Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies
Abstract
1. Introduction: Neural and Mesenchymal Stem Cells, and Neuroregenerative Cell Therapies
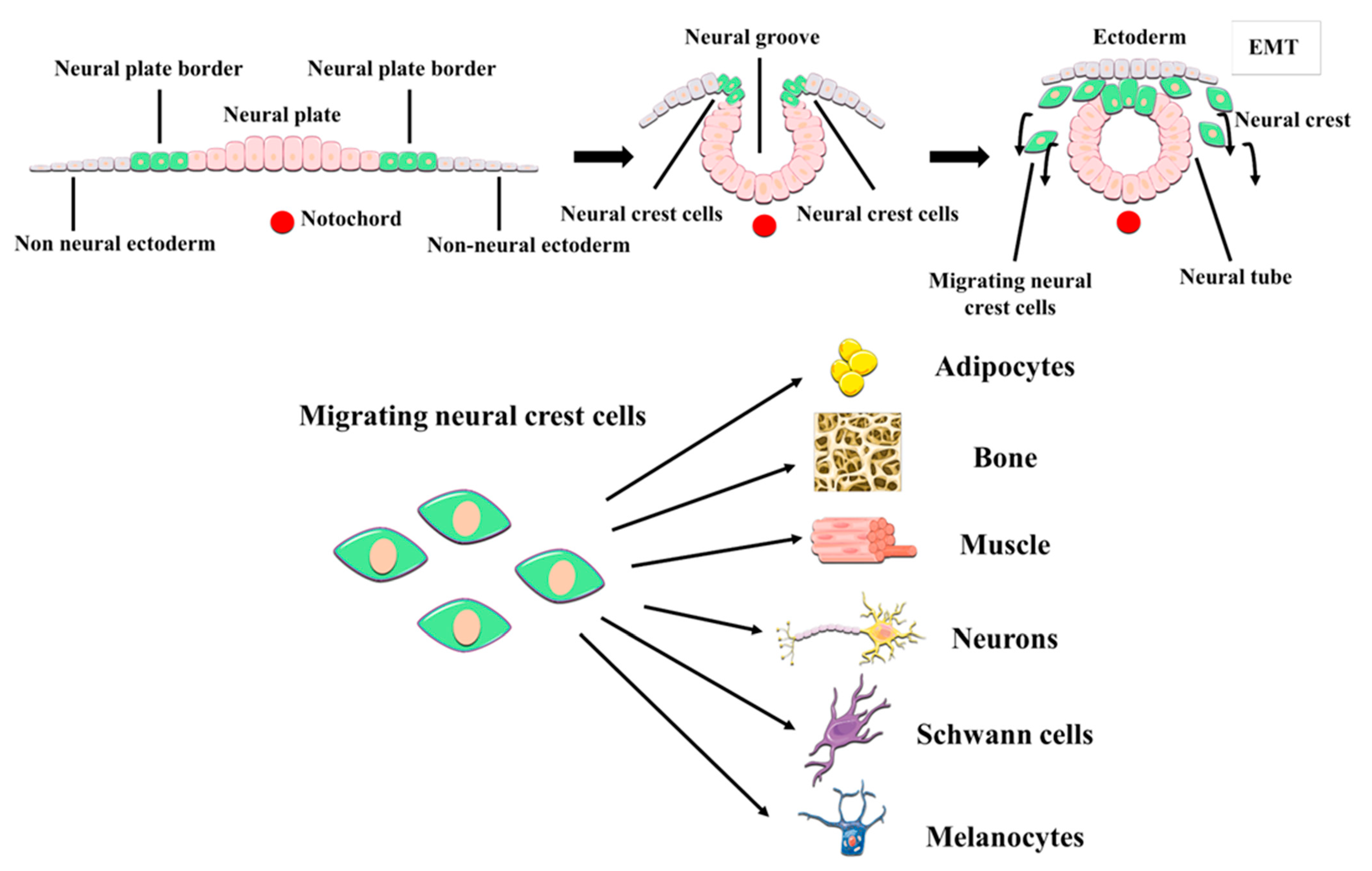
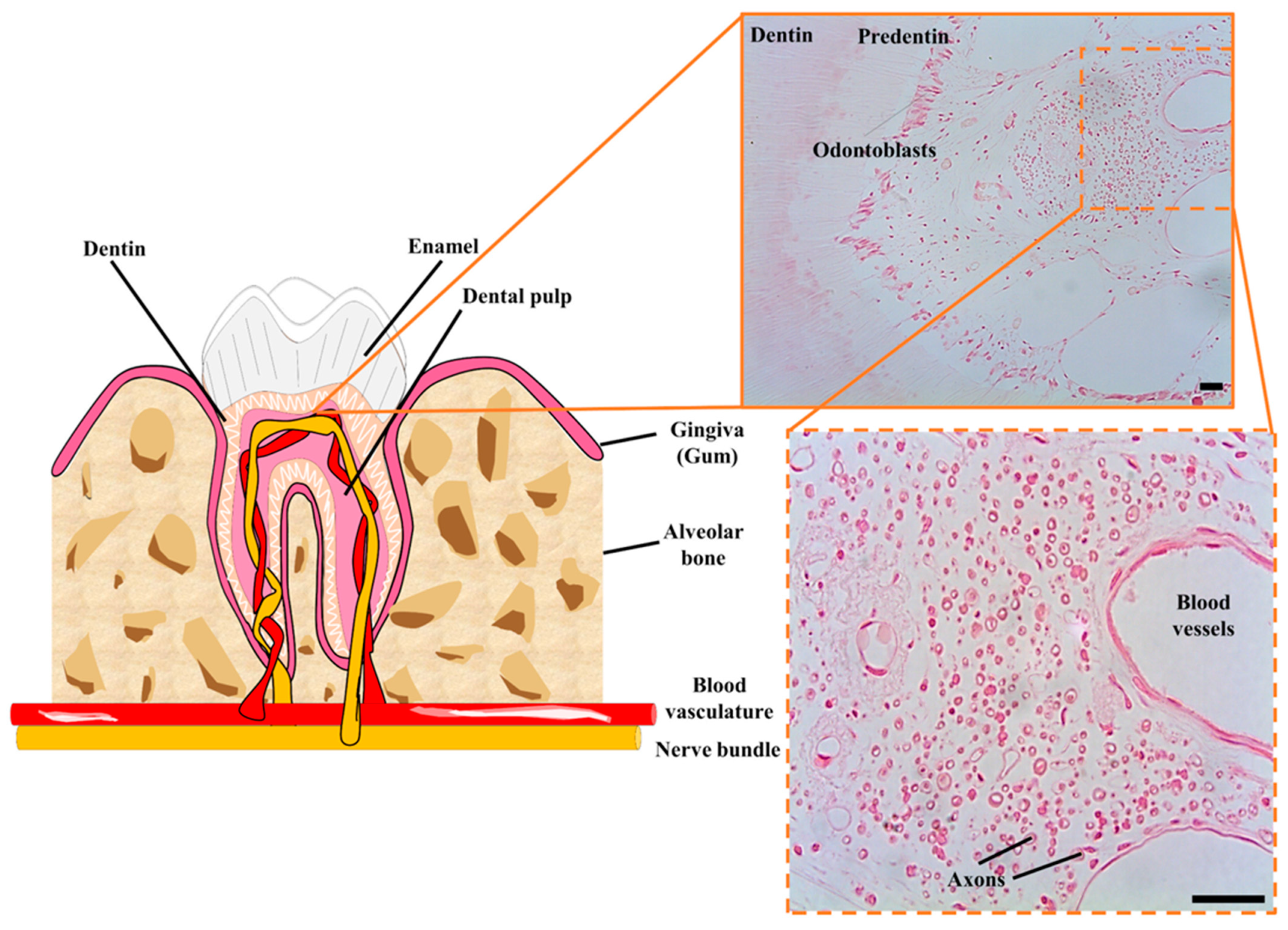
2. DPSCs as Neural Crest Stem Cells. The postnatal DPSC Niche
3. Multilineage Differentiation Characteristics and Protocols
4. Results of Neuronal and Glial Differentiation
5. Neuronal-Like Differentiation from hDPSCs: Problems and Confusions
6. Neuroprotective and Immunomodulatory Capacity. Extracellular Vesicles
7. DPSCs in Combination with Scaffold Materials for Neuroregeneration
8. Perspectives on the Applications of hDPSCs in Neuroregenerative Therapies
9. Conclusions and Future Directions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Yang, W.; Hamilton, J.L.; Kopil, C.; Beck, J.C.; Tanner, C.M.; Albin, R.L.; Ray Dorsey, E.; Dahodwala, N.; Cintina, I.; Hogan, P.; et al. Current and Projected Future Economic Burden of Parkinson’s Disease in the U.S. NPJ Park. Dis. 2020, 6, 1–9. [Google Scholar] [CrossRef]
- Glidden, A.M.; Luebbe, E.A.; Elson, M.J.; Goldenthal, S.B.; Snyder, C.W.; Zizzi, C.E.; Dorsey, E.R.; Heatwole, C.R. Patient-Reported Impact of Symptoms in Huntington Disease: PRISM-HD. Neurology 2020, 94, e2045–e2053. [Google Scholar] [CrossRef]
- Carnahan, J.L.; Judge, K.S.; Daggy, J.K.; Slaven, J.E.; Coleman, N.; Fortier, E.L.; Suelzer, C.; Fowler, N.R. Supporting Caregivers of Veterans with Alzheimer’s Disease and Traumatic Brain Injury: Study Protocol for a Randomized Controlled Trial. Trials 2020, 21, 340. [Google Scholar] [CrossRef] [PubMed]
- Sivanandam, T.M.; Thakur, M.K. Traumatic Brain Injury: A Risk Factor for Alzheimer’s Disease. Neurosci. Biobehav. Rev. 2012, 36, 1376–1381. [Google Scholar] [CrossRef] [PubMed]
- Smith, D.H.; Johnson, V.E.; Stewart, W. Chronic Neuropathologies of Single and Repetitive TBI: Substrates of Dementia? Nat. Rev. Neurol. 2013, 9, 211–221. [Google Scholar] [CrossRef]
- Chiti, F.; Dobson, C.M. Protein Misfolding, Amyloid Formation, and Human Disease: A Summary of Progress Over the Last Decade. Annu. Rev. Biochem. 2017, 86, 27–68. [Google Scholar] [CrossRef]
- Nieto-Sampedro, M. Neurite Outgrowth Inhibitors in Gliotic Tissue. Adv. Exp. Med. Biol. 1999, 468, 207–224. [Google Scholar] [CrossRef]
- Bovolenta, P.; Fernaud-Espinosa, I.; Méndez-Otero, R.; Nieto-Sampedro, M. Neurite Outgrowth Inhibitor of Gliotic Brain Tissue. Mode of Action and Cellular Localization, Studied with Specific Monoclonal Antibodies. Eur. J. Neurosci. 1997, 9, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Arvidsson, A.; Collin, T.; Kirik, D.; Kokaia, Z.; Lindvall, O. Neuronal Replacement from Endogenous Precursors in the Adult Brain after Stroke. Nat. Med. 2002, 8, 963–970. [Google Scholar] [CrossRef] [PubMed]
- Solleiro-Villavicencio, H.; Rivas-Arancibia, S. Effect of Chronic Oxidative Stress on Neuroinflammatory Response Mediated by CD4+T Cells in Neurodegenerative Diseases. Front. Cell. Neurosci. 2018, 12. [Google Scholar] [CrossRef] [PubMed]
- Subramanian, L.; Calcagnotto, M.E.; Paredes, M.F. Cortical Malformations: Lessons in Human Brain Development. Front. Cell. Neurosci. 2020, 13, 576. [Google Scholar] [CrossRef] [PubMed]
- Doetsch, F.; García-Verdugo, J.M.; Alvarez-Buylla, A. Cellular Composition and Three-Dimensional Organization of the Subventricular Germinal Zone in the Adult Mammalian Brain. J. Neurosci. 1997, 17, 5046–5061. [Google Scholar] [CrossRef] [PubMed]
- Seri, B.; García-Verdugo, J.M.; McEwen, B.S.; Alvarez-Buylla, A. Astrocytes Give Rise to New Neurons in the Adult Mammalian Hippocampus. J. Neurosci. 2001, 21, 7153–7160. [Google Scholar] [CrossRef] [PubMed]
- Van Praag, H.; Schinder, A.F.; Christie, B.R.; Toni, N.; Palmer, T.D.; Gage, F.H. Functional Neurogenesis in the Adult Hippocampus. Nature 2002, 415, 1030–1034. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; You, Y.; Qi, D.; Zhou, X.; Wang, L.; Wei, S.; Zhang, Z.; Huang, W.; Liu, Z.; Liu, F.; et al. Human and Monkey Striatal Interneurons Are Derived from the Medial Ganglionic Eminence But Not from the Adult Subventricular Zone. J. Neurosci. 2014, 34, 10906–10923. [Google Scholar] [CrossRef] [PubMed]
- Sorrells, S.F.; Paredes, M.F.; Cebrian-Silla, A.; Sandoval, K.; Qi, D.; Kelley, K.W.; James, D.; Mayer, S.; Chang, J.; Auguste, K.I.; et al. Human Hippocampal Neurogenesis Drops Sharply in Children to Undetectable Levels in Adults. Nature 2018, 555, 377–381. [Google Scholar] [CrossRef] [PubMed]
- Sanai, N.; Nguyen, T.; Ihrie, R.A.; Mirzadeh, Z.; Tsai, H.-H.; Wong, M.; Gupta, N.; Berger, M.S.; Huang, E.; Garcia-Verdugo, J.-M.; et al. Corridors of Migrating Neurons in the Human Brain and Their Decline during Infancy. Nature 2011, 478, 382–386. [Google Scholar] [CrossRef] [PubMed]
- Moreno-Jiménez, E.P.; Flor-García, M.; Terreros-Roncal, J.; Rábano, A.; Cafini, F.; Pallas-Bazarra, N.; Ávila, J.; Llorens-Martín, M. Adult Hippocampal Neurogenesis Is Abundant in Neurologically Healthy Subjects and Drops Sharply in Patients with Alzheimer’s Disease. Nat. Med. 2019, 25, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Cipriani, S.; Journiac, N.; Nardelli, J.; Verney, C.; Delezoide, A.-L.; Guimiot, F.; Gressens, P.; Adle-Biassette, H. Dynamic Expression Patterns of Progenitor and Neuron Layer Markers in the Developing Human Dentate Gyrus and Fimbria. Cereb. Cortex 2017. [Google Scholar] [CrossRef] [PubMed]
- Knoth, R.; Singec, I.; Ditter, M.; Pantazis, G.; Capetian, P.; Meyer, R.P.; Horvat, V.; Volk, B.; Kempermann, G. Murine Features of Neurogenesis in the Human Hippocampus across the Lifespan from 0 to 100 Years. PLoS ONE 2010, 5, e8809. [Google Scholar] [CrossRef]
- Blümcke, I.; Schewe, J.-C.; Normann, S.; Brüstle, O.; Schramm, J.; Elger, C.E.; Wiestler, O.D. Increase of Nestin-Immunoreactive Neural Precursor Cells in the Dentate Gyrus of Pediatric Patients with Early-Onset Temporal Lobe Epilepsy: Increased Hippocampal Neurogenesis in Human TLE. Hippocampus 2001, 11, 311–321. [Google Scholar] [CrossRef]
- Sierra, A.; Martín-Suárez, S.; Valcárcel-Martín, R.; Pascual-Brazo, J.; Aelvoet, S.-A.; Abiega, O.; Deudero, J.J.; Brewster, A.L.; Bernales, I.; Anderson, A.E.; et al. Neuronal Hyperactivity Accelerates Depletion of Neural Stem Cells and Impairs Hippocampal Neurogenesis. Cell Stem Cell 2015, 16, 488–503. [Google Scholar] [CrossRef] [PubMed]
