Glass and Jamming Rheology in Soft Particles Made of PNIPAM and Polyacrylic Acid
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Microgel Synthesis
2.3. Rheological Measurements
2.4. Steady Shear Measurements
2.4.1. Cross and Carreau-Yasuda Models for Viscous Liquids
2.4.2. Yield Stress Materials: The Herschel-Bulkley Model
2.5. Oscillatory Measurements: Storage and Loss Moduli
2.6. Arrhenius and Vogel-Fulcher-Tamman Models for the Concentration Dependence of Viscosity and Relaxation Time
2.7. Characteristic Stress and Time Scales
2.8. Dynamic Light Scattering Measurements
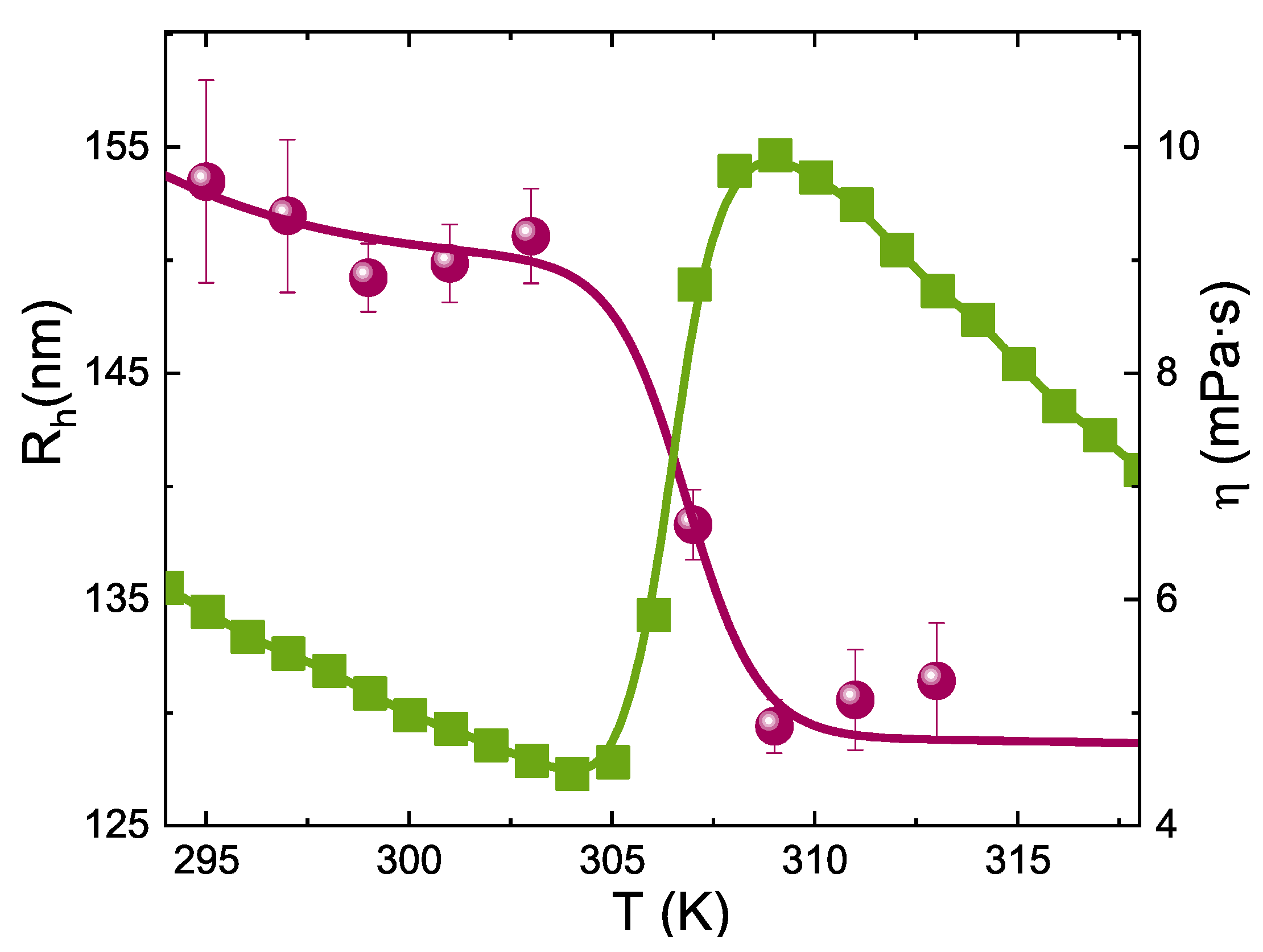
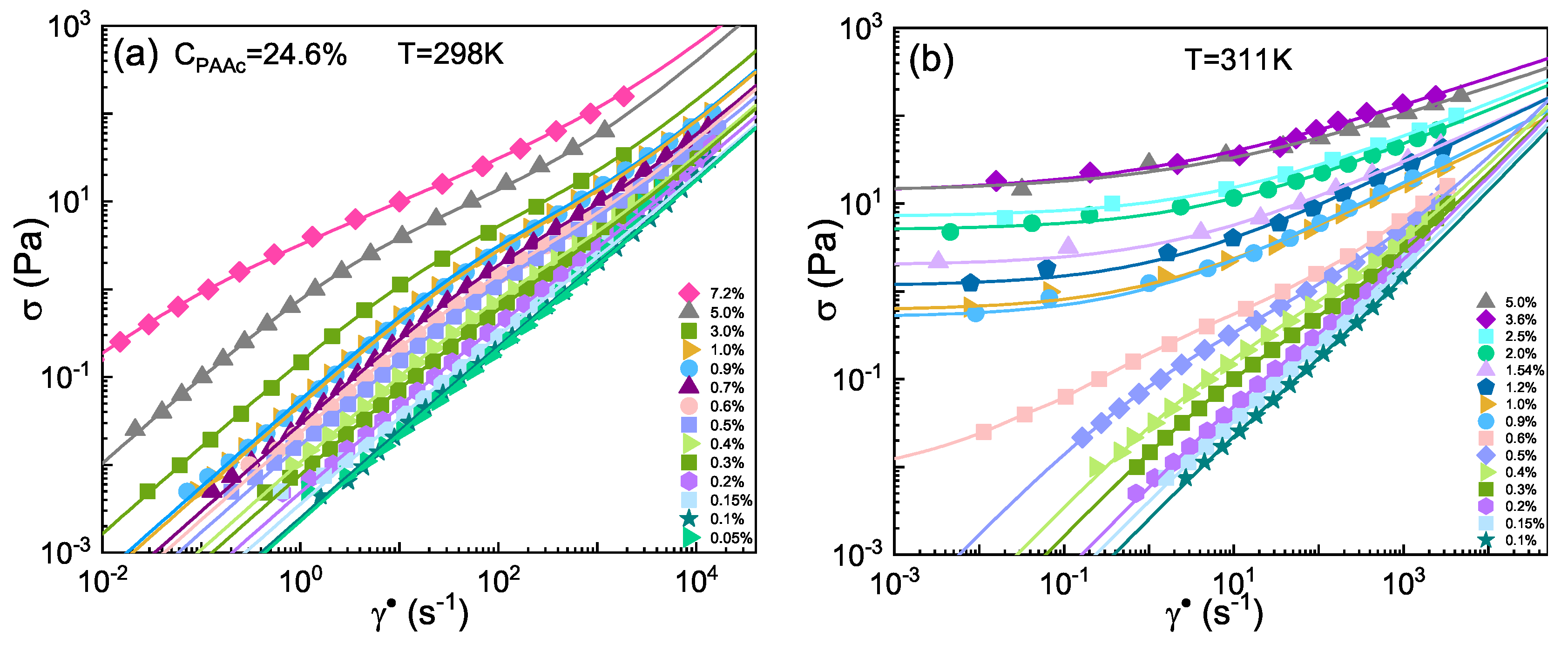
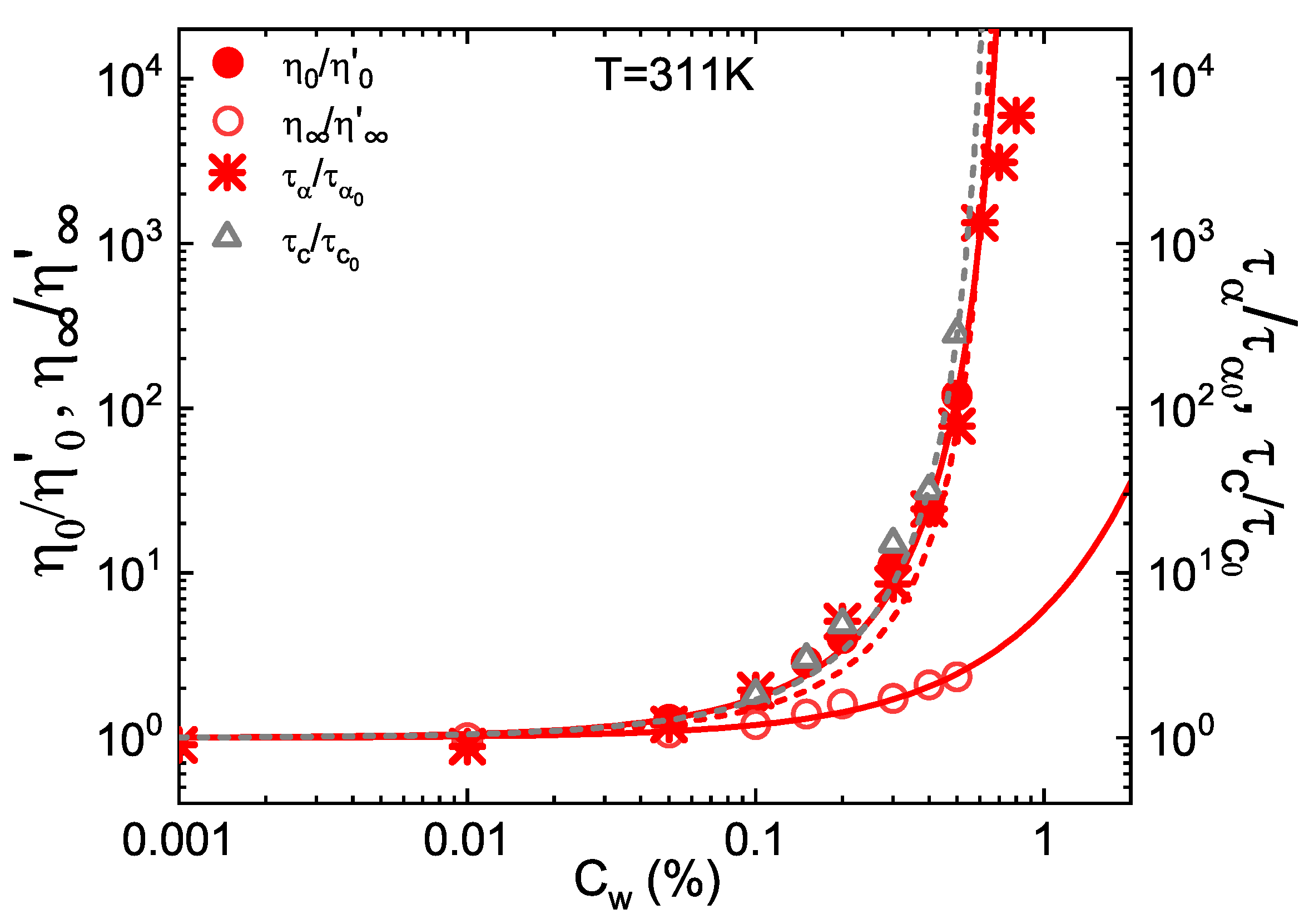
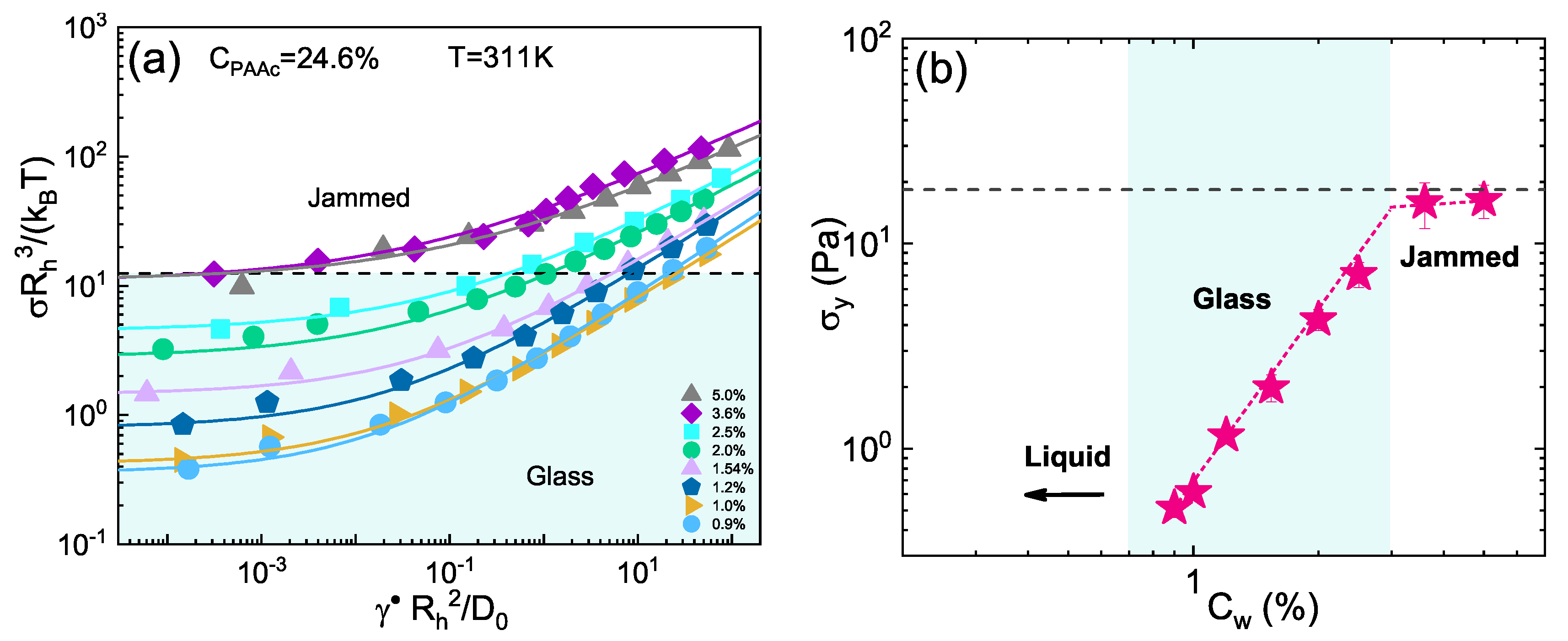
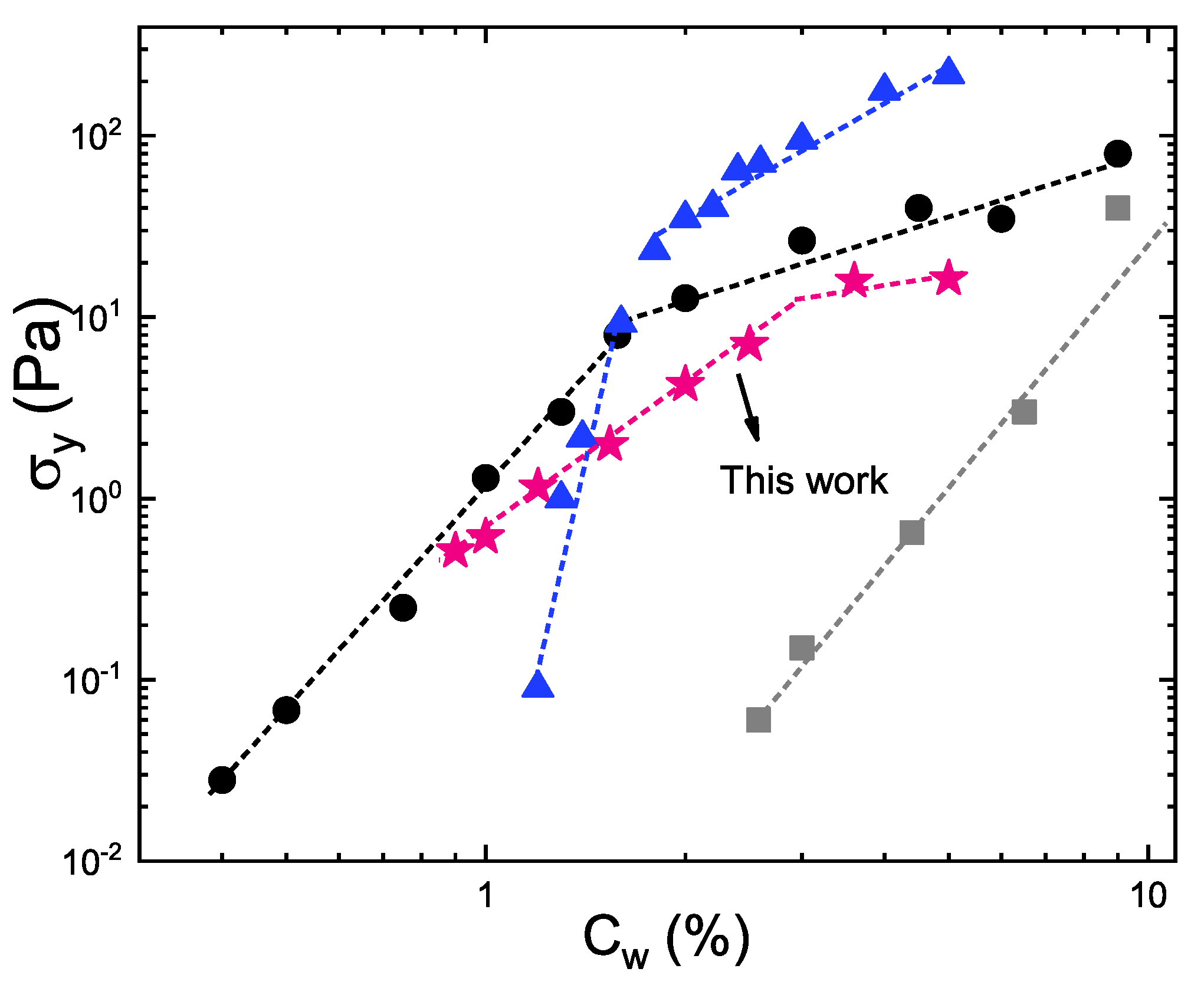
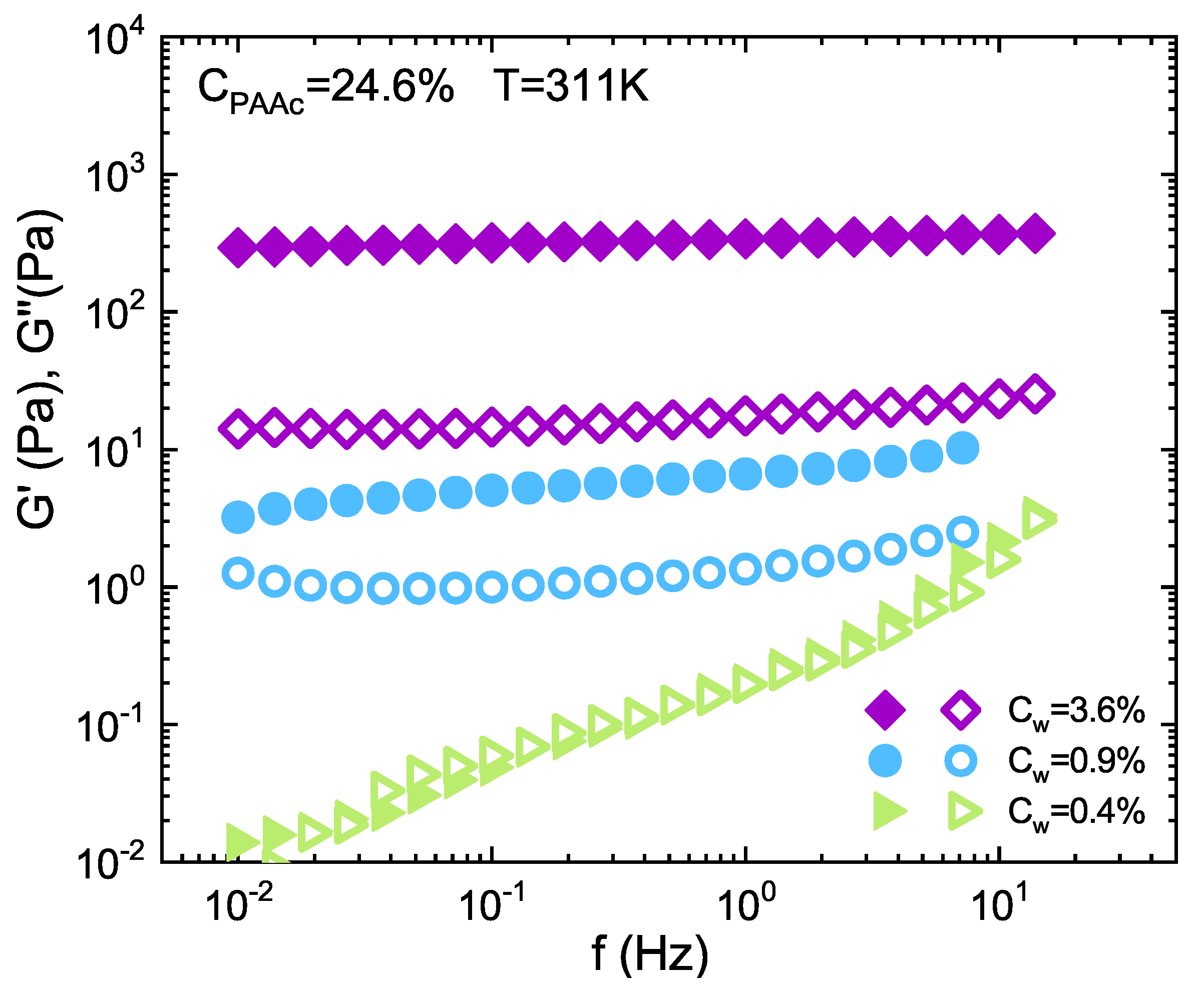
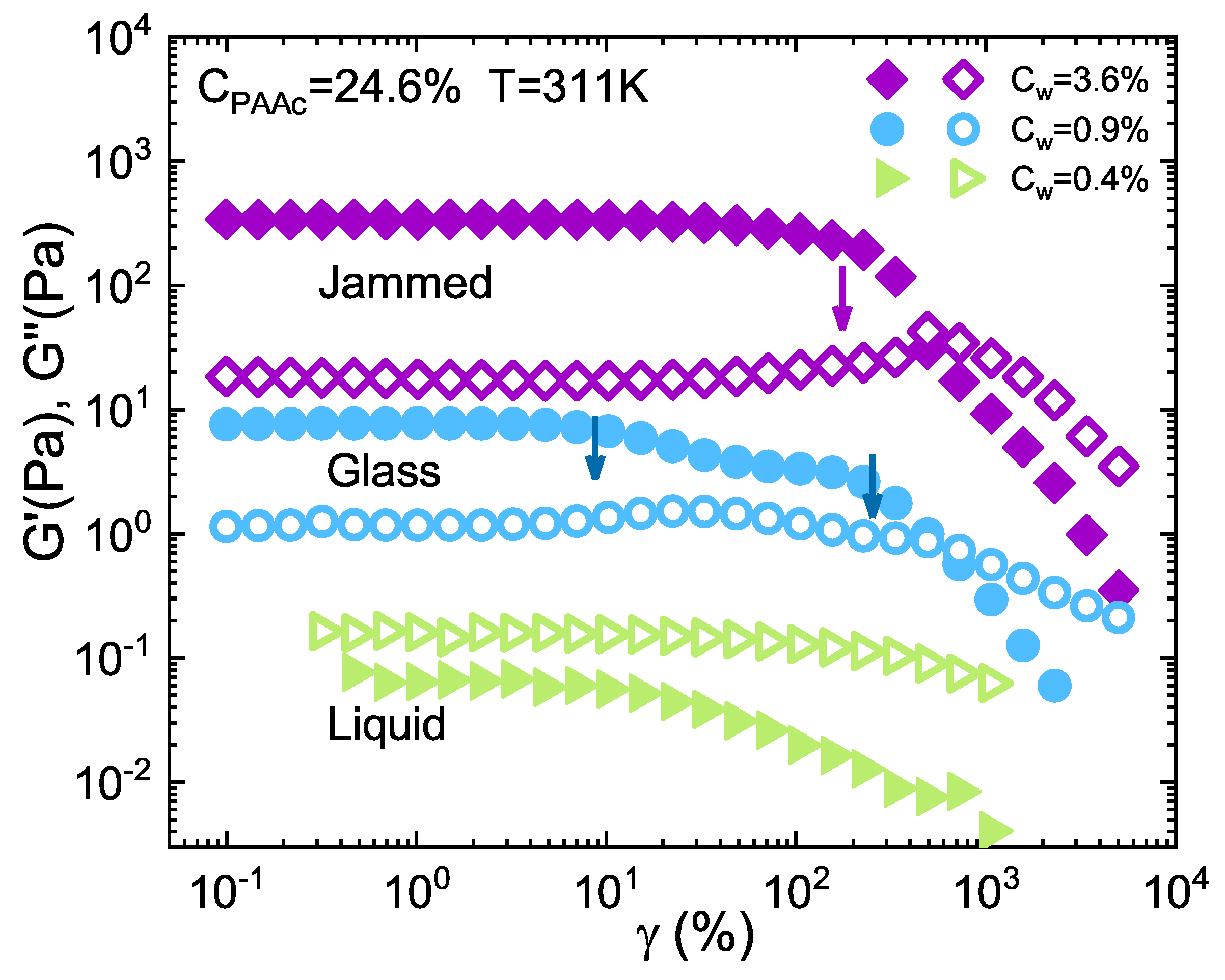
3. Results and Discussion
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Pusey, P.N.; van Megen, W. Phase Behavior of Concentrated Suspensions of Nearly Hard Colloidal Spheres. Nature 1986, 320, 340–342. [Google Scholar] [CrossRef]
- Pusey, P.N.; van Megen, W. Observation of a glass transition in suspensions of spherical colloidal particles. Phys. Rev. Lett. 1987, 59, 2083. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.F. Model hard-sphere dispersions: Statistical mechanical theory, simulations, and experiments. Curr. Opin. Colloid Interface Sci. 1996, 1, 472–480. [Google Scholar] [CrossRef]
- Pham, K.; Petekidis, G.; Vlassopoulos, D.; Egelhaaf, S.U.; Poon, W.C.K.; Pusey, P.N. Yielding behaviour of repulsion- and attraction-dominated colloidal glasses. J. Rheol. 2008, 52, 649. [Google Scholar] [CrossRef]
- Royall, C.P.; Poon, W.C.K.; Weeks, E.R. In search of colloidal hard spheres. Soft Matter 2013, 9, 17–27. [Google Scholar] [CrossRef]
- Senff, H.; Richtering, W. Temperature sensitive microgel suspensions: Colloidal phase behavior and rheology of soft spheres. J. Chem. Phys. 1999, 111, 1705–1711. [Google Scholar] [CrossRef]
- Likos, C.N. Effective interactions in soft condensed matter physics. Phys. Rep. 2001, 348, 267–439. [Google Scholar] [CrossRef]
- Crassous, J.J.; Siebenbürger, M.; Ballauff, M.; Drechsler, M.; Hajnal, D.; Henrich, O.; Fuchs, M. Shear stresses of colloidal dispersions at the glass transition in equilibrium and in flow. J. Chem. Phys. 2008, 120, 204902. [Google Scholar] [CrossRef] [PubMed]
- Mattsson, J.; Wyss, H.M.; Fernandez-Nieves, A.; Miyazaki, K.; Hu, Z.; Reichman, D.; Weitz, D.A. Soft colloids make strong glasses. Nature 2009, 462, 83–86. [Google Scholar] [CrossRef] [PubMed]
