Paving the Way for Fertilization: The Role of the Transmitting Tract
Abstract
1. Introduction
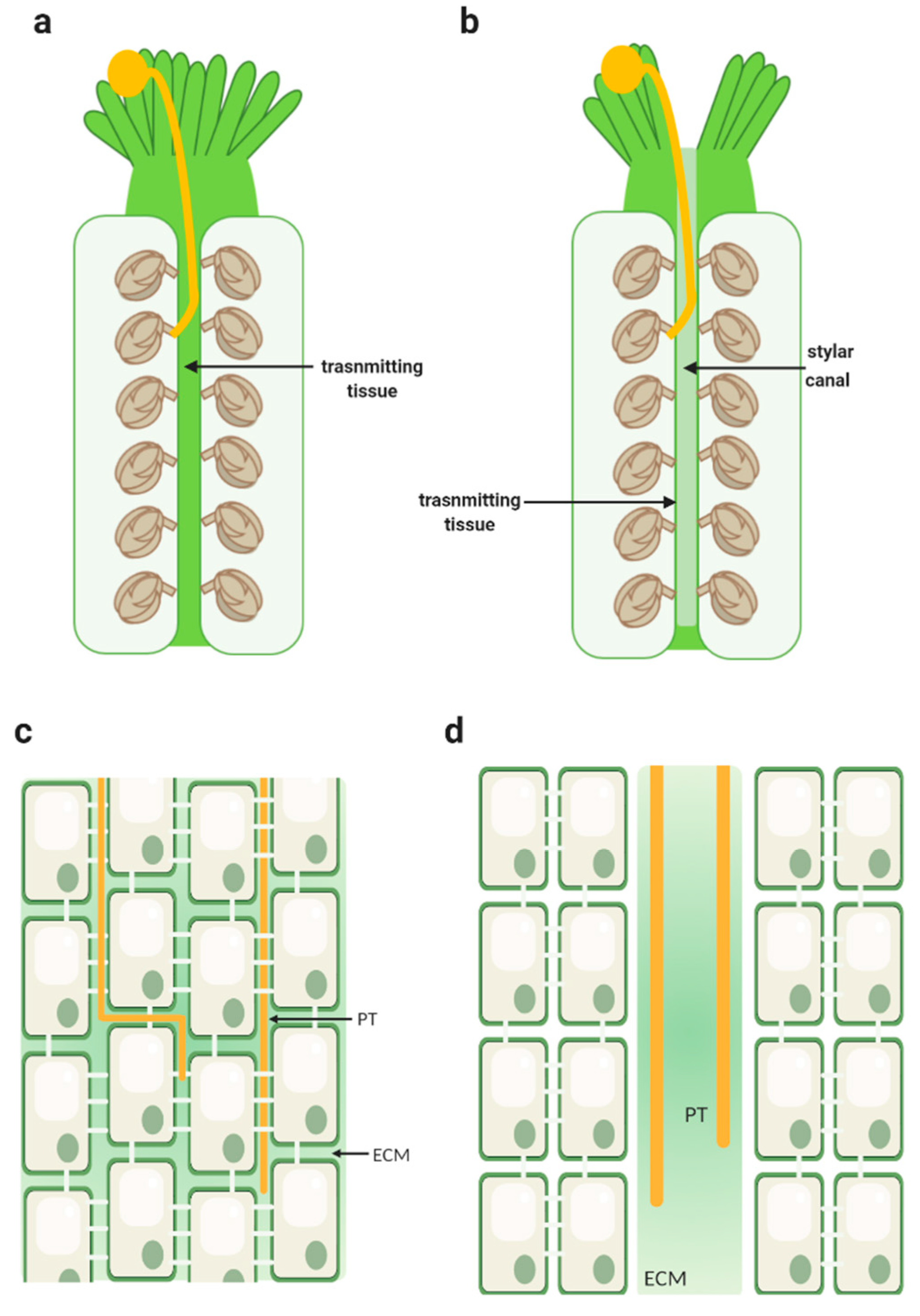
2. The Transmitting Tract
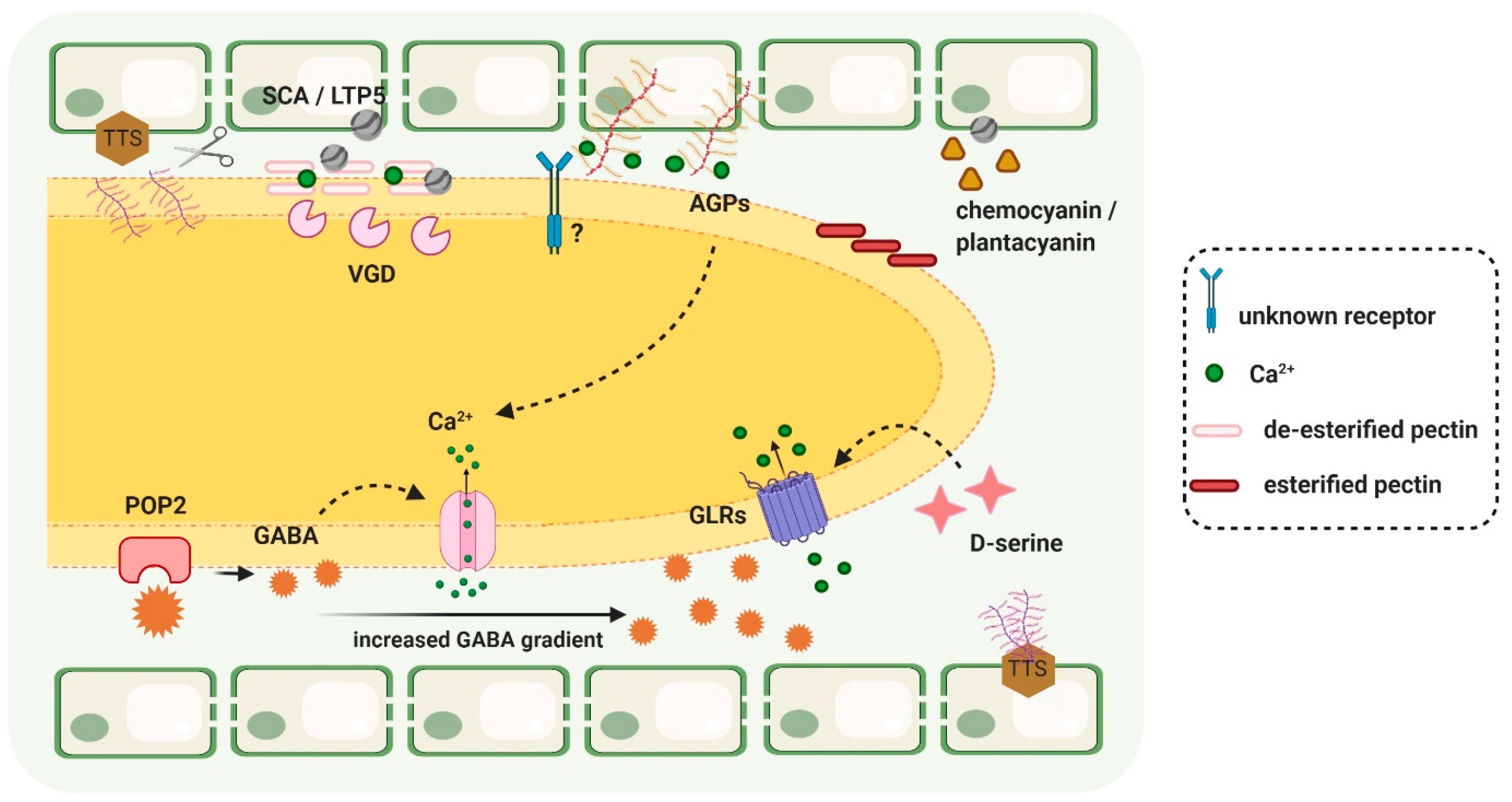
3. Key to the Highway: Pollen–Pistil Interactions in the TT
4. Concluding Remarks
Author Contributions
Funding
Conflicts of Interest
References
- Russell, S.D. Double Fertilization. Adv. Appl. Microbiol. 1992, 140, 357–388. [Google Scholar] [CrossRef]
- Yadegari, R.; Drews, G. Female gametophyte development. Plant Cell 2004, 16, S133–S141. [Google Scholar] [CrossRef] [PubMed]
- Palanivelu, R.; Tsukamoto, T. Pathfinding in angiosperm reproduction: Pollen tube guidance by pistils ensures successful double fertilization. Wiley Interdiscip. Rev. Dev. Biol. 2012, 1, 96–113. [Google Scholar] [CrossRef]
- Beale, K.M.; Johnson, M.A. Speed dating, rejection, and finding the perfect mate: Advice from flow-ering plants. Curr. Opin. Plant Biol. 2013, 16, 590–597. [Google Scholar] [CrossRef]
- Dresselhaus, T.; Franklin-Tong, N. Male–Female Crosstalk during Pollen Germination, Tube Growth and Guidance, and Double Fertilization. Mol. Plant 2013, 6, 1018–1036. [Google Scholar] [CrossRef]
- Pereira, A.M.; Masiero, S.; Nobre, M.S.; Costa, M.L.; Solís, M.-T.; Testillano, P.S.; Sprunck, S.; Coimbra, S. Differential expression patterns of arabinogalactan proteins in Arabidopsis thaliana re-productive tissues. J. Exp. Bot. 2014, 65, 5459–5471. [Google Scholar] [CrossRef]
- Pereira, A.M.; Lopes, A.L.; Coimbra, S.V. Arabinogalactan Proteins as Interactors along the Crosstalk between the Pollen Tube and the Female Tissues. Front. Plant Sci. 2016, 7, 1895. [Google Scholar] [CrossRef] [PubMed]
- Sprunck, S. Twice the fun, double the trouble: Gamete interactions in flowering plants. Curr. Opin. Plant Biol. 2020, 53, 106–116. [Google Scholar] [CrossRef]
- Higashiyama, T.; Takeuchi, H. The mechanism and key molecules involved in pollen tube guid-ance. Annu. Rev. Plant Biol. 2015, 66, 393–413. [Google Scholar] [CrossRef]
- Mizuta, Y.; Higashiyama, T. Chemical signaling for pollen tube guidance at a glance. J. Cell Sci. 2018, 131, jcs208447. [Google Scholar] [CrossRef]
- Sage, T.L.; Hristova-Sarkovski, K.; Koehl, V.; Lyew, J.; Pontieri, V.; Bernhardt, P.; Weston, P.; Bagha, S.; Chiu, G. Transmitting tissue architecture in basal-relictual angiosperms: Implications for transmitting tissue origins. Am. J. Bot. 2009, 96, 183–206. [Google Scholar] [CrossRef]
- Sassen, M.M.A. The stylar transmitting tissue. Acta Bot. Neerl. 1974, 23, 99–108. [Google Scholar] [CrossRef]
