Design and Development of Nanomaterial-Based Drug Carriers to Overcome the Blood–Brain Barrier by Using Different Transport Mechanisms
Abstract
:1. Introduction
2. Crossing the BBB by Transportation and Disruption
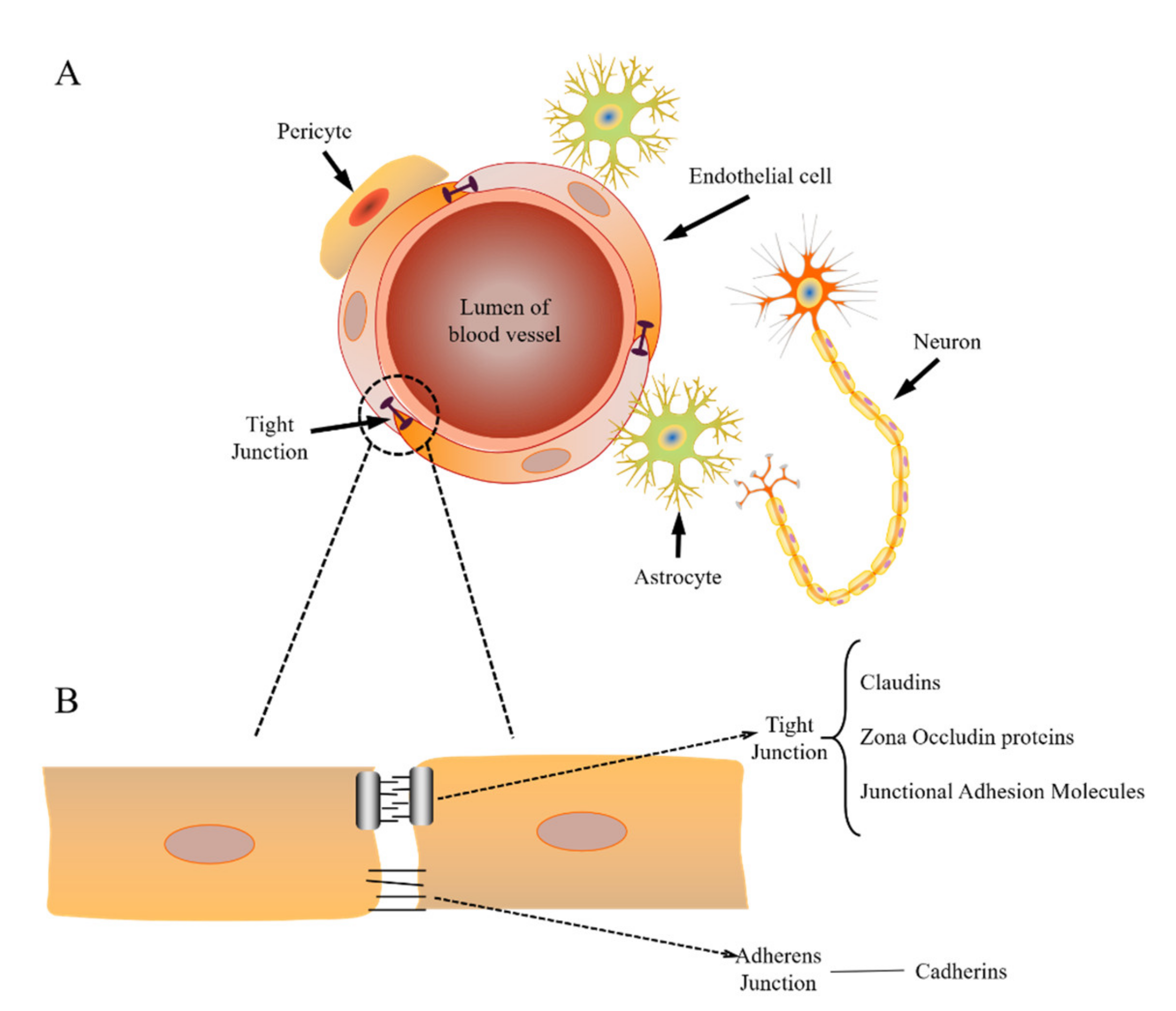
2.1. Structure of the BBB
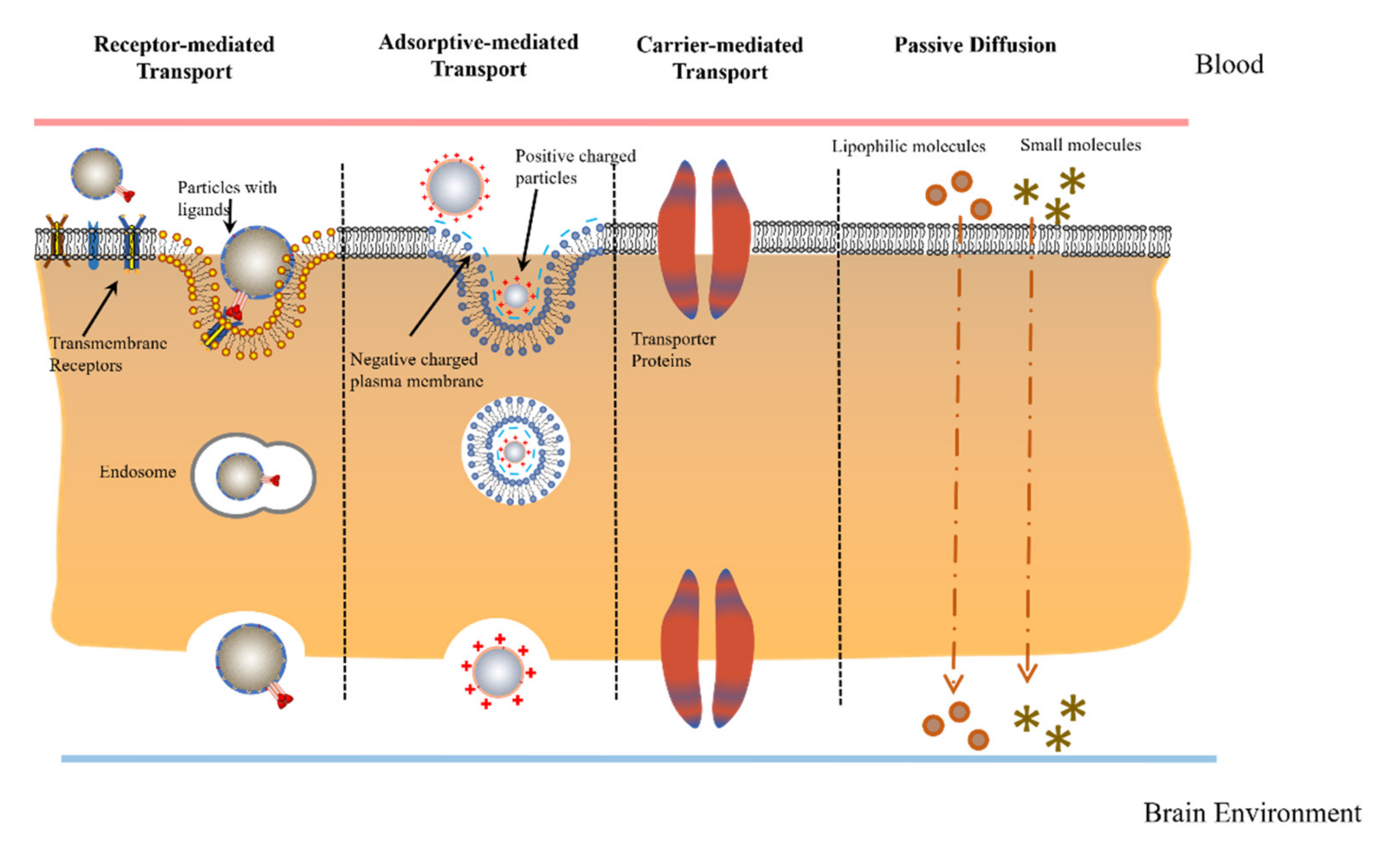
2.2. Physiological Transport Mechanisms
2.3. Viral Disruption Mechanism
3. Drug-Loaded Nanocarriers across the BBB
3.1. Carrier-Mediated Transcytosis
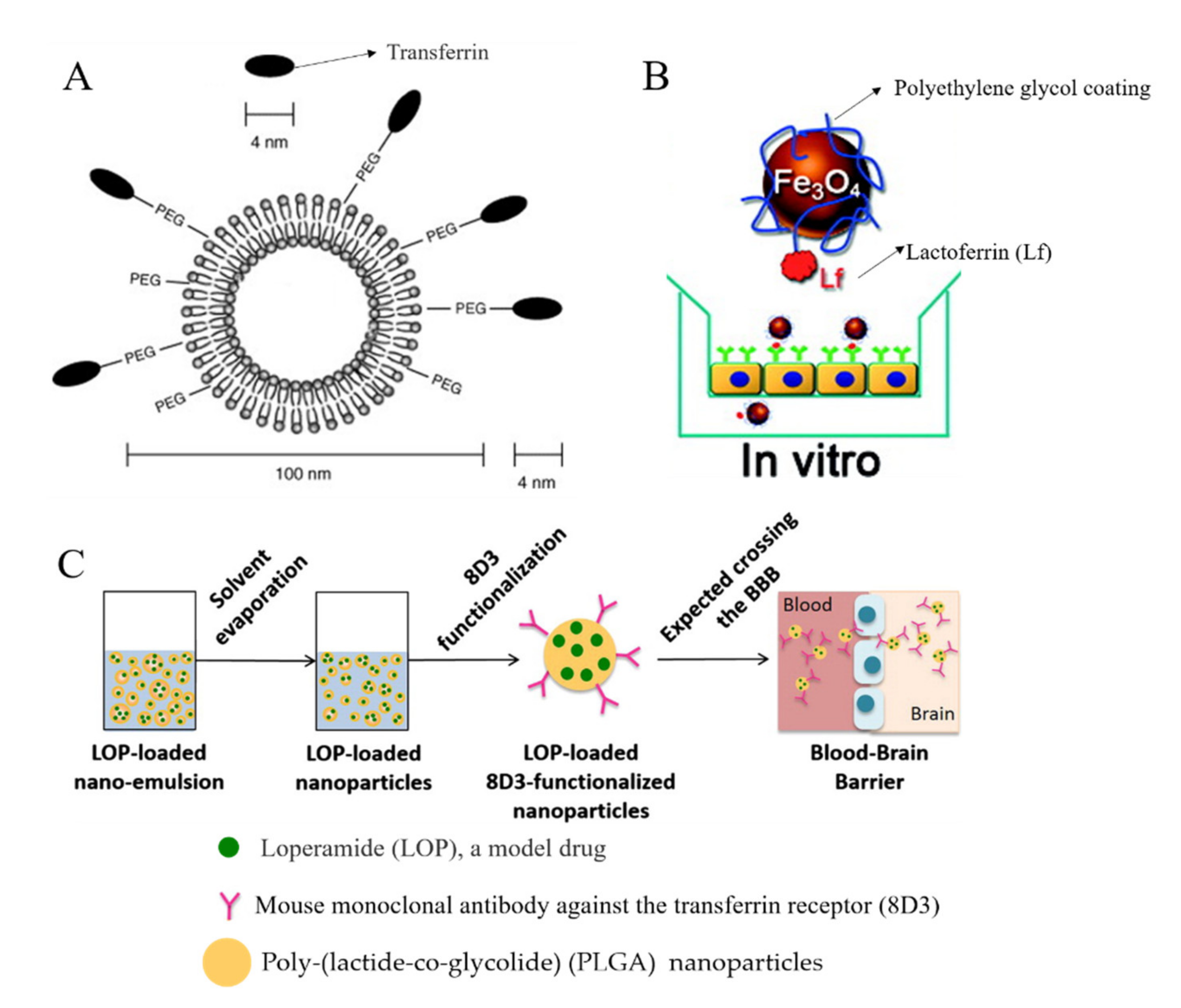
3.2. Adsorptive-Mediated Transcytosis (AMT)
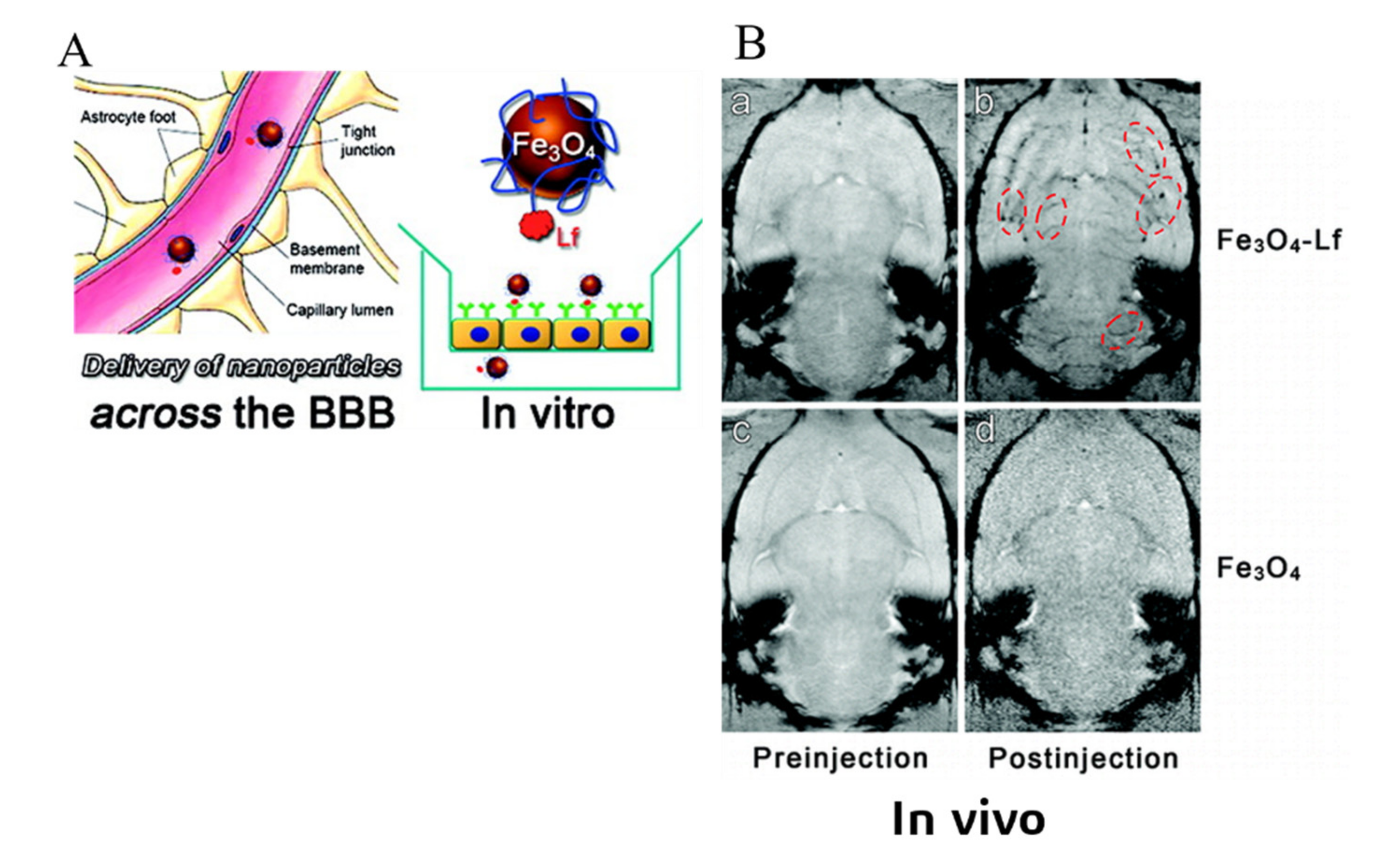
3.3. Receptor-Mediated Transcytosis (RMT)
4. Drug Delivery Strategy by the Manipulation Virus
| Virus | Design of Drug Carrier | Effects on Crossing the BBB | |
|---|---|---|---|
| Rabies Virus | modified rabies virus glycoprotein (RVG) | Poly(mannitol-co-PEI) or chitosan nano carrier as non-viral vector | Enhanced receptor-mediated transcytosis by stimulating the caveolar endocytosis [35,106]. |
| RVG-conjugated polyamidoamine dendrimers—PEG as gene transporter | A clathrin and caveolae mediated energy-depending endocytosis. Higher blood–brain barrier (BBB)-crossing efficiency [59]. | ||
| Herpes simplex virus (HSV) | Non/deficient-replication HSV vector | Vector conjugated with Nerve growth factor (injected into cerebrospinal fluid) | BBB score was largely decreased. A gradual limit recovery of motor function [133]. |
| Vector engineered with vascular endothelial growth factor | Lower infarct volume. Without aggravating cerebral edema. Potent for the therapy of stroke [134]. | ||
| HSV type 1 antibody | Possibly plays a protective role in the early stages of AD [135]. | ||
| HIV | negative factor (Nef) peptide of HIV | Nanomedicine -based delivery | Disrupted the apical blood–brain barrier and reduced transendothelial electrical resistance. Reduced expression of the tight junction protein, ZO-1 [132]. |
| HIV cell-penetrating peptide Tat | Attached on the exterior of the nanocontainer | More than one uptake mechanism via receptor-mediated endocytic pathways [136]. |
5. Routes of Administration of Nanoformulated Drugs Delivered to the Central Nervous System
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Sweeney, M.D.; Sagare, A.P.; Zlokovic, B.V. Cerebrospinal Fluid Biomarkers of Neurovascular Dysfunction in Mild Dementia and Alzheimer’s Disease. J. Cereb. Blood Flow Metab. 2015, 35, 1055–1068. [Google Scholar] [CrossRef] [Green Version]
- Xie, J.; Shen, Z.; Anraku, Y.; Kataoka, K.; Chen, X. Nanomaterial-based blood-brain-barrier (BBB) crossing strategies. Biomaterials 2019, 224, 119491. [Google Scholar] [CrossRef] [PubMed]
- Reddy, P.H.; Tonk, S.; Kumar, S.; Vijayan, M.; Kandimalla, R.; Kuruva, C.S.; Reddy, A.P. A critical evaluation of neuroprotective and neurodegenerative MicroRNAs in Alzheimer’s disease. Biochem. Biophys. Res. Commun. 2017, 483, 1156–1165. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van Tellingen, O.; Yetkin-Arik, B.; de Gooijer, M.C.; Wesseling, P.; Wurdinger, T.; de Vries, H.E. Overcoming the blood–brain tumor barrier for effective glioblastoma treatment. Drug Resist. Updates 2015, 19, 1–12. [Google Scholar] [CrossRef]
- Pardridge, W.M. The Blood-Brain Barrier: Bottleneck in Brain Drug Development. NeuroRx 2005, 2, 12. [Google Scholar] [CrossRef] [PubMed]
- Saraiva, C.; Praça, C.; Ferreira, R.; Santos, T.; Ferreira, L.; Bernardino, L. Nanoparticle-mediated brain drug delivery: Overcoming blood–brain barrier to treat neurodegenerative diseases. J. Control. Release 2016, 235, 34–47. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Abbott, N.J.; Rönnbäck, L.; Hansson, E. Astrocyte–endothelial interactions at the blood–brain barrier. Nat. Rev. Neurosci. 2006, 7, 41–53. [Google Scholar] [CrossRef]
- Abbott, N.J.; Patabendige, A.A.K.; Dolman, D.E.M.; Yusof, S.R.; Begley, D.J. Structure and function of the blood–brain barrier. Neurobiol. Dis. 2010, 37, 13–25. [Google Scholar] [CrossRef] [PubMed]
- Grabrucker, A.M.; Ruozi, B.; Belletti, D.; Pederzoli, F.; Forni, F.; Vandelli, M.A.; Tosi, G. Nanoparticle transport across the blood brain barrier. Tissue Barriers 2016, 4, e1153568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wohlfart, S.; Gelperina, S.; Kreuter, J. Transport of drugs across the blood–brain barrier by nanoparticles. J. Control. Release 2012, 161, 264–273. [Google Scholar] [CrossRef]
- Spindler, K.R.; Hsu, T.-H. Viral disruption of the blood–brain barrier. Trends Microbiol. 2012, 20, 282–290. [Google Scholar] [CrossRef] [Green Version]
- Ivask, A.; Pilkington, E.H.; Blin, T.; Käkinen, A.; Vija, H.; Visnapuu, M.; Quinn, J.F.; Whittaker, M.R.; Qiao, R.; Davis, T.P.; et al. Uptake and transcytosis of functionalized superparamagnetic iron oxide nanoparticles in an in vitro blood brain barrier model. Biomater. Sci. 2018, 6, 314–323. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, H.-C.; Ho, M.-Y.; Tsen, C.-M.; Huang, C.-C.; Wu, C.-C.; Huang, Y.-J.; Hsiao, I.L.; Chuang, C.-Y. From the Cover: Comparative Proteomics Reveals Silver Nanoparticles Alter Fatty Acid Metabolism and Amyloid Beta Clearance for Neuronal Apoptosis in a Triple Cell Coculture Model of the Blood–Brain Barrier. Toxicol. Sci. 2017, 158, 151–163. [Google Scholar] [CrossRef]
