Modeling In Vitro Osteoarthritis Phenotypes in a Vascularized Bone Model Based on a Bone-Marrow Derived Mesenchymal Cell Line and Endothelial Cells
Abstract
:1. Introduction
2. Results
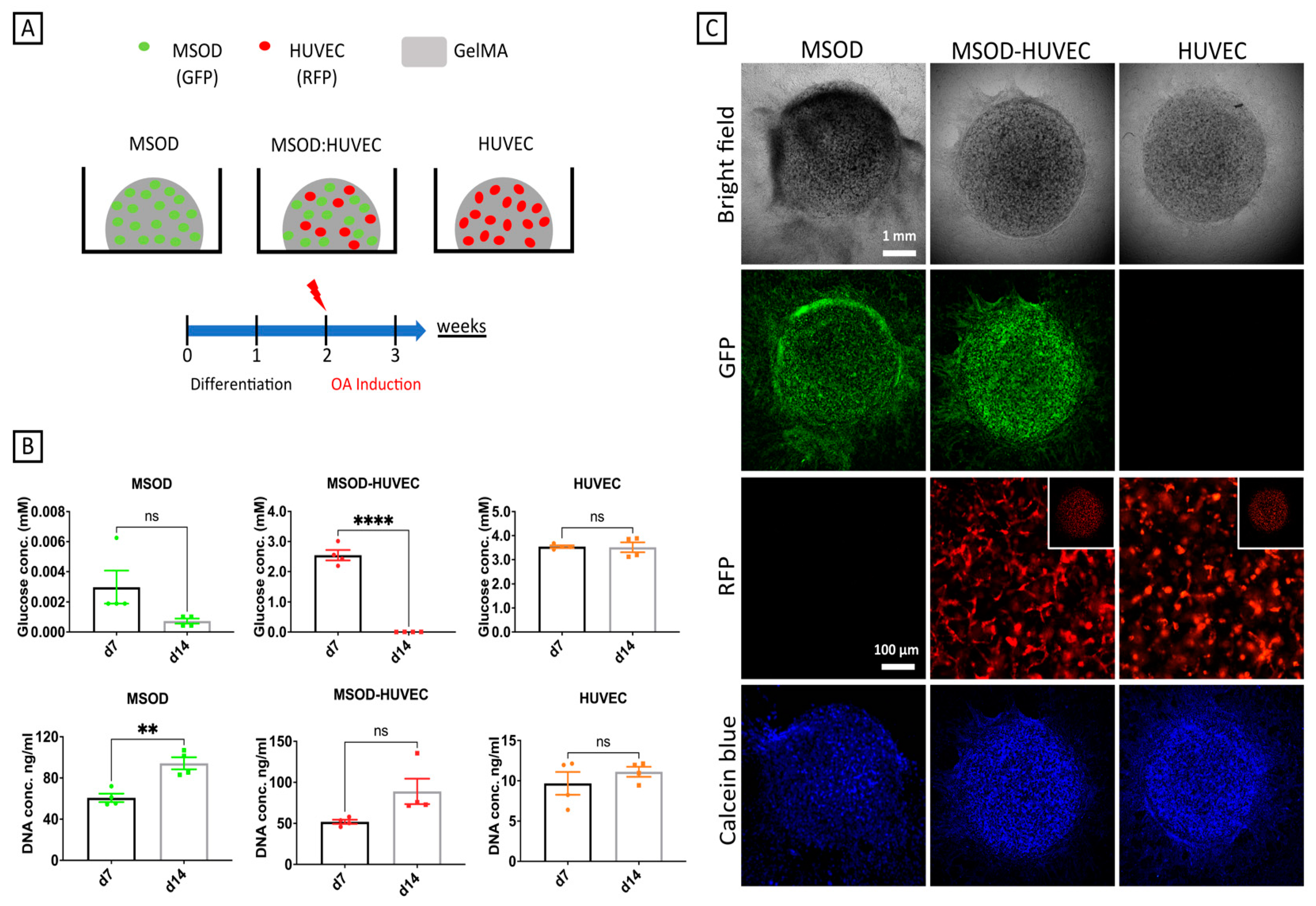
2.1. Generation of 3D Constructs Made of MSODs and HUVECs Co-Cultured in gelMA
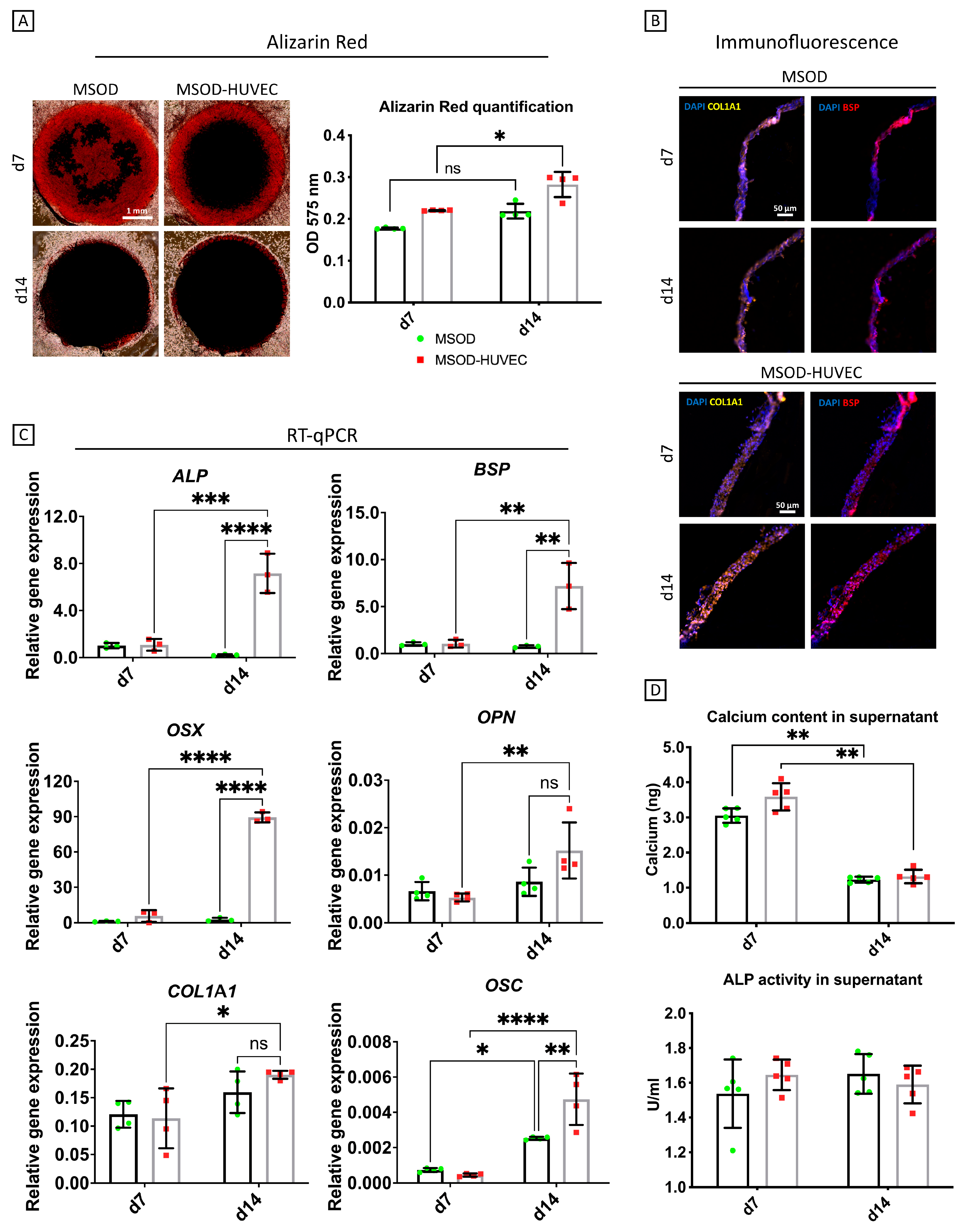
2.2. HUVECs Enhanced Osteogenic Differentiation of MSODs in the Co-Culture System
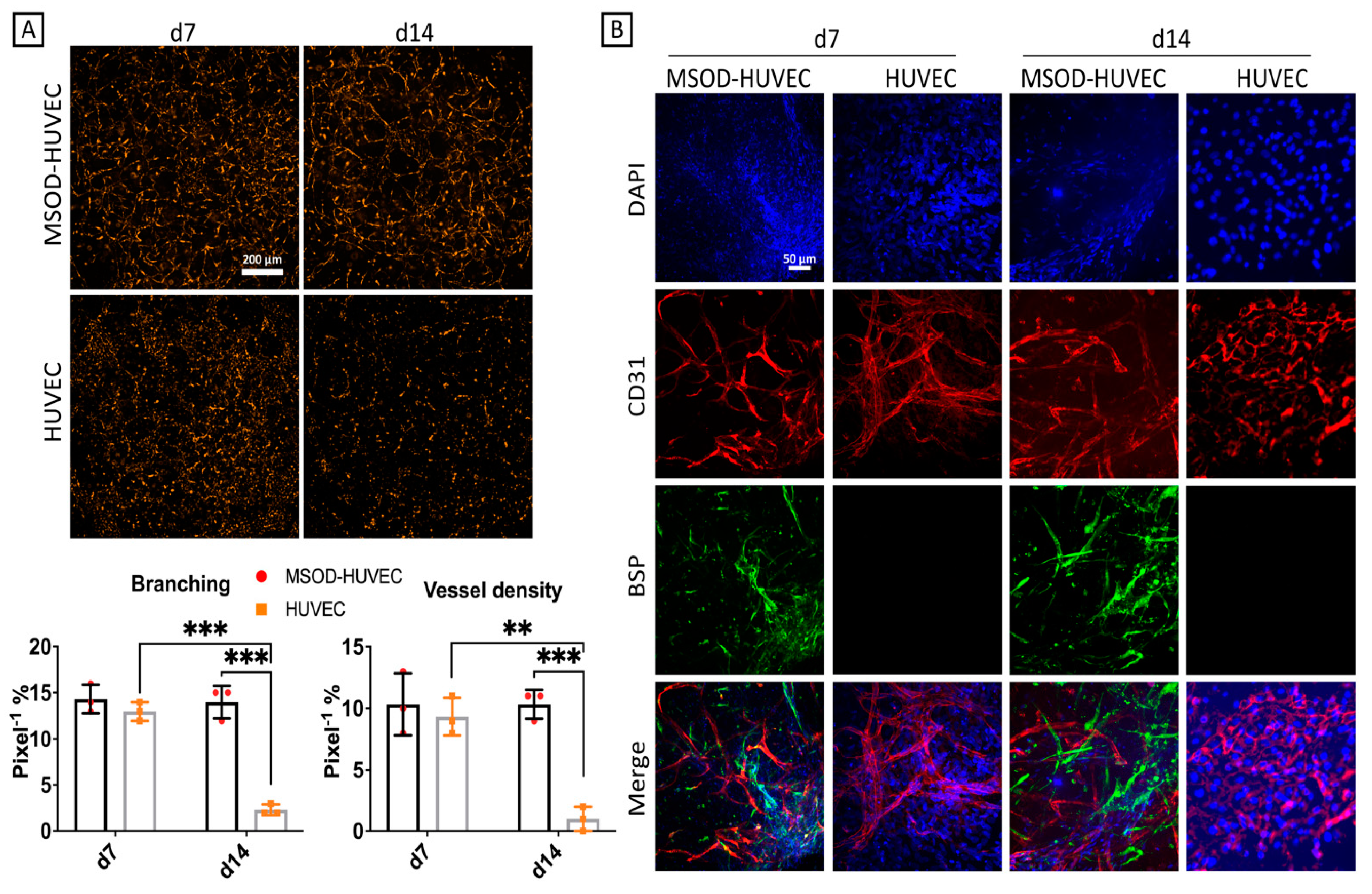
2.3. MSOD Helped Stabilization of the HUVEC Network Formation after 2 Weeks of Culture
2.4. OA Phenotype Induction though Administration of an Inflammatory Cocktail
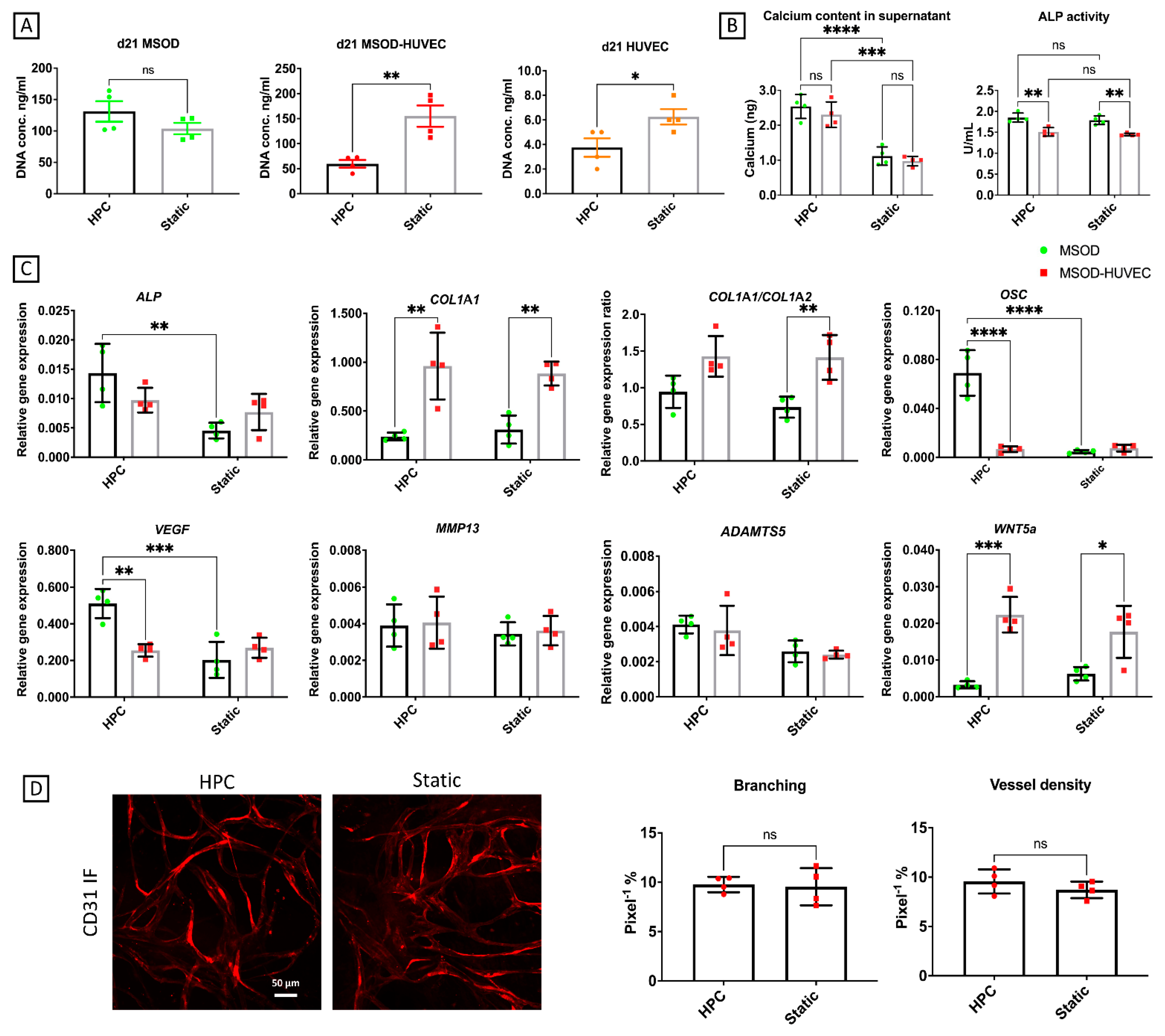
2.5. Effects of Conditioned Medium from OA Cartilage-on-Chip
3. Discussion
4. Materials and Methods
4.1. Generation of the Constructs and OA Induction
4.2. Cell Viability
4.3. Quantitative RT-PCR Analysis
4.4. Alizarin Red Staining and Quantification
4.5. Immunofluorescence
4.6. Quantification of Alkaline Phosphate (ALP) Activity and Calcium Content in the Surnatants
4.7. Angiogenesis Analysis
4.8. OA Cartilage-on-Chip Model Setup
4.9. Microfluidic Devices Fabrication
4.10. Supernatant Analyses
4.11. Statistical Analysis
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Bijlsma, J.W.; Berenbaum, F.; Lafeber, F.P. Osteoarthritis: An update with relevance for clinical practice. Lancet 2011, 377, 2115–2126. [Google Scholar] [CrossRef]
- Kim, J.-R.; Yoo, J.J.; Kim, H.A. Therapeutics in Osteoarthritis Based on an Understanding of Its Molecular Pathogenesis. Int. J. Mol. Sci. 2018, 19, 674. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chen, D.; Shen, J.; Zhao, W.; Wang, T.; Han, L.; Hamilton, J.L.; Im, H.-J. Osteoarthritis: Toward a comprehensive understanding of pathological mechanism. Bone Res. 2017, 5, 16044. [Google Scholar] [CrossRef]
