Novel Coumarin-Thiadiazole Hybrids and Their Cu(II) and Zn(II) Complexes as Potential Antimicrobial Agents and Acetylcholinesterase Inhibitors
Abstract
:1. Introduction
2. Results and Discussion
2.1. Synthesis
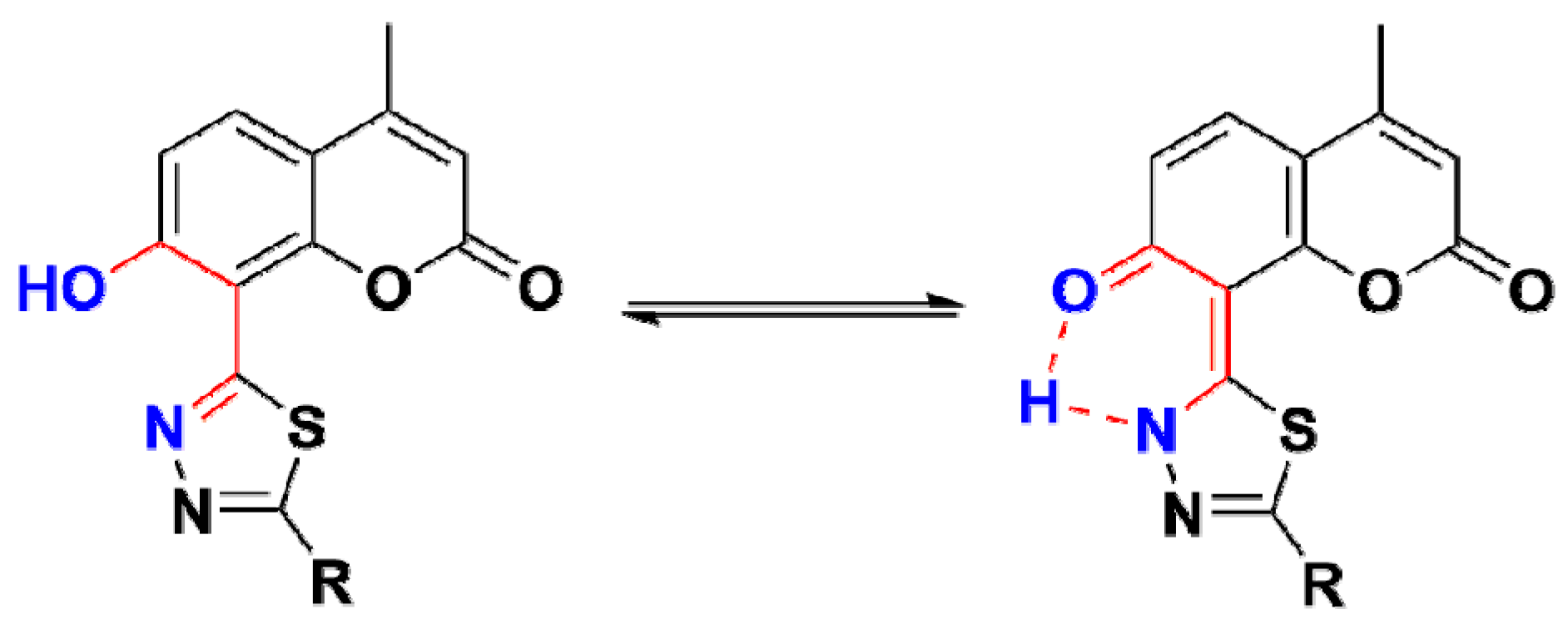
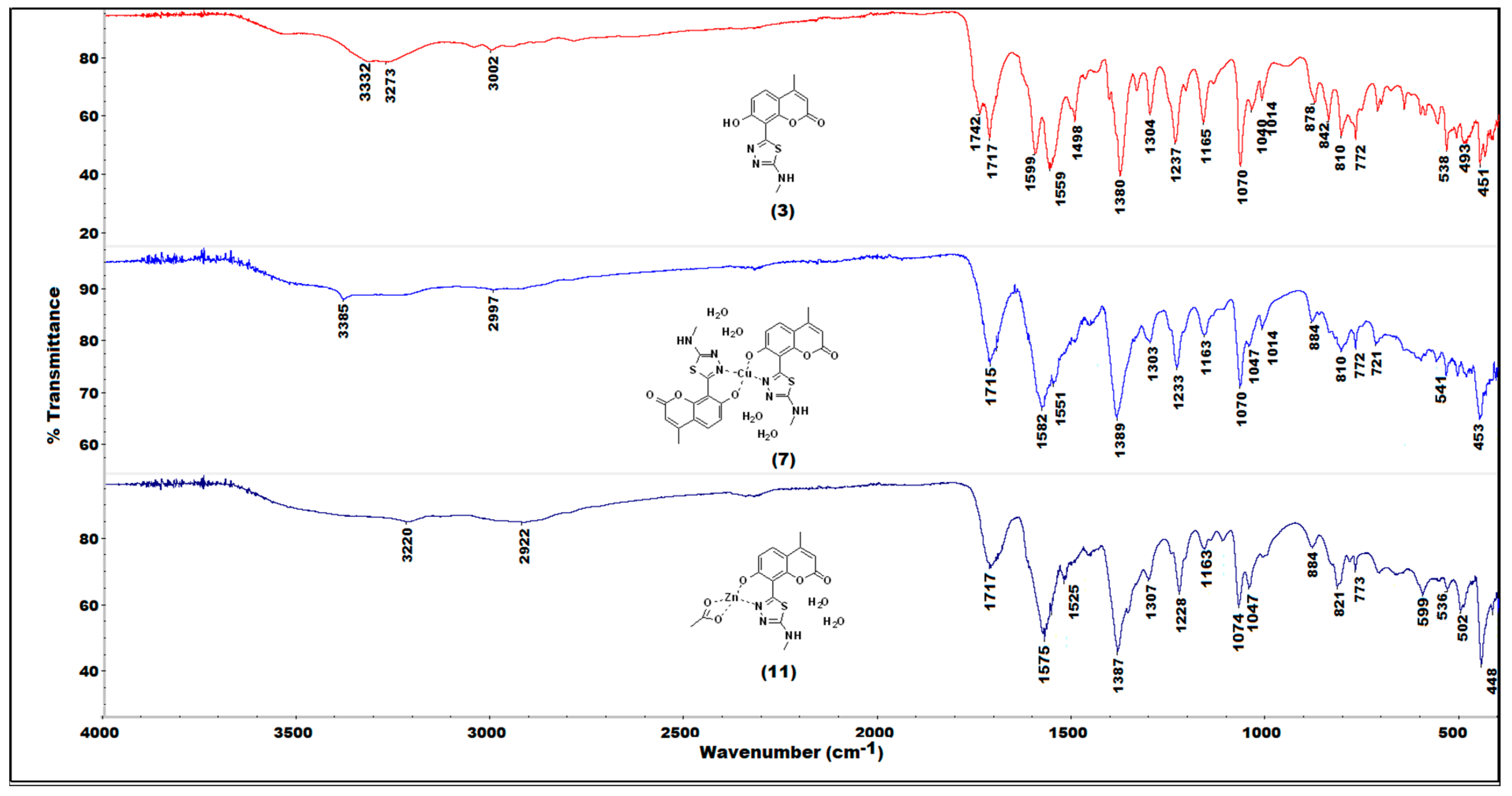
2.2. IR(ATR) Spectroscopy
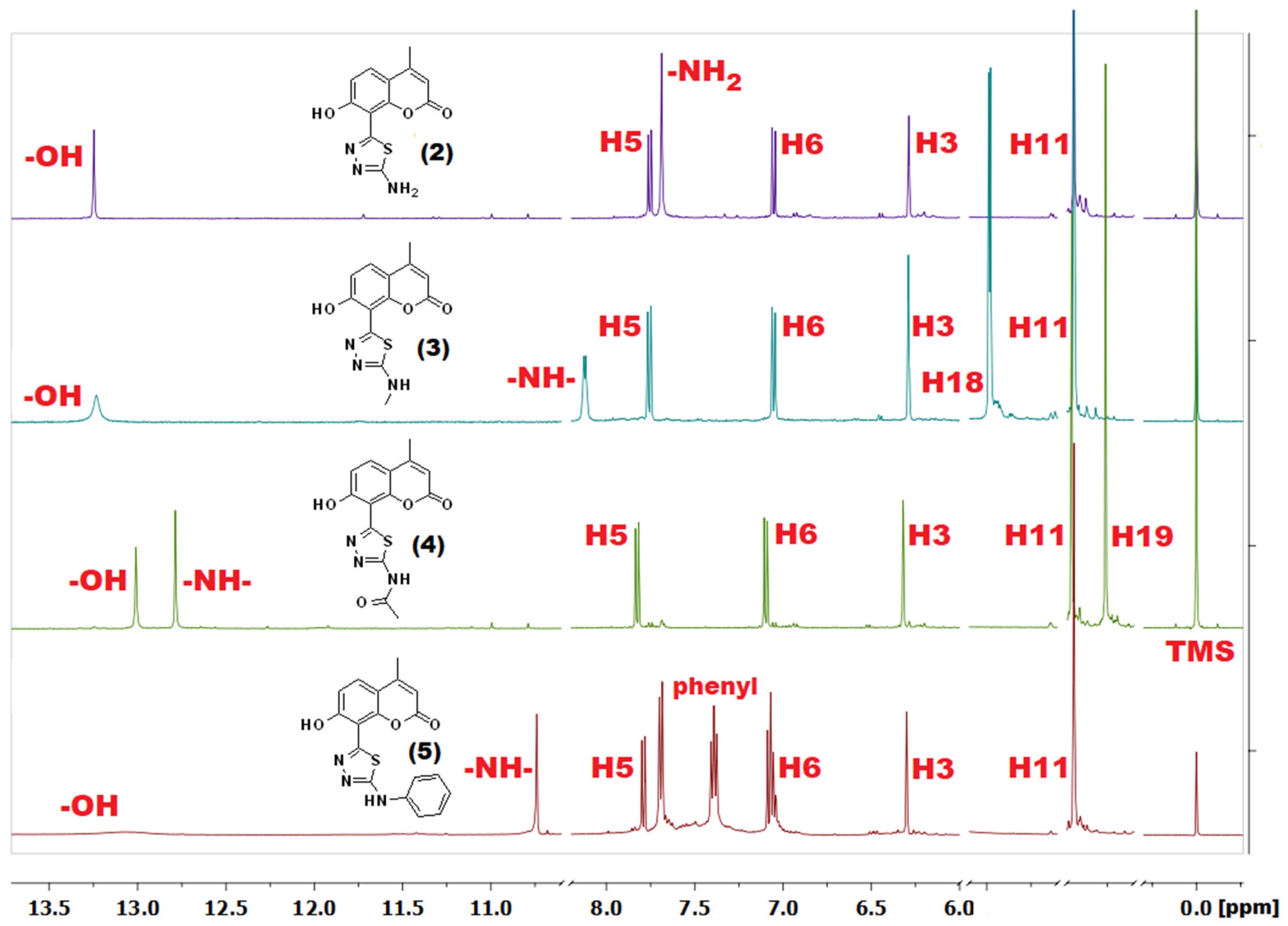
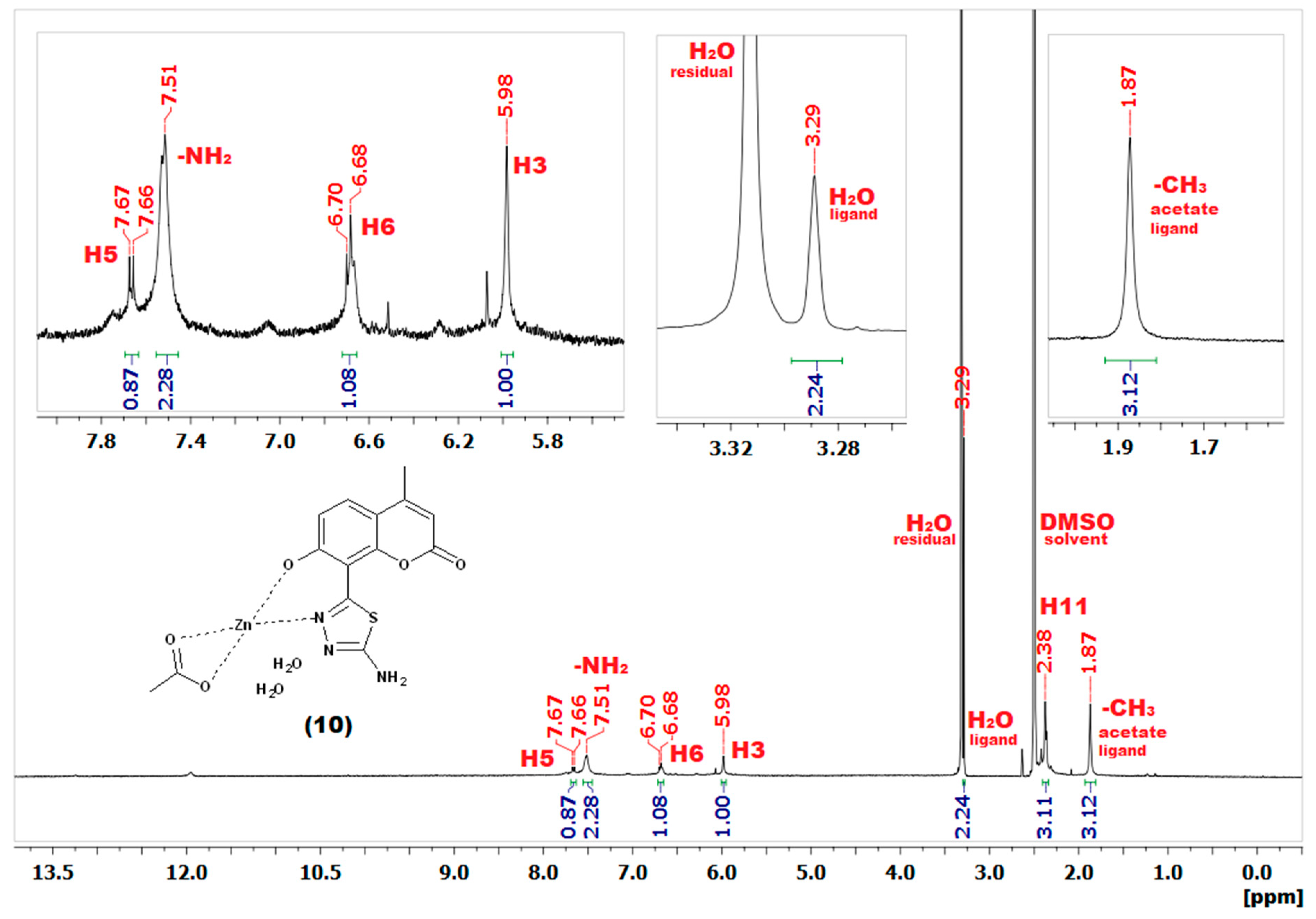
2.3. NMR Spectroscopy
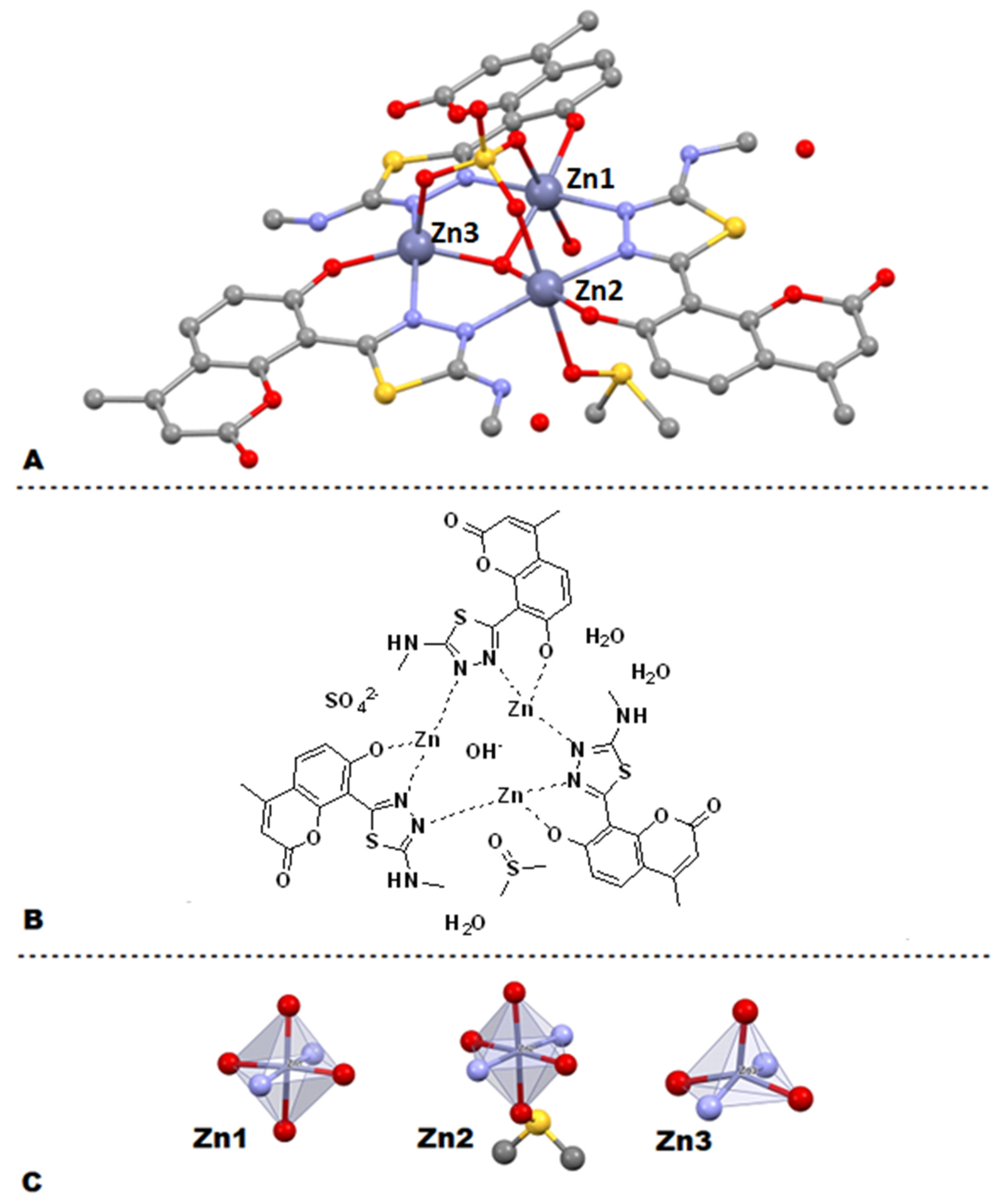
2.4. X-ray Crystallography
2.5. Microanalysis (C, H, N, and S) and Atomic Absorption Spectroscopy (AAS)
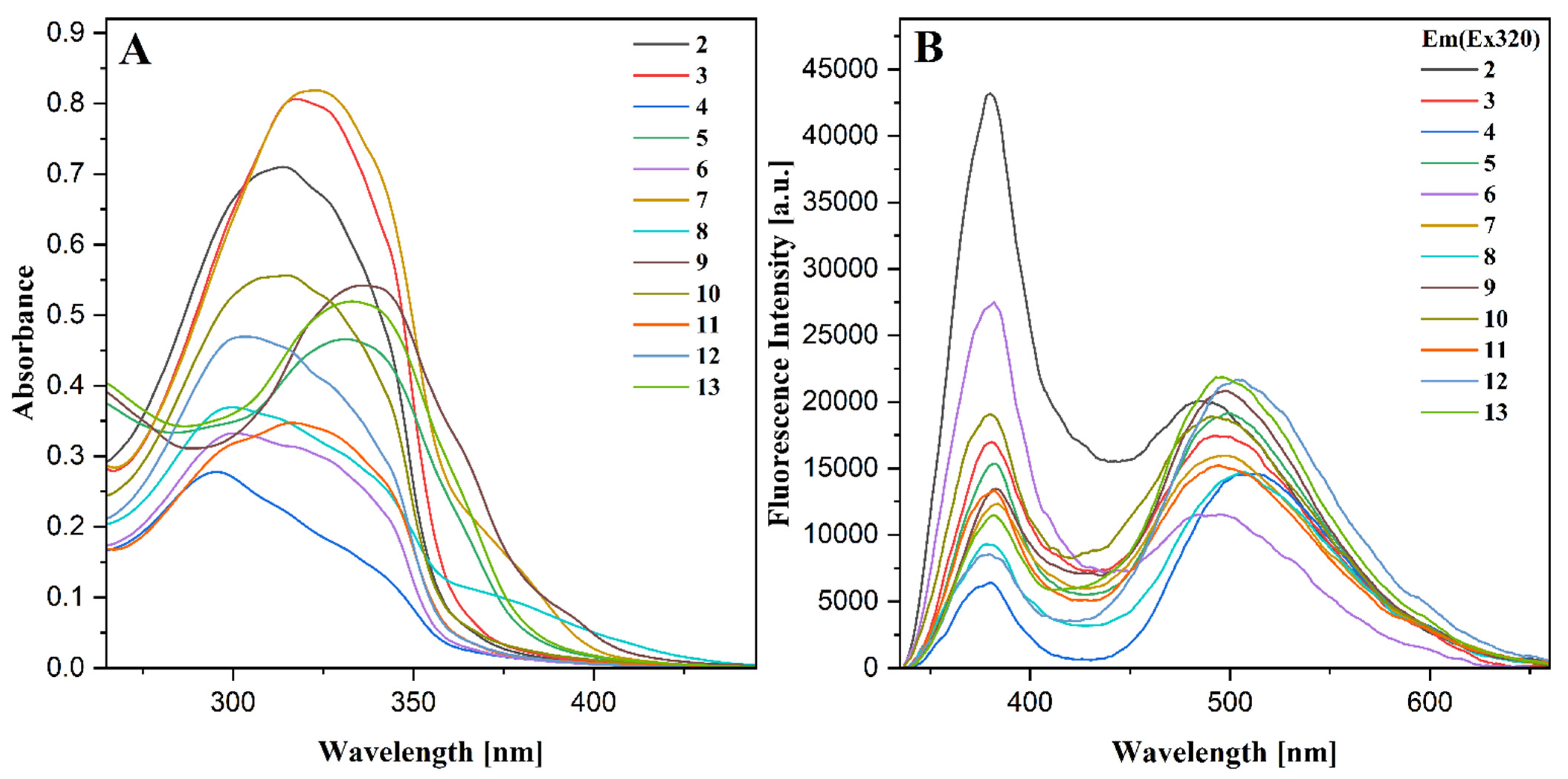
2.6. UV-Vis Spectroscopy
2.7. Fluorescence Spectroscopy
2.8. Antibacterial Activity
2.9. AChE Inhibition Activity
3. Experimental
3.1. Materials and Methods
3.2. Synthesis of 7-Hydroxy-4-methyloumarin-8-carboxylic Acid (1)
3.3. Synthesis of Coumarin-Thiadiazole Hybrids (2, 3, and 5)
3.4. Synthesis of Coumarin-Thiadiazole Hybrid (4)
3.5. Synthesis of Cu(II) (6–9) and Zn(II) (10–13) Complexes
3.6. Determination of Minimum Inhibitory Concentration (MIC) and Minimum Bactericidal Concentration (MBC)
3.7. Determination of AChE Inhibition Activity
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Carneiro, A.; Matos, M.J.; Uriarte, E.; Santana, L. Trending Topics on Coumarin and Its Derivatives in 2020. Molecules 2021, 26, 501. [Google Scholar] [CrossRef]
- Ioannis, F.; Dimitra, H.-L. Hybrids of Coumarin Derivatives as Potent and Multifunctional Bioactive Agents: A Review. Med. Chem. 2020, 16, 272–306. [Google Scholar]
- Patil, P.O.; Bari, S.B.; Firke, S.D.; Deshmukh, P.K.; Donda, S.T.; Patil, D.A. A comprehensive review on synthesis and designing aspects of coumarin derivatives as monoamine oxidase inhibitors for depression and Alzheimer’s disease. Bioorg. Med. Chem. 2013, 21, 2434–2450. [Google Scholar] [CrossRef]
- Sandhu, S.; Bansal, Y.; Silakari, O.; Bansal, G. Coumarin hybrids as novel therapeutic agents. Bioorg. Med. Chem. 2014, 22, 3806–3814. [Google Scholar] [CrossRef] [PubMed]
- Revankar, H.M.; Bukhari, S.N.A.; Kumar, G.B.; Qin, H.-L. Coumarins scaffolds as COX inhibitors. Bioorg. Chem. 2017, 71, 146–159. [Google Scholar] [CrossRef] [PubMed]
- Calcio Gaudino, E.; Tagliapietra, S.; Martina, K.; Palmisano, G.; Cravotto, G. Recent advances and perspectives in the synthesis of bioactive coumarins. RSC Adv. 2016, 6, 46394–46405. [Google Scholar] [CrossRef]
- Yusufzai, S.K.; Khan, M.S.; Sulaiman, O.; Osman, H.; Lamjin, D.N. Molecular docking studies of coumarin hybrids as potential acetylcholinesterase, butyrylcholinesterase, monoamine oxidase A/B and β-amyloid inhibitors for Alzheimer’s disease. Chem. Cent. J. 2018, 12, 1–57. [Google Scholar] [CrossRef]
- Hu, Y.; Li, C.-Y.; Wang, X.-M.; Yang, Y.-H.; Zhu, H.-L. 1,3,4-Thiadiazole: Synthesis, Reactions, and Applications in Medicinal, Agricultural, and Materials Chemistry. Chem. Rev. 2014, 114, 5572–5610. [Google Scholar] [CrossRef]
