Cathepsin X Activity Does Not Affect NK-Target Cell Synapse but Is Rather Distributed to Cytotoxic Granules
Abstract
1. Introduction
2. Results
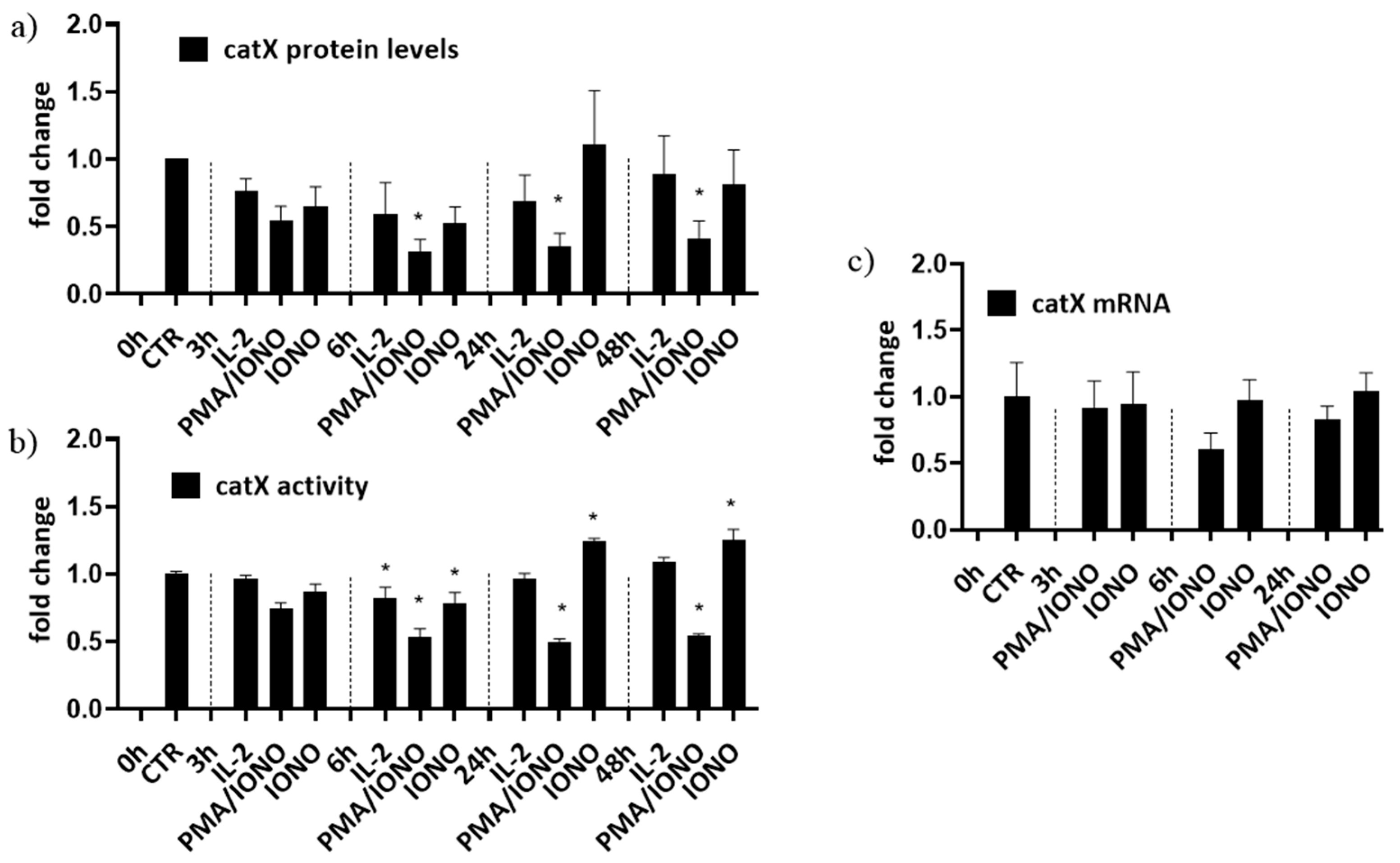
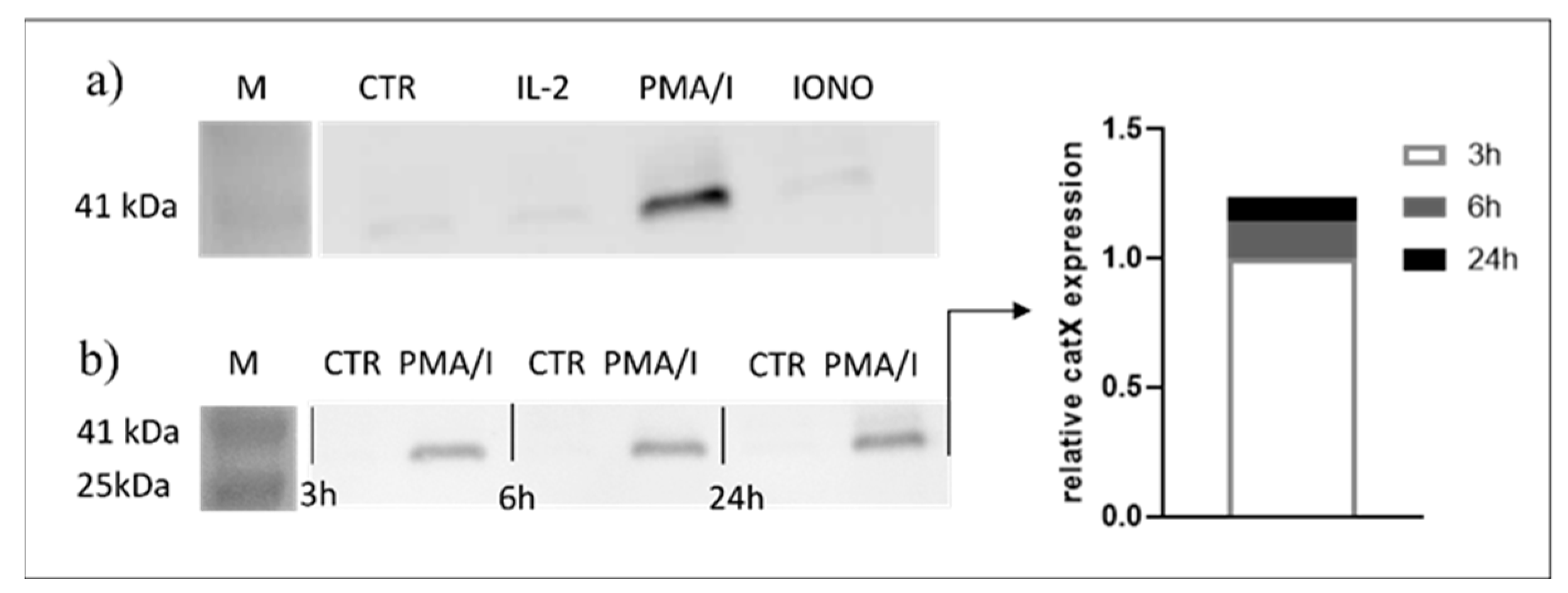
2.1. Regulation of Cathepsin X Expression and Activity in Activated NK-92 Cells
2.2. Cathepsin X Is Secreted upon Stimulation
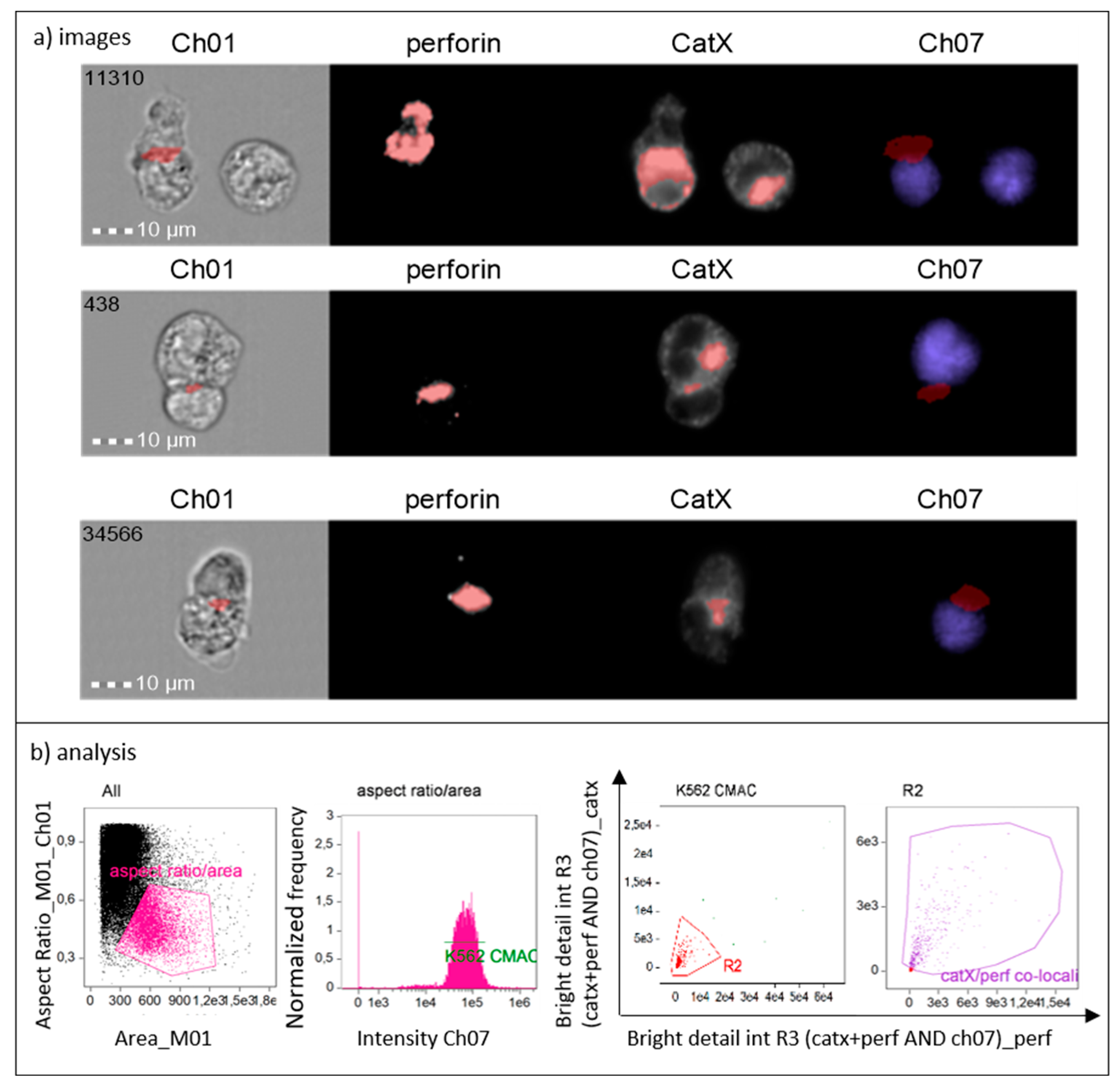
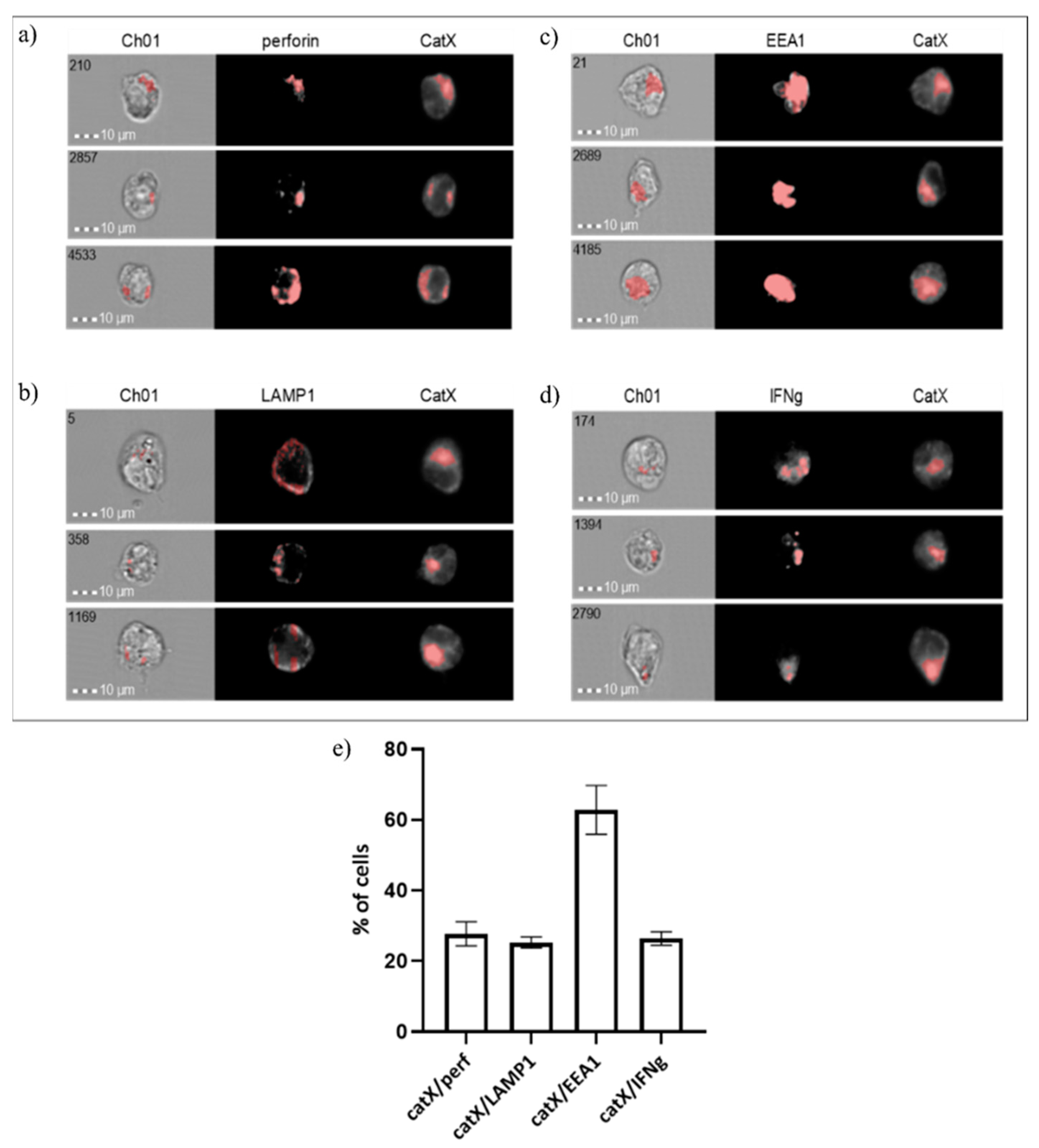
2.3. Cathepsin X Localization during NK-92 Cell Degranulation
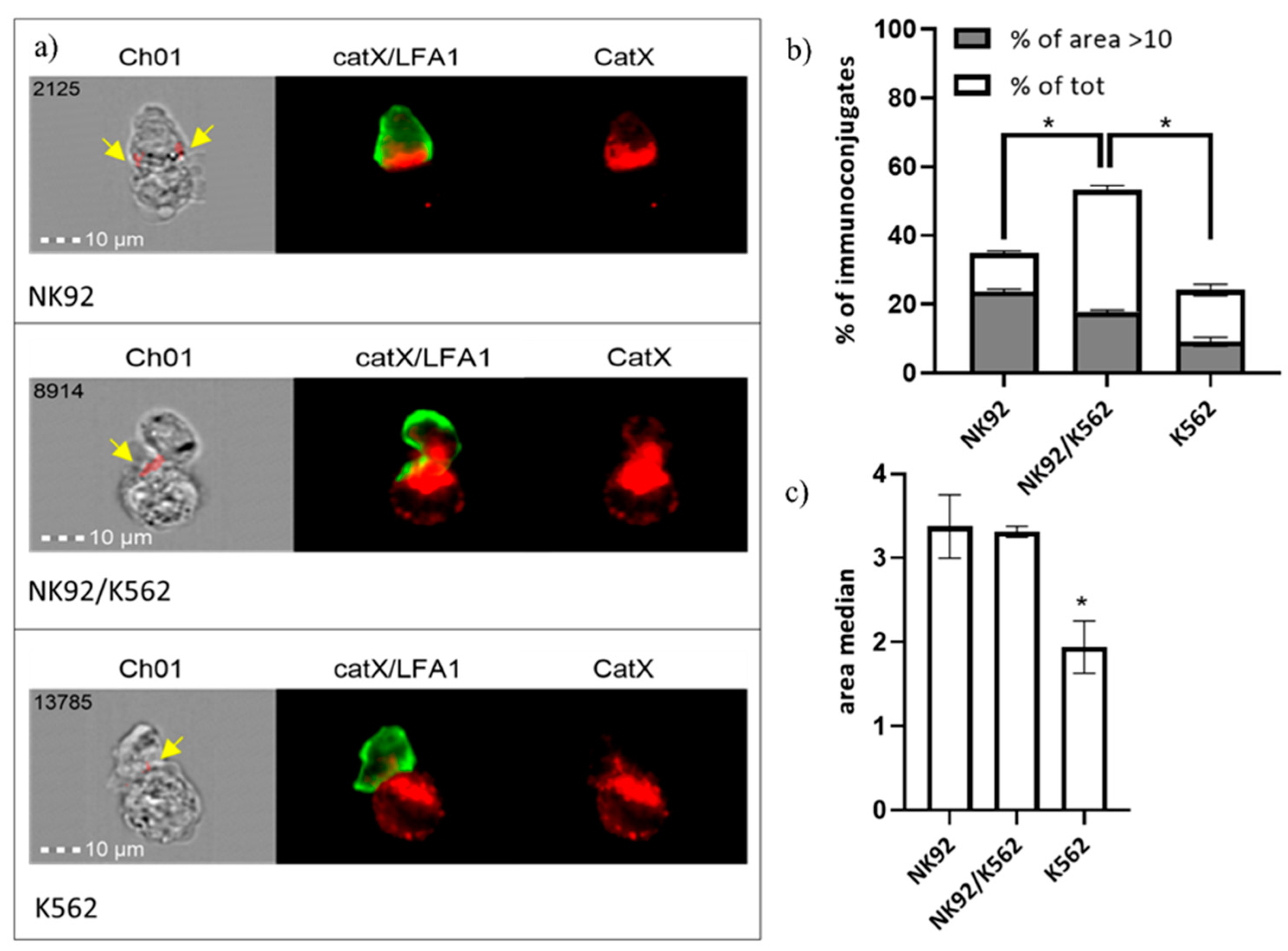
2.4. Cathepsin X Vesicular Distribution and Co-Localization with LFA-1
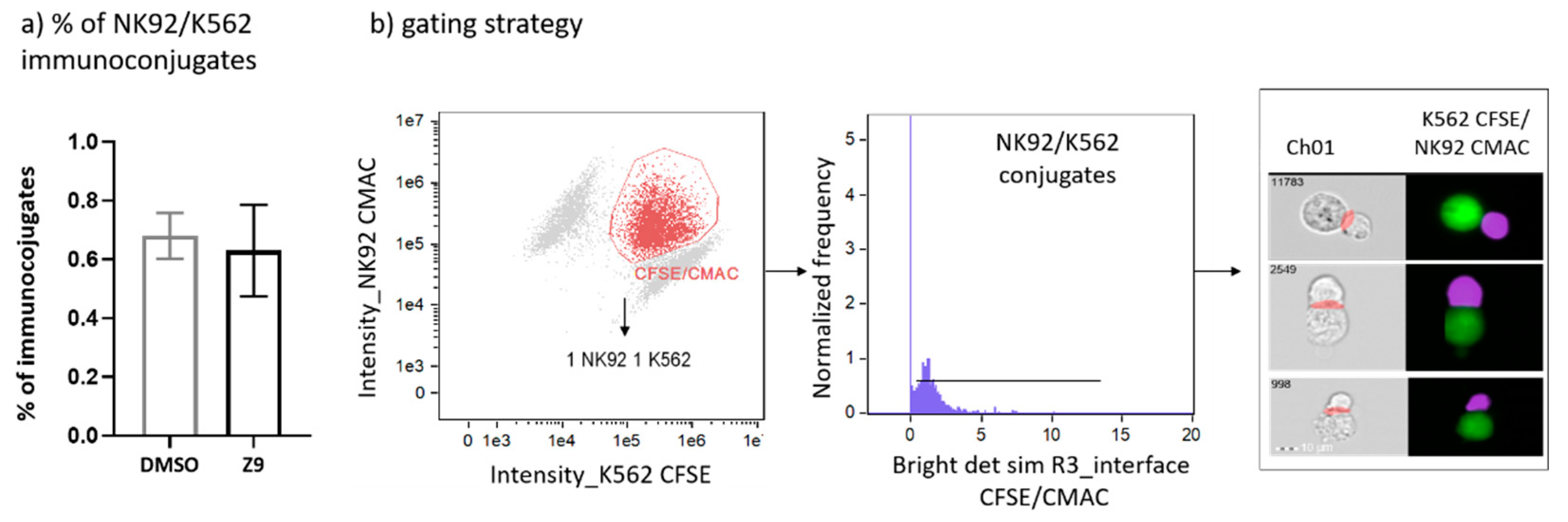
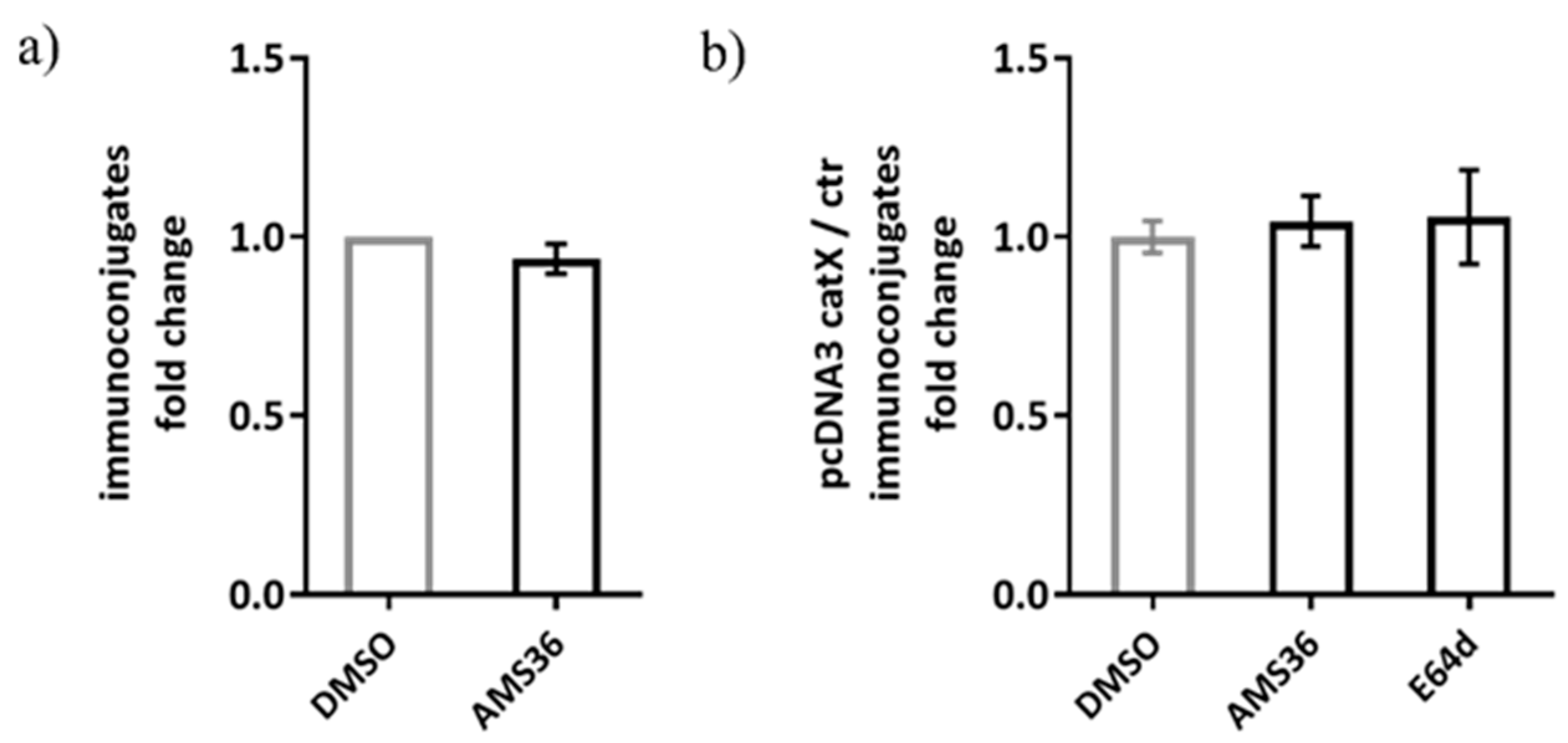
2.5. Cathepsin X Inhibition Does Not Perturb Formation of Stable Immunoconjugates
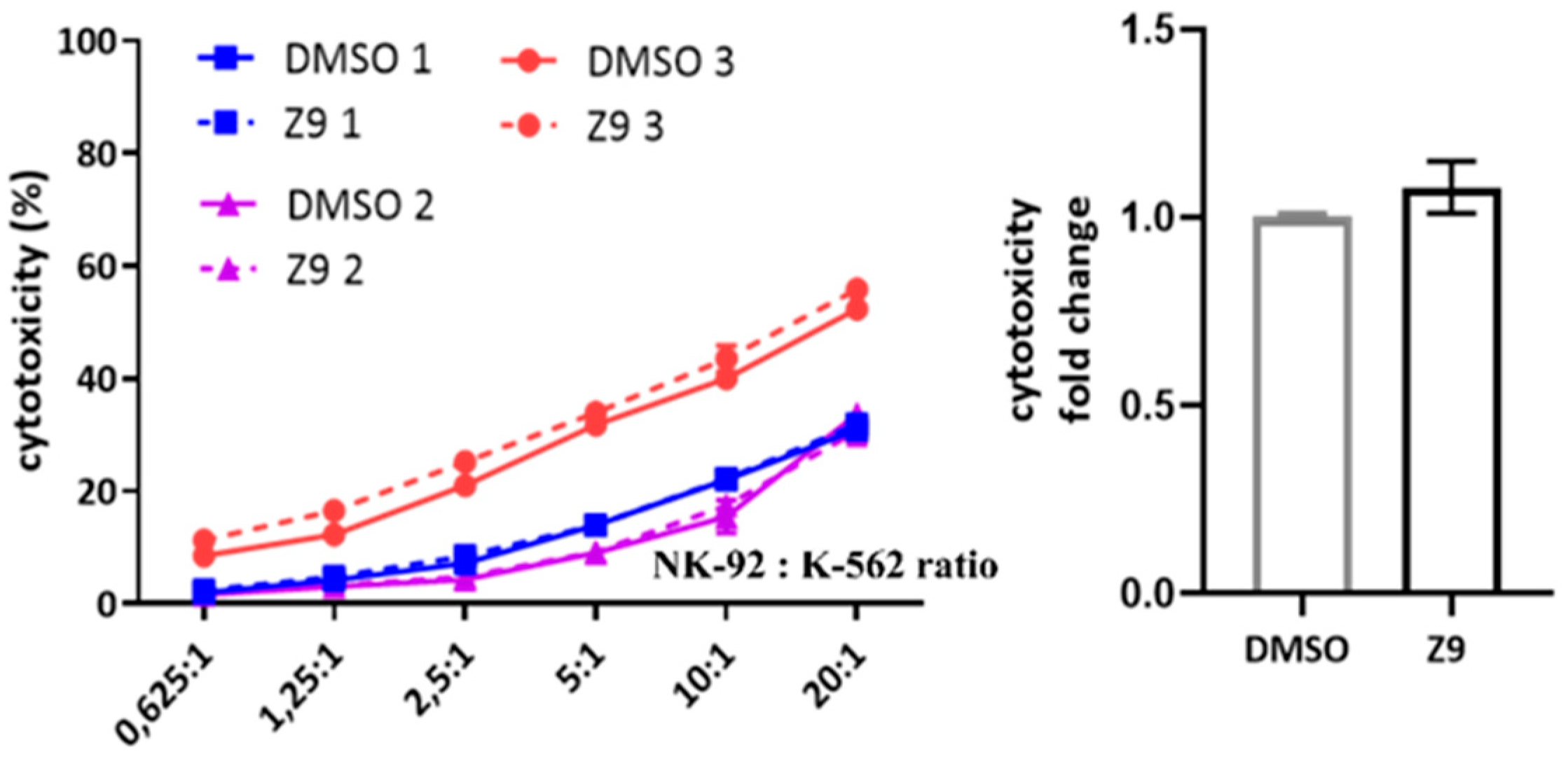
2.6. Cathepsin X Inhibition Does Not Perturb NK-92 Cell Lytic Activity
3. Discussion
4. Materials and Methods
4.1. Cell Lines
4.2. Cell Lysate Preparation for Measuring Cathepsin X Activity, mRNA Expression and Protein Levels
4.3. Kinetic Assay
4.4. Western Blotting
4.5. RNA Isolation and Quantitative Real-Time PCR Analysis
4.6. Collection of Concentrated Cell CULTURE Media
4.7. Imaging Flow Cytometry for Co-Localization Analysis
4.8. Immunoconjugate Stability Assay
4.9. Transient Transfection
4.10. Cytotoxicity Assay
4.11. Statistics
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
References
- Bonam, S.R.; Wang, F.; Muller, S. Lysosomes as a Therapeutic Target. Nat. Rev. Drug Discov. 2019, 18, 923–948. [Google Scholar] [CrossRef]
- Inpanathan, S.; Botelho, R.J. The Lysosome Signaling Platform: Adapting With the Times. Front. Cell Dev. Biol. 2019, 7, 113. [Google Scholar] [CrossRef]
- Matsumoto, F.; Saitoh, S.; Fukui, R.; Kobayashi, T.; Tanimura, N.; Konno, K.; Kusumoto, Y.; Akashi-Takamura, S.; Miyake, K. Cathepsins Are Required for Toll-like Receptor 9 Responses. Biochem. Biophys. Res. Commun. 2008, 367, 693–699. [Google Scholar] [CrossRef] [PubMed]
- Garcia-Cattaneo, A.; Gobert, F.-X.; Muller, M.; Toscano, F.; Flores, M.; Lescure, A.; Del Nery, E.; Benaroch, P. Cleavage of Toll-like Receptor 3 by Cathepsins B and H Is Essential for Signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 9053–9058. [Google Scholar] [CrossRef] [PubMed]
