Trace Element Selenium Effectively Alleviates Intestinal Diseases
Abstract
:1. Introduction
2. Se and Selenoproteins Have a Close Connection with the Organism
2.1. Basic Information about Se
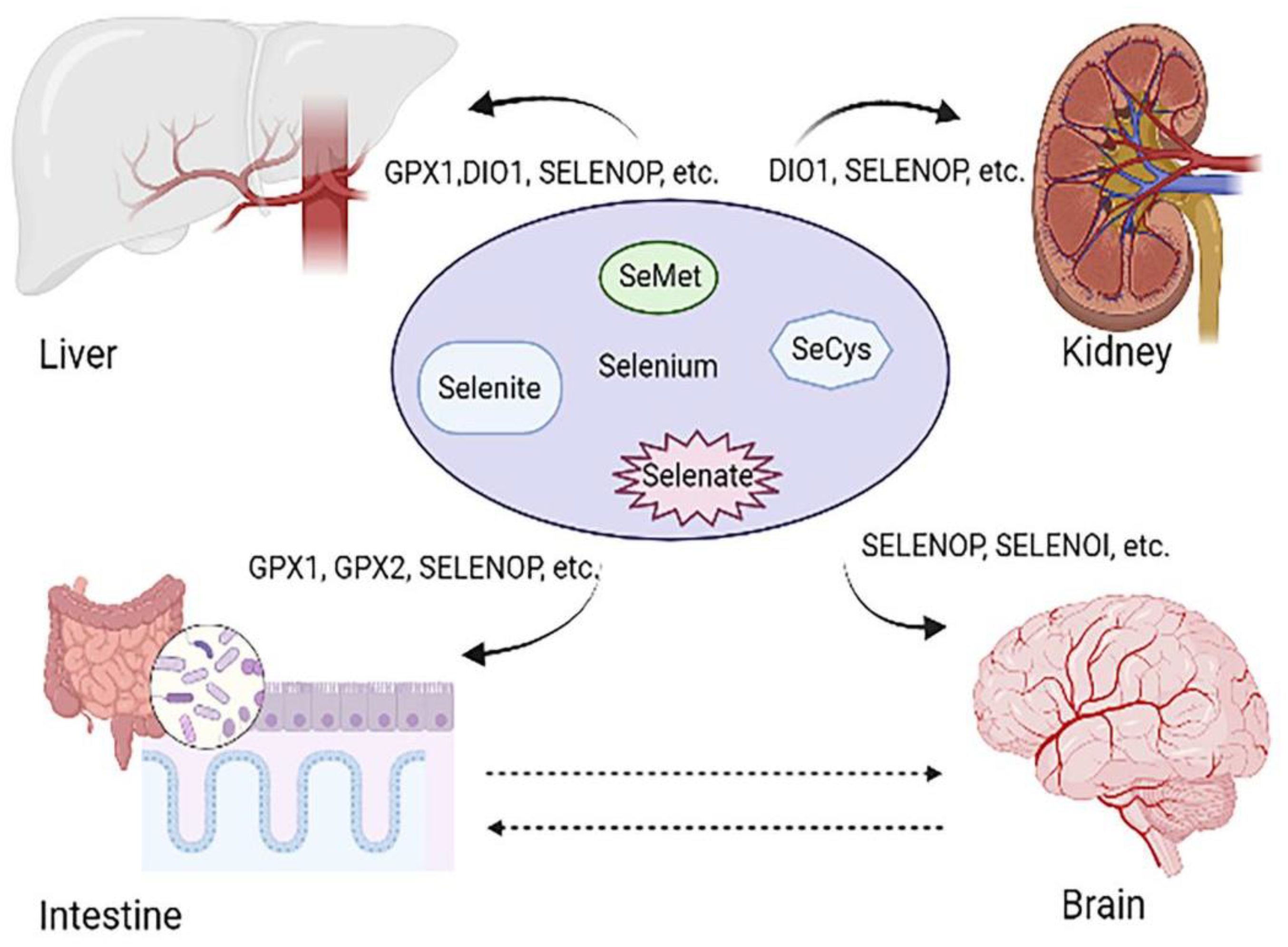
2.2. Selenoproteins: The Main Form of Se Utilization
3. Se Is Involved in the Regulation of Intestine-Related Diseases
3.1. Se Is Associated with the Occurrence of Intestinal Cancer
3.2. Se Can Affect Inflammatory Bowel Diseases
4. Disease-Related Mechanisms of Se Regulation
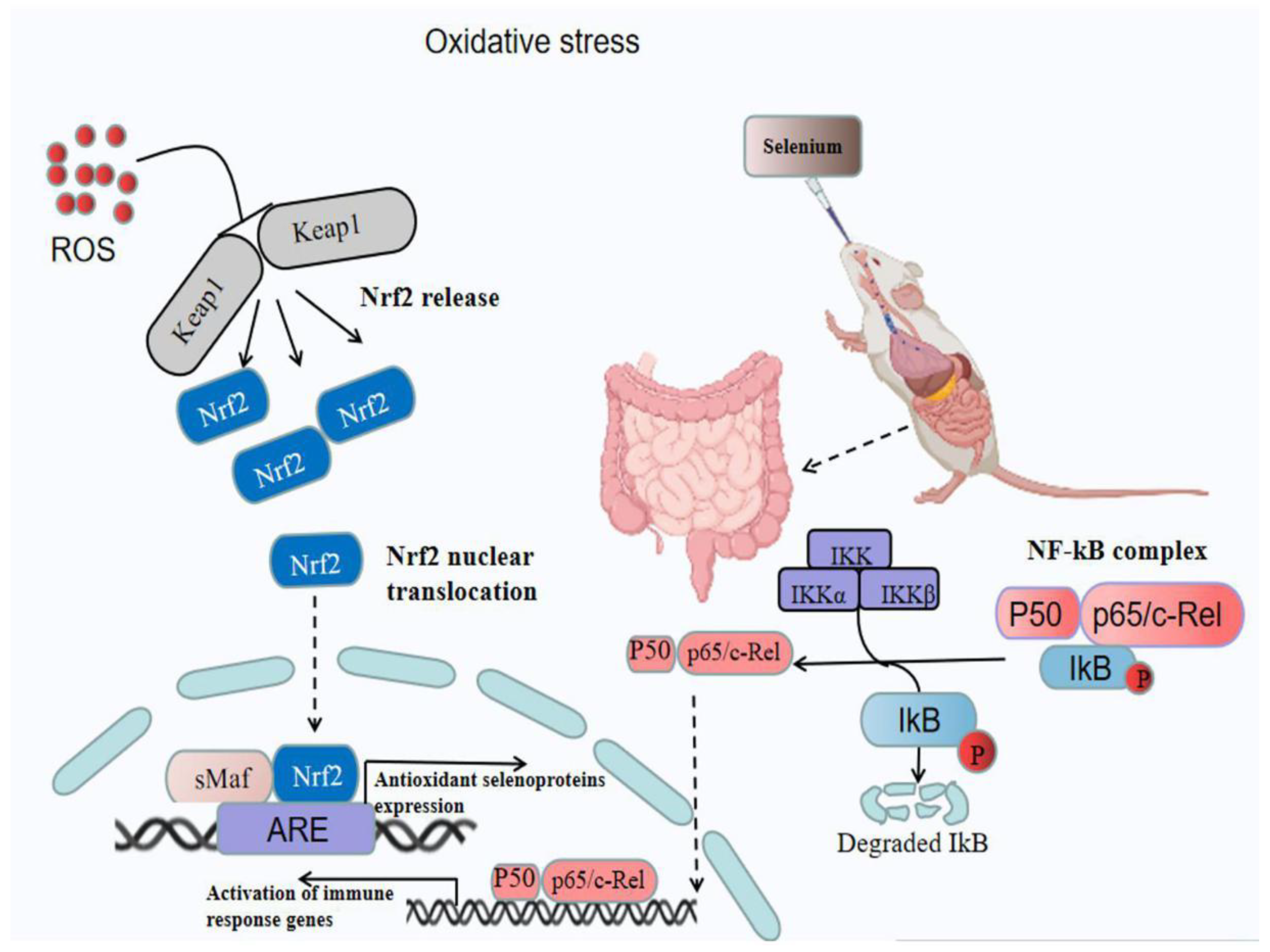
4.1. Se Acts as an Antioxidant
4.2. Se Has an Effect on the Diversity of Intestinal Microbiota
4.3. Se Regulates the Nervous System
4.4. Se Regulates Endothelial Function
5. Nano-Se Alleviates the Degree of Disease
5.1. Comparison of Modification of Nano-Se Particles by Different Biomacromolecules
5.2. Surface Modification of Nano-Se Particles Alleviates the Degree of Disease
5.3. Comparison of Nano-Se and Conventional Se Supplements
6. Conclusions and Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Prabhu, K.S.; Lei, X.G. Selenium. Adv. Nutr. 2016, 7, 415–417. [Google Scholar] [CrossRef] [Green Version]
- Rayman, M.P. Selenium and human health. Lancet 2012, 379, 1256–1268. [Google Scholar] [CrossRef]
- Schwarz, K.; Foltz, C.M. Selenium as an integral part of factor 3 against dietary necrotic liver degeneration. Nutrition 1999, 15, 255. [Google Scholar]
- Zhou, H.; Wang, T.; Li, D. Prevention of Keshan Disease by Selenium Supplementation: A Systematic Review and Meta-analysis. Biol. Trace Elem. Res. 2018, 186, 98–105. [Google Scholar] [CrossRef]
- Hou, J.; Zhu, L.; Sum, D. Association of selenium levels with the prevention and control of Keshan disease: A cross-sectional study. J. Trace Elem. Med. Biol. 2021, 68, 126832. [Google Scholar] [CrossRef] [PubMed]
- Avery, J.C.; Hoffmann, P.R. Selenium, Selenoproteins, and Immunity. Nutrients 2018, 10, 1203. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rayman, M.P. The importance of selenium to human health. Lancet 2000, 356, 233–241. [Google Scholar] [CrossRef] [Green Version]
- Foster, H.D.; Zhang, L. Longevity and selenium deficiency: Evidence from the People’s Republic of China. Sci. Total Environ. 1995, 170, 133–139. [Google Scholar] [CrossRef]
- Roman, M.; Jitaru, P.; Barbante, C. Selenium biochemistry and its role for human health. Metallomics 2014, 6, 25–54. [Google Scholar] [CrossRef] [PubMed]
- Rayman, M.P. Food-chain selenium and human health: Emphasis on intake. Br. J. Nutr. 2008, 100, 254–268. [Google Scholar] [CrossRef] [Green Version]
- Rayman, M.P. Selenium intake, status, and health: A complex relationship. Hormones 2020, 19, 9–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Papp, L.V.; Lu, J.; Holmgren, A. From selenium to selenoproteins: Synthesis, identity, and their role in human health. Antioxid. Redox Signal. 2007, 9, 775–806. [Google Scholar] [CrossRef]
- Zhang, Y.Z.; Li, Y.Y. Inflammatory bowel disease: Pathogenesis. World J. Gastroenterol. 2014, 20, 91–99. [Google Scholar] [CrossRef]
- Khor, B.; Gardet, A.; Xavier, R.J. Genetics and pathogenesis of inflammatory bowel disease. Nature 2011, 474, 307–317. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hodson, R. Inflammatory bowel disease. Nature 2016, 540, S97. [Google Scholar] [CrossRef]
