Gastric Carcinomas with Stromal B7-H3 Expression Have Lower Intratumoural CD8+ T Cell Density
Abstract
1. Introduction
2. Results
2.1. B7-H3 Expression and Its Association with Clinicopathological Characteristics
2.2. Survival Analysis
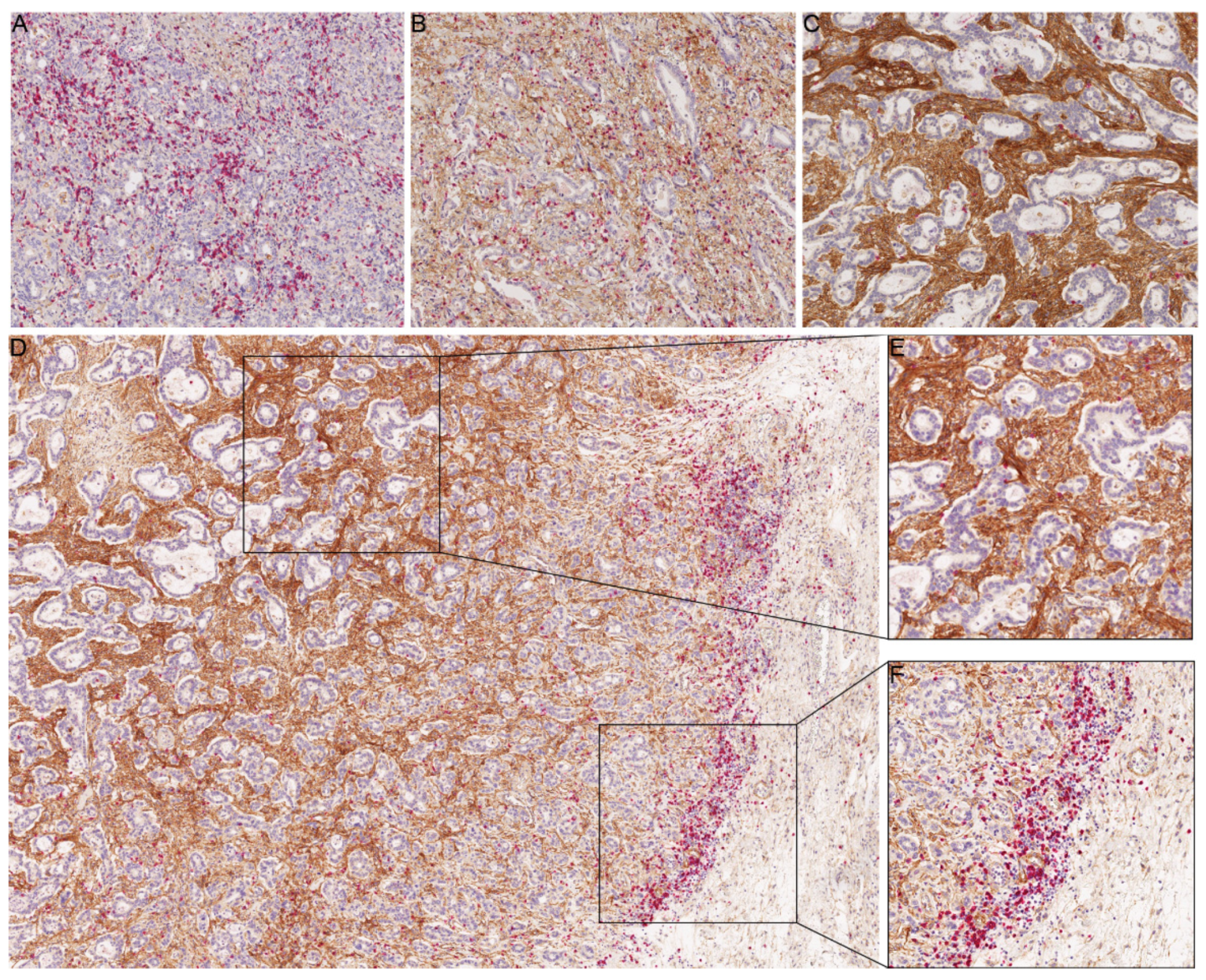
2.3. CD8+ T Cell Infiltration in Relation to B7-H3 Expression
2.4. TCGA Dataset Analysis
3. Discussion
4. Materials and Methods
4.1. Study Population
4.2. Immunohistochemistry
4.3. Evaluation of B7-H3 Immunostaining
4.4. Digital Image Analysis
4.5. Statistical Analysis
4.6. Bioinformatic Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Binnewies, M.; Roberts, E.W.; Kersten, K.; Chan, V.; Fearon, D.F.; Merad, M.; Coussens, L.M.; Gabrilovich, D.I.; Ostrand-Rosenberg, S.; Hedrick, C.C.; et al. Understanding the tumor immune microenvironment (TIME) for effective therapy. Nat. Med. 2018, 24, 541–550. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Cao, M.; Duan, Y.; Bai, H.; Li, X.; Wang, Y. Prognostic role of tumor-infiltrating lymphocytes in gastric cancer: A meta-analysis and experimental validation. Arch. Med. Sci. 2020, 16, 1092–1103. [Google Scholar] [CrossRef] [PubMed]
- Mansuri, N.; Birkman, E.M.; Heuser, V.D.; Lintunen, M.; Ålgars, A.; Sundström, J.; Ristamäki, R.; Lehtinen, L.; Carpén, O. Association of tumor-infiltrating T lymphocytes with intestinal-type gastric cancer molecular subtypes and outcome. Virchows Arch. 2020, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bonaventura, P.; Shekarian, T.; Alcazer, V.; Valladeau-Guilemond, J.; Valsesia-Wittmann, S.; Amigorena, S.; Caux, C.; Depil, S. Cold Tumors: A Therapeutic Challenge for Immunotherapy. Front. Immunol. 2019, 10, 168. [Google Scholar] [CrossRef] [PubMed]
- Castellanos, J.R.; Purvis, I.J.; Labak, C.M.; Guda, M.R.; Tsung, A.J.; Velpula, K.K.; Asuthkar, S. B7-H3 role in the immune landscape of cancer. Am. J. Clin. Exp. Immunol. 2017, 6, 66–75. [Google Scholar] [PubMed]
- Ni, L.; Dong, C. New B7 Family Checkpoints in Human Cancers. Mol. Cancer Ther. 2017, 16, 1203–1211. [Google Scholar] [CrossRef] [PubMed]
- Lee, Y.H.; Martin-Orozco, N.; Zheng, P.; Li, J.; Zhang, P.; Tan, H.; Park, H.J.; Jeong, M.; Chang, S.H.; Kim, B.S.; et al. Inhibition of the B7-H3 immune checkpoint limits tumor growth by enhancing cytotoxic lymphocyte function. Cell Res. 2017, 27, 1034–1045. [Google Scholar] [CrossRef] [PubMed]