- Orlowski, G.M.; Colbert, J.D.; Sharma, S.; Bogyo, M.; Robertson, S.A.; Rock, K.L. Multiple Cathepsins Promote Pro–IL-1β Synthesis and NLRP3-Mediated IL-1β Activation. J. Immunol. 2015, 195, 1685–1697. [Google Scholar] [CrossRef]
- Ha, S.-D.; Martins, A.; Khazaie, K.; Han, J.; Chan, B.M.C.; Kim, S.O. Cathepsin B Is Involved in the Trafficking of TNF- -Containing Vesicles to the Plasma Membrane in Macrophages. J. Immunol. 2008, 181, 690–697. [Google Scholar] [CrossRef] [PubMed]
- Müller, S.; Faulhaber, A.; Sieber, C.; Pfeifer, D.; Hochberg, T.; Gansz, M.; Deshmukh, S.D.; Dauth, S.; Brix, K.; Saftig, P.; et al. The Endolysosomal Cysteine Cathepsins L and K Are Involved in Macrophage-Mediated Clearance of Staphylococcus Aureus and the Concomitant Cytokine Induction. FASEB J. 2014, 28, 162–175. [Google Scholar] [CrossRef]
- Riese, R.J.; Mitchell, R.N.; Villadangos, J.A.; Shi, G.P.; Palmer, J.T.; Karp, E.R.; De Sanctis, G.T.; Ploegh, H.L.; Chapman, H.A. Cathepsin S Activity Regulates Antigen Presentation and Immunity. J. Clin. Investig. 1998, 101, 2351–2363. [Google Scholar] [CrossRef]
- Maher, K.; Konjar, S.; Watts, C.; Turk, B.; Kopitar-Jerala, N. Cystatin F Regulates Proteinase Activity in IL-2-Activated Natural Killer Cells. Protein Pept. Lett. 2014, 21, 957–965. [Google Scholar] [CrossRef]
- Olson, O.C.; Joyce, J.A. Cysteine Cathepsin Proteases: Regulators of Cancer Progression and Therapeutic Response. Nat. Rev. Cancer 2015, 15, 712–729. [Google Scholar] [CrossRef] [PubMed]
- Kramer, L.; Turk, D.; Turk, B. The Future of Cysteine Cathepsins in Disease Management. Trends Pharmacol. Sci. 2017, 38, 873–898. [Google Scholar] [CrossRef]
- Allan, E.R.O.; Campden, R.I.; Ewanchuk, B.W.; Tailor, P.; Balce, D.R.; McKenna, N.T.; Greene, C.J.; Warren, A.L.; Reinheckel, T.; Yates, R.M. A Role for Cathepsin Z in Neuroinflammation Provides Mechanistic Support for an Epigenetic Risk Factor in Multiple Sclerosis. J. Neuroinflammation 2017, 14, 103. [Google Scholar] [CrossRef] [PubMed]
- Kos, J.; Sekirnik, A.; Premzl, A.; Bergant, V.Z.; Langerholc, T.; Repnik, U.; Turk, B.; Werle, B.; Golouh, R.; Jeras, M.; et al. Carboxypeptidases Cathepsins {X} and {B} Display Distinct Protein Profile in Human Cells and Tissues. Exp. Cell Res. 2005, 306, 103–113. [Google Scholar] [CrossRef]