- Labunskyy, V.M.; Hatfield, D.L.; Gladyshev, V.N. Selenoproteins: Molecular pathways and physiological roles. Physiol. Rev. 2014, 94, 739–777. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Reeves, M.A.; Hoffmann, P.R. The human selenoproteome: Recent insights into functions and regulation. Cell Mol. Life Sci. 2009, 66, 2457–2478. [Google Scholar] [CrossRef] [Green Version]
- Dinh, Q.T.; Cui, Z.; Huang, J. Selenium distribution in the Chinese environment and its relationship with human health: A review. Environ. Int. 2018, 112, 294–309. [Google Scholar] [CrossRef]
- Kim, J.H.; Dong, Y.K. Comparison of toxic effects of dietary organic or inorganic selenium and prediction of selenium intake and tissue selenium concentrations in broiler chickens using feather selenium concentrations. Poult. Sci. 2020, 99, 6462–6473. [Google Scholar] [CrossRef]
- Emmanuelle, B.; Virginie, M.; Fabienne, S. Selenium exposure in subjects living in areas with high selenium concentrated drinking water: Results of a French integrated exposure assessment survey. Environ. Int. 2012, 40, 155–161. [Google Scholar] [CrossRef] [PubMed]
- Weekley, C.M.; Harris, H.H. Which form is that? The importance of selenium speciation and metabolism in the prevention and treatment of disease. Chem. Soc. Rev. 2013, 42, 8870–8894. [Google Scholar] [CrossRef]
- Hadrup, N.; Ravn-Haren, G. Absorption, distribution, metabolism and excretion (ADME) of oral selenium from organic and inorganic sources: A review. J. Trace Elem. Med. Biol. 2021, 67, 126801. [Google Scholar] [CrossRef]
- Panchal, S.K.; Wanyonyi, S.; Brown, L. Selenium, Vanadium, and Chromium as Micronutrients to Improve Metabolic Syndrome. Curr. Hypertens. Rep. 2017, 19, 10. [Google Scholar] [CrossRef]
- Mehdi, Y.; Hornick, J.L.; Istasse, L.; Dufrasne, I. Selenium in the environment, metabolism and involvement in body functions. Molecules 2013, 18, 3292–3311. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tan, J.; Zhu, W.; Wang, W. Selenium in soil and endemic diseases in China. Sci. Total Environ. 2002, 284, 227–235. [Google Scholar] [CrossRef]
- Hesketh, J. Nutrigenomics and selenium: Gene expression patterns, physiological targets, and genetics. Annu. Rev. Nutr. 2008, 28, 157–177. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shahid, M.; Niazi, N.K.; Rashid, M.I. A critical review of selenium biogeochemical behavior in soil-plant system with an inference to human health. Environ. Pollut. 2018, 234, 915–934. [Google Scholar]
- Ullah, H.; Liu, G.; Ahmad, R. A comprehensive review on environmental transformation of selenium: Recent advances and research perspectives. Environ. Geochem. Health 2019, 41, 1003–1035. [Google Scholar] [CrossRef]
- Hatfield, D.L.; Tsuji, P.A.; Carlson, B.A. Selenium and selenocysteine: Roles in cancer, health, and development. Trends Biochem. Sci. 2014, 39, 112–120. [Google Scholar] [CrossRef] [Green Version]
- Yao, Y.; Pei, F.; Kang, P. Selenium, iodine, and the relation with Kashin-Beck disease. Nutrition 2011, 27, 1095–1100. [Google Scholar] [CrossRef]
- Driscoll, D.M.; Copeland, P.R. Mechanism and regulation of selenoprotein synthesis. Annu. Rev. Nutr. 2003, 23, 17–40. [Google Scholar] [CrossRef]