- Serban, G.; Stanasel, O.; Serban, E.; Bota, S. 2-Amino-1,3,4-thiadiazole as a potential scaffold for promising antimicrobial agents. Drug Des. Dev. Ther. 2018, 12, 1545–1566. [Google Scholar] [CrossRef] [Green Version]
- Dawood, K.M.; Farghaly, T.A. Thiadiazole inhibitors: A patent review. Expert Opin. Ther. Pat. 2017, 27, 477–505. [Google Scholar] [CrossRef] [PubMed]
- Matysiak, J.; Skrzypek, A.; Niewiadomy, A. Synthesis and antifungal activity of novel 5-substituted 4-(1,3,4-thiadiazol-2-yl) benzene-1,3-diols. Heteroat. Chem. 2010, 21, 533–540. [Google Scholar] [CrossRef]
- Chudzik, B.; Bonio, K.; Dabrowski, W.; Pietrzak, D.; Niewiadomy, A.; Olender, A.; Malodobry, K.; Gagoś, M. Synergistic antifungal interactions of amphotericin B with 4-(5-methyl-1,3,4-thiadiazole-2-yl) benzene-1,3-diol. Sci. Rep. 2019, 9, 12945. [Google Scholar] [CrossRef]
- Szeliga, M.; Karpińska, M.; Rola, R.; Niewiadomy, A. Design, synthesis and biological evaluation of novel 1,3,4-thiadiazole derivatives as anti-glioblastoma agents targeting the AKT pathway. Bioorg. Chem. 2020, 105, 104362. [Google Scholar] [CrossRef]
- Skrzypek, A.; Matysiak, J.; Niewiadomy, A.; Bajda, M.; Szymański, P. Synthesis and biological evaluation of 1,3,4-thiadiazole analogues as novel AChE and BuChE inhibitors. Eur. J. Med. Chem. 2013, 62, 311–319. [Google Scholar] [CrossRef] [PubMed]
- Skrzypek, A.; Matysiak, J.; Karpińska, M.; Czarnecka, K.; Kręcisz, P.; Stary, D.; Kukułowicz, J.; Paw, B.; Bajda, M.; Szymański, P.; et al. Biological evaluation and molecular docking of novel 1,3,4-thiadiazole-resorcinol conjugates as multifunctional cholinesterases inhibitors. Bioorg. Chem. 2021, 107, 104617. [Google Scholar] [CrossRef]
- Creaven, B.S.; Egan, D.A.; Karcz, D.; Kavanagh, K.; McCann, M.; Mahon, M.; Noble, A.; Thati, B.; Walsh, M. Synthesis, characterisation and antimicrobial activity of copper (II) and manganese (II) complexes of coumarin-6,7-dioxyacetic acid (cdoaH2) and 4-methylcoumarin-6,7-dioxyacetic acid (4-MecdoaH2): X-ray crystal structures of [Cu(cdoa)(phen)2] 8.8H2O and [Cu(4-Mecdoa)(phen)2] 13H2O (phen = 1,10-phenanthroline). J. Inorg. Biochem. 2007, 101, 1108–1119. [Google Scholar] [PubMed] [Green Version]
- Creaven, B.S.; Devereux, M.; Karcz, D.; Kellett, A.; McCann, M.; Noble, A.; Walsh, M. Copper (II) complexes of coumarin-derived Schiff bases and their anti-Candida activity. J. Inorg. Biochem. 2009, 103, 1196–1203. [Google Scholar] [CrossRef] [Green Version]
- Creaven, B.S.; Czeglédi, E.; Devereux, M.; Enyedy, E.A.; Foltyn-Arfa Kia, A.; Karcz, D.; Kellett, A.; McClean, S.; Nagy, N.V.; Noble, A.; et al. Biological activity and coordination modes of copper (II) complexes of Schiff base-derived coumarin ligands. Dalton Trans. 2010, 39, 10854–10865. [Google Scholar] [CrossRef] [PubMed]
- Dömötör, O.; Tuccinardi, T.; Karcz, D.; Walsh, M.; Creaven, B.S.; Enyedy, É.A. Interaction of anticancer reduced Schiff base coumarin derivatives with human serum albumin investigated by fluorescence quenching and molecular modeling. Bioorg. Chem. 2014, 52, 16–23. [Google Scholar] [CrossRef] [Green Version]