- Fernandez-Nieves, A.; Wyss, H.M.; Mattsson, J.; Weitz, D. Microgel Suspensions: Fundamental and Applications; Wiley-VCH Verlag: Hoboken, NJ, USA, 2011. [Google Scholar]
- Lyon, L.A.; Fernandez-Nieves, A. The Polymer/Colloid Duality of Microgel Suspensions. Annu. Rev. Phys. Chem. 2012, 63, 25–43. [Google Scholar] [CrossRef] [PubMed]
- Vlassopoulos, D.; Cloitre, M. Tunable rheology of dense soft deformable colloids. Curr. Opin. Colloid Interface Sci. 2014, 19, 561–574. [Google Scholar] [CrossRef]
- Bonn, D.; Denn, M.M.; Berthier, L.; Divoux, T.; Manneville, S. Yield stress materials in soft condensed matter. Phys. Rev. Lett. 2017, 89, 1035005–1035040. [Google Scholar] [CrossRef]
- Nigro, V.; Angelini, R.; Bertoldo, M.; Bruni, F.; Ricci, M.; Ruzicka, B. Dynamical behavior of microgels of Interpenetrated Polymer Networks. Soft Matter 2017, 13, 5185–5193. [Google Scholar] [CrossRef] [PubMed]
- Bergman, M.J.; Gnan, N.; Obiols-Rabasa, M.; Meijer, J.M.; Rovigatti, L.; Zaccarelli, E.; Schurtenberger, P. A new look at effective interactions between microgel particles. Nat. Commun. 2018, 9, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Karg, M.; Pich, A.; Hellweg, T.; Hoare, T.; Lyon, L.A.; Crassous, J.J.; Suzuki, D.; Gumerov, R.A.; Schneider, S.; Potemkin, I.I.; et al. Microgels: From Model Colloids to Applications, Recent Developments, and Future Trends. Langmuir 2019, 35, 6231–6255. [Google Scholar] [CrossRef] [PubMed]
- Gnan, N.; Zaccarelli, E. The microscopic role of deformation in the dynamics of soft colloids. Nat. Phys. 2019, 15, 683–688. [Google Scholar] [CrossRef]
- Ninarello, A.; Crassous, J.J.; Paloli, D.; Camerin, F.; Gnan, N.; Rovigatti, L.; Schurtenberger, P.; Zaccarelli, E. Modeling Microgels with a Controlled Structure across the Volume Phase Transition. Macromolecules 2019, 52, 7584–7592. [Google Scholar] [CrossRef] [PubMed]
- Nigro, V.; Ruzicka, B.; Ruta, B.; Zontone, F.; Bertoldo, M.; Buratti, E.; Angelini, R. Relaxation Dynamics, Softness, and Fragility of Microgels with Interpenetrated Polymer Networks. Macromolecules 2020, 53, 1596–1603. [Google Scholar] [CrossRef]
- Ikeda, A.; Berthier, L.; Sollich, P. Disentangling glass and jamming physics in the rheology of soft materials. Soft Matter 2013, 9, 17669–17683. [Google Scholar] [CrossRef]
- Olsson, P.; Teitel, S. Critical Scaling of Shear Viscosity at the Jamming Transition. Phys. Rev. Lett. 2007, 99, 178001–178004. [Google Scholar] [CrossRef] [PubMed]
- O’Hern, C.S.; Silbert, L.E.; Liu, A.J.; Nagel, S.R. Jamming at zero temperature and zero applied stress: The epitome of disorder. Phys. Rev. E 2003, 68, 011306. [Google Scholar] [CrossRef] [PubMed]
