Preeclampsia: Cardiotonic Steroids, Fibrosis, Fli1 and Hint to Carcinogenesis
Abstract
1. Introduction
2. Bufadienolide Cardiotonic Steroids
3. Cardiotonic Steroids, Pregnancy and Preeclampsia
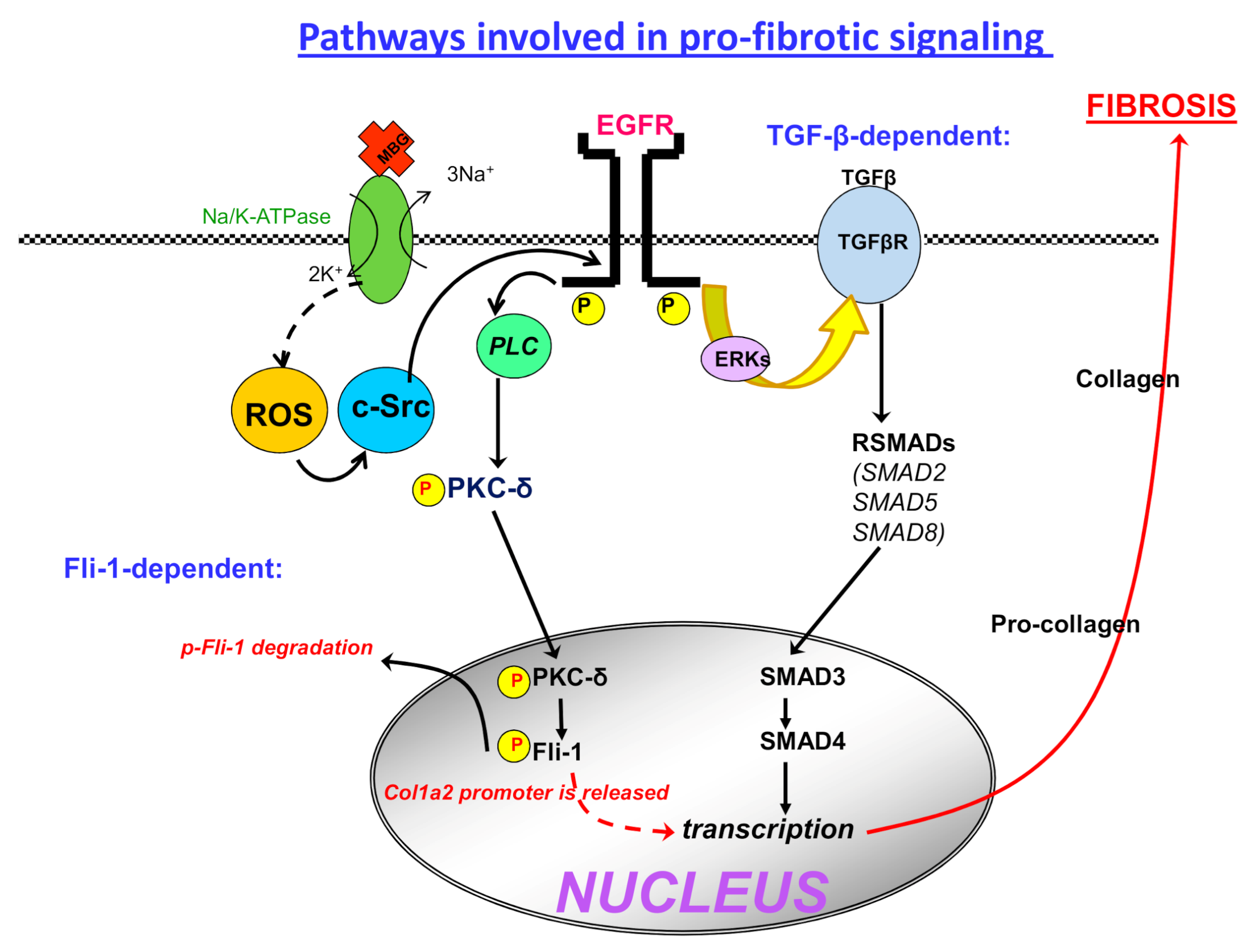
4. Fibrosis and Preeclampsia
5. Immunoneutralization of Cardiotonic Steroids
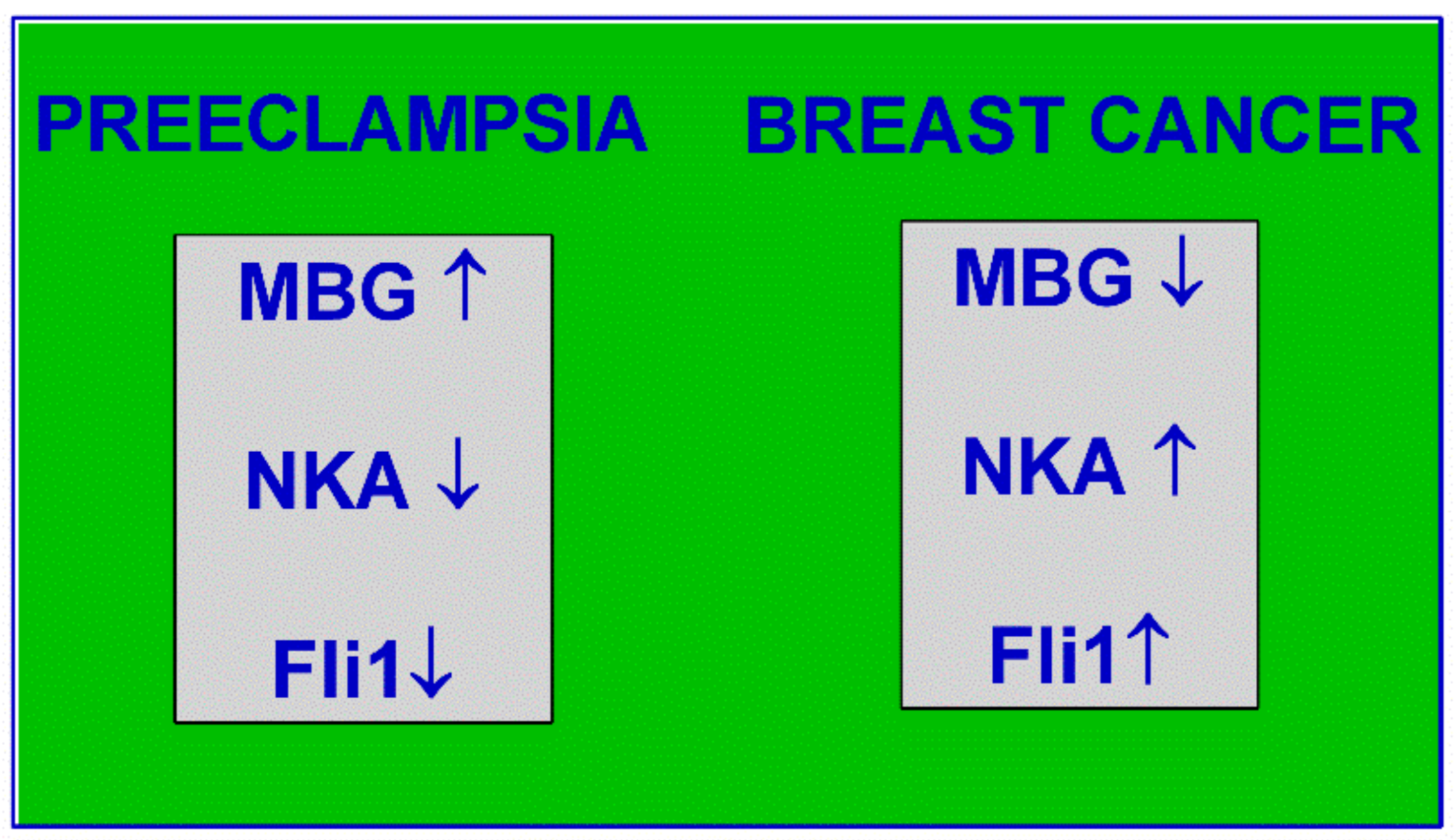
6. Interaction of CS and Fli1 and a Hint for Cancer
7. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| CS | Cardiotonic steroids |
| EGRF | Epidermal growth factor receptor |
| ERK | Extracellular signal-regulated kinases |
| ETS | Erythroblast transformation specific family |
| Fli1 | Friend leukemia integration 1 transcription factor |
| MBG | Marinobufagenin |
| SMAD | Proteins, main signal transducers for receptors of the transforming growth factor beta |
| Src | A family of proto-oncogenic tyrosine kinases |
| PKC | Protein kinase C |
| PLC | Phospholipase C |
| PE | Preeclampsia |
| TGF-β | Transforming growth factor beta 1 |
References
- Sibai, B.; Dekker, G.; Kupferminc, M. Pre-eclampsia. Lancet 2005, 365, 785–799. [Google Scholar] [CrossRef]
- MacKay, A.P.; Berg, C.J.; Atrash, H.K. Pregnancy-related mortality from preeclampsia and eclampsia. Obstet. Gynecol. 2001, 97, 533–538. [Google Scholar] [CrossRef]
- Thangaratinam, S.; Coomarasamy, A.; O’Mahony, F.; Sharp, S.; Zamora, J.; Khan, K.S.; Ismail, K.M.K. Estimation of proteinuria as a predictor of complications of pre-eclampsia: A systematic review. BMC Med. 2009, 7, 10. [Google Scholar] [CrossRef]
- Wagner, S.J.; Craici, I.M.; Grande, J.P.; Garovic, V.D. From placenta to podocyte: Vascular and podocyte pathophysiology in preeclampsia. Clin. Nephrol. 2012, 78, 241–249. [Google Scholar] [CrossRef]
- Meekins, J.W.; Pijnenborg, R.; Hanssens, M.; McFadyen, I.R.; van Asshe, A. A study of placental bed spiral arteries and trophoblast invasion in normal and severe pre-eclamptic pregnancies. Brit. J. Obstet. Gynaecol. 1994, 101, 669–674. [Google Scholar] [CrossRef]
- Bagrov, A.Y.; Shapiro, J.I.; Fedorova, O.V. Endogenous cardiotonic steroids: Physiology, pharmacology and novel therapeutic targets. Pharmacol. Rev. 2009, 61, 9–38. [Google Scholar] [CrossRef] [PubMed]
- Flier, J.; Edwards, M.W.; Daly, J.W.; Myers, C.W. Widespread occurrence in frogs and toads of skin compounds interacting with the ouabain site of Na+, K+-ATPase. Science 1980, 208, 503–505. [Google Scholar] [CrossRef]
- Kieval, R.S.; Butler, V.P., Jr.; Derguini, F.; Bruening, R.C.; Rosen, M.R. Cellular electrophysiologic effects of vertebrate digitalis-like substances. J. Am. Coll. Cardiol. 1988, 11, 637–643. [Google Scholar] [CrossRef]
- Bagrov, A.Y.; Fedorova, O.V.; Dmitrieva, R.I.; Howald, W.N.; Hunter, A.P.; Kuznetsova, E.A.; Shpen, V.M. Bufodienolide nature of endogenous inhibitor of Na/K ATPase in the urine from patients with acute myocardial infarction. Hypertension 1998, 31, 1097–1103. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Simbirtsev, A.S.; Kolodkin, N.I.; Kotov, A.Y.; Agalakova, N.I.; Kashkin, V.A.; Tapilskaya, N.I.; Bzhelyansky, A.; Reznik, V.A.; Frolova, E.V.; et al. Monoclonal antibody to an endogenous bufadienolide, marinobufagenin, reverses preeclampsia-induced Na/K-ATPase inhibition in lowers blood pressure in NaCl-sensitive hypertension. J. Hypertens. 2008, 26, 2414–2425. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Zernetkina, V.I.; Shilova, V.Y.; Grigorova, Y.N.; Juhasz, O.; Wei, W.; Marshall, C.A.; Lakatta, E.G.; Bagrov, A.Y. Synthesis of an endogenous steroidal Na pump inhibitor marinobufagenin, implicated in human cardiovascular diseases, is initiated by CYP27A1 via bile acid pathway. Circ. Cardiovasc. Genet. 2015, 8, 736–745. [Google Scholar] [CrossRef] [PubMed]