- Jure, I.; De Nicola, A.F.; Encinas, J.M.; Labombarda, F. Spinal Cord Injury Leads to Hippocampal Glial Alterations and Neural Stem Cell Inactivation. Cell. Mol. Neurobiol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Faiz, M.; Sachewsky, N.; Gascón, S.; Bang, K.W.A.; Morshead, C.M.; Nagy, A. Adult Neural Stem Cells from the Subventricular Zone Give Rise to Reactive Astrocytes in the Cortex after Stroke. Cell Stem Cell 2015, 17, 624–634. [Google Scholar] [CrossRef] [PubMed]
- Llorens-Bobadilla, E.; Chell, J.M.; Le Merre, P.; Wu, Y.; Zamboni, M.; Bergenstråhle, J.; Stenudd, M.; Sopova, E.; Lundeberg, J.; Shupliakov, O.; et al. A Latent Lineage Potential in Resident Neural Stem Cells Enables Spinal Cord Repair. Science 2020, 370. [Google Scholar] [CrossRef] [PubMed]
- Zamboni, M.; Llorens-Bobadilla, E.; Magnusson, J.P.; Frisén, J. A Widespread Neurogenic Potential of Neocortical Astrocytes Is Induced by Injury. Cell Stem Cell 2020, 27, 605–617.e5. [Google Scholar] [CrossRef] [PubMed]
- Chareyron, L.J.; Amaral, D.G.; Lavenex, P. Selective Lesion of the Hippocampus Increases the Differentiation of Immature Neurons in the Monkey Amygdala. Proc. Natl. Acad. Sci. USA 2016, 113, 14420–14425. [Google Scholar] [CrossRef] [PubMed]
- Kawabori, M.; Weintraub, A.H.; Imai, H.; Zinkevych, L.; McAllister, P.; Steinberg, G.K.; Frishberg, B.M.; Yasuhara, T.; Chen, J.W.; Cramer, S.C.; et al. Cell Therapy for Chronic TBI: Interim Analysis of the Randomized Controlled STEMTRA Trial. Neurology 2021. [Google Scholar] [CrossRef] [PubMed]
- Dewan, S.; Schimmel, S.; Borlongan, C.V. Treating Childhood Traumatic Brain Injury with Autologous Stem Cell Therapy. Expert Opin. Biol. Ther. 2018, 18, 515–524. [Google Scholar] [CrossRef] [PubMed]
- Bachoud-Lévi, A.-C.; Massart, R.; Rosser, A. Cell Therapy in Huntington’s Disease: Taking Stock of Past Studies to Move the Field Forward. Stem Cells Dayt. Ohio 2021, 39, 144–155. [Google Scholar] [CrossRef]
- Bachoud-Lévi, A.-C. From Open to Large-Scale Randomized Cell Transplantation Trials in Huntington’s Disease: Lessons from the Multicentric Intracerebral Grafting in Huntington’s Disease Trial (MIG-HD) and Previous Pilot Studies. Prog. Brain Res. 2017, 230, 227–261. [Google Scholar] [CrossRef]
- Parmar, M. Towards Stem Cell Based Therapies for Parkinson’s Disease. Dev. Camb. Engl. 2018, 145. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.W.; Koo, S.Y.; Studer, L. Pluripotent Stem Cell Therapies for Parkinson Disease: Present Challenges and Future Opportunities. Front. Cell Dev. Biol. 2020, 8, 729. [Google Scholar] [CrossRef] [PubMed]
- Fernández-Muñoz, B.; Rosell-Valle, C.; Ferrari, D.; Alba-Amador, J.; Montiel, M.Á.; Campos-Cuerva, R.; Lopez-Navas, L.; Muñoz-Escalona, M.; Martín-López, M.; Profico, D.C.; et al. Retrieval of Germinal Zone Neural Stem Cells from the Cerebrospinal Fluid of Premature Infants with Intraventricular Hemorrhage. Stem Cells Transl. Med. 2020, 9, 1085–1101. [Google Scholar] [CrossRef] [PubMed]
- Si, Z.; Wang, X. Stem Cells Therapies in Alzheimer’s Disease: Applications for Disease Modeling. J. Pharmacol. Exp. Ther. 2021. [Google Scholar] [CrossRef]
- Vasic, V.; Barth, K.; Schmidt, M.H.H. Neurodegeneration and Neuro-Regeneration-Alzheimer’s Disease and Stem Cell Therapy. Int. J. Mol. Sci. 2019, 20, 4272. [Google Scholar] [CrossRef]
- Bachoud-Lévi, A.; Bourdet, C.; Brugières, P.; Nguyen, J.P.; Grandmougin, T.; Haddad, B.; Jény, R.; Bartolomeo, P.; Boissé, M.F.; Barba, G.D.; et al. Safety and Tolerability Assessment of Intrastriatal Neural Allografts in Five Patients with Huntington’s Disease. Exp. Neurol. 2000, 161, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Gaura, V.; Bachoud-Lévi, A.-C.; Ribeiro, M.-J.; Nguyen, J.-P.; Frouin, V.; Baudic, S.; Brugières, P.; Mangin, J.-F.; Boissé, M.-F.; Palfi, S.; et al. Striatal Neural Grafting Improves Cortical Metabolism in Huntington’s Disease Patients. Brain J. Neurol. 2004, 127, 65–72. [Google Scholar] [CrossRef]
- Li, Y.; Shen, P.-P.; Wang, B. Induced Pluripotent Stem Cell Technology for Spinal Cord Injury: A Promising Alternative Therapy. Neural Regen. Res. 2021, 16, 1500–1509. [Google Scholar] [CrossRef] [PubMed]
- Morizane, A. Cell therapy for Parkinson’s disease with induced pluripotent stem cells. Rinsho Shinkeigaku 2019, 59, 119–124. [Google Scholar] [CrossRef] [PubMed]
- Lavazza, A.; Massimini, M. Cerebral Organoids: Ethical Issues and Consciousness Assessment. J. Med. Ethics 2018, 44, 606–610. [Google Scholar] [CrossRef]
- Ferrari, D.; Gelati, M.; Profico, D.C.; Vescovi, A.L. Human Fetal Neural Stem Cells for Neurodegenerative Disease Treatment. Results Probl. Cell Differ. 2018, 66, 307–329. [Google Scholar] [CrossRef] [PubMed]
- Shorr, A.F. Abortion and Fetal Tissue Research: Some Ethical Concerns. Fetal Diagn. Ther. 1994, 9, 196–203. [Google Scholar] [CrossRef] [PubMed]
- Panikkar, B.; Smith, N.; Brown, P. Reflexive Research Ethics in Fetal Tissue Xenotransplantation Research. Account. Res. 2012, 19, 344–369. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yamaguchi, S.; Marumoto, T.; Nii, T.; Kawano, H.; Liao, J.; Nagai, Y.; Okada, M.; Takahashi, A.; Inoue, H.; Sasaki, E.; et al. Characterization of Common Marmoset Dysgerminoma-like Tumor Induced by the Lentiviral Expression of Reprogramming Factors. Cancer Sci 2014, 105, 402–408. [Google Scholar] [CrossRef] [PubMed]
- Qiao, Y.; Agboola, O.S.; Hu, X.; Wu, Y.; Lei, L. Tumorigenic and Immunogenic Properties of Induced Pluripotent Stem Cells: A Promising Cancer Vaccine. Stem Cell Rev. Rep. 2020, 16, 1049–1061. [Google Scholar] [CrossRef] [PubMed]
- Guo, X.; Johe, K.; Molnar, P.; Davis, H.; Hickman, J. Characterization of a Human Fetal Spinal Cord Stem Cell Line NSI-566RSC and Its Induction to Functional Motoneurons. J. Tissue Eng. Regen. Med. 2010, 4, 181–193. [Google Scholar] [CrossRef] [PubMed]
- Curtis, E.; Martin, J.R.; Gabel, B.; Sidhu, N.; Rzesiewicz, T.K.; Mandeville, R.; Van Gorp, S.; Leerink, M.; Tadokoro, T.; Marsala, S.; et al. A First-in-Human, Phase I Study of Neural Stem Cell Transplantation for Chronic Spinal Cord Injury. Cell Stem Cell 2018, 22, 941–950. [Google Scholar] [CrossRef]
