Biomimetic Chromatographic Studies Combined with the Computational Approach to Investigate the Ability of Triterpenoid Saponins of Plant Origin to Cross the Blood–Brain Barrier
Abstract
1. Introduction
2. Results
2.1. Division of the Dataset for the QSAR Studies
2.2. BBB Descriptors Calculated In Silico
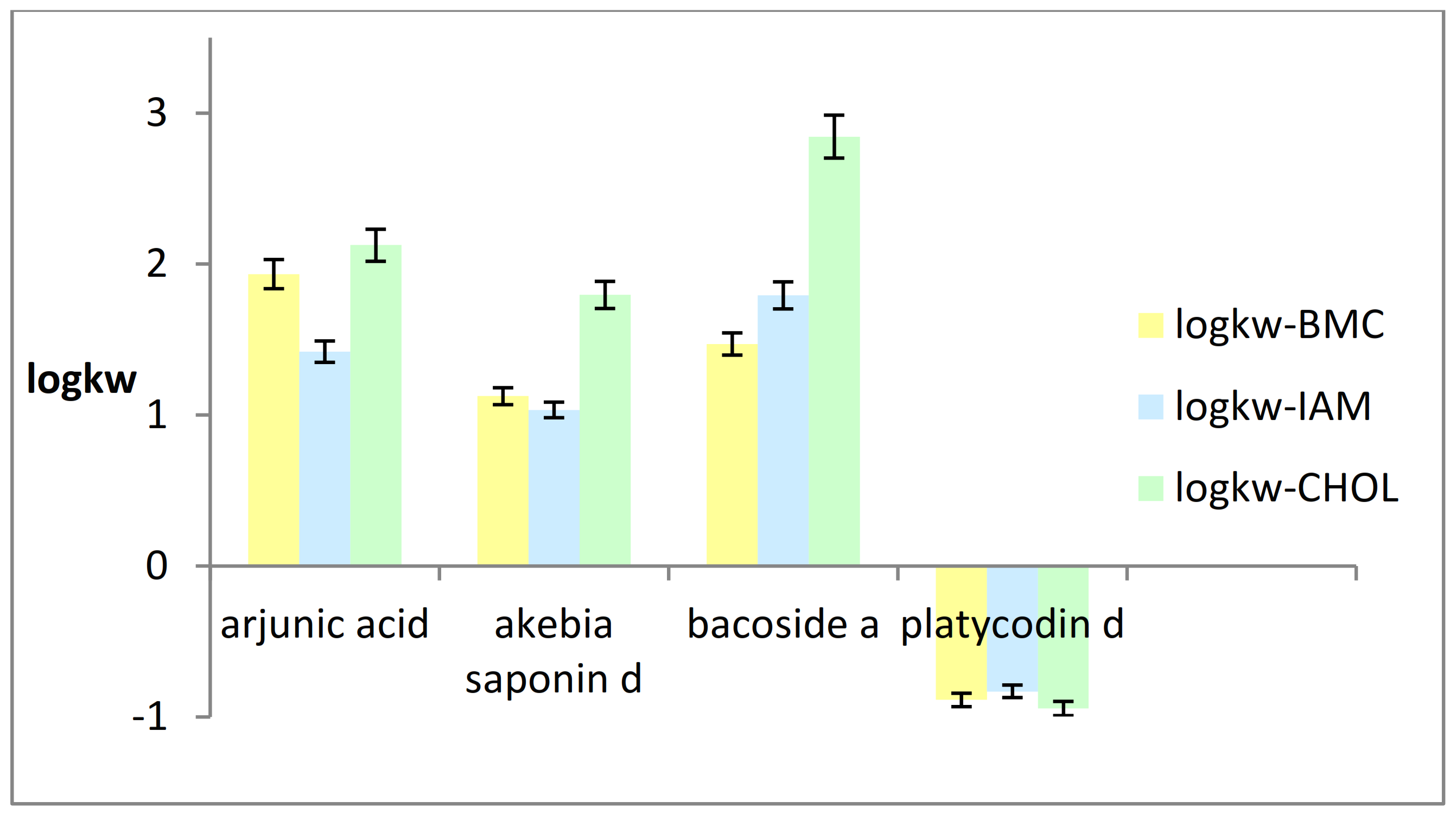
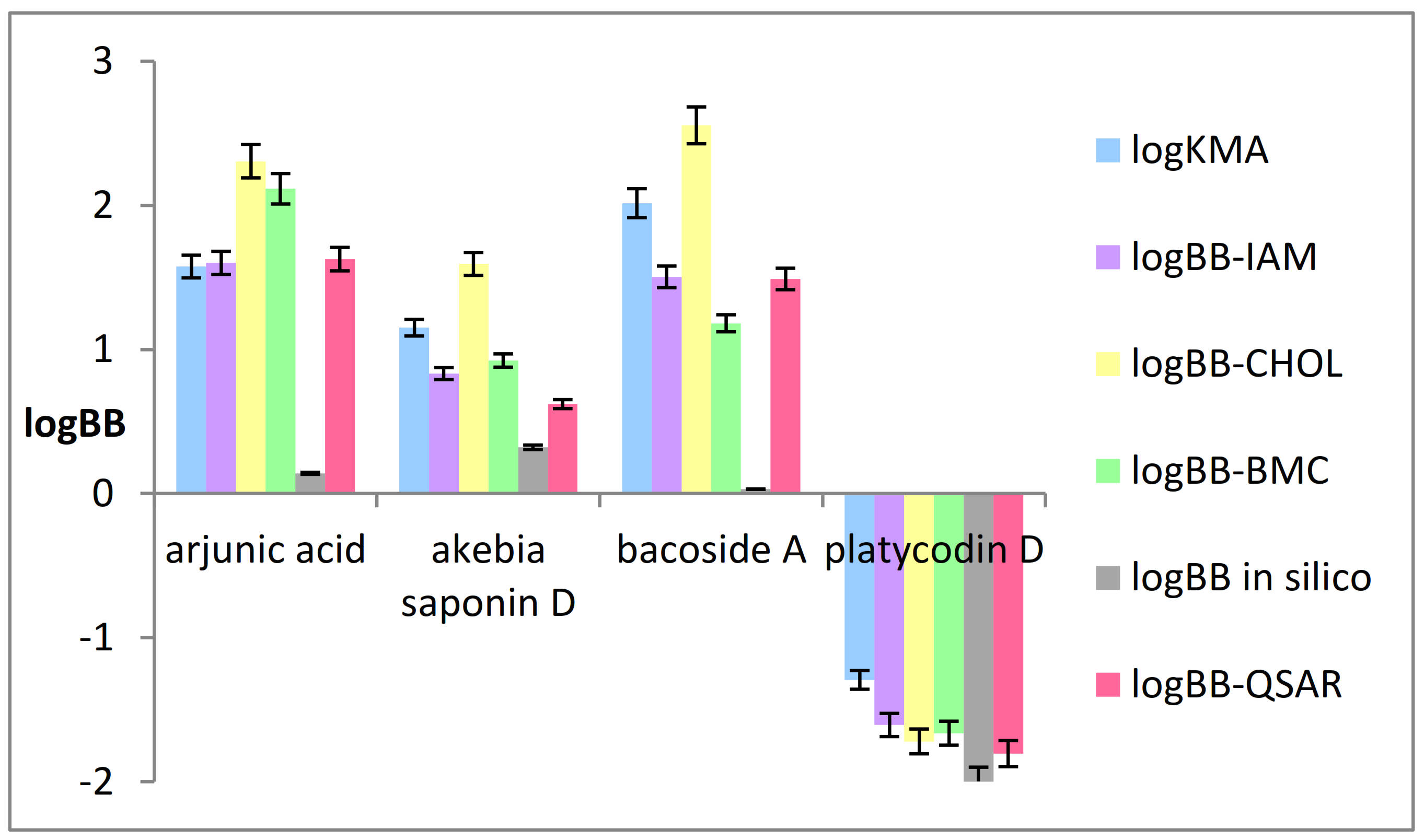
2.3. Chromatographic Biomimetic Studies
3. Discussion
4. Materials and Methods
4.1. Chemicals
4.2. Instrumentation
4.3. Chromatographic Conditions
4.4. Computer Programs
5. Conclusions
Supplementary Materials
Funding
Conflicts of Interest
References
- WHO. Dementia: A Public Health Priority; World Health Organization and Alzheimer’s Disease International Report; WHO: Geneva, Switzerland, 2012. [Google Scholar]
- Tao, L.; Zhang, P.; Qin, C.; Chen, S.Y.; Zhang, C.; Chen, Z.; Zhu, F.; Yang, S.Y.; We, Y.Q.; Chen, Y.Z. Recent progresses in the exploration of machine learning methods as in-silico ADME prediction tools. Adv. Drug Deliv. Rev. 2015, 86, 83–100. [Google Scholar] [CrossRef] [PubMed]
- Etkins, S.; Waller, C.L.; Swaan, P.W.; Cruciani, G.; Wrighton, S.A.; Wikel, J.H. Progress in predicting human ADME parameters in silico. J. Pharmacol. Toxicol. Methods 2000, 44, 251–272. [Google Scholar] [CrossRef]
- Etkins, S.; Ring, B.J.; Grace, J.; McRobie-Belle, D.J.; Wrighton, S.A. Present and future in vitro approaches for drug metabolism. J. Pharmacol. Toxicol. Methods 2000, 44, 313–324. [Google Scholar] [CrossRef]
- Drews, J. Drug discovery: A historical perspective. Science 2000, 287, 1960–1964. [Google Scholar] [CrossRef] [PubMed]
- Van de Waterbeemd, H.; Gifford, E. ADMET in silico modelling: Towards prediction paradise? Nat. Rev. Drug Discov. 2003, 2, 192–204. [Google Scholar] [CrossRef] [PubMed]
- Stepensky, D. Prediction of drug disposition on the basis of its chemical structure. Clin. Pharmacokinet. 2013, 52, 415–431. [Google Scholar] [CrossRef]
- Iyer, M.; Mishra, R.; Han, Y.; Hopfinger, A.J. Predicting Blood–Brain Barrier Partitioning of Organic Molecules Using Membrane-Interaction QSAR Analysis. Pharm. Res. 2002, 19, 1611–1621. [Google Scholar] [CrossRef]
- Hou, T.J.; Xu, X.J. ADME evaluation in drug discovery 1. Applications of genetic algorithms to the prediction of blood–brain partitioning of a large set of drugs. J. Mol. Model. 2002, 8, 337–349. [Google Scholar] [CrossRef]
- Hou, T.; Wang, J.; Zhang, W.; Wang, W.; Xu, X. Recent advances in computational prediction of drug absorption and permeability in drug discovery. Curr. Med. Chem. 2006, 13, 2653–2667. [Google Scholar] [CrossRef] [PubMed]
- Worth, A.P.; Bassan, A.; Gallegos, A.; Netzeva, T.I.; Patlewicz, G.; Pavan, M.; Tsakovska, I.; Vracko, M. The Characterisation of (Quantitative) Structure-Activity Relationships: Preliminary Guidance; JRC Report EUR 21866 EN; European Chemicals Bureau: Ispra, Italy, 2005. [Google Scholar]
- Zhou, Y.Q.; Yang, Z.L.; Xu, L.; Li, P.; Hu, Y.Z. Akebia saponin D, a saponin component from Dipsacus asper Wall, protects PC 12 cells against amyloid-beta induced cytotoxicity. Cell Biol. Int. 2009, 33, 1102–1110. [Google Scholar] [CrossRef]
- Ramasamy, S.; Chin, S.P.; Sukumaran, S.D.; Buckle, M.J.C.; Kiew, L.V.; Chung, L.Y. In Silico and In Vitro Analysis of Bacoside A Aglycones and Its Derivatives as the Constituents Responsible for the Cognitive Effects of Bacopa monnieri. PLoS ONE 2015, 10, e0126565. [Google Scholar] [CrossRef] [PubMed]
- Madhu, K.; Prakash, T.; Maya, S. Bacoside-A inhibits inflammatory cytokines and chemokine in experimental autoimmune encephalomyelitis. Biomed. Pharmacother. 2019, 109, 1339–1345. [Google Scholar] [CrossRef]
- Wang, G.; Guo, H.; Wang, X. Platycodin D protects cortical neurons against oxygen-glucose deprivation/reperfusion in neonatal hypoxic-ischemic encephalopathy. J. Cell. Biochem. 2019, 120, 14028–14034. [Google Scholar] [CrossRef]
- Thomas, R.B.; Joy, S.; Ajayan, M.S.; Paulose, C.S. Neuroprotective potential of Bacopa monnieri and Bacoside A against dopamine receptor dysfunction in the cerebral cortex of neonatal hypoglycaemic rats. Cell. Mol. Neurobiol. 2013, 33, 1065–1074. [Google Scholar] [CrossRef]
- Dubey, T.; Chinnathambi, S. Brahmi (Bacopa monnieri): An ayurvedic herb against the Alzheimer’s disease. Arch. Biochem. Biophys. 2019, 676, 108153. [Google Scholar] [CrossRef] [PubMed]
- Sekhar, V.C.; Viswanathan, G.; Baby, S. Insights Into the Molecular Aspects of Neuroprotective Bacoside A and Bacopaside I. Curr. Neuropharmacol. 2019, 17, 438–446. [Google Scholar] [CrossRef]
- Gupta, D.; Kumar, M. Evaluation of in vitro antimicrobial potential and GC-MS analysis of Camellia sinensis and Terminalia arjuna. Biotechnol. Rep. 2016, 13, 19–25. [Google Scholar] [CrossRef] [PubMed]
