Natural Products Counteracting Cardiotoxicity during Cancer Chemotherapy: The Special Case of Doxorubicin, a Comprehensive Review
Abstract
1. Introduction
2. Cardioprotective Potential of Foods and Phytochemicals during DOX Chemotherapy
2.1. Cardioprotective Potential of Fruits
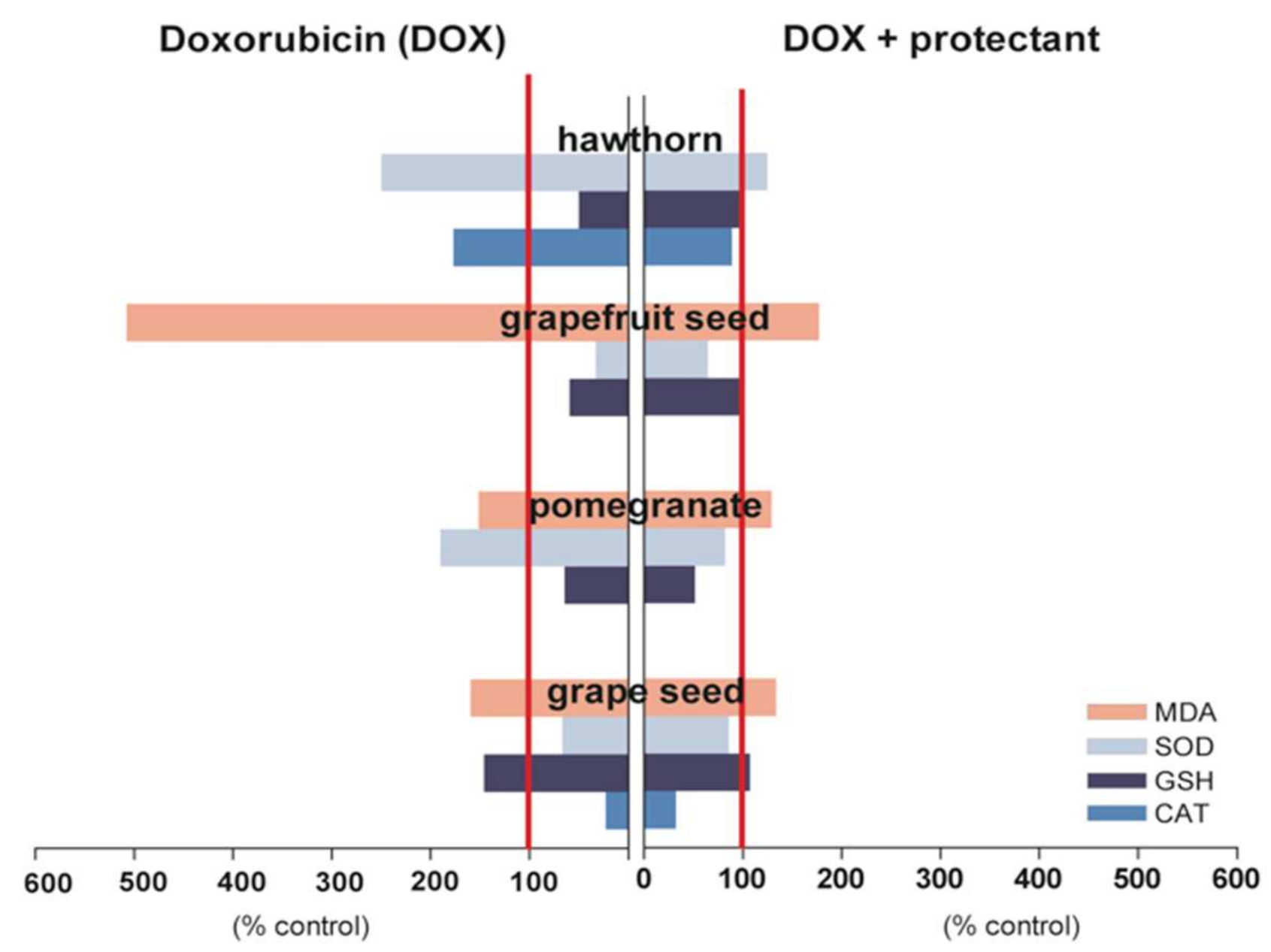
2.1.1. Grapes
2.1.2. Pomegranate
2.1.3. Hawthorn
2.1.4. Grapefruit
2.2. Cardioprotective Potential of Vegetables
2.2.1. Garlic
2.2.2. Tomato
2.2.3. Spinach
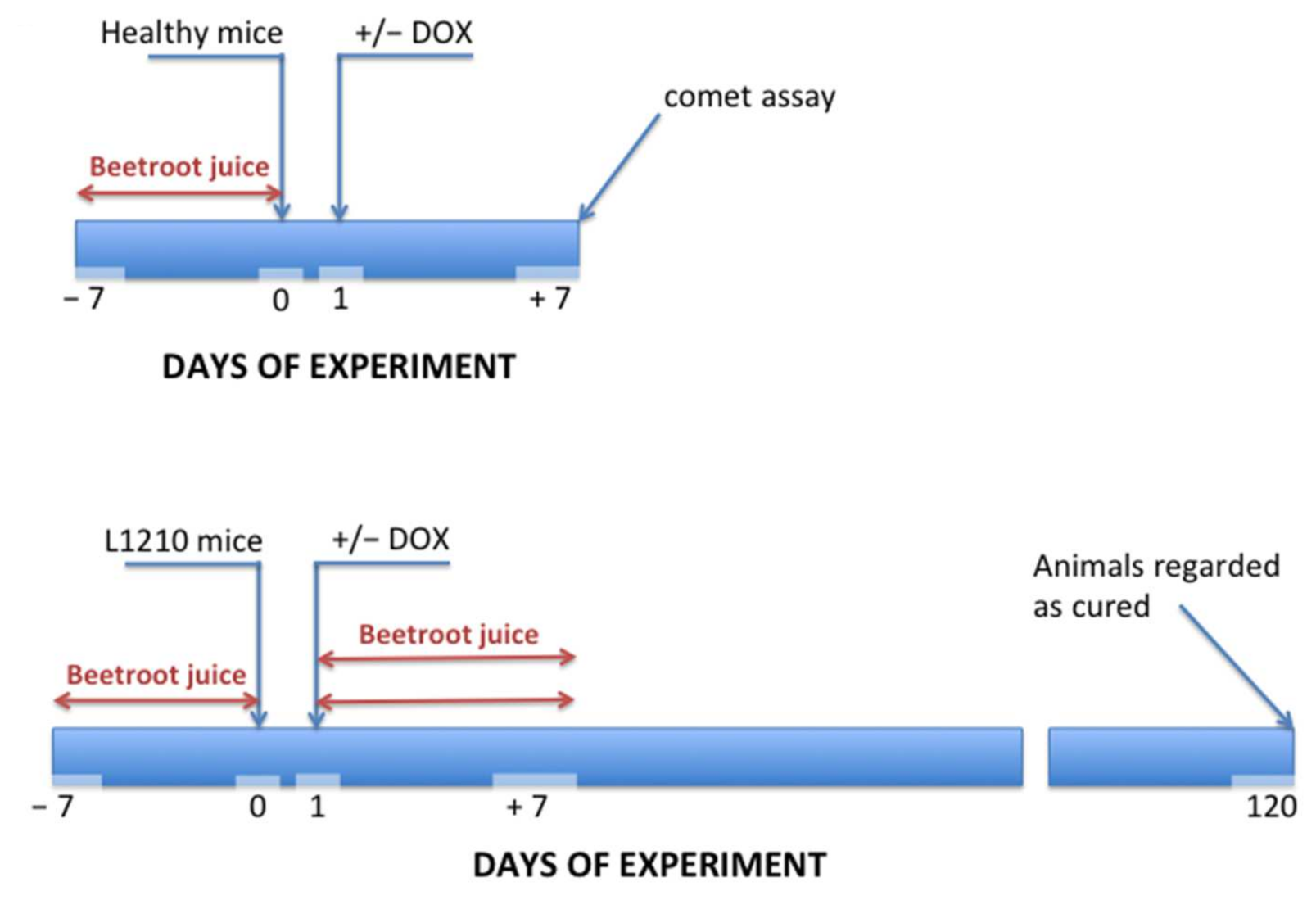
2.2.4. Beetroot
2.3. Cardioprotective Potential of Medicinal Mushrooms
2.4. Cardioprotective Potential of Wheat
2.5. Cardioprotective Potential of Herbs and Spices
2.5.1. Ginkgo Biloba
2.5.2. Red Sage
2.5.3. Saffron
2.5.4. Ginger
2.5.5. Phyllanthus urinaria
2.5.6. Spirulina
2.6. Cardioprotective Dietary Supplement—CardiPro
2.7. Phytochemicals Isolated from Edible Plants with Cardioprotective Potential
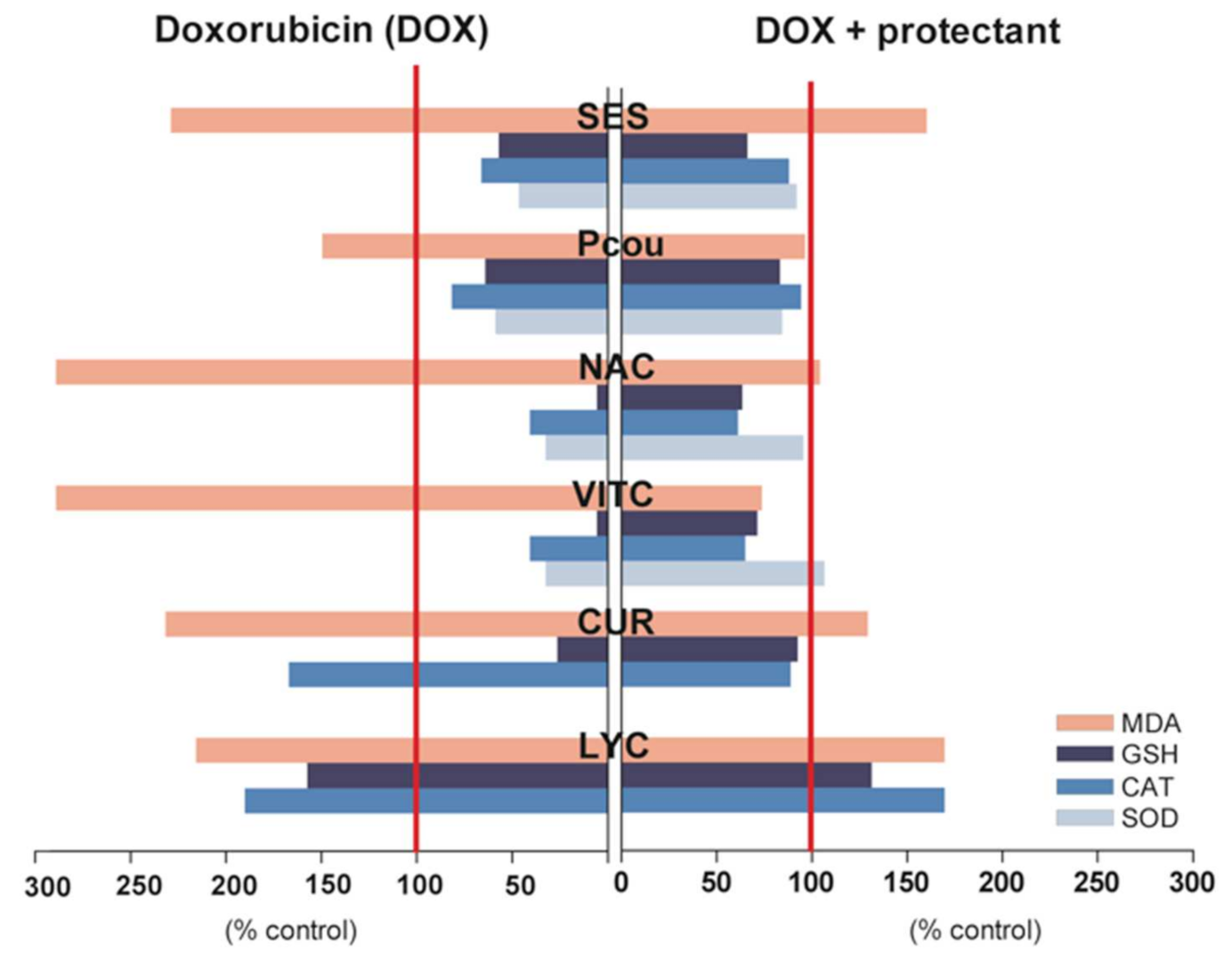
2.7.1. Sesamol
2.7.2. p-Coumaric Acid
2.7.3. Curcumin
2.7.4. Chalcones
2.7.5. Catechins
- A scheme of 20 mg/kg catechin, followed 30 min by DOX;
- A scheme of 10 mg/kg catechin 30 min before and 1 h after DOX treatment;
- A scheme of 200 mg/kg catechin, followed 30 min by DOX;
- A scheme of 100 mg/kg catechin 30 min before and 1 h after administration of DOX;
- A scheme of 500 mg/kg catechin, followed 30 min by DOX.
