Vitamin D and Cardiovascular Disease: An Updated Narrative Review
Abstract
1. Introduction
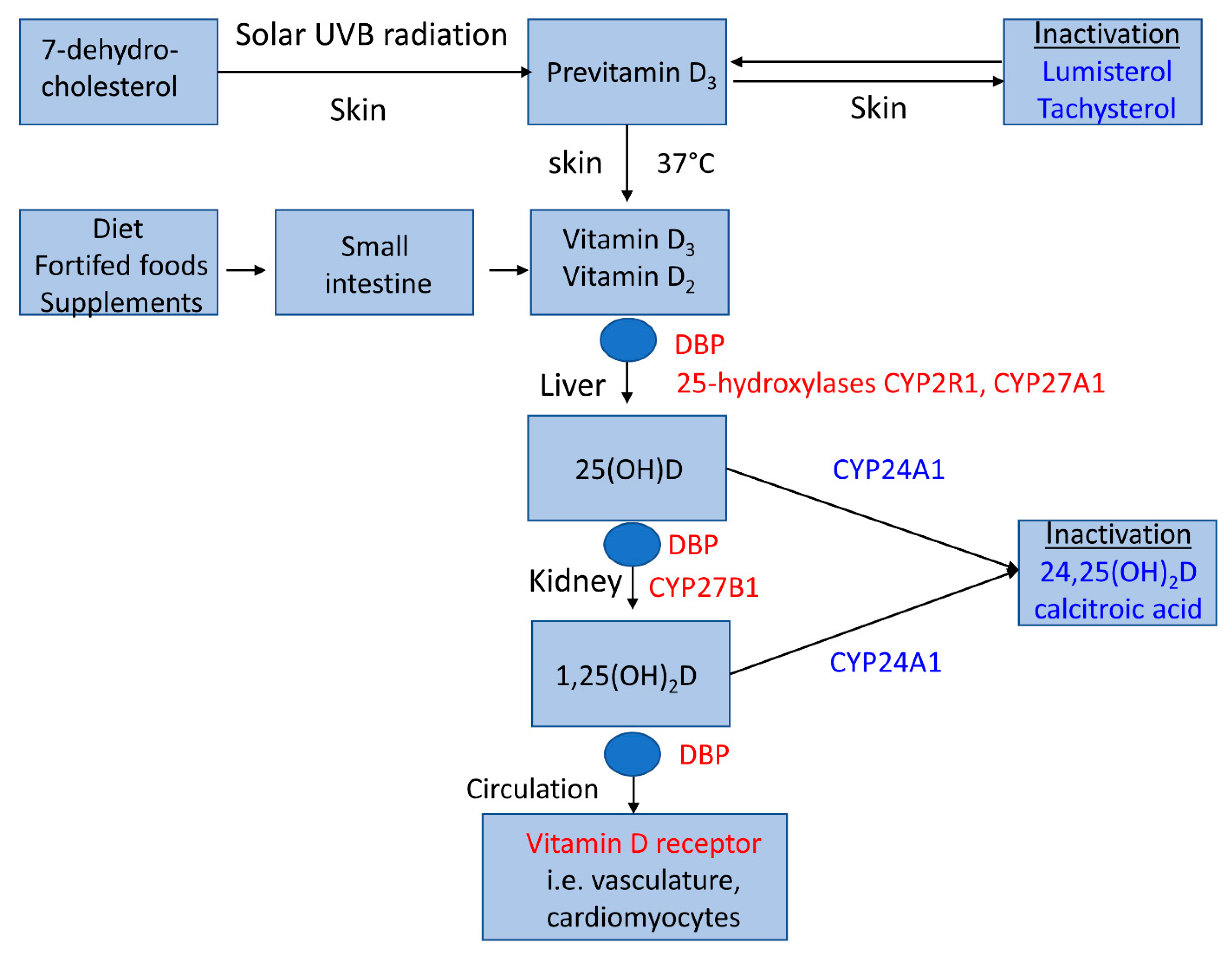
2. Vitamin D Metabolism
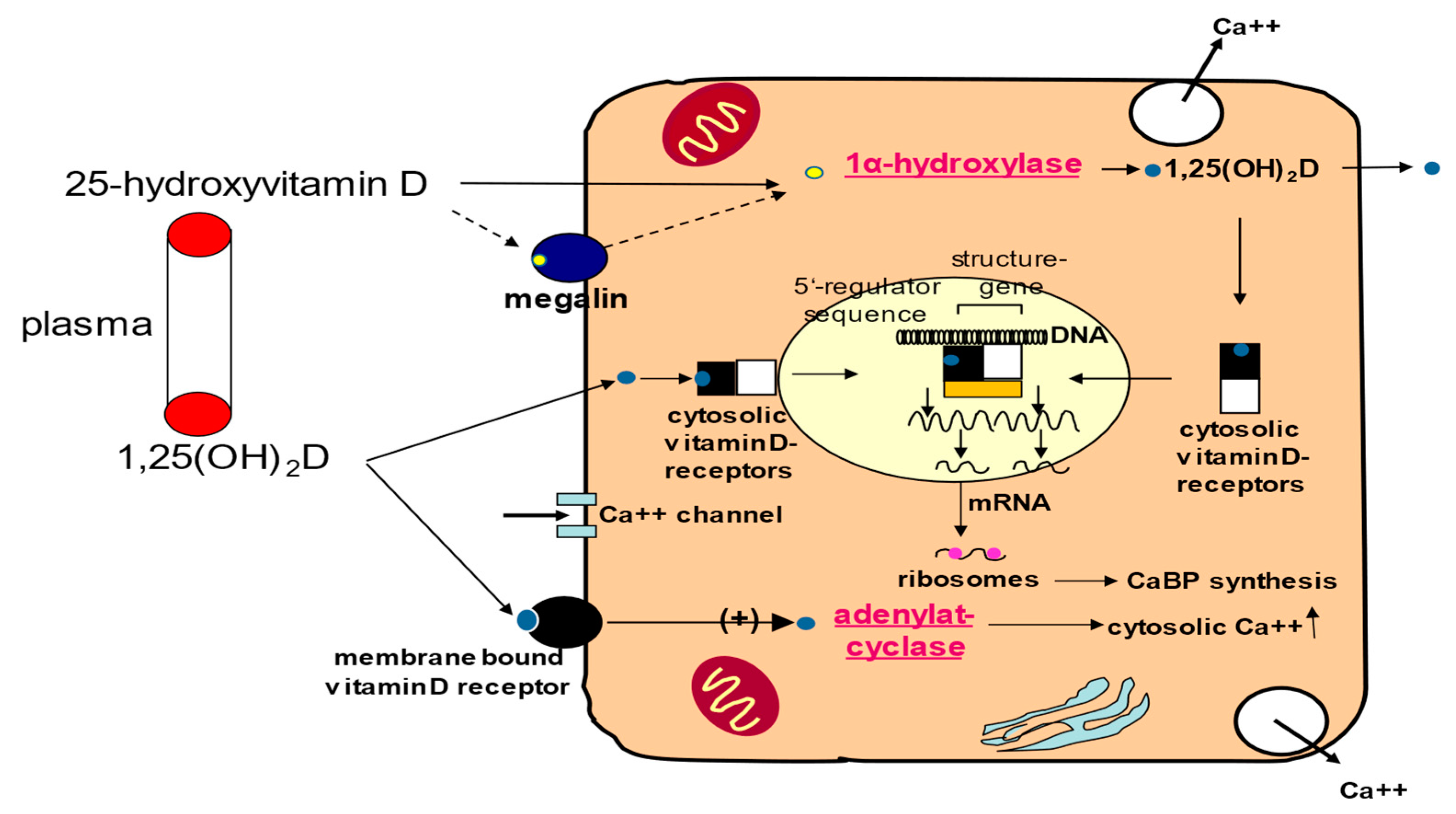
3. Vitamin D Effects on The Cardiovascular System
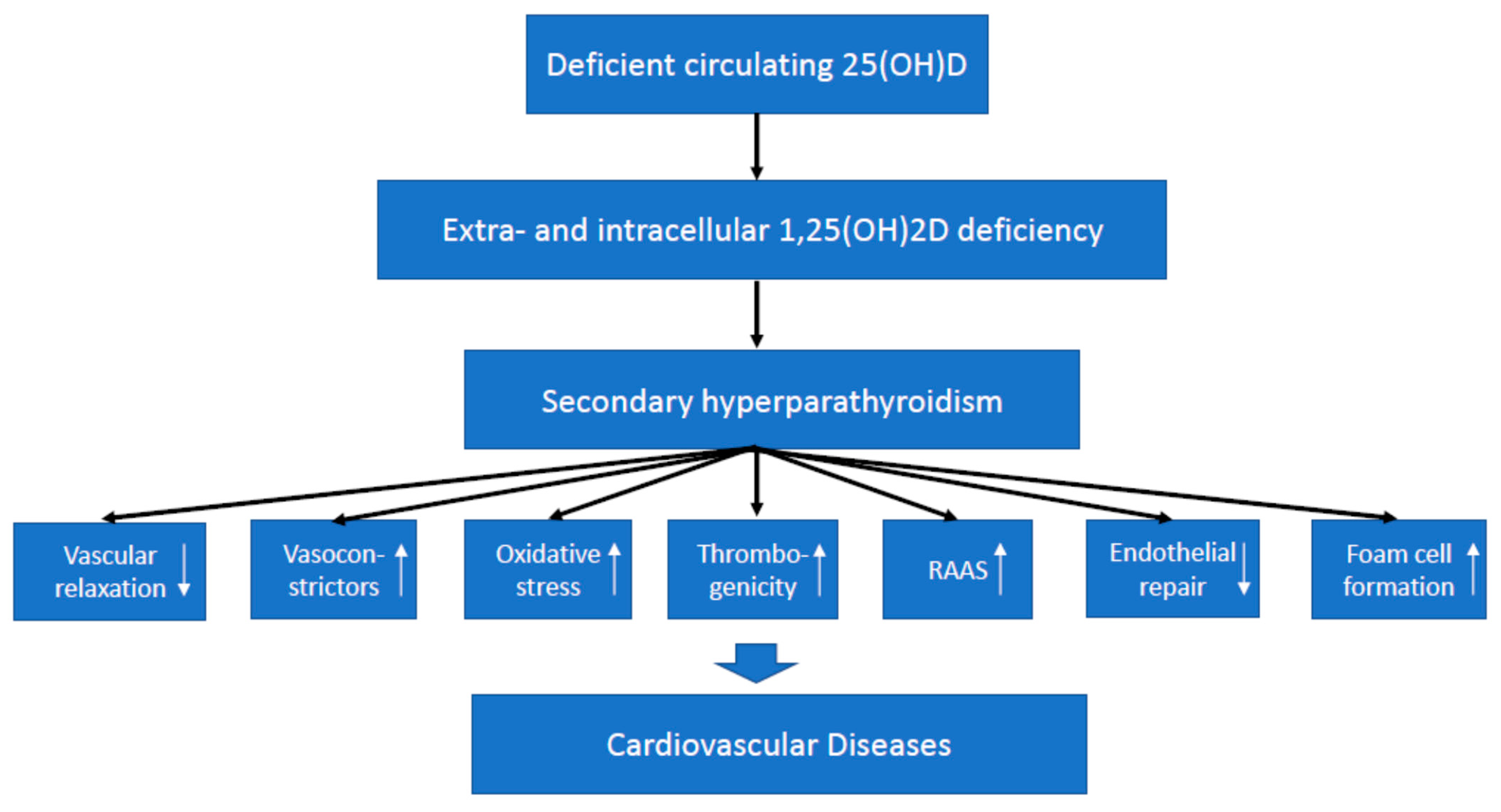
4. Preclinical Data on Vitamin D and Cardiovascular Diseases
5. Data on Vitamin D and Cardiovascular Diseases in Humans
5.1. Cardiovascular Disease in Severe Vitamin D Deficiency Such as Rickets and Osteomalcia
5.2. Cardiovascular Events in General Populations According to Vitamin D Status
5.3. Cardiovascular Disease According to Genetic Analyses of Vitamin D Metabolism
5.4. Cardiovascular Surrogate Parameters in Vitamin D Supplemenation Trials
5.4.1. Vascular Parameters
5.4.2. Cardiac Parameters
5.4.3. Lipid Parameters
6. Cardiovascular Events in Large Vitamin D Supplementation Trials
7. Cardiovascular Events in Patients with Chronic Kidney Disease (CKD)
8. Discussion of the Available Evidence
9. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Jahreis, G.; Hesse, V. Vitamin D-induced tissue calcinosis and arteriosclerosis changes. I: A contribution to the 60-year history of vitamin D research with special reference to childhood. Padiatr. Grenzgeb. 1990, 29, 203–211. (In German) [Google Scholar] [PubMed]
- Autier, P.; Boniol, M.; Pizot, C.; Mullie, P. Vitamin D status and ill health: A systematic review. Lancet Diabetes Endocrinol. 2014, 2, 76–89. [Google Scholar] [CrossRef]
- Roth, G.A.; Johnson, C.; Abajobir, A.; Abd-Allah, F.; Abera, S.F.; Abyu, G.; Ahmed, M.; Aksut, B.; Alam, T.; Alam, K.; et al. Global, Regional, and National Burden of Cardiovascular Diseases for 10 Causes, 1990 to 2015. J. Am. Coll. Cardiol. 2017, 70, 1–25. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; März, W.; Cashman, K.D.; Kiely, M.E.; Whiting, S.J.; Holick, M.F.; Grant, W.B.; Pludowski, P.; Hiligsmann, M.; Trummer, C.; et al. Rationale and Plan for Vitamin D Food Fortification: A Review and Guidance Paper. Front. Endocrinol. 2018, 9, 373. [Google Scholar] [CrossRef] [PubMed]
- Holick, M.F. Sunlight, UV Radiation, Vitamin D, and Skin Cancer: How Much Sunlight Do We Need? Adv. Exp. Med. Biol. 2020, 1268, 19–36. [Google Scholar] [PubMed]
- Mawer, E.B.; Backhouse, J.; Holman, C.A.; Lumb, G.A.; Stanbury, S.W. The distribution and storage of vitamin D and its metabolites in human tissues. Clin. Sci. 1972, 43, 413–431. [Google Scholar] [CrossRef]
- Levine, B.S.; Singer, F.R.; Bryce, G.F.; Mallon, J.P.; Miller, O.N.; Coburn, J.W. Pharmacokinetics and biologic effects of calcitriol in normal humans. J. Lab. Clin. Med. 1985, 105, 239–246. [Google Scholar] [PubMed]
- Bacchetta, J.; Salusky, I.B.; Hewison, M. Beyond mineral metabolism, is there an interplay between FGF23 and vitamin D in innate immunity? Pediatr. Nephrol. 2013, 28, 577–582. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Ernst, J.B. Calciotropic and phosphaturic hormones in heart failure. Nutr. Metab. Cardiovasc. Dis. 2016, 26, 971–979. [Google Scholar] [CrossRef] [PubMed]
- Tuckey, R.C.; Cheng, C.Y.S.; Slominski, A.T. The serum vitamin D metabolome: What we know and what is still to discover. J. Setroid. Biochem. Mol. Biol. 2019, 186, 4–21. [Google Scholar] [CrossRef] [PubMed]
- Saponaro, F.; Saba, A.; Zucchi, R. An Update on Vitamin D Metabolism. Int. J. Mol. Sci. 2020, 21, 6573. [Google Scholar] [CrossRef] [PubMed]
- Martinaityte, I.; Kamycheva, E.; Didriksen, A.; Jakobsen, J.; Jorde, R. Vitamin D Stored in Fat Tissue During a 5-Year Intervention Affects Serum 25-Hydroxyvitamin D Levels the Following Year. J. Clin. Endocrinol. Metab. 2017, 102, 3731–3738. [Google Scholar] [CrossRef] [PubMed]
- Jorde, R.; Grimnes, G. Serum cholecalciferol may be a better marker of vitamin D status than 25-hydroxyvitamin D. Med. Hypotheses 2018, 111, 61–65. [Google Scholar] [CrossRef] [PubMed]
- Latic, N.; Erben, R.G. Vitamin D and Cardiovascular Disease, with Emphasis on Hypertension, atherosclerosis, and Heart Failure. Int. J. Mol. Sci. 2020, 21, 6483. [Google Scholar] [CrossRef]
- Institute of Medicine. Dietary Reference Intakes: Calcium and Vitamin D; National Academies Press: Washington, DC, USA, 2011. [Google Scholar]
