Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19 †
Abstract
1. Introduction
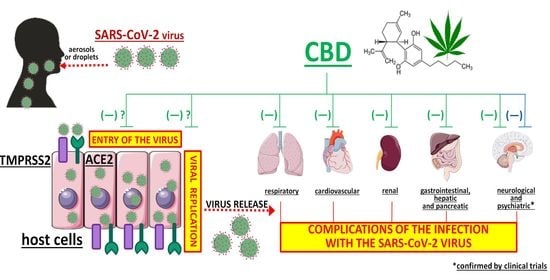
2. Mode of Infection and Symptoms of COVID-19
3. Cannabidiol—Pharmacological Potential and Mechanism of Action
4. Potential Antiviral Activity of CBD
5. Preclinical Studies on the Use of CBD for COVID-19 Treatment
6. Use of CBD for COVID-19 Treatment? Clinical Studies
7. Opportunities, Challenges and Pitfalls of Cannabidiol Use as a COVID-19 Therapy
- Numerous preclinical findings (Table 1) and reviews regarding the potential use of CBD in COVID-19 treatment [14,15,56,57,58,59,60] suggest that CBD has beneficial anti-inflammatory and antioxidative effects, which can be expected to improve the systemic symptoms that are characteristic of SARS-CoV-2 infection. Unfortunately, a comparison of the results of preclinical (Table 1) and clinical (Table 2) studies demonstrates that the favorable preclinical properties may not translate into the clinical setting (or that the appropriate clinical studies have not been conducted; see also reviews [58,61,62]). Above all, there is no confirmation of such beneficial effects of CBD with regard to its effects on respiratory failure. In addition, careful examination of whether the influence of CBD on the immune system could exacerbate viral infection is required (reviews [64,65]). Indeed, viral, fungal infections and pneumonia infections are listed among the side effects of CBD [166,167,168,169]. Moreover, it has to be considered that CBD, which was found to have a pro-oxidative effect in one preclinical study ([83]; Table 1), might even aggravate the feared cytokine storm.
- Anxiolytic and antinociceptive properties of CBD, given alone or together with THC, which have been identified in clinical studies (Table 2), suggest that it may be used as an adjuvant treatment to improve the quality of life of patients with COVID-19 and, even after recovery, may limit post-traumatic stress symptoms. However, well-designed double-blind, placebo-controlled clinical trials regarding the efficacy of CBD against COVID-19-associated panic, anxiety, depression and neurological complications are so far missing [63].
- The decreases in ACE2 and TMPRSS2 protein expression in a human tissue model are extremely interesting but so far this effect has only been shown in an in vitro study by Wang et al. [67]. Even if this mechanism was also found to occur in vivo, certain issues would have to be considered, for example, the mechanism was found for some CBD-rich extracts but did not occur when pure CBD was used. The consequence would be that an extract would need to be administered instead of a pure substance and oral administration would not be possible (for problems associated with topical administration, see below). Next, the question arises about the extent to which ACE2 and TMPRSS2 have to be decreased in order to obtain a robust antiviral effect. Moreover, it is unclear whether a reduction in ACE2 (the importance of which is highlighted in Figure 1) will lead to problems other than the struggle against the virus.
- Inhibition of the replication of SARS-CoV-2 by CBD in a cell line also represents an interesting mechanism, although this effect has only been shown in a study by Raj et al. [68] and transfer to the in vivo situation of the human body is unclear.
8. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| 5-HT1A-R | serotonin receptor type 1A |
| A1A-R | adenosine receptor type A1A |
| A2A-R | adenosine receptor type A2A |
| ACE2 | angiotensin-converting enzyme 2 |
| ACE-I | angiotensin-converting-enzyme inhibitors |
| AGEs | advanced glycation endproducts |
| ALI | acute lung injury |
| ALT | alanine transaminase |
| Ang II | angiotensin II |
| Ang 1–7 | angiotensin 1–7 |
| AST | aspartate transaminase |
| ARDS | acute respiratory distress syndrome |
| BALF | bronchoalveolar lavage fluid |
| CB-R | cannabinoid receptor |
| CB1-R | cannabinoid CB1 receptor |
| CB2-R | cannabinoid CB2 receptor |
| CBD | cannabidiol |
| CBF | cerebral blood flow |
| CCL2, CCL5 | C-C motif chemokine ligand 2 and 5 |
| CINV | chemotherapy-induced nausea and vomiting |
| COPD | chronic obstructive pulmonary disease |
| COVID-19 | coronavirus disease 2019 |
| COX-2 | cyclooxygenase 2 |
| CRP | C-reactive protein |
| CVD | cardiovascular disease |
| d | day(s) |
| DOCA | deoxycorticosterone acetate |
| DPCPX | 8-cyclopentyl-1,3-dipropylxanthine |
| EEG | electroencephalography |
| EP4 | prostanoid EP4 receptor |
| FAAH | fatty acid amide hydrolase |
| GCSF | granulocyte colony stimulating factor |
| HDL | high density lipoprotein |
| HR | heart rate |
| i.a. | intra-arterially |
| IFN-γ | interferon γ |
| IL-n | interleukin n,e.g., IL-1, interleukin-1 |
| IL-1β | interleukin-1ß |
| iNOS | inducible nitric oxide synthase |
| IP | prostacyclin receptor |
| i.p. | intraperitoneally |
| i.v. | intravenously |
| LAD | left anterior descending artery |
| LCx | left circumflex coronary artery |
| LDL | low-density lipoprotein |
| LPS | lipopolysaccharide |
| MCA | middle cerebral artery |
| MCP-1 | monocyte chemoattractant protein-1 |
| MERS | middle east respiratory syndrome |
| MIP-2 | macrophage inflammatory protein-2 |
| MPO | myeloperoxidase |
| MS | multiple sclerosis |
| NF-κB | nuclear factor κB |
| NO | nitric oxide |
| NRP1 | protein neuropilin-1 |
| PAI-1 | plasminogen activator inhibitor-1 |
| pCO2 | partial pressure of carbon dioxide |
| p.o. | per os, orally |
| pO2 | partial pressure of oxygen |
| poly(I:C) | polyriboinosinic:polyribocytidylic acid |
| PPARγ | peroxisome proliferator-activated receptor type γ |
| SARS | severe acute respiratory syndrome |
| SARS-CoV-2 | severe acute respiratory syndrome coronavirus 2 |
| SHR | spontaneously hypertensive rats |
| STZ | streptozotocin |
| TGs | triglycerides |
| THC | Δ9-tetrahydrocannabinol |
| TLC | total lung capacity |
| TMEV | Theiler’s murine encephalomyelitis virus-induced demyelinating disease |
| TMPRSS2 | transmembrane serine protease 2 |
| TNF-α | tumor necrosis factor α |
| t-PA | tissue plasminogen activator |
| TRP | transient receptor potential |
| TRPV1 | transient receptor potential vanilloid subfamily member 1 |
| UVA | ultraviolet A |
| VCAM-1 | vascular cell adhesion molecule-1 |
References
- Baj, J.; Karakuła-Juchnowicz, H.; Teresiński, G.; Buszewicz, G.; Ciesielka, M.; Sitarz, E.; Forma, A.; Karakuła, K.; Flieger, W.; Portincasa, P.; et al. Specific and non-specific clinical manifestations and symptoms: The current state of knowledge. J. Clin. Med. 2020, 9, 1753. [Google Scholar] [CrossRef] [PubMed]
- Tu, Y.F.; Chien, C.S.; Yarmishyn, A.A.; Lin, Y.Y.; Luo, Y.H.; Lin, Y.T.; Lai, W.Y.; Yang, D.M.; Chou, S.J.; Yang, Y.P.; et al. A review of SARS-CoV-2 and the ongoing clinical trials. Int. J. Mol. Sci. 2020, 21, 2657. [Google Scholar] [CrossRef]
- Peng, M. Outbreak of COVID-19: An emerging global pandemic threat. Biomed. Pharmacother. 2020, 129, 110499. [Google Scholar] [CrossRef] [PubMed]
- List of Epidemics. Available online: https://en.wikipedia.org/wiki/List_of_epidemics (accessed on 1 February 2021).
- Cannalire, R.; Stefanelli, I.; Cerchia, C.; Beccari, A.R.; Pelliccia, S.; Summa, V. Entry inhibitors: Small molecules and peptides targeting virus or host cells. Int. J. Mol. Sci. 2020, 21, 5707. [Google Scholar] [CrossRef]
- World Health Organisation. Draft Landscape of COVID-19 Candidate Vaccines. Available online: https://www.who.int/publications/m/item/draft-landscape-of-covid-19-candidate-vaccines (accessed on 1 February 2021).
- Krammer, F. SARS-CoV-2 vaccines in development. Nature 2020, 586, 516–527. [Google Scholar] [CrossRef]
- Weisberg, E.; Sattler, M.; Yang, P.L.; Parent, A.; Gray, N.; Griffin, J.D. Current therapies under investigation for COVID-19: Potential COVID-19 treatments. Can. J. Physiol. Pharmacol. 2020, 98, 483–489. [Google Scholar] [CrossRef]
- Asai, A.; Konno, M.; Ozaki, M.; Otsuka, C.; Vecchione, A.; Arai, T.; Kitagawa, T.; Ofusa, K.; Yabumoto, M.; Hirotsu, T.; et al. COVID-19 drug discovery using intensive approaches. Int. J. Mol. Sci. 2020, 21, 2839. [Google Scholar] [CrossRef]
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. ACTT-1 study group members. Remdesivir for the treatment of Covid-19-final report. N. Engl. J. Med. 2020, 383, 1813–1826. [Google Scholar] [CrossRef]
- Recovery Collaborative Group; Horby, P.; Lim, W.S.; Emberson, J.R.; Mafham, M.; Bell, J.L.; Linsell, L.; Staplin, N.; Brightling, C.; Ustianowski, A.; Elmahi, E.; et al. Dexamethasone in hospitalized patients with Covid-19—Preliminary report. N. Engl. J. Med. 2020. [Google Scholar] [CrossRef]
- McKee, D.L.; Sternberg, A.; Stange, U.; Laufer, S.; Naujokat, C. Candidate drugs against SARS-CoV-2 and COVID-19. Pharmacol. Res. 2020, 157, 104859. [Google Scholar] [CrossRef]
- He, Y.Q.; Zhou, C.C.; Yu, L.Y.; Wang, L.; Deng, J.L.; Tao, Y.L.; Zhang, F.; Chen, W.S. Natural product derived phytochemicals in managing acute lung injury by multiple mechanisms. Pharmacol. Res. 2020, 163, 105224. [Google Scholar] [CrossRef]
- Mamber, S.W.; Krakowka, S.; Osborn, J.; Saberski, L.; Rhodes, R.G.; Dahlberg, A.E.; Pond-Tor, S.; Fitzgerald, K.; Wright, N.; Beseme, S.; et al. Can unconventional immunomodulatory agents help alleviate COVID-19 symptoms and severity? MSphere 2020, 5, e00288. [Google Scholar] [CrossRef]
- Onaivi, E.S.; Sharma, V. Cannabis for COVID-19: Can cannabinoids quell the cytokine storm? Future Sci. 2020, 6, FSO625. [Google Scholar] [CrossRef] [PubMed]
- Ni, W.; Yang, X.; Yang, D.; Bao, J.; Li, R.; Xiao, Y.; Hou, C.; Wang, H.; Liu, J.; Yang, D.; et al. Role of angiotensin-converting enzyme 2 (ACE2) in COVID-19. Crit. Care 2020, 24, 422. [Google Scholar] [CrossRef]
- Kielian, M. Enhancing host cell infection by SARS-CoV-2. Science 2020, 370, 765–766. [Google Scholar] [CrossRef]
- Penna, C.; Mercurio, V.; Tocchetti, C.G.; Pagliaro, P. Sex-related differences in COVID-19 lethality. Br. J. Pharmacol. 2020, 177, 4375–4385. [Google Scholar] [CrossRef] [PubMed]
- Gemmati, D.; Bramanti, B.; Serino, M.L.; Secchiero, P.; Zauli, G.; Tisato, V. COVID-19 and individual genetic susceptibility/receptivity: Role of ACE1/ACE2 genes, immunity, inflammation and coagulation. Might the double X-chromosome in females be protective against SARS-CoV-2 compared to the single X-Chromosome in males? Int. J. Mol. Sci. 2020, 21, 3474. [Google Scholar] [CrossRef] [PubMed]
- Pagliaro, P.; Penna, C. ACE/ACE2 ratio: A key also in 2019 Coronavirus disease (Covid-19)? Front. Med. 2020, 7, 335. [Google Scholar] [CrossRef]
- Gheblawi, M.; Wang, K.; Viveiros, A.; Nguyen, Q.; Zhong, J.C.; Turner, A.J.; Raizada, M.K.; Grant, M.B.; Oudit, G.Y. Angiotensin-Converting Enzyme 2: SARS-CoV-2 receptor and regulator of the renin-angiotensin system: Celebrating the 20th anniversary of the discovery of ACE2. Circ. Res. 2020, 126, 1456–1474. [Google Scholar] [CrossRef]
- Guzik, T.J.; Mohiddin, S.A.; Dimarco, A.; Patel, V.; Savvatis, K.; Marelli-Berg, F.M.; Madhur, M.S.; Tomaszewski, M.; Maffia, P.; D’Acquisto, F.; et al. COVID-19 and the cardiovascular system: Implications for risk assessment, diagnosis, and treatment options. Cardiovasc. Res. 2020, 16, 1666–1687. [Google Scholar] [CrossRef]
- Coperchini, F.; Chiovato, L.; Croce, L.; Magri, F.; Rotondi, M. The cytokine storm in COVID-19: An overview of the involvement of the chemokine/chemokine-receptor system. Cytokine Growth Factor Rev. 2020, 53, 25–32. [Google Scholar] [CrossRef]
- Arendse, L.B.; Danser, A.; Poglitsch, M.; Touyz, R.M.; Burnett, J.C.; Llorens-Cortes, C.; Ehlers, M.R.; Sturrock, E.D. Novel therapeutic approaches targeting the renin-angiotensin system and associated peptides in hypertension and heart failure. Pharmacol. Rev. 2019, 71, 539–570. [Google Scholar] [CrossRef]
- Verdecchia, P.; Cavallini, C.; Spanevello, A.; Angeli, F. COVID-19: ACE2centric infective disease? Hypertension 2020, 76, 294–299. [Google Scholar] [CrossRef]
- Samidurai, A.; Das, A. Cardiovascular Complications associated with COVID-19 and potential therapeutic strategies. Int. J. Mol. Sci. 2020, 21, 6790. [Google Scholar] [CrossRef]
- Iwasaki, M.; Saito, J.; Zhao, H.; Sakamoto, A.; Hirota, K.; Ma, D. Inflammation triggered by SARS-CoV-2 and ACE2 augment drives multiple organ failure of severe COVID-19: Molecular mechanisms and implications. Inflammation 2020, 44, 13–34. [Google Scholar] [CrossRef]
- Cremer, P.C. SARS-CoV-2 and myocardial injury: Few answers, many questions. Cleve. Clin. J. Med. 2020, 87, 521–525. [Google Scholar] [CrossRef] [PubMed]
- Evans, P.C.; Rainger, G.E.; Mason, J.C.; Guzik, T.J.; Osto, E.; Stamataki, Z.; Neil, D.; Hoefer, I.E.; Fragiadaki, M.; Waltenberger, J.; et al. Endothelial dysfunction in COVID-19: A position paper of the ESC Working Group for Atherosclerosis and Vascular Biology, and the ESC Council of Basic Cardiovascular Science. Cardiovasc. Res. 2020, 116, 2177–2184. [Google Scholar] [CrossRef] [PubMed]
- Kochi, A.N.; Tagliari, A.P.; Forleo, G.B.; Fassini, G.M.; Tondo, C. Cardiac and arrhythmic complications in patients with COVID-19. J. Cardiovasc. Electrophysiol. 2020, 31, 1003–1008. [Google Scholar] [CrossRef] [PubMed]
- Zheng, K.I.; Feng, G.; Liu, W.Y.; Targher, G.; Byrne, C.D.; Zheng, M.H. Extrapulmonary complications of COVID-19: A multisystem disease? J. Med. Virol 2020, 10. [Google Scholar] [CrossRef]
- De Vries, A. SARS-CoV-2/COVID-19: A primer for cardiologists. Neth. Heart J. 2020, 28, 366–383. [Google Scholar] [CrossRef]