- Neogi, T. Clinical significance of bone changes in osteoarthritis. Ther. Adv. Musculoskelet. Dis. 2012, 4, 259–267. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malfait, A. Osteoarthritis year in review 2015: Biology. Osteoarthr. Cartil. 2016, 24, 21–26. [Google Scholar] [CrossRef] [Green Version]
- Findlay, D.M.; Kuliwaba, J.S. Bone–cartilage crosstalk: A conversation for understanding osteoarthritis. Bone Res. 2016, 4, 16028. [Google Scholar] [CrossRef] [Green Version]
- Felson, D.T.; Neogi, T. Osteoarthritis: Is it a disease of cartilage or of bone? Arthritis Rheum. 2004, 50, 341–344. [Google Scholar] [CrossRef]
- Li, G.; Yin, J.; Gao, J.; Cheng, T.S.; Pavlos, N.J.; Zhang, C.; Zheng, M.H. Subchondral bone in osteoarthritis: Insight into risk factors and microstructural changes. Arthritis Res. Ther. 2013, 15, 223. [Google Scholar] [CrossRef] [Green Version]
- Bianco, D.; Todorov, A.; Čengić, T.; Pagenstert, G.; Schären, S.; Netzer, C.; Hügle, T.; Geurts, J. Alterations of Subchondral Bone Progenitor Cells in Human Knee and Hip Osteoarthritis Lead to a Bone Sclerosis Phenotype. Int. J. Mol. Sci. 2018, 19, 475. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Chen, G.; Li, Y.-P. TGF-β and BMP signaling in osteoblast, skeletal development, and bone formation, homeostasis and disease. Bone Res. 2016, 4, 16009. [Google Scholar] [CrossRef]
- Sun, K.; Luo, J.; Guo, J.; Yao, X.; Jing, X.; Guo, F. The PI3K/AKT/mTOR signaling pathway in osteoarthritis: A narrative review. Osteoarthr. Cartil. 2020, 28, 400–409. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Fan, X.; Xing, L.; Tian, F. Wnt signaling: A promising target for osteoarthritis therapy. Cell Commun. Signal. 2019, 17, 97. [Google Scholar] [CrossRef] [Green Version]