- EFSA NDA Panel (EFSA Panel on Dietetic Products, Nutrition and Allergies). Scientific opinion on dietary reference values for vitamin D. EFSA J. 2016, 14, 4547. [Google Scholar]
- DGE (German Nutrition Society), Österreichische Gesellschaft für Ernährung, Schweizerische Gesellschaft für Ernährungsforschung, Schweizerische Vereinigung für Ernährung. D-A-CH Referenzwerte für die Nährstoffzufuhr, 1st ed.; 5. korrigierter Nachdruck 2013; Neuer Umschau Buchverlag: Neustadt an der Weinstraße, Germany, 2012. [Google Scholar]
- Nordic Council of Ministers. Nordic Nutrition Recommendations 2012, 5th ed.; Narayana Press: Odder, Denmark, 2014. [Google Scholar]
- Holick, M.F.; Binkley, N.C.; Bischoff-Ferrari, H.A.; Gordon, C.M.; Hanley, D.A.; Heaney, R.P.; Murad, M.H.; Weaver, C.M. Endocrine Society: Evaluation, treatment, and prevention of vitamin D deficiency: An Endocrine Society clinical practice guideline. J. Clin. Endocrinol. Metab. 2011, 96, 1911–1930. [Google Scholar] [CrossRef] [PubMed]
- Pilz, S.; Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; Keppel, M.H.; Grübler, M.R.; März, W.; Pandis, M. Vitamin D testing and treatment: A narrative review of current evidence. Endocr. Connect. 2019, 8, R27–R43. [Google Scholar] [CrossRef] [PubMed]
- Van Schoor, N.; Lips, P. Global Overview of Vitamin D Status. Endocrinol. Metab. Clin. North Am. 2017, 46, 845–870. [Google Scholar] [CrossRef]
- Cashman, K.D.; Dowling, K.G.; Škrabáková, Z.; Gonzalez-Gross, M.; Valtueña, J.; De Henauw, S.; Moreno, L.; Damsgaard, C.T.; Michaelsen, K.F.; Mølgaard, C.; et al. Vitamin D deficiency in Europe: Pandemic? Am. J. Clin. Nutr. 2016, 103, 1033–1044. [Google Scholar] [CrossRef] [PubMed]
- Cashman, K.D.; Sheehy, T.; O’Neill, C.M. Is vitamin D deficiency a public health concern for low middle income countries? A systematic literature review. Eur. J. Nutr. 2019, 58, 433–453. [Google Scholar] [CrossRef] [PubMed]
- Norman, P.E.; Powell, J.T. Vitamin D and cardiovascular disease. Circ. Res. 2014, 114, 379–393. [Google Scholar] [CrossRef]
- Pilz, S.; Verheyen, N.; Grübler, M.R.; Tomaschitz, A.; März, W. Vitamin D and cardiovascular disease prevention. Nat. Rev. Cardiol. 2016, 13, 404–417. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Glenn, D.J.; Ni, W.; Grigsby, C.L.; Olsen, K.; Nishimoto, M.; Law, C.S.; Gardner, D.G. Expression of the vitamin D receptor is increased in the hypertrophic heart. Hypertension 2008, 52, 1106–1112. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Schleithoff, S.S.; Tenderich, G.; Berthold, H.K.; Körfer, R.; Stehle, P. Low vitamin D status: A contributing factor in the pathogenesis of congestive heart failure? J. Am. Coll. Cardiol. 2003, 41, 105–112. [Google Scholar] [CrossRef]
- Chanakul, A.; Zhang, M.Y.H.; Louw, A.; Armbrecht, H.J.; Miller, W.L.; Portale, A.A.; Perwad, F. FGF-23 regulates CYP27B1 transcription in the kidney and in extra-renal tissues. PLoS ONE 2013, 8, e72816. [Google Scholar] [CrossRef] [PubMed]
- Somjen, D.; Weisman, Y.; Kohen, F.; Gayer, B.; Limor, R.; Sharon, O.; Jaccard, N.; Knoll, E.; Stern, N. 25-Hydroxyvitamin D3-1-Hydroxylase Is Expressed in Human Vascular Smooth Muscle Cells and Is Upregulated by Parathyroid Hormone and Estrogenic Compounds. Circulation 2005, 111, 1666–1671. [Google Scholar] [CrossRef] [PubMed]
- Scialla, J.J.; Wolf, M. Roles of phosphate and fibroblast growth factor 23 in cardiovascular disease. Nat. Rev. Nephrol. 2014, 10, 268–278. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Kong, J.; Wei, M.; Chen, Z.F.; Liu, S.Q.; Cao, L.P. 1,25-Dihydroxyvitamin D(3) is a negative endocrine regulator of the renin-angiotensin system. J. Clin. Investig. 2002, 110, 229–238. [Google Scholar] [CrossRef]