- MacLean, L.; Karcz, D.; Jenkins, H.; McClean, S.; Devereux, M.; Howe, O.; Pereira, M.D.; May, N.V.; Enyedy, É.A.; Creaven, B.S. Copper (II) complexes of coumarin-derived Schiff base ligands: Pro- or antioxidant activity in MCF-7 cells? J. Inorg. Biochem. 2019, 197, 110702. [Google Scholar] [CrossRef] [Green Version]
- Sullivan, M.; Kia, A.F.-A.; Long, M.; Walsh, M.; Kavanagh, K.; McClean, S.; Creaven, B.S. Isolation and characterisation of silver (I) complexes of substituted coumarin-4-carboxylates which are effective against Pseudomonas aeruginosa biofilms. Polyhedron 2014, 67, 549–559. [Google Scholar] [CrossRef] [Green Version]
- Mujahid, M.; Kia, A.F.A.; Duff, B.; Egan, D.A.; Devereux, M.; McClean, S.; Walsh, M.; Trendafilova, N.; Georgieva, I.; Creaven, B.S. Spectroscopic studies, DFT calculations, and cytotoxic activity of novel silver (I) complexes of hydroxy ortho-substituted-nitro-2 H-chromen-2-one ligands and a phenanthroline adduct. J. Inorg. Biochem. 2015, 153, 103–113. [Google Scholar] [CrossRef]
- Mujahid, M.; Trendafilova, N.; Arfa-Kia, A.F.; Rosair, G.; Kavanagh, K.; Devereux, M.; Walsh, M.; McClean, S.; Creaven, B.S.; Georgieva, I. Novel silver (I) complexes of coumarin oxyacetate ligands and their phenanthroline adducts: Biological activity, structural and spectroscopic characterisation. J. Inorg. Biochem. 2016, 163, 53–67. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Starzak, K.; Matwijczuk, A.; Creaven, B.; Matwijczuk, A.; Wybraniec, S.; Karcz, D. Fluorescence quenching-based mechanism for determination of hypochlorite by coumarin-derived sensors. Int. J. Mol. Sci. 2019, 20, 281. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Starzak, K.; Creaven, B.; Matwijczuk, A.; Matwijczuk, A.; Karcz, D. Anti-hypochlorite and catalytic activity of commercially available moringa oleifera diet supplement. Molecules 2019, 24, 3330. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Starzak, K.; Świergosz, T.; Matwijczuk, A.; Creaven, B.; Podleśny, J.; Karcz, D. Anti-hypochlorite, antioxidant, and catalytic activity of three polyphenol-rich super-foods investigated with the use of coumarin-based sensors. Biomolecules 2020, 10, 723. [Google Scholar] [CrossRef]
- Karcz, D.; Matwijczuk, A.; Boroń, B.; Creaven, B.; Fiedor, L.; Niewiadomy, A.; Gagoś, M. Isolation and spectroscopic characterization of Zn (II), Cu (II), and Pd (II) complexes of 1,3,4-thiadiazole-derived ligand. J. Mol. Struct. 2017, 1128, 44–50. [Google Scholar] [CrossRef]
- Karcz, D.; Starzak, K.; Matwijczuk, A.; Gładyszewska, B.; Matwijczuk, A.; Niewiadomy, A.; Gagoś, M. Preparation, spectroscopic characteristics and antioxidant activity of novel 1,3,4-thiadiazole-based coumarin hybrids. Przem. Chem. 2017, 96, 2157–2161. [Google Scholar]
- Karcz, D.; Matwijczuk, A.; Kamiński, D.; Creaven, B.; Ciszkowicz, E.; Lecka-Szlachta, K.; Starzak, K. Structural features of 1,3,4-thiadiazole-derived ligands and their Zn (II) and Cu (II) complexes which demonstrate synergistic antibacterial effects with kanamycin. Int. J. Mol. Sci. 2020, 21, 5735. [Google Scholar] [CrossRef]