- Fedorova, O.V.; Bagrov, A.Y. Inhibition of Na/K ATPase from rat aorta by two endogenous Na/K pump inhibitors, ouabain and marinobufagenin. Evidence of interaction with different alpha-subinit isoforms. Am. J. Hypertens. 1997, 10, 929–935. [Google Scholar] [CrossRef]
- Fridman, A.I.; Matveev, S.A.; Agalakova, N.I.; Fedorova, O.V.; Lakatta, E.G.; Bagrov, A.Y. Marinobufagenin, an endogenous ligand of alfa-1 Na/K-ATPase, is a marker of congestive heart failure severity. J. Hypertens. 2002, 20, 1189–1194. [Google Scholar] [CrossRef] [PubMed]
- Haller, S.T.; Kennedy, D.J.; Shidyak, A.; Budny, G.V.; Malhotra, D.; Fedorova, O.V.; Shapiro, J.I.; Bagrov, A.Y. Monoclonal antibody against marinobufagenin reverses cardiac fibrosis in rats with chronic renal failure. Am. J. Hypertens. 2012, 25, 690–696. [Google Scholar] [CrossRef]
- Mottelson, M.N.; Lundsgaard, C.C.; Møller, S. Mechanisms in fluid retention—Towards a mutual concept. Clin. Physiol. Funct. Imaging 2020, 40, 67–75. [Google Scholar] [CrossRef] [PubMed]
- Graves, S.W. The possible role of digitalislike factors in pregnancy-induced hypertension. Hypertension 1987, 10, I84–I86. [Google Scholar] [CrossRef]
- Poston, L.; Morris, J.F.; Wolfe, C.D.; Hilton, P.J. Serum digoxin-like substances in pregnancy-induced hypertension. Clin. Sci. Lond. 1989, 77, 189–194. [Google Scholar] [CrossRef]
- Gilson, G.J.; Graves, S.W.; Qualls, C.R.; Curet, L.B. Digoxin-like immunoreactive substance and sodium-potassium-adenosine triphosphatase inhibition in normal pregnancy: A longitudinal study. Obstet. Gynecol. 1997, 89, 743–746. [Google Scholar] [CrossRef]
- Goodlin, R.C. Antidigoxin antibodies in eclampsia. N. Engl. J. Med. 1988, 318, 518–519. [Google Scholar] [CrossRef]
- Kaminski, K.; Rechberger, T. Concentration of digoxin-like immunoreactive substance in patients with preeclampsia and its relation to severity of pregnancy-induced hypertension. Am. J. Obstet. Gynecol. 1991, 165, 733–736. [Google Scholar] [CrossRef]
- Ujhelyi, M.R.; Robert, S.; Cummings, D.M.; Colucci, R.D.; Green, P.J.; Sailstad, J.; Vlasses, P.H.; Zarowitz, B.J. Influence of digoxin immune Fab therapy and renal dysfunction on the disposition of total and free digoxin. Ann. Intern. Med. 1993, 119, 273–277. [Google Scholar] [CrossRef]
- Yamada, K.; Goto, A.; Hui, C.; Yagi, N.; Sugimoto, T. Effects of the Fab fragment of digoxin antibody on the natriuresis and increase in blood pressure induced by intracerebroventricular infusion of hypertonic saline solution in rats. Clin. Sci. Lond. 1992, 82, 625–630. [Google Scholar] [CrossRef]
- Soszynski, P.; Ensign, A.; Graves, S.W.; Hollenberg, N.K. Specificity of the vascular smooth muscle contractile response to a labile digitalis-like factor in peritoneal dialysate: The influence of potassium. Am. J. Hypertens. 1997, 10, 1342–1348. [Google Scholar] [CrossRef]
- Morris, J.F.; Poston, L.; Wolfe, C.D.; Hilton, P.J. A comparison of endogenous digoxin-like immunoreactivity and sodium transport inhibitory activity in umbilical arterial and venous serum. Clin. Sci. Lond. 1988, 75, 577–579. [Google Scholar] [CrossRef]
- Di Grande, A.; Boura, A.L.; Read, M.A.; Malatino, L.S.; Walters, W.A. Release of a substance from the human placenta having digoxin-like immunoreactivity. Clin. Exp. Pharmacol. Physiol. 1993, 20, 603–607. [Google Scholar] [CrossRef] [PubMed]
- Armler, E.; Cester, N.; Salvolini, E.; Staffolani, R.; Burkhard, M.; Mazzanti, L.; Kotyk, A.; Romanini, C. Human hypertensive placenta contains an increased amount of Na,K-ATPase with higher affinity for cardiac glycosides. Cell Biol. Int. 1994, 18, 723–727. [Google Scholar] [CrossRef] [PubMed]
