Genetics of Host Protection against Helicobacter pylori Infections
Abstract
1. Introduction
2. Helicobacter pylori–Human Host Interaction
3. Why Study Infectious Disease Genes?
4. Detection of Infectious Disease Genes: Candidate Gene Studies
5. Detection of Infectious Disease Genes: Genome-Wide Association Studies (GWAS)
6. Presence in the Population of Different Strains of the Same Pathogen
7. Infectious Disease Genes Controlled at the Transcriptional Level
8. Hp Modulates Gene Expression through Epigenetics and Co-Infection
9. Resistance to Pathogen May Be Ephemeral
10. Potential Role of Hp against Inflammatory and Autoimmune Diseases
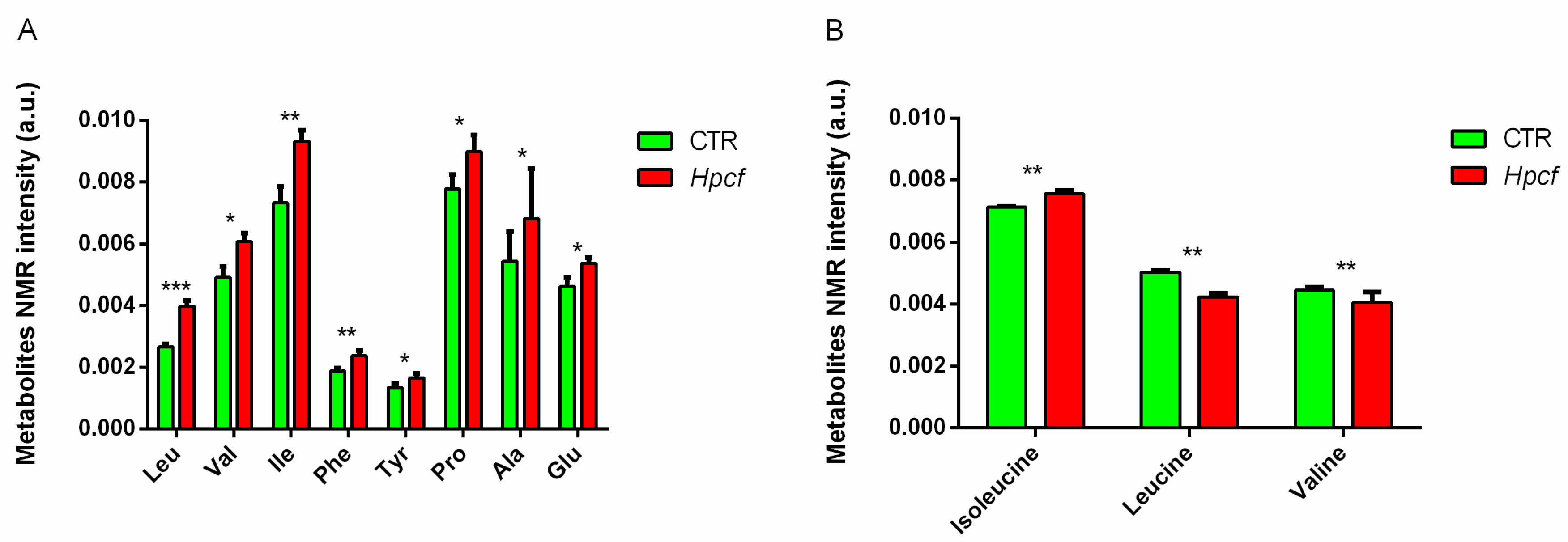
11. Hp and Metabolic Diseases
12. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Archer, N.M.; Petersen, N.; Clark, M.A.; Buckee, C.O.; Childs, L.M.; Duraisingh, M.T. Resistance to Plasmodium falciparum in sickle cell trait erythrocytes is driven by oxygen-dependent growth inhibition. Proc. Natl. Acad. Sci. USA 2018, 115, 7350–7355. [Google Scholar] [CrossRef] [PubMed]
- Miller, L.H.; Mason, S.J.; Clyde, D.F.; McGinniss, M.H. The Resistance Factor to Plasmodium vivax in Blacks. N. Engl. J. Med. 1976, 295, 302–304. [Google Scholar] [CrossRef]
- Dean, M.; Carrington, M.; Winkler, C.; Huttley, G.A.; Smith, M.W.; Allikmets, R.; Goedert, J.J.; Buchbinder, S.P.; Vittinghoff, E.; Gomperts, E.; et al. Genetic restriction of HIV-1 infection and progression to AIDS by a deletion allele of the CKR5 structural gene. Science 1996, 273, 1856–1862. [Google Scholar] [CrossRef]
- Kodaman, N.; Pazos, A.; Schneider, B.G.; Blanca Piazuelo, M.; Mera, R.; Sobota, R.S.; Sicinschi, L.A.; Shaffer, C.L.; Romero-Gallo, J.; De Sablet, T.; et al. Human and Helicobacter pylori coevolution shapes the risk of gastric disease. Proc. Natl. Acad. Sci. USA 2014, 111, 1455–1460. [Google Scholar] [CrossRef] [PubMed]
- Bik, E.M.; Eckburg, P.B.; Gill, S.R.; Nelson, K.E.; Purdom, E.A.; Francois, F.; Perez-Perez, G.; Blaser, M.J.; Relman, D.A. Molecular analysis of the bacterial microbiota in the human stomach. Proc. Natl. Acad. Sci. USA 2006, 103, 732–737. [Google Scholar] [CrossRef] [PubMed]
- Stewart, A.D.; Logsdon, J.M.; Kelley, S.E. An empirical study of the evolution of virulence under both horizontal and vertical transmission. Evolution 2005, 59, 730–739. [Google Scholar] [CrossRef]
- Dorer, M.S.; Talarico, S.; Salama, N.R. Helicobacter pylori’s unconventional role in health and disease. PLoS Pathog. 2009, 5, e1000544. [Google Scholar] [CrossRef]
- Linz, B.; Windsor, H.M.; McGraw, J.J.; Hansen, L.M.; Gajewski, J.P.; Tomsho, L.P.; Hake, C.M.; Solnick, J.V.; Schuster, S.C.; Marshall, B.J. A mutation burst during the acute phase of Helicobacter pylori infection in humans and rhesus macaques. Nat. Commun. 2014, 5, 1–8. [Google Scholar] [CrossRef]
