Towards 3D Bioprinted Spinal Cord Organoids
Abstract
:1. Introduction
2. Results
2.1. Hydrogel Gelation Time
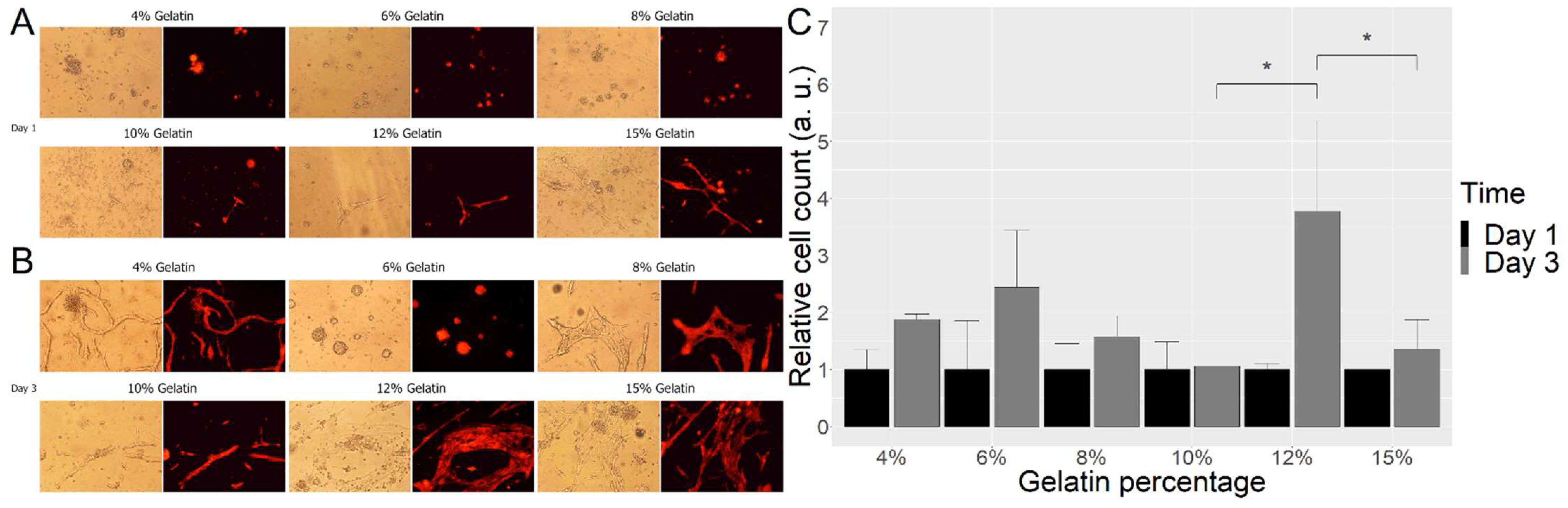
2.2. Cell Survival and Proliferation
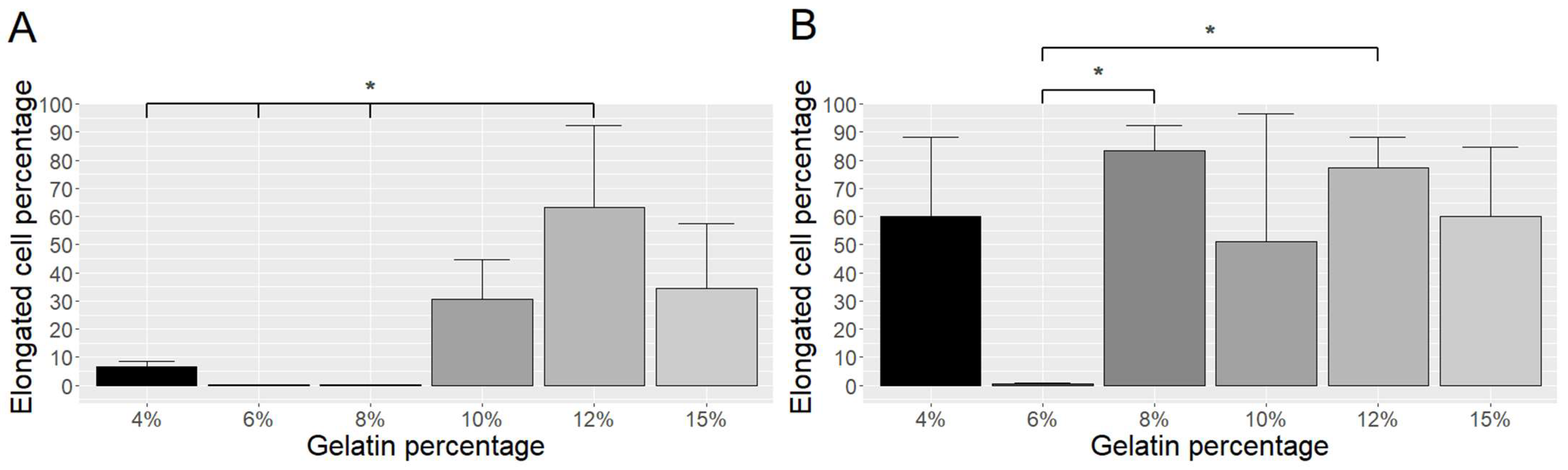
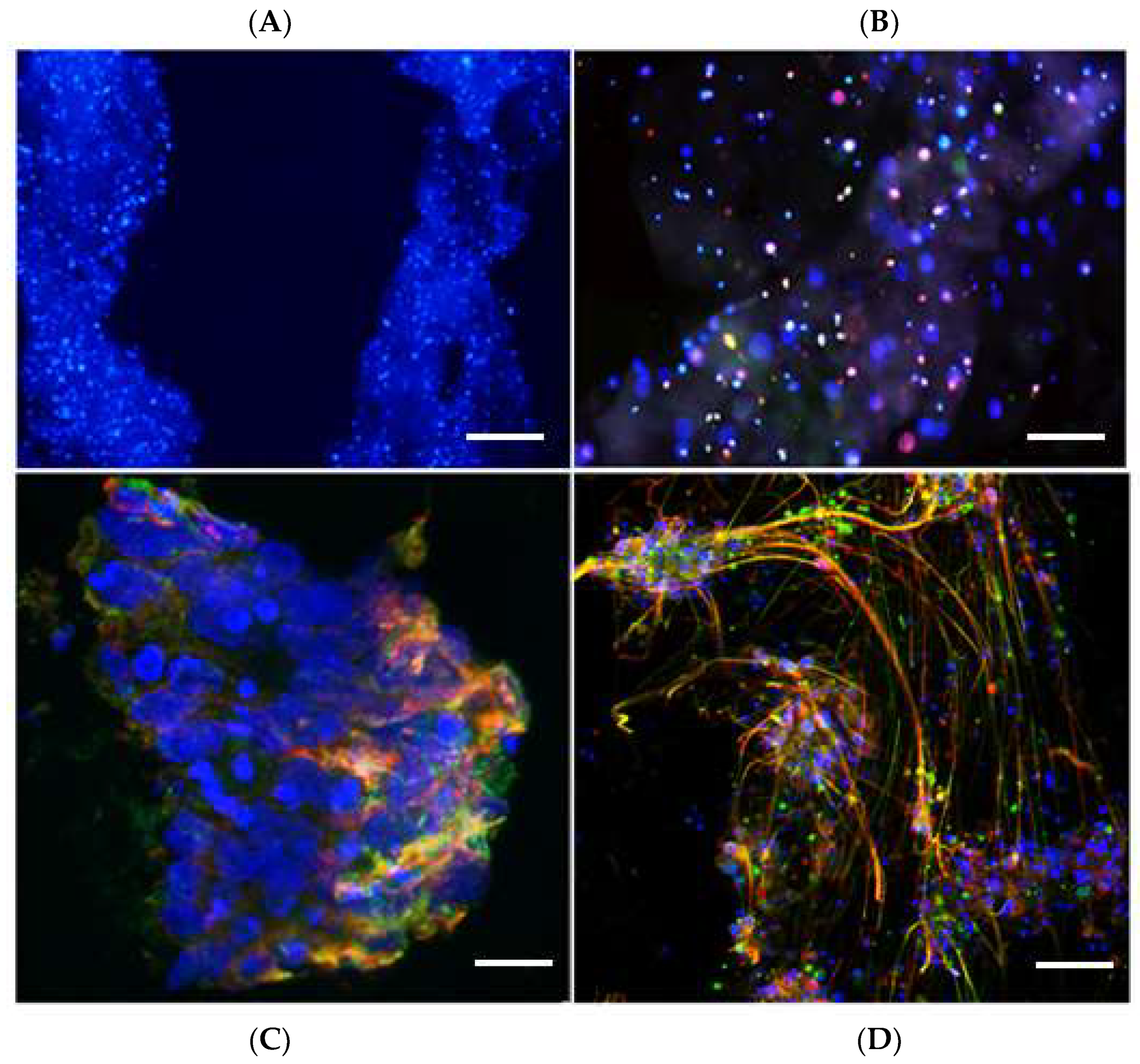
2.3. Cell Differentiation
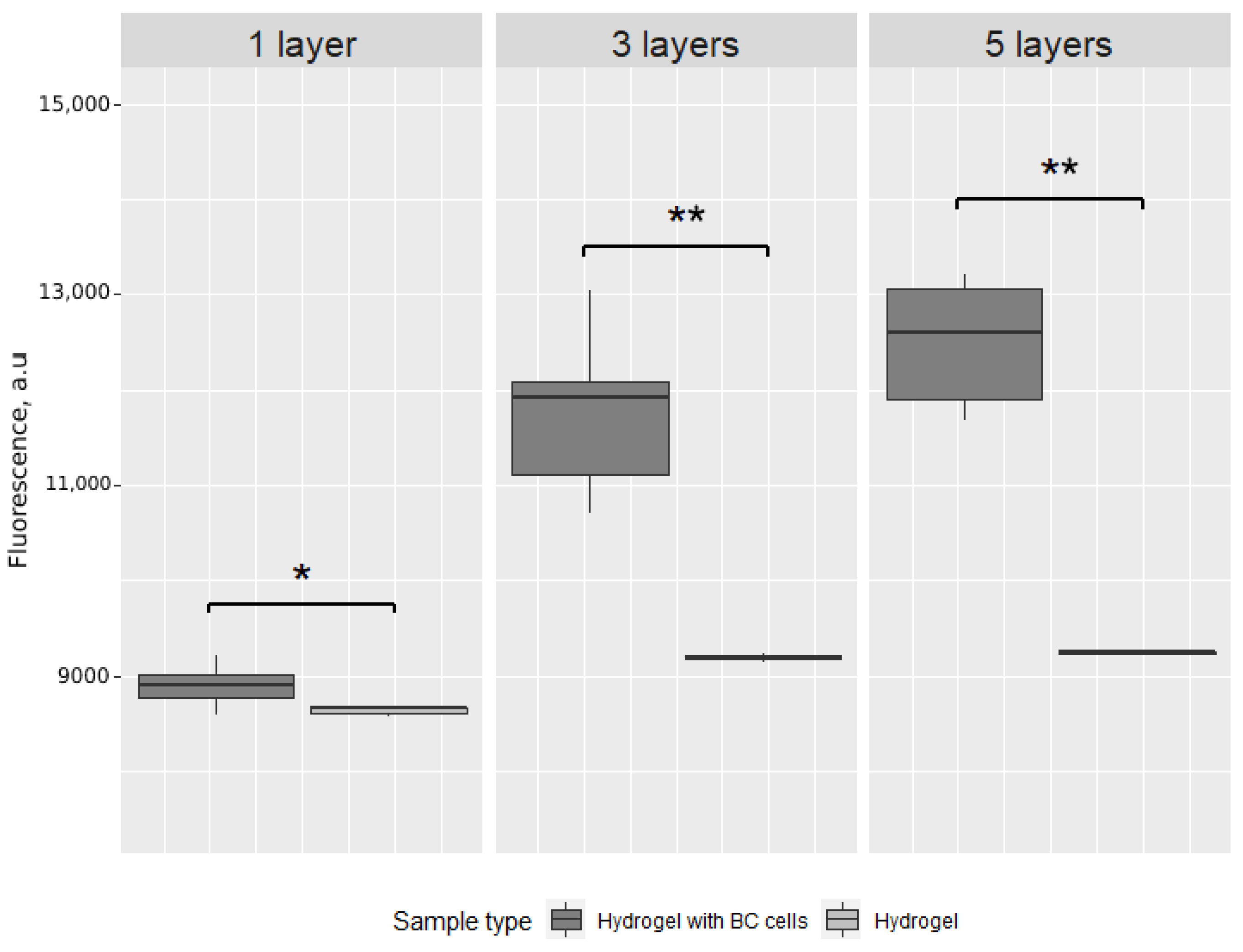
2.4. Alamar Blue Evaluation
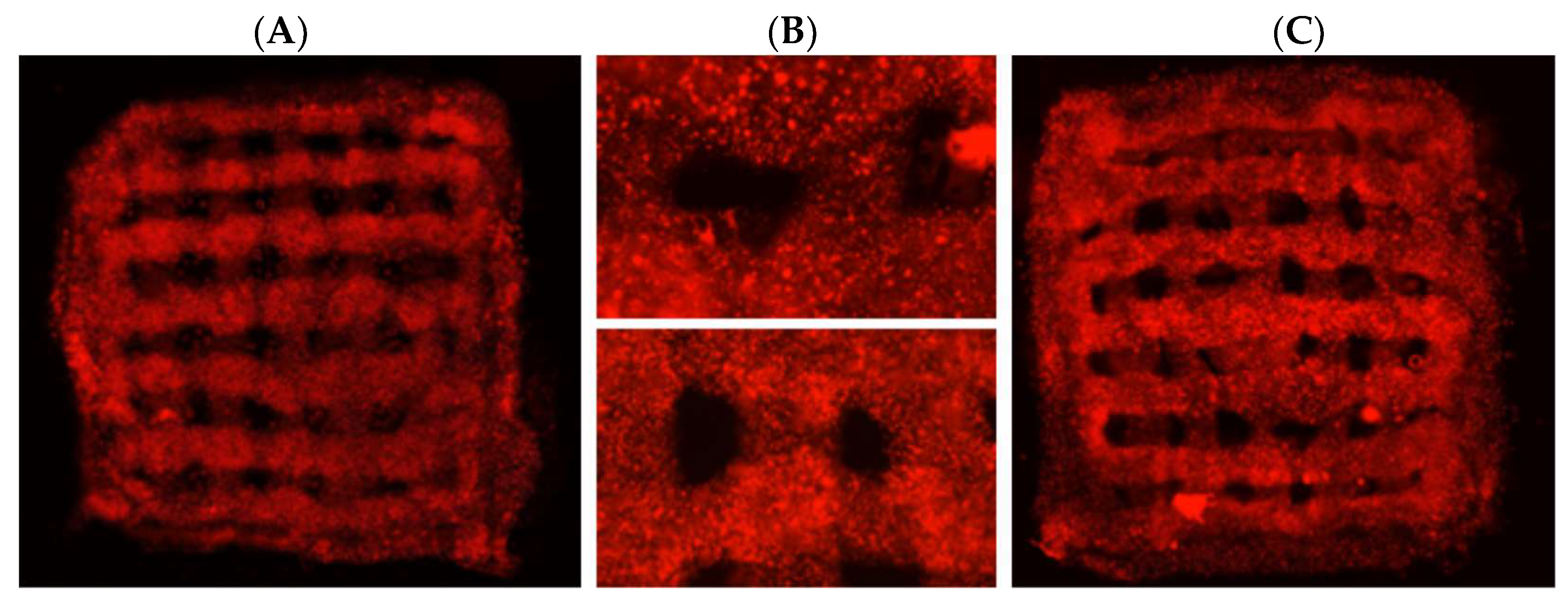
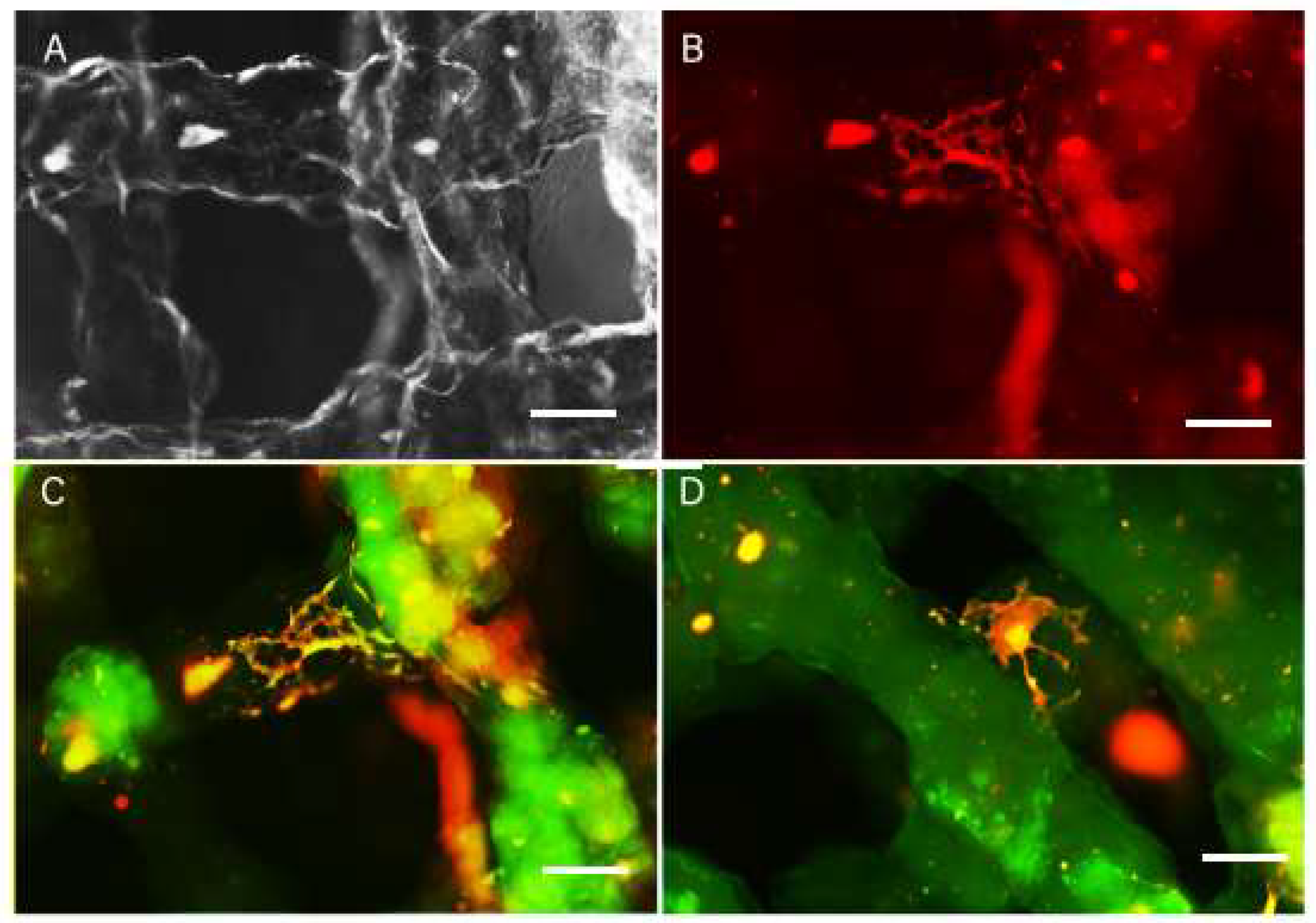
2.5. Cell and Hydrogel Imaging
3. Discussion
4. Materials and Methods
4.1. Cell Culture
4.1.1. Boundary Cap Neural Crest Stem Cell Culture
4.1.2. iPSC-Derived Motor Neuron Cultures
4.1.3. iPSC-Derived Astrocyte Cultures
4.2. Hydrogel Ink Preparation
4.3. 3D Printing of Cells
4.3.1. 3D Printing of BC
4.3.2. 3D Printing of iPSC-Derived Cells
4.4. Cell Differentiation Assay
4.5. Viability Assays
4.6. Immunohistochemistry
4.7. Live Imaging
4.8. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bose, R.; Banerjee, S.; Dunbar, G.L. Modeling Neurological Disorders in 3D Organoids Using Human-Derived Pluripotent Stem Cells. Front. Cell Dev. Biol. 2021, 9, 640212. [Google Scholar] [CrossRef] [PubMed]
- Porciúncula, L.O.; Goto-Silva, L.; Ledur, P.F.; Rehen, S.K. The Age of Brain Organoids: Tailoring Cell Identity and Functionality for Normal Brain Development and Disease Modeling. Front. Neurosci. 2021, 15, 674563. [Google Scholar] [CrossRef] [PubMed]
- Martins, J.-M.F.; Fischer, C.; Urzi, A.; Vidal, R.; Kunz, S.; Ruffault, P.-L.; Kabuss, L.; Hube, I.; Gazzerro, E.; Birchmeier, C.; et al. Self-Organizing 3D Human Trunk Neuromuscular Organoids. Cell Stem Cell 2020, 26, 172–186.e6. [Google Scholar] [CrossRef] [PubMed]
- Andersen, J.; Revah, O.; Miura, Y.; Thom, N.; Amin, N.D.; Kelley, K.W.; Singh, M.; Chen, X.; Thete, M.V.; Walczak, E.M.; et al. Generation of Functional Human 3D Cortico-Motor Assembloids. Cell 2020, 183, 1913–1929.e26. [Google Scholar] [CrossRef]
