The Effects of Sperm and Seminal Fluid of Immunized Male Mice on In Vitro Fertilization and Surrogate Mother–Embryo Interaction
Abstract
:1. Introduction
2. Results
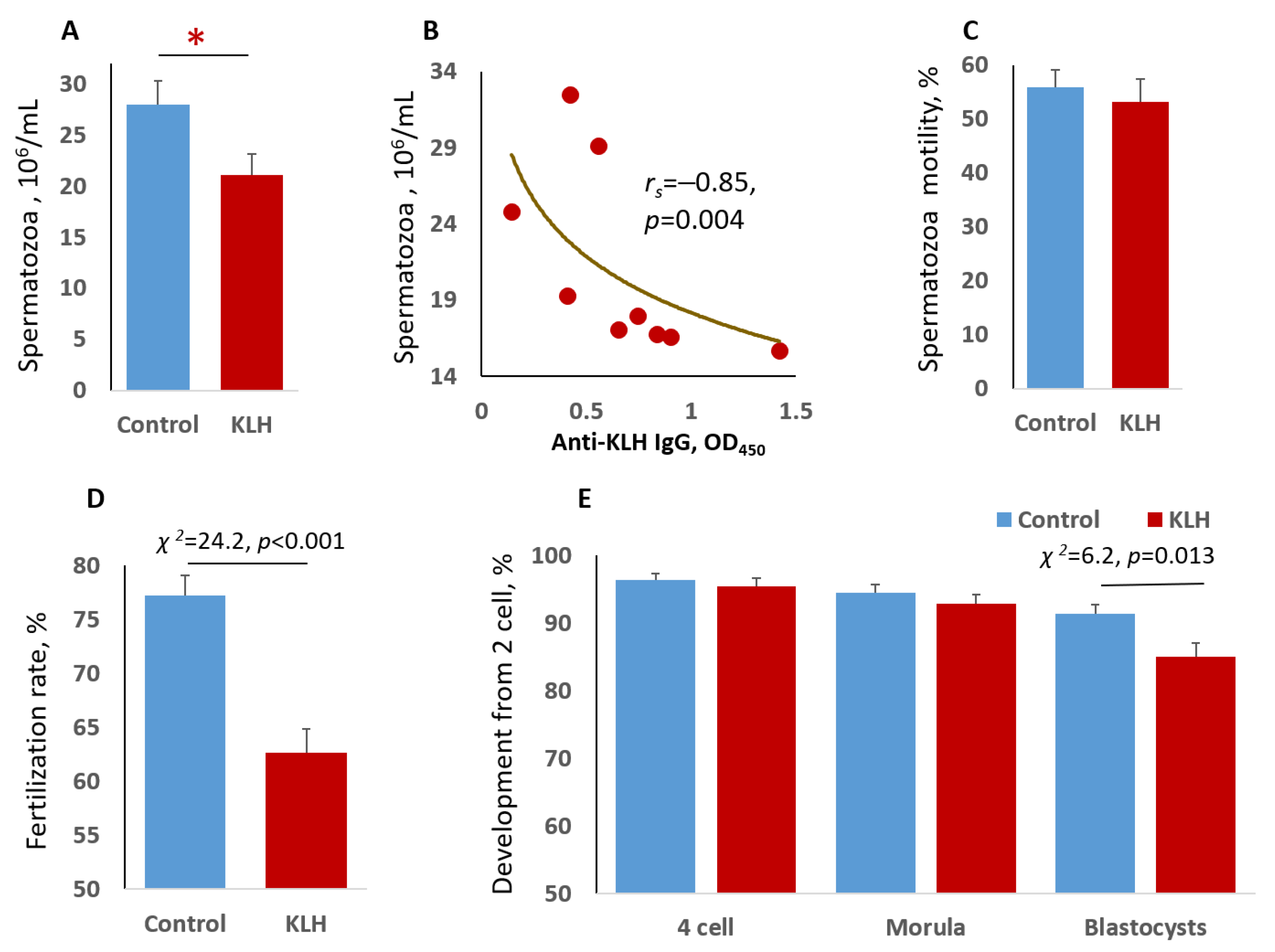
2.1. Impact of the Male Mice Antigenic Challenge on Spermatozoa
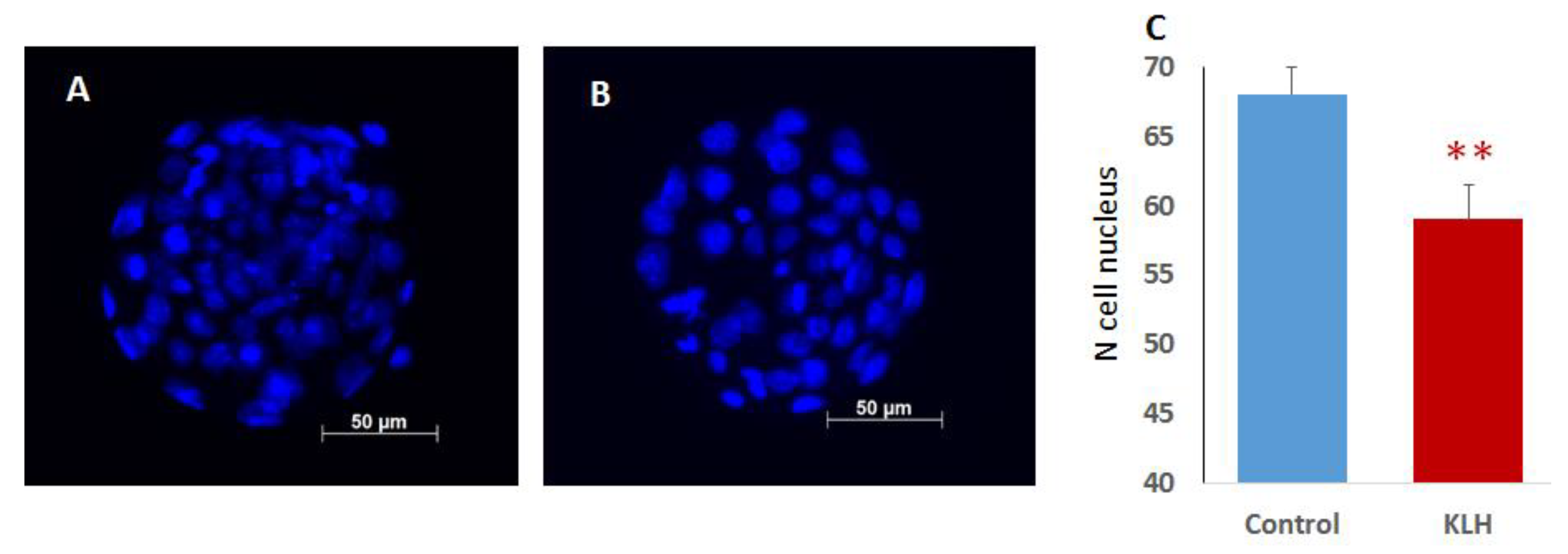
2.2. The Impact of the Male Mice Antigenic Challenge on IVF and Blastocyst Cell Divisions
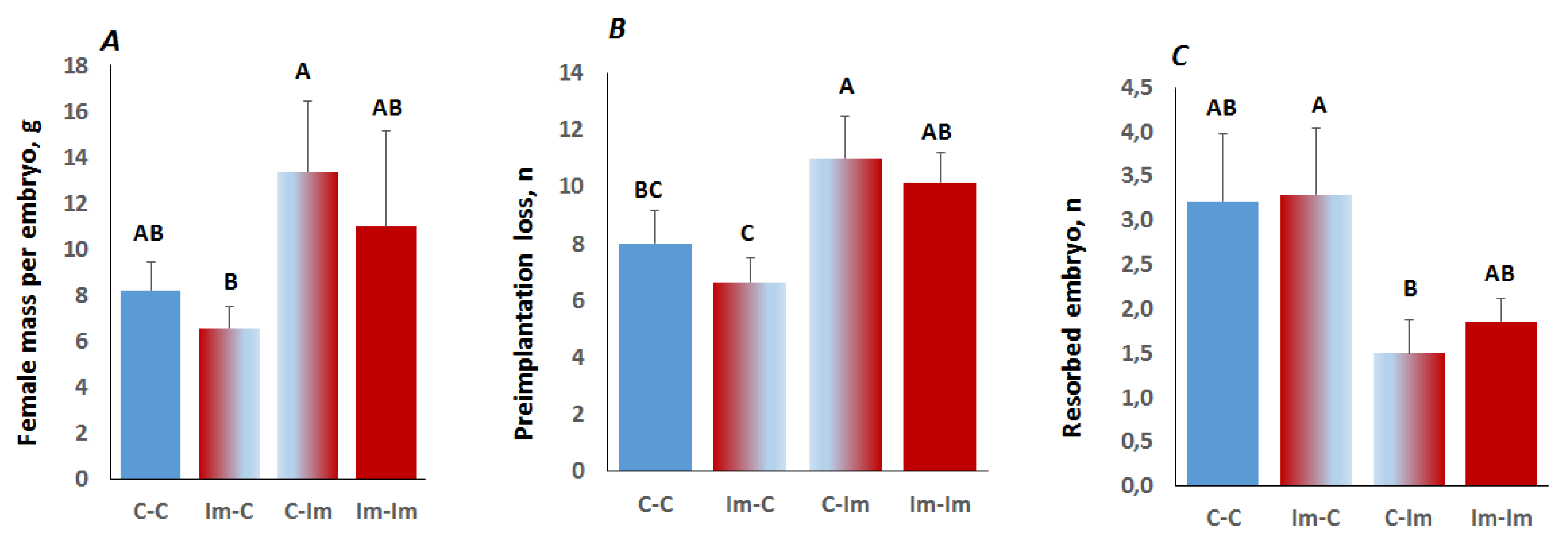
2.3. Interaction between Spermatozoa and Seminal Fluid of The Immune-Primed Males Determined Embryo Implantation Efficiency
2.4. Progesterone, Testosterone, and GM-CSF in Pregnant Female Mice
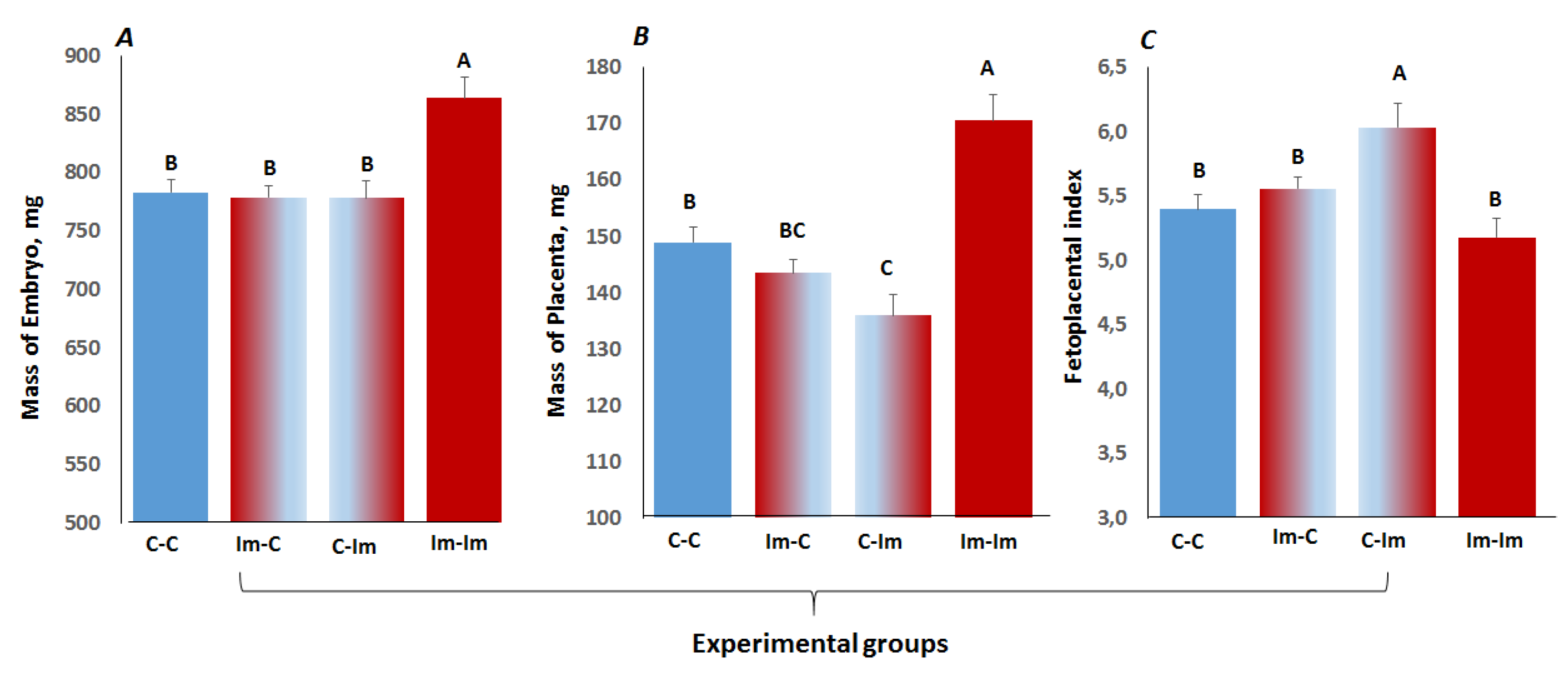
2.5. The Impact of Cross-Talk between Spermatozoa and Seminal Fluid of Immune Primed CD1 Male Mice on Embryo Development
3. Discussion
4. Materials and Methods
4.1. Animals and Immunization
4.2. Spermatozoa Parameters: In Vitro Fertilization
4.3. Experimental Groups, Embryo Transfer, Embryonic Development
4.4. Testosterone, Progesterone, and GM-CSF Immunoassays
4.5. Statistical Analysis
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Wu, L.; Lu, Y.; Jiao, Y.; Liu, B.; Li, S.; Li, Y.; Li, X. Paternal Psychological Stress Reprograms Hepatic Gluconeogenesis in Offspring. Cell Metab. 2016, 23, 735–743. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, W.; Dong, H.; Becker, A.S.; Dapito, D.H.; Modica, S.; Grandl, G.; Wolfrum, C. Cold-induced epigenetic programming of the sperm enhances brown adipose tissue activity in the offspring. Nat. Med. 2018, 24, 1372–1383. [Google Scholar] [CrossRef] [PubMed]
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Terashima, M.; Barbour, S.; Ren, J.; Yu, W.; Han, Y.; Muegge, K. Effect of high fat diet on paternal sperm histone distribution and male offspring liver gene expression. Epigenetics 2015, 10, 861–871. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gerlinskaia, L.A.; Maslennikova, S.O.; Zav’ialov, E.L.; Kontsevaia, G.V.; Moshkin, M.P. Reproductive success of males of the ICR outbred line during propagation against the background of antigenic stimulation. Ontogenez 2012, 43, 357–365. [Google Scholar] [CrossRef] [PubMed]
