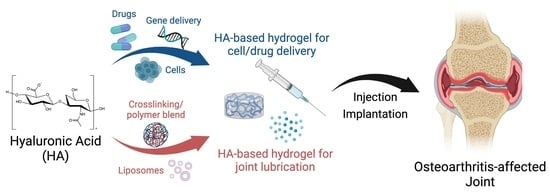
Engineering Hyaluronic Acid for the Development of New Treatment Strategies for Osteoarthritis
Abstract
1. Introduction
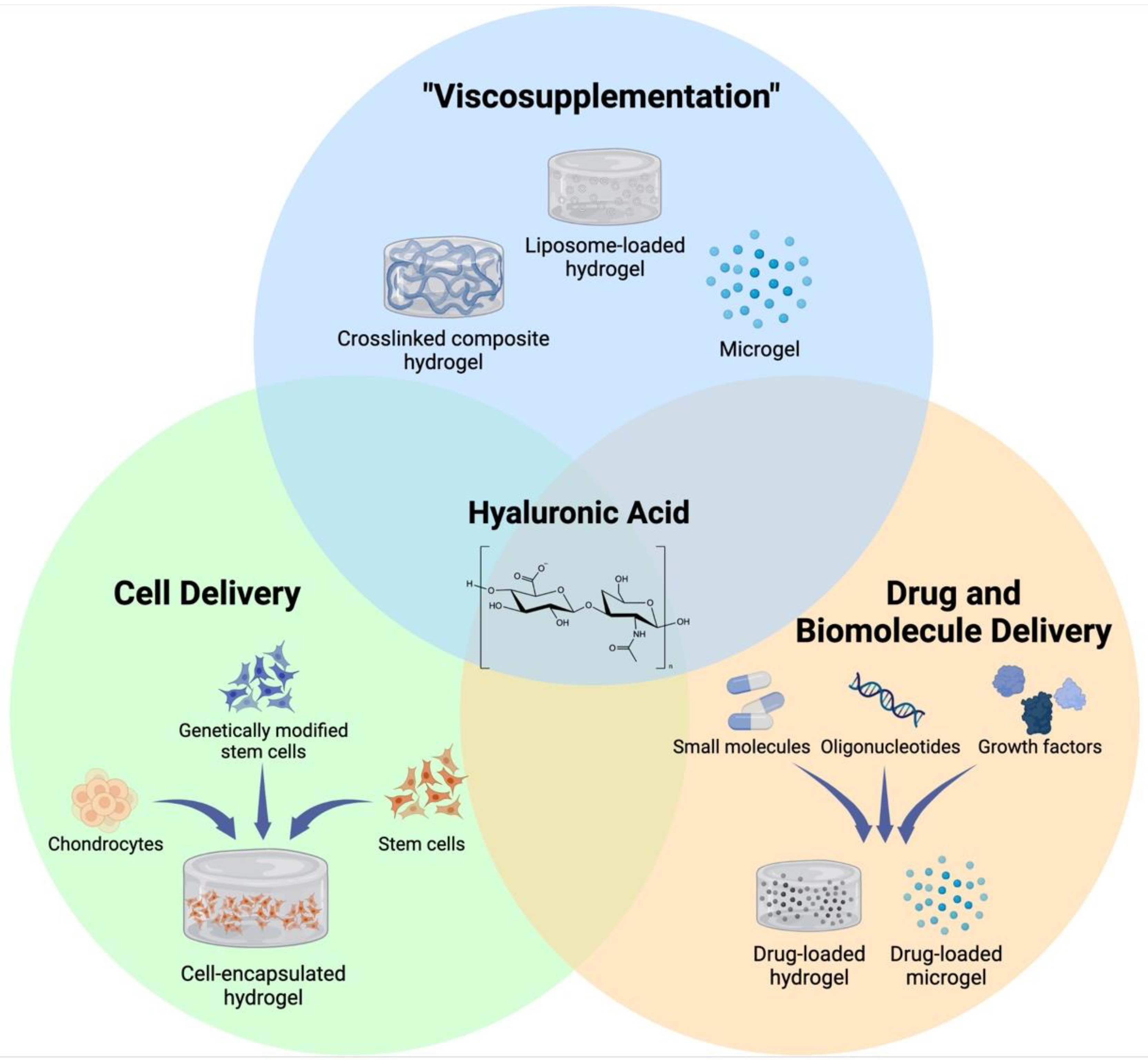
2. HA Hydrogels for Joint Lubrication
3. HA-Based Hydrogels for Biologics Delivery
4. HA-Based Hydrogels for Cell Delivery
5. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Loeser, R.F.; Goldring, S.R.; Scanzello, C.R.; Goldring, M.B. Osteoarthritis: A disease of the joint as an organ. Arthritis Rheum. 2012, 64, 1697–1707. [Google Scholar] [CrossRef] [PubMed]
- Favero, M.; Ramonda, R.; Goldring, M.B.; Goldring, S.R.; Punzi, L. Early knee osteoarthritis: Figure 1. RMD Open 2015, 1 (Suppl. 1), e000062. [Google Scholar] [CrossRef] [PubMed]
- Poole, A.R. Osteoarthritis as a Whole Joint Disease. HSS J. 2012, 8, 4–6. [Google Scholar] [CrossRef]
- Long, H.; Liu, Q.; Yin, H.; Wang, K.; Diao, N.; Zhang, Y.; Lin, J.; Guo, A. Prevalence Trends of Site-Specific Osteoarthritis from 1990 to 2019: Findings from the Global Burden of Disease Study 2019. Arthritis Rheumatol. 2022, 74, 1172–1183. [Google Scholar] [CrossRef] [PubMed]
- Katz, J.N.; Arant, K.R.; Loeser, R.F. Diagnosis and Treatment of Hip and Knee Osteoarthritis: A Review. JAMA 2021, 325, 568–578. [Google Scholar] [CrossRef] [PubMed]
- Lespasio, M.J.; Piuzzi, N.S.; Husni, M.E.; Muschler, G.F.; Guarino, A.; Mont, M.A. Knee Osteoarthritis: A Primer. Perm. J. 2017, 21, 16–183. [Google Scholar] [CrossRef] [PubMed]
- Millett, P.J.; Gobezie, R.; Boykin, R.E. Shoulder osteoarthritis: Diagnosis and management. Am. Fam. Physician 2008, 78, 605–611. [Google Scholar] [PubMed]
- Grayson, C.W.; Decker, R.C. Total Joint Arthroplasty for Persons with Osteoarthritis. PM&R 2012, 4, S97–S103. [Google Scholar] [CrossRef]
- Lützner, J.; Kasten, P.; Günther, K.-P.; Kirschner, S. Surgical options for patients with osteoarthritis of the knee. Nat. Rev. Rheumatol. 2009, 5, 309–316. [Google Scholar] [CrossRef]
- Kalamegam, G.; Memic, A.; Budd, E.; Abbas, M.; Mobasheri, A. A Comprehensive Review of Stem Cells for Cartilage Regeneration in Osteoarthritis. In Cell Biology and Translational Medicine, Volume 2. Advances in Experimental Medicine and Biology; Turksen, K., Ed.; Springer International Publishing: Cham, Switzerland, 2018; pp. 23–36. ISBN 978-3-030-04170-0. [Google Scholar]
- Luyten, F.P.; Vanlauwe, J. Tissue engineering approaches for osteoarthritis. Bone 2012, 51, 289–296. [Google Scholar] [CrossRef]
- Kou, L.; Xiao, S.; Sun, R.; Bao, S.; Yao, Q.; Chen, R. Biomaterial-engineered intra-articular drug delivery systems for osteoarthritis therapy. Drug Deliv. 2019, 26, 870–885. [Google Scholar] [CrossRef]
- Maihöfer, J.; Madry, H.; Rey-Rico, A.; Venkatesan, J.K.; Goebel, L.; Schmitt, G.; Speicher-Mentges, S.; Cai, X.; Meng, W.; Zurakowski, D.; et al. Hydrogel-Guided, rAAV-Mediated IGF-I Overexpression Enables Long-Term Cartilage Repair and Protection against Perifocal Osteoarthritis in a Large-Animal Full-Thickness Chondral Defect Model at One Year In Vivo. Adv. Mater. 2021, 33, 2008451. [Google Scholar] [CrossRef]
- Thangprasert, A.; Tansakul, C.; Thuaksubun, N.; Meesane, J. Mimicked hybrid hydrogel based on gelatin/PVA for tissue engineering in subchondral bone interface for osteoarthritis surgery. Mater. Des. 2019, 183, 108113. [Google Scholar] [CrossRef]
- Bierbrauer, K.L.; Alasino, R.V.; Barclay, F.E.; Belotti, E.M.; Ortega, H.H.; Beltramo, D.M. Biocompatible Hydrogel for Intra-Articular Implantation Comprising Cationic and Anionic Polymers of Natural Origin: In Vivo Evaluation in a Rabbit Model. Polymers 2021, 13, 4426. [Google Scholar] [CrossRef]
- Liang, R.; Zhao, J.; Li, B.; Cai, P.; Loh, X.J.; Xu, C.; Chen, P.; Kai, D.; Zheng, L. Implantable and degradable antioxidant poly(ε-caprolactone)-lignin nanofiber membrane for effective osteoarthritis treatment. Biomaterials 2020, 230, 119601. [Google Scholar] [CrossRef]
- Zhao, C.F.; Li, Z.H.; Li, S.J.; Li, J.A.; Hou, T.T.; Wang, Y. PLGA scaffold carrying icariin to inhibit the progression of osteoarthritis in rabbits. R. Soc. Open Sci. 2019, 6, 181877. [Google Scholar] [CrossRef]
