Chronic Oral Selegiline Treatment Mitigates Age-Related Hearing Loss in BALB/c Mice
Abstract
1. Introduction
2. Results
2.1. Effect of Chronic Oral Administration of Selegiline on Hearing Function in BALB/c and DBA/2J Mice
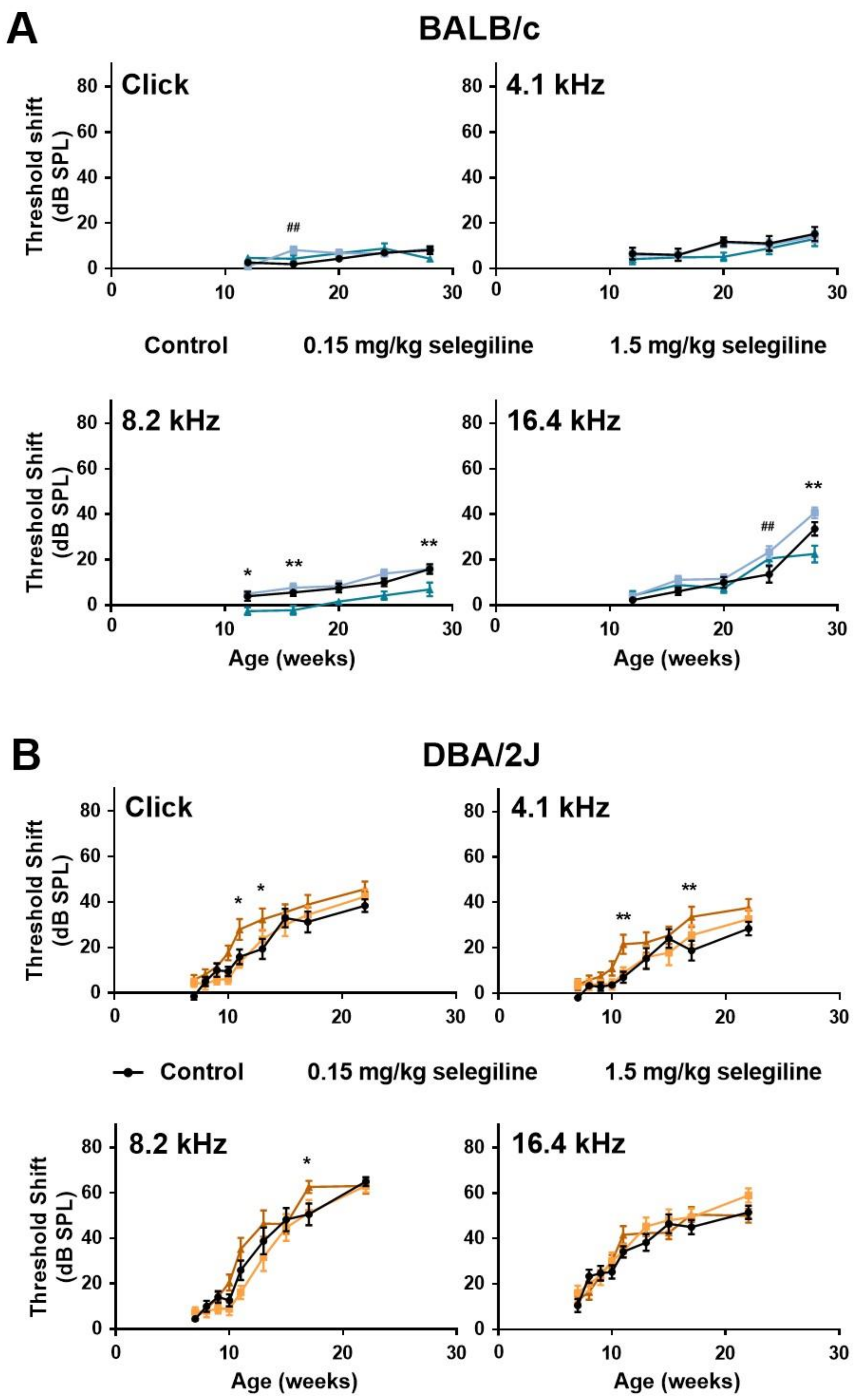
2.1.1. Experiment I (0.15 and 1.5 mg/kg Selegiline)
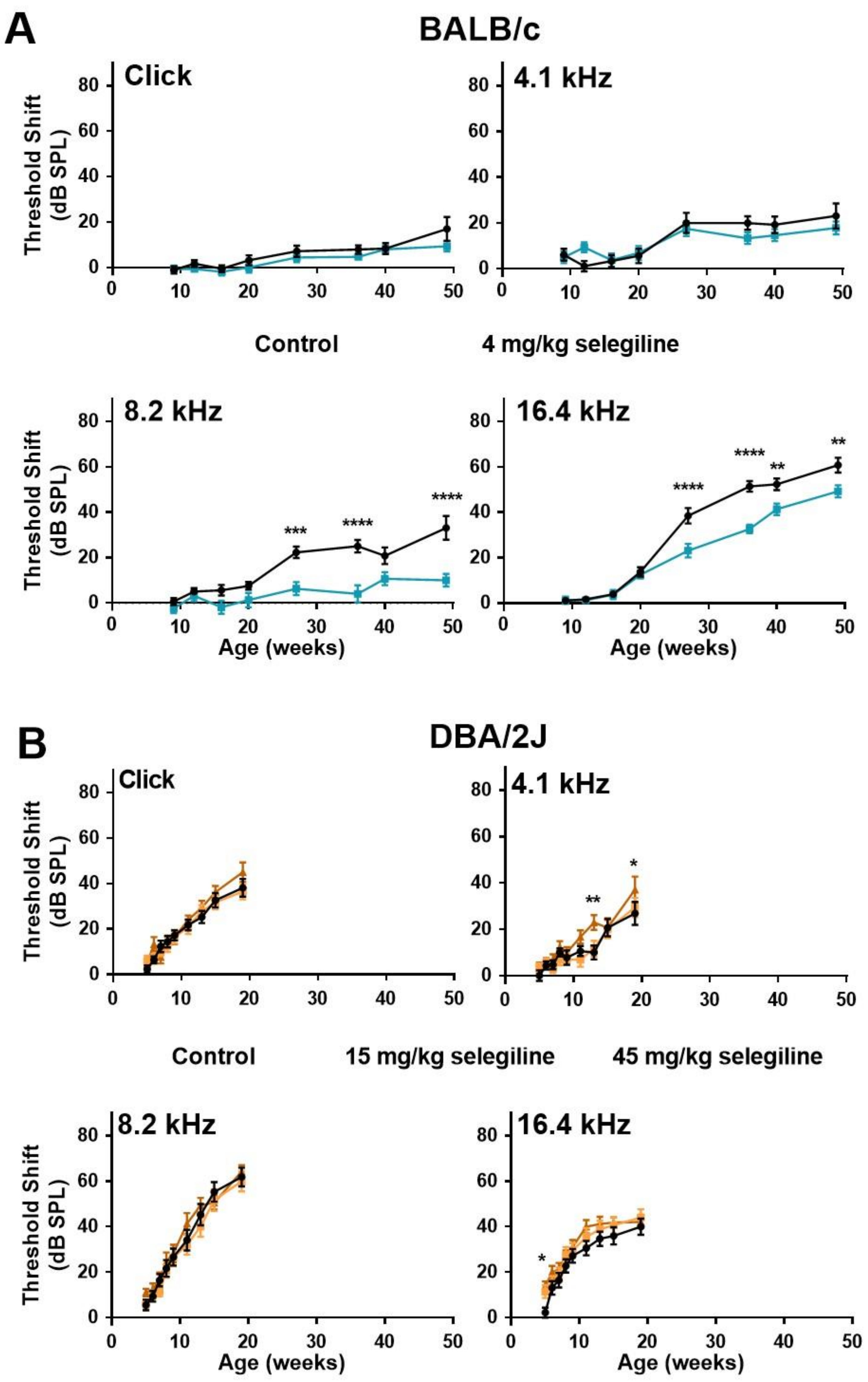
2.1.2. Experiment II (4 and 15, 45 mg/kg Selegiline)
2.2. Effect of Chronic Oral Selegiline Administration on Water Intake, Body Weight and Survival Rate of BALB/c and DBA/2J Mice
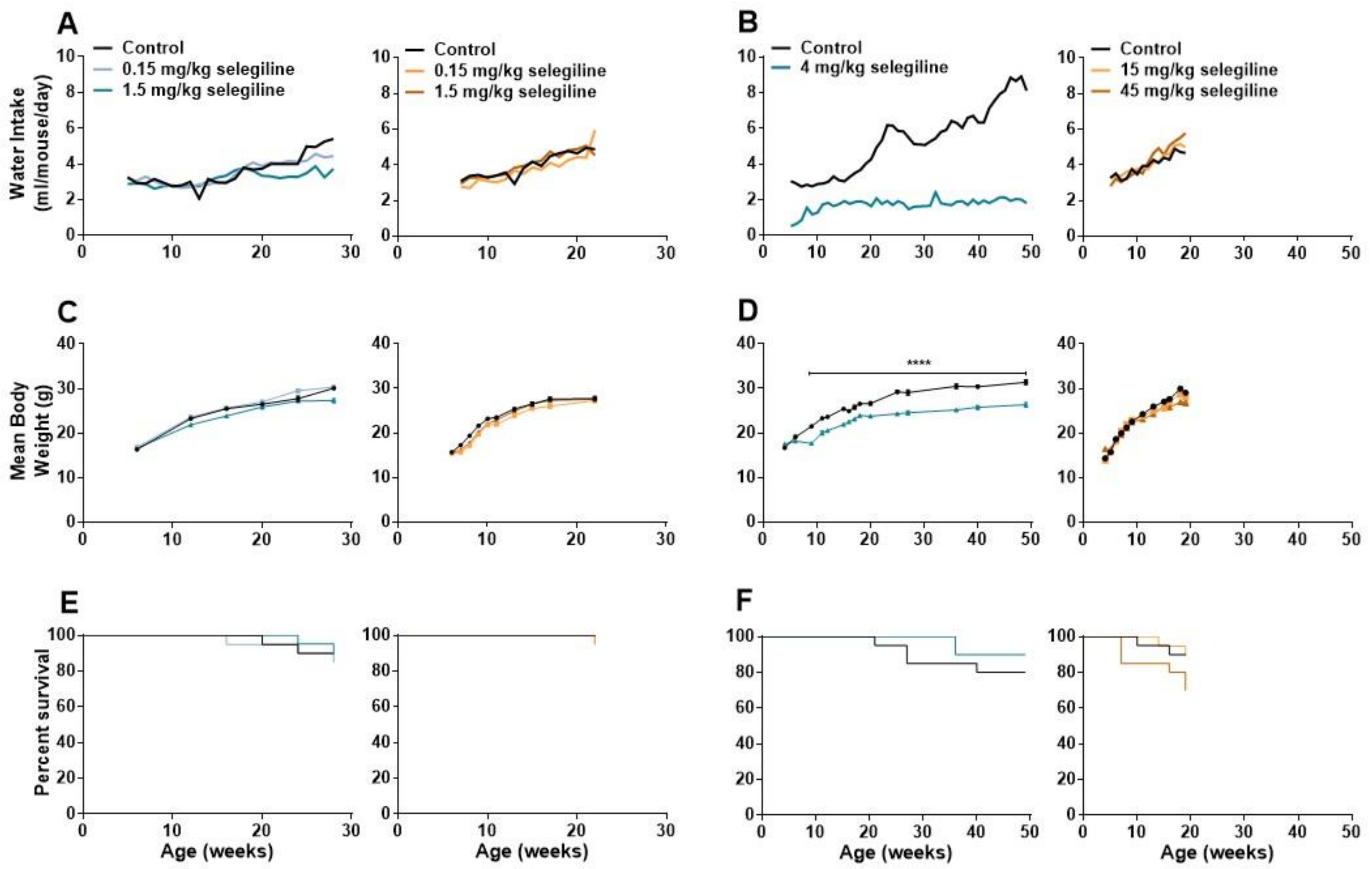
2.2.1. Changes in Water Intake
2.2.2. Changes in Body Weight
2.2.3. Survival Rate
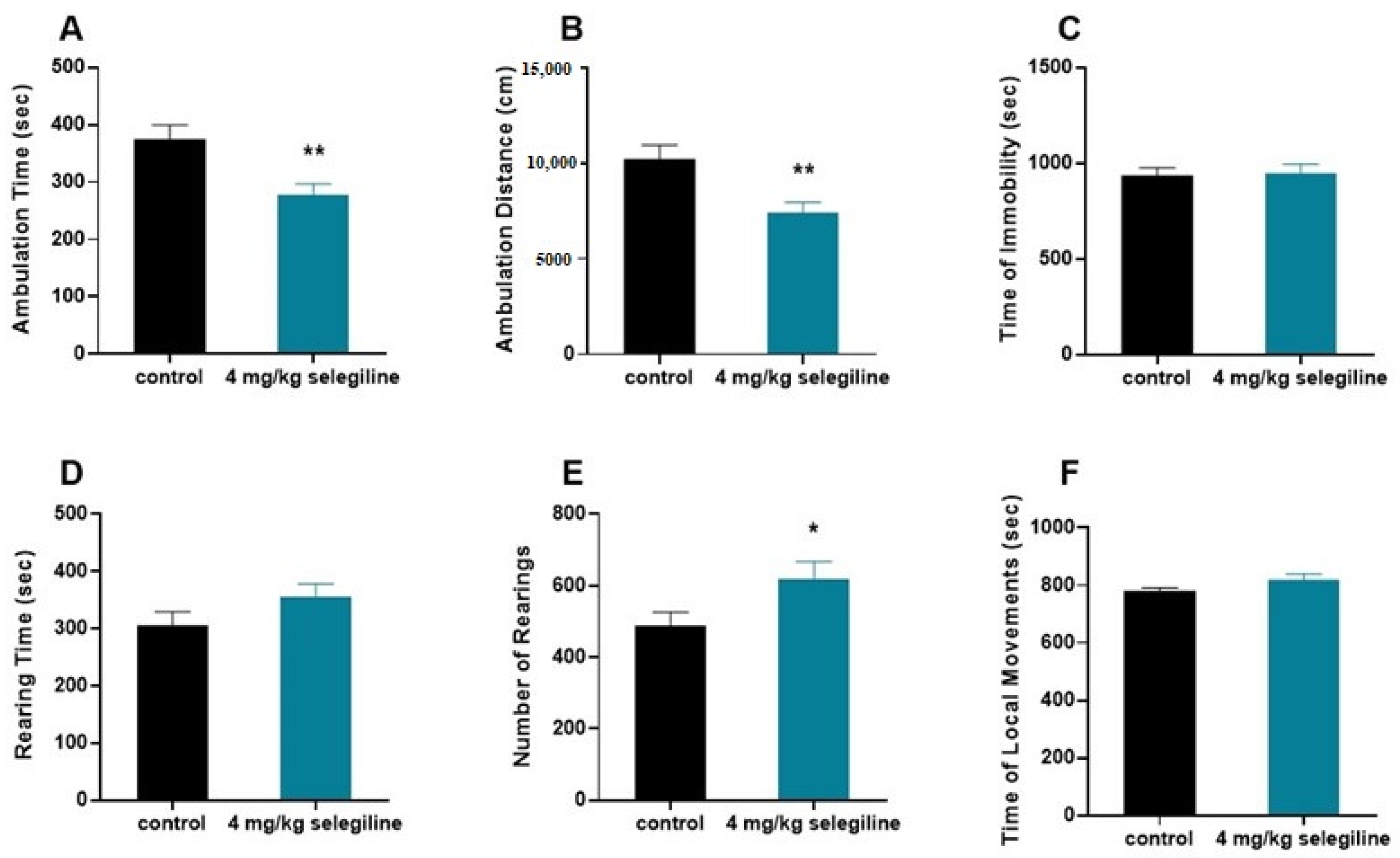
2.3. Effect of 4 mg/kg Selegiline on Locomotor Activity
3. Discussion
4. Materials and Methods
4.1. Ethics Statement
4.2. Experimental Animals and Housing Conditions
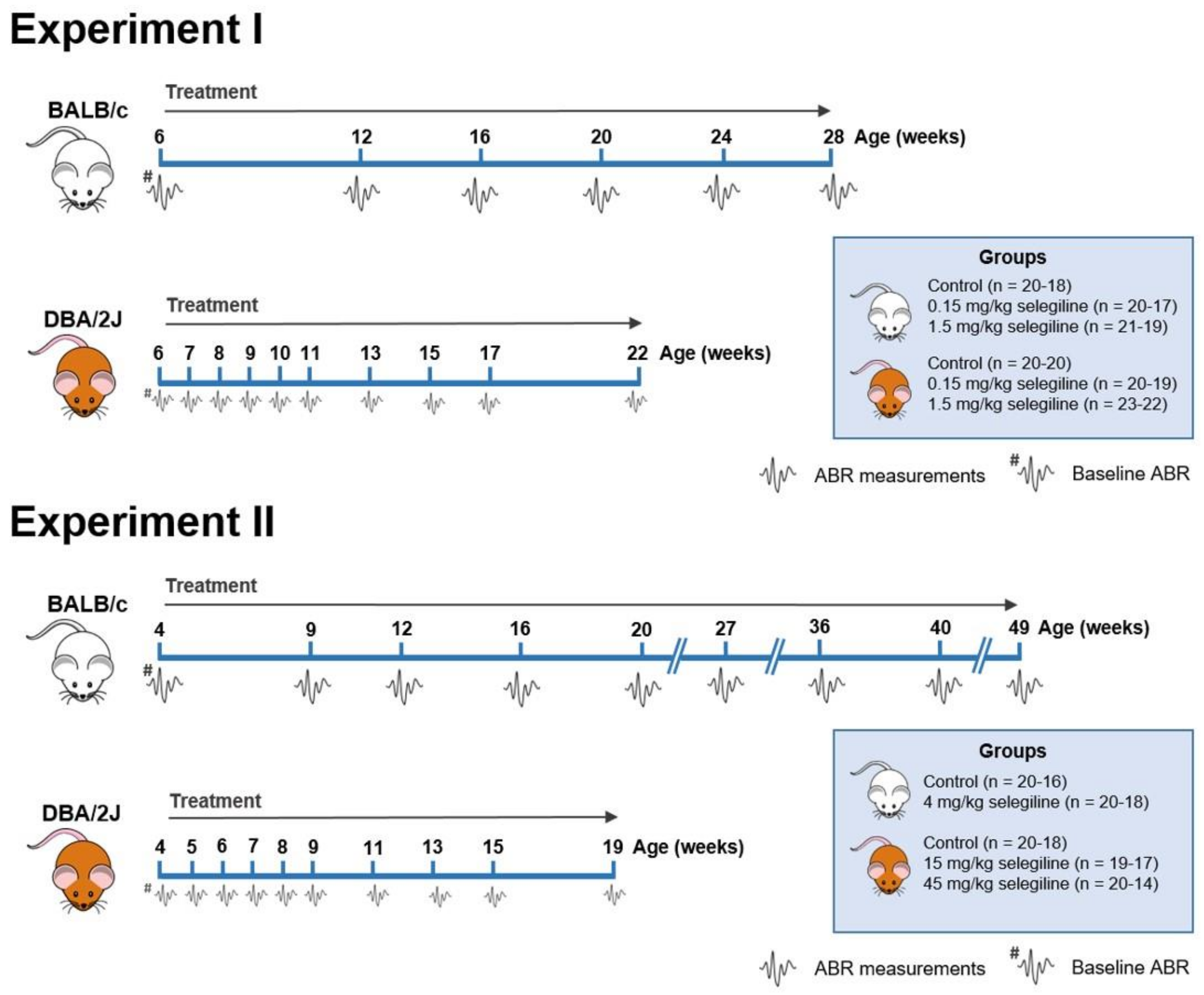
4.3. Experimental Design of Hearing Function Measurements and Selegiline Administration
4.4. Auditory Brainstem Response (ABR) Recordings
4.5. Survival Analysis
4.6. Test of Locomotor Activity
4.7. Data Analysis
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gopinath, B.; Rochtchina, E.; Wang, J.J.; Schneider, J.; Leeder, S.R.; Mitchell, P. Prevalence of age-related hearing loss in older adults: Blue mountains study. Arch. Intern. Med. 2009, 169, 415–416. [Google Scholar] [CrossRef]
- World Health Organization. Deafness and Hearing Loss. Available online: https://www.who.int/news-room/fact-sheets/detail/deafness-and-hearing-loss (accessed on 2 March 2021).
- Peelle, J.E.; Wingfield, A. The Neural Consequences of Age-Related Hearing Loss. Trends Neurosci. 2016, 39, 486–497. [Google Scholar] [CrossRef] [PubMed]
- Dalton, D.S.; Cruickshanks, K.J.; Klein, B.E.K.; Klein, R.; Wiley, T.L.; Nondahl, D.M. The Impact of Hearing Loss on Quality of Life in Older Adults. Gerontologist 2003, 43, 661–668. [Google Scholar] [CrossRef]
- Van Eyken, E.; Van Camp, G.; Van Laer, L. The complexity of age-related hearing impairment: Contributing environmental and genetic factors. Audiol. Neurotol. 2007, 12, 345–358. [Google Scholar] [CrossRef]
- Gates, G.A.; Mills, J.H. Presbycusis. Lancet 2005, 366, 1111–1120. [Google Scholar] [CrossRef]
- Someya, S.; Tanokura, M.; Weindruch, R.; Prolla, T.; Yamasoba, T. Effects of Caloric Restriction on Age-Related Hearing Loss in Rodents and Rhesus Monkeys. Curr. Aging Sci. 2010, 3, 20–25. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Puel, J.L. Toward cochlear therapies. Physiol. Rev. 2018, 98, 2477–2522. [Google Scholar] [CrossRef]