- Lopatin, D.A.; Ailamazian, E.K.; Dmitrieva, R.I.; Shpen, V.M.; Fedorova, O.V.; Doris, P.A.; Bagrov, A.Y. Circulating bufodienolide and cardenolide sodium pump inhibitors in preeclampsia. J. Hypertens. 1999, 17, 1179–1187. [Google Scholar] [CrossRef]
- Averina, I.V.; Tapilskaya, N.I.; Reznik, V.A.; Frolova, E.V.; Fedorova, O.V.; Lakatta, E.G.; Bagrov, A.Y. Endogenous Na/K-ATPase inhibitors in patients with preeclampsia. Cell. Mol. Biol. Noisy-le-grand 2006, 52, 19–23. [Google Scholar] [PubMed]
- Hausvater, A.; Giannone, T.; Sandoval, Y.H.; Doonan, R.J.; Antonopoulos, C.N.; Matsoukis, I.L.; Petridou, E.T.; Daskalopoulou, S.S. The association between preeclampsia and arterial stiffness. J. Hypertens. 2012, 30, 17–33. [Google Scholar] [CrossRef]
- Orabona, R.; Sciatti, E.; Vizzardi, E.; Bonadei, I.; Valcamonico, A.; Metra, M.; Frusca, T. Endothelial dysfunction and vascular stiffness in women with previous pregnancy complicated by early or late pre-eclampsia. Ultrasound Obstet. Gynecol. 2017, 49, 116–123. [Google Scholar] [CrossRef]
- Elkareh, J.; Kennedy, D.J.; Yashaswi, B.; Vetteth, S.; Shidyak, A.; Kim, E.G.; Smaili, S.; Periyasamy, S.M.; Hariri, I.M.; Fedorova, L.; et al. Marinobufagenin stimulates fibroblast collagen production and causes fibrosis in experimental uremic cardiomyopathy. Hypertension 2007, 49, 215–224. [Google Scholar] [CrossRef]
- Elkareh, J.; Periyasamy, S.M.; Shidyak, A.; Vetteth, S.; Schroeder, J.; Raju, V.; Hariri, I.M.; El-Okdi, N.; Gupta, S.; Fedorova, L.; et al. Marinobufagenin induces increases in procollagen expression in a process involving protein kinase C and Fli-1: Implications for uremic cardiomyopathy. Am. J. Physiol. Renal Physiol. 2009, 296, F1219–F1226. [Google Scholar] [CrossRef]
- Nikitina, E.R.; Mikhailov, A.V.; Nikandrova, E.S.; Frolova, E.V.; Fadeev, A.V.; Shman, V.V.; Shilova, V.Y.; Tapilskaya, N.I.; Shapiro, J.I.; Fedorova, O.V.; et al. In preeclampsia endogenous cardiotonic steroids induce vascular fibrosis and impair relaxation of umbilical arteries. J. Hypertens. 2011, 29, 769–776. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Emelianov, I.V.; Bagrov, K.A.; Grigorova, Y.N.; Wei, W.; Juhasz, O.; Frolova, E.V.; Marshall, C.A.; Lakatta, E.G.; Konradi, A.O.; et al. Marinobufagenin-induced vascular fibrosis is a likely target for mineralocorticoid antagonists. J. Hypertens. 2015, 33, 1602–1610. [Google Scholar] [CrossRef] [PubMed]
- Trojanowska, M. Cellular and molecular aspects of vascular dysfunction in systemic sclerosis. Nat. Rev. Rheumatol. 2010, 6, 453–460. [Google Scholar] [CrossRef] [PubMed]
- Omatsu, J.; Saigusa, R.; Miyagawa, T.; Fukui, Y.; Toyama, S.; Awaji, K.; Ikawa, T.; Norimatsu, Y.; Yoshizaki, A.; Sato, S.; et al. Serum S100A12 levels: Possible association with skin sclerosis and interstitial lung disease in systemic sclerosis. Exp. Dermatol. 2020. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Ishkaraeva, V.V.; Grigorova, Y.N.; Reznik, V.A.; Kolodkin, N.I.; Zazerskaya, I.E.; Zernetkina, V.; Agalakova, N.I.; Tapilskaya, N.I.; Adair, C.D.; et al. Antibody to Marinobufagenin Reverses Placenta-Induced Fibrosis of Umbilical Arteries in Preeclampsia. Int. J. Mol. Sci. 2018, 19, 2377. [Google Scholar] [CrossRef] [PubMed]
- Kolmakova, E.V.; Haller, S.T.; Kennedy, D.J.; Isachkina, A.N.; Budny, G.V.; Frolova, E.V.; Nikitina, E.R.; Piecha, G.; Malhotra, D.; Fedorova, O.V.; et al. Endogenous cardiotonic steroids in chronic renal failure. Nephrol. Dial. Transplant. 2011, 26, 2912–2919. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Kolodkin, N.I.; Agalakova, N.I.; Namikas, A.R.; Bzhelyansky, A.; St-Louis, J.; Lakatta, E.G.; Bagrov, A.Y. Antibody to marinobufagenin lowers blood pressure in pregnant rats on a high NaCl intake. J. Hypertens. 2005, 23, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Orabona, R.; Sciatti, E.; Prefumo, F.; Vizzardi, E.; Bonadei, I.; Valcamonico, A.; Metra, M.; Frusca, T. Pre-eclampsia and heart failure: A close relationship. Ultrasound Obstet. Gynecol. 2018, 52, 297–301. [Google Scholar] [CrossRef] [PubMed]