- Fair, R.J.; Tor, Y. Antibiotics and bacterial resistance in the 21st century. Perspect. Med. Chem. 2014, 6, 25–64. [Google Scholar] [CrossRef] [Green Version]
- Ventola, C.L. The antibiotic resistance crisis: Part 1: Causes and threats. Pharm. Ther. 2015, 40, 277–283. [Google Scholar]
- Creaven, B.S.; Egan, D.A.; Kavanagh, K.; McCann, M.; Noble, A.; Thati, B.; Walsh, M. Synthesis, characterization and antimicrobial activity of a series of substituted coumarin-3-carboxylatosilver (I) complexes. Inorg. Chim. Acta 2006, 359, 3976–3984. [Google Scholar] [CrossRef]
- Hureau, C.; Dorlet, P. Coordination of redox active metal ions to the amyloid precursor protein and to amyloid-β peptides involved in Alzheimer disease. Part 2: Dependence of Cu (II) binding sites with Aβ sequences. Coord. Chem. Rev. 2012, 256, 2175–2187. [Google Scholar] [CrossRef]
- Kozlowski, H.; Janicka-Klos, A.; Brasun, J.; Gaggelli, E.; Valensin, D.; Valensin, G. Copper, iron, and zinc ions homeostasis and their role in neurodegenerative disorders (metal uptake, transport, distribution and regulation). Coord. Chem. Rev. 2009, 253, 2665–2685. [Google Scholar] [CrossRef]
- Kozlowski, H.; Luczkowski, M.; Remelli, M.; Valensin, D. Copper, zinc and iron in neurodegenerative diseases (Alzheimer’s, Parkinson’s and prion diseases). Coord. Chem. Rev. 2012, 256, 2129–2141. [Google Scholar] [CrossRef]
- Valensin, D.; Gabbiani, C.; Messori, L. Metal compounds as inhibitors of β-amyloid aggregation. Perspectives for an innovative metallotherapeutics on Alzheimer’s disease. Coord. Chem. Rev. 2012, 256, 2357–2366. [Google Scholar] [CrossRef]
- Zatta, P.; Drago, D.; Bolognin, S.; Sensi, S.L. Alzheimer’s disease, metal ions and metal homeostatic therapy. Trends Pharmacol. Sci. 2009, 30, 346–355. [Google Scholar] [CrossRef] [PubMed]
- Morrison, R.; Al-Rawi, J.M.A. Synthesis, structure elucidation, DNA-PK, PI3K, anti-platelet and anti-bacteria activity of linear 5, 6, and 10-substituted-2-morpholino-chromen-oxazine-dione and angular 3, 4, 6-substituted-8-morpholino-chromen-oxazine-2,10-dione. J. Enzym. Inhib. Med. Chem. 2016, 31, 86–95. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Matwijczuk, A.; Karcz, D.; Walkowiak, R.; Furso, J.; Gladyszewska, B.; Wybraniec, S.; Niewiadomy, A.; Karwasz, G.P.; Gagos, M. Effect of Solvent Polarizability on the Keto/Enol Equilibrium of Selected Bioactive Molecules from the 1,3,4-Thiadiazole Group with a 2,4-Hydroxyphenyl Function. J. Phys. Chem. A 2017, 121, 1402–1411. [Google Scholar] [CrossRef] [PubMed]
- Matwijczuk, A.; Górecki, A.; Kamiński, D.; Myśliwa-Kurdziel, B.; Fiedor, L.; Niewiadomy, A.; Karwasz, G.P.; Gagoś, M. Influence of Solvent Polarizability on the Keto-Enol Equilibrium in 4-[5-(naphthalen-1-ylmethyl)-1,3,4-thiadiazol-2-yl] benzene-1,3-diol. J. Fluoresc. 2015, 25, 1867–1874. [Google Scholar] [CrossRef]
