Constitutive, Basal, and β-Alanine-Mediated Activation of the Human Mas-Related G Protein-Coupled Receptor D Induces Release of the Inflammatory Cytokine IL-6 and Is Dependent on NF-κB Signaling
Abstract
:1. Introduction
2. Results
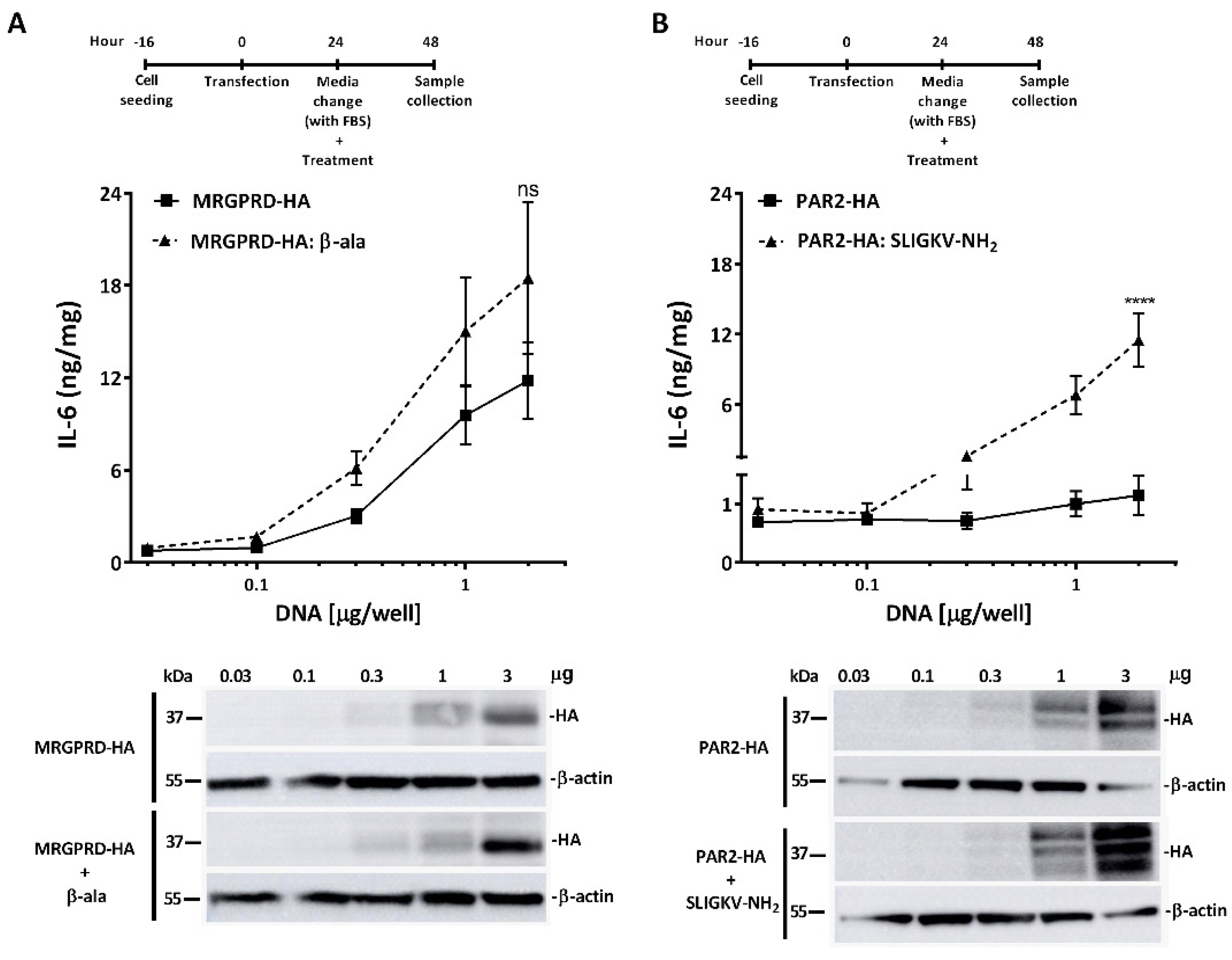
2.1. β-Alanine-Mediated Activation of Human MRGPRD Induces Release of IL-6
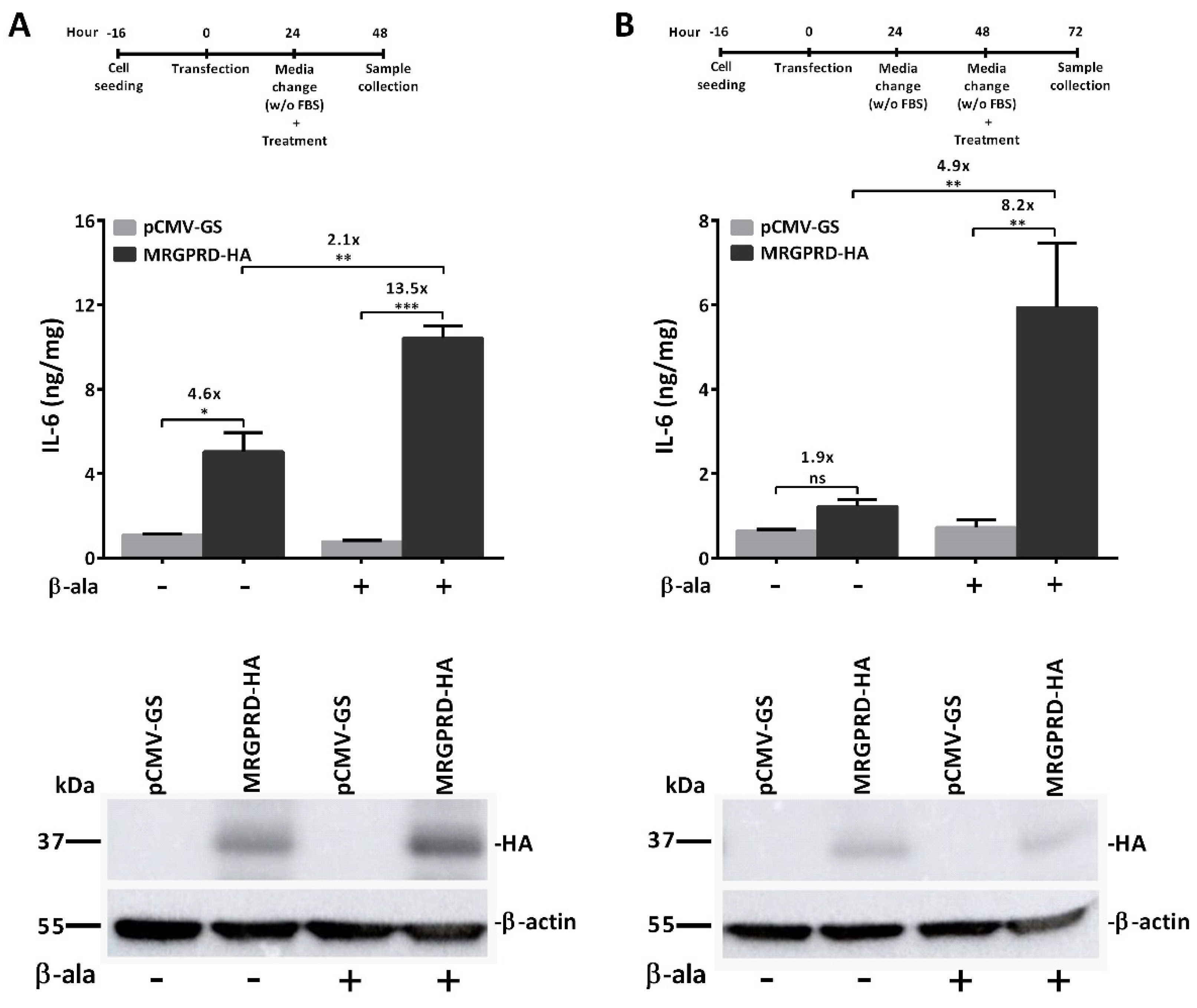
2.2. Demonstration of Basal Activity of MRGPRD and Assay Optimization to Maximize the IL-6 Detection Window
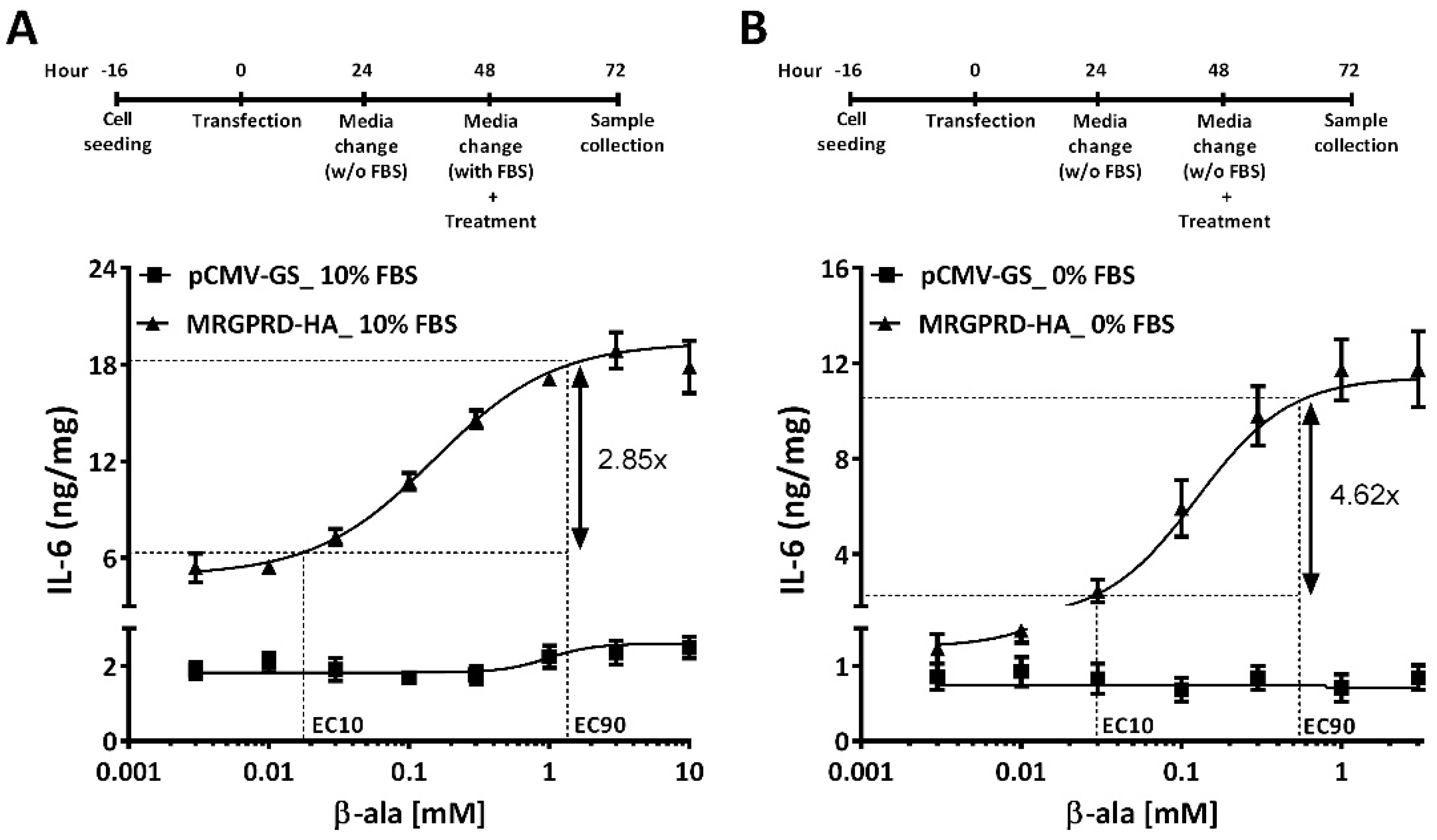
2.3. FBS Is Associated with Activation of MRGPRD and Induces Release of IL-6
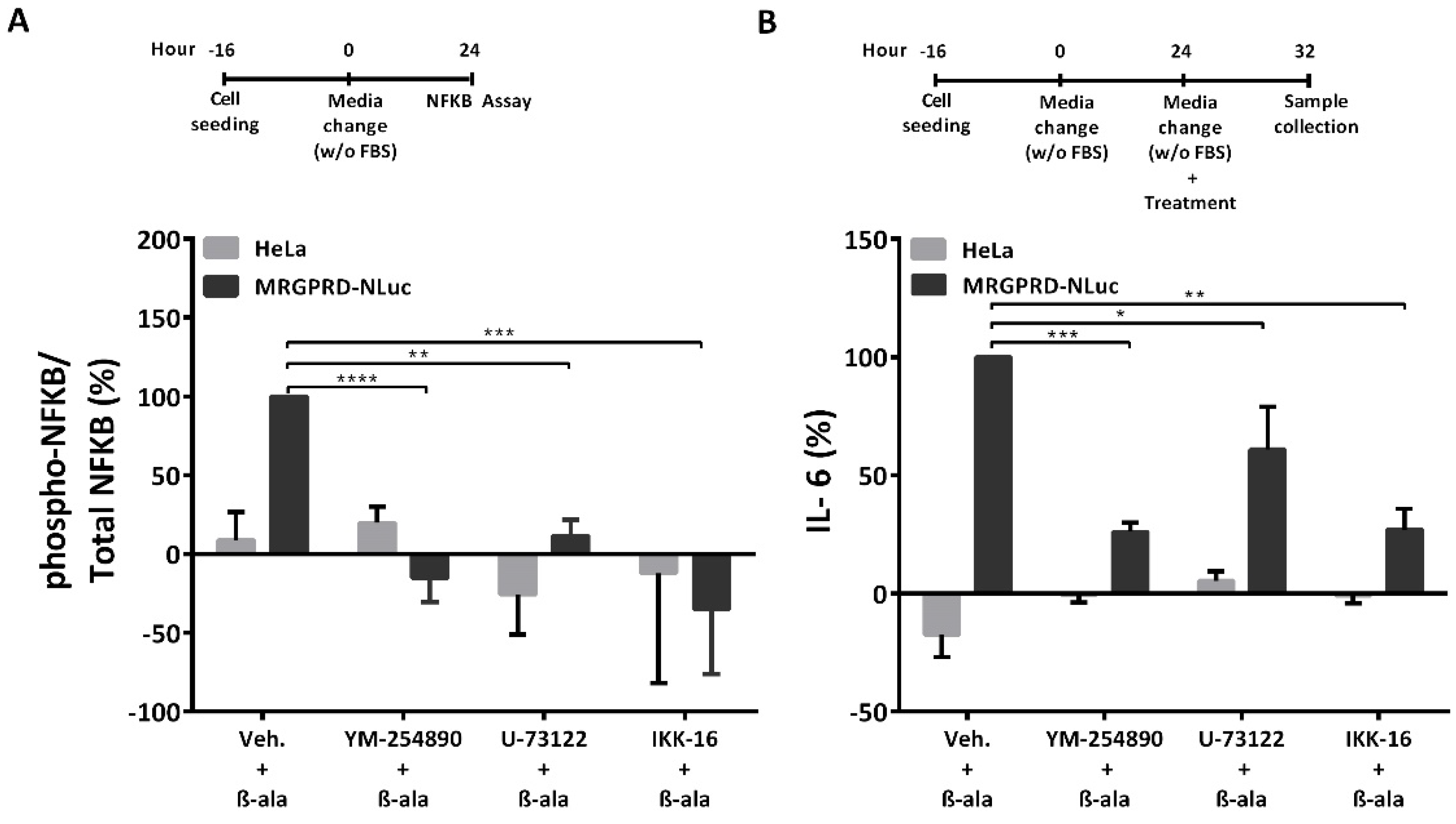
2.4. Inhibiting MRGPRD-Gαq/PLC/IKK/NF-κB Signaling Impedes the Release of IL-6
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Plasmid Preparation
4.3. Cell Culture, Transfections, Treatments, and IL-6 Detection
4.4. Immunoblotting
4.5. MRGPRD Stable Cell Preparation and IL-6 Inhibition
4.6. NF-kB Detection and Inhibition Assay
4.7. Data Analysis
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| β-AIBA | β-amino-isobutyric acid |
| cAMP | Cyclic adenosine monophosphate |
| DAG | Diacyl glycerol |
| DMEM | Dulbecco’s modified eagle medium |
| DRG | Dorsal root ganglia |
| ELISA | Enzyme-linked immunosorbent assay |
| FBS | Fetal bovine serum |
| GABA | Gamma-aminobutyric acid |
| GPCR | G protein-coupled receptor |
| HA | Hemagglutinin |
| IBS | Irritable bowel syndrome |
| IKK | IkB kinase |
| IL-1β | Interleukin-1 beta |
| IL-6 | Interleukin-6 |
| IP | Inositol phosphate |
| LPS | Lipopolysaccharide |
| MRGPRD | Mas-related G-protein coupled receptor D |
| NF-κB | Nuclear factor kappa-B |
| NLuc | NanoLuc |
| NO | Nitric oxide |
| PAR2 | Protease-activated receptor 2 |
| PBS | Phosphate buffered saline |
| PIP2 | Phosphatidylinositol 4,5-biphosphate |
| PLC | Phospholipase C |
| PUFA | Polyunsaturated fatty acid |
| RAS | Renin-angiotensin system |
| TG | Trigeminal ganglia |
| TNF-α | Tumor necrosis factor-alpha |
| TRPA1 | Transient receptor potential cation channel-A1 |
| 5-oxoETE | 5-oxoeicosatetraenoic acid |
References
- Sun, L.; Ye, R.D. Role of G protein-coupled receptors in inflammation. Acta Pharmacol. Sin. 2012, 33, 342–350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dong, X.; Han, S.; Zylka, M.J.; Simon, M.I.; Anderson, D.J. A diverse family of GPCRs expressed in specific subsets of nociceptive sensory neurons. Cell 2001, 106, 619–632. [Google Scholar] [CrossRef] [Green Version]
