Mitogen-Activated Protein Kinases Associated Sites of Tobacco Repression of Shoot Growth Regulates Its Localization in Plant Cells
Abstract
1. Introduction
2. Results
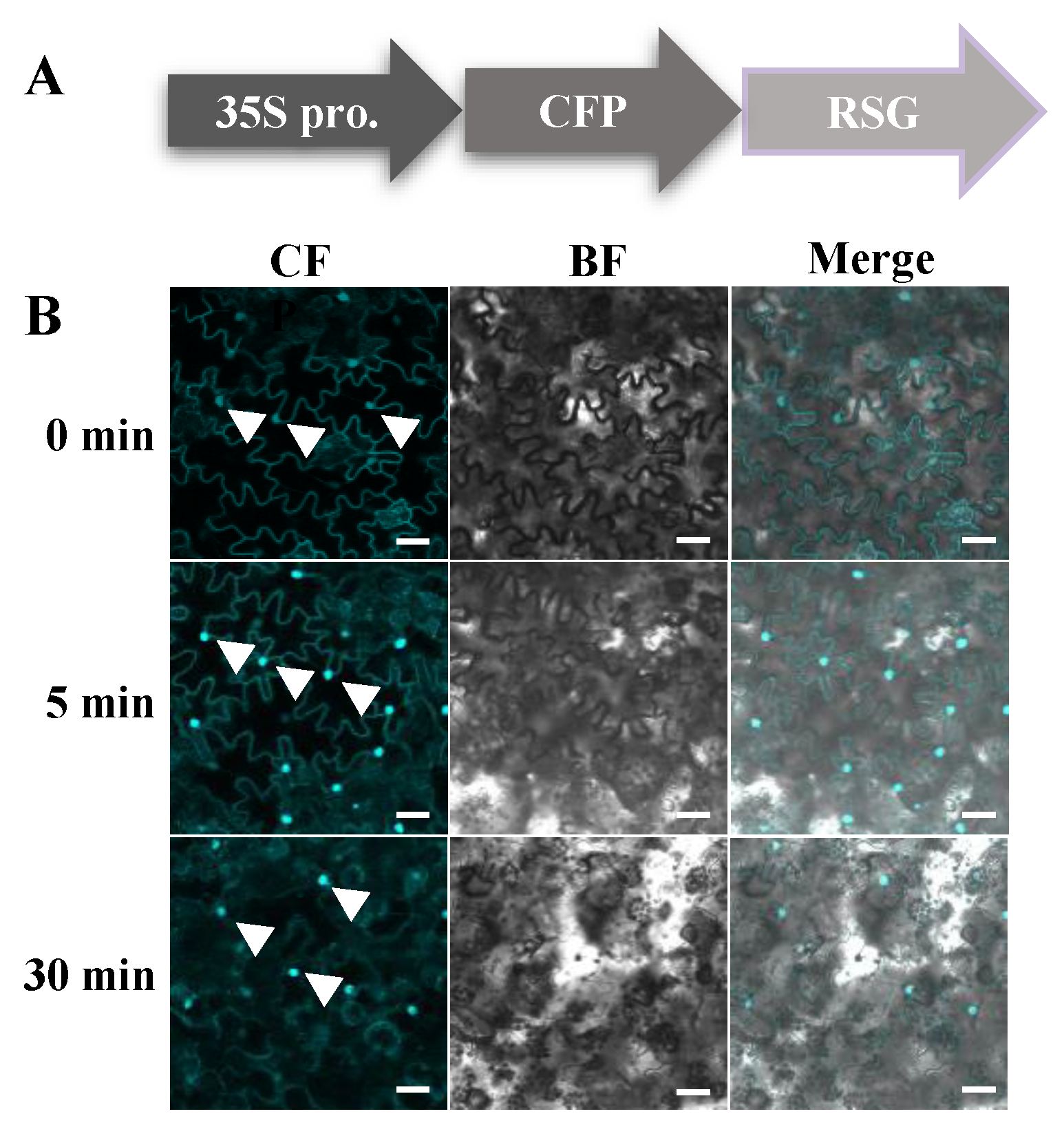
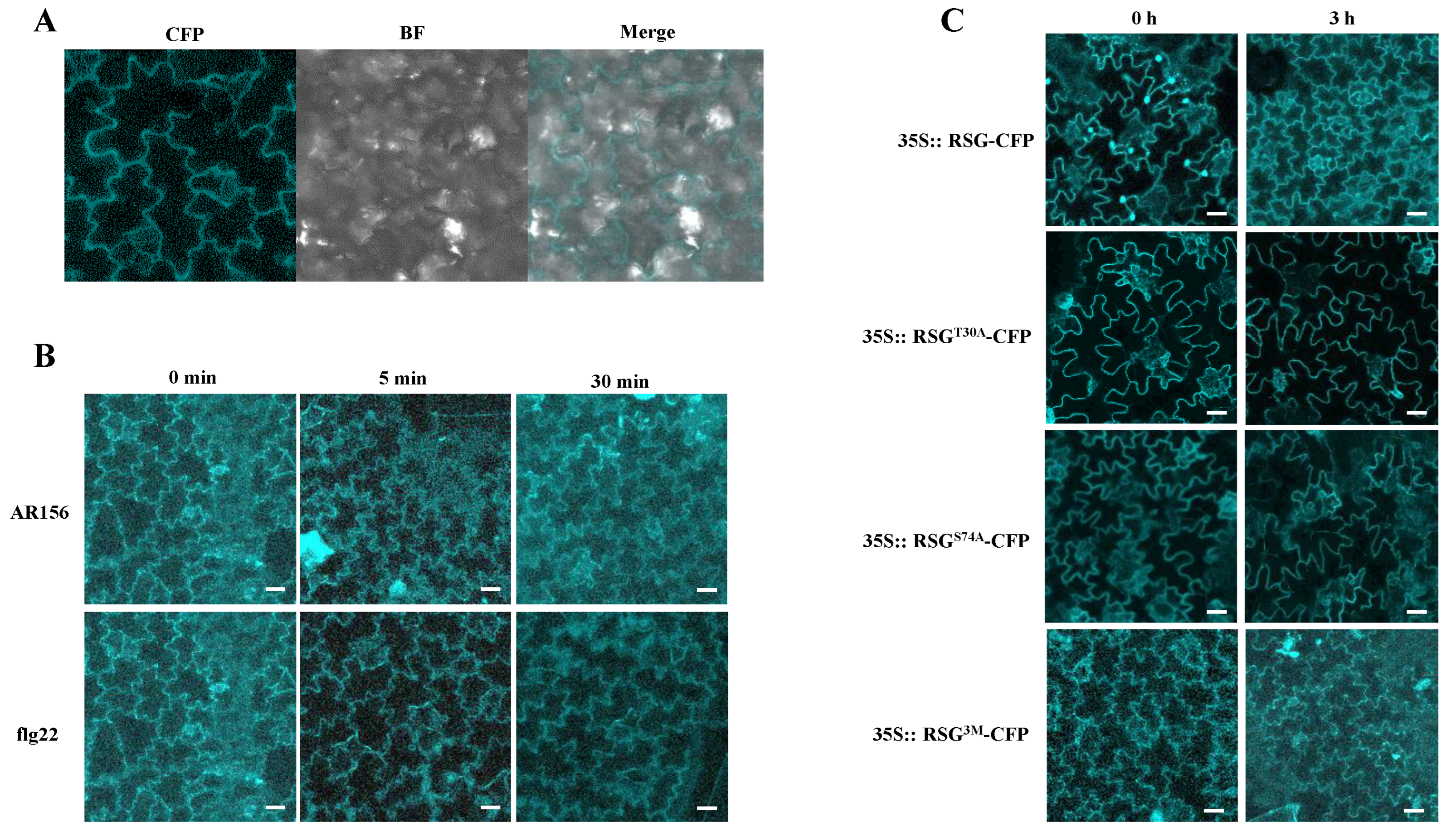
2.1. Bacillus cereus AR156 Induces Nuclear Transport of RSG
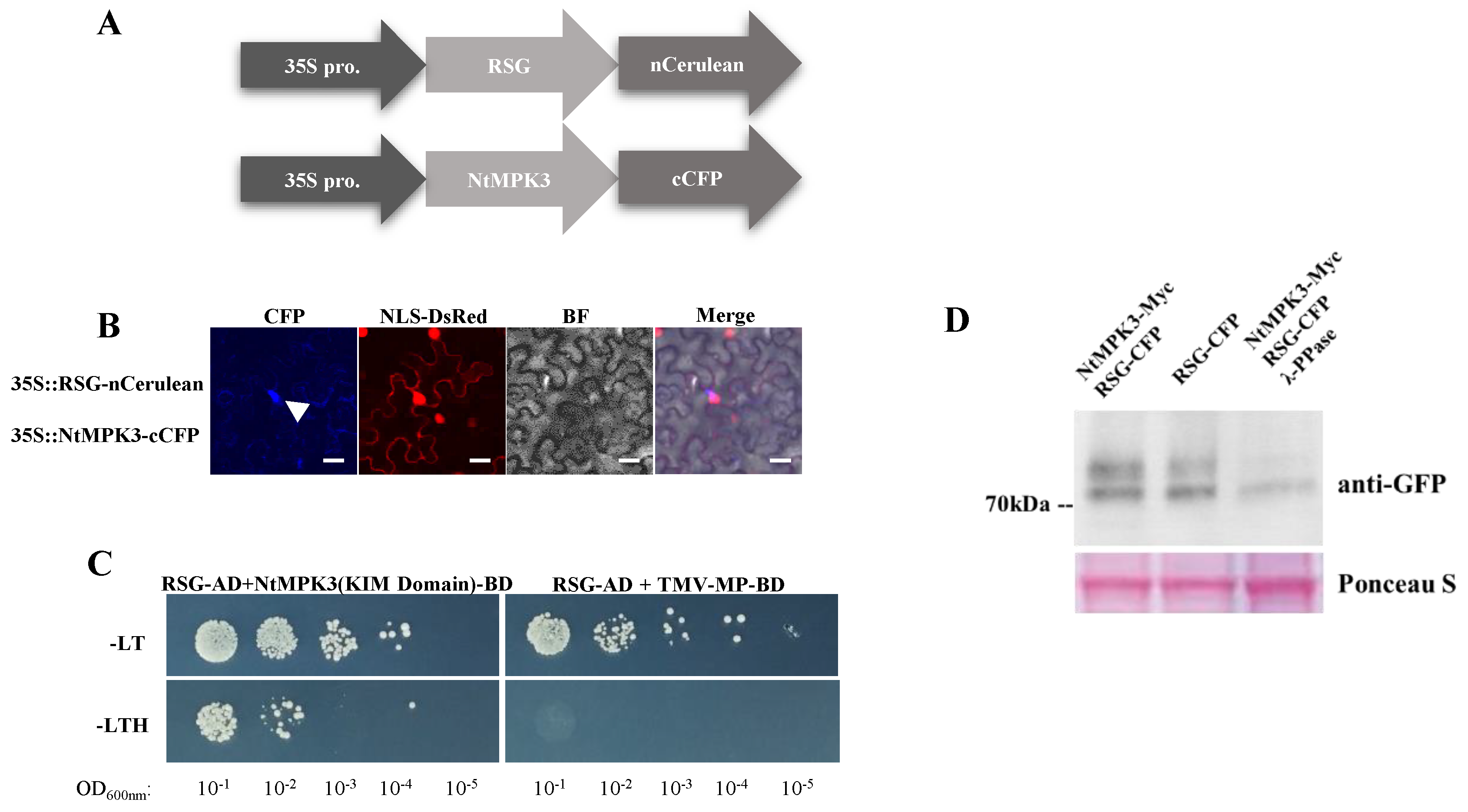
2.2. NtMPK3 Interacts with RSG as a Potential RSG Kinase
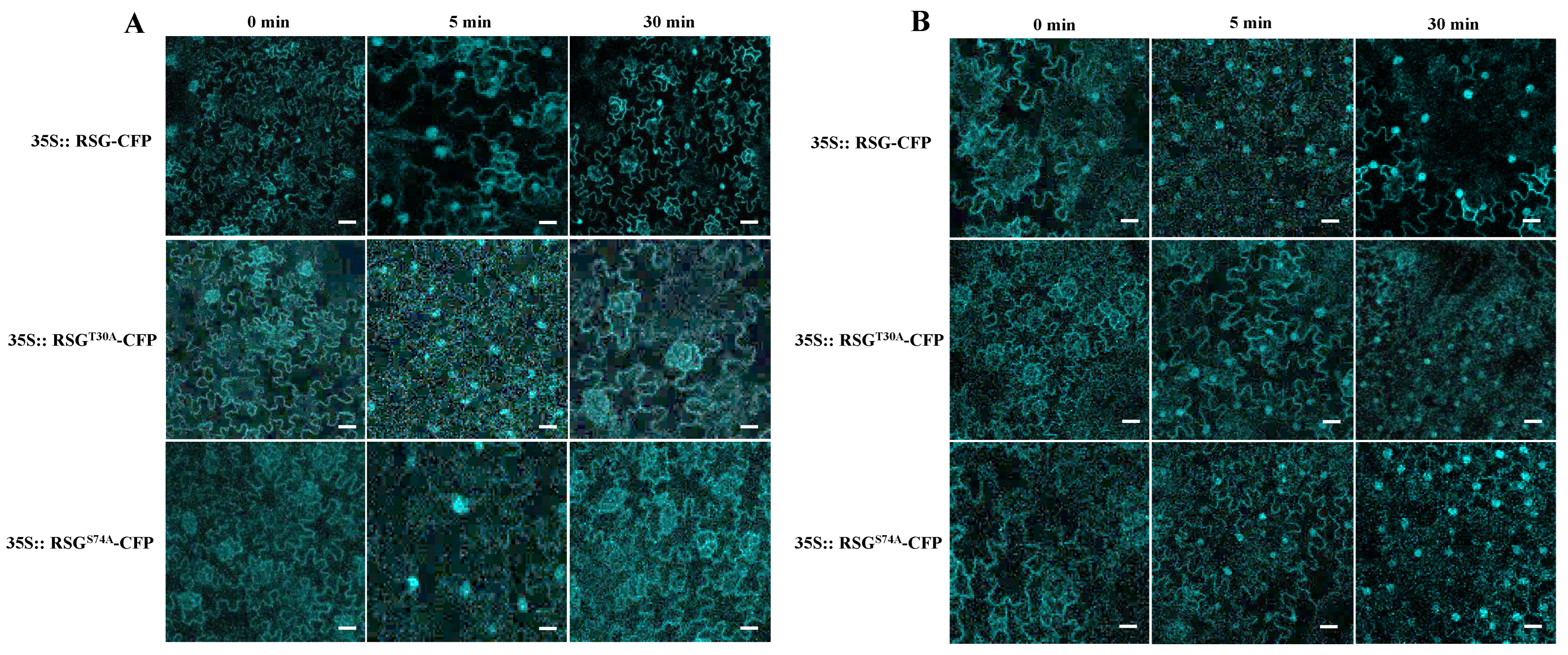
2.3. MAPK-Associated Phosphorylation Sites in RSG Regulates Its Intracellular Localization with NtMPK3
2.4. RSGT30A and RSGS74A Show Durable Localization in Tobacco Nuclear after B. cereus AR156 Treatment
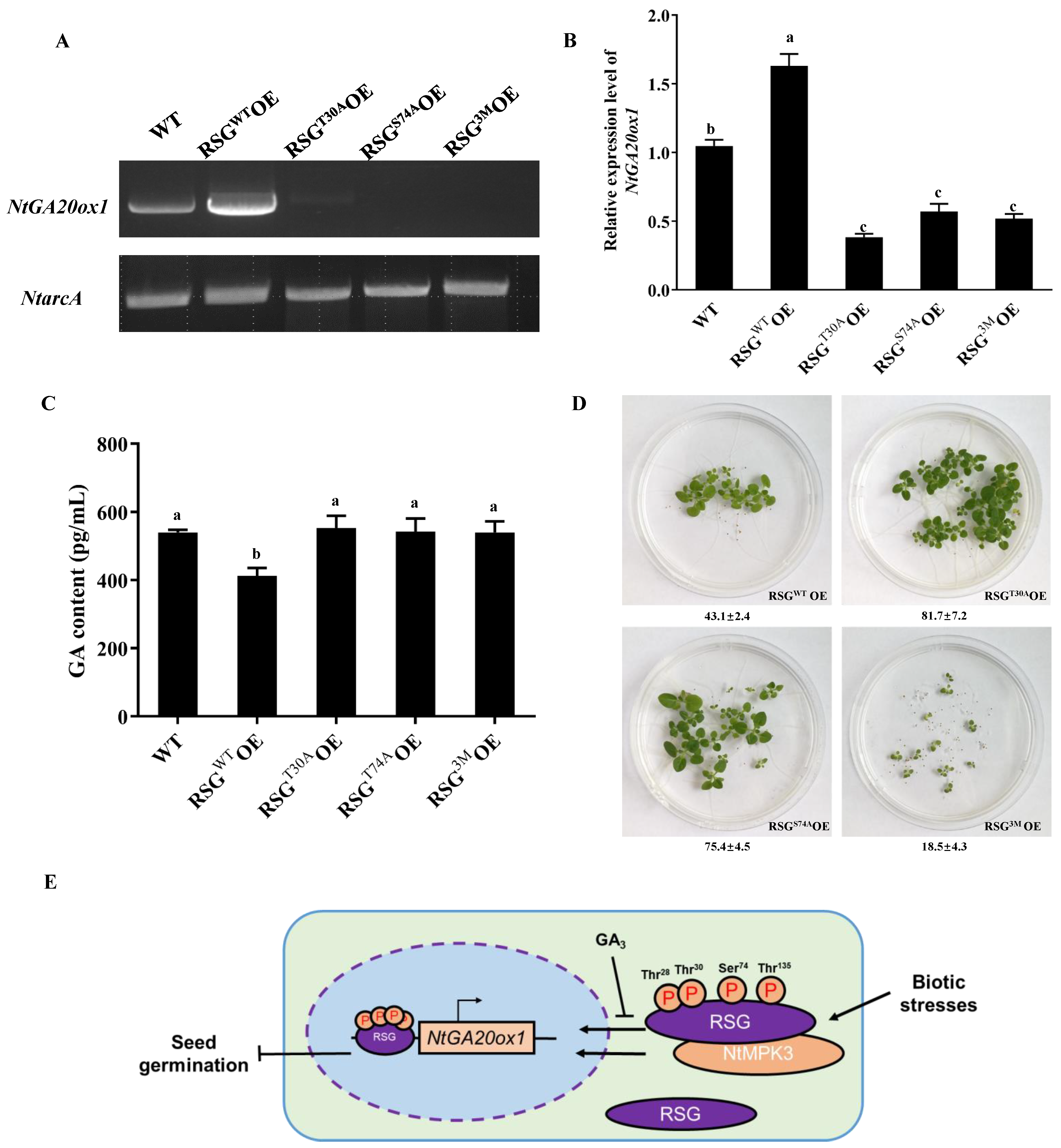
2.5. Mimic Constant MAPK-Associated Unphosphorylation in RSG Leads Pre-Dominant Cytoplasm Localization
2.6. Constant MAPK-Associated Phosphorylation in RSG Reduces Expression of Downstream Genes
3. Discussion
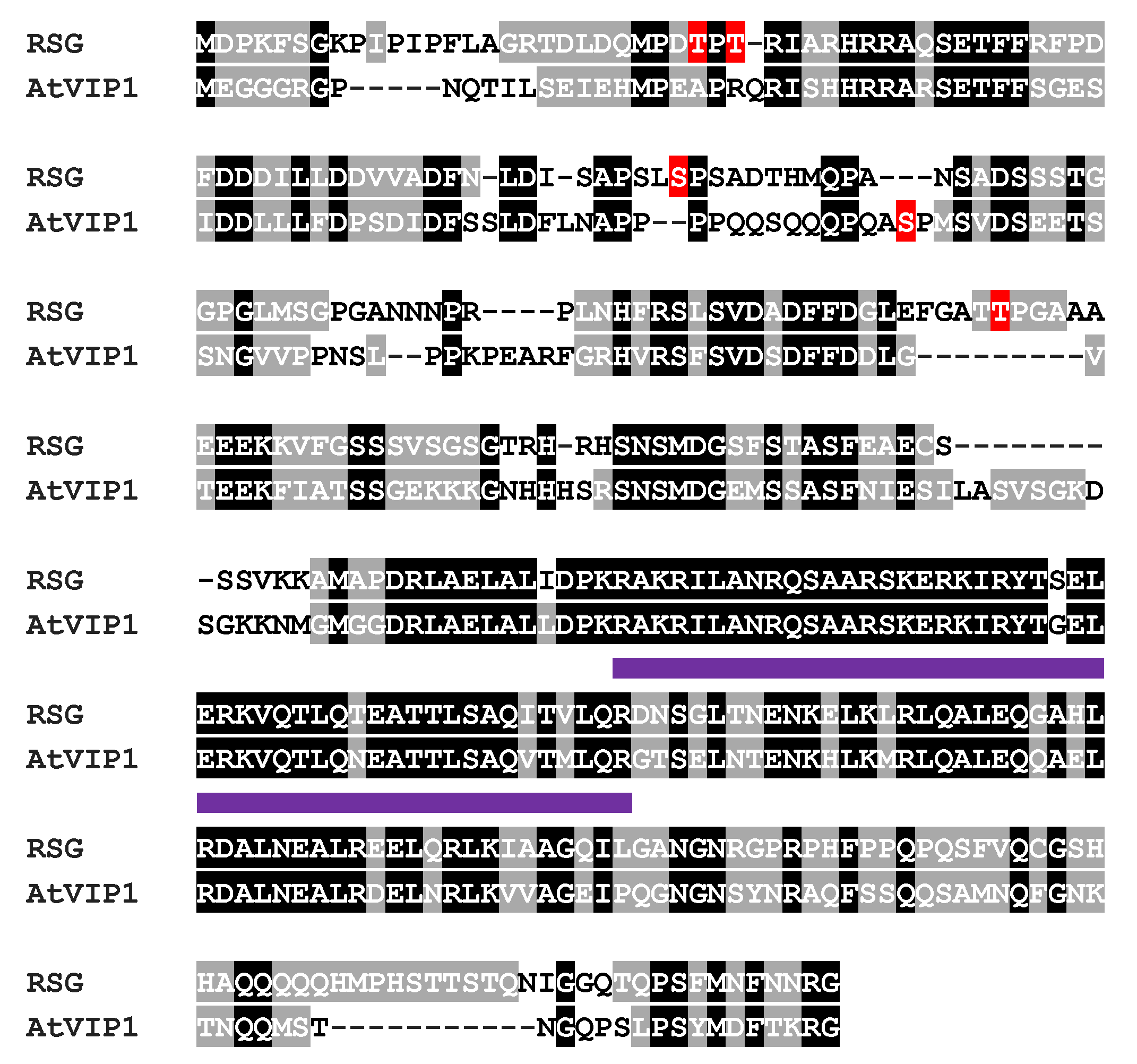
3.1. Biological Functions of RSG in Tobacco and VIP1 in Arabidopsis
3.2. Intracellular Movement of RSG under Biotic Stresses
3.3. Biological Roles of MAPK-Associated Phosphorylation Sites in RSG
4. Material and Methods
4.1. Bacterial Strains, Plants, and Growth Conditions
4.2. Plasmid Construction and Mutagenesis
4.3. Yeast-Two-Hybrid Protein Interaction Assay
4.4. Biomolecular Fluorescence Complementation Assay
4.5. Semi-Quantitative PCR
4.6. Gibberellin Content Assay
4.7. Confocal Fluorescence Microscopy