- Zaim, S.; Chong, J.H.; Sankaranarayanan, V.; Harky, A. COVID-19 and multiorgan response. Curr. Prob. Cardiol. 2020, 45, 100618. [Google Scholar] [CrossRef]
- Gąsecka, A.; Borovac, J.A.; Guerreiro, R.A.; Giustozzi, M.; Parker, W.; Caldeira, D.; Chiva-Blanch, G. Thrombotic complications in patients with COVID-19: Pathophysiological mechanisms, diagnosis, and treatment. Cardiovasc. Drugs Ther. 2020, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Jayarangaiah, A.; Kariyanna, P.T.; Chen, X.; Jayarangaiah, A.; Kumar, A. COVID-19-associated coagulopathy: An exacerbated immunothrombosis response. Clin. Appl. Thromb. Hemost. 2020, 26, 1076029620943293. [Google Scholar] [CrossRef]
- Vinayagam, S.; Sattu, K. SARS-CoV-2 and coagulation disorders in different organs. Life Sci. 2020, 260, 118431. [Google Scholar] [CrossRef]
- Chueh, T.I.; Zheng, C.M.; Hou, Y.C.; Lu, K.C. Novel evidence of acute kidney injury in COVID-19. J. Clin. Med. 2020, 9, 3547. [Google Scholar] [CrossRef] [PubMed]
- Gan, R.; Rosoman, N.P.; Henshaw, D.; Noble, E.P.; Georgius, P.; Sommerfeld, N. COVID-19 as a viral functional ACE2 deficiency disorder with ACE2 related multi-organ disease. Med. Hypotheses 2020, 144, 110024. [Google Scholar] [CrossRef]
- Galanopoulos, M.; Gkeros, F.; Doukatas, A.; Karianakis, G.; Pontas, C.; Tsoukalas, N.; Viazis, N.; Liatsos, C.; Mantzaris, G.J. COVID-19 pandemic: Pathophysiology and manifestations from the gastrointestinal tract. World J. Gastroenterol. 2020, 26, 4579–4588. [Google Scholar] [CrossRef]
- Hunt, R.H.; East, J.E.; Lanas, A.; Malfertheiner, P.; Satsangi, J.; Scarpignato, C.; Webb, G.J. COVID-19 and gastrointestinal disease. Implications for the gastroenterologist. Dig. Dis. 2020, 1–21. [Google Scholar] [CrossRef]
- Asadi-Pooya, A.A. Seizures associated with coronavirus infections. Seizure 2020, 79, 49–52. [Google Scholar] [CrossRef] [PubMed]
- Pennisi, M.; Lanza, G.; Falzone, L.; Fisicaro, F.; Ferri, R.; Bella, R. SARS-CoV-2 and the nervous system: From clinical features to molecular mechanisms. Int. J. Mol. Sci. 2020, 21, 5475. [Google Scholar] [CrossRef]
- Varatharaj, A.; Thomas, N.; Ellul, M.A.; Davies, N.W.S.; Pollak, T.A.; Tenorio, E.L.; Sultan, M.; Easton, A.; Breen, G.; Zandi, M.; et al. CoroNerve Study Group. Neurological and neuropsychiatric complications of COVID-19 in 153 patients: A UK-wide surveillance study. Lancet Psychiatry 2020, 7, 875–882. [Google Scholar] [CrossRef]
- Luís, M.E.; Hipólito-Fernandes, D.; Mota, C.; Maleita, D.; Xavier, C.; Maio, T.; Cunha, J.P.; Tavares Ferreira, J. A review of neuro-ophthalmological manifestations of human coronavirus infection. Eye Brain 2020, 12, 129–137. [Google Scholar] [CrossRef] [PubMed]
- Somasundaram, N.P.; Ranathunga, I.; Ratnasamy, V.; Wijewickrama, P.; Dissanayake, H.A.; Yogendranathan, N.; Gamage, K.; de Silva, N.L.; Sumanatilleke, M.; Katulanda, P.; et al. The impact of SARS-Cov-2 virus infection on the endocrine system. J. Endocr. Soc. 2020, 4, bvaa082. [Google Scholar] [CrossRef]
- Lundholm, M.D.; Poku, C.; Emanuele, N.; Emanuele, M.A.; Lopez, N. SARS-CoV-2 (COVID-19) and the endocrine system. J. Endocr. Soc. 2020, 4, bvaa144. [Google Scholar] [CrossRef] [PubMed]
- Juan, J.; Gil, M.M.; Rong, Z.; Zhang, Y.; Yang, H.; Poon, L.C. Effect of coronavirus disease 2019 (COVID-19) on maternal, perinatal and neonatal outcome: Systematic review. Ultrasound Obstet. Gynecol. 2020, 56, 15–27. [Google Scholar] [CrossRef]
- Gonzalez, A.; Orozco-Aguilar, J.; Achiardi, O.; Simon, F.; Cabello-Verrugio, C. SARS-CoV-2/renin-angiotensin system: Deciphering the clues for a couple with potentially harmful effects on skeletal muscle. Int. J. Mol. Sci. 2020, 21, 7904. [Google Scholar] [CrossRef]
- Rahimi, H.; Tehranchinia, Z. A comprehensive review of cutaneous manifestations associated with COVID-19. Biomed. Res. Int. 2020, 2020, 1236520. [Google Scholar] [CrossRef]
- Mawhirt, S.L.; Frankel, D.; Diaz, A.M. Cutaneous manifestations in adult patients with COVID-19 and dermatologic conditions related to the COVID-19 pandemic in health care workers. Curr. Allergy Asthma Rep. 2020, 20, 75. [Google Scholar] [CrossRef]
- Kicman, A.; Toczek, M. The effects of cannabidiol, a non-intoxicating compound of cannabis, on the cardiovascular system in health and disease. Int. J. Mol. Sci. 2020, 21, 6740. [Google Scholar] [CrossRef] [PubMed]
- Atalay, S.; Jarocka-Karpowicz, I.; Skrzydlewska, E. Antioxidative and anti-inflammatory properties of cannabidiol. Antioxidants 2019, 9, 21. [Google Scholar] [CrossRef]
- Ligresti, A.; De Petrocellis, L.; Di Marzo, V. Phytocannabinoids to cannabinoid receptors and endocannabinoids: Pleiotropic physiological and pathological roles through complex pharmacology. Physiol. Rev. 2016, 96, 1593–1659. [Google Scholar] [CrossRef]
- Britch, S.C.; Babalonis, S.; Walsh, S.L. Cannabidiol: Pharmacology and therapeutic targets. Psychopharmacology 2020, 238, 9–28. [Google Scholar] [CrossRef]
- Pisanti, S.; Malfitano, A.M.; Ciaglia, E.; Lamberti, A.; Ranieri, R.; Cuomo, G.; Abate, M.; Faggiana, G.; Proto, M.C.; Fiore, D.; et al. Cannabidiol: State of the art and new challenges for therapeutic applications. Pharmacol. Ther. 2017, 175, 133–150. [Google Scholar] [CrossRef] [PubMed]
- Esposito, G.; Pesce, M.; Seguella, L.; Sanseverino, W.; Lu, J.; Corpetti, C.; Sarnelli, G. The potential of cannabidiol in the COVID-19 pandemic. Br. J. Pharmacol. 2020, 177, 4967–4970. [Google Scholar] [CrossRef]
- Janmohamed, K.; Soale, A.N.; Forastiere, L.; Tang, W.; Sha, Y.; Demant, J.; Airoldi, E.; Kumar, N. Intersection of the web-based vaping narrative with COVID-19: Topic modeling study. J. Med. Internet Res. 2020, 22, e21743. [Google Scholar] [CrossRef] [PubMed]
- Sexton, M. Cannabis in the time of coronavirus disease 2019: The Yin and Yang of the endocannabinoid system in immunocompetence. J. Altern. Complement. Med. 2020, 26, 444–448. [Google Scholar] [CrossRef] [PubMed]
- Costiniuk, C.T.; Jenabian, M.A. Acute inflammation and pathogenesis of SARS-CoV-2 infection: Cannabidiol as a potential anti-inflammatory treatment? Cytokine Growth Factor Rev. 2020, 53, 63–65. [Google Scholar] [CrossRef] [PubMed]
- Rossi, F.; Tortora, C.; Argenziano, M.; Di Paola, A.; Punzo, F. Cannabinoid receptor type 2: A possible target in SARS-CoV-2 (CoV-19) infection? Int. J. Mol. Sci. 2020, 21, 3809. [Google Scholar] [CrossRef]
- Shover, C.L.; Humphreys, K. Debunking cannabidiol as a treatment for COVID-19: Time for the FDA to adopt a focused deterrence model? Cureus 2020, 12, e8671. [Google Scholar] [CrossRef]
- Biali, M.; Broers, B.; Besson, M.; Demeules, J. Cannabinoids and COVID-19. Med. Cannabis Cannabinoids 2020, 3, 111–115. [Google Scholar] [CrossRef]
- Khalsa, J.H.; Bunt, G.; Maggirwar, S.B.; Kottilil, S. COVID-19 and cannabidiol (CBD). J. Addict. Med. 2020. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.D. Cannabidiol as prophylaxis for SARS-CoV-2 and COVID-19? Unfounded claims versus potential risks of medications during the pandemic. Res. Soc. Adm. Pharm. 2021, 17, 2053. [Google Scholar] [CrossRef]
- Hill, K.P. Cannabinoids and the coronavirus. Cannabis Cannabinoid Res. 2020, 5, 118–120. [Google Scholar] [CrossRef]
- Booz, G.W. Cannabidiol as an emergent therapeutic strategy for lessening the impact of inflammation on oxidative stress. Free Radic. Biol. Med. 2011, 51, 1054–1061. [Google Scholar] [CrossRef]
- Wang, B.; Kovalchuk, A.; Li, D.; Rodriguez-Juarez, R.; Ilnytskyy, Y.; Kovalchuk, I.; Kovalchuk, O. In search of preventive strategies: Novel high-CBD Cannabis sativa extracts modulate ACE2 expression in COVID-19 gateway tissues. Aging 2020, 2, 22425–22444. [Google Scholar] [CrossRef]
- Raj, V.; Park, J.G.; Cho, K.H.; Choi, P.; Kim, T.; Ham, J.; Lee, J. Assessment of antiviral potencies of cannabinoids against SARS-CoV-2 using computational and in vitro approaches. Int. J. Biol. Macromol. 2020, 168, 474–485. [Google Scholar] [CrossRef] [PubMed]
- Lowe, H.I.; Toyang, N.J.; McLaughlin, W. Potential of cannabidiol for the treatment of viral hepatitis. Pharmacognosy. Res. 2017, 9, 116–118. [Google Scholar] [CrossRef] [PubMed]
- Maor, Y.; Yu, J.; Kuzontkoski, P.M.; Dezube, B.J.; Zhang, X.; Groopman, J.E. Cannabidiol inhibits growth and induces programmed cell death in kaposi sarcoma-associated herpesvirus-infected endothelium. Genes. Cancer 2012, 3, 512–520. [Google Scholar] [CrossRef]
- Khodadadi, H.; Salles, É.L.; Jarrahi, A.; Chibane, F.; Costigliola, V.; Yu, J.C.; Vaibhav, K.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol modulates cytokine storm in acute respiratory distress syndrome induced by simulated viral infection using synthetic RNA. Cannabis Cannabinoid Res. 2020, 5, 197–201. [Google Scholar] [CrossRef] [PubMed]
- Salles, É.L.; Khodadadi, H.; Jarrahi, A.; Ahluwalia, M.; Paffaro, V.A.; Costigliola, V.; Yu, J.C.; Hess, D.C.; Dhandapani, K.M.; Baban, B. Cannabidiol (CBD) modulation of apelin in acute respiratory distress syndrome. J. Cell. Mol. Med. 2020, 24, 12869–12872. [Google Scholar] [CrossRef]
- Patel, D.C.; Wallis, G.; Fujinami, R.S.; Wilcox, K.S.; Smith, M.D. Cannabidiol reduces seizures following CNS infection with Theiler’s murine encephalomyelitis virus. Epilepsia Open 2019, 4, 431–442. [Google Scholar] [CrossRef]
- Mecha, M.; Feliú, A.; Iñigo, P.M.; Mestre, L.; Carrillo-Salinas, F.J.; Guaza, C. Cannabidiol provides long-lasting protection against the deleterious effects of inflammation in a viral model of multiple sclerosis: A role for A2A receptors. Neurobiol. Dis. 2013, 59, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.; Ferraz-de-Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Mariano-Souza, D.P.; Quinteiro-Filho, W.M.; Akamine, A.T.; Almeida, V.I.; Quevedo, J.; Dal-Pizzol, F.; et al. Cannabidiol, a non-psychotropic plant-derived cannabinoid, decreases inflammation in a murine model of acute lung injury: Role for the adenosine A2A receptor. Eur. J. Pharmacol. 2012, 678, 78–85. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro, A.; Almeida, V.I.; Costola De Souza, C.; Ferraz De Paula, V.; Pinheiro, M.L.; Vitoretti, L.B.; Gimenes-Junior, J.A.; Akamine, A.T.; Crippa, J.A.; Tavares De Lima, W.; et al. Cannabidiol improves lung function and inflammation in mice submitted to LPS-induced acute lung injury. Immunopharmacol. Immunotoxicol. 2015, 37, 35–41. [Google Scholar] [CrossRef]
- Sadowska, O.; Baranowska-Kuczko, M.; Gromotowicz-Popławska, A.; Biernacki, M.; Kicman, A.; Malinowska, B.; Kasacka, I.; Krzyżewska, A.; Kozłowska, H. Cannabidiol ameliorates monocrotaline-induced pulmonary hypertension in rats. Int. J. Mol. Sci. 2020, 21, 7077. [Google Scholar] [CrossRef] [PubMed]
- Arruza, L.; Pazos, M.R.; Mohammed, N.; Escribano, N.; Lafuente, H.; Santos, M.; Alvarez-Díaz, F.J. Cannabidiol reduces lung injury induced by hypoxic-ischemic brain damage in newborn piglets. Pediatr. Res. 2017, 82, 79–86. [Google Scholar] [CrossRef] [PubMed]
- Vuolo, F.; Petronilho, F.; Sonai, B.; Ritter, C.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.; Dal-Pizzol, F. Evaluation of serum cytokines levels and the role of cannabidiol treatment in animal model of asthma. Mediators Inflamm. 2015, 2015, 538670. [Google Scholar] [CrossRef] [PubMed]
- Vuolo, F.; Abreu, S.C.; Michels, M.; Xisto, D.G.; Blanco, N.G.; Hallak, J.E.; Zuardi, A.W.; Crippa, J.A.; Reis, C.; Bahl, M.; et al. Cannabidiol reduces airway inflammation and fibrosis in experimental allergic asthma. Eur. J. Pharmacol. 2019, 843, 251–259. [Google Scholar] [CrossRef]
- Dudášová, A.; Keir, S.D.; Parsons, M.E.; Molleman, A.; Page, C.P. The effects of cannabidiol on the antigen-induced contraction of airways smooth muscle in the guinea-pig. Pulm. Pharmacol. Ther. 2013, 26, 373–379. [Google Scholar] [CrossRef] [PubMed]
- Baranowska-Kuczko, M.; Kozłowska, H.; Kloza, M.; Sadowska, O.; Kozłowski, M.; Kusaczuk, M.; Kasacka, I.; Malinowska, B. Vasodilatory effects of cannabidiol in human pulmonary and rat small mesenteric arteries: Modification by hypertension and the potential pharmacological opportunities. J. Hypertens. 2020, 38, 896–911. [Google Scholar] [CrossRef]
- Karmaus, P.W.; Wagner, J.G.; Harkema, J.R.; Kaminski, N.E.; Kaplan, B.L. Cannabidiol (CBD) enhances lipopolysaccharide (LPS)-induced pulmonary inflammation in C57BL/6 mice. J. Immunotoxicol. 2013, 10, 321–328. [Google Scholar] [CrossRef] [PubMed]
- Makwana, R.; Venkatasamy, R.; Spina, D.; Page, C. The effect of phytocannabinoids on airway hyper-responsiveness, airway inflammation, and cough. J. Pharmacol. Expl. Ther. 2015, 353, 169–180. [Google Scholar] [CrossRef]
- Feng, Y.; Chen, F.; Yin, T.; Xia, Q.; Liu, Y.; Huang, G.; Zhang, J.; Oyen, R.; Ni, Y.J. Effects of cannabidiol on acute reperfused myocardial infarction in rabbits: Evaluated with 3.0T Cardiac Magnetic Resonance Imaging and Histopathology. J. Cardiovasc. Pharmacol. 2015, 66, 354–363. [Google Scholar] [CrossRef] [PubMed]
- Durst, R.; Danenberg, H.; Gallily, R.; Mechoulam, R.; Meir, K.; Grad, E.; Beeri, R.; Pugatsch, T.; Tarsish, E.; Lotan, C. Cannabidiol, a nonpsychoactive Cannabis constituent, protects against myocardial ischemic reperfusion injury. Am. J. Physiol. Heart Circ. Physiol. 2007, 293, 3602–3607. [Google Scholar] [CrossRef]
- Walsh, S.K.; Hepburn, C.Y.; Kane, K.A.; Wainwright, C.L. Acute administration of cannabidiol in vivo suppresses ischaemia-induced cardiac arrhythmias and reduces infarct size when given at reperfusion. Br. J. Pharmacol. 2010, 160, 1234–1242. [Google Scholar] [CrossRef]
- Gonca, E.; Darici, F. The effect of cannabidiol on ischemia/reperfusion-induced ventricular arrhythmias: The role of adenosine A1 receptors. J. Cardiovasc. Pharmacol. Ther. 2015, 20, 76–83. [Google Scholar] [CrossRef]
