Lipid Self-Assemblies under the Atomic Force Microscope †
Abstract
1. Introduction: Membranes
2. Lipids: Building Blocks and More
3. Membrane Biophysics: Model Membranes
4. Membrane Biophysics: Techniques
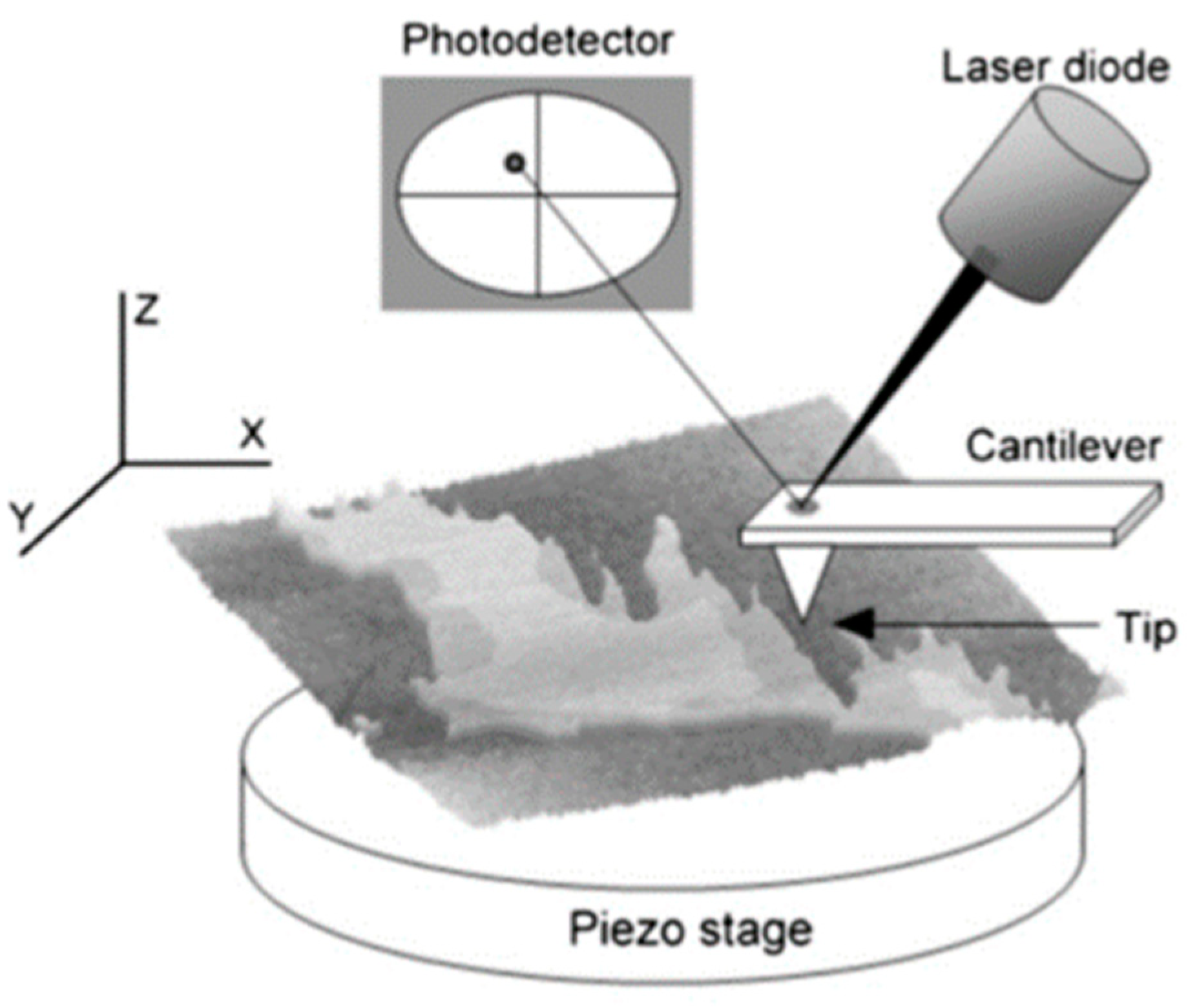
5. Membranes under AFM: The Basics
6. Membranes under the AFM: Findings
7. Impact on Membrane Biophysics ‘Hot Topics’
8. Conclusions and Perspectives
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Overton, C.E. Über die osmotischen Eigenschaften der lebenden Pflanzen-und Tierzelle; Fäsi & Beer: Zurich, Switzerland, 1895. [Google Scholar]
- Singer, S.J.; Nicolson, G.L. The fluid mosaic model of the structure of cell membranes. Science 1972, 175, 720–731. [Google Scholar] [CrossRef]
- Goñi, F.M. The basic structure and dynamics of cell membranes: An update of the Singer—Nicolson model. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2014, 1838, 1467–1476. [Google Scholar] [CrossRef] [PubMed]
- Chapman, D.; Gomez-Fernandez, J.C.; Goni, F.M. Intrinsic protein—Lipid interactions: Physical and biochemical evidence. FEBS Lett. 1979, 98, 211–223. [Google Scholar] [CrossRef]
- Engelman, D.M. Membranes are more mosaic than fluid. Nature 2005, 438, 578–580. [Google Scholar] [CrossRef]
- Bagatolli, L.A.; Mouritsen, O.G. Is the fluid mosaic (and the accompanying raft hypothesis) a suitable model to describe fundamental features of biological membranes? What may be missing? Front. Plant Sci. 2013, 4, 457. [Google Scholar] [CrossRef] [PubMed]
- Van Meer, G. Dynamic transbilayer lipid asymmetry. Cold Spring Harb. Perspect. Biol. 2011, 3, a004671. [Google Scholar] [CrossRef]
- Goñi, F.M.; Alonso, A.; Contreras, F.X. Membrane Nanodomains. In eLS; John Wiley & Sons, Ltd.: Chichester, UK, 2020. [Google Scholar]
- Simons, K.; Ikonen, E. Functional rafts in cell membranes. Nature 1997, 387, 569–572. [Google Scholar] [CrossRef]
- Shimshick, E.J.; McConnell, H.M. Lateral phase separations in binary mixtures of cholesterol and phospholipids. Biochem. Biophys. Res. Commun. 1973, 53, 446–451. [Google Scholar] [CrossRef]
- Chapman, D.; Collin, D.T. Differential thermal analysis of phospholipids. Nature 1965, 206, 189. [Google Scholar] [CrossRef]
- Epand, R.M. Lipid polymorphism and protein-lipid interactions. Biochim. Et Biophys. Acta (BBA)-Rev. Biomembr. 1998, 1376, 353–368. [Google Scholar] [CrossRef]
- Chernomordik, L.V.; Zimmerberg, J.; Kozlov, M.M. Membranes of the world unite! J. Cell Biol. 2006, 175, 201–207. [Google Scholar] [CrossRef]
- Yang, Y.; Lee, M.; Fairn, G.D. Phospholipid subcellular localization and dynamics. J. Biol. Chem. 2018, 293, 6230–6240. [Google Scholar] [CrossRef]
- Thudichum, J.L.W. A Treatise on the Chemical Constitution of the Brain: Based Throughout Upon Original Researches; Baillière, Tindall, and Cox: London, UK, 1884. [Google Scholar]
- Merrill, A.; Sereni, A.; Stevens, V.; Hannun, Y.; Bell, R.; Kinkade, J. Inhibition of phorbol ester-dependent differentiation of human promyelocytic leukemic (HL-60) cells by sphinganine and other long-chain bases. J. Biol. Chem. 1986, 261, 12610–12615. [Google Scholar] [CrossRef]
- Hannun, Y.A.; Loomis, C.R.; Merrill, A.H., Jr.; Bell, R.M. Sphingosine inhibition of protein kinase C activity and of phorbol dibutyrate binding in vitro and in human platelets. J. Biol. Chem. 1986, 261, 12604–12609. [Google Scholar] [CrossRef]
