Repercussions of Bisphenol A on the Physiology of Human Osteoblasts
Abstract
:1. Introduction
2. Results
2.1. Effect of BPA on Growth and Cellular Viability
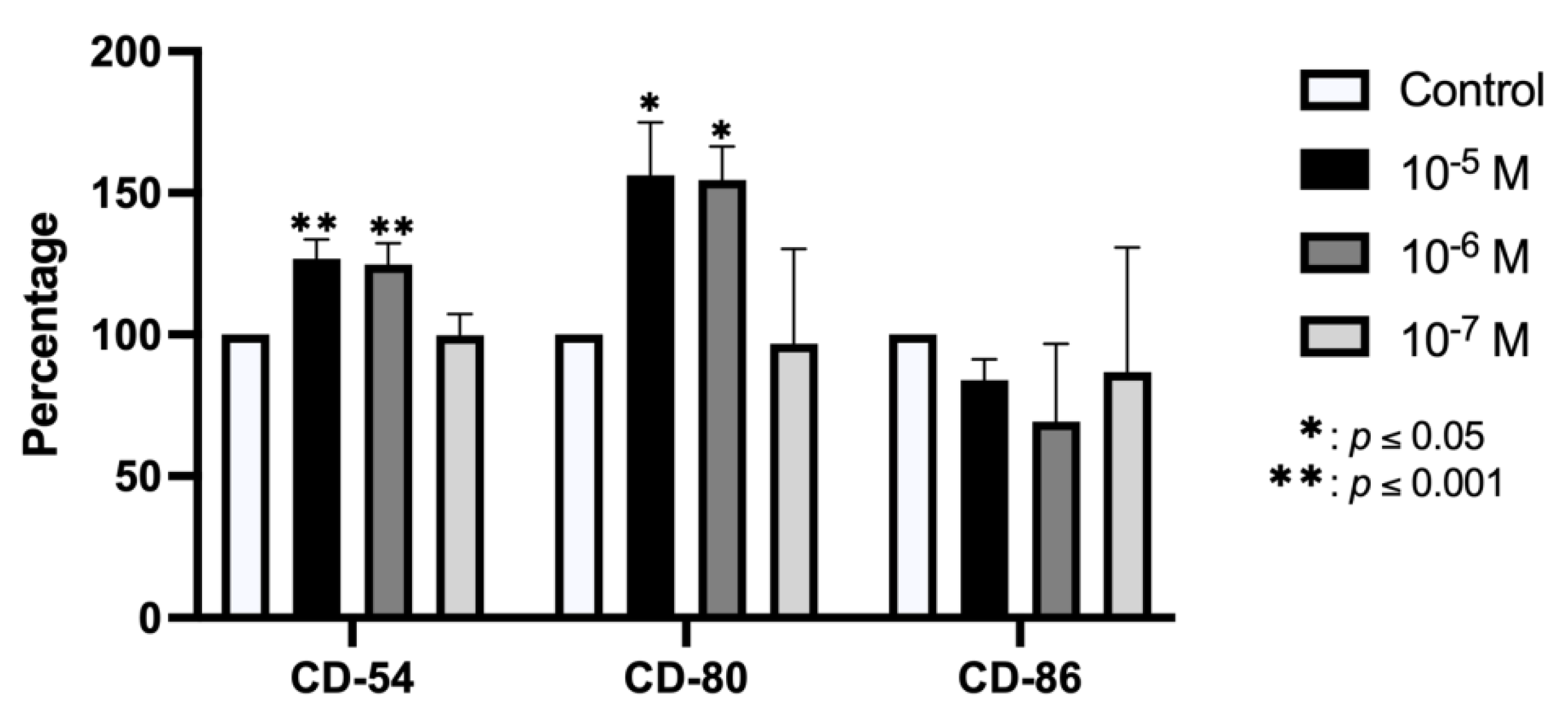
2.2. Effect of BPA on Antigenic Profile
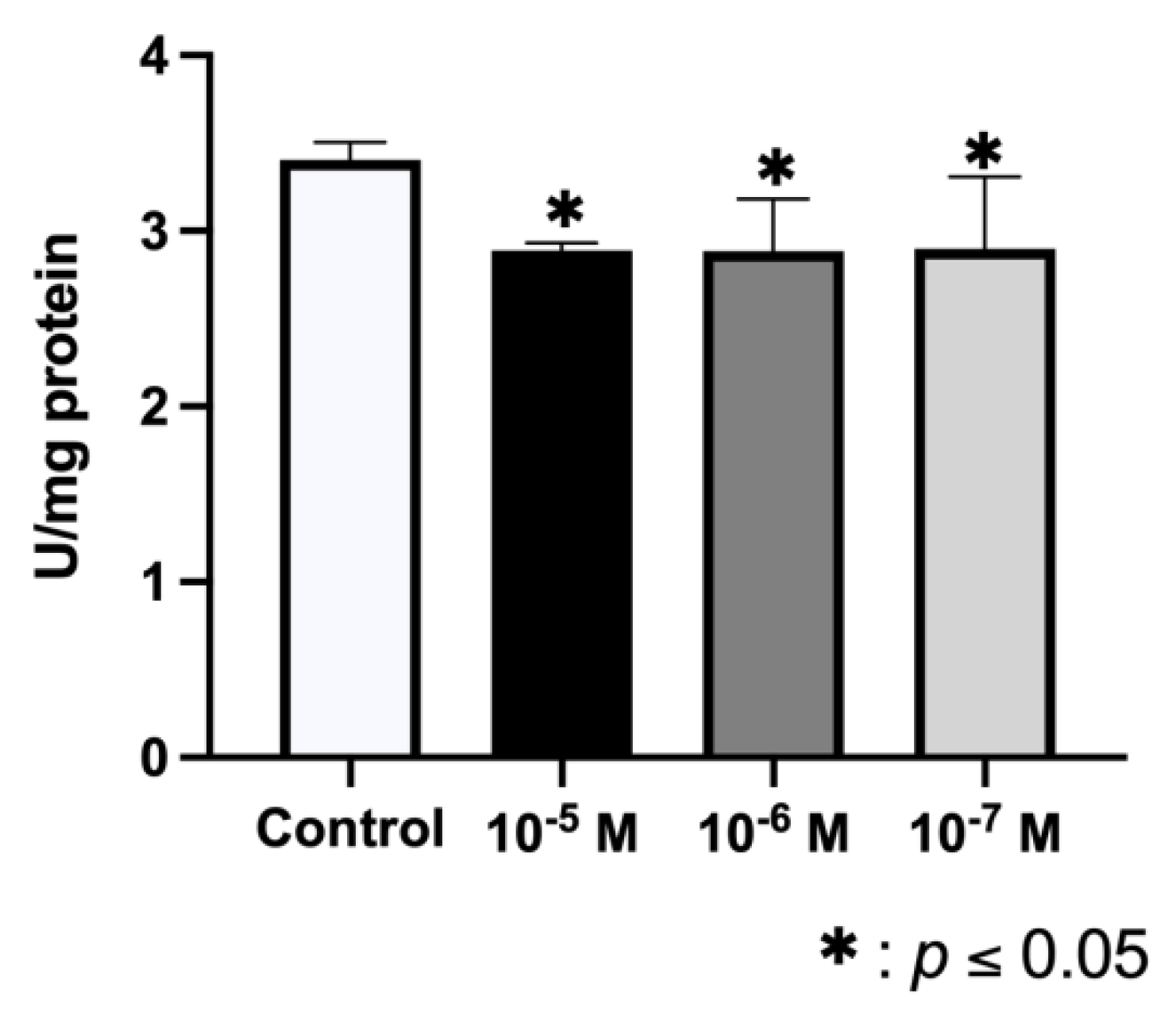
2.3. Effect of BPA on ALP Synthesis
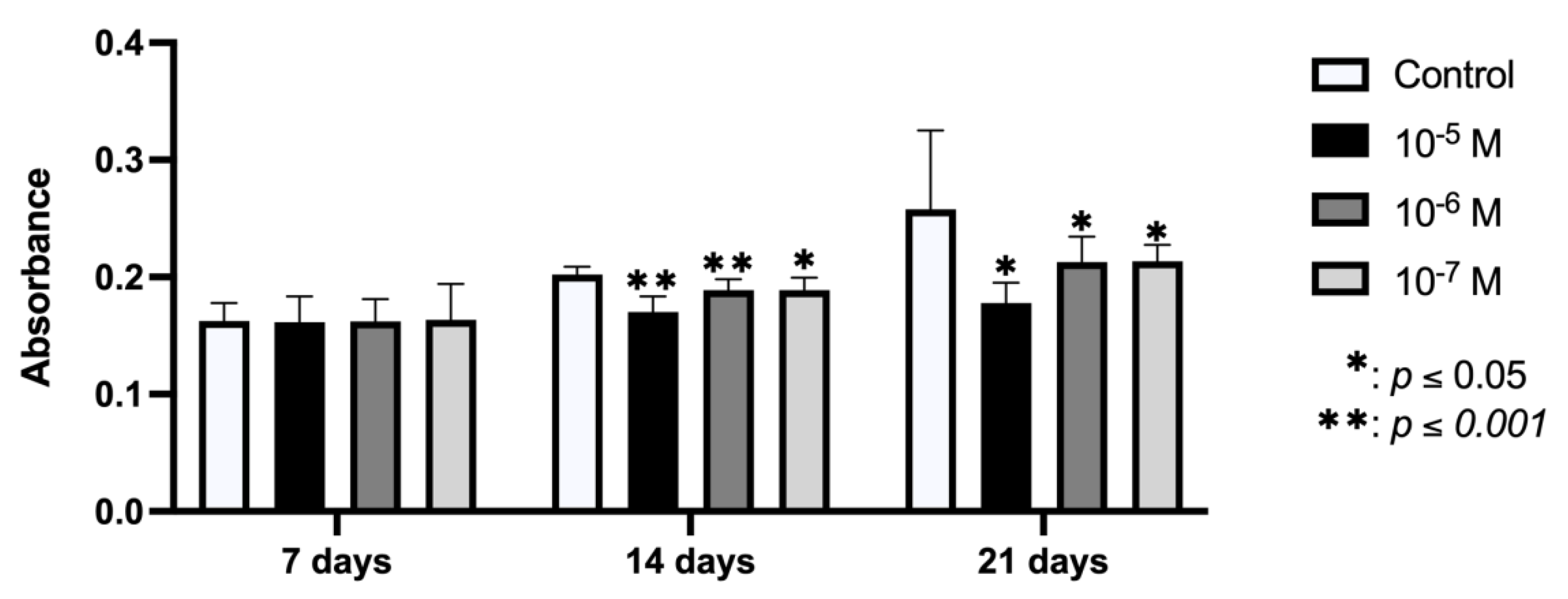
2.4. Effect of BPA on In Vitro Mineralization
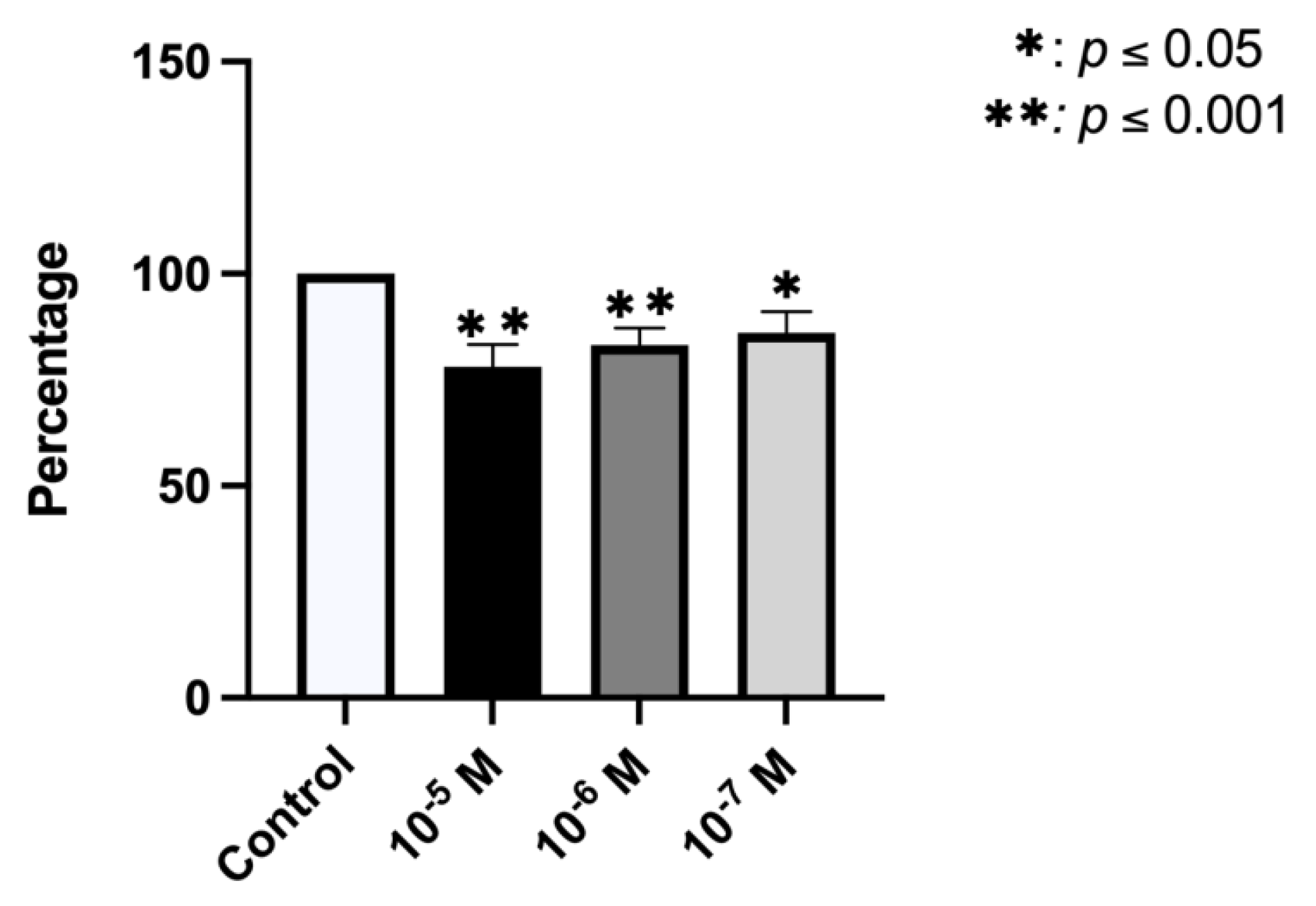
2.5. Effect of BPA on Phagocytic Capacity
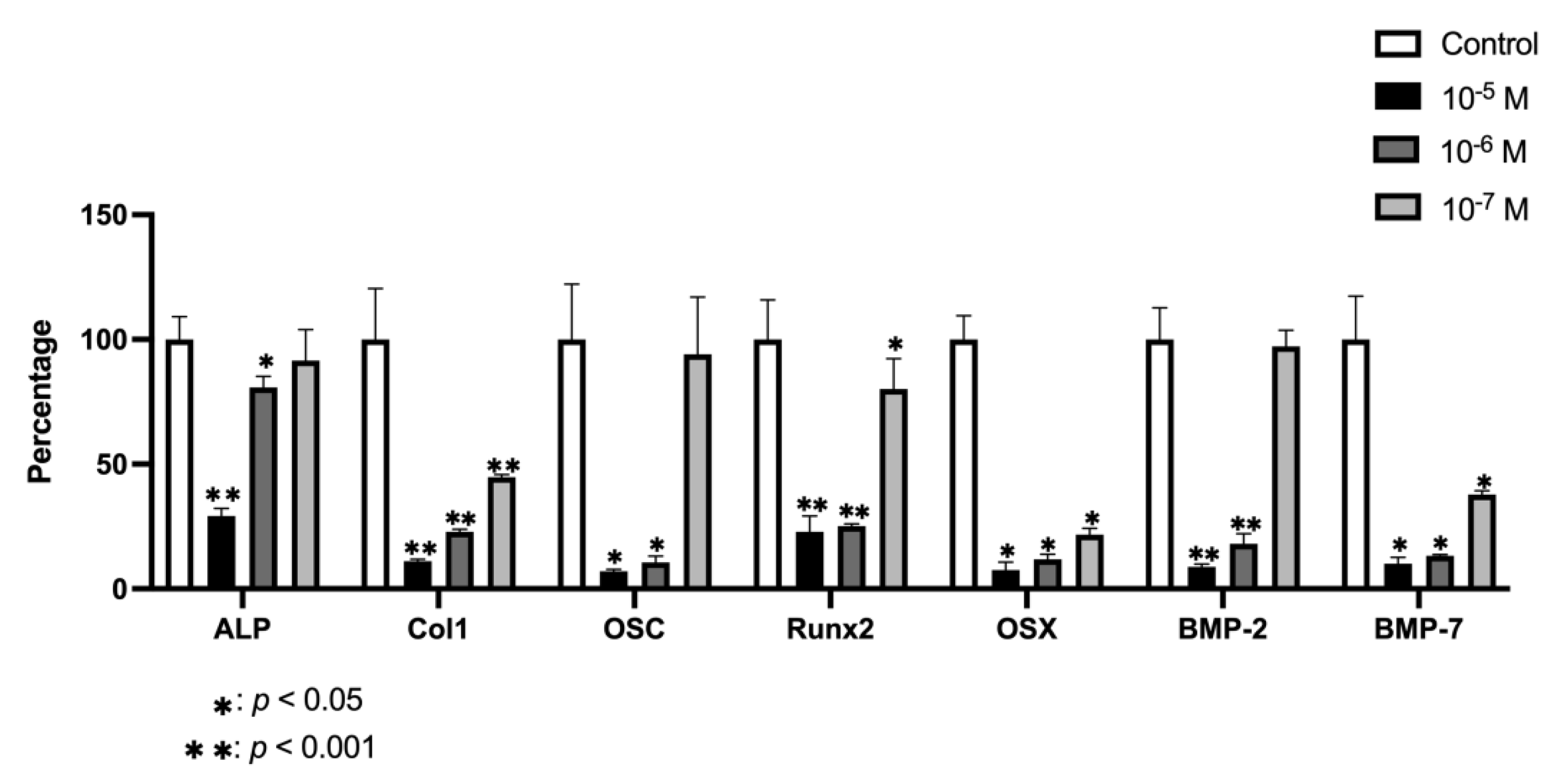
2.6. Effect of BPA on Gene Expression
3. Discussion
4. Materials and Methods
4.1. Chemical
4.2. Primary Human Osteoblasts
4.3. Treatments
4.4. Cell Proliferation
4.5. Apoptosis and Necrosis Analysis
4.6. Antigenic Phenotype
4.7. ALP Activity
4.8. Mineralization Assay
4.9. Phagocytic Activity
4.10. Gene Expression Analysis
4.11. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Acknowledgments
Conflicts of Interest
References
- Dodson, R.E.; Setzer, R.W.; Spengler, J.D.; Brody, J.G.; Rudel, R.A.; Cedeño Laurent, J.G. Influence of Living in the Same Home on Biomonitored Levels of Consumer Product Chemicals. J. Exp. Sci Environ. Epidemiol. 2021. [Google Scholar] [CrossRef]
- Sathyanarayana, S.; Alcedo, G.; Saelens, B.E.; Zhou, C.; Dills, R.L.; Yu, J.; Lanphear, B. Unexpected Results in a Randomized Dietary Trial to Reduce Phthalate and Bisphenol A Exposures. J. Exp. Sci Environ. Epidemiol. 2013, 23, 378–384. [Google Scholar] [CrossRef]
- Bisphenol A|EFSA. Available online: https://www.efsa.europa.eu/en/topics/topic/bisphenol (accessed on 4 May 2022).
- Thent, Z.C.; Froemming, G.R.A.; Muid, S. Bisphenol A Exposure Disturbs the Bone Metabolism: An Evolving Interest towards an Old Culprit. Life Sci. 2018, 198, 1–7. [Google Scholar] [CrossRef]
