Roles of the C-Terminal Amino Acids of Non-Hexameric Helicases: Insights from Escherichia coli UvrD
Abstract
1. Introduction
2. Helicase Superfamily
2.1. Superfamily 1 Helicases
2.2. Superfamily 2 Helicases
2.3. Proposed Models for the Functional Unit of Some SF1 and SF2 Helicases
2.4. Dimeric-Helicase and Monomeric-Helicase Models for SF1 and SF2 Helicases Other than E. coli SF1 UvrD Helicase
2.4.1. PcrA
2.4.2. RecQ
3. C-Terminal Amino Acids
4. C-Terminus Truncated UvrD
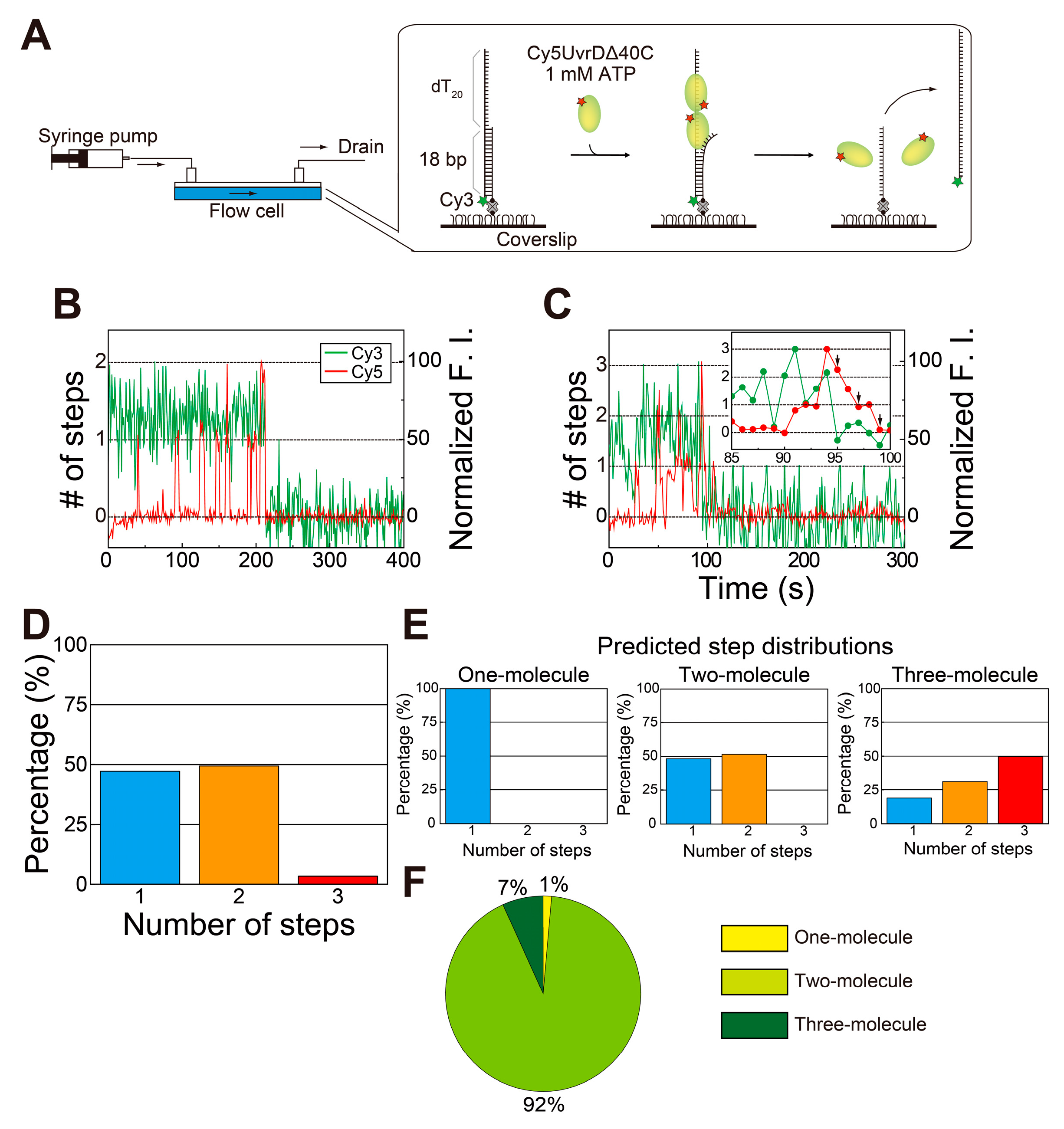
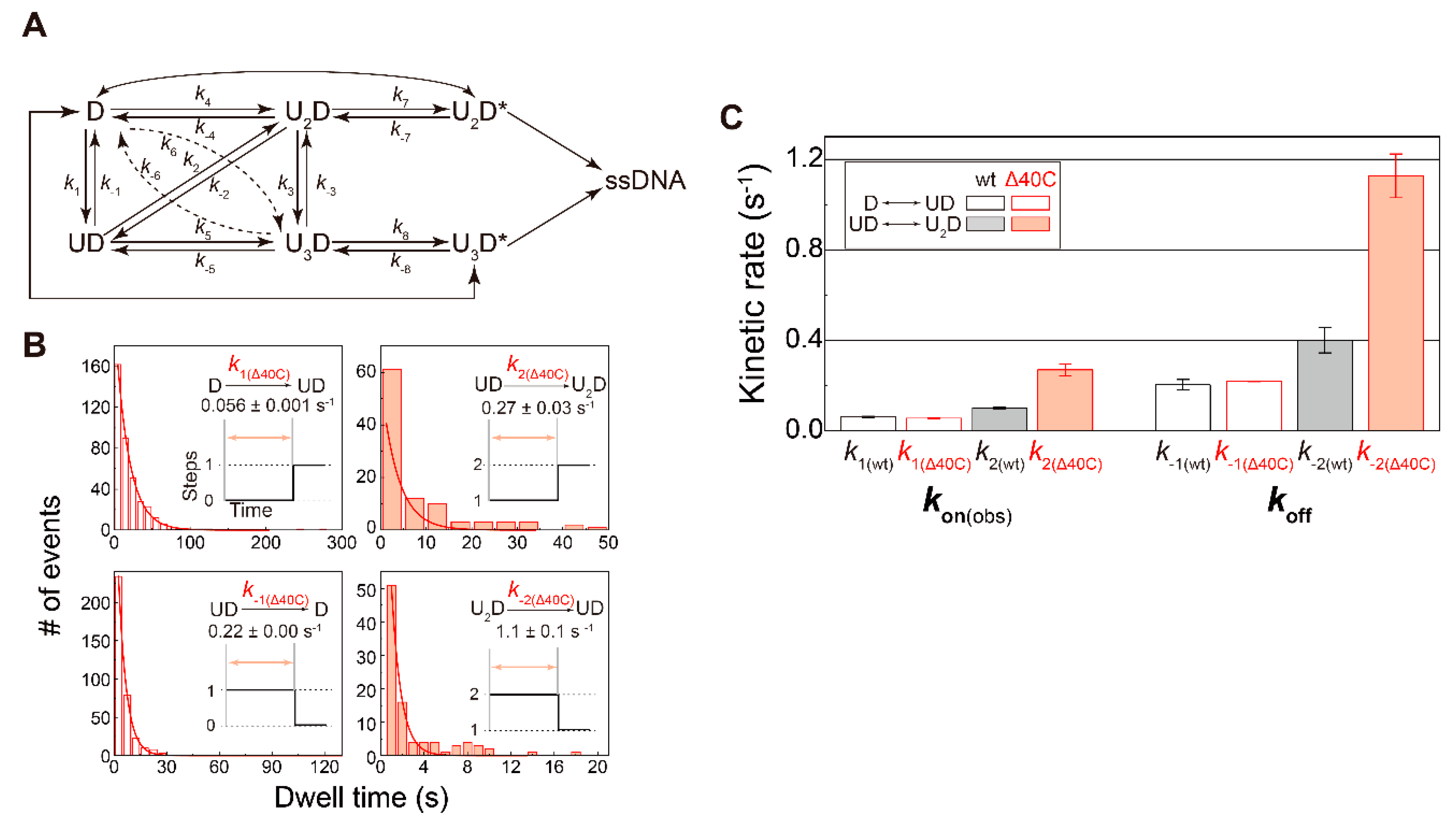
5. Single-Molecule Direct Visualization of UvrDΔ40C
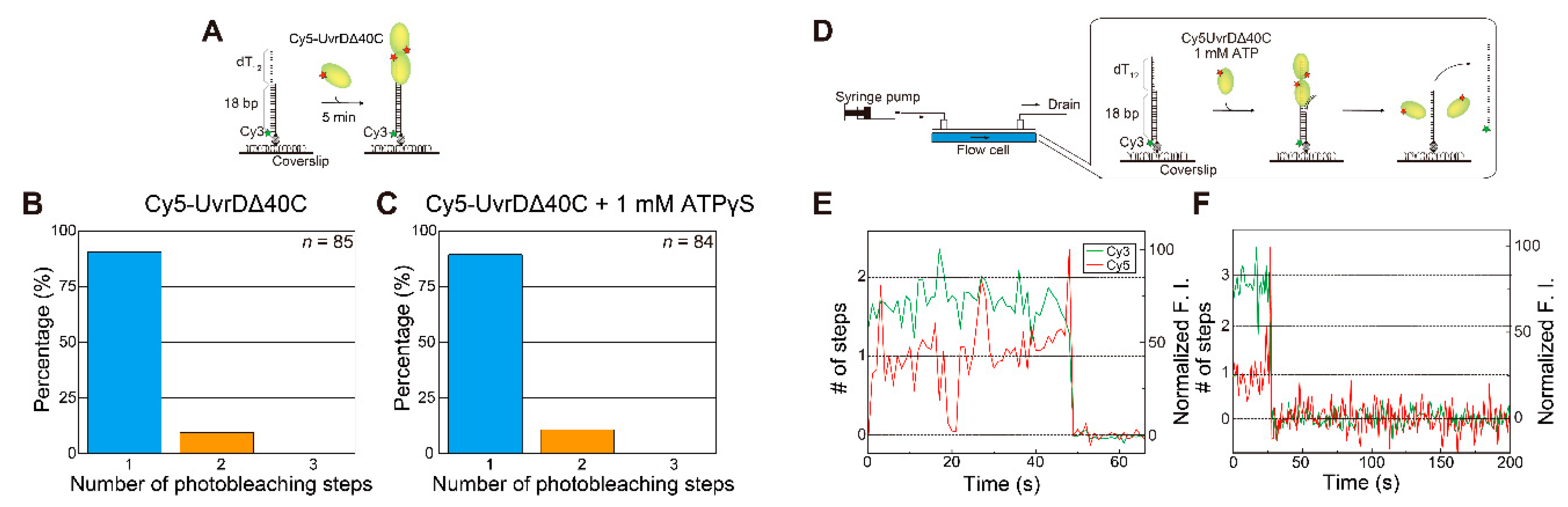
5.1. Observation of Multiple UvrDΔ40C Molecules that Bound to DNA in the Absence of ATP
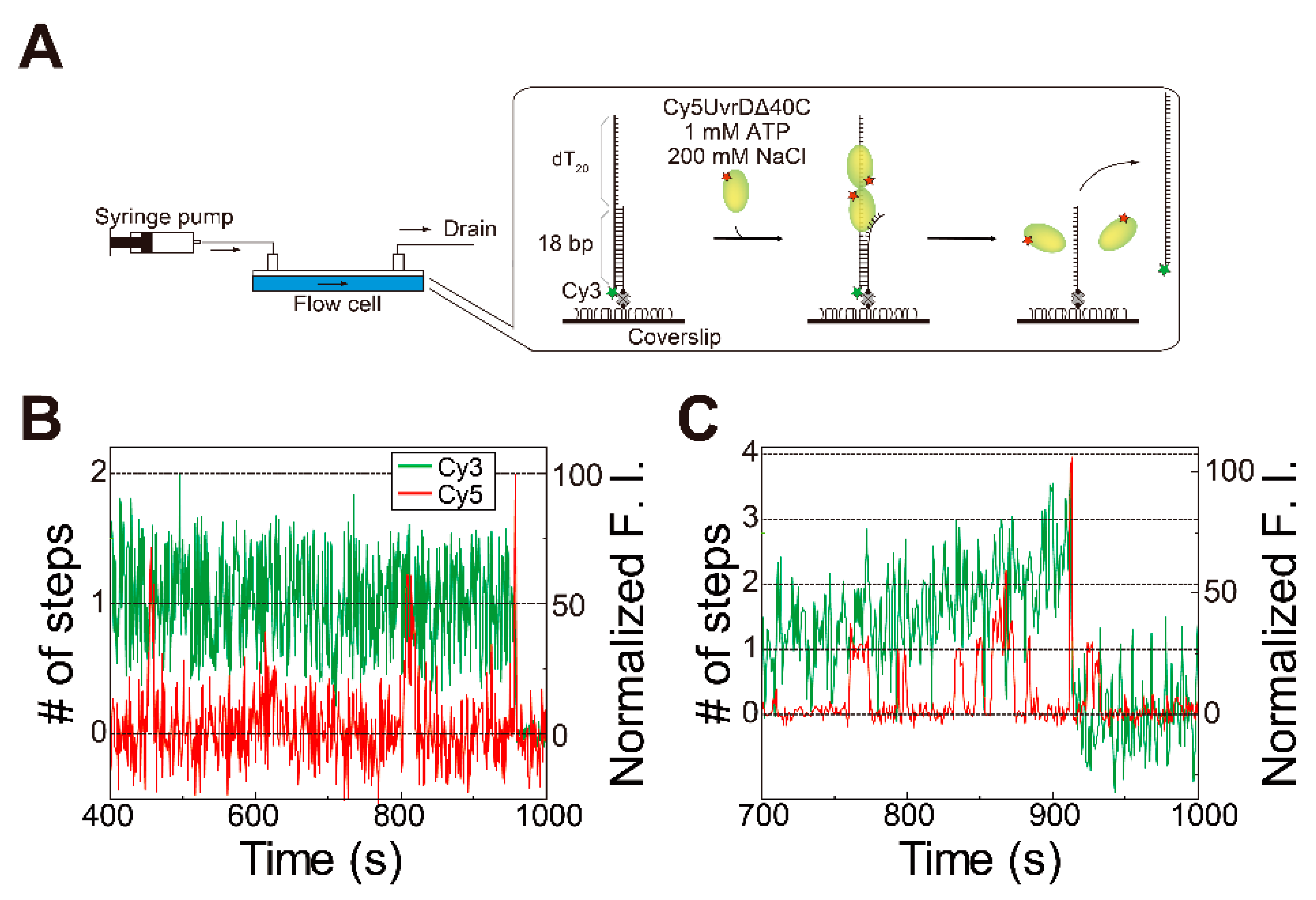
5.2. Observation of Multiple UvrDΔ40C Molecules’ Association to DNA, which Synchronized with the Unwinding Activity in the Presence of ATP
5.3. Multiple UvrDΔ40C Molecules that Bound to the DNA Are Likely to Form an Oligomer
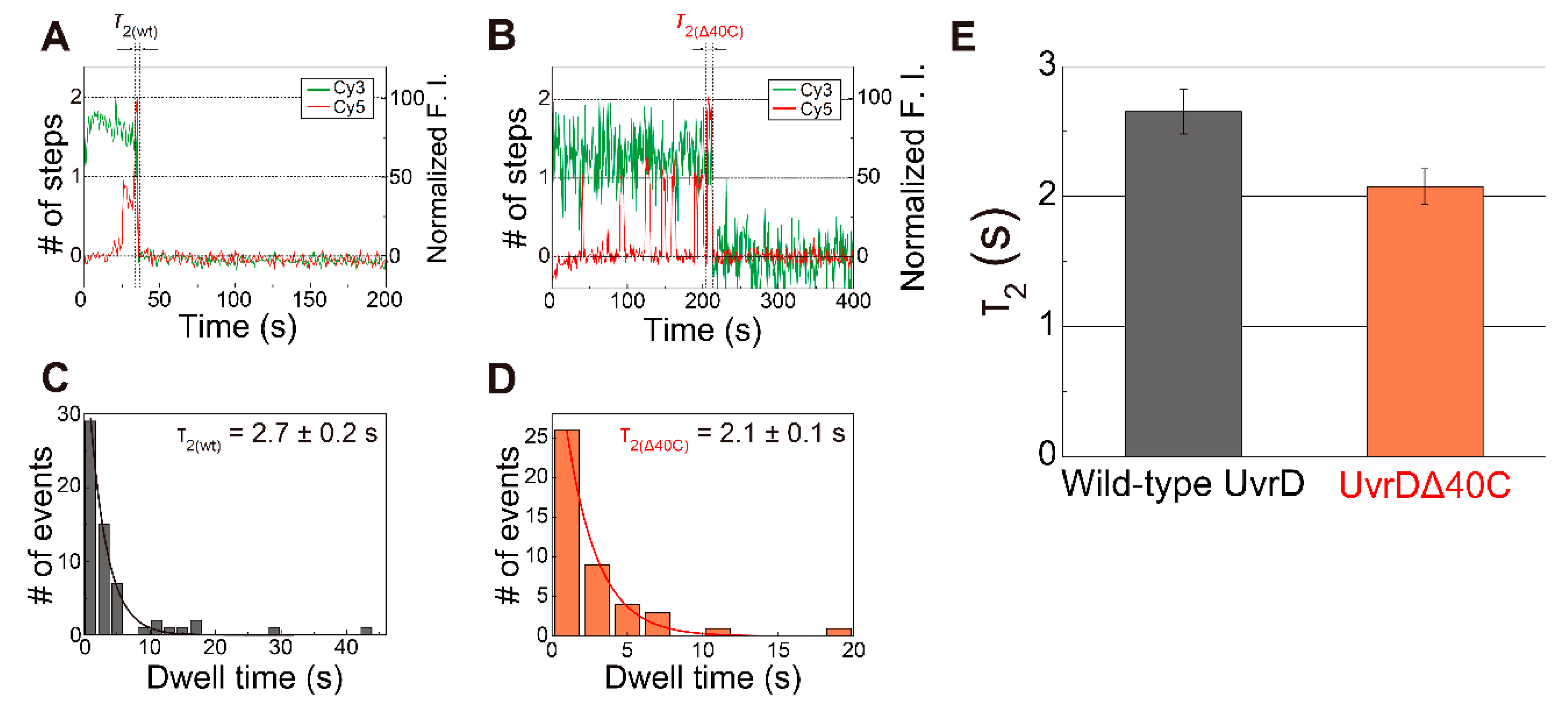
5.4. Transient Two UvrD Bound State Just before Completion of the DNA-Unwinding Process
5.5. Association/Dissociation Rates for the UvrDΔ40C–DNA Interaction
6. Summary
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ATP | adenosine triphosphate |
| ATPγS | adenosine 5′-(γ-thio)triphosphate |
| B. stearothermophilus | Bacillus stearothermophilus |
| bp | base pair |
| dsDNA | double-stranded DNA |
| E. coli | Escherichia coli |
| F.I. | fluorescence intensities |
| FCCS | fluorescence cross-correlation spectroscopy |
| FRET | fluorescence resonance energy transfer |
| G. stearothermophilus | Geobacillus stearothermophilus |
| N.D. | not determined |
| nt | nucleotide |
| SF | superfamily |
| ssDNA | single-stranded DNA |
| T. thermophilus | Thermus thermophilus |
| UvrDΔ102C | UvrD mutant lacking the C-terminal 102 amino acids |
| UvrDΔ107C | UvrD mutant lacking the C-terminal 107 amino acids |
| UvrDΔ40C | UvrD mutant lacking the C-terminal 40 amino acids |
| UvrDΔ73C | UvrD mutant lacking the C-terminal 73 amino acids |
References
- Singleton, M.R.; Dillingham, M.S.; Wigley, D.B. Structure and Mechanism of Helicases and Nucleic Acid Translocases. Annu. Rev. Biochem. 2007, 76, 23–50. [Google Scholar] [CrossRef] [PubMed]
- Fairman-Williams, M.E.; Guenther, U.-P.; Jankowsky, E. SF1 and SF2 helicases: Family matters. Curr. Opin. Struct. Biol. 2010, 20, 313–324. [Google Scholar] [CrossRef] [PubMed]