- Ford, E.; Pearlman, J.; Ruan, T.; Manion, J.; Waller, M.; Neely, G.G.; Caron, L. Human Pluripotent Stem Cells-Based Therapies for Neurodegenerative Diseases: Current Status and Challenges. Cells 2020, 9, 2517. [Google Scholar] [CrossRef] [PubMed]
- Sepehrimanesh, M.; Ding, B. Generation and Optimization of Highly Pure Motor Neurons from Human Induced Pluripotent Stem Cells via Lentiviral Delivery of Transcription Factors. Am. J. Physiol. Cell Physiol. 2020, 319, C771–C780. [Google Scholar] [CrossRef]
- Klapper, S.D.; Garg, P.; Dagar, S.; Lenk, K.; Gottmann, K.; Nieweg, K. Astrocyte Lineage Cells Are Essential for Functional Neuronal Differentiation and Synapse Maturation in Human IPSC-Derived Neural Networks. Glia 2019, 67, 1893–1909. [Google Scholar] [CrossRef]
- Toft, M. Advances in Genetic Diagnosis of Neurological Disorders. Acta Neurol. Scand. Suppl. 2014, 20–25. [Google Scholar] [CrossRef]
- Fatima, A.; Gutiérrez-Garcia, R.; Vilchez, D. Induced Pluripotent Stem Cells from Huntington’s Disease Patients: A Promising Approach to Define and Correct Disease-Related Alterations. Neural Regen. Res. 2019, 14, 769–770. [Google Scholar] [CrossRef] [PubMed]
- Dabrowska, S.; Andrzejewska, A.; Janowski, M.; Lukomska, B. Immunomodulatory and Regenerative Effects of Mesenchymal Stem Cells and Extracellular Vesicles: Therapeutic Outlook for Inflammatory and Degenerative Diseases. Front. Immunol. 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Dominici, M.; Le Blanc, K.; Mueller, I.; Slaper-Cortenbach, I.; Marini, F.; Krause, D.; Deans, R.; Keating, A.; Prockop, D.; Horwitz, E. Minimal Criteria for Defining Multipotent Mesenchymal Stromal Cells. The International Society for Cellular Therapy Position Statement. Cytotherapy 2006, 8, 315–317. [Google Scholar] [CrossRef] [PubMed]
- Anitua, E.; Troya, M.; Zalduendo, M. Progress in the Use of Dental Pulp Stem Cells in Regenerative Medicine. Cytotherapy 2018, 20, 479–498. [Google Scholar] [CrossRef]
- Kawashima, N. Characterisation of Dental Pulp Stem Cells: A New Horizon for Tissue Regeneration? Arch. Oral Biol. 2012, 57, 1439–1458. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; He, Y.; Wang, X.; Key, B.; Lee, B.H.; Li, H.; Ye, Q. Potential Roles of Dental Pulp Stem Cells in Neural Regeneration and Repair. Stem Cells Int. 2018, 2018, 1731289. [Google Scholar] [CrossRef] [PubMed]
- Gronthos, S.; Brahim, J.; Li, W.; Fisher, L.W.; Cherman, N.; Boyde, A.; DenBesten, P.; Robey, P.G.; Shi, S. Stem Cell Properties of Human Dental Pulp Stem Cells. J. Dent. Res. 2002, 81, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Vaquero, J.; Zurita, M.; Rico, M.A.; Bonilla, C.; Aguayo, C.; Montilla, J.; Bustamante, S.; Carballido, J.; Marin, E.; Martinez, F.; et al. An Approach to Personalized Cell Therapy in Chronic Complete Paraplegia: The Puerta de Hierro Phase I/II Clinical Trial. Cytotherapy 2016, 18, 1025–1036. [Google Scholar] [CrossRef]
- Gronthos, S.; Mankani, M.; Brahim, J.; Robey, P.G.; Shi, S. Postnatal Human Dental Pulp Stem Cells (DPSCs) in vitro and in Vivo. Proc. Natl. Acad. Sci. USA 2000, 97, 13625–13630. [Google Scholar] [CrossRef] [PubMed]
- Seo, B.-M.; Miura, M.; Gronthos, S.; Bartold, P.M.; Batouli, S.; Brahim, J.; Young, M.; Robey, P.G.; Wang, C.-Y.; Shi, S. Investigation of Multipotent Postnatal Stem Cells from Human Periodontal Ligament. Lancet Lond. Engl. 2004, 364, 149–155. [Google Scholar] [CrossRef]
- Zhang, Q.Z.; Nguyen, A.L.; Yu, W.H.; Le, A.D. Human Oral Mucosa and Gingiva: A Unique Reservoir for Mesenchymal Stem Cells. J. Dent. Res. 2012, 91, 1011–1018. [Google Scholar] [CrossRef] [PubMed]
- Huang, G.T.-J.; Sonoyama, W.; Liu, Y.; Liu, H.; Wang, S.; Shi, S. The Hidden Treasure in Apical Papilla: The Potential Role in Pulp/Dentin Regeneration and Bioroot Engineering. J. Endod. 2008, 34, 645–651. [Google Scholar] [CrossRef]
- Morsczeck, C.; Götz, W.; Schierholz, J.; Zeilhofer, F.; Kühn, U.; Möhl, C.; Sippel, C.; Hoffmann, K.H. Isolation of Precursor Cells (PCs) from Human Dental Follicle of Wisdom Teeth. Matrix Biol. J. Int. Soc. Matrix Biol. 2005, 24, 155–165. [Google Scholar] [CrossRef]
- Miura, M.; Gronthos, S.; Zhao, M.; Lu, B.; Fisher, L.W.; Robey, P.G.; Shi, S. SHED: Stem Cells from Human Exfoliated Deciduous Teeth. Proc. Natl. Acad. Sci. USA 2003, 100, 5807–5812. [Google Scholar] [CrossRef]
- Huang, G.T.-J.; Gronthos, S.; Shi, S. Mesenchymal Stem Cells Derived from Dental Tissues vs. Those from Other Sources: Their Biology and Role in Regenerative Medicine. J. Dent. Res. 2009, 88, 792–806. [Google Scholar] [CrossRef]
- Janebodin, K.; Horst, O.V.; Ieronimakis, N.; Balasundaram, G.; Reesukumal, K.; Pratumvinit, B.; Reyes, M. Isolation and Characterization of Neural Crest-Derived Stem Cells from Dental Pulp of Neonatal Mice. PLoS ONE 2011, 6, e27526. [Google Scholar] [CrossRef] [PubMed]
- Méndez-Maldonado, K.; Vega-López, G.A.; Aybar, M.J.; Velasco, I. Neurogenesis From Neural Crest Cells: Molecular Mechanisms in the Formation of Cranial Nerves and Ganglia. Front. Cell Dev. Biol. 2020, 8, 635. [Google Scholar] [CrossRef]
- Ibarretxe, G.; Crende, O.; Aurrekoetxea, M.; García-Murga, V.; Etxaniz, J.; Unda, F. Neural Crest Stem Cells from Dental Tissues: A New Hope for Dental and Neural Regeneration. Stem Cells Int. 2012, 2012, 103503. [Google Scholar] [CrossRef]
- Luzuriaga, J.; Pineda, J.R.; Irastorza, I.; Uribe-Etxebarria, V.; García-Gallastegui, P.; Encinas, J.M.; Chamero, P.; Unda, F.; Ibarretxe, G. BDNF and NT3 Reprogram Human Ectomesenchymal Dental Pulp Stem Cells to Neurogenic and Gliogenic Neural Crest Progenitors Cultured in Serum-Free Medium. Cell. Physiol. Biochem. Int. J. Exp. Cell. Physiol. Biochem. Pharmacol. 2019, 52, 1361–1380. [Google Scholar] [CrossRef]
- Martens, W.; Wolfs, E.; Struys, T.; Politis, C.; Bronckaers, A.; Lambrichts, I. Expression Pattern of Basal Markers in Human Dental Pulp Stem Cells and Tissue. Cells Tissues Organs 2012, 196, 490–500. [Google Scholar] [CrossRef]
- Uribe-Etxebarria, V.; Luzuriaga, J.; García-Gallastegui, P.; Agliano, A.; Unda, F.; Ibarretxe, G. Notch/Wnt Cross-Signalling Regulates Stemness of Dental Pulp Stem Cells through Expression of Neural Crest and Core Pluripotency Factors. Eur. Cell. Mater. 2017, 34, 249–270. [Google Scholar] [CrossRef]