- Szepesy, J.; Miklós, G.; Farkas, J.; Kucsera, D.; Giricz, Z.; Gáborján, A.; Polony, G.; Szirmai, Á.; Tamás, L.; Köles, L.; et al. Anti-PD-1 Therapy Does Not Influence Hearing Ability in the Most Sensitive Frequency Range, but Mitigates Outer Hair Cell Loss in the Basal Cochlear Region. Int. J. Mol. Sci. 2020, 21, 6701. [Google Scholar] [CrossRef]
- Watson, N.; Ding, B.; Zhu, X.; Frisina, R.D. Chronic inflammation—Inflammaging—In the ageing cochlea: A novel target for future presbycusis therapy. Ageing Res. Rev. 2017, 40, 142–148. [Google Scholar] [CrossRef]
- Schuknecht, H.F.; Gacek, M.R. Cochlear pathology in presbycusis. Ann. Otol. Rhinol. Laryngol. 1993, 102, 1–16. [Google Scholar] [CrossRef]
- Otte, J.; Schunknecht, H.F.; Kerr, A.G. Ganglion cell populations in normal and pathological human cochleae. Implications for cochlear implantation. Laryngoscope 1978, 88, 1231–1246. [Google Scholar] [CrossRef]
- Steel, K.P. Similarities between mice and humans with hereditary deafness. Ann. N. Y. Acad. Sci. 1991, 630, 68–79. [Google Scholar] [CrossRef]
- Steel, K.P.; Bock, G.R. Hereditary inner-ear abnormalities in animals. Relationships with human abnormalities. Arch. Otolaryngol. 1983, 109, 22–29. [Google Scholar] [CrossRef]
- Keithley, E.M.; Canto, C.; Zheng, Q.Y.; Fischel-Ghodsian, N.; Johnson, K.R. Age-related hearing loss and the ahl locus in mice. Hear. Res. 2004, 188, 21–28. [Google Scholar] [CrossRef]
- Willott, J.F.; Turner, J.G.; Carlson, S.; Ding, D.; Seegers Bross, L.; Falls, W.A. The BALB/C mouse as an animal model for progressive sensorineural hearing loss. Hear. Res. 1998, 115, 162–174. [Google Scholar] [CrossRef]
- Ralls, K. Auditory sensitivity in mice: Peromyscus and Mus musculus. Anim. Behav. 1967, 15, 123–128. [Google Scholar] [CrossRef]
- Knoll, J.; Magyar, K. Some puzzling pharmacological effects of monoamine oxidase inhibitors. Adv. Biochem. Psychopharmacol. 1972, 5, 393–408. [Google Scholar]
- Alexander Bodkin, J.; Amsterdam, J.D. Transdermal Selegiline in Major Depression: A Double-Blind., Placebo-Controlled, Parallel-Group Study in Outpatients. Am. J. Psychiatry 2020, 159, 1869–1875. [Google Scholar] [CrossRef]
- Miklya, I. The significance of selegiline/(-)-deprenyl after 50 years in research and therapy (1965–2015). Mol. Psychiatry 2016, 21, 1499–1503. [Google Scholar] [CrossRef]
- Knoll, J. (-)Deprenyl-medication: A strategy to modulate the age-related decline of the striatal dopaminergic system. J. Am. Geriatr. Soc. 1992, 40, 839–847. [Google Scholar] [CrossRef]
- Ruehl, W.W.; Entriken, T.L.; Muggenburg, B.A.; Bruyette, D.S.; Griffith, W.C.; Hahn, F.F. Treatment with L-deprenyl prolongs life in elderly dogs. Life Sci. 1997, 61, 1037–1044. [Google Scholar] [CrossRef]