- Gerlinskaya, L.A.; Shnayder, E.P.; Dotsenko, A.S.; Maslennikova, S.O.; Zavjalov, E.L.; Moshkin, M.P. Antigen-induced changes in odor attractiveness and reproductive output in male mice. Brain Behav. Immun. 2012, 26, 451–458. [Google Scholar] [CrossRef] [PubMed]
- Gerlinskaya, L.A.; Maslennikova, S.O.; Anisimova, M.V.; Feofanova, N.A.; Zavjalov, E.L.; Kontsevaya, G.V.; Moshkin, Y.M.; Moshkin, M.P. Modulation of embryonic development due to mating with immunised males. Reprod. Fertil. Dev. 2017, 29, 565–574. [Google Scholar] [CrossRef]
- Gerlinskaya, L.; Anisimova, M.; Kontsevaya, G.; Maslennikova, S.; Romashchenko, A.; Gong, Y.; Moshkin, M. Mating with immunised male mice affects the phenotype of adult progeny. Reproduction 2020, 160, 117–127. [Google Scholar] [CrossRef]
- Rokade, S.; Upadhya, M.; Bhat, D.S.; Subhedar, N.; Yajnik, C.S.; Ghose, A.; Bal, V. Transient systemic inflammation in adult male mice results in underweight progeny. Am. J. Reprod. Immunol. 2021, 86, e13401. [Google Scholar] [CrossRef]
- Winnall, W.R.; Muir, J.A.; Hedger, M.P. Differential responses of epithelial Sertoli cells of the rat testis to Toll-like receptor 2 and 4 ligands: Implications for studies of testicular inflammation using bacterial lipopolysaccharides. Innate Immun. 2011, 17, 123–136. [Google Scholar] [CrossRef] [Green Version]
- Silva, E.J.R.; Ribeiro, C.M.; Mirim, A.F.M.; Silva, A.A.S.; Romano, R.M.; Hallak, J.; Avellar, M.C.W. Lipopolysaccharide and lipotheicoic acid differentially modulate epididymal cytokine and chemokine profiles and sperm parameters in experimental acute epididymitis. Sci. Rep. 2018, 8, 103. [Google Scholar] [CrossRef]