- Xiang, W.; Kong, J.; Chen, S.; Cao, L.P.; Qiao, G.; Zheng, W.; Liu, W.; Li, X.; Gardner, D.G.; Li, J.C. Cardiac hypertrophy in vitamin D receptor knockout mice: Role of the systemic and cardiac renin-angiotensin systems. Am. J. Physiol. Endocrinol. Metab. 2005, 288, E125–E132. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.; Law, C.S.; Grigsby, C.L.; Olsen, K.; Hong, T.T.; Zhang, Y.; Yeghiazarians, Y.; Gardner, D.G. Cardiomyocyte-specific deletion of the vitamin D receptor gene results in cardiac hypertrophy. Circulation 2011, 124, 1838–1847. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.C.; Qiao, G.; Uskokovic, M.; Xiang, W.; Zheng, W.; Kong, J. Vitamin D: A negative endocrine regulator of the renin-angiotensin system and blood pressure. J. Steroid. Biochem. Mol. Biol. 2004, 89–90, 387–392. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.; Brandsch, C.; Kühne, H.; Thiele, A.; Hirche, F.; Stangl, G.I. Vitamin D receptor deficiency and low vitamin D diet stimulate aortic calcification and osteogenic key factor expression in mice. PLoS ONE 2012, 7, e35316. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, N.; Brandsch, C.; Schutkowski, A.; Hirche, F.; Stangl, G.I. Dietary vitamin D inadequacy accelerates calcification and osteoblast-like cell formation in the vascular system of LDL receptor knockout and wild-type mice. J. Nutr. 2014, 144, 638–646. [Google Scholar] [CrossRef] [PubMed]
- Aihara, K.; Azuma, H.; Akaike, M.; Ikeda, Y.; Yamashita, M.; Sudo, T.; Hayashi, H.; Yamada, Y.; Endoh, F.; Fujimura, M.; et al. Disruption of nuclear vitamin D receptor gene causes enhanced thrombogenicity in mice. J. Biol. Chem. 2004, 279, 35798–35802. [Google Scholar] [CrossRef] [PubMed]
- Zhou, C.; Lu, F.; Cao, K.; Xu, D.; Goltzman, D.; Miao, D. Calcium-independent and 1,25(OH)2D3-dependent regulation of the renin-angiotensin system in 1α-hydroxylase knockout mice. Kidney Int. 2008, 74, 170–179. [Google Scholar] [CrossRef] [PubMed]
- Elidrissy, A.T.; Munawarah, M.; Alharbi, K.M. Hypocalcemic rachitic cardiomyopathy in infants. J. Saudi Heart Assoc. 2013, 25, 25–33. [Google Scholar] [CrossRef]
- Shedeed, S.A. Vitamin D supplementation in infants with chronic congestive heart failure. Pediatr. Cardiol. 2012, 33, 713–719. [Google Scholar] [CrossRef] [PubMed]
- Acar, S.; Demir, K.; Shi, Y. Genetic Causes of Rickets. J. Clin. Res. Pediatr. Endocrinol. 2017, 9 (Suppl. S2), 88–105. [Google Scholar] [CrossRef] [PubMed]
- Avery, P.G.; Arnold, I.R.; Hubner, P.J.; Iqbal, S.J. Cardiac failure secondary to hypocalcaemia of nutritional osteomalacia. Eur. Heart J. 1992, 13, 426–427. [Google Scholar] [CrossRef]
- Jan de Beur, S.M. Tumor-induced osteomalacia. JAMA 2005, 294, 1260–1267. [Google Scholar] [CrossRef] [PubMed]
- Nasu, T.; Kurisu, S.; Matsuno, S.; Tatsumi, K.; Kakimoto, T.; Kobayashi, M.; Nakano, Y.; Wakasaki, H.; Furuta, H.; Nishi, M.; et al. Tumor-induced hypophosphatemic osteomalacia diagnosed by the combinatory procedures of magnetic resonance imaging and venous sampling for FGF23. Intern. Med. 2008, 47, 957–961. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, Y.; Suzuki, H.; Ogura, S.; Imai, R.; Yamazaki, Y.; Yamashita, T.; Miyamoto, Y.; Okazaki, H.; Nakamura, K.; Nakahara, K.; et al. Venous sampling for fibroblast growth factor-23 confirms preoperative diagnosis of tumor-induced osteomalacia. J. Clin. Endocrinol. Metab. 2004, 89, 3979–3982. [Google Scholar] [CrossRef] [PubMed]
- Zhang, R.; Li, B.; Gao, X.; Tian, R.; Pan, Y.; Jiang, Y.; Gu, H.; Wang, Y.; Wang, Y.; Liu, G. Serum 25-hydroxyvitamin D and the risk of cardiovascular disease: Dose-response meta-analysis of prospective studies. Am. J. Clin. Nutr. 2017, 105, 810–819. [Google Scholar] [CrossRef]
- Wang, L.; Song, Y.; Manson, J.E.; Pilz, S.; März, W.; Michaëlsson, K.; Lundqvist, A.; Jassal, S.K.; Barrett-Connor, E.; Zhang, C.; et al. Circulating 25-hydroxy-vitamin D and risk of cardiovascular disease: A meta-analysis of prospective studies. Circ. Cardiovasc. Qual. Outcomes. 2012, 5, 819–829. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Ou-Yang, J.; Huang, J. Low serum vitamin D levels increase the mortality of cardiovascular disease in older adults: A dose-response meta-analysis of prospective studies. Medicine (Baltimore) 2019, 98, e16733. [Google Scholar] [CrossRef]
- Zhang, D.; Cheng, C.; Wang, Y.; Sun, H.; Yu, S.; Xue, Y.; Liu, Y.; Li, W.; Li, X. Effect of Vitamin D on Blood Pressure and Hypertension in the General Population: An Update Meta-Analysis of Cohort Studies and Randomized Controlled Trials. Prev. Chronic. Dis. 2020, 17, E03. [Google Scholar] [CrossRef] [PubMed]
