Stromal CCL5 Promotes Breast Cancer Progression by Interacting with CCR3 in Tumor Cells
Abstract
1. Introduction
2. Results
2.1. Expression of CCL5, CCR1, CCR3 and CCR5 in 111 Breast Carcinoma Tissues
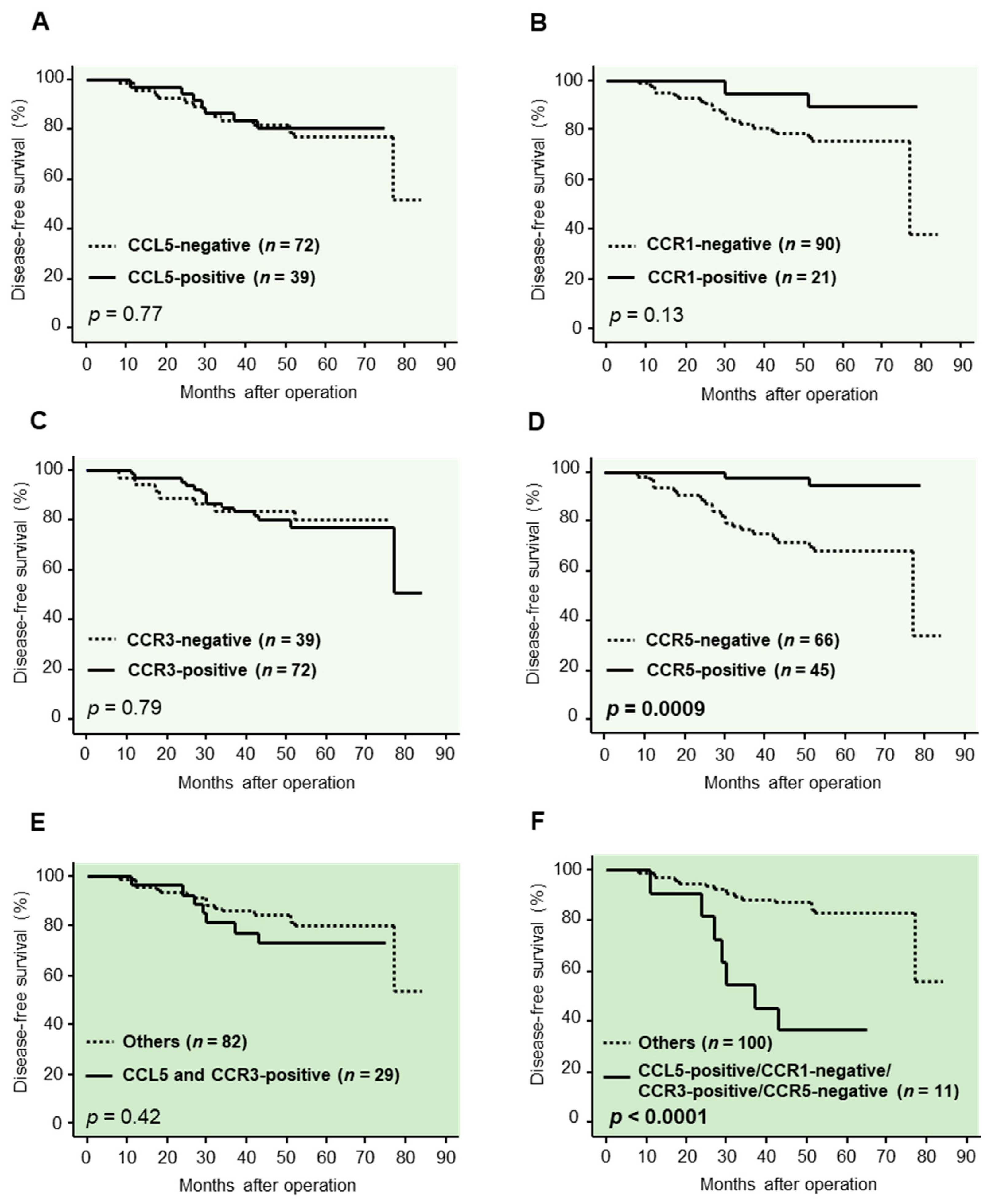
2.2. Association between Immunoreactivity for CCL5, CCR1 and CCR3 and CCR5 and the Clinical Outcome of 111 Breast Cancer Patients
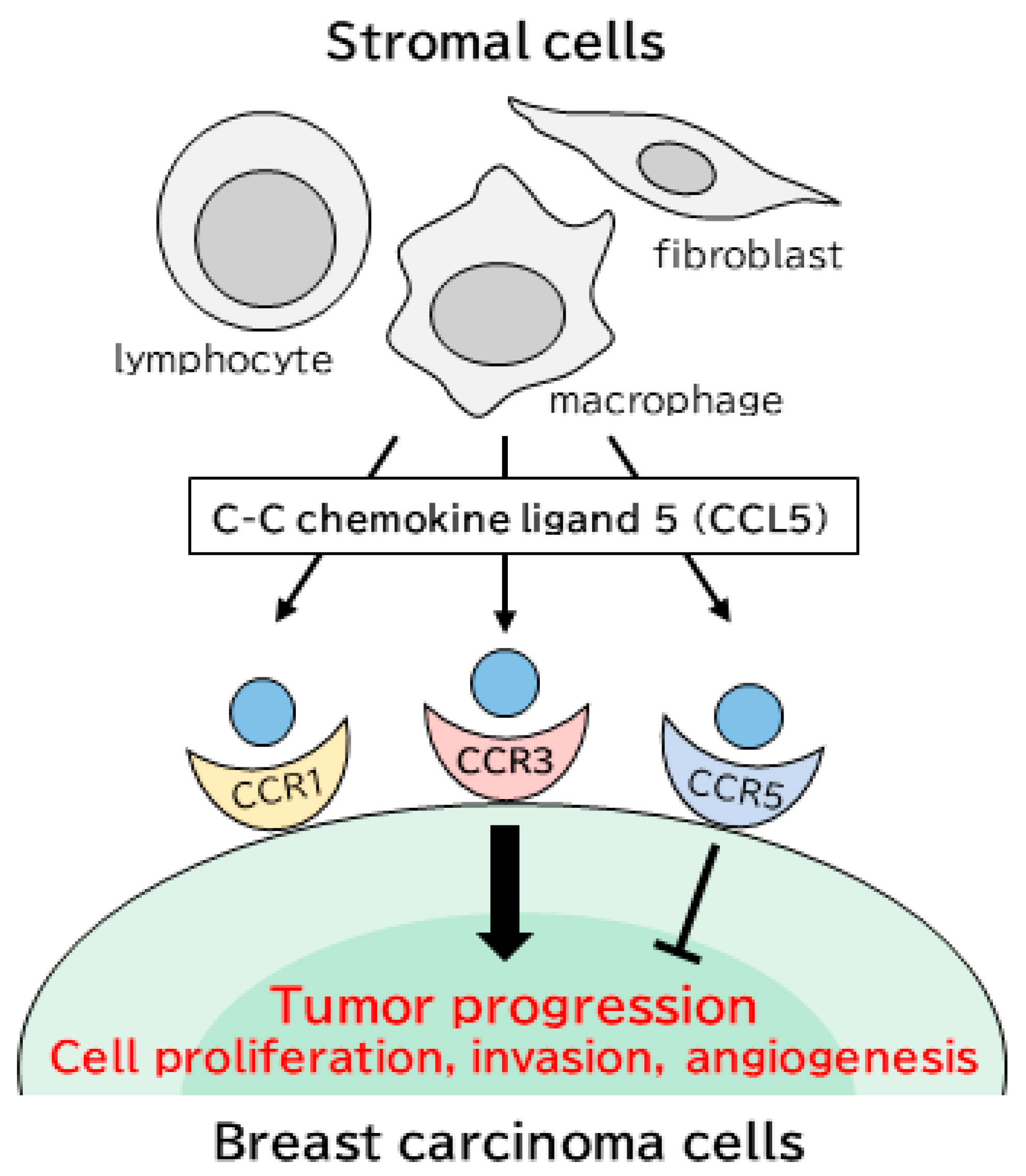
3. Discussion
4. Materials and Methods
4.1. Patients and Tissues
4.2. Immunohistochemistry
4.3. Scoring of Immunoreactivity
4.4. Statistical Analyses
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- Luqmani, Y.A.; Alam-Eldin, N. Overcoming Resistance to Endocrine Therapy in Breast Cancer: New Approaches to a Nagging Problem. Med. Princ. Pract. 2016, 25, 28–40. [Google Scholar] [CrossRef]
- Tevaarwerk, A.J.; Gray, R.J.; Schneider, B.P.; Smith, M.L.; Wagner, L.I.; Fetting, J.H.; Davidson, N.; Goldstein, L.J.; Miller, K.D.; Sparano, J.A. Survival in patients with metastatic recurrent breast cancer after adjuvant chemotherapy: Little evidence of improvement over the past 30 years. Cancer 2013, 119, 1140–1148. [Google Scholar] [CrossRef] [PubMed]
- De Palma, M.; Biziato, D.; Petrova, T.V. Microenvironmental regulation of tumour angiogenesis. Nat. Rev. Cancer 2017, 17, 457–474. [Google Scholar] [CrossRef]
- Viola, A.; Luster, A.D. Chemokines and their receptors: Drug targets in immunity and inflammation. Annu. Rev. Pharmacol. Toxicol. 2008, 48, 171–197. [Google Scholar] [CrossRef]
- Balkwill, F. Cancer and the chemokine network. Nat. Rev. Cancer 2004, 4, 540–550. [Google Scholar] [CrossRef] [PubMed]
- Aldinucci, D.; Colombatti, A. The inflammatory chemokine CCL5 and cancer progression. Mediat. Inflamm. 2014, 2014, 292376. [Google Scholar] [CrossRef]
- Huang, R.; Wang, S.; Wang, N.; Zheng, Y.; Zhou, J.; Yang, B.; Wang, X.; Zhang, J.; Guo, L.; Wang, S.; et al. CCL5 derived from tumor-associated macrophages promotes prostate cancer stem cells and metastasis via activating beta-catenin/STAT3 signaling. Cell Death Dis. 2020, 11, 234. [Google Scholar] [CrossRef] [PubMed]
