Loss of Nckx3 Exacerbates Experimental DSS-Induced Colitis in Mice through p53/NF-κB Pathway
Abstract
1. Introduction
2. Results
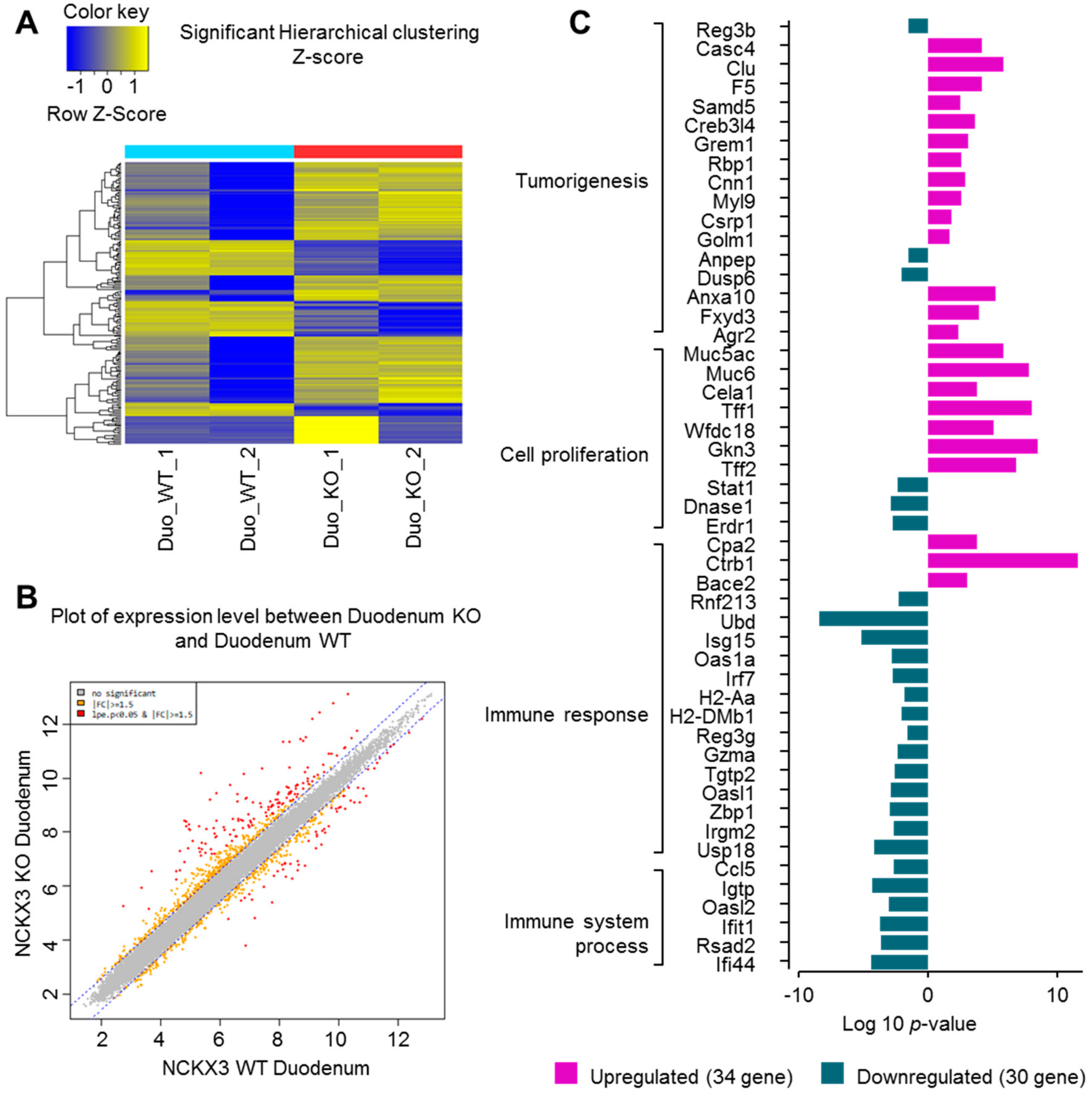
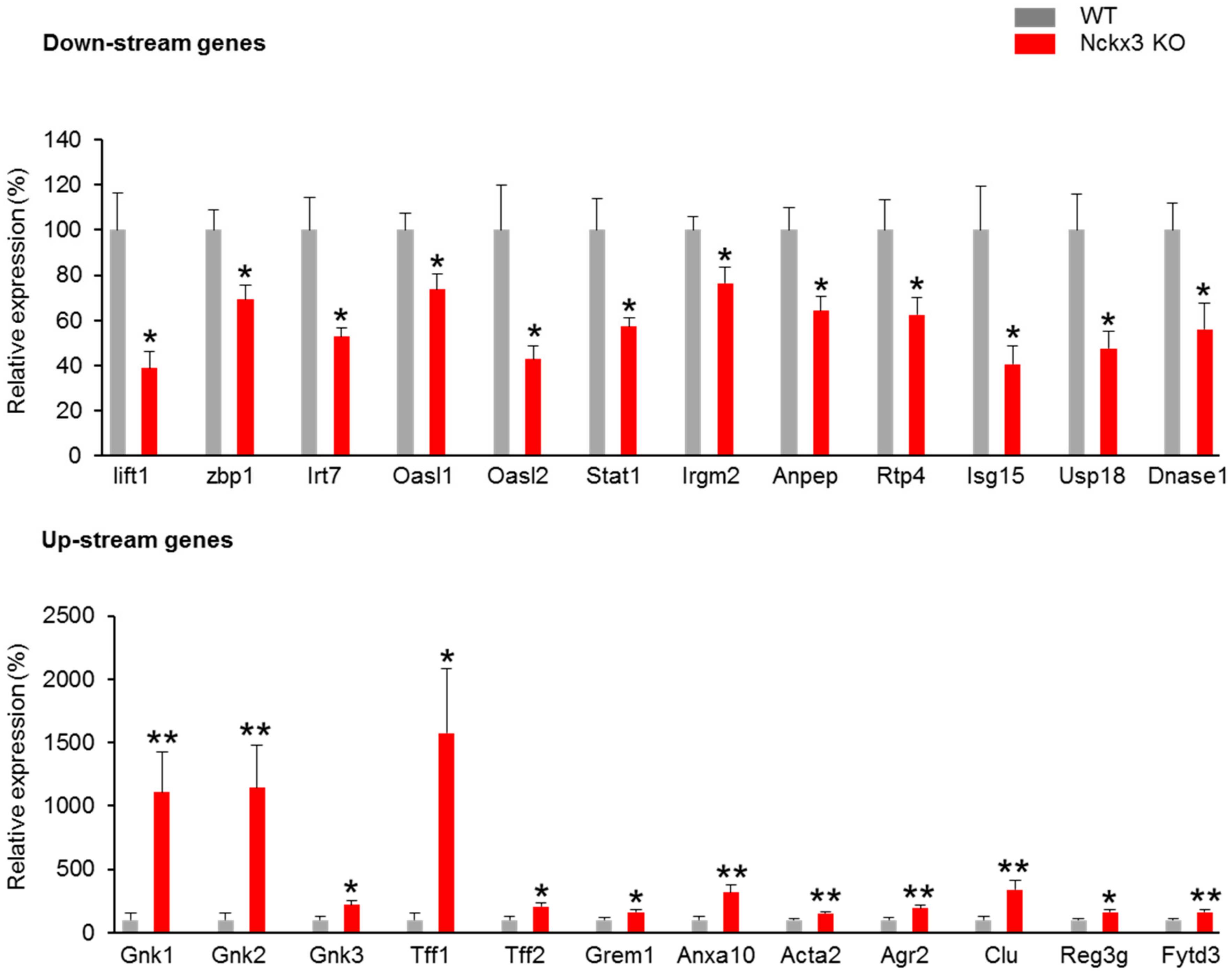
2.1. Nckx3 KO Mice Exhibit Distinct Transcriptional Profiles of Proliferation- and Inflammation-Associated Genes
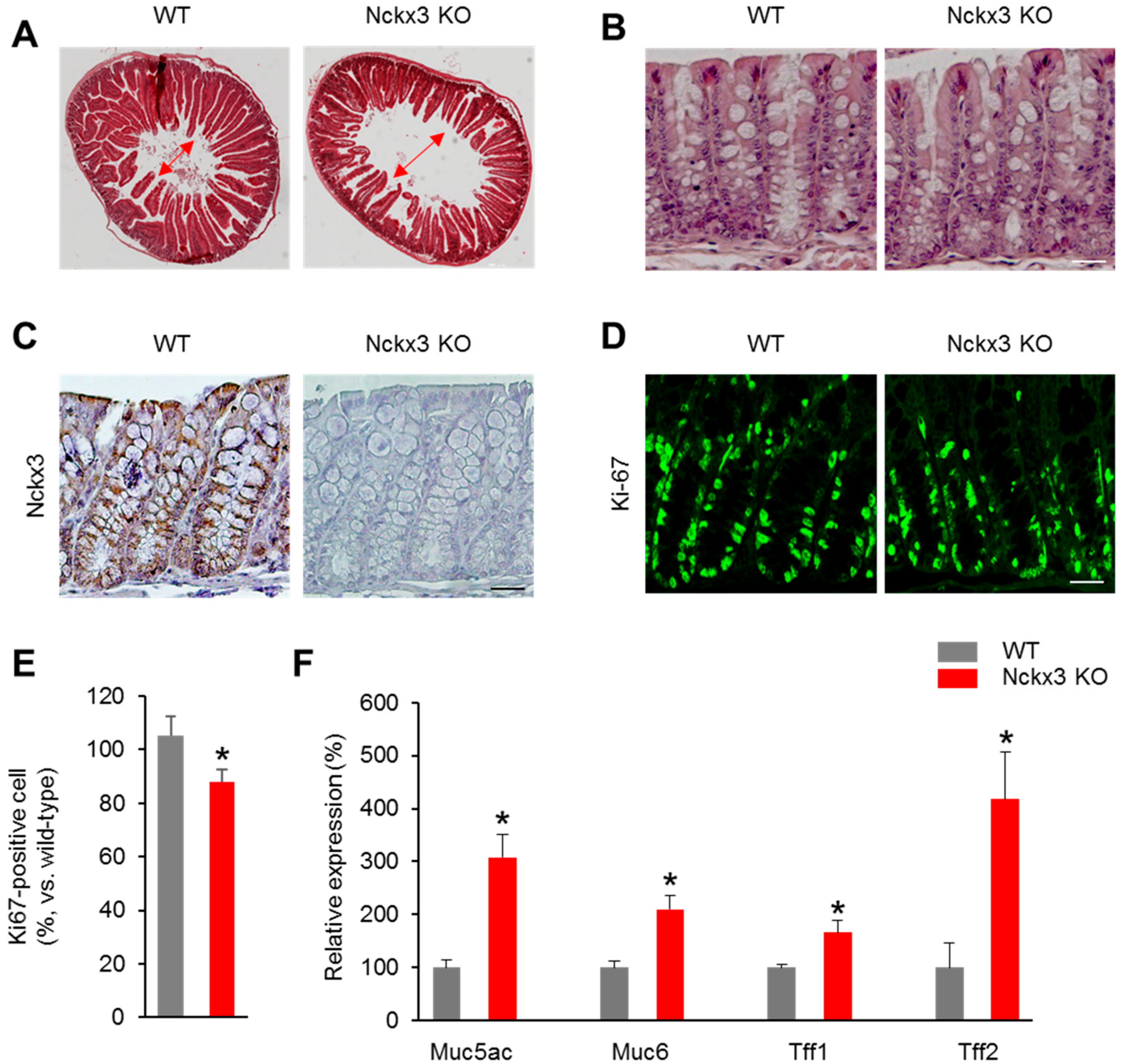
2.2. Nckx3 Loss Impairs the Morphology and Cell Proliferation of the Gut
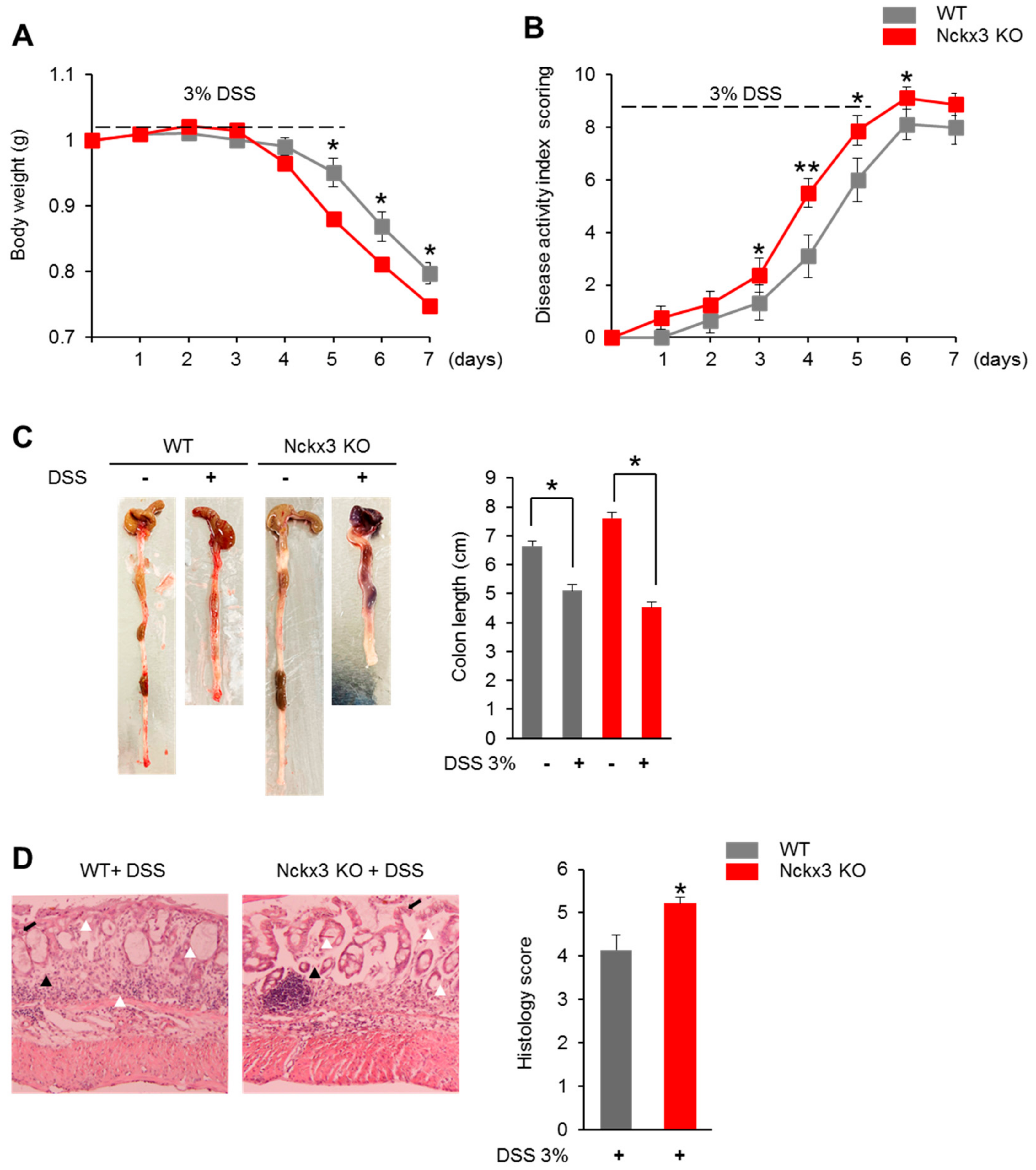
2.3. Nckx3 Loss Promotes Acute DSS-Induced Colitis
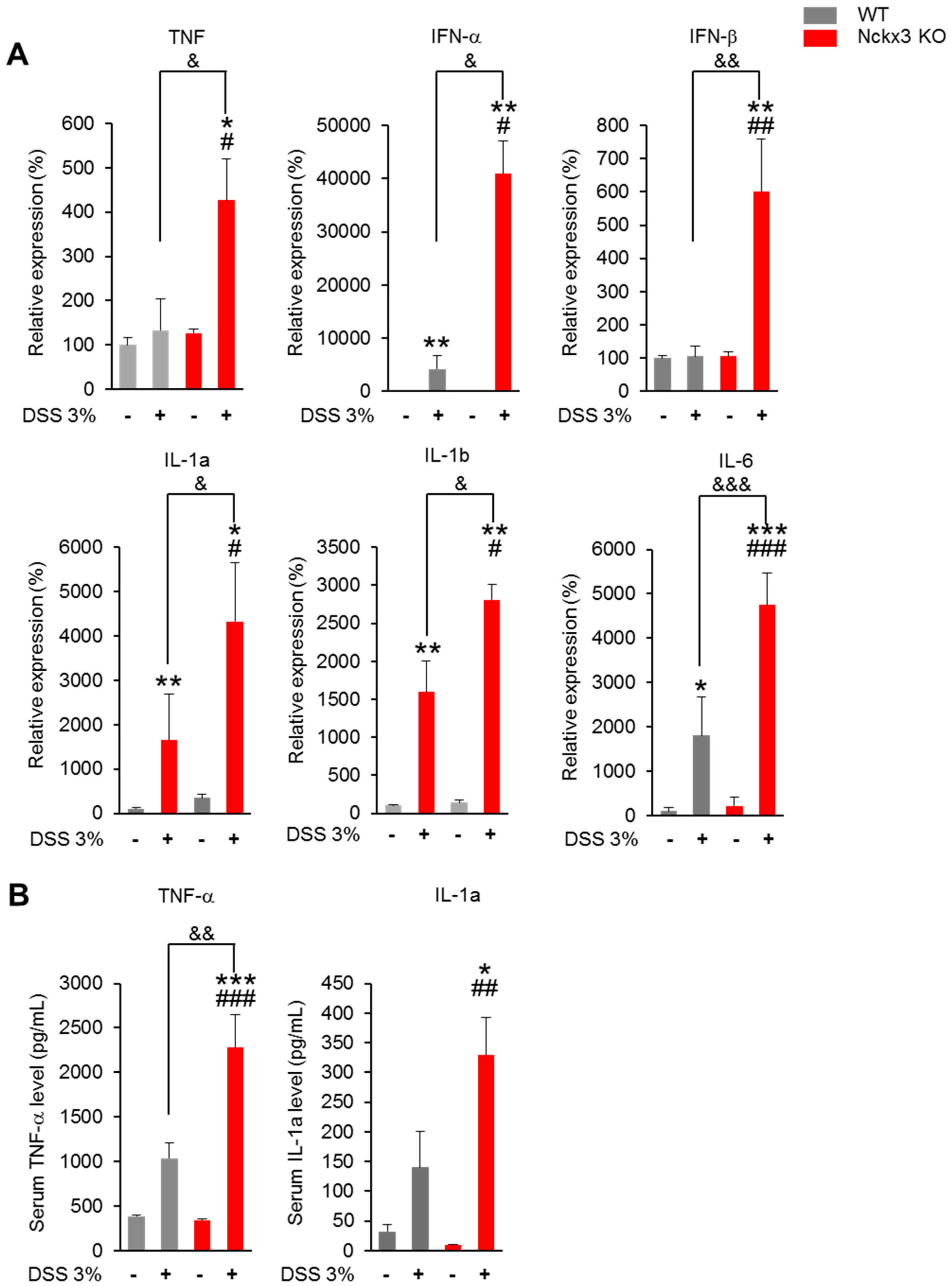
2.4. Nckx3 Deficiency Increased the Production of Dextran Sodium Sulfate (DSS)-Induced Proinflammatory Mediators
2.5. Nckx3 Loss Upregulated NF-κB Signaling in Acute DSS-Induced Colitis
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Microarray Assay
4.3. DSS-Induced Colitis
4.4. Colitis Scores and Histologic Analysis
4.5. Immunofluorescence
4.6. RNA Extraction and Quantitative Real-Time PCR
4.7. Western Blot Analysis
4.8. ELISA Assay
4.9. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Rigoli, L.; Caruso, R.A. Inflammatory Bowel Disease in Pediatric and Adolescent Patients: A Biomolecular and Histopathological Review. World J. Gastroenterol. 2014, 20, 10262. [Google Scholar] [CrossRef]
- Molodecky, N.A.; Soon, S.; Rabi, D.M.; Ghali, W.A.; Ferris, M.; Chernoff, G.; Benchimol, E.I.; Panaccione, R.; Ghosh, S.; Barkema, H.W. Increasing Incidence and Prevalence of the Inflammatory Bowel Diseases with Time, Based on Systematic Review. Gastroenterology 2012, 142, 46–54. [Google Scholar] [CrossRef]
- Dahlhamer, J.M.; Zammitti, E.P.; Ward, B.W.; Wheaton, A.G.; Croft, J.B. Prevalence of Inflammatory Bowel Disease Among Adults Aged≥ 18 years—United States 2015. Morb. Mortal. Wkly. Rep. 2016, 65, 1166–1169. [Google Scholar] [CrossRef]
- Molodecky, N.A.; Kaplan, G.G. Environmental Risk Factors for Inflammatory Bowel Disease. Gastroenterol. Hepatol. 2010, 339. [Google Scholar]
- Hogan, P.G.; Chen, L.; Nardone, J.; Rao, A. Transcriptional Regulation by Calcium, Calcineurin, and NFAT. Genes Dev. 2003, 17, 2205–2232. [Google Scholar] [CrossRef]