4.8. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Matsuoka, M. Gibberellin signaling: How do plant cells respond to GA signals? J. Plant Growth Regul. 2003, 22, 123–125. [Google Scholar] [CrossRef]
- Fukazawa, J.; Nakata, M.; Ito, T.; Yamaguchi, S.; Takahashi, Y. The transcription factor RSG regulates negative feedback of NtGA20ox1 encoding GA 20-oxidase. Plant J. 2010, 62, 1035–1045. [Google Scholar] [PubMed]
- Ishida, S.; Fukazawa, J.; Yuasa, T.; Takahashi, Y. Involvement of 14-3-3 signaling protein binding in the functional regulation of the transcriptional activator repression of shoot growth by gibberellins. Plant Cell 2004, 16, 2641–2651. [Google Scholar] [CrossRef] [PubMed]
- Ishida, S.; Yuasa, T.; Nakata, M.; Takahashi, Y. A tobacco calcium-dependent protein kinase, CDPK1, regulates the transcription factor repression of shoot growth in response to gibberellins. Plant Cell 2008, 20, 3273–3288. [Google Scholar] [CrossRef]
- Andreasson, E.; Ellis, B. Convergence and specificity in the Arabidopsis MAPK nexus. Trends Plant Sci. 2010, 15, 106–113. [Google Scholar] [CrossRef]
- Cheong, Y.H.; Kim, M.C. Functions of MAPK cascade pathways in plant defense signaling. Plant Pathol. J. 2010, 26, 101–109. [Google Scholar] [CrossRef][Green Version]
- Sinha, A.K.; Jaggi, M.; Raghuram, B.; Tuteja, N. Mitogen-activated protein kinase signaling in plants under abiotic stress. Plant Signal. Behav. 2011, 6, 196–203. [Google Scholar] [CrossRef]
