Pharmacological Screening of Venoms from Five Brazilian Micrurus Species on Different Ion Channels
Abstract
1. Introduction
2. Results
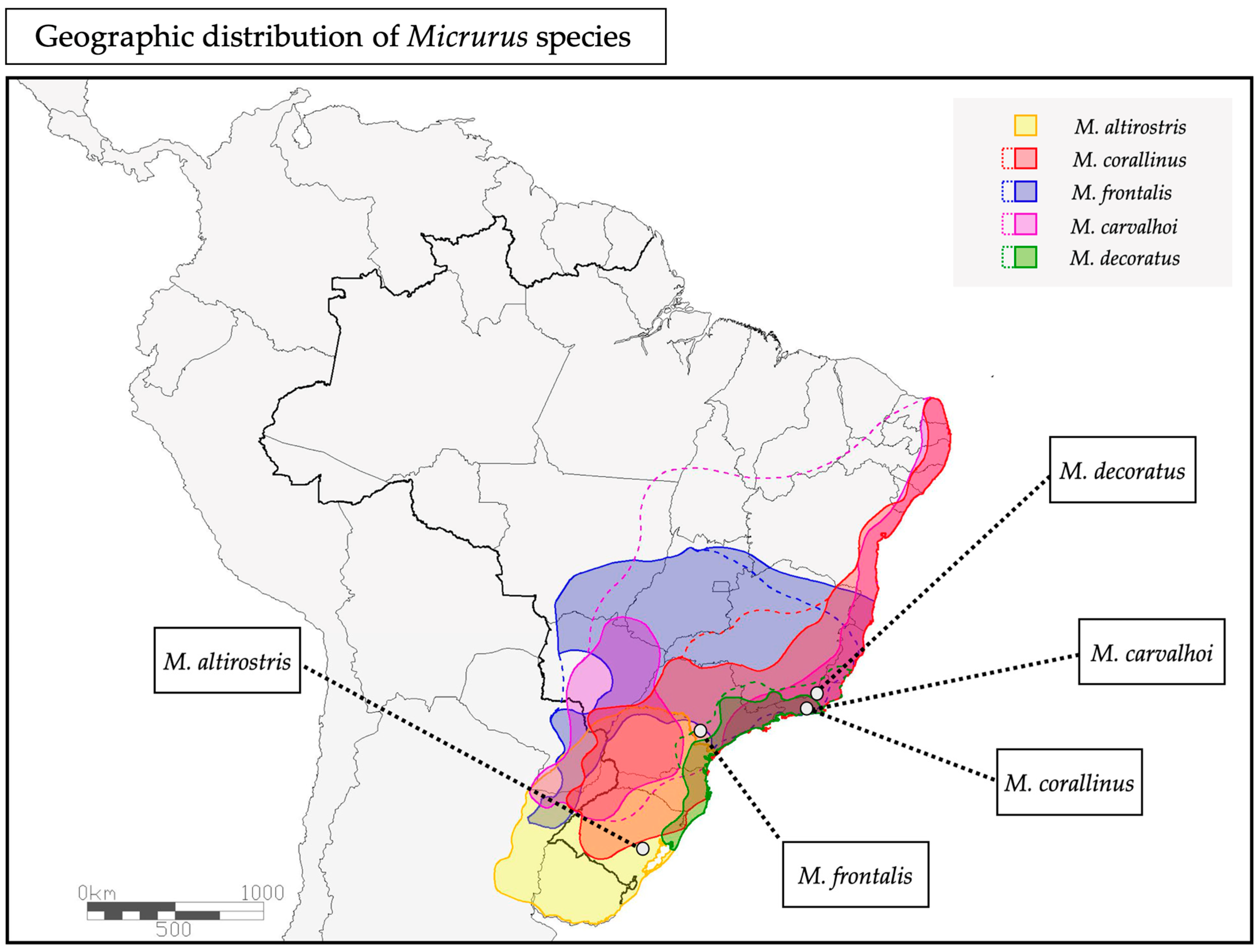
2.1. Chromatographic Profile of Five Micrurus Venoms from the South and Southeast Brazil
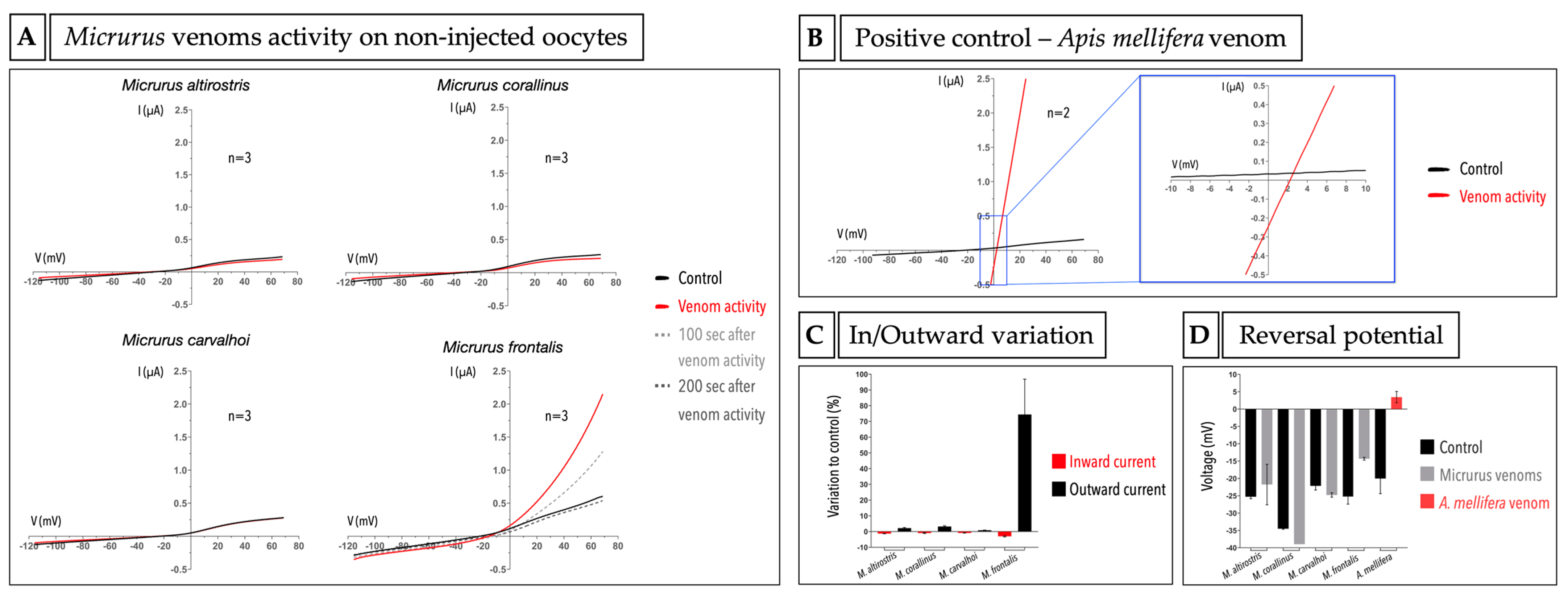
2.2. Micrurus Venoms Are Not Cytotoxic to X. laevis Oocytes
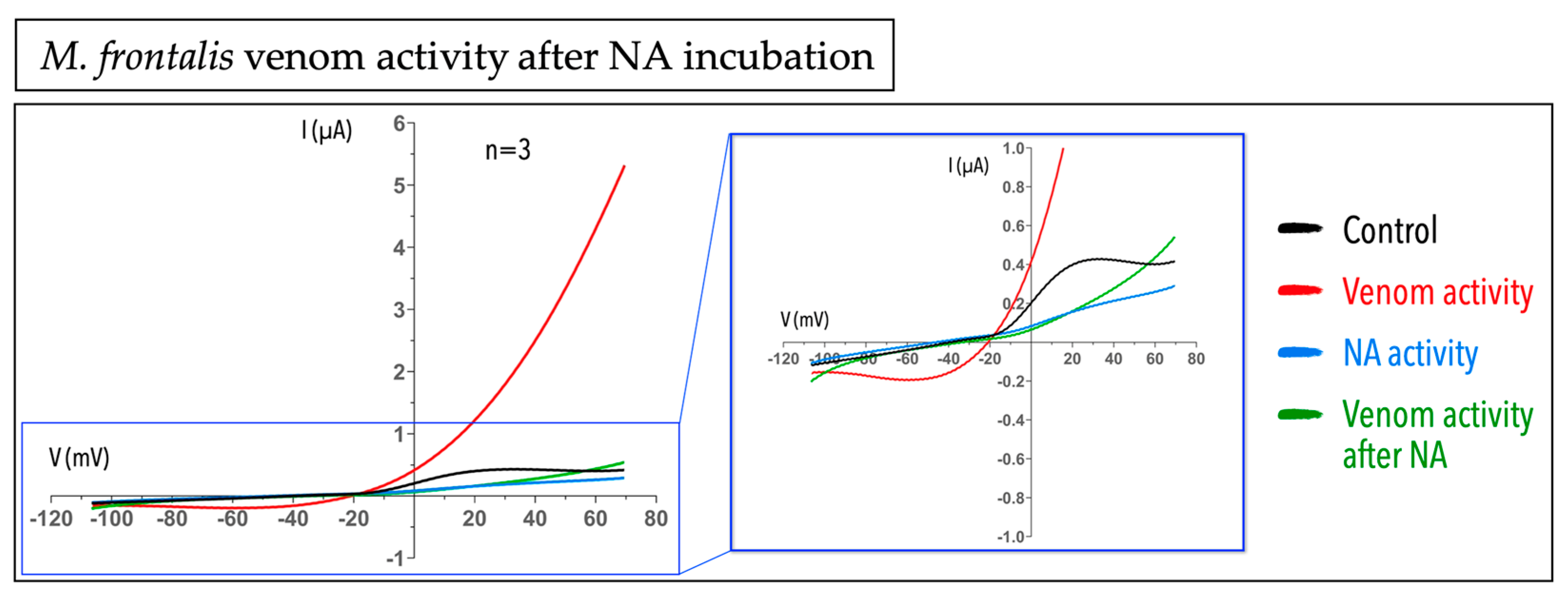
2.3. Micrurus Frontalis Venom Evoked Cl− Outward Current by Acting Directly or Indirectly on Ca2+-Activated Cl− Channels
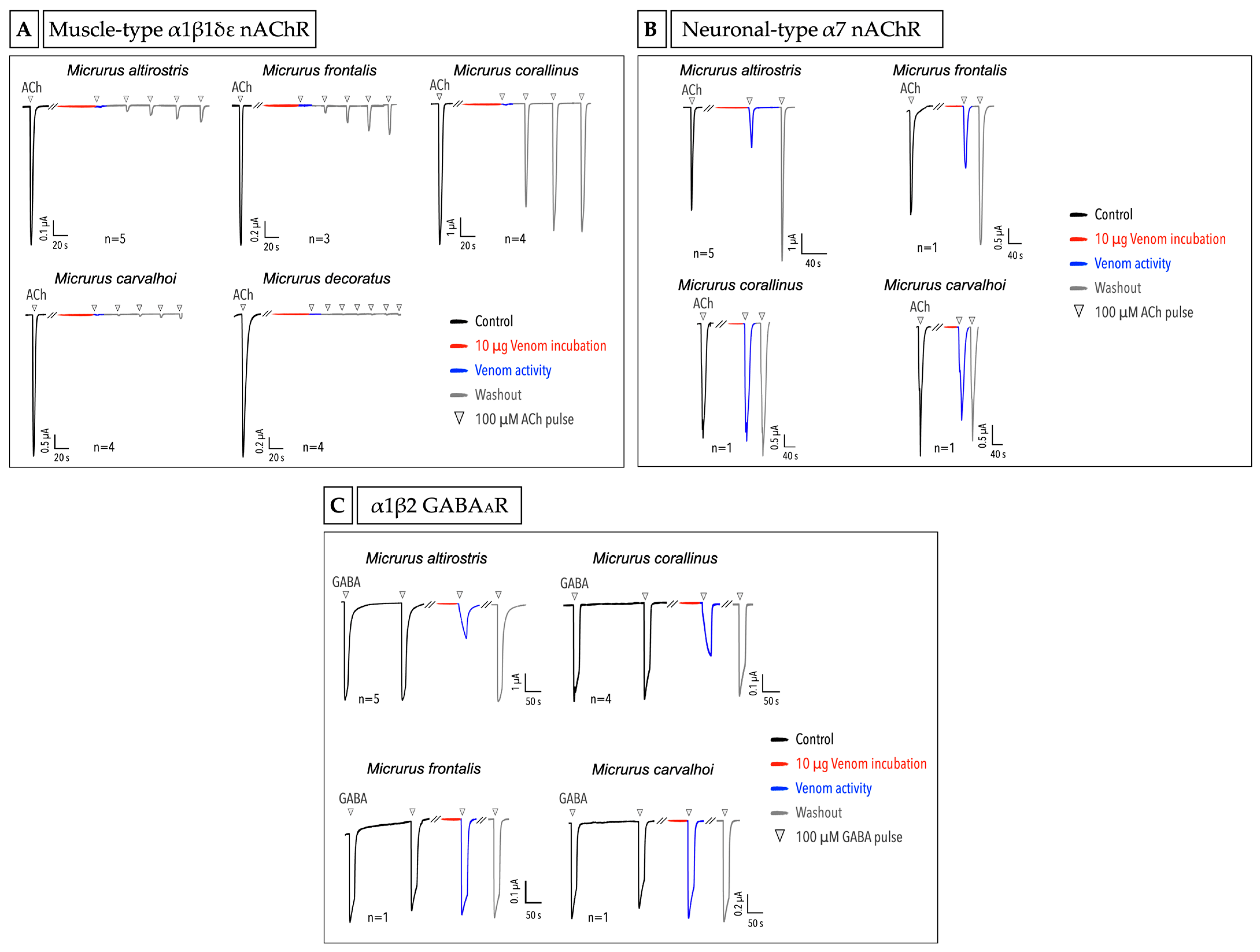
2.4. Micrurus Venoms Act in Different Ion Channels
3. Discussion
4. Conclusions
5. Materials and Methods
5.1. Venoms
5.2. RP-HPLC Profile
5.3. Electrophysiologic Recordings on Xenopus laevis Oocytes
5.3.1. Cytotoxic Experiments with Non-Injected Oocytes
5.3.2. Niflumic Acid Assay
5.3.3. Ion Channels Heterologous Expression and Recordings in Oocytes
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Uetz, P.; Koo, M.S.; Aguilar, R.; Catenazzi, A. A Quarter Century of Reptile and Amphibian Databases. Herpetol. Rev. 2021, 52, 246–255. [Google Scholar]
- Lomonte, B.; Rangel, J. Snake Venom Lys49 Myotoxins: From Phospholipases A 2 to Non-Enzymatic Membrane Disruptors. Toxicon 2012, 60, 520–530. [Google Scholar] [CrossRef]
- Kini, R.M.; Doley, R. Structure, Function and Evolution of Three-Finger Toxins: Mini Proteins with Multiple Targets. Toxicon 2010, 56, 855–867. [Google Scholar] [CrossRef]
- Utkin, Y.N. Three-Finger Toxins, a Deadly Weapon of Elapid Venom-Milestones of Discovery. Toxicon 2013, 62, 50–55. [Google Scholar] [CrossRef]
- Baudou, F.G.; Rodriguez, J.P.; Fusco, L.; de Roodt, A.R.; de Marzi, M.C.; Leiva, L. South American Snake Venoms with Abundant Neurotoxic Components. Composition and Toxicological Properties. A Literature Review. Acta Trop. 2021, 224, 106119. [Google Scholar] [CrossRef]
- Lomonte, B.; Rey-Suárez, P.; Fernández, J.; Sasa, M.; Pla, D.; Vargas, N.; Bénard-Valle, M.; Sanz, L.; Corrêa-Netto, C.; Núñez, V.; et al. Venoms of Micrurus Coral Snakes: Evolutionary Trends in Compositional Patterns Emerging from Proteomic Analyses. Toxicon 2016, 122, 7–25. [Google Scholar] [CrossRef]
- Sanz, L.; Quesada-Bernat, S.; Ramos, T.; Casais-e-Silva, L.L.; Corrêa-Netto, C.; Silva-Haad, J.J.; Sasa, M.; Lomonte, B.; Calvete, J.J. New Insights into the Phylogeographic Distribution of the 3FTx/PLA 2 Venom Dichotomy across Genus Micrurus in South America. J. Proteom. 2019, 200, 90–101. [Google Scholar] [CrossRef]
- Aird, S.D.; da Silva, N.J.; Qiu, L.; Villar-Briones, A.; Saddi, V.A.; de Telles, M.P.C.; Grau, M.L.; Mikheyev, A.S. Coralsnake Venomics: Analyses of Venom Gland Transcriptomes and Proteomes of Six Brazilian Taxa. Toxins 2017, 9, 187. [Google Scholar] [CrossRef]
- Corrêa-Netto, C.; Junqueira-de-Azevedo, I.d.L.M.; Silva, D.A.; Ho, P.L.; Leitão-de-Araújo, M.; Alves, M.L.M.; Sanz, L.; Foguel, D.; Zingali, R.B.; Calvete, J.J. Snake Venomics and Venom Gland Transcriptomic Analysis of Brazilian Coral Snakes, Micrurus Altirostris and M. Corallinus. J. Proteom. 2011, 74, 1795–1809. [Google Scholar] [CrossRef]
- Sanz, L.; de Freitas-Lima, L.N.; Quesada-Bernat, S.; Graça-de-Souza, V.K.; Soares, A.M.; Calderón, L.d.A.; Calvete, J.J.; Caldeira, C.A.S. Comparative Venomics of Brazilian Coral Snakes: Micrurus Frontalis, Micrurus Spixii Spixii, and Micrurus Surinamensis. Toxicon 2019, 166, 39–45. [Google Scholar] [CrossRef]
