CCL4 Stimulates Cell Migration in Human Osteosarcoma via the mir-3927-3p/Integrin αvβ3 Axis
Abstract
1. Introduction
2. Results
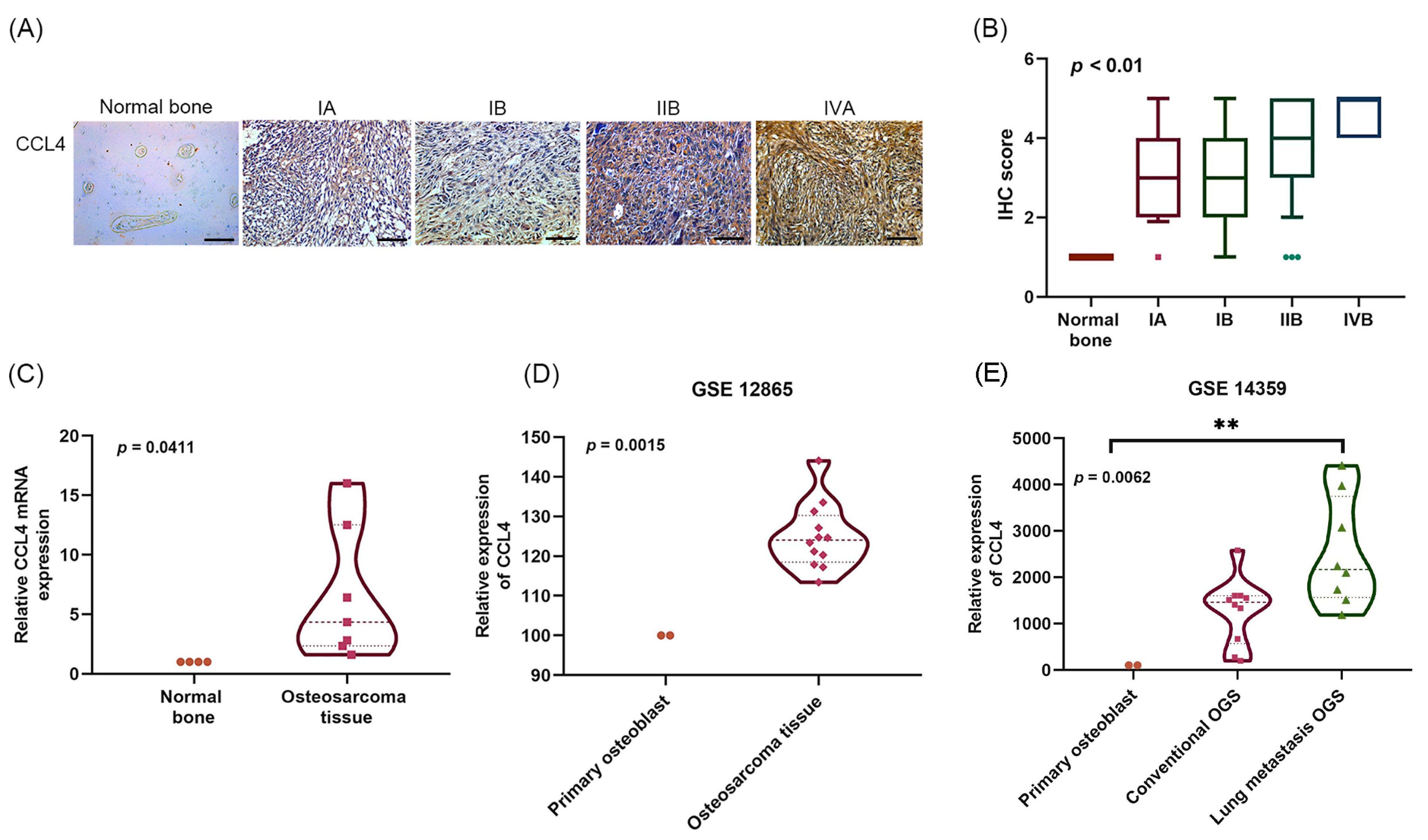
2.1. Clinicopathological Characteristics of CCL4 in Osteosarcoma Tissue
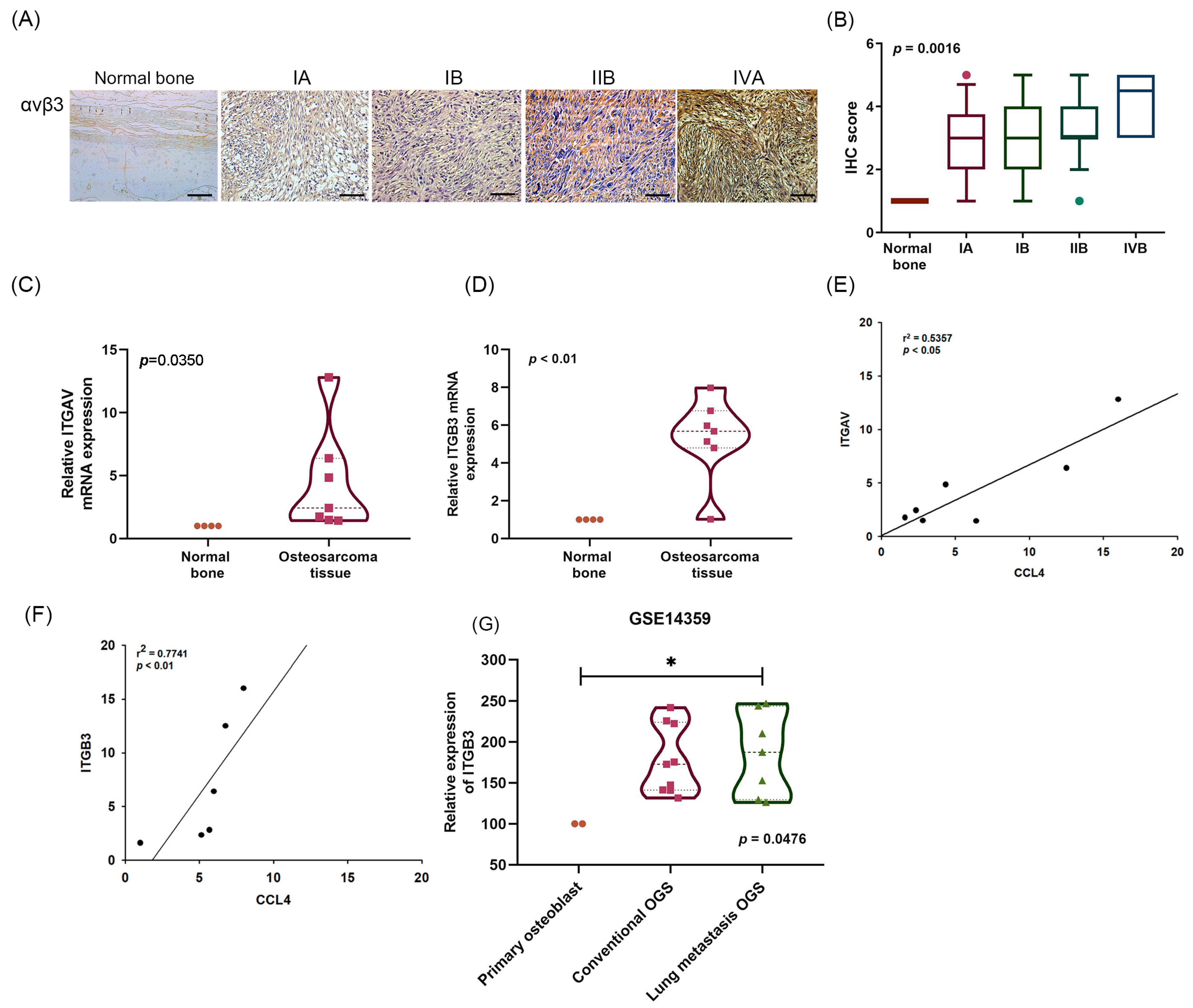
2.2. A Positive Correlation between CCL4 and Integrin αvβ3 Expression in Osteosarcoma Tissue
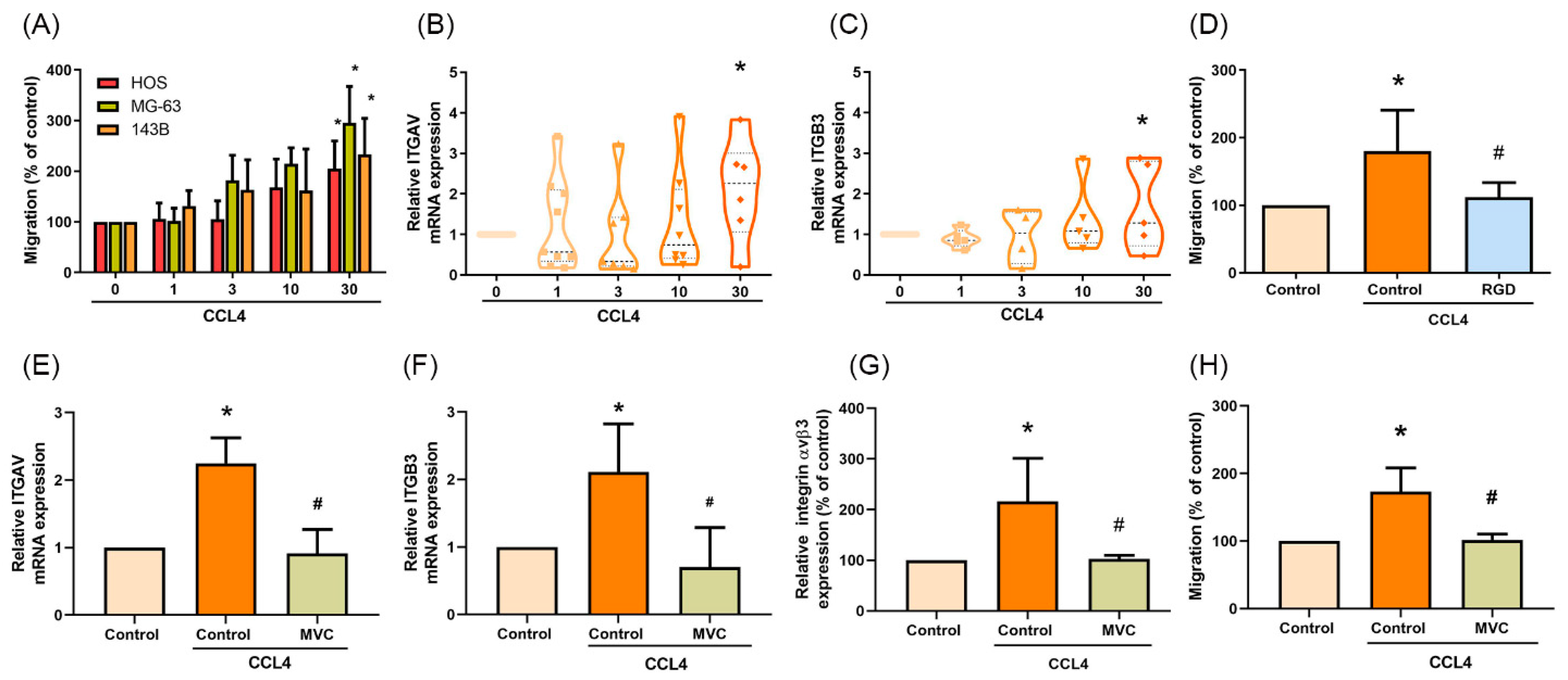
2.3. CCL4 Promotes Integrin αvβ3 Expression and Cell Migration through CCR5
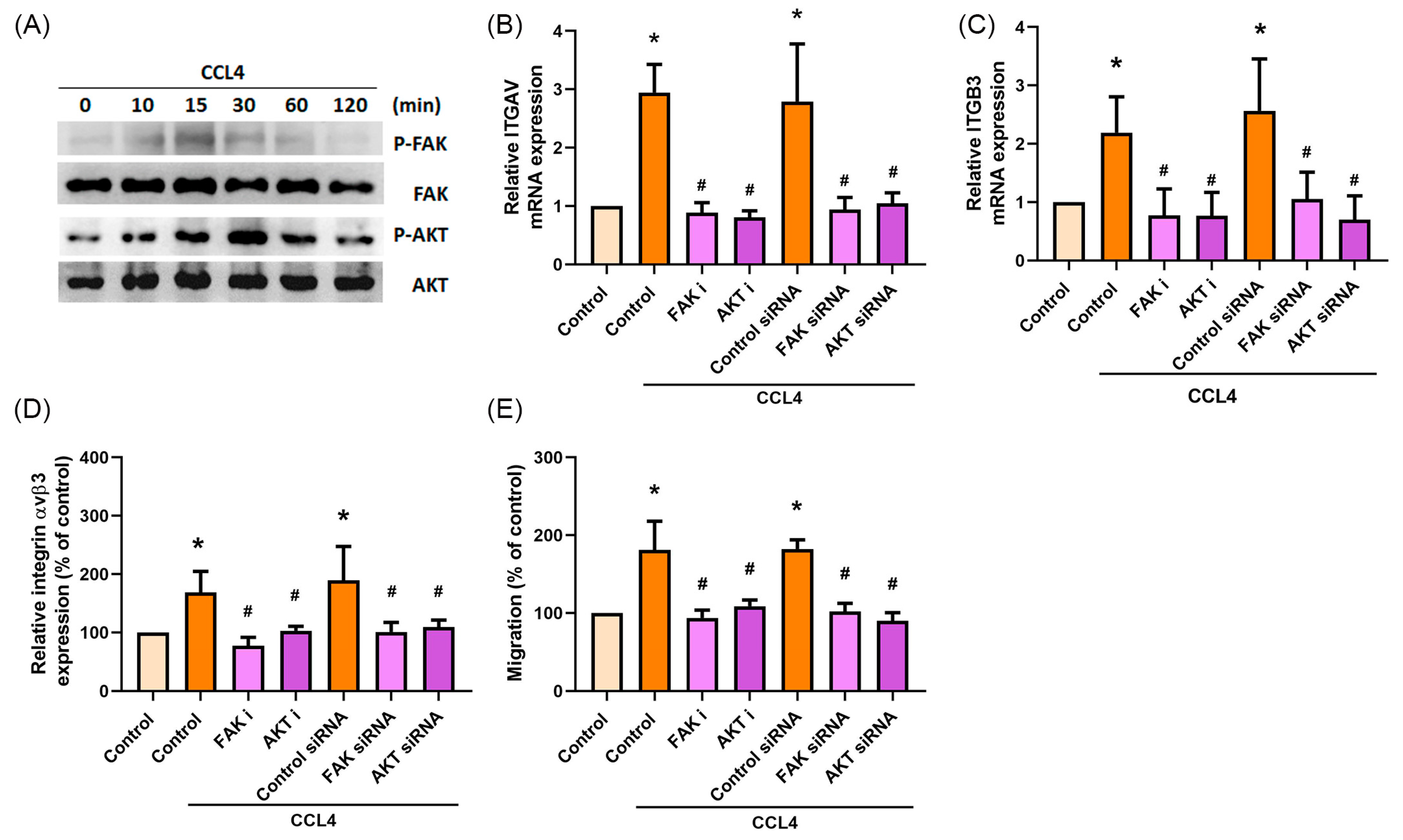
2.4. CCL4 Promotes Cell Migration by Activating FAK and AKT Signaling
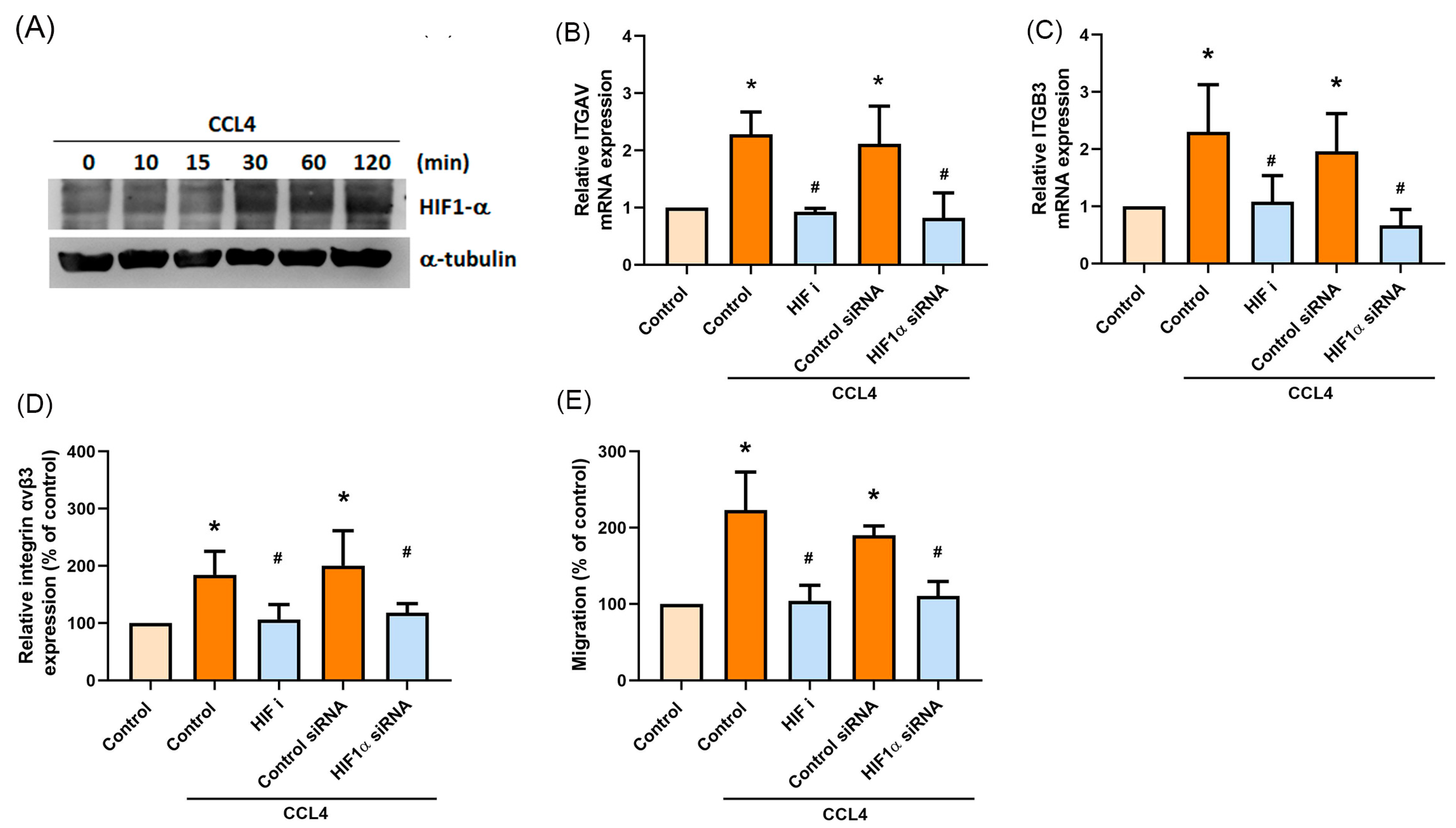
2.5. HIF-1α Is Involved in CCL4-Mediated Expression of Integrin αvβ3 and Migratory Activities of Osteosarcoma Cells
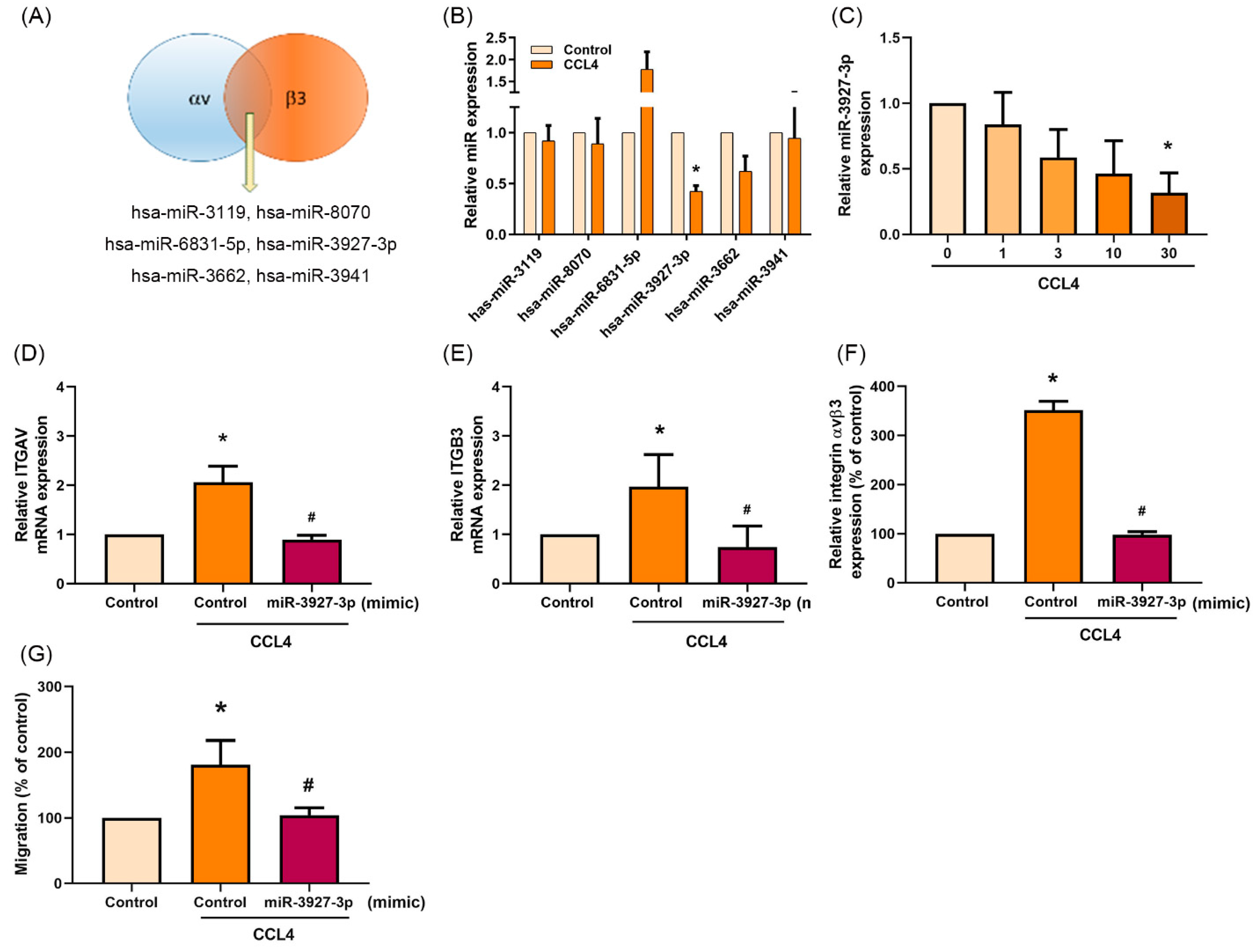
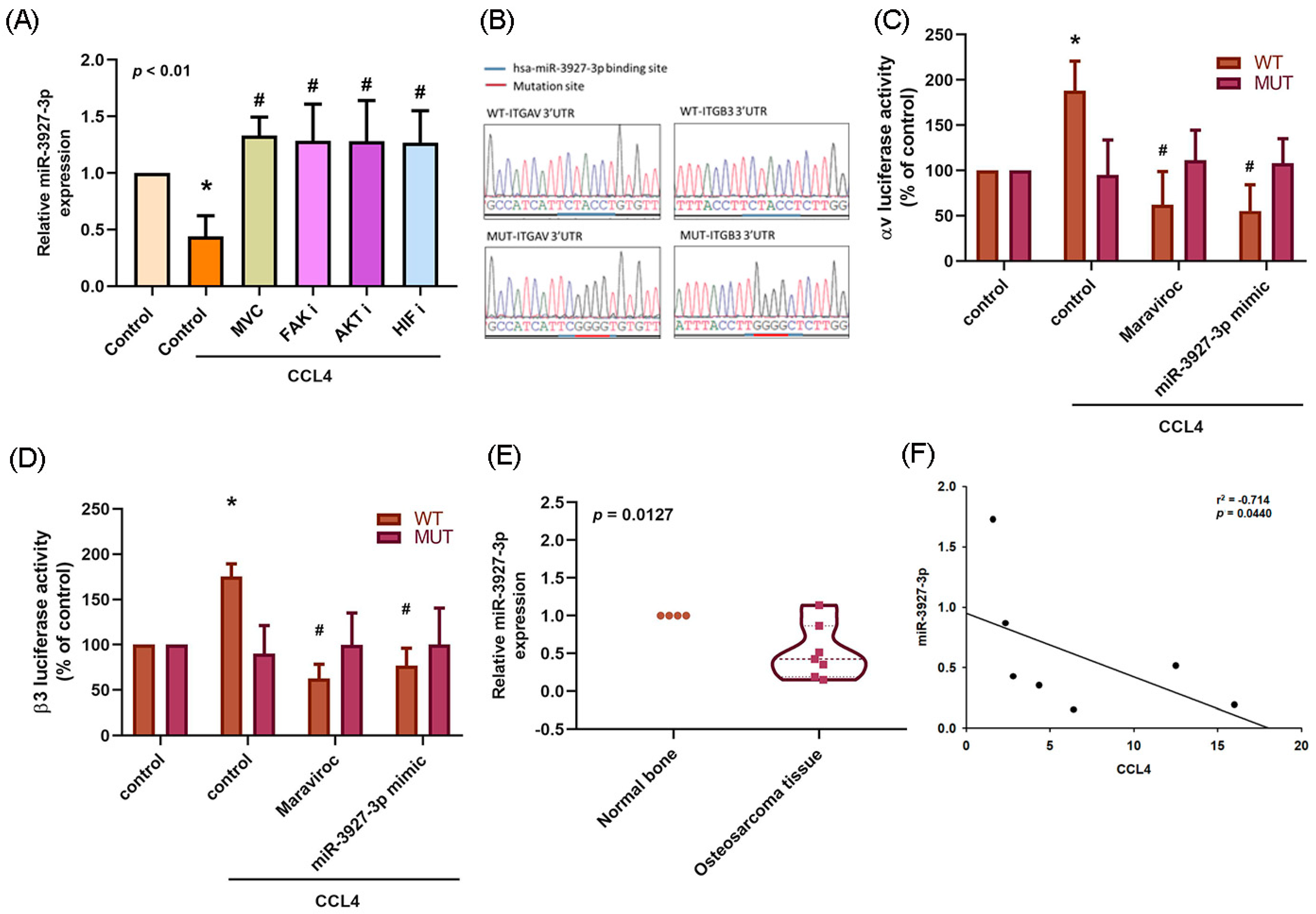
2.6. Downregulation of miR-3927-3p Increases Levels of Integrin αvβ3 Expression and Osteosarcoma Cell Migration
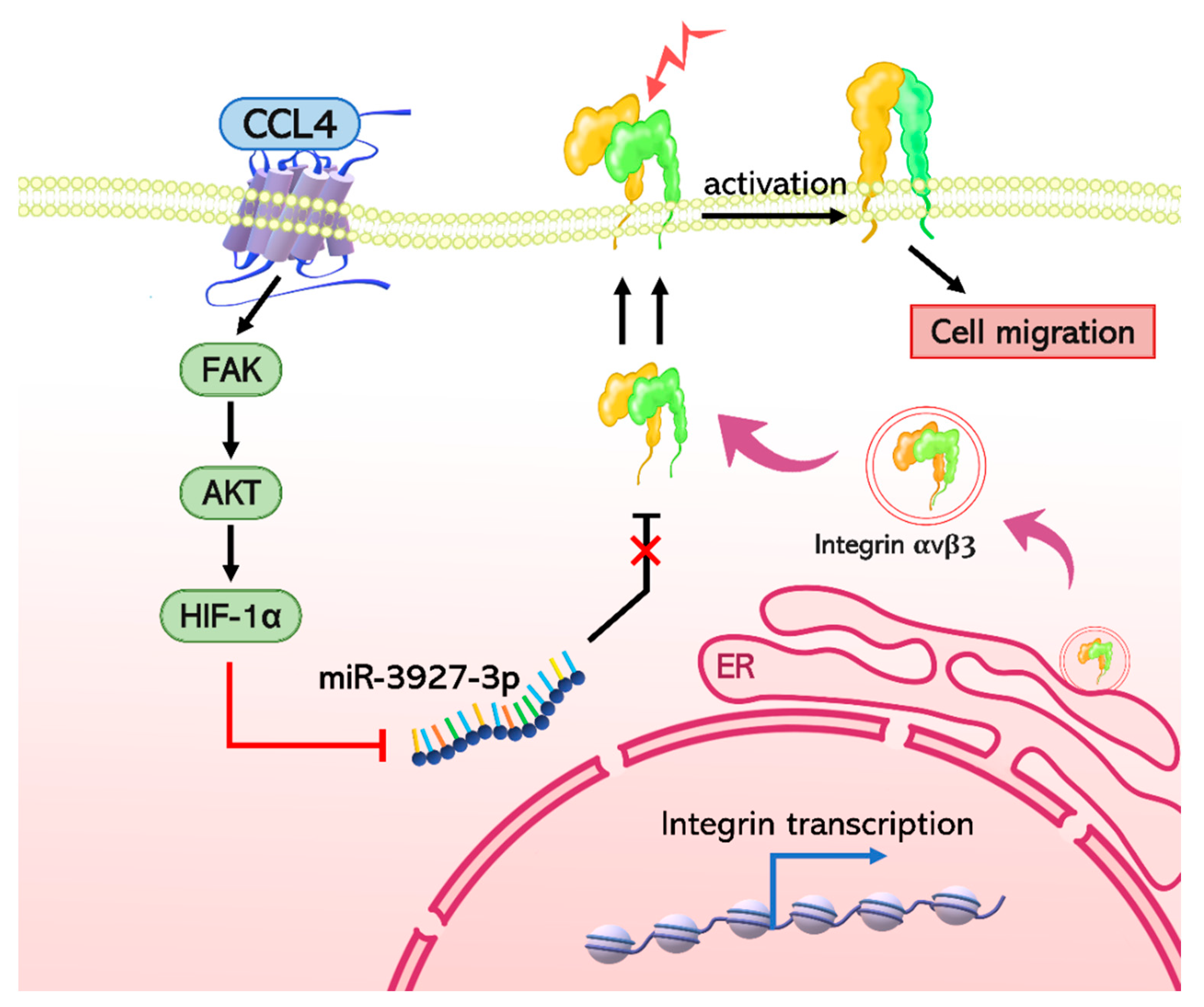
3. Discussion
4. Materials and Methods
4.1. Materials
4.2. Cell Culture
4.3. Immunohistochemistry
4.4. Analysis of Messenger RNA (mRNA) Expression Profiles from the GEO Database
4.5. Migration Assay
4.6. Quantitative Real-Time PCR
4.7. Flow Cytometry
4.8. Western Blot Analysis
4.9. Luciferase Reporter Assay
4.10. Statistics
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Norregaard, K.S.; Jurgensen, H.J.; Gardsvoll, H.; Engelholm, L.H.; Behrendt, N.; Soe, K. Osteosarcoma and Metastasis Associated Bone Degradation-A Tale of Osteoclast and Malignant Cell Cooperativity. Int. J. Mol. Sci. 2021, 22, 6865. [Google Scholar] [CrossRef]
- Taran, S.J.; Taran, R.; Malipatil, N.B. Pediatric Osteosarcoma: An Updated Review. Indian J. Med. Paediatr. Oncol. 2017, 38, 33–43. [Google Scholar] [CrossRef]