- Raney, K.D.; Byrd, A.K.; Aarattuthodiyil, S. Structure and Mechanisms of SF1 DNA Helicases. Neurotransm. Interact. Cogn. Funct. 2012, 767, 17–46. [Google Scholar] [CrossRef]
- Singleton, M.R.; Sawaya, M.R.; Ellenberger, T.; Wigley, D.B. Crystal Structure of T7 Gene 4 Ring Helicase Indicates a Mechanism for Sequential Hydrolysis of Nucleotides. Cell 2000, 101, 589–600. [Google Scholar] [CrossRef]
- Enemark, E.J.; Joshua-Tor, L. On helicases and other motor proteins. Curr. Opin. Struct. Biol. 2008, 18, 243–257. [Google Scholar] [CrossRef]
- Lee, J.Y.; Yang, W. UvrD Helicase Unwinds DNA One Base Pair at a Time by a Two-Part Power Stroke. Cell 2006, 127, 1349–1360. [Google Scholar] [CrossRef] [PubMed]
- Jia, H.; Korolev, S.; Niedziela-Majka, A.; Maluf, N.K.; Gauss, G.H.; Myong, S.; Ha, T.; Waksman, G.; Lohman, T.M. Rotations of the 2B Sub-domain of E. coli UvrD Helicase/Translocase Coupled to Nucleotide and DNA Binding. J. Mol. Biol. 2011, 411, 633–648. [Google Scholar] [CrossRef]
- Korolev, S.; Hsieh, J.; Gauss, G.H.; Lohman, T.M.; Waksman, G. Major Domain Swiveling Revealed by the Crystal Structures of Complexes of E. coli Rep Helicase Bound to Single-Stranded DNA and ADP. Cell 1997, 90, 635–647. [Google Scholar] [CrossRef]
- Velankar, S.S.; Soultanas, P.; Dillingham, M.S.; Subramanya, H.S.; Wigley, D.B. Crystal Structures of Complexes of PcrA DNA Helicase with a DNA Substrate Indicate an Inchworm Mechanism. Cell 1999, 97, 75–84. [Google Scholar] [CrossRef]
- Yokota, H. DNA-Unwinding Dynamics of Escherichia coli UvrD Lacking the C-Terminal 40 Amino Acids. Biophys. J. 2020, 118, 1634–1648. [Google Scholar] [CrossRef]
- Gorbalenya, A.E.; Koonin, E.V. Helicases: Amino acid sequence comparisons and structure-function relationships. Curr. Opin. Struct. Biol. 1993, 3, 419–429. [Google Scholar] [CrossRef]
- Gilhooly, N.S.; Gwynn, E.J.; Dillingham, M.S. Superfamily 1 helicases. Front. Biosci. 2013, 5, 206–216. [Google Scholar] [CrossRef] [PubMed]
- Pettersen, E.F.; Goddard, T.D.; Huang, C.C.; Couch, G.S.; Greenblatt, D.M.; Meng, E.C.; Ferrin, T.E. UCSF Chimera—A visualization system for exploratory research and analysis. J. Comput. Chem. 2004, 25, 1605–1612. [Google Scholar] [CrossRef] [PubMed]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; Mcgettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef]