- Lewis, R.S. The Molecular Choreography of a Store-Operated Calcium Channel. Nature 2007, 446, 284–287. [Google Scholar] [CrossRef] [PubMed]
- Hogan, P.G.; Rao, A. Dissecting ICRAC, a Store-Operated Calcium Current. Trends Biochem. Sci. 2007, 32, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Vig, M.; Kinet, P.J. Calcium Signaling in Immune Cells. Nat. Immunol. 2009, 10, 21. [Google Scholar] [CrossRef] [PubMed]
- Macian, F. NFAT Proteins: Key Regulators of T-Cell Development and Function. Nat. Rev. Immunol. 2005, 5, 472–484. [Google Scholar] [CrossRef] [PubMed]
- Park, Y.; Jun, C.; Kang, H.; Kim, H.; Kim, H.; Chung, H. Role of Intracellular Calcium as a Priming Signal for the Induction of Nitric Oxide Synthesis in Murine Peritoneal Macrophages. Immunology 1996, 87, 296–302. [Google Scholar] [CrossRef] [PubMed]
- Kenny, E.F.; Quinn, S.R.; Doyle, S.L.; Vink, P.M.; van Eenennaam, H.; O’Neill, L.A. Bruton’s Tyrosine Kinase Mediates the Synergistic Signalling Between TLR9 and the B Cell Receptor by Regulating Calcium and Calmodulin. PLoS ONE 2013, 8, 74103. [Google Scholar] [CrossRef] [PubMed]
- Shintani, Y.; Drexler, H.C.; Kioka, H.; Terracciano, C.M.; Coppen, S.R.; Imamura, H.; Akao, M.; Nakai, J.; Wheeler, A.P.; Higo, S. Toll-Like Receptor 9 Protects Non-Immune Cells from Stress by Modulating Mitochondrial ATP Synthesis Through the Inhibition of SERCA 2. EMBO Rep. 2014, 15, 438–445. [Google Scholar] [CrossRef] [PubMed]
- Krawitt, E.L.; Beeken, W.L.; Janney, D.C. Calcium Absorption in Crohn’s Disease. Gastroenterology 1976, 71, 251–254. [Google Scholar] [CrossRef]
- Wang, Y.; Li, J.-X.; Ji, G.-J.; Zhai, K.; Wang, H.-H.; Liu, X.-G. The Involvement of Ca2+ Signal Pathways in Distal Colonic Myocytes in a Rat Model of Dextran Sulfate Sodium-Induced Colitis. Chin. Med. J. 2016, 129, 1185. [Google Scholar] [CrossRef]
- Shi, W.; Zou, R.; Yang, M.; Mai, L.; Ren, J.; Wen, J.; Liu, Z.; Lai, R. Analysis of Genes Involved in Ulcerative Colitis Activity and Tumorigenesis Through Systematic Mining of Gene Co-expression Networks. Front. Physiol. 2019, 10, 662. [Google Scholar] [CrossRef]