- Morille, M.; Toupet, K.; Montero-Menei, C.N.; Jorgensen, C.; Noël, D. PLGA-based microcarriers induce mesenchymal stem cell chondrogenesis and stimulate cartilage repair in osteoarthritis. Biomaterials 2016, 88, 60–69. [Google Scholar] [CrossRef]
- Rodriguez-Marquez, C.D.; Arteaga-Marin, S.; Rivas-Sánchez, A.; Autrique-Hernández, R.; Castro-Muñoz, R. A Review on Current Strategies for Extraction and Purification of Hyaluronic Acid. Int. J. Mol. Sci. 2022, 23, 6038. [Google Scholar] [CrossRef]
- Sze, J.H.; Brownlie, J.C.; Love, C.A. Biotechnological production of hyaluronic acid: A mini review. 3 Biotech 2016, 6, 67. [Google Scholar] [CrossRef]
- Payan, E.; Jouzeau, J.Y.; Lapicque, F.; Muller, N.; Netter, P. Hyaluronidase degradation of hyaluronic acid from different sources: Influence of the hydrolysis conditions on the production and the relative proportions of tetra- and hexasaccharide produced. Int. J. Biochem. 1993, 25, 325–329. [Google Scholar] [CrossRef]
- Burdick, J.A.; Chung, C.; Jia, X.; Randolph, M.A.; Langer, R. Controlled Degradation and Mechanical Behavior of Photopolymerized Hyaluronic Acid Networks. Biomacromolecules 2005, 6, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Moreland, L.W. Intra-articular hyaluronan (hyaluronic acid) and hylans for the treatment of osteoarthritis: Mechanisms of action. Arthritis Res. Ther. 2003, 5, 54–67. [Google Scholar] [CrossRef] [PubMed]
- Migliore, A.; Procopio, S. Effectiveness and utility of hyaluronic acid in osteoarthritis. Clin. Cases Miner. Bone Metab. 2015, 12, 31–33. [Google Scholar] [CrossRef] [PubMed]
- Strauss, E.J.; Hart, J.A.; Miller, M.D.; Altman, R.D.; Rosen, J.E. Hyaluronic Acid Viscosupplementation and Osteoarthritis: Current Uses and Future Directions. Am. J. Sports Med. 2009, 37, 1636–1644. [Google Scholar] [CrossRef]
- Hayes, A.J.; Melrose, J. Glycosaminoglycan and Proteoglycan Biotherapeutics in Articular Cartilage Protection and Repair Strategies: Novel Approaches to Visco-supplementation in Orthobiologics. Adv. Ther. 2019, 2, 1900034. [Google Scholar] [CrossRef]
- Evanich, J.D.; Evanich, C.J.; Wright, M.B.; Rydlewicz, J.A. Efficacy of Intraarticular Hyaluronic Acid Injections in Knee Osteoarthritis. Clin. Orthop. Relat. Res. 2001, 390, 173–181. [Google Scholar] [CrossRef]
- Watterson, J.R.; Esdaile, J.M. Viscosupplementation: Therapeutic Mechanisms and Clinical Potential in Osteoarthritis of the Knee. JAAOS-J. Am. Acad. Orthop. Surg. 2000, 8, 277–284. [Google Scholar] [CrossRef]
- Lo, G.H.; LaValley, M.; McAlindon, T.; Felson, D.T. Intra-Articular Hyaluronic Acid in Treatment of Knee Osteoarthritis: A Meta-Analysis. JAMA 2003, 290, 3115–3121. [Google Scholar] [CrossRef]
- Arrich, J.; Piribauer, F.; Mad, P.; Schmid, D.; Klaushofer, K.; Müllner, M. Intra-articular hyaluronic acid for the treatment of osteoarthritis of the knee: Systematic review and meta-analysis. CMAJ Can. Med. Assoc. J. 2005, 172, 1039–1043. [Google Scholar] [CrossRef]
- Zhao, K.; Wen, Y.; Bunpetch, V.; Lin, J.; Hu, Y.; Zhang, X.; Xie, Y.; Zhang, S.; Hongwei, O. Hype or hope of hyaluronic acid for osteoarthritis: Integrated clinical evidence synthesis with multi-organ transcriptomics. J. Orthop. Transl. 2022, 32, 91–100. [Google Scholar] [CrossRef]
- Pereira, T.V.; Jüni, P.; Saadat, P.; Xing, D.; Yao, L.; Bobos, P.; Agarwal, A.; Hincapié, C.A.; da Costa, B.R. Viscosupplementation for knee osteoarthritis: Systematic review and meta-analysis. BMJ 2022, 378, e069722. [Google Scholar] [CrossRef]
- Li, H.; Qi, Z.; Zheng, S.; Chang, Y.; Kong, W.; Fu, C.; Yu, Z.; Yang, X.; Pan, S. The Application of Hyaluronic Acid-Based Hydrogels in Bone and Cartilage Tissue Engineering. Adv. Mater. Sci. Eng. 2019, 2019, e3027303. [Google Scholar] [CrossRef]
- Tsanaktsidou, E.; Kammona, O.; Kiparissides, C. Recent Developments in Hyaluronic Acid-Based Hydrogels for Cartilage Tissue Engineering Applications. Polymers 2022, 14, 839. [Google Scholar] [CrossRef]
- Schuurmans, C.C.L.; Mihajlovic, M.; Hiemstra, C.; Ito, K.; Hennink, W.E.; Vermonden, T. Hyaluronic acid and chondroitin sulfate (meth)acrylate-based hydrogels for tissue engineering: Synthesis, characteristics and pre-clinical evaluation. Biomaterials 2021, 268, 120602. [Google Scholar] [CrossRef]
- Tiwari, S.; Bahadur, P. Modified hyaluronic acid based materials for biomedical applications. Int. J. Biol. Macromol. 2019, 121, 556–571. [Google Scholar] [CrossRef]