- Castro, B.M.; Prieto, M.; Silva, L.C. Ceramide: A simple sphingolipid with unique biophysical properties. Prog. Lipid Res. 2014, 54, 53–67. [Google Scholar] [CrossRef]
- Obeid, L.M.; Linardic, C.M.; Karolak, L.A.; Hannun, Y.A. Programmed cell death induced by ceramide. Science 1993, 259, 1769–1771. [Google Scholar] [CrossRef] [PubMed]
- Summers, S.A.; Chaurasia, B.; Holland, W.L. Metabolic messengers: Ceramides. Nat. Metab. 2019, 1, 1051–1058. [Google Scholar] [CrossRef] [PubMed]
- Barth, B.M.; Cabot, M.C.; Kester, M. Ceramide-based therapeutics for the treatment of cancer. Anti-Cancer Agents Med. Chem. 2011, 11, 911–919. [Google Scholar] [CrossRef] [PubMed]
- Shaw, J.; Costa-Pinheiro, P.; Patterson, L.; Drews, K.; Spiegel, S.; Kester, M. Novel sphingolipid-based cancer therapeutics in the personalized medicine era. In Advances in Cancer Research; Elsevier: Amsterdam, The Netherlands, 2018; Volume 140, pp. 327–366. [Google Scholar]
- Gómez-Muñoz, A. Ceramide-1-phosphate: A novel regulator of cell activation. FEBS Lett. 2004, 562, 5–10. [Google Scholar] [CrossRef]
- Gómez-Muñoz, A.; Gangoiti, P.; Arana, L.; Ouro, A.; Rivera, I.G.; Ordoñez, M.; Trueba, M. New insights on the role of ceramide 1-phosphate in inflammation. Biochim. Et Biophys. Acta (BBA)-Mol. Cell Biol. Lipids 2013, 1831, 1060–1066. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Boppart, S.A. Dynamic Signatures of Lipid Droplets as New Markers to Quantify Cellular Metabolic Changes. Anal. Chem. 2020, 92, 15943–15952. [Google Scholar] [CrossRef]
- Cloherty, A.P.; Olmstead, A.D.; Ribeiro, C.; Jean, F. Hijacking of Lipid Droplets by Hepatitis C, Dengue and Zika Viruses—From Viral Protein Moonlighting to Extracellular Release. Int. J. Mol. Sci. 2020, 21, 7901. [Google Scholar] [CrossRef] [PubMed]
- Chaurasia, B.; Talbot, C.L.; Summers, S.A. Adipocyte Ceramides—The Nexus of Inflammation and Metabolic Disease. Front. Immunol. 2020, 11, 2282. [Google Scholar] [CrossRef] [PubMed]
- Hannun, Y.A.; Obeid, L.M. Sphingolipids and their metabolism in physiology and disease. Nat. Rev. Mol. Cell Biol. 2018, 19, 175. [Google Scholar] [CrossRef]
- Fidorra, M.; Duelund, L.; Leidy, C.; Simonsen, A.C.; Bagatolli, L.A. Absence of fluid-ordered/fluid-disordered phase coexistence in ceramide/POPC mixtures containing cholesterol. Biophys. J. 2006, 90, 4437–4451. [Google Scholar] [CrossRef] [PubMed]
- García-Arribas, A.B.; Busto, J.V.; Alonso, A.; Goñi, F.M. Atomic force microscopy characterization of palmitoylceramide and cholesterol effects on phospholipid bilayers: A topographic and nanomechanical study. Langmuir 2015, 31, 3135–3145. [Google Scholar] [CrossRef]
- Sot, J.; Bagatolli, L.A.; Goñi, F.M.; Alonso, A. Detergent-resistant, ceramide-enriched domains in sphingomyelin/ceramide bilayers. Biophys. J. 2006, 90, 903–914. [Google Scholar] [CrossRef] [PubMed]
- Fanani, M.L.; Maggio, B. The many faces (and phases) of ceramide and sphingomyelin II—Binary mixtures. Biophys. Rev. 2017, 9, 601–616. [Google Scholar] [CrossRef]
- Al Sazzad, M.A.; Yasuda, T.; Murata, M.; Slotte, J.P. The long-chain sphingoid base of ceramides determines their propensity for lateral segregation. Biophys. J. 2017, 112, 976–983. [Google Scholar] [CrossRef]
- González-Ramírez, E.J.; García-Arribas, A.B.; Sot, J.; Goñi, F.M.; Alonso, A. C24: 0 and C24: 1 sphingolipids in cholesterol-containing, five-and six-component lipid membranes. Sci. Rep. 2020, 10, 14085. [Google Scholar] [CrossRef]
