Homogentisic Acid and Gentisic Acid Biosynthesized Pyomelanin Mimics: Structural Characterization and Antioxidant Activity
Abstract
1. Introduction
2. Results
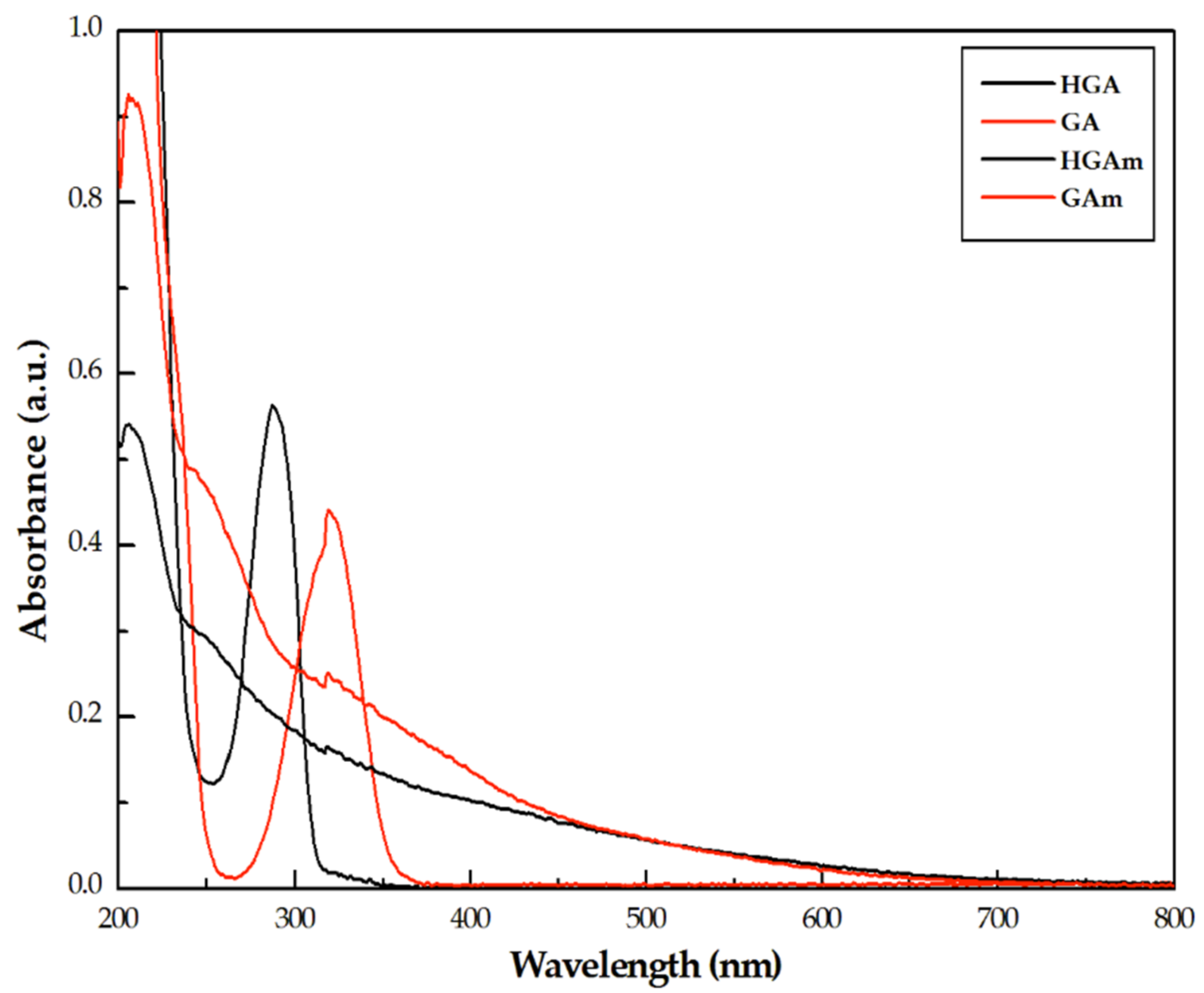
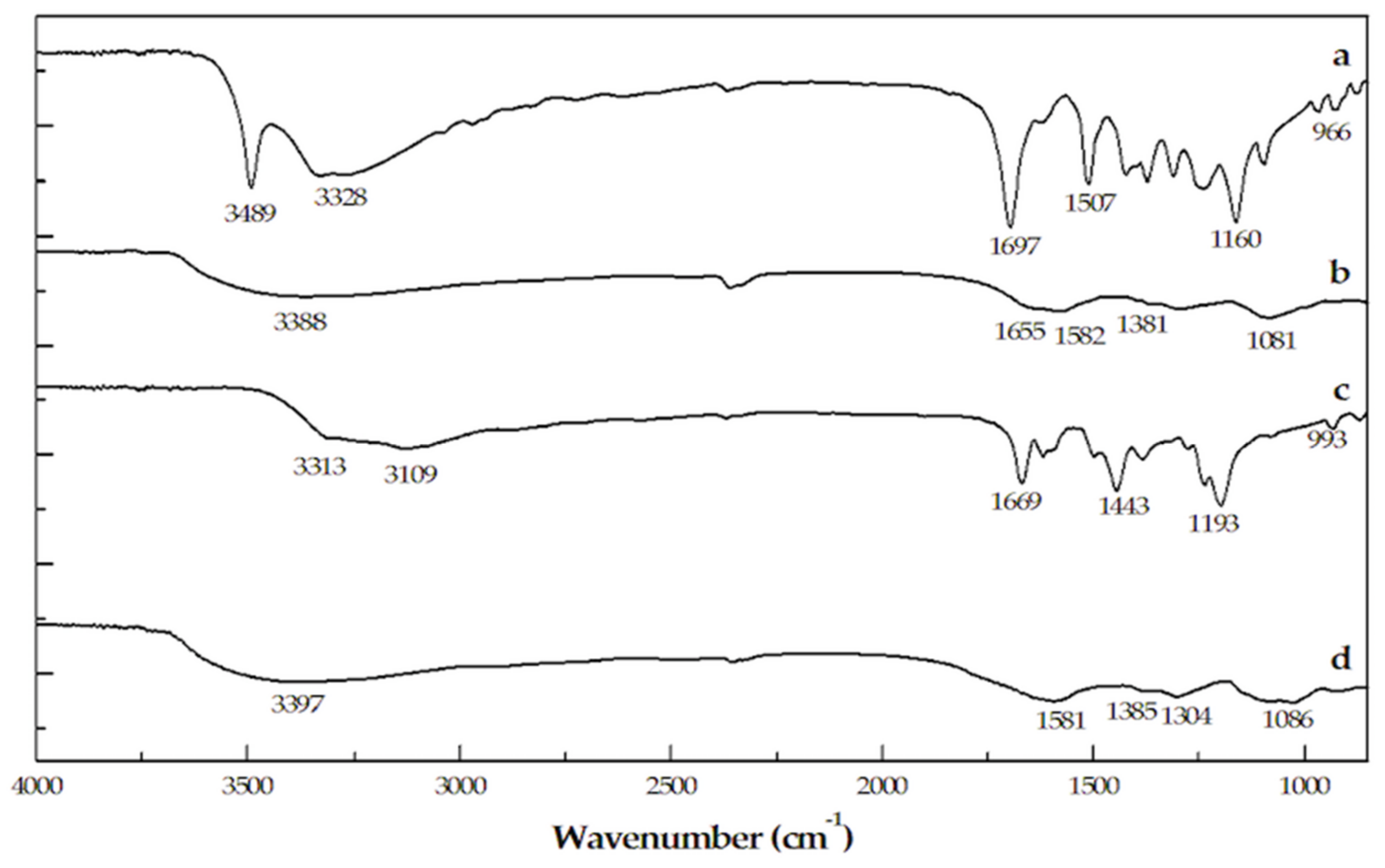
2.1. UV–Vis and FT-IR Characterization
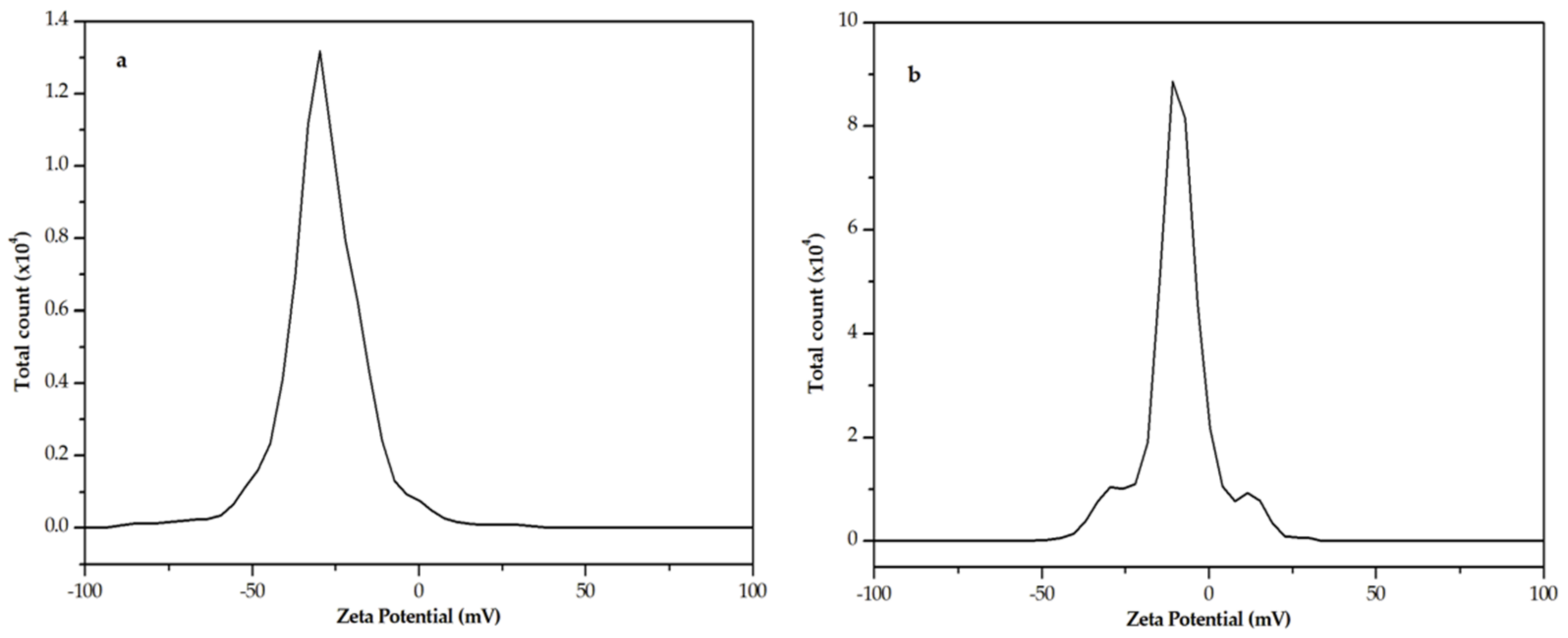
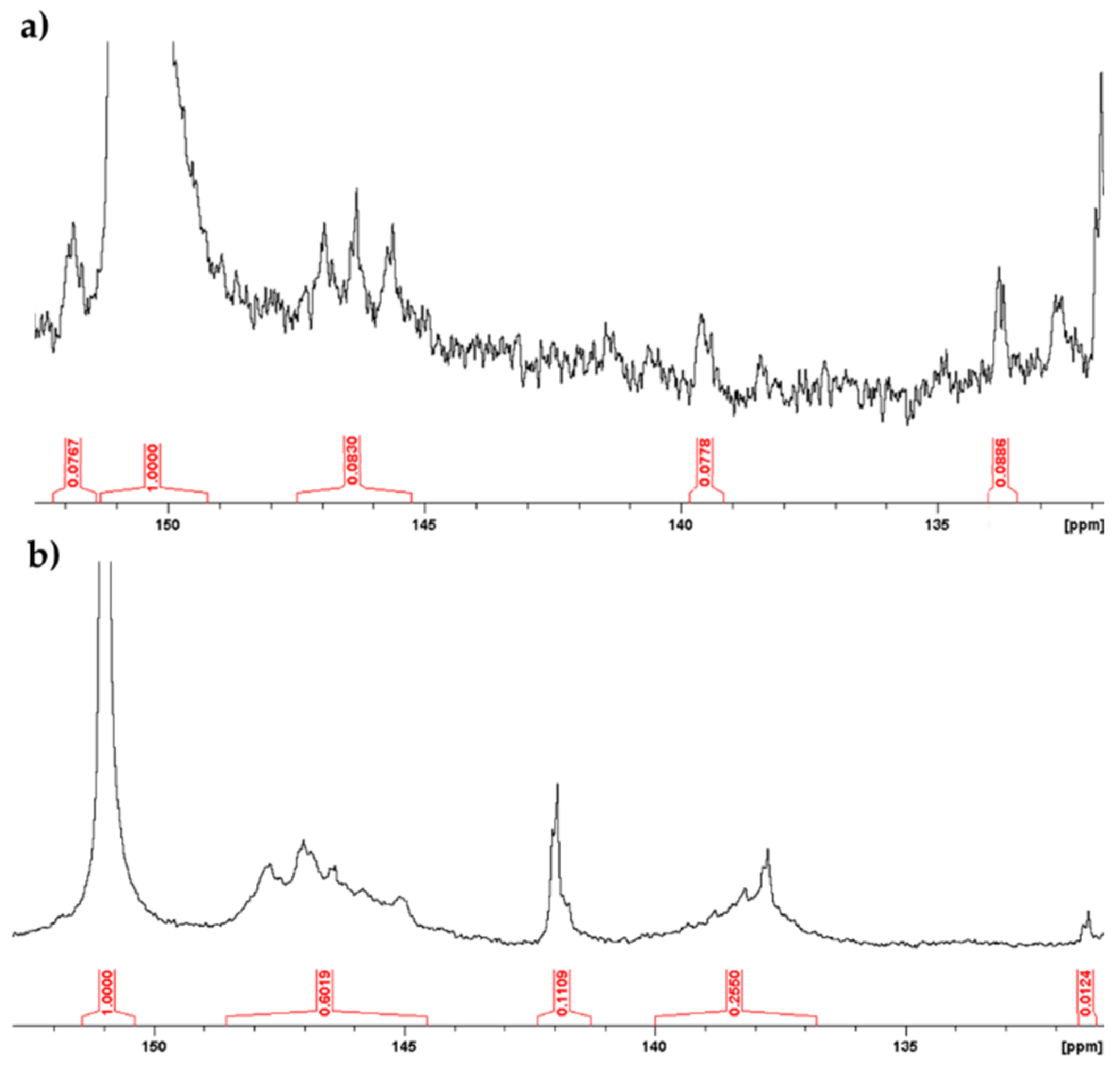
2.2. Dynamic Light Scattering (DLS) and Nuclear Magnetic Resonance (NMR) Analysis
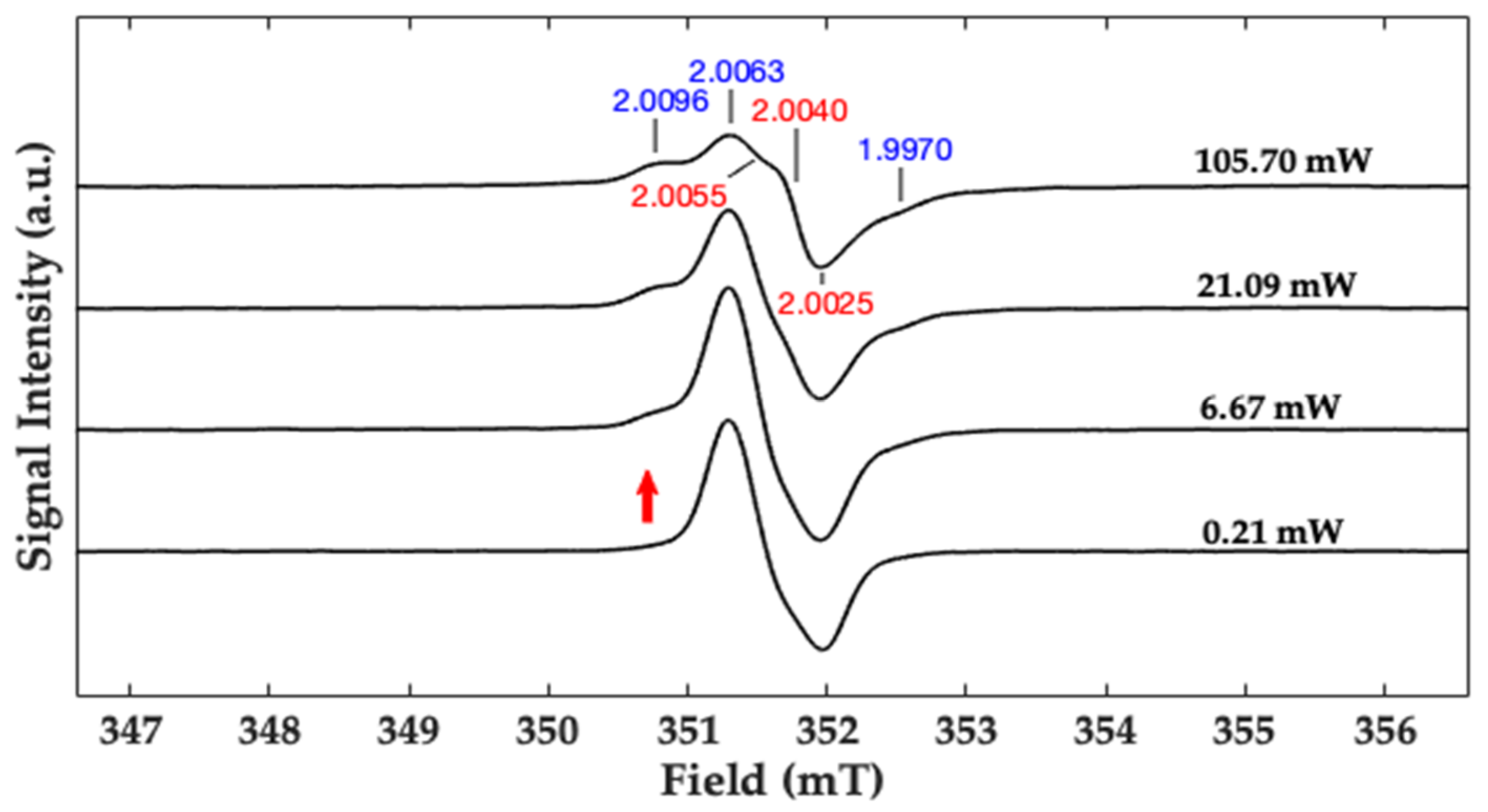
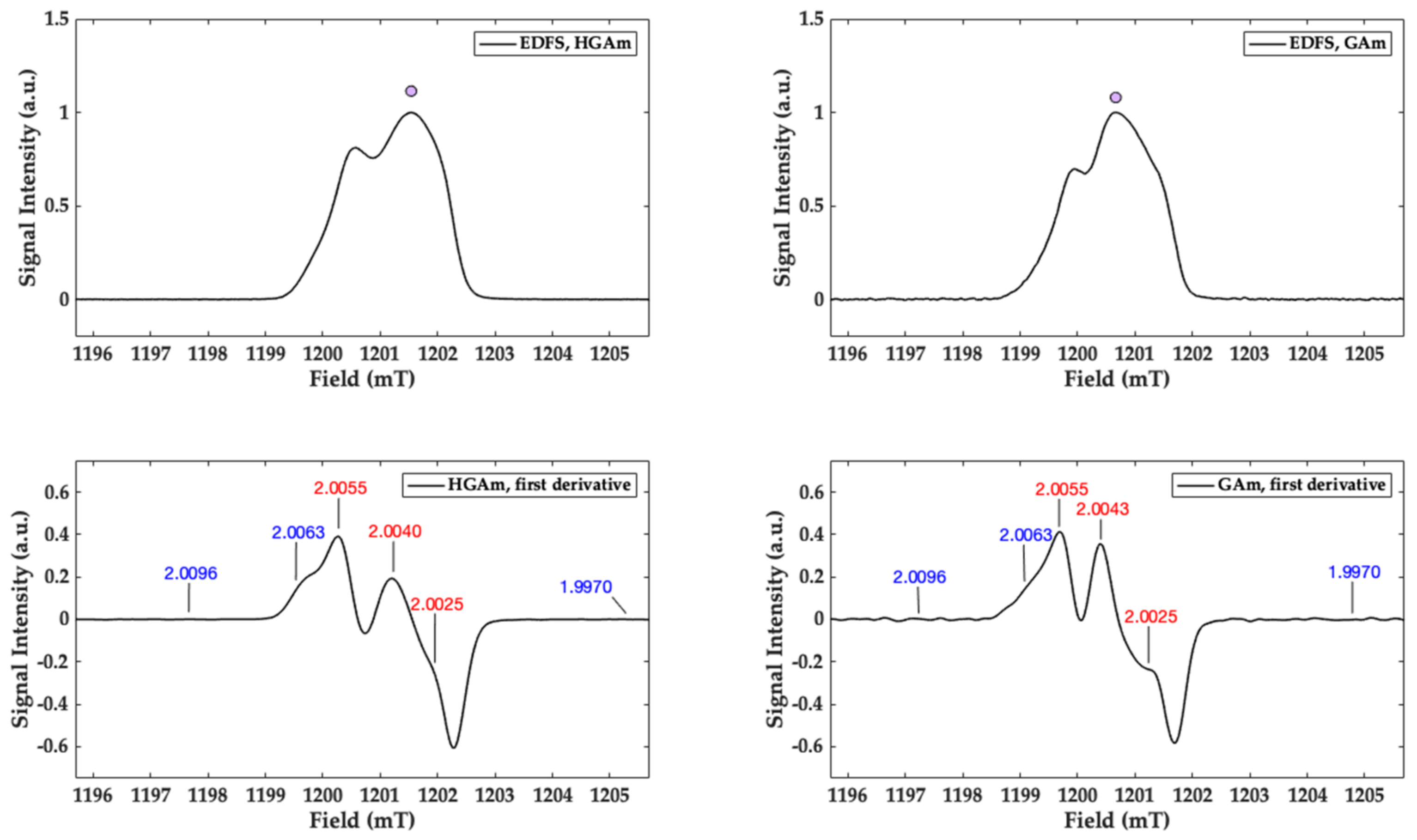
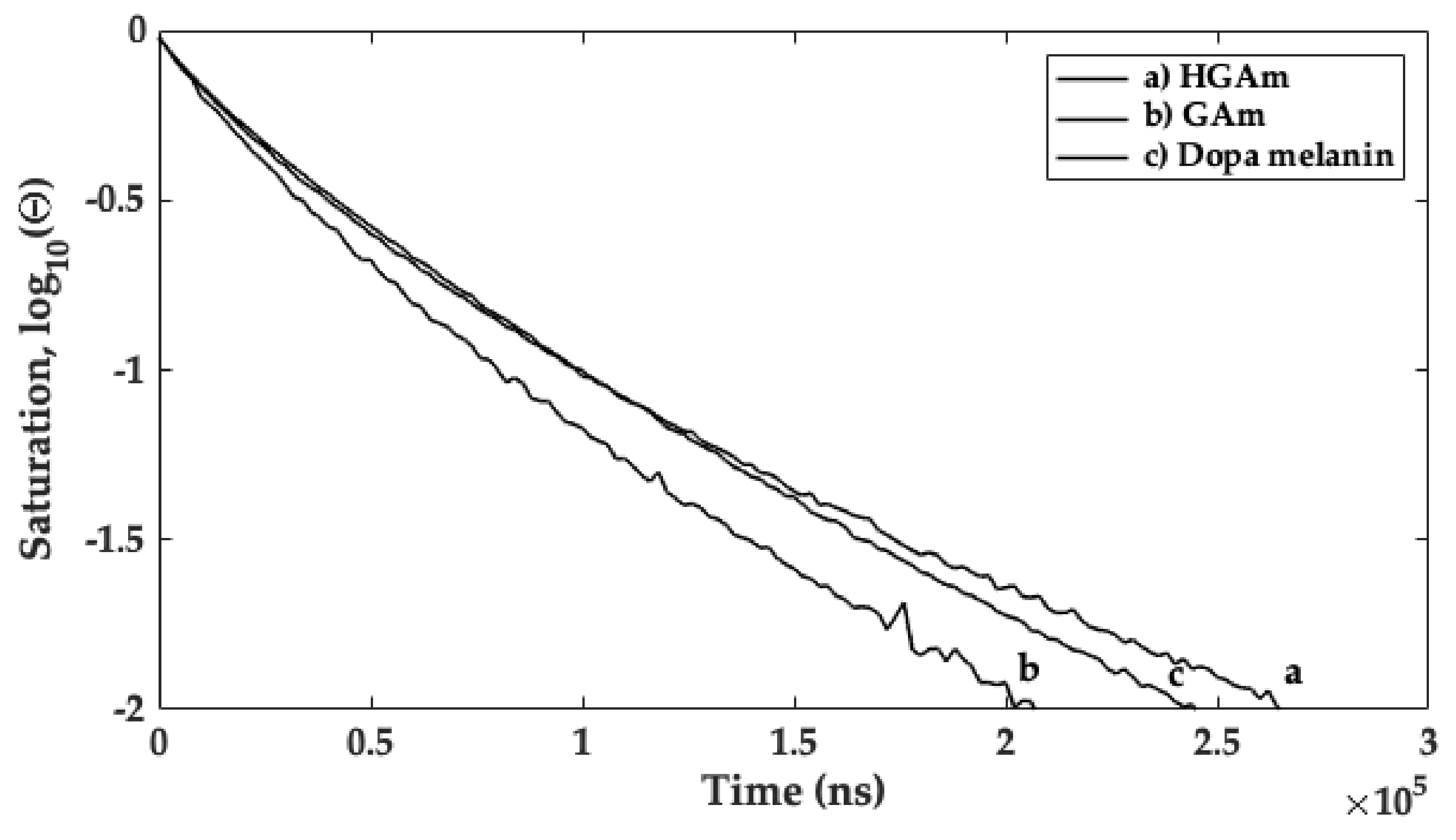
2.3. CW X-Band and Q-Band Pulse EPR Measurements
2.4. Antioxidant Properties and Scanning Electron Microscopy Analysis
3. Discussion
4. Materials and Methods
4.1. Synthetic Procedure and Spectrophotometric Analysis
4.2. DLS and NMR Analysis
4.3. Multifrequency Continuous Wave (CW) and Pulse EPR Analysis
4.4. EC50 Determination
4.5. Scanning Electron Microscopy (SEM) Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Solano, F. Melanins: Skin pigments and much more—types, structural models, biological functions, and formation routes. New J. Sci. 2014, 2014, 1–28. [Google Scholar] [CrossRef]
- D’Ischia, M.; Wakamatsu, K.; Napolitano, A.; Briganti, S.; Garcia-Borron, J.C.; Kovacs, D.; Meredith, P.; Pezzella, A.; Picardo, M.; Sarna, T.; et al. Melanins and melanogenesis: Methods, standards, protocols. Pigment Cell Melanoma Res. 2013, 26, 616–633. [Google Scholar] [CrossRef] [PubMed]
- Turick, C.E.; Knox, A.S.; Becnel, J.M.; Ekechukwu, A.A.; Millike, C.E. Properties and function of pyomelanin. Biopolymers 2010, 449, 72. [Google Scholar]