- Patel, T.; Zhou, J.; Piepmeier, J.M.; Saltzman, W.M. Polymeric nanoparticles for drug delivery to the central nervous system. Adv. Drug Deliv. Rev. 2012, 64, 701–705. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsai, Y.-C.; Vijayaraghavan, P.; Chiang, W.-H.; Chen, H.-H.; Liu, T.-I.; Shen, M.-Y.; Omoto, A.; Kamimura, M.; Soga, K.; Chiu, H.-C. Targeted Delivery of Functionalized Upconversion Nanoparticles for Externally Triggered Photothermal/Photodynamic Therapies of Brain Glioblastoma. Theranostics 2018, 8, 1435–1448. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Yan, F.; Liang, X.; Wu, M.; Shen, Y.; Chen, M.; Xu, Y.; Zou, G.; Jiang, P.; Tang, C.; et al. Localized delivery of curcumin into brain with polysorbate 80-modified cerasomes by ultrasound-targeted microbubble destruction for improved Parkinson’s disease therapy. Theranostics 2018, 8, 2264–2277. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, P.-P.; Romme, E.; van der Spek, P.J.; Dirven, C.M.F.; Willemsen, R.; Kros, J.M. Glut1/SLC2A1 is crucial for the development of the blood-brain barrier in vivo. Ann. Neurol. 2010, 68, 835–844. [Google Scholar] [CrossRef]
- Peura, L.; Malmioja, K.; Huttunen, K.; Leppänen, J.; Hämäläinen, M.; Forsberg, M.M.; Gynther, M.; Rautio, J.; Laine, K. Design, synthesis and brain uptake of LAT1-targeted amino acid prodrugs of dopamine. Pharm. Res. 2013, 30, 2523–2537. [Google Scholar] [CrossRef] [PubMed]
- Fletcher, N.F.; Wilson, G.K.; Murray, J.; Hu, K.; Lewis, A.; Reynolds, G.M.; Stamataki, Z.; Meredith, L.W.; Rowe, I.A.; Luo, G.; et al. Hepatitis C Virus Infects the Endothelial Cells of the Blood-Brain Barrier. Gastroenterology 2012, 142, 634–643.e6. [Google Scholar] [CrossRef] [Green Version]
- Wang, P.; Dai, J.; Bai, F.; Kong, K.-F.; Wong Susan, J.; Montgomery Ruth, R.; Madri Joseph, A.; Fikrig, E. Matrix Metalloproteinase 9 Facilitates West Nile Virus Entry into the Brain. J. Virol. 2008, 82, 8978–8985. [Google Scholar] [CrossRef] [Green Version]
- Yang, C.-M.; Lin, C.-C.; Lee, I.T.; Lin, Y.-H.; Yang, C.M.; Chen, W.-J.; Jou, M.-J.; Hsiao, L.-D. Japanese encephalitis virus induces matrix metalloproteinase-9 expression via a ROS/c-Src/PDGFR/PI3K/Akt/MAPKs-dependent AP-1 pathway in rat brain astrocytes. J. Neuroinflamm. 2012, 9, 12. [Google Scholar] [CrossRef] [Green Version]
- Chaturvedi, U.C.; Dhawan, R.; Khanna, M.; Mathur, A. Breakdown of the blood-brain barrier during dengue virus infection of mice. J. Gen. Virol. 1991, 72, 859–866. [Google Scholar] [CrossRef]
- Chiu, C.-F.; Chu, L.-W.; Liao, I.-C.; Simanjuntak, Y.; Lin, Y.-L.; Juan, C.-C.; Ping, Y.-H. The Mechanism of the Zika Virus Crossing the Placental Barrier and the Blood-Brain Barrier. Front. Microbiol. 2020, 11, 214. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ou, X.; Liu, Y.; Lei, X.; Li, P.; Mi, D.; Ren, L.; Guo, L.; Guo, R.; Chen, T.; Hu, J.; et al. Characterization of spike glycoprotein of SARS-CoV-2 on virus entry and its immune cross-reactivity with SARS-CoV. Nat. Commun. 2020, 11, 1–12. [Google Scholar] [CrossRef] [Green Version]
- Letko, M.; Marzi, A.; Munster, V. Functional assessment of cell entry and receptor usage for SARS-CoV-2 and other lineage B betacoronaviruses. Nat. Microbiol. 2020, 5, 562–569. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krüger, N.; Herrler, T.; Erichsen, S.; Schiergens, T.S.; Herrler, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280.e8. [Google Scholar] [CrossRef] [PubMed]
- Wrapp, D.; Wang, N.; Corbett, K.S.; Goldsmith, J.A.; Hsieh, C.-L.; Abiona, O.; Graham, B.S.; McLellan, J.S. Cryo-EM structure of the 2019-nCoV spike in the prefusion conformation. Science 2020, 367, 1260–1263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Desforges, M.; Le Coupanec, A.; Stodola, J.K.; Meessen-Pinard, M.; Talbot, P.J. Human coronaviruses: Viral and cellular factors involved in neuroinvasiveness and neuropathogenesis. Virus Res. 2014, 194, 145–158. [Google Scholar] [CrossRef] [PubMed]
- Collins, A.R. In Vitro Detection of Apoptosis in Monocytes/Macrophages Infected with Human Coronavirus. Clin. Vaccine Immunol. 2002, 9, 1392–1395. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lassnig, C.; Sanchez, C.M.; Egerbacher, M.; Walter, I.; Majer, S.; Kolbe, T.; Pallares, P.; Enjuanes, L.; Muller, M. From The Cover: Development of a transgenic mouse model susceptible to human coronavirus 229E. Proc. Natl. Acad. Sci. USA 2005, 102, 8275–8280. [Google Scholar] [CrossRef] [Green Version]
- Gu, J.; Gong, E.; Zhang, B.; Zheng, J.; Gao, Z.; Zhong, Y.; Zou, W.; Zhan, J.; Wang, S.; Xie, Z.; et al. Multiple organ infection and the pathogenesis of SARS. J. Exp. Med. 2005, 202, 415–424. [Google Scholar] [CrossRef]
- Nicholls, J.M.; Butany, J.; Poon, L.L.M.; Chan, K.H.; Beh, S.L.; Poutanen, S.; Peiris, J.S.M.; Wong, M. Time Course and Cellular Localization of SARS-CoV Nucleoprotein and RNA in Lungs from Fatal Cases of SARS. PLoS Med. 2006, 3, e27. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piacentini, R.; De Chiara, G.; Li Puma, D.D.; Ripoli, C.; Marcocci, M.E.; Garaci, E.; Palamara, A.T.; Grassi, C. HSV-1 and Alzheimer’s disease: More than a hypothesis. Front. Pharmacol. 2014, 5, 97. [Google Scholar] [CrossRef] [PubMed]
- Martínez-Torres, F.J.; Wagner, S.; Haas, J.; Kehm, R.; Sellner, J.; Hacke, W.; Meyding-Lamadé, U. Increased presence of matrix metalloproteinases 2 and 9 in short- and long-term experimental herpes simplex virus encephalitis. Neurosci. Lett. 2004, 368, 274–278. [Google Scholar] [CrossRef]
- Park, T.-E.; Singh, B.; Li, H.; Lee, J.-Y.; Kang, S.-K.; Choi, Y.-J.; Cho, C.-S. Enhanced BBB permeability of osmotically active poly(mannitol-co-PEI) modified with rabies virus glycoprotein via selective stimulation of caveolar endocytosis for RNAi therapeutics in Alzheimer’s disease. Biomaterials 2015, 38, 61–71. [Google Scholar] [CrossRef]