- Erbar, C. Pollen Tube Transmitting Tissue: Place of Competition of Male Gametophytes. Int. J. Plant Sci. 2003, 164, S265–S277. [Google Scholar] [CrossRef]
- Mascarenhas, J.P. The biochemistry of angiosperm pollen development. Bot. Rev. 1975, 41, 259–314. [Google Scholar] [CrossRef]
- Mascarenhas, J.P. Molecular mechanisms of pollen tube growth and differentiation. Plant Cell 1993, 5, 1303. [Google Scholar] [CrossRef]
- Heslopharrison, J. Pollen Germination and Pollen-Tube Growth. Adv. Appl. Microbiol. 1987, 107, 1–78. [Google Scholar] [CrossRef]
- Johnson, M.A.; Preuss, D. Plotting a course: Multiple signals guide pollen tubes to their targets. Dev. Cell 2002, 2, 273–281. [Google Scholar] [CrossRef]
- Johnson, M.A.; Lord, E. Extracellular guidance cues and intracellular signaling pathways that di-rect pollen tube growth. In The Pollen Tube; Springer: Berlin/Heidelberg, Germany, 2006; pp. 223–242. [Google Scholar]
- Crawford, B.C.; Yanofsky, M.F. The Formation and Function of the Female Reproductive Tract in Flowering Plants. Curr. Biol. 2008, 18, R972–R978. [Google Scholar] [CrossRef]
- Bowman, J.L.; Baum, S.F.; Eshed, Y.; Putterill, J.; Alvarez, J. 4 Molecular Genetics of Gynoecium Development in Arabidopsis. Protein Kinases Dev. Dis. 1999, 45, 155–205. [Google Scholar] [CrossRef]
- Alvarez, J.; Smyth, D.R. Crabs claw and spatula genes regulate growth and pattern for-mation during gynoecium development in Arabidopsis thaliana. Int. J. Plant Sci. 2002, 163, 17–41. [Google Scholar] [CrossRef]
- Reyes-Olalde, J.I.; Zuñiga-Mayo, V.M.; Montes, R.A.C.; Marsch-Martínez, N.; De Folter, S. Inside the gynoecium: At the carpel margin. Trends Plant Sci. 2013, 18, 644–655. [Google Scholar] [CrossRef]
- Ciampolini, F.; Shivanna, K.R.; Cresti, M. Organization of the Stigma and Transmitting Tissue of Rice, Oryza sativa (L.). Plant Biol. 2001, 3, 149–155. [Google Scholar] [CrossRef]
- Wang, H.; Wu, H.-M.; Cheung, A.Y. Pollination induces mRNA poly(A) tail-shortening and cell deterioration in flower transmitting tissue. Plant J. 1996, 9, 715–727. [Google Scholar] [CrossRef] [PubMed]
- Crawford, B.C.; Ditta, G.; Yanofsky, M.F. The NTT gene is required for transmitting-tract devel-opment in carpels of Arabidopsis thaliana. Curr. Biol. 2007, 17, 1101–1108. [Google Scholar] [CrossRef] [PubMed]
- A Sessions, R.; Zambryski, P.C. Arabidopsis gynoecium structure in the wild and in ettin mutants. Development 1995, 121, 1519–1532. [Google Scholar] [PubMed]