- Vindry, C.; Ohlmann, T.; Chavatte, L. Translation regulation of mammalian selenoproteins. Biochim. Biophys. Acta Gen. Subj. 2018, 1862, 2480–2492. [Google Scholar] [CrossRef]
- Reszka, E.; Jablonska, E.; Gromadzinska, J. Relevance of selenoprotein transcripts for selenium status in humans. Genes Nutr. 2012, 7, 127–137. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Short, S.P.; Pilat, J.M.; Williams, C.S. Roles for selenium and selenoprotein P in the development, progression, and prevention of intestinal disease. Free Radic. Biol. Med. 2018, 127, 26–35. [Google Scholar] [CrossRef]
- Cardoso, B.R.; Roberts, B.R.; Bush, A.I. Selenium, selenoproteins and neurodegenerative diseases. Metallomics 2015, 7, 1213–1228. [Google Scholar] [CrossRef] [Green Version]
- Pillai, R.; Uyehara-Lock, J.H.; Bellinger, F.P. Selenium and selenoprotein function in brain disorders. IUBMB Life 2014, 66, 229–239. [Google Scholar] [CrossRef]
- Hughes, D.J.; Kunická, T.; Schomburg, L. Expression of Selenoprotein Genes and Association with Selenium Status in Colorectal Adenoma and Colorectal Cancer. Nutrients 2018, 10, 1812. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shchedrina, V.A.; Zhang, Y.; Labunskyy, V.M. Structure–Function Relations, Physiological Roles, and Evolution of Mammalian ER-Resident Selenoproteins. Antioxid. Redox Signal. 2010, 12, 839–849. [Google Scholar] [CrossRef] [PubMed]
- Steinbrenner, H.; Speckmann, B.; Klotz, L.O. Selenoproteins: Antioxidant selenoenzymes and beyond. Arch. Biochem. Biophys. 2016, 595, 113–119. [Google Scholar] [CrossRef] [PubMed]
- Murawaki, Y.; Tsuchiya, H.; Kanbe, T. Aberrant expression of selenoproteins in the progression of colorectal cancer. Cancer Lett. 2008, 259, 218–230. [Google Scholar] [CrossRef] [Green Version]
- Ying, H.; Zhang, Y. Systems Biology of Selenium and Complex Disease. Biol. Trace Elem. Res. 2019, 192, 38–50. [Google Scholar] [CrossRef] [PubMed]
- Ha, H.Y.; Alfulaij, N.; Seale, L.A. From Selenium Absorption to Selenoprotein Degradation. Biol. Trace Elem. Res. 2019, 192, 26–37. [Google Scholar] [CrossRef]
- Ramos, G.P.; Papadakis, K.A. Mechanisms of Disease: Inflammatory Bowel Diseases. Mayo Clin. Proc. 2019, 94, 155–165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Geerling, B.J.; Badart-Smook, A.; Stockbrügger, R.W. Comprehensive nutritional status in recently diagnosed patients with inflammatory bowel disease compared with population controls. Eur. J. Clin. Nutr. 2000, 54, 514–521. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nettleford, S.K.; Prabhu, K.S. Selenium and Selenoproteins in Gut Inflammation-A Review. Antioxidants 2018, 7, 36. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kudva, A.K.; Shay, A.E.; Prabhu, K.S. Selenium and inflammatory bowel disease. Am. J. Physiol. Gastrointest. Liver Physiol. 2015, 309, G71–G77. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Santesmasses, D.; Mariotti, M.; Gladyshev, V.N. Bioinformatics of Selenoproteins. Antioxid. Redox Signal. 2020, 33, 525–536. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.T.; Navarro-Alarcón, M.; Quesada, G.J. Ulcerative Colitis and Crohn’s Disease Are Associated with Decreased Serum Selenium Concentrations and Increased Cardiovascular Risk. Nutrients 2016, 8, 780. [Google Scholar] [CrossRef]
