Naturally Occurring Ecdysteroids in Triticum aestivum L. and Evaluation of Fenarimol as a Potential Inhibitor of Their Biosynthesis in Plants
Abstract
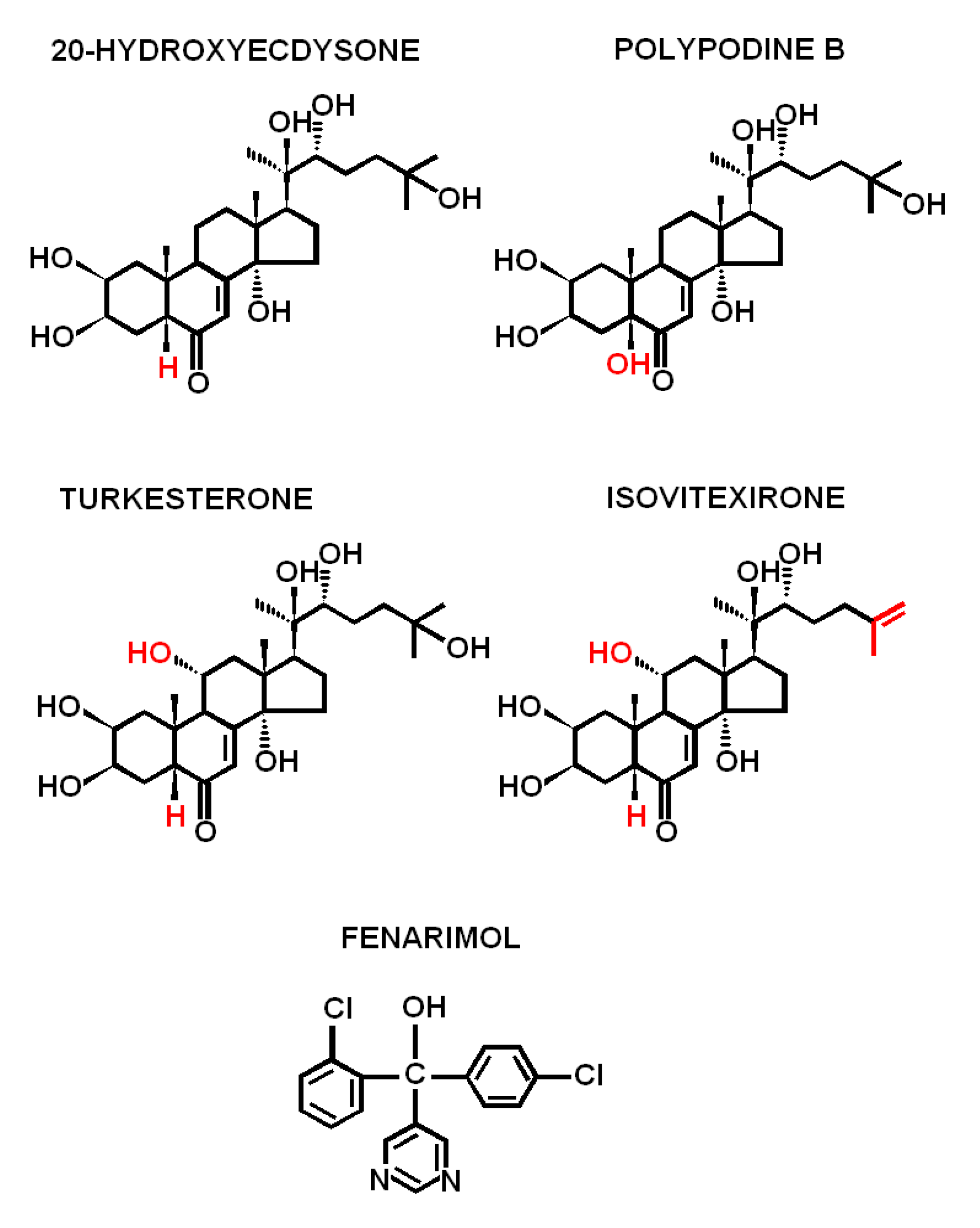
1. Introduction
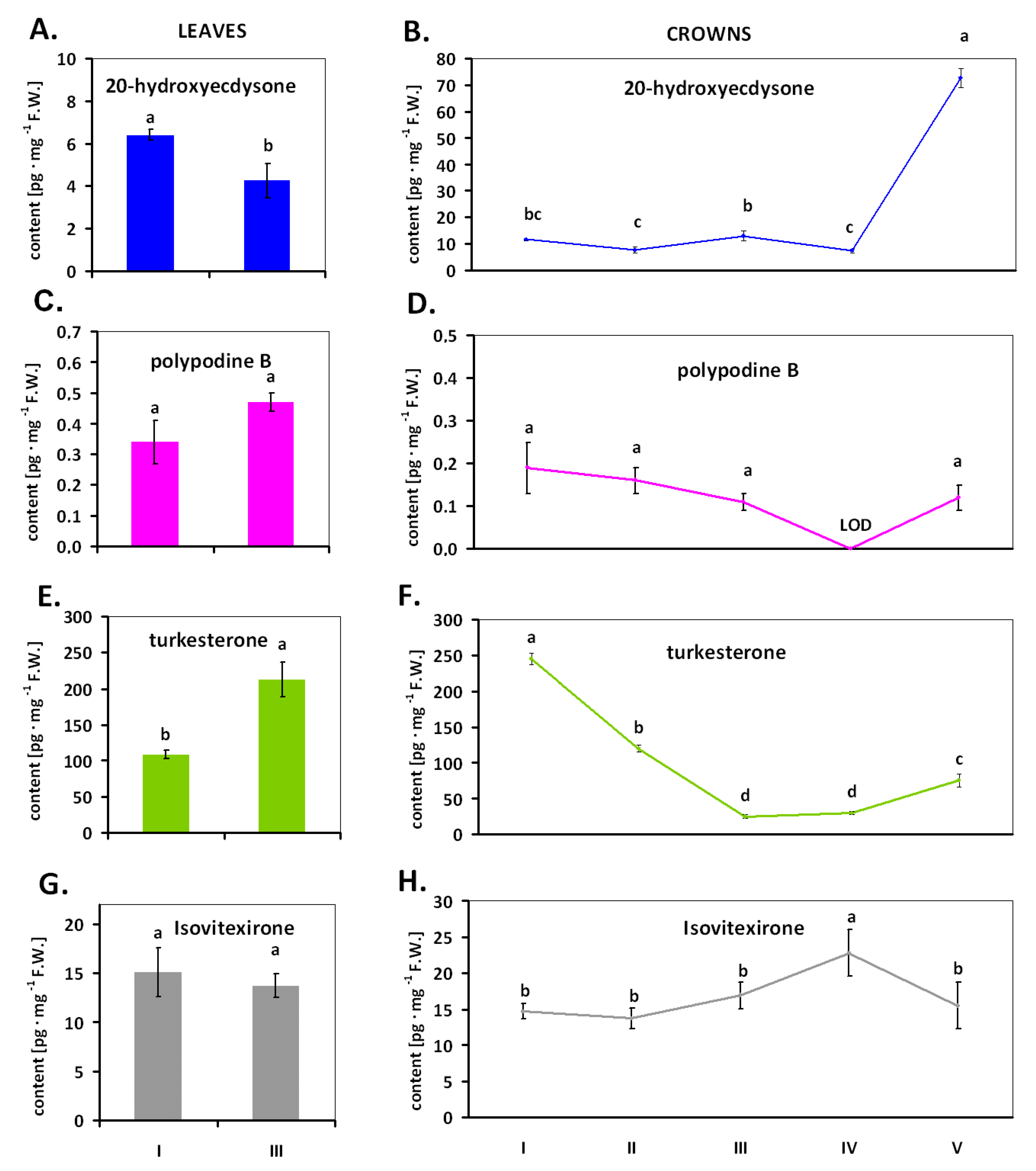
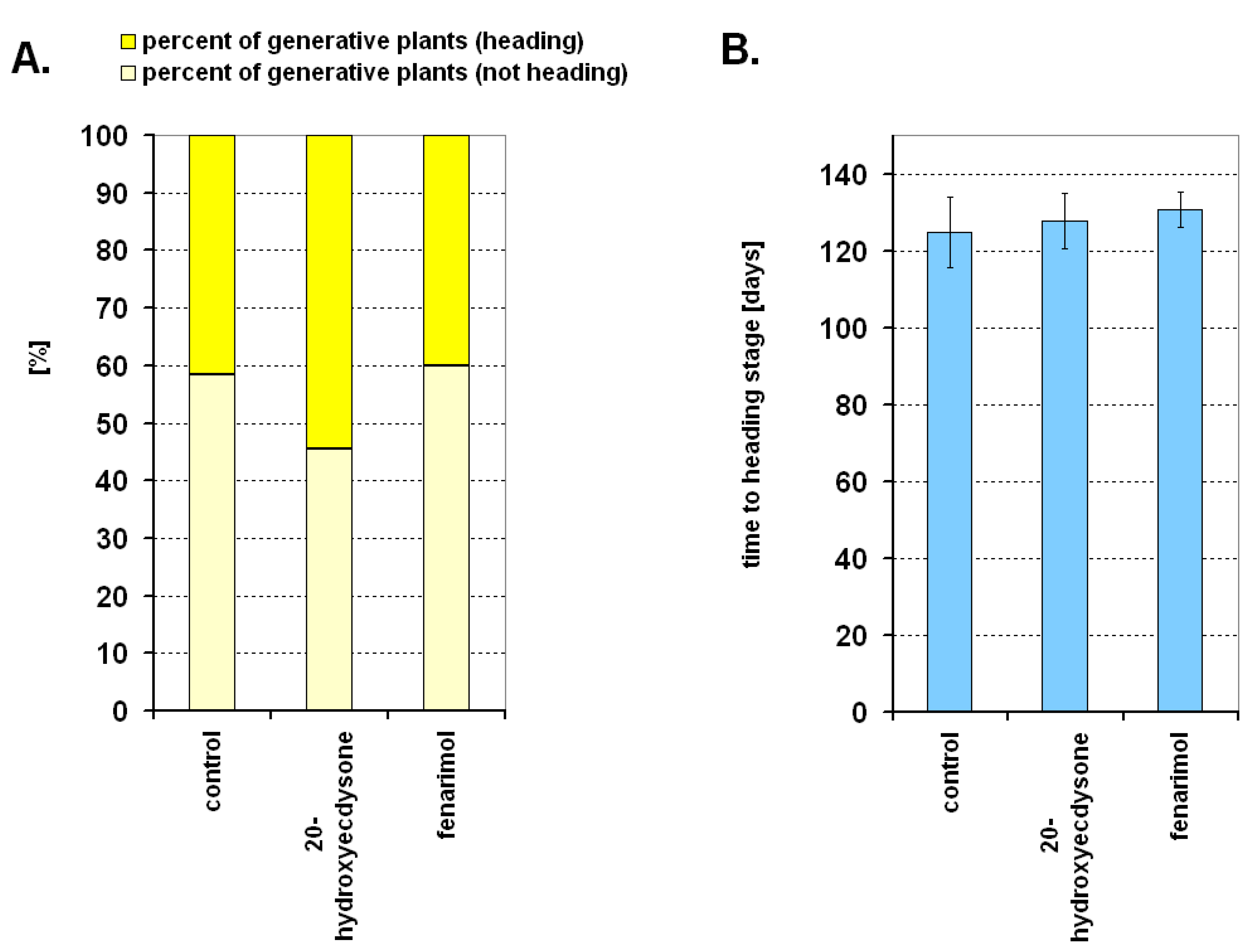
2. Results
3. Discussion
4. Material and Methods
4.1. Characteristic of Plant Material
4.2. Experimental Design and Sampling
4.2.1. Experiment 1. The Presence and Changes of ECs in Winter Wheat
4.2.2. Experiment 2. The Effect of Fenarimol on ECs Content in Wheat
4.2.3. Experiment 3. The Effect of 20-hydroxyecdysone and Fenarimol on Generative Development of Winter Wheat
4.3. Measurements and Observations
4.3.1. Extraction and Analysis of Ecdysteroids in Experiments 1 and 2
4.3.2. Evaluation of the Advancement of Wheat Development in Experiment 3
4.4. Statistical Approach
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Brown, M.R.; Sieglaff, D.H.; Rees, H.H. Gonadal Ecdysteroidogenesis in Arthropoda: Occurrence and Regulation. Annu. Rev. Entomol. 2009, 54, 105–125. [Google Scholar] [CrossRef]
- Lafont, R.; Koolman, J. Diversity of Ecdysteroids in Animal Species. In Ecdysone: Structures and Functions; Smagghe, G., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 47–71. [Google Scholar] [CrossRef]
- Spindler, K.-D.; Hönl, C.; Tremmel, C.; Braun, S.; Ruff, H.; Spindler-Barth, M. Ecdysteroid hormone action. Cell. Mol. Life Sci 2009, 66, 3837–3850. [Google Scholar] [CrossRef] [PubMed]
- Dinan, L. Phytoecdysteroids: Biological aspects. Phytochemistry 2001, 57, 325–339. [Google Scholar] [CrossRef]
- Dinan, L.; Savchenko, T.; Whiting, P. On the distribution of phytoecdysteroids in plants. CMLS Cell. Mol. Life Sci. 2001, 58, 1121–1132. [Google Scholar] [CrossRef] [PubMed]
- Hopkins, P.M. Crustacean Ecdysteroids and Their Receptors. In Ecdysone: Structures and Functions; Smagghe, G., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 73–97. [Google Scholar] [CrossRef]
