New Therapeutics in Endometriosis: A Review of Hormonal, Non-Hormonal, and Non-Coding RNA Treatments
Abstract
:1. Introduction
2. Pathophysiology of Endometriosis
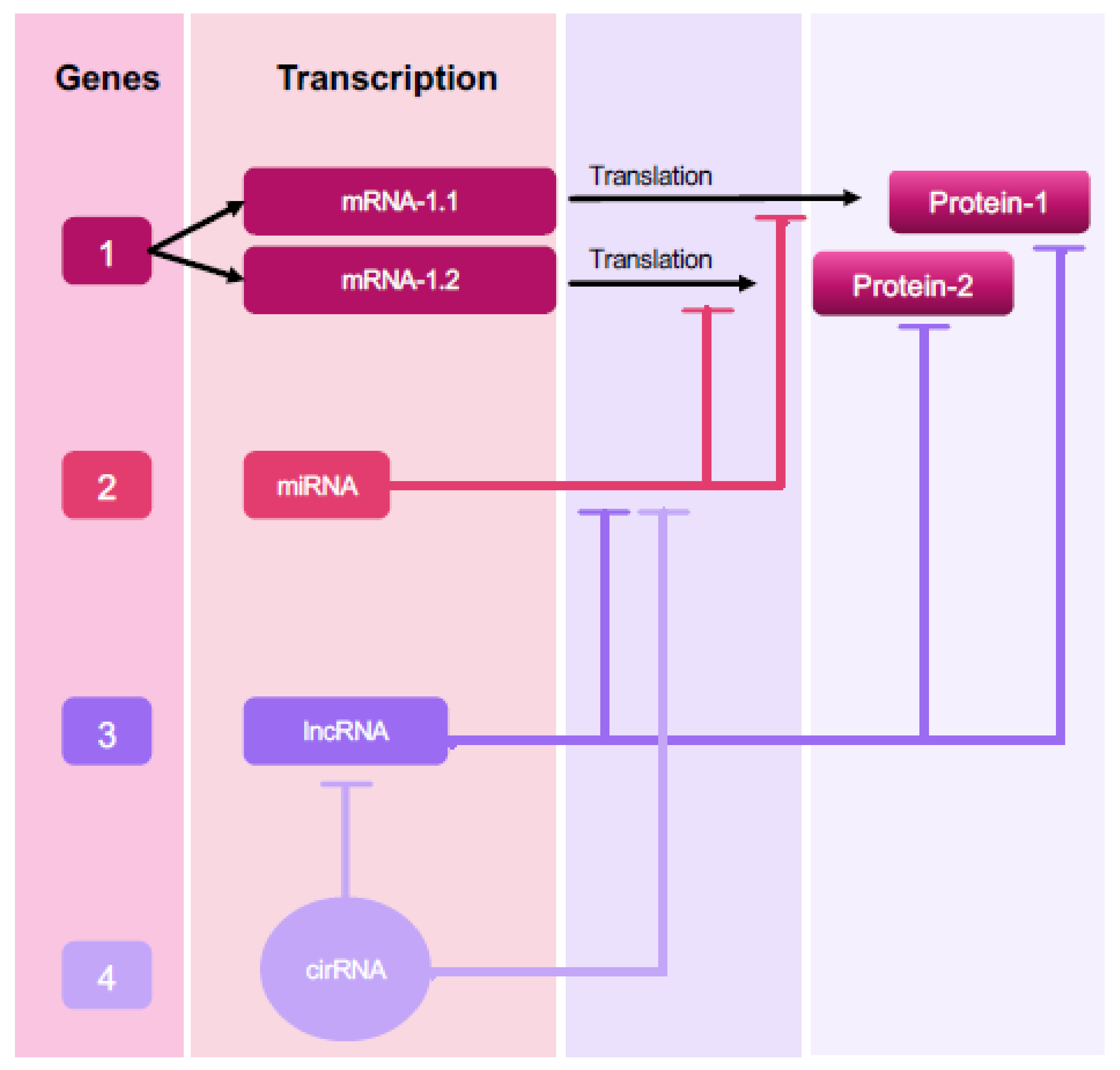
3. Non-Coding RNAs
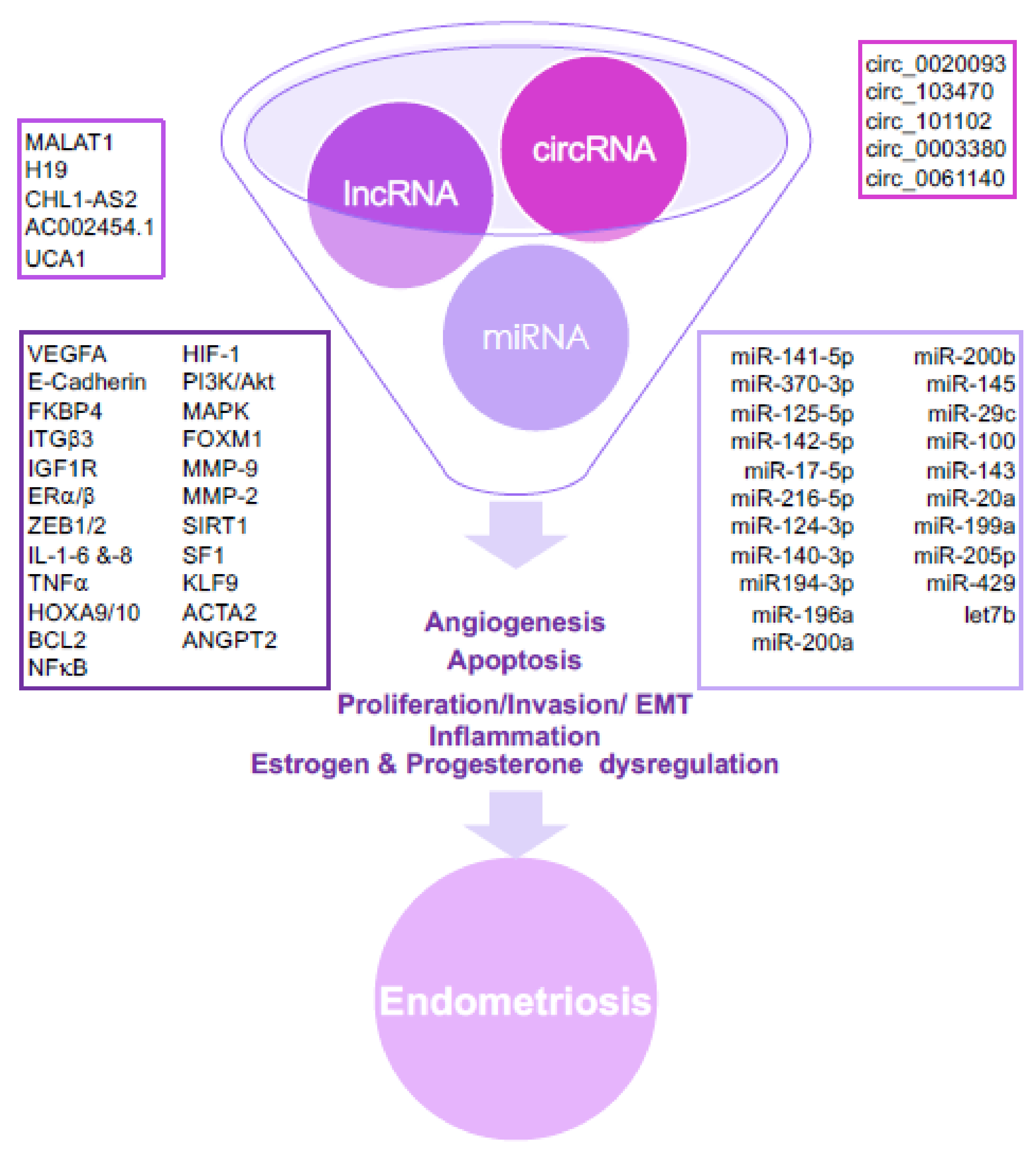
3.1. Insights into ncRNA Classes and Their Association with Endometriosis
3.2. MicroRNAs (miRNAs)
3.2.1. MicroRNAs and Endometriotic Cell Proliferation
3.2.2. Endometriotic Cell Migration
3.2.3. Progesterone Resistance
3.2.4. Endometriosis and Inflammation
3.3. Long ncRNAs (lncRNAs)
3.4. Circular RNAs (circRNAs)
4. Hormonal Treatment
4.1. Progestins
4.2. GnRH Agonists
4.3. GnRH Antagonists
4.4. Dopamine Agonists
4.5. Selective Estrogen Receptor Modulators
4.6. Selective Progesterone Receptor Modulators
5. Non-Hormonal Treatment
5.1. Angiogenesis
5.2. Fibrosis
5.3. Inflammatory Cytokines and Immune Cells
5.4. Stem Cells
5.5. Migration
5.6. Apoptosis
6. Conclusions
Funding
Acknowledgments
Conflicts of Interest
References
- Giudice, L.C.; Kao, L.C. Endometriosis. Lancet 2004, 364, 1789–1799. [Google Scholar] [CrossRef]
- Jamali, N.; Zal, F.; Mostafavi-Pour, Z.; Samare-Najaf, M.; Poordast, T.; Dehghanian, A. Ameliorative effects of quercetin and metformin and their combination against experimental endometriosis in rats. Reprod. Sci. 2021, 28, 683–692. [Google Scholar] [CrossRef]
- Miller, J.E.; Ahn, S.H.; Monsanto, S.P.; Khalaj, K.; Koti, M.; Tayade, C. Implications of immune dysfunction on endometriosis associated infertility. Oncotarget 2016, 8, 7138–7147. [Google Scholar] [CrossRef] [Green Version]
- Becker, C.M.; Gattrell, W.; Gude, K.; Singh, S.S. Reevaluating response and failure of medical treatment of endometriosis: A systematic review. Fertil. Steril. 2017, 108, 125–136. [Google Scholar] [CrossRef] [Green Version]
- Brichant, G.; Nervo, P.; Albert, A.; Munaut, C.; Foidart, J.-M.; Nisolle, M. Heterogeneity of estrogen receptor α and progesterone receptor distribution in lesions of deep infiltrating endometriosis of untreated women or during exposure to various hormonal treatments. Gynecol. Endocrinol. 2018, 34, 651–655. [Google Scholar] [CrossRef]
- Bedaiwy, M.A.; Alfaraj, S.; Yong, P.; Casper, R. New developments in the medical treatment of endometriosis. Fertil. Steril. 2017, 107, 555–565. [Google Scholar] [CrossRef] [Green Version]
- Van Langendonckt, A.; Casanas-Roux, F.; Fau-Donnez, J.; Donnez, J. Oxidative stress and peritoneal endometriosis. Fertil. Steril. 2002, 77, 861–870. [Google Scholar] [CrossRef]
- Santanam, N.; Murphy, A.A.; Parthasarathy, S. Macrophages, oxidation, and endometriosis. Ann. N. Y. Acad. Sci. 2002, 955, 183–198. [Google Scholar] [CrossRef]
- Sampson, J.A. Peritoneal endometriosis due to menstrual dissemination of endometrial tissue into the pelvic cavity. Am. J. Obstet. Gynecol. 1927, 14, 422–469. [Google Scholar] [CrossRef]
- Barra, F.; Ferrero, S. Adhesion proteins: Suitable therapeutic targets or biomarkers of therapy response for endometriosis? Acta Obstet. Gynecol. Scand. 2019, 98, 810–811. [Google Scholar] [CrossRef]
- Pitsos, M.; Kanakas, N. The role of matrix metalloproteinases in the pathogenesis of endometriosis. Reprod. Sci. 2009, 16, 717–726. [Google Scholar] [CrossRef]
- Ezzati, M.; Carr, B.R. Elagolix a novel, orally bioavailable GnRH antagonist under investigation for the treatment of endometriosis-related pain. Women’s Health 2015, 11, 19–28. [Google Scholar] [CrossRef] [PubMed]
- Vercellini, P.; Viganò, P.; Somigliana, E.; Fedele, L. Endometriosis: Pathogenesis and treatment. Nat. Rev. Endocrinol. 2014, 10, 261–275. [Google Scholar] [CrossRef] [PubMed]
- Reis, F.M.; Coutinho, L.M.; Vannuccini, S.; Batteux, F.; Chapron, C.; Petraglia, F. Progesterone receptor ligands for the treatment of endometriosis: The mechanisms behind therapeutic success and failure. Hum. Reprod. Update 2020, 26, 565–585. [Google Scholar] [CrossRef]
