The Less We Eat, the Longer We Live: Can Caloric Restriction Help Us Become Centenarians?
Abstract
1. Introduction
1.1. Populations of Centenarians
1.1.1. The Okinawan Population
1.1.2. The Sardinian Population
1.1.3. The Ikarian Population
1.2. Human Studies
CALERIETM Study
1.3. CR Impact on Longevity in Long-Living Primates
2. Physiological Processes Underlying Positive Effects of CR on Health and Lifespan
2.1. CR, Insulin Sensitivity, and Longevity
2.2. CR, Endocrine Factors, and Longevity
2.3. CR, Immune Function, and Longevity
2.4. CR, Gut Microbiota, and Longevity
3. Molecular Mechanisms Underlying CR Effects on Health and Lifespan
3.1. Insulin/IGF1 Signaling
3.2. AMPK
3.3. mTOR
3.4. Sirtuins
3.5. Cross-Regulation of Molecular Mechanisms
3.6. New Determinants (Multi-Omics Approach)
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ADP | adenosine diphosphate |
| Akt1 | protein kinase B |
| AMP | adenosine monophosphate |
| AMPK | AMP-activated protein kinase |
| ATP | adenosine triphosphate |
| CALERIE | Comprehensive Assessment of Long-term Effects of Reducing Intake of Energy |
| COX-2 | cyclooxygenase 2 |
| CR | caloric restriction |
| CRP | C-reactive protein |
| CXCL16 | chemokine ligand 16 |
| DHEA | dehydroepiandrosterone |
| DHEAS | dehydroepiandrosterone sulfate |
| FMT | fecal microbiota transfer |
| FOXO | forkhead box protein O |
| GABA | γ-aminobutyric acid |
| GH | growth hormone |
| GLUT | glucose transporter |
| HDL | high density lipoprotein |
| IGF1 | insulin-like growth factor 1 |
| IL | interleukin |
| INFγ | interferon gamma |
| iNOS | inducible nitric oxide synthase |
| Irs, IRS | insulin receptor substrate |
| LBP | lipopolysaccharide—binding protein |
| LBZ | Longevity Blue Zones |
| LDL | low dencity lipoprotein |
| LKB1 | liver kinase B1 |
| MCP-1 | monocyte chemoattractant protein 1 |
| MedDiet | Mediterranean diet |
| MIP-1α | macrophage inflammatory protein 1α |
| mTOR | mammalian target of rapamycin kinase |
| mTORC | mTOR complex |
| NAD+ | nicotinamide adenine dinucleotide |
| NF-κB | nuclear factor-κB |
| NIA | National Institute on Aging |
| PGC-1a | peroxisome proliferator-activated receptor gamma coactivator 1-alpha |
| PI3K | phosphoinositide 3-kinase |
| PPAR | peroxisome proliferator-activated receptor |
| RAGs | recombination-activating genes |
| RANTES | regulated on activation, normal T cell expressed and secreted |
| S6K1 | S6 kinase 1 |
| Sirt, SIRT | sirtuins |
| SOD2 | superoxide dismutase 2 |
| SREPB1c | sterol regulatory element-binding protein 1c |
| T3 | triiodothyronine |
| T4 | thyroxine |
| TNF | tumor necrosis factor |
| TNO | Toxicology and Nutrition Institute in the Netherlands |
| TSC2 | tuberous sclerosis complex 2 |
| TSH | thyroid stimulating hormone |
| UCP2 | mitochondrial uncoupling protein 2 |
| WHO | World Health Organization |
References
- Harrison, J.; Ticknor, C. The Philosophy of Health; or, an Exposition of the Physical and Mental Constitution of Man, with a View to the Promotion of Human Longevity and Happiness. Br. Foreign Med. Rev. 1838, 5, 380–413. [Google Scholar]
- Longo, V.D.; Anderson, R.M. Nutrition, Longevity and Disease: From Molecular Mechanisms to Interventions. Cell 2022, 185, 1455–1470. [Google Scholar] [CrossRef] [PubMed]
- Trepanowski, J.F.; Bloomer, R.J. The impact of religious fasting on human health. Nutr. J. 2010, 9, 57. [Google Scholar] [CrossRef] [PubMed]
- Persynaki, A.; Karras, S.; Pichard, C. Unraveling the metabolic health benefits of fasting related to religious beliefs: A narrative review. Nutrition 2017, 35, 14–20. [Google Scholar] [CrossRef] [PubMed]
- Teixeira, L.; Araújo, L.; Jopp, D.; Ribeiro, O. Centenarians in Europe. Maturitas 2017, 104, 90–95. [Google Scholar] [CrossRef] [PubMed]
- Heshmati, H. The Centenarians: An Emerging Population. In Update in Geriatrics; Amornyotin, S., Ed.; IntechOpen: London, UK, 2021; ISBN 978-1-83962-308-0. [Google Scholar]
- Sebastiani, P.; Federico, A.; Morris, M.; Gurinovich, A.; Tanaka, T.; Chandler, K.B.; Andersen, S.L.; Denis, G.; Costello, C.E.; Ferrucci, L.; et al. Protein signatures of centenarians and their offspring suggest centenarians age slower than other humans. Aging Cell 2021, 20, e13290. [Google Scholar] [CrossRef]
- Borras, C.; Ingles, M.; Mas-Bargues, C.; Dromant, M.; Sanz-Ros, J.; Román-Domínguez, A.; Gimeno-Mallench, L.; Gambini, J.; Viña, J. Centenarians: An excellent example of resilience for successful ageing. Mech. Ageing Dev. 2019, 186, 111199. [Google Scholar] [CrossRef]
- Andersen, S.L. Centenarians as Models of Resistance and Resilience to Alzheimer’s Disease and Related Dementias. Adv. Geriatr. Med. Res. 2020, 2, e200018. [Google Scholar] [CrossRef]
- Robert, L.; Fulop, T. Longevity and Its Regulation: Centenarians and Beyond. In Interdisciplinary Topics in Gerontology; Robert, L., Fulop, T., Eds.; S. KARGER AG: Basel, Switzerland, 2014; Volume 39, pp. 198–211. ISBN 978-3-318-02652-8. [Google Scholar]
- Frame, L.T.; Hart, R.W.; Leakey, J.E. Caloric restriction as a mechanism mediating resistance to environmental disease. Environ. Health Perspect. 1998, 106, 313–324. [Google Scholar] [CrossRef]
- Bucci, L.; Ostan, R.; Cevenini, E.; Pini, E.; Scurti, M.; Vitale, G.; Mari, D.; Caruso, C.; Sansoni, P.; Fanelli, F.; et al. Centenarians’ offspring as a model of healthy aging: A reappraisal of the data on Italian subjects and a comprehensive overview. Aging 2016, 8, 510–519. [Google Scholar] [CrossRef]
- Garagnani, P.; Marquis, J.; Delledonne, M.; Pirazzini, C.; Marasco, E.; Kwiatkowska, K.M.; Iannuzzi, V.; Bacalini, M.G.; Valsesia, A.; Carayol, J.; et al. Whole-genome sequencing analysis of semi-supercentenarians. eLife 2021, 10, e57849. [Google Scholar] [CrossRef] [PubMed]
- Capri, M.; Santoro, A.; Garagnani, P.; Bacalini, M.G.; Pirazzini, C.; Olivieri, F.; Procopio, A.; Salvioli, S.; Franceschi, C. Genes of human longevity: An endless quest? Curr. Vasc. Pharmacol. 2013, 12, 707–717. [Google Scholar] [CrossRef] [PubMed]
- Salvioli, S.; Olivieri, F.; Marchegiani, F.; Cardelli, M.; Santoro, A.; Bellavista, E.; Mishto, M.; Invidia, L.; Capri, M.; Valensin, S.; et al. Genes, ageing and longevity in humans: Problems, advantages and perspectives. Free Radic. Res. 2006, 40, 1303–1323. [Google Scholar] [CrossRef]
- Balistreri, C.; Candore, G.; Accardi, G.; Buffa, S.; Bulati, M.; Martorana, A.; Colonna-Romano, G.; Lio, D.; Caruso, C. Centenarian offspring: A model for understanding longevity. Curr. Vasc. Pharmacol. 2013, 12, 718–725. [Google Scholar] [CrossRef] [PubMed]
- Robine, J.-M.; Cubaynes, S. Worldwide demography of centenarians. Mech. Ageing Dev. 2017, 165, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Poulain, M.; Herm, A.; Pes, G.M. The Blue Zones: Areas of exceptional longevity around the world. Vienna Yearb. Popul. Res. 2014, 11, 87–108. [Google Scholar] [CrossRef]
- Psaltopoulou, T.; Orfanos, P.; Naska, A.; Lenas, D.; Trichopoulos, D.; Trichopoulou, A. Prevalence, awareness, treatment and control of hypertension in a general population sample of 26 913 adults in the Greek EPIC study. Int. J. Epidemiol. 2004, 33, 1345–1352. [Google Scholar] [CrossRef]
- Bendjilali, N.; Hsueh, W.-C.; He, Q.; Willcox, D.C.; Nievergelt, C.M.; Donlon, T.A.; Kwok, P.-Y.; Suzuki, M.; Willcox, B.J. Who Are the Okinawans? Ancestry, Genome Diversity, and Implications for the Genetic Study of Human Longevity from a Geographically Isolated Population. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2014, 69, 1474–1484. [Google Scholar] [CrossRef]