- Kuusk, S.; Sohlberg, J.J.; Magnus Eklund, D.; Sundberg, E.J. Functionally redundant SHI family genes regulate Arabidopsis gynoecium development in a dose-dependent manner. Plant J. 2006, 47, 99–111. [Google Scholar] [CrossRef]
- Alvarez, J.; Smyth, D.R. Crabs claw and spatula, two arabidopsis genes that control carpel development in parallel with agamous. Development 1999, 126, 2377–2386. [Google Scholar]
- Azhakanandam, S.; Nole-Wilson, S.; Bao, F.; Franks, R.G. Seuss and aintegumenta Mediate Patterning and Ovule Initiation during Gynoecium Medial Domain Development. Plant Physiol. 2008, 146, 1165–1181. [Google Scholar] [CrossRef]
- Groszmann, M.; Paicu, T.; Alvarez, J.P.; Swain, S.M.; Smyth, D.R. Spatula and alcatraz, are partially redundant, functionally diverging bHLH genes required for Arabidopsis gynoecium and fruit development. Plant J. 2011, 68, 816–829. [Google Scholar] [CrossRef]
- Kay, P.; Groszmann, M.; Ross, J.J.; Parish, R.W.; Swain, S.M. Modifications of a conserved regulatory network involving INDEHISCENT controls multiple aspects of reproductive tissue development in Arabidopsis. New Phytol. 2012, 197, 73–87. [Google Scholar] [CrossRef] [PubMed]
- Lennon, K.A.; Roy, S.; Hepler, P.K.; Lord, E.J. The structure of the transmitting tissue of Ara-bidopsis thaliana (L.) and the path of pollen tube growth. Sex. Plant Reprod. 1998, 11, 49–59. [Google Scholar] [CrossRef]
- Gremski, K.; Ditta, G.; Yanofsky, M.F. The HECATE genes regulate female reproductive tract de-velopment in Arabidopsis thaliana. Developtment 2007, 134, 3593–3601. [Google Scholar]
- Crawford, B.C.; Yanofsky, M.F. Half filled promotes reproductive tract development and ferti-lization efficiency in Arabidopsis thaliana. Development 2011, 138, 2999–3009. [Google Scholar] [CrossRef] [PubMed]
- Di Marzo, M.; Roig-Villanova, I.; Zanchetti, E.; Caselli, F.; Gregis, V.; Bardetti, P.; Chiara, M.; Guazzotti, A.; Caporali, E.; Mendes, M.A.; et al. MADS-Box and bHLH Transcription Factors Co-ordinate Transmitting Tract Development in Arabidopsis thaliana. Front. Plant Sci. 2020, 11, 526. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Ubaldo, H.; Lozano-Sotomayor, P.; Ezquer, I.; Di Marzo, M.; Montes, R.A.C.; Gómez-Felipe, A.; Pablo-Villa, J.; Diaz-Ramirez, D.; Ballester, P.; Ferrándiz, C. New roles of no transmitting tract and seedstick during medial domain development in Arabidopsis fruits. Development 2019, 146. [Google Scholar] [CrossRef]
- Di Marzo, M.; Herrera-Ubaldo, H.; Caporali, E.; Novák, O.; Strnad, M.; Balanzà, V.; Ezquer, I.; Mendes, M.A.; de Folter, S.; Colombo, L. Seedstick controls Arabidopsis fruit size by regulat-ing cytokinin levels and fruitfull. Cell. Rep. 2020, 30, 2846–2857. [Google Scholar] [CrossRef]