- Nisolle, M.; Donnez, J. Reprint of: Peritoneal endometriosis, ovarian endometriosis, and adenomyotic nodules of the rectovaginal septum are three different entities. Fertil. Steril. 2019, 112, e125–e136. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tosti, C.; Pinzauti, S.; Santulli, P.; Chapron, C.; Petraglia, F. Pathogenetic mechanisms of deep infiltrating endometriosis. Reprod. Sci. 2015, 22, 1053–1059. [Google Scholar] [CrossRef]
- Bernacchioni, C.; Capezzuoli, T.; Vannuzzi, V.; Malentacchi, F.; Castiglione, F.; Cencetti, F.; Ceccaroni, M.; Donati, C.; Bruni, P.; Petraglia, F. Sphingosine 1-phosphate receptors are dysregulated in endometriosis: Possible implication in transforming growth factor β–induced fibrosis. Fertil. Steril. 2021, 115, 501–511. [Google Scholar] [CrossRef]
- Massarotti, C.; Badenier, I.M.; Paudice, M.; Scaglione, G.; Remorgida, V.; Vellone, V.G. Steroids receptors immunohistochemical expression in different sites of endometriosis. J. Gynecol. Obstet. Hum. Reprod. 2021, 50, 101861. [Google Scholar] [CrossRef]
- Adams, B.D.; Parsons, C.; Walker, L.; Zhang, W.C.; Slack, F.J. Targeting noncoding RNAs in disease. J. Clin. Investig. 2017, 127, 761–771. [Google Scholar] [CrossRef]
- Bartel, D.P. Metazoan MicroRNAs. Cell 2018, 173, 20–51. [Google Scholar] [CrossRef] [Green Version]
- Evans, J.R.; Feng, F.Y.; Chinnaiyan, A.M. The bright side of dark matter: lncRNAs in cancer. J. Clin. Investig. 2016, 126, 2775–2782. [Google Scholar] [CrossRef] [Green Version]
- Kiss, T. Small nucleolar RNA-guided post-transcriptional modification of cellular RNAs. EMBO J. 2001, 20, 3617–3622. [Google Scholar] [CrossRef]
- Rinn, J.L.; Chang, H.Y. Genome Regulation by Long Noncoding RNAs. Annu. Rev. Biochem. 2012, 81, 145–166. [Google Scholar] [CrossRef] [Green Version]
- Memczak, S.; Jens, M.; Elefsinioti, A.; Torti, F.; Krueger, J.; Rybak, A.; Maier, L.; Mackowiak, S.D.; Gregersen, L.H.; Munschauer, M.; et al. Circular RNAs are a large class of animal RNAs with regulatory potency. Nature 2013, 495, 333–338. [Google Scholar] [CrossRef]
- Mattick, J.S.; Makunin, I.V. Non-coding RNA. Hum. Mol. Genet. 2006, 15, R17–R29. [Google Scholar] [CrossRef] [Green Version]
- Yamamura, S.; Imai-Sumida, M.; Tanaka, Y.; Dahiya, R. Interaction and cross-talk between non-coding RNAs. Cell Mol. Life Sci. 2018, 75, 467–484. [Google Scholar] [CrossRef]
- Aljubran, F.; Nothnick, W.B. Long non-coding RNAs in endometrial physiology and pathophysiology. Mol. Cell Endocrinol. 2021, 525, 111190. [Google Scholar] [CrossRef] [PubMed]
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–222. [Google Scholar] [CrossRef] [PubMed]
- Adams, D.; Gonzalez-Duarte, A.; O’Riordan, W.D.; Yang, C.C.; Ueda, M.; Kristen, A.V.; Tournev, I.; Schmidt, H.H.; Coelho, T.; Berk, J.L.; et al. An RNAi Therapeutic, for hereditary transthyretin amyloidosis. N. Engl. J. Med. 2018, 379, 11–21. [Google Scholar] [CrossRef] [PubMed]
- Balwani, M.; Sardh, E.; Ventura, P.; Peiró, P.A.; Rees, D.C.; Stölzel, U.; Bissell, D.M.; Bonkovsky, H.L.; Windyga, J.; Anderson, K.E.; et al. Phase 3 Trial of RNAi therapeutic givosiran for acute intermittent porphyria. N. Engl. J. Med. 2020, 382, 2289–2301. [Google Scholar] [CrossRef] [PubMed]
- Garrelfs, S.F.; Frishberg, Y.; Hulton, S.A.; Koren, M.J.; O’Riordan, W.D.; Cochat, P.; Deschênes, G.; Shasha-Lavsky, H.; Saland, J.M.; Hoff, W.G.V.; et al. Lumasiran, an RNAi Therapeutic for primary hyperoxaluria type. N. Engl. J. Med. 2021, 384, 1216–1226. [Google Scholar] [CrossRef] [PubMed]
- Bartel, D.P. MicroRNAs: Target recognition and regulatory functions. Cell 2009, 136, 215–233. [Google Scholar] [CrossRef] [Green Version]
- Lai, E.C. Micro RNAs are complementary to 3′ UTR sequence motifs that mediate negative post-transcriptional regulation. Nat. Genet. 2002, 30, 363–364. [Google Scholar] [CrossRef]
- Santamaria, X.; Taylor, H. MicroRNA and gynecological reproductive diseases. Fertil. Steril. 2014, 101, 1545–1551. [Google Scholar] [CrossRef] [PubMed]