- World Health Organization, United Nations Environment Programme, Inter-Organization Programme for the Sound Management of Chemicals; Bergman, Å.; Heindel, J.J. State of the Science of Endocrine Disrupting Chemicals 2012: Summary for Decision-Makers; World Health Organization: Geneva, Switzerland, 2013. [Google Scholar]
- Krüger, T.; Long, M.; Bonefeld-Jørgensen, E.C. Plastic Components Affect the Activation of the Aryl Hydrocarbon and the Androgen Receptor. Toxicology 2008, 246, 112–123. [Google Scholar] [CrossRef]
- Matsushima, A.; Kakuta, Y.; Teramoto, T.; Koshiba, T.; Liu, X.; Okada, H.; Tokunaga, T.; Kawabata, S.-I.; Kimura, M.; Shimohigashi, Y. Structural Evidence for Endocrine Disruptor Bisphenol A Binding to Human Nuclear Receptor ERR. J. Biochem. 2007, 142, 517–524. [Google Scholar] [CrossRef]
- Vrzal, R.; Zenata, O.; Doricakova, A.; Dvorak, Z. Environmental Pollutants Parathion, Paraquat and Bisphenol A Show Distinct Effects towards Nuclear Receptors-Mediated Induction of Xenobiotics-Metabolizing Cytochromes P450 in Human Hepatocytes. Toxicol. Lett. 2015, 238, 43–53. [Google Scholar] [CrossRef]
- Bolli, A.; Galluzzo, P.; Ascenzi, P.; Del Pozzo, G.; Manco, I.; Vietri, M.T.; Mita, L.; Altucci, L.; Mita, D.G.; Marino, M. Laccase Treatment Impairs Bisphenol A-Induced Cancer Cell Proliferation Affecting Estrogen Receptor Alpha-Dependent Rapid Signals. IUBMB Life 2008, 60, 843–852. [Google Scholar] [CrossRef]
- Murata, M.; Kang, J.-H. Bisphenol A (BPA) and Cell Signaling Pathways. Biotechnol. Adv. 2018, 36, 311–327. [Google Scholar] [CrossRef]
