Acute Cycling Exercise Induces Changes in Red Blood Cell Deformability and Membrane Lipid Remodeling
Abstract
1. Introduction
2. Results
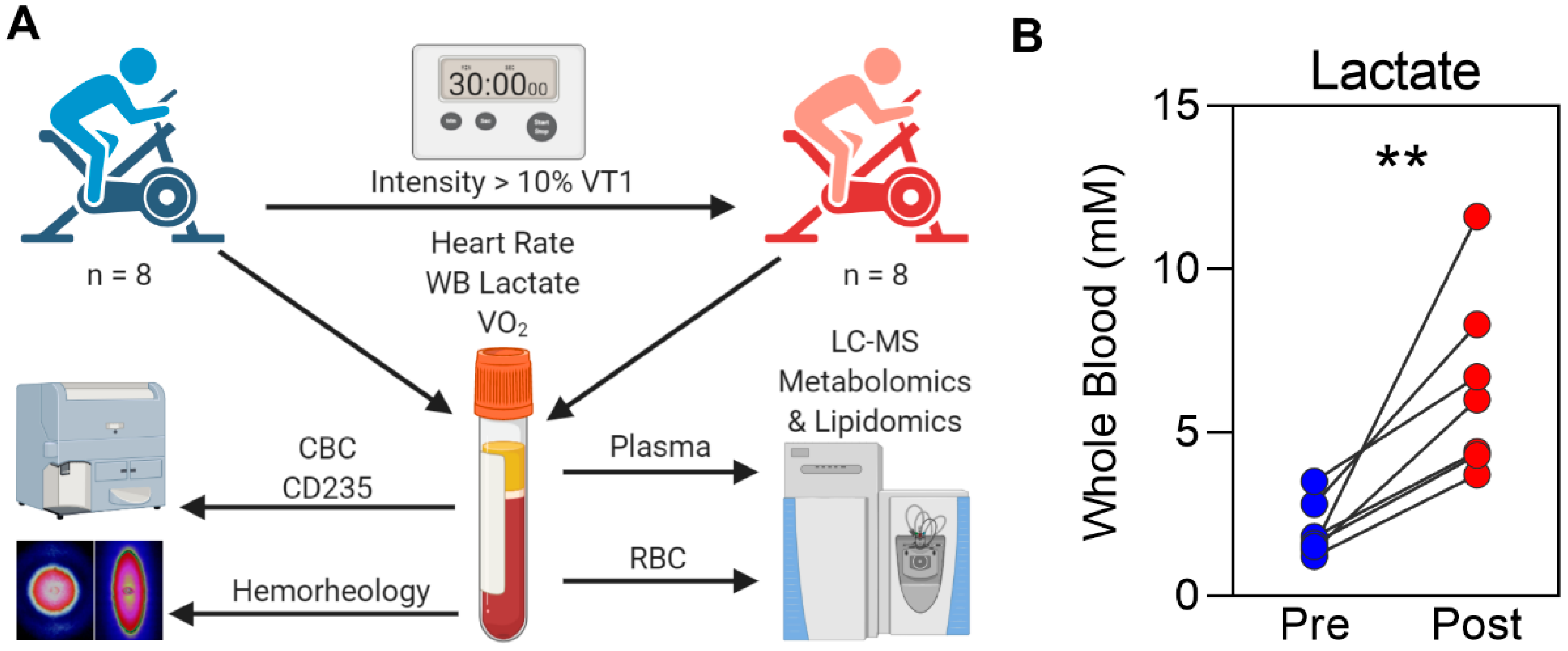
2.1. Exercise Testing
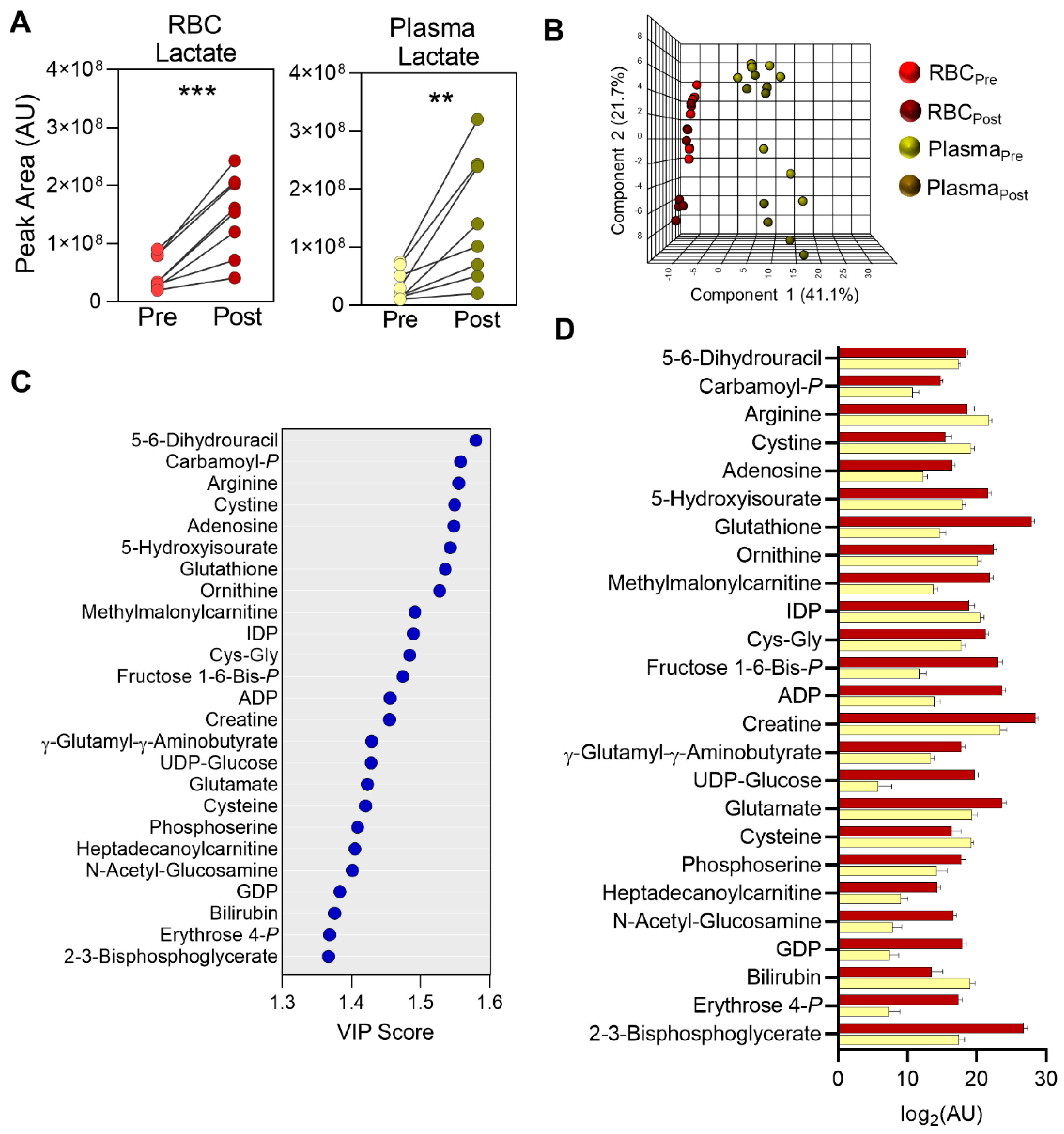
2.2. Distinct Metabolite Profiles in RBC and Plasma Fractions
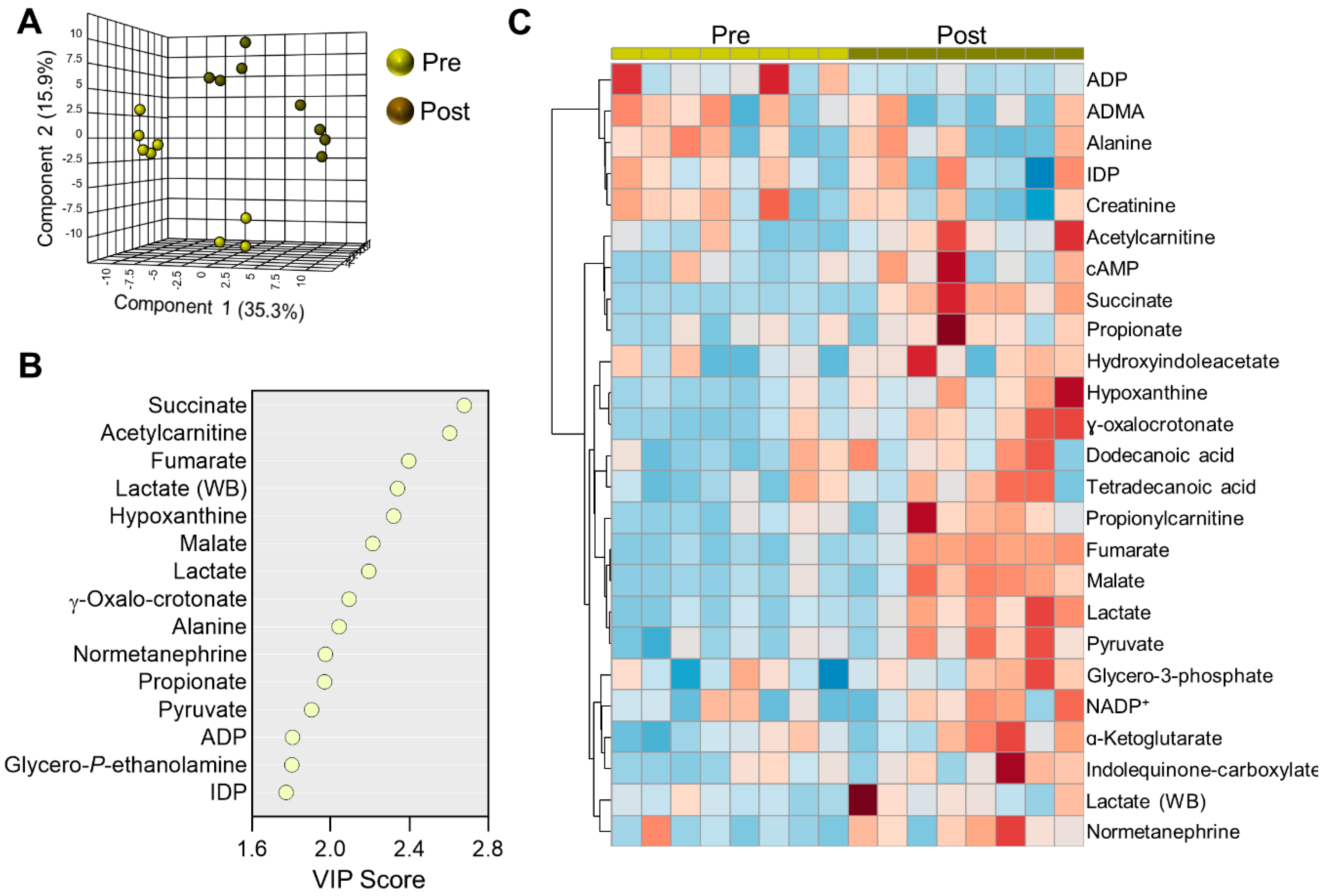
2.3. Acute Metabolic Alterations in Plasma during Cycling
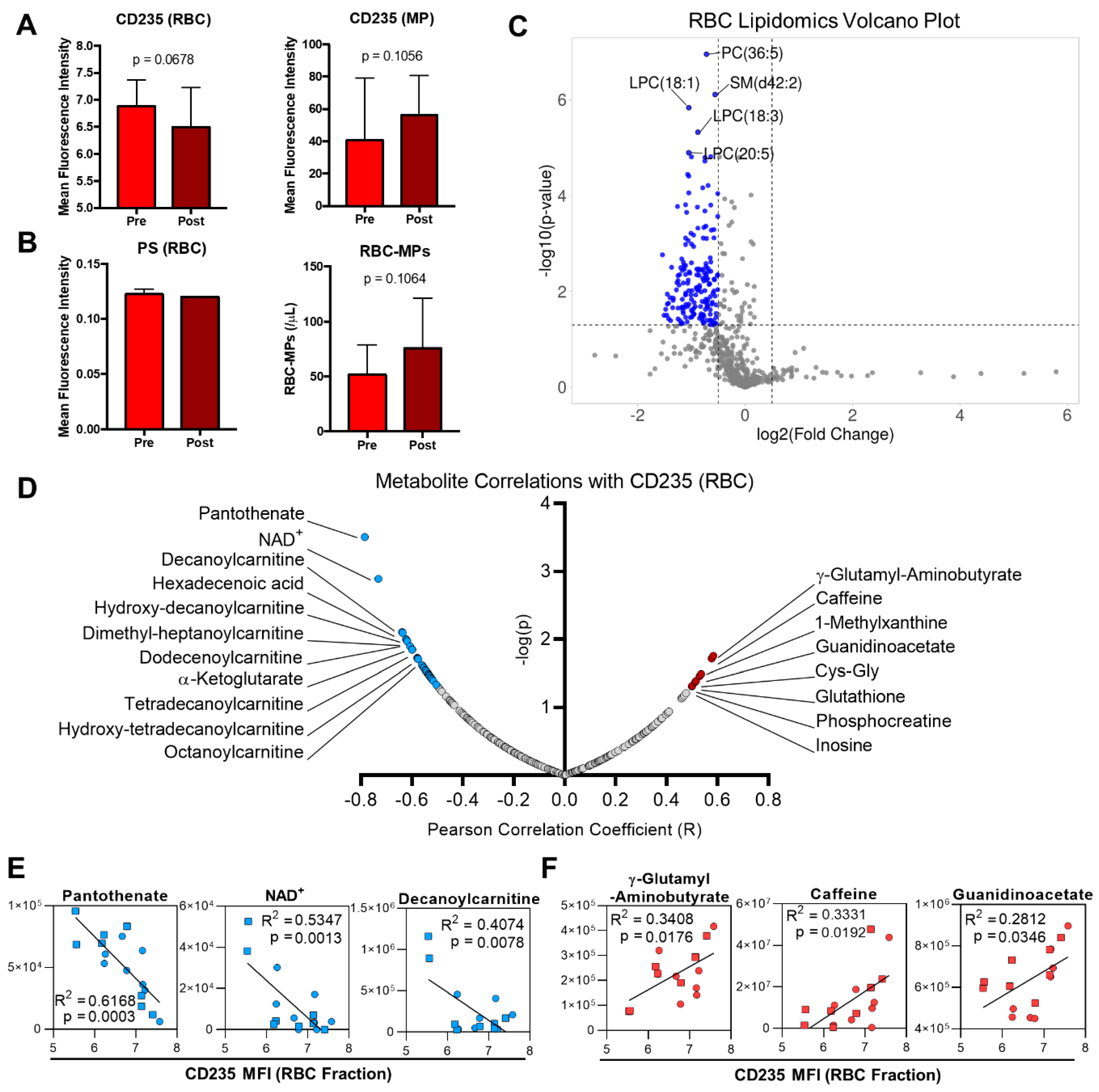
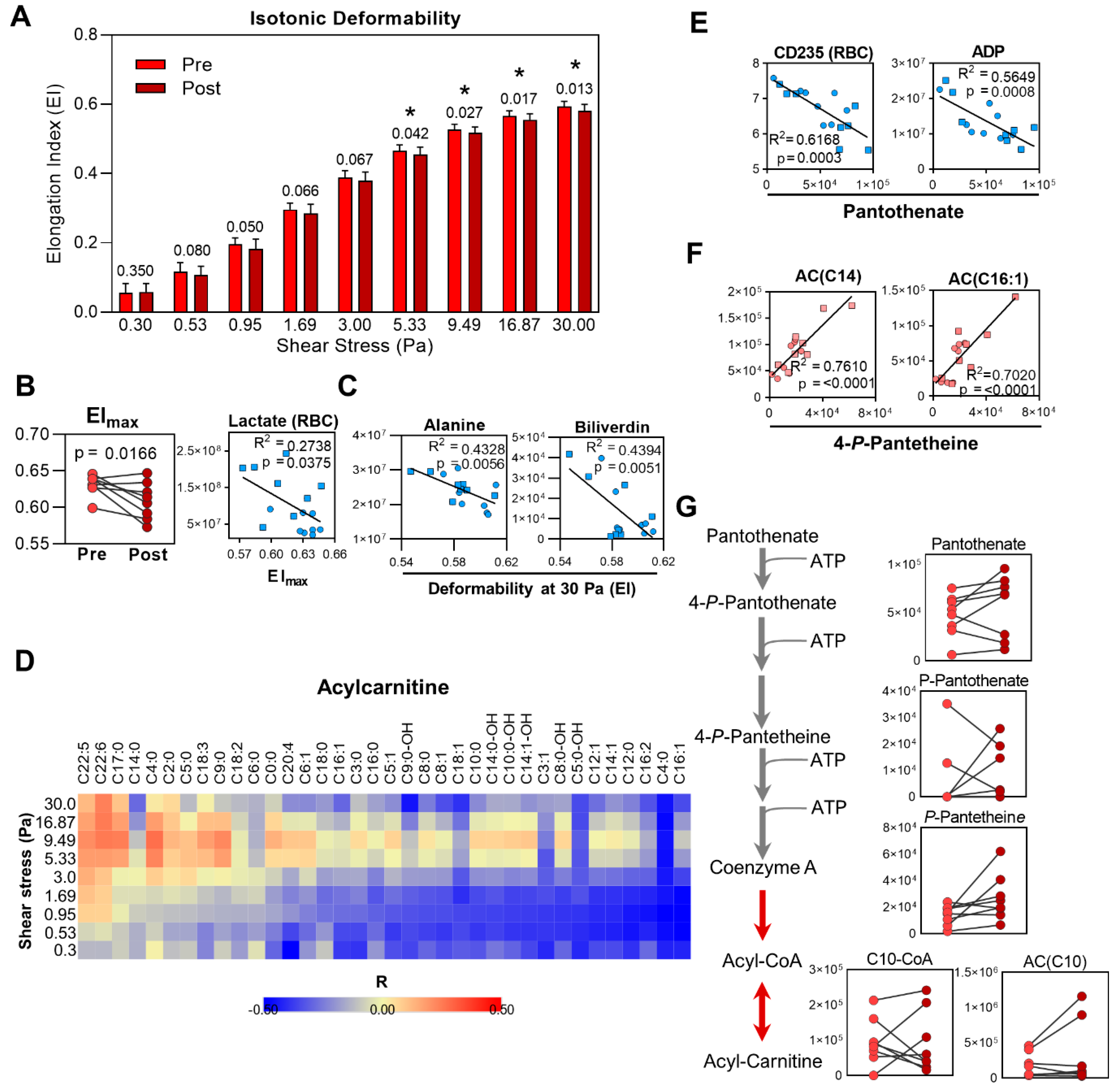
2.4. Metabolic Markers of Membrane Remodeling in RBC
2.5. Formation of Red Blood Cell Microparticles during Exercise
2.6. Increased White Blood Cells (WBCs), RBCs, Hemoglobin (Hb), and Hematocrit (HCT) Following Exercise
2.7. Reduced RBC Deformability and Increased RBC Aggregation during Exercise
3. Discussion
4. Materials and Methods
4.1. Study Subjects
4.2. Cycling Exercise
4.3. Incremental Maximal Cycling Test
4.4. Submaximal Cycling Exercise
4.5. Blood Sampling and Whole Blood Lactate Measurement
4.6. Metabolomics and Lipidomics Assessment
4.6.1. Sample Collection
4.6.2. Sample Preparation
4.7. UHPLC–MS Analysis for Metabolomics
4.8. UHPLC–MS Analysis for Lipidomics
4.9. Hematological and RBC Rheological Parameters
4.10. Analysis of PS and CD235a Exposure
4.11. MPs Extraction and Quantification
4.12. Statistical Analyses for Hematological, RBC Rheological, PS, CD235a, and MP Data
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| AC | Acylcarnitine |
| ADP | Adenosine diphosphate |
| ALT | Alanine aminotransferase |
| AMPD3 | Adenosine deaminase 3 |
| CoA | Coenzyme A |
| CD235a | Gycophorin A |
| CPT | Carnitine palmitoyltransferase |
| DPG | Diphosphoglycerate |
| EImax | Maximum elongation index |
| FA | Fatty acid |
| HCA | Hierarchical clustering analysis |
| IDP | Inosine diphosphate |
| LC-MS | Liquid chromatography–mass spectrometry |
| LPC | Lysophatidylcholine |
| LPLAT | Lysophospholipid acyltransferase |
| MAP | Maximal aerobic power |
| MCHC | Mean corpuscular hemoglobin concentration |
| MCV | Mean corpuscular volume |
| MFI | Mean fluorescence intensity |
| MP | Microparticle |
| NAD+PLS-DA | Nicotinamide adenine dinucleotidePartial least-squares discriminant analysis |
| PS | Phosphatidylserine |
| PVP | Polyvinylpyrrolidone |
| RBC | Red blood cell |
| RDW | Red blood cell distribution width |
| SpO2 | Blood oxygen saturation |
| TCA | Tricarboxylic acid |
| UDP | Uridine diphosphate |
| VO2max | Maximal oxygen uptake |
| VIP | Variable importance in project |
| VT1 | Ventilatory threshold 1 |
| WB | Whole blood |
| WBC | White blood cell |
References
- Cho, Y.-I.; Mooney, M.P.; Cho, D.J. Hemorheological Disorders in Diabetes Mellitus. J. Diabetes Sci. Technol. 2008, 2, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Connes, P.; Simmonds, M.J.; Brun, J.-F.; Baskurt, O.K. Exercise hemorheology: Classical data, recent findings and unresolved issues. Clin. Hemorheol. Microcirc. 2013, 53, 187–199. [Google Scholar] [CrossRef] [PubMed]
- Waltz, X.; Hardy-Dessources, M.-D.; Lemonne, N.; Mougenel, D.E.; Lalanne-Mistrih, M.-L.; Lamarre, Y.; Tarer, V.; Tressières, B.; Etienne-Julan, M.; Hue, O.; et al. Is there a relationship between the hematocrit-to-viscosity ratio and microvascular oxygenation in brain and muscle? Clin. Hemorheol. Microcirc. 2015, 59, 37–43. [Google Scholar] [CrossRef] [PubMed]
- Reinhart, W.H.; Stäubli, M.; Straub, P.W. Impaired red cell filterability with elimination of old red blood cells during a 100-km race. J. Appl. Physiol. 1983, 54, 827–830. [Google Scholar] [CrossRef] [PubMed]