- Hor, J.-H.; Ng, S.-Y. Generating ventral spinal organoids from human induced pluripotent stem cells. In Methods in Cell Biology; Academic Press: Cambridge, MA, USA, 2020; Volume 159, pp. 257–277. [Google Scholar] [CrossRef]
- Ao, Z.; Cai, H.; Wu, Z.; Krzesniak, J.; Tian, C.; Lai, Y.Y.; Mackie, K.; Guo, F. Human Spinal Organoid-on-a-Chip to Model Nociceptive Circuitry for Pain Therapeutics Discovery. Anal. Chem. 2022, 94, 1365–1372. [Google Scholar] [CrossRef]
- Son, J.; Park, S.J.; Ha, T.; Lee, S.-N.; Cho, H.-Y.; Choi, J.-W. Electrophysiological Monitoring of Neurochemical-Based Neural Signal Transmission in a Human Brain–Spinal Cord Assembloid. ACS Sens. 2022, 7, 409–414. [Google Scholar] [CrossRef]
- Jägers, C.; Roelink, H. Generation and Analysis of Mosaic Spinal Cord Organoids Derived from Mouse Embryonic Stem Cells. In Hedgehog Signaling; Humana: New York, NY, USA, 2022; Volume 2374, pp. 1–11. [Google Scholar] [CrossRef]
- Bron, R.; Vermeren, M.; Kokot, N.; Andrews, W.; Little, G.E.; Mitchell, K.J.; Cohen, J. Boundary cap cells constrain spinal motor neuron somal migration at motor exit points by a semaphorin-plexin mechanism. Neural Dev. 2007, 2, 21. [Google Scholar] [CrossRef] [Green Version]
- Mauti, O.; Domanitskaya, E.; Andermatt, I.; Sadhu, R.; Stoeckli, E.T. Semaphorin6A acts as a gate keeper between the central and the peripheral nervous system. Neural Dev. 2007, 2, 28. [Google Scholar] [CrossRef] [Green Version]
- Leffler, J.H.; Marmigere, F.; Heglind, M.; Cederberg, A.; Koltzenburg, M.; Enerbäck, S.; Ernfors, P. The boundary cap: A source of neural crest stem cells that generate multiple sensory neuron subtypes. Development 2005, 132, 2623–2632. [Google Scholar] [CrossRef] [Green Version]
- Radomska, K.; Topilko, P. Boundary cap cells in development and disease. Curr. Opin. Neurobiol. 2017, 47, 209–215. [Google Scholar] [CrossRef]