- Yaglova, N.V.; Yaglov, V.V. Endocrine Disruptors as a New Etiologic Factor of Bone Tissue Diseases (Review). Sovrem Tekhnologii Med. 2021, 13, 84–94. [Google Scholar] [CrossRef]
- Araiza, V.H.D.R.; Mendoza, M.S.; Castro, K.E.N.; Cruz, S.M.; Rueda, K.C.; de Leon, C.T.G.; Morales Montor, J. Bisphenol A, an Endocrine-Disruptor Compund, That Modulates the Immune Response to Infections. Front. Biosci. 2021, 26, 346–362. [Google Scholar] [CrossRef]
- Chin, K.-Y.; Pang, K.-L.; Mark-Lee, W.F. A Review on the Effects of Bisphenol A and Its Derivatives on Skeletal Health. Int. J. Med. Sci. 2018, 15, 1043–1050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hwang, J.K.; Min, K.H.; Choi, K.H.; Hwang, Y.C.; Jeong, I.-K.; Ahn, K.J.; Chung, H.-Y.; Chang, J.S. Bisphenol A Reduces Differentiation and Stimulates Apoptosis of Osteoclasts and Osteoblasts. Life Sci. 2013, 93, 367–372. [Google Scholar] [CrossRef]
- Miki, Y.; Hata, S.; Nagasaki, S.; Suzuki, T.; Ito, K.; Kumamoto, H.; Sasano, H. Steroid and Xenobiotic Receptor-Mediated Effects of Bisphenol A on Human Osteoblasts. Life Sci. 2016, 155, 29–35. [Google Scholar] [CrossRef]
- Adoamnei, E.; Mendiola, J.; Vela-Soria, F.; Fernández, M.F.; Olea, N.; Jørgensen, N.; Swan, S.H.; Torres-Cantero, A.M. Urinary Bisphenol A Concentrations Are Associated with Reproductive Parameters in Young Men. Environ. Res. 2018, 161, 122–128. [Google Scholar] [CrossRef]
- Ma, Y.; Liu, H.; Wu, J.; Yuan, L.; Wang, Y.; Du, X.; Wang, R.; Marwa, P.W.; Petlulu, P.; Chen, X.; et al. The Adverse Health Effects of Bisphenol A and Related Toxicity Mechanisms. Environ. Res. 2019, 176, 108575. [Google Scholar] [CrossRef]