- Zylka, M.J.; Dong, X.; Southwell, A.L.; Anderson, D.J. Atypical expansion in mice of the sensory neuron-specific Mrg G protein-coupled receptor family. Proc. Natl. Acad. Sci. USA 2003, 100, 10043–10048. [Google Scholar] [CrossRef] [Green Version]
- Avula, L.R.; Buckinx, R.; Favoreel, H.; Cox, E.; Adriaensen, D.; van Nassauw, L.; Timmermans, J.P. Expression and distribution patterns of Mas-related gene receptor subtypes A-H in the mouse intestine: Inflammation-induced changes. Histochem. Cell Biol. 2013, 139, 639–658. [Google Scholar] [CrossRef]
- Solinski, H.J.; Gudermann, T.; Breit, A. Pharmacology and signaling of MAS-related G protein-coupled receptors. Pharmacol. Rev. 2014, 66, 570–597. [Google Scholar] [CrossRef] [Green Version]
- Bader, M.; Alenina, N.; Andrade-Navarro, M.A.; Santos, R.A. MAS and its related G protein-coupled receptors, Mrgprs. Pharmacol. Rev. 2014, 66, 1080–1105. [Google Scholar] [CrossRef] [PubMed]
- Alexander, S.P.; Christopoulos, A.; Davenport, A.P.; Kelly, E.; Mathie, A.; Peters, J.A.; Veale, E.L.; Armstrong, J.F.; Faccenda, E.; Harding, S.D.; et al. The concise guide to pharmacology 2021/22: G protein-coupled receptors. Br. J. Pharmacol. 2021, 178 (Suppl. 1), S27–S156. [Google Scholar] [CrossRef]
- Van Remoortel, S.; Ceuleers, H.; Arora, R.; van Nassauw, L.; de Man, J.G.; Buckinx, R.; de Winter, B.Y.; Timmermans, J.P. Mas-related G protein-coupled receptor C11 (Mrgprc11) induces visceral hypersensitivity in the mouse colon: A novel target in gut nociception? Neurogastroenterol. Motil. 2019, 31, e13623. [Google Scholar] [CrossRef]
- Castro, J.; Harrington, A.M.; Lieu, T.; Garcia-Caraballo, S.; Maddern, J.; Schober, G.; O’Donnell, T.; Grundy, L.; Lumsden, A.L.; Miller, P.; et al. Activation of pruritogenic TGR5, MrgprA3, and MrgprC11 on colon-innervating afferents induces visceral hypersensitivity. JCI Insight 2019, 4, e131712. [Google Scholar] [CrossRef]
- Grundy, L.; Caldwell, A.; Garcia-Caraballo, S.; Grundy, D.; Spencer, N.J.; Dong, X.; Castro, J.; Harrington, A.M.; Brierley, S.M. Activation of MrgprA3 and MrgprC11 on Bladder-Innervating Afferents Induces Peripheral and Central Hypersensitivity to Bladder Distension. J. Neurosci. 2021, 41, 3900–3916. [Google Scholar] [CrossRef] [PubMed]