- Panir, K.E.; Schjenken, J.; Robertson, A.S.; Hull, M.L. Non-coding RNAs in endometriosis: A narrative review. Hum. Reprod. Update 2018, 24, 497–515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ohlsson Teague, E.M.; Van der Hoek, K.H.; Van der Hoek, M.B.; Perry, N.; Wagaarachchi, P.; Robertson, S.A.; Print, C.G.; Hull, L.M. MicroRNA-regulated pathways associated with endometriosis. Mol. Endocrinol. 2009, 23, 265–275. [Google Scholar] [CrossRef] [PubMed]
- Filigheddu, N.; Gregnanin, I.; Porporato, P.E.; Surico, D.; Perego, B.; Galli, L.; Patrignani, C.; Graziani, A.; Surico, N. Differential expression of MicroRNAs between eutopic and ectopic endometrium in ovarian endometriosis. J. Biomed. Biotechnol. 2010, 2010, 1–29. [Google Scholar] [CrossRef]
- Hu, Z.; Mamillapalli, R.; Taylor, H.S. Increased circulating miR-370-3p regulates steroidogenic factor 1 in endometriosis. Am. J. Physiol. Metab. 2019, 316, E373–E382. [Google Scholar] [CrossRef] [PubMed]
- Ma, L.; Li, Z.; Li, W.; Ai, J.; Chen, X. MicroRNA-142-3p suppresses endometriosis by regulating KLF9-mediated autophagy in vitro and in vivo. RNA Biol. 2019, 16, 1733–1748. [Google Scholar] [CrossRef]
- Zhou, C.-F.; Liu, M.-J.; Wang, W.; Wu, S.; Huang, Y.-X.; Chen, G.-B.; Liu, L.-M.; Peng, D.-X.; Wang, X.-F.; Cai, X.-Z.; et al. miR-205-5p inhibits human endometriosis progression by targeting ANGPT2 in endometrial stromal cells. Stem Cell Res. Ther. 2019, 10, 1–13. [Google Scholar] [CrossRef]
- Adammek, M.; Greve, B.; Kässens, N.; Schneider, C.; Brüggemann, K.; Schüring, A.N.; Starzinski-Powitz, A.; Kiesel, L.; Götte, M. MicroRNA miR-145 inhibits proliferation, invasiveness, and stem cell phenotype of an in vitro endometriosis model by targeting multiple cytoskeletal elements and pluripotency factors. Fertil. Steril. 2013, 99, 1346–1355.e5. [Google Scholar] [CrossRef]
- Agrawal, S.; Tapmeier, T.T.; Rahmioglu, N.; Kirtley, S.; Zondervan, K.T.; Becker, C.M. The miRNA mirage: How close are we to finding a non-invasive diagnostic biomarker in endometriosis? A systematic review. Int. J. Mol. Sci. 2018, 19, 599. [Google Scholar] [CrossRef] [Green Version]
- Hawkins, S.M.; Creighton, C.J.; Han, D.Y.; Zariff, A.; Anderson, M.; Gunaratne, P.H.; Matzuk, M.M. Functional MicroRNA Involved in Endometriosis. Mol. Endocrinol. 2011, 25, 821–832. [Google Scholar] [CrossRef] [Green Version]
- Hu, W.; Xie, Q.; Xu, Y.; Tang, X.; Zhao, H. Integrated bioinformatics analysis reveals function and regulatory network of miR-200b-3p in endometriosis. BioMed Res. Int. 2020, 2020, 1–7. [Google Scholar] [CrossRef]
- Yang, Y.-M.; Yang, W.-X. Epithelial-to-mesenchymal transition in the development of endometriosis. Oncotarget 2017, 8, 41679–41689. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pei, T.; Liu, C.; Liu, T.; Xiao, L.; Luo, B.; Tan, J.; Li, X.; Zhou, G.; Duan, C.; Huang, W. MiR-194-3p represses the progesterone receptor and decidualization in eutopic endometrium from women with endometriosis. Endocrinology 2018, 159, 2554–2562. [Google Scholar] [CrossRef]
- Joshi, N.R.; Miyadahira, E.H.; Afshar, Y.; Jeong, J.-W.; Young, S.L.; Lessey, B.A.; Serafini, P.C.; Fazleabas, A.T. Progesterone resistance in endometriosis is modulated by the altered expression of microRNA-29c and FKBP. J. Clin. Endocrinol. Metab. 2016, 102, 141–149. [Google Scholar] [CrossRef] [Green Version]
- Zhou, M.; Fu, J.; Xiao, L.; Yang, S.; Song, Y.; Zhang, X.; Feng, X.; Sun, H.; Xu, W.; Huang, W. MiR-196a overexpression activates the MEK/ERK signal and represses the progesterone receptor and decidualization in eutopic endometrium from women with endometriosis. Hum. Reprod. 2016, 31, 2598–2608. [Google Scholar] [CrossRef] [PubMed]
- Cosar, E.; Mamillapalli, R.; Ersoy, G.S.; Cho, S.; Seifer, B.; Taylor, H.S. Serum microRNAs as diagnostic markers of endometriosis: A comprehensive array-based analysis. Fertil. Steril. 2016, 106, 402–409. [Google Scholar] [CrossRef] [Green Version]