- Ding, H.; Zhao, L.; Dai, S.; Li, L.; Wang, F.; Shan, B. CCL5 secreted by tumor associated macrophages may be a new target in treatment of gastric cancer. Biomed. Pharmacotherapy 2016, 77, 142–149. [Google Scholar] [CrossRef]
- Liu, C.; Yao, Z.; Wang, J.; Zhang, W.; Yang, Y.; Zhang, Y.; Qu, X.; Zhu, Y.; Zou, J.; Peng, S.; et al. Macrophage-derived CCL5 facilitates immune escape of colorectal cancer cells via the p65/STAT3-CSN5-PD-L1 pathway. Cell Death Differ. 2020, 27, 1765–1781. [Google Scholar] [CrossRef]
- Zhou, B.; Sun, C.; Li, N.; Shan, W.; Lu, H.; Guo, L.; Guo, E.; Xia, M.; Weng, D.; Meng, L.; et al. Cisplatin-induced CCL5 secretion from CAFs promotes cisplatin-resistance in ovarian cancer via regulation of the STAT3 and PI3K/Akt signaling pathways. Int. J. Oncol. 2016, 48, 2087–2097. [Google Scholar] [CrossRef]
- Sugasawa, H.; Ichikura, T.; Kinoshita, M.; Ono, S.; Majima, T.; Tsujimoto, H.; Chochi, K.; Hiroi, S.; Takayama, E.; Saitoh, D.; et al. Gastric cancer cells exploit CD4+ cell-derived CCL5 for their growth and prevention of CD8+ cell-involved tumor elimination. Int. J. Cancer 2008, 122, 2535–2541. [Google Scholar] [CrossRef]
- Lin, S.; Sun, L.; Lyu, X.; Ai, X.; Du, D.; Su, N.; Li, H.; Zhang, L.; Yu, J.; Yuan, S. Lactate-activated macrophages induced aerobic glycolysis and epithelial-mesenchymal transition in breast cancer by regulation of CCL5-CCR5 axis: A positive metabolic feedback loop. Oncotarget 2017, 8, 110426–110443. [Google Scholar] [CrossRef]
- D’Esposito, V.; Liguoro, D.; Ambrosio, M.R.; Collina, F.; Cantile, M.; Spinelli, R.; Raciti, G.A.; Miele, C.; Valentino, R.; Campiglia, P.; et al. Adipose microenvironment promotes triple negative breast cancer cell invasiveness and dissemination by producing CCL5. Oncotarget 2016, 7, 24495–24509. [Google Scholar] [CrossRef]
- Gao, D.; Rahbar, R.; Fish, E.N. CCL5 activation of CCR5 regulates cell metabolism to enhance proliferation of breast cancer cells. Open Biol. 2016, 6, 160122. [Google Scholar] [CrossRef]