- Rodríguez-Martínez, H.; Kvist, U.; Ernerudh, J.; Sanz, L.; Calvete, J.J. Seminal plasma proteins: What role do they play? Am. J. Reprod. Immunol. 2011, 66 (Suppl. 1), 11–22. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sharkey, D.J.; Macpherson, A.M.; Tremellen, K.P.; Robertson, S.A. Seminal plasma differentially regulates inflammatory cytokine gene expression in human cervical and vaginal epithelial cells. Mol. Hum. Reprod. 2007, 13, 491–501. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Robertson, S.A. Seminal fluid signaling in the female reproductive tract: Lessons from rodents and pigs. J. Anim. Sci. 2007, 85 (Suppl. 13), E36–E44. [Google Scholar] [CrossRef]
- Lundy, S.K.; Lukacs, N.W. Chronic schistosome infection leads to modulation of granuloma formation and systemic immune suppression. Front. Immunol. 2013, 4, 39. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sego, T.J.; Aponte-Serrano, J.O.; Ferrari Gianlupi, J.; Heaps, S.R.; Breithaupt, K.; Brusch, L.; Crawshaw, J.; Osborne, J.M.; Quardokus, E.M.; Plemper, R.K.; et al. A modular framework for multiscale, multicellular, spatiotemporal modeling of acute primary viral infection and immune response in epithelial tissues and its application to drug therapy timing and effectiveness. PLoS Comput. Biol. 2020, 16, e1008451. [Google Scholar] [CrossRef] [PubMed]
- Loveland, K.L.; Klein, B.; Pueschl, D.; Indumathy, S.; Bergmann, M.; Loveland, B.E.; Hedger, M.P.; Schuppe, H.-C. Cytokines in Male Fertility and Reproductive Pathologies: Immunoregulation and Beyond. Front. Endocrinol. 2017, 8, 307. [Google Scholar] [CrossRef]
- Naz, R.K.; Sellamuthu, R. Receptors in spermatozoa: Are they real? J. Androl. 2006, 27, 627–636. [Google Scholar] [CrossRef]
- Abu Elhija, M.; Lunenfeld, E.; Huleihel, M. LPS increases the expression levels of IL-18, ICE and IL-18 R in mouse testes. Am. J. Reprod. Immunol. 2008, 60, 361–371. [Google Scholar] [CrossRef]
- Collodel, G.; Moretti, E.; Brecchia, G.; Kuželová, L.; Arruda, J.; Mourvaki, E.; Castellini, C. Cytokines release and oxidative status in semen samples from rabbits treated with bacterial lipopolysaccharide. Theriogenology 2015, 83, 1233–1240. [Google Scholar] [CrossRef]