- Braconi, D.; Millucci, L.; Bernardini, G.; Santucci, A. Oxidative stress and mechanisms of ochronosis in alkaptonuria. Free Radic. Biol. Med. 2015, 88, 70–80. [Google Scholar] [CrossRef] [PubMed]
- Chow, W.Y.; Norman, B.P.; Roberts, N.B.; Ranganath, L.R.; Teutloff, C.; Bittl, R.; Duer, M.J.; Gallagher, J.A.; Oschkinat, H. Pigmentation chemistry and radical-based collagen degradation in alkaptonuria and osteoarthritic cartilage. Angew. Chemie Int. Ed. 2020, 59, 11937–11942. [Google Scholar] [CrossRef]
- Bernardus Mostert, A.; Davy, K.J.P.; Ruggles, J.L.; Powell, B.J.; Gentle, I.R.; Meredith, P. Gaseous adsorption in melanins: Hydrophilic biomacromolecules with high electrical conductivities. Langmuir 2010, 26, 412–416. [Google Scholar] [CrossRef]
- Pradhan, S.; Brooks, A.K.; Yadavalli, V.K. Nature-derived materials for the fabrication of functional biodevices. Mater. Today Bio 2020, 7, 100065–100088. [Google Scholar] [CrossRef] [PubMed]
- Caldas, M.; Santos, A.C.; Veiga, F.; Rebelo, R.; Reis, R.L.; Correlo, V.M. Melanin nanoparticles as a promising tool for biomedical applications–a review. Acta Biomater. 2020, 105, 26–43. [Google Scholar] [CrossRef] [PubMed]
- D’Ischia, M.; Napolitano, A.; Pezzella, A.; Meredith, P.; Sarna, T. Chemical and structural diversity in eumelanins: Unexplored bio-optoelectronic materials. Angew. Chemie Int. Ed. 2009, 48, 3914–3921. [Google Scholar] [CrossRef]
