Mechanisms of Systemic Osteoporosis in Rheumatoid Arthritis
Abstract
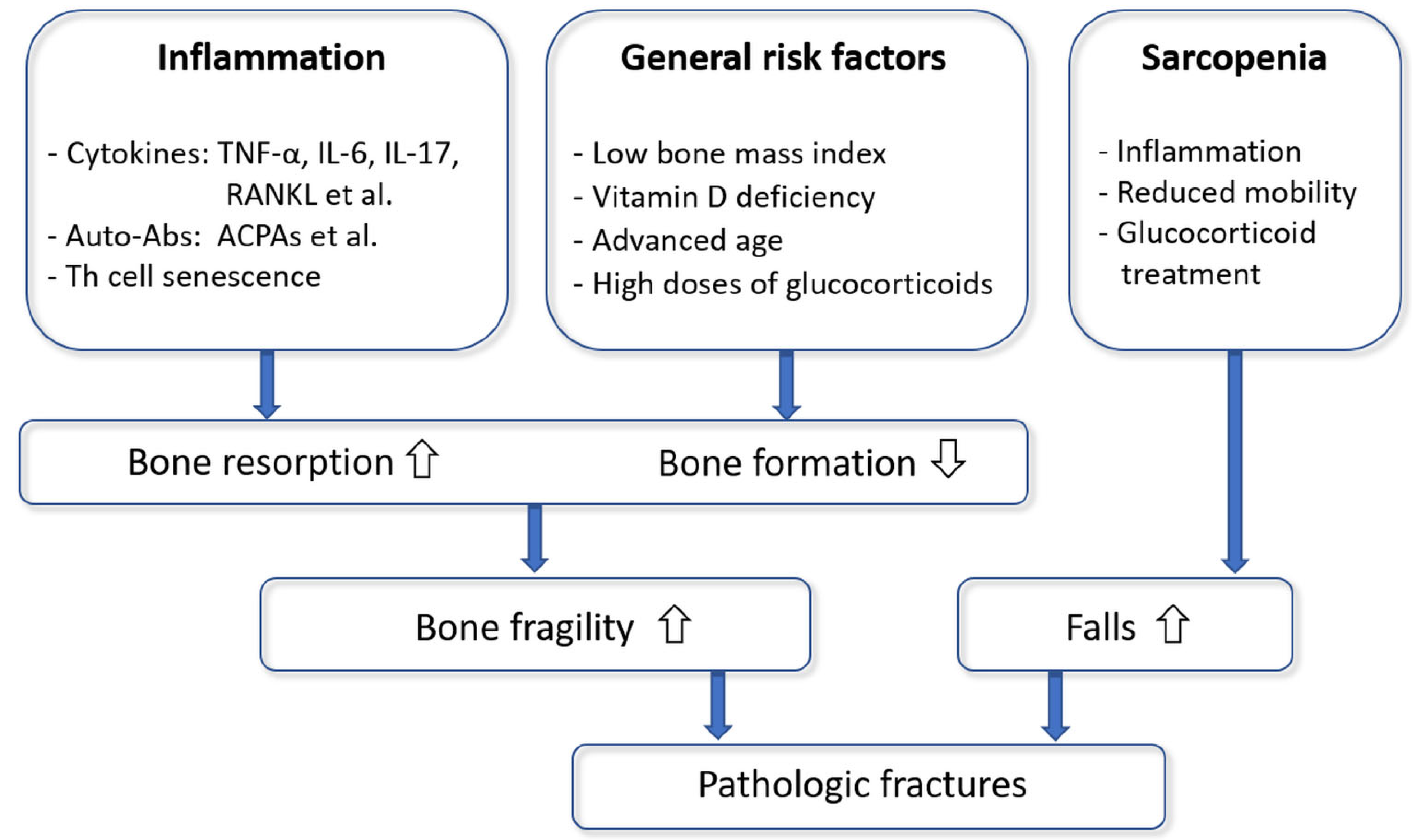
:1. Introduction
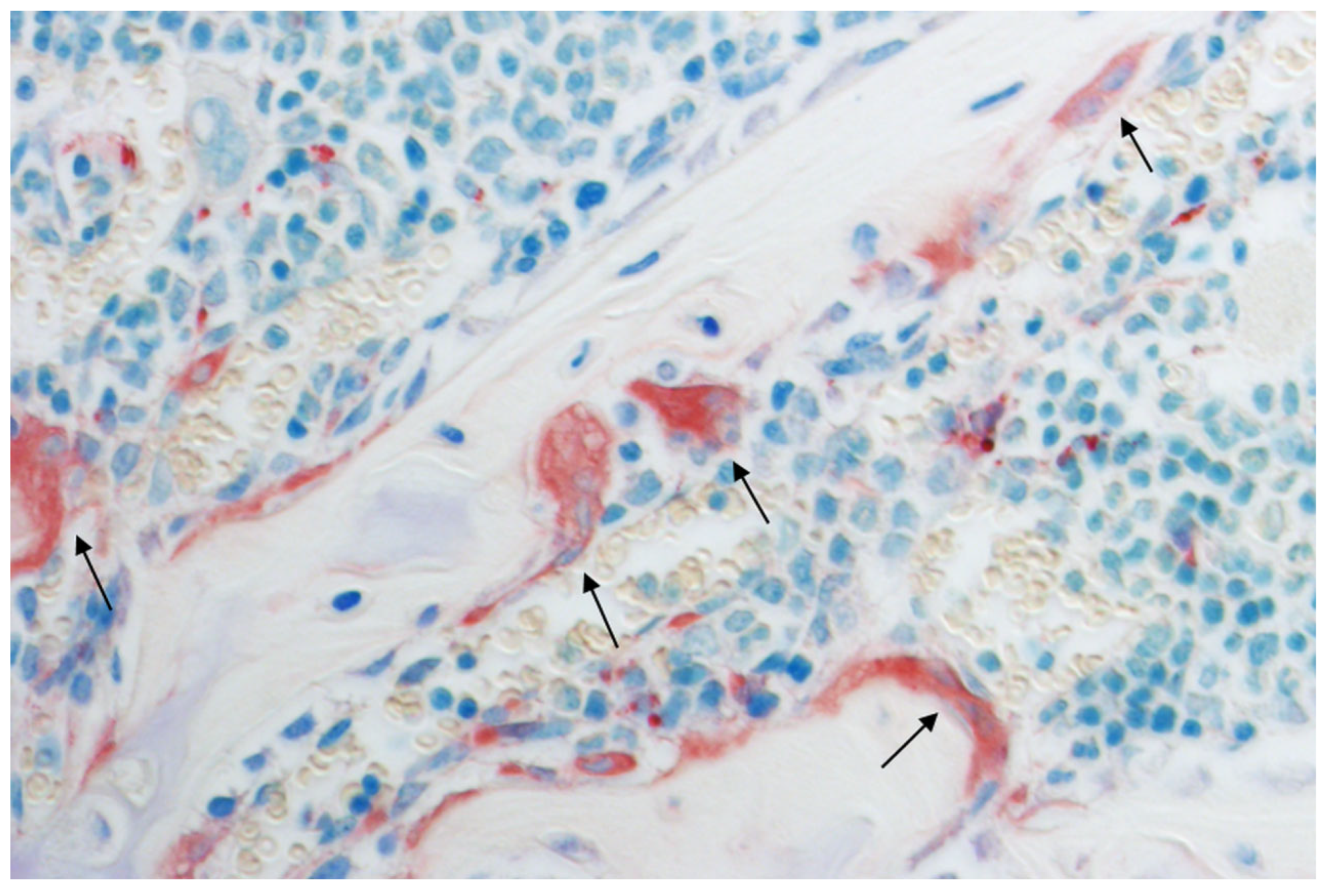
2. Animal Studies
3. Conclusions from Animal Studies
4. Human Association Studies
5. Proof of Concept: Human Pharmacologic Intervention Studies
5.1. Bone Mineral Density
5.2. Non-Vertebral Fracture Risk
5.3. Vertebral Fracture Risk
5.4. Fracture Healing
5.5. Sarcopenia
6. Non-Pharmacologic Intervention Studies
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- McInnes, I.B.; Schett, G. Pathogenetic insights from the treatment of rheumatoid arthritis. Lancet 2017, 389, 2328–2337. [Google Scholar] [CrossRef] [Green Version]
- Smolen, J.S.; Aletaha, D.; McInnes, I.B. Rheumatoid arthritis. Lancet 2016, 388, 2023–2038, Erratum in Lancet 2016, 388, 1984. [Google Scholar] [CrossRef]
- Omata, Y.; Frech, M.; Saito, T.; Schett, G.; Zaiss, M.M.; Tanaka, S. Inflammatory Arthritis and Bone Metabolism Regulated by Type 2 Innate and Adaptive Immunity. Int. J. Mol. Sci. 2022, 23, 1104. [Google Scholar] [CrossRef]
- NIH Consensus Development Panel on Osteoporosis Prevention, Diagnosis, and Therapy. Osteoporosis Prevention, Diagnosis, and Therapy. JAMA 2001, 285, 785–795. [Google Scholar] [CrossRef] [PubMed]
- De Sire, A.; Invernizzi, M.; Baricich, A.; Lippi, L.; Ammendolia, A.; Grassi, F.A.; Leigheb, M. Optimization of transdisciplinary management of elderly with femur proximal extremity fracture: A patient-tailored plan from orthopaedics to rehabilitation. World J. Orthop. 2021, 12, 456–466. [Google Scholar] [CrossRef] [PubMed]
- Jin, S.; Hsieh, E.; Peng, L.; Yu, C.; Wang, Y.; Wu, C.; Wang, Q.; Li, M.; Zeng, X. Incidence of fractures among patients with rheumatoid arthritis: A systematic review and meta-analysis. Osteoporos. Int. 2018, 29, 1263–1275. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Jiang, J.; Mo, M.; Sun, X.; Yu, K. Incidence and risk factors for vertebral fracture in rheumatoid arthritis: An update meta-analysis. Clin. Rheumatol. 2022, 41, 1313–1322. [Google Scholar] [CrossRef]
- Heinlen, L.; Humphrey, M.B. Skeletal complications of rheumatoid arthritis. Osteoporos. Int. 2017, 28, 2801–2812. [Google Scholar] [CrossRef]
- Kocijan, R.; Finzel, S.; Englbrecht, M.; Engelke, K.; Rech, J.; Schett, G. Decreased Quantity and Quality of the Periarticular and Nonperiarticular Bone in Patients with Rheumatoid Arthritis: A Cross-Sectional HR-pQCT Study. J. Bone Miner. Res. 2014, 29, 1005–1014. [Google Scholar] [CrossRef]
- Jaschke, N.; Sipos, W.; Hofbauer, L.C.; Rachner, T.D.; Rauner, M. Skeletal endocrinology: Where evolutionary advantage meets disease. Bone Res. 2021, 9, 28. [Google Scholar] [CrossRef]
- Föger-Samwald, U.; Kerschan-Schindl, K.; Butylina, M.; Pietschmann, P. Age Related Osteoporosis: Targeting Cellular Senescence. Int. J. Mol. Sci. 2022, 23, 2701. [Google Scholar] [CrossRef]
- Föger-Samwald, U.; Dovjak, P.; Azizi-Semrad, U.; Kerschan-Schindl, K.; Pietschmann, P. Osteoporosis: Patho-physiology and therapeutic options. EXCLI J. 2020, 19, 1017–1037. [Google Scholar]
- McDonald, M.M.; Kim, A.S.; Mulholland, B.S.; Rauner, M. New Insights into Osteoclast Biology. JBMR Plus 2021, 5, e10539. [Google Scholar] [CrossRef]
- Pietschmann, P.; Mechtcheriakova, D.; Meshcheryakova, A.; Föger-Samwald, U.; Ellinger, I. Immunology of Osteoporosis: A Mini-Review. Gerontology 2016, 62, 128–137. [Google Scholar] [CrossRef] [Green Version]
- Rauner, M.; Sipos, W.; Thiele, S.; Pietschmann, P. Advances in Osteoimmunology: Pathophysiologic Concepts and Treatment Opportunities. Int. Arch. Allergy Immunol. 2013, 160, 114–125. [Google Scholar] [CrossRef]
- Gemmati, D.; Varani, K.; Bramanti, B.; Piva, R.; Bonaccorsi, G.; Trentini, A.; Manfrinato, M.C.; Tisato, V.; Carè, A.; Bellini, T. “Bridging the Gap” Everything that Could Have Been Avoided If We Had Applied Gender Medicine, Pharmacogenetics and Personalized Medicine in the Gender-Omics and Sex-Omics Era. Int. J. Mol. Sci. 2019, 21, 296. [Google Scholar] [CrossRef] [Green Version]