- Ngadimin, K.D.; Stokes, A.; Gentile, P.; Ferreira-Duarte, A.M. Biomimetic hydrogels designed for cartilage tissue engineering. Biomater. Sci. 2021, 9, 4246–4259. [Google Scholar] [CrossRef]
- Bland, J.H.; Cooper, S.M. Osteoarthritis: A review of the cell biology involved and evidence for reversibility. Management rationally related to known genesis and pathophysiology. Semin. Arthritis Rheum. 1984, 14, 106–133. [Google Scholar] [CrossRef]
- Martel-Pelletier, J.; Boileau, C.; Pelletier, J.-P.; Roughley, P.J. Cartilage in normal and osteoarthritis conditions. Best Pract. Res. Clin. Rheumatol. 2008, 22, 351–384. [Google Scholar] [CrossRef]
- Bowman, S.; Awad, M.E.; Hamrick, M.W.; Hunter, M.; Fulzele, S. Recent advances in hyaluronic acid based therapy for osteoarthritis. Clin. Transl. Med. 2018, 7, 6. [Google Scholar] [CrossRef]
- Shimizu, H.; Shimoda, M.; Mochizuki, S.; Miyamae, Y.; Abe, H.; Chijiiwa, M.; Yoshida, H.; Shiozawa, J.; Ishijima, M.; Kaneko, K.; et al. Hyaluronan-Binding Protein Involved in Hyaluronan Depolymerization Is Up-Regulated and Involved in Hyaluronan Degradation in Human Osteoarthritic Cartilage. Am. J. Pathol. 2018, 188, 2109–2119. [Google Scholar] [CrossRef]
- Shiozawa, J.; de Vega, S.; Cilek, M.Z.; Yoshinaga, C.; Nakamura, T.; Kasamatsu, S.; Yoshida, H.; Kaneko, H.; Ishijima, M.; Kaneko, K.; et al. Implication of HYBID (Hyaluronan-Binding Protein Involved in Hyaluronan Depolymerization) in Hyaluronan Degradation by Synovial Fibroblasts in Patients with Knee Osteoarthritis. Am. J. Pathol. 2020, 190, 1046–1058. [Google Scholar] [CrossRef]
- Brown, T.J.; Laurent, U.B.; Fraser, J.R. Turnover of hyaluronan in synovial joints: Elimination of labelled hyaluronan from the knee joint of the rabbit. Exp. Physiol. 1991, 76, 125–134. [Google Scholar] [CrossRef]
- Papakonstantinou, E.; Roth, M.; Karakiulakis, G. Hyaluronic acid: A key molecule in skin aging. Dermato-Endocrinol. 2012, 4, 253–258. [Google Scholar] [CrossRef]
- Liu, W.; Ma, M.; Lei, Z.; Xiong, Z.; Tao, T.; Lei, P.; Hu, Y.; Jiang, X.; Xiao, J. Intra-articular injectable hydroxypropyl chitin/hyaluronic acid hydrogel as bio-lubricant to attenuate osteoarthritis progression. Mater. Des. 2022, 217, 110579. [Google Scholar] [CrossRef]
- Ziadlou, R.; Rotman, S.; Teuschl, A.; Salzer, E.; Barbero, A.; Martin, I.; Alini, M.; Eglin, D.; Grad, S. Optimization of hyaluronic acid-tyramine/silk-fibroin composite hydrogels for cartilage tissue engineering and delivery of anti-inflammatory and anabolic drugs. Mater. Sci. Eng. C 2021, 120, 111701. [Google Scholar] [CrossRef]
- Cai, Z.; Zhang, H.; Wei, Y.; Wu, M.; Fu, A. Shear-thinning hyaluronan-based fluid hydrogels to modulate viscoelastic properties of osteoarthritis synovial fluids. Biomater. Sci. 2019, 7, 3143–3157. [Google Scholar] [CrossRef]
- Zhou, T.; Xiong, H.; Wang, S.Q.; Zhang, H.L.; Zheng, W.W.; Gou, Z.R.; Fan, C.Y.; Gao, C.Y. An injectable hydrogel dotted with dexamethasone acetate-encapsulated reactive oxygen species-scavenging micelles for combinatorial therapy of osteoarthritis. Mater. Today Nano 2022, 17, 100164. [Google Scholar] [CrossRef]
- Kandel, L.; Agar, G.; Elkayam, O.; Sharipov, A.; Slevin, O.; Rivkin, G.; Dahan, M.; Aloush, V.; Pyeser, A.B.; Brin, Y.; et al. A novel approach for knee osteoarthritis using high molecular weight hyaluronic acid conjugated to plasma fibrinogen—Interim findings of a double-blind clinical study. Heliyon 2020, 6, e04475. [Google Scholar] [CrossRef]
- Leone, G.; Pepi, S.; Consumi, M.; Lamponi, S.; Fragai, M.; Martinucci, M.; Baldoneschi, V.; Francesconi, O.; Nativi, C.; Magnani, A. Sodium hyaluronate-g-2-((N-(6-aminohexyl)-4-methoxyphenyl)sulfonamido)-N-hydroxyacetamide with enhanced affinity towards MMP12 catalytic domain to be used as visco-supplement with increased degradation resistance. Carbohydr. Polym. 2021, 271, 118452. [Google Scholar] [CrossRef]
- Schmidt, T.A.; Gastelum, N.S.; Nguyen, Q.T.; Schumacher, B.L.; Sah, R.L. Boundary lubrication of articular cartilage: Role of synovial fluid constituents. Arthritis Rheum. 2007, 56, 882–891. [Google Scholar] [CrossRef]