- Wang, F.; Liu, W.; Jiang, Q.; Gong, M.; Chen, R.; Wu, H.; Han, D. Lipopolysaccharide-induced testicular dysfunction and epididymitis in mice: A critical role of tumor necrosis factor alpha†. Biol. Reprod. 2019, 100, 849–861. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A.; Gagnon, C. Formation of reactive oxygen species in spermatozoa of infertile patients. Fertil. Steril. 1992, 57, 409–416. [Google Scholar] [CrossRef]
- Plante, M.; de Lamirande, E.; Gagnon, C. Reactive oxygen species released by activated neutrophils, but not by deficient spermatozoa, are sufficient to affect normal sperm motility. Fertil. Steril. 1994, 62, 387–393. [Google Scholar] [CrossRef]
- Tunc, O.; Tremellen, K. Oxidative DNA damage impairs global sperm DNA methylation in infertile men. J. Assist. Reprod. Genet. 2009, 26, 537–544. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Menezo, Y.J.; Silvestris, E.; Dale, B.; Elder, K. Oxidative stress and alterations in DNA methylation: Two sides of the same coin in reproduction. Reprod. Biomed. Online 2016, 33, 668–683. [Google Scholar] [CrossRef] [Green Version]
- Wyck, S.; Herrera, C.; Requena, C.E.; Bittner, L.; Hajkova, P.; Bollwein, H.; Santoro, R. Oxidative stress in sperm affects the epigenetic reprogramming in early embryonic development. Epigenetics Chromatin 2018, 11, 60. [Google Scholar] [CrossRef] [Green Version]
- Schütte, B.; El Hajj, N.; Kuhtz, J.; Nanda, I.; Gromoll, J.; Hahn, T.; Dittrich, M.; Schorsch, M.; Müller, T.; Haaf, T. Broad DNA methylation changes of spermatogenesis, inflammation and immune response-related genes in a subgroup of sperm samples for assisted reproduction. Andrology 2013, 1, 822–829. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Zhong, L.; Xu, B.; Yang, Y.; Ban, R.; Zhu, J.; Cooke, H.J.; Hao, Q.; Shi, Q. Spermatogenesis Online 1.0: A resource for spermatogenesis based on manual literature curation and genome-wide data mining. Nucleic Acids Res. 2013, 41, D1055–D1062. [Google Scholar] [CrossRef] [Green Version]