- García-Arribas, A.B.; González-Ramírez, E.J.; Sot, J.; Areso, I.; Alonso, A.; Goñi, F.M. Complex Effects of 24:1 Sphingolipids in Membranes Containing Dioleoylphosphatidylcholine and Cholesterol. Langmuir 2017, 33, 5545–5554. [Google Scholar] [CrossRef]
- Veiga, M.P.; Arrondo, J.L.R.; Goñi, F.M.; Alonso, A. Ceramides in phospholipid membranes: Effects on bilayer stability and transition to nonlamellar phases. Biophys. J. 1999, 76, 342–350. [Google Scholar] [CrossRef]
- Siskind, L.J.; Kolesnick, R.N.; Colombini, M. Ceramide channels increase the permeability of the mitochondrial outer membrane to small proteins. J. Biol. Chem. 2002, 277, 26796–26803. [Google Scholar] [CrossRef] [PubMed]
- Montes, L.R.; Ruiz-Arguello, M.B.; Goñi, F.M.; Alonso, A. Membrane restructuring via ceramide results in enhanced solute efflux. J. Biol. Chem. 2002, 277, 11788–11794. [Google Scholar] [CrossRef]
- Colombini, M. Ceramide channels and mitochondrial outer membrane permeability. J. Bioenerg. Biomembr. 2017, 49, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Artetxe, I.; Ugarte-Uribe, B.; Gil, D.; Valle, M.; Alonso, A.; García-Sáez, A.J.; Goñi, F.M. Does ceramide form channels? The ceramide-induced membrane permeabilization mechanism. Biophys. J. 2017, 113, 860–868. [Google Scholar] [CrossRef]
- Contreras, F.X.; Villar, A.V.; Alonso, A.; Kolesnick, R.N.; Goñi, F.M. Sphingomyelinase activity causes transbilayer lipid translocation in model and cell membranes. J. Biol. Chem. 2003, 278, 37169–37174. [Google Scholar] [CrossRef]
- Ohvo-Rekilä, H.; Ramstedt, B.; Leppimäki, P.; Peter Slotte, J. Cholesterol interactions with phospholipids in membranes. Prog. Lipid Res. 2002, 41, 66–97. [Google Scholar] [CrossRef]
- Bienias, K.; Fiedorowicz, A.; Sadowska, A.; Prokopiuk, S.; Car, H. Regulation of sphingomyelin metabolism. Pharmacol. Rep. 2016, 68, 570–581. [Google Scholar] [CrossRef] [PubMed]
- Megha; London, E. Ceramide selectively displaces cholesterol from ordered lipid domains (rafts): Implications for lipid raft structure and function. J. Biol. Chem. 2004, 279, 9997–10004. [Google Scholar] [CrossRef] [PubMed]
- Silva, L.C.; de Almeida, R.F.; Castro, B.M.; Fedorov, A.; Prieto, M. Ceramide-domain formation and collapse in lipid rafts: Membrane reorganization by an apoptotic lipid. Biophys. J. 2007, 92, 502–516. [Google Scholar] [CrossRef]
- Sot, J.; Ibarguren, M.; Busto, J.V.; Montes, L.; Goñi, F.M.; Alonso, A. Cholesterol displacement by ceramide in sphingomyelin-containing liquid-ordered domains, and generation of gel regions in giant lipidic vesicles. FEBS Lett. 2008, 582, 3230–3236. [Google Scholar] [CrossRef] [PubMed]
- García-Arribas, A.B.; Axpe, E.; Mujika, J.I.; Mérida, D.; Busto, J.V.; Sot, J.; Alonso, A.; Lopez, X.; García, J.Á.; Ugalde, J.M.; et al. Cholesterol–Ceramide Interactions in Phospholipid and Sphingolipid Bilayers As Observed by Positron Annihilation Lifetime Spectroscopy and Molecular Dynamics Simulations. Langmuir 2016, 32, 5434–5444. [Google Scholar] [CrossRef] [PubMed]
- Busto, J.V.; Garcia-Arribas, A.B.; Sot, J.; Torrecillas, A.; Gomez-Fernandez, J.C.; Goñi, F.M.; Alonso, A. Lamellar gel (Lb) phases of ternary lipid composition containing ceramide and cholesterol. Biophys. J. 2014, 106, 621–630. [Google Scholar] [CrossRef]
