CD147 Is Essential for the Development of Psoriasis via the Induction of Th17 Cell Differentiation
Abstract
:1. Introduction
2. Results
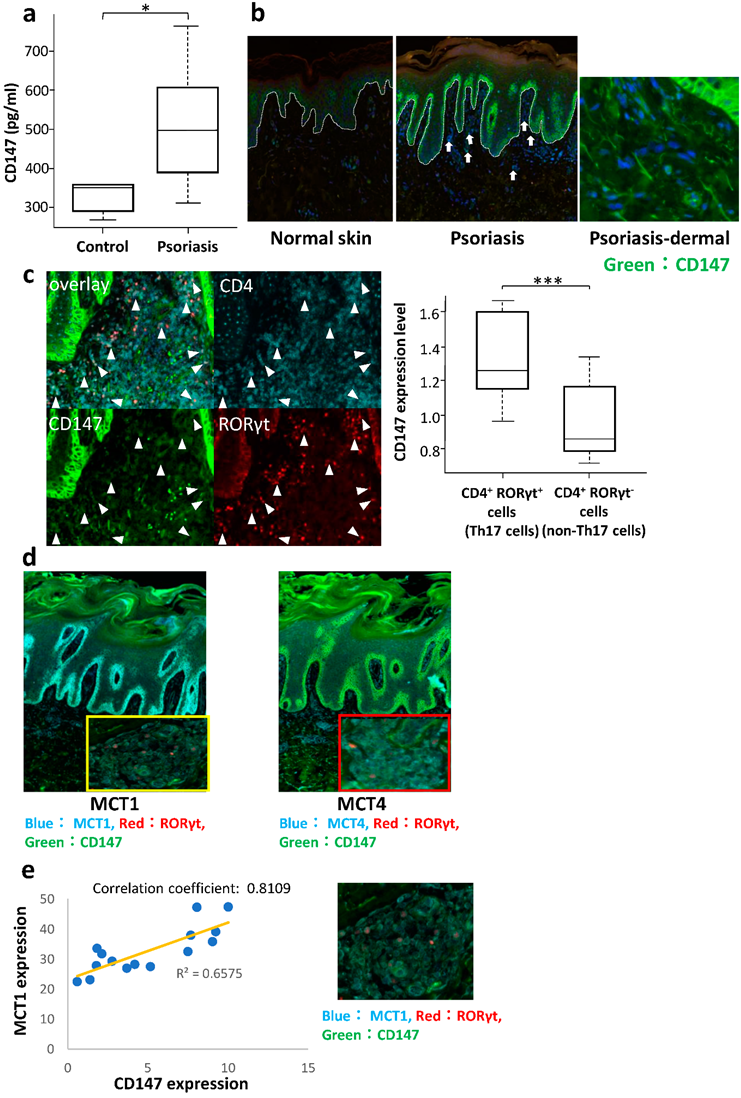
2.1. Serum Level of CD147 in Patients with Psoriasis
2.2. Expression of CD147 and MCTs on the Lymphocytes in the Lesional Skin of Psoriasis
2.3. Effect of CD147 Deficiency on MCT-1 and RORγt expression in CD4+ T Cells—In Vitro Studies
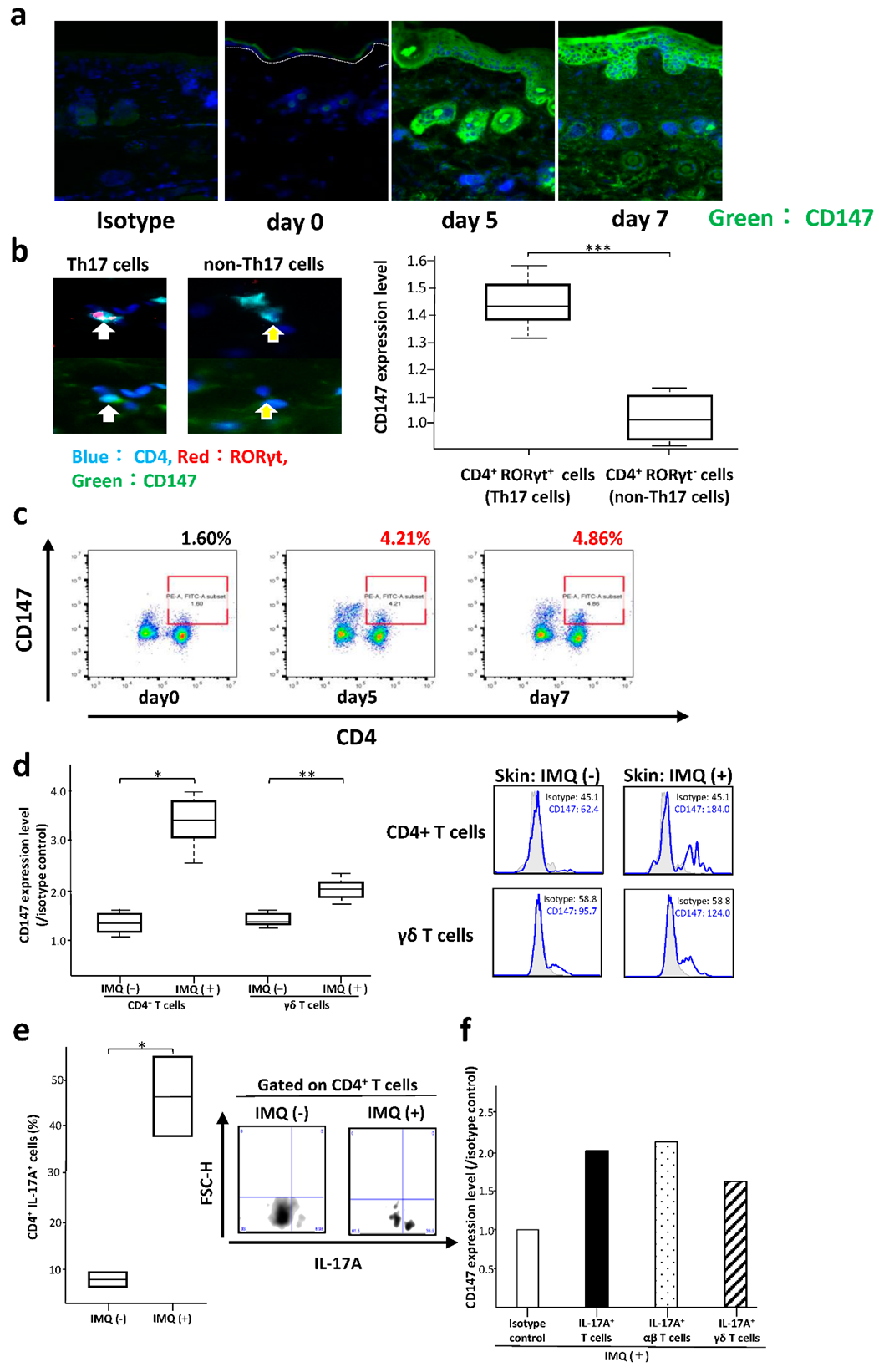
2.4. Imiquimod-Induced Psoriatic Lesions in Model Mice
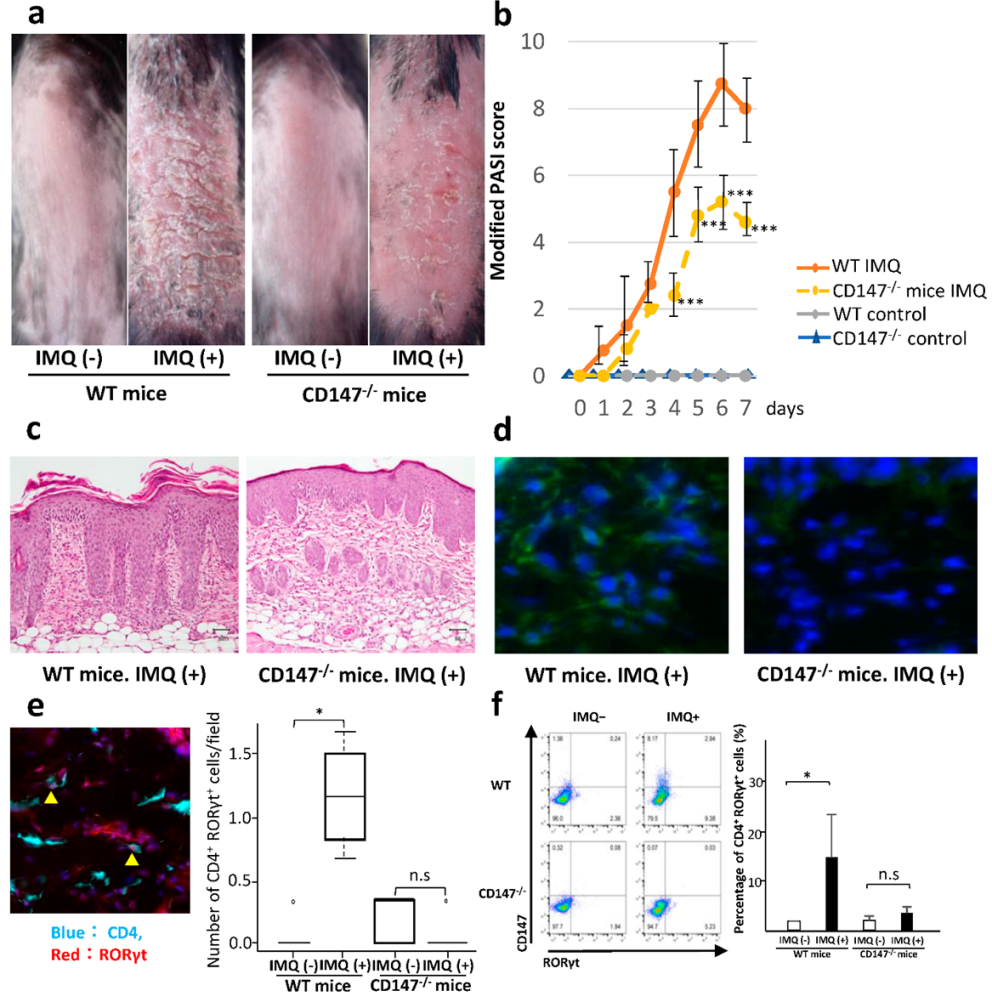
2.5. Effect of CD147 Deficiency on IMQ-Induced Dermatitis
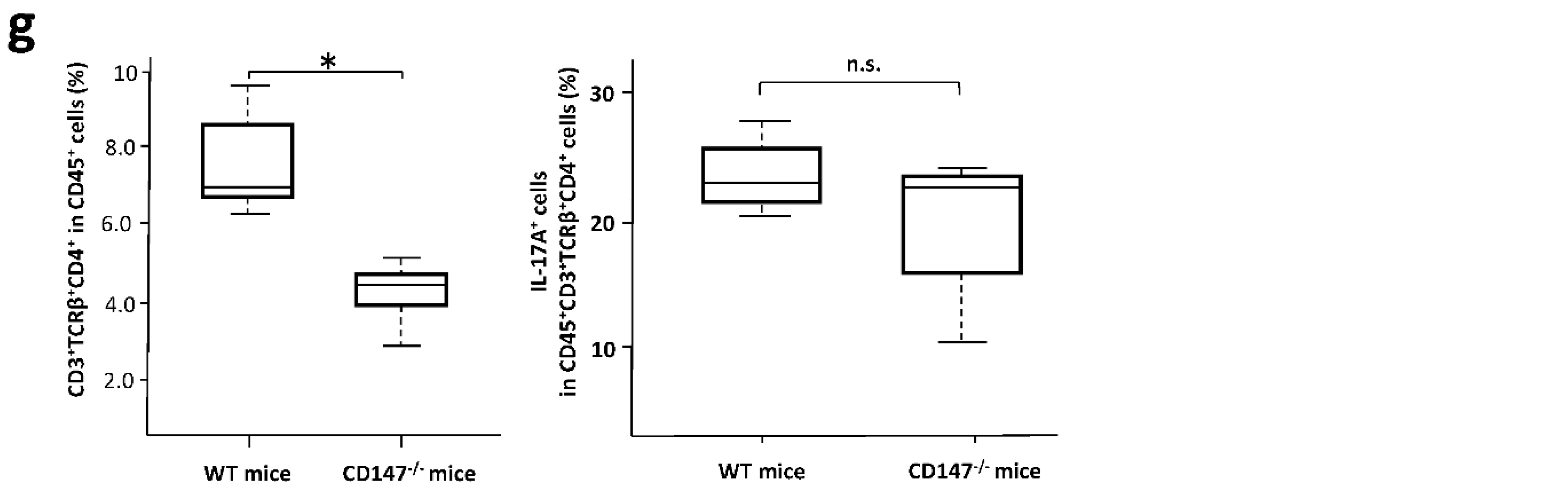
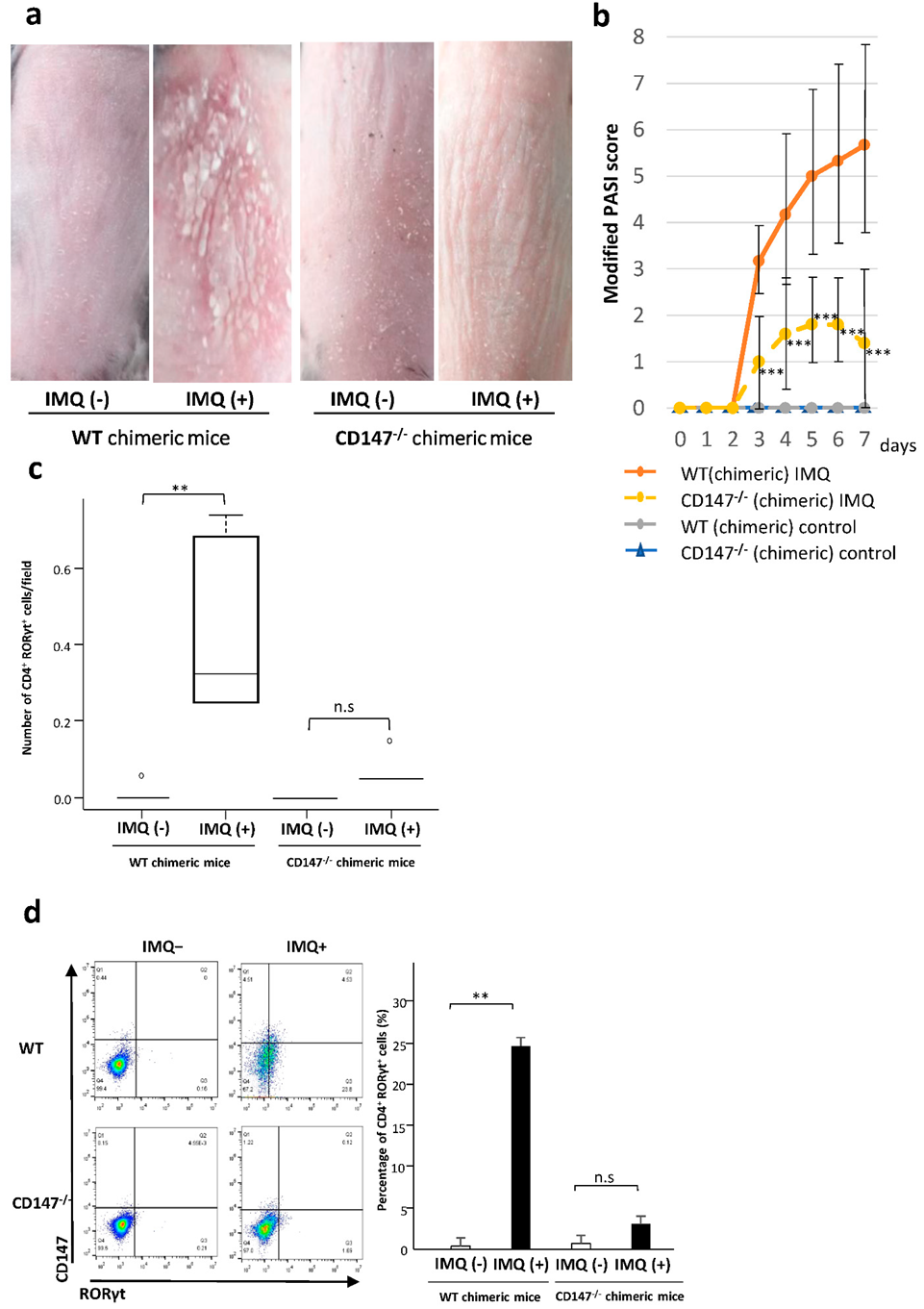
2.6. Effect of CD147 Deficiency in Hematopoietic Cells on the Development of IMQ-Induced Dermatitis
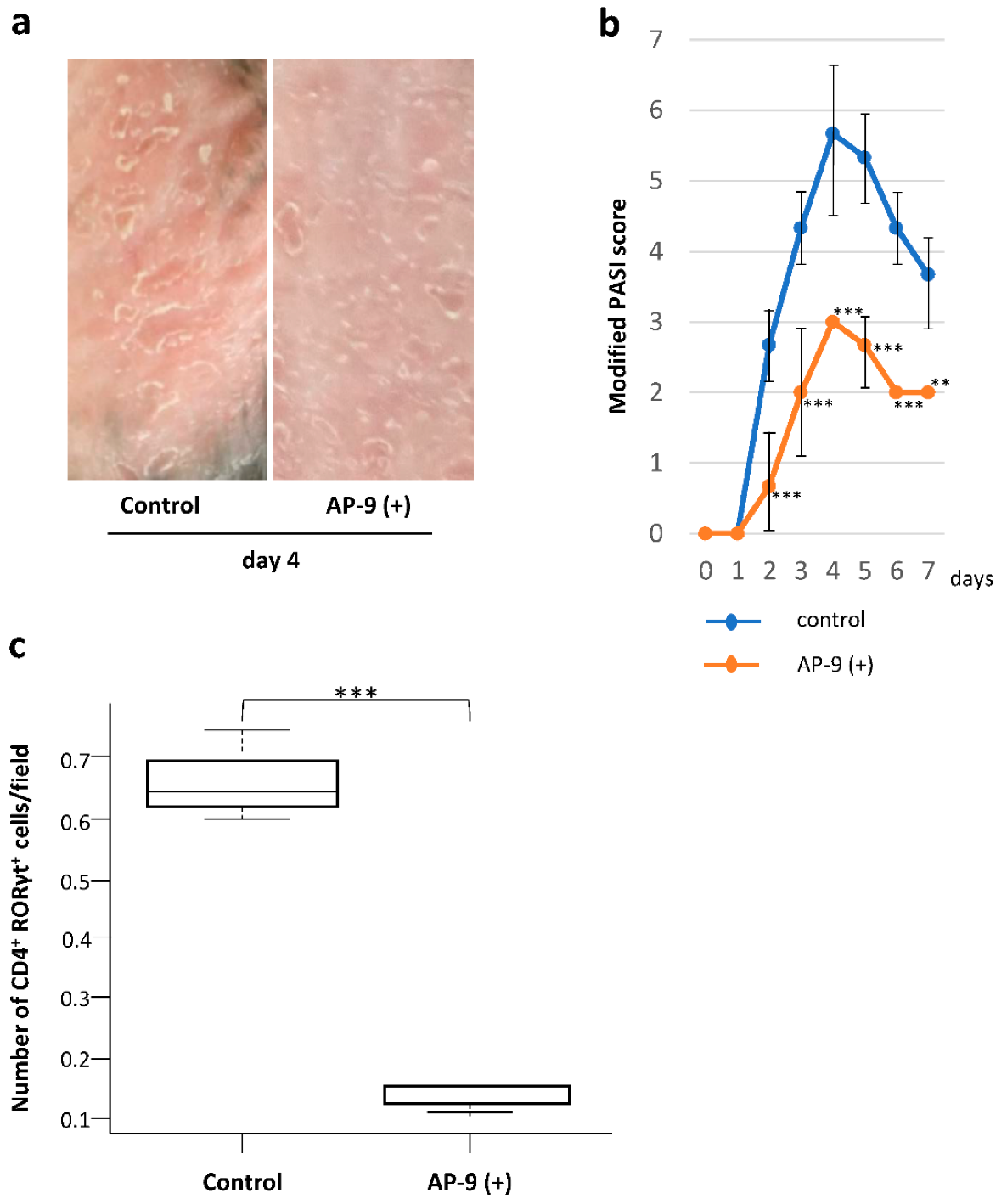
2.7. Effect of AP-9 on the Development of IMQ-Induced Dermatitis
3. Discussion
4. Materials and Methods
4.1. Ethical Considerations
4.2. Enzyme-Linked Immunosorbent Assay (ELISA)
4.3. Animals
4.4. Induction of Psoriasis in Mice
4.5. Light and Confocal Immunofluorescence Microscopy
4.6. Preparation of Skin Single-Cell Suspensions
4.7. Flow Cytometry