- Galea, G.; Davidson, R. Hemorrheology of Marathon Running. Int. J. Sports Med. 1985, 6, 136–138. [Google Scholar] [CrossRef] [PubMed]
- Gueguen-Duchesne, M.; Durand, F.; Beillot, J.; Dezier, J.; Rochcongar, P.; Legoff, M. Could maximal exercise be a hemorhe-ological risk factor? Clin. Hemorheol. 1987, 7, 418. [Google Scholar]
- Brun, J.; Fons, C.; Supparo, I.; Mallard, C.; Orsetti, A. Could exercise-induced increase in blood viscosity at high shear rate be entirely explained by hematocrit and plasma viscosity changes? Clin. Hemorheol. Microcirc. 1993, 13, 187–199. [Google Scholar] [CrossRef]
- Brun, J.; Micallef, J.; Orsetti, A. Hemorheologic effects of light prolonged exercise. Clin. Hemorheol. Microcirc. 1994, 14, 807–818. [Google Scholar] [CrossRef]
- Oostenbrug, G.S.; Mensink, R.P.; Hardeman, M.R.; De Vries, T.; Brouns, F.; Hornstra, G. Exercise performance, red blood cell deformability, and lipid peroxidation: Effects of fish oil and vitamin E. J. Appl. Physiol. 1997, 83, 746–752. [Google Scholar] [CrossRef]
- Yalcin, O.; Bor-Kucukatay, M.; Senturk, U.K.; Baskurt, O.K. Effects of swimming exercise on red blood cell rheology in trained and untrained rats. J. Appl. Physiol. 2000, 88, 2074–2080. [Google Scholar] [CrossRef]
- Yalcin, O.; Erman, A.; Muratli, S.; Bor-Kucukatay, M.; Baskurt, O.K. Time course of hemorheological alterations after heavy anaerobic exercise in untrained human subjects. J. Appl. Physiol. 2003, 94, 997–1002. [Google Scholar] [CrossRef] [PubMed]
- Van Beaumont, W.; Underkofler, S.; Van Beaumont, S. Erythrocyte volume, plasma volume, and acid-base changes in exercise and heat dehydration. J. Appl. Physiol. 1981, 50, 1255–1262. [Google Scholar] [CrossRef] [PubMed]
- Lipovac, V.; Gavella, M.; Turk, Z.; Škrabalo, Z. Influence of lactate on the insulin action on red blood cell filterability. Clin. Hemorheol. Microcirc. 2016, 5, 421–428. [Google Scholar] [CrossRef]
- Smith, J.A.; Telford, R.D.; Kolbuch-Braddon, M.; Weidemann, M.J. Lactate/H + uptake by red blood cells during exercise alters their physical properties. Graefe’s Arch. Clin. Exp. Ophthalmol. 1996, 75, 54–61. [Google Scholar] [CrossRef] [PubMed]
- Connes, P.; Bouix, D.; Py, G.; Prefaut, C.; Mercier, J.; Brun, J.-F.; Caillaud, C. Opposite effects of in vitro lactate on erythro-cyte deformability in athletes and untrained subjects. Clin. Hemorheol. Microcirc. 2004, 31, 311–318. [Google Scholar] [PubMed]
- Şentürk, Ü.K.; Gündüz, F.; Kuru, O.; Koçer, G.; Özkaya, Y.G.; Yeşilkaya, A.; Bor-Küçükatay, M.; Üyüklü, M.; Yalcin, O.; Baskurt, O.K.; et al. Exercise-induced oxidative stress leads hemolysis in sedentary but not trained humans. J. Appl. Physiol. 2005, 99, 1434–1441. [Google Scholar] [CrossRef]
- Lang, E.; Lang, F. Triggers, Inhibitors, Mechanisms, and Significance of Eryptosis: The Suicidal Erythrocyte Death. BioMed Res. Int. 2015, 2015, 513518. [Google Scholar] [CrossRef]
- Nemkov, T.; Qadri, S.M.; Sheffield, W.P.; D’Alessandro, A. Decoding the metabolic landscape of pathophysiological stress-induced cell death in anucleate red blood cells. Blood Transfus. 2020, 18, 130–142. [Google Scholar]
- Highton, P.J.; Martin, N.; Smith, A.C.; Burton, J.O.; Bishop, N.C. Microparticles and Exercise in Clinical Populations. Exerc. Immunol. Rev. 2018, 24, 46–58. [Google Scholar]