- Ailloud, F.; Didelot, X.; Woltemate, S.; Pfaffinger, G.; Overmann, J.; Bader, R.C.; Schulz, C.; Malfertheiner, P.; Suerbaum, S. Within-host evolution of Helicobacter pylori shaped by niche-specific adaptation, intragastric migrations and selective sweeps. Nat. Commun. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Nejati, S.; Karkhah, A.; Darvish, H.; Validi, M.; Ebarhimpour, S.; Nouri, H.R. Influence of Helicobacter pylori virulence factors CagA and VacA on pathogenesis of gastrointestinal disorders. Microb. Pathog. 2018, 117, 43–48. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.; Yamaoka, Y. Survival of Helicobacter pylori in gastric acidic territory. Helicobacter 2017, 22, e12386. [Google Scholar] [CrossRef]
- Lina, T.T. Immune evasion strategies used by Helicobacter pylori. World J. Gastroenterol. 2014, 20, 12753. [Google Scholar] [CrossRef]
- Noto, J.M.; Chopra, A.; Loh, J.; Romero-Gallo, J.; Blanca Piazuelo, M.; Watson, M.; Leary, S.; Beckett, A.; Wilson, K.; Cover, T.; et al. Pan-genomic analyses identify key Helicobacter pylori pathogenic loci modified by carcinogenic host microenvironments. Gut 2018, 67, 1793–1804. [Google Scholar] [CrossRef]
- Cuomo, P.; Papaianni, M.; Sansone, C.; Iannelli, A.; Iannelli, D.; Medaglia, C.; Paris, D.; Motta, A.; Capparelli, R. An In Vitro Model to Investigate the Role of Helicobacter pylori in Type 2 Diabetes, Obesity, Alzheimer’s Disease and Cardiometabolic Disease. Int. J. Mol. Sci. 2020, 21, 8369. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.A.; Murphy, S.J.; Johnston, B.T.; Watson, R.G.P.; Ferguson, H.R.; Bamford, K.B.; Ghazy, A.; McCarron, P.; McGuigan, J.; Reynolds, J.V.; et al. Relationship between Helicobacter pylori infection and gastric atrophy and the stages of the oesophageal inflammation, metaplasia, adenocarcinoma sequence: Results from the FINBAR case-control study. Gut 2008, 57, 734–739. [Google Scholar] [CrossRef]
- Huang, Y.; Paxton, W.A.; Wolinsky, S.M.; Neumann, A.U.; Zhang, L.; He, T.; Kang, S.; Ceradini, D.; Jin, Z.; Yazdanbakhsh, K.; et al. The role of a mutant CCR5 allele in HIV-1 transmission and disease progression. Nat. Med. 1996, 2, 1240–1243. [Google Scholar] [CrossRef] [PubMed]
- Kellam, P.; Weiss, R.A. Infectogenomics: Insights from the host genome into infectious diseases. Cell 2006, 124, 695–697. [Google Scholar] [CrossRef] [PubMed]
- Cain, J. Perspectives on genetics: Anecdotal, historical, and critical commentaries, 1987–1998. Med Hist. 2002, 46, 453–454. [Google Scholar] [CrossRef]
- Malaty, H.M.; Engstrand, L.; Pedersen, N.L.; Graham, D.Y. Helicobacter pylori infection: Genetic and environmental influences. A study of twins. Ann. Intern. Med. 1994, 120, 982–986. [Google Scholar] [CrossRef] [PubMed]