- Yu, X.; Yustein, J.T.; Xu, J. Research models and mesenchymal/epithelial plasticity of osteosarcoma. Cell Biosci. 2021, 11, 94. [Google Scholar] [CrossRef]
- Zhao, X.; Wu, Q.; Gong, X.; Liu, J.; Ma, Y. Osteosarcoma: A review of current and future therapeutic approaches. Biomed. Eng. Online 2021, 20, 24. [Google Scholar] [CrossRef]
- Li, M.; Wang, Y.; Li, M.; Wu, X.; Setrerrahmane, S.; Xu, H. Integrins as attractive targets for cancer therapeutics. Acta Pharm. Sin. B 2021, 11, 2726–2737. [Google Scholar] [CrossRef]
- Seguin, L.; Desgrosellier, J.S.; Weis, S.M.; Cheresh, D.A. Integrins and cancer: Regulators of cancer stemness, metastasis, and drug resistance. Trends Cell Biol. 2015, 25, 234–240. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.F.; Chen, P.C.; Chang, T.M.; Hou, C.H. Thrombospondin-2 stimulates MMP-9 production and promotes osteosarcoma metastasis via the PLC, PKC, c-Src and NF-kappaB activation. J. Cell Mol. Med. 2020, 24, 12826–12839. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Luo, Y. LINC01354 Promotes Osteosarcoma Cell Invasion by Up-regulating Integrin beta1. Arch. Med. Res. 2020, 51, 115–123. [Google Scholar] [CrossRef]
- Li, R.; Shi, Y.; Zhao, S.; Shi, T.; Zhang, G. NF-kappaB signaling and integrin-beta1 inhibition attenuates osteosarcoma metastasis via increased cell apoptosis. Int. J. Biol. Macromol. 2019, 123, 1035–1043. [Google Scholar] [CrossRef] [PubMed]
- Yang, J.; Zhang, W. New molecular insights into osteosarcoma targeted therapy. Curr. Opin. Oncol. 2013, 25, 398–406. [Google Scholar] [CrossRef] [PubMed]
- Hou, C.H.; Yang, R.S.; Tsao, Y.T. Connective tissue growth factor stimulates osteosarcoma cell migration and induces osteosarcoma metastasis by upregulating VCAM-1 expression. Biochem. Pharmacol. 2018, 155, 71–81. [Google Scholar] [CrossRef]