- Uribe-Etxebarria, V.; García-Gallastegui, P.; Pérez-Garrastachu, M.; Casado-Andrés, M.; Irastorza, I.; Unda, F.; Ibarretxe, G.; Subirán, N. Wnt-3a Induces Epigenetic Remodeling in Human Dental Pulp Stem Cells. Cells 2020, 9, 652. [Google Scholar] [CrossRef] [PubMed]
- Uribe-Etxebarria, V.; Agliano, A.; Unda, F.; Ibarretxe, G. Wnt Signaling Reprograms Metabolism in Dental Pulp Stem Cells. J. Cell. Physiol. 2019, 234, 13068–13082. [Google Scholar] [CrossRef] [PubMed]
- Mitsiadis, T.A.; Woloszyk, A. Odyssey of Human Dental Pulp Stem Cells and Their Remarkable Ability to Survive in Extremely Adverse Conditions. Front. Physiol. 2015, 6, 99. [Google Scholar] [CrossRef] [PubMed]
- Nancy, A. Ten Cate’s Oral Histology, 9th ed.; Elsevier: Amsterdam, The Netherlands, 2017; ISBN 978-0-323-48524-1. [Google Scholar]
- Shi, S.; Gronthos, S. Perivascular Niche of Postnatal Mesenchymal Stem Cells in Human Bone Marrow and Dental Pulp. J. Bone Miner. Res. Off. J. Am. Soc. Bone Miner. Res. 2003, 18, 696–704. [Google Scholar] [CrossRef] [PubMed]
- Kaukua, N.; Shahidi, M.K.; Konstantinidou, C.; Dyachuk, V.; Kaucka, M.; Furlan, A.; An, Z.; Wang, L.; Hultman, I.; Ahrlund-Richter, L.; et al. Glial Origin of Mesenchymal Stem Cells in a Tooth Model System. Nature 2014, 513, 551–554. [Google Scholar] [CrossRef] [PubMed]
- Luzuriaga, J.; Pastor-Alonso, O.; Encinas, J.M.; Unda, F.; Ibarretxe, G.; Pineda, J.R. Human Dental Pulp Stem Cells Grown in Neurogenic Media Differentiate Into Endothelial Cells and Promote Neovasculogenesis in the Mouse Brain. Front. Physiol. 2019, 10, 347. [Google Scholar] [CrossRef] [PubMed]
- Luzuriaga, J.; Irurzun, J.; Irastorza, I.; Unda, F.; Ibarretxe, G.; Pineda, J.R. Vasculogenesis from Human Dental Pulp Stem Cells Grown in Matrigel with Fully Defined Serum-Free Culture Media. Biomedicines 2020, 8, 483. [Google Scholar] [CrossRef]
- Pisciotta, A.; Bertani, G.; Bertoni, L.; Di Tinco, R.; De Biasi, S.; Vallarola, A.; Pignatti, E.; Tupler, R.; Salvarani, C.; de Pol, A.; et al. Modulation of Cell Death and Promotion of Chondrogenic Differentiation by Fas/FasL in Human Dental Pulp Stem Cells (HDPSCs). Front. Cell Dev. Biol. 2020, 8, 279. [Google Scholar] [CrossRef] [PubMed]
- Zordani, A.; Pisciotta, A.; Bertoni, L.; Bertani, G.; Vallarola, A.; Giuliani, D.; Puliatti, S.; Mecugni, D.; Bianchi, G.; de Pol, A.; et al. Regenerative Potential of Human Dental Pulp Stem Cells in the Treatment of Stress Urinary Incontinence: In Vitro and in Vivo Study. Cell Prolif. 2019, 52, e12675. [Google Scholar] [CrossRef] [PubMed]
- Pisciotta, A.; Riccio, M.; Carnevale, G.; Beretti, F.; Gibellini, L.; Maraldi, T.; Cavallini, G.M.; Ferrari, A.; Bruzzesi, G.; De Pol, A. Human Serum Promotes Osteogenic Differentiation of Human Dental Pulp Stem Cells in vitro and in vivo. PLoS ONE 2012, 7, e50542. [Google Scholar] [CrossRef] [PubMed]
- Király, M.; Porcsalmy, B.; Pataki, A.; Kádár, K.; Jelitai, M.; Molnár, B.; Hermann, P.; Gera, I.; Grimm, W.-D.; Ganss, B.; et al. Simultaneous PKC and CAMP Activation Induces Differentiation of Human Dental Pulp Stem Cells into Functionally Active Neurons. Neurochem. Int. 2009, 55, 323–332. [Google Scholar] [CrossRef]
- Arthur, A.; Rychkov, G.; Shi, S.; Koblar, S.A.; Gronthos, S. Adult Human Dental Pulp Stem Cells Differentiate toward Functionally Active Neurons under Appropriate Environmental Cues. Stem Cells Dayt. Ohio 2008, 26, 1787–1795. [Google Scholar] [CrossRef] [PubMed]
- Gregory, C.A.; Reyes, E.; Whitney, M.J.; Spees, J.L. Enhanced Engraftment of Mesenchymal Stem Cells in a Cutaneous Wound Model by Culture in Allogenic Species-Specific Serum and Administration in Fibrin Constructs. Stem Cells Dayt. Ohio 2006, 24, 2232–2243. [Google Scholar] [CrossRef] [PubMed]
- Horwitz, E.M.; Gordon, P.L.; Koo, W.K.K.; Marx, J.C.; Neel, M.D.; McNall, R.Y.; Muul, L.; Hofmann, T. Isolated Allogeneic Bone Marrow-Derived Mesenchymal Cells Engraft and Stimulate Growth in Children with Osteogenesis Imperfecta: Implications for Cell Therapy of Bone. Proc. Natl. Acad. Sci. USA 2002, 99, 8932–8937. [Google Scholar] [CrossRef] [PubMed]
- Shi, K.; Tian, D.-C.; Li, Z.-G.; Ducruet, A.F.; Lawton, M.T.; Shi, F.-D. Global Brain Inflammation in Stroke. Lancet Neurol. 2019, 18, 1058–1066. [Google Scholar] [CrossRef]
- Pacini, S. Deterministic and Stochastic Approaches in the Clinical Application of Mesenchymal Stromal Cells (MSCs). Front. Cell Dev. Biol. 2014, 2. [Google Scholar] [CrossRef] [PubMed]
- Bonnamain, V.; Thinard, R.; Sergent-Tanguy, S.; Huet, P.; Bienvenu, G.; Naveilhan, P.; Farges, J.-C.; Alliot-Licht, B. Human Dental Pulp Stem Cells Cultured in Serum-Free Supplemented Medium. Front. Physiol. 2013, 4, 357. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Gervois, P.; Struys, T.; Hilkens, P.; Bronckaers, A.; Ratajczak, J.; Politis, C.; Brône, B.; Lambrichts, I.; Martens, W. Neurogenic Maturation of Human Dental Pulp Stem Cells Following Neurosphere Generation Induces Morphological and Electrophysiological Characteristics of Functional Neurons. Stem Cells Dev. 2015, 24, 296–311. [Google Scholar] [CrossRef] [PubMed]
- Mokry, J.; Soukup, T.; Micuda, S.; Karbanova, J.; Visek, B.; Brcakova, E.; Suchanek, J.; Bouchal, J.; Vokurkova, D.; Ivancakova, R. Telomere Attrition Occurs during Ex Vivo Expansion of Human Dental Pulp Stem Cells. J. Biomed. Biotechnol. 2010, 2010, 673513. [Google Scholar] [CrossRef] [PubMed]
- Xiao, L.; Tsutsui, T. Characterization of Human Dental Pulp Cells-Derived Spheroids in Serum-Free Medium: Stem Cells in the Core. J. Cell. Biochem. 2013, 114, 2624–2636. [Google Scholar] [CrossRef]
- Li, D.; Zou, X.-Y.; El-Ayachi, I.; Romero, L.O.; Yu, Z.; Iglesias-Linares, A.; Cordero-Morales, J.F.; Huang, G.T.-J. Human Dental Pulp Stem Cells and Gingival Mesenchymal Stem Cells Display Action Potential Capacity In Vitro after Neuronogenic Differentiation. Stem Cell Rev. Rep. 2019, 15, 67–81. [Google Scholar] [CrossRef] [PubMed]
- Kanafi, M.; Majumdar, D.; Bhonde, R.; Gupta, P.; Datta, I. Midbrain Cues Dictate Differentiation of Human Dental Pulp Stem Cells towards Functional Dopaminergic Neurons. J. Cell. Physiol. 2014, 229, 1369–1377. [Google Scholar] [CrossRef] [PubMed]
- Niapour, N.; Mohammadi-Ghalehbin, B.; Golmohammadi, M.G.; Gholami, M.R.; Amani, M.; Niapour, A. An Efficient System for Selection and Culture of Schwann Cells from Adult Rat Peripheral Nerves. Cytotechnology 2016, 68, 629–636. [Google Scholar] [CrossRef] [PubMed]