- Airi, S.D.; Jorge, N.; Silva, D.A. Comparative Enzymatic Composition Of Brazilian Coral Snake (Micrurus) Venoms. Comp. Biochem. Physiol. Part B Comp. Biochem. 1991, 99, 287–294. [Google Scholar]
- Margres, M.J.; Aronow, K.; Loyacano, J.; Rokyta, D.R. The Venom-Gland Transcriptome of the Eastern Coral Snake (Micrurus Fulvius) Reveals High Venom Complexity in the Intragenomic Evolution of Venoms. BMC Genom. 2013, 14, 531. [Google Scholar] [CrossRef]
- Aird, S.D. Ophidian Envenomation Strategies and the Role of Purines. Toxicon 2002, 40, 335–393. [Google Scholar] [CrossRef]
- da Silva, N.J., Jr.; Aird, S.D. Prey Specificity, Comparative Lethality and Compositional Differences of Coral Snake Venoms. Comp. Biochem. Physiol. Part C Toxicol. Pharmacol. 2001, 128, 425–456. [Google Scholar] [CrossRef]
- Rey-Suárez, P.; Núñez, V.; Gutiérrez, J.M.; Lomonte, B. Proteomic and Biological Characterization of the Venom of the Redtail Coral Snake, Micrurus Mipartitus (Elapidae), from Colombia and Costa Rica. J. Proteom. 2011, 75, 655–667. [Google Scholar] [CrossRef]
- Kleiz-Ferreira, J.M.; Cirauqui, N.; Trajano, E.A.; Almeida, M.d.S.; Zingali, R.B. Three-Finger Toxins from Brazilian Coral Snakes: From Molecular Framework to Insights in Biological Function. Toxins 2021, 13, 328. [Google Scholar] [CrossRef]
- Coelho Da Silva, D.; Araújo De Medeiros, W.A.; de Fátima Correia Batista, I.; Pimenta, D.C.; Lebrun, I.; Francis Abdalla, F.M.; Lopes Sandoval, M.R. Characterization of a New Muscarinic Toxin from the Venom of the Brazilian Coral Snake Micrurus Lemniscatus in Rat Hippocampus. Life Sci. 2011, 89, 931–938. [Google Scholar] [CrossRef]
- Silveira De Oliveira, J.; Rossan De Brandão Prieto Da Silva, Á.; Soares, M.B.; Stephano, M.A.; de Oliveira Dias, W.; Raw, I.; Ho, P.L. Cloning and Characterization of an α-Neurotoxin-Type Protein Specific for the Coral Snake Micrurus Corallinus. Biochem. Biophys. Res. Commun. 2000, 267, 887–891. [Google Scholar] [CrossRef]
- Moreira, K.G.; Prates, M.V.; Andrade, F.A.C.; Silva, L.P.; Beirão, P.S.L.; Kushmerick, C.; Naves, L.A.; Bloch, C. Frontoxins, Three-Finger Toxins from Micrurus Frontalis Venom, Decrease Miniature Endplate Potential Amplitude at Frog Neuromuscular Junction. Toxicon 2010, 56, 55–63. [Google Scholar] [CrossRef]
- Terra, A.L.C.; Moreira-Dill, L.S.; Simões-Silva, R.; Monteiro, J.R.N.; Cavalcante, W.L.G.; Gallacci, M.; Barros, N.B.; Nicolete, R.; Teles, C.B.G.; Medeiros, P.S.M.; et al. Biological Characterization of the Amazon Coral Micrurus Spixii Snake Venom: Isolation of a New Neurotoxic Phospholipase A2. Toxicon 2015, 103, 1–11. [Google Scholar] [CrossRef]
- Bucaretchi, F.; de Capitani, E.M.; Vieira, R.J.; Rodrigues, C.K.; Zannin, M.; da Silva, N.J.; Casais-E-Silva, L.L.; Hyslop, S. Coral Snake Bites (Micrurus spp.) in Brazil: A Review of Literature Reports. Clin. Toxicol. 2016, 54, 222–234. [Google Scholar] [CrossRef]
- Cecchini, A.L.; Marcussi, S.; Silveira, L.B.; Borja-Oliveira, C.R.; Rodrigues-Simioni, L.; Amara, S.; Stábeli, R.G.; Giglio, J.R.; Arantes, E.C.; Soares, A.M. Biological and Enzymatic Activities of Micrurus Sp. (Coral) Snake Venoms. Comp. Biochem. Physiol. -A Mol. Integr. Physiol. 2005, 140, 125–134. [Google Scholar] [CrossRef]