- Jäger, S.; Stange, E.F.; Wehkamp, J. Inflammatory bowel disease: An impaired barrier disease. Langenbeck’s Arch. Surg. 2013, 398, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Kipp, A.P. Selenium in colorectal and differentiated thyroid cancer. Hormones 2020, 19, 41–46. [Google Scholar] [CrossRef]
- Peters, K.M.; Carlson, B.A.; Gladyshev, V.N. Selenoproteins in colon cancer. Free Radic. Biol. Med. 2018, 127, 14–25. [Google Scholar] [CrossRef] [PubMed]
- Barrett, C.W.; Reddy, V.K.; Short, S.P. Selenoprotein P influences colitis-induced tumorigenesis by mediating stemness and oxidative damage. J. Clin. Investig. 2015, 125, 2646–2660. [Google Scholar] [CrossRef] [Green Version]
- Ma, Y.M.; Guo, Y.Z.; Ibeanu, G. Overexpression of selenoprotein H prevents mitochondrial dynamic imbalance induced by glutamate exposure. Int. J. Biol. Sci. 2017, 13, 1458–1469. [Google Scholar] [CrossRef] [Green Version]
- Qazi, I.H.; Angel, C.; Yang, H. Selenium, Selenoproteins, and Female Reproduction: A Review. Molecules 2018, 23, 3053. [Google Scholar] [CrossRef] [Green Version]
- Bertz, M.; Kühn, K.; Koeberle, S.C. Selenoprotein H controls cell cycle progression and proliferation of human colorectal cancer cells. Free Radic. Biol. Med. 2018, 127, 98–107. [Google Scholar] [CrossRef] [PubMed]
- Fedirko, V.; Jenab, M.; Hughes, D.J. Association of Selenoprotein and Selenium Pathway Genotypes with Risk of Colorectal Cancer and Interaction with Selenium Status. Nutrients 2019, 11, 935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Méplan, C.; Hesketh, J. The influence of selenium and selenoprotein gene variants on colorectal cancer risk. Mutagenesis 2012, 27, 177–186. [Google Scholar] [CrossRef] [Green Version]
- Barrett, C.W.; Short, S.P.; Williams, C.S. Selenoproteins and oxidative stress-induced inflammatory tumorigenesis in the gut. Cell Mol. Life Sci. 2017, 74, 607–616. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Steinbrenner, H.; Speckmann, B.; Sies, H. Toward understanding success and failures in the use of selenium for cancer prevention. Antioxid. Redox Signal. 2013, 19, 181–191. [Google Scholar] [CrossRef]
- Irons, R.; Tsuji, P.A.; Davis, C.D. Deficiency in the 15-kDa selenoprotein inhibits tumorigenicity and metastasis of colon cancer cells. Cancer Prev. Res. 2010, 3, 630–639. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Han, Y.M.; Koh, J.; Kim, J.W. NF-kappa B activation correlates with disease phenotype in Crohn’s disease. PLoS ONE 2017, 12, e0182071. [Google Scholar] [CrossRef] [Green Version]
- Barrett, C.W.; Singh, K.; Motley, A.K. Dietary selenium deficiency exacerbates DSS-induced epithelial injury and AOM/DSS-induced tumorigenesis. PLoS ONE 2013, 8, e67845. [Google Scholar] [CrossRef] [Green Version]
- Huang, L.J.; Mao, X.T.; Li, Y.Y. Multiomics analyses reveal a critical role of selenium in controlling T cell differentiation in Crohn’s disease. Immunity 2021, 54, 1728–1744.e7. [Google Scholar] [CrossRef]
- Atreya, I.; Atreya, R.; Neurath, M.F. NF-kappaB in inflammatory bowel disease. J. Intern. Med. 2008, 263, 591–596. [Google Scholar] [CrossRef]