- Brøndum-Jacobsen, P.; Nordestgaard, B.G.; Schnohr, P.; Benn, M. 25-hydroxyvitamin D and symptomatic ischemic stroke: An original study and meta-analysis. Ann. Neurol. 2013, 73, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Schöttker, B.; Jorde, R.; Peasey, A.; Thorand, B.; Jansen, E.H.; de Groot, L.; Streppel, M.; Gardiner, J.; Ordóñez-Mena, J.M.; Perna, L.; et al. Vitamin D and mortality: Meta-analysis of individual participant data from a large consortium of cohort studies from Europe and the United States. BMJ 2014, 348, g3656. [Google Scholar] [CrossRef] [PubMed]
- Chowdhury, R.; Kunutsor, S.; Vitezova, A.; Oliver-Williams, C.; Chowdhury, S.; Kiefte-de-Jong, J.C.; Khan, H.; Baena, C.P.; Prabhakaran, D.; Hoshen, M.B.; et al. Vitamin D and risk of cause specific death: Systematic review and meta-analysis of observational cohort and randomised intervention studies. BMJ 2014, 348, g1903. [Google Scholar] [CrossRef] [PubMed]
- Qi, D.; Nie, X.L.; Wu, S.; Cai, J. Vitamin D and hypertension: Prospective study and meta-analysis. PLoS ONE 2017, 12, e0174298. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Darssan, D.; Pascoe, E.M.; Johnson, D.W.; Pi, H.; Dong, J. Vitamin D status and mortality risk among patients on dialysis: A systematic review and meta-analysis of observational studies. Nephrol. Dial. Transplant. 2018, 33, 1742–1751. [Google Scholar] [CrossRef] [PubMed]
- Gholami, F.; Moradi, G.; Zareei, B.; Rasouli, M.A.; Nikkhoo, B.; Roshani, D.; Ghaderi, E. The association between circulating 25-hydroxyvitamin D and cardiovascular diseases: A meta-analysis of prospective cohort studies. BMC Cardiovasc. Disord. 2019, 19, 248. [Google Scholar] [CrossRef]
- Bouillon, R. Vitamin D and cardiovascular disorders. Osteoporos. Int. 2019, 30, 2167–2181. [Google Scholar] [CrossRef] [PubMed]
- Brøndum-Jacobsen, P.; Benn, M.; Afzal, S.; Nordestgaard, B.G. No evidence that genetically reduced 25-hydroxyvitamin D is associated with increased risk of ischaemic heart disease or myocardial infarction: A Mendelian randomization study. Int. J. Epidemiol. 2015, 44, 651–661. [Google Scholar] [CrossRef] [PubMed]
- Afzal, S.; Brøndum-Jacobsen, P.; Bojesen, S.E.; Nordestgaard, B.G. Genetically low vitamin D concentrations and increased mortality: Mendelian randomisation analysis in three large cohorts. BMJ 2014, 349, g6330. [Google Scholar] [CrossRef]
- Manousaki, D.; Mokry, L.E.; Ross, S.; Goltzman, D.; Richards, J.B. Mendelian randomization studies Do not support a role for vitamin D in coronary artery disease. Circ. Cardiovasc. Genet. 2016, 9, 349–356. [Google Scholar] [CrossRef] [PubMed]
- Leong, A.; Rehman, W.; Dastani, Z.; Greenwood, C.; Timpson, N.; Langsetmo, L.; Berger, C.; METASTROKE; Fu, L.; Wong, B.Y.L.; et al. The causal effect of vitamin D binding protein (DBP) levels on calcemic and cardiometabolic diseases: A Mendelian randomization study. PLoS Med. 2014, 11, e1001751. [Google Scholar]
- Vimaleswaran, K.S.; Cavadino, A.; Berry, D.J.; LifeLines Cohort Study investigators; Jorde, R.; Dieffenbach, A.K.; Lu, C.; Alves, A.C.; Heerspink, H.J.; Tikkanen, E.; et al. Association of vitamin D status with arterial blood pressure and hypertension risk: A Mendelian randomisation study. Lancet Diabetes Endocrinol. 2014, 2, 719–729. [Google Scholar] [CrossRef]
- Davies, N.M.; Holmes, M.; Davey Smith, G. Reading Mendelian randomization studies: A guide, glossary, and checklist for clinicians. BMJ 2018, 362, k601. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Liu, L.; Chen, X.; He, W.; Yu, X. Associations study of vitamin D receptor gene polymorphisms with diabetic microvascular complications: A meta-analysis. Gene 2014, 546, 6–10. [Google Scholar] [CrossRef] [PubMed]
- Lu, S.; Guo, S.; Hu, F.; Guo, Y.; Yan, L.; Ma, W.; Wang, Y.; Wei, Y.; Zhang, Z.; Wang, Z. The Associations Between the Polymorphisms of Vitamin D Receptor and Coronary Artery Disease: A Systematic Review and Meta-Analysis. Medicine (Baltimore) 2016, 95, e3467. [Google Scholar] [CrossRef] [PubMed]