- Charles River Laboratories. Available online: https://www.criver.com/products-services/find-model/balbc-mouse?region=3631 (accessed on 2 March 2021).
- Talbot, S.R.; Biernot, S.; Bleich, A.; van Dijk, R.M.; Ernst, L.; Häger, C.; Helgers, S.O.A.; Koegel, B.; Koska, I.; Kuhla, A.; et al. Defining body-weight reduction as a humane endpoint: A critical appraisal. Lab. Anim. 2020, 54, 99–110. [Google Scholar] [CrossRef]
- Morton, D.B. A systematic approach for establishing humane endpoints. ILAR J. 2000, 41, 80–86. [Google Scholar] [CrossRef]
- Morton, D.B.; Griffiths, P.H. Guidelines on the recognition of pain, distress and discomfort in experimental animals and a hypothesis for assessment. Vet. Rec. 1985, 116, 431–436. [Google Scholar] [CrossRef]
- Kachele, D.L.; Lasiter, P.S. Murine strain differences in taste responsivity and organization of the rostral nucleus of the solitary tract. Brain Res. Bull. 1990, 24, 239–247. [Google Scholar] [CrossRef]
- Someya, S.; Prolla, T.A. Mitochondrial oxidative damage and apoptosis in age-related hearing loss. Mech. Ageing Dev. 2010, 131, 480–486. [Google Scholar] [CrossRef]
- Wang, J.; Puel, J.-L. Presbycusis: An Update on Cochlear Mechanisms and Therapies. J. Clin. Med. 2020, 9, 2018. [Google Scholar] [CrossRef]
- Marie, A.; Meunier, J.; Brun, E.; Malmstrom, S.; Baudoux, V.; Flaszka, E.; Naert, G.; Roman, F.; Cosnier-Pucheu, S.; Gonzalez-Gonzalez, S. N-acetylcysteine Treatment Reduces Age-related Hearing Loss and Memory Impairment in the Senescence-Accelerated Prone 8 (SAMP8) Mouse Model. Aging Dis. 2018, 9, 664–673. [Google Scholar] [CrossRef] [PubMed]
- Ding, D.; Jiang, H.; Chen, G.-D.; Longo-Guess, C.; Muthaiah, V.P.K.; Tian, C.; Sheppard, A.; Salvi, R.; Johnson, K.R. N-acetyl-cysteine prevents age-related hearing loss and the progressive loss of inner hair cells in γ-glutamyl transferase 1 deficient mice. Aging 2016, 8, 730–750. [Google Scholar] [CrossRef]
- Ruan, Q.; Zeng, S.; Liu, A.; Chen, Z.; Yu, Z.; Zhang, R.; He, J.; Bance, M.; Robertson, G.; Yin, S.; et al. Overexpression of X-Linked Inhibitor of Apoptotic Protein (XIAP) reduces age-related neuronal degeneration in the mouse cochlea. Gene Ther. 2014, 21, 967–974. [Google Scholar] [CrossRef][Green Version]
- Kitani, K.; Minami, C.; Yamamoto, T.; Kanai, S.; Ivy, G.O.; Carrillo, M.-C. Pharmacological interventions in aging and age-associated disorders: Potentials of propargylamines for human use. Ann. N. Y. Acad. Sci. 2002, 959, 295–307. [Google Scholar] [CrossRef]