- Koeberle, S.C.; Gollowitzer, A.; Laoukili, J. Distinct and overlapping functions of glutathione peroxidases 1 and 2 in limiting NF-κB-driven inflammation through redox-active mechanisms. Redox Biol. 2020, 28, 101388. [Google Scholar] [CrossRef]
- Zhu, C.; Zhang, S.; Song, C. Selenium nanoparticles decorated with Ulva lactuca polysaccharide potentially attenuate colitis by inhibiting NF-κB mediated hyper inflammation. J. Nanobiotechnol. 2017, 15, 20. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Song, Y.; Kurose, A.; Li, R. Ablation of Selenbp1 Alters Lipid Metabolism via the Pparα Pathway in Mouse Kidney. Int. J. Mol. Sci. 2021, 22, 5334. [Google Scholar] [CrossRef] [PubMed]
- Arias-Borrego, A.; Callejón-Leblic, B.; Calatayud, M.; Gómez-Ariza, J.L.; Collado, M.C.; García-Barrera, T. Insights into cancer and neurodegenerative diseases through selenoproteins and the connection with gut microbiota-current analytical methodologies. Expert Rev. Proteom. 2019, 16, 805–814. [Google Scholar] [CrossRef] [PubMed]
- Bellinger, F.P.; Raman, A.V.; Rueli, R.H. Changes in selenoprotein P in substantia nigra and putamen in Parkinson’s disease. J. Parkinsons Dis. 2012, 2, 115–126. [Google Scholar] [CrossRef]
- Fredericks, G.J.; Hoffmann, P.R. Selenoprotein K and protein palmitoylation. Antioxid. Redox Signal. 2015, 23, 854–862. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, J.H.; Jang, J.K.; Ko, K.Y. Degradation of selenoprotein S and selenoprotein K through PPARγ-mediated ubiquitination is required for adipocyte differentiation. Cell Death Differ. 2019, 26, 1007–1023. [Google Scholar] [CrossRef] [PubMed]
- Foong, J.P.P.; Hung, L.Y.; Poon, S. Early life interaction between the microbiota and the enteric nervous system. Am. J. Physiol. Gastrointest. Liver Physiol. 2020, 319, G541–G548. [Google Scholar] [CrossRef]
- Takahashi, K.; Suzuki, N.; Ogra, Y. Effect of gut microflora on nutritional availability of selenium. Food Chem. 2020, 319, 126537. [Google Scholar] [CrossRef] [PubMed]
- Lv, C.H.; Wang, T.; Liao, S.F. Effects of dietary supplementation of selenium-enriched probiotics on production performance and intestinal microbiota of weanling piglets raised under high ambient temperature. J. Anim. Physiol. Anim. Nutr. 2015, 99, 1161–1171. [Google Scholar] [CrossRef]
- Li, Z.; Dong, Y.; Xu, S. Organic Selenium Increased Gilts Antioxidant Capacity, Immune Function, and Changed Intestinal Microbiota. Front Microbiol. 2021, 12, 723190. [Google Scholar] [CrossRef] [PubMed]
- Keogh, C.E.; Kim, D.H.J.; Pusceddu, M.M. Myelin as a regulator of development of the microbiota-gut-brain axis. Brain Behav. Immun. 2021, 91, 437–450. [Google Scholar] [CrossRef]
- Li, X.J.; You, X.Y.; Wang, C.Y. Bidirectional Brain-gut-microbiota Axis in increased intestinal permeability induced by central nervous system injury. CNS Neurosci. Ther. 2020, 26, 783–790. [Google Scholar] [CrossRef]
- Stakenborg, N.; Boeckxstaens, G.E. Bioelectronics in the brain-gut axis: Focus on inflammatory bowel disease (IBD). Int. Immunol. 2021, 33, 337–348. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.; Berry, M.J. Selenium and selenoproteins in the brain and brain diseases. J. Neurochem. 2003, 86, 1–12. [Google Scholar] [CrossRef]