- Shi, K.; Wang, S.L.; Shen, B.; Yu, F.Q.; Weng, D.F.; Lin, J.H. Clinicopathological and prognostic values of fibronectin and integrin αvβ3 expression in primary osteosarcoma. World J. Surg. Oncol. 2019, 17, 23. [Google Scholar] [CrossRef]
- Lei, Y.; Junxin, C.; Yongcan, H.; Xiaoguang, L.; Binsheng, Y. Role of microRNAs in the crosstalk between osteosarcoma cells and the tumour microenvironment. J. Bone Oncol. 2020, 25, 100322. [Google Scholar] [CrossRef] [PubMed]
- Cai, W.; Xu, Y.; Zuo, W.; Su, Z. MicroR-542-3p can mediate ILK and further inhibit cell proliferation, migration and invasion in osteosarcoma cells. Aging 2019, 11, 18–32. [Google Scholar] [CrossRef]
- Wang, D.; Tang, L.; Wu, H.; Wang, K.; Gu, D. MiR-127-3p inhibits cell growth and invasiveness by targeting ITGA6 in human osteosarcoma. IUBMB Life 2018, 70, 411–419. [Google Scholar] [CrossRef] [PubMed]
- Luo, Z.; Li, D.; Luo, X.; Li, L.; Gu, S.; Yu, L.; Ma, Y. Decreased Expression of miR-548c-3p in Osteosarcoma Contributes to Cell Proliferation Via Targeting ITGAV. Cancer Biother. Radiopharm. 2016, 31, 153–158. [Google Scholar] [CrossRef]
- Mukaida, N.; Sasaki, S.I.; Baba, T. CCL4 Signaling in the Tumor Microenvironment. Adv. Exp. Med. Biol. 2020, 1231, 23–32. [Google Scholar] [CrossRef]
- Korbecki, J.; Grochans, S.; Gutowska, I.; Barczak, K.; Baranowska-Bosiacka, I. CC Chemokines in a Tumor: A Review of Pro-Cancer and Anti-Cancer Properties of Receptors CCR5, CCR6, CCR7, CCR8, CCR9, and CCR10 Ligands. Int. J. Mol. Sci. 2020, 21, 7619. [Google Scholar] [CrossRef]
- Lee, D.; Shin, K.J.; Kim, D.W.; Yoon, K.A.; Choi, Y.J.; Lee, B.N.R.; Cho, J.Y. CCL4 enhances preosteoclast migration and its receptor CCR5 downregulation by RANKL promotes osteoclastogenesis. Cell Death Dis. 2018, 9, 495. [Google Scholar] [CrossRef] [PubMed]