- Lories, R.J.; Luyten, F.P. The bone–cartilage unit in osteoarthritis. Nat. Rev. Rheumatol. 2010, 7, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Dicarlo, E.F.; Kahn, L.B. Inflammatory diseases of the bones and joints. Semin. Diagn. Pathol. 2011, 28, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Teichtahl, A.J.; Wluka, A.; Wijethilake, P.; Wang, Y.; Ghasem-Zadeh, A.; Cicuttini, F.M. Wolff’s law in action: A mechanism for early knee osteoarthritis. Arthritis Res. Ther. 2015, 17, 207. [Google Scholar] [CrossRef] [Green Version]
- Su, W.; Liu, G.; Liu, X.; Zhou, Y.; Sun, Q.; Zhen, G.; Wang, X.; Hu, Y.; Gao, P.; Demehri, S.; et al. Angiogenesis stimulated by elevated PDGF-BB in subchondral bone contributes to osteoarthritis development. JCI Insight 2020, 5, e135446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Malfait, A.-M.; Little, C.B. On the predictive utility of animal models of osteoarthritis. Arthritis Res. Ther. 2015, 17, 225. [Google Scholar] [CrossRef] [Green Version]
- Karsdal, M.; Michaelis, M.; Ladel, C.; Siebuhr, A.; Bihlet, A.; Andersen, J.; Guehring, H.; Christiansen, C.; Bay-Jensen, A.; Kraus, V. Disease-modifying treatments for osteoarthritis (DMOADs) of the knee and hip: Lessons learned from failures and opportunities for the future. Osteoarthr. Cartil. 2016, 24, 2013–2021. [Google Scholar] [CrossRef]
- Samvelyan, H.J.; Hughes, D.; Stevens, C.; Staines, K.A. Models of Osteoarthritis: Relevance and New Insights. Calcif. Tissue Int. 2021, 109, 243–256. [Google Scholar] [CrossRef] [Green Version]
- Occhetta, P.; Mainardi, A.; Votta, E.; Vallmajo-Martin, Q.; Ehrbar, M.; Martin, I.; Barbero, A.; Rasponi, M. Hyperphysiological compression of articular cartilage induces an osteoarthritic phenotype in a cartilage-on-a-chip model. Nat. Biomed. Eng. 2019, 3, 545–557. [Google Scholar] [CrossRef]