- Ebadi, M.; Sharma, S.; Shavali, S.; El Refaey, H. Neuroprotective actions of selegiline. J. Neurosci. Res. 2002, 67, 285–289. [Google Scholar] [CrossRef]
- Szökő, É.; Tábi, T.; Riederer, P.; Vécsei, L.; Magyar, K. Pharmacological aspects of the neuroprotective effects of irreversible MAO-B inhibitors, selegiline and rasagiline, in Parkinson’s disease. J. Neural Transm. 2018, 125, 1735–1749. [Google Scholar] [CrossRef]
- Tábi, T.; Vécsei, L.; Youdim, M.B.; Riederer, P.; Szökő, É. Selegiline: A molecule with innovative potential. J. Neural Transm. 2020, 127, 831–842. [Google Scholar] [CrossRef]
- Zheng, Q.Y.; Johnson, K.R.; Erway, L.C. Assessment of hearing in 80 inbred strains of mice by ABR threshold analyses. Hear. Res. 1999, 130, 94–107. [Google Scholar] [CrossRef]
- Bespalov, A.; Michel, M.C.; Steckler, T. (Eds.) Handbook of Experimental Pharmacology: Good Research Practice in Non-Clinical Pharmacology and Biomedicine; Springer International Publishing: Cham, Switzerland, 2020; Volume 257, ISBN 978-3-030-33655-4. [Google Scholar]
- Ehret, G. Age-dependent hearing loss in normal hearing mice. Naturwissenschaften 1974, 61, 506–507. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Johnson, K.R.; Longo-Guess, C.; Gagnon, L.H.; Yu, H.; Zheng, Q.Y. A locus on distal chromosome 11 (ahl8) and its interaction with Cdh23ahl underlie the early onset, age-related hearing loss of DBA/2J mice. Genomics 2008, 92, 219–225. [Google Scholar] [CrossRef]
- Nagtegaal, A.P.; Spijker, S.; Crins, T.T.H.; Neuro-Bsik Mouse Phenomics Consortium; Borst, J.G.G. A novel QTL underlying early-onset, low-frequency hearing loss in BXD recombinant inbred strains. Genes. Brain. Behav. 2012, 11, 911–920. [Google Scholar] [CrossRef]
- Wang, Q.; Zhao, H.; Zheng, T.; Wang, W.; Zhang, X.; Wang, A.; Li, B.; Wang, Y.; Zheng, Q. Otoprotective effects of mouse nerve growth factor in DBA/2J mice with early-onset progressive hearing loss. J. Neurosci. Res. 2017, 95, 1937–1950. [Google Scholar] [CrossRef]
- Fetoni, A.R.; Picciotti, P.M.; Paludetti, G.; Troiani, D. Pathogenesis of presbycusis in animal models: A review. Exp. Gerontol. 2011, 46, 413–425. [Google Scholar] [CrossRef]
- Ohlemiller, K.K.; Gagnon, P.M. Apical-to-basal gradients in age-related cochlear degeneration and their relationship to “primary” loss of cochlear neurons. J. Comp. Neurol. 2004, 479, 103–116. [Google Scholar] [CrossRef]
- Duan, M.; Agerman, K.; Ernfors, P.; Canlon, B. Complementary roles of neurotrophin 3 and a N-methyl-D-aspartate antagonist in the protection of noise and aminoglycoside-induced ototoxicity. Proc. Natl. Acad. Sci. USA 2000, 97, 7597–7602. [Google Scholar] [CrossRef] [PubMed]