- Cerbantez-Bueno, V.E.; Zúñiga-Mayo, V.M.; Reyes-Olalde, J.I.; Lozano-Sotomayor, P.; Herrera-Ubaldo, H.; Marsch-Martinez, N.; De Folter, S. Redundant and Non-redundant Functions of the AHK Cytokinin Receptors During Gynoecium Development. Front. Plant. Sci. 2020, 11, 568277. [Google Scholar] [CrossRef]
- Higashiyama, T.; Kuroiwa, H.; Kawano, S.; Kuroiwa, T.; Prymakowska-Bosak, M.; Przewloka, M.R.; Ślusarczyk, J.; Kuraś, M.; Lichota, J.; Kiliańczyk, B.; et al. Guidance in Vitro of the Pollen Tube to the Naked Embryo Sac of Torenia fournieri. Plant. Cell 1998, 10, 2019–2031. [Google Scholar] [CrossRef]
- Palanivelu, R.; Preuss, D. Distinct short-range ovule signals attract or repel Arabidopsis thaliana pollen tubes in vitro. BMC Plant. Biol. 2006, 6, 7. [Google Scholar] [CrossRef] [PubMed]
- Zhong, S.; Qu, L.-J. Peptide/receptor-like kinase-mediated signaling involved in male–female in-teractions. Curr. Opin. Plant Biol. 2019, 51, 7–14. [Google Scholar] [CrossRef]
- Qin, Y.; Leydon, A.R.; Manziello, A.; Pandey, R.; Mount, D.; Denic, S.; Vasic, B.; Johnson, M.A.; Palanivelu, R. Penetration of the Stigma and Style Elicits a Novel Transcriptome in Pollen Tubes, Pointing to Genes Critical for Growth in a Pistil. PLoS Genet. 2009, 5, e1000621. [Google Scholar] [CrossRef] [PubMed]
- Tung, C.-W.; Dwyer, K.G.; Nasrallah, M.E.; Nasrallah, J.B. Genome-Wide Identification of Genes Expressed in Arabidopsis Pistils Specifically along the Path of Pollen Tube Growth. Plant. Physiol. 2005, 138, 977–989. [Google Scholar] [CrossRef] [PubMed]
- Mizukami, A.G.; Inatsugi, R.; Jiao, J.; Kotake, T.; Kuwata, K.; Ootani, K.; Okuda, S.; Sankaranarayanan, S.; Sato, Y.; Maruyama, D.; et al. The AMOR Arabinogalactan Sugar Chain Induces Pollen-Tube Competency to Respond to Ovular Guidance. Curr. Biol. 2016, 26, 1091–1097. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.; Hicks, G. Transmitting tissue in the pistil of tobacco: Light and electron microscopic obser-vations. Planta 1976, 131, 187–200. [Google Scholar] [CrossRef]
- Kroh, M.; Van Bakel, C. Incorporation of label into the intercellular substance of stylar transmit-ting tissue from Petunia pistils labeled with tritiated myo-inositol. An electronmicroscopic autora-diographic study. Acta Bot. Neerl. 1973, 22, 106–111. [Google Scholar] [CrossRef]
- Lord, E.; Sanders, L. Roles for the extracellular matrix in plant development and pollination: A special case of cell movement in plants. Dev. Biol. 1992, 153, 16–28. [Google Scholar] [CrossRef]