- Martens, W.; Sanen, K.; Georgiou, M.; Struys, T.; Bronckaers, A.; Ameloot, M.; Phillips, J.; Lambrichts, I. Human Dental Pulp Stem Cells Can Differentiate into Schwann Cells and Promote and Guide Neurite Outgrowth in an Aligned Tissue-Engineered Collagen Construct in Vitro. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2014, 28, 1634–1643. [Google Scholar] [CrossRef] [PubMed]
- Pisciotta, A.; Bertoni, L.; Vallarola, A.; Bertani, G.; Mecugni, D.; Carnevale, G. Neural Crest Derived Stem Cells from Dental Pulp and Tooth-Associated Stem Cells for Peripheral Nerve Regeneration. Neural Regen. Res. 2020, 15, 373–381. [Google Scholar] [CrossRef] [PubMed]
- Wang, D.-R.; Wang, Y.-H.; Pan, J.; Tian, W.-D. Neurotrophic Effects of Dental Pulp Stem Cells in Repair of Peripheral Nerve after Crush Injury. World J. Stem Cells 2020, 12, 1196–1213. [Google Scholar] [CrossRef]
- Sakai, K.; Yamamoto, A.; Matsubara, K.; Nakamura, S.; Naruse, M.; Yamagata, M.; Sakamoto, K.; Tauchi, R.; Wakao, N.; Imagama, S.; et al. Human Dental Pulp-Derived Stem Cells Promote Locomotor Recovery after Complete Transection of the Rat Spinal Cord by Multiple Neuro-Regenerative Mechanisms. J. Clin. Investig. 2012, 122, 80–90. [Google Scholar] [CrossRef]
- Kolar, M.K.; Itte, V.N.; Kingham, P.J.; Novikov, L.N.; Wiberg, M.; Kelk, P. The Neurotrophic Effects of Different Human Dental Mesenchymal Stem Cells. Sci. Rep. 2017, 7, 12605. [Google Scholar] [CrossRef] [PubMed]
- Ibarretxe, G.; Perrais, D.; Jaskolski, F.; Vimeney, A.; Mulle, C. Fast Regulation of Axonal Growth Cone Motility by Electrical Activity. J. Neurosci. Off. J. Soc. Neurosci. 2007, 27, 7684–7695. [Google Scholar] [CrossRef]
- Gritti, A.; Parati, E.A.; Cova, L.; Frolichsthal, P.; Galli, R.; Wanke, E.; Faravelli, L.; Morassutti, D.J.; Roisen, F.; Nickel, D.D.; et al. Multipotential Stem Cells from the Adult Mouse Brain Proliferate and Self-Renew in Response to Basic Fibroblast Growth Factor. J. Neurosci. Off. J. Soc. Neurosci. 1996, 16, 1091–1100. [Google Scholar] [CrossRef]
- Rutecki, P.A. Neuronal Excitability: Voltage-Dependent Currents and Synaptic Transmission. J. Clin. Neurophysiol. Off. Publ. Am. Electroencephalogr. Soc. 1992, 9, 195–211. [Google Scholar] [CrossRef]
- Pagella, P.; Miran, S.; Neto, E.; Martin, I.; Lamghari, M.; Mitsiadis, T.A. Human Dental Pulp Stem Cells Exhibit Enhanced Properties in Comparison to Human Bone Marrow Stem Cells on Neurites Outgrowth. FASEB J. Off. Publ. Fed. Am. Soc. Exp. Biol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Liebau, S.; Vaida, B.; Storch, A.; Boeckers, T.M. Maturation of Synaptic Contacts in Differentiating Neural Stem Cells. Stem Cells Dayt. Ohio 2007, 25, 1720–1729. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, G.; Trubert, C.; Terrien, J.; Pifferi, F.; Leroy, D.; Loyens, A.; Migaud, M.; Baroncini, M.; Maurage, C.-A.; Fontaine, C.; et al. A Comparative Study of the Neural Stem Cell Niche in the Adult Hypothalamus of Human, Mouse, Rat and Gray Mouse Lemur (Microcebus Murinus). J. Comp. Neurol. 2018, 526, 1419–1443. [Google Scholar] [CrossRef] [PubMed]
- Rafiee, F.; Pourteymourfard-Tabrizi, Z.; Mahmoudian-Sani, M.-R.; Mehri-Ghahfarrokhi, A.; Soltani, A.; Hashemzadeh-Chaleshtori, M.; Jami, M.-S. Differentiation of Dental Pulp Stem Cells into Neuron-like Cells. Int. J. Neurosci. 2020, 130, 107–116. [Google Scholar] [CrossRef]
- Kogo, Y.; Seto, C.; Totani, Y.; Mochizuki, M.; Nakahara, T.; Oka, K.; Yoshioka, T.; Ito, E. Rapid Differentiation of Human Dental Pulp Stem Cells to Neuron-like Cells by High K+ Stimulation. Biophys. Phys. 2020, 17, 132–139. [Google Scholar] [CrossRef]
- Gomes, M.d.M. Franz Nissl (1860–1919), Noted Neuropsychiatrist and Neuropathologist, Staining the Neuron, but Not Limiting It. Dement. Neuropsychol. 2019, 13, 352–355. [Google Scholar] [CrossRef]
- Inada, E.; Saitoh, I.; Kubota, N.; Iwase, Y.; Kiyokawa, Y.; Shibasaki, S.; Noguchi, H.; Yamasaki, Y.; Sato, M. PiggyBac Transposon-Based Immortalization of Human Deciduous Tooth Dental Pulp Cells with Multipotency and Non-Tumorigenic Potential. Int. J. Mol. Sci. 2019, 20, 4904. [Google Scholar] [CrossRef]
- Mead, B.; Logan, A.; Berry, M.; Leadbeater, W.; Scheven, B.A. Paracrine-Mediated Neuroprotection and Neuritogenesis of Axotomised Retinal Ganglion Cells by Human Dental Pulp Stem Cells: Comparison with Human Bone Marrow and Adipose-Derived Mesenchymal Stem Cells. PLoS ONE 2014, 9, e109305. [Google Scholar] [CrossRef]
- Caseiro, A.R.; Pereira, T.; Ivanova, G.; Luís, A.L.; Maurício, A.C. Neuromuscular Regeneration: Perspective on the Application of Mesenchymal Stem Cells and Their Secretion Products. Stem Cells Int. 2016, 2016, 16. [Google Scholar] [CrossRef] [PubMed]
- Nosrat, I.V.; Widenfalk, J.; Olson, L.; Nosrat, C.A. Dental Pulp Cells Produce Neurotrophic Factors, Interact with Trigeminal Neurons in Vitro, and Rescue Motoneurons after Spinal Cord Injury. Dev. Biol. 2001, 238, 120–132. [Google Scholar] [CrossRef] [PubMed]
- Arthur, A.; Shi, S.; Zannettino, A.C.W.; Fujii, N.; Gronthos, S.; Koblar, S.A. Implanted Adult Human Dental Pulp Stem Cells Induce Endogenous Axon Guidance. Stem Cells Dayt. Ohio 2009, 27, 2229–2237. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Lee, J.-H.; Bae, J.; Bu, Y.; Kim, E.-C. Human Dental Pulp Stem Cells Are More Effective Than Human Bone Marrow-Derived Mesenchymal Stem Cells in Cerebral Ischemic Injury. Cell Transplant. 2017, 26, 1001–1016. [Google Scholar] [CrossRef] [PubMed]
- Heneka, M.T.; Carson, M.J.; El Khoury, J.; Landreth, G.E.; Brosseron, F.; Feinstein, D.L.; Jacobs, A.H.; Wyss-Coray, T.; Vitorica, J.; Ransohoff, R.M.; et al. Neuroinflammation in Alzheimer’s Disease. Lancet Neurol. 2015, 14, 388–405. [Google Scholar] [CrossRef]
- Andre, R.; Carty, L.; Tabrizi, S.J. Disruption of Immune Cell Function by Mutant Huntingtin in Huntington’s Disease Pathogenesis. Curr. Opin. Pharmacol. 2016, 26, 33–38. [Google Scholar] [CrossRef] [PubMed]
- Amor, S.; Peferoen, L.A.N.; Vogel, D.Y.S.; Breur, M.; van der Valk, P.; Baker, D.; van Noort, J.M. Inflammation in Neurodegenerative Diseases—An Update. Immunology 2014, 142, 151–166. [Google Scholar] [CrossRef]