2.7.6. Rutin
2.7.7. Caffeic Acid Phenylethyl Ester
2.7.8. Oleuropein
2.7.9. Cannabinoids
2.8. Other Cardioprotective Food Ingredients
2.8.1. Selenium and Vitamins A, C, E, D
2.8.2. Melatonin
- Left ventricular diastolic pressure (LVEDP);
- Left ventricular systolic pressure (LVESP);
- the first derivative of left-ventricular pressure after time (± dP/dt);
- Ejection volume (SV);
- Cardiac efficiency (CO).
2.8.3. CoQ10 and l-Carnitine
2.9. Semi-Synthetic Compounds with Cardioprotective Properties
2.9.1. MonoHER and Its Derivative Frederine
2.9.2. Metformin
3. Strengths and Limitations
4. Conclusions and Future Perspectives
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, H.; Ferlay, J.; Siegel, R.L.; Laversanne, M.; Soerjomataram, I.; Jemal, A.; Bray, F. Global Cancer Statistics 2020: GLOBOCAN Estimates of Incidence and Mortality Worldwide for 36 Cancers in 185 Countries. CA Cancer J. Clin. 2021, 71, 209–249. [Google Scholar] [CrossRef]
- Bray, F.; Ferlay, J.; Soerjomataram, I.; Siegel, R.L.; Torre, L.A.; Jemal, A. Global cancer statistics 2018: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J. Clin. 2018, 68, 394–424. [Google Scholar] [CrossRef]
- World Health Organization. World Cancer Report; Stewart, B.W., Wild, C.P., Eds.; IARC: Lyon, France, 2014; pp. 1–632. [Google Scholar]
- Agrawal, M.; Garg, R.J.; Cortes, J.; Quintas-Cardama, A. Tyrosine Kinase Inhibitors: The First Decade. Curr. Hematol. Malign. Rep. 2010, 5, 70–80. [Google Scholar] [CrossRef]
- Howlader, N.; Noone, A.M.; Krapcho, M.; Garshell, J.; Miller, D.; Altekruse, S.F.; Kosary, C.L.; Yu, M.; Ruhl, J.; Tatalovich, Z.; et al. SEER Cancer Statistics Review, 1975–2012; National Cancer Institute: Bethesda, MD, USA, 2021. Available online: https://seer.cancer.gov/archive/csr/1975_2012/ (accessed on 6 December 2020).
- Alfarouk, O.K.; Stock, C.-M.; Taylor, S.; Walsh, M.; Muddathir, A.K.; Verduzco, D.; Bashir, A.H.H.; Mohammed, O.Y.; ElHassan, O.G.; Harguindey, S.; et al. Resistance to cancer chemotherapy: Failure in drug response from ADME to P-gp. Cancer Cell Int. 2015, 15, 71. [Google Scholar] [CrossRef] [PubMed]
- Weaver, K.E.; Forsythe, L.P.; Reeve, B.B.; Alfano, C.M.; Rodriguez, J.L.; Sabatino, S.A.; Hawkins, N.A.; Rowland, J.H. Mental and Physical Health–Related Quality of Life among U.S. Cancer Survivors: Population Estimates from the 2010 National Health Interview Survey. Cancer Epidemiol. Biomark. Prev. 2012, 21, 2108–2117. [Google Scholar] [CrossRef]
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016, 66, 271–289. [Google Scholar] [CrossRef]
- Schimmel, K.J.; Richel, D.J.; Brink, R.B.V.D.; Guchelaar, H.-J. Cardiotoxicity of cytotoxic drugs. Cancer Treat. Rev. 2004, 30, 181–191. [Google Scholar] [CrossRef]
- Birtle, A.J. Anthracyclines and cardiotoxicity. Clin. Oncol. 2000, 12, 146–152. [Google Scholar]
- Minow, R.A.; Benjamin, R.S.; Gottlieb, J.A. Adriamycin (NSC 123127) cardiomyopathy—An overview with determination of risk factors. Cancer Chemother. Rep. 1975, 6, 198–211. [Google Scholar]
- Ewer, M.S.; Lenihan, D.J. Left Ventricular Ejection Fraction and Cardiotoxicity: Is Our Ear Really to the Ground? J. Clin. Oncol. 2008, 26, 1201–1203. [Google Scholar] [CrossRef] [PubMed]
- Ewer, M.S.; Ewer, S.M. Troponin I Provides Insight Into Cardiotoxicity and the Anthracycline-Trastuzumab Interaction. J. Clin. Oncol. 2010, 28, 3901–3904. [Google Scholar] [CrossRef]
- Štěrba, M.; Popelová, O.; Vávrová, A.; Jirkovsky, E.; Kovaříková, P.; Geršl, V.; Šimůnek, T. Oxidative Stress, Redox Signaling, and Metal Chelation in Anthracycline Cardiotoxicity and Pharmacological Cardioprotection. Antioxid. Redox Signal. 2013, 18, 899–929. [Google Scholar] [CrossRef]
- Khan, M.; Shobha, J.C.; Mohan, I.K.; Naidu, M.U.R.; Sundaram, C.; Singh, S.; Kuppusamy, P.; Kutala, V.K. Protective effect of spirulina against doxorubicin-induced cardiotoxicity. Phytother. Res. 2005, 19, 1030–1037. [Google Scholar] [CrossRef] [PubMed]
- Peng, X.; Chen, B.; Lim, C.C.; Sawyer, D.B. The cardiotoxicology of anthracycline chemotherapeutics: Translating molecular mechanism into preventative medicine. Mol. Interv. 2005, 5, 163–171. [Google Scholar] [CrossRef]
- Chaiswing, L.; Cole, M.P.; Clair, D.K.S.; Ittarat, W.; Szweda, L.I.; Oberley, T.D. Oxidative Damage Precedes Nitrative Damage in Adriamycin-Induced Cardiac Mitochondrial Injury. Toxicol. Pathol. 2004, 32, 536–547. [Google Scholar] [CrossRef]
- Ito, H.; Miller, S.C.; Billingham, M.E.; Akimoto, H.; Torti, S.V.; Wade, R.; Gahlmann, R.; Lyons, G.; Kedes, L.; Torti, F.M. Doxorubicin selectively inhibits muscle gene expression in cardiac muscle cells in vivo and in vitro. Proc. Natl. Acad. Sci. USA 1990, 87, 4275–4279. [Google Scholar] [CrossRef]
- Burke, B.E.; Mushlin, P.S.; Cusack, B.J.; Olson, S.J.; Gambliel, H.A.; Olson, R.D. Decreased sensitivity of neonatal rabbit sarco-plasmic reticulum to anthracycline cardiotoxicity. Cardiovasc. Toxicol. 2002, 2, 41–51. [Google Scholar] [CrossRef]
- Hahm, S.; Dresner, H.S.; Podwall, D.; Golden, M.; Winiarsky, R.; Moosikasuwan, M.; Cajigas, A.; Steinberg, J.J. DNA biomarkers antecede semiquantitative anthracycline cardiomyopathy. Cancer Investig. 2003, 21, 53–67. [Google Scholar] [CrossRef] [PubMed]
- L’Ecuyer, T.; Sanjeev, S.; Thomas, R.; Novak, R.; Das, L.; Campbell, W.; Heide, R.V. DNA damage is an early event in doxo-rubicin-induced cardiac myocyte death. Am. J. Physiol. Heart Circ. Physiol. 2006, 291, 1273–1280. [Google Scholar] [CrossRef]
- Mercurio, V.; Pirozzi, F.; Lazzarini, E.; Marone, G.; Rizzo, P.; Agnetti, G.; Tocchetti, C.G.; Ghigo, A.; Ameri, P. Models of Heart Failure Based on the Cardiotoxicity of Anticancer Drugs. J. Card. Fail. 2016, 22, 449–458. [Google Scholar] [CrossRef] [PubMed]
- Karimi, G.; Ramezani, M.; Abdi, A. Protective effects of lycopene and tomato extract against doxorubicin-induced cardiotoxicity. Phytother. Res. 2005, 19, 912–914. [Google Scholar] [CrossRef] [PubMed]
- Maheshwari, R.K.; Singh, A.K.; Gaddipati, J.; Srimal, R.C. Multiple biological activities of curcumin: A short review. Life Sci. 2006, 78, 2081–2087. [Google Scholar] [CrossRef] [PubMed]
- Silvestrini, A.; Meucci, E.; Vitali, A.; Giardina, B.; Mordente, A. Chalcone Inhibition of Anthracycline Secondary Alcohol Metabolite Formation in Rabbit and Human Heart Cytosol. Chem. Res. Toxicol. 2006, 19, 1518–1524. [Google Scholar] [CrossRef] [PubMed]
- Zhao, Y. Berry fruit. In Value-Added Products for Health Promotion; CRC Press Taylor & Francis Group: Boca Raton, FL, USA, 2000. [Google Scholar]
- Rechner, A.R.; Kuhnle, G.; Bremner, P.; Hubbard, G.P.; Moore, K.P.; Rice-Evans, C.A. The metabolic fate of dietary polyphenols in humans. Free. Radic. Biol. Med. 2002, 33, 220–235. [Google Scholar] [CrossRef]
- Kang, W.; Weiss, M. Modeling the metabolism of idarubicin to idarubicinol in rat heart: Effect of rutin and phenobarbital. Drug Metab. Dispos. 2003, 31, 462–468. [Google Scholar] [CrossRef]