- Pruthi, D.; Khankin, E.V.; Blanton, R.M.; Aronovitz, M.; Burke, S.D.; McCurley, A.; Karumanchi, S.A.; Jaffe, I.Z. Exposure to experimental preeclampsia in mice enhances the vascular response to future injury. Hypertension 2015, 65, 863–870. [Google Scholar] [CrossRef] [PubMed]
- Grigorova, Y.N.; Wei, W.; Petrashevskaya, N.; Zernetkina, V.; Juhasz, O.; Fenner, R.; Gilbert, C.; Lakatta, E.G.; Shapiro, J.I.; Bagrov, A.Y.; et al. Dietary Sodium Restriction Reduces Arterial Stiffness, Vascular TGF-β-Dependent Fibrosis and Marinobufagenin in Young Normotensive Rats. Int. J. Mol. Sci. 2018, 19, 3168. [Google Scholar] [CrossRef]
- Zhang, Y.; Wei, W.; Shilova, V.; Petrashevskaya, N.N.; Zernetkina, V.I.; Grigorova, Y.N.; Marshall, C.A.; Fenner, R.C.; Lehrmann, E.; Wood, W.H., 3rd; et al. Monoclonal Antibody to Marinobufagenin Downregulates TGFβ Profibrotic Signaling in Left Ventricle and Kidney and Reduces Tissue Remodeling in Salt-Sensitive Hypertension. J. Am. Heart Assoc. 2019, 8, e012138. [Google Scholar] [CrossRef]
- Fedorova, O.V.; Fadeev, A.V.; Grigorova, Y.N.; Marshall, C.A.; Zernetkina, V.; Kolodkin, N.I.; Agalakova, N.I.; Konradi, A.O.; Lakatta, E.G.; Bagrov, A.Y. Cardiotonic Steroids Induce Vascular Fibrosis Via Pressure-Independent Mechanism in NaCl-Loaded Diabetic Rats. J. Cardiovasc. Pharmacol. 2019, 74, 436–442. [Google Scholar] [CrossRef]
- Adair, C.D.; Buckalew, V.M.; Kipikasa, J.; Torres, C.; Stallings, S.P.; Briery, C.M. Repeated dosing of digoxin-fragmented antibody in preterm eclampsia. J. Perinatol. 2009, 29, 163–165. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Adair, C.D.; Buckalew, V.; Taylor, K.; Ernest, J.M.; Frye, A.H.; Evans, C.; Veille, J.C. Elevated endoxin-like factor complicating a multifetal second trimester pregnancy: Treatment with digoxin-binding immunoglobulin. Am. J. Nephrol. 1996, 16, 529–531. [Google Scholar] [CrossRef]
- Roberts, D.M.; Gallapatthy, G.; Dunuwille, A.; Chan, B.S. Pharmacological treatment of cardiac glycoside poisoning. Br. J. Clin. Pharmacol. 2016, 81, 488–495. [Google Scholar] [CrossRef]
- Chen, K.K.; Kovarikova, A. Pharmacology and toxicology of toad venom. J. Pharm. Sci. 1967, 56, 1535–1541. [Google Scholar] [CrossRef] [PubMed]
- Brubacher, J.R.; Lachmanen, D.; Ravikumar, P.R.; Hoffman, R.S. Efficacy of digoxin specific Fab fragments (Digibind) in the treatment of toad venom poisoning. Toxicon 1999, 37, 931–942. [Google Scholar] [CrossRef]
- Krep, H.; Price, D.A.; Soszynski, P.; Tao, Q.-F.; Graves, S.W.; Hollenberg, N.K. Volume sensitive hypertension and the digoxin-like factor reversal by a fab directed against digoxin in DOCA-salt hypertensive rats. Am. J. Hypertens. 1995, 8, 921–927. [Google Scholar] [CrossRef]
- Ma, J.; Esplin, M.S.; Adair, C.D.; Mason, L.A.; Graves, S.W. Increasing evidence for and regulation of a human placental endogenous digitalis-like factor. Reprod. Sci. 2012, 19, 437–448. [Google Scholar] [CrossRef]
- Adair, C.D.; Buckalew, V.M.; Graves, S.W.; Lam, G.K.; Johnson, D.D.; Saade, G.; Lewis, D.F.; Robinson, C.; Danoff, T.M.; Chauhan, N.; et al. Digoxin immune fab treatment for severe preeclampsia. Am. J. Perinatol. 2010, 27, 655–662. [Google Scholar] [CrossRef] [PubMed]
- Adair, D.; Hinshaw, A.; Russell, G.; Rose, J.; Veille, J.; Buckalew, V. Effects of Fab digoxin-specific antibodies on mean arterial pressure in severe preeclampsia. Am. J. Hypertens. 1997, 10, 11A. [Google Scholar] [CrossRef]
