Consecutive Low Doses of Streptozotocin Induce Polycystic Ovary Syndrome Features in Mice
Abstract
1. Introduction
2. Results
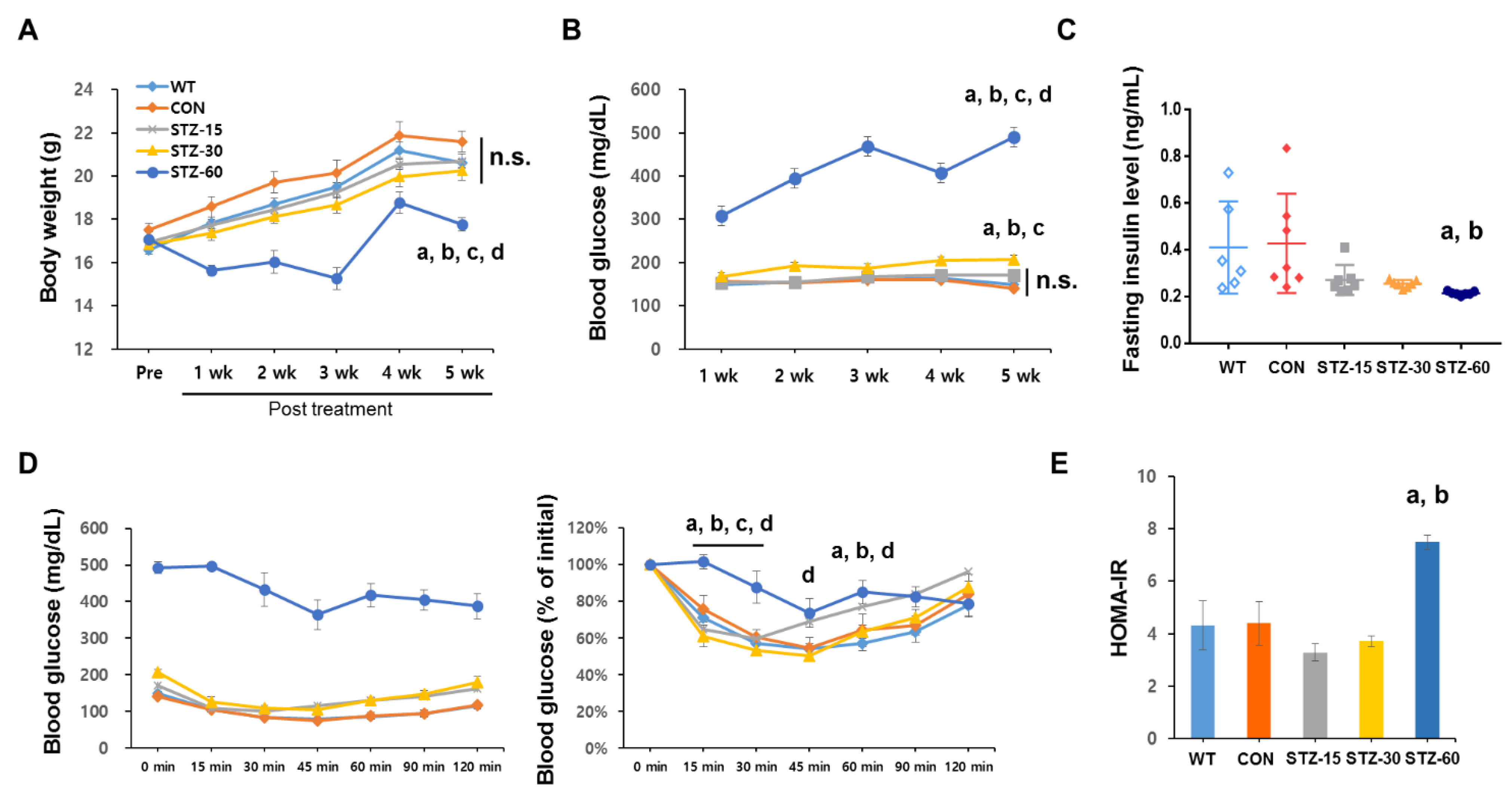
2.1. Multiple Low-Dose STZ Treatments Do Not Induce Typical Diabetic Profiles in Mice
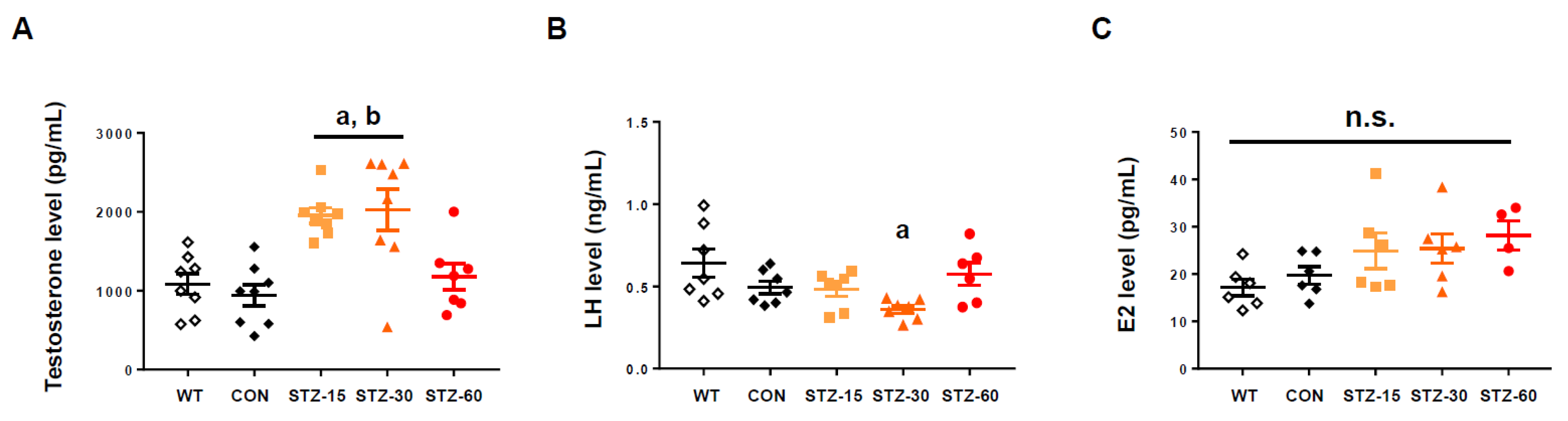
2.2. Low Doses of STZ Induces Hyperandrogenism but Do Not Increase LH or E2 Levels
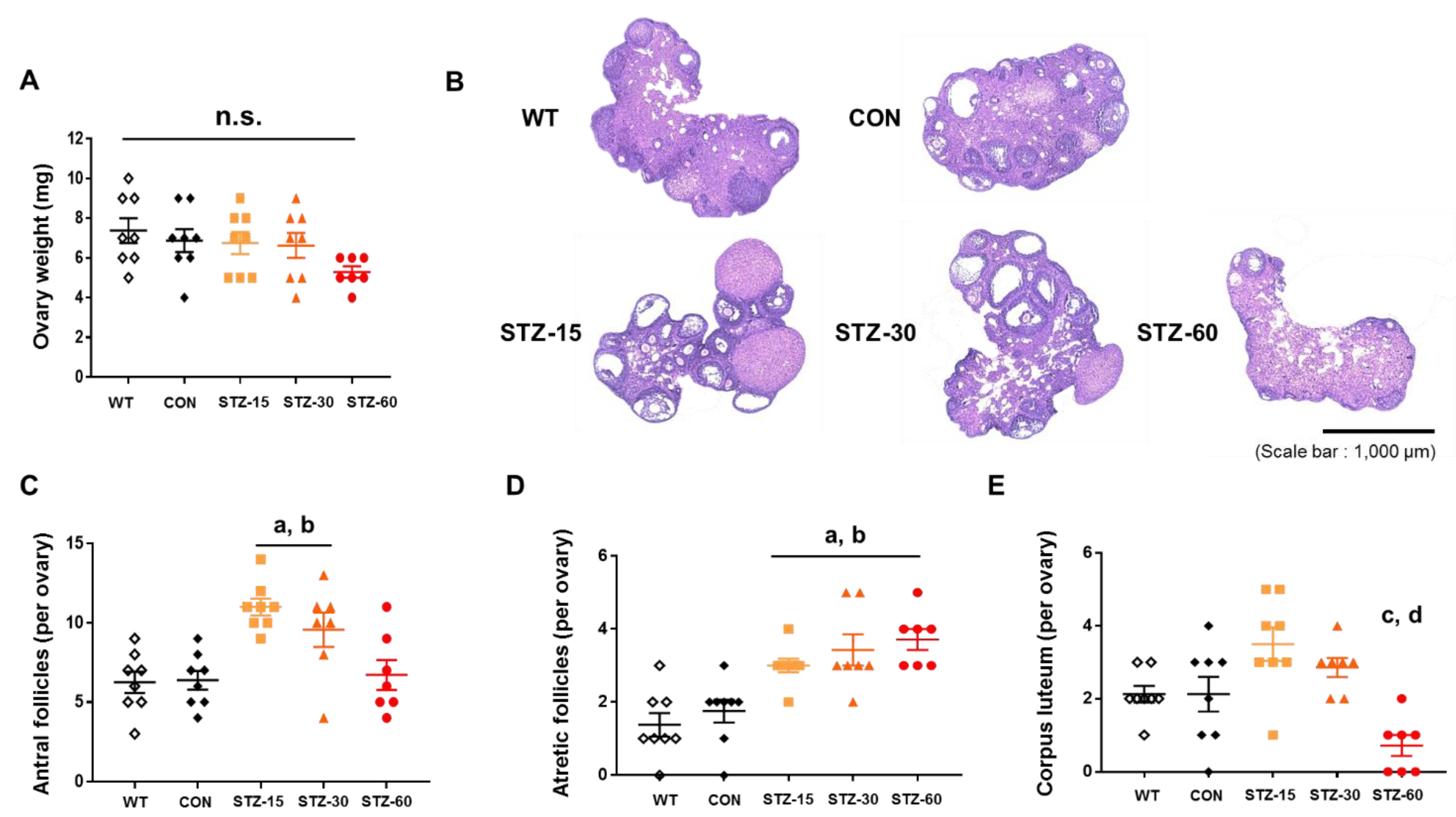
2.3. Polycystic Ovaries in Mice Treated with Low Doses of STZ
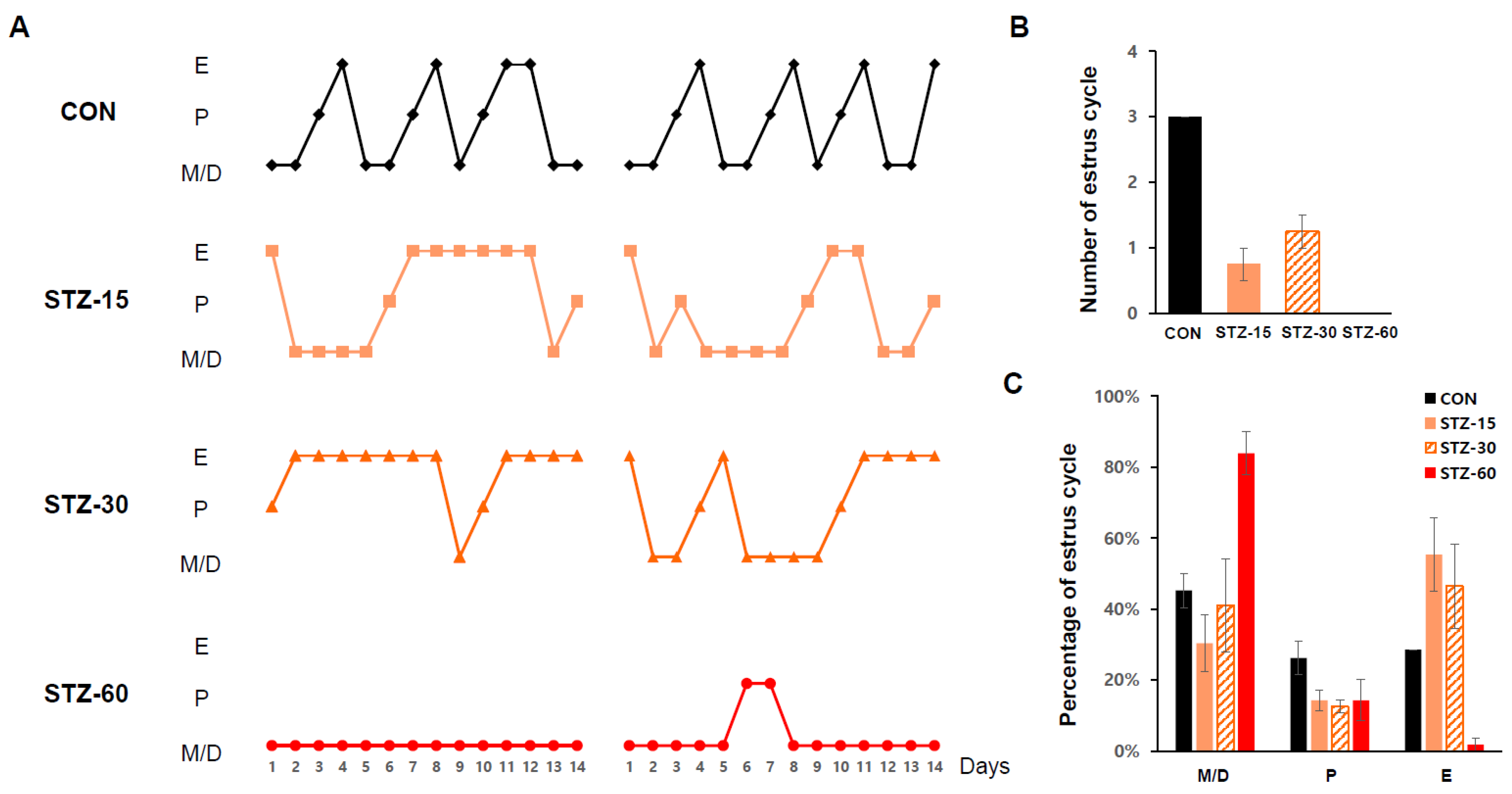
2.4. STZ Treatment Disrupts Mice Estrous Cycle
2.5. Systemic STZ Treatment Enhances Intraovarian PI3K-AKT and IRS-1signals
3. Discussion
4. Materials and Methods
4.1. Animals
4.2. Chemicals and Antibodies
4.3. Streptozotocin Treatment
4.4. Bodyweight, Blood Glucose Level, Insulin Tolerance Tests, and Homeostasis Model Assessment of Insulin Resistance Index
4.5. Estrous Cycle Analysis
4.6. Hormonal Assays
4.7. Histological Analysis
4.8. Immunoblotting
4.9. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Azziz, R.; Carmina, E.; Chen, Z.; Dunaif, A.; Laven, J.S.; Legro, R.S.; Lizneva, D.; Natterson-Horowtiz, B.; Teede, H.J.; Yildiz, B.O. Polycystic ovary syndrome. Nat. Rev. Dis. Primers 2016, 2, 16057. [Google Scholar] [CrossRef]
- The Rotterdam ESHRE/ASRM-Sponsored PCOS Consensus Workshop Group. Revised 2003 consensus on diagnostic criteria and long-term health risks related to polycystic ovary syndrome (PCOS). Hum. Reprod. 2004, 19, 41–47. [Google Scholar] [CrossRef]
- Ehrmann, D.A. Polycystic ovary syndrome. N. Engl. J. Med. 2005, 352, 1223–1236. [Google Scholar] [CrossRef] [PubMed]
- The Amsterdam ESHRE/ASRM-Sponsored 3rd PCOS Consensus Workshop Group. Consensus on women’s health aspects of polycystic ovary syndrome (PCOS). Hum. Reprod. 2012, 27, 14–24. [Google Scholar] [CrossRef] [PubMed]