- Silverstein, R.M.; Webster, F.X.; Kiemle, D. Spectrometric Identification of Organic Compounds, 7th ed.; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Fulmer, G.R.; Miller, A.J.M.; Sherden, N.H.; Gottlieb, H.E.; Nudelman, A.; Stoltz, B.M.; Bercaw, J.E.; Goldberg, K.I. NMR Chemical Shifts of Trace Impurities: Common Laboratory Solvents, Organics, and Gases in Deuterated Solvents Relevant to the Organometallic Chemist. Organometallics 2010, 29, 2176–2179. [Google Scholar] [CrossRef] [Green Version]
- Ishioka, T.; Shibata, Y.; Takahashi, M.; Kanesaka, I.; Kitagawa, Y.; Nakamura, K.T. Vibrational spectra and structures of zinc carboxylates I. Zinc acetate dihydrate. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 1998, 54, 1827–1835. [Google Scholar] [CrossRef]
- Matwijczuk, A.; Karcz, D.; Pustuła, K.; Makowski, M.; Górecki, A.; Kluczyk, D.; Karpińska, M.M.; Niewiadomy, A.; Gagoś, M. Spectroscopic and theoretical studies of fluorescence effects in bio-active: 4-(5-(methyl-1,3,4-thiadiazol-2-yl)) benzene-1,3-diol and 4-(5-(methylamino-1,3,4-thiadiazol-2-yl)) benzene-1,3-diol compounds: Effect of molecular aggregation and amino group position. J. Lumin. 2018, 201, 44–56. [Google Scholar]
- Czernel, G.; Budziak, I.; Oniszczuk, A.; Karcz, D.; Pustuła, K.; Górecki, A.; Matwijczuk, A.; Gładyszewska, B.; Gagoś, M.; Niewiadomy, A.; et al. ESIPT-Related Origin of Dual Fluorescence in the Selected Model 1,3,4-Thiadiazole Derivatives. Molecules 2020, 25, 4168. [Google Scholar] [CrossRef]
- Budziak, I.; Karcz, D.; Makowski, M.; Myśliwa-Kurdziel, B.; Kasprzak, K.; Matwijczuk, A.; Chruściel, E.; Oniszczuk, A.; Adwent, L.; Matwijczuk, A. Spectroscopic and theoretical investigation into substituent- and aggregation-related dual fluorescence effects in the selected 2-amino-1,3,4-thiadiazoles. J. Mol. Liq. 2019, 291, 111261. [Google Scholar] [CrossRef]
- Matwijczuk, A.; Kaminski, D.; Gorecki, A.; Ludwiczuk, A.; Niewiadomy, A.; Mackowski, S.; Gagos, M. Spectroscopic Studies of Dual Fluorescence in 2-((4-Fluorophenyl)amino)-5-(2,4-dihydroxybenzeno)-1,3,4-thiadiazole. J. Phys. Chem. A 2015, 119, 10791–10805. [Google Scholar] [CrossRef]
- Czernel, G.; Matwijczuk, A.; Karcz, D.; Gorecki, A.; Niemczynowicz, A.; Szczes, A.; Gladyszewski, G.; Matwijczuk, A.; Gladyszewska, B.; Niewiadomy, A. Spectroscopic Studies of Dual Fluorescence in 2-(4-Fluorophenylamino)-5-(2,4-dihydroxybenzeno)-1,3,4-thiadiazole: Effect of Molecular Aggregation in a Micellar System. Molecules 2018, 23, 2861. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bochkov, A.Y.; Akchurin, I.O.; Dyachenko, O.A.; Traven, V.F. NIR-fluorescent coumarin-fused BODIPY dyes with large Stokes shifts. Chem. Commun. 2013, 49, 11653–11655. [Google Scholar] [CrossRef]
- Chen, W.; Yue, X.; Li, W.; Hao, Y.; Zhang, L.; Zhu, L.; Sheng, J.; Song, X. A phenothiazine coumarin-based red emitting fluorescent probe for nanomolar detection of thiophenol with a large Stokes shift. Sens. Actuators B Chem. 2017, 245, 702–710. [Google Scholar] [CrossRef]