- Vital Brazil, O. Coral Snake Venoms: Mode of Action and Pathophysiology of Experimental Envenomation (1). Rev. Do Inst. De Med. Trop. De Sao Paulo 1987, 29, 119–126. [Google Scholar] [CrossRef]
- Moraes, F.V.; Sousa-E-Silva, M.C.C.; Barbaro, K.C.; Leitão, M.A.; Furtado, M.F.D. Biological and Immunochemical Characterization of Micrurus Altirostris Venom and Serum Neutralization of Its Toxic Activities. Toxicon 2002, 41, 71–79. [Google Scholar] [CrossRef]
- Bénard-Valle, M.; Carbajal-Saucedo, A.; de Roodt, A.; López-Vera, E.; Alagón, A. Biochemical Characterization of the Venom of the Coral Snake Micrurus Tener and Comparative Biological Activities in the Mouse and a Reptile Model. Toxicon 2014, 77, 6–15. [Google Scholar] [CrossRef]
- Francis, B.R.; da Silva, N.J., Jr.; Seebart, C.; Silva, L.L.C.E.; Schmidt, J.J.; Kaiser, I.I. Toxins isolated from the venom of the brazilian coral snake (m1crurus frontalis frontalis) include hemorrhagic type phospholipases a, and postsynaptic neurotoxins. Toxicon 1997, 35, 1193–1203. [Google Scholar] [CrossRef]
- Carbajal-Saucedo, A.; Floriano, R.S.; Dal Belo, C.A.; Olvera-Rodríguez, A.; Alagón, A.; Rodrigues-Simioni, L. Neuromuscular Activity of Micrurus Laticollaris (Squamata: Elapidae) Venom In Vitro. Toxins 2013, 6, 359–370. [Google Scholar] [CrossRef]
- Dal Belo, C.A.; Toyama, M.H.; Toyama, D.D.O.; Marangoni, S.; Moreno, F.B.; Cavada, B.S.; Fontana, M.D.; Hyslop, S.; Carneiro, E.M.; Boschero, A.C. Determination of the Amino Acid Sequence of a New Phospholipase A 2 (MIDCA1) Isolated from Micrurus Dumerilii Carinicauda Venom. Protein J. 2005, 24, 147–153. [Google Scholar] [CrossRef]
- Oliveira, D.A.; Harasawa, C.; Seibert, C.S.; Casais e Silva, L.L.; Pimenta, D.C.; Lebrun, I.; Sandoval, M.R.L. Phospholipases A2 Isolated from Micrurus Lemniscatus Coral Snake Venom: Behavioral, Electroencephalographic, and Neuropathological Aspects. Brain Res. Bull. 2008, 75, 629–639. [Google Scholar] [CrossRef]
- dos Santos, R.T.F.; Silva, M.F.P.; Porto, R.M.; Lebrun, I.; de Camargo Gonçalves, L.R.; de Fátima Correia Batista, I.; Sandoval, M.R.L.; Abdalla, F.M.F. Effects of Mlx-8, a Phospholipase A2 from Brazilian Coralsnake Micrurus Lemniscatus Venom, on Muscarinic Acetylcholine Receptors in Rat Hippocampus. J. Venom. Anim. Toxins Incl. Trop. Dis. 2020, 26, e20190041. [Google Scholar] [CrossRef]
- Kessler, P.; Marchot, P.; Silva, M.; Servent, D. The Three-Finger Toxin Fold: A Multifunctional Structural Scaffold Able to Modulate Cholinergic Functions. J. Neurochem. 2017, 142, 7–18. [Google Scholar] [CrossRef]
- Utkin, Y.N. Last Decade Update for Three-Finger Toxins: Newly Emerging Structures and Biological Activities. World J. Biol. Chem. 2019, 10, 17–27. [Google Scholar] [CrossRef]
- Rosso, J.-P.; Schwarz, J.R.; Diaz-Bustamante, M.; Céard, B.; Gutiérrez, J.M.; Kneussel, M.; Pongs, O.; Bosmans, F.; Bougis, P.E. MmTX1 and MmTX2 from Coral Snake Venom Potently Modulate GABAA Receptor Activity. Proc. Natl. Acad. Sci. USA 2015, 112, E891–E900. [Google Scholar] [CrossRef]
- Hannan, S.; Mortensen, M.; Smart, T.G. Snake Neurotoxin α-Bungarotoxin Is an Antagonist at Native GABAA Receptors. Neuropharmacology 2015, 93, 28–40. [Google Scholar] [CrossRef]