- Caselli, G.; Pozzi, L.; Vaupel, J.; Deiana, L.; Pes, G.M.; Carru, C.; Franceschi, C.; Baggio, G. Family clustering in Sardinian longevity: A genealogical approach. Exp. Gerontol. 2006, 41, 727–736. [Google Scholar] [CrossRef]
- Chan, Y.C.; Suzuki, M.; Yamamoto, S. Dietary, anthropometric, hematological and biochemical assessment of the nutritional status of centenarians and elderly people in Okinawa, Japan. J. Am. Coll. Nutr. 1997, 16, 229–235. [Google Scholar] [CrossRef]
- Fastame, M.C. Well-being, food habits, and lifestyle for longevity. Preliminary evidence from the sardinian centenarians and long-lived people of the Blue Zone. Psychol. Health Med. 2022, 27, 728–733. [Google Scholar] [CrossRef] [PubMed]
- Hokama, T.; Arakaki, H.; Sho, H.; Inafuku, M. Nutrition Survey of School Children in Okinawa. Sci. B Coll. Agr. Univ. Ryukyus. 1967, 14, 1–15. [Google Scholar]
- Kagawa, Y. Impact of westernization on the nutrition of Japanese: Changes in physique, cancer, longevity and centenarians. Prev. Med. 1978, 7, 205–217. [Google Scholar] [CrossRef]
- Legrand, R.; Manckoundia, P.; Nuemi, G.; Poulain, M. Assessment of the Health Status of the Oldest Olds Living on the Greek Island of Ikaria: A Population Based-Study in a Blue Zone. Curr. Gerontol. Geriatr. Res. 2019, 2019, 8194310. [Google Scholar] [CrossRef] [PubMed]
- Legrand, R.; Nuemi, G.; Poulain, M.; Manckoundia, P. Description of Lifestyle, Including Social Life, Diet and Physical Activity, of People ≥90 years Living in Ikaria, a Longevity Blue Zone. Int. J. Environ. Res. Public Health 2021, 18, 6602. [Google Scholar] [CrossRef] [PubMed]
- Montesanto, A.; De Rango, F.; Pirazzini, C.; Guidarelli, G.; Domma, F.; Franceschi, C.; Passarino, G. Demographic, genetic and phenotypic characteristics of centenarians in Italy: Focus on gender differences. Mech. Ageing Dev. 2017, 165, 68–74. [Google Scholar] [CrossRef]
- Nieddu, A.; Vindas, L.; Errigo, A.; Vindas, J.; Pes, G.M.; Dore, M.P. Dietary Habits, Anthropometric Features and Daily Performance in Two Independent Long-Lived Populations from Nicoya peninsula (Costa Rica) and Ogliastra (Sardinia). Nutrients 2020, 12, 1621. [Google Scholar] [CrossRef]
- Okamoto, K.; Sasaki, R. Geographical Epidemiologic Studies on Factors Associated with Centenarians in Japan. Nippon. Ronen Igakkai Zasshi 1995, 32, 485–490. [Google Scholar] [CrossRef][Green Version]
- Panagiotakos, D.B.; Chrysohoou, C.; Siasos, G.; Zisimos, K.; Skoumas, J.; Pitsavos, C.; Stefanadis, C. Sociodemographic and Lifestyle Statistics of Oldest Old People (>80 Years) Living in Ikaria Island: The Ikaria Study. Cardiol. Res. Pract. 2011, 2011, 679187. [Google Scholar] [CrossRef]
- Passarino, G.; Underhill, P.A.; Cavalli-Sforza, L.L.; Semino, O.; Pes, G.M.; Carru, C.; Ferrucci, L.; Bonafè, M.; Franceschi, C.; Deiana, L.; et al. Y chromosome binary markers to study the high prevalence of males in Sardinian centenarians and the genetic structure of the Sardinian population. Hum. Hered. 2001, 52, 136–139. [Google Scholar] [CrossRef]
- Pes, G.; Poulain, M.; Errigo, A.; Dore, M. Evolution of the Dietary Patterns across Nutrition Transition in the Sardinian Longevity Blue Zone and Association with Health Indicators in the Oldest Old. Nutrients 2021, 13, 1495. [Google Scholar] [CrossRef] [PubMed]
- Poulain, M.; Pes, G.M.; Grasland, C.; Carru, C.; Ferrucci, L.; Baggio, G.; Franceschi, C.; Deiana, L. Identification of a geographic area characterized by extreme longevity in the Sardinia island: The AKEA study. Exp. Gerontol. 2004, 39, 1423–1429. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Curb, J.D.; Suzuki, M. Caloric restriction and human longevity: What can we learn from the Okinawans? Biogerontology 2006, 7, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Willcox, B.J.; Willcox, D.C.; Todoriki, H.; Fujiyoshi, A.; Yano, K.; He, Q.; Curb, J.D.; Suzuki, M. Caloric Restriction, the Traditional Okinawan Diet, and Healthy Aging: The Diet of the World’s Longest-Lived People and Its Potential Impact on Morbidity and Life Span. Ann. N. Y. Acad. Sci. 2007, 1114, 434–455. [Google Scholar] [CrossRef] [PubMed]
- Willcox, B.J.; Willcox, D.C.; Todoriki, H.; Yano, K.; Curb, J.D.; Suzuki, M. Caloric Restriction, Energy Balance and Healthy Aging in Okinawans and Americans: Biomarker Differences in Septuagenarians. Okinawan J. Am. Stud. 2007, 4, 60–72. [Google Scholar]
- Willcox, D.C.; Willcox, B.J.; He, Q.; Wang, N.-C.; Suzuki, M. They Really Are That Old: A Validation Study of Centenarian Prevalence in Okinawa. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2008, 63, 338–349. [Google Scholar] [CrossRef]
- Willcox, B.J.; Willcox, D.C.; Suzuki, M. Demographic, phenotypic, and genetic characteristics of centenarians in Okinawa and Japan: Part 1—Centenarians in Okinawa. Mech. Ageing Dev. 2017, 165, 75–79. [Google Scholar] [CrossRef]
- Trepanowski, J.F.; Canale, R.E.; Marshall, K.E.; Kabir, M.M.; Bloomer, R.J. Impact of caloric and dietary restriction regimens on markers of health and longevity in humans and animals: A summary of available findings. Nutr. J. 2011, 10, 107. [Google Scholar] [CrossRef]
- Das, S.K.; Balasubramanian, P.; Weerasekara, Y.K. Nutrition modulation of human aging: The calorie restriction paradigm. Mol. Cell. Endocrinol. 2017, 455, 148–157. [Google Scholar] [CrossRef]
- Wierik, E.J.V.-T.; Berg, H.V.D.; Schaafsma, G.; Hendriks, H.F.; Brouwer, A. Energy restriction, a useful intervention to retard human ageing? Results of a feasibility study. Eur. J. Clin. Nutr. 1994, 48, 138–148. [Google Scholar]
- Roth, G.S.; Ingram, D.K.; Lane, M.A. Calorie restriction in primates: Will it work and how will we know? J. Am. Geriatr. Soc. 1999, 47, 896–903. [Google Scholar] [CrossRef] [PubMed]
- Hamilton-Williams, E.E.; Serreze, D.V.; Charlton, B.; Johnson, E.A.; Marron, M.P.; Müllbacher, A.; Slattery, R.M. Transgenic rescue implicates β2 -microglobulin as a diabetes susceptibility gene in nonobese diabetic (NOD) mice. Proc. Natl. Acad. Sci. USA 2001, 98, 11533–11538. [Google Scholar] [CrossRef] [PubMed]
- Walford, R.L.; Harris, S.B.; Gunion, M.W. The calorically restricted low-fat nutrient-dense diet in Biosphere 2 significantly lowers blood glucose, total leukocyte count, cholesterol, and blood pressure in humans. Proc. Natl. Acad. Sci. USA 1992, 89, 11533–11537. [Google Scholar] [CrossRef] [PubMed]
- Verdery, R.B.; Walford, R.L. Changes in plasma lipids and lipoproteins in humans during a 2-year period of dietary restriction in Biosphere 2. Arch. Intern. Med. 1998, 158, 900–906. [Google Scholar] [CrossRef] [PubMed]
- Rickman, A.D.; Williamson, D.A.; Martin, C.K.; Gilhooly, C.H.; Stein, R.; Bales, C.W.; Roberts, S.; Das, S.K. The CALERIE Study: Design and methods of an innovative 25% caloric restriction intervention. Contemp. Clin. Trials 2011, 32, 874–881. [Google Scholar] [CrossRef]
- Heilbronn, L.; de Jonge, L.; Frisard, M.I.; DeLany, J.; Larson-Meyer, D.E.; Rood, J.; Nguyen, T.; Martin, C.K.; Volaufova, J.; Most, M.M.; et al. Effect of 6-Month Calorie Restriction on Biomarkers of Longevity, Metabolic Adaptation, and Oxidative Stress in Overweight Individuals: A Randomized Controlled Trial. JAMA 2006, 295, 1539–1548. [Google Scholar] [CrossRef]
- Racette, S.B.; Weiss, E.P.; Villareal, D.T.; Arif, H.; Steger-May, K.; Schechtman, K.B.; Fontana, L.; Klein, S.; Holloszy, J.O.; The Washington University School of Medicine CALERIE Group. One Year of Caloric Restriction in Humans: Feasibility and Effects on Body Composition and Abdominal Adipose Tissue. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2006, 61, 943–950. [Google Scholar] [CrossRef]