- Pelch, K.; Wignall, J.A.; Goldstone, A.E.; Ross, P.K.; Blain, R.B.; Shapiro, A.J.; Holmgren, S.D.; Hsieh, J.-H.; Svoboda, D.; Auerbach, S.S.; et al. A Scoping Review of the Health and Toxicological Activity of Bisphenol A (BPA) Structural Analogues and Functional Alternatives. Toxicology 2019, 424, 152235. [Google Scholar] [CrossRef] [PubMed]
- Thent, Z.C.; Froemming, G.R.A.; Ismail, A.B.M.; Fuad, S.B.S.A.; Muid, S. Employing Different Types of Phytoestrogens Improve Bone Mineralization in Bisphenol A Stimulated Osteoblast. Life Sci. 2018, 210, 214–223. [Google Scholar] [CrossRef] [PubMed]
- Papa, V.; Bimonte, V.M.; Wannenes, F.; D’Abusco, A.S.; Fittipaldi, S.; Scandurra, R.; Politi, L.; Crescioli, C.; Lenzi, A.; Di Luigi, L.; et al. The Endocrine Disruptor Cadmium Alters Human Osteoblast-like Saos-2 Cells Homeostasis in Vitro by Alteration of Wnt/β-Catenin Pathway and Activation of Caspases. J. Endocrinol. Investig. 2015, 38, 1345–1356. [Google Scholar] [CrossRef]
- Boyce, B.F.; Zuscik, M.J.; Xing, L. Chapter 11—Biology of Bone and Cartilage. In Genetics of Bone Biology and Skeletal Disease, 2nd ed.; Thakker, R.V., Whyte, M.P., Eisman, J.A., Igarashi, T., Eds.; Academic Press: Cambridge, MA, USA, 2018; pp. 173–195. ISBN 978-0-12-804182-6. [Google Scholar]
- Hadjidakis, D.J.; Androulakis, I.I. Bone Remodeling. Ann. N. Y. Acad. Sci. 2006, 1092, 385–396. [Google Scholar] [CrossRef]
- Xiao, W.; Wang, Y.; Pacios, S.; Li, S.; Graves, D.T. Cellular and Molecular Aspects of Bone Remodeling. Front. Oral Biol. 2016, 18, 9–16. [Google Scholar] [CrossRef] [PubMed]
- Jayakumar, P.; Di Silvio, L. Osteoblasts in Bone Tissue Engineering. Proc. Inst. Mech. Eng. H 2010, 224, 1415–1440. [Google Scholar] [CrossRef]