- Al Naggar, Y.; Ghorab, M.; Mohamed, K. Phytoecdysteroids: Isolation and Biological Applications. Am. J. Life Sci 2017, 5, 7–10. [Google Scholar] [CrossRef][Green Version]
- Hornok, S.; Csorba, A.; Kováts, D.; Csörgő, T.; Hunyadi, A. Ecdysteroids are present in the blood of wild passerine birds. Sci. Rep. 2019, 9, 17002. [Google Scholar] [CrossRef] [PubMed]
- Kreis, W.; Müller-Uri, F. Biochemistry of Sterols, Cardiac Glycosides, Brassinosteroids, Phytoecdysteroids and Steroid saponins. Annu. Plant Rev. 2010, 40, 304–363. [Google Scholar] [CrossRef]
- Thiem, B.; Kikowska, M.; Maliński, M.P.; Kruszka, D.; Napierała, M.; Florek, E. Ecdysteroids: Production in plant in vitro cultures. Phytochem. Rev. 2017, 16, 603–622. [Google Scholar] [CrossRef]
- Wu, P.; Xie, H.; Tao, W.; Miao, S.; Wei, X. Phytoecdysteroids from the rhizomes of Brainea insignis. Phytochemistry 2010, 71, 975–981. [Google Scholar] [CrossRef]
- Dang, N.H.; Tuyen, P.T.; Loan, V.T.; Dac, L.X. The contents of 20-hydroecdysone (20 E), quercetin and essential oils in Asteraceae species gwowing in Tamdao District, Vinh Phuc Province, Vietnam. Manag. For. Resour. Environ. 2019, 7, 72–81. [Google Scholar]
- Guibout, L.; Mamadalieva, N.; Balducci, C.; Girault, J.-P.; Lafont, R. The minor ecdysteroids from Ajuga turkestanica. Phytochem. Anal. 2015, 26, 293–300. [Google Scholar] [CrossRef]
- Lafont, R.; Dinan, L. Practical uses for ecdysteroids in mammals including humans: An update. J. Insect Sci. 2003, 3, 7. [Google Scholar] [CrossRef] [PubMed]
- Golovatskaya, I.F. Effect of Ecdysterone on Morphological and Physiological Processes in Plants. Russ. J. Plant Physiol. 2004, 51, 407–413. [Google Scholar] [CrossRef]
- Bajguz, A.; Dinan, L. Effects of ecdysteroids on Chlorella vulgaris. Physiol. Plant 2004, 121, 349–357. [Google Scholar] [CrossRef]