- Pellet, C.; Cloitre, M. The glass and jamming transitions of soft polyelectrolyte microgel suspensions. Soft Matter 2016, 12, 3710–3720. [Google Scholar] [CrossRef] [PubMed]
- Scotti, A.; Brugnoni, M.; Lopez, C.G.; Bochenek, S.; Crassous, J.J.; Richtering, W. Flow properties reveal the particle-to-polymer transition of ultra-low crosslinked microgels. Soft Matter 2020, 16, 668–678. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, A.; Chaudhary, G.; Kang, J.G.; Braun, P.V.; Ewoldt, R.H.; Schweizer, K.S. Linear and nonlinear rheology and structural relaxation in dense glassy and jammed soft repulsive pNIPAM microgel suspensions. Soft Matter 2019, 15, 1038–1052. [Google Scholar] [CrossRef] [PubMed]
- Braibanti, M.; Kim, H.S.; Şenbil, N.; Pagenkopp, M.J.; Mason, T.G.; Scheffold, F. The liquid-glass-jamming transition in disordered ionic nanoemulsions. Sci. Rep. 2017, 7, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Tighe, B.P.; Woldhuis, E.; Remmers, J.J.C.; van Saarloos, W.; van Hecke, M. Model for the Scaling of Stresses and Fluctuations in Flows near Jamming. Phys. Rev. Lett. 2010, 105, 088303. [Google Scholar] [CrossRef]
- Lerner, E.; Düring, G.; Wyart, M. A unified framework for non-Brownian suspension flows and soft amorphous solids. Proc. Natl. Acad. Sci. USA 2012, 109, 4798–4803. [Google Scholar] [CrossRef] [PubMed]
- Olsson, P.; Teitel, S. Herschel-Bulkley Shearing Rheology Near the Athermal Jamming Transition. Phys. Rev. Lett. 2012, 109, 108001. [Google Scholar] [CrossRef] [PubMed]
- Charbonneau, P.; Kurchan, J.; Parisi, G.; Urbani, P.; Zamponi, F. Glass and Jamming Transitions: From Exact Results to Finite-Dimensional Descriptions. Ann. Rev. Condens. Matter Phys. 2016, 8, 1–26. [Google Scholar] [CrossRef]
- Pusey, P.N. Colloidal glasses. J. Phys. Condens. Matter 2008, 20, 494202. [Google Scholar] [CrossRef]
- Conley, G.M.; Zhang, C.; Aebischer1, P.; Harden, J.L.; Scheffold, F. Relationship between rheology and structure of interpenetrating, deforming and compressing microgels. Nat. Commun. 2019, 10, 2436. [Google Scholar] [CrossRef] [PubMed]
- Petekidis, G.; Vlassopoulos, D.; Pusey, P.N. Yielding and flow of sheared colloidal glasses. J. Phys. Condens. Matter 2004, 16, S3955–S3963. [Google Scholar] [CrossRef]
- Karg, M.; Hellweg, T.; Mulvaney, P. Self-Assembly of Tunable Nanocrystal Superlattices Using Poly-(NIPAM) Spacers. Adv. Funct. Mater. 2011, 21, 4668–4676. [Google Scholar] [CrossRef]
- Philippe, A.M.; Truzzolillo, D.; Galvan-Myoshi, J.; Dieudonné-George, P.; Trappe, V.; Berthier, L.; Cipelletti, L. Glass transition of soft colloids. Phys. Rev. E 2018, 97, 040601. [Google Scholar] [CrossRef]