- Rathke, C.; Baarends, W.M.; Awe, S.; Renkawitz-Pohl, R. Chromatin dynamics during spermiogenesis. Biochim. Biophys. Acta 2014, 1839, 155–168. [Google Scholar] [CrossRef] [Green Version]
- Henkel, R.; Bastiaan, H.S.; Schüller, S.; Hoppe, I.; Starker, W.; Menkveld, R. Leucocytes and intrinsic ROS production may be factors compromising sperm chromatin condensation status. Andrologia 2010, 42, 69–75. [Google Scholar] [CrossRef]
- Fournier, C.; Labrune, E.; Lornage, J.; Soignon, G.; Giscard d’Estaing, S.; Guérin, J.F.; Benchaib, M. The impact of histones linked to sperm chromatin on embryo development and ART outcome. Andrology 2018, 6, 436–445. [Google Scholar] [CrossRef] [Green Version]
- Moshkin, M.P.; Kondratiuk, E.I.; Litvinova, E.A.; Gerlinskaia, L.A. The activation of specific immunity in male mice stimulates fertility of their breeding partners: The phenomenon of Lot’s daughters. Zh. Obshch. Biol. 2010, 71, 425–435. [Google Scholar] [CrossRef] [PubMed]
- Bromfield, J.J.; Schjenken, J.E.; Chin, P.Y.; Care, A.S.; Jasper, M.J.; Robertson, S.A. Maternal tract factors contribute to paternal seminal fluid impact on metabolic phenotype in offspring. Proc. Natl. Acad. Sci. USA 2014, 111, 2200–2205. [Google Scholar] [CrossRef] [Green Version]
- Fattahi, A.; Latifi, Z.; Darabi, M.; Salmassi, A.; Farzadi, L.; Shaaker, M.; Nouri, M. Mating with seminal vesicle-excised male can affect the uterus phospholipid fatty-acids composition during implantation in an experimental mouse model. Int. Braz. J. Urol. 2019, 45, 825–833. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schjenken, J.E.; Robertson, S.A. Seminal fluid and immune adaptation for pregnancy--comparative biology in mammalian species. Reprod. Domest. Anim. 2014, 49 (Suppl. 3), 27–36. [Google Scholar] [CrossRef]
- Schjenken, J.E.; Sharkey, D.J.; Green, E.S.; Chan, H.Y.; Matias, R.A.; Moldenhauer, L.M.; Robertson, S.A. Sperm modulate uterine immune parameters relevant to embryo implantation and reproductive success in mice. Commun. Biol. 2021, 4, 572. [Google Scholar] [CrossRef]