- Wu, W.-M.; Lu, L.; Long, Y.; Wang, T.; Liu, L.; Chen, Q.; Wang, R. Free radical scavenging and antioxidative activities of caffeic acid phenethyl ester (CAPE) and its related compounds in solution and membranes: A structure–activity insight. Food Chem. 2007, 105, 107–115. [Google Scholar] [CrossRef]
- Liu, X.; Chen, Z.; Chua, C.C.; Ma, Y.-S.; Youngberg, G.A.; Hamdy, R.; Chua, B.H.L. Melatonin as an effective protector against doxorubicin-induced cardiotoxicity. Am. J. Physiol. Circ. Physiol. 2002, 283, H254–H263. [Google Scholar] [CrossRef]
- Fallowfield, L. Quality of life: A new perspective for cancer patients. Nat. Rev. Cancer 2002, 2, 873–879. [Google Scholar] [CrossRef]
- Delmas, D.; Passilly-Degrace, P.; Jannin, B.; Malki, M.C.; Latruffe, N. Resveratrol, a chemopreventive agent, disrupts the cell cycle control of human SW480 colorectal tumor cells. Int. J. Mol. Med. 2002, 10, 193–199. [Google Scholar] [CrossRef]
- Baur, J.; Sinclair, D. Therapeutic potential of resveratrol: The in vivo evidence. Nat. Rev. Drug Discov. 2006, 5, 493–506. [Google Scholar] [CrossRef]
- Rezk, Y.A.; Balulad, S.S.; Keller, R.S.; Bennett, J.A. Use of Resveratrol to improve the effectiveness of cisplatin and doxorubicin: Study in human gynecologic cancer cell lines and in rodent heart. Am. J. Obstet. Gynecol. 2006, 194, e23–e26. [Google Scholar] [CrossRef]
- Al-Harthi, S.; AlArabi, O.M.; Ramadan, W.S.; Alaama, M.N.; Al-Kreathy, H.M.; Damanhouri, Z.A.; Khan, L.; Osman, A.-M.M. Amelioration of doxorubicin-induced cardiotoxicity by resveratrol. Mol. Med. Rep. 2014, 10, 1455–1460. [Google Scholar] [CrossRef]
- Bagchi, D.; Sen, C.K.; Ray, S.D.; Das, D.K.; Bagchi, M.; Preuss, H.G.; Vinson, A.J. Molecular mechanisms of cardioprotection by a novel grape seed proanthocyanidin extract. Mutat. Res. Mol. Mech. Mutagen. 2003, 523–524, 87–97. [Google Scholar] [CrossRef]
- Ray, S.D.; Patel, D.; Wong, V.; Bagchi, D. In vivo protection of DNA damage associated apoptotic and necrotic cell deaths during acetaminophen-induced nephrotoxicity, amiodarone-induced lung toxicity and doxorubicin-induced cardiotoxicity by a novel IH636 grape seed pro- anthocyanidin extract. Res. Commun. Mol. Pathol. Pharmacol. 2000, 107, 137–166. [Google Scholar] [PubMed]
- Ammar, S.M.; Said, S.A.; El-Damarawy, S.L.; Suddek, G.M. Cardioprotective effect of grape-seed proanthocyanidins on dox-orubicin-induced cardiac toxicity in rats. Pharm. Biol. 2013, 51, 339–344. [Google Scholar] [CrossRef] [PubMed]
- Lin, H.; Zhang, J.; Ni, T.; Lin, N.; Meng, L.; Gao, F.; Luo, H.; Liu, X.; Chi, J.; Guo, H. Yellow Wine Polyphenolic Compounds prevents Doxorubicin-induced cardiotoxicity through activation of the Nrf2 signalling pathway. J. Cell. Mol. Med. 2019, 23, 6034–6047. [Google Scholar] [CrossRef]
- Mahboubi, A.; Asgarpanah, J.; Sadaghiyani, P.N.; Faizi, M. Total phenolic and flavonoid content and antibacterial activity of Punica granatum L. var. pleniflora flowers (Golnar) against bacterial strains causing foodborne diseases. BMC Complement. Altern. Med. 2015, 15, 366. [Google Scholar] [CrossRef]
- Fard, M.H.; Ghule, A.E.; Bodhankar, S.L.; Dikshit, M. Cardioprotective effect of whole fruit extract of pomegranate on doxorubicin-induced toxicity in rat. Pharm. Biol. 2011, 49, 377–382. [Google Scholar] [CrossRef]
- Mahmud, S.A.; Al-Habib, O.A.; Bugoni, S.; Clericuzio, M.; Vidari, G. A New Ursane-type Triterpenoid and Other Constituents from the Leaves of Crataegus azarolus var. aronia. Nat. Prod. Commun. 2016, 11, 1637–1639. [Google Scholar] [CrossRef]
- Mostafa, D.; Khaleel, E.; Abdel-Aleem, G. Inhibition of the hepatic glucose output is responsible for the hypoglycemic effect of Crataegus aronia against type 2 diabetes mellitus in rats. Arch. Biol. Sci. 2018, 70, 277–287. [Google Scholar] [CrossRef]
- Dallak, M. Crataegus aronia enhances sperm parameters and preserves testicular architecture in both control and non-alcoholic fatty liver disease-induced rats. Pharm. Biol. 2018, 56, 535–547. [Google Scholar] [CrossRef]
- Shatoor, A.S.; Ahmed, M.A.A.S. Cardioprotective effect of Crataegus aronia syn. Azarolus (L) aqueous extract against doxoru-bicin-induced cardiotoxicity and heart failure in Wistar rats. J. Basic. Appl. Sci. Res. 2014, 4, 102–114. [Google Scholar]
- Mink, P.J.; Scrafford, C.G.; Barraj, L.M.; Harnack, L.; Hong, C.P.; Nettleton, J.A.; Jacobs, D.J. Flavonoid intake and cardio-vascular disease mortality: A prospective study in postmenopausal women. Am. J. Clin. Nutr. 2007, 85, 895–909. [Google Scholar] [CrossRef] [PubMed]
- Saalu, L.; Ajayi, G.; Adeneye, A.; Imosemi, I.; Osinubi, A. Ethanolic Seed Extract of Grapefruit (Citrus paradisi Macfad) as an Effective Attenuator of Doxorubicin-Induced Oxidative Stress in the Rat Heart. Int. J. Cancer Res. 2009, 5, 44–52. [Google Scholar] [CrossRef][Green Version]
- Tattelman, E. Health effects of garlic. Am. Fam. Physician 2005, 72, 103–106. [Google Scholar]
- Gorinstein, S.; Leontowicz, M.; Leontowicz, H.; Najman, K.; Namiesnik, J.; Park, Y.-S.; Jung, S.-T.; Kang, S.-G.; Trakhtenberg, S. Supplementation of garlic lowers lipids and increases antioxidant capacity in plasma of rats. Nutr. Res. 2006, 26, 362–368. [Google Scholar] [CrossRef]
- Kojima, R.; Toyama, Y.; Ohnishi, S.T. Protective effects of an aged garlic extract on doxorubicin-induced cardiotoxicity in the mouse. Nutr. Cancer 1994, 22, 163–173. [Google Scholar] [CrossRef] [PubMed]
- Chularojmontri, L.; Wattanapitayakul, S.K.; Herunsalee, A.; Charuchongkolwongse, S.; Niumsakul, S.; Srichairat, S. Antioxi-dative and cardioprotective effects of Phyllanthus urinaria L. on doxorubicin-induced cardiotoxicity. Biol. Pharm. Bull. 2005, 28, 1165–1171. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Yilmaz, S.; Ateşşahin, A.; Sahna, E.; Karahan, I.; Ozer, S. Protective effect of lycopene on adriamycin-induced cardiotoxicity and nephrotoxicity. Toxicology 2006, 218, 164–171. [Google Scholar] [CrossRef] [PubMed]
- Pandjaitan, N.; Howard, L.R.; Morelock, T.; Gil, M.I. Antioxidant Capacity and Phenolic Content of Spinach As Affected by Genetics and Maturation. J. Agric. Food Chem. 2005, 53, 8618–8623. [Google Scholar] [CrossRef]
- Breitbart, E.; Lomnitski, L.; Nyska, A.; Malik, Z.; Bergman, M.; Sofer, Y.; Haseman, J.K.; Grossman, S. Effects of water-soluble antioxidant from spinach, NAO, on doxorubicin-induced heart injury. Hum. Exp. Toxicol. 2001, 20, 337–345. [Google Scholar] [CrossRef]
- Kujala, T.; Loponen, J.; Pihlaja, K. Betalains and phenolics in red beetroot (Beta vulgaris) peel extracts: Extraction and charac-terization. Z. Nat. C. J. Biosci. 2001, 56, 343–348. [Google Scholar]
- Kujala, T.S.; Loponen, J.M.; Klika, K.D.; Pihlaja, K. Phenolics and betacyanins in red beetroot (Beta vulgaris) root: Distribution and effect of cold storage on the content of total phenolics and three individual compounds. J. Agric. Food Chem. 2000, 48, 5338–5342. [Google Scholar] [CrossRef]
- Stintzing, F.C.; Carle, R. Functional properties of anthocyanins and betalains in plants, food, and in human nutrition. Trends Food Sci. Technol. 2004, 15, 19–38. [Google Scholar] [CrossRef]