- Washington University School of Medicine CALERIE Group; Weiss, E.P.; Racette, S.; Villareal, D.T.; Fontana, L.; Steger-May, K.; Schechtman, K.B.; Klein, S.; Holloszy, J.O. Improvements in glucose tolerance and insulin action induced by increasing energy expenditure or decreasing energy intake: A randomized controlled trial. Am. J. Clin. Nutr. 2006, 84, 1033–1042. [Google Scholar] [CrossRef]
- Dorling, J.L.; Van Vliet, S.; Huffman, K.M.; Kraus, W.E.; Bhapkar, M.; Pieper, C.F.; Stewart, T.; Das, S.K.; Racette, S.B.; Roberts, S.B.; et al. Effects of caloric restriction on human physiological, psychological, and behavioral outcomes: Highlights from CALERIE phase 2. Nutr. Rev. 2020, 79, 98–113. [Google Scholar] [CrossRef]
- Kraus, W.E.; Bhapkar, M.; Huffman, K.M.; Pieper, C.F.; Das, S.K.; Redman, L.M.; Villareal, D.T.; Rochon, J.; Roberts, S.B.; Ravussin, E.; et al. 2 years of calorie restriction and cardiometabolic risk (CALERIE): Exploratory outcomes of a multicentre, phase 2, randomised controlled trial. Lancet Diabetes Endocrinol. 2019, 7, 673–683. [Google Scholar] [CrossRef]
- Grigolon, R.B.; Brietzke, E.; Trevizol, A.P.; McIntyre, R.S.; Mansur, R.B. Caloric restriction, resting metabolic rate and cognitive performance in Non-obese adults: A post-hoc analysis from CALERIE study. J. Psychiatry Res. 2020, 128, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Shen, W.; Chen, J.; Zhou, J.; Martin, C.K.; Ravussin, E.; Redman, L.M. Effect of 2-year caloric restriction on organ and tissue size in nonobese 21- to 50-year-old adults in a randomized clinical trial: The CALERIE study. Am. J. Clin. Nutr. 2021, 114, 1295–1303. [Google Scholar] [CrossRef] [PubMed]
- Roth, G.S.; Lane, M.A.; Ingram, D.K.; Mattison, J.A.; Elahi, D.; Tobin, J.D.; Muller, D.; Metter, E.J. Biomarkers of Caloric Restriction May Predict Longevity in Humans. Science 2002, 297, 811. [Google Scholar] [CrossRef] [PubMed]
- Soare, A.; Cangemi, R.; Omodei, D.; Holloszy, J.O.; Fontana, L. Long-term calorie restriction, but not endurance exercise, lowers core body temperature in humans. Aging 2011, 3, 374–379. [Google Scholar] [CrossRef] [PubMed]
- Zainal, T.A.; Oberley, T.D.; Allison, D.B.; Szweda, L.I.; Weindruch, R. Caloric restriction of rhesus monkeys lowers oxidative damage in skeletal muscle. FASEB J. 2000, 14, 1825–1836. [Google Scholar] [CrossRef]
- Dandona, P.; Mohanty, P.; Ghanim, H.; Aljada, A.; Browne, R.; Hamouda, W.; Prabhala, A.; Afzal, A.; Garg, R. The Suppressive Effect of Dietary Restriction and Weight Loss in the Obese on the Generation of Reactive Oxygen Species by Leukocytes, Lipid Peroxidation, and Protein Carbonylation1. J. Clin. Endocrinol. Metab. 2001, 86, 355–362. [Google Scholar] [CrossRef]
- Ravussin, E.; Redman, L.M.; Rochon, J.; Das, S.K.; Fontana, L.; Kraus, W.E.; Romashkan, S.; Williamson, D.A.; Meydani, S.N.; Villareal, D.T.; et al. A 2-Year Randomized Controlled Trial of Human Caloric Restriction: Feasibility and Effects on Predictors of Health Span and Longevity. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2015, 70, 1097–1104. [Google Scholar] [CrossRef]
- Weindruch, R. Will dietary restriction work in primates? Biogerontology 2006, 7, 169–171. [Google Scholar] [CrossRef]
- Ingram, D.K.; Cutler, R.G.; Weindruch, R.; Renquist, D.M.; Knapka, J.J.; April, M.; Belcher, C.T.; Clark, M.A.; Hatcherson, C.D.; Marriott, B.M.; et al. Dietary Restriction and Aging: The Initiation of a Primate Study. J. Gerontol. 1990, 45, B148–B163. [Google Scholar] [CrossRef]
- Kemnitz, J.W.; Weindruch, R.; Roecker, E.B.; Crawford, K.; Kaufman, P.L.; Ershler, W.B. Dietary Restriction of Adult Male Rhesus Monkeys: Design, Methodology, and Preliminary Findings from the First Year of Study. J. Gerontol. 1993, 48, B17–B26. [Google Scholar] [CrossRef]
- Cefalu, W.T.; Wagner, J.D.; Wang, Z.Q.; Bell-Farrow, A.D.; Collins, J.; Haskell, D.; Bechtold, R.; Morgan, T. A Study of Caloric Restriction and Cardiovascular Aging in Cynomolgus Monkeys (Macacafascicularis): A Potential Model for Aging Research. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 1997, 52, B10–B19. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.A.; Ingram, D.K.; Roth, G.S. Beyond the rodent model: Calorie restriction in rhesus monkeys. AGE 1997, 20, 45–56. [Google Scholar] [CrossRef] [PubMed]
- Colman, R.J.; Anderson, R.M.; Johnson, S.C.; Kastman, E.K.; Kosmatka, K.J.; Beasley, T.M.; Allison, D.B.; Cruzen, C.; Simmons, H.A.; Kemnitz, J.W.; et al. Caloric Restriction Delays Disease Onset and Mortality in Rhesus Monkeys. Science 2009, 325, 201–204. [Google Scholar] [CrossRef] [PubMed]
- Colman, R.J.; Beasley, T.M.; Kemnitz, J.W.; Johnson, S.C.; Weindruch, R.; Anderson, R.M. Caloric restriction reduces age-related and all-cause mortality in rhesus monkeys. Nat. Commun. 2014, 5, 3557. [Google Scholar] [CrossRef]
- Mattison, J.A.; Roth, G.S.; Beasley, T.M.; Tilmont, E.M.; Handy, A.M.; Herbert, R.L.; Longo, D.L.; Allison, D.B.; Young, J.E.; Bryant, M.; et al. Impact of caloric restriction on health and survival in rhesus monkeys from the NIA study. Nature 2012, 489, 318–321. [Google Scholar] [CrossRef]
- Todorovic, S.; Smiljanic, K.R.; Ruzdijic, S.D.; Djordjevic, A.N.M.; Kanazir, S.D. Effects of Different Dietary Protocols on General Activity and Frailty of Male Wistar Rats During Aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2018, 73, 1036–1044. [Google Scholar] [CrossRef]
- Prvulovic, M.R.; Milanovic, D.J.; Vujovic, P.Z.; Jovic, M.S.; Kanazir, S.D.; Todorovic, S.T.; Mladenovic, A.N. Late-Onset Calorie Restriction Worsens Cognitive Performances and Increases Frailty Level in Female Wistar Rats. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2021, 77, 947–955. [Google Scholar] [CrossRef]
- Prvulovic, M.; Todorovic, S.; Milanovic, D.; Jovic, M.; Vujovic, P.; Kanazir, S.; Mladenovic, A. Calorie restriction changes the anxiety-like behaviour of ageing male Wistar rats in an onset- and duration-dependent manner. Mech. Ageing Dev. 2022, 204, 111666. [Google Scholar] [CrossRef]
- Longo, V.D.; Antebi, A.; Bartke, A.; Barzilai, N.; Brown-Borg, H.M.; Caruso, C.; Curiel, T.J.; de Cabo, R.; Franceschi, C.; Gems, D.; et al. Interventions to Slow Aging in Humans: Are We Ready? Aging Cell 2015, 14, 497–510. [Google Scholar] [CrossRef]
- Shanley, D.P.; Kirkwood, T.B.L. Caloric restriction does not enhance longevity in all species and is unlikely to do so in humans. Biogerontology 2006, 7, 165–168. [Google Scholar] [CrossRef]
- Zeng, Y.; Feng, Q.; Gu, D.; Vaupel, J. Demographics, phenotypic health characteristics and genetic analysis of centenarians in China. Mech. Ageing Dev. 2017, 165, 86–97. [Google Scholar] [CrossRef] [PubMed]
- Donlon, T.A.; Morris, B.J.; Chen, R.; Masaki, K.H.; Allsopp, R.C.; Willcox, D.C.; Tiirikainen, M.; Willcox, B.J. Analysis of Polymorphisms in 59 Potential Candidate Genes for Association with Human Longevity. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2017, 73, 1459–1464. [Google Scholar] [CrossRef] [PubMed]
- Franceschi, C.; Ostan, R.; Santoro, A. Nutrition and Inflammation: Are Centenarians Similar to Individuals on Calorie-Restricted Diets? Annu. Rev. Nutr. 2018, 38, 329–356. [Google Scholar] [CrossRef] [PubMed]
- Ormazabal, V.; Nair, S.; Elfeky, O.; Aguayo, C.; Salomon, C.; Zuñiga, F.A. Association between insulin resistance and the development of cardiovascular disease. Cardiovasc. Diabetol. 2018, 17, 122. [Google Scholar] [CrossRef]
- Park, C.Y.; Park, S.; Kim, M.S.; Kim, H.-K.; Han, S.N. Effects of mild calorie restriction on lipid metabolism and inflammation in liver and adipose tissue. Biochem. Biophys. Res. Commun. 2017, 490, 636–642. [Google Scholar] [CrossRef]