- Song, N.; Yang, S.; Wang, Y.Y.; Tang, S.Q.; Zhu, Y.Q.; Dai, Q.; Zhang, H. The Impact of Vitamin D Receptor Gene Polymorphisms on the Susceptibility of Diabetic Vascular Complications: A Meta-Analysis. Genet. Test Mol. Biomark. 2019, 23, 533–556. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Y.B.; Li, Z.Q.; Ding, N.; Yi, H.L. The association between vitamin D receptor gene polymorphism and susceptibility to hypertension: A meta-analysis. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 9066–9074. [Google Scholar] [PubMed]
- Alizadeh, S.; Djafarian, K.; Alizadeh, H.; Mohseni, R.; Shab-Bidar, S. Common variants of Vitamin D receptor gene polymorphisms and susceptibility to coronary artery disease: A systematic review and meta-analysis. J. Nutr. Nutr. 2017, 10, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Shen, H.; Bielak, L.F.; Ferguson, J.F.; Streeten, E.A.; Yerges-Armstrong, L.M.; Liu, J.; Post, W.; O’Connell, J.R.; Hixson, J.E.; Kardia, S.L.; et al. Association of the vitamin D metabolism gene CYP24A1 with coronary artery calcification. Arterioscler. Thromb. Vasc. Biol. 2010, 30, 2648–2654. [Google Scholar] [CrossRef]
- Stojanović, M.; Radenković, M. Vitamin D versus placebo in improvement of endothelial dysfunction: A meta-analysis of randomized clinical trials. Cardiovasc. Ther. 2015, 33, 145–154. [Google Scholar] [CrossRef]
- Rodríguez, A.J.; Scott, D.; Srikanth, V.; Ebeling, P. Effect of vitamin D supplementation on measures of arterial stiffness: A systematic review and meta-analysis of randomized controlled trials. Clin. Endocrinol. 2016, 84, 645–657. [Google Scholar] [CrossRef] [PubMed]
- Mirhosseini, N.; Rainsbury, J.; Kimball, S.M. Vitamin D Supplementation, Serum 25(OH)D Concentrations and Cardiovascular Disease Risk Factors: A Systematic Review and Meta-Analysis. Front. Cardiovasc. Med. 2018, 5, 87. [Google Scholar] [CrossRef] [PubMed]
- Dou, D.; Yang, B.; Gan, H.; Xie, D.; Lei, H.; Ye, N. Vitamin D supplementation for the improvement of vascular function in patients with chronic kidney disease: A meta-analysis of randomized controlled trials. Int. Urol. Nephrol. 2019, 51, 851–858. [Google Scholar] [CrossRef]
- Beveridge, L.A.; Khan, F.; Struthers, A.D.; Armitage, J.; Barchetta, I.; Bressendorff, I.; Cavallo, M.G.; Clarke, R.; Dalan, R.; Dreyer, G.; et al. Effect of Vitamin D Supplementation on Markers of Vascular Function: A Systematic Review and Individual Participant Meta-Analysis. J. Am. Heart Assoc. 2018, 7, e008273. [Google Scholar] [CrossRef] [PubMed]
- Pincombe, N.L.; Pearson, M.J.; Smart, N.A.; King, N.; Dieberg, G. Effect of vitamin D supplementation on endothelial function —An updated systematic review with meta-analysis and meta-regression. Nutr. Metab. Cardiovasc. Dis. 2019, 29, 1261–1272. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.C.; Hsu, C.Y.; Mao, P.C.; Dreyer, G.; Wu, F.Z.; Chen, C.L. The effects of correction of vitamin D deficiency on arterial stiffness: A systematic review and updated meta-analysis of randomized controlled trials. J. Steroid. Biochem. Mol. Biol. 2020, 198, 105561. [Google Scholar] [CrossRef] [PubMed]
- Jiang, W.L.; Gu, H.B.; Zhang, Y.F.; Xia, Q.Q.; Qi, J.; Chen, J.C. Vitamin D Supplementation in the Treatment of Chronic Heart Failure: A Meta-analysis of Randomized Controlled Trials. Clin. Cardiol. 2016, 39, 56–61. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.; Liu, Z.; Fu, J.; Min, Z. Meta-analysis of vitamin D supplementation in the treatment of chronic heart failure. Scand. Cardiovasc. J. 2019, 53, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Zhao, J.D.; Jia, J.J.; Dong, P.S.; Zhao, D.; Yang, X.M.; Li, D.L.; Zhang, H.F. Effect of vitamin D on ventricular remodelling in heart failure: A meta-analysis of randomised controlled trials. BMJ Open 2018, 8, e020545. [Google Scholar] [CrossRef] [PubMed]
- Lutsey, P.L.; Alonso, A.; Michos, E.D.; Loehr, L.R.; Astor, B.C.; Coresh, J.; Folsom, A.R. Serum magnesium, phosphorus, and calcium are associated with risk of incident heart failure: The Atherosclerosis Risk in Communities (ARIC) Study. Am. J. Clin. Nutr. 2014, 100, 756–764. [Google Scholar] [CrossRef]
- Zittermann, A.; Ernst, J.B.; Prokop, S.; Fuchs, U.; Gruszka, A.; Dreier, J.; Kuhn, J.; Knabbe, C.; Berthold, H.K.; Gouni-Berthold, I.; et al. Vitamin D supplementation of 4000 IU daily and cardiac function in patients with advanced heart failure: The EVITA trial. Int. J. Cardiol. 2019, 280, 117–123. [Google Scholar] [CrossRef]