- Łukowicz, J.; Peszyńska-Sularz, G.; Cieślak, A.; Piasek, A.; Popadiuk, S.; Grajek, W.; Bartoszek, A. Dietary intervention with red beet juice ameliorates side effects resulting from oxidative stress during cancer chemotherapy with doxorubicin in vivo. Acta Biochim. Pol. 2006, 53, 133. [Google Scholar]
- Mahajna, J.; Dotan, N.; Zaidman, B.-Z.; Petrova, R.D.; Wasser, S.P. Pharmacological Values of Medicinal Mushrooms for Prostate Cancer Therapy: The Case of Ganoderma Lucidum. Nutr. Cancer 2008, 61, 16–26. [Google Scholar] [CrossRef]
- Zaidman, B.-Z.; Wasser, S.P.; Nevo, E.; Mahajna, J. Androgen receptor-dependent and -independent mechanisms mediate Ganoderma lucidum activities in LNCaP prostate cancer cells. Int. J. Oncol. 2007, 31, 959–967. [Google Scholar] [CrossRef]
- Zaidman, B.-Z.; Wasser, S.P.; Nevo, E.; Mahajna, J. Coprinus comatus and Ganoderma lucidum interfere with androgen receptor function in LNCaP prostate cancer cells. Mol. Biol. Rep. 2007, 35, 107–117. [Google Scholar] [CrossRef] [PubMed]
- Veena, R.K.; Ajith, T.A.; Janardhanan, K.K. Lingzhi or Reishi Medicinal Mushroom, Ganoderma lucidum (Agaricomycetes), Prevents Doxorubicin-Induced Cardiotoxicity in Rats. Int. J. Med. Mushrooms 2018, 20, 761–774. [Google Scholar] [CrossRef]
- Arunachalam, S.; Kim, S.Y.; Lee, S.H.; Lee, Y.H.; Kim, M.S.; Yun, B.S.; Yi, H.K.; Hwang, P.H. Davallialactone protects against adriamycin-induced cardiotoxicity in vitro and in vivo. J. Nat. Med. 2012, 66, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Laddomada, B.; Caretto, S.; Mita, G. Wheat Bran Phenolic Acids: Bioavailability and Stability in Whole Wheat-Based Foods. Molecules 2015, 20, 15666–15685. [Google Scholar] [CrossRef] [PubMed]
- Sahu, R.; Dua, T.K.; Das, S.; De Feo, V.; Dewanjee, S. Wheat phenolics suppress doxorubicin-induced cardiotoxicity via inhi-bition of oxidative stress, MAP kinase activation, NF-κB pathway, PI3K/Akt/mTOR impairment, and cardiac apoptosis. Food Chem. Toxicol. 2019, 125, 503–519. [Google Scholar] [CrossRef]
- Dubey, A.K.; Shankar, P.R.; Upadhyaya, D.; Deshpande, V.Y. Ginkgo biloba—An appraisal. Kathmandu Univ. Med. J. 2004, 2, 225–229. [Google Scholar]
- Timioğlu, O.; Kutsal, S.; Ozkur, M.; Uluoğlu, O.; Aricioğlu, A.; Cevik, C.; Düzgün, E.; Sancak, B.; Poyraz, A. The effect of EGb 761 on the doxorubicin cardiomyopathy. Res. Commun. Mol. Pathol. Pharmacol. 1999, 106, 181–192. [Google Scholar] [PubMed]
- Jasim, S.T.; Al-Kuraishy, H.M.; Al-Gareeb, A.I. Gingko Biloba protects cardiomyocytes against acute doxorubicin induced car-diotoxicity by suppressing oxidative stress. J. Pak. Med. Assoc. 2019, 69, 103–107. [Google Scholar]
- Zhou, L.; Zuo, Z.; Chow, M.S.S. Danshen: An Overview of Its Chemistry, Pharmacology, Pharmacokinetics, and Clinical Use. J. Clin. Pharmacol. 2005, 45, 1345–1359. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.-Y.; Ma, F.-L.; Liu, J.-T.; Tian, J.-W.; Fu, F.-H. Protective Effect of Salvianic Acid A on Acute Liver Injury Induced by Carbon Tetrachloride in Rats. Biol. Pharm. Bull. 2007, 30, 44–47. [Google Scholar] [CrossRef] [PubMed]
- Jiang, B.; Zhang, L.; Li, M.; Wu, W.; Yang, M.; Wang, J.; Guo, D.-A. Salvianolic acids prevent acute doxorubicin cardiotoxicity in mice through suppression of oxidative stress. Food Chem. Toxicol. 2008, 46, 1510–1515. [Google Scholar] [CrossRef] [PubMed]
- Karimi, E.; Oskoueian, E.; Hendra, R.; Jaafar, H.Z. Evaluation of Crocus sativus L. Stigma Phenolic and Flavonoid Compounds and Its Antioxidant Activity. Molecules 2010, 15, 6244–6256. [Google Scholar] [CrossRef]
- Amin, B.; Abnous, K.; Motamedshariaty, V.; Hosseinzadeh, H. Attenuation of oxidative stress, inflammation and apoptosis by ethanolic and aqueous extracts of Crocus sativus L. stigma after chronic constriction injury of rats. An. Acad. Bras. Ciências 2014, 86, 1821–1832. [Google Scholar] [CrossRef]
- Samarghandian, S.; Boskabady, M.H.; Davoodi, S. Use of in vitro assays to assess the potential antiproliferative and cytotoxic effects of saffron (Crocus sativus L.) in human lung cancer cell line. Pharmacogn. Mag. 2010, 6, 309–314. [Google Scholar] [CrossRef]
- Chahine, N.; Makhlouf, H.; Duca, L.; Martiny, L.; Chahine, R. Cardioprotective effect of saffron extracts against acute doxo-rubicin toxicity in isolated rabbit hearts submitted to ischemia-reperfusion injury. Z Nat. C. J. Biosci. 2014, 69, 459–470. [Google Scholar]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef] [PubMed]
- Ajith, T.A.; Hema, U.; Aswathi, S. Zingiber officinale Roscoe ameliorates anticancer antibiotic doxorubicin-induced acute car-diotoxicity in rat. J. Exp. Ther. Oncol. 2016, 11, 171–175. [Google Scholar]
- Calixto, J.B.; Santos, A.R.S.; Filho, V.C.; Yunes, R.A. ChemInform Abstract: A Review of the Plants of the Genus Phyllanthus: Their Chemistry, Pharmacology, and Therapeutic Potential. ChemInform 2010, 29, 225–258. [Google Scholar] [CrossRef]
- Yao, Q.Q.; Zuo, C.X. Chemical studies on the constituents of Phyllanthus urinaria L. Acta Pharm. Sin. 1993, 28, 829–835. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.Z.; Guo, Y.J.; Tu, G.Z.; Guo, W.B.; Miao, F. Studies on chemical constituents of Phyllanthus urinaria L. China J. Chin. Mater. Med. 2000, 25, 615–617. [Google Scholar]
- Wattanapitayakul, S.K.; Chularojmontri, L.; Herunsalee, A.; Charuchongkolwongse, S.; Niumsakul, S.; Bauer, J.A. Screening of Antioxidants from Medicinal Plants for Cardioprotective Effect against Doxorubicin Toxicity. Basic Clin. Pharmacol. Toxicol. 2005, 96, 80–87. [Google Scholar] [CrossRef] [PubMed]
- Rasool, M.; Sabina, E.P.; Lavanya, B. Anti-inflammatory Effect of Spirulina fusiformis on Adjuvant-Induced Arthritis in Mice. Biol. Pharm. Bull. 2006, 29, 2483–2487. [Google Scholar] [CrossRef]
- Chamorro, G.; Salazar, M.; Araújo, K.G.D.L.; Dos Santos, C.P.; Ceballos, G.; Castillo, L.F. Update on the pharmacology of Spirulina (Arthrospira), an unconventional food. Arch. Latinoam. Nutr. 2002, 52, 232–240. [Google Scholar]
- Mohan, I.; Kumar, K.; Naidu, M.; Khan, M.; Sundaram, C. Protective effect of CardiPro against doxorubicin-induced cardiotoxicity in mice. Phytomedicine 2006, 13, 222–229. [Google Scholar] [CrossRef]
- Majdalawieh, A.F.; Mansour, Z.R. Sesamol, a major lignan in sesame seeds (Sesamum indicum): Anti-cancer properties and mechanisms of action. Eur. J. Pharmacol. 2019, 855, 75–89. [Google Scholar] [CrossRef]
- Chennuru, A.; Saleem, M.T. Antioxidant, lipid lowering, and membrane stabilization effect of sesamol against doxorubi-cin-induced cardiomyopathy in experimental rats. Biomed. Res. Int. 2013, 2013, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Abdel-Wahab, M.; El-Mahdy, M.; Abd-Ellah, M.; Helal, G.; Khalifa, F.; Hamada, F. Influence of p-coumaric acid on doxorubicin-induced oxidative stress in rat’s heart. Pharmacol. Res. 2003, 48, 461–465. [Google Scholar] [CrossRef]