- McNeil, B.D.; Pundir, P.; Meeker, S.; Han, L.; Undem, B.J.; Kulka, M.; Dong, X. Identification of a mast-cell-specific receptor crucial for pseudo-allergic drug reactions. Nature 2015, 519, 237–241. [Google Scholar] [CrossRef] [Green Version]
- Karhu, T.; Akiyama, K.; Vuolteenaho, O.; Bergmann, U.; Naito, T.; Tatemoto, K.; Herzig, K.H. Mast cell degranulation via MRGPRX2 by isolated human albumin fragments. Biochim. Biophys. Acta Gen. Subj. 2017, 1861, 2530–2534. [Google Scholar] [CrossRef] [PubMed]
- Arifuzzaman, M.; Mobley, Y.R.; Choi, H.W.; Bist, P.; Salinas, C.A.; Brown, Z.D.; Chen, S.L.; Staats, H.F.; Abraham, S.N. MRGPR-mediated activation of local mast cells clears cutaneous bacterial infection and protects against reinfection. Sci. Adv. 2019, 5, eaav0216. [Google Scholar] [CrossRef] [Green Version]
- Elst, J.; Sabato, V.; Faber, M.A.; Bridts, C.H.; Mertens, C.; van Houdt, M.; van Gasse, A.L.; Hagendorens, M.M.; van Tendeloo, V.; Maurer, M.; et al. MRGPRX2 and Immediate Drug Hypersensitivity: Insights from Cultured Human Mast Cells. J. Investig. Allergol. Clin. Immunol. 2020, 31, 1–25. [Google Scholar] [CrossRef] [PubMed]
- De Carvalho Santuchi, M.; Dutra, M.F.; Vago, J.P.; Lima, K.M.; Galvao, I.; de Souza-Neto, F.P.; Morais, E.S.M.; Oliveira, A.C.; de Oliveira, F.C.B.; Goncalves, R.; et al. Angiotensin-(1-7) and Alamandine Promote Anti-inflammatory Response in Macrophages In Vitro and In Vivo. Mediat. Inflamm. 2019, 2019, 2401081. [Google Scholar] [CrossRef] [Green Version]
- Hrenak, J.; Paulis, L.; Simko, F. Angiotensin A/Alamandine/MrgD Axis: Another Clue to Understanding Cardiovascular Pathophysiology. Int. J. Mol. Sci. 2016, 17, 1098. [Google Scholar] [CrossRef]
- Oliveira, A.C.; Melo, M.B.; Motta-Santos, D.; Peluso, A.A.; Souza-Neto, F.; da Silva, R.F.; Almeida, J.F.Q.; Canta, G.; Reis, A.M.; Goncalves, G.; et al. Genetic deletion of the alamandine receptor MRGD leads to dilated cardiomyopathy in mice. Am. J. Physiol. Heart Circ. Physiol. 2019, 316, H123–H133. [Google Scholar] [CrossRef] [Green Version]
- Lansu, K.; Karpiak, J.; Liu, J.; Huang, X.P.; McCorvy, J.D.; Kroeze, W.K.; Che, T.; Nagase, H.; Carroll, F.I.; Jin, J.; et al. In silico design of novel probes for the atypical opioid receptor MRGPRX2. Nat. Chem. Biol. 2017, 13, 529–536. [Google Scholar] [CrossRef]