- Sipos, W. RANKL Inhibition: Preclinical Data. In Principles of Osteoimmunology: Molecular Mechanisms and Clinical Applications; Pietschmann, P., Ed.; Springer International Publishing: Vienna, Austria, 2012; pp. 197–215. [Google Scholar] [CrossRef]
- Zhang, X.; Wu, X.; Min, Y.; Lu, J.; Zhang, X.; Chen, W.; Zou, L.; Lv, X.; Cui, L.; Xu, B. Effect of prednisone treatment for 30 and 90 days on bone metabolism in collagen-induced arthritis (CIA) rats. J. Bone Miner. Metab. 2018, 36, 628–639. [Google Scholar] [CrossRef]
- Sipos, W.; Schmoll, F.; Stumpf, I. Minipigs and Potbellied Pigs as Pets in the Veterinary Practice—A Retrospective Study. J. Vet. Med. Ser. A 2007, 54, 504–511. [Google Scholar] [CrossRef]
- Shaughnessy, M.L.; Sample, S.J.; Abicht, C.; Heaton, C.; Muir, P. Clinical features and pathological joint changes in dogs with erosive immune-mediated polyarthritis: 13 cases (2004–2012). J. Am. Vet. Med. Assoc. 2016, 249, 1156–1164. [Google Scholar] [CrossRef] [Green Version]
- Campagnuolo, G.; Bolon, B.; Feige, U. Kinetics of bone protection by recombinant osteoprotegerin therapy in Lewis rats with adjuvant arthritis. Arthritis Care Res. 2002, 46, 1926–1936. [Google Scholar] [CrossRef]
- Ohshima, S.; Saeki, Y.; Mima, T.; Sasai, M.; Nishioka, K.; Nomura, S.; Kopf, M.; Katada, Y.; Tanaka, T.; Suemura, M.; et al. Interleukin 6 plays a key role in the development of antigen-induced arthritis. Proc. Natl. Acad. Sci. USA 1998, 95, 8222–8226. [Google Scholar] [CrossRef] [Green Version]
- Cremer, M.A.; Hernandez, A.D.; Townes, A.S.; Stuart, J.M.; Kang, A.H. Collagen-induced arthritis in rats: Anti-gen-specific suppression of arthritis and immunity by intravenously injected native type II collagen. J. Immunol. 1983, 131, 2995–3000. [Google Scholar] [PubMed]
- Stolina, M.; Schett, G.; Dwyer, D.; Vonderfecht, S.; Middleton, S.; Duryea, D.; Pacheco, E.; Van, G.; Bolon, B.; Feige, U.; et al. RANKL inhibition by osteoprotegerin prevents bone loss without affecting local or systemic inflammation parameters in two rat arthritis models: Comparison with anti-TNFα or anti-IL-1 therapies. Arthritis Res. Ther. 2009, 11, R187. [Google Scholar] [CrossRef] [Green Version]
- Stolina, M.; Bolon, B.; Middleton, S.; Dwyer, D.; Brown, H.; Duryea, D.; Zhu, L.; Rohner, A.; Pretorius, J.; Kostenuik, P.; et al. The Evolving Systemic and Local Biomarker Milieu at Different Stages of Disease Progression in Rat Adjuvant-Induced Arthritis. J. Clin. Immunol. 2009, 29, 158–174. [Google Scholar] [CrossRef]
- Stolina, M.; Bolon, B.; Dwyer, D.; Middleton, S.; Duryea, D.; Kostenuik, P.J.; Feige, U.; Zack, D.J. The evolving systemic and local biomarker milieu at different stages of disease progression in rat collagen-induced arthritis. Biomarkers 2009, 13, 692–712. [Google Scholar] [CrossRef]
- Sarkar, S.; Cooney, L.A.; White, P.; Dunlop, D.B.; Endres, J.; Jorns, J.M.; Wasco, M.J.; Fox, D.A. Regulation of pathogenic IL-17 responses in collagen-induced arthritis: Roles of endogenous interferon-gamma and IL-4. Arthritis Res. Ther. 2009, 11, R158. [Google Scholar] [CrossRef] [Green Version]
- Corr, M.; Crain, B. The Role of FcγR Signaling in the K/B × N Serum Transfer Model of Arthritis. J. Immunol. 2002, 169, 6604–6609. [Google Scholar] [CrossRef] [Green Version]
- Chen, X.-X.; Baum, W.; Dwyer, D.; Stock, M.; Schwabe, K.; Ke, H.-Z.; Stolina, M.; Schett, G.; Bozec, A. Sclerostin inhibition reverses systemic, periarticular and local bone loss in arthritis. Ann. Rheum. Dis. 2013, 72, 1732–1736. [Google Scholar] [CrossRef] [Green Version]
- Moon, N.; Effiong, L.; Song, L.; Gardner, T.R.; Soung, D.Y. Tart Cherry Prevents Bone Loss through Inhibition of RANKL in TNF-Overexpressing Mice. Nutrients 2019, 11, 63. [Google Scholar] [CrossRef] [Green Version]
- Redlich, K.; Hayer, S.; Maier, A.; Dunstan, C.; Tohidast-Akrad, M.; Lang, S.; Türk, B.; Pietschmann, P.; Woloszczuk, W.; Haralambous, S.; et al. Tumor necrosis factor α-mediated joint destruction is inhibited by targeting osteoclasts with osteoprotegerin. Arthritis Care Res. 2002, 46, 785–792. [Google Scholar] [CrossRef]