- Kanno, S.; Hirano, S.; Kayama, F. Effects of Phytoestrogens and Environmental Estrogens on Osteoblastic Differentiation in MC3T3-E1 Cells. Toxicology 2004, 196, 137–145. [Google Scholar] [CrossRef]
- Seeman, E.; Delmas, P.D. Bone Quality—the Material and Structural Basis of Bone Strength and Fragility. N. Engl. J. Med. 2006, 354, 2250–2261. [Google Scholar] [CrossRef] [Green Version]
- De Luna-Bertos, E.; Ramos-Torrecillas, J.; Garcia-Martinez, O.; Guildford, A.; Santin, M.; Ruiz, C. Therapeutic Doses of Nonsteroidal Anti-Inflammatory Drugs Inhibit Osteosarcoma MG-63 Osteoblast-Like Cells Maturation, Viability, and Biomineralization Potential. Sci. World J. 2013, 2013, 809891. [Google Scholar] [CrossRef] [Green Version]
- Garcia-Martinez, O.; Reyes-Botella, C.; Diaz-Rodriguez, L.; De Luna-Bertos, E.; Ramos-Torrecillas, J.; Francisco Vallecillo-Capilla, M.; Ruiz, C. Effect of Platelet-Rich Plasma on Growth and Antigenic Profile of Human Osteoblasts and Its Clinical Impact. J. Oral Maxillofac. Surg. 2012, 70, 1558–1564. [Google Scholar] [CrossRef]
- Melguizo-Rodriguez, L.; Ramos-Torrecillas, J.; Javier Manzano-Moreno, F.; Illescas-Montes, R.; Rivas, A.; Ruiz, C.; De Luna-Bertos, E.; Garcia-Martinez, O. Effect of Phenolic Extracts from Different Extra-Virgin Olive Oil Varieties on Osteoblast-like Cells. PLoS ONE 2018, 13, e0196530. [Google Scholar] [CrossRef] [Green Version]
- Pérez, E.; García-Martínez, O.; Arroyo-Morales, M.; Reyes-Botella, C.; Ruiz, C. Modulation of Antigenic Phenotype in Cultured Human Osteoblast-like Cells by FGFb, TGFbeta1, PDGF-BB, IL-2, IL-1beta, LPS and IFNgamma. Biosci. Rep. 2006, 26, 281–289. [Google Scholar] [CrossRef]