- Kudryavtsev, D.S.; Shelukhina, I.V.; Son, L.V.; Ojomoko, L.O.; Kryukova, E.V.; Lyukmanova, E.N.; Zhmak, M.N.; Dolgikh, D.A.; Ivanov, I.A.; Kasheverov, I.E.; et al. Neurotoxins from Snake Venoms and α-Conotoxin ImI Inhibit Functionally Active Ionotropic γ-Aminobutyric Acid (GABA) Receptors. J. Biol. Chem. 2015, 290, 22747–22758. [Google Scholar] [CrossRef]
- Blanchet, G.; Upert, G.; Mourier, G.; Gilquin, B.; Gilles, N.; Servent, D. New α-Adrenergic Property for Synthetic MTβ and CM-3 Three-Finger Fold Toxins from Black Mamba. Toxicon 2013, 75, 160–167. [Google Scholar] [CrossRef]
- Maïga, A.; Mourier, G.; Quinton, L.; Rouget, C.; Gales, C.; Denis, C.; Lluel, P.; Sénard, J.-M.; Palea, S.; Servent, D.; et al. G Protein-Coupled Receptors, an Unexploited Animal Toxin Targets: Exploration of Green Mamba Venom for Novel Drug Candidates Active against Adrenoceptors. Toxicon 2012, 59, 487–496. [Google Scholar] [CrossRef]
- Rouget, C.; Quinton, L.; Maïga, A.; Gales, C.; Masuyer, G.; Malosse, C.; Chamot-Rooke, J.; Thai, R.; Mourier, G.; de Pauw, E.; et al. Identification of a Novel Snake Peptide Toxin Displaying High Affinity and Antagonist Behaviour for the A2-Adrenoceptors. Br. J. Pharmacol. 2010, 161, 1361–1374. [Google Scholar] [CrossRef]
- Rivera-Torres, I.O.; Jin, T.B.; Cadene, M.; Chait, B.T.; Poget, S.F. Discovery and Characterisation of a Novel Toxin from Dendroaspis Angusticeps, Named Tx7335, That Activates the Potassium Channel KcsA. Sci. Rep. 2016, 6, 23904. [Google Scholar] [CrossRef]
- Diochot, S.; Baron, A.; Salinas, M.; Douguet, D.; Scarzello, S.; Dabert-Gay, A.-S.; Debayle, D.; Friend, V.; Alloui, A.; Lazdunski, M.; et al. Black Mamba Venom Peptides Target Acid-Sensing Ion Channels to Abolish Pain. Nature 2012, 490, 552–555. [Google Scholar] [CrossRef]
- Soliman, C.; Eastwood, S.; Truong, V.K.; Ramsland, P.A.; Elbourne, A. The Membrane Effects of Melittin on Gastric and Colorectal Cancer. PLoS ONE 2019, 14, e0224028. [Google Scholar] [CrossRef] [PubMed]
- Borojeni, S.K.; Zolfagharian, H.; Babaie, M.; Javadi, I. Cytotoxic Effect of Bee (A. mellifera) Venom on Cancer Cell Lines. J. Pharmacopunct. 2020, 23. [Google Scholar] [CrossRef] [PubMed]
- Adade, C.M.; Chagas, G.S.F.; Souto-Padrón, T. Apis Mellifera Venom Induces Different Cell Death Pathways in Trypanosoma Cruzi. Parasitol. 2012, 139, 1444–1461. [Google Scholar] [CrossRef] [PubMed]
- Park, C.; Lee, D.G. Melittin Induces Apoptotic Features in Candida Albicans. Biochem. Biophys. Res. Commun. 2010, 394, 170–172. [Google Scholar] [CrossRef] [PubMed]
- Maher, S.; McClean, S. Melittin Exhibits Necrotic Cytotoxicity in Gastrointestinal Cells Which Is Attenuated by Cholesterol. Biochem. Pharmacol. 2008, 75, 1104–1114. [Google Scholar] [CrossRef]
- Lee, Y.J.; Kang, S.J.; Kim, B.M.; Kim, Y.J.; Woo, H.D.; Chung, H.W. Cytotoxicity of Honeybee (Apis Mellifera) Venom in Normal Human Lymphocytes and HL-60 Cells. Chem. -Biol. Interact. 2007, 169, 189–197. [Google Scholar] [CrossRef]
- White, M.M.; Aylwin, M. Niflumic and Flufenamic Acids Are Potent Reversible Blockers of Ca2(+)-Activated Cl- Channels in Xenopus Oocytes. Mol. Pharmacol. 1990, 37, 720–724. [Google Scholar]
- Hiu, J.J.; Yap, M.K.K. Cytotoxicity of Snake Venom Enzymatic Toxins: Phospholipase A2 and L-Amino Acid Oxidase. Biochem. Soc. Trans. 2020, 48, 719–731. [Google Scholar] [CrossRef]