- Zhan, S.; Liu, Z.; Zhang, M.; Guo, T.; Quan, Q.; Huang, L.; Guo, L.; Cao, L.; Zhang, X. Overexpression of B7-H3 in α-SMA-Positive Fibroblasts Is Associated With Cancer Progression and Survival in Gastric Adenocarcinomas. Front. Oncol. 2019, 9, 1466. [Google Scholar] [CrossRef] [PubMed]
- Guo, L.; Liu, Z.; Zhang, Y.; Quan, Q.; Huang, L.; Xu, Y.; Cao, L.; Zhang, X. Association of increased B7 protein expression by infiltrating immune cells with progression of gastric carcinogenesis. Medicine 2019, 98, e14663. [Google Scholar] [CrossRef] [PubMed]
- Goldman, M.J.; Craft, B.; Hastie, M.; Repečka, K.; McDade, F.; Kamath, A.; Banerjee, A.; Luo, Y.; Rogers, D.; Brooks, A.N.; et al. Visualizing and interpreting cancer genomics data via the Xena platform. Nat. Biotechnol. 2020, 38, 675–678. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Fu, J.; Zeng, Z.; Cohen, D.; Li, J.; Chen, Q.; Li, B.; Liu, X.S. TIMER2.0 for analysis of tumor-infiltrating immune cells. Nucleic Acids Res. 2020, 48, W509–W514. [Google Scholar] [CrossRef] [PubMed]
- Lee, J.S.; Won, H.S.; Sun, S.; Hong, J.H.; Ko, Y.H. Prognostic role of tumor-infiltrating lymphocytes in gastric cancer: A systematic review and meta-analysis. Medicine 2018, 97, e11769. [Google Scholar] [CrossRef] [PubMed]
- Lin, S.J.; Gagnon-Bartsch, J.A.; Tan, I.B.; Earle, S.; Ruff, L.; Pettinger, K.; Ylstra, B.; van Grieken, N.; Rha, S.Y.; Chung, H.C.; et al. Signatures of tumour immunity distinguish Asian and non-Asian gastric adenocarcinomas. Gut 2015, 64, 1721–1731. [Google Scholar] [CrossRef] [PubMed]
- Heo, Y.J.; Lee, T.; Byeon, S.J.; Kim, E.J.; Shin, H.C.; Kim, B.; Kang, S.Y.; Ha, S.Y.; Kim, K.M. Digital image analysis in pathologist-selected regions of interest predicts survival more accurately than whole-slide analysis: A direct comparison study in 153 gastric carcinomas. J. Pathol. Clin. Res. 2020. [Google Scholar] [CrossRef] [PubMed]
- Derks, S.; de Klerk, L.K.; Xu, X.; Fleitas, T.; Liu, K.X.; Liu, Y.; Dietlein, F.; Margolis, C.; Chiaravalli, A.M.; Da Silva, A.C.; et al. Characterizing diversity in the tumor-immune microenvironment of distinct subclasses of gastroesophageal adenocarcinomas. Ann. Oncol. 2020, 31, 1011–1020. [Google Scholar] [CrossRef] [PubMed]
- Gullo, I.; Oliveira, P.; Athelogou, M.; Gonçalves, G.; Pinto, M.L.; Carvalho, J.; Valente, A.; Pinheiro, H.; Andrade, S.; Almeida, G.M.; et al. New insights into the inflamed tumor immune microenvironment of gastric cancer with lymphoid stroma: From morphology and digital analysis to gene expression. Gastric Cancer 2019, 22, 77–90. [Google Scholar] [CrossRef] [PubMed]