- Fujii, S.; Kubo, K.-I.; Takayama, S. Non-self- and self-recognition models in plant self-incompatibility. Nat. Plants 2016, 2, 16130. [Google Scholar] [CrossRef] [PubMed]
- Johnson, M.A.; Harper, J.F.; Palanivelu, R. A fruitful journey: Pollen tube navigation from germination to fertilization. Annu. Rev. Plant Biol. 2019, 70, 809–837. [Google Scholar] [CrossRef]
- Cheung, A.Y.; Wang, H.; Wu, H.-M. A floral transmitting tissue-specific glycoprotein attracts pollen tubes and stimulates their growth. Cell 1995, 82, 383–393. [Google Scholar] [CrossRef]
- Wu, H.-M.; Wang, H.; Cheung, A.Y. A pollen tube growth stimulatory glycoprotein is deglycosylated by pollen tubes and displays a glycosylation gradient in the flower. Cell 1995, 82, 395–403. [Google Scholar] [CrossRef]
- Wu, H.M.; Wong, E.; Ogdahl, J.; Cheung, A.Y. A pollen tube growth-promoting arabinogalactan pro-tein from Nicotiana alata is similar to the tobacco TTS protein. Plant J. 2000, 22, 165–176. [Google Scholar] [CrossRef]
- Ma, Y.; Yan, C.; Li, H.; Wu, W.; Liu, Y.; Wang, Y.; Chen, Q.; Ma, H. Bioinformatics Prediction and Evolution Analysis of Arabinogalactan Proteins in the Plant Kingdom. Front. Plant. Sci. 2017, 8, 66. [Google Scholar] [CrossRef]
- Gao, M.; Showalter, A.M. Yariv reagent treatment induces programmed cell death in Arabidopsis cell cultures and implicates arabinogalactan protein involvement. Plant J. 1999, 19, 321–331. [Google Scholar] [CrossRef]
- Lamport, D.T.A.; Varnai, P.; Seal, C.E. Back to the future with the AGP–Ca2+ flux capacitor. Ann. Bot. 2014, 114, 1069–1085. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Hernandez, F.; Tryfona, T.; Rizza, A.; Xiaolan, L.Y.; Harris, M.O.; Webb, A.A.; Kotake, T.; Dupree, P.J. Calcium binding by arabinogalactan polysaccharides is important for normal plant de-velopment. Plant Cell 2020, 32, 3346–3369. [Google Scholar] [CrossRef] [PubMed]
- Lord, E.; Walling, L.; Jauh, G. Cell Adhesion in Plants and Its Role in pollination. Membranes: Specialized Functions in Plants; BIOS Scientific Publishers: Oxford, UK, 1996; pp. 21–37. [Google Scholar]
- Mollet, J.-C.; Park, S.-Y.; Nothnagel, E.A.; Lord, E.M. A lily stylar pectin is necessary for pollen tube adhesion to an in vitro stylar matrix. Plant Cell 2000, 12, 1737–1749. [Google Scholar] [CrossRef] [PubMed]
- Park, S.-Y.; Jauh, G.-Y.; Mollet, J.-C.; Eckard, K.J.; Nothnagel, E.A.; Walling, L.L.; Lord, E.M. A lipid transfer–like protein is necessary for lily pollen tube adhesion to an in vitro stylar matrix. Plant Cell 2000, 12, 151–163. [Google Scholar] [PubMed]