- Chae, S.J.; Kim, J.J.; Choi, Y.M.; Hwang, K.R.; Jee, B.C.; Ku, S.Y.; Suh, C.S.; Kim, S.H.; Kim, J.G.; Moon, S.Y. Clinical and biochemical characteristics of polycystic ovary syndrome in Korean women. Hum. Reprod. 2008, 23, 1924–1931. [Google Scholar] [CrossRef] [PubMed]
- Guo, M.; Chen, Z.J.; Macklon, N.S.; Shi, Y.H.; Westerveld, H.E.; Eijkemans, M.J.; Fauser, B.C.; Goverde, A.J. Cardiovascular and metabolic characteristics of infertile Chinese women with PCOS diagnosed according to the Rotterdam consensus criteria. Reprod. BioMed. Online 2010, 21, 572–580. [Google Scholar] [CrossRef] [PubMed]
- Kubota, T. Update in polycystic ovary syndrome: New criteria of diagnosis and treatment in Japan. Reprod. Med. Biol. 2013, 12, 71–77. [Google Scholar] [CrossRef] [PubMed]
- McNeilly, A.S.; Duncan, W.C. Rodent models of polycystic ovary syndrome. Mol. Cell. Endocrinol. 2013, 373, 2–7. [Google Scholar] [CrossRef]
- Paixao, L.; Ramos, R.B.; Lavarda, A.; Morsh, D.M.; Spritzer, P.M. Animal models of hyperandrogenism and ovarian morphology changes as features of polycystic ovary syndrome: A systematic review. Reprod. Biol. Endocrinol. 2017, 15, 12. [Google Scholar] [CrossRef]
- Shi, D.; Vine, D.F. Animal models of polycystic ovary syndrome: A focused review of rodent models in relationship to clinical phenotypes and cardiometabolic risk. Fertil. Steril. 2012, 98, 185–193. [Google Scholar] [CrossRef]
- Walters, K.A.; Allan, C.M.; Handelsman, D.J. Rodent models for human polycystic ovary syndrome. Biol. Reprod. 2012, 86, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Ryu, Y.; Kim, S.W.; Kim, Y.Y.; Ku, S.Y. Animal models for human polycystic ovary syndrome (PCOS) focused on the use of indirect hormonal perturbations: A review of the literature. Int. J. Mol. Sci. 2019, 20, 2720. [Google Scholar] [CrossRef]
