A Lack of GD3 Synthase Leads to Impaired Renal Expression of Connexins and Pannexin1 in St8sia1 Knockout Mice
Abstract
1. Introduction
2. Results
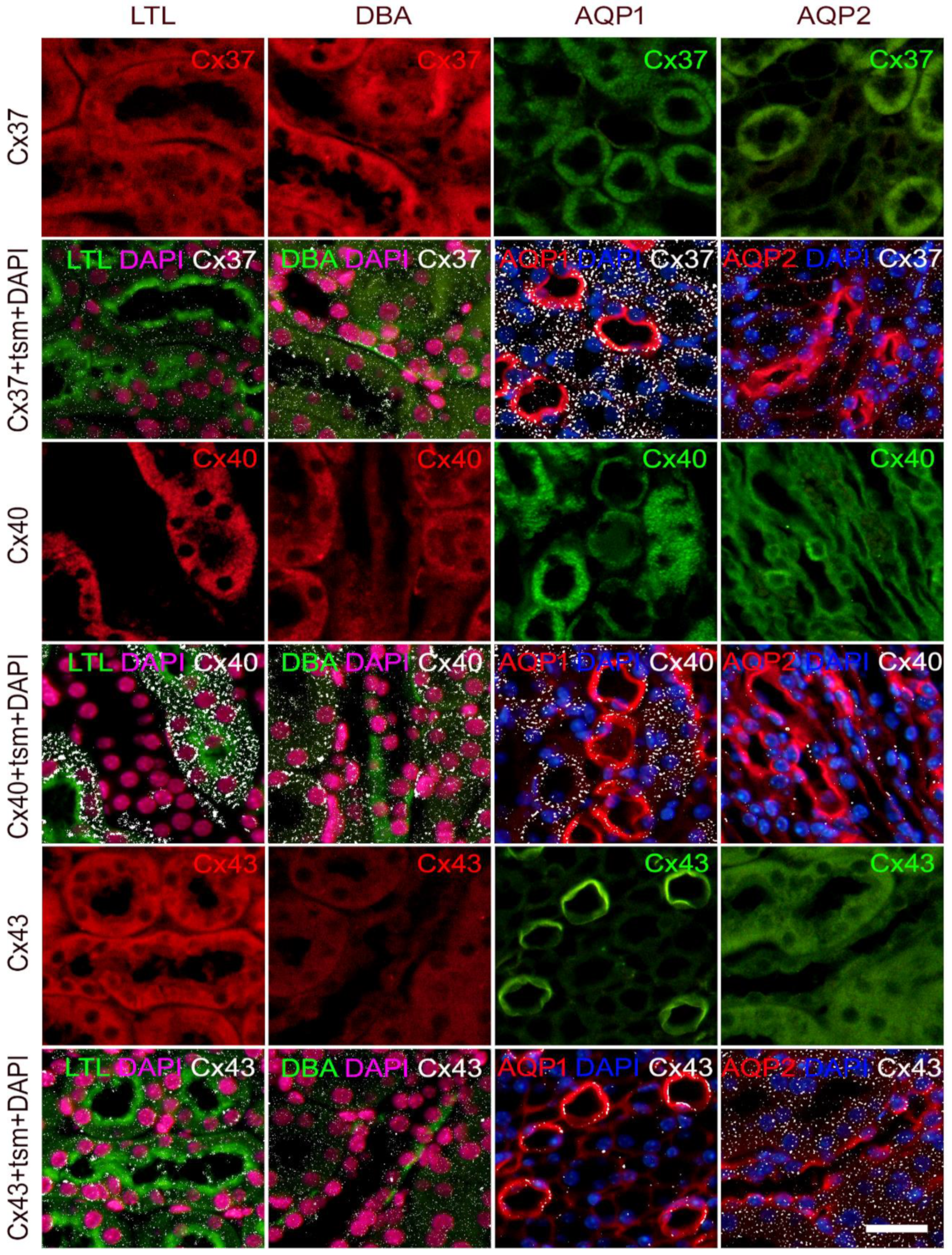
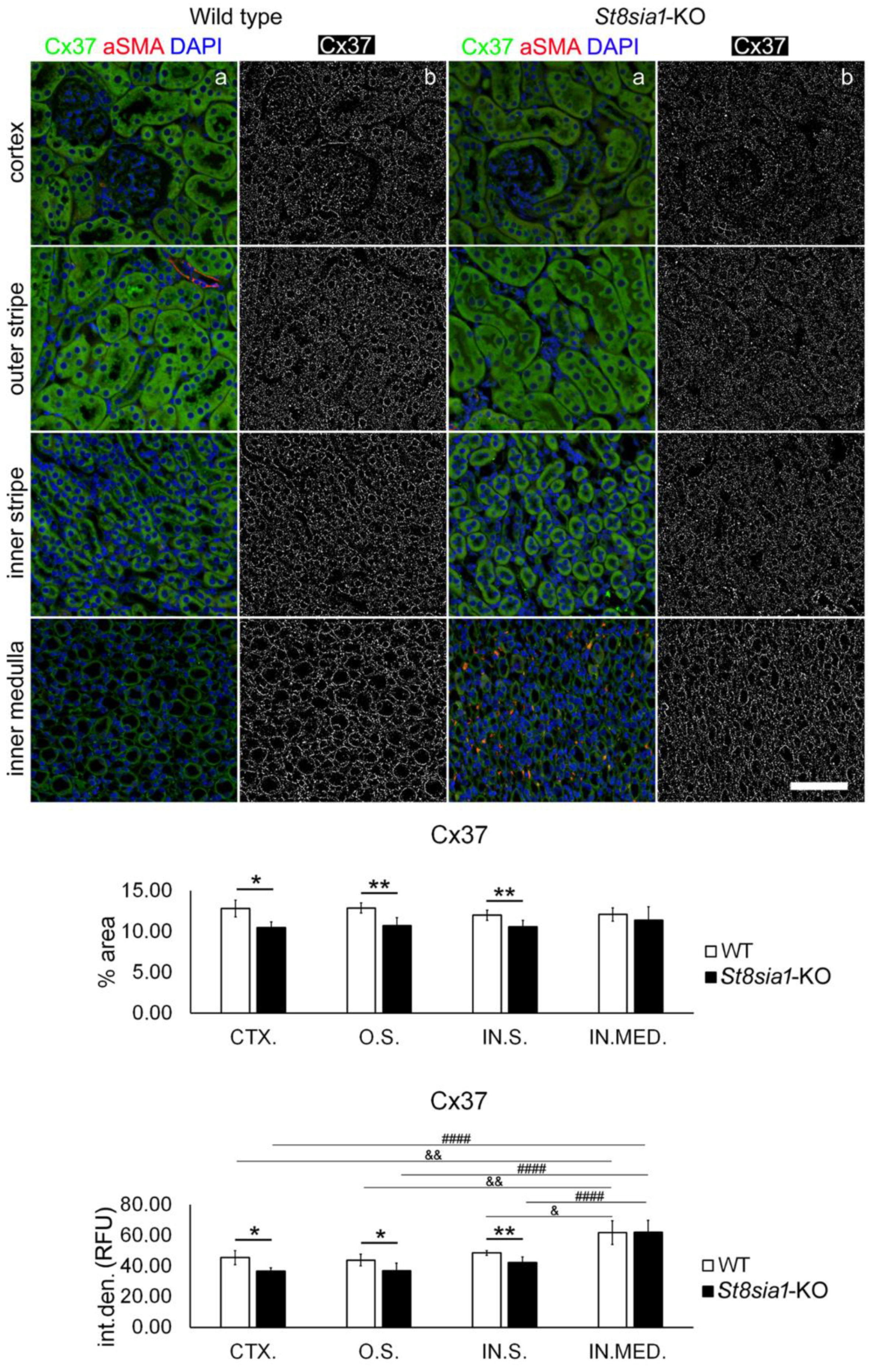
2.1. Expression of Connexin 37 in Kidney of Wild Type and St8sia1-KO Mice
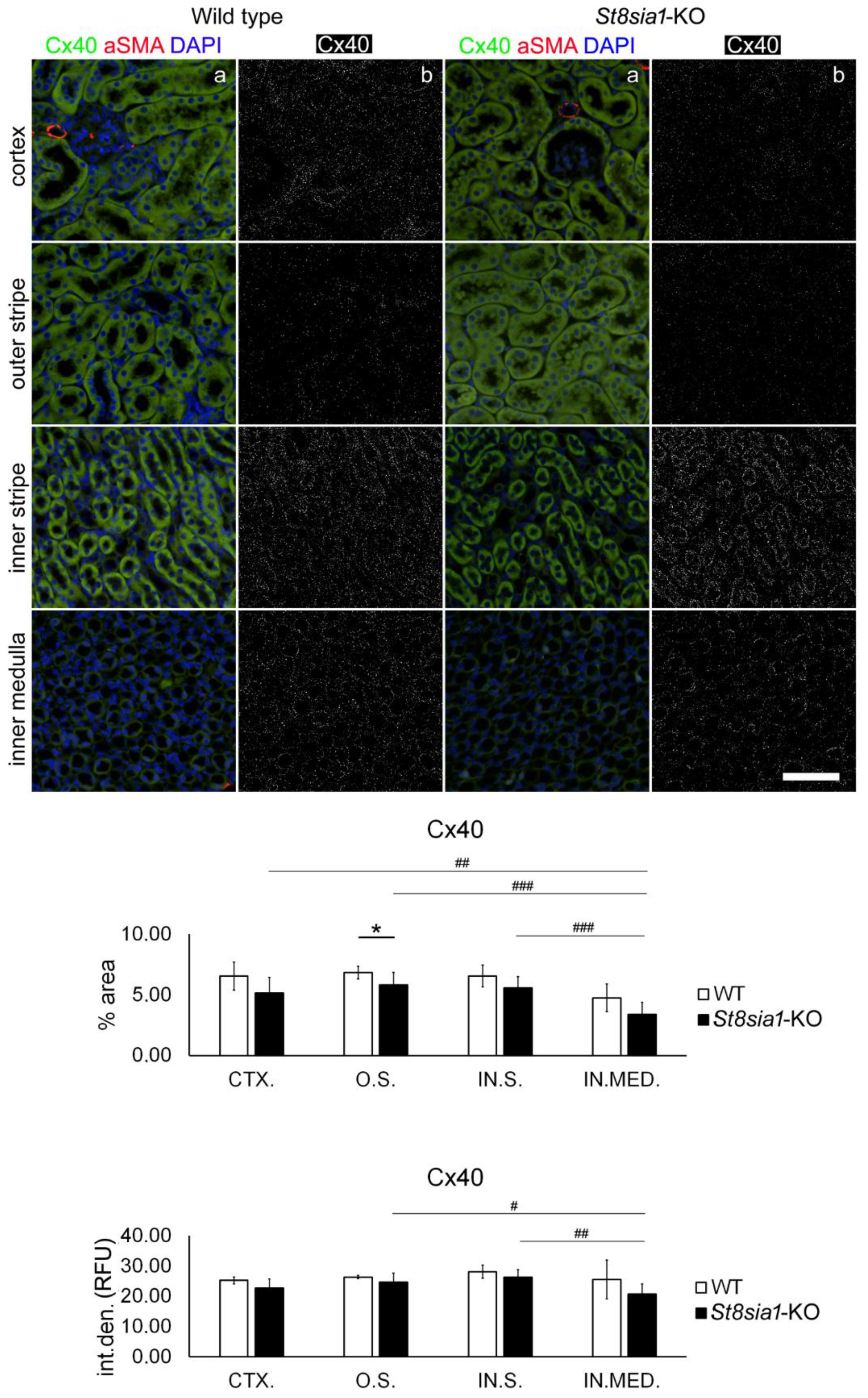
2.2. Expression of Connexin 40 in the Kidney of Wild-Type and St8sia1-KO Mice
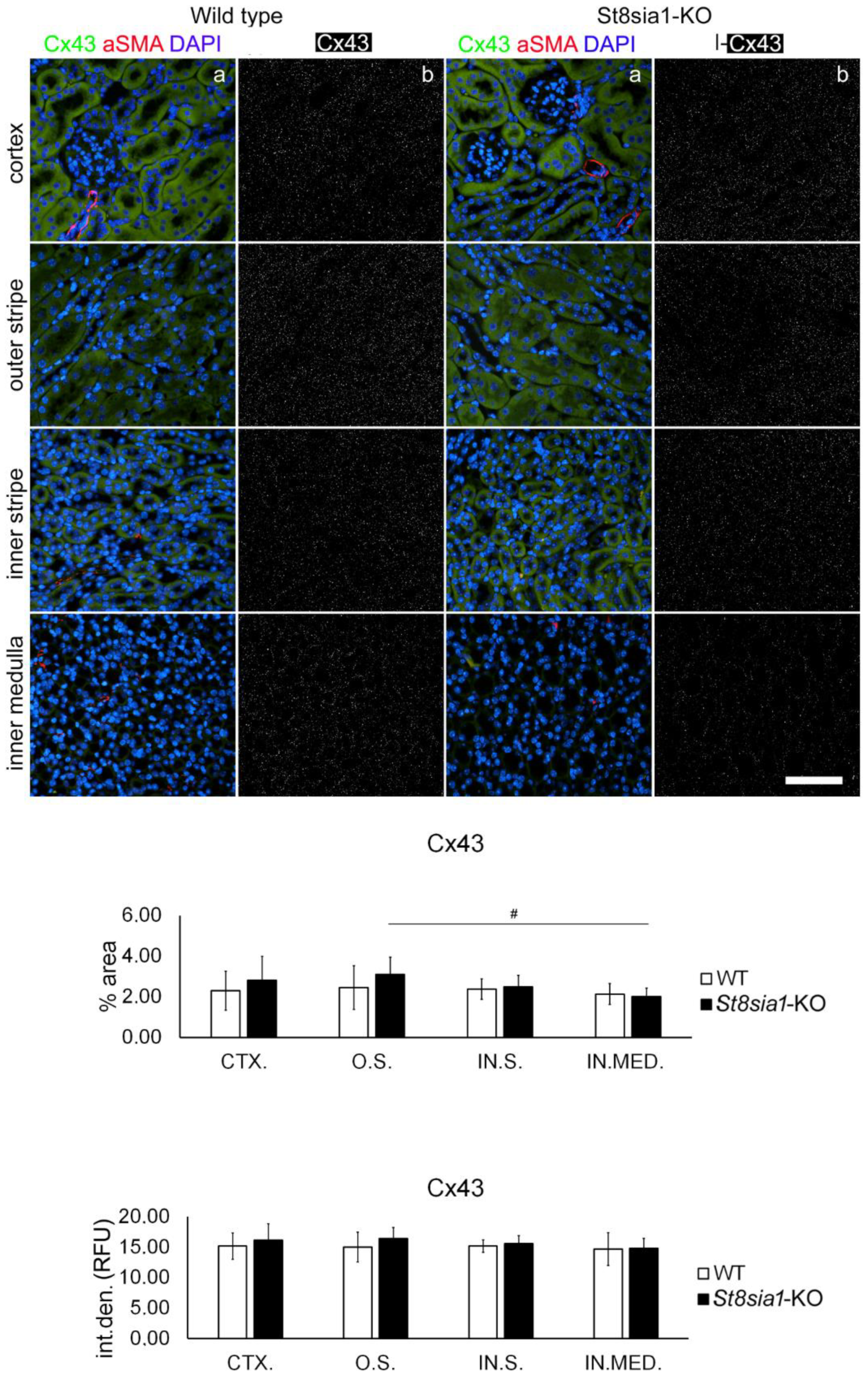
2.3. Expression of Connexin 43 in the Kidney of Wild-Type and St8sia1-KO Mice
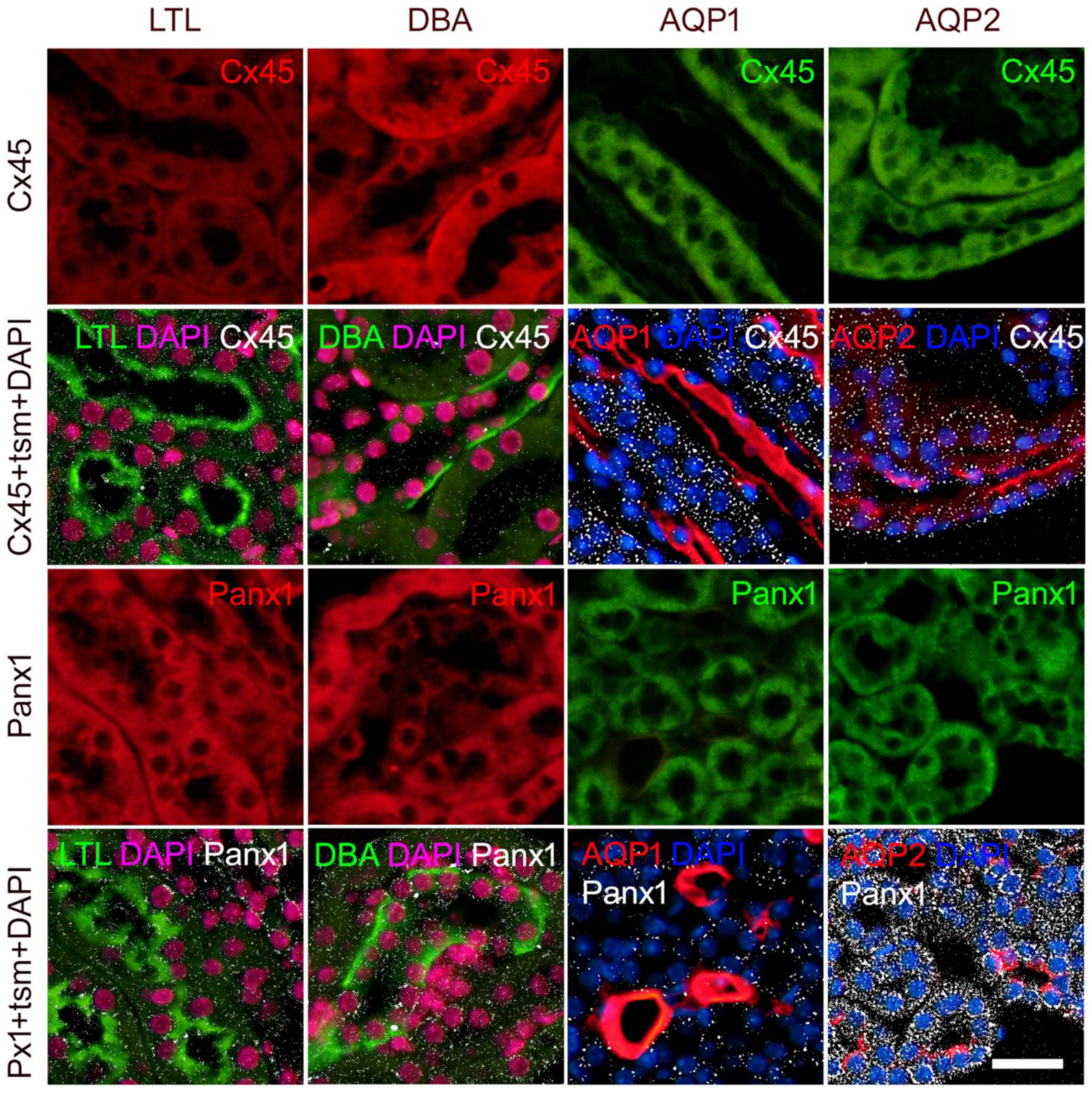
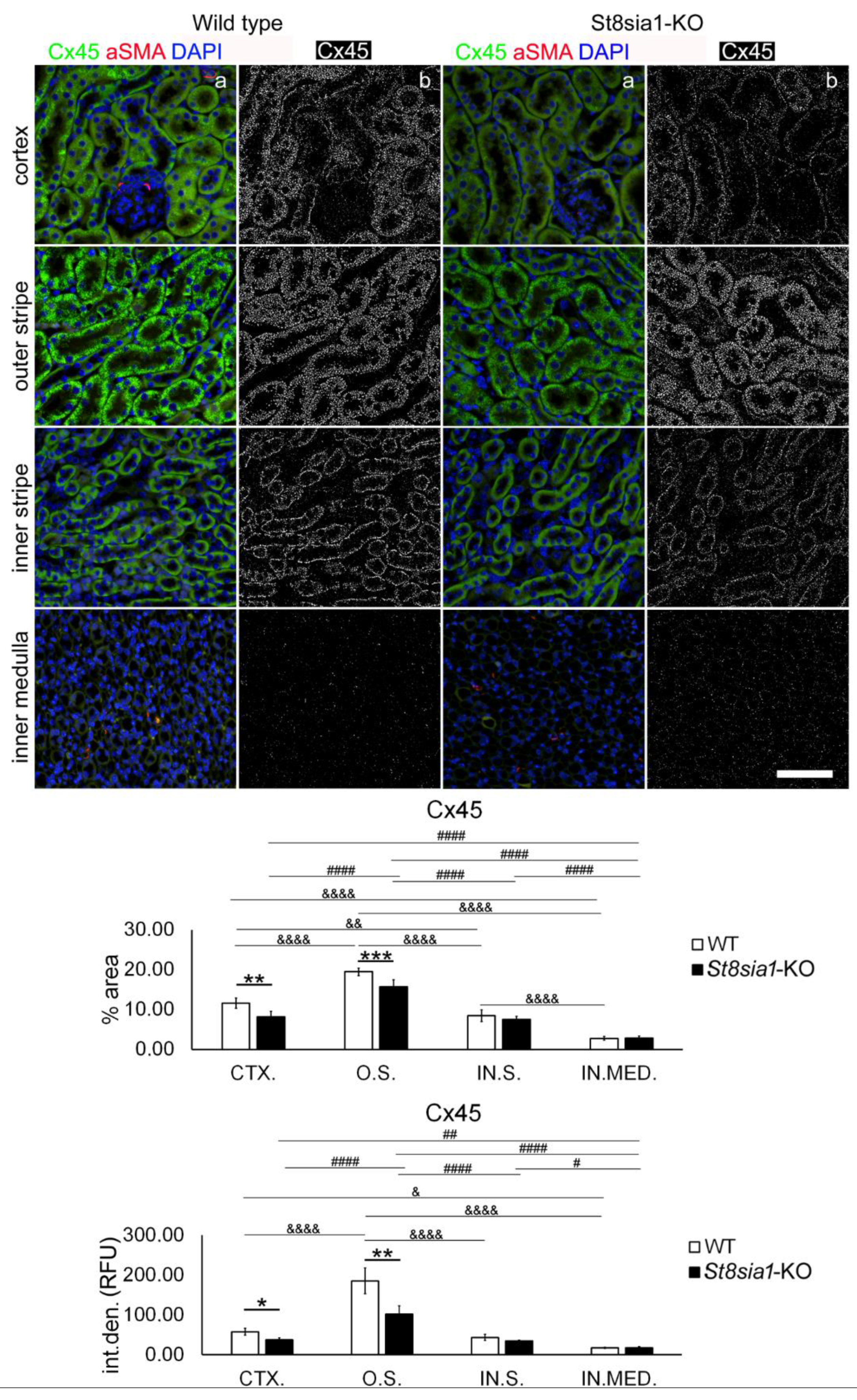
2.4. Expression of Connexin 45 in the Kidney of Wild-Type and St8sia1-KO Mice
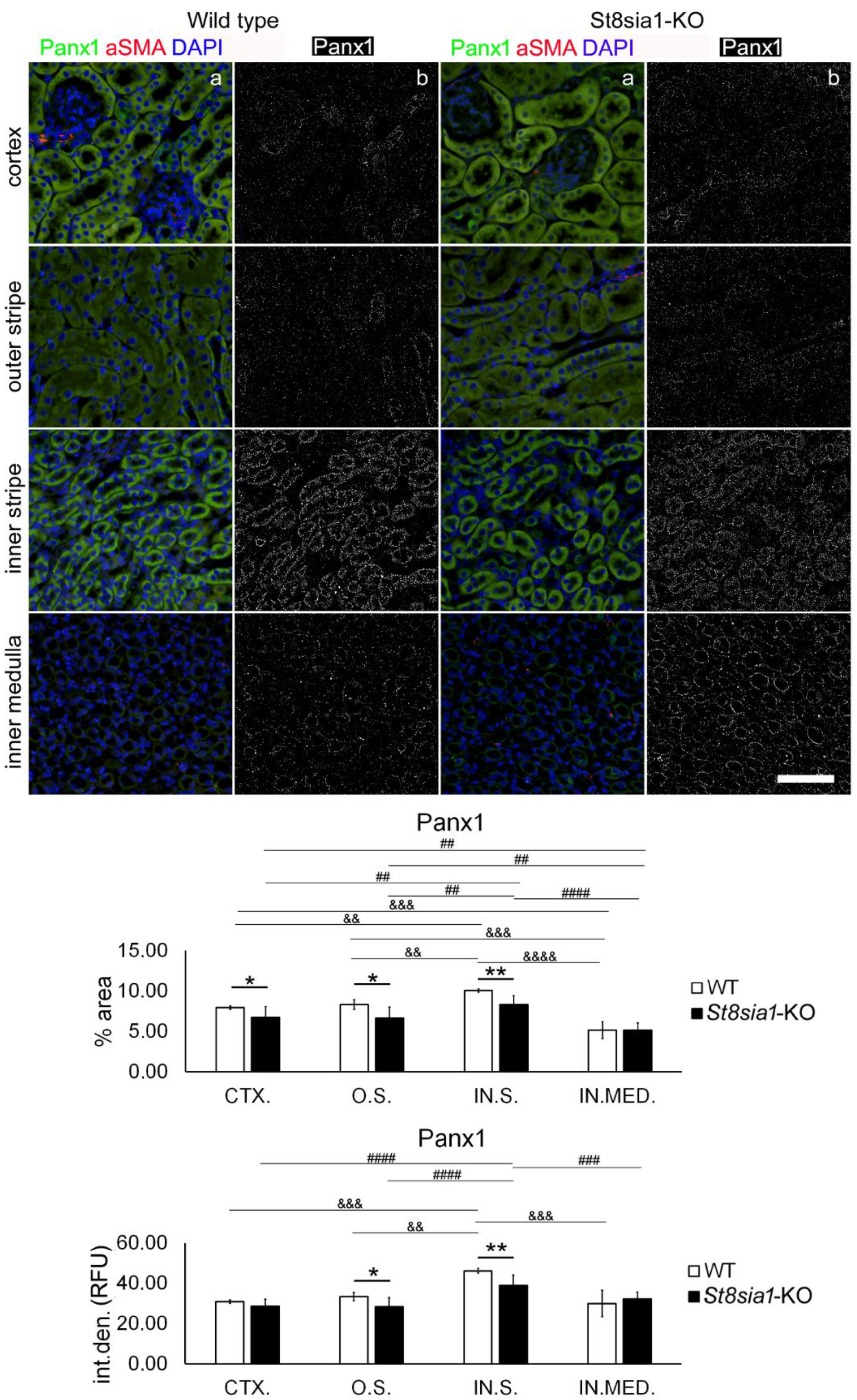
2.5. Expression of Pannexin 1 in the Kidney of Wild-Type and St8sia1-KO Mice
3. Discussion
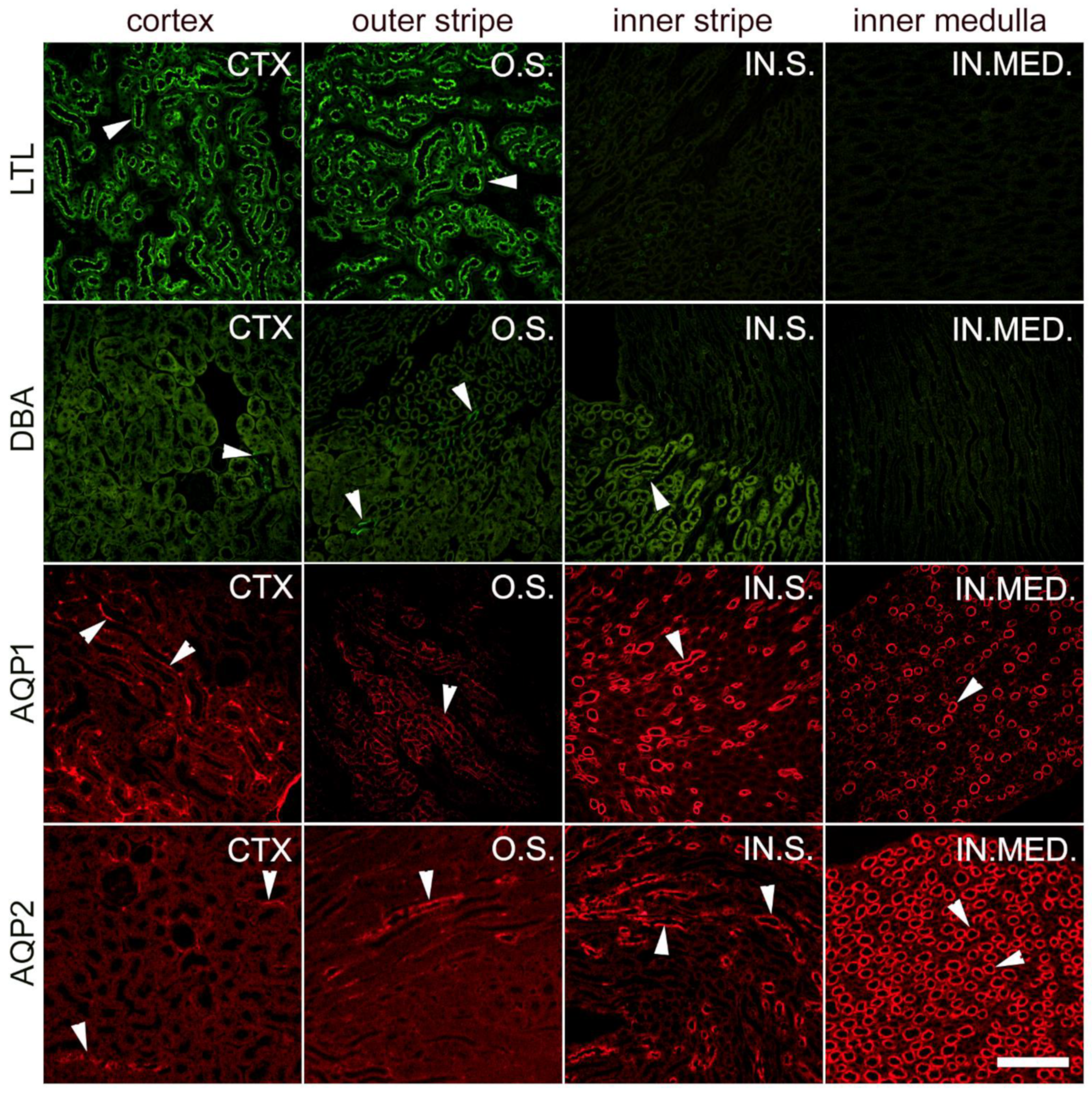
4. Materials and Methods
4.1. Ethics
4.2. Experimental Animals
4.3. Sample Collection and Preparation of Kidney Tissue for Immunohistochemistry
4.4. Immunohistochemistry
4.5. Data Acquisition and Analysis
4.6. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hanner, F.; Sorensen, C.M.; Holstein-Rathlou, N.H.; Peti-Peterdi, J. Connexins and the kidney. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 298, R1143–R1155. [Google Scholar] [CrossRef]
- Wright, J.A.; Richards, T.; Becker, D.L. Connexins and diabetes. Cardiol. Res. Pract. 2012, 2012, 496904. [Google Scholar] [CrossRef] [PubMed]
- Saez, J.C.; Berthoud, V.M.; Branes, M.C.; Martinez, A.D.; Beyer, E.C. Plasma membrane channels formed by connexins: Their regulation and functions. Physiol. Rev. 2003, 83, 1359–1400. [Google Scholar] [CrossRef] [PubMed]
- Evans, W.H.; Martin, P.E. Gap junctions: Structure and function (Review). Mol. Membr. Biol. 2002, 19, 121–136. [Google Scholar] [CrossRef] [PubMed]
- Loewenstein, W.R.; Rose, B. The cell-cell channel in the control of growth. Semin. Cell Biol. 1992, 3, 59–79. [Google Scholar] [CrossRef]
- Trosko, J.E.; Chang, C.C.; Wilson, M.R.; Upham, B.; Hayashi, T.; Wade, M. Gap junctions and the regulation of cellular functions of stem cells during development and differentiation. Methods 2000, 20, 245–264. [Google Scholar] [CrossRef]
- Meda, P. The role of gap junction membrane channels in secretion and hormonal action. J. Bioenerg. Biomembr. 1996, 28, 369–377. [Google Scholar] [CrossRef]
- Mesnil, M.; Rideout, D.; Kumar, N.M.; Gilula, N.B. Non-communicating human and murine carcinoma cells produce alpha 1 gap junction mRNA. Carcinogenesis 1994, 15, 1541–1547. [Google Scholar] [CrossRef]
- Hertzberg, E.L.; Gilula, N.B. Isolation and characterization of gap junctions from rat liver. J. Biol. Chem. 1979, 254, 2138–2147. [Google Scholar] [CrossRef]