- Mandal, S.; Patra, A.; Samanta, A.; Roy, S.; Mandal, A.; Mahapatra, T.D.; Pradhan, S.; Das, K.; Nandi, D.K. Analysis of phytochemical profile of Terminalia arjuna bark extract with antioxidative and antimicrobial properties. Asian Pac. J. Trop. Biomed. 2013, 12, 960–966. [Google Scholar] [CrossRef]
- Dube, N.; Nimgulkar, C.; Bharatraj, D.K. Validation of therapeutic anti-inflammatory potential of Arjuna Ksheera Paka—A traditional Ayurvedic formulation of Terminalia arjuna. J. Tradit. Complementary Med. 2017, 7, 414–420. [Google Scholar] [CrossRef] [PubMed]
- Ahmad, M.S.; Ahmad, S.; Gautam, B.; Arshad, M.; Afzal, M. Terminalia arjuna, a herbal remedy against environmental carcinogenicity: An in vitro and in vivo study. Egypt J. Med. Hum. Genet. 2014, 15, 61–67. [Google Scholar] [CrossRef]
- Pugazhendhi, A.; Shafreen, R.B.; Devi, K.P.; Suganthy, N. Assessment of antioxidant, anticholinesterase and antiamyloidogenic effect of Terminalia chebula, Terminalia arjuna and its bioactive constituent 7-Methyl gallic acid—An in vitro and in silico studies. J. Mol. Liq. 2018, 257, 69–81. [Google Scholar] [CrossRef]
- Yaidikar, L.; Thakur, S. Arjunolic acid, a pentacyclic triterpenoidal saponin of Terminalia arjuna bark protects neurons from oxidative stress associated damage in focal cerebral ischemia and reperfusion. Pharmacol. Rep. 2015, 67, 890–895. [Google Scholar] [CrossRef]
- Mohanty, I.R.; Borde, M.; Kumar, C.S.; Maheshwari, U. Dipeptidyl peptidase IV Inhibitory activity of Terminalia arjuna attributes to its cardioprotective effects in experimental diabetes: In silico, in vitro and in vivo analyses. Phytomedicine 2019, 57, 158–165. [Google Scholar] [CrossRef]
- Kapoor, D.; Vijayvergiya, R.; Dhawan, V. Terminalia arjuna in coronary artery disease: Ethnopharmacology, pre-clinical, clinical & safety evaluation. J. Ethnopharmacol. 2014, 155, 1029–1045. [Google Scholar] [CrossRef]
- Bhattacharjee, B.; Pal, P.K.; Ghosh, A.K.; Mishra, S.; Chattopadhyay, A.; Bandyopadhyay, D. Aqueous bark extract of Terminalia arjuna protects against cadmium-induced hepatic and cardiac injuries in male Wistar rats through antioxidative mechanisms. Food Chem. Toxicol. 2019, 124, 249–264. [Google Scholar] [CrossRef]
- Daneman, R.; Prat, A. The blood–brain barrier. Cold Spring Harb. Perspect. Biol. 2015, 7, a020412. [Google Scholar] [CrossRef] [PubMed]
- Delsing, L.; Herland, A.; Falk, A.; Hicks, R.; Synnergren, J.; Zetterberg, H. Models of the blood-brain barrier using iPSC-derived cells. Mol. Cell. Neurosci. 2020, 107, 103533. [Google Scholar] [CrossRef]
- Ballabh, P.; Braun, A.; Nedergaard, M. The blood–brain barrier: An overview. Structure, regulation, and clinical implications. Neurobiol. Dis. 2004, 16, 1–13. [Google Scholar] [CrossRef] [PubMed]
- Van Leeuwen, E.; Hampton, M.B.; Smyth, L.C.D. Redox signalling and regulation of the blood-brain barrier. Int. J. Biochem. Cell Biol. 2020, 125, 105794. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Blood–brain barrier biology and methodology. J. Neurovirol. 1999, 5, 556–569. [Google Scholar] [CrossRef] [PubMed]
- Cereijido, M.; Ruiz, O.; González-Mariscal, L.; Contreras, R.; Susana Balda, M.; García-Villegas, M.R. The Paracellular Pathway. In Biological Barriers to Protein Delivery. Pharmaceutical Biotechnology; Audus, K.L., Raub, T.J., Eds.; Springer: Boston, MA, USA, 1993; Volume 4. [Google Scholar] [CrossRef]
- Laksitorini, M.; Prasasty, V.D.; Kiptoo, P.K.; Siahaan, T.J. Pathways and Progress in Improving Drug Delivery through the Intestinal Mucosa and Blood-Brain Barriers. Ther. Deliv. 2014, 10, 1143–1163. [Google Scholar] [CrossRef]
- Abbott, N.J.; Ronnback, L.; Hansson, E. Astrocyte-endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef] [PubMed]
- Cardoso, L.F.; Brites, D.; Brito, M.A. Looking at the blood–brain barrier: Molecular anatomy and possible investigation approaches. Brain Res. Rev. 2010, 64, 328–363. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Liu, L. Modern methods for delivery of drugs across the blood–brain barrier. Adv. Drug Deliv. Rev. 2012, 64, 640–665. [Google Scholar] [CrossRef] [PubMed]
- Zheng, W.; Aschner, M.; Ghersi-Egea, J.F. Brain barrier systems: A new frontier in metal neurotoxicological research. Toxicol. Appl. Pharmacol. 2003, 192, 1–11. [Google Scholar] [CrossRef]
- Norsted, E.; Gomuc, B.; Meister, B. Protein components of the blood–brain barrier (BBB) in the mediobasal hypothalamus. J. Chem. Neuroanat. 2008, 36, 107–121. [Google Scholar] [CrossRef]