- Karhu, T.; Akiyama, K.; Vuolteenaho, O.; Bergmann, U.; Naito, T.; Tatemoto, K.; Herzig, K.H. Isolation of new ligands for orphan receptor MRGPRX1-hemorphins LVV-H7 and VV-H7. Peptides 2017, 96, 61–66. [Google Scholar] [CrossRef]
- Zylka, M.J.; Rice, F.L.; Anderson, D.J. Topographically distinct epidermal nociceptive circuits revealed by axonal tracers targeted to Mrgprd. Neuron 2005, 45, 17–25. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crozier, R.A.; Ajit, S.K.; Kaftan, E.J.; Pausch, M.H. MrgD activation inhibits KCNQ/M-currents and contributes to enhanced neuronal excitability. J. Neurosci 2007, 27, 4492–4496. [Google Scholar] [CrossRef]
- Wang, C.; Gu, L.; Ruan, Y.; Geng, X.; Xu, M.; Yang, N.; Yu, L.; Jiang, Y.; Zhu, C.; Yang, Y.; et al. Facilitation of MrgprD by TRP-A1 promotes neuropathic pain. FASEB J. 2019, 33, 1360–1373. [Google Scholar] [CrossRef] [PubMed]
- Bautzova, T.; Hockley, J.R.F.; Perez-Berezo, T.; Pujo, J.; Tranter, M.M.; Desormeaux, C.; Barbaro, M.R.; Basso, L.; le Faouder, P.; Rolland, C.; et al. 5-oxoETE triggers nociception in constipation-predominant irritable bowel syndrome through MAS-related G protein-coupled receptor D. Sci. Signal. 2018, 11, eaal2171. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liu, Q.; Sikand, P.; Ma, C.; Tang, Z.; Han, L.; Li, Z.; Sun, S.; LaMotte, R.H.; Dong, X. Mechanisms of itch evoked by beta-alanine. J. Neurosci. 2012, 32, 14532–14537. [Google Scholar] [CrossRef]
- Lautner, R.Q.; Villela, D.C.; Fraga-Silva, R.A.; Silva, N.; Verano-Braga, T.; Costa-Fraga, F.; Jankowski, J.; Jankowski, V.; Sousa, F.; Alzamora, A.; et al. Discovery and characterization of alamandine: A novel component of the renin-angiotensin system. Circ. Res. 2013, 112, 1104–1111. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Villela, D.C.; Passos-Silva, D.G.; Santos, R.A. Alamandine: A new member of the angiotensin family. Curr. Opin. Nephrol. Hypertens. 2014, 23, 130–134. [Google Scholar] [CrossRef] [PubMed]
- Tetzner, A.; Gebolys, K.; Meinert, C.; Klein, S.; Uhlich, A.; Trebicka, J.; Villacanas, O.; Walther, T. G-Protein-Coupled Receptor MrgD Is a Receptor for Angiotensin-(1-7) Involving Adenylyl Cyclase, cAMP, and Phosphokinase A. Hypertension 2016, 68, 185–194. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, C.; Wu, X.; Shen, Y.; Liu, C.; Kong, X.; Li, P. Alamandine attenuates angiotensin II-induced vascular fibrosis via inhibiting p38 MAPK pathway. Eur. J. Pharmacol. 2020, 883, 173384. [Google Scholar] [CrossRef]