- Nagler, D.K.; Kruger, S.; Kellner, A.; Ziomek, E.; Menard, R.; Buhtz, P.; Krams, M.; Roessner, A.; Kellner, U. Up-Regulation of Cathepsin X in Prostate Cancer and Prostatic Intraepithelial Neoplasia. Prostate 2004, 60, 109–119. [Google Scholar] [CrossRef]
- Krueger, S.; Kalinski, T.; Hundertmark, T.; Wex, T.; Kuster, D.; Peitz, U.; Ebert, M.; Nagler, D.K.; Kellner, U.; Malfertheiner, P.; et al. Up-Regulation of Cathepsin X in Helicobacter Pylori Gastritis and Gastric Cancer. J. Pathol. 2005, 207, 32–42. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Chen, L.; Li, Y.; Guan, X.-Y. Overexpression of Cathepsin Z Contributes to Tumor Metastasis by Inducing Epithelial-Mesenchymal Transition in Hepatocellular Carcinoma. PLoS ONE 2011, 6, e24967. [Google Scholar] [CrossRef]
- Decock, J.; Obermajer, N.; Vozelj, S.; Hendrickx, W.; Paridaens, R.; Kos, J. Cathepsin B, Cathepsin H, Cathepsin X and Cystatin C in Sera of Patients with Early-Stage and Inflammatory Breast Cancer. Int. J. Biol. Markers 2008, 23, 161–168. [Google Scholar] [CrossRef]
- Vižin, T.; Christensen, I.J.; Wilhelmsen, M.; Nielsen, H.J.; Kos, J. Prognostic and Predictive Value of Cathepsin X in Serum from Colorectal Cancer Patients. BMC Cancer 2014, 14, 259. [Google Scholar] [CrossRef] [PubMed]
- Teller, A.; Jechorek, D.; Hartig, R.; Adolf, D.; Reissig, K.; Roessner, A.; Franke, S. Dysregulation of Apoptotic Signaling Pathways by Interaction of RPLP0 and Cathepsin X/Z in Gastric Cancer. Pathol. Res. Pract. 2015, 211, 62–70. [Google Scholar] [CrossRef]
- Kraus, S.; Fruth, M.; Bunsen, T.; Nagler, D.K. IGF-I Receptor Phosphorylation Is Impaired in Cathepsin X-Deficient Prostate Cancer Cells. Biol. Chem. 2012, 393, 1457–1462. [Google Scholar] [CrossRef][Green Version]
- Lechner, A.M.; Assfalg-Machleidt, I.; Zahler, S.; Stoeckelhuber, M.; Machleidt, W.; Jochum, M.; Nägler, D.K. RGD-Dependent Binding of Procathepsin X to Integrin Alpha v Beta 3 Mediates Cell-Adhesive Properties. J. Biol. Chem. 2006, 281, 39588–39597. [Google Scholar] [CrossRef] [PubMed]
- Lines, K.E.; Chelala, C.; Dmitrovic, B.; Wijesuriya, N.; Kocher, H.M.; Marshall, J.F.; Crnogorac-Jurcevic, T. S100P-Binding Protein, S100PBP, Mediates Adhesion through Regulation of Cathepsin Z in Pancreatic Cancer Cells. Am. J. Pathol. 2012, 180, 1485–1494. [Google Scholar] [CrossRef]