- Robert, X.; Gouet, P. Deciphering key features in protein structures with the new ENDscript server. Nucleic Acids Res. 2014, 42, W320–W324. [Google Scholar] [CrossRef]
- Maluf, N.K.; Fischer, C.J.; Lohman, T.M. A Dimer of Escherichia coli UvrD is the Active Form of the Helicase In Vitro. J. Mol. Biol. 2003, 325, 913–935. [Google Scholar] [CrossRef]
- Sun, B.; Wei, K.-J.; Zhang, B.; Zhang, X.-H.; Dou, S.-X.; Ming, L.; Xi, X.G. Impediment of E. coli UvrD by DNA-destabilizing force reveals a strained-inchworm mechanism of DNA unwinding. EMBO J. 2008, 27, 3279–3287. [Google Scholar] [CrossRef]
- Yokota, H.; Chujo, Y.A.; Harada, Y. Single-Molecule Imaging of the Oligomer Formation of the Nonhexameric Escherichia coli UvrD Helicase. Biophys. J. 2013, 104, 924–933. [Google Scholar] [CrossRef]
- Lee, K.S.; Balci, H.; Jia, H.; Lohman, T.M.; Ha, T. Direct imaging of single UvrD helicase dynamics on long single-stranded DNA. Nat. Commun. 2013, 4, 1878. [Google Scholar] [CrossRef]
- Comstock, M.J.; Whitley, K.D.; Jia, H.; Sokoloski, J.; Lohman, T.M.; Ha, T.; Chemla, Y.R. Direct observation of structure-function relationship in a nucleic acid-processing enzyme. Science 2015, 348, 352–354. [Google Scholar] [CrossRef]
- Nguyen, B.; Ordabayev, Y.; Sokoloski, J.E.; Weiland, E.; Lohman, T.M. Large domain movements upon UvrD dimerization and helicase activation. Proc. Natl. Acad. Sci. USA 2017, 114, 12178–12183. [Google Scholar] [CrossRef] [PubMed]
- Wong, I.; Lohman, T.M. Allosteric Effects of Nucleotide Cofactors on Escherichia coli Rep Helicase & DNA Binding. Science 1992, 256, 350–355. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Hsieh, J.; Brendza, K.M.; Lohman, T.M. E. coli Rep oligomers are required to initiate DNA unwinding in vitro. J. Mol. Biol. 2001, 310, 327–350. [Google Scholar] [CrossRef] [PubMed]
- Ilangovan, A.; Kay, C.W.M.; Roier, S.; El Mkami, H.; Salvadori, E.; Zechner, E.L.; Zanetti, G.; Waksman, G. Cryo-EM Structure of a Relaxase Reveals the Molecular Basis of DNA Unwinding during Bacterial Conjugation. Cell 2017, 169, 708–721. [Google Scholar] [CrossRef] [PubMed]
- Yang, Y.; Dou, S.-X.; Ren, H.; Wang, P.-Y.; Zhang, X.-D.; Qian, M.; Pan, B.-Y.; Xi, X.G. Evidence for a functional dimeric form of the PcrA helicase in DNA unwinding. Nucleic Acids Res. 2008, 36, 1976–1989. [Google Scholar] [CrossRef]