- Lee, W.S.; Erdelyi, K.; Matyas, C.; Mukhopadhyay, P.; Varga, Z.V.; Liaudet, L.; Haskú, G.; Čiháková, D.; Mechoulam, R.; Pacher, P. Cannabidiol limits T cell-mediated chronic autoimmune myocarditis: Implications to autoimmune disorders and organ transplantation. Mol. Med. 2016, 22, 136–146. [Google Scholar] [CrossRef] [PubMed]
- Rajesh, M.; Mukhopadhyay, P.; Bátkai, S.; Patel, V.; Saito, K.; Matsumoto, S.; Kashiwaya, Y.; Horváth, B.; Mukhopadhyay, B.; Becker, L.; et al. Cannabidiol attenuates cardiac dysfunction, oxidative stress, fibrosis, and inflammatory and cell death signaling pathways in diabetic cardiomyopathy. J. Am. Coll. Cardiol. 2010, 56, 2115–2125. [Google Scholar] [CrossRef]
- Remiszewski, P.; Jarocka-Karpowicz, I.; Biernacki, M.; Jastrząb, A.; Schlicker, E.; Toczek, M.; Harasim-Symbor, E.; Pędzińska-Betiuk, A.; Malinowska, B. Chronic Cannabidiol administration fails to diminish blood pressure in rats with primary and secondary hypertension despite its effects on cardiac and plasma endocannabinoid system, oxidative stress and lipid metabolism. Int. J. Mol. Sci. 2020, 21, 1295. [Google Scholar] [CrossRef]
- Pędzińska-Betiuk, A.; Weresa, J.; Schlicker, E.; Harasim-Symbor, E.; Toczek, M.; Kasacka, I.; Gajo, B.; Malinowska, B. Chronic cannabidiol treatment reduces the carbachol-induced coronary constriction and left ventricular cardiomyocyte width of the isolated hypertensive rat heart. Toxicol. Appl. Pharmacol. 2020, 411, 115368. [Google Scholar] [CrossRef]
- Wheal, A.J.; Jadoon, K.; Randall, M.D.; O’Sullivan, S.E. In Vivo cannabidiol treatment improves endothelium-dependent vasorelaxation in mesenteric arteries of zucker diabetic fatty rats. Front. Pharmacol. 2017, 8, 248. [Google Scholar] [CrossRef] [PubMed]
- Grambow, E.; Strüder, D.; Klar, E.; Hinz, B.; Vollmar, B. Differential effects of endogenous, phyto and synthetic cannabinoids on thrombogenesis and platelet activity. BioFactors 2016, 42, 581–590. [Google Scholar] [CrossRef]
- Fouad, A.A.; Al-Mulhim, A.S.; Jresat, I. Cannabidiol treatment ameliorates ischemia/reperfusion renal injury in rats. Life Sci. 2012, 91, 284–292. [Google Scholar] [CrossRef]
- Soares, R.Z.; Vuolo, F.; Dall’Igna, D.M.; Michels, M.; Crippa, J.A.; Hallak, J.E.; Zuardi, A.W.; Dal-Pizzol, F. Evaluation of the role of the cannabidiol system in an animal model of ischemia/reperfusion kidney injury. Rev. Bras. Ter. Intensiva 2015, 27, 383–389. [Google Scholar] [CrossRef] [PubMed]
- Baban, B.; Khodadadi, H.; Vaibhav, K.; Marchetti, C.; Riccardi, C.; Mozaffari, M.S. Regulation of innate lymphoid cells in acute kidney injury: Crosstalk between cannabidiol and GILZ. J. Immunol. Res. 2020, 2020, 6056373. [Google Scholar] [CrossRef] [PubMed]
- Rock, E.M.; Sullivan, M.T.; Collins, S.A.; Goodman, H.; Limebeer, C.L.; Mechoulam, R.; Parker, L.A. Evaluation of repeated or acute treatment with cannabidiol (CBD), cannabidiolic acid (CBDA) or CBDA methyl ester (HU-580) on nausea and/or vomiting in rats and shrews. Psychopharmacology 2020, 37, 2621–2631. [Google Scholar] [CrossRef]
- Kossakowski, R.; Schlicker, E.; Toczek, M.; Weresa, J.; Malinowska, B. Cannabidiol affects the Bezold-Jarisch reflex via TRPV1 and 5-HT3 receptors and has peripheral sympathomimetic effects in spontaneously hypertensive and normotensive rats. Front. Pharmacol. 2019, 10, 500. [Google Scholar] [CrossRef]
- Mukhopadhyay, P.; Rajesh, M.; Horváth, B.; Bátkai, S.; Park, O.; Tanchian, G.; Gao, R.Y.; Patel, V.; Wink, D.A.; Liaudet, L.; et al. Cannabidiol protects against hepatic ischemia/reperfusion injury by attenuating inflammatory signaling and response, oxidative/nitrative stress, and cell death. Free Radic. Biol. Med. 2011, 50, 1368–1381. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Mukhopadhyay, P.; Cao, Z.; Wang, H.; Feng, D.; Haskó, G.; Mechoulam, R.; Gao, B.; Pacher, P. Cannabidiol attenuates alcohol-induced liver steatosis, metabolic dysregulation, inflammation and neutrophil-mediated injury. Sci. Rep. 2017, 7, 12064. [Google Scholar] [CrossRef] [PubMed]
- Avraham, Y.; Grigoriadis, N.; Poutahidis, T.; Vorobiev, L.; Magen, I.; Ilan, Y.; Mechoulam, R.; Berry, E. Cannabidiol improves brain and liver function in a fulminant hepatic failure-induced model of hepatic encephalopathy in mice. Br. J. Pharmacol. 2010, 162, 1650–1658. [Google Scholar] [CrossRef]
- Vilela, L.R.; Gomides, L.F.; David, B.A.; Antunes, M.M.; Diniz, A.B.; Moreira, F.; Menezes, G.B. Cannabidiol rescues acute hepatic toxicity and seizure induced by cocaine. Mediators Inflamm. 2015, 2015, 523418. [Google Scholar] [CrossRef]
- Li, K.; Feng, J.Y.; Li, Y.Y.; Yuece, B.; Lin, X.H.; Yu, L.Y.; Li, Y.N.; Feng, Y.J.; Storr, M. Anti-inflammatory role of cannabidiol and O-1602 in cerulein-induced acute pancreatitis in mice. Pancreas 2013, 42, 123–129. [Google Scholar] [CrossRef] [PubMed]
- Capasso, R.; Borrelli, F.; Aviello, G.; Romano, B.; Scalisi, C.; Capasso, F.; Izzo, A.A. Cannabidiol, extracted from Cannabis sativa, selectively inhibits inflammatory hypermotility in mice. Br. J. Pharmacol. 2008, 154, 1001–1008. [Google Scholar] [CrossRef] [PubMed]
- Pagano, E.; Capasso, R.; Piscitelli, F.; Romano, B.; Parisi, O.A.; Finizio, S.; Lauritano, A.; Marzo, V.D.; Izzo, A.A.; Borrelli, F. An orally active cannabis extract with high content in cannabidiol attenuates chemically-induced intestinal inflammation and hypermotility in the mouse. Front. Pharmacol. 2016, 7, 341. [Google Scholar] [CrossRef] [PubMed]
- Gáll, Z.; Farkas, S.; Albert, Á.; Ferencz, E.; Vancea, S.; Urkon, M.; Kolcsár, M. Effects of chronic cannabidiol treatment in the rat chronic unpredictable mild stress model of depression. Biomolecules 2020, 10, 801. [Google Scholar] [CrossRef] [PubMed]
- Fiani, B.; Sarhadi, K.J.; Soula, M.; Zafar, A.; Quadri, S.A. Current application of cannabidiol (CBD) in the management and treatment of neurological disorders. Neurol. Sci. 2020, 41, 3085–3098. [Google Scholar] [CrossRef]
- Patra, P.H.; Barker-Haliski, M.; White, H.S.; Whalley, B.J.; Glyn, S.; Sandhu, H.; Jones, N.; Bazelot, M.; Williams, C.M.; McNeish, A.J. Cannabidiol reduces seizures and associated behavioral comorbidities in a range of animal seizure and epilepsy models. Epilepsia 2019, 60, 303–314. [Google Scholar] [CrossRef]
- Vilela, L.R.; Lima, I.V.; Kunsch, É.B.; Pinto, H.; De Miranda, A.S.; Vieira, É.; De Oliveira, A.; Moraes, M.; Teixeira, A.L.; Moreira, F.A. Anticonvulsant effect of cannabidiol in the pentylenetetrazole model: Pharmacological mechanisms, electroencephalographic profile, and brain cytokine levels. Epilepsy Behav. 2017, 75, 29–35. [Google Scholar] [CrossRef]
- Pazos, M.R.; Mohammed, N.; Lafuente, H.; Santos, M.; Martínez-Pinilla, E.; Moreno, E.; Valdizan, E.; Romero, J.; Pazos, A.; Franco, R.; et al. Mechanisms of cannabidiol neuroprotection in hypoxic-ischemic newborn pigs: Role of 5HT1A and CB2 receptors. Neuropharmacology 2013, 71, 282–291. [Google Scholar] [CrossRef]
- Lafuente, H.; Pazos, M.R.; Alvarez, A.; Mohammed, N.; Santos, M.; Arizti, M.; Alvarez, F.J.; Martinez-Orgado, J.A. Effects of cannabidiol and hypothermia on short-term brain damage in new-born piglets after acute hypoxia-ischemia. Front. Neurosci. 2016, 10, 323. [Google Scholar] [CrossRef]
- Ruiz-Valdepeñas, L.; Martínez-Orgado, J.A.; Benito, C.; Millán, A.; Tolón, R.M.; Romero, J. Cannabidiol reduces lipopolysaccharide-induced vascular changes and inflammation in the mouse brain: An intravital microscopy study. J. Neuroinflamm. 2011, 8, 5. [Google Scholar] [CrossRef] [PubMed]
- Hayakawa, K.; Mishima, K.; Irie, K.; Hazekawa, M.; Mishima, S.; Fujioka, M.; Orito, K.; Egashira, N.; Katsurabayashi, S.; Takasaki, K.; et al. Cannabidiol prevents a post-ischemic injury progressively induced by cerebral ischemia via a high-mobility group box1-inhibiting mechanism. Neuropharmacology 2008, 55, 1280–1286. [Google Scholar] [CrossRef] [PubMed]
- Mishima, K.; Hayakawa, K.; Abe, K.; Ikeda, T.; Egashira, N.; Iwasaki, K.; Fujiwara, M. Cannabidiol prevents cerebral infarction via a serotonergic 5-hydroxytryptamine 1A receptor-dependent mechanism. Stroke 2005, 36, 1077–1082. [Google Scholar] [CrossRef] [PubMed]
- Liou, G.; El-Remessy, A.; Ibrahim, A.; Caldwell, R.; Khalifa, Y.; Gunes, A.; Nussbaum, J. Cannabidiol as a putative novel therapy for diabetic retinopathy: A postulated mechanism of action as an entry point for biomarker-guided clinical development. Curr. Pharmacogen. Person Med. 2009, 7, 215–222. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Liou, G.I.; Auchampach, J.A.; Hillard, C.J.; Zhu, G.; Yousufzai, B.; Mian, S.; Khan, S.; Khalifa, Y. Mediation of cannabidiol anti-inflammation in the retina by equilibrative nucleoside transporter and A2A adenosine receptor. Invest. Ophthalmol. Vis. Sci. 2008, 9, 5526–5531. [Google Scholar] [CrossRef]
- Bielawiec, P.; Harasim-Symbor, E.; Konstantynowicz-Nowicka, K.; Sztolsztener, K.; Chabowski, A. Chronic cannabidiol administration attenuates skeletal muscle de novo ceramide synthesis pathway and related metabolic effects in a rat model of high-fat diet-induced obesity. Biomolecules 2020, 10, 1241. [Google Scholar] [CrossRef]
- Zorzenon, M.; Santiago, A.N.; Mori, M.A.; Piovan, S.; Jansen, C.A.; Perina Padilha, M.E.; Ciotta, S.R.; Cezar De Freitas Mathias, P.; Guimarães, F.S.; Weffort De Oliveira, R.M.; et al. Cannabidiol improves metabolic dysfunction in middle-aged diabetic rats submitted to a chronic cerebral hypoperfusion. Chem. Biol. Interact. 2019, 312, 108819. [Google Scholar] [CrossRef]
- Paoli, A.; Gorini, S.; Caprio, M. The dark side of the spoon-glucose, ketones and COVID-19: A possible role for ketogenic diet? J. Transl. Med. 2020, 18, 441. [Google Scholar] [CrossRef]
- Iannotti, F.A.; Pagano, E.; Moriello, A.S.; Alvino, F.G.; Sorrentino, N.C.; D’Orsi, L.; Gazzerro, E.; Capasso, R.; De Leonibus, E.; De Petrocellis, L.; et al. Effects of non-euphoric plant cannabinoids on muscle quality and performance of dystrophic mdx mice. Br. J. Pharmacol. 2019, 176, 1568–1584. [Google Scholar] [CrossRef]
- Atalay, S.; Gęgotek, A.; Wroński, A.; Domigues, P.; Skrzydlewska, E. Therapeutic application of cannabidiol on UVA and UVB irradiated rat skin. A proteomic study. J. Pharm. Biomed. Anal. 2021, 192, 113656. [Google Scholar] [CrossRef]
- Oláh, A.; Tóth, B.I.; Borbíró, I.; Sugawara, K.; Szöllõsi, A.G.; Czifra, G.; Pál, B.; Ambrus, L.; Kloepper, J.; Camera, E.; et al. Cannabidiol exerts sebostatic and antiinflammatory effects on human sebocytes. J. Clin. Invest. 2014, 124, 3713–3724. [Google Scholar] [CrossRef]
- Petrosino, S.; Verde, R.; Vaia, M.; Allarà, M.; Iuvone, T.; Di Marzo, V. Anti-inflammatory properties of cannabidiol, a nonpsychotropic cannabinoid, in experimental allergic contact dermatitis. J. Pharmacol. Exp. Ther. 2018, 365, 652–663. [Google Scholar] [CrossRef]
- Cassol, O.J.; Comim, C.M.; Silva, B.R.; Hermani, F.V.; Constantino, L.S.; Felisberto, F.; Petronilho, F.; Hallak, J.E.; De Martinis, B.S.; Zuardi, A.W.; et al. Treatment with cannabidiol reverses oxidative stress parameters, cognitive impairment and mortality in rats submitted to sepsis by cecal ligation and puncture. Brain Res. 2010, 1348, 128–138. [Google Scholar] [CrossRef]
- Abdallah, S.J.; Smith, B.M.; Ware, M.A.; Moore, M.; Li, P.Z.; Bourbeau, J.; Jensen, D. Effect of vaporized cannabis on exertional breathlessness and exercise endurance in advanced chronic obstructive pulmonary disease. A Randomized Controlled Trial. Ann. Am. Thorac. Soc. 2018, 15, 1146–1158. [Google Scholar] [CrossRef]
- Pickering, E.E.; Semple, S.J.; Nazir, M.S.; Murphy, K.; Snow, T.M.; Cummin, A.R.; Moosavi, S.H.; Guz, A.; Holdcroft, A. Cannabinoid effects on ventilation and breathlessness: A pilot study of efficacy and safety. Chron. Respir. Dis. 2011, 8, 109–118. [Google Scholar] [CrossRef] [PubMed]
- Duran, M.; Pérez, E.; Abanades, S.; Vidal, X.; Saura, C.; Majem, M.; Arriola, E.; Rabanal, M.; Pastor, A.; Farré, M.; et al. Preliminary efficacy and safety of an oromucosal standardized cannabis extract in chemotherapy-induced nausea and vomiting. Br. J. Clin. Pharmacol. 2010, 70, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Luftner, D.; Possinger, K.; Ernst, G.; Ruhstaller, T.; Meissner, W.; Ko, Y.D.; Schnelle, M.; Reif, M.; Cerny, T. Comparison of orally administered cannabis extract and delta-9-tetrahydrocannabinol in treating patients with cancer-related anorexia-cachexia syndrome: A multicenter, phase III, randomized, double-blind, placebo-controlled clinical trial from the Cannabis-In-Cachexia-Study-Group. J. Clin. Oncol. 2006, 24, 3394–3400. [Google Scholar] [CrossRef]
- Couch, D.G.; Cook, H.; Ortori, C.; Barrett, D.; Lund, J.N.; O’Sullivan, S.E. Palmitoylethanolamide and cannabidiol prevent inflammation-induced hyperpermeability of the human gut in vitro and in vivo—A randomized, placebo-controlled, double-blind controlled trial. Inflamm. Bowel Dis. 2019, 25, 1006–1018. [Google Scholar] [CrossRef]
- Naftali, T.; Mechulam, R.; Marii, A.; Gabay, G.; Stein, A.; Bronshtain, M.; Laish, I.; Benjaminov, F.; Konikoff, F.M. Low-dose cannabidiol is safe but not effective in the treatment for Crohn’s disease, a randomized controlled trial. Dig. Dis. Sci. 2017, 62, 1615–1620. [Google Scholar] [CrossRef] [PubMed]
- Irving, P.M.; Iqbal, T.; Nwokolo, C.; Subramanian, S.; Bloom, S.; Prasad, N.; Hart, A.; Murray, C.; Lindsay, J.O.; Taylor, A.; et al. A randomized, double-blind, placebo-controlled, parallel-group, pilot study of cannabidiol-rich botanical extract in the symptomatic treatment of ulcerative colitis. Inflamm. Bowel Dis. 2018, 24, 714–724. [Google Scholar] [CrossRef]