- Adair, C.D.; Luper, A.; Rose, J.C.; Russell, G.; Veille, J.C.; Buckalew, V.M. The hemodynamic effects of intravenous digoxin-binding fab immunoglobulin in severe preeclampsia: A double-blind, randomized, clinical trial. J. Perinatol. 2009, 29, 284–289. [Google Scholar] [CrossRef][Green Version]
- Hopoate-Sitake, M.; Adair, C.D.; Buckalew, V.M.; Johnson, D.D.; Lewis, D.F.; Robinson, C.J.; Saade, G.R.; Graves, S.W. Digoxin antibody fragment, antigen binding (Fab), treatment of preeclampsia in women with endogenous digitalis-like factor: A secondary analysis of the DEEP Trial. Am. J. Obstet. Gynecol. 2013, 209, 119. [Google Scholar] [CrossRef]
- Ishkaraeva-Yakovleva, V.V.; Fedorova, O.V.; Solodovnikova, N.G.; Frolova, E.V.; Bzhelyansky, A.M.; Emelyanov, I.V.; Adair, C.D.; Zazerskaya, I.E.; Bagrov, A.Y. DigiFab interacts with endogenous cardiotonic steroids and reverses preeclampsia-induced Na/K-ATPase inhibition. Reprod. Sci. 2012, 19, 1260–1267. [Google Scholar] [CrossRef]
- Uddin, M.N.; Wages, J.M.; Pantho, A.F.; Larrick, J.W.; Drever, N.; Allen, S.R.; Kuehl, T.J. Novel anti-MBG antibodies protect cytotrophoblast cells from a marinobufagenin-induced preeclampsia phenotype. Am. J. Obstet. Gynecol. 2018, 218, S199. [Google Scholar] [CrossRef]
- Agalakova, N.I.; Reznik, V.A.; Nadei, O.V.; Ershov, I.A.; Rassokha, O.S.; Vasyutina, M.L.; Ivanov, D.O.; Adair, E.D.; Galagudza, M.M.; Bagrov, A.Y. Antibody against Na/K-ATPase inhibitor lowers blood pressure and increases vascular Fli1 in experimental preeclampsia. Am. J. Hypertens. 2020, 33, 514–519. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Q.; Contreras, R.G.; Wang, R.; Fernandez, S.V.; Shoshani, L.; Russo, I.H.; Cereijido, M.; Russo, J. Sodium/potassium ATPase (Na+, K+-ATPase) and ouabain/related cardiac glycosides: A new paradigm for development of anti- breast cancer drugs? Breast Cancer Res. Treat. 2006, 96, 1–15. [Google Scholar] [CrossRef]
- Troisi, R.; Gulbech Ording, A.; Grotmol, T.; Glimelius, I.; Engeland, A.; Gissler, M.; Trabert, B.; Ekbom, A.; Madanat-Harjuoja, L.; Sørensen, H.T.; et al. Pregnancy complications and subsequent breast cancer risk in the mother: A Nordic population-based case-control study. Int. J. Cancer. 2018, 143, 1904–1913. [Google Scholar] [CrossRef] [PubMed]
- Audette, M.; Pillai, K.; Wrana, J.; Kingdom, J. Does Preeclampsia Reduce the Risk of Breast Cancer? J. Obstet. Gynaecol. Can. 2015, 37, 736–739. [Google Scholar] [CrossRef][Green Version]
- Wright, L.B.; Schoemaker, M.J.; Jones, M.E.; Ashworth, A.; Swerdlow, A.J. Breast cancer risk in relation to history of preeclampsia and hyperemesis gravidarum: Prospective analysis in the Generations Study. Int. J. Cancer 2018, 143, 782–792. [Google Scholar] [CrossRef] [PubMed]
- Garcia, I.J.P.; de Oliveira, G.C.; de Moura Valadares, J.M.; Banfi, F.F.; Andrade, S.N.; Freitas, T.R.; Dos Santos Monção Filho, E.; Lima Santos, H.; Júnior, G.M.V.; Chaves, M.H.; et al. New bufadienolides extracted from Rhinella marina inhibit Na,K-ATPase and induce apoptosis by activating caspases 3 and 9 in human breast and ovarian cancer cells. Steroids 2019, 152, 108490. [Google Scholar] [CrossRef]
- Li, Y.; Tian, X.; Liu, X.; Gong, P. Bufalin inhibits human breast cancer tumorigenesis by inducing cell death through the ROS-mediated RIP1/RIP3/PARP-1 pathways. Carcinogenesis 2018, 39, 700–707. [Google Scholar] [CrossRef]
- Khajah, M.A.; Mathew, P.M.; Luqmani, Y.A. Na+/K+ ATPase activity promotes invasion of endocrine resistant breast cancer cells. PLoS ONE 2018, 13, e0193779. [Google Scholar] [CrossRef] [PubMed]