- Schepens, M.A.; Schonewille, A.J.; Vink, C.; van Schothorst, E.M.; Kramer, E.; Hendriks, T.; Brummer, R.-J.; Keijer, J.; van der Meer, R.; Bovee-Oudenhoven, I.M. Supplemental Calcium Attenuates the Colitis-Related Increase in Diarrhea, Intestinal Permeability, and Extracellular Matrix Breakdown in HLA-B27 Transgenic Rats. J. Nutr. 2009, 139, 1525–1533. [Google Scholar] [CrossRef]
- Bjarnason, I.; Macpherson, A.; Mackintosh, C.; Buxton-Thomas, M.; Forgacs, I.; Moniz, C. Reduced Bone Density in Patients with Inflammatory Bowel Disease. Gut 1997, 40, 228–233. [Google Scholar] [CrossRef]
- Lima, C.A.; Lyra, A.C.; Rocha, R.; Santana, G.O. Risk Factors for Osteoporosis in Inflammatory Bowel Disease Patients. World J. Gastrointest. Pathophysiol. 2015, 6, 210. [Google Scholar] [CrossRef] [PubMed]
- Abitbol, V.; Mary, J.; Roux, C.; Soule, J.; Belaiche, J.; Dupas, J.L.; Gendre, J.; Lerebours, E.; Chaussade, S.; Digestives, G.D.T.A.I. Osteoporosis in Inflammatory Bowel Disease: Effect of Calcium and Vitamin D with or Without Fluoride. Aliment. Pharmacol. Ther. 2002, 16, 919–927. [Google Scholar] [CrossRef]
- Aneiros, E.; Philipp, S.; Lis, A.; Freichel, M.; Cavalié, A. Modulation of Ca2+ Signaling by Na+/Ca2+ Exchangers in Mast Cells. J. Immunol. 2005, 174, 119–130. [Google Scholar] [CrossRef]
- Schnetkamp, P.P. The SLC24 Gene Family of Na+/Ca2+–K+ Exchangers: From Sight and Smell to Memory Consolidation and Skin Pigmentation. Mol. Asp. Med. 2013, 34, 455–464. [Google Scholar] [CrossRef]
- Yang, H.; Yoo, Y.M.; Jung, E.M.; Choi, K.C.; Jeung, E.B. Uterine Expression of Sodium/Potassium/Calcium Exchanger 3 and its Regulation by Sex-Steroid Hormones During the Estrous Cycle of Rats. Mol. Reprod. Dev. 2010, 77, 971–977. [Google Scholar] [CrossRef]
- Altimimi, H.F.; Schnetkamp, P.P. Na+/Ca2+-K+ Exchangers (NCKX): Functional Properties and Physiological Roles. Channels 2007, 1, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Yang, H.; An, B.-S.; Choi, K.-C.; Jeung, E.-B. Change of Genes in Calcium Transport Channels Caused by Hypoxic Stress in the Placenta, Duodenum, and Kidney of Pregnant Rats. Biol. Reprod. 2013, 88, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ahn, C.; Choi, J.S.; Jeung, B.E. Organ-Specific Expression of the Divalent Ion Channel Proteins NCKX3, TRPV2, CTR1, ATP7A, IREG1 and HEPH in Various Canine Organs. Mol. Med. Rep. 2018, 18, 1773–1781. [Google Scholar] [CrossRef]