- Oliveira, J.C.R.; Montes De Oca, H.; Duarte, M.M.; Diniz, C.R.; Fortes-Dias, C.L. Toxicity of South American Snake Venoms Measured by an in Vitro Cell Culture Assay. Toxicon 2001, 40, 321–325. [Google Scholar] [CrossRef]
- Chong, H.P.; Tan, K.Y.; Tan, C.H. Cytotoxicity of Snake Venoms and Cytotoxins From Two Southeast Asian Cobras (Naja Sumatrana, Naja Kaouthia): Exploration of Anticancer Potential, Selectivity, and Cell Death Mechanism. Front. Mol. Biosci. 2020, 7, 583587. [Google Scholar] [CrossRef]
- Li, P.-L. Cytotoxicity of Cobra (Naja Naja Kaouthia) Venom on Rabbit Red Blood Cells and S-180 Tumor Cells in the Presence of Tetracaine, Lidocaine and Procaine. Z. Nat. C Biosci. 1980, 35, 268–272. [Google Scholar] [CrossRef]
- Khalid, H.; Mukhtar, M.M.; Konstantakopoulos, N. Cytotoxiciy of Naja Nubiae (Serpentes: Elapidae) and Echis Ocellatus (Serpentes: Viperidae) Venoms from Sudan. J. Toxins 2015, 2015, 1–7. [Google Scholar] [CrossRef][Green Version]
- Thangam, R.; Gunasekaran, P.; Kaveri, K.; Sridevi, G.; Sundarraj, S.; Paulpandi, M.; Kannan, S. A Novel Disintegrin Protein from Naja Naja Venom Induces Cytotoxicity and Apoptosis in Human Cancer Cell Lines In Vitro. Process Biochem. 2012, 47, 1243–1249. [Google Scholar] [CrossRef]
- Gutiérrez, J.M.; Rojas, G.; da Silva Júnior, N.J.; Núñez, J. Experimental Myonecrosis Induced By The Venoms Of South American Micrurus (Coral Snakes). Toxicon 1992, 30, 1299–1302. [Google Scholar] [CrossRef]
- de Roodt, A.R.; Lago, N.R.; Stock, R.P. Myotoxicity and Nephrotoxicity by Micrurus Venoms in Experimental Envenomation. Toxicon 2012, 59, 356–364. [Google Scholar] [CrossRef]
- Gutiérrez, J.; Lomonte, B.; Portilla, E.; Cerdas, L.; Rojas, E. Local Effects Induced By Coral Snake Venoms: Evidence Of Myonecrosis After Experimental Inoculations Of Venoms From Five Species. Toxicon 1983, 21, 777–783. [Google Scholar] [CrossRef]
- Barros, A.C.; Fernandes, D.P.; Ferreira, L.C.; Dos Santos, M.C. local effects induced by venoms from five species of genus micrurus sp. (coral snakes). Toxicon 1994, 32, 445–452. [Google Scholar] [CrossRef]
- Casais-e-Silva, L.L.; Teixeira, C.F.P.; Lebrun, I.; Lomonte, B.; Alape-Girón, A.; Gutiérrez, J.M. Lemnitoxin, the Major Component of Micrurus Lemniscatus Coral Snake Venom, Is a Myotoxic and pro-Inflammatory Phospholipase A2. Toxicol. Lett. 2016, 257, 60–71. [Google Scholar] [CrossRef]
- Barber, C.M.; Isbister, G.K.; Hodgson, W.C. Alpha Neurotoxins. Toxicon 2013, 66, 47–58. [Google Scholar] [CrossRef]
- Kini, R.M. Excitement Ahead: Structure, Function and Mechanism of Snake Venom Phospholipase A2 Enzymes. Toxicon 2003, 42, 827–840. [Google Scholar] [CrossRef]
- Mackessy, S.P. Handbook of Venoms and Toxins of Reptiles; CRC Press: Boca Raton, FL, USA, 2016. [Google Scholar]
- Carbajal-Saucedo, A.; López-Vera, E.; Bénard-Valle, M.; Smith, E.N.; Zamudio, F.; de Roodt, A.R.; Olvera-Rodríguez, A. Isolation, Characterization, Cloning and Expression of an Alpha-Neurotoxin from the Venom of the Mexican Coral Snake Micrurus Laticollaris (Squamata: Elapidae). Toxicon 2013, 66, 64–74. [Google Scholar] [CrossRef] [PubMed]
- Son, L.; Kryukova, E.; Ziganshin, R.; Andreeva, T.; Kudryavtsev, D.; Kasheverov, I.; Tsetlin, V.; Utkin, Y. Novel Three-Finger Neurotoxins from Naja Melanoleuca Cobra Venom Interact with GABAA and Nicotinic Acetylcholine Receptors. Toxins 2021, 13, 164. [Google Scholar] [CrossRef] [PubMed]