- Chen, N.; Zhao, G.; Yan, X.; Lu, Z.; Yin, H.; Zhang, S.; Song, W.; Li, X.; Li, L.; Du, Z.; et al. A novel FLI1 exonic circular RNA promotes metastasis in breast cancer by coordinately regulating TET1 and DNMT1. Genome Biol. 2018, 19, 218. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Wang, Y.; Yu, C.; Cao, Y.; Yu, Y.; Pan, Y.; Su, D.; Lu, Q.; Yang, W.; Zuo, Y.; et al. Characterization of the relationship between FLI1 and immune infiltrate level in tumour immune microenvironment for breast cancer. J. Cell. Mol. Med. 2020, 24, 5501–5514. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Yu, Y.; Li, L.; Chen, N.; Song, W.; He, H.; Dong, J.; Liu, X.; Cui, J. Friend leukemia virus integration 1 is a predictor of poor prognosis of breast cancer and promotes metastasis and cancer stem cell properties of breast cancer cells. Cancer Med. 2018, 7, 3548–3560. [Google Scholar] [CrossRef]
- Keskin, T.; Bakaric, A.; Waszyk, P.; Boulay, G.; Torsello, M.; Cornaz-Buros, S.; Chevalier, N.; Geiser, T.; Martin, P.; Volorio, A.; et al. LIN28B Underlies the Pathogenesis of a Subclass of Ewing Sarcoma LIN28B Control of EWS-FLI1 Stability. Cell Rep. 2020, 30, 4567–4583. [Google Scholar] [CrossRef] [PubMed]
- García-Domínguez, D.J.; Hontecillas-Prieto, L.; León, E.A.; Sánchez-Molina, S.; Rodríguez-Núñez, P.; Morón, F.J.; Hajji, N.; Mackintosh, C.; de Álava, E. An inducible ectopic expression system of EWSR1-FLI1 as a tool for understanding Ewing sarcoma oncogenesis. PLoS ONE 2020, 15, e0234243. [Google Scholar] [CrossRef]
- Machado, K.D.C.; Sousa, L.Q.; Lima, D.J.B.; Soares, B.M.; Cavalcanti, B.C.; Maranhão, S.S.; Noronha, J.D.C.; Rodrigues, D.J.; Militão, G.C.G.; Chaves, M.H.; et al. Marinobufagin, a molecule from poisonous frogs, causes biochemical, morphological and cell cycle changes in human neoplasms and vegetal cells. Toxicol. Lett. 2018, 285, 121–131. [Google Scholar] [CrossRef]
- Lan, Y.L.; Wang, X.; Lou, J.C.; Xing, J.S.; Zou, S.; Yu, Z.L.; Ma, X.C.; Wang, H.; Zhang, B. Marinobufagenin inhibits glioma growth through sodium pump α1 subunit and ERK signaling-mediated mitochondrial apoptotic pathway. Cancer Med. 2018, 7, 2034–2047. [Google Scholar] [CrossRef]
- Reddy, D.; Kumavath, R.; Barh, D.; Azevedo, V.; Ghosh, P. Anticancer and Antiviral Properties of Cardiac Glycosides: A Review to Explore the Mechanism of Actions. Molecules 2020, 25, 3596. [Google Scholar] [CrossRef] [PubMed]
- Soumoy, L.; Wells, M.; Najem, A.; Krayem, M.; Ghanem, G.; Hambye, S.; Saussez, S.; Blankert, B.; Journe, F. Toad Venom Antiproliferative Activities on Metastatic Melanoma: Bio-Guided Fractionation and Screening of the Compounds of Two Different Venoms. Biology 2020, 9, 218. [Google Scholar] [CrossRef] [PubMed]
- Weidemann, H. Na/K-ATPase, endogenous digitalis like compounds and cancer development—A hypothesis. Front. Biosci. 2005, 10, 2165–2176. [Google Scholar] [CrossRef]
- Weidemann, H. “The Lower Threshold” phenomenon in tumor cells toward endogenous digitalis-like compounds: Responsible for tumorigenesis? J. Carcinog. 2012, 11, 2. [Google Scholar] [CrossRef]
- Numazawa, S.; Honma, Y.; Yamamoto, T.; Yoshida, T.; Kuroiwa., Y. A cardiotonic steroid bufalin-like factor in human plasma induces leukemia cell differentiation. Leuk. Res. 1995, 19, 945–953. [Google Scholar] [CrossRef]
- Han, M.; Yang, G.; Lin, Q.; Yang, Y.; Zhang, H.; Su, Y. Determination of endogenous bufalin in serum of patients with hepatocellular carcinoma based on HPLC-MS/MS. Front. Oncol. 2019, 9, 1572. [Google Scholar] [CrossRef]
- Oda, M.; Kurosawa, M.; Numazawa, S.; Tanaka, S.; Akizawa, T.; Ito, K.; Maeda, M.; Yoshida, T. Determination of bufalin-like immunoreactivity in serum of humans and rats by time-resolved fluoroimmunoassay for using a monoclonal antibody. Life Sci. 2001, 68, 1107–1117. [Google Scholar] [CrossRef]