- Wang, X.; Wu, J.; Liu, X. Engineered liposomes targeting the gut–CNS Axis for comprehensive therapy of spinal cord injury. J. Control Release 2021, 331, 390–403. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Z.H.; Song, G.L. Roles of Selenoproteins in Brain Function and the Potential Mechanism of Selenium in Alzheimer’s Disease. Front Neurosci. 2021, 15, 646518. [Google Scholar] [CrossRef] [PubMed]
- Solovyev, N.; Blume, B. Selenium at the Neural Barriers: A Review. Front. Neurosci. 2021, 15, 630016. [Google Scholar] [CrossRef]
- Pitts, M.W.; Byrns, C.N.; Berry, M.J. Selenoproteins in nervous system development and function. Biol. Trace Elem. Res. 2014, 161, 231–245. [Google Scholar] [CrossRef] [Green Version]
- Steinbrenner, H.; Alili, L.; Bilgic, E. Involvement of selenoprotein P in protection of human astrocytes from oxidative damage. Free Radic. Biol Med. 2006, 40, 1513–1523. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.Y.; Al-Khayat, A.; Al-Murshedi, F. A mutation of EPT1 (SELENOI) underlies a new disorder of Kennedy pathway phospholipid biosynthesis. Brain 2017, 140, 547–554. [Google Scholar] [PubMed] [Green Version]
- Horibata, Y.; Elpeleg, O.; Eran, A. EPT1 (selenoprotein I) is critical for the neural development and maintenance of plasmalogen in humans. J. Lipid Res. 2018, 59, 1015–1026. [Google Scholar] [CrossRef] [Green Version]
- Kieliszek, M. Selenium–Fascinating Microelement, Properties and Sources in Food. Molecules 2019, 24, 1298. [Google Scholar] [CrossRef] [Green Version]
- Avery, J.C.; Yamazaki, Y.; Hoffmann, F.W. Selenoprotein I is essential for murine embryogenesis. Arch. Biochem. Biophys. 2020, 689, 108444. [Google Scholar] [CrossRef]
- Bourassa, M.W.; Alim, I.; Bultman, S.J. Butyrate, neuroepigenetics and the gut microbiome: Can a high fiber diet improve brain health? Neurosci. Lett. 2016, 625, 56–63. [Google Scholar] [CrossRef] [Green Version]
- Chanaday, N.L.; Bem, A.F.; Roth, G.A. Effect of diphenyl diselenide on the development of experimental autoimmune encephalomyelitis. Neurochem. Int. 2011, 59, 1155–1162. [Google Scholar] [CrossRef]
- Ma, C.; Hoffmann, F.W.; Marciel, M.P. Upregulated ethanolamine phospholipid synthesis via selenoprotein I is required for effective metabolic reprogramming during T cell activation. Mol. Metab. 2021, 47, 101170. [Google Scholar] [CrossRef] [PubMed]
- Fairweather-Tait, S.J.; Bao, Y.; Broadley, M.R. Selenium in human health and disease. Antioxid. Redox Signal. 2011, 14, 1337–1383. [Google Scholar] [CrossRef]
- Tian, T.; Wang, Z.; Zhang, J. Pathomechanisms of Oxidative Stress in Inflammatory Bowel Disease and Potential Antioxidant Therapies. Oxid. Med. Cell Longev. 2017, 2017, 4535194. [Google Scholar] [CrossRef] [PubMed]
- Fukata, M.; Arditi, M. The role of pattern recognition receptors in intestinal inflammation. Mucosal Immunol. 2013, 6, 451–463. [Google Scholar] [CrossRef] [PubMed]
- Garrett, W.S.; Gordon, J.I.; Glimcher, L.H. Homeostasis and inflammation in the intestine. Cell 2010, 140, 859–870. [Google Scholar] [CrossRef] [Green Version]
- Huang, J.Q.; Ren, F.Z.; Jiang, Y.Y. Selenoproteins protect against avian nutritional muscular dystrophy by metabolizing peroxides and regulating redox/apoptotic signaling. Free Radic. Biol. Med. 2015, 83, 129–138. [Google Scholar] [CrossRef]