- Eugen-Olsen, J.; Andersen, O.; Linneberg, A.; Ladelund, S.; Hansen, T.W.; Langkilde, A.; Petersen, J.; Pielak, T.; Møller, L.N.; Jeppesen, J.; et al. Circulating Soluble Urokinase Plasminogen Activator Receptor Predicts Cancer, Cardiovascular Disease, Diabetes and Mortality in the General Population. J. Intern. Med. 2010, 268, 296–308. [Google Scholar] [CrossRef]
- Koch, A.; Voigt, S.; Kruschinski, C.; Sanson, E.; Dückers, H.; Horn, A.; Yagmur, E.; Zimmermann, H.; Trautwein, C.; Tacke, F. Circulating Soluble Urokinase Plasminogen Activator Receptor Is Stably Elevated during the First Week of Treatment in the Intensive Care Unit and Predicts Mortality in Critically Ill Patients. Crit. Care Lond. Engl. 2011, 15, R63. [Google Scholar] [CrossRef] [PubMed]
- Isola, G.; Polizzi, A.; Alibrandi, A.; Williams, R.C.; Leonardi, R. Independent Impact of Periodontitis and Cardiovascular Disease on Elevated Soluble Urokinase-Type Plasminogen Activator Receptor (SuPAR) Levels. J. Periodontol. 2020. [Google Scholar] [CrossRef]
- Rasmussen, L.J.H.; Caspi, A.; Ambler, A.; Danese, A.; Elliott, M.; Eugen-Olsen, J.; Hariri, A.R.; Harrington, H.; Houts, R.; Poulton, R.; et al. Association between Elevated SuPAR, a New Biomarker of Inflammation, and Accelerated Aging. J. Gerontol. A. Biol. Sci. Med. Sci. 2021, 76, 318–327. [Google Scholar] [CrossRef] [PubMed]
- Bianco, J.; De Berdt, P.; Deumens, R.; des Rieux, A. Taking a Bite out of Spinal Cord Injury: Do Dental Stem Cells Have the Teeth for It? Cell. Mol. Life Sci. CMLS 2016, 73, 1413–1437. [Google Scholar] [CrossRef] [PubMed]
- Özdemir, A.T.; Özgül Özdemir, R.B.; Kırmaz, C.; Sarıboyacı, A.E.; Ünal Halbutoğlları, Z.S.; Özel, C.; Karaöz, E. The Paracrine Immunomodulatory Interactions between the Human Dental Pulp Derived Mesenchymal Stem Cells and CD4 T Cell Subsets. Cell. Immunol. 2016, 310, 108–115. [Google Scholar] [CrossRef]
- Tomic, S.; Djokic, J.; Vasilijic, S.; Vucevic, D.; Todorovic, V.; Supic, G.; Colic, M. Immunomodulatory Properties of Mesenchymal Stem Cells Derived from Dental Pulp and Dental Follicle Are Susceptible to Activation by Toll-like Receptor Agonists. Stem Cells Dev. 2011, 20, 695–708. [Google Scholar] [CrossRef]
- Kwack, K.H.; Lee, J.M.; Park, S.H.; Lee, H.W. Human Dental Pulp Stem Cells Suppress Alloantigen-Induced Immunity by Stimulating T Cells to Release Transforming Growth Factor Beta. J. Endod. 2017, 43, 100–108. [Google Scholar] [CrossRef]
- Sugita, S.; Kamao, H.; Iwasaki, Y.; Okamoto, S.; Hashiguchi, T.; Iseki, K.; Hayashi, N.; Mandai, M.; Takahashi, M. Inhibition of T-Cell Activation by Retinal Pigment Epithelial Cells Derived from Induced Pluripotent Stem Cells. Investig. Ophthalmol. Vis. Sci. 2015, 56, 1051–1062. [Google Scholar] [CrossRef]
- He, W.; Qu, T.; Yu, Q.; Wang, Z.; Lv, H.; Zhang, J.; Zhao, X.; Wang, P. LPS Induces IL-8 Expression through TLR4, MyD88, NF-KappaB and MAPK Pathways in Human Dental Pulp Stem Cells. Int. Endod. J. 2013, 46, 128–136. [Google Scholar] [CrossRef] [PubMed]
- Demircan, P.C.; Sariboyaci, A.E.; Unal, Z.S.; Gacar, G.; Subasi, C.; Karaoz, E. Immunoregulatory Effects of Human Dental Pulp-Derived Stem Cells on T Cells: Comparison of Transwell Co-Culture and Mixed Lymphocyte Reaction Systems. Cytotherapy 2011, 13, 1205–1220. [Google Scholar] [CrossRef]
- Pierdomenico, L.; Bonsi, L.; Calvitti, M.; Rondelli, D.; Arpinati, M.; Chirumbolo, G.; Becchetti, E.; Marchionni, C.; Alviano, F.; Fossati, V.; et al. Multipotent Mesenchymal Stem Cells with Immunosuppressive Activity Can Be Easily Isolated from Dental Pulp. Transplantation 2005, 80, 836–842. [Google Scholar] [CrossRef] [PubMed]
- Di Nicola, M.; Carlo-Stella, C.; Magni, M.; Milanesi, M.; Longoni, P.D.; Matteucci, P.; Grisanti, S.; Gianni, A.M. Human Bone Marrow Stromal Cells Suppress T-Lymphocyte Proliferation Induced by Cellular or Nonspecific Mitogenic Stimuli. Blood 2002, 99, 3838–3843. [Google Scholar] [CrossRef]
- Le Blanc, K.; Mougiakakos, D. Multipotent Mesenchymal Stromal Cells and the Innate Immune System. Nat. Rev. Immunol. 2012, 12, 383–396. [Google Scholar] [CrossRef] [PubMed]
- Selmani, Z.; Naji, A.; Zidi, I.; Favier, B.; Gaiffe, E.; Obert, L.; Borg, C.; Saas, P.; Tiberghien, P.; Rouas-Freiss, N.; et al. Human Leukocyte Antigen-G5 Secretion by Human Mesenchymal Stem Cells Is Required to Suppress T Lymphocyte and Natural Killer Function and to Induce CD4+CD25highFOXP3+ Regulatory T Cells. Stem Cells Dayt. Ohio 2008, 26, 212–222. [Google Scholar] [CrossRef] [PubMed]
- Casiraghi, F.; Azzollini, N.; Cassis, P.; Imberti, B.; Morigi, M.; Cugini, D.; Cavinato, R.A.; Todeschini, M.; Solini, S.; Sonzogni, A.; et al. Pretransplant Infusion of Mesenchymal Stem Cells Prolongs the Survival of a Semiallogeneic Heart Transplant through the Generation of Regulatory T Cells. J. Immunol. 2008, 181, 3933–3946. [Google Scholar] [CrossRef] [PubMed]
- Heldring, N.; Mäger, I.; Wood, M.J.A.; Le Blanc, K.; Andaloussi, S.E.L. Therapeutic Potential of Multipotent Mesenchymal Stromal Cells and Their Extracellular Vesicles. Hum. Gene Ther. 2015, 26, 506–517. [Google Scholar] [CrossRef] [PubMed]
- Buzas, E.I.; György, B.; Nagy, G.; Falus, A.; Gay, S. Emerging Role of Extracellular Vesicles in Inflammatory Diseases. Nat. Rev. Rheumatol. 2014, 10, 356–364. [Google Scholar] [CrossRef]
- Thompson, A.G.; Gray, E.; Heman-Ackah, S.M.; Mäger, I.; Talbot, K.; Andaloussi, S.E.; Wood, M.J.; Turner, M.R. Extracellular Vesicles in Neurodegenerative Disease—Pathogenesis to Biomarkers. Nat. Rev. Neurol. 2016, 12, 346–357. [Google Scholar] [CrossRef] [PubMed]
- Schorey, J.S.; Cheng, Y.; Singh, P.P.; Smith, V.L. Exosomes and Other Extracellular Vesicles in Host-Pathogen Interactions. EMBO Rep. 2015, 16, 24–43. [Google Scholar] [CrossRef]
- Tolar, J.; Le Blanc, K.; Keating, A.; Blazar, B.R. Concise Review: Hitting the Right Spot with Mesenchymal Stromal Cells. Stem Cells Dayt. Ohio 2010, 28, 1446–1455. [Google Scholar] [CrossRef]
- De Jong, O.G.; Van Balkom, B.W.M.; Schiffelers, R.M.; Bouten, C.V.C.; Verhaar, M.C. Extracellular Vesicles: Potential Roles in Regenerative Medicine. Front. Immunol. 2014, 5, 608. [Google Scholar] [CrossRef] [PubMed]
- Jin, T.; Gu, J.; Li, Z.; Xu, Z.; Gui, Y. Recent Advances on Extracellular Vesicles in Central Nervous System Diseases. Clin. Interv. Aging 2021, 16, 257–274. [Google Scholar] [CrossRef] [PubMed]