4.8. Intracellular IL-17A Staining
4.9. Treatment with CD147 Inhibitor
4.10. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Perera, G.K.; Meglio, P.D.; Nestle, F.O. Psoriasis. Annu. Rev. Pathol. 2012, 7, 385–422. [Google Scholar] [CrossRef]
- Lynde, C.W.; Poulin, Y.; Vender, R.; Bourcier, M.; Khalil, S. Interleukin 17A: Toward a new understanding of psoriasis pathogenesis. J. Am. Acad. Dermatol. 2014, 71, 141–150. [Google Scholar] [CrossRef]
- Martin, D.A.; Towne, J.E.; Krikorian, G.; Klekotka, P.; Gudjonsson, J.E.; Krueger, J.G.; Russel, C.B. The emerging role of IL-17 in the pathogenesis of psoriasis: Preclinical and clinical findings. J. Invest. Dermatol. 2013, 133, 17–26. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ivanov, I.I.; McKenzie, B.S.; Zhou, L.; Tadokoro, C.E.; Lepelley, A.; Lafaille, J.J.; Cua, D.J.; Littman, D.R. The orphan nuclear receptor RORγt directs the differentiation program of proinflammatory IL-17+ T helper cells. Cell 2006, 126, 1121–1133. [Google Scholar] [CrossRef] [Green Version]
- Donnelly, R.P.; Finlay, D.K. Glucose, glycolysis and lymphocyte responses. Mol. Immunol. 2015, 68, 513–519. [Google Scholar] [CrossRef] [PubMed]