- Bakrim, A.; Lamhamdi, M.; Sayah, F.; Chib, F. Effects of plant hormones and 20-hydroxyecdysone on tomato (Lycopersicum esculentum) seed germination and seedlings growth. Afr. J. Biotechnol. 2007, 6, 2792–2802. [Google Scholar]
- Lamhamdi, M.; Lafont, R.; Rharrabe, K.; Sayah, F.; Aarab, A.; Bakrim, A. 20-Hydroxyecdysone protects wheat seedlings (Triticum aestivum L.) against lead stress. Plant Physiol. Biochem. 2016, 98, 64–71. [Google Scholar] [CrossRef]
- Li, J.-T.; Han., X.-P.; Wang, C.; Zhang, W.-Y.; Ma, J.-H. 20-Hydroxyecdysone protects wheat seedlings from salt stress. Arch. Biol. Sci. 2018, 70, 379–386. [Google Scholar] [CrossRef]
- Holá, D.; Frimlová, K.; Kocová, M.; Marková, H.; Rothová, O.; Tumová, L. Effect of exogenously applied 20-hydroxyecdysone on the efficiency of primary photosynthetic processes substantially differs across plant species. Photosynthetica 2020, 58, 961–973. [Google Scholar] [CrossRef]
- Hirsch, K.S.; Adams, E.R.; Hoffman, D.G.; Markham, J.K.; Owen, N.V. Studies to elucidate the mechanism of fenarimol-induced infertility in male rats. Toxicol. Appl. Pharmacol. 1986, 86, 391–399. [Google Scholar] [CrossRef]
- Hirsch, K.S.; Weaver, D.E.; Black, L.J.; Falcone, J.F.; MacLusky, N.J. Inhibition of central nervous system aromatase activity: A mechanism for fenarimol-induced infertility in the male rat. Toxicol. Appl. Pharmacol. 1987, 91, 235–245. [Google Scholar] [CrossRef]
- Grieneisen, M.L.; Warren, J.T.; Gilbert, L.I. Early steps in ecdysteroid biosynthesis: Evidence for the involvement of cytochrome P-450 enzymes. Insect Biochem. Mol. Biol 1993, 23, 13–23. [Google Scholar] [CrossRef]
- Paolini, M.; Pozzetti, L.; Mesirca, R.; Sapone, A.; Cantelli-Forti, G. Testosterone hydroxylase in evaluating induction and suppression of murine CYP isoenzymes by fenarimol. Arch. Toxicol. 1996, 70, 451–456. [Google Scholar] [CrossRef] [PubMed]