- Rad, B.; Forget, A.L.; Baskin, R.J.; Kowalczykowski, S.C. Single-molecule visualization of RecQ helicase reveals DNA melting, nucleation, and assembly are required for processive DNA unwinding. Proc. Natl. Acad. Sci. USA 2015, 112, E6852–E6861. [Google Scholar] [CrossRef]
- Sikora, B.; Chen, Y.; Lichti, C.F.; Harrison, M.K.; Jennings, T.A.; Tang, Y.; Tackett, A.J.; Jordan, J.B.; Sakon, J.; Cameron, C.E.; et al. Hepatitis C virus NS3 helicase forms oligomeric structures that exhibit optimal DNA unwinding activity in vitro. J. Biol. Chem. 2008, 283, 11516–11525. [Google Scholar] [CrossRef]
- Xu, L.; Wang, L.; Peng, J.; Li, F.; Wu, L.; Zhang, B.; Lv, M.; Zhang, J.; Gong, Q.; Zhang, R.; et al. Insights into the Structure of Dimeric RNA Helicase CsdA and Indispensable Role of Its C-Terminal Regions. Structure 2017, 25, 1795–1808.e5. [Google Scholar] [CrossRef]
- Liou, G.-G.; Chang, H.-Y.; Lin, C.-S.; Lin-Chao, S. DEAD Box RhlB RNA Helicase Physically Associates with Exoribonuclease PNPase to Degrade Double-stranded RNA Independent of the Degradosome-assembling Region of RNase E. J. Biol. Chem. 2002, 277, 41157–41162. [Google Scholar] [CrossRef]
- Huen, J.; Lin, C.-L.; Golzarroshan, B.; Yi, W.-L.; Yang, W.-Z.; Yuan, H.S. Structural Insights into a Unique Dimeric DEAD-Box Helicase CshA that Promotes RNA Decay. Structure 2017, 25, 469–481. [Google Scholar] [CrossRef]
- Klostermeier, D.; Rudolph, M.G. A novel dimerization motif in the C-terminal domain of the Thermus thermophilus DEAD box helicase Hera confers substantial flexibility. Nucleic Acids Res. 2008, 37, 421–430. [Google Scholar] [CrossRef] [PubMed]
- Mechanic, L.E.; Hall, M.C.; Matson, S.W. Escherichia coli DNA Helicase II Is Active as a Monomer. J. Biol. Chem. 1999, 274, 12488–12498. [Google Scholar] [CrossRef] [PubMed]
- Chisty, L.T.; Toseland, C.P.; Fili, N.; Mashanov, G.I.; Dillingham, M.S.; Molloy, J.E.; Webb, M.R. Monomeric PcrA helicase processively unwinds plasmid lengths of DNA in the presence of the initiator protein RepD. Nucleic Acids Res. 2013, 41, 5010–5023. [Google Scholar] [CrossRef] [PubMed]
- Sikora, B.; Eoff, R.L.; Matson, S.W.; Raney, K.D. DNA Unwinding by Escherichia coli DNA Helicase I (TraI) Provides Evidence for a Processive Monomeric Molecular Motor. J. Biol. Chem. 2006, 281, 36110–36116. [Google Scholar] [CrossRef] [PubMed]