- Brosky, G.; Logothetopoulos, J. Streptozotocin diabetes in the mouse and guinea pig. Diabetes 1969, 18, 606–611. [Google Scholar] [CrossRef] [PubMed]
- Kazumi, T.; Yoshino, G.; Fujii, S.; Baba, S. Tumorigenic action of streptozotocin on the pancreas and kidney in male Wistar rats. Cancer Res. 1978, 38, 2144–2147. [Google Scholar] [PubMed]
- Sharma, M.; Gupta, Y.K. Intracerebroventricular injection of streptozotocin in rats produces both oxidative stress in the brain and cognitive impairment. Life Sci. 2001, 68, 1021–1029. [Google Scholar] [CrossRef]
- Bolzan, A.D.; Bianchi, M.S. Genotoxicity of streptozotocin. Mutat. Res. 2002, 512, 121–134. [Google Scholar] [CrossRef]
- Leaming, A.B.; Mathur, R.S.; Levine, J.H. Increased plasma testosterone in streptozotocin-diabetic female rats. Endocrinology 1982, 111, 1329–1333. [Google Scholar] [CrossRef]
- Valdes, C.T.; Elkind-Hirsch, K.E.; Rogers, D.G.; Adelman, J.P. The hypothalamic-pituitary axis of streptozotocin-induced diabetic female rats is not normalized by estradiol replacement. Endocrinology 1991, 128, 433–440. [Google Scholar] [CrossRef]
- Tesone, M.; Ladenheim, R.G.; Oliveira-Filho, R.M.; Chiauzzi, V.A.; Foglia, V.G.; Charreau, E.H. Ovarian dysfunction in streptozotocin-induced diabetic rats. Proc. Soc. Exp. Biol. Med. 1983, 174, 123–130. [Google Scholar] [CrossRef]
- Diamanti-Kandarakis, E.; Papavassiliou, A.G. Molecular mechanisms of insulin resistance in polycystic ovary syndrome. Trends Mol. Med. 2006, 12, 324–332. [Google Scholar] [CrossRef]
- Makker, A.; Goel, M.M.; Das, V.; Agarwal, A. PI3K-Akt-mTOR and MAPK signaling pathways in polycystic ovarian syndrome, uterine leiomyomas and endometriosis: An update. Gynecol. Endocrinol. 2012, 28, 175–181. [Google Scholar] [CrossRef] [PubMed]