- Lozito, T.P.; Alexander, P.G.; Lin, H.; Gottardi, R.; Cheng, A.W.-M.; Tuan, R.S. Three-dimensional osteochondral microtissue to model pathogenesis of osteoarthritis. Stem Cell Res. Ther. 2013, 4, S6. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lin, H.; Lozito, T.P.; Alexander, P.G.; Gottardi, R.; Tuan, R.S. Stem Cell-Based Microphysiological Osteochondral System to Model Tissue Response to Interleukin-1β. Mol. Pharm. 2014, 11, 2203–2212. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pirosa, A.; Gottardi, R.; Alexander, P.G.; Puppi, D.; Chiellini, F.; Tuan, R.S. An in vitro chondro-osteo-vascular triphasic model of the osteochondral complex. Biomaterials 2021, 272, 120773. [Google Scholar] [CrossRef]
- Lin, Z.; Li, Z.; Li, E.N.; Li, X.; Del Duke, C.J.; Shen, H.; Hao, T.; O’Donnell, B.; Bunnell, B.; Goodman, S.B.; et al. Osteochondral Tissue Chip Derived From iPSCs: Modeling OA Pathologies and Testing Drugs. Front. Bioeng. Biotechnol. 2019, 7, 411. [Google Scholar] [CrossRef]
- Caplan, A.I. Mesenchymal Stem Cells: Time to Change the Name! STEM CELLS Transl. Med. 2017, 6, 1445–1451. [Google Scholar] [CrossRef] [Green Version]
- Doss, M.X.; Sachinidis, A. Current Challenges of iPSC-Based Disease Modeling and Therapeutic Implications. Cells 2019, 8, 403. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bourgine, P.; Le Magnen, C.; Pigeot, S.; Geurts, J.; Scherberich, A.; Martin, I. Combination of immortalization and inducible death strategies to generate a human mesenchymal stromal cell line with controlled survival. Stem Cell Res. 2014, 12, 584–598. [Google Scholar] [CrossRef] [Green Version]
- Bourgine, P.E.; Gaudiello, E.; Pippenger, B.; Jaquiery, C.; Klein, T.; Pigeot, S.; Todorov, A.; Feliciano, S.; Banfi, A.; Martin, I. Engi-neered Extracellular Matrices as Biomaterials of Tunable Composition and Function. Adv. Funct. Mater. 2017, 27, 1605486. [Google Scholar] [CrossRef]
- Liu, J.; Chuah, Y.J.; Fu, J.; Zhu, W.; Wang, D.A. Co-culture of human umbilical vein endothelial cells and human bone marrow stromal cells into a micro-cavitary gelatin-methacrylate hydrogel system to enhance angiogenesis. Mater. Sci. Eng. C Mater. Biol. Appl. 2019, 102, 906–916. [Google Scholar] [CrossRef] [PubMed]