- Dinkgreve, M.; Paredes, J.; Michels, M.A.J.; Bonn, D. Universal rescaling of flow curves for yield-stress fluids close to jamming. Phys. Rev. E 2015, 92, 012305. [Google Scholar] [CrossRef]
- Mason, T.G.; Bibette, J.; Weitz, D.A. Elasticity of Compressed Emulsions. Phys. Rev. Lett. 1995, 75, 2051–2054. [Google Scholar] [CrossRef]
- Shao, Z.; Negi, A.S.; Osuji, C.O. Role of interparticle attraction in the yielding response of microgel suspensions. Soft Matter 2013, 9, 5492. [Google Scholar] [CrossRef]
- Coussot, P.; Ovarlez, G. Physical origin of shear-banding in jammed systems. Eur. Phys. J. E 2010, 33, 183–188. [Google Scholar] [CrossRef] [PubMed]
- Rovigatti, L.; Gnan, N.; Tavagnacco, L.; Moreno, A.J.; Zaccarelli, E. Numerical modelling of non-ionic microgels: An overview. Soft Matter 2019, 15, 1108–1119. [Google Scholar] [CrossRef] [PubMed]
- Stieger, M.; Pedersen, J.S.; Lindner, P.; Richtering, W. Are Thermoresponsive Microgels Model Systems for Concentrated Colloidal Suspensions? A Rheology and Small-Angle Neutron Scattering Study. Langmuir 2004, 20, 7283–7292. [Google Scholar] [CrossRef] [PubMed]
- Vinogradov, S.V. Colloidal microgels in drug delivery applications. Curr. Pharm. Des. 2006, 12, 4703–4712. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Wang, G.; Zou, L.; Tang, L.; Marquez, M.; Hu, Z. Viscoelastic Behavior and In Vivo Release Study of Microgel Dispersions with Inverse Thermoreversible Gelation. Biomacromolecules 2008, 9, 142–148. [Google Scholar] [CrossRef]
- Baglioni, M.; Giorgi, R.; Berti, D.; Baglioni, P. Smart cleaning of cultural heritage: A new challenge for soft nanoscience. Nanoscale 2012, 4, 42–53. [Google Scholar] [CrossRef] [PubMed]
- Park, J.S.; Yang, H.N.; Woo, D.G.; Jeon, S.Y.; Park, K.H. Poly(N-isopropylacrylamide-co-acrylic acid) nanogels for tracing and delivering genes to human mesenchymal stem cells. Biomaterials 2013, 34, 8819–8834. [Google Scholar] [CrossRef] [PubMed]
- Napoli, B.D.; Franco, S.; Severini, L.; Tumiati, M.; Buratti, E.; Titubante, M.; Nigro, V.; Gnan, N.; Micheli, L.; Ruzicka, B.; et al. Gellan Gum Microgels as Effective Agents for a Rapid Cleaning of Paper. ACS Appl. Polym. Mater. 2020, 2, 2791–2801. [Google Scholar] [CrossRef]
- Tavagnacco, L.; Zaccarelli, E.; Chiessi, E. On the molecular origin of the cooperative coil-to-globule transition of poly (N-isopropylacrylamide) in water. Phys. Chem. Chem. Phys. 2018, 20, 9997–10010. [Google Scholar] [CrossRef]