- Cho, S.; Mutlu, L.; Grechukhina, O.; Taylor, H.S. Circulating microRNAs as potential biomarkers for endometriosis. Fertil. Steril. 2015, 103, 1252–1260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nematian, E.S.; Mamillapalli, R.; Kadakia, T.S.; Zolbin, M.M.; Moustafa, S.; Taylor, H.S. Systemic inflammation induced by microRNAs: Endometriosis-derived alterations in circulating microRNA 125b-5p and Let-7b-5p regulate macrophage cytokine production. J. Clin. Endocrinol. Metab. 2018, 103, 64–74. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wang, X.; Zhang, J.; Liu, X.; Wei, B.; Zhan, L. Long noncoding RNAs in endometriosis: Biological functions, expressions, and mechanisms. J. Cell Physiol. 2021, 236, 6–14. [Google Scholar] [CrossRef]
- Yan, W.; Hu, H.; Tang, B. Progress in understanding the relationship between long noncoding RNA and endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. X 2019, 5, 100067. [Google Scholar] [CrossRef] [PubMed]
- Kopp, F.; Mendell, J.T. Functional classification and experimental dissection of long noncoding RNAs. Cell 2018, 172, 393–407. [Google Scholar] [CrossRef] [Green Version]
- Kallen, A.; Zhou, X.-B.; Xu, J.; Qiao, C.; Ma, J.; Yan, L.; Lu, L.; Liu, C.; Yi, J.-S.; Zhang, H.; et al. The Imprinted H19 LncRNA Antagonizes Let-7 MicroRNAs. Mol. Cell 2013, 52, 101–112. [Google Scholar] [CrossRef] [Green Version]
- Ghazal, S.; McKinnon, B.; Zhou, J.; Mueller, M.; Men, Y.; Yang, L.; Mueller, M.; Flannery, C.; Huang, Y.; Taylor, H.S. H19 lncRNA alters stromal cell growth via IGF signaling in the endometrium of women with endometriosis. EMBO Mol. Med. 2015, 7, 996–1003. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Qiu, J.; Tang, X.; Cui, H.; Zhang, Q.; Yang, Q. LncRNA-H19 regulates cell proliferation and invasion of ectopic endometrium by targeting ITGB3 via modulating miR-124-3p. Exp. Cell Res. 2019, 381, 215–222. [Google Scholar] [CrossRef] [PubMed]
- Xu, Z.; Zhang, L.; Yu, Q.; Zhang, Y.; Yan, L.; Chen, Z.-J. The estrogen-regulated lncRNA H19/miR-216a-5p axis alters stromal cell invasion and migration via ACTA2 in endometriosis. Mol. Hum. Reprod. 2019, 25, 550–561. [Google Scholar] [CrossRef]
- Liu, S.; Xin, W.; Lu, Q.; Tang, X.; Wang, F.; Shao, W.; Zhang, Y.; Qiu, J.; Hua, K. Knockdown of lncRNA H19 suppresses endometriosis in vivo. Braz. J. Med. Biol. Res. 2021, 54, e10117. [Google Scholar] [CrossRef]
- Hubé, F.; Guo, J.; Chooniedass-Kothari, S.; Cooper, C.; Hamedani, M.K.; Dibrov, A.A.; Blanchard, A.A.; Wang, X.; Deng, G.; Myal, Y.; et al. Alternative splicing of the first intron of the steroid receptor RNA activator (SRA) participates in the generation of coding and noncoding RNA isoforms in breast cancer cell lines. DNA Cell Biol. 2006, 25, 418–428. [Google Scholar] [CrossRef]
- Lin, K.; Zhan, H.; Ma, J.; Xu, K.; Wu, R.; Zhou, C.; Lin, J. Silencing of SRA1 Regulates ER expression and attenuates the growth of stromal cells in ovarian endometriosis. Reprod. Sci. 2016, 24, 836–843. [Google Scholar] [CrossRef] [PubMed]
- Bossi, L.; Figueroa-Bossi, N. Competing endogenous RNAs: A target-centric view of small RNA regulation in bacteria. Nat. Rev. Genet. 2016, 14, 775–784. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Greene, J.; Baird, A.-M.; Brady, L.; Lim, M.; Gray, S.; McDermott, R.; Finn, S. Circular RNAs: Biogenesis, Function and Role in Human Diseases. Front. Mol. Biosci. 2017, 4, 38. [Google Scholar] [CrossRef] [PubMed]
- Barrett, S.P.; Salzman, J. Circular RNAs: Analysis, expression and potential functions. Development 2016, 143, 1838–1847. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, H.; Tsukahara, T. A view of pre-mRNA splicing from RNase R resistant RNAs. Int. J. Mol. Sci. 2014, 15, 9331–9342. [Google Scholar] [CrossRef] [Green Version]