- He, S.; Huang, Q.; Hu, J.; Li, L.; Xiao, Y.; Yu, H.; Han, Z.; Wang, T.; Zhou, W.; Wei, H.; et al. EWS-FLI1-mediated tenascin-C expression promotes tumour progression by targeting MALAT1 through integrin α5β1-mediated YAP activation in Ewing sarcoma. Br. J. Cancer. 2019, 121, 922–933. [Google Scholar] [CrossRef]
- Blair, D.G.; Athanasiou, M. Ets and retroviruses—Transduction and activation of members of the Ets oncogene family in viral oncogenesis. Oncogene 2000, 19, 6472–6481. [Google Scholar] [CrossRef] [PubMed]
- Oikawa, T.; Yamada, T. Molecular biology of the Ets family of transcription factors. Gene 2003, 303, 11–34. [Google Scholar] [CrossRef]
- Ohno, T.; Rao, V.N.; Reddy, E.S. EWS/Fli-1 chimeric protein is a transcriptional activator. Cancer Res. 1993, 53, 5859–5863. [Google Scholar] [PubMed]
- Gonçalves-de-Albuquerque, C.F.; Ribeiro Silva, A.; Ignácio da Silva, C.; Caire Castro-Faria-Neto, H.; Burth, P. Na/K Pump and Beyond: Na/K-ATPase as a Modulator of Apoptosis and Autophagy. Molecules 2017, 22, 578. [Google Scholar] [CrossRef] [PubMed]
- Fahr, L.; Sunami, Y.; Maeritz, N.; Steiger, K.; Grünewald, T.G.P.; Gericke, M.; Kong, B.; Raulefs, S.; Mayerle, J.; Michalski, C.W.; et al. Expression of the EWSR1-FLI1 fusion oncogene in pancreas cells drives pancreatic atrophy and lipomatosis. Pancreatology 2020, 20, 1673–1681. [Google Scholar] [CrossRef]
- Marchetto, A.; Grünewald, T.G.P. SOX6: A double-edged sword for Ewing sarcoma. Mol. Cell. Oncol. 2020, 7, 1783081. [Google Scholar] [CrossRef] [PubMed]
- Xie, B.; Hu, F.; Li, M.; Mo, L.; Xu, C.; Xiao, Y.; Wang, X.; Nie, J.; Yang, L.; He, Y. FLI-1 mediates tumor suppressor function via Klotho signaling in regulating CRC. Cell. Biol. Int. 2020, 44, 1514–1522. [Google Scholar] [CrossRef] [PubMed]
- Haux, J.; Klepp, O.; Spigset, O.; Tretli, S. Digitoxin medication and cancer; case control and internal dose-response studies. BMC Cancer 2001, 1, 11. [Google Scholar] [CrossRef]
- Mijatovic, T.; Dufrasne, F.; Kiss, R. Cardiotonic steroids-mediated targeting of the Na(+)/K(+)-ATPase to combat chemoresistant cancers. Curr. Med. Chem. 2012, 19, 627–646. [Google Scholar] [CrossRef]
- Banerjee, M.; Cui, X.; Li, Z.; Yu, H.; Cai, L.; Jia, X.; He, D.; Wang, C.; Gao, T.; Xie, Z. Na/K-ATPase Y260 Phosphorylation-mediated Src Regulation in Control of Aerobic Glycolysis and Tumor Growth. Sci. Rep. 2018, 8, 12322. [Google Scholar] [CrossRef]
- De Souza, W.F.; Barbosa, L.A.; Liu, L.; de Araujo, W.M.; de-Freitas-Junior, J.C.; Fortunato-Miranda, N.; Fontes, C.F.; Morgado-Díaz, J.A. Ouabain-induced alterations of the apical junctional complex involve α1 and β1 Na,K-ATPase downregulation and ERK1/2 activation independent of caveolae in colorectal cancer cells. J. Membr. Biol. 2014, 247, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Z.; Xie, J.X.; Li, X.; Tian, J.; Cai, T.; Cui, H.; Ding, H.; Shapiro, J.I.; Xie, Z. Na/K-ATPase mimetic pNaKtide peptide inhibits the growth of human cancer cells. J. Biol. Chem. 2011, 286, 32394–32403. [Google Scholar] [CrossRef] [PubMed]
- Benfante, R.; Antonini, R.A.; Vaccari, M.; Flora, A.; Chen, F.; Clementi, F.; Fornasari, D. The expression of the human neuronal alpha3 Na+, K+-ATPase subunit gene is regulated by the activity of the Sp1 and NF-Y transcription factors. Biochem. J. 2005, 386, 63–72. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Suñol, M.; Cusi, V.; Cruz, O.; Kiss, R.; Lefranc, F. Immunohistochemical analyses of alpha1 and alpha3 Na+/K+-ATPase subunit expression in medulloblastomas. Anticancer Res. 2011, 31, 953–958. [Google Scholar] [PubMed]