- Remes Lenicov, F.; Rodriguez Rodrigues, C.; Sabatté, J.; Cabrini, M.; Jancic, C.; Ostrowski, M.; Ceballos, A. Semen promotes the differentiation of tolerogenic dendritic cells. J. Immunol. 2012, 189, 4777–4786. [Google Scholar] [CrossRef] [Green Version]
- Robertson, S.A.; Chin, P.Y.; Femia, J.G.; Brown, H.M. Embryotoxic cytokines—Potential roles in embryo loss and fetal programming. J. Reprod. Immunol. 2018, 125, 80–88. [Google Scholar] [CrossRef] [PubMed]
- O’leary, S.; Jasper, M.J.; Warnes, G.M.; Armstrong, D.T.; Robertson, S.A. Seminal plasma regulates endometrial cytokine expression, leukocyte recruitment and embryo development in the pig. Reproduction 2004, 128, 237–247. [Google Scholar] [CrossRef] [Green Version]
- Schjenken, J.E.; Glynn, D.J.; Sharkey, D.J.; Robertson, S.A. TLR4 Signaling Is a Major Mediator of the Female Tract Response to Seminal Fluid in Mice. Biol. Reprod. 2015, 93, 68. [Google Scholar] [CrossRef]
- Scott, J.L.; Ketheesan, N.; Summers, P.M. Spermatozoa and seminal plasma induce a greater inflammatory response in the ovine uterus at oestrus than dioestrus. Reprod. Fertil. Dev. 2009, 21, 817–826. [Google Scholar] [CrossRef] [PubMed]
- Maslennikova, S.O.; Gerlinskaya, L.A.; Kontsevaya, G.V.; Anisimova, M.V.; Nedospasov, S.A.; Feofanova, N.A.; Moshkin, M.P.; Moshkin, Y.M. TNFα is responsible for the canonical offspring number-size trade-off. Sci. Rep. 2019, 9, 4568. [Google Scholar] [CrossRef] [PubMed]
- Montano, L.; Donato, F.; Bianco, P.M.; Lettieri, G.; Guglielmino, A.; Motta, O.; Bonapace, I.M.; Piscopo, M. Air Pollution and COVID-19: A Possible Dangerous Synergy for Male Fertility. Int. J. Environ. Res. Public Health 2021, 18, 6846. [Google Scholar] [CrossRef] [PubMed]
- Montano, L.; Donato, F.; Bianco, P.M.; Lettieri, G.; Guglielmino, A.; Motta, O.; Bonapace, I.M.; Piscopo, M. Semen quality as a potential susceptibility indicator to SARS-CoV-2 insults in polluted areas. Environ. Sci. Pollut. Res. Int. 2021, 28, 37031–37040. [Google Scholar] [CrossRef]