- Zuo, C.; Huang, Y.; Bajis, R.; Sahih, M.; Li, Y.-P.; Dai, K.; Zhang, X. Osteoblastogenesis Regulation Signals in Bone Remodeling. Osteoporos. Int. 2012, 23, 1653–1663. [Google Scholar] [CrossRef]
- Abraham, A.; Chakraborty, P. A Review on Sources and Health Impacts of Bisphenol A. Rev. Environ. Health 2020, 35, 201–210. [Google Scholar] [CrossRef]
- Den Braver-Sewradj, S.P.; van Spronsen, R.; Hessel, E.V.S. Substitution of Bisphenol A: A Review of the Carcinogenicity, Reproductive Toxicity, and Endocrine Disruption Potential of Alternative Substances. Crit. Rev. Toxicol. 2020, 50, 128–147. [Google Scholar] [CrossRef]
- Clynes, M.A.; Harvey, N.C.; Curtis, E.M.; Fuggle, N.R.; Dennison, E.M.; Cooper, C. The Epidemiology of Osteoporosis. Br. Med. Bull. 2020, 133, 105–117. [Google Scholar] [CrossRef] [PubMed]
- Cauley, J.A. Osteoporosis: Fracture Epidemiology Update 2016. Curr. Opin. Rheumatol. 2017, 29, 150–156. [Google Scholar] [CrossRef] [PubMed]
- Föger-Samwald, U.; Dovjak, P.; Azizi-Semrad, U.; Kerschan-Schindl, K.; Pietschmann, P. Osteoporosis: Pathophysiology and Therapeutic Options. EXCLI J. 2020, 19, 1017–1037. [Google Scholar] [CrossRef] [PubMed]
- Mahmoudi, A.; Hadrich, F.; Feki, I.; Ghorbel, H.; Bouallagui, Z.; Marrekchi, R.; Fourati, H.; Sayadi, S. Oleuropein and Hydroxytyrosol Rich Extracts from Olive Leaves Attenuate Liver Injury and Lipid Metabolism Disturbance in Bisphenol A-Treated Rats. Food Funct. 2018, 9, 3220–3234. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Martinez, O.; Diaz-Rodriguez, L.; Rodriguez-Perez, L.; De Luna-Bertos, E.; Reyes Botella, C.; Concepcion Ruiz, C. Effect of Acetaminophen, Ibuprofen and Methylprednisolone on Different Parameters of Human Osteoblast-like Cells. Arch. Oral Biol. 2011, 56, 317–323. [Google Scholar] [CrossRef]