- Gralinski, L.E.; Ashley, S.L.; Dixon, S.D.; Spindler, K.R. Mouse Adenovirus Type 1-Induced Breakdown of the Blood-Brain Barrier. J. Virol. 2009, 83, 9398–9410. [Google Scholar] [CrossRef] [Green Version]
- Kajon, A.E.; Brown, C.C.; Spindler, K.R. Distribution of Mouse Adenovirus Type 1 in Intraperitoneally and Intranasally Infected Adult Outbred Mice. J. Virol. 1998, 72, 1219–1223. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ashley, S.L.; Welton, A.R.; Harwood, K.M.; Rooijen, N.V.; Spindler, K.R. Mouse adenovirus type 1 infection of macrophages. Virology 2009, 390, 307–314. [Google Scholar] [CrossRef] [Green Version]
- Johnson, H.L.; Jin, F.; Pirko, I.; Johnson, A.J. Theiler’s murine encephalomyelitis virus as an experimental model system to study the mechanism of blood-brain barrier disruption. J. Neurovirol. 2014, 20, 107–112. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maximova, O.A.; Pletnev, A.G. Flaviviruses and the Central Nervous System: Revisiting Neuropathological Concepts. Annu. Rev. Virol. 2018, 5, 255–272. [Google Scholar] [CrossRef] [PubMed]
- Balin, B.J.; Hudson, A.P. Herpes viruses and Alzheimer’s disease: New evidence in the debate. Lancet Neurol. 2018, 17, 839–841. [Google Scholar] [CrossRef]
- Mustafá, Y.M.; Meuren, L.M.; Coelho, S.V.A.; de Arruda, L.B. Pathways Exploited by Flaviviruses to Counteract the Blood-Brain Barrier and Invade the Central Nervous System. Front. Microbiol. 2019, 10, 525. [Google Scholar] [CrossRef] [PubMed]
- St-Jean, J.R.; Jacomy, H.; Desforges, M.; Vabret, A.; Freymuth, F.; Talbot, P.J. Human Respiratory Coronavirus OC43: Genetic Stability and Neuroinvasion. J. Virol. 2004, 78, 8824–8834. [Google Scholar] [CrossRef] [Green Version]
- Desforges, M.; Miletti, T.C.; Gagnon, M.; Talbot, P.J. Activation of human monocytes after infection by human coronavirus 229E. Virus Res. 2007, 130, 228–240. [Google Scholar] [CrossRef]
- Singh, S.K.; Ruzek, D. Neuroviral Infections: RNA Viruses and Retroviruses; CRC: Boca Raton, FL, USA, 2013; Volume 2. [Google Scholar]
- Arbour, N.; Côté, G.; Lachance, C.; Tardieu, M.; Cashman, N.R.; Talbot, P.J. Acute and Persistent Infection of Human Neural Cell Lines by Human Coronavirus OC43. J. Virol. 1999, 73, 3338–3350. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Baig, A.M.; Khaleeq, A.; Ali, U.; Syeda, H. Evidence of the COVID-19 Virus Targeting the CNS: Tissue Distribution, Host–Virus Interaction, and Proposed Neurotropic Mechanisms. ACS Chem. Neurosci. 2020, 11, 995–998. [Google Scholar] [CrossRef] [Green Version]
- Wu, A.; Peng, Y.; Huang, B.; Ding, X.; Wang, X.; Niu, P.; Meng, J.; Zhu, Z.; Zhang, Z.; Wang, J.; et al. Genome Composition and Divergence of the Novel Coronavirus (2019-nCoV) Originating in China. Cell Host Microbe 2020, 27, 325–328. [Google Scholar] [CrossRef] [Green Version]
- Li, Y.C.; Bai, W.Z.; Hashikawa, T. The neuroinvasive potential of SARS-CoV2 may play a role in the respiratory failure of COVID-19 patients. J. Med Virol. 2020, 92, 552–555. [Google Scholar] [CrossRef] [PubMed]
- Natoli, S.; Oliveira, V.; Calabresi, P.; Maia, L.F.; Pisani, A. Does SARS-Cov-2 invade the brain? Translational lessons from animal models. Eur. J. Neurol. 2020, 27, 1764–1773. [Google Scholar] [CrossRef] [PubMed]
- Xia, H.; Lazartigues, E. Angiotensin-converting enzyme 2 in the brain: Properties and future directions. J. Neurochem. 2008, 107, 1482–1494. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, C.; Wang, Y.; Li, X.; Ren, L.; Zhao, J.; Hu, Y.; Zhang, L.; Fan, G.; Xu, J.; Gu, X.; et al. Clinical features of patients infected with 2019 novel coronavirus in Wuhan, China. Lancet 2020, 395, 497–506. [Google Scholar] [CrossRef] [Green Version]
- Zanin, L.; Saraceno, G.; Panciani, P.P.; Renisi, G.; Signorini, L.; Migliorati, K.; Fontanella, M.M. SARS-CoV-2 can induce brain and spine demyelinating lesions. Acta Neurochir. 2020, 162, 1491–1494. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Huang, J.; Dai, D.; Feng, Y.; Liu, L.; Nie, S. Acute myelitis after SARS-CoV-2 infection: A case report. MedRxiv 2020. [Google Scholar] [CrossRef] [Green Version]
- Paniz-Mondolfi, A.; Bryce, C.; Grimes, Z.; Gordon, R.E.; Reidy, J.; Lednicky, J.; Sordillo, E.M.; Fowkes, M. Central Nervous System Involvement by Severe Acute Respiratory Syndrome Coronavirus-2 (SARS-CoV-2). J. Med Virol. 2020, 92, 699–702. [Google Scholar] [CrossRef] [Green Version]
- Panciani, P.P.; Saraceno, G.; Zanin, L.; Renisi, G.; Signorini, L.; Battaglia, L.; Fontanella, M.M. SARS-CoV-2: “Three-steps” infection model and CSF diagnostic implication. Brain Behav. Immun. 2020, 87, 128. [Google Scholar] [CrossRef] [PubMed]
- Hui, D.S.C.; Zumla, A. Severe Acute Respiratory Syndrome. Infect. Dis. Clin. N. Am. 2019, 33, 869–889. [Google Scholar] [CrossRef]
- Netland, J.; Meyerholz, D.K.; Moore, S.; Cassell, M.; Perlman, S. Severe Acute Respiratory Syndrome Coronavirus Infection Causes Neuronal Death in the Absence of Encephalitis in Mice Transgenic for Human ACE2. J. Virol. 2008, 82, 7264–7275. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Huang, R.; Han, L.; Ke, W.; Shao, K.; Ye, L.; Lou, J.; Jiang, C. Brain-targeting gene delivery and cellular internalization mechanisms for modified rabies virus glycoprotein RVG29 nanoparticles. Biomaterials 2009, 30, 4195–4202. [Google Scholar] [CrossRef]
- Schnell, M.J.; McGettigan, J.P.; Wirblich, C.; Papaneri, A. The cell biology of rabies virus: Using stealth to reach the brain. Nat. Rev. Microbiol. 2010, 8, 51–61. [Google Scholar] [CrossRef]