- He, Y.; Wang, J.; Ren, J.; Zhao, Y.; Chen, J.; Chen, X. Effect of COVID-19 on Male Reproductive System—A Systematic Review. Front. Endocrinol. 2021, 12, 677701. [Google Scholar] [CrossRef]
- Aldinger, K.A.; Sokoloff, G.; Rosenberg, D.M.; Palmer., A.A.; Millen, K.J. Genetic variation and population substructure in outbred CD-1 mice: Implications for genome-wide association studies. PLoS ONE 2009, 4, e4729. [Google Scholar] [CrossRef] [Green Version]
- Fowden, A.L.; Sferruzzi-Perri, A.N.; Coan, P.M.; Constancia, M.; Burton, G.J. Placental efficiency and adaptation: Endocrine regulation. J. Physiol. 2009, 587 Pt 14, 3459–3472. [Google Scholar] [CrossRef]

| Traits | Control (n = 9) Mean ± SEM | KLH (n = 9) Mean ± SEM |
|---|---|---|
| Size | 32.8 ± 1.6 | 33.3 ± 1.4 |
| Elongation | 63.1 ± 1.1 | 63.6 ±0.8 |
| VAP | 94.3 ± 7.0 | 93.5 ± 3.9 |
| VSL | 66.2 ± 5.3 | 66.8 ± 2.8 |
| VCL | 190.0 ± 11.4 | 184.7 ± 7.5 |
| ALH | 14.8 ± 0.5 | 14.6 ± 0.3 |
| BSF | 39.5 ± 0.3 | 38.9 ± 0.33 |
| STR | 64.7 ± 1.6 | 64.9 ± 0.5 |
| LIN | 34.5 ± 1.4 | 35.1 ± 0.6 |
| Groups | n | Female Mass Mean ± SEM | Implanted Embryos Mean ± SEM | Litter Size Mean ± SEM |
|---|---|---|---|---|
| C–C | 14 | 46.4 ± 1.5 | 10.5 ± 1.3 | 7.23 ± 1.09 |
| Im–C | 14 | 48.7 ± 1.6 | 11.9 ± 1.2 | 8.57 ± 0.76 |
| C–Im | 12 | 46.1 ± 2.3 | 8.1 ± 1.6 | 7.18 ± 1.46 |
| Im–Im | 7 | 44.2 ± 1.8 | 8.2 ± 1.1 | 7.17 ± 0.91 |
| Factors | Female Mass/Litter Size | Pre-Implantation Losses | Post-Implantation Losses (Resorption) | |||
|---|---|---|---|---|---|---|
| F1,44 | p | F1,44 | p | F1,44 | p | |
| Spermatozoa donor (KLH vs. control) | 0.76 | 0.39 | 0.95 | 0.33 | 0.07 | 0.79 |
| Vasectomized male (KLH vs. control) | 4.74 | 0.03 | 7.01 | 0.01 | 5.42 | 0.02 |
| Traits | Day | Groups | |||
|---|---|---|---|---|---|
| C–C Mean ± SEM (n) | Im–C Mean ± SEM (n) | C–ImC Mean ± SEM (n) | Im–Im Mean ± SEM (n) | ||
| Progesterone | 5 | 37.7 ± 9.4(14) | 36.8 ± 6.5(14) | 31.8 ± 6.5(12) | 43.6 ± 2.8(6) |
| Progesterone | 16 | 50.9 ± 5.4(14) | 57.7 ± 7.1(14) | 56.5 ± 6.9(12) | 56.8 ± 3.0(7) |