- Wu, Q.; Xiong, X.; Zhang, X.; Lu, J.; Zhang, X.; Chen, W.; Wu, T.; Cui, L.; Liu, Y.; Xu, B. Secondary osteoporosis in collagen-induced arthritis rats. J. Bone Miner. Metab. 2016, 34, 500–516. [Google Scholar] [CrossRef] [PubMed]
- Oelzner, P.; Fleissner-Richter, S.; Bräuer, R.; Hein, G.; Wolf, G.; Neumann, T. Combination therapy with dexamethasone and osteoprotegerin protects against arthritis-induced bone alterations in antigen-induced arthritis of the rat. Agents Actions 2010, 59, 731–741. [Google Scholar] [CrossRef] [PubMed]
- Sugahara, S.; Hanaoka, K.; Emori, T.; Takeshita, N.; Fujii, Y.; Nakano, M.; Suzuki, T.; Takahashi, J.; Nakamura, Y. Peficitinib improves bone fragility by recovering bone turnover imbalance in arthritic mice. J. Pharmacol. Sci. 2022, 148, 134–141. [Google Scholar] [CrossRef]
- Yoshida, H.; Suzuki, M.; Tanaka, K.; Takeda, S.; Yogo, K.; Matsumoto, Y. Anti-interleukin-6 receptor antibody prevents loss of bone structure and bone strength in collagen-induced arthritis mice. Scand. J. Rheumatol. 2018, 47, 384–391. [Google Scholar] [CrossRef]
- Qiu, J.; Lu, C.; Zhang, L.; Zhou, X.; Zou, H. Osteoporosis in patients with rheumatoid arthritis is associated with serum immune regulatory cellular factors. Clin. Rheumatol. 2022, 1–9. [Google Scholar] [CrossRef]
- Cheng, T.; Zhang, S.-X.; Wang, J.; Qiao, J.; Chang, M.-J.; Niu, H.-Q.; Liu, G.-Y.; Li, X.-F. Abnormalities of Peripheral Lymphocyte Subsets in Rheumatoid Arthritis Patients Complicated with Osteoporosis. Rheumatol. Ther. 2022, 9, 1049–1059. [Google Scholar] [CrossRef]
- Verbruggen, A.; De Clerck, L.S.; Bridts, C.H.; Van Offel, J.F.; Stevens, W.J. Flow cytometrical determination of interleukin 1β, interleukin 6 and tumour necrosis factor α in monocytes of rheumatoid arthritis patients; relation with parameters of osteoporosis. Cytokine 1999, 11, 869–874. [Google Scholar] [CrossRef]
- Meguid, M.H.A.; Hamad, Y.H.; Swilam, R.S.; Barakat, M.S. Relation of interleukin-6 in rheumatoid arthritis patients to systemic bone loss and structural bone damage. Rheumatol. Int. 2013, 33, 697–703. [Google Scholar] [CrossRef]
- Caetano-Lopes, J.; Rodrigues, A.; Lopes, A.; Vale, A.C.; Pitts-Kiefer, M.A.; Vidal, B.; Perpétuo, I.; Monteiro, J.; Konttinen, Y.T.; Vaz, M.F.; et al. Rheumatoid Arthritis Bone Fragility Is Associated with Upregulation of IL17 and DKK1 Gene Expression. Clin. Rev. Allergy Immunol. 2014, 47, 38–45. [Google Scholar] [CrossRef]
- Fessler, J.; Husic, R.; Schwetz, V.; Lerchbaum, E.; Aberer, F.; Fasching, P.; Ficjan, A.; Obermayer-Pietsch, B.; Duftner, C.; Graninger, W.; et al. Senescent T-Cells Promote Bone Loss in Rheumatoid Arthritis. Front. Immunol. 2018, 9, 95. [Google Scholar] [CrossRef] [Green Version]
- Oelzner, P.; Franke, S.; Lehmann, G.; Eidner, T.; Müller, A.; Wolf, G.; Hein, G. Soluble receptor activator of NFkappa B-ligand and osteoprotegerin in rheumatoid arthritis—Relationship with bone mineral density, disease activity and bone turnover. Clin. Rheumatol. 2007, 26, 2127–2135. [Google Scholar] [CrossRef] [PubMed]
- Nava-Valdivia, C.A.; Ponce-Guarneros, J.M.; Saldaña-Cruz, A.M.; Corona-Sanchez, E.G.; Ramirez-Villafaña, M.; Perez-Guerrero, E.E.; Murillo-Saich, J.D.; Contreras-Haro, B.; Vazquez-Villegas, M.L.; Gonzalez-Ponce, F.; et al. Assessment of Serum sRANKL, sRANKL/OPG Ratio, and Other Bone Turnover Markers with the Estimated 10-Year Risk of Major and Hip Osteoporotic Fractures in Rheumatoid Arthritis: A Cross-Sectional Study. BioMed Res. Int. 2021, 2021, 5567666. [Google Scholar] [CrossRef] [PubMed]
- Xu, S.; Wang, Y.; Lu, J.; Xu, J. Osteoprotegerin and RANKL in the pathogenesis of rheumatoid arthritis-induced osteoporosis. Rheumatol. Int. 2011, 32, 3397–3403. [Google Scholar] [CrossRef] [PubMed]
- Yang, L.; Zhang, J.; Tao, J.; Lu, T. Elevated serum levels of Interleukin-37 are associated with inflammatory cytokines and disease activity in rheumatoid arthritis. APMIS 2015, 123, 1025–1031. [Google Scholar] [CrossRef]