- Nagel, S.; Su, Y.; Horstmann, S.; Heiland, S.; Gardner, H.; Koziol, J.; Martinez-Torres, F.J.; Wagner, S. Minocycline and hypothermia for reperfusion injury after focal cerebral ischemia in the rat—Effects on BBB breakdown and MMP expression in the acute and subacute phase. Brain Res. 2008, 1188, 198–206. [Google Scholar] [CrossRef]
- Ayala-Nunez, N.V.; Gaudin, R. A viral journey to the brain: Current considerations and future developments. PLOS Pathog. 2020, 16, e1008434. [Google Scholar] [CrossRef]
- Liu, D.; Lin, B.; Shao, W.; Zhu, Z.; Ji, T.; Yang, C. In Vitro and in Vivo Studies on the Transport of PEGylated Silica Nanoparticles across the Blood–Brain Barrier. ACS Appl. Mater. Interfaces 2014, 6, 2131–2136. [Google Scholar] [CrossRef]
- Sonavane, G.; Tomoda, K.; Makino, K. Biodistribution of colloidal gold nanoparticles after intravenous administration: Effect of particle size. Colloids Surf. B Biointerfaces 2008, 66, 274–280. [Google Scholar] [CrossRef]
- Huang, N.; Cheng, S.; Zhang, X.; Tian, Q.; Pi, J.; Tang, J.; Huang, Q.; Wang, F.; Chen, J.; Xie, Z.; et al. Efficacy of NGR peptide-modified PEGylated quantum dots for crossing the blood–brain barrier and targeted fluorescence imaging of glioma and tumor vasculature. Nanomed. Nanotechnol. Biol. Med. 2017, 13, 83–93. [Google Scholar] [CrossRef]
- Mishra, M.K.; Beaty, C.A.; Lesniak, W.G.; Kambhampati, S.P.; Zhang, F.; Wilson, M.A.; Blue, M.E.; Troncoso, J.C.; Kannan, S.; Johnston, M.V.; et al. Dendrimer Brain Uptake and Targeted Therapy for Brain Injury in a Large Animal Model of Hypothermic Circulatory Arrest. ACS Nano 2014, 8, 2134–2147. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Patel, T.R.; Sirianni, R.W.; Strohbehn, G.; Zheng, M.-Q.; Duong, N.; Schafbauer, T.; Huttner, A.J.; Huang, Y.; Carson, R.E.; et al. Highly penetrative, drug-loaded nanocarriers improve treatment of glioblastoma. Proc. Natl. Acad. Sci. USA 2013, 110, 11751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sarvaiya, J.; Agrawal, Y.K. Chitosan as a suitable nanocarrier material for anti-Alzheimer drug delivery. Int. J. Biol. Macromol. 2015, 72, 454–465. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.; Ahn, S.I.; Kim, Y. Nanotherapeutics engineered to cross the blood-brain barrier for advanced drug delivery to the central nervous system. J. Ind. Eng. Chem. 2019, 73, 8–18. [Google Scholar] [CrossRef] [PubMed]
- Agrawal, M.; Tripathi, D.K.; Saraf, S.; Saraf, S.; Antimisiaris, S.G.; Mourtas, S.; Hammarlund-Udenaes, M.; Alexander, A. Recent advancements in liposomes targeting strategies to cross blood-brain barrier (BBB) for the treatment of Alzheimer’s disease. J. Control. Release 2017, 260, 61–77. [Google Scholar] [CrossRef] [PubMed]
- Geldenhuys, W.; Mbimba, T.; Bui, T.; Harrison, K.; Sutariya, V. Brain-targeted delivery of paclitaxel using glutathione-coated nanoparticles for brain cancers. J. Drug Target. 2011, 19, 837–845. [Google Scholar] [CrossRef]
- Geldenhuys, W.; Wehrung, D.; Groshev, A.; Hirani, A.; Sutariya, V. Brain-targeted delivery of doxorubicin using glutathione-coated nanoparticles for brain cancers. Pharm. Dev. Technol. 2015, 20, 497–506. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. Blood-Brain Barrier Carrier-Mediated Transport and Brain Metabolism of Amino Acids. Neurochem. Res. 1998, 23, 635–644. [Google Scholar] [CrossRef] [PubMed]
- Vannucci, S.J.; Maher, F.; Simpson, I.A. Glucose transporter proteins in brain: Delivery of glucose to neurons and glia. Glia 1997, 21, 2–21. [Google Scholar] [CrossRef]
- Jiang, X.; Xin, H.; Ren, Q.; Gu, J.; Zhu, L.; Du, F.; Feng, C.; Xie, Y.; Sha, X.; Fang, X. Nanoparticles of 2-deoxy-d-glucose functionalized poly(ethylene glycol)-co-poly(trimethylene carbonate) for dual-targeted drug delivery in glioma treatment. Biomaterials 2014, 35, 518–529. [Google Scholar] [CrossRef] [PubMed]
- Du, D.; Chang, N.; Sun, S.; Li, M.; Yu, H.; Liu, M.; Liu, X.; Wang, G.; Li, H.; Liu, X.; et al. The role of glucose transporters in the distribution of p-aminophenyl-α-d-mannopyranoside modified liposomes within mice brain. J. Control. Release 2014, 182, 99–110. [Google Scholar] [CrossRef] [PubMed]
- Hao, Z.-F.; Cui, Y.-X.; Li, M.-H.; Du, D.; Liu, M.-F.; Tao, H.-Q.; Li, S.; Cao, F.-Y.; Chen, Y.-L.; Lei, X.-H.; et al. Liposomes modified with P-aminophenyl-α-d-mannopyranoside: A carrier for targeting cerebral functional regions in mice. Eur. J. Pharm. Biopharm. 2013, 84, 505–516. [Google Scholar] [CrossRef]
- Singh, I.; Swami, R.; Jeengar, M.K.; Khan, W.; Sistla, R. p-Aminophenyl-α-d-mannopyranoside engineered lipidic nanoparticles for effective delivery of docetaxel to brain. Chem. Phys. Lipids 2015, 188, 1–9. [Google Scholar] [CrossRef]
- Zhang, C.-X.; Zhao, W.-Y.; Liu, L.; Ju, R.-J.; Mu, L.-M.; Zhao, Y.; Zeng, F.; Xie, H.-J.; Yan, Y.; Lu, W.-L. A nanostructure of functional targeting epirubicin liposomes dually modified with aminophenyl glucose and cyclic pentapeptide used for brain glioblastoma treatment. Oncotarget 2015, 6, 32681. [Google Scholar] [CrossRef]
- Lei, F.; Fan, W.; Li, X.K.; Wang, S.; Hai, L.; Wu, Y. Design, synthesis and preliminary bio-evaluation of glucose–cholesterol derivatives as ligands for brain targeting liposomes. Chin. Chem. Lett. 2011, 22, 831–834. [Google Scholar] [CrossRef]
- Qin, Y.; Fan, W.; Chen, H.; Yao, N.; Tang, W.; Tang, J.; Yuan, W.; Kuai, R.; Zhang, Z.; Wu, Y.; et al. In vitro and in vivo investigation of glucose-mediated brain-targeting liposomes. J. Drug Target. 2010, 18, 536–549. [Google Scholar] [CrossRef]
- Xie, F.; Yao, N.; Qin, Y.; Zhang, Q.; Chen, H.; Yuan, M.; Tang, J.; Li, X.; Fan, W.; Zhang, Q. Investigation of glucose-modified liposomes using polyethylene glycols with different chain lengths as the linkers for brain targeting. Int. J. Nanomed. 2012, 7, 163. [Google Scholar] [CrossRef] [Green Version]