- Mohanty, I.; Singh, A.D.; Dinda, A.; Joshi, S.; Talwar, K.K.; Gupta, S.K. Protective effects of Curcuma longa on ische-mia-reperfusion induced myocardial injuries and their mechanisms. Life Sci. 2004, 75, 1701–1711. [Google Scholar] [CrossRef]
- Ashraf, M.Z.; Hussain, M.; Fahim, M. Antiatherosclerotic effects of dietary supplementations of garlic and turmeric: Restoration of endothelial function in rats. Life Sci. 2005, 77, 837–857. [Google Scholar] [CrossRef]
- Venkatesan, N. Curcumin attenuation of acute adriamycin myocardial toxicity in rats. Br. J. Pharmacol. 1998, 124, 425–427. [Google Scholar] [CrossRef]
- He, H.; Luo, Y.; Qiao, Y.; Zhang, Z.; Yin, D.; Yao, J.; You, J.; He, M. Curcumin attenuates doxorubicin-induced cardiotoxicity via suppressing oxidative stress and preventing mitochondrial dysfunction mediated by 14-3-3γ. Food Funct. 2018, 9, 4404–4418. [Google Scholar] [CrossRef] [PubMed]
- Go, M.L.; Wu, X.; Liu, X.L. Chalcones: An Update on Cytotoxic and Chemoprotective Properties. Curr. Med. Chem. 2005, 12, 483–499. [Google Scholar] [CrossRef] [PubMed]
- Sabzevari, O.; Galati, G.; Moridani, M.Y.; Siraki, A.; O’Brien, P.J. Molecular cytotoxic mechanisms of anticancer hydroxychal-cones. Chem. Biol. Interact. 2004, 148, 57–67. [Google Scholar] [CrossRef]
- El Hassan, M.A.; Kedde, M.A.; Zwiers, U.T.; Bast, A.; Van Der Vijgh, W.J. The cardioprotector monoHER does not interfere with the pharmacokinetics or the metabolism of the cardiotoxic agent doxorubicin in mice. Cancer Chemother. Pharmacol. 2003, 51, 306–310. [Google Scholar] [CrossRef] [PubMed]
- Hrelia, S.; Bordoni, A.; Angeloni, C.; Leoncini, E.; Toschi, T.; Lercker, G.; Biagi, P. Green tea extracts can counteract the modification of fatty acid composition induced by doxorubicin in cultured cardiomyocytes. Prostaglandins Leukot. Essent. Fat. Acids 2002, 66, 519–524. [Google Scholar] [CrossRef]
- Li, H.L.; Huang, Y.; Zhang, C.N.; Liu, G.; Wei, Y.S.; Wang, A.B.; Liu, Y.Q.; Hui, R.T.; Wei, C.; Williams, G.M.; et al. Epigallocatechin-3 gallate inhibits cardiac hypertrophy through blocking reactive oxidative species-dependent and -independent signal pathways. Free Radic. Biol. Med. 2006, 40, 1756–1775. [Google Scholar] [CrossRef] [PubMed]
- Pagnotta, E.; Calonghi, N.; Hrelia, S.; Masotti, L.; Biagi, P.; Angeloni, C. Green Tea Protects Cytoskeleton from Oxidative Injury in Cardiomyocytes. J. Agric. Food Chem. 2006, 54, 10159–10163. [Google Scholar] [CrossRef] [PubMed]
- Kozluca, O.; Olcay, E.; Sürücü, S.; Güran, Z.; Kulaksiz, T.; Üskent, N. Prevention of doxorubicin induced cardiotoxicity by catechin. Cancer Lett. 1996, 99, 1–6. [Google Scholar] [CrossRef]
- Ozyurt, H.; Ozyurt, B.; Koca, K.; Ozgocmen, S. Caffeic acid phenethyl ester (CAPE) protects rat skeletal muscle against is-chemia-reperfusion-induced oxidative stress. Vascul. Pharmacol. 2007, 47, 108–112. [Google Scholar] [CrossRef] [PubMed]
- Fadillioglu, E.; Oztas, E.; Erdogan, H.; Yagmurca, M.; Sogut, S.; Ucar, M.; Irmak, M.K. Protective effects of caffeic acid phenethyl ester on doxorubicin-induced cardiotoxicity in rats. J. Appl. Toxicol. 2004, 24, 47–52. [Google Scholar] [CrossRef] [PubMed]
- Andreadou, I.; Mikros, E.; Ioannidis, K.; Sigala, F.; Naka, K.; Kostidis, S.; Farmakis, D.; Tenta, R.; Kavantzas, N.; Bibli, S.-I.; et al. Oleuropein prevents doxorubicin-induced cardiomyopathy interfering with signaling molecules and cardiomyocyte metabolism. J. Mol. Cell. Cardiol. 2014, 69, 4–16. [Google Scholar] [CrossRef] [PubMed]
- Ostadhadi, S.; Rahmatollahi, M.; Dehpour, A.R.; Rahimian, R. Therapeutic Potential of Cannabinoids in Counteracting Chemotherapy-induced Adverse Effects: An Exploratory Review. Phytother. Res. 2015, 29, 332–338. [Google Scholar] [CrossRef]
- Durst, R.; Danenberg, H.; Gallily, R.; Mechoulam, R.; Meir, K.; Grad, E.; Beeri, R.; Pugatsch, T.; Tarsish, E.; Lotan, C. Cannabidiol, a nonpsychoactive Cannabis constituent, protects against myocardial ischemic reperfusion injury. Am. J. Physiol. Circ. Physiol. 2007, 293, H3602–H3607. [Google Scholar] [CrossRef]
- Fouad, A.A.; Albuali, W.H.; Al-Mulhim, A.S.; Jresat, I. Cardioprotective effect of cannabidiol in rats exposed to doxorubicin toxicity. Environ. Toxicol. Pharmacol. 2013, 36, 347–357. [Google Scholar] [CrossRef]
- Hao, E.; Mukhopadhyay, P.; Cao, Z.; Erdélyi, K.; Holovac, E.; Liaudet, L.; Eileen, H.; Haskó, G.; Mechoulam, R.; Pacher, P. Cannabidiol Protects against Doxorubicin-Induced Cardiomyopathy by Modulating Mitochondrial Function and Biogenesis. Mol. Med. 2015, 21, 38–45. [Google Scholar] [CrossRef] [PubMed]
- Rajesh, M.; Mukhopadhyay, P.; Bátkai, S.; Patel, V.; Saito, K.; Matsumoto, S.; Kashiwaya, Y.; Horváth, B.; Mukhopadhyay, B.; Becker, L.; et al. Cannabidiol Attenuates Cardiac Dysfunction, Oxidative Stress, Fibrosis, and Inflammatory and Cell Death Signaling Pathways in Diabetic Cardiomyopathy. J. Am. Coll. Cardiol. 2010, 56, 2115–2125. [Google Scholar] [CrossRef]
- Wagner, J.A.; Abesser, M.; Harvey-White, J.; Ertl, G. 2-Arachidonylglycerol Acting on CB1 Cannabinoid Receptors Mediates Delayed Cardioprotection Induced by Nitric Oxide in Rat Isolated Hearts. J. Cardiovasc. Pharmacol. 2006, 47, 650–655. [Google Scholar] [CrossRef] [PubMed]
- Conklin, K.A. Dietary Antioxidants during Cancer Chemotherapy: Impact on Chemotherapeutic Effectiveness and Development of Side Effects. Nutr. Cancer 2000, 37, 1–18. [Google Scholar] [CrossRef]
- Tesoriere, L.; Ciaccio, M.; Valenza, M.; Bongiorno, A.; Maresi, E.; Albiero, R.; Livrea, A.M. Effect of vitamin A administration on resistance of rat heart against doxorubicin-induced cardiotoxicity and lethality. J. Pharmacol. Exp. Ther. 1994, 269, 430–436. [Google Scholar]
- Van Vleet, J.F.; Ferrans, V.J.; Weirich, W.E. Cardiac disease induced by chronic adriamycin administration in dogs and an evaluation of vitamin E and selenium as cardioprotectants. Am. J. Pathol. 1980, 99, 13–42. [Google Scholar]
- Gül, S.S.; Aygün, H. Cardioprotective effect of vitamin D and melatonin on doxorubicin-induced cardiotoxicity in rat model: An electrocardiographic, scintigraphic and biochemical study. Eur. Res. J. 2018, 5, 649–657. [Google Scholar] [CrossRef]
- Quiles, J.L.; Huertas, J.R.; Battino, M.; Mataix, J.; Ramirez-Tortosa, M.C. Antioxidant nutrients and adriamycin toxicity. Toxicology 2002, 180, 79–95. [Google Scholar] [CrossRef]
- Legha, S.S.; Wang, Y.-M.; Mackay, B.; Ewer, M.S.; Hortobagyi, G.N.; Benjamin, R.S.; Ali, M.K. Clinical and Pharmacologic Investigation of the Effects of α-Tocopherol on Adriamycin Cardiotoxicity. Ann. N. Y. Acad. Sci. 1982, 393, 411–417. [Google Scholar] [CrossRef]
- Wagdi, P.; Fluri, M.; Aeschbacher, B.; Fikrle, A.; Meier, B. Cardioprotection in Patients Undergoing Chemo- and/or Radiotherapy for Neoplastic Disease. A Pilot Study. Jpn. Hear. J. 1996, 37, 353–359. [Google Scholar] [CrossRef][Green Version]
- Norman, H.A.; Butrum, R.R.; Feldman, E.; Heber, D.; Nixon, D.; Picciano, M.F.; Rivlin, R.; Simopoulos, A.; Wargovich, M.J.; Weisburger, E.K.; et al. The Role of Dietary Supplements during Cancer Therapy. J. Nutr. 2003, 133, 3794S–3799S. [Google Scholar] [CrossRef]