- Meng, X.; Zhang, S. MAPK cascades in plant disease resistance signaling. Annu. Rev. Phytopathol. 2013, 51, 245–266. [Google Scholar] [CrossRef]
- Opdenakker, K.; Remans, T.; Vangronsveld, J.; Cuypers, A. Mitogen-activated protein (MAP) kinases in plant metal stress: Regulation and responses in comparison to other biotic and abiotic stresses. Int. J. Mol. Sci. 2012, 13, 7828–7853. [Google Scholar] [CrossRef]
- Pitzschke, A. Modes of MAPK substrate recognition and control. Trends Plant Sci. 2015, 20, 49–55. [Google Scholar] [CrossRef]
- Xu, J.; Zhang, S. Mitogen-activated protein kinase cascades in signaling plant growth and development. Trends Plant Sci. 2015, 20, 56–64. [Google Scholar] [CrossRef] [PubMed]
- Galletti, R.; Ferrari, S.; De Lorenzo, G. Arabidopsis MPK3 and MPK6 play different roles in basal and oligogalacturonide-or flagellin-induced resistance against Botrytis cinerea. Plant Physiol. 2011, 157, 804–814. [Google Scholar] [CrossRef] [PubMed]
- Meng, X.; Wang, H.; He, Y.; Liu, Y.; Walker, J.C.; Torii, K.U.; Zhang, S. A MAPK cascade downstream of ERECTA receptor-like protein kinase regulates Arabidopsis inflorescence architecture by promoting localized cell proliferation. Plant Cell 2012, 24, 4948–4960. [Google Scholar] [CrossRef] [PubMed]
- Ren, D.; Liu, Y.; Yang, K.-Y.; Han, L.; Mao, G.; Glazebrook, J.; Zhang, S. A fungal-responsive MAPK cascade regulates phytoalexin biosynthesis in Arabidopsis. Proc. Natl. Acad. Sci. USA 2008, 105, 5638–5643. [Google Scholar] [CrossRef]