- Illescas-Montes, R.; Melguizo-Rodríguez, L.; Manzano-Moreno, F.J.; García-Martínez, O.; Ruiz, C.; Ramos-Torrecillas, J. Cultured Human Fibroblast Biostimulation Using a 940 Nm Diode Laser. Materials 2017, 10, 793. [Google Scholar] [CrossRef] [Green Version]
- Costela-Ruiz, V.J.; Melguizo-Rodríguez, L.; Illescas-Montes, R.; Ramos-Torrecillas, J.; Manzano-Moreno, F.J.; Ruiz, C.; Bertos, E.D.L.-. Effects of Therapeutic Doses of Celecoxib on Several Physiological Parameters of Cultured Human Osteoblasts. Int. J. Med. Sci. 2019, 16, 1466–1472. [Google Scholar] [CrossRef] [Green Version]
- Manzano-Moreno, F.J.; Ramos-Torrecillas, J.; De Luna-Bertos, E.; Reyes-Botella, C.; Ruiz, C.; García-Martínez, O. Nitrogen-Containing Bisphosphonates Modulate the Antigenic Profile and Inhibit the Maturation and Biomineralization Potential of Osteoblast-like Cells. Clin. Oral. Investig. 2015, 19, 895–902. [Google Scholar] [CrossRef]
- Manzano-Moreno, F.J.; Ramos-Torrecillas, J.; Melguizo-Rodríguez, L.; Illescas-Montes, R.; Ruiz, C.; García-Martínez, O. Bisphosphonate Modulation of the Gene Expression of Different Markers Involved in Osteoblast Physiology: Possible Implications in Bisphosphonate-Related Osteonecrosis of the Jaw. Int. J. Med. Sci. 2018, 15, 359–367. [Google Scholar] [CrossRef] [Green Version]
- Fromme, H.; Küchler, T.; Otto, T.; Pilz, K.; Müller, J.; Wenzel, A. Occurrence of Phthalates and Bisphenol A and F in the Environment. Water Res. 2002, 36, 1429–1438. [Google Scholar] [CrossRef]

| Gene | Sense Primer | Antisense Primer | Amplicon (bp) |
|---|---|---|---|
| ALP | 5′-CCCATATTCCCTGCACTTTG-3′ | 5′-ACCTTGACCTCTCAGCCTCA-3′ | 195 |