- Olerud, J.; Kanaykina, N.; Vasilovska, S.; King, D.; Sandberg, M.; Jansson, L.; Kozlova, E.N. Neural crest stem cells increase beta cell proliferation and improve islet function in co-transplanted murine pancreatic islets. Diabetologia 2009, 52, 2594–2601, Erratum in: Diabetologia 2010, 53, 396. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grouwels, G.; Vasylovska, S.; Olerud, J.; Leuckx, G.; Ngamjariyawat, A.; Yuchi, Y.; Jansson, L.; Van de Casteele, M.; Kozlova, E.N.; Heimberg, H. Differentiating neural crest stem cells induce proliferation of cultured rodent islet beta cells. Diabetologia 2012, 55, 2016–2025. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ngamjariyawat, A.; Turpaev, K.; Welsh, N.; Kozlova, E.N. Coculture of Insulin-Producing RIN5AH Cells With Neural Crest Stem Cells Protects Partially Against Cytokine-Induced Cell Death. Pancreas 2012, 41, 490–492. [Google Scholar] [CrossRef]
- Ngamjariyawat, A.; Turpaev, K.; Vasylovska, S.; Kozlova, E.N.; Welsh, N. Co-Culture of Neural Crest Stem Cells (NCSC) and Insulin Producing Beta-TC6 Cells Results in Cadherin Junctions and Protection against Cytokine-Induced Beta-Cell Death. PLoS ONE 2013, 8, e61828. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lau, J.; Vasylovska, S.; Kozlova, E.; Carlsson, P.-O. Surface Coating of Pancreatic Islets with Neural Crest Stem Cells Improves Engraftment and Function after Intraportal Transplantation. Cell Transplant. 2015, 24, 2263–2272. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Grapensparr, L.; Vasylovska, S.; Li, Z.; Olerud, J.; Jansson, L.; Kozlova, E.; Carlsson, P.-O. Co-transplantation of Human Pancreatic Islets With Post-migratory Neural Crest Stem Cells Increases β-Cell Proliferation and Vascular And Neural Regrowth. J. Clin. Endocrinol. Metab. 2015, 100, E583–E590. [Google Scholar] [CrossRef] [PubMed]
- Aggarwal, T.; Hoeber, J.; Ivert, P.; Vasylovska, S.; Kozlova, E.N. Boundary Cap Neural Crest Stem Cells Promote Survival of Mutant SOD1 Motor Neurons. Neurotherapeutics 2017, 14, 773–783. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schizas, N.; König, N.; Andersson, B.; Vasylovska, S.; Hoeber, J.; Kozlova, E.N.; Hailer, N.P. Neural crest stem cells protect spinal cord neurons from excitotoxic damage and inhibit glial activation by secretion of brain-derived neurotrophic factor. Cell Tissue Res. 2018, 372, 493–5055. [Google Scholar] [CrossRef] [Green Version]