- Wang, Y.; Wang, X.; Li, T.; Ma, P.; Zhang, S.; Du, M.; Dong, W.; Xie, Y.; Chen, M. Effects of melanin on optical behavior of polymer: From natural pigment to materials applications. ACS Appl. Mater. Interfaces 2018, 10, 13100–13106. [Google Scholar] [CrossRef]
- Manini, P.; Lino, V.; Franchi, P.; Gentile, G.; Sibillano, T.; Giannini, C.; Picardi, E.; Napolitano, A.; Valgimigli, L.; Chiappe, C.; et al. A robust fungal allomelanin mimic: An antioxidant and potent π-electron donor with free-radical properties that can be tuned by ionic liquids. Chempluschem 2019, 84, 1331–1337. [Google Scholar] [CrossRef]
- Weidenfeld, I.; Zakian, C.; Duewell, P.; Chmyrov, A.; Ntziachristos, V.; Stiel, A.C. Homogentisic acid-derived pigment as a biocompatible label for optoacoustic imaging of macrophages. Nat. Commun. 2019, 10, 5056–5068. [Google Scholar] [CrossRef]
- Janusz, G.; Pawlik, A.; Świderska-Burek, U.; Polak, J.; Sulej, J.; Jarosz-Wilkołazka, A.; Paszczyński, A. Laccase properties, physiological functions, and evolution. Int. J. Mol. Sci. 2020, 21, 966. [Google Scholar] [CrossRef]
- Marcus, R.A.; Sutin, N. Electron transfers in chemistry and biology. Biochim. Biophys. Acta 1985, 811, 265–322. [Google Scholar] [CrossRef]
- Eslami, M.; Namazian, M.; Zare, H.R. Electrooxidation of homogentisic acid in aqueous and mixed solvent solutions: Experimental and theoretical studies. J. Phys. Chem. B 2013, 117, 2757–2763. [Google Scholar] [CrossRef] [PubMed]