- Sá-Pereira, I.; Brites, D.; Brito, M.A. Neurovascular unit: A focus on pericytes. Mol. Neurobiol. 2012, 45, 327–347. [Google Scholar] [CrossRef] [PubMed]
- Nikolakopoulou, A.M.; Montagne, A.; Kisler, K.; Dai, Z.; Wang, Y.; Huuskonen, M.T.; Sagare, A.P.; Lazic, D.; Sweeney, M.D.; Kong, P.; et al. Pericyte loss leads to circulatory failure and pleiotrophin depletion causing neuron loss. Nat. Neurosci. 2019, 22, 1089–1098. [Google Scholar] [CrossRef] [PubMed]
- Bell, R.D.; Winkler, E.A.; Sagare, A.P.; Singh, I.; LaRue, B.; Deane, R.; Zlokovic, B.V. Pericytes control key neurovascular functions and neuronal phenotype in the adult brain and during brain aging. Neuron 2010, 68, 409–427. [Google Scholar] [CrossRef]
- Platts, J.A.; Abraham, M.H.; Zhao, Y.H.; Hersey, A.; Ijaz, L.; Butina, D. Correlation and prediction of a large blood-brain distribution data set--an LFER study. Eur. J. Med. Chem. 2001, 36, 719–730. [Google Scholar] [CrossRef]
- Pangalos, M.N.; Schechter, L.E.; Hurko, O. Drug development for CNS disorders: Strategies for balancing risk and reducing attrition. Nat. Rev. Drug Discov. 2007, 6, 521–532. [Google Scholar] [CrossRef]
- Molero-Monfort, M.; Escuder-Gilabert, L.; Villanueva-Camanas, R.M.; Sagrado, S.; Medina-Hernández, M.J. Biopartitioning micellar chromatography: An in vitro technique for predicting human drug absorption. J. Chromatogr. B 2001, 753, 225–236. [Google Scholar] [CrossRef]
- Escuder-Gilabert, L.; Martinez-Pla, J.J.; Sagrado, S.; Villanueva-Camanas, R.M.; Medina-Hernández, M.J. Biopartitioning micellar separation methods: Modelling drug absorption, J. Chromatogr. B 2003, 797, 21–35. [Google Scholar] [CrossRef]
- Quiñones-Torrelo, C.; Martin-Biosca, Y.; Martínez-Pla, J.J.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. QRAR models for central nervous system drugs using biopartitioning micellar chromatography. Mini Rev. Med. Chem. 2002, 2, 145–161. [Google Scholar] [CrossRef]
- Escuder-Gilabert, L.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Quantitative Retention−Structure and Retention−Activity Relationship Studies of Local Anesthetics by Micellar Liquid Chromatography. Anal. Chem. 1998, 70, 28–34. [Google Scholar] [CrossRef]
- Escuder-Gilabert, L.; Sanchis-Mallols, J.M.; Sagrado, S.; Medina-Hernández, M.J.; Villanueva-Camañas, R.M. Chromatographic quantitation of the hydrophobicity of ionic compounds by the use of micellar mobile phases. J. Chromatogr. A 1998, 823, 549–559. [Google Scholar] [CrossRef]
- Quiñones-Torrelo, C.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Development of Predictive Retention−Activity Relationship Models of Tricyclic Antidepressants by Micellar Liquid Chromatography. J. Med. Chem. 1999, 42, 3154–3162. [Google Scholar] [CrossRef]
- Martin-Biosca, Y.; Molero-Monfort, M.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Development of predictive retention–activity relationship models of antipsychotic drugs by micellar liquid chromatography. Biomed. Chromatogr. 1999, 13, 478–492. [Google Scholar] [CrossRef]
- Escuder-Gilabert, L.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Development of predictive retention–activity relationship models of non-steroidal anti-inflammatory drugs by micellar liquid chromatography: Comparison with immobilized artificial membrane columns. J. Chromatogr. B 2000, 740, 59–70. [Google Scholar] [CrossRef]
- Martınez-Pla, J.J.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Retention–property relationships of anticonvulsant drugs by biopartitioning micellar chromatography. J. Chromatogr. B 2001, 757, 89–99. [Google Scholar] [CrossRef]
- Arunyanart, M.; Cline-Love, L. Model for micellar effects on liquid chromatography capacity factors and for determination of micelle-solute equilibrium constants. Anal. Chem. 1984, 56, 1557–1561. [Google Scholar] [CrossRef]
- López-Grío, S.; Baeza-Baeza, J.J.; García-Alvarez-Coque, M.C. Influence of the addition of modifiers on solute-micelle interaction in hybrid micellar liquid chromatography. Chromatographia 1998, 48, 655–663. [Google Scholar] [CrossRef]
- Medina-Hernández, M.J.; García-Álvarez-Coque, M.C. Solute-mobile phase and solute-stationary phase interactions in micellar liquid chromatography. A review. Analyst 1992, 117, 831–837. [Google Scholar] [CrossRef]
- Berthod, A.; Girard, I.; Gonnet, G. Additive effects on surfactant adsorption and ionic solute retention in micellar liquid chromatography. Anal. Chem. 1986, 58, 1362–1367. [Google Scholar] [CrossRef]
- Hansch, C. Quantitative approach to biochemical structure-activity relationships. Acc. Chem. Res. 1969, 2, 232–239. [Google Scholar] [CrossRef]
- Liu, R.; Li, X.; Lam, K.S. Combinatorial Chemistry in Drug Discovery. Curr. Opin. Chem. Biol. 2017, 38, 117–126. [Google Scholar] [CrossRef]