- Khor, C.C.; Chapman, S.J.; Vannberg, F.O.; Dunne, A.; Murphy, C.; Ling, E.Y.; Frodsham, A.J.; Walley, A.J.; Kyrieleis, O.; Khan, A.; et al. A Mal functional variant is associated with protection against invasive pneumococcal disease, bacteremia, malaria and tuberculosis. Nat. Genet. 2007, 39, 523–528. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Okada, S.; Kong, X.F.; Kreins, A.Y.; Cypowyj, S.; Abhyankar, A.; Toubiana, J.; Itan, Y.; Audry, M.; Nitschke, P.; et al. Gain-of-function human STAT1 mutations impair IL-17 immunity and underlie chronic mucocutaneous candidiasis. J. Exp. Med. 2011, 208, 1635–1648. [Google Scholar] [CrossRef] [PubMed]
- Manolio, T.A.; Collins, F.S.; Cox, N.J.; Goldstein, D.B.; Hindorff, L.A.; Hunter, D.J.; McCarthy, M.I.; Ramos, E.M.; Cardon, L.R.; Chakravarti, A.; et al. Finding the missing heritability of complex diseases. Nature 2009, 461, 747–753. [Google Scholar] [CrossRef]
- Zhang, F.-R.; Huang, W.; Chen, S.-M.; Sun, L.-D.; Liu, H.; Li, Y.; Cui, Y.; Yan, X.-X.; Yang, H.-T.; Rong-De, Y.; et al. Genomewide Association Study of Leprosy. N. Engl. J. Med. 2009, 361, 2609–2618. [Google Scholar] [CrossRef] [PubMed]
- Hill, A.V.S. Evolution, revolution and heresy in the genetics of infectious disease susceptibility. Philos. Trans. R. Soc. B Biol. Sci. 2012, 367, 840–849. [Google Scholar] [CrossRef]
- Monot, M.; Honoré, N.; Garnier, T.; Zidane, N.; Sherafi, D.; Paniz-Mondolfi, A.; Matsuoka, M.; Taylor, G.M.; Donoghue, H.D.; Bouwman, A.; et al. Comparative genomic and phylogeographic analysis of Mycobacterium leprae. Nat. Genet. 2009, 41, 1282–1289. [Google Scholar] [CrossRef]
- Caws, M.; Thwaites, G.; Dunstan, S.; Hawn, T.R.; Lan, N.T.N.; Thuong, N.T.T.; Stepniewska, K.; Huyen, M.N.T.; Nguyen, D.B.; Tran, H.L.; et al. The influence of host and bacterial genotype on the development of disseminated disease with Mycobacterium tuberculosis. PLoS Pathog. 2008, 4. [Google Scholar] [CrossRef] [PubMed]
- McGuire, W.; Hill, A.V.S.; Allsopp, C.E.M.; Greenwood, B.M.; Kwjatkowski, D. Variation in the TNF-α promoter region associated with susceptibility to cerebral malaria. Nature 1994, 371, 508–511. [Google Scholar] [CrossRef] [PubMed]
- Roederer, M.; Quaye, L.; Mangino, M.; Beddall, M.H.; Mahnke, Y.; Chattopadhyay, P.; Tosi, I.; Napolitano, L.; Terranova Barberio, M.; Menni, C.; et al. The genetic architecture of the human immune system: A bioresource for autoimmunity and disease pathogenesis. Cell 2015, 161, 387–403. [Google Scholar] [CrossRef] [PubMed]
- Algood, H.M.S.; Cover, T.L. Helicobacter pylori persistence: An overview of interactions between H. pylori and host immune defenses. Clin. Microbiol. Rev. 2006, 19, 597–613. [Google Scholar] [CrossRef]