- Fonović, U.P.; Jevnikar, Z.; Rojnik, M.; Doljak, B.; Fonović, M.; Jamnik, P.; Kos, J. Profilin 1 as a Target for Cathepsin X Activity in Tumor Cells. PLoS ONE 2013, 8, e53918. [Google Scholar] [CrossRef]
- Mitrovic, A.; Fonovic, U.P.; Kos, J. Cysteine Cathepsins B and X Promote Epithelial-Mesenchymal Transition of Tumor Cells. Eur. J. Cell Biol. 2017, 96, 622–631. [Google Scholar] [CrossRef]
- Jakoš, T.; Pišlar, A.; Fonović, U.P.; Švajger, U.; Kos, J. Cysteine Cathepsins L and X Differentially Modulate Interactions between Myeloid-Derived Suppressor Cells and Tumor Cells. Cancer Immunol. Immunother. 2020, 69, 1869–1880. [Google Scholar] [CrossRef]
- Jevnikar, Z.; Obermajer, N.; Doljak, B.; Turk, S.; Gobec, S.; Svajger, U.; Hailfinger, S.; Thome, M.; Kos, J. Cathepsin X Cleavage of the 2 Integrin Regulates Talin-Binding and LFA-1 Affinity in T Cells. J. Leukoc. Biol. 2011, 90, 99–109. [Google Scholar] [CrossRef]
- Obermajer, N.; Premzl, A.; Zavašnik Bergant, T.; Turk, B.; Kos, J. Carboxypeptidase Cathepsin X Mediates Β2-Integrin-Dependent Adhesion of Differentiated U-937 Cells. Exp. Cell Res. 2006, 312, 2515–2527. [Google Scholar] [CrossRef] [PubMed]
- Obermajer, N.; Svajger, U.; Bogyo, M.; Jeras, M.; Kos, J. Maturation of Dendritic Cells Depends on Proteolytic Cleavage by Cathepsin {X}. J. Leukoc. Biol. 2008, 84, 1306–1315. [Google Scholar] [CrossRef] [PubMed]
- Obermajer, N.; Jevnikar, Z.; Doljak, B.; Sadaghiani, A.M.; Bogyo, M.; Kos, J. Cathepsin X-Mediated Β2 Integrin Activation Results in Nanotube Outgrowth. Cell. Mol. Life Sci. 2009, 66, 1126–1134. [Google Scholar] [CrossRef]
- Jevnikar, Z.; Obermajer, N.; Bogyo, M.; Kos, J. The Role of Cathepsin X in the Migration and Invasiveness of T Lymphocytes. J. Cell Sci. 2008, 121, 2652–2661. [Google Scholar] [CrossRef]
- Mace, E.M.; Monkley, S.J.; Critchley, D.R.; Takei, F. A Dual Role for Talin in NK Cell Cytotoxicity: Activation of LFA-1-Mediated Cell Adhesion and Polarization of NK Cells. J. Immunol. 2009, 182, 948–956. [Google Scholar] [CrossRef]
- Barber, D.F.; Faure, M.; Long, E.O. LFA-1 Contributes an Early Signal for NK Cell Cytotoxicity. J. Immunol. 2004, 173, 3653–3659. [Google Scholar] [CrossRef] [PubMed]