- Goldschmidt-Arzi, M.; Shimoni, E.; Sabanay, H.; Futerman, A.H.; Addadi, L. Intracellular localization of organized lipid domains of C16-ceramide/cholesterol. J. Struct. Biol. 2011, 175, 21–30. [Google Scholar] [CrossRef]
- García-Arribas, A.B.; Alonso, A.; Goñi, F.M. Cholesterol interactions with ceramide and sphingomyelin. Chem. Phys. Lipids 2016, 199, 26–34. [Google Scholar] [CrossRef] [PubMed]
- Chipuk, J.E.; McStay, G.P.; Bharti, A.; Kuwana, T.; Clarke, C.J.; Siskind, L.J.; Obeid, L.M.; Green, D.R. Sphingolipid metabolism cooperates with BAK and BAX to promote the mitochondrial pathway of apoptosis. Cell 2012, 148, 988–1000. [Google Scholar] [CrossRef]
- Lee, H.; Rotolo, J.A.; Mesicek, J.; Penate-Medina, T.; Rimner, A.; Liao, W.-C.; Yin, X.; Ragupathi, G.; Ehleiter, D.; Gulbins, E. Mitochondrial ceramide-rich macrodomains functionalize Bax upon irradiation. PLoS ONE 2011, 6, e19783. [Google Scholar] [CrossRef]
- Jain, A.; Beutel, O.; Ebell, K.; Korneev, S.; Holthuis, J.C. Diverting CERT-mediated ceramide transport to mitochondria triggers Bax-dependent apoptosis. J. Cell Sci. 2017, 130, 360–371. [Google Scholar] [CrossRef]
- Jain, A.; Dadsena, S.; Holthuis, J.C.M. A switchable ceramide transfer protein for dissecting the mechanism of ceramide-induced mitochondrial apoptosis. FEBS Lett. 2020, 594, 3739–3750. [Google Scholar] [CrossRef]
- Cremesti, A.; Paris, F.; Grassme, H.; Holler, N.; Tschopp, J.; Fuks, Z.; Gulbins, E.; Kolesnick, R. Ceramide enables fas to cap and kill. J. Biol. Chem. 2001, 276, 23954–23961. [Google Scholar] [CrossRef] [PubMed]
- Israelachvili, J.N.; Marcelja, S.; Horn, R.G. Physical principles of membrane organization. Q. Rev. Biophys. 1980, 13, 121–200. [Google Scholar] [CrossRef]
- Luzzati, V. Biological significance of lipid polymorphism: The cubic phases. Curr. Opin. Struct. Biol. 1997, 7, 661–668. [Google Scholar] [CrossRef]
- Luzzati, V. X-ray diffraction studies of lipid-water systems. Biol. Membr. 1968, 1, 71–123. [Google Scholar]
- Tan, M.L.; Choong, P.F.; Dass, C.R. Recent developments in liposomes, microparticles and nanoparticles for protein and peptide drug delivery. Peptides 2010, 31, 184–193. [Google Scholar] [CrossRef]
- Pardi, N.; Tuyishime, S.; Muramatsu, H.; Kariko, K.; Mui, B.L.; Tam, Y.K.; Madden, T.D.; Hope, M.J.; Weissman, D. Expression kinetics of nucleoside-modified mRNA delivered in lipid nanoparticles to mice by various routes. J. Control. Release 2015, 217, 345–351. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Cong, H.; Wang, S.; Yu, B.; Shen, Y. Liposomes modified with bio-substances for cancer treatment. Biomater. Sci. 2020, 8, 6442–6468. [Google Scholar] [CrossRef]
- Mulligan, M.J.; Lyke, K.E.; Kitchin, N.; Absalon, J.; Gurtman, A.; Lockhart, S.; Neuzil, K.; Raabe, V.; Bailey, R.; Swanson, K.A. Phase I/II study of COVID-19 RNA vaccine BNT162b1 in adults. Nature 2020, 586, 589–593. [Google Scholar] [CrossRef]
- Jackson, L.A.; Anderson, E.J.; Rouphael, N.G.; Roberts, P.C.; Makhene, M.; Coler, R.N.; McCullough, M.P.; Chappell, J.D.; Denison, M.R.; Stevens, L.J.; et al. An mRNA Vaccine against SARS-CoV-2—Preliminary Report. N. Engl. J. Med. 2020, 383, 1920–1931. [Google Scholar] [CrossRef]
- Montes, L.R.; Ahyayauch, H.; Ibarguren, M.; Sot, J.; Alonso, A.; Bagatolli, L.A.; Goñi, F.M. Electroformation of giant unilamellar vesicles from native membranes and organic lipid mixtures for the study of lipid domains under physiological ionic-strength conditions. Methods Mol. Biol. 2010, 606, 105–114. [Google Scholar]