- Abdulkhaliq, A.; Cheikh, M.; Almuntashri, F.; Alzahrani, H.; Nadwi, H.; Kadi, E.; Abed, M.; Janaini, M.; Monjed, A.; Janoudi, N.; et al. A Comparison of Demographics, Disease Activity, Disability, and Treatment Among Rheumatoid Arthritis Patients with and without Osteoporosis. Open Access Rheumatol. Res. Rev. 2021, 13, 275–283. [Google Scholar] [CrossRef]
- Wysham, K.D.; Shofer, J.; Lui, G.; Trupin, L.; Andrews, J.S.; Black, D.M.; Graf, J.; Shoback, D.M.; Katz, P.P. Low cumulative disease activity is associated with higher bone mineral density in a majority Latinx and Asian US rheumatoid arthritis cohort. Semin. Arthritis Rheum. 2022, 53, 151972. [Google Scholar] [CrossRef]
- Guañabens, N.; Olmos, J.M.; Hernández, J.L.; Cerdà, D.; Calleja, C.H.; López, J.A.M.; Arboleya, L.; Del Rey, F.J.A.; Pardo, S.M.; Vilamajó, I.R.; et al. Vertebral fractures are increased in rheumatoid arthritis despite recent therapeutic advances: A case-control study. Osteoporos. Int. 2021, 32, 1333–1342. [Google Scholar] [CrossRef]
- Tong, J.-J.; Xu, S.-Q.; Zong, H.-X.; Pan, M.-J.; Teng, Y.-Z.; Xu, J.-H. Prevalence and risk factors associated with vertebral osteoporotic fractures in patients with rheumatoid arthritis. Clin. Rheumatol. 2020, 39, 357–364. [Google Scholar] [CrossRef]
- Tomizawa, T.; Ito, H.; Murata, K.; Hashimoto, M.; Tanaka, M.; Murakami, K.; Nishitani, K.; Azukizawa, M.; Okahata, A.; Doi, K.; et al. Distinct biomarkers for different bones in osteoporosis with rheumatoid arthritis. Arthritis Res. Ther. 2019, 21, 174. [Google Scholar] [CrossRef] [Green Version]
- Coiffier, G.; Bouvard, B.; Chopin, F.; Biver, E.; Funck-Brentano, T.; Garnero, P.; Guggenbuhl, P. Common bone turnover markers in rheumatoid arthritis and ankylosing spondylitis: A literature review. Jt. Bone Spine 2013, 80, 250–257. [Google Scholar] [CrossRef]
- Garnero, P.; Jouvenne, P.; Buchs, N.; Delmas, P.D.; Miossec, P. Uncoupling of bone metabolism in rheumatoid arthritis patients with or without joint destruction: Assessment with serum type I collagen breakdown products. Bone 1999, 24, 381–385. [Google Scholar] [CrossRef]
- Schett, G. The role of ACPAs in at-risk individuals: Early targeting of the bone and joints. Best Pract. Res. Clin. Rheumatol. 2017, 31, 53–58. [Google Scholar] [CrossRef]
- D’Onofrio, B.; Di Lernia, M.; De Stefano, L.; Bugatti, S.; Montecucco, C.; Bogliolo, L. Personalized Therapeutic Strategies in the Management of Osteoporosis in Patients with Autoantibody-Positive Rheumatoid Arthritis. J. Clin. Med. 2022, 11, 2341. [Google Scholar] [CrossRef]
- Nam, J.L.; Hunt, L.; Hensor, E.; Emery, P. Enriching case selection for imminent RA: The use of anti-CCP antibodies in individuals with new non-specific musculoskeletal symptoms—A cohort study. Ann. Rheum. Dis. 2016, 75, 1452–1456. [Google Scholar] [CrossRef]
- Kleyer, A.; Finzel, S.; Rech, J.; Manger, B.; Krieter, M.; Faustini, F.; Araujo, E.; Hueber, A.; Harre, U.; Engelke, K.; et al. Bone loss before the clinical onset of rheumatoid arthritis in subjects with anticitrullinated protein antibodies. Ann. Rheum. Dis. 2014, 73, 854–860. [Google Scholar] [CrossRef] [Green Version]
- Stemmler, F.; Simon, D.; Liphardt, A.-M.; Englbrecht, M.; Rech, J.; Hueber, A.; Engelke, K.; Schett, G.; Kleyer, A. Biomechanical properties of bone are impaired in patients with ACPA-positive rheumatoid arthritis and associated with the occurrence of fractures. Ann. Rheum. Dis. 2018, 77, 973–980. [Google Scholar] [CrossRef]
- Bugatti, S.; Bogliolo, L.; Vitolo, B.; Manzo, A.; Montecucco, C.; Caporali, R. Anti-citrullinated protein antibodies and high levels of rheumatoid factor are associated with systemic bone loss in patients with early untreated rheumatoid arthritis. Arthritis Res. Ther. 2016, 18, 226. [Google Scholar] [CrossRef] [Green Version]
- Bruno, D.; Fedele, A.L.; Tolusso, B.; Barini, A.; Petricca, L.; Di Mario, C.; Barini, A.; Mirone, L.; Ferraccioli, G.; Alivernini, S.; et al. Systemic Bone Density at Disease Onset Is Associated With Joint Erosion Progression in Early Naive to Treatment Rheumatoid Arthritis: A Prospective 12-Month Follow-Up Open-Label Study. Front. Med. 2021, 8, 613889. [Google Scholar] [CrossRef]