- Warburg, O. On respiratory impairment in cancer cells. Science 1956, 124, 269–270. [Google Scholar] [CrossRef] [PubMed]
- Miyauchi, T.; Kanekura, T.; Yamaoka, A.; Ozawa, M.; Miyazawa, S.; Muramatsu, T. Basigin, a new, broadly distributed member of the immunoglobulin superfamily, has strong homology with both the immunoglobulin V domain and the β-chain of major histocompatibility complex class II antigen. J. Biochem. 1990, 107, 316–323. [Google Scholar] [CrossRef]
- Kanekura, T.; Miyauchi, T.; Tashiro, M.; Muramatsu, T. Basigin, a new member of the immunoglobulin super family: Genes in different mammalian species, glycosylation changes in the molecule from adult organs and possible variation in the N-terminal sequences. Cell Struct. Funct. 1991, 16, 23–30. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Peng, C.; Zhang, S.; Lei, L.; Zhang, X.; Jia, X.; Luo, Z.; Huang, X.; Kuang, Y.; Zeng, W.; Su, J.; et al. Epidermal CD147 expression plays a key role in IL-22-induced psoriatic dermatitis. Sci. Rep. 2017, 7, 1–14. [Google Scholar]
- Muramatsu, T. Basigin (CD147), a multifunctional transmembrane glycoprotein with various binding partners. J. Biochem. 2016, 159, 481–490. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kirk, P.; Wilson, M.C.; Heddle, C.; Brown, M.H.; Barcley, A.N.; Halestrap, A.P. CD147 is tightly associated with lactate transporters MCT1 and MCT4 and facilitates their cell surface expression. EMBO J. 2000, 19, 3896–3904. [Google Scholar] [CrossRef]
- Su, J.; Chen, X.; Kanekura, T. A CD147-targeting siRNA inhibits the proliferation, invasiveness, and VEGF production of human malignant melanoma cells by down-regulating glycolysis. Cancer Lett. 2009, 273, 140–147. [Google Scholar] [CrossRef] [PubMed]
- Gallagher, S.M.; Castorino, J.J.; Wang, D.; Philp, N.N. Monocarboxylate transporter 4 regulates maturation and trafficking of CD147 to the plasma membrane in the metastatic breast cancer cell line MDA-MB-231. Cancer Res. 2007, 67, 4182–4189. [Google Scholar] [CrossRef] [Green Version]
- Baba, M.; Inoue, M.; Itoh, K.; Nishizawa, Y. CD147 induces cell death in cancer cells through impairment of glycolytic energy metabolism. Biochem. Biophys. Res. Commun. 2008, 374, 111–116. [Google Scholar] [CrossRef] [PubMed]