- Kim, S.; Mollet, J.-C.; Dong, J.; Zhang, K.; Park, S.-Y.; Lord, E.M. Chemocyanin, a small basic protein from the lily stigma, induces pollen tube chemotropism. Proc. Natl. Acad. Sci. USA 2003, 100, 16125–16130. [Google Scholar] [CrossRef]
- Dong, J.; Kim, S.T.; Lord, E.M. Plantacyanin Plays a Role in Reproduction in Arabidopsis. Plant. Physiol. 2005, 138, 778–789. [Google Scholar] [CrossRef] [PubMed]
- Chae, K.; Kieslich, C.A.; Morikis, D.; Kim, S.-C.; Lord, E.M. A gain-of-function mutation of Ara-bidopsis lipid transfer protein 5 disturbs pollen tube tip growth and fertilization. Plant Cell 2009, 21, 3902–3914. [Google Scholar] [CrossRef]
- Jiang, L.; Yang, S.-L.; Xie, L.-F.; San Puah, C.; Zhang, X.-Q.; Yang, W.-C.; Sundaresan, V.; Ye, D.J. VANGUARD1 encodes a pectin methylesterase that enhances pollen tube growth in the Ara-bidopsis style and transmitting tract. Plant Cell 2005, 17, 584–596. [Google Scholar] [CrossRef]
- Goldberg, R.; Morvan, C.; Jauneau, A.; Jarvis, M. Methyl-esterification, de-esterification and gela-tion of pectins in the primary cell wall. In Progress in Biotechnology; Elsevier: Amsterdam, The Netherlands, 1996; Volume 14, pp. 151–172. [Google Scholar]
- Lu, Y.; Lauter, A.N.M.; Makkena, S.; Scott, M.P.; Evans, M.M.S. Insights into the molecular control of cross-incompatibility in Zea mays. Plant. Reprod. 2020, 33, 117–128. [Google Scholar] [CrossRef] [PubMed]
- Palanivelu, R.; Brass, L.; Edlund, A.F.; Preuss, D. Pollen Tube Growth and Guidance Is Regulated by POP2, an Arabidopsis Gene that Controls GABA Levels. Cell 2003, 114, 47–59. [Google Scholar] [CrossRef]
- Yu, G.-H.; Zou, J.; Feng, J.; Peng, X.-B.; Wu, J.-Y.; Wu, Y.-L.; Palanivelu, R.; Sun, M.-X.J. Exoge-nous γ-aminobutyric acid (GABA) affects pollen tube growth via modulating putative Ca2+-permeable membrane channels and is coupled to negative regulation on glutamate decarboxylase. J. Exp. Bot. 2014, 65, 3235–3248. [Google Scholar] [CrossRef]
- Michard, E. Channels in Pollen 2+ Like Genes Form Ca− Glutamate Receptor. Science 2011, 1201101, 332. [Google Scholar]
- Vogler, F.; Schmalzl, C.; Englhart, M.; Bircheneder, M.; Sprunck, S. Brassinosteroids promote Ar-abidopsis pollen germination and growth. Plant Repro. 2014, 27, 153–167. [Google Scholar] [CrossRef]
- Lu, Y.; Chanroj, S.; Zulkifli, L.; Johnson, M.A.; Uozumi, N.; Cheung, A.; Sze, H. Pollen Tubes Lacking a Pair of K+ Transporters Fail to Target Ovules in Arabidopsis. Plant. Cell 2011, 23, 81–93. [Google Scholar] [CrossRef]