- El-Sayed, E.R.; Abdelhakim, H.K.; Ahmed, A.S. Solid-state fermentation for enhanced production of selenium nanoparticles by gamma-irradiated Monascus purpureus and their biological evaluation and photocatalytic activities. Bioprocess. Biosyst. Eng. 2020, 43, 797–809. [Google Scholar] [CrossRef]
- Hu, S.; Hu, W.; Li, Y. Construction and structure-activity mechanism of polysaccharide nano-selenium carrier. Carbohydr. Polym. 2020, 236, 116052. [Google Scholar] [CrossRef]
- Hosnedlova, B.; Kepinska, M.; Skalickova, S. Nano-selenium and its nanomedicine applications: A critical review. Int. J. Nanomed. 2018, 13, 2107–2128. [Google Scholar] [CrossRef] [Green Version]
- Kaur, G.; Iqbal, M.; Bakshi, M.S. Biomineralization of Fine Selenium Crystalline Rods and Amorphous Spheres. J. Phys. Chem. C 2009, 113, 13670–13676. [Google Scholar] [CrossRef]
- Song, D.; Cheng, Y.; Li, X. Biogenic Nanoselenium Particles Effectively Attenuate Oxidative Stress-Induced Intestinal Epithelial Barrier Injury by Activating the Nrf2 Antioxidant Pathway. ACS Appl. Mater. Interfaces 2017, 9, 14724–14740. [Google Scholar] [CrossRef]
- Zhang, Y.; Wang, J.; Zhang, L. Creation of Highly Stable Selenium Nanoparticles Capped with Hyperbranched Polysaccharide in Water. Langmuir 2010, 26, 17617. [Google Scholar] [CrossRef] [PubMed]
- Wang, J.; Zhang, Y.; Yuan, Y. Immunomodulatory of selenium nano-particles decorated by sulfated Ganoderma lucidum polysaccharides. Food Chem. Toxicol. 2014, 68, 183–189. [Google Scholar] [CrossRef]
- Yu, S.; Wang, Y.; Zhang, W. pH-Assisted surface functionalization of selenium nanoparticles with curcumin to achieve enhanced cancer chemopreventive activity. RSC Adv. 2016, 6, 72213–72223. [Google Scholar] [CrossRef]
- Rao, L.; Ma, Y.; Zhuang, M. Chitosan-Decorated Selenium Nanoparticles as Protein Carriers to Improve the In Vivo Half-Life of the Peptide Therapeutic BAY 55-9837 for Type 2 Diabetes Mellitus. Int. J. Nanomed. 2014, 9, 4819–4828. [Google Scholar]
- Zhang, X.; Yan, H.; Ma, L. Preparation and characterization of selenium nanoparticles decorated by Spirulina platensis polysaccharide. J. Food Biochem. 2020, 44, e13363. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, X.; Xu, T. Elemental selenium at nano size (Nano-Se) as a potential chemopreventive agent with reduced risk of selenium toxicity: Comparison with se-methylselenocysteine in mice. Toxicol. Sci. 2008, 101, 22–31. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Li, X.; Zhi, H. Enhancement of cell permeabilization apoptosis-inducing activity of selenium nanoparticles by ATP surface decoration. Nanomedicine 2013, 9, 74–84. [Google Scholar] [CrossRef]
- Fang, Y.; Tang, Q.; Zhong, X. Surface decoration by Spirulina polysaccharide enhances the cellular uptake and anticancer efficacy of selenium nanoparticles. Int. J. Nanomed. 2012, 7, 835–844. [Google Scholar]