- Weitz-Schmidt, G.; Chreng, S.; Riek, S. Allosteric LFA-1 Inhibitors Modulate Natural Killer Cell Function. Mol. Pharmacol. 2009, 75, 355–362. [Google Scholar] [CrossRef]
- Zhang, C.; Oberoi, P.; Oelsner, S.; Waldmann, A.; Lindner, A.; Tonn, T.; Wels, W.S. Chimeric Antigen Receptor-Engineered NK-92 Cells: An Off-the-Shelf Cellular Therapeutic for Targeted Elimination of Cancer Cells and Induction of Protective Antitumor Immunity. Front. Immunol. 2017, 8, 533. [Google Scholar] [CrossRef] [PubMed]
- Klingemann, H.; Boissel, L.; Toneguzzo, F. Natural Killer Cells for Immunotherapy—Advantages of the NK-92 Cell Line over Blood NK Cells. Front. Immunol. 2016, 7, 91. [Google Scholar] [CrossRef] [PubMed]
- Tang, X.; Yang, L.; Li, Z.; Nalin, A.P.; Dai, H.; Xu, T.; Yin, J.; You, F.; Zhu, M.; Shen, W.; et al. First-in-Man Clinical Trial of CAR NK-92 Cells: Safety Test of CD33-CAR NK-92 Cells in Patients with Relapsed and Refractory Acute Myeloid Leukemia. Am. J. Cancer Res. 2018, 8, 1083–1089. [Google Scholar] [PubMed]
- Jewett, A.; Kos, J.; Kaur, K.; Turnsek, T.L.; Breznik, B.; Senjor, E.; Wong, P.; Nguyen, K.Y.; Ko, M.-W. Multiple Defects of Natural Killer Cells in Cancer Patients: Anarchy, Dysregulated Systemic Immunity, and Immunosuppression in Metastatic Cancer. Crit. Rev. Immunol. 2020, 40, 93–133. [Google Scholar] [CrossRef] [PubMed]
- Yan, Y.; Steinherz, P.; Klingemann, H.G.; Dennig, D.; Childs, B.H.; McGuirk, J.; O’Reilly, R.J. Antileukemia Activity of a Natural Killer Cell Line against Human Leukemias. Clin. Cancer Res. 1998, 4, 2859–2868. [Google Scholar]
- Romera-Cárdenas, G.; Thomas, L.M.; Lopez-Cobo, S.; García-Cuesta, E.M.; Long, E.O.; Reyburn, H.T. Ionomycin Treatment Renders NK Cells Hyporesponsive. PLoS ONE 2016, 11, e0150998. [Google Scholar] [CrossRef]
- Obermajer, N.; Repnik, U.; Jevnikar, Z.; Turk, B.; Kreft, M.; Kos, J. Cysteine Protease Cathepsin X Modulates Immune Response via Activation of β 2 Integrins. Immunology 2008, 124, 76–88. [Google Scholar] [CrossRef]
- Fonović, U.P.; Mitrović, A.; Knez, D.; Jakoš, T.; Pišlar, A.; Brus, B.; Doljak, B.; Stojan, J.; Žakelj, S.; Trontelj, J.; et al. Identification and Characterization of the Novel Reversible and Selective Cathepsin X Inhibitors. Sci. Rep. 2017, 7, 11459. [Google Scholar] [CrossRef]