- Chapoval, A.I.; Ni, J.; Lau, J.S.; Wilcox, R.A.; Flies, D.B.; Liu, D.; Dong, H.; Sica, G.L.; Zhu, G.; Tamada, K.; et al. B7-H3: A costimulatory molecule for T cell activation and IFN-gamma production. Nat. Immunol. 2001, 2, 269–274. [Google Scholar] [CrossRef] [PubMed]
- Hashiguchi, M.; Kobori, H.; Ritprajak, P.; Kamimura, Y.; Kozono, H.; Azuma, M. Triggering receptor expressed on myeloid cell-like transcript 2 (TLT-2) is a counter-receptor for B7-H3 and enhances T cell responses. Proc. Natl. Acad. Sci. USA 2008, 105, 10495–10500. [Google Scholar] [CrossRef] [PubMed]
- Leitner, J.; Klauser, C.; Pickl, W.F.; Stöckl, J.; Majdic, O.; Bardet, A.F.; Kreil, D.P.; Dong, C.; Yamazaki, T.; Zlabinger, G.; et al. B7-H3 is a potent inhibitor of human T-cell activation: No evidence for B7-H3 and TREML2 interaction. Eur. J. Immunol. 2009, 39, 1754–1764. [Google Scholar] [CrossRef] [PubMed]
- Hofmeyer, K.A.; Ray, A.; Zang, X. The contrasting role of B7-H3. Proc. Natl. Acad. Sci. USA 2008, 105, 10277–10278. [Google Scholar] [CrossRef] [PubMed]

| Characteristics | Valid/Missing | B7-H3 Low | B7-H3 High | p-Value | |||
|---|---|---|---|---|---|---|---|
| n | (%) | n | (%) | n | (%) | ||
| Total | 96 | (100) | 55 | (57.3) | 41 | (42.7) | |
| Gender | 96/0 | 0.061 2 | |||||
| Male | 57 | (59.4) | 28 | (49.1) | 29 | (50.9) | |
| Female | 39 | (40.6) | 27 | (69.2) | 12 | (30.8) | |
| Age | 96/0 | 0.410 1 | |||||
| <68 years | 50 | (52.1) | 31 | (62.0) | 19 | (38.0) | |
| ≥68 years | 46 | (47.9) | 24 | (52.2) | 22 | (47.8) | |
| Location | 95/1 | 0.003 2 | |||||
| Proximal stomach | 37 | (38.9) | 14 | (37.8) | 23 | (62.2) | |
| Distal stomach | 58 | (61.1) | 40 | (69.0) | 18 | (31.0) | |
| Laurén phenotype | 96/0 | <0.001 2 | |||||
| Intestinal | 48 | (50.0) | 17 | (35.4) | 31 | (64.6) | |
| Diffuse | 27 | (28.1) | 25 | (92.6) | 2 | (7.4) | |
| Mixed | 6 | (6.3) | 2 | (33.3) | 4 | (66.7) | |
| Unclassified | 15 | (15.6) | 11 | (73.3) | 4 | (26.7) | |
| Grade | 90/6 | 0.173 1 | |||||
| G1/G2 | 17 | (18.9) | 7 | (41.2) | 10 | (58.8) | |
| G3/G4 | 73 | (81.1) | 45 | (61.6) | 28 | (38.4) | |
| pT category | 96/0 | 0.013 1 | |||||
| pT1/pT2 | 22 | (22.9) | 18 | (81.8) | 4 | (18.2) | |
| pT3/pT4 | 74 | (77.1) | 37 | (50.0) | 37 | (50.0) | |
| pN category | 96/0 | 1.000 1 | |||||
| pN0 | 32 | (33.3) | 18 | (56.2) | 14 | (43.8) | |
| pN+ | 64 | (66.7) | 37 | (57.8) | 27 | (42.2) | |
| M category | 91/5 | 0.591 1 | |||||
| M0 | 74 | (81.3) | 41 | (55.4) | 33 | (44.6) | |