- Chang, D.P.; Abu-Lail, N.I.; Coles, J.M.; Guilak, F.; Jay, G.D.; Zauscher, S. Friction force microscopy of lubricin and hyaluronic acid between hydrophobic and hydrophilic surfaces. Soft Matter 2009, 5, 3438–3445. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Dellatore, S.M.; Miller, W.M.; Messersmith, P.B. Mussel-Inspired Surface Chemistry for Multifunctional Coatings. Science 2007, 318, 426–430. [Google Scholar] [CrossRef] [PubMed]
- Guarise, C.; Barbera, C.; Pavan, M.; Pluda, S.; Celestre, M.; Galesso, D. Dopamine-functionalized sulphated hyaluronic acid as a titanium implant coating enhances biofilm prevention and promotes osseointegration. Biofouling 2018, 34, 719–730. [Google Scholar] [CrossRef] [PubMed]
- Guarise, C.; Maglio, M.; Sartori, M.; Galesso, D.; Barbera, C.; Pavan, M.; Martini, L.; Giavaresi, G.; Sambri, V.; Fini, M. Titanium implant coating based on dopamine-functionalized sulphated hyaluronic acid: In vivo assessment of biocompatibility and antibacterial efficacy. Mater. Sci. Eng. C 2021, 128, 112286. [Google Scholar] [CrossRef]
- Ren, K.; Wan, H.; Kaper, H.J.; Sharma, P.K. Dopamine-conjugated hyaluronic acid delivered via intra-articular injection provides articular cartilage lubrication and protection. J. Colloid Interface Sci. 2022, 619, 207–218. [Google Scholar] [CrossRef]
- Faust, H.J.; Sommerfeld, S.D.; Rathod, S.; Rittenbach, A.; Banerjee, S.R.; Tsui, B.M.; Pomper, M.; Amzel, M.L.; Singh, A.; Elisseeff, J.H. A hyaluronic acid binding peptide-polymer system for treating osteoarthritis. Biomaterials 2018, 183, 93–101. [Google Scholar] [CrossRef]
- Goldberg, R.; Klein, J. Liposomes as lubricants: Beyond drug delivery. Chem. Phys. Lipids 2012, 165, 374–381. [Google Scholar] [CrossRef]
- Duan, Y.; Liu, Y.; Zhang, C.; Chen, Z.; Wen, S. Insight into the Tribological Behavior of Liposomes in Artificial Joints. Langmuir 2016, 32, 10957–10966. [Google Scholar] [CrossRef]
- Sivan, S.; Schroeder, A.; Verberne, G.; Merkher, Y.; Diminsky, D.; Priev, A.; Maroudas, A.; Halperin, G.; Nitzan, D.; Etsion, I.; et al. Liposomes Act as Effective Biolubricants for Friction Reduction in Human Synovial Joints. Langmuir 2010, 26, 1107–1116. [Google Scholar] [CrossRef]
- Lei, Y.; Wang, X.; Liao, J.; Shen, J.; Li, Y.; Cai, Z.; Hu, N.; Luo, X.; Cui, W.; Huang, W. Shear-responsive boundary-lubricated hydrogels attenuate osteoarthritis. Bioact. Mater. 2022, 16, 472–484. [Google Scholar] [CrossRef]
- Hilšer, P.; Suchánková, A.; Mendová, K.; Filipič, K.E.; Daniel, M.; Vrbka, M. A new insight into more effective viscosupplementation based on the synergy of hyaluronic acid and phospholipids for cartilage friction reduction. Biotribology 2021, 25, 100166. [Google Scholar] [CrossRef]
- Lin, W.; Mashiah, R.; Seror, J.; Kadar, A.; Dolkart, O.; Pritsch, T.; Goldberg, R.; Klein, J. Lipid-hyaluronan synergy strongly reduces intrasynovial tissue boundary friction. Acta Biomater. 2019, 83, 314–321. [Google Scholar] [CrossRef]
- Maudens, P.; Meyer, S.; Seemayer, C.A.; Jordan, O.; Allémann, E. Self-assembled thermoresponsive nanostructures of hyaluronic acid conjugates for osteoarthritis therapy. Nanoscale 2018, 10, 1845–1854. [Google Scholar] [CrossRef]
- Mota, A.H.; Direito, R.; Carrasco, M.P.; Rijo, P.; Ascensão, L.; Viana, A.S.; Rocha, J.; Eduardo-Figueira, M.; Rodrigues, M.J.; Custódio, L.; et al. Combination of hyaluronic acid and PLGA particles as hybrid systems for viscosupplementation in osteoarthritis. Int. J. Pharm. 2019, 559, 13–22. [Google Scholar] [CrossRef]
- Porcello, A.; Gonzalez-Fernandez, P.; Jordan, O.; Allémann, E. Nanoforming Hyaluronan-Based Thermoresponsive Hydrogels: Optimized and Tunable Functionality in Osteoarthritis Management. Pharmaceutics 2022, 14, 659. [Google Scholar] [CrossRef]
- Lei, Y.; Wang, Y.; Shen, J.; Cai, Z.; Zhao, C.; Chen, H.; Luo, X.; Hu, N.; Cui, W.; Huang, W. Injectable hydrogel microspheres with self-renewable hydration layers alleviate osteoarthritis. Sci. Adv. 2022, 8, eabl6449. [Google Scholar] [CrossRef]
- Heskins, M.; Guillet, J.E. Solution Properties of Poly(N-isopropylacrylamide). J. Macromol. Sci. Part A-Chem. 1968, 2, 1441–1455. [Google Scholar] [CrossRef]
- Cui, Z.; Lee, B.H.; Vernon, B.L. New Hydrolysis-Dependent Thermosensitive Polymer for an Injectable Degradable System. Biomacromolecules 2007, 8, 1280–1286. [Google Scholar] [CrossRef]