- Han, H.-W.; Hou, Y.-T.; Hsu, S.-H. Angiogenic potential of co-spheroids of neural stem cells and endothelial cells in injectable gelatin-based hydrogel. Mater. Sci. Eng. C 2019, 99, 140–149. [Google Scholar] [CrossRef]
- Bale, S.S.; Golberg, I.; Jindal, R.; McCarty, W.; Luitje, M.; Hegde, M.; Bhushan, A.; Usta, O.B.; Yarmush, M.L. Long-Term Coculture Strategies for Primary Hepatocytes and Liver Sinusoidal Endothelial Cells. Tissue Eng. Part C Methods 2015, 21, 413–422. [Google Scholar] [CrossRef] [Green Version]
- Miron, R.J.; Zhang, Y.F. Osteoinduction: A review of old concepts with new standards. J. Dent. Res. 2012, 91, 736–744. [Google Scholar] [CrossRef]
- Tanikake, Y.; Akahane, M.; Furukawa, A.; Tohma, Y.; Inagaki, Y.; Kira, T.; Tanaka, Y. Calcium Concentration in Culture Medium as a Nondestructive and Rapid Marker of Osteogenesis. Cell Transplant. 2017, 26, 1067–1076. [Google Scholar] [CrossRef] [Green Version]
- Blocki, A.; Wang, Y.; Koch, M.K.; Peh, P.; Beyer, S.; Law, P.; Hui, J.; Raghunath, M. Not All MSCs Can Act as Pericytes: Functional In Vitro Assays to Distinguish Pericytes from Other Mesenchymal Stem Cells in Angiogenesis. Stem Cells Dev. 2013, 22, 2347–2355. [Google Scholar] [CrossRef] [Green Version]
- Hoff, P.; Buttgereit, F.; Burmester, G.-R.; Jakstadt, M.; Gaber, T.; Andreas, K.; Matziolis, G.; Perka, C.; Röhner, E. Osteoarthritis synovial fluid activates pro-inflammatory cytokines in primary human chondrocytes. Int. Orthop. 2013, 37, 145–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Walsh, D.A.; Bonnet, C.S.; Turner, E.L.; Wilson, D.; Situ, M.; McWilliams, D.F. Angiogenesis in the synovium and at the oste-ochondral junction in osteoarthritis. Osteoarthr. Cartil. 2007, 15, 743–751. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, H.-M.; Lee, J.-H.; Lee, Y.-J. Positive Association of Serum Alkaline Phosphatase Level with Severe Knee Osteoarthritis: A Nationwide Population-Based Study. Diagnostics 2020, 10, 1016. [Google Scholar] [CrossRef]