- Harrell, C.R.; Volarevic, A.; Djonov, V.; Volarevic, V. Mesenchymal Stem Cell-Derived Exosomes as New Remedy for the Treatment of Neurocognitive Disorders. Int. J. Mol. Sci. 2021, 22, 1433. [Google Scholar] [CrossRef]
- Garbuzova-Davis, S.; Borlongan, C.V. Stem Cell-Derived Extracellular Vesicles as Potential Mechanism for Repair of Microvascular Damage within and Outside of the Central Nervous System in Amyotrophic Lateral Sclerosis: Perspective Schema. Neural Regen. Res. 2021, 16, 680–681. [Google Scholar] [CrossRef] [PubMed]
- Merckx, G.; Hosseinkhani, B.; Kuypers, S.; Deville, S.; Irobi, J.; Nelissen, I.; Michiels, L.; Lambrichts, I.; Bronckaers, A. Angiogenic Effects of Human Dental Pulp and Bone Marrow-Derived Mesenchymal Stromal Cells and Their Extracellular Vesicles. Cells 2020, 9, 312. [Google Scholar] [CrossRef] [PubMed]
- Ciregia, F.; Urbani, A.; Palmisano, G. Extracellular Vesicles in Brain Tumors and Neurodegenerative Diseases. Front. Mol. Neurosci. 2017, 10, 276. [Google Scholar] [CrossRef]
- Guervilly, C.; Bonifay, A.; Burtey, S.; Sabatier, F.; Cauchois, R.; Abdili, E.; Arnaud, L.; Lano, G.; Pietri, L.; Robert, T.; et al. Dissemination of Extreme Levels of Extracellular Vesicles: Tissue Factor Activity in Patients with Severe COVID-19. Blood Adv. 2021, 5, 628–634. [Google Scholar] [CrossRef] [PubMed]
- Couch, Y.; Akbar, N.; Roodselaar, J.; Evans, M.C.; Gardiner, C.; Sargent, I.; Romero, I.A.; Bristow, A.; Buchan, A.M.; Haughey, N.; et al. Circulating Endothelial Cell-Derived Extracellular Vesicles Mediate the Acute Phase Response and Sickness Behaviour Associated with CNS Inflammation. Sci. Rep. 2017, 7, 9574. [Google Scholar] [CrossRef] [PubMed]
- Henriques, D.; Moreira, R.; Schwamborn, J.; Pereira de Almeida, L.; Mendonça, L.S. Successes and Hurdles in Stem Cells Application and Production for Brain Transplantation. Front. Neurosci. 2019, 13, 1194. [Google Scholar] [CrossRef]
- Luo, L.; He, Y.; Jin, L.; Zhang, Y.; Guastaldi, F.P.; Albashari, A.A.; Hu, F.; Wang, X.; Wang, L.; Xiao, J.; et al. Application of Bioactive Hydrogels Combined with Dental Pulp Stem Cells for the Repair of Large Gap Peripheral Nerve Injuries. Bioact. Mater. 2021, 6, 638–654. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.; Feng, G.; Zhang, J.; Xing, J.; Huang, D.; Lian, M.; Zhang, W.; Wu, W.; Hu, Y.; Lu, X.; et al. Basic Fibroblast Growth Factor Promotes Human Dental Pulp Stem Cells Cultured in 3D Porous Chitosan Scaffolds to Neural Differentiation. Int. J. Neurosci. 2020, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Lu, X.; Feng, G.; Gu, Z.; Sun, Y.; Bao, G.; Xu, G.; Lu, Y.; Chen, J.; Xu, L.; et al. Chitosan Scaffolds Induce Human Dental Pulp Stem Cells to Neural Differentiation: Potential Roles for Spinal Cord Injury Therapy. Cell Tissue Res. 2016, 366, 129–142. [Google Scholar] [CrossRef]
- Tian, J.-L.; Zhao, Y.-Z.; Jin, Z.; Lu, C.-T.; Tang, Q.-Q.; Xiang, Q.; Sun, C.-Z.; Zhang, L.; Xu, Y.-Y.; Gao, H.-S.; et al. Synthesis and Characterization of Poloxamer 188-Grafted Heparin Copolymer. Drug Dev. Ind. Pharm. 2010, 36, 832–838. [Google Scholar] [CrossRef] [PubMed]
- Luo, L.; Albashari, A.A.; Wang, X.; Jin, L.; Zhang, Y.; Zheng, L.; Xia, J.; Xu, H.; Zhao, Y.; Xiao, J.; et al. Effects of Transplanted Heparin-Poloxamer Hydrogel Combining Dental Pulp Stem Cells and BFGF on Spinal Cord Injury Repair. Stem Cells Int. 2018, 2018, 2398521. [Google Scholar] [CrossRef] [PubMed]
- Albashari, A.; He, Y.; Zhang, Y.; Ali, J.; Lin, F.; Zheng, Z.; Zhang, K.; Cao, Y.; Xu, C.; Luo, L.; et al. Thermosensitive BFGF-Modified Hydrogel with Dental Pulp Stem Cells on Neuroinflammation of Spinal Cord Injury. ACS Omega 2020, 5, 16064–16075. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Dai, J. Bridging the Gap with Functional Collagen Scaffolds: Tuning Endogenous Neural Stem Cells for Severe Spinal Cord Injury Repair. Biomater. Sci. 2018, 6, 265–271. [Google Scholar] [CrossRef] [PubMed]
- Carnevale, G.; Pisciotta, A.; Riccio, M.; Bertoni, L.; De Biasi, S.; Gibellini, L.; Zordani, A.; Cavallini, G.M.; La Sala, G.B.; Bruzzesi, G.; et al. Human Dental Pulp Stem Cells Expressing STRO-1, c-Kit and CD34 Markers in Peripheral Nerve Regeneration. J. Tissue Eng. Regen. Med. 2018, 12, e774–e785. [Google Scholar] [CrossRef] [PubMed]
- Sanen, K.; Martens, W.; Georgiou, M.; Ameloot, M.; Lambrichts, I.; Phillips, J. Engineered Neural Tissue with Schwann Cell Differentiated Human Dental Pulp Stem Cells: Potential for Peripheral Nerve Repair? J. Tissue Eng. Regen. Med. 2017, 11, 3362–3372. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.; Aoki, S.; Yamato, M.; Uchiyama, H.; Wada, K.; Okano, T.; Ogiuchi, H. Tubulation with Dental Pulp Cells Promotes Facial Nerve Regeneration in Rats. Tissue Eng. Part A 2008, 14, 1141–1147. [Google Scholar] [CrossRef]
- Sasaki, R.; Matsumine, H.; Watanabe, Y.; Takeuchi, Y.; Yamato, M.; Okano, T.; Miyata, M.; Ando, T. Electrophysiologic and Functional Evaluations of Regenerated Facial Nerve Defects with a Tube Containing Dental Pulp Cells in Rats. Plast. Reconstr. Surg. 2014, 134, 970–978. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, R.; Aoki, S.; Yamato, M.; Uchiyama, H.; Wada, K.; Ogiuchi, H.; Okano, T.; Ando, T. PLGA Artificial Nerve Conduits with Dental Pulp Cells Promote Facial Nerve Regeneration. J. Tissue Eng. Regen. Med. 2011, 5, 823–830. [Google Scholar] [CrossRef] [PubMed]
- Rosa, V.; Xie, H.; Dubey, N.; Madanagopal, T.T.; Rajan, S.S.; Morin, J.L.P.; Islam, I.; Castro Neto, A.H. Graphene Oxide-Based Substrate: Physical and Surface Characterization, Cytocompatibility and Differentiation Potential of Dental Pulp Stem Cells. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2016, 32, 1019–1025. [Google Scholar] [CrossRef]
- Seonwoo, H.; Jang, K.-J.; Lee, D.; Park, S.; Lee, M.; Park, S.; Lim, K.-T.; Kim, J.; Chung, J.H. Neurogenic Differentiation of Human Dental Pulp Stem Cells on Graphene-Polycaprolactone Hybrid Nanofibers. Nanomaterials 2018, 8, 554. [Google Scholar] [CrossRef] [PubMed]
- Yam, G.H.-F.; Peh, G.S.-L.; Singhal, S.; Goh, B.-T.; Mehta, J.S. Dental Stem Cells: A Future Asset of Ocular Cell Therapy. Expert Rev. Mol. Med. 2015, 17, e20. [Google Scholar] [CrossRef] [PubMed]
- Gnanasegaran, N.; Govindasamy, V.; Simon, C.; Gan, Q.F.; Vincent-Chong, V.K.; Mani, V.; Krishnan Selvarajan, K.; Subramaniam, V.; Musa, S.; Abu Kasim, N.H. Effect of Dental Pulp Stem Cells in MPTP-Induced Old-Aged Mice Model. Eur. J. Clin. Investig. 2017, 47, 403–414. [Google Scholar] [CrossRef]