- Ott, B.; Skurk, T.; Hastreiter, L.; Lagkouvardos, I.; Fischer, S.; Büttner, J.; Kellerer, T.; Clavel, T.; Rychlik, M.; Haller, D.; et al. Effect of caloric restriction on gut permeability, inflammation markers, and fecal microbiota in obese women. Sci. Rep. 2017, 7, 11955. [Google Scholar] [CrossRef]
- Chaudhuri, J.; Bains, Y.; Guha, S.; Kahn, A.; Hall, D.; Bose, N.; Gugliucci, A.; Kapahi, P. The Role of Advanced Glycation End Products in Aging and Metabolic Diseases: Bridging Association and Causality. Cell Metab. 2018, 28, 337–352. [Google Scholar] [CrossRef]
- McCarter, R.; Mejia, W.; Ikeno, Y.; Monnier, V.; Kewitt, K.; Gibbs, M.; McMahan, A.; Strong, R. Plasma Glucose and the Action of Calorie Restriction on Aging. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2007, 62, 1059–1070. [Google Scholar] [CrossRef]
- Templeman, N.M.; Flibotte, S.; Chik, J.H.; Sinha, S.; Lim, G.; Foster, L.J.; Nislow, C.; Johnson, J.D. Reduced Circulating Insulin Enhances Insulin Sensitivity in Old Mice and Extends Lifespan. Cell Rep. 2017, 20, 451–463. [Google Scholar] [CrossRef]
- Masternak, M.M.; Panici, J.A.; Bonkowski, M.S.; Hughes, L.F.; Bartke, A. Insulin Sensitivity as a Key Mediator of Growth Hormone Actions on Longevity. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2009, 64, 516–521. [Google Scholar] [CrossRef]
- Blüher, M.; Kahn, B.B.; Kahn, C.R. Extended Longevity in Mice Lacking the Insulin Receptor in Adipose Tissue. Science 2003, 299, 572–574. [Google Scholar] [CrossRef] [PubMed]
- Selman, C.; Partridge, L.; Withers, D. Replication of Extended Lifespan Phenotype in Mice with Deletion of Insulin Receptor Substrate 1. PLoS ONE 2011, 6, e16144. [Google Scholar] [CrossRef] [PubMed]
- Taguchi, A.; Wartschow, L.M.; White, M.F. Brain IRS2 Signaling Coordinates Life Span and Nutrient Homeostasis. Science 2007, 317, 369–372. [Google Scholar] [CrossRef] [PubMed]
- Nojima, A.; Yamashita, M.; Yoshida, Y.; Shimizu, I.; Ichimiya, H.; Kamimura, N.; Kobayashi, Y.; Ohta, S.; Ishii, N.; Minamino, T. Haploinsufficiency of Akt1 Prolongs the Lifespan of Mice. PLoS ONE 2013, 8, e69178. [Google Scholar] [CrossRef] [PubMed]
- Yu, D.; Tomasiewicz, J.L.; Yang, S.E.; Miller, B.R.; Wakai, M.H.; Sherman, D.S.; Cummings, N.E.; Baar, E.L.; Brinkman, J.A.; Syed, F.A.; et al. Calorie-Restriction-Induced Insulin Sensitivity Is Mediated by Adipose mTORC2 and Not Required for Lifespan Extension. Cell Rep. 2019, 29, 236–248.e3. [Google Scholar] [CrossRef]
- Yamaza, H.; Shimokawa, I. Role of Insulin and Growth Hormone/Insulin-Like Growth Factor-I Signaling in Lifespan Extension: Rodent Longevity Models for Studying Aging and Calorie Restriction. Curr. Genom. 2007, 8, 423–428. [Google Scholar] [CrossRef][Green Version]
- Merry, T.L.; Kuhlow, D.; Laube, B.; Pöhlmann, D.; Pfeiffer, A.F.H.; Kahn, C.R.; Ristow, M.; Zarse, K. Impairment of insulin signalling in peripheral tissue fails to extend murine lifespan. Aging Cell 2017, 16, 761–772. [Google Scholar] [CrossRef]
- Bruss, M.D.; Khambatta, C.F.; Ruby, M.A.; Aggarwal, I.; Hellerstein, M.K. Calorie restriction increases fatty acid synthesis and whole body fat oxidation rates. Am. J. Physiol. Metab. 2010, 298, E108–E116. [Google Scholar] [CrossRef]
- Roberts, M.N.; Wallace, M.A.; Tomilov, A.A.; Zhou, Z.; Marcotte, G.R.; Tran, D.; Perez, G.; Gutierrez-Casado, E.; Koike, S.; Knotts, T.A.; et al. A Ketogenic Diet Extends Longevity and Healthspan in Adult Mice. Cell Metab. 2017, 26, 539–546.e5. [Google Scholar] [CrossRef]
- Breese, C.R.; Ingram, R.L.; Sonntag, W. Influence of Age and Long-term Dietary Restriction on Plasma Insulin-like Growth Factor-1 (IGF-1), IGF-1 Gene Expression, and IGF-1 Binding Proteins. J. Gerontol. 1991, 46, B180–B187. [Google Scholar] [CrossRef]
- Weiss, E.P.; Villareal, D.T.; Racette, S.B.; Steger-May, K.; Premachandra, B.N.; Klein, S.; Fontana, L. Caloric Restriction But Not Exercise-Induced Reductions in Fat Mass Decrease Plasma Triiodothyronine Concentrations: A Randomized Controlled Trial. Rejuvenation Res. 2008, 11, 605–609. [Google Scholar] [CrossRef] [PubMed]
- Fontana, L.; Klein, S.; Holloszy, J.O.; Premachandra, B.N. Effect of Long-Term Calorie Restriction with Adequate Protein and Micronutrients on Thyroid Hormones. J. Clin. Endocrinol. Metab. 2006, 91, 3232–3235. [Google Scholar] [CrossRef] [PubMed]
- Redman, L.M.; Smith, S.R.; Burton, J.H.; Martin, C.K.; Il’Yasova, D.; Ravussin, E. Metabolic Slowing and Reduced Oxidative Damage with Sustained Caloric Restriction Support the Rate of Living and Oxidative Damage Theories of Aging. Cell Metab. 2018, 27, 805–815.e4. [Google Scholar] [CrossRef] [PubMed]
- Barzilai, N.; Huffman, D.M.; Muzumdar, R.H.; Bartke, A. The Critical Role of Metabolic Pathways in Aging. Diabetes 2012, 61, 1315–1322. [Google Scholar] [CrossRef]
- Kebbe, M.; Sparks, J.R.; Flanagan, E.W.; Redman, L.M. Beyond weight loss: Current perspectives on the impact of calorie restriction on healthspan and lifespan. Expert Rev. Endocrinol. Metab. 2021, 16, 95–108. [Google Scholar] [CrossRef] [PubMed]
- Noiret, A.; Puch, L.; Riffaud, C.; Costantini, D.; Riou, J.-F.; Aujard, F.; Terrien, J. Sex-Specific Response to Caloric Restriction After Reproductive Investment in Microcebus murinus: An Integrative Approach. Front. Physiol. 2020, 11, 506. [Google Scholar] [CrossRef]
- Redman, L.M.; Ravussin, E. Caloric Restriction in Humans: Impact on Physiological, Psychological, and Behavioral Outcomes. Antioxid. Redox Signal. 2011, 14, 275–287. [Google Scholar] [CrossRef]
- Gesing, A.; Lewi¿ski, A.; Karbownik-Lewi¿ska, M. The thyroid gland and the process of aging; what is new? Thyroid Res. 2012, 5, 16. [Google Scholar] [CrossRef]
- Bano, A.; Dhana, K.; Chaker, L.; Kavousi, M.; Ikram, M.A.; Mattace-Raso, F.U.S.; Peeters, R.P.; Franco, O. Association of Thyroid Function with Life Expectancy with and without Cardiovascular Disease: The Rotterdam Study. JAMA Intern. Med. 2017, 177, 1650–1657. [Google Scholar] [CrossRef]
- Garasto, S.; Montesanto, A.; Corsonello, A.; Lattanzio, F.; Fusco, S.; Passarino, G.; Giarritta, V.P.; Corica, F. Thyroid hormones in extreme longevity. Mech. Ageing Dev. 2017, 165, 98–106. [Google Scholar] [CrossRef]
- Janssen, J.A. IGF-I and the endocrinology of aging. Curr. Opin. Endocr. Metab. Res. 2018, 5, 1–6. [Google Scholar] [CrossRef]
- Barbieri, M.; Gambardella, A.; Paolisso, G.; Varricchio, M. Metabolic aspects of the extreme longevity. Exp. Gerontol. 2008, 43, 74–78. [Google Scholar] [CrossRef] [PubMed]
- Lane, M.A.; Ingram, D.K.; Ball, S.S.; Roth, G.S. Dehydroepiandrosterone Sulfate: A Biomarker of Primate Aging Slowed by Calorie Restriction. J. Clin. Endocrinol. Metab. 1997, 82, 2093–2096. [Google Scholar] [CrossRef] [PubMed]
- Urbanski, H.F.; Downs, J.L.; Garyfallou, V.T.; Mattison, A.J.; Lane, M.A.; Roth, G.S.; Ingram, D.K. Effect of Caloric Restriction on the 24-Hour Plasma DHEAS and Cortisol Profiles of Young and Old Male Rhesus Macaques. Ann. N. Y. Acad. Sci. 2004, 1019, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Paolisso, G.; Ammendola, S.; Rotondi, M.; Gambardella, A.; Rizzo, M.R.; Mazziotti, G.; Tagliamonte, M.R.; Carella, C.; Varrichio, M. Insulin resistance and advancing age: What role for dehydroepiandrosterone sulfate? Metabolism 1997, 46, 1281–1286. [Google Scholar] [CrossRef]
- Kim, D.H.; Bang, E.; Jung, H.J.; Noh, S.G.; Yu, B.P.; Choi, Y.J.; Chung, H.Y. Anti-Aging Effects of Calorie Restriction (CR) and CR Mimetics Based on the Senoinflammation Concept. Nutrients 2020, 12, 422. [Google Scholar] [CrossRef] [PubMed]