- Nekoonam, S.; Naji, M.; Nashtaei, M.S.; Mortezaee, K.; Koruji, M.; Safdarian, L.; Amidi, F. Expression of AKT1 along with AKT2 in granulosa-lutein cells of hyperandrogenic PCOS patients. Arch. Gynecol. Obstet. 2017, 295, 1041–1050. [Google Scholar] [CrossRef]
- Lan, Z.J.; Krause, M.S.; Redding, S.D.; Li, X.; Wu, G.Z.; Zhou, H.X.; Bohler, H.C.; Ko, C.; Cooney, A.J.; Zhou, J.; et al. Selective deletion of Pten in theca-interstitial cells leads to androgen excess and ovarian dysfunction in mice. Mol. Cell. Endocrinol. 2017, 444, 26–37. [Google Scholar] [CrossRef] [PubMed]
- Nteeba, J.; Ross, J.W.; Perfield, J.W., 2nd; Keating, A.F. High fat diet induced obesity alters ovarian phosphatidylinositol-3 kinase signaling gene expression. Reprod. Toxicol. 2013, 42, 68–77. [Google Scholar] [CrossRef] [PubMed]
- Li, T.; Mo, H.; Chen, W.; Li, L.; Xiao, Y.; Zhang, J.; Li, X.; Lu, Y. Role of the PI3K-Akt signaling pathway in the pathogenesis of polycystic ovary syndrome. Reprod. Sci. 2017, 24, 646–655. [Google Scholar] [CrossRef]
- Munir, I.; Yen, H.W.; Geller, D.H.; Torbati, D.; Bierden, R.M.; Weitsman, S.R.; Agarwal, S.K.; Magoffin, D.A. Insulin augmentation of 17α-hydroxylase activity is mediated by phosphatidyl inositol 3-kinase but not extracellular signal-regulated kinase-1/2 in human ovarian theca cells. Endocrinology 2004, 145, 175–183. [Google Scholar] [CrossRef]
- Liu, K.; Rajareddy, S.; Liu, L.; Jagarlamudi, K.; Boman, K.; Selstam, G.; Reddy, P. Control of mammalian oocyte growth and early follicular development by the oocyte PI3 kinase pathway: New roles for an old timer. Dev. Biol. 2006, 299, 1–11. [Google Scholar] [CrossRef]
- Magoffin, D.A. Ovarian theca cell. Int. J. Biochem. Cell Biol. 2005, 37, 1344–1349. [Google Scholar] [CrossRef]