- Herrmann, A. Dynamic Combinatorial/Covalent Chemistry: A Tool to Read, Generate and Modulate the Bioactivity of Compounds and Compound Mixtures. Chem. Soc. Rev. 2014, 43, 1899–1933. [Google Scholar] [CrossRef] [PubMed]
- Russo, G.; Grumetto, L.; Szucs, R.; Barbato, F.; Lynen, F. Screening therapeutics according to their uptake across the blood-brain barrier: A high throughput method based on immobilized artificial membrane liquid chromatography-diode-array-detection coupled to electrospray-time-of-flight mass spectrometry. Eur. J. Pharm. Biopharm. 2018, 127, 72–84. [Google Scholar] [CrossRef]
- Pidgeon, C.; Venkataram, U.V. Immobilized artificial membrane chromatography: Supports composed of membrane lipids. Anal. Biochem. 1989, 176, 36–47. [Google Scholar] [CrossRef]
- Janicka, M.; Sztanke, M.; Sztanke, K. Predicting the Blood-Brain Barrier Permeability of New Drug-Like Compounds via HPLC with Various Stationary Phases. Molecules 2020, 25, 487. [Google Scholar] [CrossRef] [PubMed]
- Tsopelas, F.; Malaki, N.; Vallianatou, T.; Chrysanthakopoulos, M.; Vrakas, D.; Ochsenkühn-Petropoulou, M.; Tsantili-Kakoulidou, A. Insight into the retention mechanism on immobilized artificial membrane chromatography using two stationary phases. J. Chromatogr. A 2015, 1396, 25–33. [Google Scholar] [CrossRef]
- Stergiopoulos, C.; Makarouni, D.; Tsantili-Kakoulidou, A.; Ochsenkühn-Petropoulou, M.; Tsopelas, F. Immobilized artificial membrane chromatography as a tool for the prediction of ecotoxicity of pesticides. Chemosphere 2019, 224, 128–139. [Google Scholar] [CrossRef]
- Tsopelas, F.; Stergiopoulos, C.; Tsakanika, L.-A.; Ochsenkühn-Petropoulou, M.; Tsantili-Kakoulidou, A. The use of immobilized artificial membrane chromatography to predict bioconcentration of pharmaceutical compounds. Ecotoxicol. Environ. Saf. 2017, 139, 150–157. [Google Scholar] [CrossRef] [PubMed]
- Kotecha, J.; Shah, S.; Rathod, I.; Subbaiah, G. Relationship between immobilized artificial membrane chromatographic retention and human oral absorption of structurally diverse drugs. Int. J. Pharm. 2007, 333, 127–135. [Google Scholar] [CrossRef]
- Barbato, F.; Cappello, B.; Miro, A.; La Rotonda, M.I.; Quaglia, F. Chromatographic indexes on immobilized artificial membranes for the prediction of transdermal transport of drugs. Il Farmaco 1998, 53, 655–661. [Google Scholar] [CrossRef]
- Sui, X.; Sun, J.; Li, H.; Wang, Y.; Liu, J.; Liu, X.; Zhang, W.; Chen, L.; He, Z. Prediction of volume of distribution values in human using immobilized artificial membrane partitioning coefficients, the fraction of compound ionized and plasma protein binding data. Eur. J. Med. Chem. 2009, 44, 4455–4460. [Google Scholar] [CrossRef]
- Valko, K.; Nunhuck, S.; Bevan, C.; Abraham, M.H.; Reynolds, D.P. Fast Gradient HPLC Method to Determine Compounds Binding to Human Serum Albumin. Relationships with Octanol/Water and Immobilized Artificial Membrane Lipophilicity. J. Pharm. Sci. 2003, 92, 2236–2248. [Google Scholar] [CrossRef] [PubMed]
- Salminen, T.; Pulli, A.; Taskinen, J. Relationship between immobilised artificial membrane chromatographic retention and the brain penetration of structurally diverse drugs. J. Pharm. Biomed. Anal. 1997, 15, 469–477. [Google Scholar] [CrossRef]
- Cuenca-Benito, M.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Quantitative retention–structure and retention–activity relationships of barbiturates by micellar liquid chromatography. J. Chromatogr. A 1998, 814, 121–132. [Google Scholar] [CrossRef]
- Sanchis-Mallols, J.M.; Sagrado, S.; Villanueva-Camanas, R.M.; Medina-Hernandez, M.J. Quantitative retention—structure and retention—activity relationship studies of ionic and non-ionic catecholamines by micellar liquid chromatography. Chromatographia 1997, 46, 605–612. [Google Scholar] [CrossRef]
- Molero-Monfort, M.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Retention–activity relationship studies of benzodiazepines by micellar liquid chromatography. Biomed. Chromatogr. 1999, 13, 394–400. [Google Scholar] [CrossRef]
- Stępnik, K.; Malinowska, I.; Rój, E. In vitro and in silico determination of oral, jejunum and Caco-2 human absorption of fatty acids and polyphenols. Micellar liquid chromatography. Talanta 2014, 130, 265–273. [Google Scholar] [CrossRef] [PubMed]
- Escuder-Gilabert, L.; Molero-Monfort, M.; Villanueva-Camañas, R.M.; Sagrado, S.; Medina-Hernández, M.J. Potential of biopartitioning micellar chromatography as an in vitro technique for predicting drug penetration across the blood–brain barrier. J. Chromatogr. B 2004, 807, 193–201. [Google Scholar] [CrossRef]