- Angelova, M.I.; Dimitrov, D.S. Liposome electroformation. Faraday Discuss. Chem. Soc. 1986, 81, 303–311. [Google Scholar] [CrossRef]
- Ruiz-Arguello, M.B.; Basañez, G.; Goñi, F.M.; Alonso, A. Different effects of enzyme-generated ceramides and diacylglycerols in phospholipid membrane fusion and leakage. J. Biol. Chem. 1996, 271, 26616–26621. [Google Scholar] [CrossRef] [PubMed]
- Alonso, A.; Sáez, R.; Villena, A.; Goñi, F.M. Increase in size of sonicated phospholipid vesicles in the presence of detergents. J. Membr. Biol. 1982, 67, 55–62. [Google Scholar] [CrossRef] [PubMed]
- Nieva, J.L.; Goni, F.M.; Alonso, A. Liposome fusion catalytically induced by phospholipase C. Biochemistry 1989, 28, 7364–7367. [Google Scholar] [CrossRef] [PubMed]
- McConnell, H.; Watts, T.; Weis, R.; Brian, A. Supported planar membranes in studies of cell-cell recognition in the immune system. Biochim. Et Biophys Acta (BBA)-Rev. Biomembr. 1986, 864, 95–106. [Google Scholar] [CrossRef]
- Jass, J.; Tjarnhage, T.; Puu, G. From liposomes to supported, planar bilayer structures on hydrophilic and hydrophobic surfaces: An atomic force microscopy study. Biophys. J. 2000, 79, 3153–3163. [Google Scholar] [CrossRef]
- Suarez-Germa, C.; Domenech, O.; Montero, M.T.; Hernandez-Borrell, J. Effect of lactose permease presence on the structure and nanomechanics of two-component supported lipid bilayers. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2014, 1838, 842–852. [Google Scholar] [CrossRef]
- Sezgin, E.; Schneider, F.; Galiani, S.; Urbančič, I.; Waithe, D.; Lagerholm, B.C.; Eggeling, C. Measuring nanoscale diffusion dynamics in cellular membranes with super-resolution STED—FCS. Nat. Protoc. 2019, 14, 1054–1083. [Google Scholar] [CrossRef]
- Buttner, M.; Lagerholm, C.B.; Waithe, D.; Galiani, S.; Schliebs, W.; Erdmann, R.; Eggeling, C.; Reglinski, K. Challenges of Using Expansion Microscopy for Super-resolved Imaging of Cellular Organelles. ChemBioChem 2021, 22, 686–693. [Google Scholar] [CrossRef]
- Barbotin, A.; Urbancic, I.; Galiani, S.; Eggeling, C.; Booth, M.; Sezgin, E. z-STED Imaging and Spectroscopy to Investigate Nanoscale Membrane Structure and Dynamics. Biophys. J. 2020, 118, 2448–2457. [Google Scholar] [CrossRef]
- Bjorkqvist, Y.J.; Brewer, J.; Bagatolli, L.A.; Slotte, J.P.; Westerlund, B. Thermotropic behavior and lateral distribution of very long chain sphingolipids. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2009, 1788, 1310–1320. [Google Scholar] [CrossRef][Green Version]
- Bagatolli, L.A. To see or not to see: Lateral organization of biological membranes and fluorescence microscopy. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2006, 1758, 1541–1556. [Google Scholar] [CrossRef]
- Chiantia, S.; Schwille, P.; Klymchenko, A.S.; London, E. Asymmetric GUVs prepared by MbetaCD-mediated lipid exchange: An FCS study. Biophys. J. 2011, 100, L1–L3. [Google Scholar] [CrossRef]
- Maula, T.; Artetxe, I.; Grandell, P.M.; Slotte, J.P. Importance of the sphingoid base length for the membrane properties of ceramides. Biophys. J. 2012, 103, 1870–1879. [Google Scholar] [CrossRef] [PubMed]
- Rog, T.; Orlowski, A.; Llorente, A.; Skotland, T.; Sylvanne, T.; Kauhanen, D.; Ekroos, K.; Sandvig, K.; Vattulainen, I. Interdigitation of long-chain sphingomyelin induces coupling of membrane leaflets in a cholesterol dependent manner. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2016, 1858, 281–288. [Google Scholar] [CrossRef] [PubMed]
