Long-Term Effects of Ionizing Radiation on the Hippocampus: Linking Effects of the Sonic Hedgehog Pathway Activation with Radiation Response
Abstract
1. Introduction
2. Results
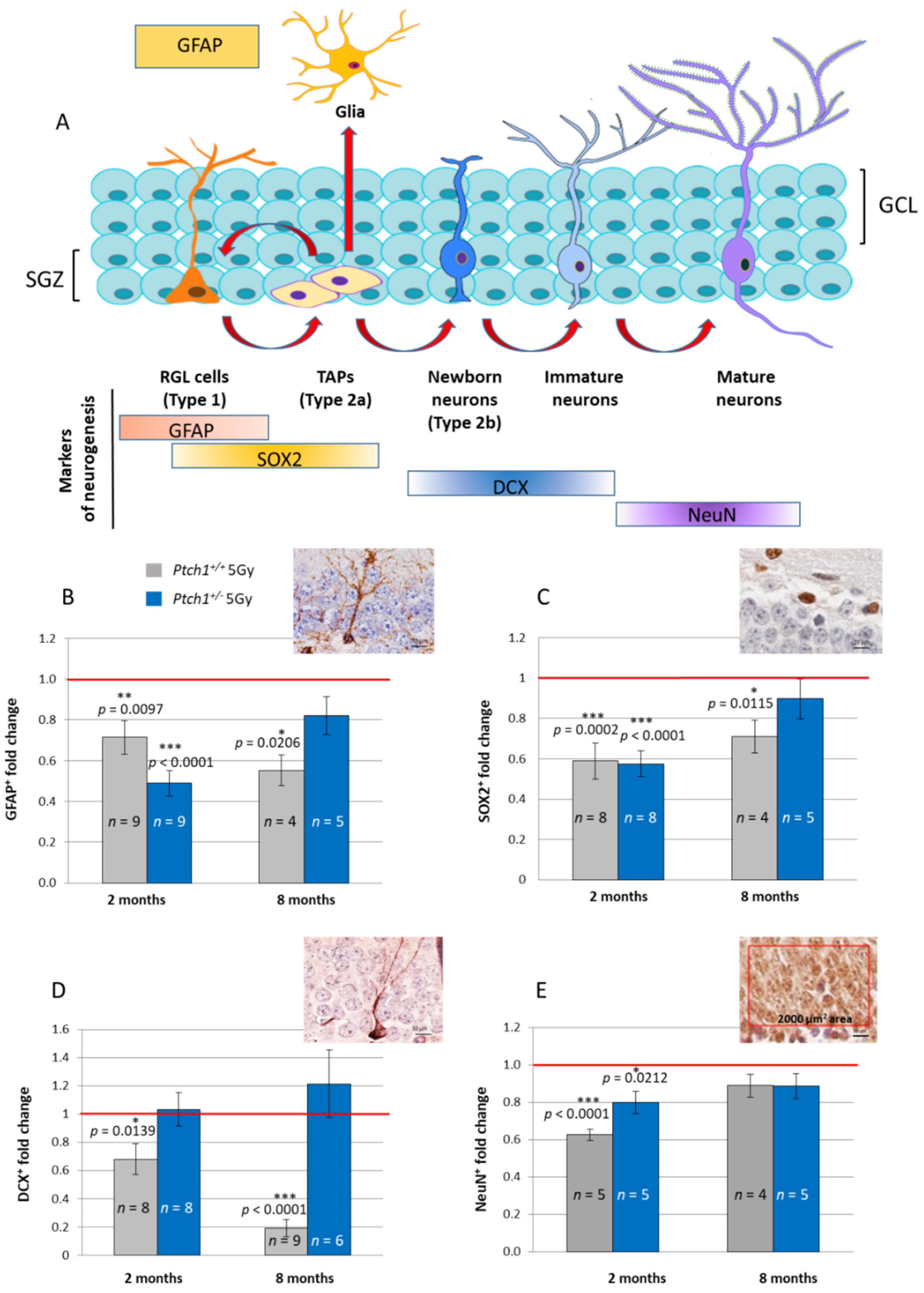
2.1. Constitutive Activation of the Shh Signaling Pathway Mitigates Radiation-Induced Inhibition of Neurogenesis in the Hippocampus
2.2. Constitutive Shh Pathway Activation Protects the Hippocampus from Radiation-Induced Astrogliosis
2.3. Long-Term Effect of Constitutive Shh Pathway Activation on the Expression of Neurogenesis-Related Genes after Irradiation
2.4. Effect of Constitutive Shh Pathway Activation on Radiation-Induced Changes in Mouse Behaviour
3. Discussion
4. Materials and Methods
4.1. Animals and Irradiation
4.2. Immunohistochemistry
4.3. RT2 Profiler PCR Array
4.4. Cognitive Tests
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| Ptch1 | Patched |
| DG | Dentate Gyrus |
| Shh | Sonic Hedgehog |
| SMO | Smoothened |
| SVZ | Subventricular zone |
| SGZ | Subgranular zone |
| GCL | Granular cell layer |
| WT | Wild Type |
| CNS | Central nervous system |
| RGLs | Radial glia-like cells |
| TAPs | Transit amplifying neural stem cells |
| DCX | Doublecortin |
| FC | Fold-changes |
| GFAP | Glial fibrillary acidic protein |
| NSC | Neural stem cells |
| SOX2 | Sex determining region Y (SRY) box 2 |
| OF | Open field test |
| EPM | Elevated plus-maze test |
| NSPC | Neural stem precursor cell |
| COUP-TFs | Chicken ovalbumin upstream promoter transcription factors |
References
- Ahles, T.A.; Root, J.C.; Ryan, E.L. Cancer- and Cancer Treatment–Associated Cognitive Change: An Update on the State of the Science. J. Clin. Oncol. 2012, 30, 3675–3686. [Google Scholar] [CrossRef]
- DeSantis, C.E.; Lin, C.C.; Mariotto, A.B.; Siegel, R.L.; Stein, K.D.; Kramer, J.L.; Alteri, R.; Robbins, A.S.; Jemal, A. Cancer treatment and survivorship statistics, 2014. CA A Cancer J. Clin. 2014, 64, 252–271. [Google Scholar] [CrossRef] [PubMed]
- Perry, A.; Schmidt, R.E. Cancer therapy-associated CNS neuropathology: An update and review of the literature. Acta Neuropathol. 2006, 111, 197–212. [Google Scholar] [CrossRef] [PubMed]
- Smart, D. Radiation Toxicity in the Central Nervous System: Mechanisms and Strategies for Injury Reduction. Semin. Radiat. Oncol. 2017, 27, 332–339. [Google Scholar] [CrossRef] [PubMed]
- Redmond, K.J.; Milano, M.T.; Kim, M.M.; Trifiletti, D.M.; Soltys, S.G.; Hattangadi-Gluth, J.A. Reducing Radiation-Induced Cognitive Toxicity: Sparing the Hippocampus and Beyond. Int. J. Radiat. Oncol. Biol. Phys. 2021, 109, 1131–1136. [Google Scholar] [CrossRef] [PubMed]
- Son, Y.; Yang, M.; Wang, H.; Moon, C. Hippocampal dysfunctions caused by cranial irradiation: A review of the experimental evidence. Brain Behav. Immun. 2015, 45, 287–296. [Google Scholar] [CrossRef]
- Goldman, S.A.; Nottebohm, F. Neuronal production, migration, and differentiation in a vocal control nucleus of the adult female canary brain. Proc. Natl. Acad. Sci. USA 1983, 80, 2390–2394. [Google Scholar] [CrossRef]
- Altman, J.; Das, G.D. Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats. J. Comp. Neurol. 1965, 124, 319–335. [Google Scholar] [CrossRef]
- Van Praag, H.; Christie, B.R.; Sejnowski, T.J.; Gage, F.H. Running enhances neurogenesis, learning, and long-term potentiation in mice. Proc. Natl. Acad. Sci. USA 1999, 96, 13427–13431. [Google Scholar] [CrossRef]
- Eriksson, P.S.; Perfilieva, E.; Bjork-Eriksson, T.; Alborn, A.-M.; Nordborg, C.; Peterson, D.A.; Gage, F.H. Neurogenesis in the adult human hippocampus. Nat. Med. 1998, 4, 1313–1317. [Google Scholar] [CrossRef]
- Spalding, K.L.; Bergmann, O.; Alkass, K.; Bernard, S.; Salehpour, M.; Huttner, H.B.; Boström, E.; Westerlund, I.; Vial, C.; Buchholz, B.A.; et al. Dynamics of Hippocampal Neurogenesis in Adult Humans. Cell 2013, 153, 1219–1227. [Google Scholar] [CrossRef] [PubMed]