- Pfeffer, M.A.; Shah, A.M.; Borlaug, B.A. Heart Failure with Preserved Ejection Fraction in Perspective. Circ. Res. 2019, 124, 1598–1617. [Google Scholar] [CrossRef] [PubMed]
- Dibaba, D.T. Effect of vitamin D supplementation on serum lipid profiles: A systematic review and meta-analysis. Nutr. Rev. 2019, 77, 890–902. [Google Scholar] [CrossRef] [PubMed]
- Greco, D.; Kocyigit, D.; Adorni, M.P.; Marchi, C.; Ronda, N.; Bernini, F.; Gurses, K.M.; Canpinar, H.; Guc, D.; Oguz, S.H.; et al. Vitamin D replacement ameliorates serum lipoprotein functions, adipokine profile and subclinical atherosclerosis in pre-menopausal women. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 822–829. [Google Scholar] [CrossRef]
- Scragg, R.; Stewart, A.W.; Waayer, D.; Lawes, C.M.M.; Toop, L.; Sluyter, J.; Murphy, J.; Khaw, K.T.; Camargo, C.A., Jr. Effect of Monthly High-Dose Vitamin D Supplementation on Cardiovascular Disease in the Vitamin D Assessment Study: A Randomized Clinical Trial. JAMA Cardiol. 2017, 2, 608–616. [Google Scholar] [CrossRef] [PubMed]
- Manson, J.E.; Cook, N.R.; Lee, I.M.; Christen, W.; Bassuk, S.S.; Mora, S.; Gibson, H.; Gordon, D.; Copeland, T.; D’Agostino, D.; et al. Vitamin D Supplements and Prevention of Cancer and Cardiovascular Disease. N. Engl. J. Med. 2019, 380, 33–44. [Google Scholar] [CrossRef] [PubMed]
- Bischoff-Ferrari, H.A.; Vellas, B.; Rizzoli, R.; Kressig, R.W.; da Silva, J.A.P.; Blauth, M.; Felson, D.T.; McCloskey, E.V.; Watzl, B.; Hofbauer, L.C.; et al. Effect of Vitamin D Supplementation, Omega-3 Fatty Acid Supplementation, or a Strength-Training Exercise Program on Clinical Outcomes in Older Adults: The DO-HEALTH Randomized Clinical Trial. JAMA 2020, 324, 1855–1868. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, L.A.; Struthers, A.D.; Khan, F.; Jorde, R.; Scragg, R.; Macdonald, H.M.; Alvarez, J.A.; Boxer, R.S.; Dalbeni, A.; Gepner, A.D.; et al. Effect of Vitamin D Supplementation on Blood Pressure: A Systematic Review and Meta-analysis Incorporating Individual Patient Data. JAMA Intern. Med. 2015, 175, 745–754. [Google Scholar] [CrossRef]
- Elamin, M.B.; Abu Elnour, N.O.; Elamin, K.B.; Fatourechi, M.M.; Alkatib, A.A.; Almandoz, J.P.; Liu, H.; Lane, M.A.; Mullan, R.J.; Hazem, A.; et al. Vitamin D and cardiovascular outcomes: A systematic review and meta-analysis. J. Clin. Endocrinol. Metab. 2011, 96, 1931–1942. [Google Scholar] [CrossRef]
- Bolland, M.J.; Grey, A.; Gamble, G.D.; Reid, I.R. The effect of vitamin D supplementation on skeletal, vascular, or cancer outcomes: A trial sequential meta-analysis. Lancet Diabetes Endocrinol. 2014, 2, 307–320. [Google Scholar] [CrossRef]
- Jenkins, D.J.A.; Spence, J.D.; Giovannucci, E.L.; Kim, Y.I.; Josse, R.; Vieth, R.; Blanco Mejia, S.; Viguiliouk, E.; Nishi, S.; Sahye-Pudaruth, S.; et al. Supplemental Vitamins and Minerals for CVD Prevention and Treatment. J. Am. Coll. Cardiol. 2018, 71, 2570–2584. [Google Scholar] [CrossRef] [PubMed]
- Barbarawi, M.; Kheiri, B.; Zayed, Y.; Barbarawi, O.; Dhillon, H.; Swaid, B.; Yelangi, A.; Sundus, S.; Bachuwa, G.; Alkotob, M.L.; et al. Vitamin D Supplementation and Cardiovascular Disease Risks in More Than 83 000 Individuals in 21 Randomized Clinical Trials: A Meta-analysis. JAMA Cardiol. 2019, 4, 765–776. [Google Scholar] [CrossRef] [PubMed]
- Rejnmark, L.; Bislev, L.S.; Cashman, K.D.; Eiríksdottir, G.; Gaksch, M.; Grübler, M.; Grimnes, G.; Gudnason, V.; Lips, P.; Pilz, S.; et al. Non-skeletal health effects of vitamin D supplementation: A systematic review on findings from meta-analyses summarizing trial data. PLoS ONE 2017, 12, e0180512. [Google Scholar] [CrossRef]
- Alfieri, C.; Ruzhytska, O.; Vettoretti, S.; Caldiroli, L.; Cozzolino, M.; Messa, P. Native Hypovitaminosis D in CKD Patients: From Experimental Evidence to Clinical Practice. Nutrients 2019, 11, 1918. [Google Scholar] [CrossRef]
- Zittermann, A.; Koerfer, R. Protective and toxic effects of vitamin D on vascular calcification: Clinical implications. Mol. Aspects Med. 2008, 29, 423–432. [Google Scholar] [CrossRef]
