Overexpression of Isl1 under the Pax2 Promoter, Leads to Impaired Sound Processing and Increased Inhibition in the Inferior Colliculus
Abstract
1. Introduction
2. Results
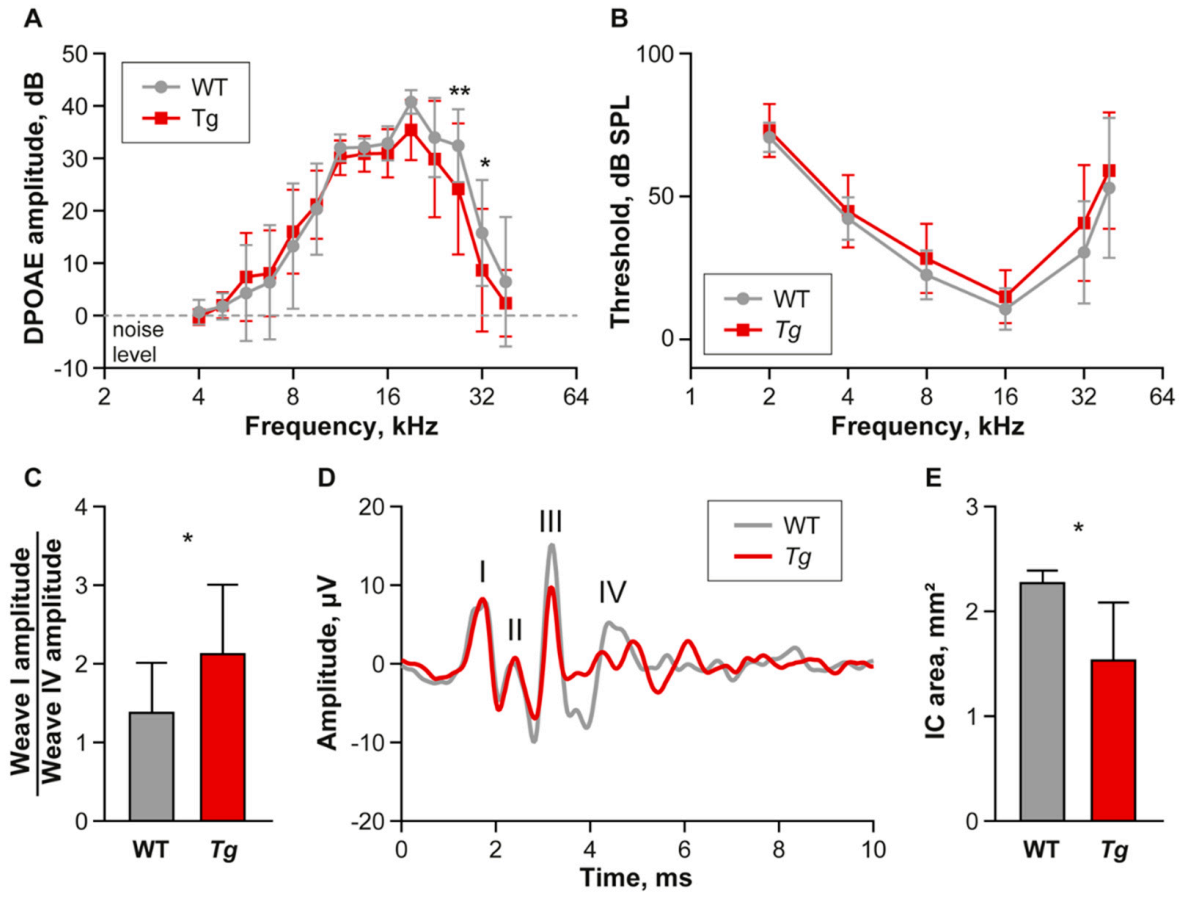
2.1. Young Tg Animals Exhibit Slightly Impaired High Frequency Hearing
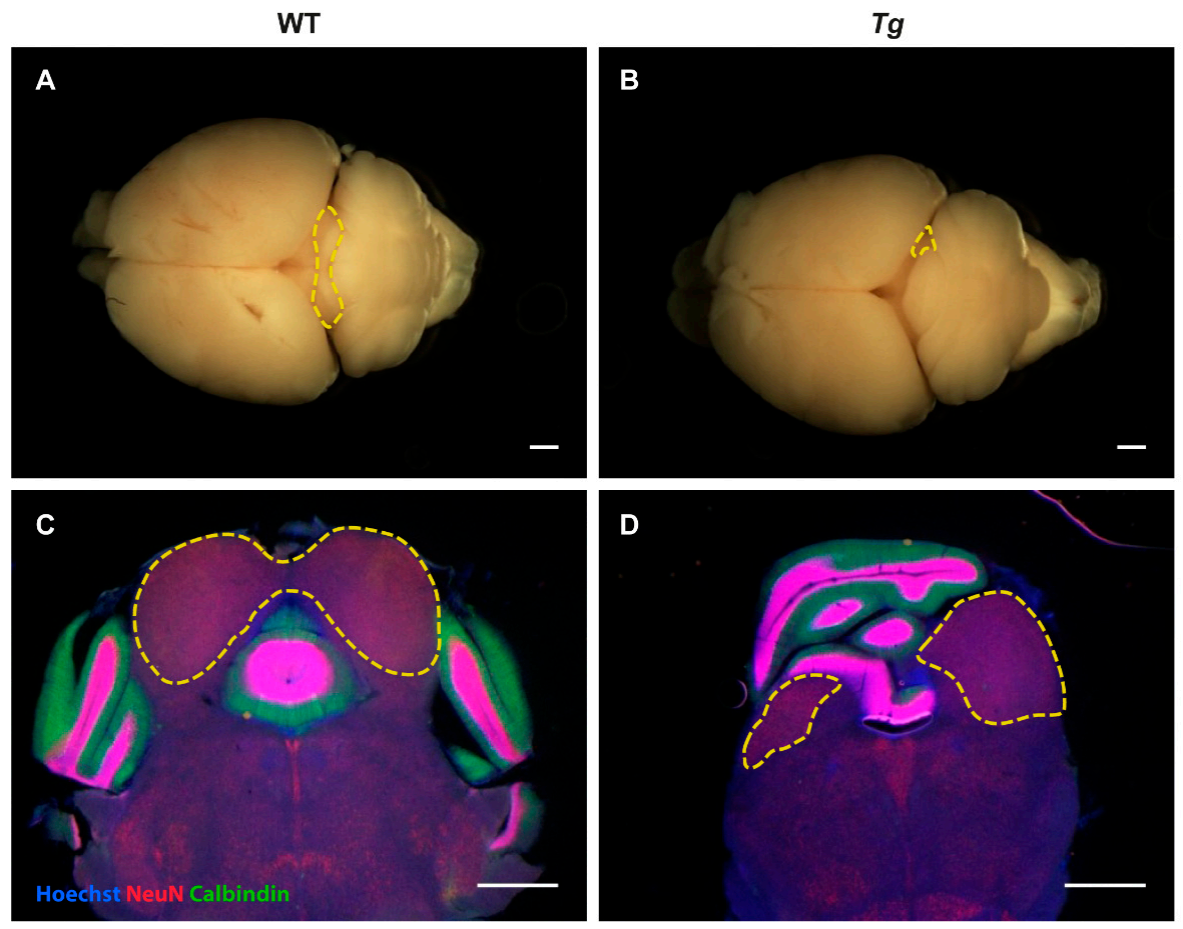
2.2. The Inferior Colliculus Is Smaller in Tg Mice
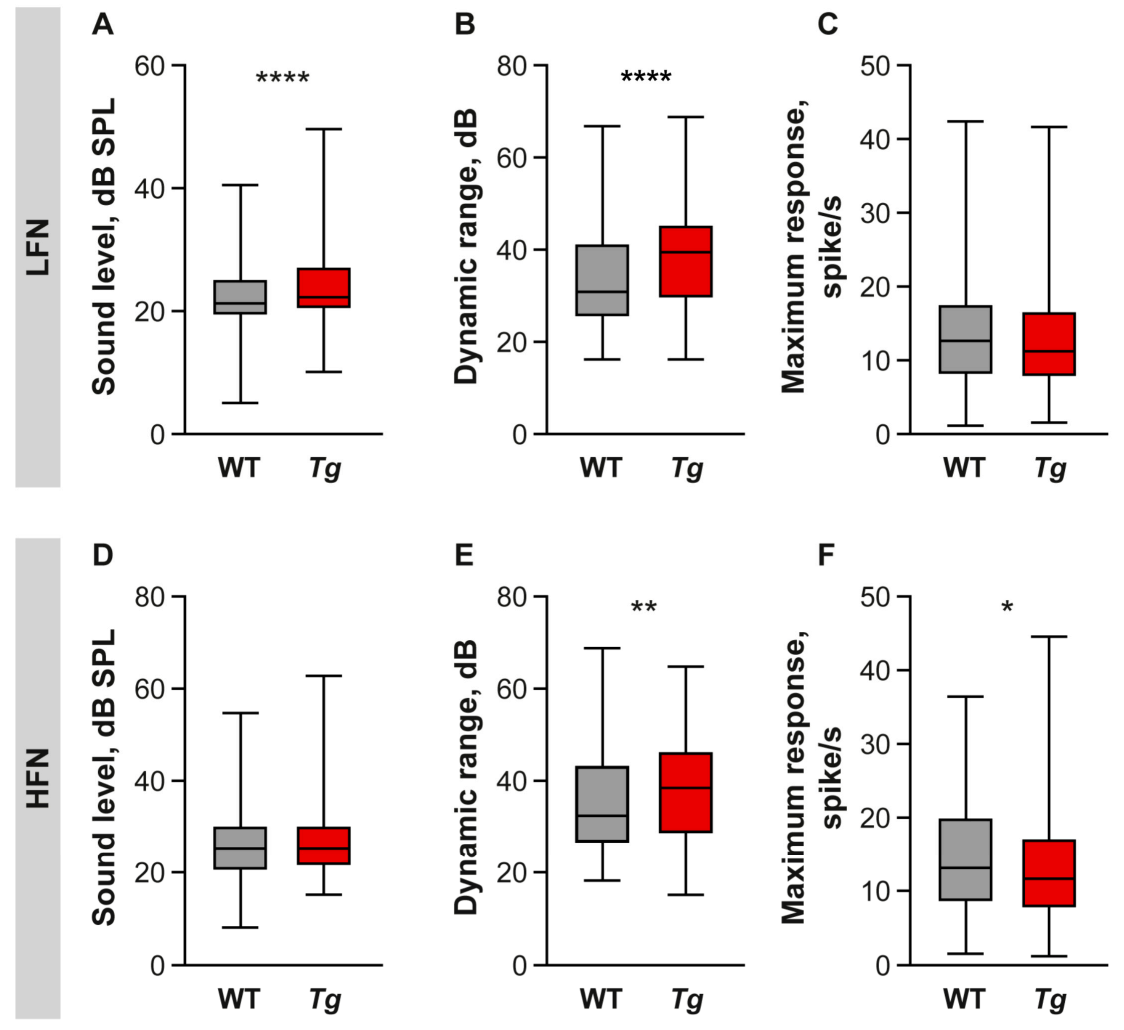
2.3. The Properties of IC Neuronal Responses Are Abnormal in Tg Mice
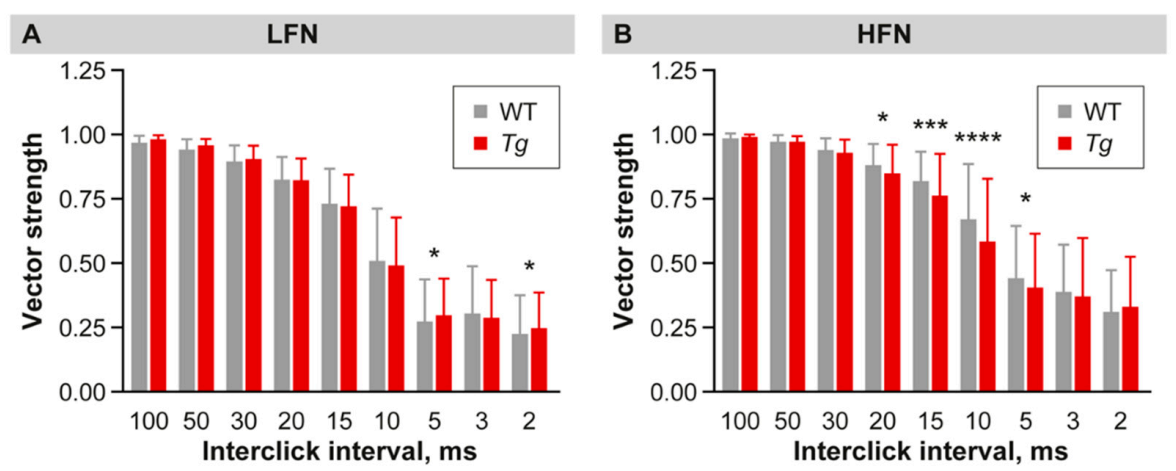
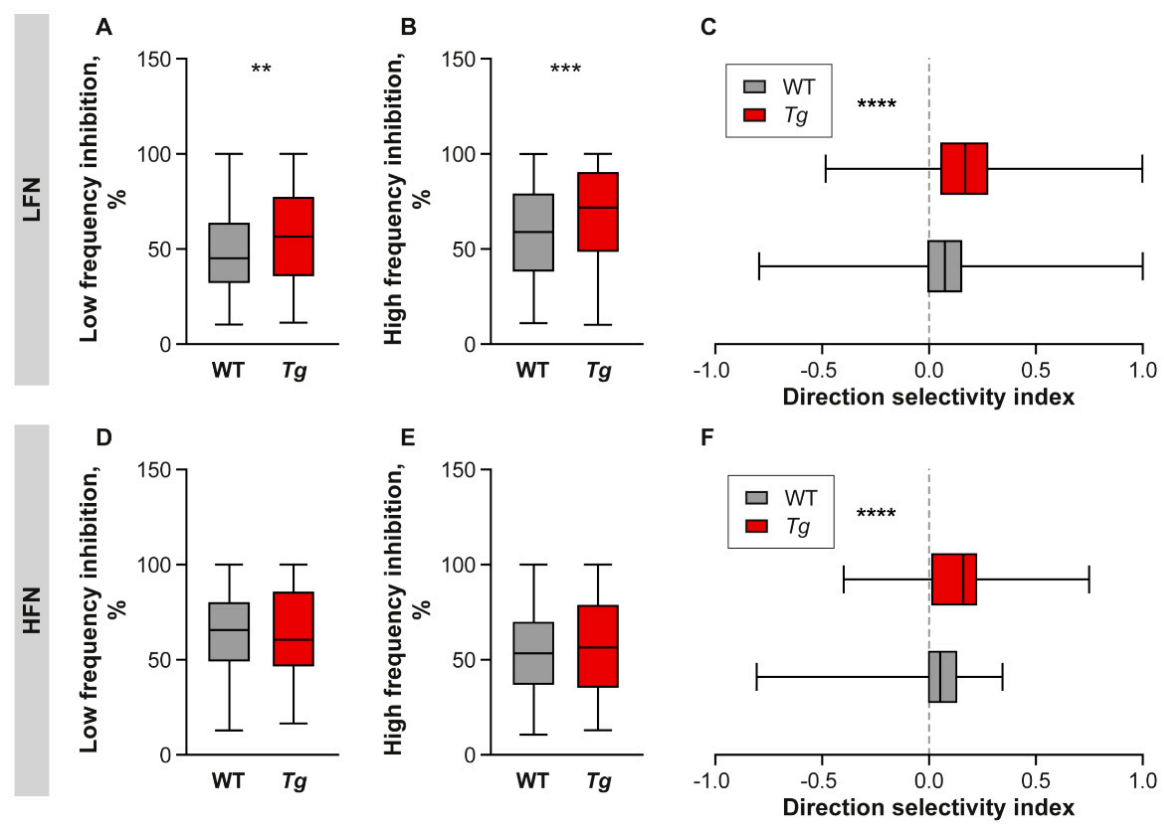
2.4. Inhibition Is Increased in the Inferior Colliculus of Mutant Mice
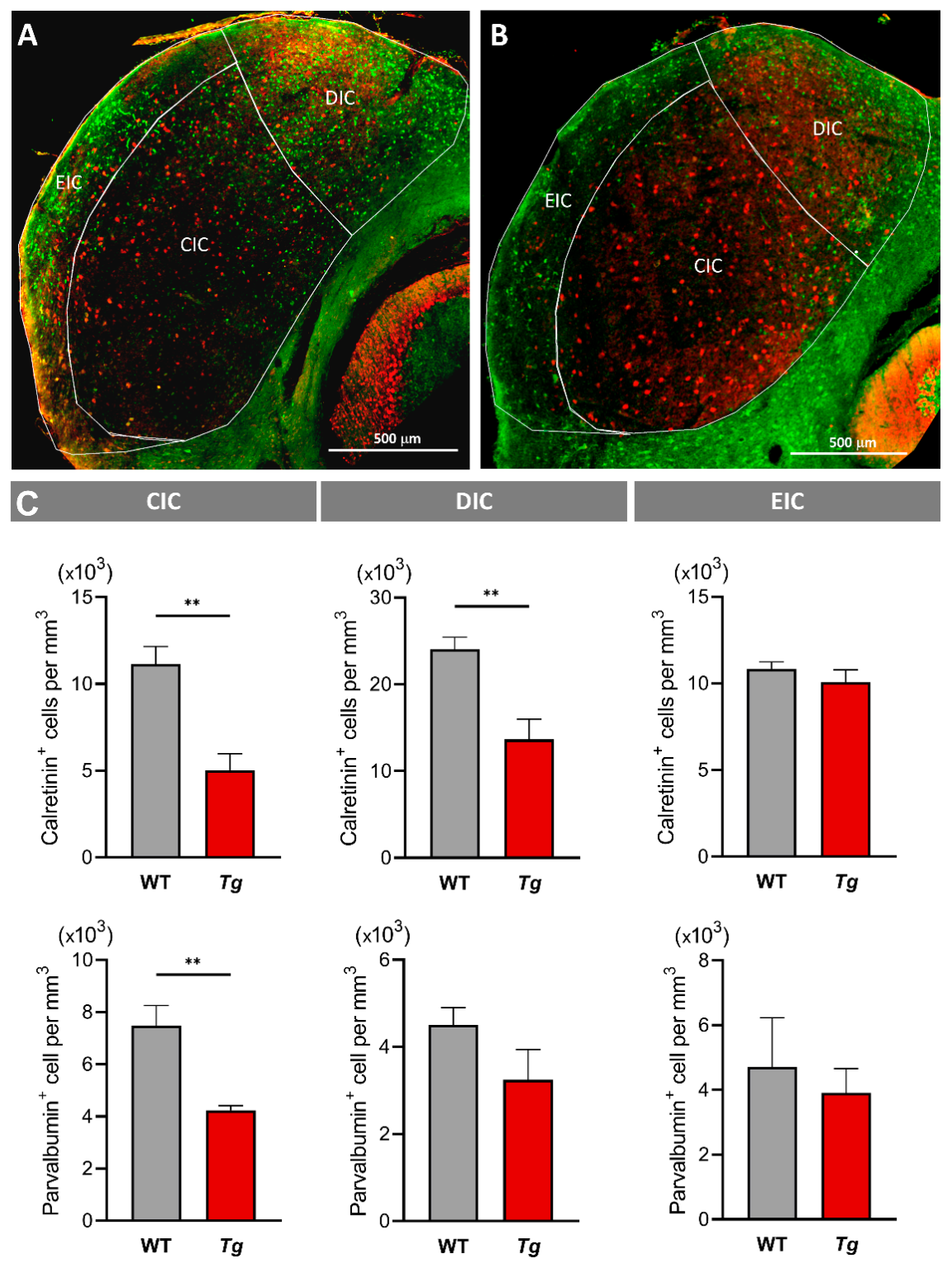
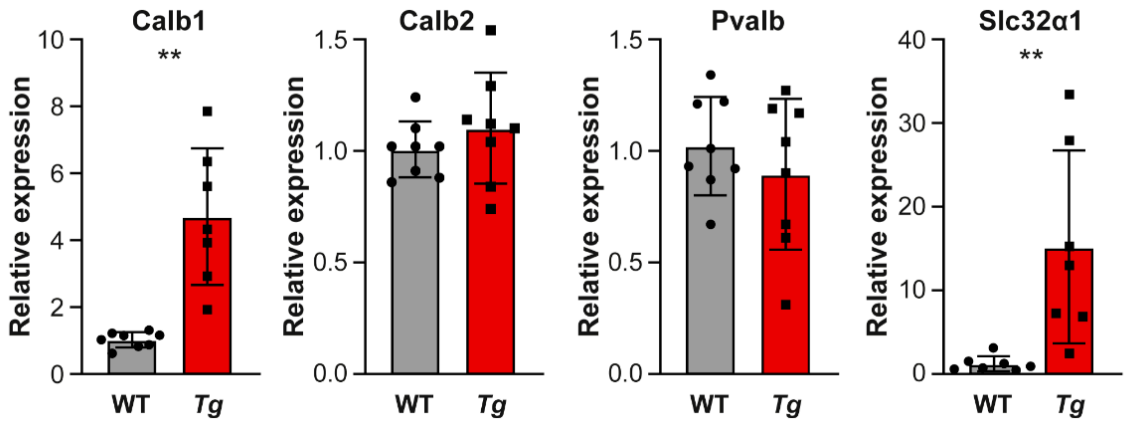
2.5. The Molecular Profile and Distribution of Neurons Are Altered in the Inferior Colliculus of Mutant Mice
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Distortion Product Otoacoustic Emissions
4.3. Auditory Brainstem Responses
4.4. Recording of the Neuronal Activity in the IC
4.5. Immunohistochemistry and Morphological Analysis
4.6. Gene Expression Analysis
4.7. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Felix, R.A.; Gourevitch, B.; Portfors, C.V. Subcortical pathways: Towards a better understanding of auditory disorders. Hear. Res. 2018, 362, 48–60. [Google Scholar] [CrossRef] [PubMed]
- Ono, M.; Bishop, D.C.; Oliver, D.L. Identified GABAergic and Glutamatergic Neurons in the Mouse Inferior Colliculus Share Similar Response Properties. J. Neurosci. 2017, 37, 8952–8964. [Google Scholar] [CrossRef] [PubMed]
- Syka, J.; Popelar, J. Inferior colliculus in the rat: Neuronal responses to stimulation of the auditory cortex. Neurosci. Lett. 1984, 51, 235–240. [Google Scholar] [CrossRef]