- Mu, X.; LeBlanc, G.A. Environmental Antiecdysteroids Alter Embryo Development in the Crustacean Daphnia magna. J. Exp. Zool. 2002, 292, 287–292. [Google Scholar] [CrossRef]
- Andersen, H.R.; Bonefeld-Jørgensen, E.C.; Nielsen, F.; Jarfeldt, K.; Jayatissa, M.N.; Vinggaard, A.M. Estrogenic effects in vitro and in vivo of the fungicide fenarimol. Toxicol. Lett. 2006, 163, 142–152. [Google Scholar] [CrossRef] [PubMed]
- de Castro, V.L.S.S.; de Mello, M.A.; Diniz, C.; Morita, L.; Zucchi, T.; Poli, P. Neurodevelopmental effects of perinatal fenarimol exposure on rats. Reprod. Toxicol. 2007, 23, 98–105. [Google Scholar] [CrossRef]
- Keenan, M.; Chaplin, J.H.; Alexander, P.W.; Abbott, M.J.; Best, W.M.; Khong, A.; Botero, A.; Perez, C.; Cornwall, S.; Thompson, A.; et al. Two Analogues of Fenarimol Show Curative Activity in an Experimental Model of Chagas Disease. J. Med. Chem. 2013, 56, 10158–10170. [Google Scholar] [CrossRef]
- Oh, K.; Matsumoto, T.; Yamagami, A.; Hoshi, T.; Nakano, T.; Yoshizawa, Y. Fenarimol, a Pyrimidine-Type Fungicide, Inhibits Brassinosteroid Biosynthesis. Int. J. Mol. Sci. 2015, 16, 17273–17288. [Google Scholar] [CrossRef]
- Bajguz, A.; Chmur, M.; Gruszka, D. Comprehensive Overview of the Brassinosteroid Biosynthesis Pathways: Substrates, Products, Inhibitors, and Connections. Front. Plant Sci. 2020, 11, 1034. [Google Scholar] [CrossRef]
- Janeczko, A. The significance of ethanol as a hormone solvent in experiments on the physiological activity of brassinosteroids. In Brassinosteroids: A Class of Plant Hormone; Hayat, S., Ahmad, A., Eds.; Springer: Dordrecht, The Netherlands, 2011; pp. 361–374. [Google Scholar] [CrossRef]
- Janeczko, A.; Swaczynová, J. Endogenous brassinosteroids in wheat treated with 24-epibrassinolide. Biol. Plant 2010, 54, 477–482. [Google Scholar] [CrossRef]