- Sasaki, S.; Baba, T.; Nishimura, T.; Hayakawa, Y.; Hashimoto, S.; Gotoh, N.; Mukaida, N. Essential roles of the interaction between cancer cell-derived chemokine, CCL4, and intra-bone CCR5-expressing fibroblasts in breast cancer bone metastasis. Cancer Lett. 2016, 378, 23–32. [Google Scholar] [CrossRef] [PubMed]
- Lien, M.Y.; Tsai, H.C.; Chang, A.C.; Tsai, M.H.; Hua, C.H.; Wang, S.W.; Tang, C.H. Chemokine CCL4 Induces Vascular Endothelial Growth Factor C Expression and Lymphangiogenesis by miR-195-3p in Oral Squamous Cell Carcinoma. Front. Immunol. 2018, 9, 412. [Google Scholar] [CrossRef] [PubMed]
- Jiang, K.; Yao, G.; Hu, L.; Yan, Y.; Liu, J.; Shi, J.; Chang, Y.; Zhang, Y.; Liang, D.; Shen, D.; et al. MOB2 suppresses GBM cell migration and invasion via regulation of FAK/Akt and cAMP/PKA signaling. Cell Death Dis. 2020, 11, 230. [Google Scholar] [CrossRef] [PubMed]
- Wu, Y.J.; Lin, S.H.; Din, Z.H.; Su, J.H.; Liu, C.I. Sinulariolide Inhibits Gastric Cancer Cell Migration and Invasion through Downregulation of the EMT Process and Suppression of FAK/PI3K/AKT/mTOR and MAPKs Signaling Pathways. Mar. Drugs 2019, 17, 668. [Google Scholar] [CrossRef]
- Luo, J.; Yao, J.F.; Deng, X.F.; Zheng, X.D.; Jia, M.; Wang, Y.Q.; Huang, Y.; Zhu, J.H. 14, 15-EET induces breast cancer cell EMT and cisplatin resistance by up-regulating integrin alphavbeta3 and activating FAK/PI3K/AKT signaling. J. Exp. Clin. Cancer Res. 2018, 37, 23. [Google Scholar] [CrossRef]
- Bhattarai, D.; Xu, X.; Lee, K. Hypoxia-inducible factor-1 (HIF-1) inhibitors from the last decade (2007 to 2016): A "structure-activity relationship" perspective. Med. Res. Rev. 2018, 38, 1404–1442. [Google Scholar] [CrossRef]
- Hayashi, Y.; Yokota, A.; Harada, H.; Huang, G. Hypoxia/pseudohypoxia-mediated activation of hypoxia-inducible factor-1alpha in cancer. Cancer Sci. 2019, 110, 1510–1517. [Google Scholar] [CrossRef]