- Joshi, R.; Gangabhagirathi, R.; Venu, S.; Adhikari, S.; Mukherjee, T. Antioxidant activity and free radical scavenging reactions of gentisic acid: In-vitro and pulse radiolysis studies. Free Radic. Res. 2012, 46, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Mostert, A.B.; Hanson, G.R.; Sarna, T.; Gentle, I.R.; Powell, B.J.; Meredith, P. Hydration-controlled X-band EPR spectroscopy: A tool for unravelling the complexities of the solid-state free radical in eumelanin. J. Phys. Chem. B 2013, 117, 4965–4972. [Google Scholar] [CrossRef]
- Meredith, P.; Sarna, T. The physical and chemical properties of eumelanin. Pigment Cell Res. 2006, 19, 572–594. [Google Scholar] [CrossRef]
- Panzella, L.; D’Errico, G.; Vitiello, G.; Perfetti, M.; Alfieri, M.L.; Napolitano, A.; D’Ischia, M. Disentangling structure-dependent antioxidant mechanisms in phenolic polymers by multiparametric EPR analysis. Chem. Commun. 2018, 54, 9426–9429. [Google Scholar] [CrossRef]
- Sarna, T.; Plonka, P.M. Biophysical Studies of Melanin. In Biomedical EPR, Part A: Free Radicals, Metals, Medicine, and Physiology; Springer: Berlin, Germany, 2005; ISBN 978-0-387-26741-8. [Google Scholar]
- Al Khatib, M.; Harir, M.; Costa, J.; Baratto, M.; Schiavo, I.; Trabalzini, L.; Pollini, S.; Rossolini, G.; Basosi, R.; Pogni, R. Spectroscopic characterization of natural melanin from a streptomyces cyaneofuscatus strain and comparison with melanin enzymatically synthesized by tyrosinase and laccase. Molecules 2018, 23, 1916. [Google Scholar] [CrossRef] [PubMed]
- Schmaler-Ripcke, J.; Sugareva, V.; Gebhardt, P.; Winkler, R.; Kniemeyer, O.; Heinekamp, T.; Brakhage, A.A. Production of pyomelanin, a second type of melanin, via the tyrosine degradation pathway in Aspergillus fumigatus. Appl. Environ. Microbiol. 2009, 75, 493–503. [Google Scholar] [CrossRef] [PubMed]