- Aitkin, L.M.; Kenyon, C.E.; Philpott, P. The representation of the auditory and somatosensory systems in the external nucleus of the cat inferior colliculus. J. Comp. Neurol. 1981, 196, 25–40. [Google Scholar] [CrossRef]
- Merchan, M.; Aguilar, L.A.; Lopez-Poveda, E.A.; Malmierca, M.S. The inferior colliculus of the rat: Quantitative immunocytochemical study of GABA and glycine. Neuroscience 2005, 136, 907–925. [Google Scholar] [CrossRef]
- Oliver, D.L.; Winer, J.A.; Beckius, G.E.; Saint Marie, R.L. Morphology of GABAergic neurons in the inferior colliculus of the cat. J. Comp. Neurol. 1994, 340, 27–42. [Google Scholar] [CrossRef]
- Ouda, L.; Profant, O.; Syka, J. Age-related changes in the central auditory system. Cell Tissue Res. 2015, 361, 337–358. [Google Scholar] [CrossRef]
- Ouda, L.; Syka, J. Immunocytochemical profiles of inferior colliculus neurons in the rat and their changes with aging. Front. Neural Circuits 2012, 6, 68. [Google Scholar] [CrossRef]
- Pal, I.; Paltati, C.R.B.; Kaur, C.; Saini, S.; Kumar, P.; Jacob, T.G.; Bhardwaj, D.N.; Roy, T.S. Morphological and neurochemical changes in GABAergic neurons of the aging human inferior colliculus. Hear. Res. 2019, 377, 318–329. [Google Scholar] [CrossRef]
- Ono, M.; Ito, T. Inhibitory Neural Circuits in the Mammalian Auditory Midbrain. J. Exp. Neurosci. 2018, 12. [Google Scholar] [CrossRef]
- Pfaff, S.L.; Mendelsohn, M.; Stewart, C.L.; Edlund, T.; Jessell, T.M. Requirement for LIM homeobox gene Isl1 in motor neuron generation reveals a motor neuron-dependent step in interneuron differentiation. Cell 1996, 84, 309–320. [Google Scholar] [CrossRef]
- Sun, Y.; Dykes, I.M.; Liang, X.; Eng, S.R.; Evans, S.M.; Turner, E.E. A central role for Islet1 in sensory neuron development linking sensory and spinal gene regulatory programs. Nat. Neurosci. 2008, 11, 1283–1293. [Google Scholar] [CrossRef]