- Jesus, I.C.G.; Scalzo, S.; Alves, F.; Marques, K.; Rocha-Resende, C.; Bader, M.; Santos, R.A.S.; Guatimosim, S. Alamandine acts via MrgD to induce AMPK/NO activation against ANG II hypertrophy in cardiomyocytes. Am. J. Physiol. Cell Physiol. 2018, 314, C702–C711. [Google Scholar] [CrossRef]
- Jesus, I.C.G.; Mesquita, T.R.R.; Monteiro, A.L.L.; Parreira, A.B.; Santos, A.K.; Coelho, E.L.X.; Silva, M.M.; Souza, L.A.C.; Campagnole-Santos, M.J.; Santos, R.S.; et al. Alamandine enhances cardiomyocyte contractility in hypertensive rats through a nitric oxide-dependent activation of CaMKII. Am. J. Physiol. Cell Physiol. 2020, 318, C740–C750. [Google Scholar] [CrossRef]
- Cavanaugh, D.J.; Lee, H.; Lo, L.; Shields, S.D.; Zylka, M.J.; Basbaum, A.I.; Anderson, D.J. Distinct subsets of unmyelinated primary sensory fibers mediate behavioral responses to noxious thermal and mechanical stimuli. Proc. Natl. Acad. Sci. USA 2009, 106, 9075–9080. [Google Scholar] [CrossRef] [Green Version]
- Nishimura, S.; Uno, M.; Kaneta, Y.; Fukuchi, K.; Nishigohri, H.; Hasegawa, J.; Komori, H.; Takeda, S.; Enomoto, K.; Nara, F.; et al. MRGD, a MAS-related G-protein coupled receptor, promotes tumorigenisis and is highly expressed in lung cancer. PLoS ONE 2012, 7, e38618. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kitase, Y.; Vallejo, J.A.; Gutheil, W.; Vemula, H.; Jahn, K.; Yi, J.; Zhou, J.; Brotto, M.; Bonewald, L.F. Beta-aminoisobutyric Acid, l-BAIBA, Is a Muscle-Derived Osteocyte Survival Factor. Cell Rep. 2018, 22, 1531–1544. [Google Scholar] [CrossRef] [Green Version]
- Reddy, V.B.; Lerner, E.A. Activation of mas-related G-protein-coupled receptors by the house dust mite cysteine protease Der p1 provides a new mechanism linking allergy and inflammation. J. Biol. Chem. 2017, 292, 17399–17406. [Google Scholar] [CrossRef] [Green Version]
- Lan, L.; Xu, M.; Li, J.; Liu, L.; Xu, M.; Zhou, C.; Shen, L.; Tang, Z.; Wan, F. Mas-related G protein-coupled receptor D participates in inflammatory pain by promoting NF-kappaB activation through interaction with TAK1 and IKK complex. Cell Signal. 2020, 76, 109813. [Google Scholar] [CrossRef] [PubMed]
- Shinohara, T.; Harada, M.; Ogi, K.; Maruyama, M.; Fujii, R.; Tanaka, H.; Fukusumi, S.; Komatsu, H.; Hosoya, M.; Noguchi, Y.; et al. Identification of a G protein-coupled receptor specifically responsive to beta-alanine. J. Biol. Chem. 2004, 279, 23559–23564. [Google Scholar] [CrossRef] [Green Version]