- Okuda, S.; Tsutsui, H.; Shiina, K.; Sprunck, S.; Takeuchi, H.; Yui, R.; Kasahara, R.D.; Hamamura, Y.; Mizukami, A.; Susaki, D.; et al. Defensin-like polypeptide LUREs are pollen tube attractants secreted from synergid cells. Nat. Cell Biol. 2009, 458, 357–361. [Google Scholar] [CrossRef] [PubMed]
- Takeuchi, H.; Higashiyama, T. Tip-localized receptors control pollen tube growth and LURE sens-ing in Arabidopsis. Nature 2016, 531, 245–248. [Google Scholar] [CrossRef] [PubMed]
- Herrera-Ubaldo, H.; de Folter, S. Exploring cell wall composition and modifications during the development of the gynoecium medial domain in Arabidopsis. Front. Plant Sci. 2018, 9, 454. [Google Scholar] [CrossRef]
- Lopes, A.L.; Moreira, D.; Ferreira, M.J.; Pereira, A.M.; Coimbra, S. Insights into secrets along the pollen tube pathway in need to be discovered. J. Exp. Bot. 2019, 70, 2979–2992. [Google Scholar] [CrossRef] [PubMed]
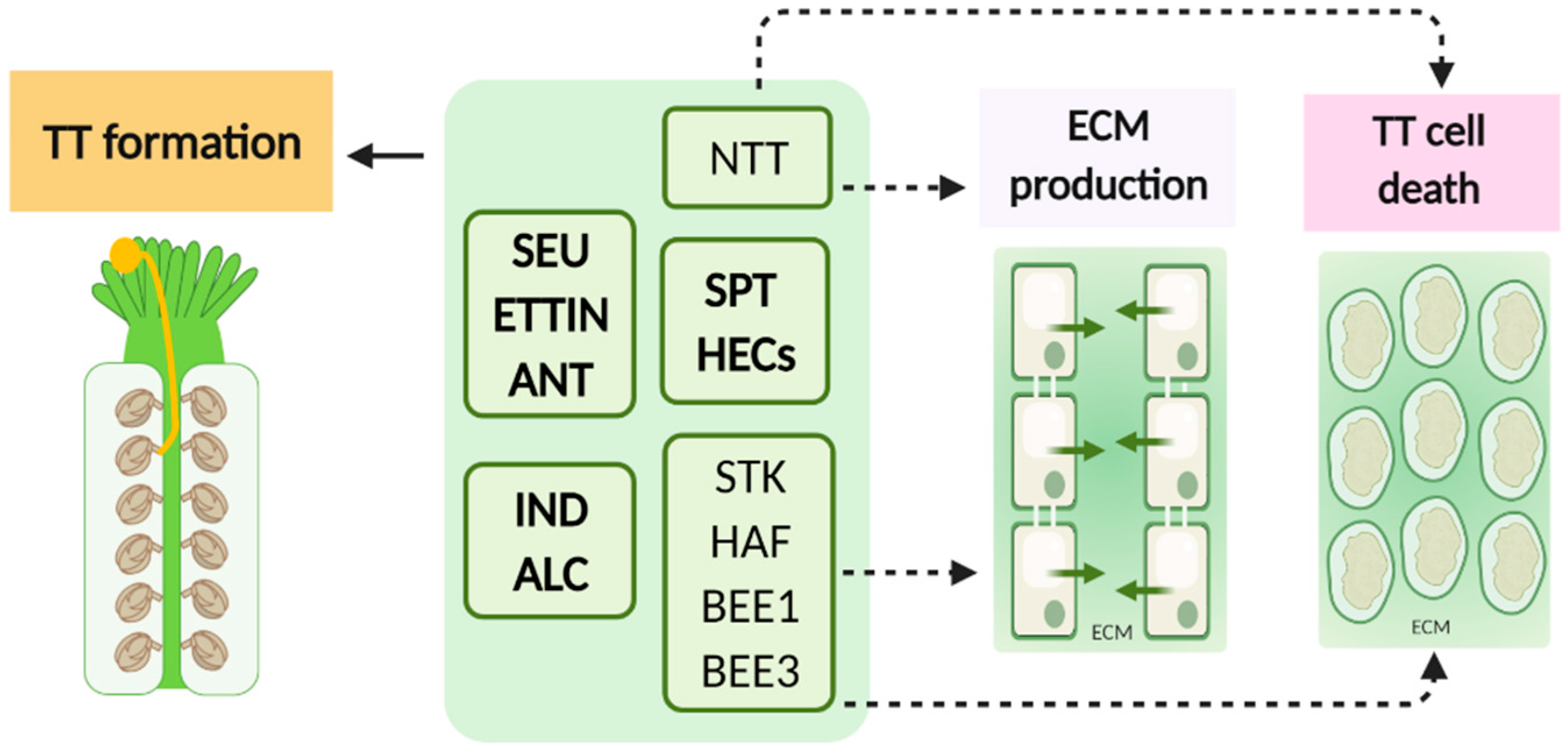
| Proteins | Type | Function | Expression Pattern | Phenotype of loss-of-function mutants | References |
|---|---|---|---|---|---|
| NO TRANSMITING TRACT (NTT) | C2H2/C2HC zinc finger transcription factor | Transmitting tract (TT) development; extracellular matrix (ECM) production and TT programed cell death (PCD) | TT | Non functional TT defective ECM production and defective PCD | [25] |
| ETTIN (ETT) | Auxin response transcription factor | Correct gynoecium formation | Gynoecium; valves; developing ovules | Everted TT develops in valve tissues & on regions with missing valves | [26] |
| SPATULA (SPT) | bHLH (basic helix-loop-helix) transcription factor | Acts redundantly with ALC to control development of carpel margin tissues | Developing carpel margin tissues | Unfused carpels; TT is absent; exhibits defects in post-genital fusion of the septum | [21,28] |
| SEUSS (SEU) | Transcriptional co-regulator | Development of the carpel margin meristem (meristematic structure located on the margins of the fused carpels and gives rise to ovules, septum, and TT) | Carpel margin meristem and ovules | Partial splitting of the gynoecial apex; seu ant double mutants exhibit ovule defects & 50% of the gynoecia do not develop a TT | [29] |
| AINTEGUMENTA (ANT) | Putative transcriptional regulator | Ovule development; TT development | Floral organs primordia & developing ovules. | Fail to produce ovules with integuments & functional female gametophyte; seu ant double mutants exhibit ovule defects & 50% of the gynoecia do not develop a TT | [29] |