- Taylor, H.S.; Giudice, L.C.; Lessey, B.A.; Abrao, M.S.; Kotarski, J.; Archer, D.F.; Diamond, M.P.; Surrey, E.; Johnson, N.P.; Watts, N.B.; et al. Treatment of endometriosis-associated pain with Elagolix, an Oral GnRH antagonist. N. Engl. J. Med. 2017, 377, 28–40. [Google Scholar] [CrossRef]
- Dragoman, M.V.; Gaffield, M.E. The safety of subcutaneously administered depot medroxyprogesterone acetate (104 mg/0.65 mL): A systematic review. Contraception 2016, 94, 202–215. [Google Scholar] [CrossRef] [Green Version]
- Bernat, A.L.; Oyama, K.; Hamdi, S.; Mandonnet, E.; Vexiau, D.; Pocard, M.; George, B.; Froelich, S. Growth stabilization and regression of meningiomas after discontinuation of cyproterone acetate: A case series of 12 patients. Acta Neurochir. 2015, 157, 1741–1746. [Google Scholar] [CrossRef]
- Schmutz, J.L. Cyproterone acetate and meningioma: The latest findings. Ann. Dermatol. Venereol. 2018, 145, 390–391. [Google Scholar] [CrossRef]
- Gezer, A.; Oral, E. Progestin therapy in endometriosis. Women’s Health 2015, 11, 643–652. [Google Scholar] [CrossRef] [PubMed]
- Vercellini, P.; Somigliana, E.; Vigano, P.; Abbiati, A.; Daguati, R.; Crosignani, P.G. Endometriosis: Current and future medical therapies. Best Pract. Res. Clin. Obstet. Gynaecol. 2008, 22, 275–306. [Google Scholar] [CrossRef]
- Quaas, A.M.; Weedin, E.A.; Hansen, K.R. On-label and off-label drug use in the treatment of endometriosis. Fertil. Steril. 2015, 103, 612–625. [Google Scholar] [CrossRef]
- Soares, S.R.; Martínez-Varea, A.; Hidalgo-Mora, J.J.; Pellicer, A. Pharmacologic therapies in endometriosis: A systematic review. Fertil. Steril. 2012, 98, 529–555. [Google Scholar] [CrossRef] [PubMed]
- Olive, D.L. Gonadotropin-releasing hormone agonists for endometriosis. N. Engl. J. Med. 2008, 359, 1136–1142. [Google Scholar] [CrossRef]
- Küpker, W.; Felberbaum, R.; Krapp, M.; Schill, T.; Malik, E.; Diedrich, K. Use of GnRH antagonists in the treatment of endometriosis. Reprod. Biomed. 2002, 5, 12–16. [Google Scholar] [CrossRef]
- Barbieri, R.L. Hormone treatment of endometriosis: The estrogen threshold hypothesis. Am. J. Obstet. Gynecol. 1992, 166, 740–745. [Google Scholar] [CrossRef]
- Struthers, R.S.; Nicholls, A.J.; Grundy, J.; Chen, T.; Jimenez, R.; Yen, S.S.C.; Bozigian, H.P. Suppression of gonadotropins and estradiol in premenopausal women by oral administration of the nonpeptide gonadotropin-releasing hormone antagonist Elagolix. J. Clin. Endocrinol. Metab. 2009, 94, 545–551. [Google Scholar] [CrossRef] [Green Version]
- Diamond, M.P.; Carr, B.; Dmowski, W.P.; Koltun, W.; O’Brien, C.; Jiang, P.; Burke, J.; Jimenez, R.; Garner, E.; Chwalisz, K. Elagolix treatment for endometriosis-associated pain: Results from a phase 2, randomized, double-blind, placebo-controlled study. Reprod. Sci. 2014, 21, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Surrey, E.; Taylor, H.S.; Giudice, L.; Lessey, B.A.; Abrao, M.S.; Archer, D.F.; Diamond, M.P.; Johnson, N.P.; Watts, N.B.; Gallagher, J.C.; et al. Long-term outcomes of Elagolix in women with endometriosis: Results from two extension studies. Obstet. Gynecol. 2018, 132, 147–160. [Google Scholar] [CrossRef]
- Riccio, L.G.C.; Jeljeli, M.; Santulli, P.; Chouzenoux, S.; Doridot, L.; Nicco, C.; Reis, F.M.; Abrão, M.S.; Chapron, C.; Batteux, F. B lymphocytes inactivation by Ibrutinib limits endometriosis progression in mice. Hum. Reprod. 2019, 34, 1225–1234. [Google Scholar] [CrossRef] [PubMed]
- Pellicer, N.; Galliano, D.; Herraiz, S.; Bagger, Y.Z.; Arce, J.-C.; Pellicer, A. Use of dopamine agonists to target angiogenesis in women with endometriosis. Hum. Reprod. 2021, 36, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Delgado-Rosas, F.; Gómez, R.; Ferrero, H.; Gaytan, F.; Garcia-Velasco, J.; Simon, C.; Pellicer, A. The effects of ergot and non-ergot-derived dopamine agonists in an experimental mouse model of endometriosis. Reproduction 2011, 142, 745–755. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Showell, M.G.; Mackenzie-Proctor, R.; Jordan, V.; Hart, R.J. Antioxidants for female subfertility. Cochrane Database Syst. Rev. 2020, 8, 007807. [Google Scholar] [CrossRef]