- Stepnik, K.; Malinowska, I. The use of biopartitioning micellar chromatography and immobilized artificial membrane column for in silico and in vitro determination of blood–brain barrier penetration of phenols. J. Chromatogr. A 2013, 1286, 127–136. [Google Scholar] [CrossRef]
- Martínez-Pla, J.J.; Martín-Biosca, Y.; Sagrado, S.; Villanueva-Camanas, R.M.; Medina-Hernandez, M.J. Evaluation of the pH effect of formulations on the skin permeability of drugs by biopartitioning micellar chromatography. J. Chromatogr. A 2004, 1047, 255–262. [Google Scholar] [CrossRef] [PubMed]
- Stępnik, K.; Malinowska, I. Skin-mimetic chromatography for prediction of human percutaneous absorption of biologically active compounds occurring in medicinal plant extracts. Biomed. Chromat. 2017, 31, e3922. [Google Scholar] [CrossRef] [PubMed]
- Hadjmohammadi, M.; Salary, M. Biopartitioning micellar chromatography with sodium dodecyl sulfate as a pseudo α1-acid glycoprotein to the prediction of protein–drug binding. J. Chromatogr. B 2013, 912, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Tsopelas, F.; Danias, P.; Pappa, A.; Tsantili-Kakoulidou, A. Biopartitioning micellar chromatography under different conditions: Insight into the retention mechanism and the potential to model biological processes. J. Chromatogr. A 2020, 1621, 461027. [Google Scholar] [CrossRef]
- Dobričić, V.; Savić, J.; Nikolic, K.; Vladimirov, S.; Vujić, Z.; Brborić, J. Application of biopartitioning micellar chromatography and QSRR modeling for prediction of gastrointestinal absorption and design of novel β-hydroxy-β-arylalkanoic acids. Eur. J. Pharm. Sci. 2017, 100, 280–284. [Google Scholar] [CrossRef] [PubMed]
- Martín-Biosca, Y.; Torres-Cartas, S.; Villanueva-Camanas, R.M.; Sagrado, S.; Medina-Hernández, M.J. Biopartitioning micellar chromatography to predict blood to lung, blood to liver, blood to fat and blood to skin partition coefficients of drugs. Anal. Chim. Acta 2009, 632, 296–303. [Google Scholar] [CrossRef]
- Dobričić, V.; Nikolic, K.; Vladimirov, S.; Čudina, O. Biopartitioningmicellar chromatographyas a predictive tool for skin and corneal permeability of newly synthesized 17β-carboxamide steroids. Eur.J. Pharm. Sci. 2014, 56, 105–112. [Google Scholar] [CrossRef]
- Martín-Biosca, Y.; Molero-Monfort, M.; Sagrado, S.; Villanueva-Camañas, R.M.; Medina-Hernández, M.J. Rapid in vitro test to predict ocular tissue permeability based on biopartitioning micellar chromatography. Eur. J. Pharm. Sci. 2003, 20, 209–216. [Google Scholar] [CrossRef]
- Lu, R.; Sun, J.; Wang, Y.; Li, H.; Liu, J.; Fang, L.; He, Z. Characterization of biopartitioning micellar chromatography system using monolithic column by linear solvation energy relationship and application to predict blood–brain barrier penetration. J. Chromatogr. A 2009, 1216, 5190–5198. [Google Scholar] [CrossRef]
- Yin, C.; Ma, L.; Huang, J.; Xu, L.; Shi, Z. Fast profiling ecotoxicity and skin permeability of benzophenone ultraviolet filters using biopartitioning micellar chromatography based on penetrable silica spheres. Anal. Chim. Acta 2013, 804, 321–327. [Google Scholar] [CrossRef]
- Li, J.; Sun, J.; He, Z. Quantitative structure–retention relationship studies with immobilized artificial membrane chromatography II: Partial least squares regression. J. Chromatogr. A 2007, 1140, 174–179. [Google Scholar] [CrossRef]
- Janicka, M.; Sztanke, M.; Sztanke, K. Reversed-phase liquid chromatography with octadecylsilyl, immobilized artificial membrane and cholesterol columns in correlation studies with in silico biological descriptors of newly synthesized antiproliferative and analgesic active compounds. J. Chromatogr. A 2013, 1318, 92–101. [Google Scholar] [CrossRef] [PubMed]
- 90. Stępnik, K.; Kukula-Koch, W. In Silico Studies on Triterpenoid Saponins Permeation through the Blood–Brain Barrier Combined with Postmortem Research on the Brain Tissues of Mice Affected by Astragaloside IV Administration. Int. J. Mol. Sci. 2020, 21, 2534. [Google Scholar] [CrossRef] [PubMed]
- Mente, S.R.; Lombardo, F. A recursive—Partitioning model for blood–brain barrier permeation. J. Comput. Aided Mol. Des. 2005, 19, 465–481. [Google Scholar] [CrossRef]
- Hansch, C.; Fujita, T. p-σ-π analysis. A Method for the correlation of biological activity and chemical structure. J. Am. Chem. Soc. 1964, 86, 1616–1626. [Google Scholar] [CrossRef]
- Abraham, M.H.; Takács-Novák, K.; Mitchell, R.C. On the partition of ampholytes: Application to blood–braindistribution. J. Pharm. Sci. 1997, 86, 310–315. [Google Scholar] [CrossRef]
- Abraham, M.H. The factors that influence permeation across the blood–brain barrier. Eur. J. Med. Chem. 2004, 39, 235–240. [Google Scholar] [CrossRef]