- Yang, H.; Ahn, C.; Shin, E.-K.; Lee, J.-S.; An, B.-S.; Jeung, E.-B. NCKX3 Was Compensated by Calcium Transporting Genes and Bone Resorption in a NCKX3 KO Mouse Model. Mol. Cell. Endocrinol. 2017, 454, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Robertson, S.Y.; Wen, X.; Yin, K.; Chen, J.; Smith, C.E.; Paine, M.L. Multiple Calcium Export Exchangers and Pumps are a Prominent Feature of Enamel Organ Cells. Front. Physiol. 2017, 8, 336. [Google Scholar] [CrossRef]
- Li, X.F.; Kraev, A.S.; Lytton, J. Molecular Cloning of a Fourth Member of the Potassium-Dependent Sodium-Calcium Exchanger Gene Family NCKX4. J. Biol. Chem. 2002, 277, 48410–48417. [Google Scholar] [CrossRef]
- Borralho, P.; Vieira, A.; Freitas, J.; Chaves, P.; Soares, J. Aberrant Gastric Apomucin Expression in Ulcerative Colitis and Associated Neoplasia. J. Crohn’s Colitis 2007, 1, 35–40. [Google Scholar] [CrossRef]
- Boltin, D.; Perets, T.T.; Vilkin, A.; Niv, Y. Mucin Function in Inflammatory Bowel Disease: An Update. J. Clin. Gastroenterol. 2013, 47, 106–111. [Google Scholar] [CrossRef]
- Buisine, M.; Desreumaux, P.; Leteurtre, E.; Copin, M.; Colombel, J.; Porchet, N.; Aubert, J. Mucin Gene Expression in Intestinal Epithelial Cells in Crohn’s Disease. Gut 2001, 49, 544–551. [Google Scholar] [CrossRef]
- Hoffmann, W. TFF2, a MUC6-Binding Lectin Stabilizing the Gastric Mucus Barrier and More. Int. J. Oncol. 2015, 47, 806–816. [Google Scholar] [CrossRef]
- Longman, R.J.; Poulsom, R.; Corfield, A.P.; Warren, B.F.; Wright, N.A.; Thomas, M.G. Alterations in the Composition of the Supramucosal Defense Barrier in Relation to Disease Severity of Ulcerative Colitis. J. Histochem. Cytochem. 2006, 54, 1335–1348. [Google Scholar] [CrossRef]
- Schmitz, S.; Hill, S.; Werling, D.; Allenspach, K. Expression of Trefoil Factor Genes in the Duodenum and Colon of Dogs with Inflammatory Bowel Disease and Healthy Dogs. Vet. Immunol. Immunopathol. 2013, 151, 168–172. [Google Scholar] [CrossRef]
- Shaoul, R.; Okada, Y.; Cutz, E.; Marcon, M.A. Colonic Expression of MUC2, MUC5AC, and TFF1 in Inflammatory Bowel Disease in Children. J. Pediatric Gastroenterol. Nutr. 2004, 38, 488–493. [Google Scholar] [CrossRef]