- Vo, T.N.; Ekenseair, A.K.; Kasper, F.K.; Mikos, A.G. Synthesis, Physicochemical Characterization, and Cytocompatibility of Bioresorbable, Dual-Gelling Injectable Hydrogels. Biomacromolecules 2014, 15, 132–142. [Google Scholar] [CrossRef]
- Yang, L.; Sun, L.; Zhang, H.; Bian, F.; Zhao, Y. Ice-Inspired Lubricated Drug Delivery Particles from Microfluidic Electrospray for Osteoarthritis Treatment. ACS Nano 2021, 15, 20600–20606. [Google Scholar] [CrossRef]
- Evans, C.H.; Kraus, V.B.; Setton, L.A. Progress in intra-articular therapy. Nat. Rev. Rheumatol. 2014, 10, 11–22. [Google Scholar] [CrossRef]
- Jin, Y.; Koh, R.H.; Kim, S.-H.; Kim, K.M.; Park, G.K.; Hwang, N.S. Injectable anti-inflammatory hyaluronic acid hydrogel for osteoarthritic cartilage repair. Mater. Sci. Eng. C 2020, 115, 111096. [Google Scholar] [CrossRef]
- Hanafy, A.S.; El-Ganainy, S.O. Thermoresponsive Hyalomer intra-articular hydrogels improve monoiodoacetate-induced osteoarthritis in rats. Int. J. Pharm. 2020, 573, 118859. [Google Scholar] [CrossRef]
- do Nascimento, M.H.M.D.; Ambrosio, F.N.; Ferraraz, D.C.; Windisch-Neto, H.; Querobino, S.M.; Nascimento-Sales, M.; Alberto-Silva, C.; Christoffolete, M.A.; Franco, M.K.; Kent, B.; et al. Sulforaphane-loaded hyaluronic acid-poloxamer hybrid hydrogel enhances cartilage protection in osteoarthritis models. Mater. Sci. Eng. C 2021, 128, 112345. [Google Scholar] [CrossRef]
- Chang, M.-C.; Chiang, P.-F.; Kuo, Y.-J.; Peng, C.-L.; Chen, K.-Y.; Chiang, Y.-C. Hyaluronan-Loaded Liposomal Dexamethasone– Diclofenac Nanoparticles for Local Osteoarthritis Treatment. Int. J. Mol. Sci. 2021, 22, 665. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Chen, M.; Yang, X.; Sun, W.; Lu, C.; Hui, Q.; Shi, C.; Li, X.; Wang, X. Modified poloxamer 407 and hyaluronic acid thermosensitive hydrogel-encapsulated keratinocyte growth factor 2 improves knee osteoarthritis in rats. Mater. Des. 2021, 210, 110086. [Google Scholar] [CrossRef]
- Yang, Y.; Zhu, Z.; Gao, R.; Yuan, J.; Zhang, J.; Li, H.; Xie, Z.; Wang, Y. Controlled release of MSC-derived small extracellular vesicles by an injectable Diels-Alder crosslinked hyaluronic acid/PEG hydrogel for osteoarthritis improvement. Acta Biomater. 2021, 128, 163–174. [Google Scholar] [CrossRef] [PubMed]
- Lu, K.-Y.; Lin, Y.-C.; Lu, H.-T.; Ho, Y.-C.; Weng, S.-C.; Tsai, M.-L.; Mi, F.-L. A novel injectable in situ forming gel based on carboxymethyl hexanoyl chitosan/hyaluronic acid polymer blending for sustained release of berberine. Carbohydr. Polym. 2019, 206, 664–673. [Google Scholar] [CrossRef]
- Wang, J.; Wang, X.; Cao, Y.; Huang, T.; Song, D.-X.; Tao, H.-R. Therapeutic potential of hyaluronic acid/chitosan nanoparticles for the delivery of curcuminoid in knee osteoarthritis and an in vitro evaluation in chondrocytes. Int. J. Mol. Med. 2018, 42, 2604–2614. [Google Scholar] [CrossRef]
- Eladawy, N.O.; Morsi, N.M.; Shamma, R.N. Diacerein-Loaded Hyaluosomes as a Dual-Function Platform for Osteoarthritis Management via Intra-Articular Injection: In Vitro Characterization and In Vivo Assessment in a Rat Model. Pharmaceutics 2021, 13, 765. [Google Scholar] [CrossRef]
- Lei, Y.; Wang, Y.; Shen, J.; Cai, Z.; Zeng, Y.; Zhao, P.; Liao, J.; Lian, C.; Hu, N.; Luo, X.; et al. Stem Cell-Recruiting Injectable Microgels for Repairing Osteoarthritis. Adv. Funct. Mater. 2021, 31, 2105084. [Google Scholar] [CrossRef]
- Takayama, K.; Kawakami, Y.; Kobayashi, M.; Greco, N.; Cummins, J.H.; Matsushita, T.; Kuroda, R.; Kurosaka, M.; Fu, F.H.; Huard, J. Local intra-articular injection of rapamycin delays articular cartilage degeneration in a murine model of osteoarthritis. Arthritis Res. Ther. 2014, 16, 482. [Google Scholar] [CrossRef]
- Caramés, B.; Taniguchi, N.; Seino, D.; Blanco, F.J.; D’Lima, D.; Lotz, M. Mechanical injury suppresses autophagy regulators and pharmacologic activation of autophagy results in chondroprotection. Arthritis Rheum. 2011, 64, 1182–1192. [Google Scholar] [CrossRef]
- Agas, D.; Laus, F.; Lacava, G.; Marchegiani, A.; Deng, S.; Magnoni, F.; Silva, G.G.; Di Martino, P.; Sabbieti, M.G.; Censi, R. Thermosensitive hybrid hyaluronan/p(HPMAm-lac)-PEG hydrogels enhance cartilage regeneration in a mouse model of osteoarthritis. J. Cell. Physiol. 2019, 234, 20013–20027. [Google Scholar] [CrossRef]