- Turick, C.E.; Tisa, L.S.; Caccavo, F. Melanin production and use as a soluble electron shuttle for Fe(III) oxide reduction and as a terminal electron acceptor by Shewanella algae BrY. Appl. Environ. Microbiol. 2002, 68, 2436–2444. [Google Scholar] [CrossRef]
- Pralea, I.E.; Moldovan, R.C.; Petrache, A.M.; Ilieș, M.; Hegheș, S.C.; Ielciu, I.; Nicoară, R.; Moldovan, M.; Ene, M.; Radu, M.; et al. From extraction to advanced analytical methods: The challenges of melanin analysis. Int. J. Mol. Sci. 2019, 20, 3943. [Google Scholar] [CrossRef] [PubMed]
- Taylor, A.M.; Vercruysse, K.P. Analysis of melanin-like pigment synthesized from homogentisic acid, with or without tyrosine, and its implications in alkaptonuria. JIMD Rep. 2017, 35, 79–85. [Google Scholar] [PubMed]
- Tokuhara, Y.; Shukuya, K.; Tanaka, M.; Sogabe, K.; Ejima, Y.; Hosokawa, S.; Ohsaki, H.; Morinishi, T.; Hirakawa, E.; Yatomi, Y.; et al. Absorbance measurements of oxidation of homogentisic acid accelerated by the addition of alkaline solution with sodium hypochlorite pentahydrate. Sci. Rep. 2018, 8, 1–10. [Google Scholar] [CrossRef] [PubMed]
- López, J.; Hernández-Alcántara, J.M.; Roquero, P.; Montiel, C.; Shirai, K.; Gimeno, M.; Bárzana, E. Trametes versicolor laccase oxidation of gallic acid toward a polyconjugated semiconducting material. J. Mol. Catal. B Enzym. 2013, 97, 100–105. [Google Scholar] [CrossRef]