- Vangelista, L.; Vento, S. The Expanding Therapeutic Perspective of CCR5 Blockade. Front. Immunol. 2017, 8, 1981. [Google Scholar] [CrossRef]
- Aldinucci, D.; Borghese, C.; Casagrande, N. The CCL5/CCR5 Axis in Cancer Progression. Cancers 2020, 12, 1765. [Google Scholar] [CrossRef] [PubMed]
- Aldinucci, D.; Casagrande, N. Inhibition of the CCL5/CCR5 Axis against the Progression of Gastric Cancer. Int. J. Mol. Sci. 2018, 19, 1477. [Google Scholar] [CrossRef]
- Haag, G.M.; Halama, N.; Springfeld, C.; Grün, B.; Apostolidis, L.; Zschaebitz, S.; Dietrich, M.; Berger, A.-K.; Weber, T.F.; Zoernig, I. Combined PD-1 inhibition (Pembrolizumab) and CCR5 inhibition (Maraviroc) for the treatment of refractory microsatellite stable (MSS) metastatic colorectal cancer (mCRC): First results of the PICCASSO phase I trial. J. Clin. Oncol. 2020, 38, 300. [Google Scholar] [CrossRef]
- Halama, N.; Zoernig, I.; Berthel, A.; Kahlert, C.; Klupp, F.; Suarez-Carmona, M.; Suetterlin, T.; Brand, K.; Krauss, J.; Lasitschka, F.; et al. Tumoral Immune Cell Exploitation in Colorectal Cancer Metastases Can Be Targeted Effectively by Anti-CCR5 Therapy in Cancer Patients. Cancer Cell 2016, 29, 587–601. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Gao, H.; Xu, R.; Wang, H.; Mei, J.; Liu, C. The interplay between HIF-1α and noncoding RNAs in cancer. J. Exp. Clin. Cancer Res. 2020, 39, 27. [Google Scholar] [CrossRef]
- Zhu, S.; He, C.; Deng, S.; Li, X.; Cui, S.; Zeng, Z.; Liu, M.; Zhao, S.; Chen, J.; Jin, Y.; et al. MiR-548an, Transcriptionally Downregulated by HIF1α/HDAC1, Suppresses Tumorigenesis of Pancreatic Cancer by Targeting Vimentin Expression. Mol. Cancer Ther. 2016, 15, 2209–2219. [Google Scholar] [CrossRef] [PubMed]