- Mañes, S.; Mira, E.; Colomer, R.; Montero, S.; Real, L.M.; Gómez-Moutón, C.; Jiménez-Baranda, S.; Garzón, A.; Lacalle, R.A.; Harshman, K.; et al. CCR5 expression influences the progression of human breast cancer in a p53-dependent manner. J. Exp. Med. 2003, 198, 1381–1389. [Google Scholar] [CrossRef]
- Place, A.E.; Jin, H.S.; Polyak, K. The microenvironment in breast cancer progression: Biology and implications for treatment. Breast Cancer Res. 2011, 13, 227. [Google Scholar] [CrossRef]
- Yeh, C.R.; Slavin, S.; Da, J.; Hsu, I.; Luo, J.; Xiao, G.Q.; Ding, J.; Chou, F.J.; Yeh, S. Estrogen receptor alpha in cancer associated fibroblasts suppresses prostate cancer invasion via reducing CCL5, IL6 and macrophage infiltration in the tumor microenvironment. Mol. Cancer 2016, 15, 7. [Google Scholar] [CrossRef]
- Yang, T.; Chen, M.; Yang, X.; Zhang, X.; Zhang, Z.; Sun, Y.; Xu, B.; Hua, J.; He, Z.; Song, Z. Down-regulation of KLF5 in cancer-associated fibroblasts inhibit gastric cancer cells progression by CCL5/CCR5 axis. Cancer Biol. Ther. 2017, 18, 806–815. [Google Scholar] [CrossRef]
- Zimmermann, N.; Conkright, J.J.; Rothenberg, M.E. CC chemokine receptor-3 undergoes prolonged ligand-induced internalization. J. Biol. Chem. 1999, 274, 12611–126118. [Google Scholar] [CrossRef]
- Elsner, J.; Dulkys, Y.; Gupta, S.; Escher, S.E.; Forssmann, W.G.; Kapp, A.; Forssmann, U. Differential pattern of CCR1 internalization in human eosinophils: Prolonged internalization by CCL5 in contrast to CCL3. Allergy 2005, 60, 1386–1393. [Google Scholar] [CrossRef]
- Mueller, A.; Kelly, E.; Strange, P.G. Pathways for internalization and recycling of the chemokine receptor CCR5. Blood 2002, 99, 785–791. [Google Scholar] [CrossRef] [PubMed]
- Vasiliadou, I.; Holen, I. The role of macrophages in bone metastasis. J. Bone Oncol. 2013, 2, 158–166. [Google Scholar] [CrossRef]
- Yang, J.; Li, X.; Liu, X.; Liu, Y. The role of tumor-associated macrophages in breast carcinoma invasion and metastasis. Int. J. Clin. Exp. Pathol. 2015, 8, 6656–6664. [Google Scholar] [PubMed]