- Tanapat, P.; Hastings, N.B.; Reeves, A.J.; Gould, E. Estrogen Stimulates a Transient Increase in the Number of New Neurons in the Dentate Gyrus of the Adult Female Rat. J. Neurosci. 1999, 19, 5792–5801. [Google Scholar] [CrossRef] [PubMed]
- Gould, E.; Tanapat, P.; Hastings, N.B.; Shors, T.J. Neurogenesis in adulthood: A possible role in learning. Trends Cogn. Sci. 1999, 3, 186–192. [Google Scholar] [CrossRef]
- Brown, J.; Cooper-Kuhn, C.M.; Kempermann, G.; van Praag, H.; Winkler, J.; Gage, F.H.; Kuhn, H.G. Enriched environment and physical activity stimulate hippocampal but not olfactory bulb neurogenesis. Eur. J. Neurosci. 2003, 17, 2042–2046. [Google Scholar] [CrossRef]
- Murphy, T.; Dias, G.P.; Thuret, S. Effects of Diet on Brain Plasticity in Animal and Human Studies: Mind the Gap. Neural Plast. 2014, 2014, 563160. [Google Scholar] [CrossRef] [PubMed]
- Kuhn, H.-G.; Dickinson-Anson, H.; Gage, F.H. Neurogenesis in the dentate gyrus of the adult rat: Age-related decrease of neuronal progenitor proliferation. J. Neurosci. 1996, 16, 2027–2033. [Google Scholar] [CrossRef]
- Haldbo-Classen, L.; Amidi, A.; Lukacova, S.; Wu, L.M.; von Oettingen, G.; Lassen-Ramshad, Y.; Zachariae, R.; Kallehauge, J.F.; Høyer, M. Cognitive impairment following radiation to hippocampus and other brain structures in adults with primary brain tumours. Radiother. Oncol. 2020, 148, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Makale, M.T.; McDonald, C.R.; Hattangadi-Gluth, J.A.; Kesari, S. Mechanisms of radiotherapy-associated cognitive disability in patients with brain tumours. Nat. Rev. Neurol. 2017, 13, 52–64. [Google Scholar] [CrossRef] [PubMed]
- Pazzaglia, S.; Briganti, G.; Mancuso, M.; Saran, A. Neurocognitive Decline Following Radiotherapy: Mechanisms and Therapeutic Implications. Cancers 2020, 12, 146. [Google Scholar] [CrossRef]
- Pereira Dias, G.; Hollywood, R.; Bevilaqua, M.C.; da Luz, A.C.; Hindges, R.; Nardi, A.E.; Thuret, S. Consequences of cancer treatments on adult hippocampal neurogenesis: Implications for cognitive function and depressive symptoms. Neuro-Oncology 2014, 16, 476–492. [Google Scholar] [CrossRef]
- Gondi, V.; Tomé, W.; Mehta, M. Why avoid the hippocampus? A comprehensive review. Radiother. Oncol. 2010, 97, 370–376. [Google Scholar] [CrossRef] [PubMed]
- Carballo, G.B.; Honorato, J.R.; De Lopes, G.P.F.; Spohr, T. A highlight on Sonic hedgehog pathway. Cell Commun. Signal. 2018, 16, 11. [Google Scholar] [CrossRef] [PubMed]
- Ihrie, R.A.; Alvarez-Buylla, A. Cells in the astroglial lineage are neural stem cells. Cell Tissue Res. 2008, 331, 179–191. [Google Scholar] [CrossRef] [PubMed]
- Zhao, C.; Deng, W.; Gage, F.H. Mechanisms and Functional Implications of Adult Neurogenesis. Cell 2008, 132, 645–660. [Google Scholar] [CrossRef]
- Machold, R.; Hayashi, S.; Rutlin, M.; Muzumdar, M.D.; Nery, S.; Corbin, J.G.; Gritli-Linde, A.; Dellovade, T.; Porter, J.A.; Rubin, L.L.; et al. Sonic Hedgehog Is Required for Progenitor Cell Maintenance in Telencephalic Stem Cell Niches. Neuron 2003, 39, 937–950. [Google Scholar] [CrossRef]
- Breunig, J.J.; Sarkisian, M.R.; Arellano, J.I.; Morozov, Y.M.; Ayoub, A.E.; Sojitra, S.; Wang, B.; Flavell, R.A.; Rakic, P.; Town, T. Primary cilia regulate hippocampal neurogenesis by mediating sonic hedgehog signaling. Proc. Natl. Acad. Sci. USA 2008, 105, 13127–13132. [Google Scholar] [CrossRef]
- Han, Y.-G.; Spassky, N.; Romaguera-Ros, M.; Garcia-Verdugo, J.M.; Aguilar, A.; Schneider-Maunoury, S.; Alvarez-Buylla, A. Hedgehog signaling and primary cilia are required for the formation of adult neural stem cells. Nat. Neurosci. 2008, 11, 277–284. [Google Scholar] [CrossRef]