- Fleschner, C.R.; Cenedella, R.J. Lipid composition of lens plasma membrane fractions enriched in fiber junctions. J. Lipid Res. 1991, 32, 45–53. [Google Scholar] [CrossRef]
- Cascio, M. Connexins and their environment: Effects of lipids composition on ion channels. Biochim. Biophys. Acta 2005, 1711, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef] [PubMed]
- Ohmi, Y.; Ohkawa, Y.; Yamauchi, Y.; Tajima, O.; Furukawa, K. Essential roles of gangliosides in the formation and maintenance of membrane microdomains in brain tissues. Neurochem. Res. 2012, 37, 1185–1191. [Google Scholar] [CrossRef]
- Lin, D.; Zhou, J.; Zelenka, P.S.; Takemoto, D.J. Protein kinase Cgamma regulation of gap junction activity through caveolin-1-containing lipid rafts. Investig. Ophthalmol. Vis. Sci. 2003, 44, 5259–5268. [Google Scholar] [CrossRef] [PubMed]
- Schubert, A.L.; Schubert, W.; Spray, D.C.; Lisanti, M.P. Connexin family members target to lipid raft domains and interact with caveolin-1. Biochemistry 2002, 41, 5754–5764. [Google Scholar] [CrossRef] [PubMed]
- Bathori, G.; Cervenak, L.; Karadi, I. Caveolae—An alternative endocytotic pathway for targeted drug delivery. Crit. Rev. Ther. Drug Carr. Syst. 2004, 21, 67–95. [Google Scholar] [CrossRef] [PubMed]
- Locke, D.; Liu, J.; Harris, A.L. Lipid rafts prepared by different methods contain different connexin channels, but gap junctions are not lipid rafts. Biochemistry 2005, 44, 13027–13042. [Google Scholar] [CrossRef] [PubMed]
- Belluardo, N.; Trovato-Salinaro, A.; Mudo, G.; Hurd, Y.L.; Condorelli, D.F. Structure, chromosomal localization, and brain expression of human Cx36 gene. J. Neurosci. Res. 1999, 57, 740–752. [Google Scholar] [CrossRef]
- Common, J.E.; O’Toole, E.A.; Leigh, I.M.; Thomas, A.; Griffiths, W.A.; Venning, V.; Grabczynska, S.; Peris, Z.; Kansky, A.; Kelsell, D.P. Clinical and genetic heterogeneity of erythrokeratoderma variabilis. J. Investig. Dermatol. 2005, 125, 920–927. [Google Scholar] [CrossRef]
- Dekel, N.; Lawrence, T.S.; Gilula, N.B.; Beers, W.H. Modulation of cell-to-cell communication in the cumulus-oocyte complex and the regulation of oocyte maturation by LH. Dev. Biol. 1981, 86, 356–362. [Google Scholar] [CrossRef]
- Sohl, G.; Odermatt, B.; Maxeiner, S.; Degen, J.; Willecke, K. New insights into the expression and function of neural connexins with transgenic mouse mutants. Brain Res. Rev. 2004, 47, 245–259. [Google Scholar] [CrossRef] [PubMed]
- Penuela, S.; Gehi, R.; Laird, D.W. The biochemistry and function of pannexin channels. Biochim. Biophys. Acta 2013, 1828, 15–22. [Google Scholar] [CrossRef] [PubMed]
- Sandilos, J.K.; Bayliss, D.A. Physiological mechanisms for the modulation of pannexin 1 channel activity. J. Physiol. 2012, 590, 6257–6266. [Google Scholar] [CrossRef] [PubMed]
- Billaud, M.; Sandilos, J.K.; Isakson, B.E. Pannexin 1 in the regulation of vascular tone. Trends Cardiovasc. Med. 2012, 22, 68–72. [Google Scholar] [CrossRef]