- Chun, S.Y.; Soker, S.; Jang, Y.-J.; Kwon, T.G.; Yoo, E.S. Differentiation of Human Dental Pulp Stem Cells into Dopaminergic Neuron-like Cells in Vitro. J. Korean Med. Sci. 2016, 31, 171–177. [Google Scholar] [CrossRef] [PubMed]
- Nosrat, I.V.; Smith, C.A.; Mullally, P.; Olson, L.; Nosrat, C.A. Dental Pulp Cells Provide Neurotrophic Support for Dopaminergic Neurons and Differentiate into Neurons in Vitro; Implications for Tissue Engineering and Repair in the Nervous System. Eur. J. Neurosci. 2004, 19, 2388–2398. [Google Scholar] [CrossRef] [PubMed]
- Fujii, H.; Matsubara, K.; Sakai, K.; Ito, M.; Ohno, K.; Ueda, M.; Yamamoto, A. Dopaminergic Differentiation of Stem Cells from Human Deciduous Teeth and Their Therapeutic Benefits for Parkinsonian Rats. Brain Res. 2015, 1613, 59–72. [Google Scholar] [CrossRef]
- Gnanasegaran, N.; Govindasamy, V.; Mani, V.; Abu Kasim, N.H. Neuroimmunomodulatory Properties of DPSCs in an in vitro Model of Parkinson’s Disease. IUBMB Life 2017, 69, 689–699. [Google Scholar] [CrossRef]
- Eskandari, N.; Boroujeni, M.E.; Abdollahifar, M.A.; Piryaei, A.; Khodagholi, F.; Mirbehbahani, S.H.; Siroosi, S.; Moghaddam, M.H.; Aliaghaei, A.; Sadeghi, Y. Transplantation of Human Dental Pulp Stem Cells Compensates for Striatal Atrophy and Modulates Neuro-Inflammation in 3-Nitropropionic Acid Rat Model of Huntington’s Disease. Neurosci. Res. 2020. [Google Scholar] [CrossRef]
- Ahmed, N.E.-M.B.; Murakami, M.; Hirose, Y.; Nakashima, M. Therapeutic Potential of Dental Pulp Stem Cell Secretome for Alzheimer’s Disease Treatment: An In Vitro Study. Stem Cells Int. 2016, 2016, 8102478. [Google Scholar] [CrossRef]
- Mita, T.; Furukawa-Hibi, Y.; Takeuchi, H.; Hattori, H.; Yamada, K.; Hibi, H.; Ueda, M.; Yamamoto, A. Conditioned Medium from the Stem Cells of Human Dental Pulp Improves Cognitive Function in a Mouse Model of Alzheimer’s Disease. Behav. Brain Res. 2015, 293, 189–197. [Google Scholar] [CrossRef] [PubMed]
- Zhou, H.; Li, X.; Yin, Y.; He, X.-T.; An, Y.; Tian, B.-M.; Hong, Y.-L.; Wu, L.-A.; Chen, F.-M. The Proangiogenic Effects of Extracellular Vesicles Secreted by Dental Pulp Stem Cells Derived from Periodontally Compromised Teeth. Stem Cell Res. Ther. 2020, 11, 110. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, T.; Kalka, C.; Masuda, H.; Chen, D.; Silver, M.; Kearney, M.; Magner, M.; Isner, J.M.; Asahara, T. Ischemia- and Cytokine-Induced Mobilization of Bone Marrow-Derived Endothelial Progenitor Cells for Neovascularization. Nat. Med. 1999, 5, 434–438. [Google Scholar] [CrossRef]
- Sugiyama, M.; Iohara, K.; Wakita, H.; Hattori, H.; Ueda, M.; Matsushita, K.; Nakashima, M. Dental Pulp-Derived CD31−/CD146− Side Population Stem/Progenitor Cells Enhance Recovery of Focal Cerebral Ischemia in Rats. Tissue Eng. Part A 2011, 17, 1303–1311. [Google Scholar] [CrossRef]
- Leong, W.K.; Henshall, T.L.; Arthur, A.; Kremer, K.L.; Lewis, M.D.; Helps, S.C.; Field, J.; Hamilton-Bruce, M.A.; Warming, S.; Manavis, J.; et al. Human Adult Dental Pulp Stem Cells Enhance Poststroke Functional Recovery through Non-Neural Replacement Mechanisms. Stem Cells Transl. Med. 2012, 1, 177–187. [Google Scholar] [CrossRef] [PubMed]
- Teijin Pharma Limited. An Exploratory, Double-Blind, Randomized, Placebo-Controlled Study to Evaluate Safety and Efficacy of JTR-161 in Patients with Acute Ischemic Stroke; Clinicaltrials.gov: U.S National Library of Medicine; NIH USA: Bethesda, MD, USA, 2020.
- Nagpal, A.; Kremer, K.L.; Hamilton-Bruce, M.A.; Kaidonis, X.; Milton, A.G.; Levi, C.; Shi, S.; Carey, L.; Hillier, S.; Rose, M.; et al. TOOTH (The Open Study Of Dental Pulp Stem Cell Therapy in Humans): Study Protocol for Evaluating Safety and Feasibility of Autologous Human Adult Dental Pulp Stem Cell Therapy in Patients with Chronic Disability after Stroke. Int. J. Stroke Off. J. Int. Stroke Soc. 2016, 11, 575–585. [Google Scholar] [CrossRef] [PubMed]
- Hasan, A.; Deeb, G.; Rahal, R.; Atwi, K.; Mondello, S.; Marei, H.E.; Gali, A.; Sleiman, E. Mesenchymal Stem Cells in the Treatment of Traumatic Brain Injury. Front. Neurol. 2017, 8, 28. [Google Scholar] [CrossRef]
- Brasil, A. Dose-Response Evaluation of the Investigational Product Cellavita HD after Intravenous Administration in Patients with Huntington’s Disease; Clinicaltrials.gov: U.S National Library of Medicine, NIH USA: Bethesda, MD, US, 2019.
- Zayed, M.; Iohara, K. Immunomodulation and Regeneration Properties of Dental Pulp Stem Cells: A Potential Therapy to Treat Coronavirus Disease 2019. Cell Transplant. 2020, 29. [Google Scholar] [CrossRef] [PubMed]
- Ye, Q.; Wang, H.; Xia, X.; Zhou, C.; Liu, Z.; Xia, Z.-E.; Zhang, Z.; Zhao, Y.; Yehenala, J.; Wang, S.; et al. Safety and Efficacy Assessment of Allogeneic Human Dental Pulp Stem Cells to Treat Patients with Severe COVID-19: Structured Summary of a Study Protocol for a Randomized Controlled Trial (Phase I/II). Trials 2020, 21, 520. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Luzuriaga, J.; Polo, Y.; Pastor-Alonso, O.; Pardo-Rodríguez, B.; Larrañaga, A.; Unda, F.; Sarasua, J.-R.; Pineda, J.R.; Ibarretxe, G. Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies. Int. J. Mol. Sci. 2021, 22, 3546. https://doi.org/10.3390/ijms22073546
Luzuriaga J, Polo Y, Pastor-Alonso O, Pardo-Rodríguez B, Larrañaga A, Unda F, Sarasua J-R, Pineda JR, Ibarretxe G. Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies. International Journal of Molecular Sciences. 2021; 22(7):3546. https://doi.org/10.3390/ijms22073546
Chicago/Turabian StyleLuzuriaga, Jon, Yurena Polo, Oier Pastor-Alonso, Beatriz Pardo-Rodríguez, Aitor Larrañaga, Fernando Unda, Jose-Ramon Sarasua, Jose Ramon Pineda, and Gaskon Ibarretxe. 2021. "Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies" International Journal of Molecular Sciences 22, no. 7: 3546. https://doi.org/10.3390/ijms22073546
APA StyleLuzuriaga, J., Polo, Y., Pastor-Alonso, O., Pardo-Rodríguez, B., Larrañaga, A., Unda, F., Sarasua, J.-R., Pineda, J. R., & Ibarretxe, G. (2021). Advances and Perspectives in Dental Pulp Stem Cell Based Neuroregeneration Therapies. International Journal of Molecular Sciences, 22(7), 3546. https://doi.org/10.3390/ijms22073546