- Hauser, B.; Riches, P.L.; Gilchrist, T.; Visconti, M.R.; Wilson, J.F.; Ralston, S.H. Autoantibodies to osteoprotegerin are associated with increased bone resorption in rheumatoid arthritis. Ann. Rheum. Dis. 2015, 74, 1631–1632. [Google Scholar] [CrossRef] [Green Version]
- Laurent, M.R.; Goemaere, S.; Verroken, C.; Bergmann, P.; Body, J.-J.; Bruyère, O.; Cavalier, E.; Rozenberg, S.; Lapauw, B.; Gielen, E. Prevention and Treatment of Glucocorticoid-Induced Osteoporosis in Adults: Consensus Recommendations from the Belgian Bone Club. Front. Endocrinol. 2022, 13, 908727. [Google Scholar] [CrossRef]
- Blavnsfeldt, A.-B.G.; de Thurah, A.; Thomsen, M.D.; Tarp, S.; Langdahl, B.; Hauge, E.-M. The effect of glucocorticoids on bone mineral density in patients with rheumatoid arthritis: A systematic review and meta-analysis of randomized, controlled trials. Bone 2018, 114, 172–180. [Google Scholar] [CrossRef]
- Wang, Y.; Zhao, R.; Gu, Z.; Dong, C.; Guo, G.; Li, L. Effects of glucocorticoids on osteoporosis in rheumatoid arthritis: A systematic review and meta-analysis. Osteoporos. Int. 2020, 31, 1401–1409. [Google Scholar] [CrossRef]
- Polito, A.; Barnaba, L.; Ciarapica, D.; Azzini, E. Osteosarcopenia: A Narrative Review on Clinical Studies. Int. J. Mol. Sci. 2022, 23, 5591. [Google Scholar] [CrossRef]
- Lian, L.; Wang, J.-X.; Xu, Y.-C.; Zong, H.-X.; Teng, Y.-Z.; Xu, S.-Q. Sarcopenia May Be a Risk Factor for Osteoporosis in Chinese Patients with Rheumatoid Arthritis. Int. J. Gen. Med. 2022, 15, 2075–2085. [Google Scholar] [CrossRef]
- Oelzner, P.; Schwabe, A.; Lehmann, G.; Eidner, T.; Franke, S.; Wolf, G.; Hein, G. Significance of risk factors for osteoporosis is dependent on gender and menopause in rheumatoid arthritis. Rheumatol. Int. 2008, 28, 1143–1150. [Google Scholar] [CrossRef]
- Zahid, P.M.; Samreen, S.; Khan, Z.A.; Salim, B.; Ahmed, S.N.; Gul, H. Factors associated with low bone mineral density in postmenopausal women with rheumatoid arthritis. J. Pak. Med. Assoc. 2022, 72, 891–895. [Google Scholar] [CrossRef]
- Kweon, S.-M.; Sohn, D.H.; Park, J.-H.; Koh, J.H.; Park, E.-K.; Lee, H.-N.; Kim, K.; Kim, Y.; Kim, G.-T.; Lee, S.-G. Male patients with rheumatoid arthritis have an increased risk of osteoporosis. Medicine 2018, 97, e11122. [Google Scholar] [CrossRef]
- Nakayama, M.; Furuya, T.; Inoue, E.; Tanaka, E.; Ikari, K.; Yamanaka, H.; Harigai, M. Vitamin D deficiency is a risk factor for new fractures in Japanese postmenopausal women with rheumatoid arthritis: Results from the IORRA cohort study. Arch. Osteoporos. 2021, 16, 119. [Google Scholar] [CrossRef]
- Hu, Z.; Zhang, L.; Lin, Z.; Zhao, C.; Xu, S.; Lin, H.; Zhang, J.; Li, W.; Chu, Y. Prevalence and risk factors for bone loss in rheumatoid arthritis patients from South China: Modeled by three methods. BMC Musculoskelet. Disord. 2021, 22, 534. [Google Scholar] [CrossRef]
- Yoshii, I.; Sawada, N.; Chijiwa, T.; Kokei, S. Impact of sustaining SDAI remission for preventing incident of bone fragility fracture in patient with rheumatoid arthritis. Ann. Rheum. Dis. 2022, 81, 296–299. [Google Scholar] [CrossRef]
- Lindner, L.; Callhoff, J.; Alten, R.; Krause, A.; Ochs, W.; Zink, A.; Albrecht, K. Osteoporosis in patients with rheumatoid arthritis: Trends in the German National Database 2007–2017. Rheumatol. Int. 2020, 40, 2005–2012. [Google Scholar] [CrossRef] [PubMed]
- Zerbini, C.A.F.; Clark, P.; Mendez-Sanchez, L.; Pereira, R.M.R.; Messina, O.D.; Uña, C.R.; Adachi, J.D.; Lems, W.F.; Cooper, C.; Lane, N.E.; et al. Biologic therapies and bone loss in rheumatoid arthritis. Osteoporos. Int. 2017, 28, 429–446. [Google Scholar] [CrossRef] [PubMed]
- Orsolini, G.; Fassio, A.; Rossini, M.; Adami, G.; Giollo, A.; Caimmi, C.; Idolazzi, L.; Viapiana, O.; Gatti, D. Effects of biological and targeted synthetic DMARDs on bone loss in rheumatoid arthritis. Pharmacol. Res. 2019, 147, 104354. [Google Scholar] [CrossRef]