- Lee, Y.B.; Kim, H.J.; Jung, H.Y.; Park, Y.G.; Kim, S.Y.; Cho, B.K.; Cho, D.; Park, H.J. Downregulation of Erythroid Differentiation Regulator 1 as a Novel Marker of Skin Tumors. Int. J. Dermatol. 2014, 53, 723–730. [Google Scholar] [CrossRef] [PubMed]
- Abo, H.; Chassaing, B.; Harusato, A.; Quiros, M.; Brazil, J.C.; Ngo, V.L.; Viennois, E.; Merlin, D.; Gewirtz, A.T.; Nusrat, A. Erythroid Differentiation Regulator-1 Induced by Microbiota in Early Life Drives Intestinal Stem Cell Proliferation and Regeneration. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kim, K.E.; Houh, Y.; Lee, J.; Kim, S.; Cho, D.; Park, H.J. Downregulation of Erythroid Differentiation Regulator 1 (Erdr1) Plays a Critical Role in Psoriasis Pathogenesis. Exp. Dermatol. 2016, 25, 570–572. [Google Scholar] [CrossRef]
- Kim, M.; Kim, K.E.; Jung, H.Y.; Jo, H.; Jeong, S.W.; Lee, J.; Kim, C.H.; Kim, H.; Cho, D.; Park, H.J. Recombinant Erythroid Differentiation Regulator 1 Inhibits Both Inflammation and Angiogenesis in a Mouse Model of Rosacea. Exp. Dermatol. 2015, 24, 680–685. [Google Scholar] [CrossRef]
- Malíčková, K.; Ďuricová, D.; Bortlík, M.; Hrušková, Z.; Svobodová, B.; Machková, N.; Komárek, V.; Fučíková, T.; Janatková, I.; Zima, T. Impaired Deoxyribonuclease I Activity in Patients with Inflammatory Bowel Diseases. Autoimmune Dis. 2011, 2011, 945861. [Google Scholar] [CrossRef] [PubMed]
- Nakashima, Y.; Yasuda, T.; Takeshita, H.; Nakajima, T.; Hosomi, O.; Mori, S.; Kishi, K. Molecular, Biochemical and Immunological Studies of Hen Pancreatic Deoxyribonuclease I. Int. J. Biochem. Cell Biol. 1999, 31, 1315–1326. [Google Scholar] [CrossRef]
- Braegger, C.; Nicholls, S.; Murch, S.; MacDonald, T.; Stephens, S. Tumour Necrosis Factor Alpha in Stool as a Marker of Intestinal Inflammation. Lancet 1992, 339, 89–91. [Google Scholar] [CrossRef]
- Derkx, B.; Taminiau, J.; Radema, S.; Stronkhorst, A.; Wortel, C. Tumour-Necrosis-Factor Antibody Treatment in Crohn’s Disease. Lancet (Br. Ed.) 1993, 342, 173–174. [Google Scholar] [CrossRef]