- Hahn, H.; Wojnowski, L.; Zimmer, A.M.; Hall, J.; Miller, G.; Zimmer, A. Rhabdomyosarcomas and radiation hypersensitivity in a mouse model of Gorlin syndrome. Nat. Med. 1998, 4, 619–622. [Google Scholar] [CrossRef]
- Antonelli, F.; Casciati, A.; Tanori, M.; Tanno, B.; Linares, V.; Serra, N.; Belles, M.; Pannicelli, A.; Saran, A.; Pazzaglia, S. Alterations in Morphology and Adult Neurogenesis in the Dentate Gyrus of Patched1 Heterozygous Mice. Front. Mol. Neurosci. 2018, 11, 168. [Google Scholar] [CrossRef]
- Gajjar, A.J.; Robinson, G.W. Medulloblastoma—Translating discoveries from the bench to the bedside. Nat. Rev. Clin. Oncol. 2014, 11, 714–722. [Google Scholar] [CrossRef]
- Gupta, S.; Takebe, N.; Lorusso, P. Targeting the Hedgehog pathway in cancer. Ther. Adv. Med. Oncol. 2010, 2, 237–250. [Google Scholar] [CrossRef] [PubMed]
- Laukkanen, M.O.; Castellone, M.D. Hijacking the Hedgehog Pathway in Cancer Therapy. Anti-Cancer Agents Med. Chem. 2016, 16, 309–317. [Google Scholar] [CrossRef] [PubMed]
- Patel, S.S.; Tomar, S.; Sharma, D.; Mahindroo, N.; Udayabanu, M. Targeting sonic hedgehog signaling in neurological disorders. Neurosci. Biobehav. Rev. 2017, 74, 76–97. [Google Scholar] [CrossRef]
- Toda, T.; Parylak, S.L.; Linker, S.B.; Gage, F.H. The role of adult hippocampal neurogenesis in brain health and disease. Mol. Psychiatry 2019, 24, 67–87. [Google Scholar] [CrossRef] [PubMed]
- Laouarem, Y.; Traiffort, E. Developmental and Repairing Production of Myelin: The Role of Hedgehog Signaling. Front. Cell. Neurosci. 2018, 12, 305. [Google Scholar] [CrossRef] [PubMed]
- Liu, L.; Zhao, B.; Xiong, X.; Xia, Z. The Neuroprotective Roles of Sonic Hedgehog Signaling Pathway in Ischemic Stroke. Neurochem. Res. 2018, 43, 2199–2211. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, V.; Sabeur, K.; Maltepe, E.; Ameri, K.; Bayraktar, O.; Rowitch, D.H. Sonic Hedgehog Agonist Protects Against Complex Neonatal Cerebellar Injury. Cerebellum 2018, 17, 213–227. [Google Scholar] [CrossRef]
- Jinno, S. Regional and laminar differences in antigen profiles and spatial distributions of astrocytes in the mouse hippocampus, with reference to aging. Neuroscience 2011, 180, 41–52. [Google Scholar] [CrossRef]
- Sofroniew, M.V.; Vinters, H.V. Astrocytes: Biology and pathology. Acta Neuropathol. 2010, 119, 7–35. [Google Scholar] [CrossRef]
- Anand, K.S.; Dhikav, V. Hippocampus in health and disease: An overview. Ann. Indian Acad. Neurol. 2012, 15, 239–246. [Google Scholar] [CrossRef]
- Lever, C.; Burton, S.; O’Keefe, J. Rearing on Hind Legs, Environmental Novelty, and the Hippocampal Formation. Rev. Neurosci. 2006, 17, 111–133. [Google Scholar] [CrossRef]
- Askins, M.A.; Moore, B.D., 3rd. Preventing Neurocognitive Late Effects in Childhood Cancer Survivors. J. Child Neurol. 2008, 23, 1160–1171. [Google Scholar] [CrossRef] [PubMed]
- Cramer, C.K.; Cummings, T.L.; Andrews, R.N.; Strowd, R.; Rapp, S.R.; Shaw, E.G.; Chan, M.D.; Lesser, G.J. Treatment of Radiation-Induced Cognitive Decline in Adult Brain Tumor Patients. Curr. Treat. Options Oncol. 2019, 20, 42. [Google Scholar] [CrossRef] [PubMed]
- Constanzo, J.; Midavaine, E.; Fouquet, J.; Lepage, M.; Descoteaux, M.; Kirby, K.; Tremblay, L.; Masson-Cote, L.; Geha, S.; Longpre, J.M.; et al. Brain irradiation leads to persistent neuroinflammation and long-term neurocognitive dysfunction in a region-specific manner. Prog. Neuropsychopharmacol. Biol. Psychiatry 2020, 102, 109954. [Google Scholar] [CrossRef] [PubMed]
- Turnquist, C.; Harris, B.T.; Harris, C.C. Radiation-induced brain injury: Current concepts and therapeutic strategies targeting neuroinflammation. Neuro-Oncol. Adv. 2020, 2, vdaa057. [Google Scholar] [CrossRef] [PubMed]
- Iyer, N.G.; Chin, S.F.; Ozdag, H.; Daigo, Y.; Hu, D.E.; Cariati, M.; Brindle, K.; Aparicio, S.; Caldas, C. p300 regulates p53-dependent apoptosis after DNA damage in colorectal cancer cells by modulation of PUMA/p21 levels. Proc. Natl. Acad. Sci. USA 2004, 101, 7386–7391. [Google Scholar] [CrossRef]