- Lipinski, C.A.; Lombardo, F.; Dominy, B.W.; Feeney, P.J. Experimental and computational approaches to estimate solubility and permeability in drug discovery and development settings. Adv. Drug Deliv. Rev. 1997, 23, 3–25. [Google Scholar] [CrossRef]
- Jandera, P.; Fischer, J.; Effenberger, H. Characterisation of retention in micellar high-performance liquid chromatography and in micellar electrokinetic chromatography using lipophilicity and polarity indices. J. Chromatogr. A 1998, 807, 57–70. [Google Scholar] [CrossRef]
- Foley, J.P. Critical compilation of solute-micelle binding constants and related parameters from micellar liquid chromatographic measurements. Anal. Chim. Acta 1990, 23, 231–237. [Google Scholar] [CrossRef]
- Li, C.; Yang, J.; Yu, S.; Chen, N.; Xue, W.; Hu, J.; Zhang, D. Triterpenoid Saponins with Neuroprotective Effects from the Roots of Polygala tenuifolia. Planta Med. 2008, 74, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Son, I.H.; Park, Y.H.; Lee, S.I.; Yang, H.D.; Moon, H.-I. Neuroprotective Activity of Triterpenoid Saponins from Platycodi radix against Glutamate-induced Toxicity in Primary Cultured Rat Cortical Cells. Molecules 2007, 12, 1147–1152. [Google Scholar] [CrossRef]
- Liu, X.-G.; Sun, Y.-Q.; Bian, J.; Han, T.; Yue, D.-D.; Li, D.-Q.; Gao, P.-Y. Neuroprotective effects of triterpenoid saponins from Medicago sativa L. against H2O2-induced oxidative stress in SH-SY5Y cells. Bioorg. Chem. 2019, 83, 468–476. [Google Scholar] [CrossRef]
- Liu, X.; Smith, B.J.; Chen, C.; Callegari, E.; Becker, S.L.; Chen, X.; Cianfrogna, J.; Doran, A.C.; Doran, S.D.; Gibbs, J.P.; et al. Use of a physiologically based pharmacokinetic model to study the time to reach brain equilibrium: An experimental analysis of the role of blood–brain barrier permeability, plasma protein binding, and brain tissue binding. Pharmacol. Exp. Ther. 2005, 313, 1254–1262. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Xin, Z.; Liu, B.; Wang, J.; Wang, J.; Zhang, X.; Wang, Y.; Li, F. Platycodin D Inhibits Inflammatory Response in LPS-Stimulated Primary Rat Microglia Cells through Activating LXRα–ABCA1 Signaling Pathway. Front. Immunol. 2018, 8, 1929. [Google Scholar] [CrossRef]
- ElAli, A.; Hermann, D.M. Liver X receptor activation enhances blood-brain barrier integrity in the ischemic brain and increases the abundance of ATP-binding cassette transporters ABCB1 and ABCC1 on brain capillary cells. Brain Pathol. 2012, 22, 175–187. [Google Scholar] [CrossRef]
- Zuo, A.H.; Cheng, M.C.; Wang, L.; Xiao, H.B. Analysis of chemical constituents of Chuanxiong Rhizoma absorbed into rat brain tissues by UPLC-Q-TOF-MS. China J. Chin. Mater. Med. 2012, 37, 3647–3650. [Google Scholar]
- Choi, J.H.; Yoo, K.-Y.; Park, O.K.; Lee, C.H.; Won, M.-H.; Hwang, I.K.; Ryu, S.Y.; Kim, Y.S.; Yi, J.-S.; Bae, Y.-S.; et al. Platycodin D and 2″-o-acetyl-polygalacin D2 isolated from Platycodon grandiflorum protect ischemia/reperfusion injury in the gerbil hippocampus. Brain Res. 2009, 1279, 197–208. [Google Scholar] [CrossRef] [PubMed]
- Choi, Y.H.; Kim, Y.S.; Yeo, S.J.; Roh, S.H.; Jeong, Y.C.; Kang, J.S.; Ryu, S.Y. Ameliorating effect of balloon flower saponin on the ethanol-induced memory impairment in mice. Phytother. Res. 2008, 22, 973–976. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, X.; Gao, S.; Qian, K.; Liu, Q.; Yin, X. Research on the neuro-protective compounds in Terminalia chebula Retz extracts in-vivo by UPLC–QTOF-MS. Acta Chromatogr. 2018, 30, 169–174. [Google Scholar] [CrossRef]
- Wang, Y.; Shen, J.; Yang, X.; Jin, Y.; Yang, Z.; Wang, R.; Zhang, F.; Linhardt, R.J. Akebia saponin D reverses corticosterone hypersecretion in an Alzheimer’s disease rat model. Biomed. Pharmacother. 2018, 107, 219–225. [Google Scholar] [CrossRef]
- Yu, X.; Wang, L.; Du, Q.; Ma, L.; Chen, L.; You, R.; Liu, L.; Ling, J.; Yang, Z.; Ji, H. Akebia Saponin D attenuates amyloid β-induced cognitive deficits and inflammatory response in rats: Involvement of Akt/NF-κB pathway. Behav. Brain Res. 2012, 235, 200–209. [Google Scholar] [CrossRef]
- Nandy, S.; Dey, A.; Mukherjeeb, A. Chapter 15—Advances in dammarane-type triterpenoid saponins from Bacopa monnieri: Structure, bioactivity, biotechnology and neuroprotection. In Studies in Natural Products Chemistry; Atta-ur-Rahman, Ed.; Elsevier: Amsterdam, The Netherlands, 2019; Volume 63, pp. 489–533. [Google Scholar]
- Uabundit, N.; Wattanathorn, J.; Mucimapura, S.; Ingkaninan, K. Cognitive enhancement and neuroprotective effects of Bacopamonnieri in Alzheimer’s disease model. J. Ethnopharmacol. 2010, 127, 26–31. [Google Scholar] [CrossRef] [PubMed]