- Hardeland, R.; Pandi-Perumal, S.R.; Cardinali, D.P. Melatonin. Int. J. Biochem. Cell Biol. 2005, 38, 313–316. [Google Scholar] [CrossRef] [PubMed]
- Blask, D.E.; Dauchy, R.T.; Sauer, L.A.; Krause, J.A. Melatonin uptake and growth prevention in rat hepatoma 7288CTC in response to dietary melatonin: Melatonin receptor-mediated inhibition of tumor linoleic acid metabolism to the growth sig-naling molecule 13-hydroxyoctadeca-dienoic acid and the potential role of phytomelatonin. Carcinogenesis 2004, 25, 951–960. [Google Scholar] [PubMed]
- Kolář, J.; Macháčková, I. Melatonin in higher plants: Occurrence and possible functions. J. Pineal Res. 2005, 39, 333–341. [Google Scholar] [CrossRef]
- Ta, D.X.; Chen, L.; Poeggeler, B.; Manchester, L.; Reiter, R.J. Melatonin: A potent, endogenous hydroxyl radical scavenger. Edcr. J. 1993, 1, 57–60. [Google Scholar]
- Romero, A.; Ramos, E.; Rios, C.D.L.; Egea, J.; Del Pino, J.; Reiter, R.J. A review of metal-catalyzed molecular damage: Protection by melatonin. J. Pineal Res. 2014, 56, 343–370. [Google Scholar] [CrossRef] [PubMed]
- Morishima, I.; Matsui, H.; Mukawa, H.; Hayashi, K.; Toki, Y.; Okumura, K.; Ito, T.; Hayakawa, T. Melatonin, a pineal hormone with antioxidant property, protects against adriamycin cardiomyopathy in rats. Life Sci. 1998, 63, 511–521. [Google Scholar] [CrossRef]
- Oz, E.; Erbas, D.; Sürücü, H.S.; Düzgün, E. Prevention of doxorubicin- induced cardiotoxicity by melatonin. Mol. Cell Biochem. 2006, 282, 31–37. [Google Scholar] [CrossRef]
- Öz, E.; Ilhan, M.N. Effects of melatonin in reducing the toxic effects of doxorubicin. Mol. Cell. Biochem. 2006, 286, 11–15. [Google Scholar] [CrossRef]
- Sahna, E.; Parlakpinar, H.; Ozer, M.K.; Ozturk, F.; Ozugurlu, F.; Acet, A. Melatonin protects against myocardial doxorubicin toxicity in rats: Role of physiological concentrations. J. Pineal Res. 2003, 35, 257–261. [Google Scholar] [CrossRef]
- Wahab, M.H.; Akoul, E.S.; Abdel-Aziz, A.A. Modulatory effects of melatonin and vitamin E on doxorubicin-induced cardio-toxicity in Ehrlich ascites carcinoma-bearing mice. Tumori 2000, 86, 157–162. [Google Scholar] [CrossRef] [PubMed]
- Dziegiel, P.; Murawska-Ciałowicz, E.; Jethon, Z.; Januszewska, L.; Podhorska-OkoŁów, M.; Surowiak, P.; Zawadzki, M.; Rabczyński, J.; Zabel, M. Melatonin stimulates the activity of protective antioxidative enzymes in myocardial cells of rats in the course of doxorubicin intoxication. J. Pineal Res. 2003, 35, 183–187. [Google Scholar] [CrossRef] [PubMed]
- Conti, V.; Izzo, V.; Corbi, G.; Russomanno, G.; Manzo, V.; De Lise, F.; Di Donato, A.; Filippelli, A. Antioxidant Supplementation in the Treatment of Aging-Associated Diseases. Front. Pharmacol. 2016, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Bhagavan, H.N.; Chopra, R.K. Coenzyme Q10: Absorption, tissue uptake, metabolism and pharmacokinetics. Free. Radic. Res. 2006, 40, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Lee, B.J.; Lin, J.S.; Lin, Y.C.; Lin, P.T. Effects of L-carnitine supplementation on oxidative stress and antioxidant enzymes ac-tivities in patients with coronary artery disease: A randomized, placebo-controlled trial. Nutr. J. 2014, 13, 79–86. [Google Scholar] [CrossRef]
- Surai, P.F. Antioxidant action of carnitine: Molecular mechanisms and practical applications. EC Vet. Sci. 2015, 2, 66–84. [Google Scholar]
- Mustafa, H.N.; Hegazy, G.A.; el Awdan, S.; AbdelBaset, M. Protective role of CoQ10 or L-carnitine on the integrity of the myocardium in doxorubicin induced toxicity. Tissue Cell 2017, 49, 410–426. [Google Scholar] [CrossRef]
- Van Acker, A.S.; Boven, E.; Kuiper, K.; Berg, D.-J.V.D.; Grimbergen, A.J.; Kramer, K.; Bast, A.; Van Der Vijgh, W.J. Monohydroxyethylrutoside, a dose-dependent cardioprotective agent, does not affect the antitumor activity of doxorubicin. Clin. Cancer Res. 1997, 3, 1747–1754. [Google Scholar]
- Fujihira, S.; Yamamoto, T.; Matsumoto, M.; Yoshizawa, K.; Oishi, Y.; Fujii, T.; Noguchi, H.; Mori, H. The High Incidence of Atrial Thrombosis in Mice Given Doxorubicin. Toxicol. Pathol. 1993, 21, 362–368. [Google Scholar] [CrossRef]
- Willems, A.M.; Bruynzeel, A.M.; Kedde, M.A.; van Groeningen, C.J.; Bast, A.; van der Vijgh, W.J. A phase I study of mono-hydroxyethyl- rutoside in healthy volunteers. Cancer Chemother. Pharmacol. 2006, 57, 678–684. [Google Scholar] [CrossRef]
- Van Acker, A.F.; Boven, E.; Kramer, K.; Haenen, G.; Bast, A.; Van Der Vijgh, W.J. Frederine, a new and promising protector against doxorubicin-induced cardiotoxicity. Clin. Cancer Res. 2001, 7, 1378–1384. [Google Scholar] [PubMed]
- Viollet, B.; Guigas, B.; Garcia, N.S.; Leclerc, J.; Foretz, M.; Andreelli, F. Cellular and molecular mechanisms of metformin: An overview. Clin. Sci. 2011, 122, 253–270. [Google Scholar] [CrossRef]
- Bailey, C.J. Metformin: Historical overview. Diabetologia 2017, 60, 1566–1576. [Google Scholar] [CrossRef]
- El Messaoudi, S.; Rongen, G.A.; De Boer, R.A.; Riksen, N.P. The cardioprotective effects of metformin. Curr. Opin. Lipidol. 2011, 22, 445–453. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.-M.; Hsieh, M.-C.; Qin, L.; Zhang, J.; Wu, S.-Y. Metformin reduces radiation-induced cardiac toxicity risk in patients having breast cancer. Am. J. Cancer Res. 2019, 9, 1017–1026. [Google Scholar] [PubMed]
- Ajzashokouhi, A.H.; Bostan, H.B.; Jomezadeh, V.; Hayes, A.W.; Karimi, G. A review on the cardioprotective mechanisms of metformin against doxorubicin. Hum. Exp. Toxicol. 2019, 39, 237–248. [Google Scholar] [CrossRef]
- Alhowail, A.; Almogbel, Y. Metformin administration increases the survival rate of doxorubicin-treated mice. Die Pharm. 2019, 74, 737–739. [Google Scholar]
- Chen, J.; Zhang, S.; Pan, G.; Lin, L.; Liu, D.; Liu, Z.; Mei, S.; Zhang, L.; Hu, Z.; Chen, J.; et al. Modulatory effect of metformin on cardiotoxicity induced by doxorubicin via the MAPK and AMPK pathways. Life Sci. 2020, 249, 117498. [Google Scholar] [CrossRef] [PubMed]
- Zilinyi, R.; Czompa, A.; Czegledi, A.; Gajtko, A.; Pituk, D.; Lekli, I.; Tosaki, A. The Cardioprotective Effect of Metformin in Doxorubicin-Induced Cardiotoxicity: The Role of Autophagy. Molecules 2018, 23, 1184. [Google Scholar] [CrossRef] [PubMed]
- Kelleni, M.T.; Amin, E.F.; Abdelrahman, A.M. Effect of Metformin and Sitagliptin on Doxorubicin-Induced Cardiotoxicity in Rats: Impact of Oxidative Stress, Inflammation, and Apoptosis. J. Toxicol. 2015, 2015, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Sheta, A.; Elsakkar, M.; Hamza, M.; Solaiman, A. Effect of metformin and sitagliptin on doxorubicin-induced cardiotoxicity in adult male albino rats. Hum. Exp. Toxicol. 2016, 35, 1227–1239. [Google Scholar] [CrossRef] [PubMed]
- Kobashigawa, L.C.; Xu, Y.C.; Padbury, J.F.; Tseng, Y.-T.; Yano, N. Metformin Protects Cardiomyocyte from Doxorubicin Induced Cytotoxicity through an AMP-Activated Protein Kinase Dependent Signaling Pathway: An In Vitro Study. PLoS ONE 2014, 9, e104888. [Google Scholar] [CrossRef]