- Naqvi, H.; Sakr, S.; Presti, T.; Krikun, G.; Komm, B.; Taylor, H.S. Treatment with Bazedoxifene and Conjugated Estrogens Results in Regression of Endometriosis in a Murine Model. Biol. Reprod. 2014, 90, 121. [Google Scholar] [CrossRef]
- Kulak, J., Jr.; Fischer, C.; Komm, B.; Taylor, H.S. Treatment with bazedoxifene, a selective estrogen receptor modulator, causes regression of endometriosis in a mouse model. Endocrinology 2011, 152, 3226–3232. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Mamillapalli, R.; Mutlu, L.; Du, H.; Taylor, H.S. Chemoattraction of bone marrow-derived stem cells towards human endometrial stromal cells is mediated by estradiol regulated CXCL12 and CXCR4 expression. Stem Cell Res. 2015, 15, 14–22. [Google Scholar] [CrossRef] [Green Version]
- Sakr, S.; Naqvi, H.; Komm, B.; Taylor, H.S. Endometriosis impairs bone marrow-derived stem cell recruitment to the uterus whereas bazedoxifene treatment leads to endometriosis regression and improved uterine stem cell engraftment. Endocrinology 2014, 155, 1489–1497. [Google Scholar] [CrossRef] [Green Version]
- Yao, Z.; Shen, X.; Capodanno, I.; Donnelly, M.; Fenyk-Melody, J.; Hausamann, J.; Nunes, C.; Strauss, J.; Vakerich, K. Validation of rat endometriosis model by using raloxifene as a positive control for the evaluation of Novel SERM compounds. J. Investig. Surg. 2005, 18, 177–183. [Google Scholar] [CrossRef]
- Stratton, P.; Sinaii, N.; Segars, J.; Koziol, D.; Wesley, R.; Zimmer, C.; Winkel, C.; Nieman, L.K. Return of chronic pelvic pain from endometriosis after raloxifene treatment: A randomized controlled trial. Obstet. Gynecol. 2008, 111, 88–96. [Google Scholar] [CrossRef] [Green Version]
- Altintas, D.; Kokcu, A.; Kandemir, B.; Tosun, M.; Cetinkaya, M.B. Comparison of the effects of raloxifene and anastrozole on experimental endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2010, 150, 84–87. [Google Scholar] [CrossRef]
- Madauss, K.P.; Stewart, E.L.; Williams, S.P. The evolution of progesterone receptor ligands. Med. Res. Rev. 2007, 27, 374–400. [Google Scholar] [CrossRef]
- Chabbert-Buffet, N.; Meduri, G.; Bouchard, P.; Spitz, I.M. Selective progesterone receptor modulators and progesterone antagonists: Mechanisms of action and clinical applications. Hum. Reprod. Update 2005, 11, 293–307. [Google Scholar] [CrossRef] [Green Version]
- Donnez, J.; Hudecek, R.; Donnez, O.; Matule, D.; Arhendt, H.-J.; Zatik, J.; Kasilovskiene, Z.; Dumitrascu, M.C.; Fernandez, H.; Barlow, D.H.; et al. Efficacy and safety of repeated use of ulipristal acetate in uterine fibroids. Fertil. Steril. 2015, 103, 519–527. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huniadi, C.A.; Pop, O.L.; Antal, T.A.; Stamatian, F. The effects of ulipristal on Bax/Bcl-2, cytochrome C, Ki-67 and cyclooxygenase-2 expression in a rat model with surgically induced endometriosis. Eur. J. Obstet. Gynecol. Reprod. Biol. 2013, 169, 360–365. [Google Scholar] [CrossRef] [PubMed]
- Liang, B.; Wu, L.; Xu, H.; Cheung, C.W.; Fung, W.Y.; Wong, S.W.; Wang, C.C. Efficacy, safety and recurrence of new progestins and selective progesterone receptor modulator for the treatment of endometriosis: A comparison study in mice. Reprod. Biol. Endocrinol. 2018, 16, 32. [Google Scholar] [CrossRef] [PubMed]
- Kettel, L.M.; Murphy, A.A.; Morales, A.J.; Ulmann, A.; Baulieu, E.E.; Yen, S.S. Treatment of endometriosis with the antiprogesterone mifepristone (RU486). Fertil. Steril. 1996, 65, 23–28. [Google Scholar] [CrossRef]
- Mei, L.; Bao, J.; Tang, L.; Zhang, C.; Wang, H.; Sun, L.; Ma, G.; Huang, L.; Yang, J.; Zhang, L.; et al. A novel mifepristone-loaded implant for long-term treatment of endometriosis: In vitro and in vivo studies. Eur. J. Pharm. Sci. 2010, 39, 421–427. [Google Scholar] [CrossRef] [PubMed]