- Tzfira, T.; Vaidya, M.; Citovsky, V. VIP1, an Arabidopsis protein that interacts with Agrobacterium VirE2, is involved in VirE2 nuclear import and Agrobacterium infectivity. EMBO J. 2001, 20, 3596–3607. [Google Scholar] [CrossRef] [PubMed]
- Djamei, A.; Pitzschke, A.; Nakagami, H.; Rajh, I.; Hirt, H. Trojan horse strategy in Agrobacterium transformation: Abusing MAPK defense signaling. Science 2007, 318, 453–456. [Google Scholar] [CrossRef] [PubMed]
- Pitzschke, A.; Djamei, A.; Teige, M.; Hirt, H. VIP1 response elements mediate mitogen-activated protein kinase 3-induced stress gene expression. Proc. Natl. Acad. Sci. USA 2009, 106, 18414–18419. [Google Scholar] [CrossRef] [PubMed]
- Tsugama, D.; Liu, S.; Takano, T. VIP1 is very important/interesting protein 1 regulating touch responses of Arabidopsis. Plant Signal. Behav. 2016, 11, e1187358. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tsugama, D.; Liu, S.; Takano, T. Analysis of functions of VIP1 and its close homologs in osmosensory responses of Arab. Thaliana. PLoS ONE 2014, 9, e103930. [Google Scholar]
- Jiang, C.-H.; Chen, Y.; Yan, F.; Fan, Z.-H.; Guo, J.-H. Whole-genome sequence of Bacillus cereus AR156, a potential biocontrol agent with high soilborne disease biocontrol efficacy and plant growth promotion. Genome Announc. 2017, 5, e00886-e17. [Google Scholar] [CrossRef]