- Lin, L.; Bu, L.; Cai, C.L.; Zhang, X.; Evans, S. Isl1 is upstream of sonic hedgehog in a pathway required for cardiac morphogenesis. Dev. Biol. 2006, 295, 756–763. [Google Scholar] [CrossRef]
- Cai, C.L.; Liang, X.; Shi, Y.; Chu, P.H.; Pfaff, S.L.; Chen, J.; Evans, S. Isl1 identifies a cardiac progenitor population that proliferates prior to differentiation and contributes a majority of cells to the heart. Dev. Cell 2003, 5, 877–889. [Google Scholar] [CrossRef]
- Elshatory, Y.; Everhart, D.; Deng, M.; Xie, X.; Barlow, R.B.; Gan, L. Islet-1 controls the differentiation of retinal bipolar and cholinergic amacrine cells. J. Neurosci. 2007, 27, 12707–12720. [Google Scholar] [CrossRef]
- Whitney, I.E.; Raven, M.A.; Ciobanu, D.C.; Poche, R.A.; Ding, Q.; Elshatory, Y.; Gan, L.; Williams, R.W.; Reese, B.E. Genetic modulation of horizontal cell number in the mouse retina. Proc. Natl. Acad. Sci. USA 2011, 108, 9697–9702. [Google Scholar] [CrossRef]
- Hobert, O.; Westphal, H. Functions of LIM-homeobox genes. Trends Genet. 2000, 16, 75–83. [Google Scholar] [CrossRef]
- Pfeffer, P.L.; Payer, B.; Reim, G.; di Magliano, M.P.; Busslinger, M. The activation and maintenance of Pax2 expression at the mid-hindbrain boundary is controlled by separate enhancers. Development 2002, 129, 307–318. [Google Scholar] [CrossRef]
- Favor, J.; Sandulache, R.; Neuhauser-Klaus, A.; Pretsch, W.; Chatterjee, B.; Senft, E.; Wurst, W.; Blanquet, V.; Grimes, P.; Sporle, R.; et al. The mouse Pax2(1Neu) mutation is identical to a human PAX2 mutation in a family with renal-coloboma syndrome and results in developmental defects of the brain, ear, eye, and kidney. Proc. Natl. Acad. Sci. USA 1996, 93, 13870–13875. [Google Scholar] [CrossRef]
- Bouchard, M.; de Caprona, D.; Busslinger, M.; Xu, P.; Fritzsch, B. Pax2 and Pax8 cooperate in mouse inner ear morphogenesis and innervation. BMC Dev. Biol. 2010, 10, 89. [Google Scholar] [CrossRef]