- Liu, J.; He, Y.; Zhang, J.; Li, J.; Yu, X.; Cao, Z.; Meng, F.; Zhao, Y.; Wu, X.; Shen, T.; et al. Functionalized nanocarrier combined seizure-specific vector with P-glycoprotein modulation property for antiepileptic drug delivery. Biomaterials 2016, 74, 64–76. [Google Scholar] [CrossRef] [PubMed]
- Vyas, A.; Jain, A.; Hurkat, P.; Jain, A.; Jain, S.K. Targeting of AIDS related encephalopathy using phenylalanine anchored lipidic nanocarrier. Colloids Surf. B Biointerfaces 2015, 131, 155–161. [Google Scholar] [CrossRef]
- Grover, A.; Hirani, A.; Pathak, Y.; Sutariya, V. Brain-Targeted Delivery of Docetaxel by Glutathione-Coated Nanoparticles for Brain Cancer. AAPS PharmSciTech 2014, 15, 1562–1568. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Venishetty, V.K.; Samala, R.; Komuravelli, R.; Kuncha, M.; Sistla, R.; Diwan, P.V. β-Hydroxybutyric acid grafted solid lipid nanoparticles: A novel strategy to improve drug delivery to brain. Nanomed. Nanotechnol. Biol. Med. 2013, 9, 388–397. [Google Scholar] [CrossRef] [PubMed]
- Devi, R.; Jain, A.; Hurkat, P.; Jain, S.K. Dual Drug Delivery Using Lactic Acid Conjugated SLN for Effective Management of Neurocysticercosis. Pharm. Res. 2015, 32, 3137–3148. [Google Scholar] [CrossRef]
- Dick, A.P.; Harik, S.I.; Klip, A.; Walker, D.M. Identification and characterization of the glucose transporter of the blood-brain barrier by cytochalasin B binding and immunological reactivity. Proc. Natl. Acad. Sci. USA 1984, 81, 7233–7237. [Google Scholar] [CrossRef] [Green Version]
- LaManna, J.C.; Harik, S.I. Regional comparisons of brain glucose influx. Brain Res. 1985, 326, 299–305. [Google Scholar] [CrossRef]
- Nii, T.; Segawa, H.; Taketani, Y.; Tani, Y.; Ohkido, M.; Kishida, S.; Ito, M.; Endou, H.; Kanai, Y.; Takeda, E.; et al. Molecular events involved in up-regulating human Na+-independent neutral amino acid transporter LAT1 during T-cell activation. Biochem. J. 2001, 358, 693–704. [Google Scholar] [CrossRef] [PubMed]
- Kido, Y.; Tamai, I.; Uchino, H.; Suzuki, F.; Sai, Y.; Tsuji, A. Molecular and functional identification of large neutral amino acid transporters LAT1 and LAT2 and their pharmacological relevance at the blood-brain barrier. J. Pharm. Pharmacol. 2001, 53, 497–503. [Google Scholar] [CrossRef] [PubMed]
- Killian, D.M.; Hermeling, S.; Chikhale, P.J. Targeting the Cerebrovascular Large Neutral Amino Acid Transporter (LAT1) Isoform Using a Novel Disulfide-Based Brain Drug Delivery System. Drug Deliv. 2007, 14, 25–31. [Google Scholar] [CrossRef]
- Peura, L.; Malmioja, K.; Laine, K.; Leppänen, J.; Gynther, M.; Isotalo, A.; Rautio, J. Large Amino Acid Transporter 1 (LAT1) Prodrugs of Valproic Acid: New Prodrug Design Ideas for Central Nervous System Delivery. Mol. Pharm. 2011, 8, 1857–1866. [Google Scholar] [CrossRef]
- Sakata, T.; Ferdous, G.; Tsuruta, T.; Satoh, T.; Baba, S.; Muto, T.; Ueno, A.; Kanai, Y.; Endou, H.; Okayasu, I. L-type amino-acid transporter 1 as a novel biomarker for high-grade malignancy in prostate cancer. Pathol. Int. 2009, 59, 7–18. [Google Scholar] [CrossRef]
- Kobayashi, K.; Ohnishi, A.; Promsuk, J.; Shimizu, S.; Kanai, Y.; Shiokawa, Y.; Nagane, M. Enhanced tumor growth elicited by L-type amino acid transporter 1 in human malignant glioma cells. Neurosurgery 2008, 62, 493–504. [Google Scholar] [CrossRef] [PubMed]
- Tsuji, A.; Tamai, I. Carrier-mediated or specialized transport of drugs across the blood–brain barrier. Adv. Drug Deliv. Rev. 1999, 36, 277–290. [Google Scholar] [CrossRef]
- Pardridge, W.M. Transport of small molecules through the blood-brain barrier: Biology and methodology. Adv. Drug Deliv. Rev. 1995, 15, 5–36. [Google Scholar] [CrossRef]
- Takanaga, H.; Tamai, I.; Inaba, S.; Sai, Y.; Higashida, H.; Yamamoto, H.; Tsuji, A. cDNA Cloning and Functional Characterization of Rat Intestinal Monocarboxylate Transporter. Biochem. Biophys. Res. Commun. 1995, 217, 370–377. [Google Scholar] [CrossRef]
- Gerhart, D.Z.; Enerson, B.E.; Zhdankina, O.Y.; Leino, R.L.; Drewes, L.R. Expression of monocarboxylate transporter MCT1 by brain endothelium and glia in adult and suckling rats. Am. J. Physiol.-Endocrinol. Metab. 1997, 273, E207–E213. [Google Scholar] [CrossRef]
- Ceña, V.; Játiva, P. Nanoparticle crossing of blood–brain barrier: A road to new therapeutic approaches to central nervous system diseases. Nanomedicine 2018, 13, 1513–1516. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lesniak, A.; Salvati, A.; Santos-Martinez, M.J.; Radomski, M.W.; Dawson, K.A.; Åberg, C. Nanoparticle Adhesion to the Cell Membrane and Its Effect on Nanoparticle Uptake Efficiency. J. Am. Chem. Soc. 2013, 135, 1438–1444. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hervé, F.; Ghinea, N.; Scherrmann, J.-M. CNS Delivery Via Adsorptive Transcytosis. AAPS J. 2008, 10, 455–472. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Patel, M.M.; Patel, B.M. Crossing the Blood–Brain Barrier: Recent Advances in Drug Delivery to the Brain. CNS Drugs 2017, 31, 109–133. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.W.; Gumbleton, M. Endocytosis at the blood–brain barrier: From basic understanding to drug delivery strategies. J. Drug Target. 2006, 14, 191–214. [Google Scholar] [CrossRef] [PubMed]
- Monsalve, Y.; Tosi, G.; Ruozi, B.; Belletti, D.; Vilella, A.; Zoli, M.; Vandelli, M.A.; Forni, F.; López, B.L.; Sierra, L. PEG-g-chitosan nanoparticles functionalized with the monoclonal antibody OX26 for brain drug targeting. Nanomedicine 2015, 10, 1735–1750. [Google Scholar] [CrossRef] [PubMed]