- Urlaub, D.; Höfer, K.; Müller, M.-L.; Watzl, C. LFA-1 Activation in NK Cells and Their Subsets: Influence of Receptors, Maturation, and Cytokine Stimulation. J. Immunol. 2017, 198, 1944–1951. [Google Scholar] [CrossRef]
- Zhang, M.; March, M.E.; Lane, W.S.; Long, E.O. A Signaling Network Stimulated by Beta2 Integrin Promotes the Polarization of Lytic Granules in Cytotoxic Cells. Sci. Signal. 2014, 7, ra96. [Google Scholar] [CrossRef] [PubMed]
- Springer, T.A.; Dustin, M.L. Integrin Inside-out Signaling and the Immunological Synapse. Curr. Opin. Cell Biol. 2012, 24, 107–115. [Google Scholar] [CrossRef]
- Nishida, N.; Xie, C.; Shimaoka, M.; Cheng, Y.; Walz, T.; Springer, T.A. Activation of Leukocyte Beta2 Integrins by Conversion from Bent to Extended Conformations. Immunity 2006, 25, 583–594. [Google Scholar] [CrossRef]
- Jevnikar, Z.; Obermajer, N.; Pečar-Fonović, U.; Karaoglanovic-Carmona, A.; Kos, J. Cathepsin X Cleaves the Β2 Cytoplasmic Tail of LFA-1 Inducing the Intermediate Affinity Form of LFA-1 and α-Actinin-1 Binding. Eur. J. Immunol. 2009, 39, 3217–3227. [Google Scholar] [CrossRef]
- Edgington-Mitchell, L.E.; Rautela, J.; Duivenvoorden, H.M.; Jayatilleke, K.M.; van der Linden, W.A.; Verdoes, M.; Bogyo, M.; Parker, B.S. Cysteine Cathepsin Activity Suppresses Osteoclastogenesis of Myeloid-Derived Suppressor Cells in Breast Cancer. Oncotarget 2015, 6, 8–10. [Google Scholar] [CrossRef] [PubMed]
- Yan, X.; Wu, C.; Chen, T.; Santos, M.M.; Liu, C.-L.L.; Yang, C.; Zhang, L.; Ren, J.; Liao, S.; Guo, H.; et al. Cathepsin S Inhibition Changes Regulatory T-Cell Activity in Regulating Bladder Cancer and Immune Cell Proliferation and Apoptosis. Mol. Immunol. 2017, 82, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Feigelson, S.W.; Pasvolsky, R.; Cemerski, S.; Shulman, Z.; Grabovsky, V.; Ilani, T.; Sagiv, A.; Lemaitre, F.; Laudanna, C.; Shaw, A.S.; et al. Occupancy of Lymphocyte LFA-1 by Surface-Immobilized ICAM-1 Is Critical for TCR- but Not for Chemokine-Triggered LFA-1 Conversion to an Open Headpiece High-Affinity State. J. Immunol. 2010, 185, 7394–7404. [Google Scholar] [CrossRef] [PubMed]