- Tabuchi, K.; Nishimura, B.; Tanaka, S.; Hayashi, K.; Hirose, Y.; Hara, A. Ischemia-reperfusion injury of the cochlea: Pharmacological strategies for cochlear protection and implications of glutamate and reactive oxygen species. Curr. Neuropharmacol. 2010, 8, 128–134. [Google Scholar] [CrossRef]
- Ruel, J.; Wang, J.; Rebillard, G.; Eybalin, M.; Lloyd, R.; Pujol, R.; Puel, J.-L. Physiology, pharmacology and plasticity at the inner hair cell synaptic complex. Hear. Res. 2007, 227, 19–27. [Google Scholar] [CrossRef]
- Lendvai, B.; Halmos, G.B.; Polony, G.; Kapocsi, J.; Horváth, T.; Aller, M.; Sylvester Vizi, E.; Zelles, T. Chemical neuroprotection in the cochlea: The modulation of dopamine release from lateral olivocochlear efferents. Neurochem. Int. 2011, 59, 150–158. [Google Scholar] [CrossRef]
- Halmos, G.; Doleviczényi, Z.; Répássy, G.; Kittel, A.; Vizi, E.S.; Lendvai, B.; Zelles, T. D2 autoreceptor inhibition reveals oxygen-glucose deprivation-induced release of dopamine in guinea-pig cochlea. Neuroscience 2005, 132, 801–809. [Google Scholar] [CrossRef]
- Halmos, G.; Gáborján, A.; Lendvai, B.; Répássy, G.; Szabó, L.Z.; Vizi, E.S. Veratridine-evoked release of dopamine from guinea pig isolated cochlea. Hear. Res. 2000, 144, 89–96. [Google Scholar] [CrossRef]
- Polony, G.; Humli, V.; Andó, R.; Aller, M.; Horváth, T.; Harnos, A.; Tamás, L.; Vizi, E.S.; Zelles, T. Protective effect of rasagiline in aminoglycoside ototoxicity. Neuroscience 2014, 265, 263–273. [Google Scholar] [CrossRef]
- Vicente-Torres, M.A.; Dávila, D.; Muñoz, E.; Gil-Loyzaga, P. Effects of aging on cochlear monoamine turnover. Adv. Otorhinolaryngol. 2002, 59, 112–115. [Google Scholar]
- Kujawa, S.G.; Liberman, M.C. Synaptopathy in the noise-exposed and aging cochlea: Primary neural degeneration in acquired sensorineural hearing loss. Hear. Res. 2015, 330, 191–199. [Google Scholar] [CrossRef] [PubMed]
- Doleviczényi, Z.; Vizi, E.S.; Gacsályi, I.; Pallagi, K.; Volk, B.; Hársing, L.G.; Halmos, G.; Lendvai, B.; Zelles, T. 5-HT6/7 Receptor Antagonists Facilitate Dopamine Release in the Cochlea via a GABAergic Disinhibitory Mechanism. Neurochem. Res. 2008, 33, 2364–2372. [Google Scholar] [CrossRef]
- Hársing, L.G.; Vizi, E.S. Release of endogenous dopamine from rat isolated striatum: Effect of clorgyline and (-)-deprenyl. Br. J. Pharmacol. 1984, 83, 741–749. [Google Scholar] [CrossRef]
- Timar, J.; Knoll, B.; Knoll, J. Long-term administration of (-)deprenyl (selegiline), a compound which facilitates dopaminergic tone in the brain, leaves the sensitivity of dopamine receptors to apomorphine unchanged. Arch. Int. Pharmacodyn. Ther. 1986, 284, 255–266. [Google Scholar]