- Byers, S.L.; Wiles, M.V.; Dunn, S.L.; Taft, R.A. Mouse estrous cycle identification tool and images. PLoS ONE 2012, 7, e35538. [Google Scholar] [CrossRef]
- Anderson, E.; Lee, G.Y.; O’Brien, K. Polycystic ovarian condition in the dehydroepiandrosterone-treated rat model: Hyperandrogenism and the resumption of meiosis are major initial events associated with cystogenesis of antral follicles. Anat. Rec. 1997, 249, 44–53. [Google Scholar] [CrossRef]
- Van Houten, E.L.; Visser, J.A. Mouse models to study polycystic ovary syndrome: A possible link between metabolism and ovarian function? Reprod. Biol. 2014, 14, 32–43. [Google Scholar] [CrossRef] [PubMed]
- Kafali, H.; Iriadam, M.; Ozardali, I.; Demir, N. Letrozole-induced polycystic ovaries in the rat: A new model for cystic ovarian disease. Arch. Med. Res. 2004, 35, 103–108. [Google Scholar] [CrossRef] [PubMed]
- Kauffman, A.S.; Thackray, V.G.; Ryan, G.E.; Tolson, K.P.; Glidewell-Kenney, C.A.; Semaan, S.J.; Poling, M.C.; Iwata, N.; Breen, K.M.; Duleba, A.J.; et al. A novel letrozole model recapitulates both the reproductive and metabolic phenotypes of polycystic ovary syndrome in female mice. Biol. Reprod. 2015, 93, 69. [Google Scholar] [CrossRef] [PubMed]
- Van Weerden, W.M.; Bierings, H.G.; van Steenbrugge, G.J.; de Jong, F.H.; Schroder, F.H. Adrenal glands of mouse and rat do not synthesize androgens. Life Sci. 1992, 50, 857–861. [Google Scholar] [CrossRef]
- Bergh, C.; Carlsson, B.; Olsson, J.H.; Selleskog, U.; Hillensjo, T. Regulation of androgen production in cultured human thecal cells by insulin-like growth factor I and insulin. Fertil. Steril. 1993, 59, 323–331. [Google Scholar] [CrossRef]
- Cai, F.; Helke, C.J. Abnormal PI3 kinase/Akt signal pathway in vagal afferent neurons and vagus nerve of streptozotocin-diabetic rats. Mol. Brain Res. 2003, 110, 234–244. [Google Scholar] [CrossRef]