| M1 | 17 | (18.7) | 11 | (64.7) | 6 | (35.3) | |
| UICC stage | 91/5 | 0.778 1 | |||||
| IA/IB | 16 | (17.6) | 13 | (81.2) | 3 | (18.8) | |
| IIA/IIB | 25 | (27.4) | 10 | (40.0) | 15 | (60.0) | |
| IIIA/IIIB/IIIC | 33 | (36.3) | 18 | (54.5) | 15 | (45.5) | |
| IV | 17 | (18.7) | 11 | (64.7) | 6 | (35.3) | |
| pR status | 89/7 | 0.231 1 | |||||
| pR0 | 82 | (92.1) | 45 | (54.9) | 37 | (45.1) | |
| pR1/pR2 | 7 | (7.9) | 6 | (85.7) | 1 | (14.3) | |
| Overall survival (months) | 0.784 3 | ||||||
| Total/events/censored | 89/64/25 | 51/38/19 | 38/26/12 | ||||
| Median survival | 17.5 | 17.5 | 16.6 | ||||
| 95% C.I. | 10.7–24.3 | 9.0–26.0 | 10.3–22.9 | ||||
| Cause-specific survival (months) | 0.990 3 | ||||||
| Total/events/censored | 82/50/32 | 47/28/19 | 35/22/13 | ||||
| Median survival | 18.4 | 17.5 | 18.4 | ||||
| 95% C.I. | 9.6–27.3 | 6.6–28.5 | 12.8–24.1 | ||||
| n | CD8+ T Cell Density (cells/mm2) | ||||||||
|---|---|---|---|---|---|---|---|---|---|
| Tumour Centre | Invasive Front | ||||||||
| Q1 | Median | Q3 | Range | Q1 | Median | Q3 | Range | ||
| Total | 96 | 33.1 | 112.5 | 214.9 | 0.0–2822.6 | 112.9 | 258.1 | 704.3 | 4.7–3560.1 |
| B7-H3 expression | |||||||||
| B7-H3 low (0/1) | 55 | 41.2 | 157.7 | 328.4 | 0.0–2822.6 | 65.2 | 218.7 | 658.3 | 4.7–3560.1 |
| B7-H3 high (2/3) | 41 | 30.4 | 86.4 | 149.9 | 0.9–1156.0 | 134.0 | 414.9 | 737.6 | 20.6–2781.4 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ulase, D.; Behrens, H.-M.; Krüger, S.; Zeissig, S.; Röcken, C. Gastric Carcinomas with Stromal B7-H3 Expression Have Lower Intratumoural CD8+ T Cell Density. Int. J. Mol. Sci. 2021, 22, 2129. https://doi.org/10.3390/ijms22042129
Ulase D, Behrens H-M, Krüger S, Zeissig S, Röcken C. Gastric Carcinomas with Stromal B7-H3 Expression Have Lower Intratumoural CD8+ T Cell Density. International Journal of Molecular Sciences. 2021; 22(4):2129. https://doi.org/10.3390/ijms22042129
Chicago/Turabian StyleUlase, Dita, Hans-Michael Behrens, Sandra Krüger, Sebastian Zeissig, and Christoph Röcken. 2021. "Gastric Carcinomas with Stromal B7-H3 Expression Have Lower Intratumoural CD8+ T Cell Density" International Journal of Molecular Sciences 22, no. 4: 2129. https://doi.org/10.3390/ijms22042129
APA StyleUlase, D., Behrens, H.-M., Krüger, S., Zeissig, S., & Röcken, C. (2021). Gastric Carcinomas with Stromal B7-H3 Expression Have Lower Intratumoural CD8+ T Cell Density. International Journal of Molecular Sciences, 22(4), 2129. https://doi.org/10.3390/ijms22042129