- Uno, M.; Nishimura, S.; Fukuchi, K.; Kaneta, Y.; Oda, Y.; Komori, H.; Takeda, S.; Haga, T.; Agatsuma, T.; Nara, F. Identification of physiologically active substances as novel ligands for MRGPRD. J. Biomed. Biotechnol. 2012, 2012, 816159. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schon, M.; Mousa, A.; Berk, M.; Chia, W.L.; Ukropec, J.; Majid, A.; Ukropcova, B.; de Courten, B. The Potential of Carnosine in Brain-Related Disorders: A Comprehensive Review of Current Evidence. Nutrients 2019, 11, 1196. [Google Scholar] [CrossRef] [Green Version]
- Blancquaert, L.; Baba, S.P.; Kwiatkowski, S.; Stautemas, J.; Stegen, S.; Barbaresi, S.; Chung, W.; Boakye, A.A.; Hoetker, J.D.; Bhatnagar, A.; et al. Carnosine and anserine homeostasis in skeletal muscle and heart is controlled by beta-alanine transamination. J. Physiol. 2016, 594, 4849–4863. [Google Scholar] [CrossRef] [Green Version]
- Reddy, V.P.; Garrett, M.R.; Perry, G.; Smith, M.A. Carnosine: A versatile antioxidant and antiglycating agent. Sci. Aging Knowl. Environ. 2005, 2005, pe12. [Google Scholar] [CrossRef]
- Desai, S.; Kumar, A.; Laskar, S.; Pandey, B.N. Cytokine profile of conditioned medium from human tumor cell lines after acute and fractionated doses of gamma radiation and its effect on survival of bystander tumor cells. Cytokine 2013, 61, 54–62. [Google Scholar] [CrossRef]
- Milligan, G. Constitutive activity and inverse agonists of G protein-coupled receptors: A current perspective. Mol. Pharmacol. 2003, 64, 1271–1276. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Martin, A.L.; Steurer, M.A.; Aronstam, R.S. Constitutive Activity among Orphan Class-A G Protein Coupled Receptors. PLoS ONE 2015, 10, e0138463. [Google Scholar] [CrossRef] [Green Version]
- Lu, S.; Jang, W.; Inoue, A.; Lambert, N.A. Constitutive G protein coupling profiles of understudied orphan GPCRs. PLoS ONE 2021, 16, e0247743. [Google Scholar] [CrossRef] [PubMed]
- Ajit, S.K.; Pausch, M.H.; Kennedy, J.D.; Kaftan, E.J. Development of a FLIPR assay for the simultaneous identification of MrgD agonists and antagonists from a single screen. J. Biomed. Biotechnol. 2010, 2010, 326020. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taniguchi, M.; Nagai, K.; Arao, N.; Kawasaki, T.; Saito, T.; Moritani, Y.; Takasaki, J.; Hayashi, K.; Fujita, S.; Suzuki, K.; et al. YM-254890, a novel platelet aggregation inhibitor produced by Chromobacterium sp. QS3666. J. Antibiot. 2003, 56, 358–363. [Google Scholar] [CrossRef] [Green Version]