- Niu, D.; Liu, H.; Jiang, C.; Wang, Y.; Wang, Q.; Jin, H.; Guo, J. The plant growth–promoting rhizobacterium Bacillus cereus AR156 induces systemic resistance in Arabidopsis thaliana by simultaneously activating salicylate-and jasmonate/ethylene-dependent signaling pathways. Mol. Plant-Microbe Interact. 2011, 24, 533–542. [Google Scholar] [CrossRef]
- Jiang, C.; Fan, Z.; Xie, P.; Guo, J. Bacillus cereus AR156 extracellular polysaccharides served as a novel micro-associated molecular pattern to induced systemic immunity to Pst DC3000 in Arabidopsis. Front. Microbiol. 2016, 7, 664. [Google Scholar] [CrossRef] [PubMed]
- Jayaswal, R.; Veluthambi, K.; Gelvin, S.B.; Slightom, J.L. Double-stranded cleavage of T-DNA and generation of single-stranded T-DNA molecules in Escherichia coli by a virD-encoded border-specific endonuclease from Agrobacterium tumefaciens. J. Bacteriol. 1987, 169, 5035–5045. [Google Scholar] [CrossRef]
- Igarashi, D.; Ishida, S.; Fukazawa, J.; Takahashi, Y. 14-3-3 proteins regulate intracellular localization of the bZIP transcriptional activator RSG. Plant Cell 2001, 13, 2483–2497. [Google Scholar] [CrossRef]
- Fukazawa, J.; Sakai, T.; Ishida, S.; Yamaguchi, I.; Kamiya, Y.; Takahashi, Y. Repression of shoot growth, a bZIP transcriptional activator, regulates cell elongation by controlling the level of gibberellins. Plant Cell 2000, 12, 901–915. [Google Scholar] [CrossRef]
- Fukazawa, J.; Nakata, M.; Ito, T.; Matsushita, A.; Yamaguchi, S.; Takahashi, Y. bZIP transcription factor RSG controls the feedback regulation of NtGA20ox1 via intracellular localization and epigenetic mechanism. Plant Signal. Behav. 2011, 6, 26–28. [Google Scholar] [CrossRef] [PubMed]