- Le Floch, R.; Chiche, J.; Marchiq, I.; Naiken, T.; Ilc, K.; Murray, C.M.; Critchlow, S.E.; Roux, D.; Simon, M.P.; Pouysségur, J. CD147 subunit of lactate/H+ symportes MCT1 and hypoxia-inducible MCT4 is critical for energetics and growth of glycolytic tumors. Proc. Natl. Acad. Sci. USA 2011, 108, 16663–16666. [Google Scholar] [CrossRef] [Green Version]
- Michalek, R.D.; Gerrets, V.A.; Jacobs, S.R.; Macintyre, A.N.; Maclver, N.J.; Mason, E.F.; Sullivan, S.A.; Nichols, A.G.; Rathmell, J.C. Cutting edge: Distinct glycolytic and lipid oxidative metabolic programs are essential for effector and regulatory CD4+ T cell subsets. J. Immunol. 2011, 186, 3299–3303. [Google Scholar] [CrossRef] [Green Version]
- Halestrap, A.P.; Wilson, M.C. The monocarboxylate transporter family—Role and regulation. IUBMB Life 2012, 64, 109–119. [Google Scholar] [CrossRef]
- Murray, C.M.; Hutchinson, R.; Bantik, J.R.; Belfield, G.P.; Benjamin, A.D.; Brazma, D.; Bundick, R.V.; Cook, I.D.; Craggs, R.I.; Edwards, S.; et al. Monocarboxylate transporter MCT1 is a target for immunosuppression. Nat. Chem. Biol. 2005, 1, 371–376. [Google Scholar] [CrossRef]
- Hahn, J.N.; Kaushik, D.K.; Yong, V.W. The role of EMMPRIN in T cell biology and immunological diseases. J. Leukocyte Biol. 2015, 98, 33–48. [Google Scholar] [CrossRef]