- Suvorava, T.; Cortese-Krott, M.M. Exercise-Induced Cardioprotection via eNOS: A Putative Role of Red Blood Cell Signaling. Curr. Med. Chem. 2018, 25, 4457–4474. [Google Scholar] [CrossRef]
- Buerck, J.P.; Burke, D.K.; Schmidtke, D.W.; Snyder, T.A.; Papavassiliou, D.V.; O’Rear, E.A. A Flow Induced Autoimmune Response and Accelerated Senescence of Red Blood Cells in Cardiovascular Devices. Sci. Rep. 2019, 9, 19443. [Google Scholar] [CrossRef] [PubMed]
- Chaar, V.; Romana, M.; Tripette, J.; Broquere, C.; Huisse, M.-G.; Hue, O.; Hardy-Dessources, M.-D.; Connes, P. Effect of strenuous physical exercise on circulating cell-derived microparticles. Clin. Hemorheol. Microcirc. 2011, 47, 15–25. [Google Scholar] [CrossRef] [PubMed]
- Nader, E.; Guillot, N.; Lavorel, L.; Hancco, I.; Fort, R.; Stauffer, E.; Renoux, C.; Joly, P.; Germain, M.; Connes, P. Eryptosis and hemorheological responses to maximal exercise in athletes: Comparison between running and cycling. Scand. J. Med. Sci. Sports 2018, 28, 1532–1540. [Google Scholar] [CrossRef] [PubMed]
- Nader, E.; Monedero, D.; Robert, M.; Skinner, S.; Stauffer, E.; Cibiel, A.; Germain, M.; Hugonnet, J.; Scheer, A.; Joly, P.; et al. Impact of a 10 km running trial on eryptosis, red blood cell rheology, and electrophysiology in endurance trained athletes: A pilot study. Graefe’s Arch. Clin. Exp. Ophthalmol. 2019, 120, 255–266. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, T.; Prudent, M.; D’Alessandro, A. Red blood cell storage lesion: Causes and potential clinical consequences. Blood Transfus. 2019, 17, 27–52. [Google Scholar]
- Nader, E.; Romana, M.; Guillot, N.; Fort, R.; Stauffer, E.; Lemonne, N.; Garnier, Y.; Skinner, S.C.; Etienne-Julan, M.; Robert, M.; et al. Association Between Nitric Oxide, Oxidative Stress, Eryptosis, Red Blood Cell Microparticles, and Vascular Function in Sickle Cell Anemia. Front. Immunol. 2020, 11, 551441. [Google Scholar] [CrossRef]
- D’Alessandro, A.; Nemkov, T.; Reisz, J.; Dzieciatkowska, M.; Wither, M.J.; Hansen, K.C. Omics markers of the red cell storage lesion and metabolic linkage. High Speed Blood Transfus. Equip. 2017, 15, 137–144. [Google Scholar]
- Sakaguchi, C.A.; Nieman, D.C.; Signini, E.F.; Abreu, R.M.; Catai, A.M. Metabolomics-Based Studies Assessing Exercise-Induced Alterations of the Human Metabolome: A Systematic Review. Metabolites 2019, 9, 164. [Google Scholar] [CrossRef]
- Heaney, L.M.; Deighton, K.; Suzuki, T. Non-targeted metabolomics in sport and exercise science. J. Sports Sci. 2017, 37, 959–967. [Google Scholar] [CrossRef]
- Nieman, D.; Scherr, J.; Luo, B.; Meaney, M.P.; Dréau, D.; Sha, W.; Dew, D.A.; Henson, D.A.; Pappan, K.L. Influence of Pistachios on Performance and Exercise-Induced Inflammation, Oxidative Stress, Immune Dysfunction, and Metabolite Shifts in Cyclists: A Randomized, Crossover Trial. PLoS ONE 2014, 9, e113725. [Google Scholar] [CrossRef]
- Nieman, D.; Gillitt, N.D.; Sha, W.; Meaney, M.P.; John, C.; Pappan, K.L.; Kinchen, J.M. Metabolomics-Based Analysis of Banana and Pear Ingestion on Exercise Performance and Recovery. J. Proteome Res. 2015, 14, 5367–5377. [Google Scholar] [CrossRef] [PubMed]
- Pechlivanis, A.; Kostidis, S.; Saraslanidis, P.; Petridou, A.; Tsalis, G.; Veselkov, K.; Mikros, E.; Mougios, V.; Theodoridis, G.A. 1H NMR Study on the Short- and Long-Term Impact of Two Training Programs of Sprint Running on the Metabolic Fingerprint of Human Serum. J. Proteome Res. 2012, 12, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Enea, C.; Seguin, F.; Petitpas-Mulliez, J.; Boildieu, N.; Boisseau, N.; Delpech, N.; Diaz, V.; Eugene, M.; Dugué, B. 1H NMR-based metabolomics approach for exploring urinary metabolome modifications after acute and chronic physical exercise. Anal. Bioanal. Chem. 2010, 396, 1167–1176. [Google Scholar] [CrossRef] [PubMed]