- Chumak, T.; Bohuslavova, R.; Macova, I.; Dodd, N.; Buckiova, D.; Fritzsch, B.; Syka, J.; Pavlinkova, G. Deterioration of the Medial Olivocochlear Efferent System Accelerates Age-Related Hearing Loss in Pax2-Isl1 Transgenic Mice. Mol. Neurobiol. 2016, 53, 2368–2383. [Google Scholar] [CrossRef] [PubMed]
- Bohuslavova, R.; Dodd, N.; Macova, I.; Chumak, T.; Horak, M.; Syka, J.; Fritzsch, B.; Pavlinkova, G. Pax2-Islet1 Transgenic Mice Are Hyperactive and Have Altered Cerebellar Foliation. Mol. Neurobiol. 2017, 54, 1352–1368. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.; Jen, P.H.; Seburn, K.L.; Frankel, W.N.; Zheng, Q.Y. Auditory brainstem responses in 10 inbred strains of mice. Brain Res. 2006, 1091, 16–26. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Q.Y.; Johnson, K.R.; Erway, L.C. Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hear. Res. 1999, 130, 94–107. [Google Scholar] [CrossRef]
- Melcher, J.R.; Guinan, J.J., Jr.; Knudson, I.M.; Kiang, N.Y. Generators of the brainstem auditory evoked potential in cat. II. Correlating lesion sites with waveform changes. Hear. Res. 1996, 93, 28–51. [Google Scholar] [CrossRef]
- Land, R.; Burghard, A.; Kral, A. The contribution of inferior colliculus activity to the auditory brainstem response (ABR) in mice. Hear. Res. 2016, 341, 109–118. [Google Scholar] [CrossRef]
- Tan, X.; Wang, X.; Yang, W.; Xiao, Z. First spike latency and spike count as functions of tone amplitude and frequency in the inferior colliculus of mice. Hear. Res. 2008, 235, 90–104. [Google Scholar] [CrossRef]
- Chumak, T.; Ruttiger, L.; Lee, S.C.; Campanelli, D.; Zuccotti, A.; Singer, W.; Popelar, J.; Gutsche, K.; Geisler, H.S.; Schraven, S.P.; et al. BDNF in Lower Brain Parts Modifies Auditory Fiber Activity to Gain Fidelity but Increases the Risk for Generation of Central Noise After Injury. Mol. Neurobiol. 2016, 53, 5607–5627. [Google Scholar] [CrossRef]