- Pelton, R.H.; Chibante, P. Preparation of aqueous lattices with N-isopropylacrylamide. Colloids Surf. 1986, 20, 247–256. [Google Scholar] [CrossRef]
- Nordstrom, K.N.; Verneuil, E.; Arratia, P.E.; Basu, A.; Zhang, Z.; Yodh, A.G.; Gollub, J.P.; Durian, D.J. Microfluidic Rheology of Soft Colloids above and below Jamming. Phys. Rev. Lett. 2010, 105, 1175701–1175704. [Google Scholar] [CrossRef] [PubMed]
- Paloli, D.; Mohanty, P.S.; Crassous, J.J.; Zaccarelli, E.; Schurtenberger, P. Fluid–solid transitions in soft-repulsive colloids. Soft Matter 2013, 9, 3000–3004. [Google Scholar] [CrossRef]
- Basu, A.; Xu, Y.; Still, T.; Arratia, P.; Zhang, Z.; Nordstrom, K.; Rieser, J.; Gollub, J.; Durian, D.; Yodh, A. Rheology of soft colloids across the onset of rigidity: Scaling behavior, thermal, and non-thermal responses. Soft Matter 2014, 10, 3027–3035. [Google Scholar] [CrossRef]
- Xia, X.; Hu, Z. Synthesis and Light Scattering Study of Microgels with Interpenetrating Polymer Networks. Langmuir 2004, 20, 2094–2098. [Google Scholar] [CrossRef]
- Hu, Z.; Xia, X. Hydrogel nanoparticle dispersions with inverse thermoreversible gelation. Adv. Mater. 2004, 16, 305–309. [Google Scholar] [CrossRef]
- Micali, N.; Bertoldo, M.; Buratti, E.; Nigro, V.; Angelini, R.; Villari, V. Interpenetrating Polymer Network Microgels in Water: Effect of Composition on the Structural Properties and Electrosteric Interactions. ChemPhysChem 2018, 19, 2894–2901. [Google Scholar] [CrossRef]
- Nigro, V.; Angelini, R.; Rosi, B.; Bertoldo, M.; E. Buratti, S.; Casciardi, S.S.; Ruzicka, B. Study of network composition in interpenetrating polymer networks of poly(N isopropylacrylamide) microgels: The role of poly(acrylic acid). J. Colloid Interface Sci. 2019, 545, 210–219. [Google Scholar] [CrossRef]
- Nigro, V.; Ripanti, F.; Angelini, R.; Sarra, A.; Bertoldo, M.; Buratti, E.; Postorino, P.; Ruzicka, B. Molecular mechanisms driving the microgels behaviour: A Raman spectroscopy and dynamic light scattering study. J. Mol. Liq. 2019, 284, 718–724. [Google Scholar] [CrossRef]
- Menut, P.; Seiffert, S.; Sprakel, J.; Weitz, D.A. Does size matter? Elasticity of compressed suspensions of colloidal- and granular-scale microgels. Soft Matter 2012, 8, 156–164. [Google Scholar] [CrossRef]
- Franco, S.; Buratti, E.; Ruzicka, B.; Nigro, V.; Zoratto, N.; Matricardi, P.; Zaccarelli, E.; Angelini, R. Volume fraction determination of microgel composed of interpenetrating polymer networks of PNIPAM and Polyacrylic acid. J. Phys. Condens. Matter 2021. [Google Scholar] [CrossRef] [PubMed]
- Makosko, C.W. Rheology: Principles, Measurements and Applications; Wiley-VCH Verlag GmbH: Weinheim, Germany, 1994. [Google Scholar]