- Hamilton, J.L.; Nagao, M.; Levine, B.R.; Chen, D.; Olsen, B.R.; Im, H.-J. Targeting VEGF and Its Receptors for the Treatment of Osteoarthritis and Associated Pain. J. Bone Miner. Res. 2016, 31, 911–924. [Google Scholar] [CrossRef] [PubMed]
- Maruotti, N.; Corrado, A.; Cantatore, F.P. Osteoblast role in osteoarthritis pathogenesis. J. Cell. Physiol. 2017, 232, 2957–2963. [Google Scholar] [CrossRef] [Green Version]
- Takahashi, A.; de Andrés, M.; Hashimoto, K.; Itoi, E.; Oreffo, R. Epigenetic regulation of interleukin-8, an inflammatory chemokine, in osteoarthritis. Osteoarthr. Cartil. 2015, 23, 1946–1954. [Google Scholar] [CrossRef] [Green Version]
- Pirosa, A.; Gottardi, R.; Alexander, P.G.; Tuan, R.S. Engineering in-vitro stem cell-based vascularized bone models for drug screening and predictive toxicology. Stem Cell Res. Ther. 2018, 9, 112. [Google Scholar] [CrossRef] [Green Version]
- Blache, U.; Metzger, S.; Vallmajo-Martin, Q.; Martin, I.; Djonov, V.; Ehrbar, M. Dual Role of Mesenchymal Stem Cells Allows for Microvascularized Bone Tissue-Like Environments in PEG Hydrogels. Adv. Health Mater. 2016, 5, 489–498. [Google Scholar] [CrossRef]
- Liu, Y.; Chan, J.K.Y.; Teoh, S.-H. Review of vascularised bone tissue-engineering strategies with a focus on co-culture systems. J. Tissue Eng. Regen. Med. 2012, 9, 85–105. [Google Scholar] [CrossRef]
- Mills, S.J.; Cowin, A.J.; Kaur, P. Pericytes, Mesenchymal Stem Cells and the Wound Healing Process. Cells 2013, 2, 621–634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Couchourel, D.; Aubry, I.; Delalandre, A.; Lavigne, M.; Martel-Pelletier, J.; Pelletier, J.-P.; Lajeunesse, D. Altered mineralization of human osteoarthritic osteoblasts is attributable to abnormal type I collagen production. Arthritis Rheum. 2009, 60, 1438–1450. [Google Scholar] [CrossRef] [PubMed]