- Nanduri, B.; Byrd, A.K.; Eoff, R.L.; Tackett, A.J.; Raney, K.D. Pre-steady-state DNA unwinding by bacteriophage T4 Dda helicase reveals a monomeric molecular motor. Proc. Natl. Acad. Sci. USA 2002, 99, 14722–14727. [Google Scholar] [CrossRef]
- Eoff, R.L.; Raney, K.D. Kinetic Mechanism for DNA Unwinding by Multiple Molecules of Dda Helicase Aligned on DNA. Biochemistry 2010, 49, 4543–4553. [Google Scholar] [CrossRef][Green Version]
- Xu, H.Q.; Deprez, E.; Zhang, A.H.; Tauc, P.; Ladjimi, M.M.; Brochon, J.C.; Auclair, C.; Xi, X.G. The Escherichia coli RecQ helicase functions as a monomer. J. Biol. Chem. 2003, 278, 34925–34933. [Google Scholar]
- Zhang, X.D.; Dou, S.X.; Xie, P.; Hu, J.S.; Wang, P.Y.; Xi, X.G. Escherichia coli RecQ is a rapid, efficient, and monomeric helicase. J. Biol. Chem. 2006, 281, 12655–12663. [Google Scholar] [CrossRef]
- Li, N.; Henry, E.; Guiot, E.; Rigolet, P.; Brochon, J.C.; Xi, X.G.; Deprez, E. Multiple Escherichia coli RecQ helicase monomers cooperate to unwind long DNA substrates: A fluorescence cross-correlation spectroscopy study. J. Biol. Chem. 2010, 285, 6922–6936. [Google Scholar] [CrossRef]
- Kim, J.L.; Morgenstern, K.A.; Griffith, J.P.; Dwyer, M.D.; Thomson, J.A.; Murcko, M.A.; Lin, C.; Caron, P.R. Hepatitis C virus NS3 RNA helicase domain with a bound oligonucleotide: The crystal structure provides insights into the mode of unwinding. Structure 1998, 6, 89–100. [Google Scholar] [CrossRef]
- Maluf, N.K.; Lohman, T.M. Self-association Equilibria of Escherichia coli UvrD Helicase Studied by Analytical Ultracentrifugation. J. Mol. Biol. 2003, 325, 889–912. [Google Scholar] [CrossRef]
- Niedziela-Majka, A.; Chesnik, M.A.; Tomko, E.J.; Lohman, T.M. Bacillus stearothermophilus PcrA Monomer Is a Single-stranded DNA Translocase but Not a Processive Helicase in Vitro. J. Biol. Chem. 2007, 282, 27076–27085. [Google Scholar] [CrossRef] [PubMed]
- Park, E.; Guzder, S.N.; Koken, M.H.; Jaspers-Dekker, I.; Weeda, G.; Hoeijmakers, J.H.; Prakash, S. RAD25 (SSL2), the yeast homolog of the human xeroderma pigmentosum group B DNA repair gene, is essential for viability. Proc. Natl. Acad. Sci. USA 1992, 89, 11416–11420. [Google Scholar] [CrossRef] [PubMed]
- Ziegelin, G.; Linderoth, N.A.; Calendar, R.; Lanka, E. Domain structure of phage P4 alpha protein deduced by mutational analysis. J. Bacteriol. 1995, 177, 4333–4341. [Google Scholar] [CrossRef] [PubMed]