- Berton, R.; Conceição, M.S.; Libardi, C.A.; Canevarolo, R.R.; Gáspari, A.F.; Chacon-Mikahil, M.P.T.; Zeri, A.C.; Cavaglieri, C.R. Metabolic time-course response after resistance exercise: A metabolomics approach. J. Sports Sci. 2016, 35, 1211–1218. [Google Scholar] [CrossRef]
- Contrepois, K.; Wu, S.; Moneghetti, K.J.; Hornburg, D.; Ahadi, S.; Tsai, M.-S.; Metwally, A.A.; Wei, E.; Lee-McMullen, B.; Quijada, J.V.; et al. Molecular Choreography of Acute Exercise. Cell 2020, 181, 1112–1130. [Google Scholar] [CrossRef]
- San-Millán, I.; Stefanoni, D.; Martinez, J.L.; Hansen, K.C.; D’Alessandro, A.; Nemkov, T. Metabolomics of Endurance Capac-ity in World Tour Professional Cyclists. Front. Physiol. 2020, 11, 578. [Google Scholar] [CrossRef]
- Meen, H.D.; Holter, P.H.; Refsum, H.E. Changes in 2,3-diphosphoglycerate (2,3-DPG) after exercise. Graefe’s Arch. Clin. Exp. Ophthalmol. 1981, 46, 177–184. [Google Scholar] [CrossRef]
- Liu, H.; Zhang, Y.; Wu, H.; D’Alessandro, A.; Yegutkin, G.G.; Song, A.; Sun, K.; Li, J.; Cheng, N.-Y.; Huang, A.; et al. Beneficial Role of Erythrocyte Adenosine A2B Receptor–Mediated AMP-Activated Protein Kinase Activation in High-Altitude Hypoxia. Circulation 2016, 134, 405–421. [Google Scholar] [CrossRef]
- Reisz, J.A.; Slaughter, A.L.; Culp-Hill, R.; Moore, E.E.; Silliman, C.C.; Fragoso, M.; Peltz, E.D.; Hansen, K.C.; Banerjee, A.; D’Alessandro, A. Red blood cells in hemorrhagic shock: A critical role for glutaminolysis in fueling alanine transamination in rats. Blood Adv. 2017, 1, 1296–1305. [Google Scholar] [CrossRef][Green Version]
- Nemkov, T.; Sun, K.; Reisz, J.A.; Yoshida, T.; Dunham, A.; Wen, E.Y.; Wen, A.Q.; Roach, R.C.; Hansen, K.C.; Xia, Y.; et al. Metabolism of Citrate and Other Carboxylic Acids in Erythrocytes As a Function of Oxygen Saturation and Refrigerated Storage. Front. Med. 2017, 4, 175. [Google Scholar] [CrossRef]
- Leal, J.K.F.; Adjobo-Hermans, M.J.W.; Bosman, G.J.C.G.M. Red Blood Cell Homeostasis: Mechanisms and Effects of Microvesicle Generation in Health and Disease. Front. Physiol. 2018, 9, 703. [Google Scholar] [CrossRef] [PubMed]
- Gray, A.B.; Telford, R.D.; Collins, M.; Weidemann, M.J. The response of leukocyte subsets and plasma hormones to interval exercise. Med. Sci. Sports Exerc. 1993, 25, 1252–1258. [Google Scholar] [CrossRef] [PubMed]
- Sand, K.L.; FlatebO, T.; Andersen, M.B.; Maghazachi, A.A. Effects of exercise on leukocytosis and blood hemostasis in 800 healthy young females and males. World J. Exp. Med. 2013, 3, 11–20. [Google Scholar] [CrossRef] [PubMed]
- Heber, S.; Volf, I. Effects of Physical (In)activity on Platelet Function. BioMed Res. Int. 2015, 2015, 165078. [Google Scholar] [CrossRef] [PubMed]
- Mairbäurl, H. Red blood cells in sports: Effects of exercise and training on oxygen supply by red blood cells. Front. Physiol. 2013, 4, 332. [Google Scholar] [CrossRef]
- Simmonds, M.J.; Connes, P.; Sabapathy, S. Exercise-Induced Blood Lactate Increase Does Not Change Red Blood Cell Deformability in Cyclists. PLoS ONE 2013, 8, e71219. [Google Scholar] [CrossRef][Green Version]
- González-Alonso, J.; Olsen, D.B.; Saltin, B. Erythrocyte and the Regulation of Human Skeletal Muscle Blood Flow and Oxygen Delivery. Circ. Res. 2002, 91, 1046–1055. [Google Scholar] [CrossRef]