- Walsh, T.; Pierce, S.B.; Lenz, D.R.; Brownstein, Z.; Dagan-Rosenfeld, O.; Shahin, H.; Roeb, W.; McCarthy, S.; Nord, A.S.; Gordon, C.R.; et al. Genomic duplication and overexpression of TJP2/ZO-2 leads to altered expression of apoptosis genes in progressive nonsyndromic hearing loss DFNA51. Am. J. Hum. Genet. 2010, 87, 101–109. [Google Scholar] [CrossRef]
- Hertzano, R.; Dror, A.A.; Montcouquiol, M.; Ahmed, Z.M.; Ellsworth, B.; Camper, S.; Friedman, T.B.; Kelley, M.W.; Avraham, K.B. Lhx3, a LIM domain transcription factor, is regulated by Pou4f3 in the auditory but not in the vestibular system. Eur. J. Neurosci. 2007, 25, 999–1005. [Google Scholar] [CrossRef]
- Ahituv, N.; Avraham, K.B. Auditory and vestibular mouse mutants: Models for human deafness. J. Basic Clin. Physiol. Pharmacol. 2000, 11, 181–191. [Google Scholar] [CrossRef] [PubMed]
- Boucher, S.; Tai, F.W.J.; Delmaghani, S.; Lelli, A.; Singh-Estivalet, A.; Dupont, T.; Niasme-Grare, M.; Michel, V.; Wolff, N.; Bahloul, A.; et al. Ultrarare heterozygous pathogenic variants of genes causing dominant forms of early-onset deafness underlie severe presbycusis. Proc. Natl. Acad. Sci. USA 2020, 117, 31278–31289. [Google Scholar] [CrossRef] [PubMed]
- Brozoski, T.J.; Spires, T.J.D.; Bauer, C.A. Vigabatrin, a GABA transaminase inhibitor, reversibly eliminates tinnitus in an animal model. J. Assoc. Res. Otolaryngol. 2007, 8, 105–118. [Google Scholar] [CrossRef] [PubMed]
- Bauer, C.A.; Brozoski, T.J. Assessing tinnitus and prospective tinnitus therapeutics using a psychophysical animal model. J. Assoc. Res. Otolaryngol. 2001, 2, 54–64. [Google Scholar] [CrossRef][Green Version]
- Bickford, P.C.; Adams, C.E.; Boyson, S.J.; Curella, P.; Gerhardt, G.A.; Heron, C.; Ivy, G.O.; Lin, A.M.L.Y.; Murphy, M.P.; Poth, K.; et al. Long-term treatment of male F344 rats with deprenyl: Assessment of effects on longevity, behavior, and brain function. Neurobiol. Aging 1997, 18, 309–318. [Google Scholar] [CrossRef]
- Speiser, Z.; Fine, T.; Litinetsky, L.; Eliash, S.; Blaugrund, E.; Cohen, S. Differential behavioral syndrome evoked in the rats after multiple doses of SSRI fluoxetine with selective MAO inhibitors rasagiline or selegiline. J. Neural Transm. 2008, 115, 107–116. [Google Scholar] [CrossRef] [PubMed]
- Bekesi, G.; Tulassay, Z.; Lengyel, G.; Schaff, Z.; Szombath, D.; Stark, J.; Marczell, I.; Nagy-Repas, P.; Adler, I.; Dinya, E.; et al. The effect of selegiline on total scavenger capacity and liver fat content: A preliminary study in an animal model. J. Neural Transm. 2012, 119, 25–30. [Google Scholar] [CrossRef]