- Meng, X.; Crestini, C.; Ben, H.; Hao, N.; Pu, Y.; Ragauskas, A.J.; Argyropoulos, D.S. Determination of hydroxyl groups in biorefinery resources via quantitative 31P NMR spectroscopy. Nat. Protoc. 2019, 14, 2627–2647. [Google Scholar] [CrossRef]
- Baiocco, P.; Barreca, A.M.; Fabbrini, M.; Galli, C.; Gentili, P. Promoting laccase activity towards non-phenolic substrates: A mechanistic investigation with some laccase-mediator systems. Org. Biomol. Chem. 2003, 1, 191–197. [Google Scholar] [CrossRef]
- Pogni, R.; Baratto, M.C.; Teutloff, C.; Giansanti, S.; Ruiz-Dueñas, F.J.; Choinowski, T.; Piontek, K.; Martínez, A.T.; Lendzian, F.; Basosi, R. A tryptophan neutral radical in the oxidized state of versatile peroxidase from Pleurotus eryngii: A combined multifrequency EPR and density functional theory study. J. Biol. Chem. 2006, 281, 9517–9526. [Google Scholar] [CrossRef]
- Pogni, R.; Teutloff, C.; Lendzian, F.; Basosi, R. Tryptophan radicals as reaction intermediates in versatile peroxidases: Multifrequency EPR, ENDOR and density functional theory studies. Appl. Magn. Reson. 2007, 31, 509–526. [Google Scholar] [CrossRef]
- Brogioni, B.; Biglino, D.; Sinicropi, A.; Reijerse, E.J.; Giardina, P.; Sannia, G.; Lubitz, W.; Basosi, R.; Pogni, R. Characterization of radical intermediates in laccase-mediator systems. a multifrequency EPR, ENDOR and DFT/PCM investigation. Phys. Chem. Chem. Phys. 2008, 10, 7284–7292. [Google Scholar] [CrossRef]
- Al Khatib, M.; Costa, J.; Baratto, M.C.; Basosi, R.; Pogni, R. Paramagnetism and relaxation dynamics in melanin biomaterials. J. Phys. Chem. B 2020, 124, 2110–2115. [Google Scholar] [CrossRef] [PubMed]