| Col-1 | 5′-CCTCATCGCAGGAGAAAAAG-3′ | 5′-CCCTGAAGTGACTGGGGTAA-3′ | 169 |
| OSC | 5′-CCTGGTCCAGACCACAGAGT-3′ | 5′-TGGAGATTTTGGGAGTACGG-3′ | 194 |
| Runx2 | 5′-CCTTGCTGCTCTACCTCCAC-3′ | 5′-CACACAGGATGGCTTGAAGA-3′ | 197 |
| OSX | 5′-TGCCTAGAAGCCCTGAGAAA-3′ | 5′-TTTAACTTGGGGCCTTGAGA-3′ | 205 |
| BMP-2 | 5′-TCGAAATTCCCCGTGACCAG-3′ | 5′-CCACTTCCACCACGAATCCA-3′ | 142 |
| BMP-7 | 5′-CTGGTCTTTGTCTGCAGTGG-3′ | 5′-GTACCCCTCAACAAGGCTTC-3′ | 202 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Recio, E.; Costela-Ruiz, V.J.; Melguizo-Rodriguez, L.; Ramos-Torrecillas, J.; García-Martínez, O.; Ruiz, C.; de Luna-Bertos, E. Repercussions of Bisphenol A on the Physiology of Human Osteoblasts. Int. J. Mol. Sci. 2022, 23, 5349. https://doi.org/10.3390/ijms23105349
García-Recio E, Costela-Ruiz VJ, Melguizo-Rodriguez L, Ramos-Torrecillas J, García-Martínez O, Ruiz C, de Luna-Bertos E. Repercussions of Bisphenol A on the Physiology of Human Osteoblasts. International Journal of Molecular Sciences. 2022; 23(10):5349. https://doi.org/10.3390/ijms23105349
Chicago/Turabian StyleGarcía-Recio, Enrique, Víctor J. Costela-Ruiz, Lucía Melguizo-Rodriguez, Javier Ramos-Torrecillas, Olga García-Martínez, Concepción Ruiz, and Elvira de Luna-Bertos. 2022. "Repercussions of Bisphenol A on the Physiology of Human Osteoblasts" International Journal of Molecular Sciences 23, no. 10: 5349. https://doi.org/10.3390/ijms23105349
APA StyleGarcía-Recio, E., Costela-Ruiz, V. J., Melguizo-Rodriguez, L., Ramos-Torrecillas, J., García-Martínez, O., Ruiz, C., & de Luna-Bertos, E. (2022). Repercussions of Bisphenol A on the Physiology of Human Osteoblasts. International Journal of Molecular Sciences, 23(10), 5349. https://doi.org/10.3390/ijms23105349