- Mak, I.; Evaniew, N.; Ghert, M. Lost in translation: Animal models and clinical trials in cancer treatment. Am. J. Transl. Res. 2014, 6, 114–118. [Google Scholar] [PubMed]
- Ledford, H. Translational research: 4 ways to fix the clinical trial. Nat. Cell Biol. 2011, 477, 526–528. [Google Scholar] [CrossRef]
- Robinson, N.B.; Krieger, K.; Khan, F.M.; Huffman, W.; Chang, M.; Naik, A.; Yongle, R.; Hameed, I.; Krieger, K.; Girardi, L.N.; et al. The current state of animal models in research: A review. Int. J. Surg. 2019, 72, 9–13. [Google Scholar] [CrossRef]
- Reagan-Shaw, S.; Nihal, M.; Ahmad, N. Dose translation from animal to human studies revisited. FASEB J. 2008, 22, 659–661. [Google Scholar] [CrossRef]
- Vandamme, T.F. Use of rodents as models of human diseases. J. Pharm. Bioallied Sci. 2014, 6, 2–9. [Google Scholar] [CrossRef]
- Madden, J.C.; Enoch, S.J.; Paini, A.; Cronin, M.T. A Review of In Silico Tools as Alternatives to Animal Testing: Principles, Resources and Applications. Altern. Lab. Anim. 2020, 48, 146–172. [Google Scholar] [CrossRef]
- Krewski, D.; Andersen, M.; Tyshenko, M.G.; Krishnan, K.; Hartung, T.; Boekelheide, K.; Wambaugh, J.; Jones, D.; Whelan, M.; Thomas, R.; et al. Toxicity testing in the 21st century: Progress in the past decade and future perspectives. Arch. Toxicol. 2019, 94, 1–58. [Google Scholar] [CrossRef]
- Štampar, M.; Breznik, B.; Filipič, M.; Žegura, B. Characterization of In Vitro 3D Cell Model Developed from Human Hepatocellular Carcinoma (HepG2) Cell Line. Cells 2020, 9, 2557. [Google Scholar] [CrossRef] [PubMed]
- Waldesch, F.G.; Konigswinter, B.S.; Remagen, H.B. Herbal Medicinal Products—Scientific and Regulatory Basis for Development Quality Assurance and Marketing Authorization; CRC Press: Boca Raton, FL, USA, 2003; pp. 37–52. [Google Scholar]
- Kobat, H.; Elkonaissi, I.; Dorak, M.T.; Nabhani-Gebara, S. Management of COVID-19 in cancer patients receiving cardiotoxic anti-cancer therapy. Future recommendations for cardio-oncology. Oncol. Rev. 2021, 15, 510–521. [Google Scholar] [CrossRef] [PubMed]
| Biochemical Marker | Function | Changes in the Amount/Activity of Biomarker That Indicate the Increase of Cardiotoxicity |
|---|---|---|
| Aldehyde reductase | Catalyzes the formation of secondary metabolites of chemotherapeutics | ↑ |
| AST—aspartate transaminase | Catalyzes the reversible transfer of an α-amino group between aspartate and glutamate | ↑ |
| Caspase-3 | Participates in cell apoptosis | ↑ |
| CAT—catalase | Catalyzes the decomposition of hydrogen peroxide, prevents the formation free radicals | ↓ |
| Carbonyl reductase | Catalyzes the formation of secondary metabolites of chemotherapeutics | ↑ |
| CK, CPK—creatine kinaze | Indicator of cardiomyocytes necrosis, catalyzes creatine phosphorylation to phosphocreatine which is an energy source for muscle cells | ↑ |
| CML—Nε-(carboxymethyl)lysine | Marker of cardiomyocytes inflammation formed during protein damage | ↑ |
| Conjugated dienes | Products of lipid peroxidation | ↑ |
| GSH—glutathione | Major cellular antioxidant | ↓ |
| GSH-Px—glutathione peroxidase | Catalyzes the decomposition of hydrogen peroxide by glutathione | ↓ |
| GST—glutathione S-transferases | Catalyze the conjugation of reduced glutathione to a wide range of substrates, usually resulting in detoxification | ↓ |
| LDH—lactate dehydrogenase | Participates in lipid peroxidation | ↑ |
| MDA and other substances reacting with thiobarbituric acid (TBARS) | Products of lipid peroxidation | ↑ |
| Myeloperoxidase | Catalyzes the oxidation of some carcinogens using hydrogen peroxide, plays an important role in their biotransformation | ↑ |
| NFκB p50/p65—transcription factor | Apoptosis inhibitor | ↑ |
| SOD—superoxide dismutase | Catalyzes dismutation of superoxide anion radical | ↓ |
| TNF-α—tumor necrosis factor | Cytotoxic for cancer cells | ↑ |
| Troponin I | Inhibitory subunit of the troponin complex, acting to inhibit actin-myosin interaction | ↑ |
| Compound | Structure | Ref. |
|---|---|---|
| Lycopene | 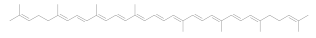 | [24] |
| Curcumin | 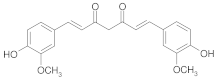 | [25] |
| 2′,4′,2-Trihydroxy chalcone |  | [26] |
| Catechin | 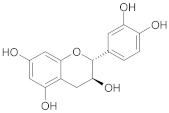 | [27] |
| p-Coumaric acid | 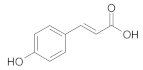 | [28] |
| Rutin |  | [29] |
| Caffeic acid phenylethyl ester | 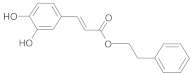 | [30] |
| Melatonin | 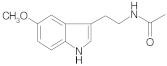 | [31] |
| Group | % CK Activity a |
|---|---|
| Control | 100 ± 2.4 |
| GSE | No change compared to the control |
| DOX | 589.6 ± 60.7 |
| GSE + DOX + GSE | 142.4 ± 19.1 |
| Group | MDA Level [%] a | GSH Level [%] b | % CAT Activity c | % SOD Activity d | % GSH-Px Activity e | TNF-α Expression |
|---|---|---|---|---|---|---|
| CON | 100.0 ± 5.7 | 100.0 ± 8.5 | 100.0 ± 5.9 | 100.0 ± 11.5 | 100.0 ± 20.7 | 0.0 |
| DOX | 161.9±17.1 | 143.6 ± 2.5 | 27.9 ± 7.5 | 66.0 ± 7.7 | 43.0 ± 1.0 | 4.0 |
| PRO + DOX | 71.5 ± 6.5 | 105.9 ± 3.6 | 29.8 ± 4.0 | 90.3 ± 11.5 | 81.9 ± 15.5 | 1.6 |
| G-250 + DOX | 50.9 ± 8.5 | 93.4 ± 3.9 | 50.8 ± 6.7 | 88.4 ± 25.0 | 77.2 ± 15.5 | 2.8 |
| G-500 + DOX | 136.2 ± 9.7 | 131.4 ± 2.4 | 31.1 ± 6.5 | 110.6 ± 13.5 | 91.7 ± 5.2 | 2.9 |
| Group | % CK Activity a |
|---|---|
| CON | 100.0 ± 6.6 |
| DOX | 781.4 ± 99.2 |
| TE (1.2 g/kg) + DOX + TE | 425.5 ± 30.5 |
| TE (2.4 g/kg) + DOX + TE | 256.1 ± 24.0 |
| LYC (1.7 mg/kg) + DOX + LYC | 464.2 ± 33.2 |
| LYC (3.5 mg/kg) + DOX + LYC | 243.6 ± 25.9 |
| Group | MDA Level [%] a | % CAT Activity b | % SOD Activity c | % GSH-Px Activity d |
|---|---|---|---|---|
| CON | 100.0 ± 10.3 | 100.0 ± 9.6 | 100.0 ± 13.4 | 100.0 ± 21.2 |
| ES | 96.0 ± 5.6 | 106.4 ± 11.7 | 118.0 ± 18.0 | 100.5 ± 5.4 |
| DOX | 148.0 ± 12.9 | 152.0 ± 15.9 | 143.6 ± 15.6 | 91.1 ± 12.6 |
| ES + DOX + ES | 111.5 ± 7.4 | 105.3 ± 8.5 | 196.4 ± 14.5 | 133.0 ± 14.1 |
| DOX + ES | 123.4 ± 11.5 | 128.7 ± 15.9 | 138.3 ± 18.0 | 100.2 ± 13.4 |
| Group | LDH Level [%] a | CK Level [%] b | AST Level [%] c | Troponin I [%] d | Troponin T [%] e |
|---|---|---|---|---|---|
| CON | 100.0 ± 9.9 | 100.0 ± 11.0 | 100.0 ± 11.2 | 100.0 ± 10.6 | 100.0 ± 10.6 |
| FA | 98.7 ± 10.6 | 98.8 ± 9.7 | 100.1 ± 10.3 | 100.0 ± 22.0 | 97.9 ± 12.8 |
| API | 98.3 ± 8.8 | 106.1 ± 11.2 | 97.9 ± 10.4 | 100.8 ± 12.9 | 100.0 ± 6.4 |
| WWGPE | 98.4 ± 9.9 | 103.5 ± 10.8 | 97.0 ± 10.7 | 102.3 ± 162.1 | 93.6 ± 6.4 |
| DOX | 129.9 ± 12.2 | 137.8 ± 13.6 | 135.9 ± 15.1 | 162.1 ± 17.4 | 142.5 ± 17.0 |
| DOX + FA | 112.4. ± 11.6 | 116.2 ± 11.9 | 115.8 ± 10.6 | 137.8 ± 14.4 | 121.3 ± 10.6 |
| DOX + API | 114.9 ± 11.9 | 119.0 ± 12.4 | 118.2 ± 11.0 | 135.6 ± 8.3 | 121.3 ± 14.9 |
| DOX + WWGPE | 108.3 ± 11.5 | 112.8 ± 10.2 | 112.6 ± 9.6 | 130.3 ± 11.4 | 110.6 ± 10.6 |
| Group | MDA Level [%] a | GSH Level [%] b | Troponin I [%] c | TNF-α Level [%] d | Caspase-3 Level [%] e |