- Senff, H.; Richtering, W. Influence of cross-link density on rheological properties of temperature-sensitive microgel suspensions. Colloid Polym. Sci. 2000, 278, 830–840. [Google Scholar] [CrossRef]
- Cross, M.M. Rheology of Non-Newtonian Fluids: A New Flow Equation for Pseudoplastic Systems. J. Colloid Sci. 2007, 20, 417–437. [Google Scholar] [CrossRef]
- Bingham, E.C. Fluidity and Plasticity; McGraw-Hill: New York, NY, USA, 1922. [Google Scholar]
- Barnes, H.; Hutton, J.; Walters, K. An Introduction to Rheology; Elsevier Science: Amsterdam, The Netherlands, 1989. [Google Scholar]
- Mueller, S.; Llewellin, E.W.; Mader, H.M. The rheology of suspensions of solid particles. R. Soc. 2010, 466, 1201–1228. [Google Scholar] [CrossRef]
- Nakaishi, A.; Minami, S.; Oura, S.; Watanabe, T.; Suzuki, D.; Urayama, K. Elastic and Flow Properties of Densely Packed Binary Microgel Mixtures with Size and Stiffness Disparities. Macromolecule 2018, 51, 9901–9914. [Google Scholar] [CrossRef]
- Fernandez-Nieves, A.; Puertas, A. Fluids, Colloids and Soft Materials: An Introduction to Soft Matter Physics; John Wiley and Sons: Hoboken, NJ, USA, 2016. [Google Scholar]
- Rubinstein, M.; Colby, R.H. Polymer Physics; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Carmona, J.A.; Ramìrez, P.; Calero, N.; Munoz, J. Large amplitude oscillatory shear of xanthan gum solutions. Effect of sodium chloride (NaCl) concentration. J. Food Eng. 2014, 126, 165–172. [Google Scholar] [CrossRef]
- Laidler, K.J. Chemical Kinetics; McGraw-Hil: New York, NY, USA, 1965. [Google Scholar]
- Angell, C.A.; Ngai, K.L.; McKenna, G.B.; McMillan, P.F.; Martin, S.W. Relaxation in glassforming liquids and amorphous solids. J. Appl. Phys. 2000, 88, 3113–3157. [Google Scholar] [CrossRef]
- Lu, H.; Huang, W.M. On the origin of the Vogel–Fulcher–Tammann law in the thermo-responsive shape memory effect of amorphous polymers. Smart Mater. Struct. 2013, 22, 105021. [Google Scholar] [CrossRef]
- Berthier, L. Theoretical perspective on the glass transition and amorphous materials. Rev. Mod. Phys. 2011, 83, 587. [Google Scholar] [CrossRef]
- Sengupta, S.; Vasconcelos, F.; Affouard, F.; Sastry, S. Dependence of the fragility of a glass former on the softness of interparticle interactions. J. Chem. Phys. 2011, 135, 94503–94509. [Google Scholar] [CrossRef] [PubMed]
- Hunter, G.L.; Weeks, E.R. The physics of the colloidal glass transition. Rev. Mod. Phys. 2012, 75, 066501. [Google Scholar] [CrossRef] [PubMed]
- van der Scheer, P.; van de Laar, T.; van der Gucht, J.; Vlassopoulos, D.; Sprakel, J. Fragility and Strength in Nanoparticle Glasses. ACS Nano 2017, 11, 6755–6763. [Google Scholar] [CrossRef]