- Nap, A.W.; Griffioen, A.W.; Dunselman, G.A.J.; Steege, J.C.A.B.-T.; Thijssen, V.L.J.L.; Evers, J.L.H.; Groothuis, P.G. Antiangiogenesis therapy for endometriosis. J. Clin. Endocrinol. Metab. 2004, 89, 1089–1095. [Google Scholar] [CrossRef] [Green Version]
- Folkman, J.; Klagsbrun, M. A family of angiogenic peptides. Nat. Cell Biol. 1987, 329, 671–672. [Google Scholar] [CrossRef]
- Becker, C.M.; Wright, R.D.; Satchi-Fainaro, R.; Funakoshi, T.; Folkman, J.; Kung, A.; D’Amato, R.J. A Novel Noninvasive Model of Endometriosis for Monitoring the Efficacy of Antiangiogenic Therapy. Am. J. Pathol. 2006, 168, 2074–2084. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Krikun, G.; Hu, Z.; Osteen, K.; Bruner-Tran, K.L.; Schatz, F.; Taylor, H.S.; Toti, P.; Arcuri, F.; Konigsberg, W.; Garen, A.; et al. The Immunoconjugate “Icon” Targets aberrantly expressed endothelial tissue factor causing regression of endometriosis. Am. J. Pathol. 2010, 176, 1050–1056. [Google Scholar] [CrossRef] [PubMed]
- Hufnagel, D.; Goetz, T.G.; Hu, Z.; Nyachieo, A.; D’Hooghe, T.; Fazleabas, A.; Duleba, A.; Krikun, G.; Taylor, H.S.; Lockwood, C.J. Icon immunoconjugate treatment results in regression of red lesions in a non-human primate (Papio anubis) model of endometriosis. Reprod. Biol. 2018, 18, 109–114. [Google Scholar] [CrossRef] [PubMed]
- Taylor, H.S.; Osteen, K.G.; Bruner-Tran, K.L.; Lockwood, C.J.; Krikun, G.; Sokalska, A.; Duleba, A.J. Novel therapies targeting endometriosis. Reprod. Sci. 2011, 18, 814–823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hull, M.L.; Charnock-Jones, D.S.; Chan, C.L.K.; Bruner-Tran, K.L.; Osteen, K.G.; Tom, B.D.M.; Fan, T.-P.D.; Smith, S.K. Antiangiogenic agents are effective inhibitors of endometriosis. J. Clin. Endocrinol. Metab. 2003, 88, 2889–2899. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ozer, H.; Boztosun, A.; Açmaz, G.; Atılgan, R.; Akkar, O.B.; Kosar, M.I. The Efficacy of Bevacizumab, Sorafenib, and Retinoic Acid on Rat Endometriosis Model. Reprod. Sci. 2012, 20, 26–32. [Google Scholar] [CrossRef] [PubMed]
- Hisrich, B.V.; Young, R.B.; Sansone, A.M.; Bowens, Z.; Green, L.J.; Lessey, B.A.; Blenda, A.V. Role of human galectins in inflammation and cancers associated with endometriosis. Biomolecules 2020, 10, 230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- De Mattos, R.M.; Machado, D.E.; Perini, J.A.; Alessandra-Perini, J.; Costa, N.D.O.M.D.; Wiecikowski, A.F.D.R.D.O.; Cabral, K.M.D.S.; Takiya, C.M.; Carvalho, R.S.; Nasciutti, L.E. Galectin-3 plays an important role in endometriosis development and is a target to endometriosis treatment. Mol. Cell Endocrinol. 2019, 486, 1–10. [Google Scholar] [CrossRef]
- Noël, J.-C.; Chapron, C.; Borghese, B.; Fayt, I.; Anaf, V. Galectin-3 is Overexpressed in Various forms of endometriosis. Appl. Immunohistochem. Mol. Morphol. 2011, 19, 253–257. [Google Scholar] [CrossRef] [PubMed]
- Nisolle, M.; Alvarez, M.L.; Colombo, M.; Foidart, J.M. Pathogenesis of endometriosis. Gynecol. Obstet. Fertil. 2007, 35, 898–903. [Google Scholar] [CrossRef]
- Harrison, C. Trial watch: BTK inhibitor shows positive results in B cell malignancies. Nature reviews. Drug Discov. 2012, 11, 96. [Google Scholar] [CrossRef]
- Ganieva, U.; Nakamura, T.; Osuka, S.; Bayasula Nakanishi, N.; Kasahara, Y.; Takasaki, N.; Muraoka, A.; Hayashi, S.; Nagai, T.; Murase, T.; et al. Involvement of transcription factor 21 in the pathogenesis of fibrosis in endometriosis. Am. J. Pathol. 2020, 190, 145–157. [Google Scholar] [CrossRef]
- Nasu, K.; Tsuno, A.; Hirao, M.; Kobayashi, H.; Yuge, A.; Narahara, H. Heparin is a promising agent for the treatment of endometriosis-associated fibrosis. Fertil. Steril. 2010, 94, 46–51. [Google Scholar] [CrossRef]