- Liu, Z.; Wang, Y.; Dou, C.; Xu, M.; Sun, L.; Wang, L.; Yao, B.; Li, Q.; Yang, W.; Tu, K.; et al. Hypoxia-induced up-regulation of VASP promotes invasiveness and metastasis of hepatocellular carcinoma. Theranostics 2018, 8, 4649–4663. [Google Scholar] [CrossRef]
- Johanson, T.M.; Lew, A.M.; Chong, M.M. MicroRNA-independent roles of the RNase III enzymes Drosha and Dicer. Open Biol. 2013, 3, 130144. [Google Scholar] [CrossRef] [PubMed]
- Shen, J.; Xia, W.; Khotskaya, Y.B.; Huo, L.; Nakanishi, K.; Lim, S.O.; Du, Y.; Wang, Y.; Chang, W.C.; Chen, C.H.; et al. EGFR modulates microRNA maturation in response to hypoxia through phosphorylation of AGO2. Nature 2013, 497, 383–387. [Google Scholar] [CrossRef] [PubMed]
- Alday-Parejo, B.; Stupp, R.; Ruegg, C. Are Integrins Still Practicable Targets for Anti-Cancer Therapy? Cancers 2019, 11, 978. [Google Scholar] [CrossRef]
- Valdembri, D.; Serini, G. The roles of integrins in cancer. Fac. Rev. 2021, 10, 45. [Google Scholar] [CrossRef]
- Desgrosellier, J.S.; Cheresh, D.A. Integrins in cancer: Biological implications and therapeutic opportunities. Nat. Rev. Cancer 2010, 10, 9–22. [Google Scholar] [CrossRef]
- Cheng, T.M.; Chang, W.J.; Chu, H.Y.; De Luca, R.; Pedersen, J.Z.; Incerpi, S.; Li, Z.L.; Shih, Y.J.; Lin, H.Y.; Wang, K.; et al. Nano-Strategies Targeting the Integrin alphavbeta3 Network for Cancer Therapy. Cells 2021, 10, 1684. [Google Scholar] [CrossRef]
- Ren, P.; Sun, D.; Xin, D.; Ma, W.; Chen, P.; Gao, H.; Zhang, S.; Gong, M. Serum amyloid A promotes osteosarcoma invasion via upregulating alphavbeta3 integrin. Mol. Med. Rep. 2014, 10, 3106–3112. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Stupp, R.; Hegi, M.E.; Gorlia, T.; Erridge, S.C.; Perry, J.; Hong, Y.K.; Aldape, K.D.; Lhermitte, B.; Pietsch, T.; Grujicic, D.; et al. Cilengitide combined with standard treatment for patients with newly diagnosed glioblastoma with methylated MGMT promoter (CENTRIC EORTC 26071-22072 study): A multicentre, randomised, open-label, phase 3 trial. Lancet Oncol. 2014, 15, 1100–1108. [Google Scholar] [CrossRef]