- Kim, J.-Y.; Choi, W.I.; Kim, Y.H.; Tae, G. Brain-targeted delivery of protein using chitosan- and RVG peptide-conjugated, pluronic-based nano-carrier. Biomaterials 2013, 34, 1170–1178. [Google Scholar] [CrossRef] [PubMed]
- Lu, W.; Wan, J.; Zhang, Q.; She, Z.; Jiang, X. Aclarubicin-loaded cationic albumin-conjugated pegylated nanoparticle for glioma chemotherapy in rats. Int. J. Cancer 2007, 120, 420–431. [Google Scholar] [CrossRef] [PubMed]
- Muniswamy, V.J.; Raval, N.; Gondaliya, P.; Tambe, V.; Kalia, K.; Tekade, R.K. ‘Dendrimer-Cationized-Albumin’ encrusted polymeric nanoparticle improves BBB penetration and anticancer activity of doxorubicin. Int. J. Pharm. 2019, 555, 77–99. [Google Scholar] [CrossRef]
- Agarwal, A.; Majumder, S.; Agrawal, H.; Majumdar, S.; Agrawal, G.P. Cationized Albumin Conjugated Solid Lipid Nanoparticles as Vectors for Brain Delivery of an Anti-Cancer Drug. Curr. Nanosci. 2011, 7, 71–80. [Google Scholar] [CrossRef]
- Agarwal, A.; Agrawal, H.; Tiwari, S.; Jain, S.; Agrawal, G.P. Cationic ligand appended nanoconstructs: A prospective strategy for brain targeting. Int. J. Pharm. 2011, 421, 189–201. [Google Scholar] [CrossRef]
- Janaszewska, A.; Ziemba, B.; Ciepluch, K.; Appelhans, D.; Voit, B.; Klajnert, B.; Bryszewska, M. The biodistribution of maltotriose modified poly(propylene imine) (PPI) dendrimers conjugated with fluorescein—Proofs of crossing blood–brain–barrier. New J. Chem. 2012, 36, 350–353. [Google Scholar] [CrossRef]
- Dombu, C.Y.; Kroubi, M.; Zibouche, R.; Matran, R.; Betbeder, D. Characterization of endocytosis and exocytosis of cationic nanoparticles in airway epithelium cells. Nanotechnology 2010, 21, 355102. [Google Scholar] [CrossRef] [PubMed]
- Kamalinia, G.; Khodagholi, F.; Shaerzadeh, F.; Tavssolian, F.; Chaharband, F.; Atyabi, F.; Sharifzadeh, M.; Amini, M.; Dinarvand, R. Cationic Albumin-Conjugated Chelating Agent as a Novel Brain Drug Delivery System in Neurodegeneration. Chem. Biol. Drug Des. 2015, 86, 1203–1214. [Google Scholar] [CrossRef] [PubMed]
- Re, F.; Cambianica, I.; Zona, C.; Sesana, S.; Gregori, M.; Rigolio, R.; La Ferla, B.; Nicotra, F.; Forloni, G.; Cagnotto, A.; et al. Functionalization of liposomes with ApoE-derived peptides at different density affects cellular uptake and drug transport across a blood-brain barrier model. Nanomed. Nanotechnol. Biol. Med. 2011, 7, 551–559. [Google Scholar] [CrossRef]
- Ulbrich, K.; Hekmatara, T.; Herbert, E.; Kreuter, J. Transferrin- and transferrin-receptor-antibody-modified nanoparticles enable drug delivery across the blood–brain barrier (BBB). Eur. J. Pharm. Biopharm. 2009, 71, 251–256. [Google Scholar] [CrossRef]
- Visser, C.C.; Stevanović, S.; van Bloois, L.; Gaillard, P.J.; Danhof, M.; Crommelin, D.J.; de Boer, A.G. Targeting liposomes with protein drugs to the blood–brain barrier in vitro. Eur. J. Pharm. Sci. 2005, 25, 299–305. [Google Scholar] [CrossRef]
- Chang, J.; Jallouli, Y.; Kroubi, M.; Yuan, X.-B.; Feng, W.; Kang, C.-S.; Pu, P.-Y.; Betbeder, D. Characterization of endocytosis of transferrin-coated PLGA nanoparticles by the blood–brain barrier. Int. J. Pharm. 2009, 379, 285–292. [Google Scholar] [CrossRef]
- Fornaguera, C.; Dols-Perez, A.; Calderó, G.; García-Celma, M.J.; Camarasa, J.; Solans, C. PLGA nanoparticles prepared by nano-emulsion templating using low-energy methods as efficient nanocarriers for drug delivery across the blood–brain barrier. J. Control. Release 2015, 211, 134–143. [Google Scholar] [CrossRef] [PubMed]
- Qiao, R.; Jia, Q.; Hüwel, S.; Xia, R.; Liu, T.; Gao, F.; Galla, H.-J.; Gao, M. Receptor-Mediated Delivery of Magnetic Nanoparticles across the Blood–Brain Barrier. ACS Nano 2012, 6, 3304–3310. [Google Scholar] [CrossRef]
- You, Y.; Yang, L.; He, L.; Chen, T. Tailored mesoporous silica nanosystem with enhanced permeability of the blood–brain barrier to antagonize glioblastoma. J. Mater. Chem. B 2016, 4, 5980–5990. [Google Scholar] [CrossRef] [PubMed]
- Boado, R.J.; Zhang, Y.; Zhang, Y.; Wang, Y.; Pardridge, W.M. GDNF fusion protein for targeted-drug delivery across the human blood–brain barrier. Biotechnol. Bioeng. 2008, 100, 387–396. [Google Scholar] [CrossRef]
- Ulbrich, K.; Knobloch, T.; Kreuter, J. Targeting the insulin receptor: Nanoparticles for drug delivery across the blood–brain barrier (BBB). J. Drug Target. 2011, 19, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Gan, C.W.; Feng, S.-S. Transferrin-conjugated nanoparticles of Poly(lactide)-d-α-Tocopheryl polyethylene glycol succinate diblock copolymer for targeted drug delivery across the blood–brain barrier. Biomaterials 2010, 31, 7748–7757. [Google Scholar] [CrossRef] [PubMed]
- Lin, T.; Zhao, P.; Jiang, Y.; Tang, Y.; Jin, H.; Pan, Z.; He, H.; Yang, V.C.; Huang, Y. Blood–Brain-Barrier-Penetrating Albumin Nanoparticles for Biomimetic Drug Delivery via Albumin-Binding Protein Pathways for Antiglioma Therapy. ACS Nano 2016, 10, 9999–10012. [Google Scholar] [CrossRef] [PubMed]
- Miura, Y.; Takenaka, T.; Toh, K.; Wu, S.; Nishihara, H.; Kano, M.R.; Ino, Y.; Nomoto, T.; Matsumoto, Y.; Koyama, H.; et al. Cyclic RGD-Linked Polymeric Micelles for Targeted Delivery of Platinum Anticancer Drugs to Glioblastoma through the Blood–Brain Tumor Barrier. ACS Nano 2013, 7, 8583–8592. [Google Scholar] [CrossRef] [PubMed]
- Pardridge, W.M. shRNA and siRNA delivery to the brain. Adv. Drug Deliv. Rev. 2007, 59, 141–152. [Google Scholar] [CrossRef] [Green Version]