- Jung, K.J.; Lee, E.K.; Kim, J.Y.; Zou, Y.; Sung, B.; Heo, H.S.; Kim, M.K.; Lee, J.; Kim, N.D.; Yu, B.P.; et al. Effect of short term calorie restriction on pro-inflammatory NF-kB and AP-1 in aged rat kidney. Agents Actions 2009, 58, 143–150. [Google Scholar] [CrossRef]
- Meydani, S.N.; Das, S.K.; Pieper, C.F.; Lewis, M.R.; Klein, S.; Dixit, V.D.; Gupta, A.K.; Villareal, D.T.; Bhapkar, M.; Huang, M.; et al. Long-term moderate calorie restriction inhibits inflammation without impairing cell-mediated immunity: A randomized controlled trial in non-obese humans. Aging 2016, 8, 1416–1431. [Google Scholar] [CrossRef]
- Sung, B.; Park, S.; Yu, B.P.; Chung, H.Y. Modulation of PPAR in Aging, Inflammation, and Calorie Restriction. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2004, 59, B997–B1006. [Google Scholar] [CrossRef]
- Pan, F.; Zhang, L.; Li, M.; Hu, Y.; Zeng, B.; Yuan, H.; Zhao, L.; Zhang, C. Predominant gut Lactobacillus murinus strain mediates anti-inflammaging effects in calorie-restricted mice. Microbiome 2018, 6, 54. [Google Scholar] [CrossRef]
- Anderson, R.M.; Weindruch, R. The caloric restriction paradigm: Implications for healthy human aging. Am. J. Hum. Biol. 2012, 24, 101–106. [Google Scholar] [CrossRef] [PubMed]
- Higami, Y.; Barger, J.L.; Page, G.; Allison, D.; Smith, S.R.; Prolla, T.A.; Weindruch, R. Energy Restriction Lowers the Expression of Genes Linked to Inflammation, the Cytoskeleton, the Extracellular Matrix, and Angiogenesis in Mouse Adipose Tissue. J. Nutr. 2006, 136, 343–352. [Google Scholar] [CrossRef] [PubMed]
- Kurki, E.; Shi, J.; Martonen, E.; Finckenberg, P.; Mervaala, E. Distinct effects of calorie restriction on adipose tissue cytokine and angiogenesis profiles in obese and lean mice. Nutr. Metab. 2012, 9, 64. [Google Scholar] [CrossRef]
- Allen, B.D.; Liao, C.; Shu, J.; Muglia, L.J.; Majzoub, J.A.; Diaz, V.; Nelson, J.F. Hyperadrenocorticism of calorie restriction contributes to its anti-inflammatory action in mice. Aging Cell 2019, 18, e12944. [Google Scholar] [CrossRef] [PubMed]
- Langille, M.G.; Meehan, C.J.; Koenig, J.E.; Dhanani, A.S.; Rose, R.A.; Howlett, S.E.; Beiko, R.G. Microbial shifts in the aging mouse gut. Microbiome 2014, 2, 50. [Google Scholar] [CrossRef] [PubMed]
- Smith, P.; Willemsen, D.; Popkes, M.; Metge, F.; Gandiwa, E.; Reichard, M.; Valenzano, D.R. Regulation of life span by the gut microbiota in the short-lived African turquoise killifish. eLife 2017, 6, e27014. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.; Benayoun, B.A. The microbiome: An emerging key player in aging and longevity. Transl. Med. Aging 2020, 4, 103–116. [Google Scholar] [CrossRef]
- Shin, J.; Noh, J.-R.; Choe, D.; Lee, N.; Song, Y.; Cho, S.; Kang, E.-J.; Go, M.-J.; Ha, S.K.; Chang, D.-H.; et al. Ageing and rejuvenation models reveal changes in key microbial communities associated with healthy ageing. Microbiome 2021, 9, 240. [Google Scholar] [CrossRef]
- Biagi, E.; Franceschi, C.; Rampelli, S.; Severgnini, M.; Ostan, R.; Turroni, S.; Consolandi, C.; Quercia, S.; Scurti, M.; Monti, D.; et al. Gut Microbiota and Extreme Longevity. Curr. Biol. 2016, 26, 1480–1485. [Google Scholar] [CrossRef]
- Bárcena, C.; Valdés-Mas, R.; Mayoral, P.; Garabaya, C.; Durand, S.; Rodríguez, F.; Fernández-García, M.T.; Salazar, N.; Nogacka, A.M.; Garatachea, N.; et al. Healthspan and lifespan extension by fecal microbiota transplantation into progeroid mice. Nat. Med. 2019, 25, 1234–1242. [Google Scholar] [CrossRef]
- Wang, N.; Li, R.; Lin, H.; Fu, C.; Wang, X.; Zhang, Y.; Su, M.; Huang, P.; Qian, J.; Jiang, F.; et al. Enriched taxa were found among the gut microbiota of centenarians in East China. PLoS ONE 2019, 14, e0222763. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Zhang, S.; Zeng, B.; Zhao, J.; Yang, M.; Zhang, M.; Li, Y.; Ni, Q.; Wu, D.; Li, Y. Transplant of microbiota from long-living people to mice reduces aging-related indices and transfers beneficial bacteria. Aging 2020, 12, 4778–4793. [Google Scholar] [CrossRef] [PubMed]
- Grajeda-Iglesias, C.; Durand, S.; Daillère, R.; Iribarren, K.; Lemaitre, F.; Derosa, L.; Aprahamian, F.; Bossut, N.; Nirmalathasan, N.; Madeo, F.; et al. Oral administration of Akkermansia muciniphila elevates systemic antiaging and anticancer metabolites. Aging 2021, 13, 6375–6405. [Google Scholar] [CrossRef] [PubMed]
- Han, B.; Sivaramakrishnan, P.; Lin, C.-C.J.; Neve, I.A.; He, J.; Tay, L.W.R.; Sowa, J.N.; Sizovs, A.; Du, G.; Wang, J.; et al. Microbial Genetic Composition Tunes Host Longevity. Cell 2017, 169, 1249–1262.e13. [Google Scholar] [CrossRef]
- Wang, S.; Huang, M.; You, X.; Zhao, J.; Chen, L.; Wang, L.; Luo, Y.; Chen, Y. Gut microbiota mediates the anti-obesity effect of calorie restriction in mice. Sci. Rep. 2018, 8, 13037. [Google Scholar] [CrossRef]
- Zheng, X.; Wang, S.; Jia, W. Calorie restriction and its impact on gut microbial composition and global metabolism. Front. Med. 2018, 12, 634–644. [Google Scholar] [CrossRef]
- Zhang, C.; Li, S.; Yang, L.; Huang, P.; Li, W.; Wang, S.; Zhao, G.; Zhang, M.; Pang, X.; Yan, Z.; et al. Structural modulation of gut microbiota in life-long calorie-restricted mice. Nat. Commun. 2013, 4, 2163. [Google Scholar] [CrossRef]
- Fraumene, C.; Manghina, V.; Cadoni, E.; Marongiu, F.; Abbondio, M.; Serra, M.; Palomba, A.; Tanca, A.; Laconi, E.; Uzzau, S. Caloric restriction promotes rapid expansion and long-lasting increase of Lactobacillus in the rat fecal microbiota. Gut Microbes 2017, 9, 104–114. [Google Scholar] [CrossRef]
- Tanca, A.; Abbondio, M.; Palomba, A.; Fraumene, C.; Marongiu, F.; Serra, M.; Pagnozzi, D.; Laconi, E.; Uzzau, S. Caloric restriction promotes functional changes involving short-chain fatty acid biosynthesis in the rat gut microbiota. Sci. Rep. 2018, 8, 14778. [Google Scholar] [CrossRef]
- Cătoi, A.F.; Corina, A.; Katsiki, N.; Vodnar, D.C.; Andreicuț, A.D.; Stoian, A.P.; Rizzo, M.; Pérez-Martínez, P. Gut microbiota and aging—A focus on centenarians. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165765. [Google Scholar] [CrossRef]
- Salazar, N.; Arboleya, S.; Fernández-Navarro, T.; Reyes-Gavilán, C.G.D.L.; Gonzalez, S.; Gueimonde, M. Age-Associated Changes in Gut Microbiota and Dietary Components Related with the Immune System in Adulthood and Old Age: A Cross-Sectional Study. Nutrients 2019, 11, 1765. [Google Scholar] [CrossRef]
- Snyder, D.L.; Pollard, M.; Wostmann, B.S.; Luckert, P. Life Span, Morphology, and Pathology of Diet-Restricted Germ-Free and Conventional Lobund-Wistar Rats. J. Gerontol. 1990, 45, B52–B58. [Google Scholar] [CrossRef] [PubMed]
- Green, C.L.; Lamming, D.W.; Fontana, L. Molecular mechanisms of dietary restriction promoting health and longevity. Nat. Rev. Mol. Cell Biol. 2021, 23, 56–73. [Google Scholar] [CrossRef]
- Aiello, A.; Accardi, G.; Candore, G.; Gambino, C.M.; Mirisola, M.; Taormina, G.; Virruso, C.; Caruso, C. Nutrient sensing pathways as therapeutic targets for healthy ageing. Expert Opin. Ther. Targets 2017, 21, 371–380. [Google Scholar] [CrossRef] [PubMed]
- Taniguchi, C.M.; Emanuelli, B.; Kahn, C.R. Critical nodes in signalling pathways: Insights into insulin action. Nat. Rev. Mol. Cell Biol. 2006, 7, 85–96. [Google Scholar] [CrossRef]
- Boucher, J.; Kleinridders, A.; Kahn, C.R. Insulin Receptor Signaling in Normal and Insulin-Resistant States. Cold Spring Harb. Perspect. Biol. 2014, 6, a009191. [Google Scholar] [CrossRef] [PubMed]
- Kimura, K.D.; Tissenbaum, H.A.; Liu, Y.; Ruvkun, G. daf-2, an Insulin Receptor-Like Gene That Regulates Longevity and Diapause in Caenorhabditis elegans. Science 1997, 277, 942–946. [Google Scholar] [CrossRef]