- Li, J.; He, K.; Liu, P.; Xu, L.X. Iron participated in breast cancer chemoresistance by reinforcing IL-6 paracrine loop. Biochem. Biophys. Res. Commun. 2016, 475, 154–160. [Google Scholar] [CrossRef] [PubMed]
- Gan, L.; Qiu, Z.; Huang, J.; Li, Y.; Huang, H.; Xiang, T.; Wan, J.; Hui, T.; Lin, Y.; Li, H.; et al. Cyclooxygenase-2 in tumor-associated macrophages promotes metastatic potential of breast cancer cells through Akt pathway. Int. J. Biol. Sci. 2016, 12, 1533–1543. [Google Scholar] [CrossRef]
- Miyagaki, T.; Sugaya, M.; Murakami, T.; Asano, Y.; Tada, Y.; Kadono, T.; Okochi, H.; Tamaki, K.; Sato, S. CCL11-CCR3 interactions promote survival of anaplastic large cell lymphoma cells via ERK1/2 activation. Cancer Res. 2011, 71, 2056–2065. [Google Scholar] [CrossRef]
- Zhu, F.; Liu, P.; Li, J.; Zhang, Y. Eotaxin-1 promotes prostate cancer cell invasion via activation of the CCR3-ERK pathway and upregulation of MMP-3 expression. Oncol. Rep. 2014, 31, 2049–2054. [Google Scholar] [CrossRef]
- Lee, Y.S.; Kim, S.Y.; Song, S.J.; Hong, H.K.; Lee, Y.; Oh, B.Y.; Lee, W.Y.; Cho, Y.B. Crosstalk between CCL7 and CCR3 promotes metastasis of colon cancer cells via ERK-JNK signaling pathways. Oncotarget 2016, 7, 36842–36853. [Google Scholar] [CrossRef]
- Murooka, T.T.; Rahbar, R.; Fish, E.N. CCL5 promotes proliferation of MCF-7 cells through mTOR-dependent mRNA translation. Biochem. Biophys. Res. Commun. 2009, 387, 381–386. [Google Scholar] [CrossRef]
- Gong, D.H.; Fan, L.; Chen, H.Y.; Ding, K.F.; Yu, K.D. Intratumoral expression of CCR3 in breast cancer is associated with improved relapse-free survival in luminal-like disease. Oncotarget 2016, 7, 28570–28578. [Google Scholar] [CrossRef][Green Version]
- Takagi, K.; Miki, Y.; Onodera, Y.; Ishida, T.; Watanabe, M.; Sasano, H.; Suzuki, T. ARHGAP15 in Human Breast Carcinoma: A Potent Tumor Suppressor Regulated by Androgens. Int. J. Mol. Sci. 2018, 19, 804. [Google Scholar] [CrossRef]