- Ito, T.; Nakata, M.; Fukazawa, J.; Ishida, S.; Takahashi, Y. Scaffold function of Ca2+—Dependent protein kinase: NtCDPK1 transfers 14-3-3 to the substrate RSG after phosphorylation. Plant Physiol. 2014, 165, 1737–1750. [Google Scholar]
- Tsugama, D.; Liu, S.; Takano, T. A bZIP protein, VIP1, is a regulator of osmosensory signaling in Arabidopsis. Plant Physiol. 2012, 159, 144–155. [Google Scholar] [CrossRef] [PubMed]
- Takeo, K.; Ito, T. Subcellular localization of VIP 1 is regulated by phosphorylation and 14-3-3 proteins. FEBS Lett. 2017, 591, 1972–1981. [Google Scholar] [CrossRef]
- Sheikh, A.H.; Raghuram, B.; Eschen-Lippold, L.; Scheel, D.; Lee, J.; Sinha, A.K. Agroinfiltration by cytokinin-producing Agrobacterium sp. strain GV3101 primes defense responses in Nicotiana tabacum. Mol. Plant-Microbe Interact. 2014, 27, 1175–1185. [Google Scholar] [CrossRef]
- Yaffe, M.B.; Rittinger, K.; Volinia, S.; Caron, P.R.; Aitken, A.; Leffers, H.; Gamblin, S.J.; Smerdon, S.J.; Cantley, L.C. The structural basis for 14-3-3: Phosphopeptide binding specificity. Cell 1997, 91, 961–971. [Google Scholar] [CrossRef]
- Nie, P.; Li, X.; Wang, S.; Guo, J.; Zhao, H.; Niu, D. Induced systemic resistance against Botrytis cinerea by Bacillus cereus AR156 through a JA/ET-and NPR1-dependent signaling pathway and activates PAMP-triggered immunity in Arabidopsis. Front. Plant Sci. 2017, 8, 238. [Google Scholar] [CrossRef] [PubMed]