- Yuan, Z.M.; Huang, Y.; Ishiko, T.; Nakada, S.; Utsugisawa, T.; Shioya, H.; Utsugisawa, Y.; Yokoyama, K.; Weichselbaum, R.; Shi, Y.; et al. Role for p300 in Stabilization of p53 in the Response to DNA Damage. J. Biol. Chem. 1999, 274, 1883–1886. [Google Scholar] [CrossRef]
- Al-Griw, M.A.; Alghazeer, R.O.; Awayn, N.; Shamlan, G.; Eskandrani, A.A.; Alnajeebi, A.M.; Babteen, N.A.; Alansari, W.S. Selective adenosine A2A receptor inhibitor SCH58261 reduces oligodendrocyte loss upon brain injury in young rats. Saudi J. Biol. Sci. 2021, 28, 310–316. [Google Scholar] [CrossRef]
- Huang, F.; Lan, Y.; Qin, L.; Dong, H.; Shi, H.; Wu, H.; Zou, Q.; Hu, Z.; Wu, X. Astragaloside IV Promotes Adult Neurogenesis in Hippocampal Dentate Gyrus of Mouse through CXCL1/CXCR2 Signaling. Molecules 2018, 23, 2178. [Google Scholar] [CrossRef]
- Weiss, J.B.; Xue, C.; Benice, T.; Xue, L.; Morris, S.W.; Raber, J. Anaplastic Lymphoma Kinase and Leukocyte Tyrosine Kinase: Functions and genetic interactions in learning, memory and adult neurogenesis. Pharmacol. Biochem. Behav. 2012, 100, 566–574. [Google Scholar] [CrossRef]
- Zhao, X.; Rouhiainen, A.; Li, Z.; Guo, S.; Rauvala, H. Regulation of Neurogenesis in Mouse Brain by HMGB1. Cells 2020, 9, 1714. [Google Scholar] [CrossRef]
- Gong, Q.; Chen, H.; Farbman, A.I. Olfactory sensory axon growth and branching is influenced by sonic hedgehog. Dev. Dyn. 2009, 238, 1768–1776. [Google Scholar] [CrossRef] [PubMed]
- Parra, L.M.; Zou, Y. Sonic hedgehog induces response of commissural axons to Semaphorin repulsion during midline crossing. Nat. Neurosci. 2010, 13, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Chen, S.-D.; Yang, J.-L.; Hwang, W.-C.; Yang, D.-I. Emerging Roles of Sonic Hedgehog in Adult Neurological Diseases: Neurogenesis and Beyond. Int. J. Mol. Sci. 2018, 19, 2423. [Google Scholar] [CrossRef] [PubMed]
- De Stefano, I.; Tanno, B.; Giardullo, P.; Leonardi, S.; Pasquali, E.; Antonelli, F.; Tanori, M.; Casciati, A.; Pazzaglia, S.; Saran, A.; et al. The Patched 1 Tumor-Suppressor Gene Protects the Mouse Lens from Spontaneous and Radiation-Induced Cataract. Am. J. Pathol. 2015, 185, 85–95. [Google Scholar] [CrossRef]
- Madhavan, L.; Daley, B.F.; Davidson, B.L.; Boudreau, R.L.; Lipton, J.W.; Cole-Strauss, A.; Steece-Collier, K.; Collier, T.J. Sonic Hedgehog Controls the Phenotypic Fate and Therapeutic Efficacy of Grafted Neural Precursor Cells in a Model of Nigrostriatal Neurodegeneration. PLoS ONE 2015, 10, e0137136. [Google Scholar] [CrossRef]
- Zhang, Y.; Dong, W.; Guo, S.; Zhao, S.; He, S.; Zhang, L.; Tang, Y.; Wang, H. Lentivirus-mediated delivery of sonic hedgehog into the striatum stimulates neuroregeneration in a rat model of Parkinson disease. Neurol. Sci. 2014, 35, 1931–1940. [Google Scholar] [CrossRef]
- Hai, B.; Zhao, Q.; Qin, L.; Rangaraj, D.; Gutti, V.R.; Liu, F. Rescue Effects and Underlying Mechanisms of Intragland Shh Gene Delivery on Irradiation-Induced Hyposalivation. Hum. Gene Ther. 2016, 27, 390–399. [Google Scholar] [CrossRef] [PubMed]
- Kiyota, T.; Ingraham, K.L.; Jacobsen, M.T.; Xiong, H.; Ikezu, T. FGF2 gene transfer restores hippocampal functions in mouse models of Alzheimer’s disease and has therapeutic implications for neurocognitive disorders. Proc. Natl. Acad. Sci. USA 2011, 108, E1339–E1348. [Google Scholar] [CrossRef] [PubMed]
- Sousa, V.H.; Fishell, G. Sonic hedgehog functions through dynamic changes in temporal competence in the developing forebrain. Curr. Opin. Genet. Dev. 2010, 20, 391–399. [Google Scholar] [CrossRef]