- Sato, A.; Takagi, K.; Miki, Y.; Yoshimura, A.; Hara, M.; Ishida, T.; Sasano, H.; Suzuki, T. Cytochrome c1 as a favorable prognostic marker in estrogen receptor-positive breast carcinoma. Histol. Histopathol. 2019, 34, 1365–1375. [Google Scholar]
- Yamaguchi, M.; Takagi, K.; Sato, A.; Miki, Y.; Miyashita, M.; Sasano, H.; Suzuki, T. Rac1 activation in human breast carcinoma as a prognostic factor associated with therapeutic resistance. Breast Cancer 2020, 27, 919–928. [Google Scholar] [CrossRef]
- Agarwal, P.; Sen, A.K.; Bhardwaj, M.; Dinand, V.; Ahuja, A.; Sood, R. Study of Proliferating cell nuclear antigen expression and Angiogenesis in Urothelial neoplasms: Correlation with tumor grade and stage. Urol. Ann. 2018, 10, 209–214. [Google Scholar] [CrossRef] [PubMed]
- Hamlin, I.M. Possible host resistance in carcinoma of the breast: A histological study. Br. J. Cancer. 1968, 22, 383–401. [Google Scholar] [CrossRef]
- Hammond, M.E.; Hayes, D.F.; Wolff, A.C.; Mangu, P.B.; Temin, S. American society of clinical oncology/college of american pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J. Oncol. Pract. 2010, 6, 195–197. [Google Scholar] [CrossRef]

| Clinicopathological Factors | CCL5 Status | p-Value | |
|---|---|---|---|
| Negative (n = 72) | Positive (n = 39) | ||
| Age * | 56 (29–82) | 56 (27–87) | 0.40 |
| Menopause | |||
| Premenopausal | 29 | 12 | 0.32 |
| Postmenopausal | 43 | 27 | |
| Stage | |||
| I | 41 | 22 | 0.56 |
| II | 17 | 12 | |
| III | 14 | 5 | |
| Pathological T Factor | |||
| pT1 | 48 | 25 | 0.79 |
| pT2-4 | 24 | 14 | |
| Lymph Node Metastasis | |||
| Negative | 49 | 26 | 0.88 |
| Positive | 23 | 13 | |
| Histological Grade | |||
| 1 (well) | 32 | 11 | 0.0061 |
| 2 (intermediate) | 31 | 13 | |
| 3 (poor) | 9 | 15 | |
| ER | |||
| Negative | 8 | 13 | 0.0043 |
| Positive | 64 | 26 | |
| PR | |||
| Negative | 15 | 20 | 0.0010 |
| Positive | 57 | 19 | |
| HER2 | |||
| Negative | 63 | 33 | 0.67 |
| Positive | 9 | 6 | |
| Ki67 LI (%) * | 9.5 (1–60) | 16 (1–49) | 0.090 |
| MVD * | 24.5 (3–101) | 32 (7–106) | 0.040 |