- Li, H.; Qian, Z.M. Transferrin/transferrin receptor-mediated drug delivery. Med. Res. Rev. 2002, 22, 225–250. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.H.; Riaz, M.K.; Xie, Y.; Zhang, X.; Liu, Q.; Chen, H.; Bian, Z.; Chen, X.; Lu, A.; Yang, Z. Review of Current Strategies for Delivering Alzheimer’s Disease Drugs across the Blood-Brain Barrier. Int. J. Mol. Sci. 2019, 20, 381. [Google Scholar] [CrossRef] [Green Version]
- Zhang, Y.; Lu, C.; Zhang, J. Lactoferrin and Its Detection Methods: A Review. Nutrients 2021, 13, 2492. [Google Scholar] [CrossRef]
- Pardridge, W.M.; Eisenberg, J.; Yang, J. Human Blood—Brain Barrier Insulin Receptor. J. Neurochem. 1985, 44, 1771–1778. [Google Scholar] [CrossRef]
- Lee, C.; Hwang, H.S.; Lee, S.; Kim, B.; Kim, J.O.; Oh, K.T.; Lee, E.S.; Choi, H.-G.; Youn, Y.S. Rabies Virus-Inspired Silica-Coated Gold Nanorods as a Photothermal Therapeutic Platform for Treating Brain Tumors. Adv. Mater. 2017, 29, 1605563. [Google Scholar] [CrossRef]
- Raymond, A.D.; Diaz, P.; Chevelon, S.; Agudelo, M.; Yndart-Arias, A.; Ding, H.; Kaushik, A.; Jayant, R.D.; Nikkhah-Moshaie, R.; Roy, U.; et al. Microglia-derived HIV Nef+ exosome impairment of the blood–brain barrier is treatable by nanomedicine-based delivery of Nef peptides. J. Neurovirol. 2016, 22, 129–139. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.-W.; Wang, H.-P.; Yuan, F.-M.; Zhang, X.; Dong, X.-J.; Xie, R.-S.; Tian, C.; Li, B.-S.; Sun, Z.-W.; Zhou, L.-H.; et al. Releasing of Herpes Simplex Virus Carrying NGF in Subarachnoid Space Promotes the Functional Repair in Spinal Cord Injured Rats. Curr. Gene Ther. 2016, 16, 263–270. [Google Scholar] [CrossRef] [PubMed]
- Miki, Y.; Nonoguchi, N.; Ikeda, N.; Coffin, R.S.; Kuroiwa, T.; Miyatake, S.-I. Vascular endothelial growth factor gene-transferred bone marrow stromal cells engineered with a herpes simplex virus type 1 vector can improve neurological deficits and reduce infarction volume in rat brain ischemia. Neurosurgery 2007, 61, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Mancuso, R.; Baglio, F.; Cabinio, M.; Calabrese, E.; Hernis, A.; Nemni, R.; Clerici, M. Titers of Herpes Simplex Virus Type 1 Antibodies Positively Correlate with Grey Matter Volumes in Alzheimer’s Disease. J. Alzheimer’s Dis. 2013, 38, 741–745. [Google Scholar] [CrossRef] [PubMed]
- Anand, P.; O’Neil, A.; Lin, E.; Douglas, T.; Holford, M. Tailored delivery of analgesic ziconotide across a blood brain barrier model using viral nanocontainers. Sci. Rep. 2015, 5, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, Y.; Liu, L. Modern methods for delivery of drugs across the blood–brain barrier. Adv. Drug Deliv. Rev. 2012, 64, 640–665. [Google Scholar] [CrossRef]
- Jones, A.R.; Shusta, E.V. Blood–Brain Barrier Transport of Therapeutics via Receptor-Mediation. Pharm. Res. 2007, 24, 1759–1771. [Google Scholar] [CrossRef] [Green Version]
- Bartanusz, V.; Jezova, D.; Alajajian, B.; Digicaylioglu, M. The blood-spinal cord barrier: Morphology and Clinical Implications. Ann. Neurol. 2011, 70, 194–206. [Google Scholar] [CrossRef]
- Claxton, A.; Baker, L.D.; Wilkinson, C.W.; Trittschuh, E.H.; Chapman, D.; Watson, G.S.; Cholerton, B.; Plymate, S.R.; Arbuckle, M.; Craft, S. Sex and ApoE Genotype Differences in Treatment Response to Two Doses of Intranasal Insulin in Adults with Mild Cognitive Impairment or Alzheimer’s Disease. J. Alzheimer’s Dis. 2013, 35, 789–797. [Google Scholar] [CrossRef] [Green Version]
- Sonvico, F.; Clementino, A.; Buttini, F.; Colombo, G.; Pescina, S.; Stanisçuaski Guterres, S.; Raffin Pohlmann, A.; Nicoli, S. Surface-Modified Nanocarriers for Nose-to-Brain Delivery: From Bioadhesion to Targeting. Pharmaceutics 2018, 10, 34. [Google Scholar] [CrossRef] [Green Version]
- Ying, W. The nose may help the brain: Intranasal drug delivery for treating neurological diseases. Future Neurol. 2008, 3, 1–4. [Google Scholar] [CrossRef] [Green Version]
- Seju, U.; Kumar, A.; Sawant, K.K. Development and evaluation of olanzapine-loaded PLGA nanoparticles for nose-to-brain delivery: In vitro and in vivo studies. Acta Biomater. 2011, 7, 4169–4176. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Q.; Jiang, X.; Jiang, W.; Lu, W.; Su, L.; Shi, Z. Preparation of nimodipine-loaded microemulsion for intranasal delivery and evaluation on the targeting efficiency to the brain. Int. J. Pharm. 2004, 275, 85–96. [Google Scholar] [CrossRef] [PubMed]
- Kozlovskaya, L.; Abou-Kaoud, M.; Stepensky, D. Quantitative analysis of drug delivery to the brain via nasal route. J. Control. Release 2014, 189, 133–140. [Google Scholar] [CrossRef] [PubMed]
- Costantino, L.; Boraschi, D. Is there a clinical future for polymeric nanoparticles as brain-targeting drug delivery agents? Drug Discov. Today 2012, 17, 367–378. [Google Scholar] [CrossRef]


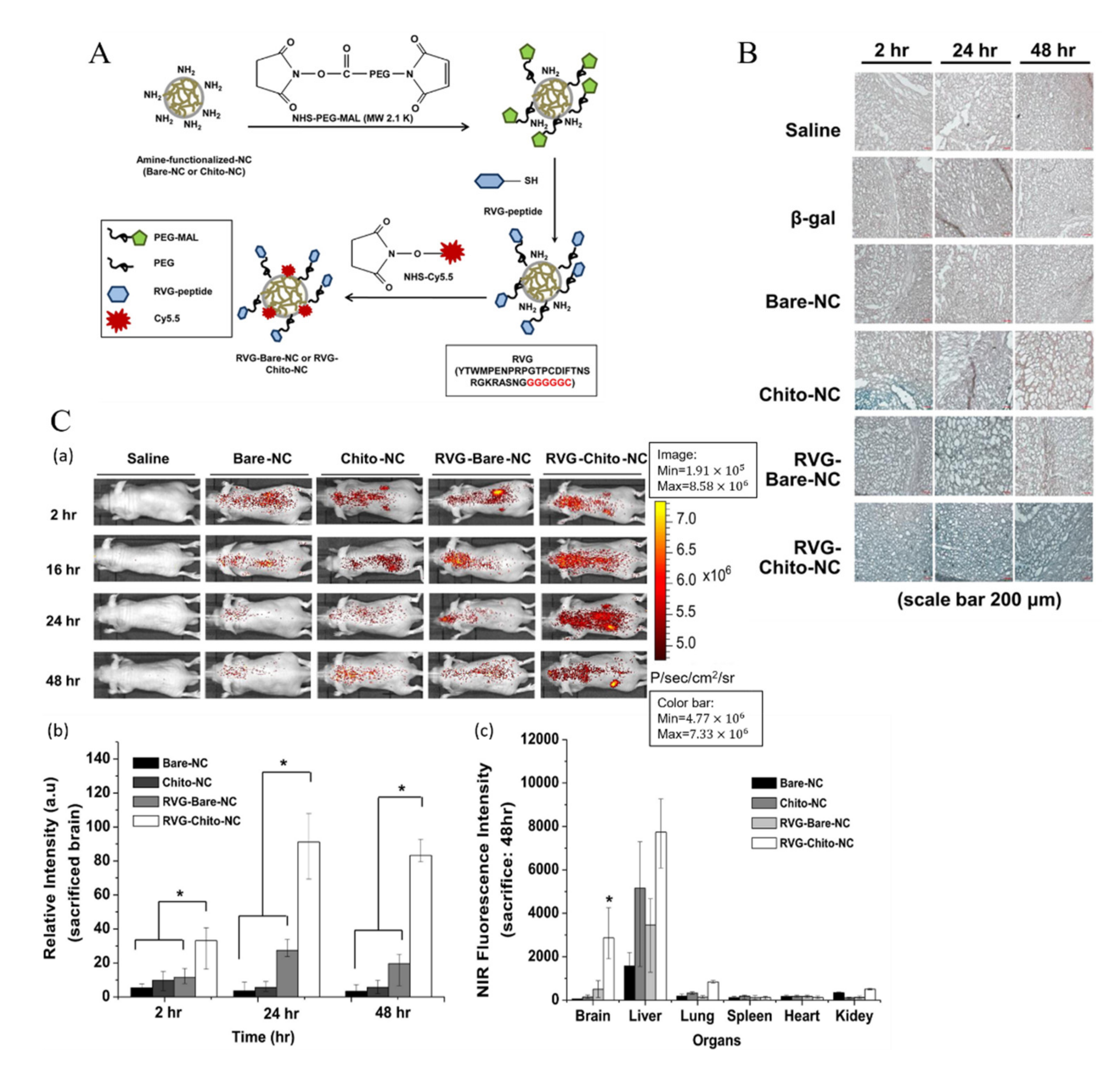
| Virus | Effects on CNS | |
|---|---|---|
| Flaviviridae | Hepatitis C virus (HCV) | Human brain endothelial cells express functional receptors that support HCV entry and replication; HCV infection promotes endothelial permeability and cellular apoptosis [19]. |
| West Nile virus (WNV) | Increase activity and mRNA expression of matrix metalloproteinases (MMP) 9 in mouse brains; a Trojan horse mechanism [20]. | |
| Japanese encephalitis virus (JEV) | Increase MMP9 expression in a reactive oxygen species (ROS)-dependent manner [21]. | |
| Dengu Virus | Mediated via the release of histamine by a virus-induced cytokine. | |
| Zika Virus | Downregulation of occludin and claudin-5 levels [22]. A cell-type-specific paracellular pathway to cross the placenta monolayer [23]. | |