- Mahar, I.; MacIsaac, A.; Kim, J.J.; Qiang, C.; Davoli, M.A.; Turecki, G.; Mechawar, N. Effects of neuregulin-1 administration on neurogenesis in the adult mouse hippocampus and characterization of immature neurons along the septotemporal axis. Sci. Rep. 2016, 6, 30467. [Google Scholar] [CrossRef] [PubMed]
- Yoo, J.Y.; Kim, H.B.; Yoo, S.Y.; Yoo, H.I.; Song, D.Y.; Baik, T.K.; Lee, J.H.; Woo, R.S. Neuregulin 1/ErbB4 signaling attenuates neuronal cell damage under oxygen-glucose deprivation in primary hippocampal neurons. Anat. Cell Biol. 2019, 52, 462–468. [Google Scholar] [CrossRef] [PubMed]
- Goldshmit, Y.; Erlich, S.; Pinkas-Kramarski, R. Neuregulin Rescues PC12-ErbB4 Cells from Cell Death Induced by H2O2. J. Biol. Chem. 2001, 276, 46379–46385. [Google Scholar] [CrossRef]
- Deakin, I.H.; Nissen, W.; Law, A.J.; Lane, T.; Kanso, R.; Schwab, M.H.; Nave, K.A.; Lamsa, K.P.; Paulsen, O.; Bannerman, D.M.; et al. Transgenic Overexpression of the Type I Isoform of Neuregulin 1 Affects Working Memory and Hippocampal Oscillations but not Long-term Potentiation. Cereb. Cortex 2012, 22, 1520–1529. [Google Scholar] [CrossRef]
- Gosso, F.M.; De Geus, E.J.; Polderman, T.J.; Boomsma, D.I.; Posthuma, D.; Heutink, P. Exploring the functional role of the CHRM2 gene in human cognition: Results from a dense genotyping and brain expression study. BMC Med. Genet. 2007, 8, 66. [Google Scholar] [CrossRef] [PubMed]
- Sudarov, A.; Zhang, X.J.; Braunstein, L.; LoCastro, E.; Singh, S.; Taniguchi, Y.; Raj, A.; Shi, S.-H.; Moore, H.; Ross, M.E. Mature Hippocampal Neurons Require LIS1 for Synaptic Integrity: Implications for Cognition. Biol. Psychiatry 2018, 83, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Baraban, S.C. Granule Cell Dispersion and Aberrant Neurogenesis in the Adult Hippocampus of an LIS1 Mutant Mouse. Dev. Neurosci. 2007, 29, 91–98. [Google Scholar] [CrossRef]
- Boyd, P.J.; Cunliffe, V.T.; Roy, S.; Wood, J.D. Sonic hedgehog functions upstream of disrupted-in-schizophrenia 1 (disc1): Implications for mental illness. Biol. Open 2015, 4, 1336–1343. [Google Scholar] [CrossRef]
- Araque, A.; Carmignoto, G.; Haydon, P.G.; Oliet, S.H.; Robitaille, R.; Volterra, A. Gliotransmitters Travel in Time and Space. Neuron 2014, 81, 728–739. [Google Scholar] [CrossRef]
- Bushong, E.A.; Martone, M.E.; Jones, Y.Z.; Ellisman, M.H. Protoplasmic Astrocytes in CA1 Stratum Radiatum Occupy Separate Anatomical Domains. J. Neurosci. 2002, 22, 183–192. [Google Scholar] [CrossRef]
- Bonni, A.; Sun, Y.; Nadal-Vicens, M.; Bhatt, A.; Frank, D.A.; Rozovsky, I.; Stahl, N.; Yancopoulos, G.D.; Greenberg, M.E. Regulation of Gliogenesis in the Central Nervous System by the JAK-STAT Signaling Pathway. Science 1997, 278, 477–483. [Google Scholar] [CrossRef]
- Ransom, B.; Behar, T.; Nedergaard, M. New roles for astrocytes (stars at last). Trends Neurosci. 2003, 26, 520–522. [Google Scholar] [CrossRef] [PubMed]
- Chiang, C.S.; Hong, J.H.; Stalder, A.; Sun, J.R.; Withers, H.R.; McBride, W.H. Delayed molecular responses to brain irradiation. Int. J. Radiat. Biol. 1997, 72, 45–53. [Google Scholar] [CrossRef]
- Hong, J.H.; Chiang, C.S.; Campbell, I.L.; Sun, J.R.; Withers, H.R.; McBride, W.H. Induction of acute phase gene expression by brain irradiation. Int. J. Radiat. Oncol. Biol. Phys. 1995, 33, 619–626. [Google Scholar] [CrossRef]
- Hwang, S.Y.; Jung, J.S.; Kim, T.H.; Lim, S.J.; Oh, E.S.; Kim, J.Y.; Ji, K.A.; Joe, E.H.; Cho, K.H.; Han, I.O. Ionizing radiation induces astrocyte gliosis through microglia activation. Neurobiol. Dis. 2006, 21, 457–467. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Gaber, M.W.; Boyd, K.; Wilson, C.M.; Kiani, M.F.; Merchant, T.E. Effects of fractionated radiation on the brain vasculature in a murine model: Blood–brain barrier permeability, astrocyte proliferation, and ultrastructural changes. Int. J. Radiat. Oncol. Biol. Phys. 2006, 66, 860–866. [Google Scholar] [CrossRef]