- Foley, R.N.; Parfrey, P.S.; Sarnaak, M.J. Clinical epidemiology of cardiovascular disease in chronic renal disease. Am. J. Kidney Dis. 1998, 32, S112–S119. [Google Scholar] [CrossRef] [PubMed]
- Zittermann, A.; Berthold, H.K.; Pilz, S. The effect of vitamin D on fibroblast growth factor 23: A systematic review and meta-analysis of randomized controlled trials. Eur. J. Clin. Nutr. 2020. [Google Scholar] [CrossRef] [PubMed]
- De Boer, I.H.; Zelnick, L.R.; Ruzinski, J.; Friedenberg, G.; Duszlak, J.; Bubes, V.Y.; Hoofnagle, A.N.; Thadhani, R.; Glynn, R.J.; Buring, J.E.; et al. Effect of vitamin D and omega-3 fatty acid supplementation on kidney function in patients with type 2 diabetes: A randomized clinical trial. JAMA 2019, 322, 1899–1909. [Google Scholar] [CrossRef]
- Strugnell, S.A.; Sprague, S.M.; Ashfaq, A.; Petkovich, M.; Bishop, C.W. Rationale for raising current clinical practice guideline target for serum 25-hydroxyvitamin D in chronic kidney disease. Am. J. Nephrol. 2019, 49, 284–293. [Google Scholar] [CrossRef]
- Lu, R.J.; Zhu, S.M.; Tang, F.L.; Zhu, X.S.; Fan, Z.D.; Wang, G.L.; Jiang, Y.F.; Zhang, Y. Effects of vitamin D or its analogues on the mortality of patients with chronic kidney disease: An updated systematic review and meta-analysis. Eur. J. Clin. Nutr. 2017, 71, 683–693. [Google Scholar] [CrossRef]
- Shoji, T.; Inaba, M.; Fukagawa, M.; Ando, R.; Emoto, M.; Fujii, H.; Fujimori, A.; Fukui, M.; Hase, H.; Hashimoto, T.; et al. Effect of oral alfacalcidol on clinical outcomes in patients without secondary hyperparathyroidism receiving maintenance hemodialysis: The J-DAVID Randomized Clinical Trial. JAMA 2018, 320, 2325–2334. [Google Scholar] [PubMed]
- Zittermann, A.; Ernst, J.B.; Prokop, S.; Fuchs, U.; Dreier, J.; Kuhn, J.; Knabbe, C.; Birschmann, I.; Schulz, U.; Berthold, H.K.; et al. Effect of vitamin D on all-cause mortality in heart failure (EVITA): A 3-year randomized clinical trial with 4000 IU vitamin D daily. Eur. Heart J. 2017, 38, 2279–2286. [Google Scholar] [CrossRef]
- Kärkkäinen, M.U.; Lamberg-Allardt, C.J.; Ahonen, S.; Välimäki, M. Does it make a difference how and when you take your calcium? The acute effects of calcium on calcium and bone metabolism. Am. J. Clin. Nutr. 2001, 74, 335–342. [Google Scholar] [CrossRef]
- Marshall, R.W.; Francis, R.M.; Hodgkinson, A. Plasma total and ionised calcium, albumin and globulin concentrations in pre- and post-menopausal women and the effects of oestrogen administration. Clin. Chim. Acta 1982, 122, 283–287. [Google Scholar] [CrossRef]
- Zorbas, Y.G.; Kakuris, K.K.; Deogenov, V.A.; Yerullis, K.B. Phosphate homeostasis in healthy subjects during prolonged periodic and continuous hypokinesia. Clin. Biochem. 2007, 40, 460–466. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Dawson-Hughes, B.; Sheehan, P.; Ware, J.H.; Knowler, W.C.; Aroda, V.R.; Brodsky, I.; Ceglia, L.; Chadha, C.; Chatterjee, R.; et al. Vitamin D Supplementation and Prevention of Type 2 Diabetes. N. Engl. J. Med. 2019, 381, 520–530. [Google Scholar] [CrossRef] [PubMed]
- Pittas, A.G.; Jorde, R.; Kawahara, T.; Dawson-Hughes, B. Vitamin D Supplementation for Prevention of Type 2 Diabetes Mellitus: To D or Not to D? J. Clin. Endocrinol. Metab. 2020, 105, 3721–3733. [Google Scholar] [CrossRef]
- Maretzke, F.; Bechthold, A.; Egert, S.; Ernst, J.B.; Melo van Lent, D.; Pilz, S.; Reichrath, J.; Stangl, G.I.; Stehle, P.; Volkert, D.; et al. Role of Vitamin D in Preventing and Treating Selected Extraskeletal Diseases-An Umbrella Review. Nutrients 2020, 12, 969. [Google Scholar] [CrossRef] [PubMed]
- Charoenngam, N.; Holick, M.F. Immunologic Effects of Vitamin D on Human Health and Disease. Nutrients 2020, 12, 2097. [Google Scholar] [CrossRef]
| Author, Year [Ref.] | Outcome | 25(OH)D Reference Category | 25(OH)D Category | Relative Risk (95%CI) |
|---|---|---|---|---|
| Wang, 2012 [47] | Cardiovascular disease | Highest category | Lowest category | 1.52 (1.30–1.77) |
| Brodum-Jacobsen, 2013 [50] | Ischemic stroke | Highest quartile | Lowest quartile | 1.54 (1.43–1.65) |
| Schöttker, 2014 [51] | CVD mortality without CVD history | Top quintile | Bottom quintile | 1.41 (1.18–1.68) |