- Angell, C.A. Formation of Glasses From Liquids and Biopolymers. Science 1995, 267, 1924–1935. [Google Scholar] [CrossRef] [PubMed]
- Ikeda, A.; Berthier, L.; Sollich, P. Unified study of glass and jamming rheology in soft particle systems. Phys. Rev. Lett. 2013, 109, 018301. [Google Scholar] [CrossRef] [PubMed]
- Kohlrausch, R. Thermoresponsive poly-(N-isopropylmethacrylamide) microgels: Tailoring particle size by interfacial tension control. Pogg. Ann. Phys. Chem. 1854, 91, 179–214. [Google Scholar] [CrossRef]
- Williams, G.; Watts, D.C. Non-symmetrical dielectric relaxation behavior arising from a simple empirical decay function. J. Chem. Soc. Faraday Trans. 1970, 66, 80–85. [Google Scholar] [CrossRef]
- Xia, X.; Hu, Z.; Marquez, M. Physically bonded nanoparticle networks: A novel drug delivery system. J. Control Release 2005, 103, 21–30. [Google Scholar] [CrossRef] [PubMed]
- Angelini, R.; Ruocco, G. Viscosity measurements in a solution undergoing inverse melting. Philos. Mag. 2007, 87, 553–558. [Google Scholar] [CrossRef][Green Version]
- Angelini, R.; Ruocco, G.; DePanfilis, S. Phase diagram of a solution undergoing inverse melting. Phys. Rev. E 2007, 78, 020502(R). [Google Scholar] [CrossRef] [PubMed]
- Howe, A.M.; Desrousseaux, S.; Lunel, L.S.; Tavacoli, J.; Yow, H.N.; Routh, A.F. Anomalous viscosity jump during the volume phase transition of poly(N-isopropylacrylamide) particles. Adv. Colloid Interface Sci. 2009, 148, 124–131. [Google Scholar] [CrossRef] [PubMed]
- Romeo, G.; Imperiali, L.; Kim, J.; Fernández-Nieves, A.; Weitz, D.A. Origin of de-swelling and dynamics of dense ionic microgel suspensions. J. Chem. Phys. 2012, 136, 124905. [Google Scholar] [CrossRef] [PubMed]
- Liu, X.; Guo, H.; Zha, L. Study of pH/temperature dual stimuli-responsive nanogels with interpenetrating polymer network structure. Polymers 2012, 61, 1144–1150. [Google Scholar] [CrossRef]
- Mason, T.G.; Weitz, D.A. Linear Viscoelasticity of Colloidal Hard Sphere Suspensions near the Glass Transition. Phys. Rev. Lett. 1995, 75, 2770–2773. [Google Scholar] [CrossRef]
- Koumakis, N.; Schofield, A.B.; Petekidis, G. Effects of shear induced crystallization on the rheology and ageing of hard sphere glasses. Soft Matter 2008, 10, 2008–2018. [Google Scholar] [CrossRef]
- Carrier, V.; Petekidis, G. Nonlinear rheology of colloidal glasses of soft thermosensitive microgel particles. J. Rheol. 2009, 53, 245–273. [Google Scholar] [CrossRef]