- Mccann, C.M.; Bracamontes, J.; Steinbach, J.H.; Sanes, J.R. The Cholinergic Antagonist-Bungarotoxin Also Binds and Blocks a Subset of GABA Receptors. Proc. Natl. Acad. Sci. USA 2006, 103, 5149–5154. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.; Ma, H.; Zhang, F.; Zhang, C.; Zou, X.; Cao, Z. Selective Voltage-Gated Sodium Channel Peptide Toxins from Animal Venom: Pharmacological Probes and Analgesic Drug Development. ACS Chem. Neurosci. 2018, 9, 187–197. [Google Scholar] [CrossRef]
- Kalia, J.; Milescu, M.; Salvatierra, J.; Wagner, J.; Klint, J.K.; King, G.F.; Olivera, B.M.; Bosmans, F. From Foe to Friend: Using Animal Toxins to Investigate Ion Channel Function. J. Mol. Biol. 2015, 427, 158–175. [Google Scholar] [CrossRef]
- Norton, R.S.; Chandy, K.G. Venom-Derived Peptide Inhibitors of Voltage-Gated Potassium Channels. Neuropharmacology 2017, 127, 124–138. [Google Scholar] [CrossRef]
- Bordon, K.d.C.F.; Cologna, C.T.; Fornari-Baldo, E.C.; Pinheiro-Júnior, E.L.; Cerni, F.A.; Amorim, F.G.; Anjolette, F.A.P.; Cordeiro, F.A.; Wiezel, G.A.; Cardoso, I.A.; et al. From Animal Poisons and Venoms to Medicines: Achievements, Challenges and Perspectives in Drug Discovery. Front. Pharmacol. 2020, 11, 1132. [Google Scholar] [CrossRef]
- Lai, H.C.; Jan, L.Y. The Distribution and Targeting of Neuronal Voltage-Gated Ion Channels. Nat. Rev. Neurosci. 2006, 7, 548–562. [Google Scholar] [CrossRef]
- Terlau, H.; Stühmer, W. The S Tructure And Function Of Voltage-Gated Na + And K + Channels. Naturwissenschaften 1998, 85, 437–444. [Google Scholar] [CrossRef]
- Yellen, G. The Voltage-Gated Potassium Channels and Their Relatives. Nature 2002, 419, 35–42. [Google Scholar] [CrossRef]
- Kwong, K.; Carr, M.J. Voltage-Gated Sodium Channels. Curr. Opin. Pharmacol. 2015, 22, 131–139. [Google Scholar] [CrossRef] [PubMed]
- Pardo, L.A.; Stühmer, W. The Roles of K + Channels in Cancer. Nat. Rev. Cancer 2014, 14, 39–48. [Google Scholar] [CrossRef] [PubMed]
- Waters, M.F.; Minassian, N.A.; Stevanin, G.; Figueroa, K.P.; Bannister, J.P.A.; Nolte, D.; Mock, A.F.; Evidente, V.G.H.; Fee, D.B.; Müller, U.; et al. Mutations in Voltage-Gated Potassium Channel KCNC3 Cause Degenerative and Developmental Central Nervous System Phenotypes. Nat. Genet. 2006, 38, 447–451. [Google Scholar] [CrossRef]
- Mao, W.; Zhang, J.; Körner, H.; Jiang, Y.; Ying, S. The Emerging Role of Voltage-Gated Sodium Channels in Tumor Biology. Front. Oncol. 2019, 9, 124. [Google Scholar] [CrossRef]
- Eijkelkamp, N.; Linley, J.E.; Baker, M.D.; Minett, M.S.; Cregg, R.; Werdehausen, R.; Rugiero, F.; Wood, J.N. Neurological Perspectives on Voltage-Gated Sodium Channels. Brain 2012, 135, 2585–2612. [Google Scholar] [CrossRef]
- Lawson, K.; Mckay, N.G. Modulation of Potassium Channels as a Therapeutic Approach. Curr. Pharm. Des. 2006, 12, 459–470. [Google Scholar] [CrossRef]
- Wickenden, A.D. K + Channels as Therapeutic Drug Targets. Pharmacol. Ther. 2002, 94, 157–182. [Google Scholar] [CrossRef]
- Wulff, H.; Castle, N.A.; Pardo, L.A. Voltage-Gated Potassium Channels as Therapeutic Targets. Nat. Rev. Drug Discov. 2009, 8, 982–1001. [Google Scholar] [CrossRef]
- Mantegazza, M.; Curia, G.; Biagini, G.; Ragsdale, D.S.; Avoli, M. Voltage-Gated Sodium Channels as Therapeutic Targets in Epilepsy and Other Neurological Disorders. Lancet Neurol. 2010, 9, 413–424. [Google Scholar] [CrossRef]