- Panzella, L.; Gentile, G.; D’Errico, G.; Della Vecchia, N.F.; Errico, M.E.; Napolitano, A.; Carfagna, C.; D’Ischia, M. Atypical structural and π-electron features of a melanin polymer that lead to superior free-radical-scavenging properties. Angew. Chemie Int. Ed. 2013, 52, 12684–12687. [Google Scholar] [CrossRef] [PubMed]
- Watt, A.A.R.; Bothma, J.P.; Meredith, P. The supramolecular structure of melanin. Soft Matter 2009, 5, 3754–3760. [Google Scholar] [CrossRef]
- Pogni, R.; Baratto, M.C.; Sinicropi, A.; Basosi, R. Spectroscopic and computational characterization of laccases and their substrate radical intermediates. Cell. Mol. Life Sci. 2015, 72, 885–896. [Google Scholar] [CrossRef]
- Carmieli, R.; Tadyszak, K. Electron spin relaxation studies of polydopamine radicals’. J. Phys. Chem. B 2021, 125, 841–849. [Google Scholar]
- Grossmann, B.; Heinze, J.; Moll, T.; Palivan, C.; Ivan, S.; Gescheidt, G. Electron delocalization in one-electron oxidized aniline oligomers, paradigms for polyaniline. a study by paramagnetic resonance in fluid solution. J. Phys. Chem. B 2004, 108, 4669–4672. [Google Scholar] [CrossRef]
- Chen, Z.; Bertin, R.; Froldi, G. EC50 estimation of antioxidant activity in DPPH* assay using several statistical programs. Food Chem. 2013, 138, 414–420. [Google Scholar] [CrossRef]

| Sample | Carboxylic Acid | Aliphatic-OH | CondensedPhenolic Units | P-hydroxyphenyl Units |
|---|---|---|---|---|
| HGAm | 4.10 | 3.98 | 1.6 | 1.9 |
| GAm | 0.52 | 31.47 | 5.64 | 13.64 |
| T1s (μs) | T1f (μs) | Af/As | |
|---|---|---|---|
| HGAm | 254 | 59 | 1.51 |