| ALCATRAZ (ALC) | myc/bHLH transcription factor-like protein | Acts redundantly with SPT to control development of carpel margin tissues; fruit dehiscence | Developing carpel margin tissues; TT; stigma | Disruption of septum & gynoecium fusion; defects in TT development. Fruit dehiscence impaired | [30] |
| INDEHISCENT (IND) | bHLH DNA-binding superfamily protein | Valve margin development and silique dehiscence. TT development | Valve margins | Disruption of septum and gynoecium fusion. Fruits fail to open on maturity. | [31] |
| HECATE 1, 2 and 3 (HEC1, HEC2, HEC3) | bHLH transcription factors | Modulate post-genital fusion of the septum, and consequently TT development | Developing septum, TT and ovules | Infertility, defects in septum, TT and stigma development and impaired pollen tube growth | [33] |
| HALF FILLED (HAF) / CESTA (CES) | Transcription factor | Acts redundantly with BEE1 & BEE3 to specify reproductive tract tissues | Septum, TT and funiculus | In haf bee1 bee3 ECM formation and PCD fail to occur within the TT; reduced pollen tube growth | [34] |
| BRASSINOSTEROID ENHANCED EXPRESSION1 and 3 (BEE1, BEE3) | bHLH transcription factor | Acts redundantly with HAF to specify reproductive tract tissues | BEE1: stigma & top of the style.BEE3 TT and style.: | In haf bee1 bee3 ECM formation and PCD fail to occur within the TT; reduced pollen tube growth | [34] |
| SEEDSTICK (STK) | MADS-box transcription factor | Ovule identity; normal seed shedding; seed coat development; TT PCD | Septum and ovules | Fail to release seeds; Separation of abscission zone cells fails; fruits are shorter; abnormal positioning of seeds | [36,37] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pereira, A.M.; Moreira, D.; Coimbra, S.; Masiero, S. Paving the Way for Fertilization: The Role of the Transmitting Tract. Int. J. Mol. Sci. 2021, 22, 2603. https://doi.org/10.3390/ijms22052603
Pereira AM, Moreira D, Coimbra S, Masiero S. Paving the Way for Fertilization: The Role of the Transmitting Tract. International Journal of Molecular Sciences. 2021; 22(5):2603. https://doi.org/10.3390/ijms22052603
Chicago/Turabian StylePereira, Ana Marta, Diana Moreira, Sílvia Coimbra, and Simona Masiero. 2021. "Paving the Way for Fertilization: The Role of the Transmitting Tract" International Journal of Molecular Sciences 22, no. 5: 2603. https://doi.org/10.3390/ijms22052603
APA StylePereira, A. M., Moreira, D., Coimbra, S., & Masiero, S. (2021). Paving the Way for Fertilization: The Role of the Transmitting Tract. International Journal of Molecular Sciences, 22(5), 2603. https://doi.org/10.3390/ijms22052603