- Chekeni, F.B.; Elliott, M.R.; Sandilos, J.K.; Walk, S.F.; Kinchen, J.M.; Lazarowski, E.R.; Armstrong, A.J.; Penuela, S.; Laird, D.W.; Salvesen, G.S.; et al. Pannexin 1 channels mediate ‘find-me’ signal release and membrane permeability during apoptosis. Nature 2010, 467, 863–867. [Google Scholar] [CrossRef]
- Sridharan, M.; Adderley, S.P.; Bowles, E.A.; Egan, T.M.; Stephenson, A.H.; Ellsworth, M.L.; Sprague, R.S. Pannexin 1 is the conduit for low oxygen tension-induced ATP release from human erythrocytes. Am. J. Physiol. Heart Circ. Physiol. 2010, 299, H1146–H1152. [Google Scholar] [CrossRef]
- Adamson, S.E.; Meher, A.K.; Chiu, Y.H.; Sandilos, J.K.; Oberholtzer, N.P.; Walker, N.N.; Hargett, S.R.; Seaman, S.A.; Peirce-Cottler, S.M.; Isakson, B.E.; et al. Pannexin 1 is required for full activation of insulin-stimulated glucose uptake in adipocytes. Mol. Metab. 2015, 4, 610–618. [Google Scholar] [CrossRef]
- Lopez, P.H.; Schnaar, R.L. Gangliosides in cell recognition and membrane protein regulation. Curr. Opin. Struct. Biol. 2009, 19, 549–557. [Google Scholar] [CrossRef]
- Toledo, M.S.; Suzuki, E.; Handa, K.; Hakomori, S. Effect of ganglioside and tetraspanins in microdomains on interaction of integrins with fibroblast growth factor receptor. J. Biol. Chem. 2005, 280, 16227–16234. [Google Scholar] [CrossRef]
- Yu, R.K.; Tsai, Y.T.; Ariga, T.; Yanagisawa, M. Structures, biosynthesis, and functions of gangliosides—An overview. J. Oleo Sci. 2011, 60, 537–544. [Google Scholar] [CrossRef]
- Matarrese, P.; Garofalo, T.; Manganelli, V.; Gambardella, L.; Marconi, M.; Grasso, M.; Tinari, A.; Misasi, R.; Malorni, W.; Sorice, M. Evidence for the involvement of GD3 ganglioside in autophagosome formation and maturation. Autophagy 2014, 10, 750–765. [Google Scholar] [CrossRef] [PubMed]
- Garofalo, T.; Matarrese, P.; Manganelli, V.; Marconi, M.; Tinari, A.; Gambardella, L.; Faggioni, A.; Misasi, R.; Sorice, M.; Malorni, W. Evidence for the involvement of lipid rafts localized at the ER-mitochondria associated membranes in autophagosome formation. Autophagy 2016, 12, 917–935. [Google Scholar] [CrossRef] [PubMed]
- Yu, R.K.; Bieberich, E.; Xia, T.; Zeng, G. Regulation of ganglioside biosynthesis in the nervous system. J. Lipid Res. 2004, 45, 783–793. [Google Scholar] [CrossRef] [PubMed]
- Sturgill, E.R.; Aoki, K.; Lopez, P.H.; Colacurcio, D.; Vajn, K.; Lorenzini, I.; Majic, S.; Yang, W.H.; Heffer, M.; Tiemeyer, M.; et al. Biosynthesis of the major brain gangliosides GD1a and GT1b. Glycobiology 2012, 22, 1289–1301. [Google Scholar] [CrossRef] [PubMed]
- Mlinac, K.; Fon Tacer, K.; Heffer, M.; Rozman, D.; Bognar, S.K. Cholesterogenic genes expression in brain and liver of ganglioside-deficient mice. Mol. Cell. Biochem. 2012, 369, 127–133. [Google Scholar] [CrossRef]