| Progesterone (AF) | 16 | 10.3 ± 2.0(14) | 11.0 ± 1.7(14) | 13.8 ± 1.9(9) | 11.9 ± 0.5(7) |
| Testosterone | 5 | 0.08 ± 0.02(14) | 0.08 ± 0.02(14) | 0.06 ± 0.01(12) | 0.05 ± 0.01(7) |
| Testosterone | 16 | 0.22 ± 0.05(14) | 0.28 ± 0.04(14) | 0.19 ± 0.01(11) | 0.20 ± 0.05(7) |
| Testosterone (AF) | 16 | 2.57 ± 0.16(14) | 2.74 ± 0.22(14) | 2.44 ± 0.15(12) | 2.72 ± 0.04(7) |
| GM-CSF (AF) | 16 | 96.1 ± 16.0AB (12) | 127.5 ± 15.9A (12) | 110.5 ± 26.6AB (9) | 57.3 ± 7.9B (6) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kontsevaya, G.V.; Gerlinskaya, L.A.; Moshkin, Y.M.; Anisimova, M.V.; Stanova, A.K.; Babochkina, T.I.; Moshkin, M.P. The Effects of Sperm and Seminal Fluid of Immunized Male Mice on In Vitro Fertilization and Surrogate Mother–Embryo Interaction. Int. J. Mol. Sci. 2021, 22, 10650. https://doi.org/10.3390/ijms221910650
Kontsevaya GV, Gerlinskaya LA, Moshkin YM, Anisimova MV, Stanova AK, Babochkina TI, Moshkin MP. The Effects of Sperm and Seminal Fluid of Immunized Male Mice on In Vitro Fertilization and Surrogate Mother–Embryo Interaction. International Journal of Molecular Sciences. 2021; 22(19):10650. https://doi.org/10.3390/ijms221910650
Chicago/Turabian StyleKontsevaya, Galina Vladimirovna, Ludmila Alekseevna Gerlinskaya, Yury Mikhailovich Moshkin, Margarita Vladimirovna Anisimova, Aliya Konstantinovna Stanova, Tatyana Ivanovna Babochkina, and Mikhail Pavlovich Moshkin. 2021. "The Effects of Sperm and Seminal Fluid of Immunized Male Mice on In Vitro Fertilization and Surrogate Mother–Embryo Interaction" International Journal of Molecular Sciences 22, no. 19: 10650. https://doi.org/10.3390/ijms221910650
APA StyleKontsevaya, G. V., Gerlinskaya, L. A., Moshkin, Y. M., Anisimova, M. V., Stanova, A. K., Babochkina, T. I., & Moshkin, M. P. (2021). The Effects of Sperm and Seminal Fluid of Immunized Male Mice on In Vitro Fertilization and Surrogate Mother–Embryo Interaction. International Journal of Molecular Sciences, 22(19), 10650. https://doi.org/10.3390/ijms221910650