- Hauser, B.; Raterman, H.; Ralston, S.H.; Lems, W.F. The Effect of Anti-rheumatic Drugs on the Skeleton. Calcif. Tissue Res. 2022, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Soós, B.; Szentpétery, Á.; Raterman, H.G.; Lems, W.F.; Bhattoa, H.P.; Szekanecz, Z. Effects of targeted therapies on bone in rheumatic and musculoskeletal diseases. Nat. Rev. Rheumatol. 2022, 18, 249–257. [Google Scholar] [CrossRef]
- Chen, J.-F.; Hsu, C.-Y.; Yu, S.-F.; Ko, C.-H.; Chiu, W.-C.; Lai, H.-M.; Chen, Y.-C.; Su, Y.-J.; Cheng, T.-T. The impact of long-term biologics/target therapy on bone mineral density in rheumatoid arthritis: A propensity score-matched analysis. Rheumatology 2020, 59, 2471–2480. [Google Scholar] [CrossRef] [Green Version]
- Kume, K.; Amano, K.; Yamada, S.; Kanazawa, T.; Ohta, H.; Hatta, K.; Kuwaba, N. The effect of tocilizumab on bone mineral density in patients with methotrexate-resistant active rheumatoid arthritis. Rheumatology 2014, 53, 900–903. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Briot, K.; Rouanet, S.; Schaeverbeke, T.; Etchepare, F.; Gaudin, P.; Perdriger, A.; Vray, M.; Steinberg, G.; Roux, C. The effect of tocilizumab on bone mineral density, serum levels of Dickkopf-1 and bone remodeling markers in patients with rheumatoid arthritis. Jt. Bone Spine 2015, 82, 109–115. [Google Scholar] [CrossRef]
- Chen, Y.-M.; Chen, H.-H.; Huang, W.-N.; Liao, T.-L.; Chen, J.-P.; Chao, W.-C.; Lin, C.-T.; Hung, W.-T.; Hsieh, C.-W.; Hsieh, T.-Y.; et al. Tocilizumab potentially prevents bone loss in patients with anticitrullinated protein antibody-positive rheumatoid arthritis. PLoS ONE 2017, 12, e0188454. [Google Scholar] [CrossRef]
- Abu-Shakra, M.; Zisman, D.; Balbir-Gurman, A.; Amital, H.; Levy, Y.; Langevitz, P.; Tishler, M.; Molad, Y.; Aamar, S.; Roser, I.; et al. Effect of Tocilizumab on Fatigue and Bone Mineral Density in Patients with Rheumatoid Arthritis. IMAJ 2018, 20, 239–244. [Google Scholar]
- Tada, M.; Inui, K.; Sugioka, Y.; Mamoto, K.; Okano, T.; Koike, T. Abatacept might increase bone mineral density at femoral neck for patients with rheumatoid arthritis in clinical practice: AIRTIGHT study. Rheumatol. Int. 2018, 38, 777–784. [Google Scholar] [CrossRef] [Green Version]
- Chen, M.-H.; Yu, S.-F.; Chen, J.-F.; Chen, W.-S.; Liou, T.-L.; Chou, C.-T.; Hsu, C.-Y.; Lai, H.-M.; Chen, Y.-C.; Tsai, C.-Y.; et al. Different Effects of Biologics on Systemic Bone Loss Protection in Rheumatoid Arthritis: An Interim Analysis of a Three-Year Longitudinal Cohort Study. Front. Immunol. 2021, 12, 783030. [Google Scholar] [CrossRef]
- Wheater, G.; Elshahaly, M.; Naraghi, K.; Tuck, S.P.; Datta, H.K.; Van Laar, J.M. Changes in bone density and bone turnover in patients with rheumatoid arthritis treated with rituximab, results from an exploratory, prospective study. PLoS ONE 2018, 13, e0201527. [Google Scholar] [CrossRef]
- Al Khayyat, S.G.; Falsetti, P.; Conticini, E.; D’Alessandro, R.; Bellisai, F.; Gentileschi, S.; Baldi, C.; Bardelli, M.; Cantarini, L.; Frediani, B. Bone-sparing effects of rituximab and body composition analysis in a cohort of postmenopausal women affected by rheumatoid arthritis—Retrospective study. Reumatologia 2021, 59, 206–210. [Google Scholar] [CrossRef]
- Hamar, A.; Szekanecz, Z.; Pusztai, A.; Czókolyová, M.; Végh, E.; Pethő, Z.; Bodnár, N.; Gulyás, K.; Horváth, Á.; Soós, B.; et al. Effects of one-year tofacitinib therapy on bone metabolism in rheumatoid arthritis. Osteoporos. Int. 2021, 32, 1621–1629. [Google Scholar] [CrossRef]
- Dore, R.K.; Cohen, S.B.; Lane, N.E.; Palmer, W.; Shergy, W.; Zhou, L.; Wang, H.; Tsuji, W.; Newmark, R.; on behalf of the Denosumab RA Study Group. Effects of denosumab on bone mineral density and bone turnover in patients with rheumatoid arthritis receiving concurrent glucocorticoids or bisphosphonates. Ann. Rheum. Dis. 2010, 69, 872–875. [Google Scholar] [CrossRef]
- Hu, Q.; Zhong, X.; Tian, H.; Liao, P. The Efficacy of Denosumab in Patients with Rheumatoid Arthritis: A Systematic Review and Pooled Analysis of Randomized or Matched Data. Front. Immunol. 2022, 12, 799575. [Google Scholar] [CrossRef]