- Okada, M.; Itoh Mi, M.; Haraguchi, M.; Okajima, T.; Inoue, M.; Oishi, H.; Matsuda, Y.; Iwamoto, T.; Kawano, T.; Fukumoto, S.; et al. b-series Ganglioside deficiency exhibits no definite changes in the neurogenesis and the sensitivity to Fas-mediated apoptosis but impairs regeneration of the lesioned hypoglossal nerve. J. Biol. Chem. 2002, 277, 1633–1636. [Google Scholar] [CrossRef]
- Prokazova, N.V.; Samovilova, N.N.; Gracheva, E.V.; Golovanova, N.K. Ganglioside GM3 and its biological functions. Biochemistry 2009, 74, 235–249. [Google Scholar] [CrossRef]
- Novak, A.; Rezic Muzinic, N.; Cikes Culic, V.; Bozic, J.; Ticinovic Kurir, T.; Ferhatovic, L.; Puljak, L.; Markotic, A. Renal distribution of ganglioside GM3 in rat models of types 1 and 2 diabetes. J. Physiol. Biochem. 2013, 69, 727–735. [Google Scholar] [CrossRef]
- Savas, B.; Astarita, G.; Aureli, M.; Sahali, D.; Ollero, M. Gangliosides in Podocyte Biology and Disease. Int. J. Mol. Sci. 2020, 21, 9645. [Google Scholar] [CrossRef]
- Vukovic, I.; Bozic, J.; Markotic, A.; Ljubicic, S.; Ticinovic Kurir, T. The missing link—Likely pathogenetic role of GM3 and other gangliosides in the development of diabetic nephropathy. Kidney Blood Press. Res. 2015, 40, 306–314. [Google Scholar] [CrossRef]
- Yamashita, T.; Hashiramoto, A.; Haluzik, M.; Mizukami, H.; Beck, S.; Norton, A.; Kono, M.; Tsuji, S.; Daniotti, J.L.; Werth, N.; et al. Enhanced insulin sensitivity in mice lacking ganglioside GM3. Proc. Natl. Acad. Sci. USA 2003, 100, 3445–3449. [Google Scholar] [CrossRef] [PubMed]
- Tagami, S.; Inokuchi Ji, J.; Kabayama, K.; Yoshimura, H.; Kitamura, F.; Uemura, S.; Ogawa, C.; Ishii, A.; Saito, M.; Ohtsuka, Y.; et al. Ganglioside GM3 participates in the pathological conditions of insulin resistance. J. Biol. Chem. 2002, 277, 3085–3092. [Google Scholar] [CrossRef] [PubMed]
- Fox, T.E.; Kester, M. Therapeutic strategies for diabetes and complications: A role for sphingolipids? Adv. Exp. Med. Biol. 2010, 688, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Masson, E.; Troncy, L.; Ruggiero, D.; Wiernsperger, N.; Lagarde, M.; El Bawab, S. a-Series gangliosides mediate the effects of advanced glycation end products on pericyte and mesangial cell proliferation: A common mediator for retinal and renal microangiopathy? Diabetes 2005, 54, 220–227. [Google Scholar] [CrossRef][Green Version]
- Andrews, P.M. Glomerular epithelial alterations resulting from sialic acid surface coat removal. Kidney Int. 1979, 15, 376–385. [Google Scholar] [CrossRef]
- Kumaran, G.K.; Hanukoglu, I. Identification and classification of epithelial cells in nephron segments by actin cytoskeleton patterns. FEBS J. 2020, 287, 1176–1194. [Google Scholar] [CrossRef]