- Kuo, R.I.; Wu, G.Y.K. The Generation of Direction Selectivity in the Auditory System. Neuron 2012, 73, 1016–1027. [Google Scholar] [CrossRef]
- Pollak, G.D.; Xie, R.L.; Gittelman, J.X.; Andoni, S.; Li, N. The dominance of inhibition in the inferior colliculus. Hear. Res. 2011, 274, 27–39. [Google Scholar] [CrossRef]
- Idrizbegovic, E.; Bogdanovic, N.; Canlon, B. Sound stimulation increases calcium-binding protein immunoreactivity in the inferior colliculus in mice. Neurosci. Lett. 1999, 259, 49–52. [Google Scholar] [CrossRef]
- Fredrich, M.; Reisch, A.; Illing, R.B. Neuronal subtype identity in the rat auditory brainstem as defined by molecular profile and axonal projection. Exp. Brain Res. 2009, 195, 241–260. [Google Scholar] [CrossRef]
- Xie, R.; Meitzen, J.; Pollak, G.D. Differing roles of inhibition in hierarchical processing of species-specific calls in auditory brainstem nuclei. J. Neurophysiol. 2005, 94, 4019–4037. [Google Scholar] [CrossRef]
- D’Angelo, W.R.; Sterbing, S.J.; Ostapoff, E.M.; Kuwada, S. Role of GABAergic inhibition in the coding of interaural time differences of low-frequency sounds in the inferior colliculus. J. Neurophysiol. 2005, 93, 3390–3400. [Google Scholar] [CrossRef]
- Palmer, A.R.; Shackleton, T.M.; Sumner, C.J.; Zobay, O.; Rees, A. Classification of frequency response areas in the inferior colliculus reveals continua not discrete classes. J. Physiol. 2013, 591, 4003–4025. [Google Scholar] [CrossRef]
- Alkhatib, A.; Biebel, U.W.; Smolders, J.W. Inhibitory and excitatory response areas of neurons in the central nucleus of the inferior colliculus in unanesthetized chinchillas. Exp. Brain Res. 2006, 174, 124–143. [Google Scholar] [CrossRef]