- Zuardi, A.W.; Cosme, R.A.; Graeff, F.G.; Guimarães, F.S. Effects of ipsapirone and cannabidiol on human experimental anxiety. J. Psychopharmacol. 1993, 7, 82–88. [Google Scholar] [CrossRef]
- Crippa, J.A.; Zuardi, A.W.; Garrido, G.E.; Wichert-Ana, L.; Guarnieri, R.; Ferrari, L.; Azevedo-Marques, P.M.; Hallak, J.E.; McGuire, P.K.; Filho Busatto, G. Effects of cannabidiol (CBD) on regional cerebral blood flow. Neuropsychopharmacology 2004, 29, 417–426. [Google Scholar] [CrossRef] [PubMed]
- Zuardi, A.W.; Rodrigues, N.P.; Silva, A.L.; Bernardo, S.A.; Hallak, J.; Guimarães, F.S.; Crippa, J. Inverted U-shaped dose-response curve of the anxiolytic effect of cannabidiol during public speaking in real life. Front. Pharmacol. 2017, 8, 259. [Google Scholar] [CrossRef]
- Bergamaschi, M.M.; Queiroz, R.H.; Chagas, M.H.; De Oliveira, D.C.; De Martinis, B.S.; Kapczinski, F.; Quevedo, J.; Roesler, R.; Schröder, N.; Nardi, A.E.; et al. Cannabidiol reduces the anxiety induced by simulated public speaking in treatment-naïve social phobia patients. Neuropsychopharmacology 2011, 36, 1219–1226. [Google Scholar] [CrossRef]
- Crippa, J.A.; Derenusson, G.N.; Ferrari, T.B.; Wichert-Ana, L.; Duran, F.L.; Martin-Santos, R.; Simões, M.V.; Bhattacharyya, S.; Fusar-Poli, P.; Atakan, Z.; et al. Neural basis of anxiolytic effects of cannabidiol (CBD) in generalized social anxiety disorder: A preliminary report. J. Psychopharmacol. 2011, 25, 121–130. [Google Scholar] [CrossRef]
- Elms, L.; Shannon, S.; Hughes, S.; Lewis, N. Cannabidiol in the treatment of post-traumatic stress disorder: A case series. J. Altern. Complement. Med. 2019, 25, 392–397. [Google Scholar] [CrossRef]
- Shannon, S.; Lewis, N.; Lee, H.; Hughes, S. Cannabidiol in anxiety and sleep: A large case series. Perm. J. 2019, 23, 18–41. [Google Scholar] [CrossRef] [PubMed]
- Appiah-Kusi, E.; Petros, N.; Wilson, R.; Colizzi, M.; Bossong, M.G.; Valmaggia, L.; Mondelli, V.; McGuire, P.; Bhattacharyya, S. Effects of short-term cannabidiol treatment on response to social stress in subjects at clinical high risk of developing psychosis. Psychopharmacology 2020, 237, 1121–1130. [Google Scholar] [CrossRef]
- Wilson, R.; Bossong, M.G.; Appiah-Kusi, E.; Petros, N.; Brammer, M.; Perez, J.; Allen, P.; McGuire, P.; Bhattacharyya, S. Cannabidiol attenuates insular dysfunction during motivational salience processing in subjects at clinical high risk for psychosis. Transl. Psychiatry 2019, 9, 203. [Google Scholar] [CrossRef] [PubMed]
- Allsop, D.J.; Copeland, J.; Lintzeris, N.; Dunlop, A.J.; Montebello, M.; Sadler, C.; Rivas, G.R.; Holland, R.M.; Muhleisen, P.; Norberg, M.M.; et al. Nabiximols as an agonist replacement therapy during cannabis withdrawal: A randomized clinical trial. JAMA Psychiatry 2014, 71, 281–291. [Google Scholar] [CrossRef]
- Solowij, N.; Broyd, S.J.; Beale, C.; Prick, J.A.; Greenwood, L.M.; Van Hell, H.; Suo, C.; Galettis, P.; Pai, N.; Fu, S.; et al. Therapeutic effects of prolonged cannabidiol treatment on psychological symptoms and cognitive function in regular cannabis users: A pragmatic open-label clinical trial. Cannabis Cannabinoid Res. 2018, 3, 21–34. [Google Scholar] [CrossRef] [PubMed]
- Hurd, Y.L.; Spriggs, S.; Alishayev, J.; Winkel, G.; Gurgov, K.; Kudrich, C.; Oprescu, A.M.; Salsitz, E. Cannabidiol for the reduction of cue-induced craving and anxiety in drug-abstinent individuals with heroin use disorder: A double-blind randomized placebo-controlled trial. Am. J. Psychiatry 2019, 176, 911–922. [Google Scholar] [CrossRef]
- Hundal, H.; Lister, R.; Evans, N.; Antley, A.; Englund, A.; Murray, R.M.; Freeman, D.; Morrison, P.D. The effects of cannabidiol on persecutory ideation and anxiety in a high trait paranoid group. J. Psychopharmacol. 2018, 32, 276–282. [Google Scholar] [CrossRef]
- Leweke, F.M.; Piomelli, D.; Pahlisch, F.; Muhl, D.; Gerth, C.W.; Hoyer, C.; Klosterkötter, J.; Hellmich, M.; Koethe, D. Cannabidiol enhances anandamide signaling and alleviates psychotic symptoms of schizophrenia. Transl. Psychiatry 2012, 2, e94. [Google Scholar] [CrossRef] [PubMed]
- McGuire, P.; Robson, P.; Cubala, W.J.; Vasile, D.; Morrison, P.D.; Barron, R.; Taylor, A.; Wright, S. Cannabidiol (CBD) as an adjunctive therapy in schizophrenia: A multicenter randomized controlled trial. Am. J. Psychiatry 2018, 175, 225–231. [Google Scholar] [CrossRef] [PubMed]
- Boggs, D.L.; Surti, T.; Gupta, A.; Gupta, S.; Niciu, M.; Pittman, B.; Schnakenberg Martin, A.M.; Thurnauer, H.; Davies, A.; D’Souza, D.C.; et al. The effects of cannabidiol (CBD) on cognition and symptoms in outpatients with chronic schizophrenia a randomized placebo controlled trial. Psychopharmacology 2018, 235, 1923–1932. [Google Scholar] [CrossRef]
- Capano, A.; Weaver, R.; Burkman, E. Evaluation of the effects of CBD hemp extract on opioid use and quality of life indicators in chronic pain patients: A prospective cohort study. Postgrad. Med. 2020, 132, 56–61. [Google Scholar] [CrossRef]
- Cuñetti, L.; Manzo, L.; Peyraube, R.; Arnaiz, J.; Curi, L.; Orihuela, S. Chronic pain treatment with cannabidiol in kidney transplant patients in Uruguay. Transplant. Proc. 2018, 50, 461–464. [Google Scholar] [CrossRef]
- Van De Donk, T.; Niesters, M.; Kowal, M.A.; Olofsen, E.; Dahan, A.; Van Velzen, M. An experimental randomized study on the analgesic effects of pharmaceutical-grade cannabis in chronic pain patients with fibromyalgia. Pain 2019, 160, 860–869. [Google Scholar] [CrossRef]
- Russo, M.; Naro, A.; Leo, A.; Sessa, E.; D’Aleo, G.; Bramanti, P.; Calabrò, R.S. Evaluating Sativex® in neuropathic pain management: A clinical and neurophysiological assessment in multiple sclerosis. Pain Med. 2016, 17, 1145–1154. [Google Scholar] [CrossRef]
- Hoggart, B.; Ratcliffe, S.; Ehler, E.; Simpson, K.H.; Hovorka, J.; Lejčko, J.; Taylor, L.; Lauder, H.; Serpell, M. A multicentre, open-label, follow-on study to assess the long-term maintenance of effect, tolerance and safety of THC/CBD oromucosal spray in the management of neuropathic pain. J. Neurol. 2015, 262, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Selvarajah, D.; Gandhi, R.; Emery, C.J.; Tesfaye, S. Randomized placebo-controlled double-blind clinical trial of cannabis-based medicinal product (Sativex) in painful diabetic neuropathy: Depression is a major confounding factor. Diabetes Care 2010, 33, 128–130. [Google Scholar] [CrossRef] [PubMed]
- Lichtman, A.H.; Lux, E.A.; McQuade, R.; Rossetti, S.; Sanchez, R.; Sun, W.; Wright, S.; Kornyeyeva, E.; Fallon, M.T. Results of a double-blind, randomized, placebo-controlled study of nabiximols oromucosal spray as an adjunctive therapy in advanced cancer patients with chronic uncontrolled pain. J. Pain Symptom Manag. 2018, 55, 179–188. [Google Scholar] [CrossRef] [PubMed]
- Miller, I.; Scheffer, I.E.; Gunning, B.; Sanchez-Carpintero, R.; Gil-Nagel, A.; Perry, M.S.; Saneto, R.P.; Checketts, D.; Dunayevich, E.; Knappertz, V. GWPCARE2 Study Group. Dose-Ranging effect of adjunctive oral cannabidiol vs placebo on convulsive seizure frequency in Dravet syndrome: A randomized clinical trial. JAMA Neurol. 2020, 77, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Devinsky, O.; Cross, J.H.; Laux, L.; Marsh, E.; Miller, I.; Nabbout, R.; Scheffer, I.E.; Thiele, E.A.; Wright, S. Cannabidiol in Dravet Syndrome Study Group. Trial of cannabidiol for drug-resistant seizures in the Dravet syndrome. N. Engl. J. Med. 2017, 376, 2011–2020. [Google Scholar] [CrossRef]
- Devinsky, O.; Patel, A.D.; Cross, J.H.; Villanueva, V.; Wirrell, E.C.; Privitera, M.; Greenwood, S.M.; Roberts, C.; Checketts, D.; Van Landingham, K.E.; et al. GWPCARE3 Study Group. Effect of Cannabidiol on drop seizures in the Lennox-Gastaut syndrome. N. Engl. J. Med. 2018, 378, 1888–1897. [Google Scholar] [CrossRef]
- Thiele, E.A.; Marsh, E.D.; French, J.A.; Mazurkiewicz-Beldzinska, M.; Benbadis, S.R.; Joshi, C.; Lyons, P.D.; Taylor, A.; Roberts, C.; GWPCARE4 Study Group; et al. Cannabidiol in patients with seizures associated with Lennox-Gastaut syndrome (GWPCARE4): A randomised, double-blind, placebo-controlled phase 3 trial. Lancet 2018, 391, 1085–1096. [Google Scholar] [CrossRef]
- Wade, D.T.; Makela, P.; Robson, P.; House, H.; Bateman, C. Do cannabis-based medicinal extracts have general or specific effects on symptoms in multiple sclerosis? A double-blind, randomized, placebo-controlled study on 160 patients. Mult. Scler. 2004, 10, 434–441. [Google Scholar] [CrossRef]
- Wade, D.T.; Makela, P.M.; House, H.; Bateman, C.; Robson, P. Long-term use of a cannabis-based medicine in the treatment of spasticity and other symptoms in multiple sclerosis. Mult. Scler. 2006, 12, 639–645. [Google Scholar] [CrossRef]
- Contin, M.; Mancinelli, L.; Perrone, A.; Sabattini, L.; Mohamed, S.; Scandellari, C.; Foschi, M.; Vacchiano, V.; Lugaresi, A.; Riva, R. Tetrahydrocannabinol/cannabidiol oromucosal spray in patients with multiple sclerosis: A pilot study on the plasma concentration-effect relationship. Clin. Neuropharmacol. 2018, 41, 171–176. [Google Scholar] [CrossRef]
- Haupts, M.; Vila, C.; Jonas, A.; Witte, K.; Álvarez-Ossorio, L. Influence of previous failed antispasticity therapy on the efficacy and tolerability of THC:CBD oromucosal spray for multiple sclerosis spasticity. Eur. Neurol. 2016, 75, 236–243. [Google Scholar] [CrossRef] [PubMed]
- Palmieri, B.; Laurino, C.; Vadalà, M. A therapeutic effect of CBD-enriched ointment in inflammatory skin diseases and cutaneous scars. Clin. Ter. 2019, 170, e93–e99. [Google Scholar] [CrossRef]
- Singh, J.; Antimisiaris, M.F. Epidiolex-induced skin rash. Epileptic Disord. 2020, 22, 511–514. [Google Scholar] [CrossRef]
- Černe, K. Toxicological properties of Δ9-tetrahydrocannabinol and cannabidiol. Arh. Ind. Hyg. Toksikol. 2020, 71, 1–11. [Google Scholar] [CrossRef]
- Chesney, E.; McGuire, P.; Freeman, T.P.; Strang, J.; Englund, A. Lack of evidence for the effectiveness or safety of over-the-counter cannabidiol products. Ther. Adv. Psychopharmacol. 2020, 10, 2045125320954992. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, R.G.; Guimarães, F.S.; Crippa, J.; Hallak, J.; Rossi, G.N.; Rocha, J.M.; Zuardi, A.W. Serious adverse effects of cannabidiol (CBD): A review of randomized controlled trials. Expert Opin. Drug Metab. Toxicol. 2020, 16, 517–526. [Google Scholar] [CrossRef]
- Huestis, M.A.; Solimini, R.; Pichini, S.; Pacifici, R.; Carlier, J.; Busardò, F.P. Cannabidiol adverse effects and toxicity. Curr. Neuropharmacol. 2019, 17, 974–989. [Google Scholar] [CrossRef] [PubMed]
- Bass, J.; Linz, D.R. A Case of toxicity from cannabidiol gummy ingestion. Cureus 2020, 12, e7688. [Google Scholar] [CrossRef]
- Herbst, J.; Musgrave, G. Respiratory depression following an accidental overdose of a CBD-labeled product: A pediatric case report. J. Am. Pharm. Assoc. 2020, 60, 248–252. [Google Scholar] [CrossRef]
- Devinsky, O.; Cilio, M.R.; Cross, H.; Fernandez-Ruiz, J.; French, J.; Hill, C.; Katz, R.; Di Marzo, V.; Jutras-Aswad, D.; Notcutt, W.G.; et al. Cannabidiol: Pharmacology and potential therapeutic role in epilepsy and other neuropsychiatric disorders. Epilepsia 2014, 55, 791–802. [Google Scholar] [CrossRef]
- Adapa, S.; Gayam, V.; Konala, V.M.; Annangi, S.; Raju, M.P.; Bezwada, V.; McMillan, C.; Dalal, H.; Mandal, A.; Naramala, S. Cannabis vaping-induced acute pulmonary toxicity: Case series and review of literature. J. Investig. Med. High. Impact Case Rep. 2020, 8, 2324709620947267. [Google Scholar] [CrossRef]
- Millar, S.A.; Stone, N.L.; Bellman, Z.D.; Yates, A.S.; England, T.J.; O’Sullivan, S.E. A systematic review of cannabidiol dosing in clinical populations. Br. J. Clin. Pharmacol. 2019, 85, 1888–1900. [Google Scholar] [CrossRef]
- Arkell, T.R.; Lintzeris, N.; Kevin, R.C.; Ramaekers, J.G.; Vandrey, R.; Irwin, C.; Haber, P.S.; McGregor, I.S. Cannabidiol (CBD) content in vaporized cannabis does not prevent tetrahydrocannabinol (THC)-induced impairment of driving and cognition. Psychopharmacology 2019, 236, 2713–2724. [Google Scholar] [CrossRef]
- Greene, N.Z.; Wiley, J.L.; Yu, Z.; Clowers, B.H.; Craft, R.M. Cannabidiol modulation of antinociceptive tolerance to Δ9-Ktetrahydrocannabinol. Psychopharmacology 2018, 235, 3289–3302. [Google Scholar] [CrossRef]
- Haney, M.; Malcolm, R.J.; Babalonis, S.; Nuzzo, P.A.; Cooper, Z.D.; Bedi, G.; Gray, K.M.; McRae-Clark, A.; Lofwall, M.R.; Sparenborg, S.; et al. Oral cannabidiol does not alter the subjective, reinforcing or cardiovascular effects of smoked Cannabis. Neuropsychopharmacology 2016, 41, 1974–1982. [Google Scholar] [CrossRef] [PubMed]
- Karschner, E.L.; Darwin, W.D.; Goodwin, R.S.; Wright, S.; Huestis, M.A. Plasma cannabinoid pharmacokinetics following controlled oral delta9-tetrahydrocannabinol and oromucosal cannabis extract administration. Clin. Chem. 2011, 57, 66–75. [Google Scholar] [CrossRef] [PubMed]
- Solowij, N.; Broyd, S.; Greenwood, L.M.; Van Hell, H.; Martelozzo, D.; Rueb, K.; Todd, J.; Liu, Z.; Galettis, P.; Martin, J.; et al. A randomised controlled trial of vaporised Δ9-tetrahydrocannabinol and cannabidiol alone and in combination in frequent and infrequent cannabis users: Acute intoxication effects. Eur. Arch. Psychiatry Clin. Neurosci. 2019, 269, 17–35. [Google Scholar] [CrossRef]
- World Health Organization. Cannabidiol (CBD). Critical Review Report. Available online: https://www.who.int/medicines/access/controlled-substances/CannabidiolCriticalReview.pdf (accessed on 1 February 2021).