- Yu, C.-E.; Oshima, J.; Fu, Y.-H.; Wijsman, E.M.; Hisama, F.; Alisch, R.; Matthews, S.; Nakura, J.; Miki, T.; Ouais, S.; et al. Positional cloning of the Werner’s syndrome gene. Science 1996, 272, 258–262. [Google Scholar] [CrossRef]
- Von Kobbe, C.; Thomä, N.H.; Czyzewski, B.K.; Pavletich, N.P.; Bohr, V.A. Werner Syndrome Protein Contains Three Structure-specific DNA Binding Domains. J. Biol. Chem. 2003, 278, 52997–53006. [Google Scholar] [CrossRef]
- Rudolph, M.G.; Klostermeier, D. When core competence is not enough: Functional interplay of the DEAD-box helicase core with ancillary domains and auxiliary factors in RNA binding and unwinding. Biol. Chem. 2015, 396, 849–865. [Google Scholar] [CrossRef]
- Giraud, C.; Hausmann, S.; Lemeille, S.; Prados, J.; Redder, P.; Linder, P. The C-terminal region of the RNA helicase CshA is required for the interaction with the degradosome and turnover of bulk RNA in the opportunistic pathogen Staphylococcus aureus. RNA Biol. 2015, 12, 658–674. [Google Scholar] [CrossRef]
- Mohr, G.; Del Campo, M.; Mohr, S.; Yang, Q.; Jia, H.; Jankowsky, E.; Lambowitz, A.M. Function of the C-terminal Domain of the DEAD-box Protein Mss116p Analyzed in Vivo and in Vitro. J. Mol. Biol. 2008, 375, 1344–1364. [Google Scholar] [CrossRef]
- Kossen, K.; Uhlenbeck, O.C. Cloning and biochemical characterization of Bacillus subtilis YxiN, a DEAD protein specifically activated by 23S rRNA: Delineation of a novel sub-family of bacterial DEAD proteins. Nucleic Acids Res. 1999, 27, 3811–3820. [Google Scholar] [CrossRef]
- Yang, L.; Yang, J.; Huang, Y.; Liu, Z.-R. Phosphorylation of p68 RNA helicase regulates RNA binding by the C-terminal domain of the protein. Biochem. Biophys. Res. Commun. 2004, 314, 622–630. [Google Scholar] [CrossRef] [PubMed]
- Lohman, T.M.; Bjornson, K.P. Mechanisms of helicase-catalyzed DNA unwinding. Annu. Rev. Biochem. 1996, 65, 169–214. [Google Scholar] [CrossRef] [PubMed]
- Manelyte, L.; Guy, C.P.; Smith, R.M.; Dillingham, M.S.; McGlynn, P.; Savery, N. The unstructured C-terminal extension of UvrD interacts with UvrB, but is dispensable for nucleotide excision repair. DNA Repair 2009, 8, 1300–1310. [Google Scholar] [CrossRef] [PubMed]
- Mechanic, L.E.; Latta, M.E.; Matson, S.W. A region near the C-terminal end of Escherichia coli DNA helicase II is required for single-stranded DNA binding. J. Bacteriol. 1999, 181, 2519–2526. [Google Scholar] [CrossRef]