- Ujan, R.; Bahadur, A.; Shabir, G.; Iqbal, S.; Saeed, A.; Channar, P.A.; Mahmood, Q.; Shoaib, M.; Arshad, I.; Saifullah, M.; et al. Facile synthesis of novel fluorescent thiazole coumarinyl compounds: Electrochemical, time resolve fluorescence, and solvatochromic study. J. Mol. Struct. 2021, 1227, 129422. [Google Scholar] [CrossRef]
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-Induced Emission: Together We Shine, United We Soar! Chem. Rev. 2015, 115, 11718–11940. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Lam, J.W.Y.; Kwok, R.T.K.; Liu, B.; Tang, B.Z. Aggregation-induced emission: Fundamental understanding and future developments. Mater. Horiz. 2019, 6, 428–433. [Google Scholar] [CrossRef]
- Asadipour, A.; Alipour, M.; Jafari, M.; Khoobi, M.; Emami, S.; Nadri, H.; Sakhteman, A.; Moradi, A.; Sheibani, V.; Homayouni Moghadam, F.; et al. Novel coumarin-3-carboxamides bearing N-benzylpiperidine moiety as potent acetylcholinesterase inhibitors. Eur. J. Med. Chem. 2013, 70, 623–630. [Google Scholar] [CrossRef] [PubMed]
- Yao, D.; Wang, J.; Wang, G.; Jiang, Y.; Shang, L.; Zhao, Y.; Huang, J.; Yang, S.; Wang, J.; Yu, Y. Design, synthesis and biological evaluation of coumarin derivatives as novel acetylcholinesterase inhibitors that attenuate H2O2-induced apoptosis in SH-SY5Y cells. Bioorg. Chem. 2016, 68, 112–123. [Google Scholar] [CrossRef] [PubMed]
- Ghanei-Nasab, S.; Khoobi, M.; Hadizadeh, F.; Marjani, A.; Moradi, A.; Nadri, H.; Emami, S.; Foroumadi, A.; Shafiee, A. Synthesis and anticholinesterase activity of coumarin-3-carboxamides bearing tryptamine moiety. Eur. J. Med. Chem. 2016, 121, 40–46. [Google Scholar] [CrossRef] [Green Version]
- Joubert, J.; Foka, G.B.; Repsold, B.P.; Oliver, D.W.; Kapp, E.; Malan, S.F. Synthesis and evaluation of 7-substituted coumarin derivatives as multimodal monoamine oxidase-B and cholinesterase inhibitors for the treatment of Alzheimer’s disease. Eur. J. Med. Chem. 2017, 125, 853–864. [Google Scholar] [CrossRef]
- de Souza, L.G.; Rennó, M.N.; Figueroa-Villar, J.D. Coumarins as cholinesterase inhibitors: A review. Chem.-Biol. Interact. 2016, 254, 11–23. [Google Scholar] [CrossRef] [PubMed]
- Xie, S.-S.; Wang, X.; Jiang, N.; Yu, W.; Wang, K.D.G.; Lan, J.-S.; Li, Z.-R.; Kong, L.-Y. Multi-target tacrine-coumarin hybrids: Cholinesterase and monoamine oxidase B inhibition properties against Alzheimer’s disease. Eur. J. Med. Chem. 2015, 95, 153–165. [Google Scholar] [CrossRef]
- Hamulakova, S.; Janovec, L.; Hrabinova, M.; Spilovska, K.; Korabecny, J.; Kristian, P.; Kuca, K.; Imrich, J. Synthesis and Biological Evaluation of Novel Tacrine Derivatives and Tacrine–Coumarin Hybrids as Cholinesterase Inhibitors. J. Med. Chem. 2014, 57, 7073–7084. [Google Scholar] [CrossRef]
- Xie, S.-S.; Lan, J.-S.; Wang, X.; Wang, Z.-M.; Jiang, N.; Li, F.; Wu, J.-J.; Wang, J.; Kong, L.-Y. Design, synthesis and biological evaluation of novel donepezil–coumarin hybrids as multi-target agents for the treatment of Alzheimer’s disease. Bioorg. Med. Chem. 2016, 24, 1528–1539. [Google Scholar] [CrossRef]