- Evans, R.; Lellouch, A.C.; Svensson, L.; McDowall, A.; Hogg, N. The Integrin LFA-1 Signals through ZAP-70 to Regulate Expression of High-Affinity LFA-1 on T Lymphocytes. Blood 2011, 117, 3331–3342. [Google Scholar] [CrossRef]
- De Clercq, S.; Zwaenepoel, O.; Martens, E.; Vandekerckhove, J.; Guillabert, A.; Gettemans, J. Nanobody-Induced Perturbation of LFA-1/L-Plastin Phosphorylation Impairs MTOC Docking, Immune Synapse Formation and T Cell Activation. Cell. Mol. Life Sci. 2013, 70, 909–922. [Google Scholar] [CrossRef]
- Nurmi, S.M.; Autero, M.; Raunio, A.K.; Gahmberg, C.G.; Fagerholm, S.C. Phosphorylation of the LFA-1 Integrin Beta2-Chain on Thr-758 Leads to Adhesion, Rac-1/Cdc42 Activation, and Stimulation of CD69 Expression in Human T Cells. J. Biol. Chem. 2007, 282, 968–975. [Google Scholar] [CrossRef] [PubMed]
- Van de Wiel-van Kemenade, E.; Te Velde, A.A.; De Boer, A.J.; Weening, R.S.; Fischer, A.; Borst, J.; Melief, C.J.; Figdor, C.G. Both LFA-1-Positive and -Deficient T Cell Clones Require the CD2/LFA-3 Interaction for Specific Cytolytic Activation. Eur. J. Immunol. 1992, 22, 1467–1475. [Google Scholar] [CrossRef]
- Matsumoto, G.; Nghiem, M.P.; Nozaki, N.; Schmits, R.; Penninger, J.M. Cooperation between CD44 and LFA-1/CD11a Adhesion Receptors in Lymphokine-Activated Killer Cell Cytotoxicity. J. Immunol. 1998, 160, 5781–5789. [Google Scholar]
- Reefman, E.; Kay, J.G.; Wood, S.M.; Offenhäuser, C.; Brown, D.L.; Roy, S.; Stanley, A.C.; Low, P.C.; Manderson, A.P.; Stow, J.L. Cytokine Secretion Is Distinct from Secretion of Cytotoxic Granules in NK Cells. J. Immunol. 2010, 184, 4852–4862. [Google Scholar] [CrossRef] [PubMed]
- Puzer, L.; Cotrin, S.S.; Cezari, M.H.S.; Hirata, I.Y.; Juliano, M.A.; Stefe, I.; Turk, D.; Turk, B.; Juliano, L.; Carmona, A.K. Recombinant Human Cathepsin X Is a Carboxymonopeptidase Only: A Comparison with Cathepsins B and L. Biol. Chem. 2005, 386, 1191–1195. [Google Scholar] [CrossRef]
- MagellanTM, version 2.0.5; Comprehensive Reader Control and Data Processing Software; Tecan: Männedorf, Switzerland, 2011.
- Gene Tools, Automatic Image Analysis for All Gel Applications Software; Syngene: Bengaluru, Karnataka, India, 2019.
- Jensen, J.; Ørntoft, T. Normalization of real-time quantitative RT-PCR data: A model based variance estimation approach to identify genes suited for normalization–applied to bladder- and colon-cancer data-sets. Cancer Res. 2004, 64, 50. [Google Scholar] [CrossRef]