- Janeczko, A.; Biesaga–Kościelniak, J.; Oklestkova, J.; Filek, M.; Dziurka, M.; Szarek-Łukaszewska, G.; Kościelniak, J. Role of 24-Epibrassinolide in wheat production: Physiological effects and uptake. J. Agron. Crop Sci. 2010, 196, 311–321. [Google Scholar] [CrossRef]
- Sadura, I.; Pociecha, E.; Dziurka, M.; Oklestkova, J.; Novak, O.; Gruszka, D.; Janeczko, A. Mutations in the HvDWARF, HvCPD and HvBRI1 genes-involved in brassinosteroid biosynthesis/signalling: Altered photosynthetic efficiency, hormonal homeostasis and tolerance to high/low temperatures in barley. J. Plant Growth Regul. 2019, 38, 1062–1081. [Google Scholar] [CrossRef]
- Janeczko, A.; Oklestkova, J.; Novak, O.; Śniegowska-Świerk, K.; Snaczke, Z.; Pociecha, E. Disturbances in production of progesterone and their implications in plant studies. Steroids 2015, 96, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Machackova, I.; Vagner, M.; Slama, K. Comparison between the effects of 20-hydroxyecdysone and phytohormones on growth and development in plants. Eur. J. Entomol. 1995, 92, 309–316. [Google Scholar]
- Schmelz, E.A.; Grebenok, R.J.; Galbraith, D.W.; Bowers, W.S. Insectinduced synthesis of phytoecdysteroids in spinach, Spinacia oleracea. J. Chem. Ecol. 1999, 25, 1739–1757. [Google Scholar] [CrossRef]
- Udalova, Z.V.; Zinov’eva, S.V.; Vasil’eva, I.S.; Paseshnichenko, V.A. Correlation between the structure of plant steroids and their effects on phytoparasitic nematodes. Appl. Biochem. Microbiol. 2004, 40, 93–97. [Google Scholar] [CrossRef]
- Soriano, I.R.; Riley, I.T.; Potter, M.J.; Bowers, W.S. Phytoecdysteroids: A novel defense against plant-parasitic nematodes. J. Chem. Ecol. 2004, 30, 651–654. [Google Scholar] [CrossRef]
- Das, N.; Mishra, S.K.; Bishayee, A.; Ali, E.S.; Bishayee, A. The phytochemical, biological, and medicinal attributes of phytoecdysteroids: An updated review. Acta Pharm. Sin. B 2020. [Google Scholar] [CrossRef]