- Yagita, M.; Morita, T.; Kumanogoh, A. Therapeutic efficacy of denosumab for rheumatoid arthritis: A systematic review and meta-analysis. Rheumatol. Adv. Pract. 2021, 5, rkab099. [Google Scholar] [CrossRef]
- Suzuki, T.; Nakamura, Y.; Kato, H. Effects of denosumab on bone metabolism and bone mineral density with anti-TNF inhibitors, tocilizumab, or abatacept in osteoporosis with rheumatoid arthritis. Ther. Clin. Risk Manag. 2018, 14, 453–459. [Google Scholar] [CrossRef] [Green Version]
- Carbone, L.; Vasan, S.; Elam, R.; Gupta, S.; Tolaymat, O.; Crandall, C.; Wactawski-Wende, J.; Johnson, K.C. The Association of Methotrexate, Sulfasalazine, and Hydroxychloroquine Use With Fracture in Postmenopausal Women With Rheumatoid Arthritis: Findings from the Women’s Health Initiative. JBMR Plus 2020, 4, e10393. [Google Scholar] [CrossRef]
- Robin, F.; Cadiou, S.; Albert, J.-D.; Bart, G.; Coiffier, G.; Guggenbuhl, P. Methotrexate osteopathy: Five cases and systematic literature review. Osteoporos. Int. 2021, 32, 225–232. [Google Scholar] [CrossRef]
- Ozen, G.; Pedro, S.; Wolfe, F.; Michaud, K. Medications associated with fracture risk in patients with rheumatoid arthritis. Ann. Rheum. Dis. 2019, 78, 1041–1047. [Google Scholar] [CrossRef]
- Kim, S.Y.; Schneeweiss, S.; Liu, J.; Solomon, D.H. Effects of disease-modifying antirheumatic drugs on nonvertebral fracture risk in rheumatoid arthritis: A population-based cohort study. J. Bone Miner. Res. 2012, 27, 789–796. [Google Scholar] [CrossRef] [Green Version]
- Shao, F.; Li, H.-C.; Wang, M.-J.; Cui, C.-M. Impact of biologic disease-modifying antirheumatic drugs on fracture risk in patients with rheumatoid arthritis: A systematic review and meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2021, 25, 3416–3424. [Google Scholar] [CrossRef]
- Pawar, A.; Desai, R.J.; He, M.; Bessette, L.; Kim, S.C. Comparative Risk of Nonvertebral Fractures Among Patients With Rheumatoid Arthritis Treated With Biologic or Targeted Synthetic Disease-Modifying Antirheumatic Drugs. ACR Open Rheumatol. 2021, 3, 531–539. [Google Scholar] [CrossRef]
- Lv, F.; Hu, S.; Lin, C.; Cai, X.; Zhu, X.; Ji, L. Association between biologic therapy and fracture incidence in patients with selected rheumatic and autoimmune diseases: A systematic review and meta-analysis of randomized controlled trials. Pharmacol. Res. 2022, 181, 106278. [Google Scholar] [CrossRef] [PubMed]
- Abtahi, S.; Cordtz, R.; Dreyer, L.; Driessen, J.H.M.; Boonen, A.; Burden, A.M. Biological Disease-Modifying Antirheumatic Drugs and Osteoporotic Fracture Risk in Patients with Rheumatoid Arthritis: A Danish Cohort Study. Am. J. Med. 2022, 135, 879–888.e3. [Google Scholar] [CrossRef] [PubMed]
- Shin, A.; Park, E.; Dong, Y.-H.; Ha, Y.-J.; Lee, Y.; Lee, E.; Song, Y.; Kang, E. Comparative risk of osteoporotic fracture among patients with rheumatoid arthritis receiving TNF inhibitors versus other biologics: A cohort study. Osteoporos. Int. 2020, 31, 2131–2139. [Google Scholar] [CrossRef]
- Murata, K.; Uozumi, R.; Hashimoto, M.; Ebina, K.; Akashi, K.; Onishi, A.; Nagai, K.; Yoshikawa, A.; Katayama, M.; Son, Y.; et al. The real-world effectiveness of anti-RANKL antibody denosumab on the clinical fracture prevention in patients with rheumatoid arthritis: The ANSWER cohort study. Mod. Rheumatol. 2022, 32, 834–838. [Google Scholar] [CrossRef] [PubMed]
- Hong, W.-J.; Chen, W.; Yeo, K.-J.; Huang, P.-H.; Chen, D.-Y.; Lan, J.-L. Increased risk of osteoporotic vertebral fracture in rheumatoid arthritis patients with new-onset cardiovascular diseases: A retrospective nationwide cohort study in Taiwan. Osteoporos. Int. 2019, 30, 1617–1625. [Google Scholar] [CrossRef]
- Kawai, V.K.; Grijalva, C.; Arbogast, P.G.; Curtis, J.R.; Solomon, D.H.; Delzell, E.; Chen, L.; Ouellet-Hellstrom, R.; Herrinton, L.; Liu, L.; et al. Initiation of tumor necrosis factor α antagonists and risk of fractures in patients with selected rheumatic and autoimmune diseases. Arthritis Care Res. 2012, 65, 1085–1094. [Google Scholar] [CrossRef] [Green Version]