- Murch, S.; Lamkin, V.; Savage, M.; Walker-Smith, J.; MacDonald, T. Serum Concentrations of Tumour Necrosis Factor Alpha in Childhood Chronic Inflammatory Bowel Disease. Gut 1991, 32, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Enss, M.-L.; Cornberg, M.; Wagner, S.; Gebert, A.; Henrichs, M.; Eisenblätter, R.; Beil, W.; Kownatzki, R.; Hedrich, H. Proinflammatory Cytokines Trigger MUC Gene Expression and Mucin Release in the Intestinal Cancer Cell Line LS180. Inflamm. Res. 2000, 49, 162–169. [Google Scholar] [CrossRef]
- Sadler, A.J.; Williams, B.R. Interferon-Inducible Antiviral Effectors. Nat. Rev. Immunol. 2008, 8, 559–568. [Google Scholar] [CrossRef]
- González-Navajas, J.M.; Lee, J.; David, M.; Raz, E. Immunomodulatory Functions of Type I Interferons. Nat. Rev. Immunol. 2012, 12, 125–135. [Google Scholar] [CrossRef]
- Katakura, K.; Lee, J.; Rachmilewitz, D.; Li, G.; Eckmann, L.; Raz, E. Toll-Like Receptor 9–Induced Type I IFN Protects Mice from Experimental Colitis. J. Clin. Investig. 2005, 115, 695–702. [Google Scholar] [CrossRef]
- Rossi, C.P.; Hanauer, S.B.; Tomasevic, R.; Hunter, J.O.; Shafran, I.; Graffner, H. Interferon Beta-1a for the Maintenance of Remission in Patients with Crohn’s Disease: Results of a Phase II Dose-Finding Study. BMC Gastroenterol. 2009, 9, 22. [Google Scholar]
- Bank, S.; Julsgaard, M.; Abed, O.K.; Burisch, J.; Broder Brodersen, J.; Pedersen, N.K.; Gouliaev, A.; Ajan, R.; Nytoft Rasmussen, D.; Honore Grauslund, C. Polymorphisms in the NF kB, TNF-Alpha, IL-1Beta, and IL-18 Pathways are Associated with Response to Anti-TNF Therapy in Danish Patients with Inflammatory Bowel Disease. Aliment. Pharmacol. Ther. 2019, 49, 890–903. [Google Scholar] [CrossRef]
- Gao, W.; Wang, C.; Yu, L.; Sheng, T.; Wu, Z.; Wang, X.; Zhang, D.; Lin, Y.; Gong, Y. Chlorogenic Acid Attenuates Dextran Sodium Sulfate-Induced Ulcerative Colitis in Mice Through MAPK/ERK/JNK Pathway. BioMed Res. Int. 2019, 2019, 6769789. [Google Scholar] [CrossRef]
- Gao, W.; Zhang, L.; Wang, X.; Yu, L.; Wang, C.; Gong, Y. The Combination of Indirubin and Isatin Attenuates Dextran Sodium Sulfate Induced Ulcerative Colitis in Mice. Biochem. Cell Biol. 2018, 96, 636–645. [Google Scholar] [CrossRef]