| Macrophage infiltration | |||
| Low | 55 | 20 | 0.0070 |
| High | 17 | 19 | |
| Clinicopathological Factors | CCR1-Positive Group | CCR3-Positive Group | CCR5-Positive Group | ||||||
|---|---|---|---|---|---|---|---|---|---|
| CCL5 Status | p-Value | CCL5 Status | p-Value | CCL5 Status | p-Value | ||||
| Negative (n = 11) | Positive (n = 10) | Negative (n = 43) | Positive (n = 29) | Negative (n = 25) | Positive (n = 20) | ||||
| Stage | |||||||||
| I | 5 | 5 | 0.36 | 24 | 17 | 0.73 | 13 | 16 | 0.13 |
| II | 4 | 5 | 10 | 8 | 7 | 3 | |||
| III | 2 | 0 | 9 | 4 | 5 | 1 | |||
| Pathological T Factor | |||||||||
| pT1 | 6 | 8 | 0.22 | 27 | 20 | 0.60 | 14 | 17 | 0.037 |
| pT2-4 | 5 | 2 | 16 | 9 | 11 | 3 | |||
| Lymph Node Metastasis | |||||||||
| Negative | 7 | 5 | 0.53 | 30 | 19 | 0.70 | 15 | 18 | 0.024 |
| Positive | 4 | 5 | 13 | 10 | 10 | 2 | |||
| Histological Grade | |||||||||
| 1 (well) | 4 | 4 | 0.14 | 15 | 8 | 0.028 | 11 | 9 | 0.92 |
| 2 (intermediate) | 5 | 1 | 22 | 9 | 10 | 7 | |||
| 3 (poor) | 2 | 5 | 6 | 12 | 4 | 4 | |||
| ER | |||||||||
| Negative | 2 | 3 | 0.53 | 6 | 10 | 0.040 | 4 | 7 | 0.14 |
| Positive | 9 | 7 | 37 | 19 | 21 | 13 | |||
| PR | |||||||||
| Negative | 3 | 5 | 0.28 | 10 | 16 | 0.0057 | 4 | 8 | 0.070 |
| Positive | 8 | 5 | 33 | 13 | 21 | 12 | |||
| HER2 | |||||||||
| Negative | 8 | 7 | 0.89 | 36 | 24 | 0.91 | 20 | 17 | 0.66 |
| Positive | 3 | 3 | 7 | 5 | 5 | 3 | |||
| Ki67 LI (%) * | 16 (1–30) | 19 (1–37) | 0.60 | 13 (1–53) | 18 (1–44) | 0.062 | 12 (1–53) | 13.5 (1–47) | 0.73 |
| MVD * | 28 (13–101) | 30.5 (18–82) | 0.84 | 23 (6–101) | 36 (7–106) | 0.044 | 28 (10–101) | 27 (7–82) | 0.62 |
| Clinicopathological Factors | CCR1-Negative/CCR3-Positive/ CCR5-Negative Cases | Others | ||||
|---|---|---|---|---|---|---|
| CCL5 Status | p-Value | CCL5 Status | p-Value | |||
| Negative (n = 23) | Positive (n = 11) | Negative (n = 49) | Positive (n = 28) | |||
| Stage | ||||||
| I | 15 | 4 | 0.27 | 26 | 18 | 0.30 |
| II | 4 | 4 | 13 | 8 | ||
| III | 4 | 3 | 10 | 2 | ||
| Pathological T Factor | ||||||