- Kang, L.-J.; Yoon, J.; Rho, J.G.; Han, H.S.; Lee, S.; Oh, Y.S.; Kim, H.; Kim, E.; Kim, S.J.; Lim, Y.T.; et al. Self-assembled hyaluronic acid nanoparticles for osteoarthritis treatment. Biomaterials 2021, 275, 120967. [Google Scholar] [CrossRef]
- Jiang, D.; Liang, J.; Noble, P.W. Hyaluronan in Tissue Injury and Repair. Annu. Rev. Cell Dev. Biol. 2007, 23, 435–461. [Google Scholar] [CrossRef]
- Garcia, J.P.; Stein, J.; Cai, Y.; Riemers, F.; Wexselblatt, E.; Wengel, J.; Tryfonidou, M.; Yayon, A.; Howard, K.A.; Creemers, L.B. Fibrin-hyaluronic acid hydrogel-based delivery of antisense oligonucleotides for ADAMTS5 inhibition in co-delivered and resident joint cells in osteoarthritis. J. Control. Release 2019, 294, 247–258. [Google Scholar] [CrossRef]
- Zhou, P.; Qiu, B.; Deng, R.; Li, H.; Xu, X.; Shang, X. Chondroprotective Effects of Hyaluronic Acid-Chitosan Nanoparticles Containing Plasmid DNA Encoding Cytokine Response Modifier A in a Rat Knee Osteoarthritis Model. Cell. Physiol. Biochem. 2018, 47, 1207–1216. [Google Scholar] [CrossRef]
- Chen, Z.; Zhang, F.; Zhang, H.; Cheng, L.; Chen, K.; Shen, J.; Qi, J.; Deng, L.; He, C.; Santos, H.A.; et al. DNA-Grafted Hyaluronic Acid System with Enhanced Injectability and Biostability for Photo-Controlled Osteoarthritis Gene Therapy. Adv. Sci. 2021, 8, 2004793. [Google Scholar] [CrossRef]
- Tortorella, M.D.; Burn, T.C.; Pratta, M.A.; Abbaszade, I.; Hollis, J.M.; Liu, R.; Rosenfeld, S.A.; Copeland, R.A.; Decicco, C.P.; Wynn, R.; et al. Purification and Cloning of Aggrecanase-1: A Member of the ADAMTS Family of Proteins. Science 1999, 284, 1664–1666. [Google Scholar] [CrossRef]
- Tortorella, M.D.; Pratta, M.; Liu, R.-Q.; Austin, J.; Ross, O.H.; Abbaszade, I.; Burn, T.; Arner, E. Sites of Aggrecan Cleavage by Recombinant Human Aggrecanase-1 (ADAMTS-4). J. Biol. Chem. 2000, 275, 18566–18573. [Google Scholar] [CrossRef]
- Tortorella, M.; Pratta, M.; Liu, R.-Q.; Abbaszade, I.; Ross, H.; Burn, T.; Arner, E. The Thrombospondin Motif of Aggrecanase-1 (ADAMTS-4) Is Critical for Aggrecan Substrate Recognition and Cleavage. J. Biol. Chem. 2000, 275, 25791–25797. [Google Scholar] [CrossRef]
- Glasson, S.S.; Askew, R.; Sheppard, B.; Carito, B.A.; Blanchet, T.; Ma, H.-L.; Flannery, C.R.; Kanki, K.; Wang, E.; Peluso, D.; et al. Characterization of and osteoarthritis susceptibility in ADAMTS-4-knockout mice. Arthritis Rheum. 2004, 50, 2547–2558. [Google Scholar] [CrossRef] [PubMed]
- Majumdar, M.K.; Askew, R.; Schelling, S.; Stedman, N.; Blanchet, T.; Hopkins, B.; Morris, E.A.; Glasson, S.S. Double-knockout of ADAMTS-4 and ADAMTS-5 in mice results in physiologically normal animals and prevents the progression of osteoarthritis. Arthritis Rheum. 2007, 56, 3670–3674. [Google Scholar] [CrossRef] [PubMed]
- Kawasaki, K.; Ochi, M.; Uchio, Y.; Adachi, N.; Matsusaki, M. Hyaluronic acid enhances proliferation and chondroitin sulfate synthesis in cultured chondrocytes embedded in collagen gels. J. Cell. Physiol. 1999, 179, 142–148. [Google Scholar] [CrossRef]
- Prasadam, I.; Mao, X.; Shi, W.; Crawford, R.; Xiao, Y. Combination of MEK-ERK inhibitor and hyaluronic acid has a synergistic effect on anti-hypertrophic and pro-chondrogenic activities in osteoarthritis treatment. J. Mol. Med. 2013, 91, 369–380. [Google Scholar] [CrossRef]
- Kikuchi, T.; Sakuta, T.; Yamaguchi, T. Effects of hyaluronan on cell proliferation and proteoglycan synthesis in rabbit ligamental cells. Int. J. Tissue React. 1996, 18, 87–95. [Google Scholar]
- Takahashi, K.; Goomer, R.S.; Harwood, F.; Kubo, T.; Hirasawa, Y.; Amiel, D. The effects of hyaluronan on matrix metalloproteinase-3 (MMP-3), interleukin-1β(IL-1β), and tissue inhibitor of metalloproteinase-1 (TIMP-1) gene expression during the development of osteoarthritis. Osteoarthr. Cartil. 1999, 7, 182–190. [Google Scholar] [CrossRef]
- Yasui, T.; Akatsuka, M.; Tobetto, K.; Umemoto, J.; Ando, T.; Yamashita, K.; Hayakawa, T. Effects of Hyaluronan on the Production of Stromelysin and Tissue Inhibitor of Metalloproteinase-1 (TIMP-1) in Bovine Articular Chondrocytes. Biomed. Res. 1992, 13, 343–348. [Google Scholar] [CrossRef]