- Lefranc, F.; Kiss, R. The sodium pump alpha1 subunit as a potential target to combat apoptosis-resistant glioblastomas. Neoplasia 2008, 10, 198–206. [Google Scholar] [CrossRef] [PubMed]
- Li, D.; Qu, X.; Hou, K.; Zhang, Y.; Dong, Q.; Teng, Y.; Zhang, J.; Liu, Y. PI3K/Akt is involved in bufalin-induced apoptosis in gastric cancer cells. Anticancer Drugs 2009, 20, 59–64. [Google Scholar] [CrossRef] [PubMed]
- Felth, J.; Rickardson, L.; Rosén, J.; Wickström, M.; Fryknäs, M.; Lindskog, M.; Bohlin, L.; Gullbo, J. Cytotoxic effects of cardiac glycosides in colon cancer cells, alone and in combination with standard chemotherapeutic drugs. J. Nat. Prod. 2009, 72, 1969–1974. [Google Scholar] [CrossRef]
- Mijatovic, T.; Kiss, R. Cardiotonic steroids-mediated Na+/K+-ATPase targeting could circumvent various chemoresistance pathways. Planta Med. 2013, 79, 189–198. [Google Scholar] [CrossRef]
- Botelho, A.F.M.; Pierezan, F.; Soto-Blanco, B.; Melo, M.M. A review of cardiac glycosides: Structure, toxicokinetics, clinical signs, diagnosis and antineoplastic potential. Toxicon 2019, 158, 63–68. [Google Scholar] [CrossRef]
- Pan, Z.; Luo, Y.; Xia, Y.; Zhang, X.; Qin, Y.; Liu, W.; Li, M.; Liu, X.; Zheng, Q.; Li, D. Cinobufagin induces cell cycle arrest at the S phase and promotes apoptosis in nasopharyngeal carcinoma cells. Biomed. Pharmacother. 2020, 122, 109763. [Google Scholar] [CrossRef]
- Guo, Y.; Liang, F.; Zhao, F.; Zhao, J. Resibufogenin suppresses tumor growth and Warburg effect through regulating miR-143-3p/HK2 axis in breast cancer. Mol. Cell. Biochem. 2020, 466, 103–115. [Google Scholar] [CrossRef]
- Fang, S.; Tao, H.; Xia, K.; Guo, W. Proscillaridin A induces apoptosis and inhibits the metastasis of osteosarcoma in vitro and in vivo. Biochem. Biophys. Res. Commun. 2020, 521, 880–886. [Google Scholar] [CrossRef] [PubMed]
- Liu, B.; Wang, W.; Fan, J.; Long, Y.; Xiaom, F.; Daniyal, M.; Tong, C.; Xie, Q.; Jian, Y.; Li, B.; et al. RBC membrane camouflaged prussian blue nanoparticles for gamabutolin loading and combined chemo/photothermal therapy of breast cancer. Biomaterials 2019, 217, 119301. [Google Scholar] [CrossRef]
- Henn, D.; Venter, A.; Botha, C. In Vitro Cytotoxicity Induced by the Bufadienolides 1α, 2α-Epoxyscillirosidine and Lanceotoxin B on Rat Myocardial and Mouse Neuroblastoma Cell Lines. Toxins 2019, 11, 14. [Google Scholar] [CrossRef] [PubMed]
- Lenaerts, C.; Bond, L.; Tuytten, R.; Blankert, B. Revealing of endogenous marinobufagin by an ultra-specific and sensitive UHPLC-MS/MS assay in pregnant women. Talanta 2018, 187, 193–199. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Agalakova, N.I.; Kolodkin, N.I.; Adair, C.D.; Trashkov, A.P.; Bagrov, A.Y. Preeclampsia: Cardiotonic Steroids, Fibrosis, Fli1 and Hint to Carcinogenesis. Int. J. Mol. Sci. 2021, 22, 1941. https://doi.org/10.3390/ijms22041941
Agalakova NI, Kolodkin NI, Adair CD, Trashkov AP, Bagrov AY. Preeclampsia: Cardiotonic Steroids, Fibrosis, Fli1 and Hint to Carcinogenesis. International Journal of Molecular Sciences. 2021; 22(4):1941. https://doi.org/10.3390/ijms22041941
Chicago/Turabian StyleAgalakova, Natalia I., Nikolai I. Kolodkin, C. David Adair, Alexander P. Trashkov, and Alexei Y. Bagrov. 2021. "Preeclampsia: Cardiotonic Steroids, Fibrosis, Fli1 and Hint to Carcinogenesis" International Journal of Molecular Sciences 22, no. 4: 1941. https://doi.org/10.3390/ijms22041941
APA StyleAgalakova, N. I., Kolodkin, N. I., Adair, C. D., Trashkov, A. P., & Bagrov, A. Y. (2021). Preeclampsia: Cardiotonic Steroids, Fibrosis, Fli1 and Hint to Carcinogenesis. International Journal of Molecular Sciences, 22(4), 1941. https://doi.org/10.3390/ijms22041941