- Stoessel, A.; Himmerkus, N.; Bleich, M.; Bachmann, S.; Theilig, F. Connexin 37 is localized in renal epithelia and responds to changes in dietary salt intake. Am. J. Physiol. Ren. Physiol. 2010, 298, F216–F223. [Google Scholar] [CrossRef]
- Zhang, J.; Hill, C.E. Differential connexin expression in preglomerular and postglomerular vasculature: Accentuation during diabetes. Kidney Int. 2005, 68, 1171–1185. [Google Scholar] [CrossRef][Green Version]
- Geis, L.; Boudriot, F.F.; Wagner, C. Connexin mRNA distribution in adult mouse kidneys. Pflugers Arch. 2021, 473, 1737–1747. [Google Scholar] [CrossRef]
- Hwan Seul, K.; Beyer, E.C. Heterogeneous localization of connexin40 in the renal vasculature. Microvasc. Res. 2000, 59, 140–148. [Google Scholar] [CrossRef]
- Silverstein, D.M.; Thornhill, B.A.; Leung, J.C.; Vehaskari, V.M.; Craver, R.D.; Trachtman, H.A.; Chevalier, R.L. Expression of connexins in the normal and obstructed developing kidney. Pediatr. Nephrol. 2003, 18, 216–224. [Google Scholar] [CrossRef] [PubMed]
- Kundu, S.; Pushpakumar, S.B.; Tyagi, A.; Coley, D.; Sen, U. Hydrogen sulfide deficiency and diabetic renal remodeling: Role of matrix metalloproteinase-9. Am. J. Physiol. Endocrinol. Metab. 2013, 304, E1365–E1378. [Google Scholar] [CrossRef] [PubMed]
- Luetic, M.; Vitlov Uljevic, M.; Masek, T.; Benzon, B.; Vukojevic, K.; Filipovic, N. PUFAs supplementation affects the renal expression of pannexin 1 and connexins in diabetic kidney of rats. Histochem. Cell Biol. 2020, 153, 165–175. [Google Scholar] [CrossRef] [PubMed]
- Sawai, K.; Mukoyama, M.; Mori, K.; Yokoi, H.; Koshikawa, M.; Yoshioka, T.; Takeda, R.; Sugawara, A.; Kuwahara, T.; Saleem, M.A.; et al. Redistribution of connexin43 expression in glomerular podocytes predicts poor renal prognosis in patients with type 2 diabetes and overt nephropathy. Nephrol. Dial. Transplant. 2006, 21, 2472–2477. [Google Scholar] [CrossRef]
- Barajas, L.; Liu, L.; Tucker, M. Localization of connexin43 in rat kidney. Kidney Int. 1994, 46, 621–626. [Google Scholar] [CrossRef]
- Guo, R.; Liu, L.; Barajas, L. RT-PCR study of the distribution of connexin 43 mRNA in the glomerulus and renal tubular segments. Am. J. Physiol. 1998, 275, R439–R447. [Google Scholar] [CrossRef]
- Butterweck, A.; Gergs, U.; Elfgang, C.; Willecke, K.; Traub, O. Immunochemical characterization of the gap junction protein connexin45 in mouse kidney and transfected human HeLa cells. J. Membr. Biol. 1994, 141, 247–256. [Google Scholar] [CrossRef]
- Vergara, L.; Bao, X.; Cooper, M.; Bello-Reuss, E.; Reuss, L. Gap-junctional hemichannels are activated by ATP depletion in human renal proximal tubule cells. J. Membr. Biol. 2003, 196, 173–184. [Google Scholar] [CrossRef]
- Vergara, L.; Bao, X.; Bello-Reuss, E.; Reuss, L. Do connexin 43 gap-junctional hemichannels activate and cause cell damage during ATP depletion of renal-tubule cells? Acta Physiol. Scand. 2003, 179, 33–38. [Google Scholar] [CrossRef]