- Siegelman, M.H.; Degrendele, H.C.; Estess, P. Activation and interaction of CD44 and hyaluronan in immunological systems. J. Leukoc. Biol. 1999, 66, 315–321. [Google Scholar] [CrossRef]
- Altman, R.; Bedi, A.; Manjoo, A.; Niazi, F.; Shaw, P.; Mease, P. Anti-Inflammatory Effects of Intra-Articular Hyaluronic Acid: A Systematic Review. Cartilage 2019, 10, 43–52. [Google Scholar] [CrossRef]
- Chow, G.; Nietfeld, J.J.; Knudson, C.B.; Knudson, W. Antisense inhibition of chondrocyte CD44 expression leading to cartilage chondrolysis. Arthritis Rheum. 1998, 41, 1411–1419. [Google Scholar] [CrossRef]
- Matas, J.; Orrego, M.; Amenabar, D.; Infante, C.; Tapia-Limonchi, R.; Cadiz, M.I.; Alcayaga-Miranda, F.; González, P.L.; Muse, E.; Khoury, M.; et al. Umbilical Cord-Derived Mesenchymal Stromal Cells (MSCs) for Knee Osteoarthritis: Repeated MSC Dosing Is Superior to a Single MSC Dose and to Hyaluronic Acid in a Controlled Randomized Phase I/II Trial. Steam Cells Transl. Med. 2019, 8, 215–224. [Google Scholar] [CrossRef]
- Zhu, Y.; Ye, L.; Cai, X.; Li, Z.; Fan, Y.; Yang, F. Icariin-Loaded Hydrogel Regulates Bone Marrow Mesenchymal Stem Cell Chondrogenic Differentiation and Promotes Cartilage Repair in Osteoarthritis. Front. Bioeng. Biotechnol. 2022, 10, 755260. [Google Scholar] [CrossRef]
- Wei, J.; Ran, P.; Li, Q.; Lu, J.; Zhao, L.; Liu, Y.; Li, X. Hierarchically structured injectable hydrogels with loaded cell spheroids for cartilage repairing and osteoarthritis treatment. Chem. Eng. J. 2022, 430, 132211. [Google Scholar] [CrossRef]
- Yang, J.; Liu, Y.; He, L.; Wang, Q.; Wang, L.; Yuan, T.; Xiao, Y.; Fan, Y.; Zhang, X. Icariin conjugated hyaluronic acid/collagen hydrogel for osteochondral interface restoration. Acta Biomater. 2018, 74, 156–167. [Google Scholar] [CrossRef]
- Wang, Z.C.; Sun, H.J.; Li, K.H.; Fu, C.; Liu, M.Z. Icariin promotes directed chondrogenic differentiation of bone marrow mesenchymal stem cells but not hypertrophy in vitro. Exp. Ther. Med. 2014, 8, 1528–1534. [Google Scholar] [CrossRef]
- Li, D.; Yuan, T.; Zhang, X.; Xiao, Y.; Wang, R.; Fan, Y.; Zhang, X. Icariin: A potential promoting compound for cartilage tissue engineering. Osteoarthr. Cartil. 2012, 20, 1647–1656. [Google Scholar] [CrossRef]
- Zhang, L.; Wei, Y.; Chi, Y.; Liu, D.; Yang, S.; Han, Z.; Li, Z. Two-step generation of mesenchymal stem/stromal cells from human pluripotent stem cells with reinforced efficacy upon osteoarthritis rabbits by HA hydrogel. Cell Biosci. 2021, 11, 6. [Google Scholar] [CrossRef]
- Blaney Davidson, E.N.; van der Kraan, P.M.; van den Berg, W.B. TGF-β and osteoarthritis. Osteoarthr. Cartil. 2007, 15, 597–604. [Google Scholar] [CrossRef]
- Ahmadi, E.D.; Raja, T.I.; Khaghani, S.A.; Soon, C.F.; Mozafari, M.; Youseffi, M.; Sefat, F. The role of photonics and natural curing agents of TGF-β1 in treatment of osteoarthritis. Mater. Today Proc. 2018, 5, 15540–15549. [Google Scholar] [CrossRef][Green Version]
- Yu, W.; Hu, B.; Boakye-Yiadom, K.O.; Ho, W.; Chen, Q.; Xu, X.; Zhang, X.-Q. Injectable hydrogel mediated delivery of gene-engineered adipose-derived stem cells for enhanced osteoarthritis treatment. Biomater. Sci. 2021, 9, 7603–7616. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Peng, L.; Li, L.; Huang, C.; Shi, K.; Meng, X.; Wang, P.; Wu, M.; Li, L.; Cao, H.; et al. 3D-bioprinted BMSC-laden biomimetic multiphasic scaffolds for efficient repair of osteochondral defects in an osteoarthritic rat model. Biomaterials 2021, 279, 121216. [Google Scholar] [CrossRef] [PubMed]
- Shi, W.; Fang, F.; Kong, Y.; Greer, S.E.; Kuss, M.A.; Liu, B.; Xue, W.; Jiang, X.; Lovell, P.; Mohs, A.M.; et al. Dynamic hyaluronic acid hydrogel with covalent linked gelatin as an anti-oxidative bioink for cartilage tissue engineering. Biofabrication 2021, 14, 014107. [Google Scholar] [CrossRef]
- Song, J.-S.; Hong, K.-T.; Kim, N.-M.; Jung, J.-Y.; Park, H.-S.; Lee, S.H.; Cho, Y.J.; Kim, S.J. Implantation of allogenic umbilical cord blood-derived mesenchymal stem cells improves knee osteoarthritis outcomes: Two-year follow-up. Regen. Ther. 2020, 14, 32–39. [Google Scholar] [CrossRef]
- Song, J.-S.; Hong, K.-T.; Kim, N.-M.; Park, H.-S.; Choi, N.-H. Human umbilical cord blood-derived mesenchymal stem cell implantation for osteoarthritis of the knee. Arch. Orthop. Trauma. Surg. 2020, 140, 503–509. [Google Scholar] [CrossRef]