- Yokota, H. Fluorescence microscopy for visualizing single-molecule protein dynamics. Biochim. Biophys. Acta Gen. Subj. 2020, 1864, 129362. [Google Scholar] [CrossRef] [PubMed]
- Ali, J.A.; Maluf, N.K.; Lohman, T.M. An oligomeric form of E. coli UvrD is required for optimal helicase activity 1 1Edited by D. E. Draper. J. Mol. Biol. 1999, 293, 815–834. [Google Scholar] [CrossRef] [PubMed]
- Maluf, N.K.; Ali, J.A.; Lohman, T.M. Kinetic Mechanism for Formation of the Active, Dimeric UvrD Helicase-DNA Complex. J. Biol. Chem. 2003, 278, 31930–31940. [Google Scholar] [CrossRef]
- Yokota, H.; Han, Y.-W.; Allemand, J.-F.; Xi, X.G.; Bensimon, D.; Croquette, V.; Ito, Y.; Harada, Y. Single-molecule Visualization of Binding Modes of Helicase to DNA on PEGylated Surfaces. Chem. Lett. 2009, 38, 308–309. [Google Scholar] [CrossRef]
- Runyon, G.T.; Wong, I.; Lohman, T.M. Overexpression, purification, DNA binding, and dimerization of the Escherichia coli uvrD gene product (Helicase II). Biochemistry 1993, 32, 602–612. [Google Scholar] [CrossRef]
- Tomko, E.J.; Lohman, T.M. Modulation of Escherichia coli UvrD Single-Stranded DNA Translocation by DNA Base Composition. Biophys. J. 2017, 113, 1405–1415. [Google Scholar] [CrossRef]
- Tomko, E.J.; Fischer, C.J.; Niedziela-Majka, A.; Lohman, T.M. A Nonuniform Stepping Mechanism for E. coli UvrD Monomer Translocation along Single-Stranded DNA. Mol. Cell 2007, 26, 335–347. [Google Scholar] [CrossRef] [PubMed]
- Ali, J.A.; Lohman, T.M. Kinetic Measurement of the Step Size of DNA Unwinding by Escherichia coli UvrD Helicase. Science 1997, 275, 377–380. [Google Scholar] [CrossRef] [PubMed]
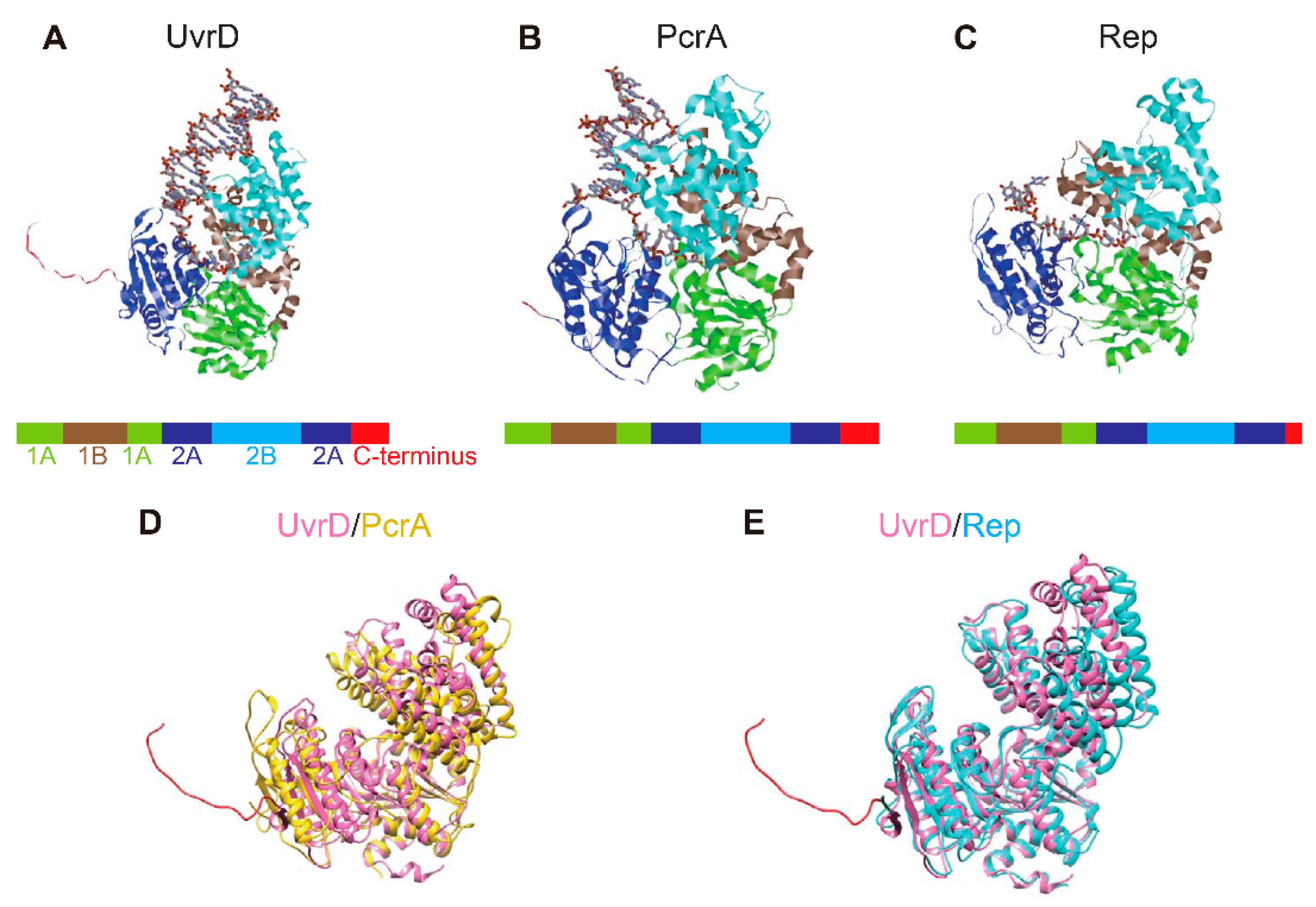
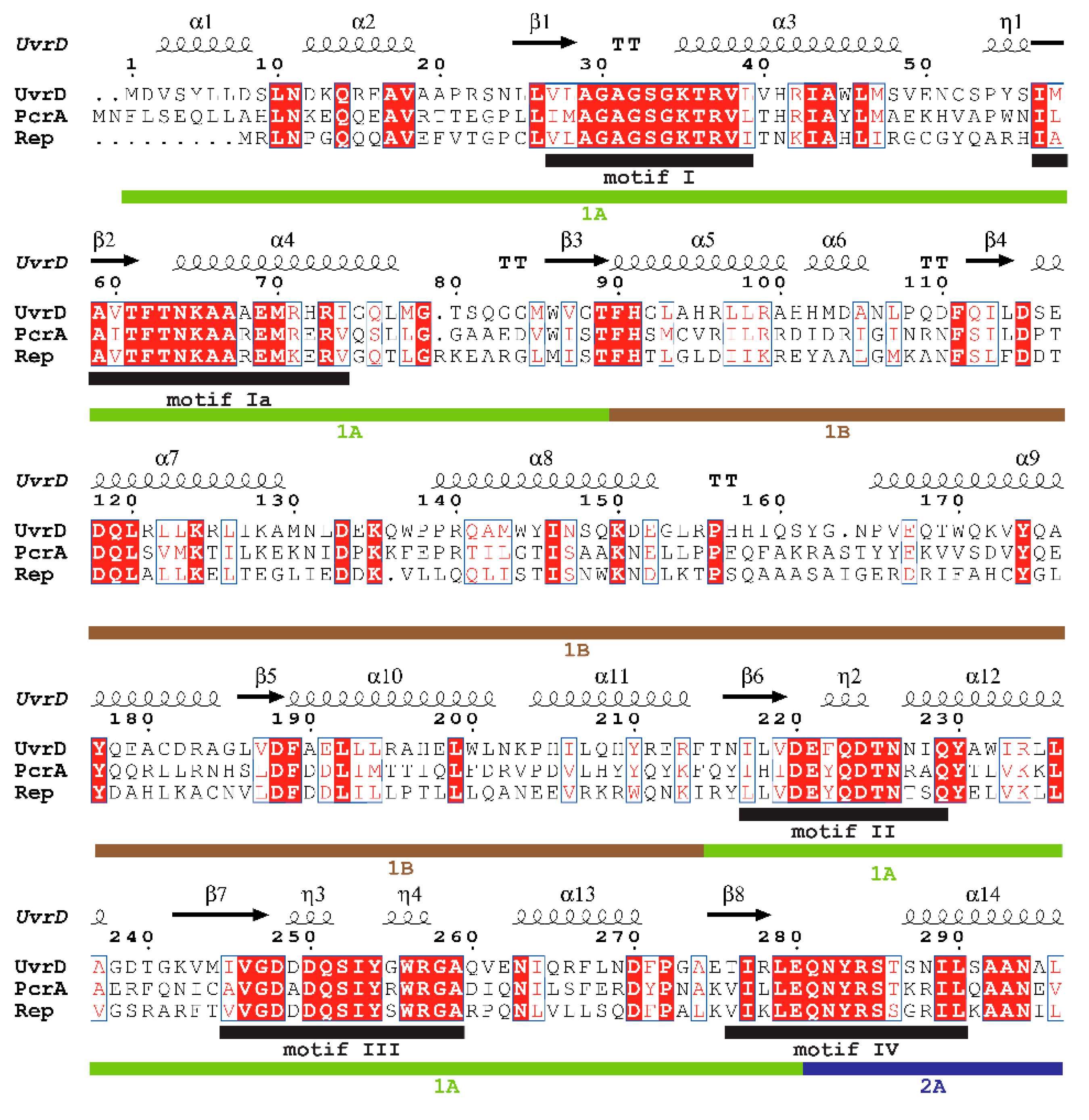
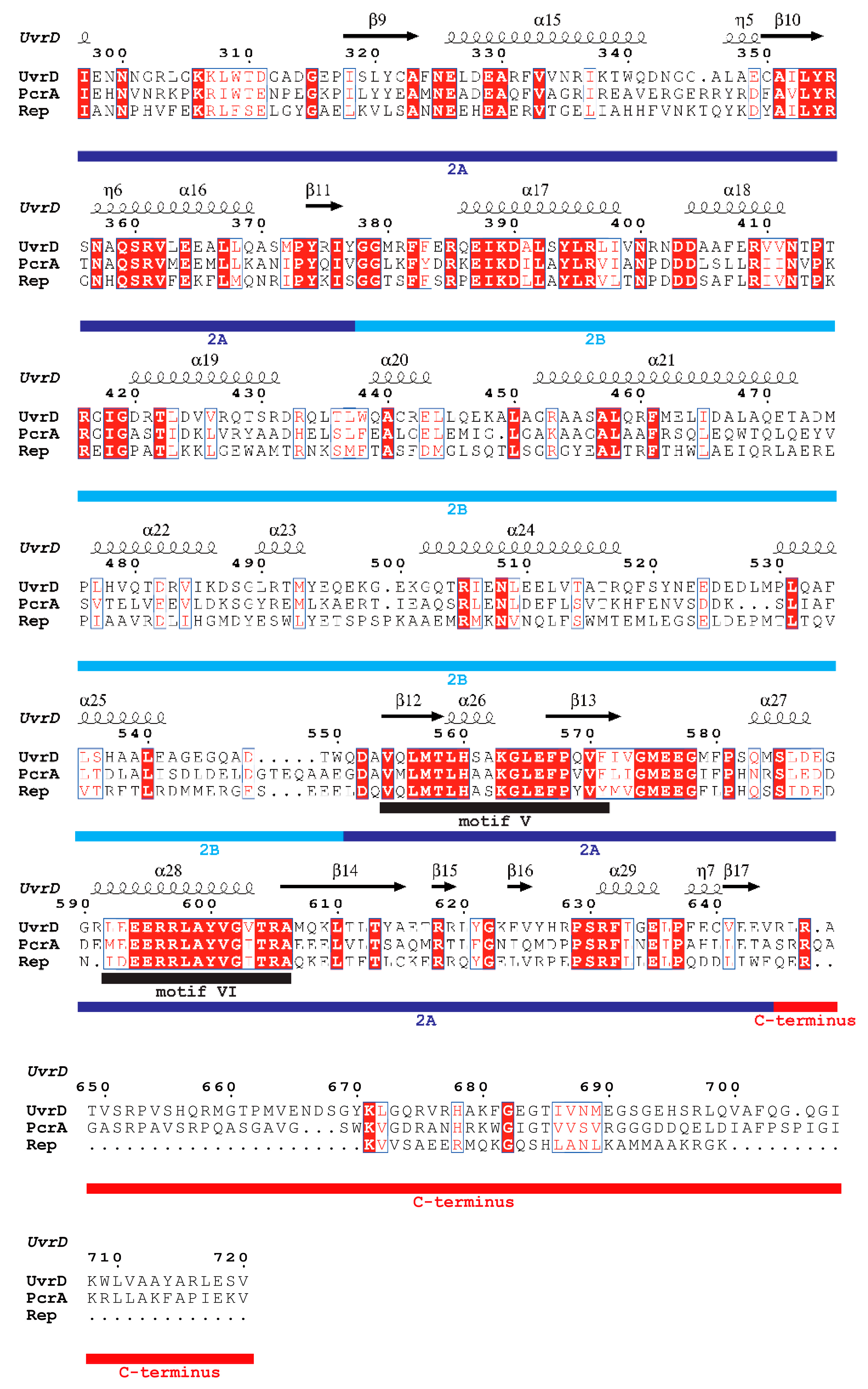
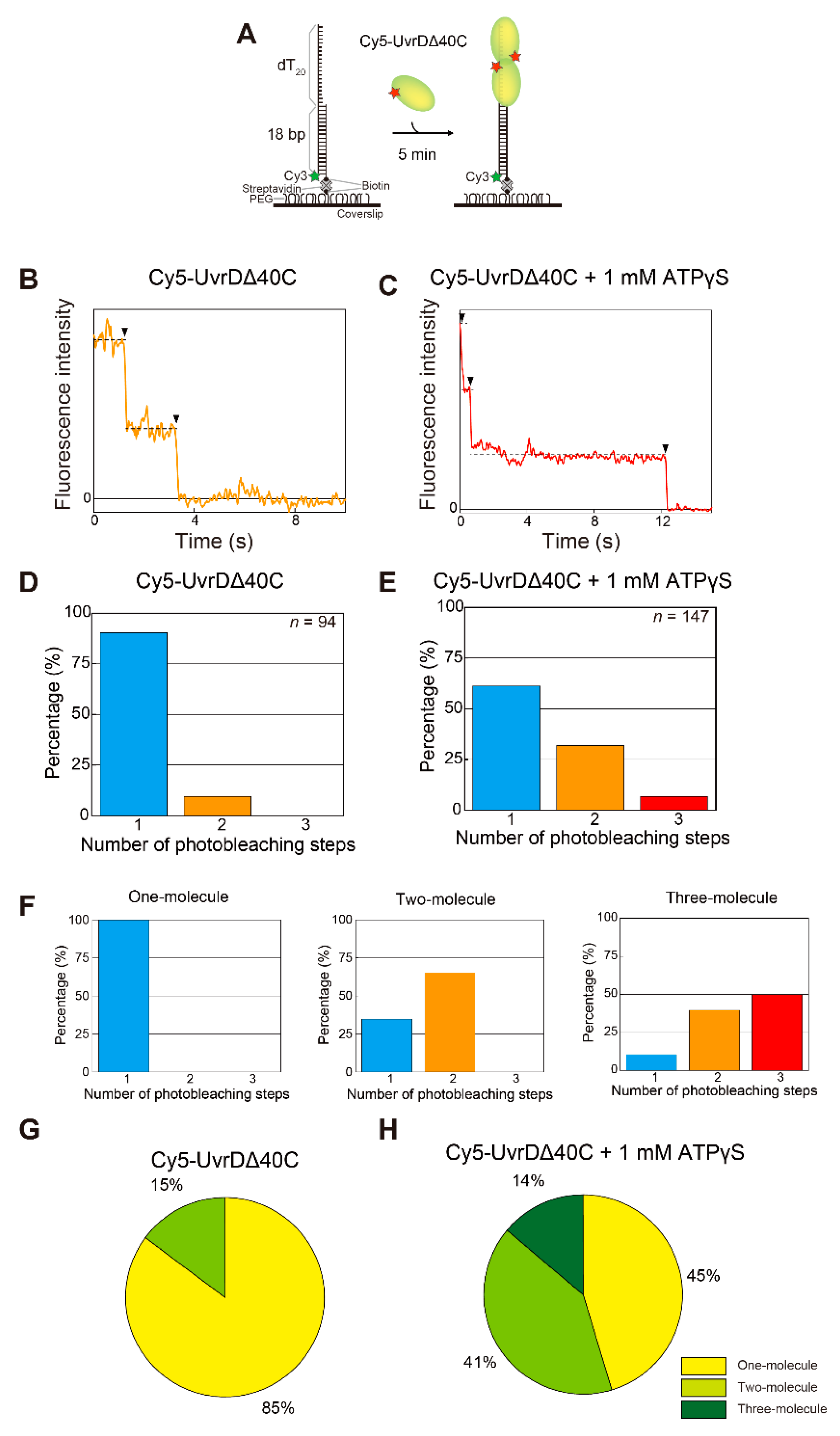
| SF | Helicase | Organism | Cellular Functions | Proposed Models for Functional Unit | References |
|---|---|---|---|---|---|
| SF1 | UvrD | E. coli | DNA repair | Monomer/Dimer | [6,10,16,17,18,19,20,21,32] |
| SF1 | Rep | E. coli | DNA replication | Dimer | [8,22,23] |
| SF1 | TraI | E. coli | DNA transfer during conjugation | Monomer/Dimer | [24,34] |
| SF1 | PcrA | B. subtilis | DNA repair | Monomer/Dimer | [9,25,33] |
| Rolling replication of plasmids | |||||
| SF1 | Dda | Phage T4 | DNA replication initiation | Monomer | [35,36] |
| DNA recombination | |||||