| CVD mortality with CVD history | Top quintile | Bottom quintile | 1.65 (1.22–2.22) | |
| Chowdhury, 2014 [52] | CVD mortality | Top thirds | Bottom thirds | 1.14 (1.01–1.29) |
| Zhang, 2017 [46] | CVD events | 75 nmol/L | 50 nmol/L | 1.00 (0.95–1.05) |
| 25 nmol/L | 1.08 (1.04–1.15) | |||
| CVD mortality | 75 nmol/L | 50 nmol/L | 1.04 (0.95–1.61) | |
| 25 nmol/L | 1.15 (1.05–1.22) | |||
| Qi, 2017 [53] | Hypertension | >75 nmol/L | 74.99–50 nmol/L | 1.09 (1.05–1.14) |
| <50 nmol/L | 1.24 (1.08–1.41) | |||
| Zhang, 2018 [54] | CVD mortality | - | Per 25 nmol/L decrease | 1.41 (1.27–1.59) |
| Yang, 2019 [48] | CVD mortality | >75 nmol/L | 25–50 nmol/L | 1.16 (1.04–1.27) |
| <25 nmol/L | 1.47 (1.15–1.81) | |||
| Golami, 2019 [55] | CVD mortality | Highest category | Lowest category | 1.54 (1.29–1.84) |
| Zhang, 2020 [49] | Hypertension | 75 nmol/L | 50 nmol/L | 1.09 (1.04–1.17) |
| 25 nmol/L | 1.30 (1.11–1.52) |
| Author, Year [Ref.] | Studies | Participants | Vitamin D Dose Equivalent | Duration | WMD or SMD (95%CI) |
|---|---|---|---|---|---|
| Flow-mediated Dilation | |||||
| Stojanović, 2015 [69] | 8 | 529 | 1788 to 5000 IU/d; 2 µg paricalcitol/d | 8 to 16 weeks | 0.96% (−1.24% to 2.06%) |
| Dou, 2019 [72] | 5 | 354 | 5042 IU/d; 1–2 µg paricalcitol/d | 4 to 17 weeks | 1.66% (−0.20% to 3.51%) |
| Pincombe, 2019, [74] | 10 | 655 | 2000 to 7114 IU/d | 4 to 24 weeks | 1.17% (−0.20% to 2.54%) |
| Beveridge, 2018 [73] | 12 | 785 | 900 to 5000 IU/d; 1–2 µg paricalcitol/d | 4 to 52 weeks | 0.49% (−0.13% to 1.11%) |
| Pulse wave velocity | |||||
| Rodríguez, 2016 [70] | 10 | 827 | 1600 to 5700 IU/d | 4 to 52 weeks | −0.10 m/s (−0.24 m/s to 0.04 m/s) |
| Mirhosseini, 2018 [71] | 11 | 1019 | 600 to 4500 IU/d | 4 to 52 weeks | −0.20 m/s (−0.46 m/s to 0.06 m/s) |
| Dou, 2019 [72] | 3 | 235 | 2473 to 5042 IU/d; 2143 IU calcifediol plus 0.5 µg calcitriol/d | 17 to 26 weeks | −0.93 m/s (− 1.71 m/s to −0.15 m/s) |
| Pincombe, 2019, [74] | 21 | 2098 | 600 to 5714 IU/d; 2,143 IU calcifediol plus 0.5 µg calcitriol/d | 8 to 57 weeks | −0.09 m/s (−0.24 m/s to 0.07 m/s) |
| Chen, 2020 [75] | 10 | 918 | 1667 to 5000 IU/d | 8 to 52 weeks | −0.29 m/s (−0.51 m/s to −0.06 m/s) |
| Beveridge, 2018 [73] | 10 | 674 | 1000 to 5700 IU/d | 5 to 52 weeks | 0.04 m/s (−0.32 m/s to 0.41 m/s) |
| Augmentation Index | |||||
| Rodríguez, 2016 [70] | 8 | 497 | 1667 to 4000 IU/d | 4 to 52 weeks | −0.15% (−0.32% to 0.02%) |
| Mirhosseini, 2018 [71] | 10 | 965 | 1000 to 4000 IU/d | 5 to 52 weeks | −0.09% (−0.37% to 0.20%) |
| Pincombe, 2019, [74] | 16 | 5117 | 600 to 7114 IU/d | 4 to 52 weeks | 0.05% (−0.10% to 0.19%) |
| Beveridge, 2018 [73] | 14 | 1030 | 1000 to 5700 IU/d | 4 to 52 weeks | 0.00% (−1.30% to 1.30%) |
| LVEF | |||||
| Jiang, 2016 [76] | 4 | 303 | 1000 to 7114 IU/d | 12 to 39 weeks | 4.11% (−0.91% to 9.12%) |
| Wang, 2019 [77] | 5 | 422 | 3552 to 7114 IU/d | 26 to 52 weeks | 2.56% (−2.18% to 7.31%) |
| Zhao, 2018 [78] | 7 | 538 | 1000 to 7114 IU/d | 10 to 52 weeks | 4.18% (0.36% to 7.99%) |
| LVEDD | |||||
| Zhao, 2018 [78] | 6 | 499 | 1000 to 7114 IU/d | 12 to 52 weeks | −2.31 mm (−4.15 mm to −0.47 mm) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zittermann, A.; Trummer, C.; Theiler-Schwetz, V.; Lerchbaum, E.; März, W.; Pilz, S. Vitamin D and Cardiovascular Disease: An Updated Narrative Review. Int. J. Mol. Sci. 2021, 22, 2896. https://doi.org/10.3390/ijms22062896
Zittermann A, Trummer C, Theiler-Schwetz V, Lerchbaum E, März W, Pilz S. Vitamin D and Cardiovascular Disease: An Updated Narrative Review. International Journal of Molecular Sciences. 2021; 22(6):2896. https://doi.org/10.3390/ijms22062896
Chicago/Turabian StyleZittermann, Armin, Christian Trummer, Verena Theiler-Schwetz, Elisabeth Lerchbaum, Winfried März, and Stefan Pilz. 2021. "Vitamin D and Cardiovascular Disease: An Updated Narrative Review" International Journal of Molecular Sciences 22, no. 6: 2896. https://doi.org/10.3390/ijms22062896
APA StyleZittermann, A., Trummer, C., Theiler-Schwetz, V., Lerchbaum, E., März, W., & Pilz, S. (2021). Vitamin D and Cardiovascular Disease: An Updated Narrative Review. International Journal of Molecular Sciences, 22(6), 2896. https://doi.org/10.3390/ijms22062896