- Yang, H.; Wang, J.; Li, Y.; Yin, Z.; Lv, T.; Zhu, P.; Zhang, Y. CD147 modulates the differentiation of T-helper 17 cells in patients with rheumatoid arthritis. APMIS 2017, 125, 24–31. [Google Scholar] [CrossRef]
- Wang, S.; Liu, C.; Liu, X.; He, Y.; Shen, D.; Luo, Q.; Dong, Y.; Dong, H.; Pang, Z. Effects of matrix metalloproteinase inhibitor doxycycline and CD147 antagonist peptide-9 on gallbladder carcinoma cell lines. Tumour Biol. 2017, 39, 1–6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Halestrap, A.P.; Meredith, D. The SLC16 gene family—From monocarboxylate transporters (MCTs) to aromatic amino acid transporters and beyond. Pflugers Arch. 2004, 447, 619–628. [Google Scholar] [CrossRef]
- Gao, M.; Liao, L.; Jia, X.; Kuang, Y.; Chen, X. Role of soluble CD147 in psoriatic patients: A preliminary study. J. Dermatol. 2018, 45, e266–e267. [Google Scholar] [CrossRef] [PubMed]
- Maeda-Hori, M.; Kosugi, T.; Kojima, H.; Sato, W.; Inaba, S.; Maeda, K.; Nagaya, H.; Sato, Y.; Ishimoto, T.; Ozaki, T.; et al. Plasma CD147 reflects histological features in patients with lupus nephritis. Lupus 2014, 23, 342–352. [Google Scholar] [CrossRef] [PubMed]