- Mason, T.G.; Weitz, D.A. Optical Measurements of Frequency-Dependent Linear Viscoelastic Moduli of Complex Fluids. Phys. Rev. Lett. 1995, 74, 1250–1253. [Google Scholar] [CrossRef] [PubMed]
- Derec, C.; Ducouret, G.; Ajdari, A.; Lequeux, F. Aging and nonlinear rheology in suspensions of polyethylene oxide-protected silica particles. Phys. Rev. E 2003, 67, 061403–061409. [Google Scholar] [CrossRef] [PubMed]
- Hyun, K.; Kim, S.H.; Ahn, K.H.; Lee, S.J. Large amplitude oscillatory shear as a way to classify the complex fluids. J. Non-Newton. Fluid Mech. 2002, 107, 51–65. [Google Scholar] [CrossRef]
- Kim, S.H.; Sim, H.G.; Ahn, K.H.; Lee, S.J. Large amplitude oscillatory shear behavior of the network model for associating polymeric systems. Rheol. J. 2002, 14, 49–55. [Google Scholar]
- Appel, J.; Folker, B.; Sprakel, J. Mechanics at the glass-to-gel transition of thermoresponsive microgel suspensions. Soft Matter 2016, 12, 2515. [Google Scholar] [CrossRef] [PubMed]
- Helgeson, M.E.; Wagner, N.J.; Vlassopoulos, D. Viscoelasticity and shear melting of colloidal star polymer glasses. J. Rheol. 2007, 51, 297–316. [Google Scholar] [CrossRef]
- Ueno, K.; Inaba, A.; Ueki, T.; Kondoh, M.; Watanabe, M. Thermosensitive, Soft Glassy and Structural Colored Colloidal Array in Ionic Liquid: Colloidal Glass to Gel Transition. Langmuir 2010, 26, 18031–18038. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Huang, G.; Hu, Z. Interparticle Potential and the Phase Behavior of Temperature-Sensitive Microgel Dispersions. Macromolecules 2003, 36, 440–448. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Franco, S.; Buratti, E.; Nigro, V.; Zaccarelli, E.; Ruzicka, B.; Angelini, R. Glass and Jamming Rheology in Soft Particles Made of PNIPAM and Polyacrylic Acid. Int. J. Mol. Sci. 2021, 22, 4032. https://doi.org/10.3390/ijms22084032
Franco S, Buratti E, Nigro V, Zaccarelli E, Ruzicka B, Angelini R. Glass and Jamming Rheology in Soft Particles Made of PNIPAM and Polyacrylic Acid. International Journal of Molecular Sciences. 2021; 22(8):4032. https://doi.org/10.3390/ijms22084032
Chicago/Turabian StyleFranco, Silvia, Elena Buratti, Valentina Nigro, Emanuela Zaccarelli, Barbara Ruzicka, and Roberta Angelini. 2021. "Glass and Jamming Rheology in Soft Particles Made of PNIPAM and Polyacrylic Acid" International Journal of Molecular Sciences 22, no. 8: 4032. https://doi.org/10.3390/ijms22084032
APA StyleFranco, S., Buratti, E., Nigro, V., Zaccarelli, E., Ruzicka, B., & Angelini, R. (2021). Glass and Jamming Rheology in Soft Particles Made of PNIPAM and Polyacrylic Acid. International Journal of Molecular Sciences, 22(8), 4032. https://doi.org/10.3390/ijms22084032