- Odje, O.E.; Ramsey, J.M. Effect of short-term strenuous exercise on erythrocyte 2,3-diphosphoglycerate in untrained men: A time-course study. Graefe’s Arch. Clin. Exp. Ophthalmol. 1995, 71, 53–57. [Google Scholar] [CrossRef]
- Hess, J.R.; Kagen, L.R.; Van Der Meer, P.F.; Simon, T.; Cardigan, R.; Greenwalt, T.J.; AuBuchon, J.P.; Brand, A.; Lockwood, W.; Zanella, A.; et al. Interlaboratory comparison of red-cell ATP, 2,3-diphosphoglycerate and haemolysis measurements. Vox Sang. 2005, 89, 44–48. [Google Scholar] [CrossRef]
- D’Alessandro, A.; Nemkov, T.; Sun, K.; Liu, H.; Song, A.; Monte, A.A.; Subudhi, A.W.; Lovering, A.T.; Dvorkin, D.; Julian, C.G.; et al. AltitudeOmics: Red Blood Cell Metabolic Adaptation to High Altitude Hypoxia. J. Proteome Res. 2016, 15, 3883–3895. [Google Scholar] [CrossRef]
- Sun, K.; Zhang, Y.; D’Alessandro, A.; Nemkov, T.; Song, A.; Wu, H.; Liu, H.; Adebiyi, M.; Huang, A.; Wen, Y.E.; et al. Sphingosine-1-phosphate promotes erythrocyte glycolysis and oxygen release for adaptation to high-altitude hypoxia. Nat. Commun. 2016, 7, 12086. [Google Scholar] [CrossRef] [PubMed]
- Song, A.; Zhang, Y.; Han, L.; Yegutkin, G.G.; Liu, H.; Sun, K.; D’Alessandro, A.; Li, J.; Karmouty-Quintana, H.; Iriyama, T.; et al. Erythrocytes retain hypoxic adenosine response for faster acclimatization upon re-ascent. Nat. Commun. 2017, 8, 14108. [Google Scholar] [CrossRef] [PubMed]
- Nemkov, T.; Sun, K.; Reisz, J.A.; Song, A.; Yoshida, T.; Dunham, A.; Wither, M.J.; Francis, R.O.; Roach, R.C.; Dzieciatkowska, M.; et al. Hypoxia modulates the purine salvage pathway and decreases red blood cell and supernatant levels of hypoxanthine during refrigerated storage. Haematologica 2017, 103, 361–372. [Google Scholar] [CrossRef] [PubMed]
- Kendrick, A.A.; Schafer, J.; Dzieciatkowska, M.; Nemkov, T.; D’Alessandro, A.; Neelakantan, D.; Ford, H.L.; Pearson, C.G.; Weekes, C.D.; Hansen, K.C.; et al. CD147: A small molecule transporter ancillary protein at the crossroad of multiple hallmarks of cancer and metabolic reprogramming. Oncotarget 2016, 8, 6742–6762. [Google Scholar] [CrossRef]
- Nemkov, T.; Hansen, K.C.; D’Alessandro, A. A three-minute method for high-throughput quantitative metabolomics and quantitative tracing experiments of central carbon and nitrogen pathways. Rapid Commun. Mass Spectrom. 2017, 31, 663–673. [Google Scholar] [CrossRef]
- Willekens, F.L.A.; Werre, J.M.; Groenen-Döpp, Y.A.M.; Roerdinkholder-Stoelwinder, B.; De Pauw, B.; Bosman, G.J.C.G.M. Erythrocyte vesiculation: A self-protective mechanism? Br. J. Haematol. 2008, 141, 549–556. [Google Scholar] [CrossRef]
- Tavazzi, B.; Amorini, A.M.; Fazzina, G.; Di Pierro, D.; Tuttobene, M.; Giardina, B.; Lazzarino, G. Oxidative Stress Induces Impairment of Human Erythrocyte Energy Metabolism through the Oxygen Radical-mediated Direct Activation of AMP-deaminase. J. Biol. Chem. 2001, 276, 48083–48092. [Google Scholar] [CrossRef]
- Sabina, R.; Wandersee, N.J.; Hillery, C.A. Ca2+-CaM activation of AMP deaminase contributes to adenine nucleotide dysregulation and phosphatidylserine externalization in human sickle erythrocytes. Br. J. Haematol. 2009, 144, 434–445. [Google Scholar] [CrossRef]
- Clark, M.R.; Mohandas, N.; Shohet, S.B. Osmotic gradient ektacytometry: Comprehensive characterization of red cell vol-ume and surface maintenance. Blood 1983, 61, 899–910. [Google Scholar] [CrossRef]