- Jiang, C.; Huang, Z.; Xie, P.; Gu, C.; Li, K.; Wang, D.; Yu, Y.; Fan, Z.; Wang, C.; Wang, Y. Transcription factors WRKY70 and WRKY11 served as regulators in rhizobacterium Bacillus cereus AR156-induced systemic resistance to Pseudomonas syringae pv. tomato DC3000 in Arabidopsis. J. Exp. Bot. 2016, 67, 157–174. [Google Scholar] [CrossRef]
- Wang, L.; Lacroix, B.; Guo, J.; Citovsky, V. The Agrobacterium VirE2 effector interacts with multiple members of the Arabidopsis VIP1 protein family. Mol. Plant Pathol. 2018, 19, 1172–1183. [Google Scholar] [CrossRef]
- Sutton, A.; Bucaria, J.; Osley, M.A.; Sternglanz, R. Yeast ASF1 protein is required for cell cycle regulation of histone gene transcription. Genetics 2001, 158, 587–596. [Google Scholar] [CrossRef] [PubMed]
- Tzfira, T.; Tian, G.-W.; Lacroix, B.; Vyas, S.; Li, J.; Leitner-Dagan, Y.; Krichevsky, A.; Taylor, T.; Vainstein, A.; Citovsky, V. pSAT vectors: A modular series of plasmids for autofluorescent protein tagging and expression of multiple genes in plants. Plant Mol. Biol. 2005, 57, 503–516. [Google Scholar] [CrossRef]
- Ballas, N.; Citovsky, V. Nuclear localization signal binding protein from Arabidopsis mediates nuclear import of Agrobacterium VirD2 protein. Proc. Natl. Acad. Sci. USA 1997, 94, 10723–10728. [Google Scholar] [CrossRef]