- Raghavan, S.; Fredriksson, M.; Svennerholm, A.M.; Holmgren, J.; Suri-Payer, E. Absence of CD4+Cd25+ regulatory T cells is associated with a loss of regulation leading to increased pathology in Helicobacter pylori-infected mice. Clin. Exp. Immunol. 2003, 132, 393–400. [Google Scholar] [CrossRef]
- Jenner, R.G.; Young, R.A. Insights into host responses against pathogens from transcriptional profiling. Nat. Rev. Microbiol. 2005, 3, 281–294. [Google Scholar] [CrossRef]
- Estibariz, I.; Overmann, A.; Ailloud, F.; Krebes, J.; Josenhans, C.; Suerbaum, S. The core genome m5C methyltransferase JHP1050 (M.Hpy99III) plays an important role in orchestrating gene expression in Helicobacter pylori. Nucleic Acids Res. 2019, 47, 2336–2348. [Google Scholar] [CrossRef]
- Niwa, T.; Tsukamoto, T.; Toyoda, T.; Mori, A.; Tanaka, H.; Maekita, T.; Ichinose, M.; Tatematsu, M.; Ushijima, T. Molecular and Cellular Pathobiology Inflammatory Processes Triggered by Helicobacter pylori Infection Cause Aberrant DNA Methylation in Gastric Epithelial Cells. Cancer Res. 2010, 70, 1430–1470. [Google Scholar] [CrossRef]
- Stein, R.A. Epigenetics-The link between infectious diseases and cancer. JAMA J. Am. Med. Assoc. 2011, 305, 1484–1485. [Google Scholar] [CrossRef] [PubMed]
- Pandey, S.; Jha, H.C.; Shukla, S.K.; Shirley, M.K.; Robertson, E.S. Epigenetic regulation of tumor suppressors by Helicobacter pylori enhances EBV-induced proliferation of gastric epithelial cells. MBio 2018, 9. [Google Scholar] [CrossRef]
- Alves, J.M.; Carneiro, M.; Cheng, J.Y.; de Matos, A.L.; Rahman, M.M.; Loog, L.; Campos, P.F.; Wales, N.; Eriksson, A.; Manica, A.; et al. Parallel adaptation of rabbit populations to myxoma virus. Science 2019, 363, 1319–1326. [Google Scholar] [CrossRef]
- Gandon, S.; Michalakis, Y. Evolution of parasite virulence against qualitative or quantitative host resistance. Proc. R. Soc. B Biol. Sci. 2000, 267, 985–990. [Google Scholar] [CrossRef]
- Kerr, P.J.; Cattadori, I.M.; Liu, J.; Sim, D.G.; Dodds, J.W.; Brooks, J.W.; Kennett, M.J.; Holmes, E.C.; Read, A.F. Next step in the ongoing arms race between myxoma virus and wild rabbits in Australia is a novel disease phenotype. Proc. Natl. Acad. Sci. USA 2017, 114, 9397–9402. [Google Scholar] [CrossRef] [PubMed]
- Gandon, S.; Mackinnon, M.J.; Nee, S.; Read, A.F. Imperfect vaccines and the evolution of pathogen virulence. Nature 2001, 414, 751–756. [Google Scholar] [CrossRef] [PubMed]
- Read, A.F.; Baigent, S.J.; Powers, C.; Kgosana, L.B.; Blackwell, L.; Smith, L.P.; Kennedy, D.A.; Walkden-Brown, S.W.; Nair, V.K. Imperfect vaccination can enhance the transmission of highly virulent pathogens. PLoS Biol. 2015, 13. [Google Scholar] [CrossRef]
- Piscione, M.; Mazzone, M.; Di Marcantonio, M.C.; Muraro, R.; Mincione, G. Eradication of Helicobacter pylori and Gastric Cancer: A Controversial Relationship. Front. Microbiol. 2021, 12. [Google Scholar] [CrossRef]