- Hulmi, J.J.; Silvennoinen, M.; Lehti, M.; Kivela, R.; Kainulainen, H. Altered REDD1, myostatin, and Akt/mTOR/FoxO/MAPK signaling in streptozotocin-induced diabetic muscle atrophy. Am. J. Physiol. Endocrinol. Metab. 2012, 302, E307–E315. [Google Scholar] [CrossRef]
- Yeon Lee, J.; Baw, C.-K.; Gupta, S.; Aziz, N.; Agarwal, A. Role of oxidative stress in polycystic ovary syndrome. Curr. Women’s Health Rev. 2010, 6, 96–107. [Google Scholar] [CrossRef]
- Fushimi, H.; Horie, H.; Inoue, T.; Kameyama, M.; Kanao, K.; Ishihara, S.; Tsujimura, T.; Nunotani, H.; Minami, T.; Okazaki, Y.; et al. Low testosterone levels in diabetic men and animals: A possible role in testicular impotence. Diabetes Res. Clin. Pract. 1989, 6, 297–301. [Google Scholar] [CrossRef]
- Kuhn-Velten, N.; Waldenburger, D.; Staib, W. Evaluation of steroid biosynthetic lesions in isolated Leydig cells from the testes of streptozotocin-diabetic rats. Diabetologia 1982, 23, 529–533. [Google Scholar] [CrossRef]
- Murri, M.; Luque-Ramirez, M.; Insenser, M.; Ojeda-Ojeda, M.; Escobar-Morreale, H.F. Circulating markers of oxidative stress and polycystic ovary syndrome (PCOS): A systematic review and meta-analysis. Hum. Reprod. Update 2013, 19, 268–288. [Google Scholar] [CrossRef] [PubMed]
- Verit, F.F.; Erel, O. Oxidative stress in nonobese women with polycystic ovary syndrome: Correlations with endocrine and screening parameters. Gynecol. Obstet. Investig. 2008, 65, 233–239. [Google Scholar] [CrossRef] [PubMed]
- Homburg, R. Androgen circle of polycystic ovary syndrome. Hum. Reprod. 2009, 24, 1548–1555. [Google Scholar] [CrossRef] [PubMed]
- Homburg, R.; Crawford, G. The role of AMH in anovulation associated with PCOS: A hypothesis. Hum. Reprod. 2014, 29, 1117–1121. [Google Scholar] [CrossRef]