|---|---|---|---|---|---|
| CON | 100.0 ± 37.6 | 100.0 ± 15.9 | 100.0 ± 20.1 | 100.0 ± 47.9 | 100.0 ± 18.1 |
| DOX | 172.7 ± 67.3 | 58.5 ± 17.4 | 247.1 ± 44.3 | 161.1 ± 38.7 | 185.1 ± 34.4 |
| GB + DOX + GB | 145.4 ± 13.6 | 77.3 ± 13.4 | 192.9 ± 24.8 | 146.7 ± 16.5 | 153.0 ± 31.6 |
| Group | MDA Level [%] a | GSH Level [%] b | % SOD Activity c | % CAT Activity d |
|---|---|---|---|---|
| CON | 100.0 ± 2.9 | 100.0 ± 2.3 | 100.0 ± 0.2 | 100.0 ± 4.0 |
| PU 1 (1 µg/mL) | 89.4 ± 1.8 | 213.5 ± 1.9 | 151.6 ± 2.2 | 136.8 ± 9.6 |
| PU 10 (10 µg/mL) | 88.5 ± 2.7 | 269.2 ± 3.8 | 170.7 ± 0.0 | 169.4 ± 12.4 |
| DOX | 289.0 ± 2.6 | 5.6 ± 3.4 | 32.6 ± 0.4 | 40.8 ± 1.5 |
| PU 1 + DOX | 55.6 ± 2.5 | 164.7 ± 0.8 | 147.3 ± 2.8 | 128.6 ± 8.5 |
| PU 10 + DOX | 30.7 ± 2.1 | 244.4 ± 7.1 | 160.9 ± 0.6 | 161.6 ± 11.7 |
| Group | MDA Level [%] a | % SOD Activity b | % GSH-Px Activity c |
|---|---|---|---|
| CON | 100.0 ± 13.9 | 100.0 ± 4.2 | 100.0 ± 7.1 |
| DOX | 167.4 ± 21.9 | 76.7 ± 8.2 | 72.6 ± 4.9 |
| spirulina | 101.6 ± 12.3 | 95.8 ± 3.7 | 102.7 ± 10.4 |
| spirulina + DOX | 138 ± 9.1 | 100.5 ± 6.3 | 92.8 ± 11.1 |
| Group | MDA Level [%] a | % SOD Activity b | % GSH-Px Activity c |
|---|---|---|---|
| CON | 100.0 ± 11.0 | 100.0 ± 9.3 | 100.0 ± 8.0 |
| DOX | 130.5 ± 3.7 | 79.5 ± 4.3 | 72.0 ± 6.4 |
| CardiPro | 79.3 ± 6.1 | 104.0 ± 2.7 | 106.0 ± 6.2 |
| DOX + CardiPro | 97.0 ± 11.0 | 101.3 ± 3.2 | 98.0 ± 3.6 |
| Compound | IC50 [µM] | |
|---|---|---|
| DOXol | DNRol | |
| 2′,4′,2-trihydroxychalcone | 21.2 ± 3.6 | 33.8 ± 4.4 |
| 2′,4′,3-trihydroxychalcone | 30.9 ± 5.5 | 83.5 ± 6.9 |
| 2′,4′,2,3- tetrahydroxychalcone | 25.3 ± 5.6 | 39.9 ± 4.5 |
| 2′,4′,2,4-tetrahydroxychalcone | 32.8 ± 8.8 | 59.1 ± 9.1 |
| quercetin | 13.7 ± 2.8 | 8.0 ± 2.0 |
| morin | 49.3 ± 6.3 | 18.8 ± 4.1 |
| Group | Body Weight [g] | Heart Weight [g] | Atrial Shrinkage df/dt [g/s] | Q-T Interval [ms] |
|---|---|---|---|---|
| CON | 245.0 ± 9.5 | 47.7 ± 1.3 | 18.0 ± 2.3 | 26.0 ± 1.0 |
| catechin (200 mg/kg) | 224.7 ± 5.3 | 40.0 ± 2.1 | 12.1 ± 1.4 | 26.1 ± 3.4 |
| DOX | 204.3 ± 60.1 | 34.1 ± 1.2 | 9.3 ± 3.4 | 40.4 ± 1.3 |
| catechin + DOX (20 mg/kg) | 241.7 ± 10.9 | 44.7 ± 1.2 | 14.1 ± 2.3 | 32.2 ± 3.3 |
| catechin (10 mg/kg) + DOX + catechin | 240.0 ± 8.6 | 43.5 ± 2.6 | 13.7 ± 2.1 | 33.2 ± 3.1 |
| catechin (200 mg/kg) + DOX | 235.0 ± 6.6 | 38.8 ± 3.1 | 13.2 ± 3.4 | 33.5 ± 2.2 |
| catechin (100 mg/kg) + DOX + catechin | 237.1 ± 5.4 | 38.6 ± 2.1 | 13.1 ± 2.3 | 35.2 ± 1.1 |
| catechin (500 mg/kg) + DOX | 219.2 ± 5.3 | 37.8 ± 3.1 | 10.5 ± 3.0 | 35.7 ± 4.4 |
| Group | % CAT Activity a | % SOD Activity b | % GSH-Px Activity c | MDA Level [%] d | % Myeloperoxidase Activity e | Content of Carbonyl Groups [%] f |
|---|---|---|---|---|---|---|
| CON | 100.0 ± 9.1 | 100.0 ± 10.2 | 100.0 ± 6.2 | 100.0 ± 10.1 | 100.0 ± 23.8 | 100.0 ± 27.3 |
| DOX | 168.8 ± 8.6 | 177.1 ± 12.1 | 116.4 ± 6.2 | 233.3 ± 13.8 | 238.0 ± 47.8 | 246.0 ± 37.4 |
| CAPE + DOX + CAPE | 207.0 ± 10.8 | 192.4 ± 12.1 | 166.7 ± 6.7 | 130.8± 8.7 | 131.2 ± 14.5 | 112.2 ± 33.8 |
| Group | LVEDP [mmHg] | LVESP [mmHg] | +dP/dt [mmHg/s] | −dP/dt [mmHg/s] | SV [µL] | CO [mL/min] |
|---|---|---|---|---|---|---|
| CON | 4.8 ± 0.7 | 64.7 ± 3.8 | 2638 ± 127 | 2010 ± 248 | 9.7 ± 0.5 | 4.3 ± 0.5 |
| DOX | 10.7 ± 2.1 | 37.3 ± 5.5 | 904 ± 156 | 727 ± 153 | 4.1 ± 0.4 | 1.6 ± 0.3 |
| MEL | 3.0 ± 0.8 | 63.2 ± 3.2 | 2664 ± 120 | 1978 ± 163 | 9.2 ± 0.7 | 4.2 ± 0.6 |
| MEL + DOX + MEL | 4.3 ± 0.8 | 60.2 ± 1.2 | 1914 ± 95 | 1629 ± 143 | 7.4 ± 0.8 | 3.2 ± 0.4 |
| 6-OH MEL | 4.5 ± 1.1 | 63.9 ± 4.4 | 2384 ± 334 | 1863 ± 299 | 9.5 ± 0.6 | 4.1 ± 0.5 |
| 6-OH MEL + DOX + 6-OH MEL | 5.8 ± 0.5 | 57.0 ± 5.2 | 1774 ± 208 | 1586 ± 121 | 7.7 ± 0.6 | 3.0 ± 0.5 |
| Group | % MDA Level a | % GSH Level b | % TNF-α Level c | % LDH Level d |
|---|---|---|---|---|
| CON | 100.0 ± 24.5 | 100.0 ± 17.9 | 100.0 ± 11.1 | 100.0 ± 12.1 |
| DOX | 555.9 ± 80.2 | 77.3 ± 16.0 | 491.9 ± 30.9 | 400.2 ± 37.5 |
| CoQ10 | 145.6 ± 14.1 | 118.9 ± 10.4 | 133.8 ± 56.6 | 95.4 ± 18.0 |
| l-car | 161.9 ± 19.0 | 123.6 ± 5.7 | 92.4 ± 9.8 | 104.2 ± 17.8 |
| CoQ10 + DOX | 345.5 ± 63.0 | 108.5 ± 9.4 | 179.5 ± 16.2 | 130.5 ± 18.2 |
| l-car + DOX | 347.7 ± 90.9 | 101.9 ± 7.5 | 282.3 ± 19.6 | 181.3 ± 15.4 |
| Plant/Compound | Markers of Oxidative Stress | Markers of Membrane Damage | Antiinflammatory Properties | Heart Cell Viability | Histopathological Features of Heart Tissue | Cardiac Function | Myocardial Infarction | Mortality |
|---|---|---|---|---|---|---|---|---|
| Grapes | ↓ | ↓ | ↑ | |||||
| Pomegranate | ↓ | ↓ | ↑ | |||||
| Grapefruit | ↓ | |||||||
| Hawthorn | ↓ | ↓ | ||||||
| Garlic | ↓ | ↓ | ↓ | |||||
| Tomato | ↓ | ↓ | ↑ | ↓ | ||||
| Spinach | ↓ | ↓ | ↓ | |||||
| Beetroot | ↓ | ↓ | ||||||
| G. lucidum | ↓ | ↑ | ↓ | ↑ | ||||
| Wheat | ↓ | ↓ | ↑ | |||||
| G. biloba | ↓ | ↓ | ↑ | ↓ | ↑ | ↓ | ||
| Red sage | ↓ | ↓ | ↓ | ↑ | ||||
| Ginger | ↓ | ↓ | ↑ | ↑ | ||||
| Saffron | ↓ | ↓ | ↑ | ↑ | ||||
| P. urinaria | ↓ | ↑ | ||||||
| Spirulina | ↓ | ↓ | ↑ | ↓ | ↓ | |||
| Seasamol | ↓ | ↓ | ||||||
| Coumaric acid | ↓ | ↓ | ||||||
| CAPE | ↓ | ↓ | ↑ | ↓ | ↑ | |||
| Oleuropein | ↓ | ↑ | ↑ | ↑ | ↓ | |||
| Cannabinoids | ↓ | ↑ | ↑ | |||||
| Vitamin C | ↓ | ↓ | ↑ | |||||
| Vitamin A | ↓ | ↓ | ||||||
| Vitamin E | ↓ | ↓ | ||||||
| Vitamin D | ↓ | ↓ | ↑ | |||||
| Curcumin | ↓ | ↓ | ↑ | ↓ | ↑ | ↓ | ||
| Catechins | ↓ | ↓ | ↑ | ↑ | ↓ | |||
| Melatonin | ↓ | ↓ | ↑ | |||||
| CoQ10 | ↓ | ↑ | ↑ | |||||
| l-carnitine | ↓ | ↑ | ↑ | |||||
| Cardi-pro | ↓ | ↓ | ↓ | ↓ | ||||
| MonoHER | ↓ | ↑ | ||||||
| Metformin | ↓ | ↓ | ↑ | ↑ | ↓ | ↑ | ↓ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koss-Mikołajczyk, I.; Todorovic, V.; Sobajic, S.; Mahajna, J.; Gerić, M.; Tur, J.A.; Bartoszek, A. Natural Products Counteracting Cardiotoxicity during Cancer Chemotherapy: The Special Case of Doxorubicin, a Comprehensive Review. Int. J. Mol. Sci. 2021, 22, 10037. https://doi.org/10.3390/ijms221810037
Koss-Mikołajczyk I, Todorovic V, Sobajic S, Mahajna J, Gerić M, Tur JA, Bartoszek A. Natural Products Counteracting Cardiotoxicity during Cancer Chemotherapy: The Special Case of Doxorubicin, a Comprehensive Review. International Journal of Molecular Sciences. 2021; 22(18):10037. https://doi.org/10.3390/ijms221810037
Chicago/Turabian StyleKoss-Mikołajczyk, Izabela, Vanja Todorovic, Sladjana Sobajic, Jamal Mahajna, Marko Gerić, Josep A. Tur, and Agnieszka Bartoszek. 2021. "Natural Products Counteracting Cardiotoxicity during Cancer Chemotherapy: The Special Case of Doxorubicin, a Comprehensive Review" International Journal of Molecular Sciences 22, no. 18: 10037. https://doi.org/10.3390/ijms221810037
APA StyleKoss-Mikołajczyk, I., Todorovic, V., Sobajic, S., Mahajna, J., Gerić, M., Tur, J. A., & Bartoszek, A. (2021). Natural Products Counteracting Cardiotoxicity during Cancer Chemotherapy: The Special Case of Doxorubicin, a Comprehensive Review. International Journal of Molecular Sciences, 22(18), 10037. https://doi.org/10.3390/ijms221810037