| GAm | 170 | 44 | 1.13 |
| Dopa melanin | 216 | 61 | 1.50 |
| Sample | EPR | UV–vis |
|---|---|---|
| HGAm | 2.7 ± 0.7 | n.d. |
| GAm | 27.2 ± 5.4 | 25.7 ± 2.6 |
| Gallic acid polymer | 14.4 ± 3.3 | 12.7 ± 4.7 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Al Khatib, M.; Costa, J.; Spinelli, D.; Capecchi, E.; Saladino, R.; Baratto, M.C.; Pogni, R. Homogentisic Acid and Gentisic Acid Biosynthesized Pyomelanin Mimics: Structural Characterization and Antioxidant Activity. Int. J. Mol. Sci. 2021, 22, 1739. https://doi.org/10.3390/ijms22041739
Al Khatib M, Costa J, Spinelli D, Capecchi E, Saladino R, Baratto MC, Pogni R. Homogentisic Acid and Gentisic Acid Biosynthesized Pyomelanin Mimics: Structural Characterization and Antioxidant Activity. International Journal of Molecular Sciences. 2021; 22(4):1739. https://doi.org/10.3390/ijms22041739
Chicago/Turabian StyleAl Khatib, Maher, Jessica Costa, Daniele Spinelli, Eliana Capecchi, Raffaele Saladino, Maria Camilla Baratto, and Rebecca Pogni. 2021. "Homogentisic Acid and Gentisic Acid Biosynthesized Pyomelanin Mimics: Structural Characterization and Antioxidant Activity" International Journal of Molecular Sciences 22, no. 4: 1739. https://doi.org/10.3390/ijms22041739
APA StyleAl Khatib, M., Costa, J., Spinelli, D., Capecchi, E., Saladino, R., Baratto, M. C., & Pogni, R. (2021). Homogentisic Acid and Gentisic Acid Biosynthesized Pyomelanin Mimics: Structural Characterization and Antioxidant Activity. International Journal of Molecular Sciences, 22(4), 1739. https://doi.org/10.3390/ijms22041739