- Berenbaum, F.; Grifka, J.; Cazzaniga, S.; D’Amato, M.; Giacovelli, G.; Chevalier, X.; Rannou, F.; Rovati, L.C.; Maheu, E. A randomised, double-blind, controlled trial comparing two intra-articular hyaluronic acid preparations differing by their molecular weight in symptomatic knee osteoarthritis. Ann. Rheum. Dis. 2012, 71, 1454–1460. [Google Scholar] [CrossRef]
- Altman, R.D.; Bedi, A.; Karlsson, J.; Sancheti, P.; Schemitsch, E. Product Differences in Intra-articular Hyaluronic Acids for Osteoarthritis of the Knee. Am. J. Sports Med. 2016, 44, 2158–2165. [Google Scholar] [CrossRef]
- Migliore, A.; Granata, M. Intra-Articular Use of Hyaluronic Acid in the Treatment of Osteoarthritis. Clin. Interv. Aging 2008, 3, 365–369. [Google Scholar] [CrossRef]
- Li, Y.; Cao, J.; Han, S.; Liang, Y.; Zhang, T.; Zhao, H.; Wang, L.; Sun, Y. ECM based injectable thermo-sensitive hydrogel on the recovery of injured cartilage induced by osteoarthritis. Artif. Cells Nanomed. Biotechnol. 2018, 46, 152–160. [Google Scholar] [CrossRef]
- Wang, C.-T.; Lin, Y.-T.; Chiang, B.-L.; Lin, Y.-H.; Hou, S.-M. High molecular weight hyaluronic acid down-regulates the gene expression of osteoarthritis-associated cytokines and enzymes in fibroblast-like synoviocytes from patients with early osteoarthritis. Osteoarthr. Cartil. 2006, 14, 1237–1247. [Google Scholar] [CrossRef]
- Kwon, M.Y.; Wang, C.; Galarraga, J.H.; Puré, E.; Han, L.; Burdick, J.A. Influence of hyaluronic acid modification on CD44 binding towards the design of hydrogel biomaterials. Biomaterials 2019, 222, 119451. [Google Scholar] [CrossRef]
- Wu, S.; Xu, R.; Duan, B.; Jiang, P. Three-dimensional hyaluronic acid hydrogel-based models for in vitro human iPSC-derived NPC culture and differentiation. J. Mater. Chem. B 2017, 5, 3870–3878. [Google Scholar] [CrossRef]
- Lam, J.; Carmichael, S.T.; Lowry, W.E.; Segura, T. Hydrogel Design of Experiments Methodology to Optimize Hydrogel for iPSC-NPC Culture. Adv. Healthc. Mater. 2015, 4, 534–539. [Google Scholar] [CrossRef]
- Adkar, S.S.; Wu, C.-L.; Willard, V.P.; Dicks, A.; Ettyreddy, A.; Steward, N.; Bhutani, N.; Gersbach, C.A.; Guilak, F. Step-Wise Chondrogenesis of Human Induced Pluripotent Stem Cells and Purification Via a Reporter Allele Generated by CRISPR-Cas9 Genome Editing. Stem Cells 2019, 37, 65–76. [Google Scholar] [CrossRef]
- Wu, C.-L.; Dicks, A.; Steward, N.; Tang, R.; Katz, D.B.; Choi, Y.-R.; Guilak, F. Single cell transcriptomic analysis of human pluripotent stem cell chondrogenesis. Nat. Commun. 2021, 12, 362. [Google Scholar] [CrossRef]
- Maleki, A.; Kjøniksen, A.-L.; Nyström, B. Characterization of the chemical degradation of hyaluronic acid during chemical gelation in the presence of different cross-linker agents. Carbohydr. Res. 2007, 342, 2776–2792. [Google Scholar] [CrossRef]
- Jeon, O.; Song, S.J.; Lee, K.-J.; Park, M.H.; Lee, S.-H.; Hahn, S.K.; Kim, S.; Kim, B.-S. Mechanical properties and degradation behaviors of hyaluronic acid hydrogels cross-linked at various cross-linking densities. Carbohydr. Polym. 2007, 70, 251–257. [Google Scholar] [CrossRef]
- Kim, I.L.; Mauck, R.L.; Burdick, J.A. Hydrogel design for cartilage tissue engineering: A case study with hyaluronic acid. Biomaterials 2011, 32, 8771–8782. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kim, Y.S.; Guilak, F. Engineering Hyaluronic Acid for the Development of New Treatment Strategies for Osteoarthritis. Int. J. Mol. Sci. 2022, 23, 8662. https://doi.org/10.3390/ijms23158662
Kim YS, Guilak F. Engineering Hyaluronic Acid for the Development of New Treatment Strategies for Osteoarthritis. International Journal of Molecular Sciences. 2022; 23(15):8662. https://doi.org/10.3390/ijms23158662
Chicago/Turabian StyleKim, Yu Seon, and Farshid Guilak. 2022. "Engineering Hyaluronic Acid for the Development of New Treatment Strategies for Osteoarthritis" International Journal of Molecular Sciences 23, no. 15: 8662. https://doi.org/10.3390/ijms23158662
APA StyleKim, Y. S., & Guilak, F. (2022). Engineering Hyaluronic Acid for the Development of New Treatment Strategies for Osteoarthritis. International Journal of Molecular Sciences, 23(15), 8662. https://doi.org/10.3390/ijms23158662