- Leyton-Jaimes, M.F.; Ivert, P.; Hoeber, J.; Han, Y.; Feiler, A.; Zhou, C.; Pankratova, S.; Shoshan-Barmatz, V.; Israelson, A.; Kozlova, E.N. Empty mesoporous silica particles significantly delay disease progression and extend survival in a mouse model of ALS. Sci. Rep. 2020, 10, 20675. [Google Scholar] [CrossRef]
- Han, Y.; Zeger, L.; Tripathi, R.; Egli, M.; Ille, F.; Lockowandt, C.; Florin, G.; Atic, E.; Redwan, I.N.; Fredriksson, R.; et al. Molecular genetic analysis of neural stem cells after space flight and simulated microgravity on earth. Biotechnol. Bioeng. 2021, 118, 3832–3846. [Google Scholar] [CrossRef]
- Billiet, T.; Vandenhaute, M.; Schelfhout, J.; Van Vlierberghe, S.; Dubruel, P. A review of trends and limitations in hydrogel-rapid prototyping for tissue engineering. Biomaterials 2012, 33, 6020–6041. [Google Scholar] [CrossRef] [PubMed]
- Moroni, L.; Burdick, J.A.; Highley, C.; Lee, S.J.; Morimoto, Y.; Takeuchi, S.; Yoo, J.J. Biofabrication strategies for 3D in vitro models and regenerative medicine. Nat. Rev. Mater. 2018, 3, 21–37. [Google Scholar] [CrossRef] [PubMed]
- Levato, R.; Jungst, T.; Scheuring, R.G.; Blunk, T.; Groll, J.; Malda, J. From Shape to Function: The Next Step in Bioprinting. Adv. Mater. 2020, 32, e1906423. [Google Scholar] [CrossRef] [PubMed]
- Guzzi, E.A.; Tibbitt, M.W. Additive Manufacturing of Precision Biomaterials. Adv. Mater. 2020, 32, e1901994. [Google Scholar] [CrossRef] [PubMed]
- Züger, F.; Marsano, A.; Poggio, M.; Gullo, M.R. Nanocomposites in 3D Bioprinting for Engineering Conductive and Stimuli-Responsive Constructs Mimicking Electrically Sensitive Tissue. Adv. NanoBiomed Res. 2021, 2, 2100108. [Google Scholar] [CrossRef]
- Grigoryan, B.; Paulsen, S.J.; Corbett, D.C.; Sazer, D.W.; Fortin, C.L.; Zaita, A.J.; Greenfield, P.T.; Calafat, N.J.; Gounley, J.P.; Ta, A.H.; et al. Multivascular networks and functional intravascular topologies within biocompatible hydrogels. Science 2019, 364, 458–464. [Google Scholar] [CrossRef]