- Boughter, J.D.; Bachmanov, A.A. Behavioral genetics and taste. BMC Neurosci. 2007, 8 (Suppl. 3), S3. [Google Scholar] [CrossRef]
- Bucher, J.R. NTP Technical Report on the Toxicity Studies of Sodium Dichromate Dihydrate Administered in Drinking Water to Male and Female F344/N Rats and B6C3F 1 Mice and Male BALB/c and am3-C57BL/6 Mice. Toxic Rep Ser. 2007, 72, 1-G4. [Google Scholar]
- Hamilton, L.W.; Flaherty, C.F. Interactive effects of deprivation in the albino rat. Learn. Motiv. 1973, 4, 148–162. [Google Scholar] [CrossRef]
- Seidman, M.D. Effects of dietary restriction and antioxidants on presbyacusis. Laryngoscope 2000, 110, 727–738. [Google Scholar] [CrossRef]
- Someya, S.; Yamasoba, T.; Weindruch, R.; Prolla, T.A.; Tanokura, M. Caloric restriction suppresses apoptotic cell death in the mammalian cochlea and leads to prevention of presbycusis. Neurobiol. Aging 2007, 28, 1613–1622. [Google Scholar] [CrossRef] [PubMed]
- Sweet, R.J.; Price, J.M.; Henry, K.R. Dietary restriction and presbyacusis: Periods of restriction and auditory threshold losses in the CBA/j mouse. Audiology 1988, 27, 305–312. [Google Scholar] [CrossRef]
- Henry, K.R. Effects of dietary restriction on presbyacusis in the mouse. Audiology 1986, 25, 329–337. [Google Scholar] [CrossRef]
- Willott, J.F.; Erway, L.C.; Archer, J.R.; Harrison, D.E. Genetics of age-related hearing loss in mice. II. Strain differences and effects of caloric restriction on cochlear pathology and evoked response thresholds. Hear. Res. 1995, 88, 143–155. [Google Scholar] [CrossRef]
- Stoll, S.; Hafner, U.; Pohl, O.; Müller, W.E. Age-related memory decline and longevity under treatment with selegiline. Life Sci. 1994, 55, 2155–2163. [Google Scholar] [CrossRef]
- Yen, T.T.; Knoll, J. Extension of lifespan in mice treated with Dinh lang (Policias fruticosum L.) and (-)deprenyl. Acta Physiol. Hung. 1992, 79, 119–124. [Google Scholar]
- Milgram, N.W.; Racine, R.J.; Nellis, P.; Mendonca, A.; Ivy, G.O. Maintenance on L-deprenyl prolongs life in aged male rats. Life Sci. 1990, 47, 415–420. [Google Scholar] [CrossRef]
- Kitani, K.; Kanai, S.; Ivy, G.O.; Carrillo, M.C. Assessing the effects of deprenyl on longevity and antioxidant defenses in different animal models. Ann. N. Y. Acad. Sci. 1998, 854, 291–306. [Google Scholar] [CrossRef] [PubMed]
- Suzuki, S.; Ishikawa, M.; Ueda, T.; Ohshiba, Y.; Miyasaka, Y.; Okumura, K.; Yokohama, M.; Taya, C.; Matsuoka, K.; Kikkawa, Y. Quantitative trait loci on chromosome 5 for susceptibility to frequency-specific effects on hearing in DBA/2J mice. Exp. Anim. 2015, 64, 241–251. [Google Scholar] [CrossRef]