- Iffland, K.; Grotenhermen, F. An update on safety and side effects of cannabidiol: A review of clinical data and relevant animal studies. Cannabis Cannabinoid Res. 2017, 2, 139–154. [Google Scholar] [CrossRef]
- Patsalos, P.N.; Szaflarski, J.P.; Gidal, B.; Van Landingham, K.; Critchley, D.; Morrison, G. Clinical implications of trials investigating drug-drug interactions between cannabidiol and enzyme inducers or inhibitors or common antiseizure drugs. Epilepsia 2020, 61, 1854–1868. [Google Scholar] [CrossRef] [PubMed]
- Cortopassi, J. Warfarin dose adjustment required after cannabidiol initiation and titration. Am. J. Health Syst. Pharm. 2020, 77, 1846–1851. [Google Scholar] [CrossRef] [PubMed]
- McNamara, N.A.; Dang, L.T.; Sturza, J.; Ziobro, J.M.; Fedak Romanowski, E.M.; Smith, G.C.; Joshi, S.M.; Leber, S.M.; Carlson, M.; Robertson, P.; et al. Thrombocytopenia in pediatric patients on concurrent cannabidiol and valproic acid. Epilepsia 2020, 61, e85–e89. [Google Scholar] [CrossRef]
- Wilson-Morkeh, H.; Al-Abdulla, A.; Sien, L.; Mohamed, H.; Youngstein, T. Important drug interactions exist between cannabidiol oil and commonly prescribed drugs in rheumatology practice. Rheumatology 2020, 59, 249–251. [Google Scholar] [CrossRef]
- Hsu, A.; Painter, N.A. Probable interaction between warfarin and inhaled and oral administration of cannabis. J. Pharm. Pract. 2020, 33, 915–918. [Google Scholar] [CrossRef]
- Dey, T.; Chatterjee, S.; Manna, S.; Nandy, A.; Basak, S.C. Identification and computational analysis of mutations in SARS-CoV-2. Comput. Biol. Med. 2020, 129, 104166. [Google Scholar] [CrossRef] [PubMed]
- Santos, R.; Sampaio, W.O.; Alzamora, A.C.; Motta-Santos, D.; Alenina, N.; Bader, M.; Campagnole-Santos, M.J. The ACE2/Angiotensin-(1-7)/MAS axis of the renin-angiotensin system: Focus on Angiotensin-(1-7). Physiol. Rev. 2018, 98, 505–553. [Google Scholar] [CrossRef] [PubMed]
- Jia, H. Pulmonary angiotensin-converting enzyme 2 (ACE2) and inflammatory lung disease. Shock 2016, 46, 239–248. [Google Scholar] [CrossRef]
- Feng, Y.; Wan, H.; Liu, J.; Zhang, R.; Ma, Q.; Han, B.; Xiang, Y.; Che, J.; Cao, H.; Fei, X.; et al. The angiotensin-converting enzyme 2 in tumor growth and tumor-associated angiogenesis in non-small cell lung cancer. Oncol. Rep. 2010, 23, 941–948. [Google Scholar] [CrossRef]
- Imai, Y.; Kuba, K.; Rao, S.; Huan, Y.; Guo, F.; Guan, B.; Yang, P.; Sarao, R.; Wada, T.; Leong-Poi, H.; et al. Angiotensin-converting enzyme 2 protects from severe acute lung failure. Nature. 2005, 436, 112–116. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Liu, L.; Chen, Q.; Liu, A.; Cai, S.; Yang, Y.; Lu, X.; Qiu, H. Mesenchymal stem cells overexpressing angiotensin-converting enzyme 2 rescue lipopolysaccharide-induced lung injury. Cell. Transplant. 2015, 24, 1699–1715. [Google Scholar] [CrossRef] [PubMed]
- Shenoy, V.; Kwon, K.C.; Rathinasabapathy, A.; Lin, S.; Jin, G.; Song, C.; Shil, P.; Nair, A.; Qi, Y.; Li, Q.; et al. Oral delivery of Angiotensin-converting enzyme 2 and Angiotensin-(1-7) bioencapsulated in plant cells attenuates pulmonary hypertension. Hypertension 2014, 64, 1248–1259. [Google Scholar] [CrossRef]
- Rey-Parra, G.J.; Vadivel, A.; Coltan, L.; Hall, A.; Eaton, F.; Schuster, M.; Loibner, H.; Penninger, J.M.; Kassiri, Z.; Oudit, G.Y.; et al. Angiotensin converting enzyme 2 abrogates bleomycin-induced lung injury. J. Mol. Med. 2012, 90, 637–647. [Google Scholar] [CrossRef]
- Marquez, A.; Wysocki, J.; Pandit, J.; Batlle, D. An update on ACE2 amplification and its therapeutic potential. Acta. Physiol. 2021, 231, e13513. [Google Scholar] [CrossRef] [PubMed]
- Marshall, R.P.; Gohlke, P.; Chambers, R.C.; Howell, D.C.; Bottoms, S.E.; Unger, T.; McAnulty, R.J.; Laurent, G.J. Angiotensin II and the fibroproliferative response to acute lung injury. Am. J. Physiol. Lung. Cell. Mol. Physiol. 2004, 286, L156–L164. [Google Scholar] [CrossRef]
- Tan, W.; Liao, W.; Zhou, S.; Mei, D.; Wong, W.F. Targeting the renin-angiotensin system as novel therapeutic strategy for pulmonary diseases. Curr. Opin. Pharmacol. 2018, 40, 9–17. [Google Scholar] [CrossRef]
- Kuba, K.; Imai, Y.; Ohto-Nakanishi, T.; Penninger, J.M. Trilogy of ACE2: A peptidase in the renin-angiotensin system, a SARS receptor, and a partner for amino acid transporters. Pharmacol. Ther. 2010, 128, 119–128. [Google Scholar] [CrossRef] [PubMed]
- Ingraham, N.E.; Barakat, A.G.; Reilkoff, R.; Bezdicek, T.; Schacker, T.; Chipman, J.G.; Tignanelli, C.J.; Puskarich, M.A. Understanding the renin-angiotensin-aldosterone-SARS-CoV axis: A comprehensive review. Eur. Respir. J. 2020, 56, 2000912. [Google Scholar] [CrossRef]
- Marshall, R.P.; Webb, S.; Bellingan, G.J.; Montgomery, H.E.; Chaudhari, B.; McAnulty, R.J.; Humphries, S.E.; Hill, M.R.; Laurent, G.J. Angiotensin converting enzyme insertion/deletion polymorphism is associated with susceptibility and outcome in acute respiratory distress syndrome. Am. J. Respir. Crit. Care. Med. 2002, 166, 646–650. [Google Scholar] [CrossRef]
- Cruces, P.; Díaz, F.; Puga, A.; Erranz, B.; Donoso, A.; Carvajal, C.; Wilhelm, J.; Repetto, G.M. Angiotensin-converting enzyme insertion/deletion polymorphism is associated with severe hypoxemia in pediatric ARDS. Intensive Care. Med. 2012, 38, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Bourgonje, A.R.; Abdulle, A.E.; Timens, W.; Hillebrands, J.L.; Navis, G.J.; Gordijn, S.J.; Bolling, M.C.; Dijkstra, G.; Voors, A.A.; Osterhaus, A.D.; et al. Angiotensin-converting enzyme 2 (ACE2), SARS-CoV-2 and the pathophysiology of coronavirus disease 2019 (COVID-19). J. Pathol. 2020, 251, 228–248. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.H.; Ou, C.Q.; He, J.X.; Liu, L.; Shan, H.; Lei, C.L.; Hui, D.; et al. Clinical characteristics of coronavirus disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef]
- Sharma, R.K.; Stevens, B.R.; Obukhov, A.G.; Grant, M.B.; Oudit, G.Y.; Li, Q.; Richards, E.M.; Pepine, C.J.; Raizada, M.K. ACE2 (angiotensin-converting enzyme 2) in cardiopulmonary diseases: Ramifications for the control of SARS-CoV-2. Hypertension 2020, 76, 651–661. [Google Scholar] [CrossRef]
- Leung, J.M.; Niikura, M.; Yang, C.; Sin, D.D. COVID-19 and COPD. Eur. Respir. J. 2020, 56, 2002108. [Google Scholar] [CrossRef]
- Bilinska, K.; Butowt., R. Anosmia in COVID-19: A bumpy road to establishing a cellular mechanism. ACS Chem. Neurosci. 2020, 11, 2152–2155. [Google Scholar] [CrossRef]
- Kimura, H.; Francisco, D.; Conway, M.; Martinez, F.D.; Vercelli, D.; Polverino, F.; Billheimer, D.; Kraft, M. Type 2 inflammation modulates ACE2 and TMPRSS2 in airway epithelial cells. J. Allergy. Clin. Immunol. 2020, 146, 80–88. [Google Scholar] [CrossRef]
- Jiang, F.; Yang, J.; Zhang, Y.; Dong, M.; Wang, S.; Zhang, Q.; Liu, F.F.; Zhang, K.; Zhang, C. Angiotensin-converting enzyme 2 and angiotensin 1-7: Novel therapeutic targets. Nat. Rev. Cardiol. 2014, 11, 413–426. [Google Scholar] [CrossRef]
- Zhong, J.; Basu, R.; Guo, D.; Chow, F.L.; Byrns, S.; Schuster, M.; Loibner, H.; Wang, X.H.; Penninger, J.M.; Kassiri, Z.; et al. Angiotensin-converting enzyme 2 suppresses pathological hypertrophy, myocardial fibrosis, and cardiac dysfunction. Circulation 2010, 122, 717–728. [Google Scholar] [CrossRef]
- Patel, V.B.; Bodiga, S.; Fan, D.; Das, S.K.; Wang, Z.; Wang, W.; Basu, R.; Zhong, J.; Kassiri, Z.; Oudit, G.Y. Cardioprotective effects mediated by angiotensin II type 1 receptor blockade and enhancing angiotensin 1-7 in experimental heart failure in angiotensin-converting enzyme 2-null mice. Hypertension 2012, 59, 1195–1203. [Google Scholar] [CrossRef] [PubMed]
- Patel, V.B.; Zhong, J.C.; Grant, M.B.; Oudit, G.Y. Role of the ACE2/Angiotensin 1-7 axis of the renin-angiotensin system in heart failure. Circ. Res. 2016, 118, 1313–1326. [Google Scholar] [CrossRef]
- Donoghue, M.; Wakimoto, H.; Maguire, C.T.; Acton, S.; Hales, P.; Stagliano, N.; Fairchild-Huntress, V.; Xu, J.; Lorenz, J.N.; Kadambi, V.; et al. Heart block, ventricular tachycardia, and sudden death in ACE2 transgenic mice with downregulated connexins. J. Mol. Cell. Cardiol. 2003, 35, 1043–1053. [Google Scholar] [CrossRef]
- Saponaro, F.; Rutigliano, G.; Sestito, S.; Bandini, L.; Storti, B.; Bizzarri, R.; Zucchi, R. ACE2 in the era of SARS-CoV-2: Controversies and novel perspectives. Front. Mol. Biosci. 2020, 7, 588618. [Google Scholar] [CrossRef]
- Oudit, G.Y.; Kassiri, Z.; Patel, M.P.; Chappell, M.; Butany, J.; Backx, P.H.; Tsushima, R.G.; Scholey, J.W.; Khokha, R.; Penninger, J.M. Angiotensin II-mediated oxidative stress and inflammation mediate the age-dependent cardiomyopathy in ACE2 null mice. Cardiovasc. Res. 2007, 75, 29–39. [Google Scholar] [CrossRef] [PubMed]
- Yamamoto, K.; Ohishi, M.; Katsuya, T.; Ito, N.; Ikushima, M.; Kaibe, M.; Tatara, Y.; Shiota, A.; Sugano, S.; Takeda, S.; et al. Deletion of angiotensin-converting enzyme 2 accelerates pressure overload-induced cardiac dysfunction by increasing local angiotensin II. Hypertension 2006, 47, 718–726. [Google Scholar] [CrossRef] [PubMed]
- Burrell, L.M.; Risvanis, J.; Kubota, E.; Dean, R.G.; MacDonald, P.S.; Lu, S.; Tikellis, C.; Grant, S.L.; Lew, R.A.; Smith, A.I.; et al. Myocardial infarction increases ACE2 expression in rat and humans. Eur. Heart. J. 2005, 26, 369–375. [Google Scholar] [CrossRef] [PubMed]
- Kassiri, Z.; Zhong, J.; Guo, D.; Basu, R.; Wang, X.; Liu, P.P.; Scholey, J.W.; Penninger, J.M.; Oudit, G.Y. Loss of angiotensin-converting enzyme 2 accelerates maladaptive left ventricular remodeling in response to myocardial infarction. Circ. Heart. Fail. 2009, 2, 446–455. [Google Scholar] [CrossRef] [PubMed]
- Epelman, S.; Shrestha, K.; Troughton, R.W.; Francis, G.S.; Sen, S.; Klein, A.L.; Tang, W.H. Soluble angiotensin-converting enzyme 2 in human heart failure: Relation with myocardial function and clinical outcomes. J. Card. Fail. 2009, 15, 565–571. [Google Scholar] [CrossRef]
- Li, Y.; Zhou, W.; Yang, L.; You, R. Physiological and pathological regulation of ACE2, the SARS-CoV-2 receptor. Pharmacol. Res. 2020, 157, 104833. [Google Scholar] [CrossRef]
- Hamming, I.; Timens, W.; Bulthuis, M.L.; Lely, A.T.; Navis, G.; van Goor, H. Tissue distribution of ACE2 protein, the functional receptor for SARS coronavirus. A first step in understanding SARS pathogenesis. J. Pathol. 2004, 203, 631–637. [Google Scholar] [CrossRef]
- Lovren, F.; Pan, Y.; Quan, A.; Teoh, H.; Wang, G.; Shukla, P.C.; Levitt, K.S.; Oudit, G.Y.; Al-Omran, M.; Stewart, D.J.; et al. Angiotensin converting enzyme-2 confers endothelial protection and attenuates atherosclerosis. Am. J. Physiol. Heart. Circ. Physiol. 2008, 295, H1377–H1384. [Google Scholar] [CrossRef]
- Sahara, M.; Ikutomi, M.; Morita, T.; Minami, Y.; Nakajima, T.; Hirata, Y.; Nagai, R.; Sata, M. Deletion of angiotensin-converting enzyme 2 promotes the development of atherosclerosis and arterial neointima formation. Cardiovasc. Res. 2014, 101, 236–246. [Google Scholar] [CrossRef]
- Tang, N.; Li, D.; Wang, X.; Sun, Z. Abnormal coagulation parameters are associated with poor prognosis in patients with novel coronavirus pneumonia. J. Thromb. Haemost. 2020, 18, 844–847. [Google Scholar] [CrossRef]
- Bonow, R.O.; Fonarow, G.C.; O’Gara, P.T.; Yancy, C.W. Association of coronavirus disease 2019 (COVID-19) with myocardial injury and mortality. JAMA Cardiol. 2020, 5, 751–753. [Google Scholar] [CrossRef]
- Sawalha, A.H.; Zhao, M.; Coit, P.; Lu, Q. Epigenetic dysregulation of ACE2 and interferon-regulated genes might suggest increased COVID-19 susceptibility and severity in lupus patients. medRxiv 2020. [Google Scholar] [CrossRef]
- Fraga-Silva, R.A.; Sorg, B.S.; Wankhede, M.; Dedeugd, C.; Jun, J.Y.; Baker, M.B.; Li, Y.; Castellano, R.K.; Katovich, M.J.; Raizada, M.K.; et al. ACE2 activation promotes antithrombotic activity. Mol. Med. 2010, 16, 210–215. [Google Scholar] [CrossRef] [PubMed]
- Samavati, L.; Uhal, B.D. ACE2, much more than just a receptor for SARS-COV-2. Front. Cell. Infect. Microbiol. 2020, 10, 317. [Google Scholar] [CrossRef]
- Konturek, P.C.; Harsch, I.A.; Neurath, M.F.; Zopf, Y. COVID-19 - more than respiratory disease: A gastroenterologist’s perspective. J. Physiol. Pharmacol. 2020, 71. [Google Scholar] [CrossRef]
- Hashimoto, T.; Perlot, T.; Rehman, A.; Trichereau, J.; Ishiguro, H.; Paolino, M.; Sigl, V.; Hanada, T.; Hanada, R.; Lipinski, S.; et al. ACE2 links amino acid malnutrition to microbial ecology and intestinal inflammation. Nature 2012, 487, 477–481. [Google Scholar] [CrossRef]
- Garg, M.; Christensen, B.; Lubel, J.S. Gastrointestinal ACE2, COVID-19 and IBD: Opportunity in the face of tragedy? Gastroenterology 2020, 159, 1623–1624. [Google Scholar] [CrossRef] [PubMed]
- Garg, M.; Royce, S.G.; Tikellis, C.; Shallue, C.; Batu, D.; Velkoska, E.; Burrell, L.M.; Patel, S.K.; Beswick, L.; Jackson, A.; et al. Imbalance of the renin-angiotensin system may contribute to inflammation and fibrosis in IBD: A novel therapeutic target? Gut 2020, 69, 841–851. [Google Scholar] [CrossRef]
- Cao, X.; Lu, X.M.; Tuo, X.; Liu, J.Y.; Zhang, Y.C.; Song, L.N.; Cheng, Z.Q.; Yang, J.K.; Xin, Z. Angiotensin-converting enzyme 2 regulates endoplasmic reticulum stress and mitochondrial function to preserve skeletal muscle lipid metabolism. Lipids. Health. Dis. 2019, 18, 207. [Google Scholar] [CrossRef] [PubMed]