- Hatanaka, M.; Higashi, Y.; Fukushige, T.; Baba, N.; Kawai, K.; Hashiguchi, T.; Su, J.; Zeng, W.; Chen, X.; Kanekura, T. Cleaved CD147 shed from the surface of malignant melanoma cells activates MMP2 produced by fibroblasts. Anticancer Res. 2014, 34, 7091–7096. [Google Scholar] [PubMed]
- Lu, H.; Kuang, Y.; Su, J.; Chang, J.; Wu, L.; Kanekura, T.; Li, D.; Cheng, M.; Chen, X. CD147 is highly expressed on peripheral blood neutrophils from patients with psoriasis and induces neutrophil chemotaxis. J. Dermatol. 2010, 37, 1053–1056. [Google Scholar] [CrossRef]
- Igakura, T.; Kafomatsu, K.; Kaname, T.; Muramatsu, H.; Fan, Q.; Miyauchi, T.; Toyama, Y.; Kuno, N.; Yuasa, S.; Takahashi, M.; et al. A null mutation in Basigin, an immunoglobulin superfamily member, indicates its important roles in peri-implantation development and spermatogenesis. Dev. Biol. 1998, 194, 152–165. [Google Scholar] [CrossRef] [Green Version]
- Chen, S.; Kadomatsu, K.; Kondo, M.; Toyama, Y.; Toshimori, K.; Ueno, S.; Miyake, Y.; Muramatsu, T. Effects of flanking genes on the phenotypes of mice deficient in basigin/CD147. Biochem. Biophys. Res. Commun. 2004, 324, 147–153. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Okubo, A.; Uchida, Y.; Higashi, Y.; Sato, T.; Ogawa, Y.; Ryuge, A.; Kadomatsu, K.; Kanekura, T. CD147 Is Essential for the Development of Psoriasis via the Induction of Th17 Cell Differentiation. Int. J. Mol. Sci. 2022, 23, 177. https://doi.org/10.3390/ijms23010177
Okubo A, Uchida Y, Higashi Y, Sato T, Ogawa Y, Ryuge A, Kadomatsu K, Kanekura T. CD147 Is Essential for the Development of Psoriasis via the Induction of Th17 Cell Differentiation. International Journal of Molecular Sciences. 2022; 23(1):177. https://doi.org/10.3390/ijms23010177
Chicago/Turabian StyleOkubo, Aoi, Youhei Uchida, Yuko Higashi, Takuya Sato, Youichi Ogawa, Akihiro Ryuge, Kenji Kadomatsu, and Takuro Kanekura. 2022. "CD147 Is Essential for the Development of Psoriasis via the Induction of Th17 Cell Differentiation" International Journal of Molecular Sciences 23, no. 1: 177. https://doi.org/10.3390/ijms23010177
APA StyleOkubo, A., Uchida, Y., Higashi, Y., Sato, T., Ogawa, Y., Ryuge, A., Kadomatsu, K., & Kanekura, T. (2022). CD147 Is Essential for the Development of Psoriasis via the Induction of Th17 Cell Differentiation. International Journal of Molecular Sciences, 23(1), 177. https://doi.org/10.3390/ijms23010177