- Clarke, J. PDGF-BB is the key to unlocking pathological angiogenesis in OA. Nat. Rev. Rheumatol. 2020, 16, 298. [Google Scholar] [CrossRef] [Green Version]
- Mapp, P.I.; Walsh, D. Mechanisms and targets of angiogenesis and nerve growth in osteoarthritis. Nat. Rev. Rheumatol. 2012, 8, 390–398. [Google Scholar] [CrossRef] [PubMed]
- Chua, J.R.; Pincus, T.; Castrejon, I.; Gibson, K.; Block, J.A. Secondary Osteoarthritis in Patients with Rheumatoid Arthritis Ap-pears as Severe as Inflammation According to Physician Visual Analog Scales, Regardless of Disease Severity. Osteoarthr. Cartil. 2017, 25, S220–S221. [Google Scholar] [CrossRef] [Green Version]
- Costa, C.; Incio, J.; Soares, R. Angiogenesis and chronic inflammation: Cause or consequence? Angiogenesis 2007, 10, 149–166. [Google Scholar] [CrossRef] [PubMed]
- Mohr, T.; Haudek-Prinz, V.; Slany, A.; Grillari, J.; Micksche, M.; Gerner, C. Proteome profiling in IL-1β and VEGF-activated human umbilical vein endothelial cells delineates the interlink between inflammation and angiogenesis. PLoS ONE 2017, 12, e0179065. [Google Scholar] [CrossRef] [Green Version]
- Carpentier, G.; Berndt, S.; Ferratge, S.; Rasband, W.; Cuendet, M.; Uzan, G.; Albanese, P. Angiogenesis Analyzer for ImageJ—A comparative morphometric analysis of “Endothelial Tube Formation Assay” and “Fibrin Bead Assay”. Sci. Rep. 2020, 10, 11568. [Google Scholar] [CrossRef] [PubMed]
- Ehrbar, M.; Rizzi, S.C.; Schoenmakers, R.G.; San Miguel, B.; Hubbell, J.A.; Weber, F.E.; Lutolf, M.P. Biomolecular Hydrogels Formed and Degraded via Site-Specific Enzymatic Reactions. Biomacromolecules 2007, 8, 3000–3007. [Google Scholar] [CrossRef] [PubMed]