- Tatar, M.; Kopelman, A.; Epstein, D.; Tu, M.-P.; Yin, C.-M.; Garofalo, R.S. A Mutant Drosophila Insulin Receptor Homolog That Extends Life-Span and Impairs Neuroendocrine Function. Science 2001, 292, 107–110. [Google Scholar] [CrossRef]
- Holzenberger, M.; Dupont, J.; Ducos, B.; Leneuve, P.; Géloën, A.; Even, P.C.; Cervera, P.; Le Bouc, Y. IGF-1 receptor regulates lifespan and resistance to oxidative stress in mice. Nature 2003, 421, 182–187. [Google Scholar] [CrossRef]
- Mao, K.; Quipildor, G.F.; Tabrizian, T.; Novaj, A.; Guan, F.; Walters, R.O.; Delahaye, F.; Hubbard, G.B.; Ikeno, Y.; Ejima, K.; et al. Late-life targeting of the IGF-1 receptor improves healthspan and lifespan in female mice. Nat. Commun. 2018, 9, 2394. [Google Scholar] [CrossRef]
- Milman, S.; Atzmon, G.; Huffman, D.M.; Wan, J.; Crandall, J.P.; Cohen, P.; Barzilai, N. Low insulin-like growth factor-1 level predicts survival in humans with exceptional longevity. Aging Cell 2014, 13, 769–771. [Google Scholar] [CrossRef] [PubMed]
- Suh, Y.; Atzmon, G.; Cho, M.-O.; Hwang, D.; Liu, B.; Leahy, D.J.; Barzilai, N.; Cohen, P. Functionally significant insulin-like growth factor I receptor mutations in centenarians. Proc. Natl. Acad. Sci. USA 2008, 105, 3438–3442. [Google Scholar] [CrossRef] [PubMed]
- Di Bona, D.; Accardi, G.; Virruso, C.; Candore, G.; Caruso, C. Association between Genetic Variations in the Insulin/Insulin-Like Growth Factor (Igf-1) Signaling Pathway and Longevity: A Systematic Review and Meta-Analysis. Curr. Vasc. Pharmacol. 2013, 12, 674–681. [Google Scholar] [CrossRef] [PubMed]
- Kolovou, V.; Bilianou, H.; Giannakopoulou, V.; Kalogeropoulos, P.; Mihas, C.; Kouris, M.; Cokkinos, D.V.; Boutsikou, M.; Hoursalas, I.; Mavrogeni, S.; et al. Five gene variants in nonagenarians, centenarians and average individuals. Arch. Med. Sci. 2017, 5, 1130–1141. [Google Scholar] [CrossRef] [PubMed]
- Selman, C.; Tullet, J.M.A.; Wieser, D.; Irvine, E.; Lingard, S.J.; Choudhury, A.I.; Claret, M.; Al-Qassab, H.; Carmignac, D.; Ramadani, F.; et al. Ribosomal Protein S6 Kinase 1 Signaling Regulates Mammalian Life Span. Science 2009, 326, 140–144. [Google Scholar] [CrossRef]
- Shimokawa, I.; Komatsu, T.; Hayashi, N.; Kim, S.-E.; Kawata, T.; Park, S.; Hayashi, H.; Yamaza, H.; Chiba, T.; Mori, R. The life-extending effect of dietary restriction requires F oxo3 in mice. Aging Cell 2015, 14, 707–709. [Google Scholar] [CrossRef]
- Kappeler, L.; Filho, C.D.M.; Dupont, J.; Leneuve, P.; Cervera, P.; Périn, L.; Loudes, C.; Blaise, A.; Klein, R.; Epelbaum, J.; et al. Brain IGF-1 Receptors Control Mammalian Growth and Lifespan through a Neuroendocrine Mechanism. PLoS Biol. 2008, 6, e254. [Google Scholar] [CrossRef]
- Hardie, D.G.; Ross, F.A.; Hawley, S.A. AMPK: A nutrient and energy sensor that maintains energy homeostasis. Nat. Rev. Mol. Cell Biol. 2012, 13, 251–262. [Google Scholar] [CrossRef]
- Ross, F.A.; MacKintosh, C.; Hardie, D.G. AMP-activated protein kinase: A cellular energy sensor that comes in 12 flavours. FEBS J. 2016, 283, 2987–3001. [Google Scholar] [CrossRef]
- Ross, F.A.; Jensen, T.E.; Hardie, D.G. Differential regulation by AMP and ADP of AMPK complexes containing different γ subunit isoforms. Biochem. J. 2016, 473, 189–199. [Google Scholar] [CrossRef]
- Jeon, S.-M. Regulation and function of AMPK in physiology and diseases. Exp. Mol. Med. 2016, 48, e245. [Google Scholar] [CrossRef] [PubMed]
- Hardie, D.G. Sensing of energy and nutrients by AMP-activated protein kinase. Am. J. Clin. Nutr. 2011, 93, 891S–896S. [Google Scholar] [CrossRef] [PubMed]
- Cantó, C.; Auwerx, J. Calorie Restriction: Is AMPK a Key Sensor and Effector? Physiology 2011, 26, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Apfeld, J.; O’Connor, G.; McDonagh, T.; DiStefano, P.S.; Curtis, R. The AMP-activated protein kinase AAK-2 links energy levels and insulin-like signals to lifespan in C. elegans. Genes Dev. 2004, 18, 3004–3009. [Google Scholar] [CrossRef]
- Greer, E.L.; Dowlatshahi, D.; Banko, M.R.; Villen, J.; Hoang, K.; Blanchard, D.; Gygi, S.P.; Brunet, A. An AMPK-FOXO Pathway Mediates Longevity Induced by a Novel Method of Dietary Restriction in C. elegans. Curr. Biol. 2007, 17, 1646–1656. [Google Scholar] [CrossRef]
- Stenesen, D.; Suh, J.M.; Seo, J.; Yu, K.; Lee, K.-S.; Kim, J.-S.; Min, K.-J.; Graff, J.M. Adenosine Nucleotide Biosynthesis and AMPK Regulate Adult Life Span and Mediate the Longevity Benefit of Caloric Restriction in Flies. Cell Metab. 2013, 17, 101–112. [Google Scholar] [CrossRef]
- Martin-Montalvo, A.; Mercken, E.M.; Mitchell, S.J.; Palacios, H.H.; Mote, P.L.; Scheibye-Knudsen, M.; Gomes, A.P.; Ward, T.M.; Minor, R.K.; Blouin, M.-J.; et al. Metformin improves healthspan and lifespan in mice. Nat. Commun. 2013, 4, 2192. [Google Scholar] [CrossRef]
- Burkewitz, K.; Zhang, Y.; Mair, W.B. AMPK at the Nexus of Energetics and Aging. Cell Metab. 2014, 20, 10–25. [Google Scholar] [CrossRef]
- Yavari, A.; Stocker, C.J.; Ghaffari, S.; Wargent, E.; Steeples, V.; Czibik, G.; Pinter, K.; Bellahcene, M.; Woods, A.; de Morentin, P.B.M.; et al. Chronic Activation of γ2 AMPK Induces Obesity and Reduces β Cell Function. Cell Metab. 2016, 23, 821–836. [Google Scholar] [CrossRef]
- Gonzalez, A.A.; Kumar, R.; Mulligan, J.D.; Davis, A.J.; Weindruch, R.; Saupe, K.W. Metabolic adaptations to fasting and chronic caloric restriction in heart, muscle, and liver do not include changes in AMPK activity. Am. J. Physiol. Metab. 2004, 287, E1032–E1037. [Google Scholar] [CrossRef][Green Version]
- To, K.; Yamaza, H.; Komatsu, T.; Hayashida, T.; Hayashi, H.; Toyama, H.; Chiba, T.; Higami, Y.; Shimokawa, I. Down-regulation of AMP-activated protein kinase by calorie restriction in rat liver. Exp. Gerontol. 2007, 42, 1063–1071. [Google Scholar] [CrossRef] [PubMed]
- Edwards, A.G.; Donato, A.J.; Lesniewski, L.A.; Gioscia, R.A.; Seals, D.R.; Moore, R.L. Life-long caloric restriction elicits pronounced protection of the aged myocardium: A role for AMPK. Mech. Ageing Dev. 2010, 131, 739–742. [Google Scholar] [CrossRef] [PubMed]
- Palacios, O.M.; Carmona, J.J.; Michan, S.; Chen, K.Y.; Manabe, Y.; Ward, J.L., 3rd; Goodyear, L.J.; Tong, Q. Diet and exercise signals regulate SIRT3 and activate AMPK and PGC-1α in skeletal muscle. Aging 2009, 1, 771–783. [Google Scholar] [CrossRef] [PubMed]
- Burkewitz, K.; Morantte, I.; Weir, H.J.; Yeo, R.; Zhang, Y.; Huynh, F.; Ilkayeva, O.R.; Hirschey, M.; Grant, A.; Mair, W.B. Neuronal CRTC-1 Governs Systemic Mitochondrial Metabolism and Lifespan via a Catecholamine Signal. Cell 2015, 160, 842–855. [Google Scholar] [CrossRef] [PubMed]
- Ulgherait, M.; Rana, A.; Rera, M.; Graniel, J.; Walker, D.W. AMPK Modulates Tissue and Organismal Aging in a Non-Cell-Autonomous Manner. Cell Rep. 2014, 8, 1767–1780. [Google Scholar] [CrossRef] [PubMed]
- Shimobayashi, M.; Hall, M.N. Making new contacts: The mTOR network in metabolism and signalling crosstalk. Nat. Rev. Mol. Cell Biol. 2014, 15, 155–162. [Google Scholar] [CrossRef]
- Robida-Stubbs, S.; Glover-Cutter, K.; Lamming, D.W.; Mizunuma, M.; Narasimhan, S.D.; Neumann-Haefelin, E.; Sabatini, D.M.; Blackwell, T.K. TOR Signaling and Rapamycin Influence Longevity by Regulating SKN-1/Nrf and DAF-16/FoxO. Cell Metab. 2012, 15, 713–724. [Google Scholar] [CrossRef]
- Jia, K.; Chen, D.; Riddle, D.L. The TOR pathway interacts with the insulin signaling pathway to regulate C. elegans larval development, metabolism and life span. Development 2004, 131, 3897–3906. [Google Scholar] [CrossRef]