- Kato, T.; Yasuda, K.; Matsushita, K.; Ishii, K.J.; Hirota, S.; Yoshimoto, T.; Shibahara, H. Interleukin-1/-33 signaling pathways as therapeutic targets for endometriosis. Front. Immunol. 2019, 10, 2021. [Google Scholar] [CrossRef] [Green Version]
- Lu, D.; Song, H.; Shi, G. Anti-TNF-α treatment for pelvic pain associated with endometriosis. Cochrane Database Syst. Rev. 2013, 3, CD008088. [Google Scholar] [CrossRef] [Green Version]
- Liu, H.; Xiong, M.; Xia, Y.-F.; Cui, N.-J.; Lu, R.-B.; Deng, L.; Lin, Y.-H.; Rong, T.-H. Studies on pentoxifylline and tocopherol combination for radiation-induced heart disease in rats. Int. J. Radiat. Oncol. 2009, 73, 1552–1559. [Google Scholar] [CrossRef]
- Taylor, H.S. Endometrial cells derived from donor stem cells in bone marrow transplant recipients. JAMA 2004, 292, 81–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, H.; Taylor, H.S. Contribution of bone marrow-derived stem cells to endometrium and endometriosis. Stem Cells 2007, 25, 2082–2086. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Zhang, Z.; Yang, F.; Wang, H.; Liang, S.; Wang, H.; Yang, J.; Lin, J. The role of endometrial stem cells in the pathogenesis of endometriosis and their application to its early diagnosis. Biol. Reprod. 2020, 102, 1153–1159. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liang, S.; Yang, F.; Sun, Y.; Niu, L.; Ren, Y.; Wang, H.; He, Y.; Du, J.; Yang, J.; et al. Biological characteristics of endometriotic mesenchymal stem cells isolated from ectopic lesions of patients with endometriosis. Stem Cell Res. Ther. 2020, 11, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Chen, P.; Mamillapalli, R.; Habata, S.; Taylor, H. Endometriosis stromal cells induce bone marrow mesenchymal stem cell differentiation and PD-1 expression through paracrine signaling. Mol. Cell Biochem. 2021, 476, 1717–1727. [Google Scholar] [CrossRef]
- Ersoy, G.S.; Zolbin, M.M.; Cosar, E.; Mamillapalli, R.; Taylor, H.S. Medical therapies for endometriosis differentially inhibit stem cell recruitment. Reprod. Sci. 2017, 24, 818–823. [Google Scholar] [CrossRef] [PubMed]
- Rahmawati, E.; Yang, W.-C.V.; Lei, Y.-P.; Maurya, P.K.; Chen, H.-W.; Tzeng, C.-R. Gonadotropin-releasing hormone agonist induces downregulation of tensin 1 in women with endometriosis. Acta Obstet. Gynecol. Scand. 2019, 98, 222–231. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pavone, M.; Malpani, S.; Dyson, M.; Bulun, S. Fenretinide: A potential treatment for endometriosis. Fertil. Steril. 2014, 102, e11. [Google Scholar] [CrossRef] [Green Version]
- Bruner-Tran, K.L.; Osteen, K.G.; Taylor, H.S.; Sokalska, A.; Haines, K.; Duleba, A.J. Resveratrol inhibits development of experimental endometriosis in vivo and reduces endometrial stromal cell invasiveness in vitro. Biol. Reprod. 2011, 84, 106–112. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Brichant, G.; Laraki, I.; Henry, L.; Munaut, C.; Nisolle, M. New Therapeutics in Endometriosis: A Review of Hormonal, Non-Hormonal, and Non-Coding RNA Treatments. Int. J. Mol. Sci. 2021, 22, 10498. https://doi.org/10.3390/ijms221910498
Brichant G, Laraki I, Henry L, Munaut C, Nisolle M. New Therapeutics in Endometriosis: A Review of Hormonal, Non-Hormonal, and Non-Coding RNA Treatments. International Journal of Molecular Sciences. 2021; 22(19):10498. https://doi.org/10.3390/ijms221910498
Chicago/Turabian StyleBrichant, Geraldine, Ines Laraki, Laurie Henry, Carine Munaut, and Michelle Nisolle. 2021. "New Therapeutics in Endometriosis: A Review of Hormonal, Non-Hormonal, and Non-Coding RNA Treatments" International Journal of Molecular Sciences 22, no. 19: 10498. https://doi.org/10.3390/ijms221910498
APA StyleBrichant, G., Laraki, I., Henry, L., Munaut, C., & Nisolle, M. (2021). New Therapeutics in Endometriosis: A Review of Hormonal, Non-Hormonal, and Non-Coding RNA Treatments. International Journal of Molecular Sciences, 22(19), 10498. https://doi.org/10.3390/ijms221910498