- Osterreicher, C.H.; Taura, K.; De Minicis, S.; Seki, E.; Penz-Osterreicher, M.; Kodama, Y.; Kluwe, J.; Schuster, M.; Oudit, G.Y.; Penninger, J.M.; et al. Angiotensin-converting-enzyme 2 inhibits liver fibrosis in mice. Hepatology 2009, 50, 929–938. [Google Scholar] [CrossRef] [PubMed]
- Chai, X.; Hu, L.; Zhang, Y.; Han, W.; Lu, Z.; Ke, A.; Zhou, J.; Shi, G.; Fang, N.; Fan, J.; et al. Specific ACE2 expression in cholangiocytes may cause liver damage after 2019-nCoV infection. medRxiv 2020. [Google Scholar]
- Nadarajah, R.; Milagres, R.; Dilauro, M.; Gutsol, A.; Xiao, F.; Zimpelmann, J.; Kennedy, C.; Wysocki, J.; Batlle, D.; Burns, K.D. Podocyte-specific overexpression of human angiotensin-converting enzyme 2 attenuates diabetic nephropathy in mice. Kidney Int. 2012, 82, 292–303. [Google Scholar] [CrossRef] [PubMed]
- Ortiz-Melo, D.I.; Gurley, S.B. Angiotensin converting enzyme 2 and the kidney. Curr. Opin. Nephrol. Hypertens. 2016, 25, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Cheng, Y.; Luo, R.; Wang, K.; Zhang, M.; Wang, Z.; Dong, L.; Li, J.; Yao, Y.; Ge, S.; Xu, G. Kidney disease is associated with in-hospital death of patients with COVID-19. Kidney Int. 2020, 97, 829–838. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Wu, M.; Yao, J.; Guo, J.; Liao, X.; Song, S.; Li, J.; Duan, G.; Zhou, Y.; Wu, X.; et al. Caution on kidney dysfunctions of COVID-19 patients. medRxiv 2020. [Google Scholar]
- Xia, H.; Lazartigues, E. Angiotensin-converting enzyme 2 in the brain: Properties and future directions. J. Neurochem. 2008, 107, 1482–1494. [Google Scholar] [CrossRef]
- Xiao, L.; Gao, L.; Lazartigues, E.; Zucker, I.H. Brain-selective overexpression of angiotensin-converting enzyme 2 attenuates sympathetic nerve activity and enhances baroreflex function in chronic heart failure. Hypertension 2011, 58, 1057–1065. [Google Scholar] [CrossRef] [PubMed]
- Katsi, V.; Maragkoudakis, S.; Marketou, M.; Tsioufis, C.; Parthenakis, F.; Tousoulis, D. The role of angiotensin-(1-7)/Mas axis and angiotensin type 2 receptors in the central nervous system in cardiovascular disease and therapeutics: A riddle to be solved. Curr. Vasc. Pharmacol. 2019, 17, 319–325. [Google Scholar] [CrossRef]
- Alenina, N.; Bader, M. ACE2 in brain physiology and pathophysiology: Evidence from transgenic animal models. Neurochem. Res. 2019, 44, 1323–1329. [Google Scholar] [CrossRef]
- Kangussu, L.M.; Marzano, L.; Souza, C.F.; Dantas, C.C.; Miranda, A.S.; Simões E Silva, A.C. The renin-angiotensin system and the cerebrovascular diseases: Experimental and clinical evidence. Protein Pept. Lett. 2020, 27, 463–475. [Google Scholar] [CrossRef]
- Gupta, A.; Madhavan, M.V.; Sehgal, K.; Nair, N.; Mahajan, S.; Sehrawat, T.S.; Bikdeli, B.; Ahluwalia, N.; Ausiello, J.C.; Wan, E.Y.; et al. Extrapulmonary manifestations of COVID-19. Nat. Med. 2020, 26, 1017–1032. [Google Scholar] [CrossRef]
- Machhi, J.; Herskovitz, J.; Senan, A.M.; Dutta, D.; Nath, B.; Oleynikov, M.D.; Blomberg, W.R.; Meigs, D.D.; Hasan, M.; Patel, M.; et al. The natural history, pathobiology, and clinical manifestations of SARS-CoV-2 infections. J. Neuroimmune Pharmacol. 2020, 15, 359–386. [Google Scholar] [CrossRef]
- Zhou, Z.; Kang, H.; Li, S.; Zhao, X. Understanding the neurotropic characteristics of SARS-CoV-2: From neurological manifestations of COVID-19 to potential neurotropic mechanisms. J. Neurol. 2020, 267, 2179–2184. [Google Scholar] [CrossRef] [PubMed]
- Najjar, S.; Najjar, A.; Chong, D.J.; Pramanik, B.K.; Kirsch, C.; Kuzniecky, R.I.; Pacia, S.V.; Azhar, S. Central nervous system complications associated with SARS-CoV-2 infection: Integrative concepts of pathophysiology and case reports. J. Neuroinflamm. 2020, 17, 231. [Google Scholar] [CrossRef]
- Holappa, M.; Vapaatalo, H.; Vaajanen, A. Local ocular renin-angiotensin-aldosterone system: Any connection with intraocular pressure? A comprehensive review. Ann. Med. 2020, 52, 191–206. [Google Scholar] [CrossRef]
- Holappa, M.; Vapaatalo, H.; Vaajanen, A. Many faces of renin-angiotensin system - Focus on eye. Open. Ophthalmol. J. 2017, 11, 122–142. [Google Scholar] [CrossRef]
- Verma, A.; Shan, Z.; Lei, B.; Yuan, L.; Liu, X.; Nakagawa, T.; Grant, M.B.; Lewin, A.S.; Hauswirth, W.W.; Raizada, M.K.; et al. ACE2 and Ang-(1-7) confer protection against development of diabetic retinopathy. Mol. Ther. 2012, 20, 28–36. [Google Scholar] [CrossRef] [PubMed]
- Zhu, P.; Verma, A.; Prasad, T.; Li, Q. Expression and function of Mas-related G Protein-Coupled Receptor D and its ligand alamandine in retina. Mol. Neurobiol. 2020, 57, 513–527. [Google Scholar] [CrossRef]
- Duan, Y.; Beli, E.; Li Calzi, S.; Quigley, J.L.; Miller, R.C.; Moldovan, L.; Feng, D.; Salazar, T.E.; Hazra, S.; Al-Sabah, J.; et al. Loss of angiotensin-converting enzyme 2 exacerbates diabetic retinopathy by promoting bone marrow dysfunction. Stem. Cells. 2018, 36, 1430–1440. [Google Scholar] [CrossRef] [PubMed]
- Verma, A.; Xu, K.; Du, T.; Zhu, P.; Liang, Z.; Liao, S.; Zhang, J.; Raizada, M.K.; Grant, M.B.; Li, Q. Expression of human ACE2 in Lactobacillus and beneficial effects in diabetic retinopathy in mice. Mol. Ther. Methods. Clin. Dev. 2019, 14, 161–170. [Google Scholar] [CrossRef]
- Lazartigues, E.; Qadir, M.; Mauvais-Jarvis, F. Endocrine significance of SARS-CoV-2’s reliance on ACE2. Endocrinology 2020, 161, bqaa108. [Google Scholar] [CrossRef] [PubMed]
- Niu, M.J.; Yang, J.K.; Lin, S.S.; Ji, X.J.; Guo, L.M. Loss of angiotensin-converting enzyme 2 leads to impaired glucose homeostasis in mice. Endocrine 2008, 34, 56–61. [Google Scholar] [CrossRef]
- Liu, F.; Long, X.; Zhang, B.; Zhang, W.; Chen, X.; Zhang, Z. ACE2 expression in pancreas may cause pancreatic damage after SARS-CoV-2 infection. Clin. Gastroenterol. Hepatol. 2020, 18, 2128–2130.e2. [Google Scholar] [CrossRef] [PubMed]
- Gupte, M.; Thatcher, S.E.; Boustany-Kari, C.M.; Shoemaker, R.; Yiannikouris, F.; Zhang, X.; Karounos, M.; Cassis, L.A. Angiotensin converting enzyme 2 contributes to sex differences in the development of obesity hypertension in C57BL/6 mice. Arterioscler. Thromb. Vasc. Biol. 2012, 32, 1392–1399. [Google Scholar] [CrossRef]
- Shoemaker, R.; Tannock, L.R.; Su, W.; Gong, M.; Gurley, S.B.; Thatcher, S.E.; Yiannikouris, F.; Ensor, C.M.; Cassis, L.A. Adipocyte deficiency of ACE2 increases systolic blood pressures of obese female C57BL/6 mice. Biol. Sex. Differ. 2019, 10, 45. [Google Scholar] [CrossRef] [PubMed]
- Pan, P.P.; Zhan, Q.T.; Le, F.; Zheng, Y.M.; Jin, F. Angiotensin-converting enzymes play a dominant role in fertility. Int. J. Mol. Sci. 2013, 14, 21071–21086. [Google Scholar] [CrossRef]
- Shan, T.; Shang, W.; Zhang, L.; Zhao, C.; Chen, W.; Zhang, Y.; Li, G. Effect of angiotensin-(1-7) and angiotensin II on the proliferation and activation of human endometrial stromal cells in vitro. Int. J. Clin. Exp. Pathol. 2015, 8, 8948–8957. [Google Scholar]
- Domińska, K. Involvement of ACE2/Ang-(1-7)/MAS1 axis in the regulation of ovarian function in mammals. Int. J. Mol. Sci. 2020, 21, 4572. [Google Scholar] [CrossRef]
- Reis, F.M.; Reis, A.M. Angiotensin-converting enzyme 2 (ACE2), angiotensin-(1-7) and Mas receptor in gonadal and reproductive functions. Clin. Sci (Lond). 2020, 134, 2929–2941. [Google Scholar] [CrossRef] [PubMed]
- Honorato-Sampaio, K.; Andrade, R.F.; Bader, M.; Martins, A.S.; Santos, R.; Reis, A.M. Genetic deletion of the Angiotensin-(1-7) receptor Mas leads to a reduced ovulatory rate. Peptides 2018, 107, 83–88. [Google Scholar] [CrossRef]
- Jing, Y.; Run-Qian, L.; Hao-Ran, W.; Hao-Ran, C.; Ya-Bin, L.; Yang, G.; Fei, C. Potential influence of COVID-19/ACE2 on the female reproductive system. Mol. Hum. Reprod. 2020, 26, 367–373. [Google Scholar] [CrossRef]
- Palumbo, A.; Ávila, J.; Naftolin, F. The Ovarian Renin-Angiotensin System (OVRAS): A Major Factor in Ovarian Function and Disease. Reprod. Sci. 2016, 23, 1644–1655. [Google Scholar] [CrossRef]
- Dhaundiyal, A.; Kumari, P.; Jawalekar, S.S.; Chauhan, G.; Kalra, S.; Navik, U. Is highly expressed ACE 2 in pregnant women “a curse” in times of COVID-19 pandemic? Life Sci. 2021, 264, 118676. [Google Scholar] [CrossRef]
- Gilbert, J.S.; LaMarca, B.B.; Granger, J.P. ACE2 and ANG-(1-7) in the gravid uterus: The new players on the block. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2008, 294, R915–R916. [Google Scholar] [CrossRef]
- Bharadwaj, M.S.; Strawn, W.B.; Groban, L.; Yamaleyeva, L.M.; Chappell, M.C.; Horta, C.; Atkins, K.; Firmes, L.; Gurley, S.B.; Brosnihan, K.B.; et al. Angiotensin-converting enzyme 2 deficiency is associated with impaired gestational weight gain and fetal growth restriction. Hypertension 2011, 58, 852–858. [Google Scholar] [CrossRef]
- Yamaleyeva, L.M.; Pulgar, V.M.; Lindsey, S.H.; Yamane, L.; Varagic, J.; McGee, C.; daSilva, M.; Lopes Bonfa, P.; Gurley, S.B.; Brosnihan, K.B. Uterine artery dysfunction in pregnant ACE2 knockout mice is associated with placental hypoxia and reduced umbilical blood flow velocity. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E84–E94. [Google Scholar] [CrossRef]
- Lumbers, E.R.; Delforce, S.J.; Pringle, K.G.; Smith, G.R. The lung, the heart, the novel coronavirus, and the renin-angiotensin system; The need for clinical trials. Front. Med. 2020, 7, 248. [Google Scholar] [CrossRef]
- Pascolo, L.; Zito, G.; Zupin, L.; Luppi, S.; Giolo, E.; Martinelli, M.; De Rocco, D.; Crovella, S.; Ricci, G. Renin angiotensin system, COVID-19 and male fertility: Any risk for conceiving? Microorganisms 2020, 8, 1492. [Google Scholar] [CrossRef]
- Nozato, S.; Yamamoto, K.; Takeshita, H.; Nozato, Y.; Imaizumi, Y.; Fujimoto, T.; Yokoyama, S.; Nagasawa, M.; Takeda, M.; Hongyo, K.; et al. Angiotensin 1-7 alleviates aging-associated muscle weakness and bone loss, but is not associated with accelerated aging in ACE2-knockout mice. Clin. Sci. 2019, 133, 2005–2018. [Google Scholar] [CrossRef] [PubMed]
- Ferrandi, P.J.; Alway, S.E.; Mohamed, J.S. The interaction between SARS-CoV-2 and ACE2 may have consequences for skeletal muscle viral susceptibility and myopathies. J. Appl. Physiol. 2020, 129, 864–867. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.; Tong, Q. Rhabdomyolysis as potential late complication associated with COVID-19. Emerg. Infect. Dis. 2020, 26, 1618–1620. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, W.O.; Nascimento, A.A.; Santos, R.A. Systemic and regional hemodynamic effects of angiotensin-(1-7) in rats. Am. J. Physiol. Heart. Circ. Physiol. 2003, 284, H1985–H1994. [Google Scholar] [CrossRef] [PubMed]
- Krueger, J.G.; Murrell, D.F.; Garcet, S.; Navrazhina, K.; Lee, P.C.; Muscianisi, E.; Blauvelt, A. Secukinumab lowers expression of ACE2 in affected skin of patients with psoriasis. J. Allergy Clin. Immunol. 2020. [Google Scholar] [CrossRef] [PubMed]
- Xue, X.; Mi, Z.; Wang, Z.; Pang, Z.; Liu, H.; Zhang, F. High expression of ACE2 on keratinocytes reveals skin as a potential target for SARS-CoV-2. J. Invest. Dermatol. 2021, 41, 206–209.e1. [Google Scholar] [CrossRef] [PubMed]
- Garduño-Soto, M.; Choreño-Parra, J.A.; Cazarin-Barrientos, J. Dermatological aspects of SARS-CoV-2 infection: Mechanisms and manifestations. Arch. Dermatol. Res. 2020, 1–12. [Google Scholar] [CrossRef]
- Tikellis, C.; Thomas, M.C. Angiotensin-converting enzyme 2 (ACE2) is a key modulator of the renin angiotensin system in health and disease. Int. J. Pept. 2012, 2012, 256294. [Google Scholar] [CrossRef] [PubMed]
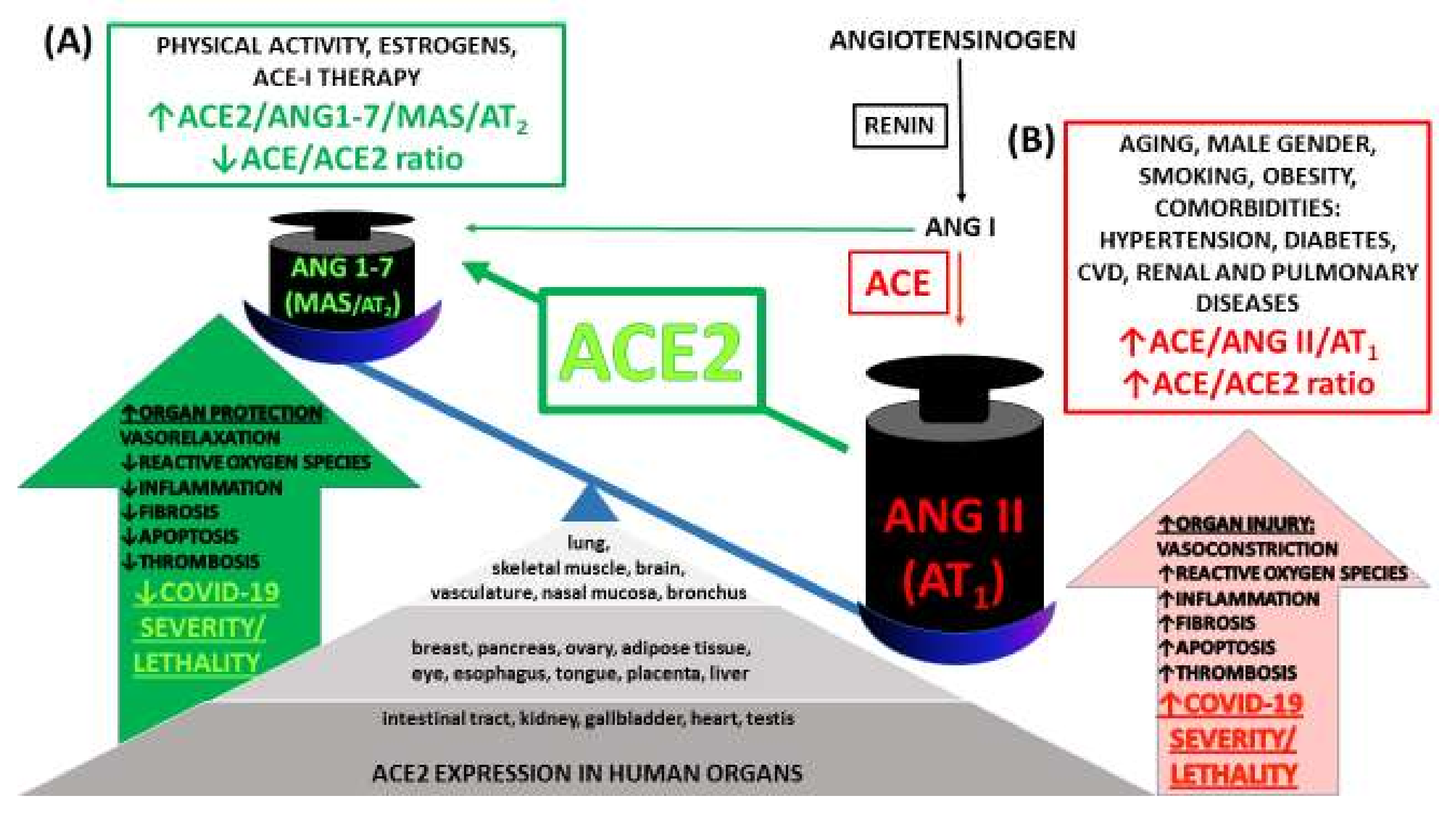
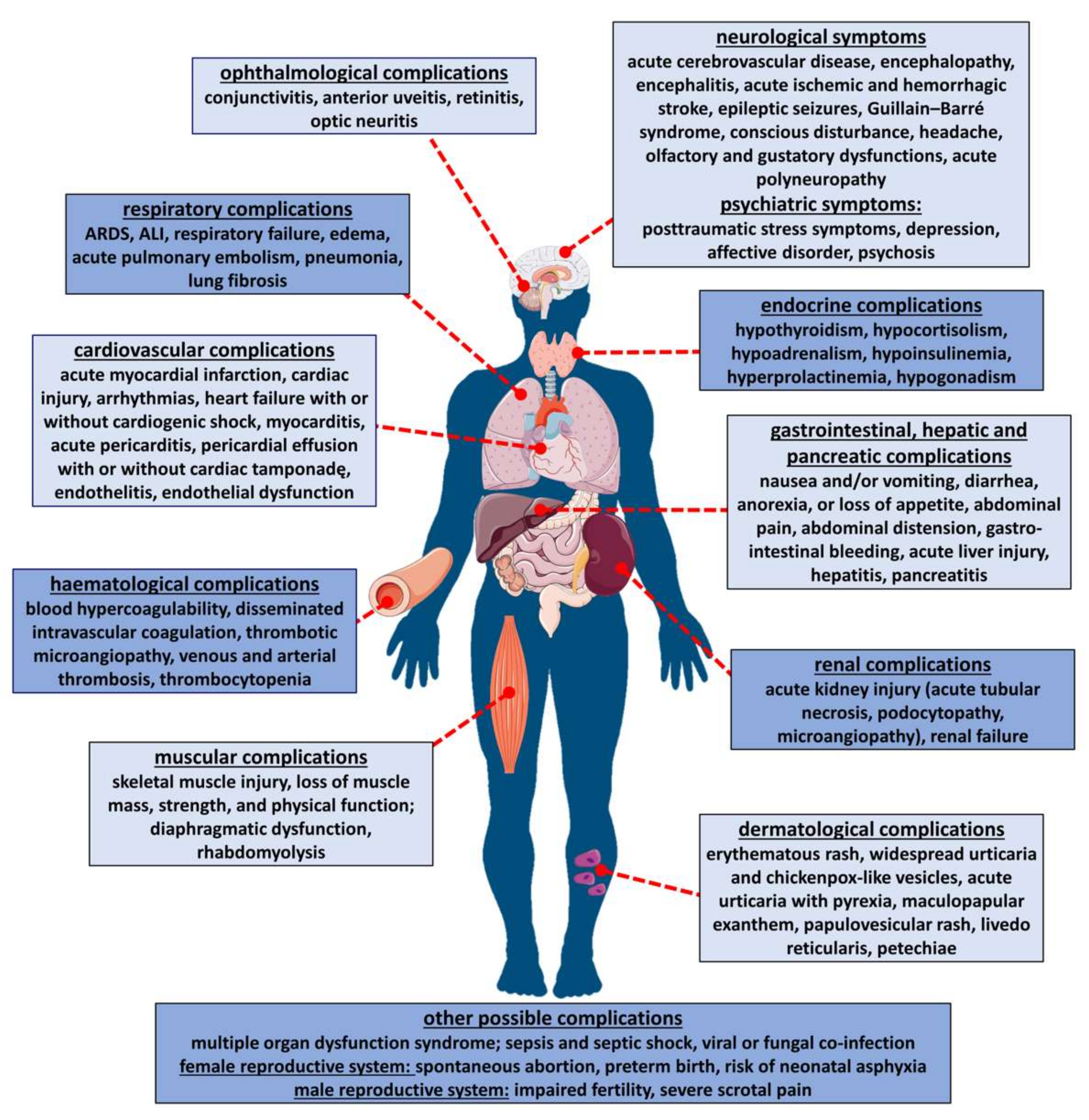
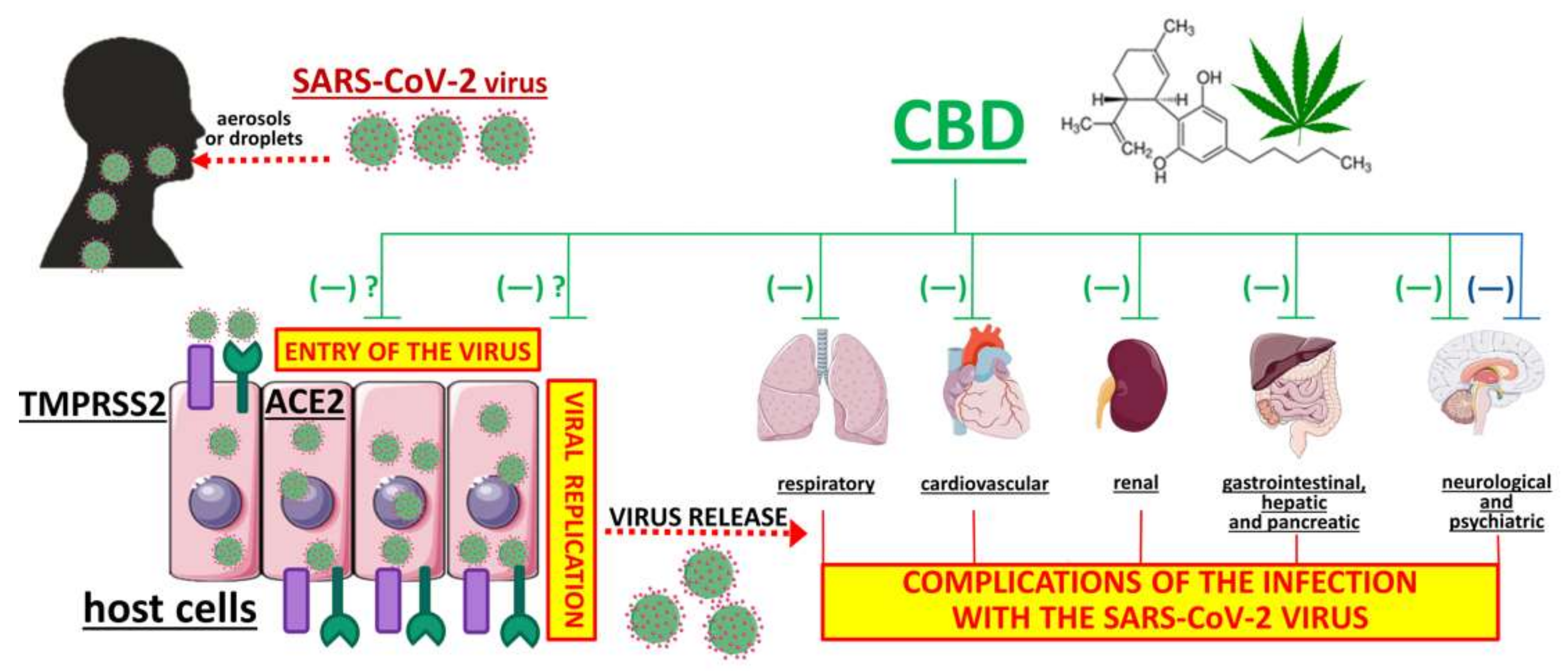
| Model | Species | Dose and Route of Administration of CBD | Effects | CBD Properties Important for COVID; Mechanisms | References | |
|---|---|---|---|---|---|---|
| Respiratory | ARDS induced by poly(I:C) | mouse | 5 mg/kg, i.p. therapeutic, once a day for 3 days | cytokine storm and ARDS symptoms totally or partially improved (blood oxygen saturation, perivascular and peribronchiolar interstitial inflammatory infiltrate, lung fibrosis, hypertrophy and pulmonary edema) ↓IL-6 expression and ↓neutrophil frequency in the lung | anti-inflammatory | [71,72] |
| 5 mg/kg, i.p. therapeutic, once a day for 3 days | improvement of lung structure ↓T cells and ↑neutrophils returned towards the normal level,↑expression of apelin in the blood | anti-inflammatory regulation of apelin level | [72] | |||
| ALI induced by LPS | mouse | 1–80 mg/kg, i.p. 20 mg/kg, i.p. prophylactic; one dose before ALI induction (effects determined 1, 2 and 4 days after LPS) | lungs:↓leukocyte count; ↓leukocyte migration into lungs; ↓MPO activity; ↓vascular permeability; BALF: ↓pro-inflammatory cytokines (TNF-α, IL-6) and chemokines (MCP-1, MIP-2) | anti-inflammatory; partially dependent on adenosine A2A-Rs | [75] | |
| 20 or 80 mg/kg, i.p. therapeutic; one dose 6 h after ALI induction (effects determined 24 h after LPS) | lungs: function improved: ↓resistance; ↓tissue damping and stiffness; ↓leukocyte migration into lungs; ↓MPO activity; ↓vascular permeability; BALF: ↓pro-inflammatory cytokines (TNF-α, IL-6) and chemokines (MCP-1, MIP-2) | anti-inflammatory; antagonists not used | [76] | |||