- Renoux, C.; Faivre, M.; Bessaa, A.; Da Costa, L.; Joly, P.; Gauthier, A.; Connes, P. Impact of surface-area-to-volume ratio, internal viscosity and membrane viscoelasticity on red blood cell deformability measured in isotonic condition. Sci. Rep. 2019, 9, 6771. [Google Scholar] [CrossRef]
- Lands, W.E. Metabolism of Glycerolipides: A Comparison of Lecithin and Triglyceride Synthesis. J. Biol. Chem. 1958, 231, 883–888. [Google Scholar] [CrossRef]
- Arduini, A.; Mancinelli, G.; Radatti, G.L.; Dottori, S.; Molajoni, F.; Ramsay, R.R. Role of carnitine and carnitine palmito-yltransferase as integral components of the pathway for membrane phospholipid fatty acid turnover in intact human erythrocytes. J. Biol. Chem. 1992, 267, 12673–12681. [Google Scholar] [CrossRef]
- Reyes, B.D.L.; Perez-García, R.; Liras, A.; Arenas, J. Reduced carnitine palmitoyl transferase activity and altered acyl-trafficking in red blood cells from hemodialysis patients. Biochim. Biophys. Acta (BBA)-Mol. Basis Dis. 1996, 1315, 37–39. [Google Scholar] [CrossRef]
- Wu, H.; Bogdanov, M.; Zhang, Y.; Sun, K.; Zhao, S.; Song, A.; Luo, R.; Parchim, N.F.; Liu, H.; Huang, A.; et al. Hypoxia-mediated impaired erythrocyte Lands’ Cycle is pathogenic for sickle cell disease. Sci. Rep. 2016, 6, 29637. [Google Scholar] [CrossRef]
- Annous, K.F.; O Song, W. Pantothenic acid uptake and metabolism by red blood cells of rats. J. Nutr. 1995, 125, 2586–2593. [Google Scholar] [PubMed]
- Brohn, F.H.; Trager, W. Coenzyme A requirement of malaria parasites: Enzymes of coenzyme A biosynthesis in normal duck erythrocytes and erythrocytes infected with Plasmodium lophurae. Proc. Natl. Acad. Sci. USA 1975, 72, 2456–2458. [Google Scholar] [CrossRef]
- Shemin, D.; Rittenberg, D. The life span of the human red blood cell. J. Biol. Chem. 1946, 166, 627–636. [Google Scholar]
- Kaestner, L.; Bogdanova, A. Regulation of red cell life-span, erythropoiesis, senescence, and clearance. Front. Physiol. 2014, 5, 5. [Google Scholar] [CrossRef][Green Version]
- Reisz, J.A.; Nemkov, T.; Dzieciatkowska, M.; Culp-Hill, R.; Stefanoni, D.; Hill, R.C.; Yoshida, T.; Dunham, A.; Kanias, T.; Dumont, L.J.; et al. Methylation of protein aspartates and deamidated asparagines as a function of blood bank storage and oxidative stress in human red blood cells. Transfusion 2018, 58, 2978–2991. [Google Scholar] [CrossRef]
- D’Alessandro, A.; Fu, X.; Reisz, J.A.; Kanias, T.; Page, G.P.; Stone, M.; Kleinman, S.; Zimring, J.C.; Busch, M.; For the Recipient Epidemiology and Donor Evaluation Study-III (REDS III). Stored RBC metabolism as a function of caffeine levels. Transfusion 2020, 60, 1197–1211. [Google Scholar] [CrossRef]
- Thomas, T.; Stefanoni, D.; Dzieciatkowska, M.; Issaian, A.; Nemkov, T.; Hill, R.C.; Francis, R.O.; Hudson, K.E.; Buehler, P.W.; Zimring, J.C.; et al. Evidence of Structural Protein Damage and Membrane Lipid Remodeling in Red Blood Cells from COVID-19 Patients. J. Proteome Res. 2020, 19, 4455–4469. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A. Exercise, Training and Red Blood Cell Turnover. Sports Med. 1995, 19, 9–31. [Google Scholar] [CrossRef] [PubMed]
- Bizjak, D.A.; Tomschi, F.; Bales, G.; Nader, E.; Romana, M.; Connes, P.; Bloch, W.; Grau, M. Does endurance training improve red blood cell aging and hemorheology in moderate-trained healthy individuals? J. Sport Health Sci. 2020, 9, 595–603. [Google Scholar] [CrossRef] [PubMed]