| Gene | Assay on Demand Ref. No. |
|---|---|
| GAPDH | Hs02758991_g1 |
| ALP | Hs01029144_m1 |
| OSX | Hs00541729_m1 |
| BSP | Hs00173720_m1 |
| OPN | Hs00959010_m1 |
| COL1A1 | Hs01097664_m1 |
| COL1A2 | Hs01028960_m1 |
| OSC | Hs01587814_g1 |
| VEGF | Hs00900055_m1 |
| WNT5a | Hs00998537_m1 |
| ADAMTS5 | Hs00199841_m1 |
| MMP-13 | Hs00233992_m1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pirosa, A.; Tankus, E.B.; Mainardi, A.; Occhetta, P.; Dönges, L.; Baum, C.; Rasponi, M.; Martin, I.; Barbero, A. Modeling In Vitro Osteoarthritis Phenotypes in a Vascularized Bone Model Based on a Bone-Marrow Derived Mesenchymal Cell Line and Endothelial Cells. Int. J. Mol. Sci. 2021, 22, 9581. https://doi.org/10.3390/ijms22179581
Pirosa A, Tankus EB, Mainardi A, Occhetta P, Dönges L, Baum C, Rasponi M, Martin I, Barbero A. Modeling In Vitro Osteoarthritis Phenotypes in a Vascularized Bone Model Based on a Bone-Marrow Derived Mesenchymal Cell Line and Endothelial Cells. International Journal of Molecular Sciences. 2021; 22(17):9581. https://doi.org/10.3390/ijms22179581
Chicago/Turabian StylePirosa, Alessandro, Esma Bahar Tankus, Andrea Mainardi, Paola Occhetta, Laura Dönges, Cornelia Baum, Marco Rasponi, Ivan Martin, and Andrea Barbero. 2021. "Modeling In Vitro Osteoarthritis Phenotypes in a Vascularized Bone Model Based on a Bone-Marrow Derived Mesenchymal Cell Line and Endothelial Cells" International Journal of Molecular Sciences 22, no. 17: 9581. https://doi.org/10.3390/ijms22179581
APA StylePirosa, A., Tankus, E. B., Mainardi, A., Occhetta, P., Dönges, L., Baum, C., Rasponi, M., Martin, I., & Barbero, A. (2021). Modeling In Vitro Osteoarthritis Phenotypes in a Vascularized Bone Model Based on a Bone-Marrow Derived Mesenchymal Cell Line and Endothelial Cells. International Journal of Molecular Sciences, 22(17), 9581. https://doi.org/10.3390/ijms22179581