- Just, A.; Kurtz, L.; de Wit, C.; Wagner, C.; Kurtz, A.; Arendshorst, W.J. Connexin 40 mediates the tubuloglomerular feedback contribution to renal blood flow autoregulation. J. Am. Soc. Nephrol. JASN 2009, 20, 1577–1585. [Google Scholar] [CrossRef]
- Hanner, F.; von Maltzahn, J.; Maxeiner, S.; Toma, I.; Sipos, A.; Kruger, O.; Willecke, K.; Peti-Peterdi, J. Connexin45 is expressed in the juxtaglomerular apparatus and is involved in the regulation of renin secretion and blood pressure. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 295, R371–R380. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Luetic, M.; Kretzschmar, G.; Grobe, M.; Jercic, L.; Bota, I.; Ivic, V.; Balog, M.; Zjalic, M.; Vitlov Uljevic, M.; Heffer, M.; et al. Sex-specific effects of metformin and liraglutide on renal pathology and expression of connexin 45 and pannexin 1 following long-term high-fat high-sugar diet. Acta Histochem. 2021, 123, 151817. [Google Scholar] [CrossRef] [PubMed]
- Lozic, M.; Filipovic, N.; Juric, M.; Kosovic, I.; Benzon, B.; Solic, I.; Kelam, N.; Racetin, A.; Watanabe, K.; Katsuyama, Y.; et al. Alteration of Cx37, Cx40, Cx43, Cx45, Panx1, and Renin Expression Patterns in Postnatal Kidneys of Dab1-/- (yotari) Mice. Int. J. Mol. Sci. 2021, 22, 1284. [Google Scholar] [CrossRef]
- Hanner, F.; Lam, L.; Nguyen, M.T.; Yu, A.; Peti-Peterdi, J. Intrarenal localization of the plasma membrane ATP channel pannexin1. Am. J. Physiol. Ren. Physiol. 2012, 303, F1454–F1459. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Scemes, E.; Suadicani, S.O.; Dahl, G.; Spray, D.C. Connexin and pannexin mediated cell-cell communication. Neuron Glia Biol. 2007, 3, 199–208. [Google Scholar] [CrossRef]
- Barbe, M.T.; Monyer, H.; Bruzzone, R. Cell-cell communication beyond connexins: The pannexin channels. Physiology 2006, 21, 103–114. [Google Scholar] [CrossRef]
- Ransford, G.A.; Fregien, N.; Qiu, F.; Dahl, G.; Conner, G.E.; Salathe, M. Pannexin 1 contributes to ATP release in airway epithelia. Am. J. Respir. Cell Mol. Biol. 2009, 41, 525–534. [Google Scholar] [CrossRef]
- Cui, H.; Liu, Y.; Qin, L.; Wang, L.; Huang, Y. Increased membrane localization of pannexin1 in human corneal synaptosomes causes enhanced stimulated ATP release in chronic diabetes mellitus. Medicine 2016, 95, e5084. [Google Scholar] [CrossRef]
- Willebrords, J.; Maes, M.; Crespo Yanguas, S.; Vinken, M. Inhibitors of connexin and pannexin channels as potential therapeutics. Pharmacol. Ther. 2017, 180, 144–160. [Google Scholar] [CrossRef]
- Qu, Y.; Misaghi, S.; Newton, K.; Gilmour, L.L.; Louie, S.; Cupp, J.E.; Dubyak, G.R.; Hackos, D.; Dixit, V.M. Pannexin-1 is required for ATP release during apoptosis but not for inflammasome activation. J. Immunol. 2011, 186, 6553–6561. [Google Scholar] [CrossRef]