- Dutagaci, B.; Becker-Baldus, J.; Faraldo-Gomez, J.D.; Glaubitz, C. Ceramide-lipid interactions studied by MD simulations and solid-state NMR. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2014, 1838, 2511–2519. [Google Scholar] [CrossRef] [PubMed]
- Sane, P.; Salonen, E.; Falck, E.; Repakova, J.; Tuomisto, F.; Holopainen, J.M.; Vattulainen, I. Probing biomembranes with positrons. J. Phys. Chem. B 2009, 113, 1810–1812. [Google Scholar] [CrossRef] [PubMed]
- Axpe, E.; García-Arribas, A.B.; Mujika, J.I.; Mérida, D.; Alonso, A.; Lopez, X.; Garcia, J.A.; Ugalde, J.M.; Goñi, F.; Plazaola, F. Ceramide increases free volume voids in DPPC membranes. RSC Adv. 2015, 5, 44282–44290. [Google Scholar] [CrossRef]
- Binnig, G.; Quate, C.F.; Gerber, C. Atomic force microscope. Phys. Rev. Lett. 1986, 56, 930. [Google Scholar] [CrossRef]
- Butt, H.-J. Measuring electrostatic, van der Waals, and hydration forces in electrolyte solutions with an atomic force microscope. Biophys. J. 1991, 60, 1438–1444. [Google Scholar] [CrossRef]
- Martin, Y.; Wickramasinghe, H.K. Magnetic imaging by ‘‘force microscopy’’ with 1000 Å resolution. Appl. Phys. Lett. 1987, 50, 1455–1457. [Google Scholar] [CrossRef]
- Di Muzio, M.; Millan-Solsona, R.; Dols-Perez, A.; Borrell, J.H.; Fumagalli, L.; Gomila, G. Dielectric properties and lamellarity of single liposomes measured by in-liquid scanning dielectric microscopy. J. Nanobiotechnol. 2021, 19, 167. [Google Scholar] [CrossRef] [PubMed]
- Ozkan, A.D.; Topal, A.E.; Dikecoglu, F.B.; Guler, M.O.; Dana, A.; Tekinay, A.B. Probe Microscopy Methods and Applications in Imaging of Biological Materials. Semin. Cell Dev. Biol. 2018, 73, 153–164. [Google Scholar] [CrossRef]
- Garcia-Manyes, S.; Sanz, F. Nanomechanics of lipid bilayers by force spectroscopy with AFM: A perspective. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2010, 1798, 741–749. [Google Scholar] [CrossRef] [PubMed]
- Simonsen, A.C.; Bagatolli, L.A. Structure of spin-coated lipid films and domain formation in supported membranes formed by hydration. Langmuir 2004, 20, 9720–9728. [Google Scholar] [CrossRef]
- Jiménez-Rojo, N.; García-Arribas, A.B.; Sot, J.; Alonso, A.; Goñi, F.M. Lipid bilayers containing sphingomyelins and ceramides of varying N-acyl lengths: A glimpse into sphingolipid complexity. Biochim. Biophys. Acta (BBA)-Biomembr. 2014, 1838, 456–464. [Google Scholar] [CrossRef]
- Garcia-Manyes, S.; Redondo-Morata, L.; Oncins, G.; Sanz, F. Nanomechanics of lipid bilayers: Heads or tails? J. Am. Chem. Soc. 2010, 132, 12874–12886. [Google Scholar] [CrossRef]
- Redondo-Morata, L.; Oncins, G.; Sanz, F. Force spectroscopy reveals the effect of different ions in the nanomechanical behavior of phospholipid model membranes: The case of potassium cation. Biophys. J. 2012, 102, 66–74. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Banerjee, S.; Lyubchenko, Y.L. Topographically smooth and stable supported lipid bilayer for high-resolution AFM studies. Methods 2021. [Google Scholar] [CrossRef]
- Attwood, S.J.; Choi, Y.; Leonenko, Z. Preparation of DOPC and DPPC Supported Planar Lipid Bilayers for Atomic Force Microscopy and Atomic Force Spectroscopy. Int. J. Mol. Sci. 2013, 14, 3514–3539. [Google Scholar] [CrossRef]