- Smith, J.A.; Martin, D.T.; Telford, R.D.; Ballas, S.K. Greater erythrocyte deformability in world-class endurance athletes. Am. J. Physiol. Circ. Physiol. 1999, 276, H2188–H2193. [Google Scholar] [CrossRef]
- Marin, M.; Roussel, C.; Dussiot, M.; Ndour, P.A.; Hermine, O.; Colin, Y.; Gray, A.; Landrigan, M.; Le Van Kim, C.; Buffet, P.A.; et al. Metabolic rejuvenation upgrades circulatory functions of red blood cells stored under blood bank conditions. Transfusion 2020, 16245. [Google Scholar] [CrossRef]
- Henry, B.; Roussel, C.; Carucci, M.; Brousse, V.; Ndour, P.A.; Buffet, P.A. The Human Spleen in Malaria: Filter or Shelter? Trends Parasitol. 2020, 36, 435–446. [Google Scholar] [CrossRef]
- D’Alessandro, A.; Blasi, B.; D’Amici, G.M.; Marrocco, C.; Zolla, L. Red blood cell subpopulations in freshly drawn blood: Application of proteomics and metabolomics to a decades-long biological issue. High Speed Blood Transfus. Equip. 2012, 11, 75–87. [Google Scholar]
- Jamshidi, N.; Xu, X.; Von Löhneysen, K.; Soldau, K.; Mohney, R.P.; Karoly, E.D.; Scott, M.; Friedman, J.S. Metabolome Changes during In Vivo Red Cell Aging Reveal Disruption of Key Metabolic Pathways. iScience 2020, 23, 101630. [Google Scholar] [CrossRef]
- Mykhailova, O.; Olafson, C.; Turner, T.R.; D’Alessandro, A.; Acker, J.P. Donor-dependent aging of young and old red blood cell subpopulations: Metabolic and functional heterogeneity. Transfusion 2020, 60, 2633–2646. [Google Scholar] [CrossRef]
- Alexander, K.; Hazegh, K.; Fang, F.; Sinchar, D.; Kiss, J.E.; Page, G.P.; Dʼalessandro, A.; Kanias, T. Testosterone replacement therapy in blood donors modulates erythrocyte metabolism and susceptibility to hemolysis in cold storage. Transfusion 2021, 61, 108–123. [Google Scholar] [CrossRef]
- D’Alessandro, A.; Fu, X.; Kanias, T.; Reisz, J.A.; Culp-Hill, R.; Guo, Y.; Gladwin, M.T.; Page, G.; Kleinman, S.; Lanteri, M.; et al. Donor sex, age and ethnicity impact stored red blood cell antioxidant metabolism through mechanisms in part explained by glucose 6-phosphate dehydrogenase levels and activity. Haematologica 2020. [Google Scholar] [CrossRef] [PubMed]
- Song, J.; Yoon, D.; Christensen, R.D.; Horvathova, M.; Thiagarajan, P.; Prchal, J.T. HIF-mediated increased ROS from reduced mitophagy and decreased catalase causes neocytolysis. J. Mol. Med. 2015, 93, 857–866. [Google Scholar] [CrossRef] [PubMed]
- Mairbäurl, H. Neocytolysis: How to Get Rid of the Extra Erythrocytes Formed by Stress Erythropoiesis Upon Descent From High Altitude. Front. Physiol. 2018, 9, 345. [Google Scholar] [CrossRef] [PubMed]
- Gaskill, S.E.; Ruby, B.C.; Walker, A.J.; Sanchez, O.A.; Serfass, R.C.; Leon, A.S. Validity and reliability of combining three methods to determine ventilatory threshold. Med. Sci. Sports Exerc. 2001, 33, 1841–1848. [Google Scholar] [CrossRef]
- Nemkov, T.; Reisz, J.A.; Gehrke, S.; Hansen, K.C.; D’Alessandro, A. High-Throughput Metabolomics: Isocratic and Gradient Mass Spectrometry-Based Methods. Methods Mol. Biol. 2019, 1978, 13–26. [Google Scholar] [CrossRef]
- Chong, J.; Soufan, O.; Li, C.; Caraus, I.; Li, S.; Bourque, G.; Wishart, D.S.; Xia, J. MetaboAnalyst 4.0: Towards more transparent and integrative metabolomics analysis. Nucleic Acids Res. 2018, 46, W486–W494. [Google Scholar] [CrossRef]
- Reisz, J.A.; Zheng, C.; D’Alessandro, A.; Nemkov, T. Untargeted and Semi-targeted Lipid Analysis of Biological Samples Using Mass Spectrometry-Based Metabolomics. Comput. Biol. 2019, 1978, 121–135. [Google Scholar] [CrossRef]