- Hollenberg, S.M.; Sternglanz, R.; Cheng, P.F.; Weintraub, H. Identification of a new family of tissue-specific basic helix-loop-helix proteins with a two-hybrid system. Mol. Cell. Biol. 1995, 15, 3813–3822. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, L.; Gui, Y.; Yang, B.; Dong, W.; Xu, P.; Si, F.; Yang, W.; Luo, Y.; Guo, J.; Niu, D.; et al. Mitogen-Activated Protein Kinases Associated Sites of Tobacco Repression of Shoot Growth Regulates Its Localization in Plant Cells. Int. J. Mol. Sci. 2022, 23, 8941. https://doi.org/10.3390/ijms23168941
Wang L, Gui Y, Yang B, Dong W, Xu P, Si F, Yang W, Luo Y, Guo J, Niu D, et al. Mitogen-Activated Protein Kinases Associated Sites of Tobacco Repression of Shoot Growth Regulates Its Localization in Plant Cells. International Journal of Molecular Sciences. 2022; 23(16):8941. https://doi.org/10.3390/ijms23168941
Chicago/Turabian StyleWang, Luyao, Ying Gui, Bingye Yang, Wenpan Dong, Peiling Xu, Fangjie Si, Wei Yang, Yuming Luo, Jianhua Guo, Dongdong Niu, and et al. 2022. "Mitogen-Activated Protein Kinases Associated Sites of Tobacco Repression of Shoot Growth Regulates Its Localization in Plant Cells" International Journal of Molecular Sciences 23, no. 16: 8941. https://doi.org/10.3390/ijms23168941
APA StyleWang, L., Gui, Y., Yang, B., Dong, W., Xu, P., Si, F., Yang, W., Luo, Y., Guo, J., Niu, D., & Jiang, C. (2022). Mitogen-Activated Protein Kinases Associated Sites of Tobacco Repression of Shoot Growth Regulates Its Localization in Plant Cells. International Journal of Molecular Sciences, 23(16), 8941. https://doi.org/10.3390/ijms23168941