- Alvarez, J.I.; Dodelet-Devillers, A.; Kebir, H.; Ifergan, I.; Fabre, P.J.; Terouz, S.; Sabbagh, M.; Wosik, K.; Bourbonnière, L.; Bernard, M.; et al. The Hedgehog Pathway Promotes Blood-Brain Barrier Integrity and CNS Immune Quiescence. Science 2011, 334, 1727–1731. [Google Scholar] [CrossRef]
- Garcia, A.D.R.; Han, Y.G.; Triplett, J.W.; Farmer, W.T.; Harwell, C.C.; Ihrie, R.A. The Elegance of Sonic Hedgehog: Emerging Novel Functions for a Classic Morphogen. J. Neurosci. 2018, 38, 9338–9345. [Google Scholar] [CrossRef]
- Hwang, S.N.; Lee, J.S.; Seo, K.; Lee, H. Astrocytic Regulation of Neural Circuits Underlying Behaviors. Cells 2021, 10, 296. [Google Scholar] [CrossRef]
- Oliveira, J.F.; Sardinha, V.M.; Guerra-Gomes, S.; Araque, A.; Sousa, N. Do stars govern our actions? Astrocyte involvement in rodent behavior. Trends Neurosci. 2015, 38, 535–549. [Google Scholar] [CrossRef]
- Woodbury, M.E.; Freilich, R.W.; Cheng, C.J.; Asai, H.; Ikezu, S.; Boucher, J.D.; Slack, F.; Ikezu, T. miR-155 Is Essential for Inflammation-Induced Hippocampal Neurogenic Dysfunction. J. Neurosci. 2015, 35, 9764–9781. [Google Scholar] [CrossRef]
- Wu, M.D.; Hein, A.M.; Moravan, M.J.; Shaftel, S.S.; Olschowka, J.A.; O’Banion, M.K. Adult murine hippocampal neurogenesis is inhibited by sustained IL-1β and not rescued by voluntary running. Brain Behav. Immun. 2012, 26, 292–300. [Google Scholar] [CrossRef]
- Kirby, E.D.; Muroy, S.E.; Sun, W.G.; Covarrubias, D.; Leong, M.J.; Barchas, L.A.; Kaufer, D. Acute stress enhances adult rat hippocampal neurogenesis and activation of newborn neurons via secreted astrocytic FGF2. eLife 2013, 2, e00362. [Google Scholar] [CrossRef]
- Palmer, T.D.; Ray, J.; Gage, F.H. FGF-2-Responsive Neuronal Progenitors Reside in Proliferative and Quiescent Regions of the Adult Rodent Brain. Mol. Cell. Neurosci. 1995, 6, 474–486. [Google Scholar] [CrossRef] [PubMed]
- Galvez-Contreras, A.Y.; Gonzalez-Castaneda, R.E.; Luquin, S.; Gonzalez-Perez, O. Role of fibroblast growth factor receptors in astrocytic stem cells. Curr. Signal Transduct. Ther. 2012, 7, 81–86. [Google Scholar] [CrossRef][Green Version]
- Zou, L.H.; Shi, Y.J.; He, H.; Jiang, S.M.; Huo, F.-F.; Wang, X.M.; Wu, F.; Ma, L. Effects of FGF2/FGFR1 Pathway on Expression of A1 Astrocytes After Infrasound Exposure. Front. Neurosci. 2019, 13, 429. [Google Scholar] [CrossRef] [PubMed]
- Lee, M.N.; Kim, J.W.; Oh, S.H.; Jeong, B.C.; Hwang, Y.C.; Koh, J.T. FGF2 Stimulates COUP-TFII Expression via the MEK1/2 Pathway to Inhibit Osteoblast Differentiation in C3H10T1/2 Cells. PLoS ONE 2016, 11, e0159234. [Google Scholar] [CrossRef]
- Krishnan, V.; Elberg, G.; Tsai, M.J.; Tsai, S.Y. Identification of a Novel Sonic Hedgehog Response Element in the Chicken Ovalbumin Upstream Promoter-Transcription Factor II Promoter. Mol. Endocrinol. 1997, 11, 1458–1466. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Yu, F.; Zhu, J.; Han, S.; Chen, J.; Wu, X.; Chen, Y.; Shen, T.; Liao, J.; Guo, W.; et al. Imbalance of Excitatory/Inhibitory Neuron Differentiation in Neurodevelopmental Disorders with an NR2F1 Point Mutation. Cell Rep. 2020, 31, 107521. [Google Scholar] [CrossRef]
- Bertacchi, M.; Gruart, A.; Kaimakis, P.; Allet, C.; Serra, L.; Giacobini, P.; Delgado-García, J.M.; Bovolenta, P.; Studer, M. Mouse Nr2f1 haploinsufficiency unveils new pathological mechanisms of a human optic atrophy syndrome. EMBO Mol. Med. 2019, 11, e10291. [Google Scholar] [CrossRef] [PubMed]
- Bonzano, S.; Crisci, I.; Podlesny-Drabiniok, A.; Rolando, C.; Krezel, W.; Studer, M.; De Marchis, S. Neuron-Astroglia Cell Fate Decision in the Adult Mouse Hippocampal Neurogenic Niche Is Cell-Intrinsically Controlled by COUP-TFI In Vivo. Cell Rep. 2018, 24, 329–341. [Google Scholar] [CrossRef] [PubMed]