- Kolesky, D.B.; Homan, K.A.; Skylar-Scott, M.A.; Lewis, J.A. Three-dimensional bioprinting of thick vascularized tissues. Proc. Natl. Acad. Sci. USA 2016, 113, 3179–3184. [Google Scholar] [CrossRef] [Green Version]
- Lin, N.Y.C.; Homan, K.A.; Robinson, S.S.; Kolesky, D.B.; Duarte, N.; Moisan, A.; Lewis, J.A. Renal reabsorption in 3D vascularized proximal tubule models. Proc. Natl. Acad. Sci. USA 2019, 116, 5399–5404. [Google Scholar] [CrossRef] [Green Version]
- Han, Y.; Baltriukienė, D.; Kozlova, E.N. Effect of scaffold properties on adhesion and maintenance of boundary cap neural crest stem cells in vitro. J. Biomed. Mater. Res. A 2020, 108, 1274–1280. [Google Scholar] [CrossRef]
- Kačarević, P.; Rider, P.M.; Alkildani, S.; Retnasingh, S.; Smeets, R.; Jung, O.; Ivanišević, Z.; Barbeck, M. An Introduction to 3D Bioprinting: Possibilities, Challenges and Future Aspects. Materials 2018, 11, 2199. [Google Scholar] [CrossRef] [Green Version]
- Esaki, S.; Katsumi, S.; Hamajima, Y.; Nakamura, Y.; Murakami, S. Transplantation of Olfactory Stem Cells with Biodegradable Hydrogel Accelerates Facial Nerve Regeneration After Crush Injury. Stem Cells Transl. Med. 2019, 8, 169–178. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fantini, V.; Bordoni, M.; Scocozza, F.; Conti, M.; Scarian, E.; Carelli, S.; Di Giulio, A.M.; Marconi, S.; Pansarasa, O.; Auricchio, F.; et al. Bioink Composition and Printing Parameters for 3D Modeling Neural Tissue. Cells 2019, 8, 830. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, X.; Zhang, C.; Xu, J.; Zhai, H.; Liu, S.; Xu, Y.; Hu, Y.; Long, H.; Bai, Y.; Quan, D. Neurotrophin-3-Loaded Multichannel Nanofibrous Scaffolds Promoted Anti-Inflammation, Neuronal Differentiation, and Functional Recovery after Spinal Cord Injury. ACS Biomater. Sci. Eng. 2020, 6, 1228–1238. [Google Scholar] [CrossRef] [PubMed]
