | 652.8 f | 62.9 c | 213.4 f | 68.7 f | 2.86 d | 0.31 e | 0.66 g | 3.16 e | |
| Fe-EDTA | 4.70 | 105.6 b | 1827.1 c | 96.0 c | 389.1 c | 99.9 cd | 4.62 b | 0.80 b | 1.10 def | 5.07 d |
| 5.70 | 133.1 a | 2286.9 a | 150.2 c | 686.6 a | 153.9 a | 4.41 bc | 1.47 a | 1.01 ef | 9.09 a | |
| 6.70 | 69.3 e | 727.0 f | 89.3 c | 265.2 e | 72.0 f | 4.23 bc | 0.36 e | 1.58 bc | 5.92 cd | |
| F-test x | A | *** | *** | *** | *** | *** | *** | ** | *** | *** |
| B | *** | *** | *** | *** | *** | ** | ** | NS | *** | |
| A × B | *** | *** | *** | *** | *** | *** | ** | *** | *** | |
| Correlation | Chlorophyll | Carotenoid | Anthocyanin |
|---|---|---|---|
| Carotenoid | 0.976 ** z | - | - |
| Anthocyanin | 0.961 ** | 0.967 ** | - |
| Fe | 0.420 * | 0.372 | 0.403 * |
| Correlation | Leaf Number | Shoot | Zn | Cu | Fe | Mn | Ca | Mg | K | P | S | NH4+ | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Length | FW | DW | ||||||||||||
| Length | 0.367 z | - | - | - | - | - | - | - | - | - | - | - | - | - |
| FW | 0.458 * | 0.370 | - | - | - | - | - | - | - | - | - | - | - | - |
| DW | −0.253 | −0.147 | 0.416 * | - | ||||||||||
| Zn | −0.040 | 0.083 | 0.556 ** | −0.639 ** | - | - | - | - | - | - | - | - | - | - |
| Cu | 0.160 | −0.247 | −0.424 * | −0.523 ** | 0.667 ** | - | - | - | - | - | - | - | - | - |
| Fe | 0.178 | −0.320 | −0.271 | −0.649 ** | 0.346 | 0.516 ** | - | - | - | - | - | - | - | - |
| Mn | 0.600 ** | −0.030 | 0.178 | −0.533 ** | −0.048 | 0.259 | 0.731 ** | - | - | - | - | - | - | - |
| Ca | 0.496 ** | −0.171 | 0.015 | −0.553 ** | 0.058 | 0.354 | 0.845 ** | 0.960 ** | - | - | - | - | - | - |
| Mg | 0.336 | −0.102 | −0.081 | −0.535 ** | 0.218 | 0.397 * | 0.803 ** | 0.828 ** | 0.883 ** | - | - | - | - | - |
| K | 0.784 ** | 0.160 | 0.286 | −0.566 ** | 0.136 | 0.398 * | 0.655 ** | 0.881 ** | 0.848 ** | 0.714 ** | - | - | - | - |
| P | 0.524 ** | −0.138 | −0.009 | −0.461 * | −0.024 | 0.319 | 0.634 ** | 0.883 ** | 0.851 ** | 0.813 ** | 0.724 ** | - | - | - |
| S | 0.112 | 0.748 ** | 0.115 | −0.280 | 0.228 | −0.259 | −0.224 | 0.012 | −0.083 | 0.130 | 0.030 | −0.026 | - | - |
| NH4+ | 0.317 | 0.157 | 0.292 | −0.069 | 0.132 | 0.161 | 0.222 | 0.075 | 0.034 | −0.019 | 0.299 | −0.080 | −0.272 | - |
| NO3− | 0.004 | −0.164 | 0.352 | 0.276 | −0.380 | −0.190 | 0.119 | 0.295 | 0.317 | 0.485 * | 0.157 | 0.296 | 0.154 | −0.398 * |
| Gene Name | Abbreviation | Locus ID |
|---|---|---|
| Ferric reduction oxidase 1 | FRO1 | AT1GO1590 |
| Ferric reduction oxidase 8 | FRO8 | AT5G50160 |
| Iron-regulated transporter 1 | IRT1 | AT4G19690 |
| Nitrate transporter 2.5 | NRT2.5 | AT1G12940 |
| Ammonium transporter 1;1 | AMT1;1 | AT4G13510 |
| Myb domain protein 113 | MYB113 | AT1G66370 |
| Deoxyhypusine synthase | DHS | AT5G05920 |
| Name | Gene Identifier | Forward (5′–3′) | Reverse (5′–3′) |
|---|---|---|---|
| PhFRO1 | Peaxi162Scf41243 | CGTCTCACAGGTCAAAACCC | TTGCTGGTTGGATTGATATTGTGG |
| PhFRO8 | Peaxi162Scf00089 | TCAGTCTTCTTGCAGGATTGAT | TGGAAGGAAAGATTCGAACAGA |
| PhIRT1 | Peaxi162Scf00623 | GCTGGGAATCATTGTTCACTC | CACCAAGTCCCATTCCTTCAAAC |
| PhNRT2.5 | Peaxi162Scf00515 | GGTGTTGAACTTACTGTGGACAA | CAAAAGTGAGGCCACAGGCA |
| PhAMT1;1 | Peaxi162Scf00043 | CCATAAACCAGCAATGCCACC | TGCAGGATCAGTTAGAGCCAA |
| PhMYB113 | Peaxi162Scf00102 | GGTGTGGAAAGAGTTGTAGGC | TGTCAGTTCGACCTGGTAATC |
| PhDHS | Peaxi162Scf00178 | TGGCACCACCACGTATTTTTC | CTGTCCATGCTGGTTCAAGG |
| PhActin11 | cn1159 | TGCACTCCCACATGCTATCCT | TCAGCCGAAGTGGTGAAAGAG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guo, G.; Xiao, J.; Jeong, B.R. Iron Source and Medium pH Affect Nutrient Uptake and Pigment Content in Petunia hybrida ‘Madness Red’ Cultured In Vitro. Int. J. Mol. Sci. 2022, 23, 8943. https://doi.org/10.3390/ijms23168943
Guo G, Xiao J, Jeong BR. Iron Source and Medium pH Affect Nutrient Uptake and Pigment Content in Petunia hybrida ‘Madness Red’ Cultured In Vitro. International Journal of Molecular Sciences. 2022; 23(16):8943. https://doi.org/10.3390/ijms23168943
Chicago/Turabian StyleGuo, Ge, Jie Xiao, and Byoung Ryong Jeong. 2022. "Iron Source and Medium pH Affect Nutrient Uptake and Pigment Content in Petunia hybrida ‘Madness Red’ Cultured In Vitro" International Journal of Molecular Sciences 23, no. 16: 8943. https://doi.org/10.3390/ijms23168943
APA StyleGuo, G., Xiao, J., & Jeong, B. R. (2022). Iron Source and Medium pH Affect Nutrient Uptake and Pigment Content in Petunia hybrida ‘Madness Red’ Cultured In Vitro. International Journal of Molecular Sciences, 23(16), 8943. https://doi.org/10.3390/ijms23168943