- Hart, R.; Doherty, D.A.; Norman, R.J.; Franks, S.; Dickinson, J.E.; Hickey, M.; Sloboda, D.M. Serum antimullerian hormone (AMH) levels are elevated in adolescent girls with polycystic ovaries and the polycystic ovarian syndrome (PCOS). Fertil. Steril. 2010, 94, 1118–1121. [Google Scholar] [CrossRef]
- Tata, B.; Mimouni, N.E.H.; Barbotin, A.L.; Malone, S.A.; Loyens, A.; Pigny, P.; Dewailly, D.; Catteau-Jonard, S.; Sundstrom-Poromaa, I.; Piltonen, T.T.; et al. Elevated prenatal anti-Mullerian hormone reprograms the fetus and induces polycystic ovary syndrome in adulthood. Nat. Med. 2018, 24, 834–846. [Google Scholar] [CrossRef]
- Caldwell, A.S.L.; Edwards, M.C.; Desai, R.; Jimenez, M.; Gilchrist, R.B.; Handelsman, D.J.; Walters, K.A. Neuroendocrine androgen action is a key extraovarian mediator in the development of polycystic ovary syndrome. Proc. Natl. Acad. Sci. USA 2017, 114, E3334–E3343. [Google Scholar] [CrossRef]
- Shen, M.; Lin, F.; Zhang, J.; Tang, Y.; Chen, W.K.; Liu, H. Involvement of the up-regulated FoxO1 expression in follicular granulosa cell apoptosis induced by oxidative stress. J. Biol. Chem. 2012, 287, 25727–25740. [Google Scholar] [CrossRef]
- Shi, F.; LaPolt, P.S. Relationship between FoxO1 protein levels and follicular development, atresia, and luteinization in the rat ovary. J. Endocrinol. 2003, 179, 195–203. [Google Scholar] [CrossRef]
- Kajihara, T.; Uchino, S.; Suzuki, M.; Itakura, A.; Brosens, J.J.; Ishihara, O. Increased ovarian follicle atresia in obese Zucker rats is associated with enhanced expression of the forkhead transcription factor FOXO1. Med. Mol. Morphol. 2009, 42, 216–221. [Google Scholar] [CrossRef]
- Vinue, A.; Gonzalez-Navarro, H. Glucose and insulin tolerance tests in the mouse. Methods Mol. Biol. 2015, 1339, 247–254. [Google Scholar] [CrossRef] [PubMed]