| SF2 | RecQ | E. coli | DNA recombination | Monomer/Dimer | [26,37,38,39] |
| SF2 | CsdA | E. coli | Ribosome biogenesis | Dimer | [28] |
| SF2 | RhlB | E. coli | RNA metabolism | Dimer | [29] |
| SF2 | NS3 | Hepatitis C virus | Viral DNA replication | Monomer/Dimer | [27,40] |
| SF2 | CshA | G. stearothermophilus | RNA metabolism | Dimer | [30] |
| SF2 | Hera | T. thermophilus | RNA metabolism | Dimer | [31] |
| Subfamily | Helicase | Organism | Roles of C-Terminal Amino Acids | References |
|---|---|---|---|---|
| DEAD-box | CsdA | E. coli | RNA binding | [28] |
| DEAD-box | CshA | G. stearothermophilus | RNA binding, RNA-dependent ATP hydrolysis, and interaction with degradosome | [30,48] |
| DEAD-box | Hera | T. thermophilus | RNA binding and dimerization | [31] |
| DEAD-box | Mss116p | S. cerevisae | RNA-dependent ATPases | [49] |
| DEAD-box | YxiN | B. subtilis | RNA binding | [50] |
| DEAD-box | p68 | Homo sapiens | RNA binding | [51] |
| UvrD | Size-Exclusion Chromatography | Sedimentation Equilibrium Experiments | Velocity Equilibrium Experiments |
|---|---|---|---|
| UvrDΔ40C | Monomer [32] | Monomer [32] | Monomer [32] |
| UvrDΔ73C | N.D. | Dimer [41] | N.D. |
| UvrD | DNA Repair | DNA-Stimulated ATPase | DNA Binding | DNA Unwinding |
|---|---|---|---|---|
| UvrDΔ40C | + 1 [32,54] | + [32,54] | + [54] | + [32,54] |
| UvrDΔ73C | + 2 [53] | + [53] | + [53] | + [53] |
| UvrDΔ102C | − 1 [54] | − [54] | − [54] | − [54] |
| UvrDΔ107C | − 1 [54] | N.D. | N.D. | N.D. |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yokota, H. Roles of the C-Terminal Amino Acids of Non-Hexameric Helicases: Insights from Escherichia coli UvrD. Int. J. Mol. Sci. 2021, 22, 1018. https://doi.org/10.3390/ijms22031018
Yokota H. Roles of the C-Terminal Amino Acids of Non-Hexameric Helicases: Insights from Escherichia coli UvrD. International Journal of Molecular Sciences. 2021; 22(3):1018. https://doi.org/10.3390/ijms22031018
Chicago/Turabian StyleYokota, Hiroaki. 2021. "Roles of the C-Terminal Amino Acids of Non-Hexameric Helicases: Insights from Escherichia coli UvrD" International Journal of Molecular Sciences 22, no. 3: 1018. https://doi.org/10.3390/ijms22031018
APA StyleYokota, H. (2021). Roles of the C-Terminal Amino Acids of Non-Hexameric Helicases: Insights from Escherichia coli UvrD. International Journal of Molecular Sciences, 22(3), 1018. https://doi.org/10.3390/ijms22031018