- Spiby, J. Screening for Hearing Loss in Older Adults. In External Review against Programme Appraisal Criteria for the UK National Screening Committee (UK NSC); The UK National Screening Committee: Waterloo Road, London, UK, 2014. [Google Scholar]
- Bagai, A.; Thavendiranathan, P.; Detsky, A.S. Does this patient have hearing impairment? J. Am. Med. Assoc. 2006, 295, 416–428. [Google Scholar] [CrossRef]
- Lycke, M.; Lefebvre, T.; Cool, L.; Van Eygen, K.; Boterberg, T.; Schofield, P.; Debruyne, P.R. Screening methods for age-related hearing loss in older patients with cancer: A review of the literature. Geriatr. 2018, 3, 48. [Google Scholar] [CrossRef]
- Zahnert, T. The Differential Diagnosis of Hearing Loss. Dtsch. Arztebl. 2011, 108, 433–444. [Google Scholar] [CrossRef]
- Olusanya, B.O.; Davis, A.C.; Hoffman, H.J. Hearing loss grades and the international classification of functioning, disability and health. Bull. World Health Organ. 2019, 97, 725–728. [Google Scholar] [CrossRef]
- World Health Organization. Guidelines for Hearing Aids and Services for Developing Countries. Available online: https://www.who.int/publications/i/item/guidelines-for-hearing-aids-and-services-for-developing-countries (accessed on 18 September 2019).
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Szepesy, J.; Humli, V.; Farkas, J.; Miklya, I.; Tímár, J.; Tábi, T.; Gáborján, A.; Polony, G.; Szirmai, Á.; Tamás, L.; et al. Chronic Oral Selegiline Treatment Mitigates Age-Related Hearing Loss in BALB/c Mice. Int. J. Mol. Sci. 2021, 22, 2853. https://doi.org/10.3390/ijms22062853
Szepesy J, Humli V, Farkas J, Miklya I, Tímár J, Tábi T, Gáborján A, Polony G, Szirmai Á, Tamás L, et al. Chronic Oral Selegiline Treatment Mitigates Age-Related Hearing Loss in BALB/c Mice. International Journal of Molecular Sciences. 2021; 22(6):2853. https://doi.org/10.3390/ijms22062853
Chicago/Turabian StyleSzepesy, Judit, Viktória Humli, János Farkas, Ildikó Miklya, Júlia Tímár, Tamás Tábi, Anita Gáborján, Gábor Polony, Ágnes Szirmai, László Tamás, and et al. 2021. "Chronic Oral Selegiline Treatment Mitigates Age-Related Hearing Loss in BALB/c Mice" International Journal of Molecular Sciences 22, no. 6: 2853. https://doi.org/10.3390/ijms22062853
APA StyleSzepesy, J., Humli, V., Farkas, J., Miklya, I., Tímár, J., Tábi, T., Gáborján, A., Polony, G., Szirmai, Á., Tamás, L., Köles, L., Vizi, E. S., & Zelles, T. (2021). Chronic Oral Selegiline Treatment Mitigates Age-Related Hearing Loss in BALB/c Mice. International Journal of Molecular Sciences, 22(6), 2853. https://doi.org/10.3390/ijms22062853