- Sukumaran, N.P.; Amalraj, A.; Gopi, S. Neuropharmacological and cognitive effects of Bacopa monnieri (L.) Wettst—A review on its mechanistic aspects. Complementary Ther. Med. 2019, 44, 68–82. [Google Scholar] [CrossRef]
- Gozalbes, R.; Pineda-Lucena, A. QSAR-based solubility model for drug-like compounds. Bioorg. Med. Chem. 2010, 18, 7078–7084. [Google Scholar] [CrossRef]
- Young, R.C.; Mitchell, R.C.; Brown, T.H.; Ganellin, C.R.; Griffiths, R.; Jones, M.; Rana, K.K.; Saunders, D.; Smith, I.R.; Sore, N.E.; et al. Development of a new physicochemical model for brain penetration and its application to the design of centrally acting H2receptor histamine antagonists. J. Med. Chem. 1988, 31, 656–671. [Google Scholar] [CrossRef]
- Rose, K.; Hall, L.H.; Kier, L.B. Modeling Blood–Brain Barrier Partitioning Using the Electrotopological State. J. Chem. Inf. Comput. Sci. 2002, 42, 651–666. [Google Scholar] [CrossRef] [PubMed]
- Lombardo, F.; Blake, J.F.; Curatolo, W.J. Computation of brain-blood partitioning of organic solutes via free energy calculations. J. Med. Chem. 1996, 39, 4750–4755. [Google Scholar] [CrossRef]
- Kaliszan, R.; Markuszewski, M. Brain/blood distribution described by a combination of partition coefficient and molecular mass. Int. J. Pharm. 1996, 145, 9–16. [Google Scholar] [CrossRef]
- Clark, D.E. Rapid calculation of polar molecular surface area and its application to the prediction of transport phenomena. 2. Prediction of blood–brain barrier penetration. J. Pharm. Sci. 1999, 88, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Feher, M.; Sourial, E.; Schmidt, J.M. A simple model for the prediction of blood brain partitioning. Int. J. Pharm. 2000, 201, 239–247. [Google Scholar] [CrossRef]
- Keseru, G.M.; Molnar, L. High-throughput prediction of blood–brain partitioning: A thermodynamic approach. J. Chem. Inf. Comput. Sci. 2001, 41, 120–128. [Google Scholar] [CrossRef] [PubMed]
- Kaznessis, Y.N.; Snow, M.E.; Blankley, C.J. Prediction of blood–brain partitioning using Monte Carlo simulations of molecules in water. J. Comput. Aided Mol. Des. 2001, 15, 697–708. [Google Scholar] [CrossRef]
- Narayanan, R.; Gunturi, S.B. In silico ADME modeling: Prediction models for blood–brain barrier permeation using a systematic variable selection method. Bioorg. Med. Chem. 2005, 13, 3017–3028. [Google Scholar] [CrossRef] [PubMed]
- Hitchcock, S.A.; Pennington, L.D. Structure-brain exposure relationships. J. Med. Chem. 2006, 49, 7559–7583. [Google Scholar] [CrossRef]
- Valko, K. Lipophilicity and biomimetic properties measured by HPLC to support drug Discovery. J. Pharm. Biomed. Anal. 2016, 130, 35–54. [Google Scholar] [CrossRef]
- Ermondi, G.; Vallaro, M.; Caron, G. Learning how to use IAM chromatography for predicting permeability. Eur. J. Pharm. Sci. 2018, 114, 385–390. [Google Scholar] [CrossRef] [PubMed]
- Ong, S.; Liu, H.; Qiu, X.; Bhat, G.; Pidgeon, C. Membrane partition coefficients chromatographically measured using immobilized artificial membrane surfaces. Anal. Chem. 1995, 67, 755–762. [Google Scholar] [CrossRef] [PubMed]
- Darrouzain, F.; Dallet, P.; Dubost, J.-P.; Ismaili, L.; Pehourcq, F.; Bannwarth, B.; Matoga, M.; Guillaume, Y.C. Molecular lipophilicity determination of a huperzine series by HPLC: Comparison of C18 and IAM stationary phases. J. Pharm. Biomed. Anal. 2006, 41, 228–232. [Google Scholar] [CrossRef] [PubMed]
| References | Drugs | BMC/IAM System | Biological Activity |
|---|---|---|---|
| [53] | Anticonvulsant drugs | Brij 35: 0.02 M; 0.04 M; 0.06 M; pH: 7.4 | Anticonvulsant properties |
| [52] | Non-steroidal anti-inflammatory drugs | Brij 35: 0.02 M; 0.04 M; 0.06 M; pH: 7.4 | Anesthetic potency |
| [48] | Local anesthetics | Brij 35: 0.02 M; 0.04 M; 0.06 M; pH: 7.4 | Anesthetic potency |
| [72] | Barbiturates | Brij35: 0.02 M; 0.04 M; 0.06 M SDS: 0.05 M; 0.1 M; 0.15 M CTAB: 0.01 M; 0.02 M; 0.05 M pH: 3.5 and 7.4 | Hypnotic activity |
| [73] | Catecholamines | SDS: 0.05 M; 0.1 M + MeOH, EtOH, 1-propanol, pentanol pH: 2–7 | β- andrenergic activity |
| [74] | Benzodiazepines | Brij35: 0.02 M; 0.04 M; 0.06 M pH: 7.4 | Toxicity and anxiolytic activity |
| [51] | Phenothiazines | Brij35: 0.02 M; 0.04 M; 0.06 M pH: 7.4 | Pharmacokinetics, preclinical pharmacology, and therapeutic efficacy parameters; antipsychotic potential |
| [45] | Structurally diverse drugs | Brij35: 0.04 M pH: 7.4 and 6.5 | Oral absorption |
| [75] | Fatty acids and polyphenols | Brij35: 0.04 M; 0.06 M; 0.08 M; 0.1 M; 0.12 M + acetonitrile CTAB: 0.04 M; 0.06 M; 0.08 M; 0.1 M; 0.12 M + acetonitrile SDS: 0.04 M; 0.06 M; 0.08 M; 0.1 M; 0.12 M + acetonitrile, dioxane, tetrahydrofuran, acetone pH: 7.4 | Oral, jejunum and Caco-2 absorption |