- Schäfer, T.; Scheuer, C.; Roemer, K.; Menger, M.; Vollmar, B. Inhibition of p53 Protects Liver Tissue Against Endotoxin-Induced Apoptotic and Mecrotic Cell Death. FASEB J. 2003, 17, 660–667. [Google Scholar] [CrossRef]
- Muñoz-Fontela, C.; Macip, S.; Martínez-Sobrido, L.; Brown, L.; Ashour, J.; García-Sastre, A.; Lee, S.W.; Aaronson, S.A. Transcriptional Role of p53 in Interferon-Mediated Antiviral Immunity. J. Exp. Med. 2008, 205, 1929–1938. [Google Scholar] [CrossRef]
- Huang, K.; Chen, L.; Zhang, J.; Wu, Z.; Lan, L.; Wang, L.; Lu, B.; Liu, Y. Elevated p53 Expression Levels Correlate with Tumor Progression and Poor Prognosis in Patients Exhibiting Esophageal Squamous Cell Carcinoma. Oncol. Lett. 2014, 8, 1441–1446. [Google Scholar] [CrossRef]
- Lu, X.; Yu, Y.; Tan, S. P53 Expression in Patients with Ulcerative Colitis-Associated with Dysplasia and Carcinoma: A Systematic Meta-Analysis. BMC Gastroenterol. 2017, 17, 111. [Google Scholar] [CrossRef] [PubMed]
- Lowe, J.M.; Menendez, D.; Bushel, P.R.; Shatz, M.; Kirk, E.L.; Troester, M.A.; Garantziotis, S.; Fessler, M.B.; Resnick, M.A. P53 and NF-κB Coregulate Proinflammatory Gene Responses in Human Macrophages. Cancer Res. 2014, 74, 2182–2192. [Google Scholar] [CrossRef] [PubMed]
- Gogna, R.; Madan, E.; Keppler, B.; Pati, U. Gallium Compound GaQ3-Induced Ca2+ Signalling Triggers p53-Dependent and-Independent Apoptosis in Cancer Cells. Br. J. Pharmacol. 2012, 166, 617–636. [Google Scholar] [CrossRef] [PubMed]
- Chang, H.; Qi, C.; Yi, Q.-L.; Reece, D.; Stewart, A.K. P53 Gene Deletion Detected by Fluorescence in Situ Hybridization is an Adverse Prognostic Factor for Patients with Multiple Myeloma Following Autologous Stem Cell Transplantation. Blood 2005, 105, 358–360. [Google Scholar] [CrossRef]
- Xiao, D.; Yun, Z.; Wu, T.; Zhang, J.; Shao, J. Helicobacter Pylori Infection Associated MiR-451 Suppressed the Proliferation of Gastric Cancer by Targeting CASC4. Int. J. Clin. Exp. Pathol. 2016, 9, 12390–12398. [Google Scholar]
- Redondo, M.; Villar, E.; Torres-Munoz, J.; Tellez, T.; Morell, M.; Petito, C.K. Overexpression of Clusterin in Human Breast Carcinoma. Am. J. Pathol. 2000, 157, 393–399. [Google Scholar] [CrossRef]