- Dinan, L.; Harmatha, J.; Volodin, V.; Lafont, R. Phytoecdysteroids: Diversity, Biosynthesis and Distribution. In Ecdysone: Structures and Functions; Smagghe, G., Ed.; Springer: Dordrecht, The Netherlands, 2009; pp. 3–45. [Google Scholar] [CrossRef]
- Grebenok, R.J.; Ripa, P.V.; Adler, J.H. Occurrence and levels of ecdysteroids in spinach. Lipids 1991, 26, 666–668. [Google Scholar] [CrossRef]
- Kholodova, Y.D.; Baltaev, U.; Volovenko, V.O.; Gorovits, M.B.; Abubakirov, N.K. Phytoecdisones of Serratula xeranthemoides. Khimiya Prir. Soedin. 1979, 2, 171–174. [Google Scholar]
- Canals, D.; Irurre-Santilari, J.; Casas, J. The first cytochrome P450 in ferns. Evidence for its involvement in phytoecdysteroid biosynthesis in Polypodium vulgare. FEBS J. 2005, 272, 4817–4825. [Google Scholar] [CrossRef]
- Skoczowski, A.M.; Filek, M. Cold-induced changes in lipids from hypocotyls of winter and spring rape. I. The lipid synthesis and fatty acid composition. Acta Physiol. Plant 1986, 8, 203–212. [Google Scholar]
- Fürtauer, L.; Weiszmann, J.; Weckwerth, W.; Nägele, T. Dynamics of Plant Metabolism during Cold Acclimation. Int. J. Mol. Sci. 2019, 20, 5411. [Google Scholar] [CrossRef]
- Livingston, D.P.; Tuong, T.D.; Owen, S. Carbohydrate changes in winter oat crowns during recovery from freezing. Dyn. Biochem. Process Biotechnol. Mol. Biol. 2009, 3, 16–22. [Google Scholar]
- Pociecha, E.; Jurczyk, B.; Dziurka, M.; Paczyński, R.; Okleštková, J.; Janeczko, A. 24-Epibrassinolide Promotes Carbohydrates Accumulation in Crowns of Perennial Ryegrass during Cold Acclimation by Regulation of Gene Expression and Enzyme Activities which Results in Increased Frost Tolerance. Procedia Environ. Sci. 2015, 29, 234–235. [Google Scholar] [CrossRef][Green Version]
- Rapacz, M.; Jurczyk, B.; Sasal, M. Deacclimation may be crucial for winter survival of cereals under warming climate. Plant Sci. J. 2017, 256, 5–15. [Google Scholar] [CrossRef]