- Nishimura, A.; Kitano, K.; Takasaki, J.; Taniguchi, M.; Mizuno, N.; Tago, K.; Hakoshima, T.; Itoh, H. Structural basis for the specific inhibition of heterotrimeric Gq protein by a small molecule. Proc. Natl. Acad. Sci. USA 2010, 107, 13666–13671. [Google Scholar] [CrossRef] [Green Version]
- Charpentier, T.H.; Waldo, G.L.; Lowery-Gionta, E.G.; Krajewski, K.; Strahl, B.D.; Kash, T.L.; Harden, T.K.; Sondek, J. Potent and Selective Peptide-based Inhibition of the G Protein Galphaq. J. Biol. Chem. 2016, 291, 25608–25616. [Google Scholar] [CrossRef] [Green Version]
- Bleasdale, J.E.; Thakur, N.R.; Gremban, R.S.; Bundy, G.L.; Fitzpatrick, F.A.; Smith, R.J.; Bunting, S. Selective inhibition of receptor-coupled phospholipase C-dependent processes in human platelets and polymorphonuclear neutrophils. J. Pharmacol. Exp. Ther. 1990, 255, 756–768. [Google Scholar]
- Waelchli, R.; Bollbuck, B.; Bruns, C.; Buhl, T.; Eder, J.; Feifel, R.; Hersperger, R.; Janser, P.; Revesz, L.; Zerwes, H.G.; et al. Design and preparation of 2-benzamido-pyrimidines as inhibitors of IKK. Bioorg. Med. Chem. Lett. 2006, 16, 108–112. [Google Scholar] [CrossRef]
- Hauser, A.S.; Attwood, M.M.; Rask-Andersen, M.; Schioth, H.B.; Gloriam, D.E. Trends in GPCR drug discovery: New agents, targets and indications. Nat. Rev. Drug Discov. 2017, 16, 829–842. [Google Scholar] [CrossRef]
- Oeckinghaus, A.; Ghosh, S. The NF-kappaB family of transcription factors and its regulation. Cold Spring Harb. Perspect Biol. 2009, 1, a000034. [Google Scholar] [CrossRef]
- Patil, D.N.; Singh, S.; Laboute, T.; Strutzenberg, T.S.; Qiu, X.; Wu, D.; Novick, S.J.; Robinson, C.V.; Griffin, P.R.; Hunt, J.F.; et al. Cryo-EM structure of human GPR158 receptor coupled to the RGS7-Gbeta5 signaling complex. Science 2021, eabl4732. [Google Scholar]
- Thomsen, A.R.B.; Plouffe, B.; Cahill, T.J., 3rd; Shukla, A.K.; Tarrasch, J.T.; Dosey, A.M.; Kahsai, A.W.; Strachan, R.T.; Pani, B.; Mahoney, J.P.; et al. GPCR-G Protein-beta-Arrestin Super-Complex Mediates Sustained G Protein Signaling. Cell 2016, 166, 907–919. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dimri, S.; Arora, R.; Jasani, A.; De, A. Dynamic monitoring of STAT3 activation in live cells using a novel STAT3 Phospho-BRET sensor. Am. J. Nucl. Med. Mol. Imaging 2019, 9, 321–334. [Google Scholar] [PubMed]