- Kapahi, P.; Zid, B.; Harper, T.; Koslover, D.; Sapin, V.; Benzer, S. Regulation of Lifespan in Drosophila by Modulation of Genes in the TOR Signaling Pathway. Curr. Biol. 2004, 14, 885–890. [Google Scholar] [CrossRef]
- Schinaman, J.M.; Rana, A.; Ja, W.W.; Clark, R.I.; Walker, D.W. Rapamycin modulates tissue aging and lifespan independently of the gut microbiota in Drosophila. Sci. Rep. 2019, 9, 7824. [Google Scholar] [CrossRef]
- Wu, J.J.; Liu, J.; Chen, E.B.; Wang, J.J.; Cao, L.; Narayan, N.; Fergusson, M.M.; Rovira, I.I.; Allen, M.; Springer, D.A.; et al. Increased Mammalian Lifespan and a Segmental and Tissue-Specific Slowing of Aging after Genetic Reduction of mTOR Expression. Cell Rep. 2013, 4, 913–920. [Google Scholar] [CrossRef] [PubMed]
- Harrison, D.E.; Strong, R.; Sharp, Z.D.; Nelson, J.F.; Astle, C.M.; Flurkey, K.; Nadon, N.L.; Wilkinson, J.E.; Frenkel, K.; Carter, C.S.; et al. Rapamycin fed late in life extends lifespan in genetically heterogeneous mice. Nature 2009, 460, 392–395. [Google Scholar] [CrossRef] [PubMed]
- González, A.; Hall, M.N.; Lin, S.-C.; Hardie, D.G. AMPK and TOR: The Yin and Yang of Cellular Nutrient Sensing and Growth Control. Cell Metab. 2020, 31, 472–492. [Google Scholar] [CrossRef] [PubMed]
- Carosi, J.M.; Fourrier, C.; Bensalem, J.; Sargeant, T.J. The mTOR–lysosome axis at the centre of ageing. FEBS Open Bio 2021, 12, 739–757. [Google Scholar] [CrossRef]
- Haigis, M.C.; Sinclair, D.A. Mammalian Sirtuins: Biological Insights and Disease Relevance. Annu. Rev. Pathol. Mech. Dis. 2010, 5, 253–295. [Google Scholar] [CrossRef] [PubMed]
- Chalkiadaki, A.; Guarente, L. The multifaceted functions of sirtuins in cancer. Nat. Cancer 2015, 15, 608–624. [Google Scholar] [CrossRef]
- Covarrubias, A.J.; Perrone, R.; Grozio, A.; Verdin, E. NAD+ metabolism and its roles in cellular processes during ageing. Nat. Rev. Mol. Cell Biol. 2020, 22, 119–141. [Google Scholar] [CrossRef]
- Qiu, X.; Brown, K.V.; Moran, Y.; Chen, D. Sirtuin regulation in calorie restriction. Biochim. Biophys. Acta Proteins Proteom. 2010, 1804, 1576–1583. [Google Scholar] [CrossRef]
- Zhao, S.; Xu, W.; Jiang, W.; Yu, W.; Lin, Y.; Zhang, T.; Yao, J.; Zhou, L.; Zeng, Y.; Li, H.; et al. Regulation of Cellular Metabolism by Protein Lysine Acetylation. Science 2010, 327, 1000–1004. [Google Scholar] [CrossRef]
- Picard, F.; Kurtev, M.; Chung, N.; Topark-Ngarm, A.; Senawong, T.; de Oliveira, R.M.; Leid, M.; McBurney, M.W.; Guarente, L. Sirt1 promotes fat mobilization in white adipocytes by repressing PPAR-γ. Nature 2004, 429, 771–776. [Google Scholar] [CrossRef]
- Gerhart-Hines, Z.; Rodgers, J.T.; Bare, O.; Lerin, C.; Kim, S.-H.; Mostoslavsky, R.; Alt, F.W.; Wu, Z.; Puigserver, P. Metabolic control of muscle mitochondrial function and fatty acid oxidation through SIRT1/PGC-1α. EMBO J. 2007, 26, 1913–1923. [Google Scholar] [CrossRef] [PubMed]
- Qiu, X.; Brown, K.; Hirschey, M.D.; Verdin, E.; Chen, D. Calorie Restriction Reduces Oxidative Stress by SIRT3-Mediated SOD2 Activation. Cell Metab. 2010, 12, 662–667. [Google Scholar] [CrossRef] [PubMed]
- Moynihan, K.A.; Grimm, A.A.; Plueger, M.M.; Bernal-Mizrachi, E.; Ford, E.; Cras-Méneur, C.; Permutt, M.A.; Imai, S.-I. Increased dosage of mammalian Sir2 in pancreatic β cells enhances glucose-stimulated insulin secretion in mice. Cell Metab. 2005, 2, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Frescas, D.; Valenti, L.; Accili, D. Nuclear Trapping of the Forkhead Transcription Factor FoxO1 via Sirt-dependent Deacetylation Promotes Expression of Glucogenetic Genes. J. Biol. Chem. 2005, 280, 20589–20595. [Google Scholar] [CrossRef] [PubMed]
- Ponugoti, B.; Kim, D.-H.; Xiao, Z.; Smith, Z.; Miao, J.; Zang, M.; Wu, S.-Y.; Chiang, C.-M.; Veenstra, T.D.; Kemper, J.K. SIRT1 Deacetylates and Inhibits SREBP-1C Activity in Regulation of Hepatic Lipid Metabolism. J. Biol. Chem. 2010, 285, 33959–33970. [Google Scholar] [CrossRef]
- López-Otín, C.; Blasco, M.A.; Partridge, L.; Serrano, M.; Kroemer, G. The Hallmarks of Aging. Cell 2013, 153, 1194–1217. [Google Scholar] [CrossRef]
- Ng, F.; Tang, B.L. Sirtuins’ modulation of autophagy. J. Cell. Physiol. 2013, 228, 2262–2270. [Google Scholar] [CrossRef]
- Tullet, J.M.A.; Araiz, C.; Sanders, M.J.; Au, C.; Benedetto, A.; Papatheodorou, I.; Clark, E.; Schmeisser, K.; Jones, D.; Schuster, E.F.; et al. DAF-16/FoxO Directly Regulates an Atypical AMP-Activated Protein Kinase Gamma Isoform to Mediate the Effects of Insulin/IGF-1 Signaling on Aging in Caenorhabditis elegans. PLoS Genet. 2014, 10, e1004109. [Google Scholar] [CrossRef]
- Hou, L.; Wang, D.; Chen, D.; Liu, Y.; Zhang, Y.; Cheng, H.; Xu, C.; Sun, N.; McDermott, J.; Mair, W.; et al. A Systems Approach to Reverse Engineer Lifespan Extension by Dietary Restriction. Cell Metab. 2016, 23, 529–540. [Google Scholar] [CrossRef]
- Ruderman, N.B.; Xu, X.J.; Nelson, L.; Cacicedo, J.M.; Saha, A.K.; Lan, F.; Ido, Y. AMPK and SIRT1: A long-standing partnership? Am. J. Physiol. Endocrinol. Metab. 2010, 298, E751–E760. [Google Scholar] [CrossRef]
- Ma, S.; Sun, S.; Geng, L.; Song, M.; Wang, W.; Ye, Y.; Ji, Q.; Zou, Z.; Wang, S.; He, X.; et al. Caloric Restriction Reprograms the Single-Cell Transcriptional Landscape of Rattus Norvegicus Aging. Cell 2020, 180, 984–1001.e22. [Google Scholar] [CrossRef] [PubMed]
- Mori, M.; Raghavan, P.; Thomou, T.; Boucher, J.; Robida-Stubbs, S.; Macotela, Y.; Russell, S.J.; Kirkland, J.L.; Blackwell, T.K.; Kahn, C.R. Role of MicroRNA Processing in Adipose Tissue in Stress Defense and Longevity. Cell Metab. 2012, 16, 336–347. [Google Scholar] [CrossRef] [PubMed]
- Ungvari, Z.; Tucsek, Z.; Sosnowska, D.; Toth, P.; Gautam, T.; Podlutsky, A.; Csiszár, A.; Losonczy, G.; Valcarcel-Ares, M.N.; Sonntag, W. Aging-Induced Dysregulation of Dicer1-Dependent MicroRNA Expression Impairs Angiogenic Capacity of Rat Cerebromicrovascular Endothelial Cells. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2012, 68, 877–891. [Google Scholar] [CrossRef]
- Victoria, B.; Dhahbi, J.M.; Lopez, Y.N.; Spinel, L.P.; Atamna, H.; Spindler, S.R.; Masternak, M.M. Circulating micro RNA signature of genotype-by-age interactions in the long-lived A mes dwarf mouse. Aging Cell 2015, 14, 1055–1066. [Google Scholar] [CrossRef] [PubMed]
- Schneider, A.; Dhahbi, J.M.; Atamna, H.; Clark, J.P.; Colman, R.J.; Anderson, R.M. Caloric restriction impacts plasma microRNAs in rhesus monkeys. Aging Cell 2017, 16, 1200–1203. [Google Scholar] [CrossRef][Green Version]
- Khanna, A.; Muthusamy, S.; Liang, R.; Sarojini, H.; Wang, E. Gain of survival signaling by down-regulation of three key miRNAs in brain of calorie-restricted mice. Aging 2011, 3, 223–236. [Google Scholar] [CrossRef]
- Wood, S.; Van Dam, S.; Craig, T.; Tacutu, R.; O’Toole, A.; Merry, B.J.; De Magalhães, J.P. Transcriptome analysis in calorie-restricted rats implicates epigenetic and post-translational mechanisms in neuroprotection and aging. Genome Biol. 2015, 16, 285. [Google Scholar] [CrossRef]
- Zhang, R.; Wang, X.; Qu, J.-H.; Liu, B.; Zhang, P.; Zhang, T.; Fan, P.-C.; Wang, X.-M.; Xiao, G.-Y.; Su, Y.; et al. Caloric Restriction Induces MicroRNAs to Improve Mitochondrial Proteostasis. IScience 2019, 17, 155–166. [Google Scholar] [CrossRef]
- Cole, J.J.; Robertson, N.; Rather, M.I.; Thomson, J.P.; McBryan, T.; Sproul, D.; Wang, T.; Brock, C.; Clark, W.; Ideker, T.; et al. Diverse interventions that extend mouse lifespan suppress shared age-associated epigenetic changes at critical gene regulatory regions. Genome Biol. 2017, 18, 58. [Google Scholar] [CrossRef]