- Taylor, S.C.; Nadeau, K.; Abbasi, M.; Lachance, C.; Nguyen, M.; Fenrich, J. The Ultimate QPCR Experiment: Producing Publication Quality, Reproducible Data the First Time. Trends Biotechnol. 2019, 37, 761–774. [Google Scholar] [CrossRef]
- IDEAS, version 6.0; Image Data Exploration and Analysis Software; Luminex: Ashland, OR, USA, 2013.
- Na, B.-R.; Jun, C.-D. In Vitro Assessment of Immunological Synapse Formation by Flow Cytometry. Bio-Protocol 2016, 6, e1758. [Google Scholar] [CrossRef]
- FlowJo, version 7.6.5; TreeStar: Ashland, OR, USA, 2002.
- Bryant, J.; Day, R.; Whiteside, T.L.; Herberman, R.B. Calculation of Lytic Units for the Expression of Cell-Mediated Cytotoxicity. J. Immunol. Methods 1992, 146, 91–103. [Google Scholar] [CrossRef]
- GraphPad Prism, version 6.0; Comprehensive Analysis and Powerful Statistics Software; GraphPad Software Inc.: San Diego, CA, USA, 2021.

| Gene | Primer Pair | Sequence (5′→ 3′) |
|---|---|---|
| CtsZ | F R | TGAACCATGGGGCGAGAGAG AGTGCTCCTCGATGGCAAGG |
| GAPDH | F R | TGCACCACCAACTGCTTAGC TGGCATGGACTGTGGTCATG |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jakoš, T.; Prunk, M.; Pišlar, A.; Kos, J. Cathepsin X Activity Does Not Affect NK-Target Cell Synapse but Is Rather Distributed to Cytotoxic Granules. Int. J. Mol. Sci. 2021, 22, 13495. https://doi.org/10.3390/ijms222413495
Jakoš T, Prunk M, Pišlar A, Kos J. Cathepsin X Activity Does Not Affect NK-Target Cell Synapse but Is Rather Distributed to Cytotoxic Granules. International Journal of Molecular Sciences. 2021; 22(24):13495. https://doi.org/10.3390/ijms222413495
Chicago/Turabian StyleJakoš, Tanja, Mateja Prunk, Anja Pišlar, and Janko Kos. 2021. "Cathepsin X Activity Does Not Affect NK-Target Cell Synapse but Is Rather Distributed to Cytotoxic Granules" International Journal of Molecular Sciences 22, no. 24: 13495. https://doi.org/10.3390/ijms222413495
APA StyleJakoš, T., Prunk, M., Pišlar, A., & Kos, J. (2021). Cathepsin X Activity Does Not Affect NK-Target Cell Synapse but Is Rather Distributed to Cytotoxic Granules. International Journal of Molecular Sciences, 22(24), 13495. https://doi.org/10.3390/ijms222413495