- Egorova, M.; Ehret, G.; Vartanian, I.; Esser, K.H. Frequency response areas of neurons in the mouse inferior colliculus. I. Threshold and tuning characteristics. Exp. Brain Res. 2001, 140, 145–161. [Google Scholar] [CrossRef]
- Le Beau, F.E.; Rees, A.; Malmierca, M.S. Contribution of GABA- and glycine-mediated inhibition to the monaural temporal response properties of neurons in the inferior colliculus. J. Neurophysiol. 1996, 75, 902–919. [Google Scholar] [CrossRef]
- LeBeau, F.E.; Malmierca, M.S.; Rees, A. Iontophoresis in vivo demonstrates a key role for GABA(A) and glycinergic inhibition in shaping frequency response areas in the inferior colliculus of guinea pig. J. Neurosci. 2001, 21, 7303–7312. [Google Scholar] [CrossRef]
- Yang, L.; Pollak, G.D.; Resler, C. GABAergic circuits sharpen tuning curves and modify response properties in the mustache bat inferior colliculus. J. Neurophysiol. 1992, 68, 1760–1774. [Google Scholar] [CrossRef]
- Williams, A.J.; Fuzessery, Z.M. Differential roles of GABAergic and glycinergic input on FM selectivity in the inferior colliculus of the pallid bat. J. Neurophysiol. 2011, 106, 2523–2535. [Google Scholar] [CrossRef][Green Version]
- Park, T.J.; Pollak, G.D. GABA shapes a topographic organization of response latency in the mustache bat’s inferior colliculus. J. Neurosci. 1993, 13, 5172–5187. [Google Scholar] [CrossRef]
- Palombi, P.S.; Caspary, D.M. GABA inputs control discharge rate primarily within frequency receptive fields of inferior colliculus neurons. J. Neurophysiol. 1996, 75, 2211–2219. [Google Scholar] [CrossRef]
- Grimsley, C.A.; Sanchez, J.T.; Sivaramakrishnan, S. Midbrain local circuits shape sound intensity codes. Front. Neural Circuit 2013, 7, 174. [Google Scholar] [CrossRef]