- Pucci, S.; Bonanno, E.; Pichiorri, F.; Angeloni, C.; Spagnoli, L.G. Modulation of Different Clusterin Isoforms in Human Colon Tumorigenesis. Oncogene 2004, 23, 2298–2304. [Google Scholar] [CrossRef]
- Liu, Y.; Liao, X.-W.; Qin, Y.-Z.; Mo, X.-W.; Luo, S.-S. Identification of F5 as a Prognostic Biomarker in Patients with Gastric Cancer. BioMed Res. Int. 2020, 2020, 9280841. [Google Scholar] [CrossRef]
- Lu, S.H.; Yuan, R.H.; Chen, Y.L.; Hsu, H.C.; Jeng, Y.M. Annexin A 10 is an Immunohistochemical Marker for Adenocarcinoma of the Upper Gastrointestinal Tract and Pancreatobiliary System. Histopathology 2013, 63, 640–648. [Google Scholar] [PubMed]
- Tran, D.N.; Jung, E.-M.; Ahn, C.; Lee, J.-H.; Yoo, Y.-M.; Jeung, E.-B. Effects of Bisphenol A and 4-Tert-Octylphenol on Embryo Implantation Failure in Mouse. Int. J. Environ. Res. Public Health 2018, 15, 1614. [Google Scholar] [CrossRef] [PubMed]
- Tran, D.N.; Jung, E.-M.; Yoo, Y.-M.; Ahn, C.; Kang, H.Y.; Choi, K.-C.; Hyun, S.-H.; Dang, V.H.; Pham, T.N.; Jeung, E.-B. Depletion of Follicles Accelerated by Combined Exposure to Phthalates and 4-Vinylcyclohexene Diepoxide, Leading to Premature Ovarian Failure. Rats Reprod. Toxicol. 2018, 80, 60–67. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tran, D.N.; Go, S.M.; Park, S.-M.; Jung, E.-M.; Jeung, E.-B. Loss of Nckx3 Exacerbates Experimental DSS-Induced Colitis in Mice through p53/NF-κB Pathway. Int. J. Mol. Sci. 2021, 22, 2645. https://doi.org/10.3390/ijms22052645
Tran DN, Go SM, Park S-M, Jung E-M, Jeung E-B. Loss of Nckx3 Exacerbates Experimental DSS-Induced Colitis in Mice through p53/NF-κB Pathway. International Journal of Molecular Sciences. 2021; 22(5):2645. https://doi.org/10.3390/ijms22052645
Chicago/Turabian StyleTran, Dinh Nam, Seon Myeong Go, Seon-Mi Park, Eui-Man Jung, and Eui-Bae Jeung. 2021. "Loss of Nckx3 Exacerbates Experimental DSS-Induced Colitis in Mice through p53/NF-κB Pathway" International Journal of Molecular Sciences 22, no. 5: 2645. https://doi.org/10.3390/ijms22052645
APA StyleTran, D. N., Go, S. M., Park, S.-M., Jung, E.-M., & Jeung, E.-B. (2021). Loss of Nckx3 Exacerbates Experimental DSS-Induced Colitis in Mice through p53/NF-κB Pathway. International Journal of Molecular Sciences, 22(5), 2645. https://doi.org/10.3390/ijms22052645