- Tsai, H.C.; Cheng, S.P.; Han, C.K.; Huang, Y.L.; Wang, S.W.; Lee, J.J.; Lai, C.T.; Fong, Y.C.; Tang, C.H. Resistin enhances angiogenesis in osteosarcoma via the MAPK signaling pathway. Aging 2019, 11, 9767–9777. [Google Scholar] [CrossRef]
- Tsai, H.C.; Chang, A.C.; Tsai, C.H.; Huang, Y.L.; Gan, L.; Chen, C.K.; Liu, S.C.; Huang, T.Y.; Fong, Y.C.; Tang, C.H. CCN2 promotes drug resistance in osteosarcoma by enhancing ABCG2 expression. J. Cell Physiol. 2019, 234, 9297–9307. [Google Scholar] [CrossRef]
- Lee, H.-P.; Liu, S.-C.; Wang, Y.-H.; Chen, B.-C.; Chen, H.-T.; Li, T.-M.; Huang, W.-C.; Hsu, C.-J.; Wu, Y.-C.; Tang, C.-H. Cordycerebroside A suppresses VCAM-dependent monocyte adhesion in osteoarthritis synovial fibroblasts by inhibiting MEK/ERK/AP-1 signaling. J. Funct. Foods 2021, 86, 104712. [Google Scholar] [CrossRef]
- Lee, H.-P.; Chen, P.-C.; Wang, S.-W.; Fong, Y.-C.; Tsai, C.-H.; Tsai, F.-J.; Chung, J.-G.; Huang, C.-Y.; Yang, J.-S.; Hsu, Y.-M.; et al. Plumbagin suppresses endothelial progenitor cell-related angiogenesis in vitro and in vivo. J. Funct. Foods 2019, 52, 537–544. [Google Scholar] [CrossRef]
- Tsai, H.C.; Huang, C.Y.; Su, H.L.; Tang, C.H. CTGF increases drug resistance to paclitaxel by upregulating survivin expression in human osteosarcoma cells. Biochim. Biophys. Acta 2014, 1843, 846–854. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.-C.; Tsai, C.-H.; Wu, T.-Y.; Tsai, C.-H.; Tsai, F.-J.; Chung, J.-G.; Huang, C.-Y.; Yang, J.-S.; Hsu, Y.-M.; Yin, M.-C. Soya-cerebroside reduces IL-1β-induced MMP-1 production in chondrocytes and inhibits cartilage degradation: Implications for the treatment of osteoarthritis. Food Agric. Immunol. 2019, 30, 620–632. [Google Scholar] [CrossRef]