| lung inflammation induced by LPS | mouse | 75 mg/kg, p.o. prophylactic; once a day for 3 days; LPS 1 h before the last dose of CBD | lungs: ↑inflammatory changes in tissue; BALF: ↑pro-inflammatory TNF-α, IL-6, IL-23, GCSF; slight ↑inflammatory cells | pro-inflammatory; antagonists not used | [83] | |
| pulmonary hypertension due to monocrotaline | rat | 10 mg/kg, i.p. prophylactic; once a day for 21 days | heart: ↓right ventricular systolic pressure; ↔ right hypertrophy and lung edema; ↑blood oxygen saturation; plasma: ↓leukocytes | ↑blood oxygen saturation; antagonists not used | [77] | |
| lung injury induced by brain hypoxic–ischemic damage | newborn piglets | 1 mg/kg, i.v. therapeutic; one dose 30 min after lung injury | improvement of gas exchange; ↑TLC, lungs: ↓ histological damage and edema; ↓leukocyte migration into lungs, ↓inflammatory changes; ↓vascular permeability; BALF: ↓pro-inflammatory cytokines (IL-1) | anti-inflammatory 5-HT1A-Rs in all parameters, except for improvement of gas exchange; site of CBD action (brain and/or lungs) unclear | [78] | |
| asthma induced by ovalbumin | rat | 5 mg/kg, i.p. therapeutic; once a day for 2 days | serum: ↓ IL-4, IL-5, IL-6, IL-13 and TNF-α; ↔ IL-10 | anti-inflammatory; antagonists not used | [79] | |
| mouse | 5 or 10 mg/kg, i.p. therapeutic; once a day for 3 days | ↓airway resistance; ↓alveolar collapse areas; ↓collagen in airways and alveolar septa; lung and BALF: ↓pro-inflammatory cytokines (IL-4, IL-5, IL-13) | anti-inflammatory; anti-fibrotic; airway resistance: CB1-Rs other effects: CB1/2-Rs | [80] | ||
| Respiratory | airway obstruction induced by ovalbumin | guinea- pig | 1 mg/kg, i.v. prophylactic; one dose | ↓airway obstruction induced by ovalbumin | bronchoprotective; reduction of the antigen-induced contractile responses | [81] |
| cough induced by aerosolized citric acid | guinea- pig | prophylactic; aerosolized solution of 10 mg/mL for 20 minutes using a nebulizer | cough inhibition only in three out of eight animals | antagonists not used | [84] | |
| isolated trachea | guinea- pig | 10 μM | ↔ resting basal tension; ↔ contractions induced electrically or by methacholine | antagonists not used | [84] | |
| isolated human pulmonary artery | human | 0.1–30 μM | almost full concentration-dependent vasorelaxation | endothelium-dependent vasodilatation mediated via K+ channels, IP, EP4, TRPV1 and PPARγ receptors | [82] | |
| Cardiovascular | myocardial infarction induced by left coronary artery occlusion | rabbit | 0.1 mg/kg, i.v. prophylactic; one dose before occlusion and one before reperfusion | heart: ↓infarct area; ↑left ventricular function; ↑blood supply to perfusion-defective region; ↓neutrophil infiltration;↓MPO activity plasma: ↓cardiac troponin I | anti-inflammatory; cardioprotective; potentiallyanti-ischemic; antagonists not used | [85] |
| rat | 5 mg/kg, i.p. prophylactic before occlusion and once a day thereafter for 7 days | heart:↓infarct size; ↔HR; ↓leukocyte infiltration; serum: ↓IL-6 CRP, TNF-α | anti-inflammatory; cardioprotective; potentially anti-ischemic; antagonists not used | [86] | ||
| 50 μg/kg, i.v. prophylactic; one dose before occlusion | heart: ↓infarct area; ↓arrhythmias; ↔HR | antiarrhythmic; cardioprotective; antagonists not used | [87] | |||
| 50 μg/kg, i.v. prophylactic; one dose before reperfusion | heart: ↓infarct area; ↔arrhythmias | |||||
| 50 μg/kg, i.v. prophylactic; one dose before occlusion | heart: ↓arrhythmias; ↔HR | antiarrhythmic; potential involvement of A1-Rs | [88] | |||
| autoimmune myocarditis | mouse | 10 mg/kg, i.p. therapeutic; once a day for 46 days | heart: improved systolic and diastolic dysfunction and myocardial stiffness; ↓left ventricular inflammatory changes; ↓necrosis; oxidative stress; ↓ fibrosis | anti-inflammatory; anti-oxidative; anti-fibrotic; cardioprotective antagonists not used | [89] | |
| diabetic cardiomyopathy induced by streptozotocin | mouse | 1, 10 or 20 mg/kg, i.p. therapeutic; once a day for 4 or 11 weeks | heart: ↑diastolic and systolic left ventricular function; ↓oxidative and nitrative stress; ↓inflammation and NF-κB activation; ↓ fibrosis; ↓expression of pro-fibrotic genes | anti-inflammatory;antioxidative; anti-nitrative; anti-fibrotic; cardioprotective; antagonists not used | [90] | |
| changes in vascular endothelium function in Zucker diabetic fatty | rat | 10 mg/kg, i.p. therapeutic for 7 days | mesenteric arteries: ↑endothelium-dependent vasorelaxation due to COX- or NO-mediated mechanisms; serum: ↓cardiovascular biomarkers (C-peptide, insulin and intracellular adhesion molecule-1); ↔glucose, body weight | vasoprotective; improvement in the profile of cardiovascular and metabolic parameters | [93] | |
| Cardiovasc. | hypertension: primary (SHR); secondary (DOCA-salt) | rat | 10 mg/kg, i.p. therapeutic; once a day for 2 weeks | ↔blood pressure, HR; heart: ↓oxidative stress; ↓carbachol-induced coronary constriction; ↓left ventricular cardiomyocyte width; ↔left ventricular hypertrophy | anti-oxidative; antagonists not used | [91,92] |
| Hematological | myocardial infarction induced by left coronary artery occlusion | rat | - 50 μg/kg, i.v. before occlusion - 50 μg/kg, i.v. before reperfusion | ↓platelet aggregation ↔platelet aggregation | antagonists not used | [87] |
| pulmonary hypertension due to monocrotaline | rat | 10 mg/kg, i.p. prophylactic; once a day for 21 days | plasma: ↓ t-PA and PAI-1 | antagonists not used | [77] | |
| pharmacologically induced thrombus in ear venules | mouse | 5 mg/kg, i.p. prophylactic; acute: one dose 30 min before thrombus induction; chronic for 3 days | acute: ↔vessel thrombogenesis chronic: ↔vessel thrombogenesis | antagonists not used | [94] | |
| platelet aggregation | human | in vitro, 0.1—10 μM | ↔resting platelets; ↔activation induced by thrombin receptor activating peptide | |||
| Renal | acute kidney injury induced by renal ischemia/reperfusion | rat | 5 mg/kg, i.v. two doses before occlusion and after reperfusion | kidney:↓tubular necrosis and dilatation; ↓inflammatory changes; ↓NF-κB, COX-2, TNF-α and iNOS; serum: ↓oxidative and nitrative stress | anti-inflammatory; anti-oxidative;anti-nitrative;nephroprotective; antagonists not used | [95] |
| 5 mg/kg, i.a. one dose after occlusion | kidney:↓MPO activity; ↓IL-1, TNF-α and NO levels; ↓lipid and protein oxidative damage; ↔nitrite/nitrate levels | anti-inflammatory; anti-oxidative; nephroprotective; CB1-Rs and CB2-Rs expression unaltered | [96] | |||
| mouse | 10 mg/kg i.p. one dose before reperfusion | kidney: pattern of innate lymphoid cells restored to control values | nephroprotective; antagonists not used | [97] | ||
| Gastrointestinal | nausea or vomiting induced by lithium chloride | rat shrew | prophylactic; acute: 5 and 20 mg/kg, s.c. chronic: 5 mg/kg s.c. for 7 days | acute and chronic: ↓ nausea and/or vomiting | anti-nausea, antiemetic; 5-HT1A-Rs | [98] |
| hepatic ischemia/reperfusion injury induced by hepatic artery and portal vein occlusion | mouse | 3 or 10 mg/kg, i.p. prophylactic; one dose before reocclusion or 90 min after | serum: ↓AST and ALT; liver: ↓inflammatory changes; ↓cell apoptosis (10 mg/kg only); ↓pro-inflammatory cytokines; ↓oxidative and nitrative stress (10 mg/kg only); ↓neutrophil migration to liver tissue | anti-inflammatory; antioxidant; anti-nitrative; hepatoprotective; independent of CB2-Rs | [100] | |
| liver injury and steatosis induced by chronic ethanol administration | mouse | 5 or 10 mg/kg, i.p. prophylactic for 11 days during the ethanol exposure | serum: ↓AST and ALT; liver: ↓inflammatory changes; ↓pro-inflammatory chemokines; ↓neutrophil accumulation; ↓oxidative burst of neutrophils; ↓ oxidative and nitrative stress | anti-inflammatory; anti-oxidative; hepatoprotective; antagonists not used | [101] | |
| Gastrointestinal | hepatic encephalopathy due to thioacetamide | mouse | 5 mg/kg, i.p. therapeutic; one dose after thioacetamide injection | liver: ↔necrosis; plasma: ↓ALT and AST, ammonia and bilirubin | partly hepatoprotective; antagonists not used | [102] |
| acute hepatic toxicity induced by cocaine | mouse | 30, 60 and 90 mg/kg, i.p. prophylactic; acute 30 min before cocaine injection | liver: ↓acute inflammation and damage (↓histological changes) serum: ↓ALT; ↓acute behavioral seizure | anti-inflammatory; FAAH inhibitor did not modify cocaine-induced changes in liver | [103] | |
| acute pancreatitis induced by cerulein | mouse | 0.5 mg/kg, i.p. prophylactic; 8 doses (2 before and 6 simultaneously with cerulein) | pancreas: ↓pathological changes, ↓MPO activity in pancreas tissue; plasma: ↓amylase and lipase; ↓Il-6 and TNF-α | anti-inflammatory; pancreas-protective; possibly via GPR55 (presence in pancreas) | [104] | |
| inflammation and intestinal hypermotility induced by croton oil | mouse | 5 and 10 mg/kg, i.p. therapeutic; one dose to mice with inflammation | intestine:↓hypermotility | involvement of CB1-Rs (but not CB2-Rs) and FAAH | [105] | |
| colitis induced by intracolonic dinitrobenzensulfonic acid | mouse | 5–30 mg/kg, i.p. and 10–60 mg/kg, p.o. therapeutic for 3 days after colitis induction | intestine:↓hypermotility; ↔colitis; ↓colon weight and MPO activity | anti-inflammatory; antagonists not used | [106] | |
| Neurological | chronic unpredictable mild stress model of depression | rat | 10 mg/kg, i.p. prophylactic for 28 days | higher rate of body weight gain and sucrose preference compared to controls | prohedonic; antagonists not used | [107] |
| various acute seizure models | rat mouse | one different i.v. dose dependent on the model | acute antiseizure activity | antiseizure activity | [109] | |
| status epilepticus—spontaneous recurrent seizures (RISESRS) model | rat | 200 mg/kg for 7 weeks | ↓seizure burden and motor comorbidities; reversal of the epilepsy-induced cognitive deficits | |||
| seizure induced by pentylene-tetrazole | mouse | 60 mg/kg, i.p. prophylactic; once before induction of epileptic attack | ↓seizure duration; ↓EEG changes | anti-convulsant; CB1, CB2 and TRPV1 receptors | [110] | |
| hepatic encephalopathy induced by thioacetamide | mouse | 5 mg/kg, i.p. therapeutic; one dose after thioacetamide injection | ↑neurological and cognitive functions; ↑activity; ↓activated astrocytes | procognitive; neuroprotective; antagonists not used | [102] | |
| sepsis-related encephalitis induced by LPS | mouse | 3 mg/kg, i.v. one dose simultaneously with LPS | ↑integrity of blood–brain barrier; ↓leukocyte margination in brain vessels; ↔level of oxidative stress; ↓TNF-α and COX-2 | anti-inflammatory; neuroprotective | [113] | |
| Neurological | perinatal hypoxic-ischemic encephalopathy induced by occlusion of carotid arteries | newborn piglet | 1 mg/kg, i.v. therapeutic; one dose 30 min after induction of brain injury | brain: ↓EEG changes; ↓neuronal mortality; ↓excitotoxicity; ↓IL-1; ↓ oxidative stress | anti-inflammatory; anti-oxidative; neuroprotective partially dependent on 5-HT1A-Rs and CB2-Rs ↔brain endocannabinoid levels | [111] |
| 1 mg/kg, i.v. therapeutic; one dose after induction of brain injury | brain: ↑activity (EEG); ↓neuronal mortality; ↓excitotoxicity; ↓oxidative stress; ↓TNF-α; effects on excitotoxicity, oxidative stress and TNF-α additive to those of hypothermia | anti-inflammatory; anti-oxidative; neuroprotective; antagonists not used | [112] | |||
| cerebral ischemia (stroke model) induced by MCA occlusion | mouse | 0.1; 1; 3 mg/kg, i.p. two doses (before and after occlusion) | brain: ↑neurological function and motor coordination; dose-dependent ↓infarct area ↓MPO activity and ↑CBF (tested only at 3 mg/kg); blood: ↔pCO2, pO2 | anti-inflammatory; neuroprotective; partially dependent on 5-HT1A-Rs independent of CB1, CB2 and TRPV1 receptors | [114,115] | |
| encephalomyelitis induced by TMEV | mouse | 180 mg/kg, i.p. twice daily starting 2 days before (prophylactic) or 3 days after infection (therapeutic) | ↓acute behavioral seizures from 5 days (prophylactic) and 6 days after infection onward (therapeutic) | anti-inflammatory, anti-oxidative (not confirmed experimentally) | [73] | |
| multiple sclerosis induced by TMEV | mouse | 5 mg/kg, i.p. therapeutic, once a day for 7 days | sub chronic effects (after 8 days): ↓transmigration of leukocytes to the nervous parenchyma by downregulating the expression of VCAM-1, CCL2 and CCL5 and the proinflammatory cytokine IL-1β and by attenuating the activation of microglia | anti-inflammatory partial involvement of adenosine A2A-Rs (experiments with an appropriate antagonist) | [74] | |
| same treatment for 10 days | chronic effects (after 80 days): improvement of motor deficits ↓microglial activation and pro-inflammatory cytokine production | |||||
| Eye | retinal inflammation due to LPS | rat | 1 mg/kg, i.p. prophylactic before LPS treatment | ↓retinal TNF-α levels | anti-inflammatory; due to A2-Rs but not A1-Rs | [117] |
| Endocrine | high-fat diet- induced obesity | rat | 10 mg/kg, i.p. prophylactic for 2 weeks | plasma: ↓insulin; skeletal muscle: improved insulin signal transduction and glycogen recovery | ↓lipotoxicity, leading to insulin-sensitization in myocytes; ↓expression of CB1, CB2, TRPV1 and 5-HT1A receptors | [118] |