- Hamid, O.A.; Eltaher, H.M.; Sottile, V.; Yang, J. 3D bioprinting of a stem cell-laden, multi-material tubular composite: An approach for spinal cord repair. Mater. Sci. Eng. C 2021, 120, 111707. [Google Scholar] [CrossRef]
- Hirano, M.; Huang, Y.; Jarquin, D.V.; Hernández, R.L.D.L.G.; Jodat, Y.A.; Cerón, E.L.; García-Rivera, L.E.; Shin, S.R. 3D bioprinted human iPSC-derived somatosensory constructs with functional and highly purified sensory neuron networks. Biofabrication 2021, 13, 035046. [Google Scholar] [CrossRef]
- Huang, B.; Peng, J.; Huang, X.; Liang, F.; Wang, L.; Shi, J.; Yamada, A.; Chen, Y. Generation of Interconnected Neural Clusters in Multiscale Scaffolds from Human-Induced Pluripotent Stem Cells. ACS Appl. Mater. Interfaces 2021, 13, 55939–55952. [Google Scholar] [CrossRef]
- Lee, H.; Han, N.R.; Hwang, J.Y.; Yun, J.I.; Kim, C.; Park, K.H.; Lee, S.T. Gelatin Directly Enhances Neurogenic Differentiation Potential in Bone Marrow-Derived Mesenchymal Stem Cells Without Stimulation of Neural Progenitor Cell Proliferation. DNA Cell Biol. 2016, 35, 530–536. [Google Scholar] [CrossRef]
- Yao, M.; Li, J.; Zhang, J.; Ma, S.; Wang, L.; Gao, F.; Guan, F. Dual-enzymatically cross-linked gelatin hydrogel enhances neural differentiation of human umbilical cord mesenchymal stem cells and functional recovery in experimental murine spinal cord injury. J. Mater. Chem. B 2021, 9, 440–452. [Google Scholar] [CrossRef]
- Li, J.; Zhang, D.; Guo, S.; Zhao, C.; Wang, L.; Ma, S.; Guan, F.; Yao, M. Dual-enzymatically cross-linked gelatin hydrogel promotes neural differentiation and neurotrophin secretion of bone marrow-derived mesenchymal stem cells for treatment of moderate traumatic brain injury. Int. J. Biol. Macromol. 2021, 187, 200–213. [Google Scholar] [CrossRef]
- Aldskogius, H.; Berens, C.; Kanaykina, N.; Liakhovitskaia, A.; Medvinsky, A.; Sandelin, M.; Schreiner, S.; Wegner, M.; Hjerling-Leffler, J.; Kozlova, E.N. Regulation of Boundary Cap Neural Crest Stem Cell Differentiation After Transplantation. Stem Cells 2009, 27, 1592–1603. [Google Scholar] [CrossRef] [Green Version]