- McKelvey, S.M.; Horgan, K.A.; Murphy, R.A. Chemical form of selenium differentially influences DNA repair pathways following exposure to lead nitrate. J. Trace Elem. Med. Biol. 2015, 29, 151–169. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Prasad, K.S. Role of nano-selenium in health and environment. J. Biotechnol. 2021, 325, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Lynch, S.J.; Horgan, K.A.; Walls, D. Selenium Source Impacts Protection of Porcine Jejunal Epithelial Cells from Cadmium-Induced DNA Damage, with Maximum Protection Exhibited with Yeast-Derived Selenium Compounds. Biol. Trace Elem. Res. 2017, 176, 311–320. [Google Scholar] [CrossRef] [PubMed]
- Gangadoo, S.; Dinev, I.; Stanley, D. Nanoparticles of selenium as high bioavailable and non-toxic supplement alternatives for broiler chickens. Environ. Sci. Pollut. Res. 2020, 27, 16159–16166. [Google Scholar] [CrossRef] [PubMed]
- Surai, P.F.; Kochish, I.I. Food for thought: Nano-selenium in poultry nutrition and health. Anim. Health Res. Rev. 2020, 21, 103–107. [Google Scholar] [CrossRef]
- Kuperman, R.G.; Checkai, R.T.; Simini, M. Selenium toxicity to survival and reproduction of Collembola and Enchytraeids in a sandy loam soil. Environ. Toxicol. Chem. 2010, 69, 119–132. [Google Scholar] [CrossRef]
| Source | Average Content | References |
|---|---|---|
| Soil | 0.01–2 mg/kg | [22,25,27,28] |
| Water | 0.02–742 μg/L | [20,25,28] |
| Cereal | 0.1–10 μg/g | [21,23,27] |
| Vegetables | 0.0008–5.37 mg/kg | [24,25,28] |
| Fruits | <0.01 μg/g | [20,21,23,27] |
| Animal-based foods | 0.3–0.5 μg/g | [18,24,27,28] |
| Selenoproteins | Abbreviation | Functions | Change | Distribution | References |
|---|---|---|---|---|---|
| Glutathione peroxidase 1 | GPX1 | Antioxidant enzymes | ↓ | Cytosol, mitochondria; widely expressed | [2,9,46] |
| Glutathione peroxidase2 | GPX2 | Antioxidant protection | ↑ | Cytosol, ER; gastrointestinal tissue, liver | [2,11,49,51] |
| Glutathione peroxidase3 | CPX3 | Antioxidant enzymes | ↓ | Secreted, plasma, gastrointestinal tissue | [12,46,51] |
| Thioredoxin reductase 1 | TXNRD1 | Reduce thioredoxin | ↑ | Cytosol, nucleus; widely distributed | [46,48,49] |
| Selenoprotein H | SELENOH | Nucleolar oxidoreductase | ↑ | Nucleus | [53,54,55] |
| Selenoprotein P | SELENOP | transport and store Se | ↓ | Secreted, cytosol; plasma, widely expressed, brain, liver, testes | [50,52] |
| 15 kDa selenoprotein | Sep15 | Regulates cell proliferation and cell cycle | ↑ | Brain, prostate, testis, liver, kidney | [57,60] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ye, R.; Huang, J.; Wang, Z.; Chen, Y.; Dong, Y. Trace Element Selenium Effectively Alleviates Intestinal Diseases. Int. J. Mol. Sci. 2021, 22, 11708. https://doi.org/10.3390/ijms222111708
Ye R, Huang J, Wang Z, Chen Y, Dong Y. Trace Element Selenium Effectively Alleviates Intestinal Diseases. International Journal of Molecular Sciences. 2021; 22(21):11708. https://doi.org/10.3390/ijms222111708
Chicago/Turabian StyleYe, Ruihua, Jiaqiang Huang, Zixu Wang, Yaoxing Chen, and Yulan Dong. 2021. "Trace Element Selenium Effectively Alleviates Intestinal Diseases" International Journal of Molecular Sciences 22, no. 21: 11708. https://doi.org/10.3390/ijms222111708
APA StyleYe, R., Huang, J., Wang, Z., Chen, Y., & Dong, Y. (2021). Trace Element Selenium Effectively Alleviates Intestinal Diseases. International Journal of Molecular Sciences, 22(21), 11708. https://doi.org/10.3390/ijms222111708