- Agilent Technologies UK Ltd. CrysAlisPRO, Oxford Diffraction; Agilent Technologies UK Ltd.: Yarnton, UK, 2014. [Google Scholar]
- Sheldrick, G.M. SHELXT—Integrated space-group and crystal-structure determination. Acta Crystallogr. A 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sheldrick, G. Crystal structure refinement with SHELXL. Acta Crystallogr. Sect. C 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Wilson, A.J.C. International Tables for Crystallograpyhy; Kluwer Academic Publishers: Dordrecht, The Netherlands, 1992. [Google Scholar]
- Bocian, A.; Ciszkowicz, E.; Hus, K.K.; Buczkowicz, J.; Lecka-Szlachta, K.; Pietrowska, M.; Petrilla, V.; Petrillova, M.; Legáth, Ľ.; Legáth, J. Antimicrobial Activity of Protein Fraction from Naja ashei Venom against Staphylococcus epidermidis. Molecules 2020, 25, 293. [Google Scholar] [CrossRef] [Green Version]
- Pusz, J.; Ciszkowicz, E.; Lecka-Szlachta, K.; Wolowiec, S.; Woźnicka, E. Synthesis and antibacterial activity of La (III), Ce (III), Pr (III), and Nd (III) complexes of chrysin-4’-sulfonate. Acta Pol. Pharm.-Drug Res. 2017, 74, 1101–1110. [Google Scholar]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem. Pharmacol. 1961, 7, 88–95. [Google Scholar] [CrossRef]



| No. | %C | %H | %N | %S | %M (AAS) | Yield | MW [g/mol] | Formula | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Calc. | Found. | Calc. | Found. | Calc. | Found. | Calc. | Found. | Calc. | Found. | [%] | |||
| 2 | 52.36 | 49.54 | 3.30 | 3.32 | 15.26 | 13.75 | 11.65 | 11.75 | - | - | 72 | 275.28 | C12H9N3O3S |
| 3 | 53.97 | 48.58 | 3.83 | 3.83 | 14.52 | 13.28 | 11.08 | 11.32 | - | - | 93 | 289.31 | C13H11N3O3S |
| 4 | 52.99 | 51.79 | 3.49 | 3.54 | 13.24 | 12.81 | 10.10 | 11.25 | - | - | 87 | 317.32 | C14H11N3O4S |
| 5 | 61.53 | 63.90 | 3.73 | 3.57 | 11.96 | 11.65 | 9.12 | 13.29 | - | - | 59 | 351.38 | C18H13N3O3S |
| 6 | 42.13 | 41.37 | 3.54 | 3.01 | 12.28 | 12.33 | 9.37 | 10.12 | 9.29 | 8.01 | 80 | 684.15 | C24H24N6O10S2Cu |
| 7 | 43.85 | 43.45 | 3.96 | 3.46 | 11.80 | 11.70 | 9.00 | 10.27 | 8.92 | 8.95 | 53 | 712.21 | C26H24N6O8S2Cu |
| 8 | 43.78 | 43.41 | 3.67 | 3.17 | 10.94 | 10.84 | 8.35 | 9.88 | 8.27 | 7.10 | 77 | 768.23 | C28H28N6O12S2Cu |
| 9 | 51.70 | 49.40 | 3.86 | 3.24 | 10.05 | 10.69 | 7.67 | 12.56 | 7.60 | 6.46 | 68 | 836.35 | C36H32N6O10S2Cu |
| 10 | 38.68 | 38.44 | 3.48 | 3.19 | 9.67 | 10.73 | 7.37 | 9.22 | 15.04 | 13.80 | 83 | 434.73 | C14H15N3O7SZn |
| 11 11 * | 40.15 37.70 | 39.26 | 3.82 3.32 | 3.23 | 9.36 9.65 | 10.16 | 7.14 12.27 | 8.16 | 14.57 15.02 | 13.70 | 78 | 448.76 1306.28 | C15H17N3O7SZn C41H43N9O18S5Zn3 |
| 12 ** | 45.82 | 45.78 | 3.30 | 3.27 | 11.45 | 11.35 | 8.74 | 10.24 | 8.91 | 6.94 | 89 | 734.03 | C28H24N6O10S2Zn |
| 13 13 ** | 47.03 53.90 | 49.61 | 3.75 3.52 | 3.40 | 8.23 10.48 | 10.77 | 6.28 7.99 | 12.66 | 12.80 8.15 | 6.86 | 71 | 510.83 802.15 | C20H19N3O8SZn C36H28N6O8S2Zn |