- Aasen, T.; Leithe, E.; Graham, S.V.; Kameritsch, P.; Mayan, M.D.; Mesnil, M.; Pogoda, K.; Tabernero, A. Connexins in cancer: Bridging the gap to the clinic. Oncogene 2019, 38, 4429–4451. [Google Scholar] [CrossRef] [PubMed]
- Alexaki, A.; Gupta, S.D.; Majumder, S.; Kono, M.; Tuymetova, G.; Harmon, J.M.; Dunn, T.M.; Proia, R.L. Autophagy regulates sphingolipid levels in the liver. J. Lipid Res. 2014, 55, 2521–2531. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A.; Yang, D.S. Autophagy failure in Alzheimer’s disease—Locating the primary defect. Neurobiol. Dis. 2011, 43, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Nixon, R.A. The role of autophagy in neurodegenerative disease. Nat. Med. 2013, 19, 983–997. [Google Scholar] [CrossRef]
- Sasseville, M.; Gagnon, M.C.; Guillemette, C.; Sullivan, R.; Gilchrist, R.B.; Richard, F.J. Regulation of gap junctions in porcine cumulus-oocyte complexes: Contributions of granulosa cell contact, gonadotropins, and lipid rafts. Mol. Endocrinol. 2009, 23, 700–710. [Google Scholar] [CrossRef]
- Penuela, S.; Bhalla, R.; Gong, X.Q.; Cowan, K.N.; Celetti, S.J.; Cowan, B.J.; Bai, D.; Shao, Q.; Laird, D.W. Pannexin 1 and pannexin 3 are glycoproteins that exhibit many distinct characteristics from the connexin family of gap junction proteins. J. Cell Sci. 2007, 120, 3772–3783. [Google Scholar] [CrossRef]
- Gehi, R.; Shao, Q.; Laird, D.W. Pathways regulating the trafficking and turnover of pannexin1 protein and the role of the C-terminal domain. J. Biol. Chem. 2011, 286, 27639–27653. [Google Scholar] [CrossRef]
- Boassa, D.; Qiu, F.; Dahl, G.; Sosinsky, G. Trafficking dynamics of glycosylated pannexin 1 proteins. Cell Commun. Adhes. 2008, 15, 119–132. [Google Scholar] [CrossRef]
- Saito, M.; Sugiyama, K. Gangliosides in rat kidney: Composition, distribution, and developmental changes. Arch. Biochem. Biophys. 2001, 386, 11–16. [Google Scholar] [CrossRef]
- Futerman, A.H.; Hannun, Y.A. The complex life of simple sphingolipids. EMBO Rep. 2004, 5, 777–782. [Google Scholar] [CrossRef]
- O’Connell, K.M.; Martens, J.R.; Tamkun, M.M. Localization of ion channels to lipid Raft domains within the cardiovascular system. Trends Cardiovasc. Med. 2004, 14, 37–42. [Google Scholar] [CrossRef] [PubMed]
- Defamie, N.; Mesnil, M. The modulation of gap-junctional intercellular communication by lipid rafts. Biochim. Biophys. Acta 2012, 1818, 1866–1869. [Google Scholar] [CrossRef] [PubMed]
| Antibody | Code No. | Host | Dilution | Source | |
|---|---|---|---|---|---|
| Primary | Anti-Cx37/GJA4 | ab181701 | Rabbit | 1:100 | Abcam, Cambridge, UK |
| Anti-Cx40/GJA5 | ab213688 | Rabbit | 1:100 | Abcam, Cambridge, UK | |
| Anti-Cx43/GJA1 | ab87645 | goat | 1:100 | Abcam, Cambridge, UK | |