- Pentinat, T.; Ramon-Krauel, M.; Cebria, J.; Diaz, R.; Jimenez-Chillaron, J.C. Transgenerational inheritance of glucose intolerance in a mouse model of neonatal overnutrition. Endocrinology 2010, 151, 5617–5623. [Google Scholar] [CrossRef] [PubMed]
- McLean, A.C.; Valenzuela, N.; Fai, S.; Bennett, S.A. Performing vaginal lavage, crystal violet staining, and vaginal cytological evaluation for mouse estrous cycle staging identification. J. Vis. Exp. 2012, e4389. [Google Scholar] [CrossRef] [PubMed]
- Yaba, A.; Demir, N. The mechanism of mTOR (mammalian target of rapamycin) in a mouse model of polycystic ovary syndrome (PCOS). J. Ovarian Res. 2012, 5, 38. [Google Scholar] [CrossRef]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ryu, Y.; Kim, Y.J.; Kim, Y.Y.; Kim, J.; Kim, S.W.; Kim, H.; Ku, S.Y. Consecutive Low Doses of Streptozotocin Induce Polycystic Ovary Syndrome Features in Mice. Int. J. Mol. Sci. 2021, 22, 1299. https://doi.org/10.3390/ijms22031299
Ryu Y, Kim YJ, Kim YY, Kim J, Kim SW, Kim H, Ku SY. Consecutive Low Doses of Streptozotocin Induce Polycystic Ovary Syndrome Features in Mice. International Journal of Molecular Sciences. 2021; 22(3):1299. https://doi.org/10.3390/ijms22031299
Chicago/Turabian StyleRyu, Youngjae, Yong Jin Kim, Yoon Young Kim, Jungwoo Kim, Sung Woo Kim, Hoon Kim, and Seung Yup Ku. 2021. "Consecutive Low Doses of Streptozotocin Induce Polycystic Ovary Syndrome Features in Mice" International Journal of Molecular Sciences 22, no. 3: 1299. https://doi.org/10.3390/ijms22031299
APA StyleRyu, Y., Kim, Y. J., Kim, Y. Y., Kim, J., Kim, S. W., Kim, H., & Ku, S. Y. (2021). Consecutive Low Doses of Streptozotocin Induce Polycystic Ovary Syndrome Features in Mice. International Journal of Molecular Sciences, 22(3), 1299. https://doi.org/10.3390/ijms22031299