| [76] | Structurally diverse drugs | Brij35: 0.04 M pH: 7.4 | BBB permeability |
| [77] | Phenols | Brij35: 0.06 M; 0.08 M; 0.1 M; 0.12 M + isobutanol (5% v/v) pH: 7.4 | BBB permeability |
| [78] | Non-steroidal anti-inflammatory drugs | Brij35: 0.04 M pH: 3.5–8 | Skin permeability |
| [79] | Fatty acids and polyphenols | Brij35: 0.04 M; 0.06 M; 0.08 M; 0.1 M; 0.12 M + acetonitrile CTAB: 0.04 M; 0.06 M; 0.08 M; 0.1 M; 0.12 M + acetonitrile SDS: 0.04 M; 0.06 M; 0.08 M; 0.1 M; 0.12 M + acetonitrile, dioxane, tetrahydrofuran, acetone pH: 7.4 | Percutaneous absorption |
| [80] | Anxiolytics, antihistamines, β-blockers, antiepileptics, antipsychotics | SDS: 0.07 M; 0.09 M pH: 7.4 | Protein drug binding properties |
| [81] | Structurally diverse drugs | PBS or PBS-acetonitrile: 5–25% v/v pH: 7.4 | Cell permeability, human oral absorption, % plasma protein binding |
| [82] | Novel β-hydroxy-β-aryl-alkanoic acids | Brij35: 0.04 M pH: 7.4 | Gastrointestinal absorption |
| [83] | Structurally diverse drugs | Brij35: 0.04 M pH 7.4 | Blood to lung; blood to liver; blood to fat; blood to skin partition coefficients |
| [84] | Newly-synthesized 17-β-carboxamide steroids | Brij35: 0.04 M pH: 5.5 and 7.5 | Skin and corneal permeability |
| [85] | Structurally diverse drugs | Brij35: 0.04 M pH: 7.4–7.7 | Ocular tissue permeability |
| [86] | Structurally diverse drugs | Brij35: 0.04 M pH: 7.4 | BBB permeability |
| [87] | Benzophenone ultraviolet filters | Brij35: 0.01 M; 0.02 M; 0.03 M pH: 7.4 and 6.5 | Ecotoxicity and skin permeability |
| [65] | Structurally diverse pesticides | Phosphate-buffered saline (PBS) or PBS-acetonitrile: 5–25% v/v pH: 7.4 | Ecotoxicity |
| [61] | Structurally diverse compounds | Buffer- MeOH: 70:30 v/v pH: 7.4 | BBB permeability |
| [66] | Structurally diverse drugs | PBS or PBS-acetonitrile: 5–25% v/v pH: 7.4 | Bioconcentration factor |
| [88] | Structurally diverse drugs | Acetonitrile-buffer pH: 7.4 | Interactions between the solutes and the immobilized phospholipid membranes |
| [67] | Structurally diverse drugs | Acetonitrile-buffer: 0–30% v/v pH: 7.4 | Human oral absorption |
| [63] | Newly-synthesized drug-like compounds | Acetonitrile-buffer pH 7.4 | Blood–brain barrier permeation |
| [89] | Newly synthesized antiproliferative and analgesic active compounds | Acetonitrile-buffer pH 7.4 | Lipophilicity |
| Name | Structure |
|---|---|
| Arjunic acid | 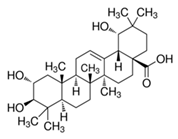 |
| Akebia saponin D | 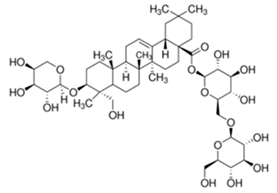 |
| Bacoside A | 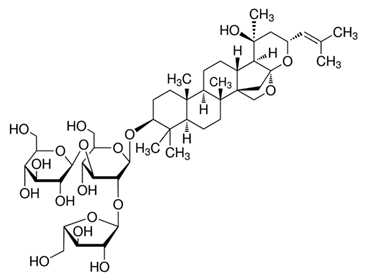 |
| Platycodin D | 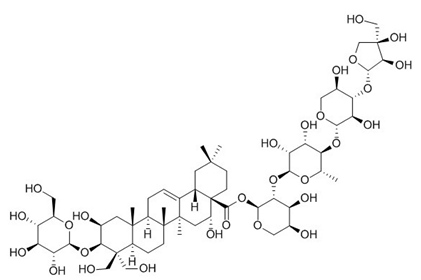 |
| Name | logBB | logPS | logPSFubrain | Fu | Fb |
|---|---|---|---|---|---|
| Arjunic acid | 0.14 | −3.2 | −4.9 | 0.012 | 0.02 |
| Akebia saponin D | 0.32 | −4.4 | −5.7 | 0.12 | 0.06 |
| Bacoside A | 0.03 | −3.6 | −4.5 | 0.14 | 0.13 |
| Platycodin D | <−2 | −3.6 | −9.0 | 0.52 | 0.98 |
| Name | LogPow (Octanol/Water) | logPhw (Heptane/Hater) | logPcw (Cyclohexane /Water) | Molecular Weight (MW) (g/mol) | Topological Polar Surface Area (TPSA) (Å2) | Polarizability |
|---|---|---|---|---|---|---|
| Arjunic acid | 5.2 | 4.179 | 4.029 | 488.70 | 97.99 | 54.15 |
| Akebia saponin D | 0.8 | −9.919 | −9.932 | 929.10 | 294.98 | 91.23 |
| Bacoside A | 2.8 | −7.553 | −7.110 | 768.97 | 215.83 | 78.66 |
| Platycodin D | −3.7 | −24.512 | −24.704 | 1225.32 | 453.28 | 114.32 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Stępnik, K. Biomimetic Chromatographic Studies Combined with the Computational Approach to Investigate the Ability of Triterpenoid Saponins of Plant Origin to Cross the Blood–Brain Barrier. Int. J. Mol. Sci. 2021, 22, 3573. https://doi.org/10.3390/ijms22073573
Stępnik K. Biomimetic Chromatographic Studies Combined with the Computational Approach to Investigate the Ability of Triterpenoid Saponins of Plant Origin to Cross the Blood–Brain Barrier. International Journal of Molecular Sciences. 2021; 22(7):3573. https://doi.org/10.3390/ijms22073573
Chicago/Turabian StyleStępnik, Katarzyna. 2021. "Biomimetic Chromatographic Studies Combined with the Computational Approach to Investigate the Ability of Triterpenoid Saponins of Plant Origin to Cross the Blood–Brain Barrier" International Journal of Molecular Sciences 22, no. 7: 3573. https://doi.org/10.3390/ijms22073573
APA StyleStępnik, K. (2021). Biomimetic Chromatographic Studies Combined with the Computational Approach to Investigate the Ability of Triterpenoid Saponins of Plant Origin to Cross the Blood–Brain Barrier. International Journal of Molecular Sciences, 22(7), 3573. https://doi.org/10.3390/ijms22073573