- Tajti, G.; Wai, D.C.C.; Panyi, G.; Norton, R.S. The Voltage-Gated Potassium Channel KV1.3 as a Therapeutic Target for Venom-Derived Peptides. Biochem. Pharmacol. 2020, 181, 114146. [Google Scholar] [CrossRef]
- Jouirou, B.; Mouhat, S.; Andreotti, N.; de Waard, M.; Sabatier, J.M. Toxin Determinants Required for Interaction with Voltage-Gated K + Channels. Toxicon 2004, 43, 909–914. [Google Scholar] [CrossRef] [PubMed]
- Mouhat, S.; Andreotti, N.; Jouirou, B.; Sabatier, J.-M. Animal Toxins Acting on Voltage-Gated Potassium Channels. Curr. Pharm. Des. 2008, 14, 2503–2518. [Google Scholar] [CrossRef] [PubMed]
- Mouhat, S.; Jouirou, B.; Mosbah, A.; de Waard, M.; Sabatier, J.-M. Diversity of Folds in Animal Toxins Acting on Ion Channels. Biochem. J. 2004, 378, 716–726. [Google Scholar] [CrossRef]
- Boldrini-França, J.; Pinheiro-Junior, E.L.; Peigneur, S.; Pucca, M.B.; Cerni, F.A.; Borges, R.J.; Costa, T.R.; Carone, S.E.I.; Fontes, M.R.d.M.; Sampaio, S.V.; et al. Beyond Hemostasis: A Snake Venom Serine Protease with Potassium Channel Blocking and Potential Antitumor Activities. Sci. Rep. 2020, 10, 4476. [Google Scholar] [CrossRef]
- An, D.; Pinheiro-Junior, E.L.; Béress, L.; Gladkikh, I.; Leychenko, E.; Undheim, E.A.B.; Peigneur, S.; Tytgat, J. AsKC11, a Kunitz Peptide from Anemonia Sulcata, Is a Novel Activator of G Protein-Coupled Inward-Rectifier Potassium Channels. Mar. Drugs 2022, 20, 140. [Google Scholar] [CrossRef] [PubMed]
- Gladkikh, I.; Peigneur, S.; Sintsova, O.; Lopes Pinheiro-Junior, E.; Klimovich, A.; Menshov, A.; Kalinovsky, A.; Isaeva, M.; Monastyrnaya, M.; Kozlovskaya, E.; et al. Kunitz-Type Peptides from the Sea Anemone Heteractis Crispa Demonstrate Potassium Channel Blocking and Anti-Inflammatory Activities. Biomedicines 2020, 8, 473. [Google Scholar] [CrossRef] [PubMed]
- Pinheiro-Junior, E.L.; Kalina, R.; Gladkikh, I.; Leychenko, E.; Tytgat, J.; Peigneur, S. A Tale of Toxin Promiscuity: The Versatile Pharmacological Effects of Hcr 1b-2 Sea Anemone Peptide on Voltage-Gated Ion Channels. Mar. Drugs 2022, 20, 147. [Google Scholar] [CrossRef]
- Teisseyre, A.; Palko-Labuz, A.; Sroda-Pomianek, K.; Michalak, K. Voltage-Gated Potassium Channel Kv1.3 as a Target in Therapy of Cancer. Front. Oncol. 2019, 9, 933. [Google Scholar] [CrossRef]
- Schmalhofer, W.A.; Bao, J.; McManus, O.B.; Green, B.; Matyskiela, M.; Wunderler, D.; Bugianesi, R.M.; Felix, J.P.; Hanner, M.; Linde-Arias, A.R.; et al. Identification of a New Class of Inhibitors of the Voltage-Gated Potassium Channel, Kv1.3, with Immunosuppressant Properties. Biochemistry 2002, 41, 7781–7794. [Google Scholar] [CrossRef]
- Pérez-Verdaguer, M.; Capera, J.; Serrano-Novillo, C.; Estadella, I.; Sastre, D.; Felipe, A. The Voltage-Gated Potassium Channel Kv1.3 Is a Promising Multitherapeutic Target against Human Pathologies. Expert Opin. Ther. Targets 2016, 20, 577–591. [Google Scholar] [CrossRef]
- Beeton, C.; Wulff, H.; Standifer, N.E.; Azam, P.; Mullen, K.M.; Pennington, M.W.; Kolski-Andreaco, A.; Wei, E.; Grino, A.; Counts, D.R.; et al. Kv1.3 Channels Are a Therapeutic Target for T Cell-Mediated Autoimmune Diseases. Proc. Natl. Acad. Sci. USA 2006, 103, 17414–17419. [Google Scholar] [CrossRef] [PubMed]