| type 1 diabetes by streptozotocin submitted to chronic cerebral hypoperfusion | rat | 10 mg/kg, i.p. 30 min before and for 30 days after cerebral hypoperfusion surgery | ↓body weight; plasma:↓hyperglycemia; ↑insulinemia; ↓AGEs and fructosamine; ↓dyslipidemia (LDL, HDL, TGs and total cholesterol levels); ↓AST and ALT; ↑memory performance | improvement of metabolic dysfunction; hepatoprotective; neuroprotective; anti-inflammatory | [119] | |
| Muscular | Duchenne muscular dystrophy caused by dystrophin deficiency | mouse | 60 mg/kg, i.p., therapeutic; three times a week for 2 weeks | muscle: prevention of the functionality loss and tissue degeneration; restoration of locomotor activity; ↓inflammation (IL-6, TNF-α); muscle strength and autophagy restored | anti-inflammatory; (involvement of TRP channels—based on in vitro experiments) | [121] |
| Skin | skin irradiated with UVA/UVB | nude rat | 2.5 g in 100 g petrolatum applied to the back of rats every 12 h for 4 weeks | ↓UV-induced changes in inflammation; apoptosis and oxidative stress | prevention of UV-induced metabolic changes in epidermal keratinocytes | [122] |
| cultured human sebocytes and human skin organ culture | human | 10 μM | ↓lipogenic actions of arachidonic acid and a combination of linoleic acid and testosterone; suppression of sebocyte proliferation | anti-inflammatory (adenosine A2aRs); sebostatic; lipostatic; antiproliferative (TRP4) | [123] | |
| experimental model of allergic contact dermatitis in keratinocytes | human | 5, 10, 20 μM | inhibition of polyinosinic-polycytidylic acid-induced release of MCP-2, IL-6, IL-8 and TNF-α; no cytotoxic effect | anti-inflammatory via CB2 and TRPV1 receptors | [124] | |
| Other | sepsis induced by cecal ligation and puncture | rat | 10 mg/kg, i.p. Acute—therapeutic; one dose (after sepsis induction) | ↓lipid peroxidation in lung, heart and kidney; ↓oxidative protein damage in spleen, liver and heart; ↓oxidative damage of proteins in striatum, cortex and hippocampus; ↑oxidative damage of proteins in lung | anti-oxidative; organo-protective; pro-oxidative in lungs | [125] |
| 10 mg/kg, i.p. Chronic—therapeutic; once daily for 9 days; first dose after sepsis induction | ↓mortality; improvement of memory-related processes; ↓lipid peroxidation in kidney; ↓oxidative protein damage in spleen, liver, heart | anti-oxidative; organo-protective; procognitive |
| Disease | n | * Study Design; Dose of CBD and THC | Application | Final Results and/or Conclusions, Properties Important for COVID-19 | Profile Safety/Side Effects | References | |
|---|---|---|---|---|---|---|---|
| Respiratory | COPD | 16 | * Cannabis 35 mg (THC, 18.2%, CBD, 0.1%); acute | vaporized | no effect on airway function, exertional breathlessness at rest and exercise | [126] | |
| 9 | * THC: CBD 2.7:2.5 mg/spray; maximum single dose of 4 sprays | oromucosal spray | no effect on simulated breathlessness in COPD subjects; ↓unpleasantness of breathlessness as judged by descriptors | [127] | |||
| Gastrointestinal | chemotherapy- induced nausea and vomiting (CINV) | 7 | * THC: CBD 2.7:2.5 mg/spray, <3 sprays within 2 h after chemotherapy plus <8 sprays each at days 2, 3 and 4 | oromucosal spray | better protection against delayed CINV compared to standard antiemetic therapy alone | well tolerated | [128] |
| cancer-related anorexia- cachexia syndrome | 99 | * THC 2.5 mg and CBD 1 mg; twice daily 1 h before meals for 6 weeks | p.o. | no effect on patients’ appetite or quality of life | well tolerated | [129] | |
| aspirin-induced increased gut permeability | 10 | * CBD:600 mg | p.o. | ↓increased gut permeability | [130] | ||
| moderately active Crohn’s disease | 20 | * CBD 10 mg/kg twice daily for 8 weeks | p.o. | no clinical improvement | excellent tolerability and safety profile | [131] | |
| Gastrointestinal | ulcerative colitis | 60 | * 250 mg CBD-rich extract (up to 4.7% THC) twice daily before meals for 10 weeks | p.o. | no effect on ulcerative colitis but ↑quality of life outcomes | mild/moderate, mainly dizziness and somnolence | [132] |
| Psychiatric | anxiety in healthy volunteers | 40 | * CBD:300 mg acute | p.o. | ↓anxiety to simulated public speaking | [133] | |
| 10 | * CBD:400 mg acute | p.o. | ↓subjective anxiety to a simulated public speaking test, ↑mental sedation | [134] | |||
| 60 | * CBD:100, 300 and 900 mg acute | p.o. | ↓subjective anxiety with a dose-dependent bell-shaped curve (effective dose:300 mg only) | CBD 300 mg has a lower sedation level than clonazepam | [135] | ||
| naïve social anxiety disorder | 24 | * CBD:600 mg acute | p.o. | ↓subjective anxiety, ↓cognitive impairment and discomfort in speech performance; no changes in blood pressure, heart rate and skin flow | absence of psychoactive or cognitive effects | [136] | |
| 10 | * CBD:400 mg acute | p.o. | ↓subjective anxiety, changes in regional cerebral flow | [137] | |||
| stress-related disorders | 11 | ** CBD: flexible doses, starting from 25 to 49 mg/d for 8 weeks | p.o. | ↓stress-related disorders (including ↓nightmares) | well tolerated, no patients discontinued treatment due to side effects; mild side effects: fatigue, reduced concentration; gastrointestinal bloating or pain | [138] | |
| psychiatric patients with anxiety or poor sleep | 103 | CBD:25 mg/d to 50–75 mg/d; for 1–3 months | p.o. | ↓anxiety in a sustained manner, ↓sleep disturbances | well tolerated, fatigue (may be related to dosing), mild sedation, dry eyes | [139] | |
| Psychiatric | patients at high risk for psychosis | 32 | * CBD:600 mg/day for 1 week | p.o. | cortisol reaction: tended to be better; anxiety: tended to be better; ↓negative self-statement | [140] | |
| 33 | * CBD:600 mg acute | p.o. | putative antipsychotic effect by normalizing motivational salience and moderating motor response | [141] | |||
| cannabis use disorder | 128 | * THC:CBD 2.7:2.5 mg/spray for 6 days, up to 32 sprays/d | oromucosal spray | ↓anxiety, ↓depression, ↓craving | no differences in adverse effects between THC:CBD and placebo group | [142] | |
| regular cannabis users | 20 | ** CBD:200 mg for 10 weeks | p.o. | ↓depressive symptoms, ↓psychotic symptoms, ↑attentional switching, ↑verbal learning, ↑memory | [143] | ||
| drug-abstinent patients with history of heroin abuse | 42 | * CBD 400 or 800 mg/d for 3 days | p.o. | ↓anxiety, ↓craving, ↓HR, ↓salivary cortisol levels | no serious adverse events; mild side effects: diarrhea, headache, tiredness, fatigue | [144] | |
| volunteers pre-selected for high paranoid traits | 32 | * CBD:600 mg acute | p.o. | no benefits on anxiety or persecutory ideation | [145] | ||
| schizophrenia | 42 | *** CBD; week 1: gradual increase to 800 mg/day; weeks 2–4:800 mg/day | p.o. | ↓positive psychotic symptoms (no difference compared to amisulpride), mitigation of psychotic symptoms | marked tolerability / safety (with respect to weight, prolactin, hepatic or cardiac functions) compared with current medications | [146] | |
| Psychiatric | schizophrenia | 88 | * CBD:1000 mg/d for 6 weeks on top of antipsychotic medication | p.o. | ↓positive psychotic symptoms, ↓impressions and severity of illness, cognitive performance and overall functioning tended to be improved | good tolerance; diarrhea, nausea, headache, infections, insomnia; mild ↓blood pressure and moderate chest pain; no significant changes in prolactin, weight, liver function, inflammatory markers or HDL cholesterol levels | [147] |
| 36 | * CBD:600 mg/day for 6 weeks | p.o. | no improvement of cognitive impairments and psychotic symptoms in stable antipsychotic-treated outpatients | well tolerated with no worsening of mood, suicidality or movement side effects; sedation | [148] | ||
| Pain | chronic pain | 94 | CBD-rich hemp extract 1 for 8 weeks | soft gels | ↓chronic pain and ↑life quality (↓opioid use, ↑sleep quality) | [149] | |
| chronic pain in kidney transplant patients | 7 | CBD increasing from 50 to 150 mg twice a day for 3 weeks | p.o. | 2 patients, total improvement of pain 4 patients, partial pain improvement | no serious adverse effects; dizziness, nausea, dry mouth, drowsiness, intermittent episodes of heat | [150] | |
| chronic pain in patients with fibromyalgia | 20 | * THC:CBD (mg) 1. 22.4: < 1 2. 13.4:17.8 3. < 1:18.4 | single vapor inhalation | small analgesic responses | limited, such as dizziness and nausea | [151] | |
| neuropathic pain in patients with MS | 20 | THC:CBD 2.7:2.5 mg/spray for 4 weeks, 8 sprays/d | sublin. spray | ↓pain rating, ↑life quality | few side effects: dizziness, nausea, dry mouth and weakness | [152] | |
| Pain | peripheral neuropathic pain associated with diabetes or allodynia | 380 | THC:CBD 2.7:2.5 mg/spray for 38 weeks, <8 sprays per 3 h and <24 sprays every 24 h | oromucosal spray | ↓pain in the majority of patients | safe and well tolerated; patients did not seek to increase their dose with time but 23% of patients ceased medication due to adverse effects (2% infections) | [153] |
| painful diabetic neuropathy | 30 | * THC:CBD 2.7:2.5 mg/spray; dose was titrated over 2 weeks, followed by a 10-week maintenance phase (4 sprays/d) | sublin. spray | no significant improvements in pain rating and life quality | [154] | ||
| advanced cancer patients with chronic pain | 199 | * THC:CBD 2.7:2.5 mg/spray: initially 4 up to 7 sprays/d | oromucosal spray | average pain score not superior to placebo | [155] | ||
| Neurological | Dravet syndrome in children | 198 | * CBD: 10 and 20 mg/kg/d for 14 weeks | p.o. | ↓frequency of seizures, improvement of patients’ condition; similar effectiveness of both doses | ↓appetite, diarrhea, somnolence, pyrexia, and fatigue, ↑aminotransferases in patients also taking valproate sodium; at 10 mg/kg/d better tolerance and safety profile | [156] |
| 108 | * CBD:20 mg/kg/d for 14 weeks | p.o. | ↓frequency of drug-resistant seizures | somnolence, ↓appetite, diarrhea, ↑aminotransferases in patients also taking valproate | [157] | ||
| Neurological | Lennox–Gastaut syndrome in children and adults | 225 | * CBD:10 or 20 mg/kg/d for 28 days | p.o. | ↓epileptic seizures in some patients, ↓total seizure frequency, improvement of patients´ condition | somnolence, ↓appetite, diarrhea (at 20 mg/kg/d), threefold ↑aminotransferases | [158] |
| 171 | * CBD:20 mg/kg/d for 14 weeks | p.o. | ↓frequency of drug-resistant seizures | in general good tolerance of CBD as add-on therapy; mild or moderate side effects: ↓appetite, vomiting, diarrhea, somnolence, fever | [159] | ||
| multiple sclerosis | 160 | * THC:CBD 2.7:2.5 mg/spray up to a maximum of 120 mg THC and 120 mg CBD/day with no more than 20 mg of each in any 3-h period for 6 weeks | oromucosal spray | ↓spasticity associated with MS | good tolerance, no effects on cognition or mood; dizziness, disturbance in attention, headache, fatigue, somnolence, disorientation, feeling drunk, vertigo, application site discomfort, nausea, diarrhea, mouth ulceration | [160] | |
| 137 | THC:CBD 2.7:2.5 mg/spray up to 48 sprays/day for 21—814 days (extension study of Wade et al. [160]) | oromucosal spray | ↓spasticity associated with MS, clinical effect maintained after a long treatment period | serious adverse effects: seizures, fall, aspiration pneumonia, gastroenteritis; mild adverse effects: sore mouth, oromucosal disorder, oral pain, altered attention, dizziness, diarrhea, nausea | [161] | ||
| 12 | THC:CBD 5.4:5.0 mg (two sublin. sprays at a 15-min interval) | oromucosal spray | ↓spasticity postural and motor tests unchanged | no differences in adverse events between THC:CBD and placebo groups | [162] | ||
| 460 | * THC:CBD 2.7:2.5 mg/spray; maximally 12 sprays/day for 12 weeks | oromucosal spray | ↓spasticity (resistant to previous antispasticity treatment) and associated symptoms in patients with moderate to severe MS | [163] | |||
| Skin | psoriasis atopic dermatitis | 5 5 | CBD-enriched ointment, twice daily, for 3 months | ointment | in both diseases, improved skin parameters | no irritant or allergic reactions | [164] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Malinowska, B.; Baranowska-Kuczko, M.; Kicman, A.; Schlicker, E. Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19. Int. J. Mol. Sci. 2021, 22, 1986. https://doi.org/10.3390/ijms22041986
Malinowska B, Baranowska-Kuczko M, Kicman A, Schlicker E. Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19. International Journal of Molecular Sciences. 2021; 22(4):1986. https://doi.org/10.3390/ijms22041986
Chicago/Turabian StyleMalinowska, Barbara, Marta Baranowska-Kuczko, Aleksandra Kicman, and Eberhard Schlicker. 2021. "Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19" International Journal of Molecular Sciences 22, no. 4: 1986. https://doi.org/10.3390/ijms22041986
APA StyleMalinowska, B., Baranowska-Kuczko, M., Kicman, A., & Schlicker, E. (2021). Opportunities, Challenges and Pitfalls of Using Cannabidiol as an Adjuvant Drug in COVID-19. International Journal of Molecular Sciences, 22(4), 1986. https://doi.org/10.3390/ijms22041986