| Compound. | MIC/MBC a [mg/mL] | ||||
|---|---|---|---|---|---|
| Gram-Negative | Gram-Positive | ||||
| E. coli | P. aeruginosa | S. aureus | S. epidermidis ATCC12228 | S. epidermidis ATCC 35984 | |
| 2 | 1.04 | 1.04 | 0.13/2.1 | 0.26 | 0.13/2.1 |
| 3 | 3.12 | 3.12 | 3.12 | 1.56 | 3.12 |
| 4 | 1.56 | 3.12 | 0.78 | 1.56 | 1.56 |
| 5 | 0.78 | 1.56 | 0.19/1.56 | 0.39 | 0.39/1.56 |
| 6 | 4.2 | 4.2 | 4.2 | 2.1 | 4.2 |
| 7 | 4.2 | 4.2 | 4.2 | 4.2 | 4.2 |
| 8 | 5.0 | 5.0 | >5.0 | 5.0 | 2.5 |
| 9 | 3.12 | >3.12 | >3.12 | 0.78 | 0.39 |
| 10 | 3.12 | 1.56 | 3.12 | 1.56 | 3.12 |
| 11 | >5.0 | 5.0 | 5.0 | 5.0 | >5.0 |
| 12 | 3.12 | 6.25 | 3.12 | 3.12 | 1.56 |
| 13 | 1.56 | 1.56 | 0.39 | 0.78 | 0.78 |
| chloramphenicol | 3.9 × 10−3 | 0.25 | 7.8 × 10−3 | 7.8 × 10−3 | 1.56 × 10−2 |
| gentamicin | 1.9 × 10−3 | 1.9 | 0.97 × 10−3 | 0.24 × 10−3 | 3.12 × 10−2 |
| kanamycin | 7.8 × 10−3 | NI | 3.9 × 10−3 | 1.9 × 10−3 | NI |
| Compound | IC50[μM] ± SD [μM] |
|---|---|
| 2 | 0.218 ± 0.0084 |
| 3 | 0.211 ± 0.0123 |
| 4 | 0.181 ± 0.0123 |
| 5 | 0.299 ± 0.0194 |
| 6 | 0.232 ± 0.0119 |
| 7 | 0.198 ± 0.0061 |
| 8 | 0.174 ± 0.0181 |
| 9 | 0.190 ± 0.0038 |
| 10 | 0.228 ± 0.0083 |
| 11 | 0.234 ± 0.0185 |
| 12 | 0.184 ± 0.0069 |
| 13 | 0.187 ± 0.0080 |
| Tacrine | 0.053 ± 0.0036 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karcz, D.; Starzak, K.; Ciszkowicz, E.; Lecka-Szlachta, K.; Kamiński, D.; Creaven, B.; Jenkins, H.; Radomski, P.; Miłoś, A.; Ślusarczyk, L.; et al. Novel Coumarin-Thiadiazole Hybrids and Their Cu(II) and Zn(II) Complexes as Potential Antimicrobial Agents and Acetylcholinesterase Inhibitors. Int. J. Mol. Sci. 2021, 22, 9709. https://doi.org/10.3390/ijms22189709
Karcz D, Starzak K, Ciszkowicz E, Lecka-Szlachta K, Kamiński D, Creaven B, Jenkins H, Radomski P, Miłoś A, Ślusarczyk L, et al. Novel Coumarin-Thiadiazole Hybrids and Their Cu(II) and Zn(II) Complexes as Potential Antimicrobial Agents and Acetylcholinesterase Inhibitors. International Journal of Molecular Sciences. 2021; 22(18):9709. https://doi.org/10.3390/ijms22189709
Chicago/Turabian StyleKarcz, Dariusz, Karolina Starzak, Ewa Ciszkowicz, Katarzyna Lecka-Szlachta, Daniel Kamiński, Bernadette Creaven, Hollie Jenkins, Piotr Radomski, Anna Miłoś, Lidia Ślusarczyk, and et al. 2021. "Novel Coumarin-Thiadiazole Hybrids and Their Cu(II) and Zn(II) Complexes as Potential Antimicrobial Agents and Acetylcholinesterase Inhibitors" International Journal of Molecular Sciences 22, no. 18: 9709. https://doi.org/10.3390/ijms22189709
APA StyleKarcz, D., Starzak, K., Ciszkowicz, E., Lecka-Szlachta, K., Kamiński, D., Creaven, B., Jenkins, H., Radomski, P., Miłoś, A., Ślusarczyk, L., & Matwijczuk, A. (2021). Novel Coumarin-Thiadiazole Hybrids and Their Cu(II) and Zn(II) Complexes as Potential Antimicrobial Agents and Acetylcholinesterase Inhibitors. International Journal of Molecular Sciences, 22(18), 9709. https://doi.org/10.3390/ijms22189709