- Rys, M.; Pociecha, E.; Oliwa, J.; Ostrowska, A.; Jurczyk, B.; Saja, D.; Janeczko, A. Deacclimation of Winter Oilseed Rape—Insight into Physiological Changes. Agronomy 2020, 10, 1565. [Google Scholar] [CrossRef]
- Murashige, T.; Skoog, F. A revised medium for rapid growth and bioassays with plant tissue culture. Physiol. Plant 1962, 15, 473–497. [Google Scholar] [CrossRef]
- Kamlar, M.; Rothova, O.; Salajkova, S.; Tarkowska, D.; Drasar, P.; Kocova, M.; Harmatha, J.; Hola, D.; Kohout, L.; Macek, T. The effect of exogenous 24-epibrassinolide on the ecdysteroid content in the leaves of Spinacia oleracea L. Steroids 2015, 97, 107–112. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Janeczko, A.; Oklestkova, J.; Tarkowská, D.; Drygaś, B. Naturally Occurring Ecdysteroids in Triticum aestivum L. and Evaluation of Fenarimol as a Potential Inhibitor of Their Biosynthesis in Plants. Int. J. Mol. Sci. 2021, 22, 2855. https://doi.org/10.3390/ijms22062855
Janeczko A, Oklestkova J, Tarkowská D, Drygaś B. Naturally Occurring Ecdysteroids in Triticum aestivum L. and Evaluation of Fenarimol as a Potential Inhibitor of Their Biosynthesis in Plants. International Journal of Molecular Sciences. 2021; 22(6):2855. https://doi.org/10.3390/ijms22062855
Chicago/Turabian StyleJaneczko, Anna, Jana Oklestkova, Danuše Tarkowská, and Barbara Drygaś. 2021. "Naturally Occurring Ecdysteroids in Triticum aestivum L. and Evaluation of Fenarimol as a Potential Inhibitor of Their Biosynthesis in Plants" International Journal of Molecular Sciences 22, no. 6: 2855. https://doi.org/10.3390/ijms22062855
APA StyleJaneczko, A., Oklestkova, J., Tarkowská, D., & Drygaś, B. (2021). Naturally Occurring Ecdysteroids in Triticum aestivum L. and Evaluation of Fenarimol as a Potential Inhibitor of Their Biosynthesis in Plants. International Journal of Molecular Sciences, 22(6), 2855. https://doi.org/10.3390/ijms22062855