- Dutka, T.; Hallberg, D.; Reeves, R.H. Chronic up-regulation of the SHH pathway normalizes some developmental effects of trisomy in Ts65Dn mice. Mech. Dev. 2015, 135, 68–80. [Google Scholar] [CrossRef] [PubMed]
- Jackson, T.W.; Bendfeldt, G.A.; Beam, K.A.; Rock, K.D.; Belcher, S.M. Heterozygous mutation of sonic hedgehog receptor (Ptch1) drives cerebellar overgrowth and sex-specifically alters hippocampal and cortical layer structure, activity, and social behavior in female mice. Neurotoxicol. Teratol. 2020, 78, 106866. [Google Scholar] [CrossRef] [PubMed]
- Eriksson, P.; Ankarberg, E.; Fredriksson, A. Exposure to nicotine during a defined period in neonatal life induces permanent changes in brain nicotinic receptors and in behaviour of adult mice. Brain Res. 2000, 853, 41–48. [Google Scholar] [CrossRef]
- Chen, S.D.; Wu, C.L.; Hwang, W.C.; Yang, D.I. More Insight into BDNF against Neurodegeneration: Anti-Apoptosis, Anti-Oxidation, and Suppression of Autophagy. Int. J. Mol. Sci. 2017, 18, 545. [Google Scholar] [CrossRef]
- Huang, S.S.; Cheng, H.; Tang, C.M.; Nien, M.W.; Huang, Y.S.; Lee, I.H.; Yin, J.H.; Kuo, T.B.; Yang, C.C.; Tsai, S.K.; et al. Anti-oxidative, anti-apoptotic, and pro-angiogenic effects mediate functional improvement by sonic hedgehog against focal cerebral ischemia in rats. Exp. Neurol. 2013, 247, 680–688. [Google Scholar] [CrossRef]
- Suwelack, D.; Hurtado-Lorenzo, A.; Millan, E.; Gonzalez-Nicolini, V.; Wawrowsky, K.; Lowenstein, P.R.; Castro, M.G. Neuronal expression of the transcription factor Gli1 using the Tα1 α-tubulin promoter is neuroprotective in an experimental model of Parkinson’s disease. Gene Ther. 2004, 11, 1742–1752. [Google Scholar] [CrossRef]
- Wu, C.L.; Chen, S.D.; Hwang, C.S.; Yang, D.I. Sonic hedgehog mediates BDNF-induced neuroprotection against mitochondrial inhibitor 3-nitropropionic acid. Biochem. Biophys. Res. Commun. 2009, 385, 112–117. [Google Scholar] [CrossRef]
- Wu, C.L.; Chen, S.D.; Yin, J.H.; Hwang, C.S.; Yang, D.I. Erythropoietin and sonic hedgehog mediate the neuroprotective effects of brain-derived neurotrophic factor against mitochondrial inhibition. Neurobiol. Dis. 2010, 40, 146–154. [Google Scholar] [CrossRef]
- Jin, Y.; Raviv, N.; Barnett, A.; Bambakidis, N.C.; Filichia, E.; Luo, Y. The Shh Signaling Pathway Is Upregulated in Multiple Cell Types in Cortical Ischemia and Influences the Outcome of Stroke in an Animal Model. PLoS ONE 2015, 10, e0124657. [Google Scholar] [CrossRef]
- Jin, Y.; Barnett, A.; Zhang, Y.; Yu, X.; Luo, Y. Poststroke Sonic Hedgehog Agonist Treatment Improves Functional Recovery by Enhancing Neurogenesis and Angiogenesis. Stroke 2017, 48, 1636–1645. [Google Scholar] [CrossRef]
- Chechneva, O.V.; Mayrhofer, F.; Daugherty, D.J.; Krishnamurty, R.G.; Bannerman, P.; Pleasure, D.E.; Deng, W. A Smoothened receptor agonist is neuroprotective and promotes regeneration after ischemic brain injury. Cell Death Dis. 2014, 5, e1481. [Google Scholar] [CrossRef]
- Das, I.; Park, J.M.; Shin, J.H.; Jeon, S.K.; Lorenzi, H.; Linden, D.J.; Worley, P.F.; Reeves, R.H. Hedgehog Agonist Therapy Corrects Structural and Cognitive Deficits in a Down Syndrome Mouse Model. Sci. Transl. Med. 2013, 5, 201ra120. [Google Scholar] [CrossRef]
- He, P.; Staufenbiel, M.; Li, R.; Shen, Y. Deficiency of Patched 1-induced Gli1 signal transduction results in astrogenesis in Swedish mutated APP transgenic mice. Hum. Mol. Genet. 2014, 23, 6512–6527. [Google Scholar] [CrossRef]
- Eriksson, P.; Ankarberg, E.; Viberg, H.; Fredriksson, A. The developing cholinergic system as target for environmental toxicants, nicotine and polychlorinated biphenyls (PCBs): Implications for neurotoxicological processes in mice. Neurotox. Res. 2001, 3, 37–51. [Google Scholar] [CrossRef] [PubMed]
- Casciati, A.; Dobos, K.; Antonelli, F.; Benedek, A.; Kempf, S.J.; Belles, M.; Balogh, A.; Tanori, M.; Heredia, L.; Atkinson, M.J.; et al. Age-related effects of X-ray irradiation on mouse hippocampus. Oncotarget 2016, 7, 28040–28058. [Google Scholar] [CrossRef] [PubMed]