- Viladomiu, M.; Bassaganya-Riera, J.; Tubau-Juni, N.; Kronsteiner, B.; Leber, A.; Philipson, C.W.; Zoccoli-Rodriguez, V.; Hontecillas, R. Cooperation of Gastric Mononuclear Phagocytes with Helicobacter pylori during Colonization. J. Immunol. 2017, 198, 3195–3204. [Google Scholar] [CrossRef]
- Facciabene, A.; Motz, G.T.; Coukos, G. T-regulatory cells: Key players in tumor immune escape and angiogenesis. Cancer Res. 2012, 72, 2162–2171. [Google Scholar] [CrossRef]
- Saini, C.; Ramesh, V.; Nath, I. Increase in TGF-β secreting CD4⁺CD25⁺ FOXP3⁺ T regulatory cells in anergic lepromatous leprosy patients. PLoS Negl. Trop. Dis. 2014, 8, e2639. [Google Scholar] [CrossRef]
- Chionh, Y.T.; Ng, G.Z.; Ong, L.; Arulmuruganar, A.; Stent, A.; Saeed, M.A.; Wee, J.L.K.; Sutton, P. Protease-activated receptor 1 suppresses Helicobacter pylori gastritis via the inhibition of macrophage cytokine secretion and interferon regulatory factor 5. Mucosal Immunol. 2015, 8, 68–79. [Google Scholar] [CrossRef] [PubMed]
- Tubau-Juni, N.; Bassaganya-Riera, J.; Leber, A.; Zoccoli-Rodriguez, V.; Kronsteiner, B.; Viladomiu, M.; Abedi, V.; Philipson, C.W.; Hontecillas, R. Identification of new regulatory genes through expression pattern analysis of a global RNA-seq dataset from a Helicobacter pylori co-culture system. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef]
- Franceschi, F.; Gasbarrini, A.; Polyzos, S.A.; Kountouras, J. Extragastric Diseases and Helicobacter pylori. Helicobacter 2015, 20, 40–46. [Google Scholar] [CrossRef]
- Eisenreich, W.; Rudel, T.; Heesemann, J.; Goebel, W. How viral and intracellular bacterial pathogens reprogram the metabolism of host cells to allow their intracellular replication. Front. Cell. Infect. Microbiol. 2019, 9, 42. [Google Scholar] [CrossRef] [PubMed]
- Ansari, S.; Yamaoka, Y. Helicobacter pylori virulence factors exploiting gastric colonization and its pathogenicity. Toxins 2019, 11, 677. [Google Scholar] [CrossRef]
- Kim, I.J.; Lee, J.; Oh, S.J.; Yoon, M.S.; Jang, S.S.; Holland, R.L.; Reno, M.L.; Hamad, M.N.; Maeda, T.; Chung, H.J.; et al. Helicobacter pylori Infection Modulates Host Cell Metabolism through VacA-Dependent Inhibition of mTORC1. Cell Host Microbe 2018, 23, 583–593.e8. [Google Scholar] [CrossRef] [PubMed]
- White, P.J.; Newgard, C.B. Branched-chain amino acids in disease. Science 2019, 363, 582–583. [Google Scholar] [CrossRef]
- Shao, D.; Villet, O.; Zhang, Z.; Choi, S.W.; Yan, J.; Ritterhoff, J.; Gu, H.; Djukovic, D.; Christodoulou, D.; Kolwicz, S.C.; et al. Glucose promotes cell growth by suppressing branched-chain amino acid degradation. Nat. Commun. 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, M.I.; Romano, M.; Basarali, M.K.; Elzagallaai, A.; Karaman, M.; Demir, Z.; Demir, M.F.; Akcay, F.; Seyrek, M.; Haksever, N.; et al. The Effect of Corrected Inflammation, Oxidative Stress and Endothelial Dysfunction on Fmd Levels in Patients with Selected Chronic Diseases: A Quasi-Experimental Study. Sci. Rep. 2020, 10. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Luc, Y.; Martinez, J.; Bi, Y.; Lian, G.; Wang, T.; Milasta, S.; Wang, J.; Yang, M.; Liu, G.; et al. Proinflammatory signal suppresses proliferation and shifts macrophage metabolism from Myc-dependent to HIF1α-dependent. Proc. Natl. Acad. Sci. USA 2016, 113, 1564–1569. [Google Scholar] [CrossRef]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef]
- D’Souza, G.; Waschina, S.; Pande, S.; Bohl, K.; Kaleta, C.; Kost, C. Less is more: Selective advantages can explain the prevalent loss of biosynthetic genes in bacteria. Evolution 2014, 68, 2559–2570. [Google Scholar] [CrossRef] [PubMed]
- Savenije, O.E.; Mahachie John, J.M.; Granell, R.; Kerkhof, M.; Dijk, F.N.; De Jongste, J.C.; Smit, H.A.; Brunekreef, B.; Postma, D.S.; Van Steen, K.; et al. Association of IL33-IL-1 receptor-like 1 (IL1RL1) pathway polymorphisms with wheezing phenotypes and asthma in childhood. J. Allergy Clin. Immunol. 2014, 134, 170–177. [Google Scholar] [CrossRef]
- Valkov, E.; Stamp, A.; DiMaio, F.; Baker, D.; Verstak, B.; Roversi, P.; Kellie, S.; Sweet, M.J.; Mansell, A.; Gay, N.J.; et al. Crystal structure of toll-like receptor adaptor MAL/TIRAP reveals the molecular basis for signal transduction and disease protection. Proc. Natl. Acad. Sci. USA 2011, 108, 14879–14884. [Google Scholar] [CrossRef] [PubMed]
- Fulgione, A.; Papaianni, M.; Cuomo, P.; Paris, D.; Romano, M.; Tuccillo, C.; Palomba, L.; Medaglia, C.; De Seta, M.; Esposito, N.; et al. Interaction between MyD88, TIRAP and IL1RL1 against Helicobacter pylori infection. Sci. Rep. 2020, 10, 1–13. [Google Scholar] [CrossRef]
| Biological Function | Gene ID | Gene Name |
|---|---|---|
| Signal transduction | APC | Adenomatous Polyposis Coli (APC) Regulator of WNT Signaling Pathway |
| RASSF1A | Ras Association Domain Family Member 1 | |
| Cell cycle regulation | CDH1 | Cadherin 1 |
| CHFR | Checkpoint with Forkhead and Ring Finger Domains | |
| P14/ARF | Cyclin-Dependent Kinase Inhibitor 2A | |
| P15/INK4B | Cyclin-Dependent Kinase Inhibitor 2B | |
| P16/INK4A | Cyclin-Dependent Kinase Inhibitor 2A | |
| Inflammatory response | COX-2 | Mitochondrially Encoded Cytochrome C Oxidase II |
| Apoptosis | DAP-K | Death-Associated Protein Kinase 1 |
| DNA repair | GSTP1 | Glutathione S-Transferase Pi 1 |
| hMLH1 | MutL Homolog 1 | |
| MGMT | O-6-Methylguanine-DNA Methyltransferase | |
| Growth factor | HPP1 | Hyperpigmentation, Progressive, 1 |
| Transcription factor | RUNX3 | RUNX Family Transcription Factor 3 |
| Angiogenesis | THBS1 | Thrombospondin 1 |
| TIMP3 | TIMP Metallopeptidase Inhibitor 3 |
| Pathway Name | Gene ID | Gene Name | Fold Regulation 1 h | Fold Regulation 2 h |