- König, N.; Åkesson, E.; Telorack, M.; Vasylovska, S.; Ngamjariyawat, A.; Sundström, E.; Oster, A.; Trolle, C.; Berens, C.; Aldskogius, H.; et al. Forced Runx1 Expression in Human Neural Stem/Progenitor Cells Transplanted to the Rat Dorsal Root Ganglion Cavity Results in Extensive Axonal Growth Specifically from Spinal Cord–Derived Neurospheres. Stem Cells Dev. 2011, 20, 1847–1857. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bennett, A.E.; Kozhevnikova, M.; König, N.; Zhou, C.; Leao, R.; Knöpfel, T.; Pankratova, S.; Trolle, C.; Berezin, V.; Bock, E.; et al. Delivery of Differentiation Factors by Mesoporous Silica Particles Assists Advanced Differentiation of Transplanted Murine Embryonic Stem Cells. STEM CELLS Transl. Med. 2013, 2, 906–915. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Bennett, A.E.; König, N.; Abrahamsson, N.; Kozhevnikova, M.; Zhou, C.; Trolle, C.; Pankratova, S.; Berezin, V.; Kozlova, E.N. In vitro generation of motor neuron precursors from mouse embryonic stem cells using mesoporous nanoparticles. Nanomedicine 2014, 9, 2457–2466. [Google Scholar] [CrossRef] [PubMed]
- Ji, S.; Almeida, E.; Guvendiren, M. 3D bioprinting of complex channels within cell-laden hydrogels. Acta Biomater. 2019, 95, 214–224. [Google Scholar] [CrossRef] [PubMed]
- Skylar-Scott, M.A.; Uzel, S.G.M.; Nam, L.L.; Ahrens, J.H.; Truby, R.L.; Damaraju, S.; Lewis, J.A. Biomanufacturing of organ-specific tissues with high cellular density and embedded vascular channels. Sci. Adv. 2019, 5, eaaw2459. [Google Scholar] [CrossRef] [Green Version]
- Vintersten, K.; Monetti, C.; Gertsenstein, M.; Zhang, P.; Laszlo, L.; Biechele, S.; Nagy, A. Mouse in red: Red fluorescent protein expression in mouse ES cells, embryos, and adult animals. Genesis 2004, 40, 241–246. [Google Scholar] [CrossRef]