- D’Orazi, G.; Cordani, M.; Cirone, M. Oncogenic pathways activated by pro-inflammatory cytokines promote mutant p53 stability: Clue for novel anticancer therapies. Cell Mol. Life Sci. 2021, 78, 1853–1860. [Google Scholar] [CrossRef] [PubMed]
- Seiler, C.Y.; Park, J.G.; Sharma, A.; Hunter, P.; Surapaneni, P.; Sedillo, C.; Field, J.; Algar, R.; Price, A.; Steel, J.; et al. DNASU plasmid and PSI: Biology-Materials repositories: Resources to accelerate biological research. Nucleic Acids Res. 2014, 42, D1253–D1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]


Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Arora, R.; Van Theemsche, K.M.; Van Remoortel, S.; Snyders, D.J.; Labro, A.J.; Timmermans, J.-P. Constitutive, Basal, and β-Alanine-Mediated Activation of the Human Mas-Related G Protein-Coupled Receptor D Induces Release of the Inflammatory Cytokine IL-6 and Is Dependent on NF-κB Signaling. Int. J. Mol. Sci. 2021, 22, 13254. https://doi.org/10.3390/ijms222413254
Arora R, Van Theemsche KM, Van Remoortel S, Snyders DJ, Labro AJ, Timmermans J-P. Constitutive, Basal, and β-Alanine-Mediated Activation of the Human Mas-Related G Protein-Coupled Receptor D Induces Release of the Inflammatory Cytokine IL-6 and Is Dependent on NF-κB Signaling. International Journal of Molecular Sciences. 2021; 22(24):13254. https://doi.org/10.3390/ijms222413254
Chicago/Turabian StyleArora, Rohit, Kenny M. Van Theemsche, Samuel Van Remoortel, Dirk J. Snyders, Alain J. Labro, and Jean-Pierre Timmermans. 2021. "Constitutive, Basal, and β-Alanine-Mediated Activation of the Human Mas-Related G Protein-Coupled Receptor D Induces Release of the Inflammatory Cytokine IL-6 and Is Dependent on NF-κB Signaling" International Journal of Molecular Sciences 22, no. 24: 13254. https://doi.org/10.3390/ijms222413254
APA StyleArora, R., Van Theemsche, K. M., Van Remoortel, S., Snyders, D. J., Labro, A. J., & Timmermans, J.-P. (2021). Constitutive, Basal, and β-Alanine-Mediated Activation of the Human Mas-Related G Protein-Coupled Receptor D Induces Release of the Inflammatory Cytokine IL-6 and Is Dependent on NF-κB Signaling. International Journal of Molecular Sciences, 22(24), 13254. https://doi.org/10.3390/ijms222413254