- Rangaraju, S.; Chi, V.; Pennington, M.W.; Chandy, K.G. Kv1.3 Potassium Channels as a Therapeutic Target in Multiple Sclerosis. Expert Opin. Ther. Targets 2009, 13, 909–924. [Google Scholar] [CrossRef]
- Gubič, Š.; Hendrickx, L.A.; Toplak, Ž.; Sterle, M.; Peigneur, S.; Tomašič, T.; Pardo, L.A.; Tytgat, J.; Zega, A.; Mašič, L.P. Discovery of KV1.3 Ion Channel Inhibitors: Medicinal Chemistry Approaches and Challenges. Med. Res. Rev. 2021, 41, 2423–2473. [Google Scholar] [CrossRef]
- Chandy, K.G.; Norton, R.S. Peptide Blockers of Kv1.3 Channels in T Cells as Therapeutics for Autoimmune Disease. Curr. Opin. Chem. Biol. 2017, 38, 97–107. [Google Scholar] [CrossRef]
- Kuzmenkov, A.I.; Grishin, E.V.; Vassilevski, A.A. Diversity of Potassium Channel Ligands: Focus on Scorpion Toxins. Biochemistry 2015, 80, 1764–1799. [Google Scholar] [CrossRef]
- Quintero-Hernández, V.; Jiménez-Vargas, J.M.; Gurrola, G.B.; Valdivia, H.H.; Possani, L.D. Scorpion Venom Components That Affect Ion-Channels Function. Toxicon 2013, 76, 328–342. [Google Scholar] [CrossRef] [PubMed]
- Kuzmenkov, A.I.; Krylov, N.A.; Chugunov, A.O.; Grishin, E.V.; Vassilevski, A.A. Kalium: A Database of Potassium Channel Toxins from Scorpion Venom. Database 2016, 2016, baw056. [Google Scholar] [CrossRef] [PubMed]
- Bergeron, Z.L.; Bingham, J.P. Scorpion Toxins Specific for Potassium (K+) Channels: A Historical Overview of Peptide Bioengineering. Toxins 2012, 4, 1082–1119. [Google Scholar] [CrossRef]
- Schütter, N.; Barreto, Y.C.; Vardanyan, V.; Hornig, S.; Hyslop, S.; Marangoni, S.; Rodrigues-Simioni, L.; Pongs, O.; Dal Belo, C.A. Inhibition of Kv2.1 Potassium Channels by MiDCA1, a Pre-Synaptically Active PLA2-Type Toxin from Micrurus Dumerilii Carinicauda Coral Snake Venom. Toxins 2019, 11, 335. [Google Scholar] [CrossRef]
- Yang, D.C.; Deuis, J.R.; Dashevsky, D.; Dobson, J.; Jackson, T.N.W.; Brust, A.; Xie, B.; Koludarov, I.; Debono, J.; Hendrikx, I.; et al. The Snake with the Scorpion’s Sting: Novel Three-Finger Toxin Sodium Channel Activators from the Venom of the Long-Glanded Blue Coral Snake (Calliophis Bivirgatus). Toxins 2016, 8, 303. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kleiz-Ferreira, J.M.; Bernaerts, H.; Pinheiro-Junior, E.L.; Peigneur, S.; Zingali, R.B.; Tytgat, J. Pharmacological Screening of Venoms from Five Brazilian Micrurus Species on Different Ion Channels. Int. J. Mol. Sci. 2022, 23, 7714. https://doi.org/10.3390/ijms23147714
Kleiz-Ferreira JM, Bernaerts H, Pinheiro-Junior EL, Peigneur S, Zingali RB, Tytgat J. Pharmacological Screening of Venoms from Five Brazilian Micrurus Species on Different Ion Channels. International Journal of Molecular Sciences. 2022; 23(14):7714. https://doi.org/10.3390/ijms23147714
Chicago/Turabian StyleKleiz-Ferreira, Jessica Matos, Hans Bernaerts, Ernesto Lopes Pinheiro-Junior, Steve Peigneur, Russolina Benedeta Zingali, and Jan Tytgat. 2022. "Pharmacological Screening of Venoms from Five Brazilian Micrurus Species on Different Ion Channels" International Journal of Molecular Sciences 23, no. 14: 7714. https://doi.org/10.3390/ijms23147714
APA StyleKleiz-Ferreira, J. M., Bernaerts, H., Pinheiro-Junior, E. L., Peigneur, S., Zingali, R. B., & Tytgat, J. (2022). Pharmacological Screening of Venoms from Five Brazilian Micrurus Species on Different Ion Channels. International Journal of Molecular Sciences, 23(14), 7714. https://doi.org/10.3390/ijms23147714