- Andressen, C.; Blumcke, I.; Celio, M.R. Calcium-binding proteins: Selective markers of nerve cells. Cell Tissue Res. 1993, 271, 181–208. [Google Scholar] [CrossRef]
- Baimbridge, K.G.; Celio, M.R.; Rogers, J.H. Calcium-binding proteins in the nervous system. Trends Neurosci. 1992, 15, 303–308. [Google Scholar] [CrossRef]
- Fuentes-Santamaria, V.; Alvarado, J.C.; Brunso-Bechtold, J.K.; Henkel, C.K. Upregulation of calretinin immunostaining in the ferret inferior colliculus after cochlear ablation. J. Comp. Neurol. 2003, 460, 585–596. [Google Scholar] [CrossRef]
- Bures, Z.; Bartosova, J.; Lindovsky, J.; Chumak, T.; Popelar, J.; Syka, J. Acoustical enrichment during early postnatal development changes response properties of inferior colliculus neurons in rats. Eur. J. Neurosci. 2014, 40, 3674–3683. [Google Scholar] [CrossRef]
- Grecova, J.; Bures, Z.; Popelar, J.; Suta, D.; Syka, J. Brief exposure of juvenile rats to noise impairs the development of the response properties of inferior colliculus neurons. Eur. J. Neurosci. 2009, 29, 1921–1930. [Google Scholar] [CrossRef]
- Bures, Z.; Pysanenko, K.; Lindovsky, J.; Syka, J. Acoustical Enrichment during Early Development Improves Response Reliability in the Adult Auditory Cortex of the Rat. Neural Plast. 2018, 2018, 5903720. [Google Scholar] [CrossRef] [PubMed]
- Ye, C.Q.; Poo, M.M.; Dan, Y.; Zhang, X.H. Synaptic mechanisms of direction selectivity in primary auditory cortex. J. Neurosci. 2010, 30, 1861–1868. [Google Scholar] [CrossRef] [PubMed]
- Paxinos, G.; Franklin, K.B.J. The Mouse Brain in Stereotaxic Coordinates, 2nd ed.; Academic Press: San Diego, CA, USA, 2001. [Google Scholar]
- Bohuslavova, R.; Skvorova, L.; Sedmera, D.; Semenza, G.L.; Pavlinkova, G. Increased susceptibility of HIF-1alpha heterozygous-null mice to cardiovascular malformations associated with maternal diabetes. J. Mol. Cell Cardiol. 2013, 60, 129–141. [Google Scholar] [CrossRef] [PubMed]