- Leonenko, Z.; Finot, E.; Ma, H.; Dahms, T.; Cramb, D. Investigation of temperature-induced phase transitions in DOPC and DPPC phospholipid bilayers using temperature-controlled scanning force microscopy. Biophys. J. 2004, 86, 3783–3793. [Google Scholar] [CrossRef]
- Lv, Z.; Banerjee, S.; Zagorski, K.; Lyubchenko, Y.L. Supported Lipid Bilayers for Atomic Force Microscopy Studies. Methods Mol. Biol. 2018, 1814, 129–143. [Google Scholar]
- De Pablo, P.J.; Hernando-Perez, M.; Carrasco, C.; Carrascosa, J.L. Direct visualization of single virus restoration after damage in real time. J. Biol. Phys. 2018, 44, 225–235. [Google Scholar] [CrossRef] [PubMed]
- González-Bullón, D.; B Uribe, K.; Largo, E.; Guembelzu, G.; García-Arribas, A.B.; Martín, C.; Ostolaza, H. Membrane permeabilization by Bordetella adenylate cyclase toxin involves pores of tunable size. Biomolecules 2019, 9, 183. [Google Scholar] [CrossRef] [PubMed]
- González-Ramírez, E.J.; Artetxe, I.; García-Arribas, A.B.; Goñi, F.l.M.; Alonso, A. Homogeneous and heterogeneous bilayers of ternary lipid compositions containing equimolar ceramide and cholesterol. Langmuir 2019, 35, 5305–5315. [Google Scholar] [CrossRef] [PubMed]
- Chiantia, S.; Ries, J.; Kahya, N.; Schwille, P. Combined AFM and two-focus SFCS study of raft-exhibiting model membranes. Chemphyschem A Eur. J. Chem. Phys. Phys. Chem. 2006, 7, 2409–2418. [Google Scholar] [CrossRef]
- Leidy, C.; Mouritsen, O.G.; Jørgensen, K.; Peters, G.H. Evolution of a rippled membrane during phospholipase A2 hydrolysis studied by time-resolved AFM. Biophys. J. 2004, 87, 408–418. [Google Scholar] [CrossRef] [PubMed]
- Picas, L.; Rico, F.; Scheuring, S. Direct measurement of the mechanical properties of lipid phases in supported bilayers. Biophys. J. 2012, 102, L01–L03. [Google Scholar] [CrossRef]
- Ando, T. High-speed atomic force microscopy. Curr. Opin. Chem. Biol. 2019, 51, 105–112. [Google Scholar] [CrossRef] [PubMed]
- Monasterio, B.G.; Jiménez-Rojo, N.; García-Arribas, A.B.; Riezman, H.; Goñi, F.M.; Alonso, A. Patches and Blebs: A Comparative Study of the Composition and Biophysical Properties of Two Plasma Membrane Preparations from CHO Cells. Int. J. Mol. Sci. 2020, 21, 2643. [Google Scholar] [CrossRef]
- Sot, J.; Goñi, F.M.; Alonso, A. Molecular associations and surface-active properties of short-and long-N-acyl chain ceramides. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2005, 1711, 12–19. [Google Scholar] [CrossRef]
- Garcia-Manyes, S.; Oncins, G.; Sanz, F. Effect of temperature on the nanomechanics of lipid bilayers studied by force spectroscopy. Biophys. J. 2005, 89, 4261–4274. [Google Scholar] [CrossRef] [PubMed]
- Balleza, D.; García-Arribas, A.B.; Sot, J.; Ruiz-Mirazo, K.; Goni, F.M. Ether- versus Ester-Linked Phospholipid Bilayers Containing either Linear or Branched Apolar Chains. Biophys. J. 2014, 107, 1364–1374. [Google Scholar] [CrossRef] [PubMed]
- Redondo-Morata, L.; Giannotti, M.I.; Sanz, F. Influence of cholesterol on the phase transition of lipid bilayers: A temperature-controlled force spectroscopy study. Langmuir 2012, 28, 12851–12860. [Google Scholar] [CrossRef] [PubMed]
- Alessandrini, A.; Seeger, H.M.; Caramaschi, T.; Facci, P. Dynamic force spectroscopy on supported lipid bilayers: Effect of temperature and sample preparation. Biophys. J. 2012, 103, 38–47. [Google Scholar] [CrossRef] [PubMed]
- Relat-Goberna, J.; Beedle, A.E.M.; Garcia-Manyes, S. The Nanomechanics of Lipid Multibilayer Stacks Exhibits Complex Dynamics. Small 2017, 13, 1700147. [Google Scholar] [CrossRef] [PubMed]