- Baskurt, O.K. (Ed.) Handbook of Hemorheology and Hemodynamics; IOS Press: Amsterdam, The Netherlands; Washington, DC, USA, 2007. [Google Scholar]
- Baskurt, O.K.; Meiselman, H.J. Data reduction methods for ektacytometry in clinical hemorheology. Clin. Hemorheol. Microcirc. 2013, 54, 99–107. [Google Scholar] [CrossRef]
- Nébor, D.; Romana, M.; Santiago, R.; Vachiery, N.; Picot, J.; Broquere, C.; Chaar, V.; Doumdo, L.; Odièvre, M.-H.; Benkerrou, M.; et al. Fetal hemoglobin and hydroxycarbamide moduate both plasma concentration and cellular origin of circulating microparticles in sickle cell anemia children. Haematologica 2013, 98, 862–867. [Google Scholar] [CrossRef]

| n | 8 |
|---|---|
| Age (years) | 35 ± 8 |
| Weight (kg) | 65.8 ± 6.4 |
| Height (cm) | 176.1 ±2.2 |
| VO2max (mL/min/kg) | 70.5 ± 5.8 |
| HRmax (BPM) | 188 ± 11 |
| MAP (W) | 373 ± 39 |
| Percent HRmax at VT1 (%) | 85.95 ± 3.4 |
| Percent MAP at VT1 (%) | 69.88 ± 5.0 |
| Exercise duration (min) | 20.1 ± 1.8 |
| Pre | Post | |
|---|---|---|
| Aggregation M | 4.95 ± 2.47 | 6.03 ± 1.35 * |
| Aggregation M1 | 8.35 ± 2.71 | 9.76 ± 1.30 * |
| WBCs (103/uL) | 4.84 ± 0.93 | 6.76 ± 1.34 ** |
| RBCs (106/uL) | 4.76 ± 0.30 | 5.01 ± 0.42 * |
| HGB (g/dL) | 14.30 ± 0.90 | 15.11 ± 1.35 * |
| HCT % | 43.26 ± 2.94 | 45.56 ± 3.84 * |
| MCV fL | 90.90 ± 3.21 | 90.97 ± 2.77 |
| MCH pg | 30.06 ± 0.96 | 30.16 ± 0.69 |
| MCHC g/dL | 33.07 ± 0.74 | 33.05 ± 0.86 |
| RDW-SD fL | 43.27 ± 2.06 | 43.26 ± 1.84 |
| PLTs (103/uL) | 204.75 ± 43.13 | 237.25 ± 60.92 |
| Alanine | Biliverdin | |||
|---|---|---|---|---|
| Shear Stress | R2 | p-Value | R2 | p-Value |
| 0.3 Pa | 0.1739 | 0.108 | 0.2426 | 0.053 |
| 0.53 Pa | 0.2507 | 0.049 | 0.3691 | 0.013 |
| 0.95 Pa | 0.4008 | 0.009 | 0.4506 | 0.004 |
| 1.69 Pa | 0.3765 | 0.012 | 0.3888 | 0.01 |
| 3 Pa | 0.3290 | 0.02 | 0.3150 | 0.024 |
| 5.33 Pa | 0.4162 | 0.007 | 0.3115 | 0.024 |
| 9.49 Pa | 0.4270 | 0.006 | 0.2878 | 0.032 |
| 16.87 Pa | 0.4630 | 0.004 | 0.3684 | 0.013 |
| 30 Pa | 0.4354 | 0.006 | 0.4398 | 0.005 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Nemkov, T.; Skinner, S.C.; Nader, E.; Stefanoni, D.; Robert, M.; Cendali, F.; Stauffer, E.; Cibiel, A.; Boisson, C.; Connes, P.; et al. Acute Cycling Exercise Induces Changes in Red Blood Cell Deformability and Membrane Lipid Remodeling. Int. J. Mol. Sci. 2021, 22, 896. https://doi.org/10.3390/ijms22020896
Nemkov T, Skinner SC, Nader E, Stefanoni D, Robert M, Cendali F, Stauffer E, Cibiel A, Boisson C, Connes P, et al. Acute Cycling Exercise Induces Changes in Red Blood Cell Deformability and Membrane Lipid Remodeling. International Journal of Molecular Sciences. 2021; 22(2):896. https://doi.org/10.3390/ijms22020896
Chicago/Turabian StyleNemkov, Travis, Sarah C. Skinner, Elie Nader, Davide Stefanoni, Mélanie Robert, Francesca Cendali, Emeric Stauffer, Agnes Cibiel, Camille Boisson, Philippe Connes, and et al. 2021. "Acute Cycling Exercise Induces Changes in Red Blood Cell Deformability and Membrane Lipid Remodeling" International Journal of Molecular Sciences 22, no. 2: 896. https://doi.org/10.3390/ijms22020896
APA StyleNemkov, T., Skinner, S. C., Nader, E., Stefanoni, D., Robert, M., Cendali, F., Stauffer, E., Cibiel, A., Boisson, C., Connes, P., & D’Alessandro, A. (2021). Acute Cycling Exercise Induces Changes in Red Blood Cell Deformability and Membrane Lipid Remodeling. International Journal of Molecular Sciences, 22(2), 896. https://doi.org/10.3390/ijms22020896