| pT1 | 18 | 4 | 0.0017 | 30 | 21 | 0.22 |
| pT2-4 | 5 | 7 | 19 | 7 | ||
| Lymph Node Metastasis | ||||||
| Negative | 18 | 5 | 0.056 | 31 | 21 | 0.29 |
| Positive | 5 | 6 | 18 | 7 | ||
| Histological Grade | ||||||
| 1 (well) | 8 | 0 | 0.0015 | 24 | 11 | 0.35 |
| 2 (intermediate) | 12 | 3 | 19 | 10 | ||
| 3 (poor) | 3 | 8 | 6 | 7 | ||
| ER | ||||||
| Negative | 2 | 5 | 0.013 | 6 | 8 | 0.074 |
| Positive | 21 | 6 | 43 | 20 | ||
| PR | ||||||
| Negative | 5 | 9 | 0.0009 | 10 | 11 | 0.074 |
| Positive | 18 | 2 | 39 | 17 | ||
| HER2 | ||||||
| Negative | 20 | 10 | 0.74 | 43 | 23 | 0.50 |
| Positive | 3 | 1 | 6 | 5 | ||
| Ki67 LI (%) * | 13 (1–49) | 22 (3–44) | 0.037 | 9 (160) | 12 (1–49) | 0.44 |
| MVD * | 22 (6–52) | 40 (14–106) | 0.025 | 27 (3–101) | 28.5 (7–82) | 0.38 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yamaguchi, M.; Takagi, K.; Narita, K.; Miki, Y.; Onodera, Y.; Miyashita, M.; Sasano, H.; Suzuki, T. Stromal CCL5 Promotes Breast Cancer Progression by Interacting with CCR3 in Tumor Cells. Int. J. Mol. Sci. 2021, 22, 1918. https://doi.org/10.3390/ijms22041918
Yamaguchi M, Takagi K, Narita K, Miki Y, Onodera Y, Miyashita M, Sasano H, Suzuki T. Stromal CCL5 Promotes Breast Cancer Progression by Interacting with CCR3 in Tumor Cells. International Journal of Molecular Sciences. 2021; 22(4):1918. https://doi.org/10.3390/ijms22041918
Chicago/Turabian StyleYamaguchi, Mio, Kiyoshi Takagi, Koki Narita, Yasuhiro Miki, Yoshiaki Onodera, Minoru Miyashita, Hironobu Sasano, and Takashi Suzuki. 2021. "Stromal CCL5 Promotes Breast Cancer Progression by Interacting with CCR3 in Tumor Cells" International Journal of Molecular Sciences 22, no. 4: 1918. https://doi.org/10.3390/ijms22041918
APA StyleYamaguchi, M., Takagi, K., Narita, K., Miki, Y., Onodera, Y., Miyashita, M., Sasano, H., & Suzuki, T. (2021). Stromal CCL5 Promotes Breast Cancer Progression by Interacting with CCR3 in Tumor Cells. International Journal of Molecular Sciences, 22(4), 1918. https://doi.org/10.3390/ijms22041918