| Anti-Connexin 45/GJA7/Cx45 | ab135474 | Rabbit | 1:100 | Abcam, Cambridge, UK | |
| Anti-pannexin 1/PANX1 | ABN242 | Rabbit | 1:100 | Merck KGaA, Darmstadt, Germany | |
| Anti-Aquaporin 1/AQP1 (B-11) | sc-25287 | Mouse | 1:50 | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | |
| Anti-Aquaporin 2/AQP2 (E-2) | sc-515770 | Mouse | 1:50 | Santa Cruz Biotechnology Inc., Santa Cruz, CA, USA | |
| Anti-Smooth Muscle Actin | M0851 | Mouse | 1:300 | Dako, Glostrup, Denmark | |
| Lectins | Fluorescein labeled Dolichos Biflorus agglutinin (DBA) | FL-1031 | - | 1:400 | Vector Laboratories Ltd., Peterborough, UK |
| Fluorescein labeled Lotus Tetragonolobus lectin (LTL) | FL-1321 | - | 1:400 | Vector Laboratories Ltd., Peterborough, UK | |
| Secondary | Alexa Fluor®488 AffiniPure Anti-Goat lgG (H+L) | 705-545-003 | Donkey | 1:400 | Jackson Immuno Research Laboratories, Inc., Baltimore, PA, USA |
| Alexa Fluor®488 AffiniPure Anti-Rabbit lgG (H+L) | 711-545-152 | Donkey | 1:400 | Jackson Immuno Research Laboratories, Inc., Baltimore, PA, USA | |
| Rhodamine Red™-X (RRX) AffiniPure Anti-Mouse IgG (H+L) | 715-295-151 | Donkey | 1:400 | Jackson Immuno Research Laboratories, Inc., Baltimore, PA, USA | |
| Rhodamine Red™-X (RRX) AffiniPure Donkey Anti-Rabbit IgG (H+L) | 711-295-152 | Donkey | 1:400 | Jackson Immuno Research Laboratories, Inc., Baltimore, PA, USA | |
| Rhodamine Red™-X (RRX) AffiniPure Anti-Goat IgG (H+L) | 705-295-003 | Donkey | 1:400 | Jackson Immuno Research Laboratories, Inc., Baltimore, PA, USA |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Meter, D.; Racetin, A.; Vukojević, K.; Balog, M.; Ivić, V.; Zjalić, M.; Heffer, M.; Filipović, N. A Lack of GD3 Synthase Leads to Impaired Renal Expression of Connexins and Pannexin1 in St8sia1 Knockout Mice. Int. J. Mol. Sci. 2022, 23, 6237. https://doi.org/10.3390/ijms23116237
Meter D, Racetin A, Vukojević K, Balog M, Ivić V, Zjalić M, Heffer M, Filipović N. A Lack of GD3 Synthase Leads to Impaired Renal Expression of Connexins and Pannexin1 in St8sia1 Knockout Mice. International Journal of Molecular Sciences. 2022; 23(11):6237. https://doi.org/10.3390/ijms23116237
Chicago/Turabian StyleMeter, Diana, Anita Racetin, Katarina Vukojević, Marta Balog, Vedrana Ivić, Milorad Zjalić, Marija Heffer, and Natalija Filipović. 2022. "A Lack of GD3 Synthase Leads to Impaired Renal Expression of Connexins and Pannexin1 in St8sia1 Knockout Mice" International Journal of Molecular Sciences 23, no. 11: 6237. https://doi.org/10.3390/ijms23116237
APA StyleMeter, D., Racetin, A., Vukojević, K., Balog, M., Ivić, V., Zjalić, M., Heffer, M., & Filipović, N. (2022). A Lack of GD3 Synthase Leads to Impaired Renal Expression of Connexins and Pannexin1 in St8sia1 Knockout Mice. International Journal of Molecular Sciences, 23(11), 6237. https://doi.org/10.3390/ijms23116237