- Lee, H.-P.; Wu, Y.-C.; Chen, B.-C.; Liu, S.-C.; Li, T.-M.; Huang, W.-C.; Hsu, C.-J.; Tang, C.-H. Soya-cerebroside reduces interleukin production in human rheumatoid arthritis synovial fibroblasts by inhibiting the ERK, NF-κB and AP-1 signalling pathways. Food Agric. Immunol. 2020, 31, 740–750. [Google Scholar] [CrossRef]
- Lee, H.-P.; Wang, S.-W.; Wu, Y.-C.; Lin, L.-W.; Tsai, F.-J.; Yang, J.-S.; Li, T.-M.; Tang, C.-H. Soya-cerebroside inhibits VEGF-facilitated angiogenesis in endothelial progenitor cells. Food Agric. Immunol. 2020, 31, 193–204. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tsai, H.-C.; Lai, Y.-Y.; Hsu, H.-C.; Fong, Y.-C.; Lien, M.-Y.; Tang, C.-H. CCL4 Stimulates Cell Migration in Human Osteosarcoma via the mir-3927-3p/Integrin αvβ3 Axis. Int. J. Mol. Sci. 2021, 22, 12737. https://doi.org/10.3390/ijms222312737
Tsai H-C, Lai Y-Y, Hsu H-C, Fong Y-C, Lien M-Y, Tang C-H. CCL4 Stimulates Cell Migration in Human Osteosarcoma via the mir-3927-3p/Integrin αvβ3 Axis. International Journal of Molecular Sciences. 2021; 22(23):12737. https://doi.org/10.3390/ijms222312737
Chicago/Turabian StyleTsai, Hsiao-Chi, Yan-You Lai, Hsuan-Chih Hsu, Yi-Chin Fong, Ming-Yu Lien, and Chih-Hsin Tang. 2021. "CCL4 Stimulates Cell Migration in Human Osteosarcoma via the mir-3927-3p/Integrin αvβ3 Axis" International Journal of Molecular Sciences 22, no. 23: 12737. https://doi.org/10.3390/ijms222312737
APA StyleTsai, H.-C., Lai, Y.-Y., Hsu, H.-C., Fong, Y.-C., Lien, M.-Y., & Tang, C.-H. (2021). CCL4 Stimulates Cell Migration in Human Osteosarcoma via the mir-3927-3p/Integrin αvβ3 Axis. International Journal of Molecular Sciences, 22(23), 12737. https://doi.org/10.3390/ijms222312737