|---|---|---|---|---|
| mTOR signaling pathway | RPTOR | Regulatory associated protein of mTOR complex 1 | −1.42 | 286.04 |
| MLST8 | mTOR associated protein, LST8 homolog (S. cerevisiae) | −1.42 | 398.95 | |
| AKT1 | V-akt murine thymoma viral oncogene homolog 1 | −1.42 | 50.13 | |
| AKT2 | V-akt murine thymoma viral oncogene homolog 2 | −1.42 | 504.97 | |
| INSR | Insulin receptor | −1.42 | 257.79 | |
| IRS1 | Insulin receptor substrate 1 | −1.42 | 278.22 | |
| PLD1 | Phospholipase D1, phosphatidylcholine-specific | −6.31 | 130.70 | |
| RPS6KA2 | Ribosomal protein S6 kinase, 90kDa, polypeptide 2 | −1.24 | 3.37 | |
| PDPK1 | 3-phosphoinositide dependent protein kinase-1 | −1.53 | 28.25 | |
| PIK3CB | Phosphoinositide-3-kinase, catalytic, beta polypeptide | −1.42 | 16.34 | |
| PIK3CD | Phosphoinositide-3-kinase, catalytic, delta polypeptide | 3.37 | 184.83 | |
| PIK3CG | Phosphoinositide-3-kinase, catalytic, gamma polypeptide | −1.42 | 215.28 | |
| CHUK | Conserved helix-loop-helix ubiquitous kinase | −4.08 | 181.03 | |
| EIF4E | Eukaryotic translation initiation factor 4E | −1.42 | 922.92 | |
| HIFIA | Hypoxia inducible factor 1, alpha subunit | 192.93 | 955.47 | |
| Inflammatory pathway | CXCL8 | Interleukin 8 | −3.29 | 2.96 |
| IL-6 | Interleukin 6 | 14.45 | 114.56 | |
| TLR2 | Toll-like receptor 2 | 58 | 72.18 | |
| TLR9 | Toll-like receptor 9 | 3.29 | 134.55 | |
| TNF | Tumor necrosis factor | 12.9 | 154.26 | |
| Oxidative stress pathway | ATOX1 | ATX1 antioxidant protein 1 homolog (yeast) | 3.57 | 37.69 |
| GPX2 | Glutathione peroxidase 2 (gastrointestinal) | 3.57 | 37.69 | |
| GPX4 | Glutathione peroxidase 4 (gastrointestinal) | 3.57 | 37.69 | |
| GSS | Glutathione synthetase | 3.57 | 9.54 | |
| NOX5 | NADPH oxidase, EF-hand calcium binding domain 5 | 3.57 | 7.54 | |
| SOD1 | Superoxide dismutase 1, soluble | −28.68 | −9.67 | |
| SOD2 | Superoxide dismutase 2, mitochondrial | 3.96 | 4.04 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Capparelli, R.; Iannelli, D. Genetics of Host Protection against Helicobacter pylori Infections. Int. J. Mol. Sci. 2021, 22, 3192. https://doi.org/10.3390/ijms22063192
Capparelli R, Iannelli D. Genetics of Host Protection against Helicobacter pylori Infections. International Journal of Molecular Sciences. 2021; 22(6):3192. https://doi.org/10.3390/ijms22063192
Chicago/Turabian StyleCapparelli, Rosanna, and Domenico Iannelli. 2021. "Genetics of Host Protection against Helicobacter pylori Infections" International Journal of Molecular Sciences 22, no. 6: 3192. https://doi.org/10.3390/ijms22063192
APA StyleCapparelli, R., & Iannelli, D. (2021). Genetics of Host Protection against Helicobacter pylori Infections. International Journal of Molecular Sciences, 22(6), 3192. https://doi.org/10.3390/ijms22063192