| coronavirus | SARS-CoV-2 | Spike protein S1 binding to ACE2 [24,25,26]. Much higher affinity [27]. |
| HCoV-OC43 | Neuronal retrograded (olfactory bulb) [28] and hematogenous pathway [29]; May have neuronal degeneration [28]. | |
| HCoV-229E | Invasion via the circulation of bloodstream [28]. Neuro-invasive under immune-suppressed environment [30]. | |
| SARS-CoV | ACE2 receptor. Both hematogenous route [31] and olfactory bulb [32]; | |
| Other viruses | HSV | Bloodstream and neuronal route [33]; Up-regulate MMP2 and MMP9 and disrupt BBB [34]; |
| Rabies virus | Rabies virus glycoprotein as brain-targeted ligand andthe nicotinic acetylcholine on neuronal cells as receptor [35]; | |
| MAV-1 | Stimulate an innate host response to induce BBB disruption [36]; Possible invasion by a Trojan horse mechanism via monocytes [37,38]; | |
| Theiler’s murine encephalomyelitis virus | Induce acute encephalitis with alterations in tight junction protein expression [39]. |
| The Compounds on the Carriers’ Surface | Drug Carrier | Transport Pathway | Effects on BBB and Brain | |
|---|---|---|---|---|
| Hexose derivatives | 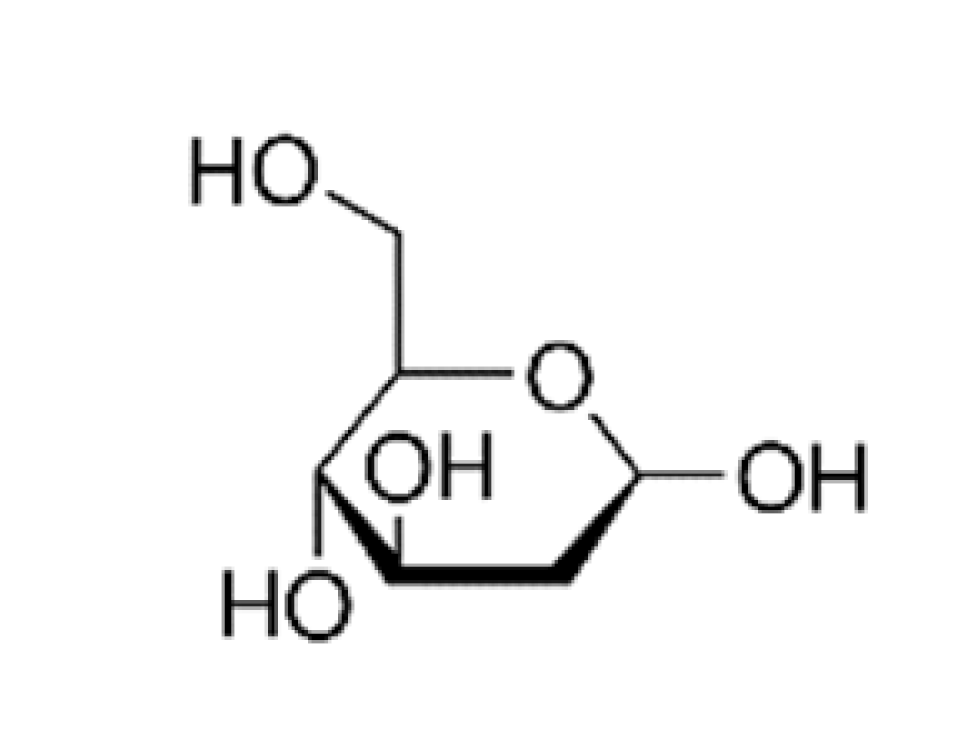 | Poly(ethylene glycol)-co-poly(trimethylene carbonate) nanoparticles | GLUT1 [75] | Higher internalization amount by glioma cells. Successful penetration of the BBB. Showing specific and efficient accumulation in intracranial tumor. |
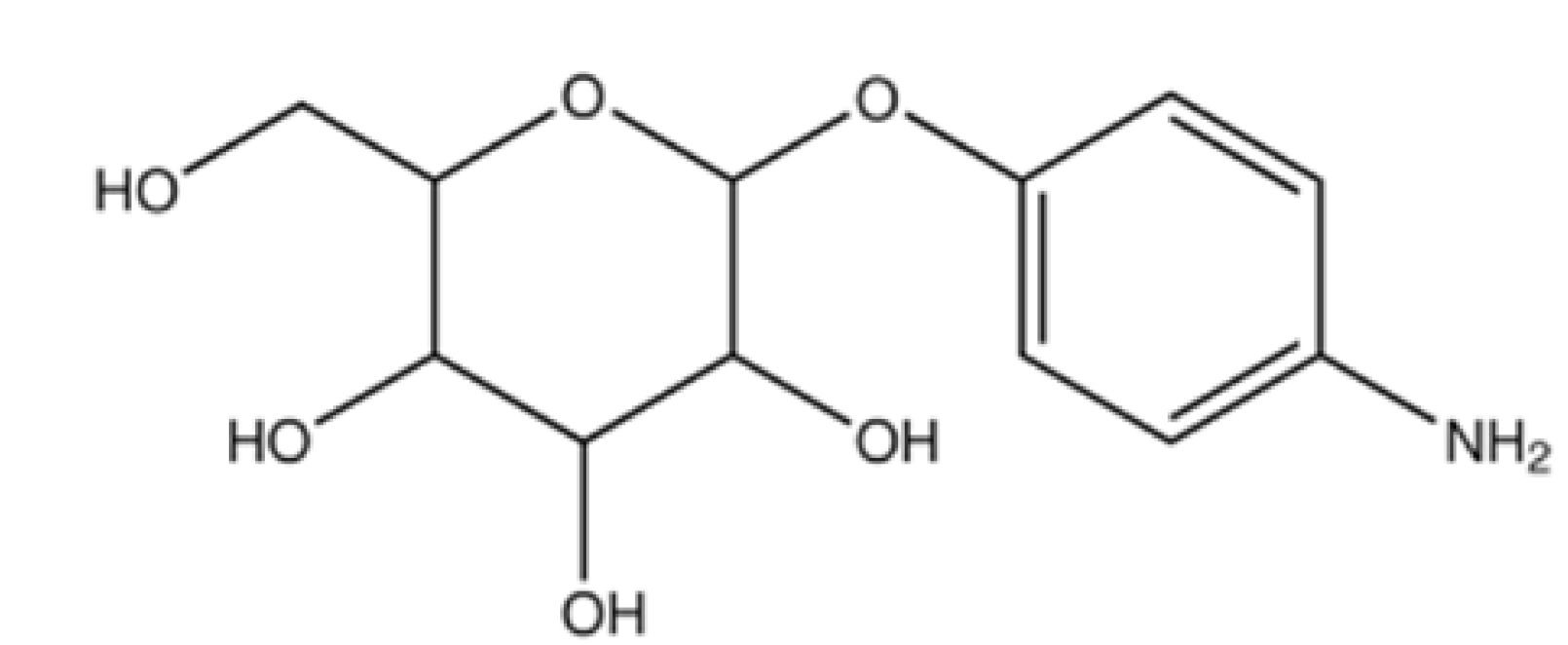 | Liposomes | GLUT1 and GLUT3 [76,77,78] | Enhanced cellular uptake and accumulation in the brain. Stronger transendothelial ability. | |
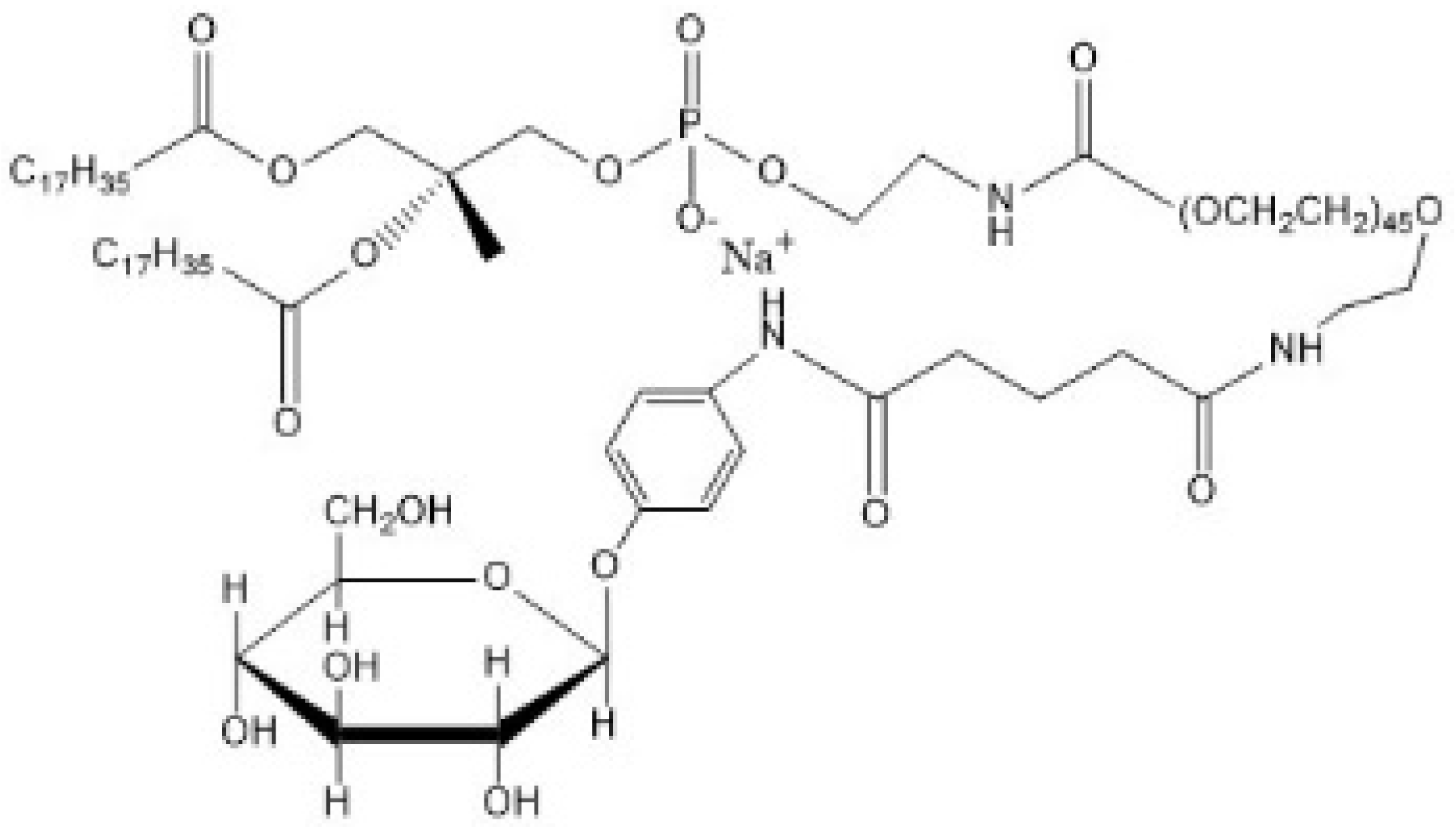 | GLUT1 [79] | Long circulation in blood. Less leakage in the blood component-containing system. Efficacies in killing glioblastoma cells. | ||
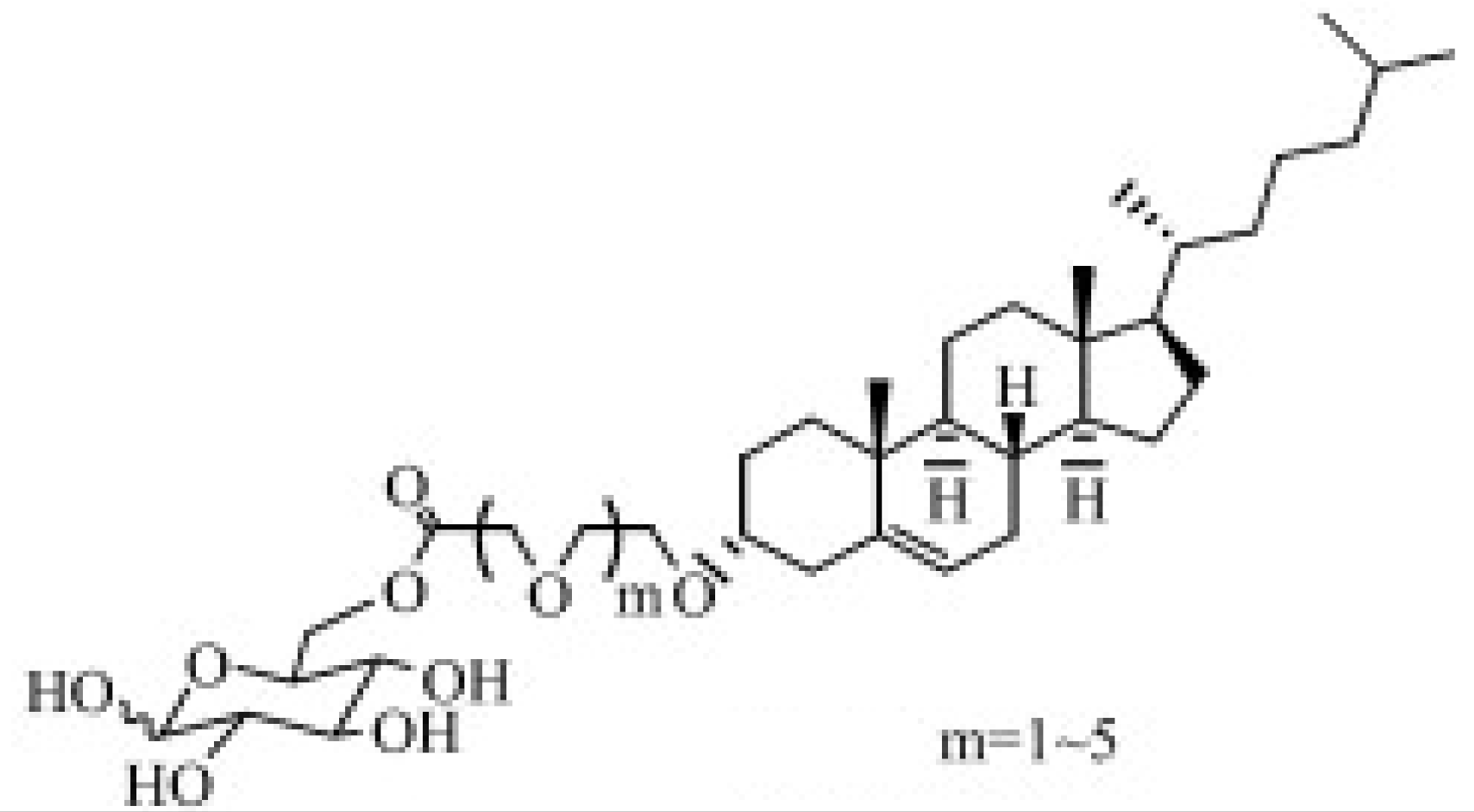 | GLUT [80,81,82] | The potential of brain targeting. Molecules with moderate chain length exhibiting the strongest brain delivery capacity. | ||
| Amino acid derivatives |  | Pluronic F127 copolymer nanoparticles | LAT1 [83] | Successful drug delivery to the hippocampus in the brain. Increased tryptophan uptake at epileptogenic focus. |
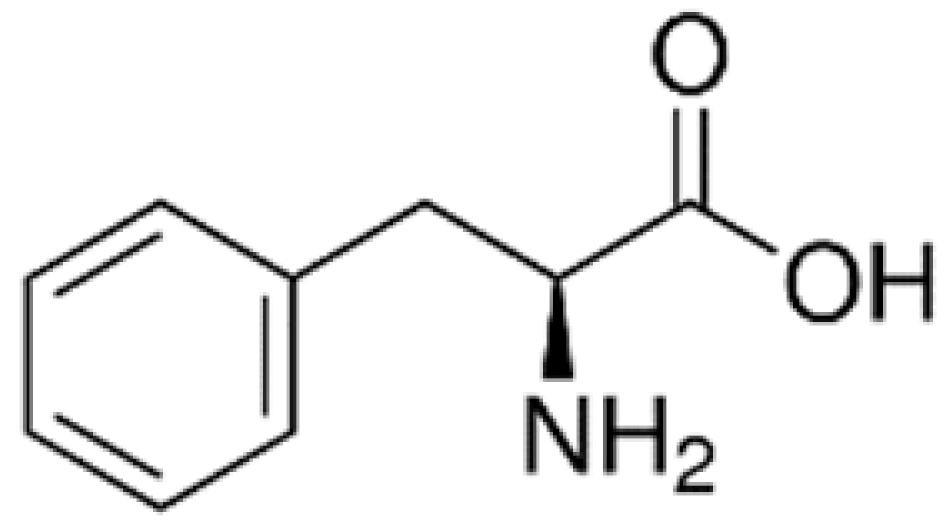 | Solid lipid nanoparticles (SLNs) | LAT1 [84] | Higher accumulation in the brain. | |
| Peptide | 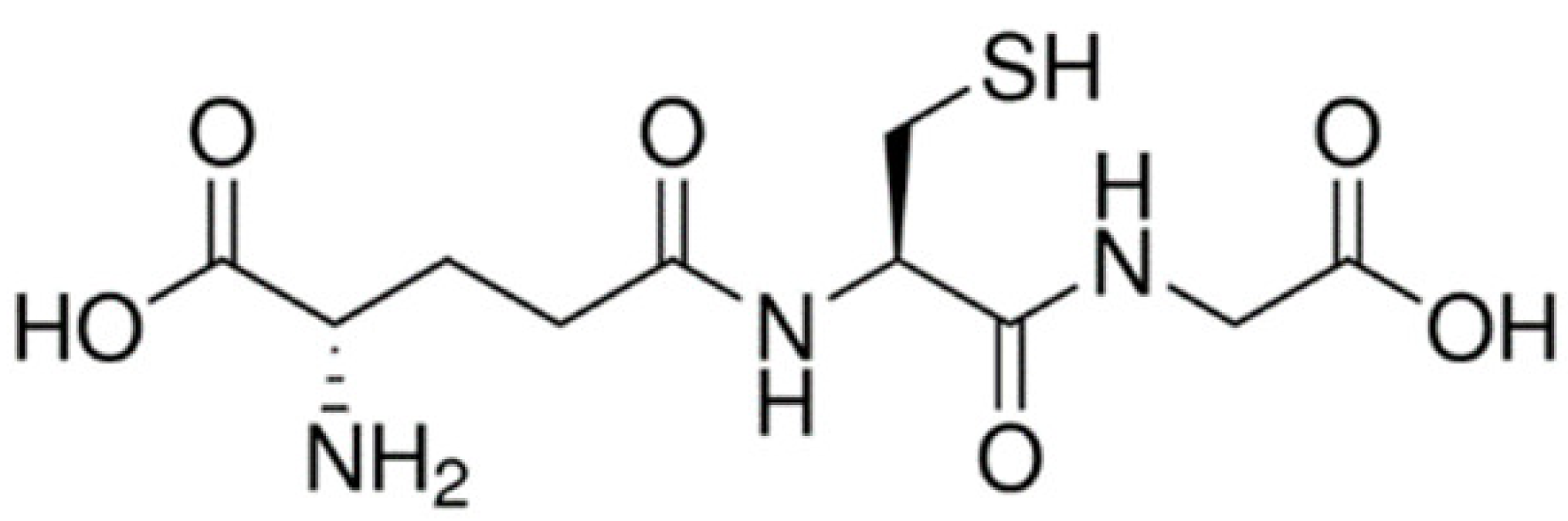 | Poly-(lactide-co-glycolide) (PLGA) nanoparticles | Glutathione transporter [71,72] | Higher BBB permeation and brain uptake. Not substrates of P-glycoprotein (P-gp) and not being effluxed by P-gp. |
| Poly(ethylene glycol)ylated PLGA | [85] | |||
| Monocarboxylic acid | 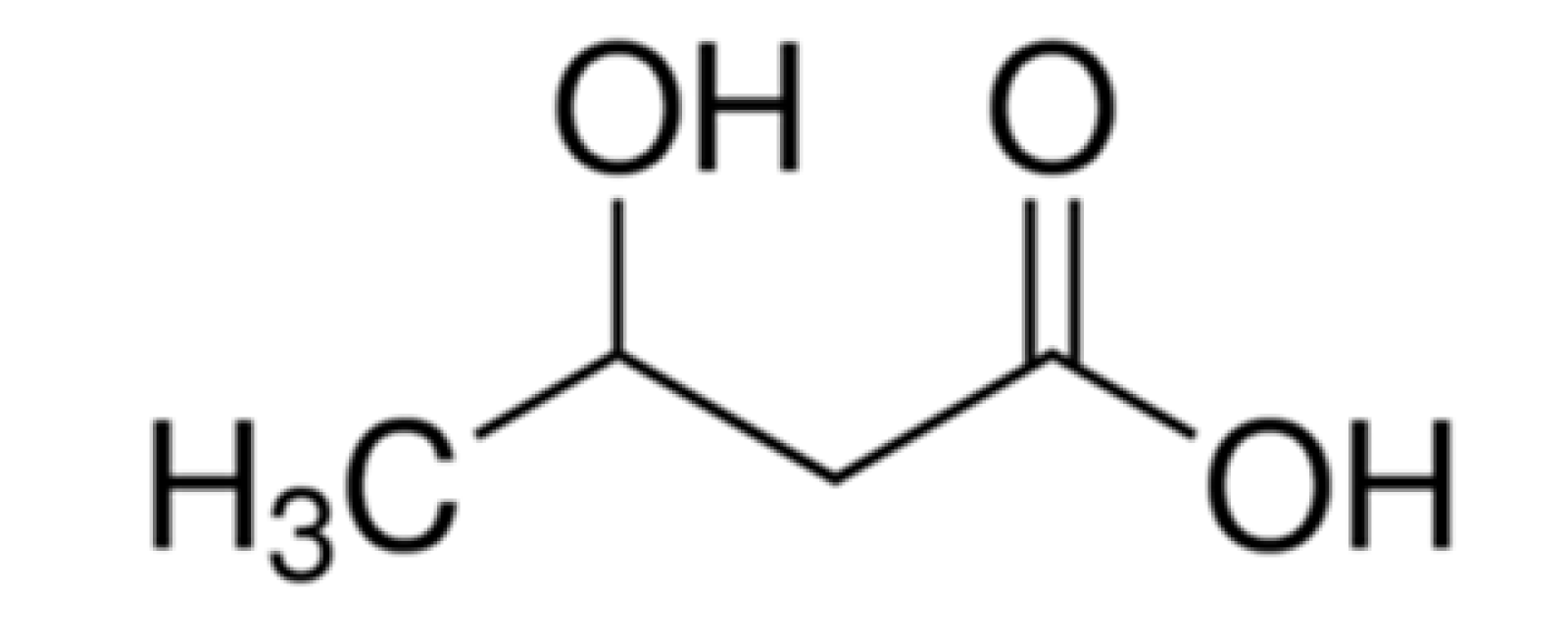 | SLNs | Monocarboxylic acid transport system (MCT) 1 [86] | Improved brain uptake. |
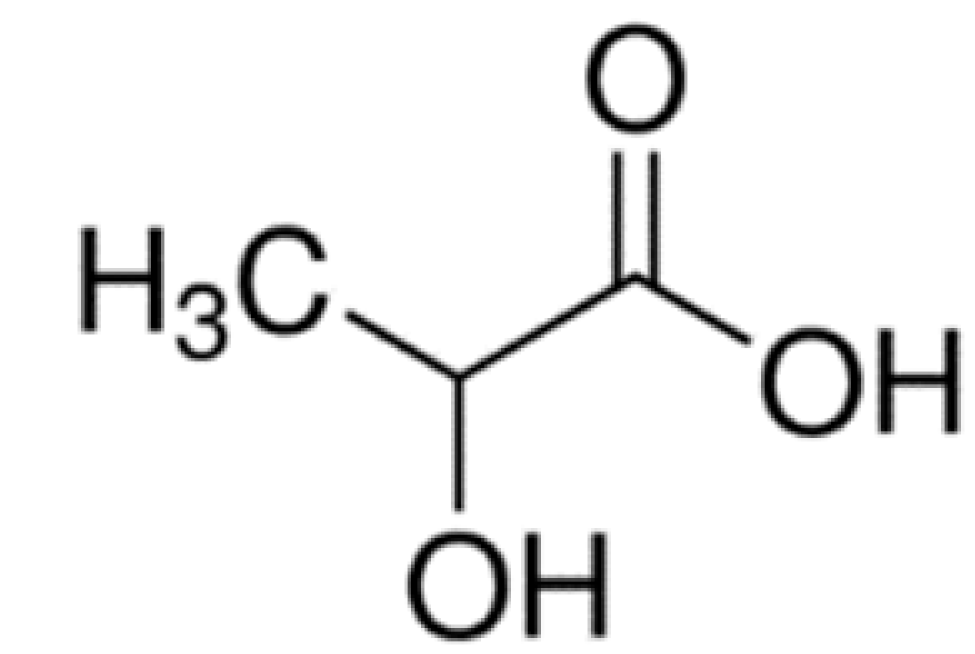 | MCT [87] | Selective brain uptake. | ||
| Drug Carrier | Modification Methods | Zeta Potential |
|---|---|---|
| poly(propylene imine) (PPI) [111] | Oligosaccharide-modified | Positively charged |
| Poly(ethylenimine) (PEI) [35] | Conjugating with rabies virus glycoprotein and PEG | Relatively neutral |
| Maltodextrin nanoparticles [112] | \ | 25 ± 1.5 |
| PEG-g-chitosan [105] | Transferrin receptor monoclonal antibodies (OX26) | 23.0 ± 0.4 |
| Solid lipid nanoparticles (SLNs) [109,110] | Conjugating bovine serum albumin | 10.3 ± 0.6 |
| Pluronic-based nano-carrier [106] | Chitosan and rabies virus glycoprotein-conjugated | 12.1 ± 0.8 |
| Drug Carrier | Ligand | Receptor |
|---|---|---|
| Human serum albumin nanoparticles [115] | Transferrin (Tf)/ transferrin receptor monoclonal antibodies (OX26 or R17217) | Transferrin receptor |
| Pegylated liposome [116] | Transferrin (Tf) | |
| Poly-(lactide-co-glycolide) (PLGA) [117] | ||
| PLGA [118] | Mouse monoclonal antibody against the transferrin receptor (8D3) | |
| Polyethylene glycol (PEG)-coated Fe3O4 [119] | Lactoferrin (Lf) | Lf receptor |
| Nanoliposomes [114] | Apolipoprotein E (ApoE)-derived peptides | Low-density lipoprotein receptor |
| Pluronic-based nano-carrier [106] | Rabies virus glycoprotein | g-aminobutyric acid (GABA) /nicotinic acetylcholine (nACh) receptor |
| Poly(ethylenimine) [35] | ||
| Porous silica nanoparticle [120] | RGD (arginine–glycine–aspartate) peptide | αvβ3 and αvβ5 integrins |
| Glial-derived neurotrophic factor [121] | Chimeric monoclonal antibody | Insulin receptor |
| Human serum albumin (HSA) nanoparticles [122] | Anti-insulin receptor monoclonal antibody (29B4) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Song, J.; Lu, C.; Leszek, J.; Zhang, J. Design and Development of Nanomaterial-Based Drug Carriers to Overcome the Blood–Brain Barrier by Using Different Transport Mechanisms. Int. J. Mol. Sci. 2021, 22, 10118. https://doi.org/10.3390/ijms221810118
Song J, Lu C, Leszek J, Zhang J. Design and Development of Nanomaterial-Based Drug Carriers to Overcome the Blood–Brain Barrier by Using Different Transport Mechanisms. International Journal of Molecular Sciences. 2021; 22(18):10118. https://doi.org/10.3390/ijms221810118
Chicago/Turabian StyleSong, Jisu, Chao Lu, Jerzy Leszek, and Jin Zhang. 2021. "Design and Development of Nanomaterial-Based Drug Carriers to Overcome the Blood–Brain Barrier by Using Different Transport Mechanisms" International Journal of Molecular Sciences 22, no. 18: 10118. https://doi.org/10.3390/ijms221810118
APA StyleSong, J., Lu, C., Leszek, J., & Zhang, J. (2021). Design and Development of Nanomaterial-Based Drug Carriers to Overcome the Blood–Brain Barrier by Using Different Transport Mechanisms. International Journal of Molecular Sciences, 22(18), 10118. https://doi.org/10.3390/ijms221810118