- Hagihara, H.; Toyama, K.; Yamasaki, N.; Miyakawa, T. Dissection of Hippocampal Dentate Gyrus from Adult Mouse. J. Vis. Exp. 2009, 33, e1543–e1549. [Google Scholar] [CrossRef] [PubMed]
- Bellés, M.; Alonso, V.; Linares, V.; Albina, M.L.; Sirvent, J.J.; Domingo, J.L.; Sánchez, D.J. Behavioral effects and oxidative status in brain regions of adult rats exposed to BDE-99. Toxicol. Lett. 2010, 194, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Vicens, P.; Ribes, D.; Torrente, M.; Domingo, J.L. Behavioral effects of PNU-282987, an alpha7 nicotinic receptor agonist, in mice. Behav. Brain Res. 2011, 216, 341–348. [Google Scholar] [CrossRef]
- Heredia, L.; Bellés, M.; Llovet, M.I.; Domingo, J.L.; Linares, V. Neurobehavioral effects of concurrent exposure to cesium-137 and paraquat during neonatal development in mice. Toxicology 2015, 329, 73–79. [Google Scholar] [CrossRef]
- Lalonde, R.; Strazielle, C. Relations between open-field, elevated plus-maze, and emergence tests in C57BL/6JIco and BALB/cAnN@Ico mice injected with ethanol. Fundam. Clin. Pharmacol. 2012, 26, 271–278. [Google Scholar] [CrossRef] [PubMed]
- Heredia, L.; Bellés, M.; Llovet, M.I.; Domingo, J.L.; Linares, V. Behavioral effects in mice of postnatal exposure to low-doses of 137-cesium and bisphenol A. Toxicology 2016, 340, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Walf, A.A.; Frye, C.A. Estradiol decreases anxiety behavior and enhances inhibitory avoidance and gestational stress produces opposite effects. Stress 2007, 10, 251–260. [Google Scholar] [CrossRef] [PubMed]




| Synaptic Functions | Cell Differentiation | Apoptosis | Growth Factors | Cell Adhesion/Neural Migration | Cell Cycle/Transcription Factors | Signal Transduction | |
|---|---|---|---|---|---|---|---|
| WT | Erbb2 | Pax6 | Adora2a Ep300 Alk | Cxcl1 | Slit2 | Ep300 | Adora2a Cxcl1 |
| Ptch1+/- | Chrm2 Fgf2 Pafah1b1 S100b Apbb1 ApoE | Nrg1 Pafah1b1 Pax5 | S100b ApoE | Fgf2 Nrg1 Ptn | Pafah1b1 Rac1 Robo1 | Ptn Flna | Nrg1 Dll1 |
| WT Ptch1+/- | App Bdnf Creb1 Dcx Drd2 Map2 Notch1 | Bdnf Mdk NeuroD1 Ntf3 Olig2 Sox2 | Notch2 | Artn Mdk Ndp | Dcx Drd2 | Mdk | App Notch1 Notch2 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Antonelli, F.; Casciati, A.; Belles, M.; Serra, N.; Linares-Vidal, M.V.; Marino, C.; Mancuso, M.; Pazzaglia, S. Long-Term Effects of Ionizing Radiation on the Hippocampus: Linking Effects of the Sonic Hedgehog Pathway Activation with Radiation Response. Int. J. Mol. Sci. 2021, 22, 12605. https://doi.org/10.3390/ijms222212605
Antonelli F, Casciati A, Belles M, Serra N, Linares-Vidal MV, Marino C, Mancuso M, Pazzaglia S. Long-Term Effects of Ionizing Radiation on the Hippocampus: Linking Effects of the Sonic Hedgehog Pathway Activation with Radiation Response. International Journal of Molecular Sciences. 2021; 22(22):12605. https://doi.org/10.3390/ijms222212605
Chicago/Turabian StyleAntonelli, Francesca, Arianna Casciati, Montserrat Belles, Noemi Serra, Maria Victoria Linares-Vidal, Carmela Marino, Mariateresa Mancuso, and Simonetta Pazzaglia. 2021. "Long-Term Effects of Ionizing Radiation on the Hippocampus: Linking Effects of the Sonic Hedgehog Pathway Activation with Radiation Response" International Journal of Molecular Sciences 22, no. 22: 12605. https://doi.org/10.3390/ijms222212605
APA StyleAntonelli, F., Casciati, A., Belles, M., Serra, N., Linares-Vidal, M. V., Marino, C., Mancuso, M., & Pazzaglia, S. (2021). Long-Term Effects of Ionizing Radiation on the Hippocampus: Linking Effects of the Sonic Hedgehog Pathway Activation with Radiation Response. International Journal of Molecular Sciences, 22(22), 12605. https://doi.org/10.3390/ijms222212605