- Meyer, K.; Ferraiuolo, L.; Miranda, C.J.; Likhite, S.; McElroy, S.; Renusch, S.; Ditsworth, D.; Lagier-Tourenne, C.; Smith, R.A.; Ravits, J.; et al. Direct conversion of patient fibroblasts demonstrates non-cell autonomous toxicity of astrocytes to motor neurons in familial and sporadic ALS. Proc. Natl. Acad. Sci. USA 2013, 111, 829–832. [Google Scholar] [CrossRef] [Green Version]
- Rampersad, S.N. Multiple Applications of Alamar Blue as an Indicator of Metabolic Function and Cellular Health in Cell Viability Bioassays. Sensors 2012, 12, 12347–12360. [Google Scholar] [CrossRef]

| Gelatin Percentage | ||||||
|---|---|---|---|---|---|---|
| mTG Concentration | 4% | 6% | 8% | 10% | 12% | 15% |
| 15 mg/mL | >2 h | >2 h | >2 h | 49.0 min | 32.0 min | 24.0 min |
| 20 mg/mL | >2 h | >2 h | 1.8 h | 47.3 min | 30.0 min | 22.3 min |
| 25 mg/mL | 1.7 h | 1.4 h | 1.2 h | 42.0 min | 27.0 min | 17.0 min |
| 30 mg/mL | 1.7 h | 1.3 h | 1 h | 38.3 min | 19.0 min | 6.3 min |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Han, Y.; King, M.; Tikhomirov, E.; Barasa, P.; Souza, C.D.S.; Lindh, J.; Baltriukiene, D.; Ferraiuolo, L.; Azzouz, M.; Gullo, M.R.; et al. Towards 3D Bioprinted Spinal Cord Organoids. Int. J. Mol. Sci. 2022, 23, 5788. https://doi.org/10.3390/ijms23105788
Han Y, King M, Tikhomirov E, Barasa P, Souza CDS, Lindh J, Baltriukiene D, Ferraiuolo L, Azzouz M, Gullo MR, et al. Towards 3D Bioprinted Spinal Cord Organoids. International Journal of Molecular Sciences. 2022; 23(10):5788. https://doi.org/10.3390/ijms23105788
Chicago/Turabian StyleHan, Yilin, Marianne King, Evgenii Tikhomirov, Povilas Barasa, Cleide Dos Santos Souza, Jonas Lindh, Daiva Baltriukiene, Laura Ferraiuolo, Mimoun Azzouz, Maurizio R. Gullo, and et al. 2022. "Towards 3D Bioprinted Spinal Cord Organoids" International Journal of Molecular Sciences 23, no. 10: 5788. https://doi.org/10.3390/ijms23105788
APA StyleHan, Y., King, M., Tikhomirov, E., Barasa, P., Souza, C. D. S., Lindh, J., Baltriukiene, D., Ferraiuolo, L., Azzouz, M., Gullo, M. R., & Kozlova, E. N. (2022). Towards 3D Bioprinted Spinal Cord Organoids. International Journal of Molecular Sciences, 23(10), 5788. https://doi.org/10.3390/ijms23105788