- Timmen, M.; Hidding, H.; Wieskötter, B.; Baum, W.; Pap, T.; Raschke, M.J.; Schett, G.; Zwerina, J.; Stange, R. Influence of antiTNF-alpha antibody treatment on fracture healing under chronic inflammation. BMC Musculoskelet. Disord. 2014, 15, 184. [Google Scholar] [CrossRef] [Green Version]
- Satoh, K.; Mark, H.; Zachrisson, P.; Rydevik, B.; Byröd, G.; Kikuchi, S.-I.; Konno, S.-I.; Sekiguchi, M. Effect of Methotrexate on Fracture Healing. Fukushima J. Med. Sci. 2011, 57, 11–18. [Google Scholar] [CrossRef] [Green Version]
- George, M.D.; Baker, J.F.; Winthrop, K.L.; Goldstein, S.; Alemao, E.; Chen, L.; Wu, Q.; Xie, F.; Curtis, J.R. Immunosuppression and the risk of readmission and mortality in patients with rheumatoid arthritis undergoing hip fracture, abdominopelvic and cardiac surgery. Ann. Rheum. Dis. 2020, 79, 573–580. [Google Scholar] [CrossRef]
- Gaber, T.; Brinkman, A.C.K.; Pienczikowski, J.; Diesing, K.; Damerau, A.; Pfeiffenberger, M.; Lang, A.; Ohrndorf, S.; Burmester, G.-R.; Buttgereit, F.; et al. Impact of Janus Kinase Inhibition with Tofacitinib on Fundamental Processes of Bone Healing. Int. J. Mol. Sci. 2020, 21, 865. [Google Scholar] [CrossRef] [Green Version]
- Stanmore, E.K.; Oldham, J.; Skelton, D.A.; O’Neill, T.; Pilling, M.; Campbell, A.J.; Todd, C. Risk Factors for Falls in Adults with Rheumatoid Arthritis: A Prospective Study. Arthritis Care Res. 2013, 65, 1251–1258. [Google Scholar] [CrossRef] [Green Version]
- Tournadre, A.; Pereira, B.; Dutheil, F.; Giraud, C.; Courteix, D.; Sapin, V.; Frayssac, T.; Mathieu, S.; Malochet-Guinamand, S.; Soubrier, M. Changes in body composition and metabolic profile during interleukin 6 inhibition in rheumatoid arthritis. J. Cachex Sarcopenia Muscle 2017, 8, 639–646. [Google Scholar] [CrossRef]
- Torii, M.; Hashimoto, M.; Hanai, A.; Fujii, T.; Furu, M.; Ito, H.; Uozumi, R.; Hamaguchi, M.; Terao, C.; Yamamoto, W.; et al. Prevalence and factors associated with sarcopenia in patients with rheumatoid arthritis. Mod. Rheumatol. 2018, 29, 589–595. [Google Scholar] [CrossRef]
- Guan, Y.; Hao, Y.; Guan, Y.; Bu, H.; Wang, H. The Effect of Vitamin D Supplementation on Rheumatoid Arthritis Patients: A Systematic Review and Meta-Analysis. Front. Med. 2020, 7, 596007. [Google Scholar] [CrossRef]
- Nguyen, Y.; Sigaux, J.; Letarouilly, J.-G.; Sanchez, P.; Czernichow, S.; Flipo, R.-M.; Soubrier, M.; Semerano, L.; Seror, R.; Sellam, J.; et al. Efficacy of Oral Vitamin Supplementation in Inflammatory Rheumatic Disorders: A Systematic Review and Meta-Analysis of Randomized Controlled Trials. Nutrients 2021, 13, 107. [Google Scholar] [CrossRef]
- De Jong, Z.; Munneke, M.; Lems, W.F.; Zwinderman, A.H.; Kroon, H.M.; Pauwels, E.K.J.; Jansen, A.; Ronday, K.H.; Dijkmans, B.A.C.; Breedveld, F.C.; et al. Slowing of bone loss in patients with rheumatoid arthritis by long-term high-intensity exercise: Results of a randomized, controlled trial. Arthritis Care Res. 2004, 50, 1066–1076. [Google Scholar] [CrossRef] [PubMed]
- Pietschmann, P.; Föger-Samwald, U.; Butylina, M.; Sipos, W. Evolution and History of Osteoimmunology. Osteologie 2021, 30, 286–291. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pietschmann, P.; Butylina, M.; Kerschan-Schindl, K.; Sipos, W. Mechanisms of Systemic Osteoporosis in Rheumatoid Arthritis. Int. J. Mol. Sci. 2022, 23, 8740. https://doi.org/10.3390/ijms23158740
Pietschmann P, Butylina M, Kerschan-Schindl K, Sipos W. Mechanisms of Systemic Osteoporosis in Rheumatoid Arthritis. International Journal of Molecular Sciences. 2022; 23(15):8740. https://doi.org/10.3390/ijms23158740
Chicago/Turabian StylePietschmann, Peter, Maria Butylina, Katharina Kerschan-Schindl, and Wolfgang Sipos. 2022. "Mechanisms of Systemic Osteoporosis in Rheumatoid Arthritis" International Journal of Molecular Sciences 23, no. 15: 8740. https://doi.org/10.3390/ijms23158740
APA StylePietschmann, P., Butylina, M., Kerschan-Schindl, K., & Sipos, W. (2022). Mechanisms of Systemic Osteoporosis in Rheumatoid Arthritis. International Journal of Molecular Sciences, 23(15), 8740. https://doi.org/10.3390/ijms23158740