- Vázquez, R.F.; Ovalle-García, E.; Antillón, A.; Ortega-Blake, I.; Bakás, L.S.; Muñoz-Garay, C.; Maté, S.M. Asymmetric bilayers mimicking membrane rafts prepared by lipid exchange: Nanoscale characterization using AFM-Force spectroscopy. Biochim. Et Biophys. Acta (BBA)-Biomembr. 2021, 1863, 183467. [Google Scholar] [CrossRef] [PubMed]
- Ahyayauch, H.; García-Arribas, A.B.; Masserini, M.E.; Pantano, S.; Goñi, F.M.; Alonso, A. β-Amyloid (1–42) peptide adsorbs but does not insert into ganglioside-containing phospholipid membranes in the liquid-disordered state: Modelling and experimental studies. Int. J. Biol. Macromol. 2020, 164, 2651–2658. [Google Scholar] [CrossRef] [PubMed]
- Gumí-Audenis, B.; Costa, L.; Ferrer-Tasies, L.; Ratera, I.; Ventosa, N.; Sanz, F.; Giannotti, M.I. Pulling lipid tubes from supported bilayers unveils the underlying substrate contribution to the membrane mechanics. Nanoscale 2018, 10, 14763–14770. [Google Scholar] [CrossRef]
- Honigmann, A.; Sadeghi, S.; Keller, J.; Hell, S.W.; Eggeling, C.; Vink, R. A lipid bound actin meshwork organizes liquid phase separation in model membranes. eLife 2014, 3, e01671. [Google Scholar] [CrossRef]
- Honigmann, A.; Mueller, V.; Hell, S.W.; Eggeling, C. STED microscopy detects and quantifies liquid phase separation in lipid membranes using a new far-red emitting fluorescent phosphoglycerolipid analogue. Faraday Discuss. 2013, 161, 77–89; discussion 113–150. [Google Scholar] [CrossRef] [PubMed]
- García-Arribas, A.B.; Ahyayauch, H.; Sot, J.; López-González, P.L.; Alonso, A.; Goñi, F.M. Ceramide-induced lamellar gel phases in fluid cell lipid extracts. Langmuir 2016, 32, 9053–9063. [Google Scholar] [CrossRef] [PubMed]
- Kusumi, A.; Fujiwara, T.K.; Morone, N.; Yoshida, K.J.; Chadda, R.; Xie, M.; Kasai, R.S.; Suzuki, K.G. Membrane mechanisms for signal transduction: The coupling of the meso-scale raft domains to membrane-skeleton-induced compartments and dynamic protein complexes. Semin. Cell Dev. Biol. 2012, 23, 126–144. [Google Scholar] [CrossRef]
- Sullan, R.M.A.; Li, J.K.; Hao, C.; Walker, G.C.; Zou, S. Cholesterol-dependent nanomechanical stability of phase-segregated multicomponent lipid bilayers. Biophys. J. 2010, 99, 507–516. [Google Scholar] [CrossRef]
- Sullan, R.M.A.; Li, J.K.; Zou, S. Direct correlation of structures and nanomechanical properties of multicomponent lipid bilayers. Langmuir 2009, 25, 7471–7477. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
García-Arribas, A.B.; Goñi, F.M.; Alonso, A. Lipid Self-Assemblies under the Atomic Force Microscope. Int. J. Mol. Sci. 2021, 22, 10085. https://doi.org/10.3390/ijms221810085
García-Arribas AB, Goñi FM, Alonso A. Lipid Self-Assemblies under the Atomic Force Microscope. International Journal of Molecular Sciences. 2021; 22(18):10085. https://doi.org/10.3390/ijms221810085
Chicago/Turabian StyleGarcía-Arribas, Aritz B., Félix M. Goñi, and Alicia Alonso. 2021. "Lipid Self-Assemblies under the Atomic Force Microscope" International Journal of Molecular Sciences 22, no. 18: 10085. https://doi.org/10.3390/ijms221810085
APA StyleGarcía-Arribas, A. B., Goñi, F. M., & Alonso, A. (2021). Lipid Self-Assemblies under the Atomic Force Microscope. International Journal of Molecular Sciences, 22(18), 10085. https://doi.org/10.3390/ijms221810085