| Hprt1 F | GCTTGCTGGTGAAAAGGACCTCTCGAAG |
| Hprt1 R | CCCTGAAGTACTCATTATAGTCAAGGGCAT |
| Calb1 F | GATTGGAGCTATCACCGGAA |
| Calb1 R | TTCCTCGCAGGACTTCAGTT |
| Calb2 F | TGATGCTGACGGAAATGGGT |
| Calb2 R | CCCTTCCTTGCCTTCTCCAG |
| Pvalb F | ATCAAGAAGGCGATAGGAGCC |
| Pvalb R | GGCCAGAAGCGTCTTTGTT |
| Slc32a1 F | ACCTCCGTGTCCAACAAGTC |
| Slc32a1 R | CAAAGTCGAGATCGTCGCAGT |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chumak, T.; Tothova, D.; Filova, I.; Bures, Z.; Popelar, J.; Pavlinkova, G.; Syka, J. Overexpression of Isl1 under the Pax2 Promoter, Leads to Impaired Sound Processing and Increased Inhibition in the Inferior Colliculus. Int. J. Mol. Sci. 2021, 22, 4507. https://doi.org/10.3390/ijms22094507
Chumak T, Tothova D, Filova I, Bures Z, Popelar J, Pavlinkova G, Syka J. Overexpression of Isl1 under the Pax2 Promoter, Leads to Impaired Sound Processing and Increased Inhibition in the Inferior Colliculus. International Journal of Molecular Sciences. 2021; 22(9):4507. https://doi.org/10.3390/ijms22094507
Chicago/Turabian StyleChumak, Tetyana, Diana Tothova, Iva Filova, Zbynek Bures, Jiri Popelar, Gabriela Pavlinkova, and Josef Syka. 2021. "Overexpression of Isl1 under the Pax2 Promoter, Leads to Impaired Sound Processing and Increased Inhibition in the Inferior Colliculus" International Journal of Molecular Sciences 22, no. 9: 4507. https://doi.org/10.3390/ijms22094507
APA StyleChumak, T., Tothova, D., Filova, I., Bures, Z., Popelar, J., Pavlinkova, G., & Syka, J. (2021). Overexpression of Isl1 under the Pax2 Promoter, Leads to Impaired Sound Processing and Increased Inhibition in the Inferior Colliculus. International Journal of Molecular Sciences, 22(9), 4507. https://doi.org/10.3390/ijms22094507