- Hahn, O.; Grönke, S.; Stubbs, T.; Ficz, G.; Hendrich, O.; Krueger, F.; Andrews, S.; Zhang, Q.; Wakelam, M.; Beyer, A.; et al. Dietary restriction protects from age-associated DNA methylation and induces epigenetic reprogramming of lipid metabolism. Genome Biol. 2017, 18, 56. [Google Scholar] [CrossRef]
- Hearn, J.; Pearson, M.; Blaxter, M.; Wilson, P.J.; Little, T.J. Genome-wide methylation is modified by caloric restriction in Daphnia magna. BMC Genom. 2019, 20, 197. [Google Scholar] [CrossRef] [PubMed]
- Gensous, N.; Franceschi, C.; Santoro, A.; Milazzo, M.; Garagnani, P.; Bacalini, M.G. The Impact of Caloric Restriction on the Epigenetic Signatures of Aging. Int. J. Mol. Sci. 2019, 20, 2022. [Google Scholar] [CrossRef] [PubMed]
- Aon, M.A.; Bernier, M.; Mitchell, S.J.; Di Germanio, C.; Mattison, J.A.; Ehrlich, M.R.; Colman, R.J.; Anderson, R.M.; de Cabo, R. Untangling Determinants of Enhanced Health and Lifespan through a Multi-omics Approach in Mice. Cell Metab. 2020, 32, 100–116.e4. [Google Scholar] [CrossRef]
- Le Couteur, D.; Solon-Biet, S.; Wahl, D.; Cogger, V.C.; Willcox, B.J.; Willcox, D.C.; Raubenheimer, D.; Simpson, S.J. New Horizons: Dietary protein, ageing and the Okinawan ratio. Age Ageing 2016, 45, 443–447. [Google Scholar] [CrossRef] [PubMed]
- Willcox, D.C.; Willcox, B.J.; Todoriki, H.; Suzuki, M. The Okinawan Diet: Health Implications of a Low-Calorie, Nutrient-Dense, Antioxidant-Rich Dietary Pattern Low in Glycemic Load. J. Am. Coll. Nutr. 2009, 28, 500S–516S. [Google Scholar] [CrossRef] [PubMed]
- Serra-Majem, L.; Roman, B.; Estruch, R. Scientific Evidence of Interventions Using the Mediterranean Diet: A Systematic Review. Nutr. Rev. 2006, 64, S27–S47. [Google Scholar] [CrossRef] [PubMed]
- Sofi, F.; Macchi, C.; Abbate, R.; Gensini, G.F.; Casini, A. Mediterranean diet and health status: An updated meta-analysis and a proposal for a literature-based adherence score. Public Health Nutr. 2013, 17, 2769–2782. [Google Scholar] [CrossRef]
- Eleftheriou, D.; Benetou, V.; Trichopoulou, A.; La Vecchia, C.; Bamia, C. Mediterranean diet and its components in relation to all-cause mortality: Meta-analysis. Br. J. Nutr. 2018, 120, 1081–1097. [Google Scholar] [CrossRef]
- Soltani, S.; Jayedi, A.; Shab-Bidar, S.; Becerra-Tomás, N.; Salas-Salvadó, J. Adherence to the Mediterranean Diet in Relation to All-Cause Mortality: A Systematic Review and Dose-Response Meta-Analysis of Prospective Cohort Studies. Adv. Nutr. Int. Rev. J. 2019, 10, 1029–1039. [Google Scholar] [CrossRef]
- Papadaki, A.; Nolen-Doerr, E.; Mantzoros, C.S. The Effect of the Mediterranean Diet on Metabolic Health: A Systematic Review and Meta-Analysis of Controlled Trials in Adults. Nutrients 2020, 12, 3342. [Google Scholar] [CrossRef]
- Bonaccio, M.; Di Castelnuovo, A.; Costanzo, S.; Gialluisi, A.; Persichillo, M.; Cerletti, C.; Donati, M.B.; de Gaetano, G.; Iacoviello, L. Mediterranean diet and mortality in the elderly: A prospective cohort study and a meta-analysis. Br. J. Nutr. 2018, 120, 841–854. [Google Scholar] [CrossRef] [PubMed]
- Murphy, K.J.; Parletta, N. Implementing a Mediterranean-Style Diet Outside the Mediterranean Region. Curr. Atheroscler. Rep. 2018, 20, 28. [Google Scholar] [CrossRef] [PubMed]
- Le Bourg, E. Dietary Restriction Studies in Humans: Focusing on Obesity, Forgetting Longevity. Gerontology 2012, 58, 126–128. [Google Scholar] [CrossRef] [PubMed]
- Le Bourg, E. Predicting whether dietary restriction would increase longevity in species not tested so far. Ageing Res. Rev. 2010, 9, 289–297. [Google Scholar] [CrossRef]
- Jankovic, N.; Geelen, A.; Streppel, M.T.; De Groot, L.C.P.G.M.; Orfanos, P.; Hooven, E.H.V.D.; Pikhart, H.; Boffetta, P.; Trichopoulou, A.; Bobak, M.; et al. Adherence to a Healthy Diet According to the World Health Organization Guidelines and All-Cause Mortality in Elderly Adults from Europe and the United States. Am. J. Epidemiol. 2014, 180, 978–988. [Google Scholar] [CrossRef]
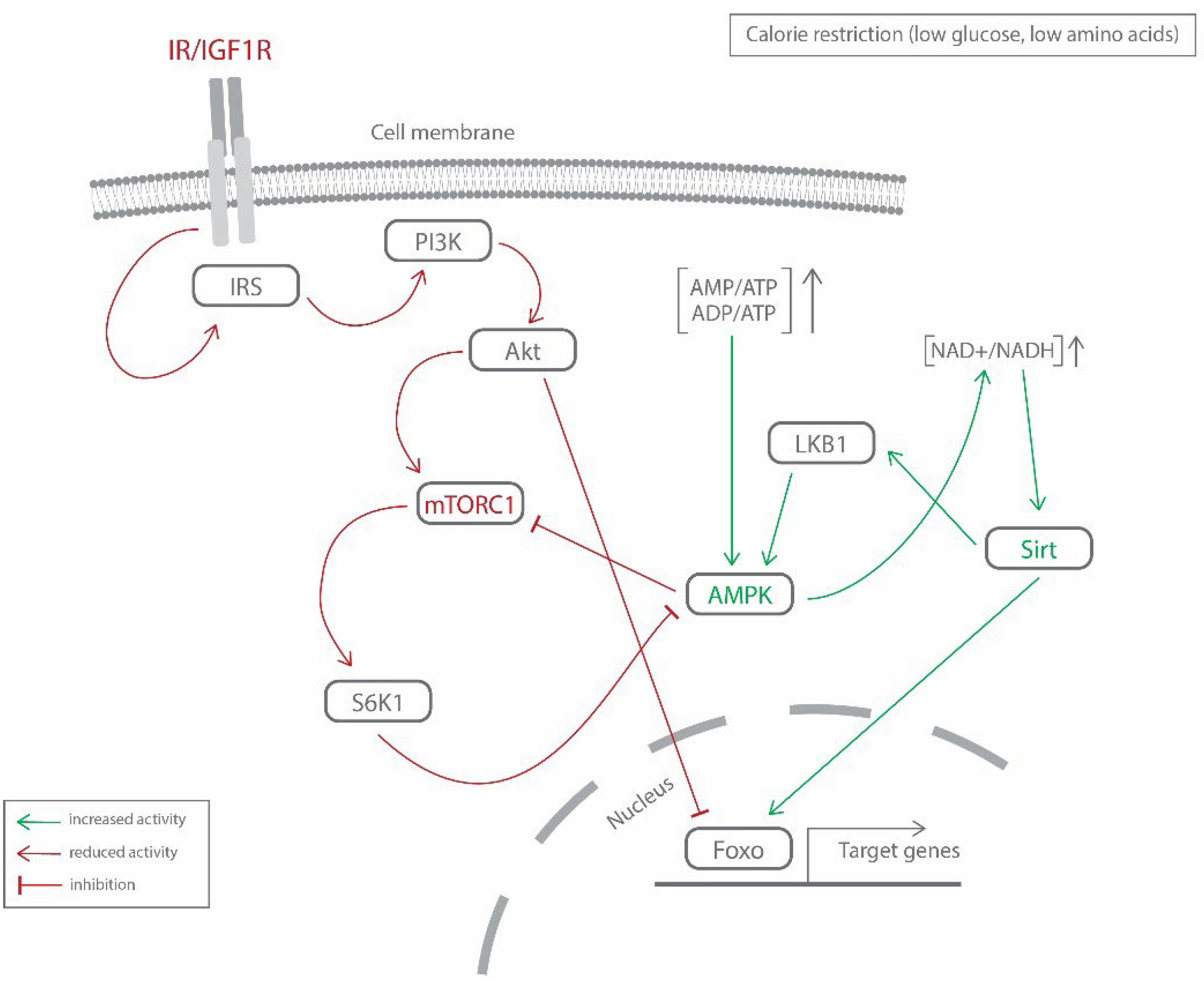
| Study | Population | Topic |
|---|---|---|
| [16] | NA | Centenarian offspring as a model for understanding longevity |
| [20] | Okinawan population | Genetic aspects of longevity |
| [21] | Sardinian population | Clustering in Sardinian longevity |
| [22] | Okinawan population | Nutritional status in Okinawan centenarians |
| [23] | Sardinian population | Food habits and lifestyle in Sardinian centenarians |
| [24] | School children in Okinawa | Nutrition factors and longevity |
| [25] | Japanese population | Impact of westernization on the nutrition of Japanese |
| [26] | Ikarian population | Centenarians’ health status |
| [27] | Ikarian population | Lifestyle and longevity |
| [28] | Italian centenarians | Gender differences in longevity |
| [29] | Costa Rican/Sardinian centenarians | Dietary habits and longevity |
| [30] | Japanese centenarians | Factors associated with longevity in Japan/ 1990 population census data |
| [31] | Ikarian population | Sociodemographic and lifestyle statistics of Ikarian old people (>80 years) |
| [32] | Sardinian male population | Y Chromosome markers and centenarians/genetic structure of the Sardinian population |
| [33] | Sardinian population | Evolution of the Sardinian dietary patterns |
| [34] | Sardinian population/AKEA study | Identification of Sardinia island as an area characterized by extreme longevity |
| [18] | Blue Zones | Blue Zones as areas of exceptional longevity |
| [19] | Greek Adults/EPIC Study | Hypertension in the Greek general population |
| [17] | NA | Worldwide demography of centenarians |
| [7] | New England population | Aging rate of centenarians |
| [35] | Okinawan population | Caloric restriction and human longevity |
| [36] | Okinawan population | Caloric restriction/Okinawan diet |
| [37] | Okinawan and American septuagenarians | Caloric restriction in Okinawans and Americans |
| [38] | Okinawan population | Validation study of centenarians in Okinawa |
| [39] | Okinawan and Japanese populations | Demographic, phenotypic, and genetic characteristics of centenarians in Japan |
| Physiological Factor | Detected during Aging in Animals and Humans | Detected during CR in Animals and Humans | Detected in Centenarians |
|---|---|---|---|
| Insulin sensitivity | ↓ | ↑ | ↑ |
| Circulation insulin | ↑ | ↓ | ↓ |
| Fasting blood glucose | ↑ | ↓ | ↓ |
| Serum IGF1 | ↓ | ↓ − | − |
| Serum GH | ↓ | − | |
| Serum glucocorticoids | ↑ | ↑ | |
| T3 | ↓ | ↓ | |
| T4 | − | − | ↓ |
| TSH | ↑ | − | ↑ |
| DHEAS | ↓ | ↑ − | − |
| Inflammation | ↑ | ↓ | ↑↓ |
| Gut microbiota diversity | ↓ | ↑ | ↑ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Dakic, T.; Jevdjovic, T.; Vujovic, P.; Mladenovic, A. The Less We Eat, the Longer We Live: Can Caloric Restriction Help Us Become Centenarians? Int. J. Mol. Sci. 2022, 23, 6546. https://doi.org/10.3390/ijms23126546
Dakic T, Jevdjovic T, Vujovic P, Mladenovic A. The Less We Eat, the Longer We Live: Can Caloric Restriction Help Us Become Centenarians? International Journal of Molecular Sciences. 2022; 23(12):6546. https://doi.org/10.3390/ijms23126546
Chicago/Turabian StyleDakic, Tamara, Tanja Jevdjovic, Predrag Vujovic, and Aleksandra Mladenovic. 2022. "The Less We Eat, the Longer We Live: Can Caloric Restriction Help Us Become Centenarians?" International Journal of Molecular Sciences 23, no. 12: 6546. https://doi.org/10.3390/ijms23126546
APA StyleDakic, T., Jevdjovic, T., Vujovic, P., & Mladenovic, A. (2022). The Less We Eat, the Longer We Live: Can Caloric Restriction Help Us Become Centenarians? International Journal of Molecular Sciences, 23(12), 6546. https://doi.org/10.3390/ijms23126546







