Aluminum Enters Mammalian Cells and Destabilizes Chromosome Structure and Number
Abstract
:1. Introduction
2. Results
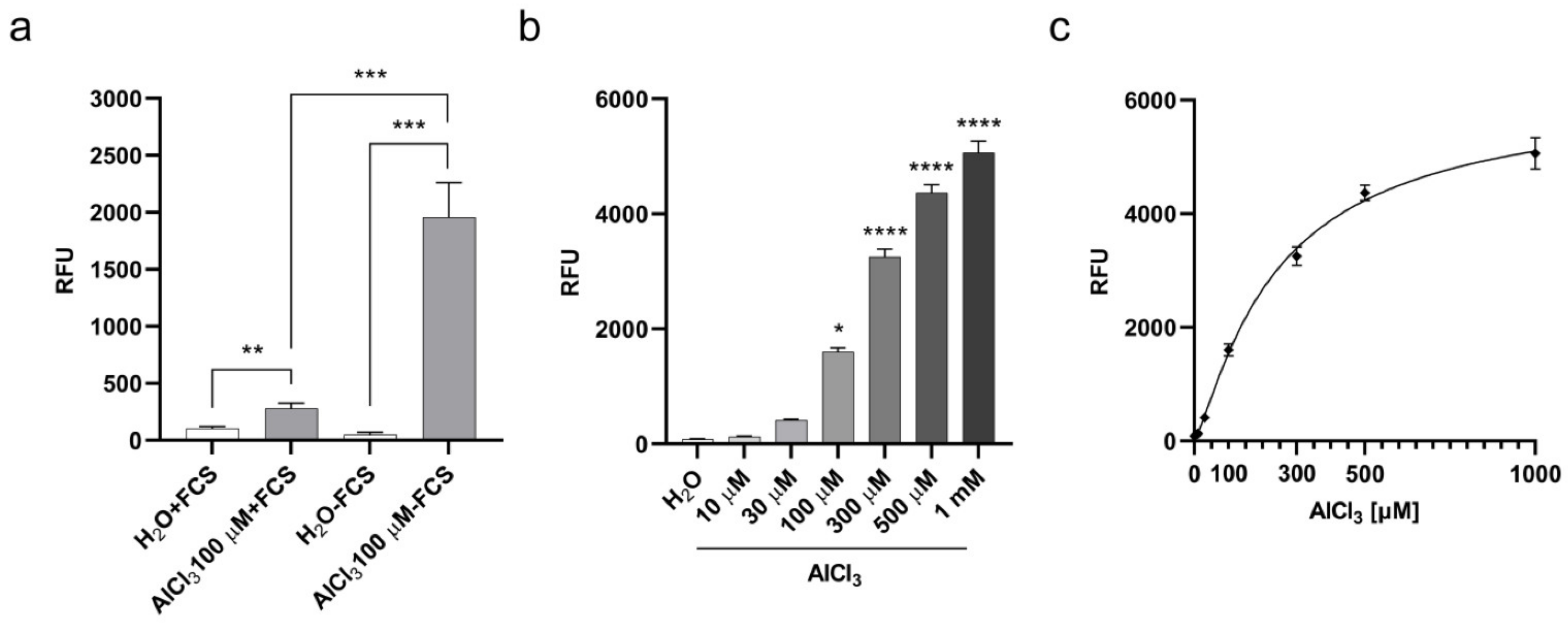
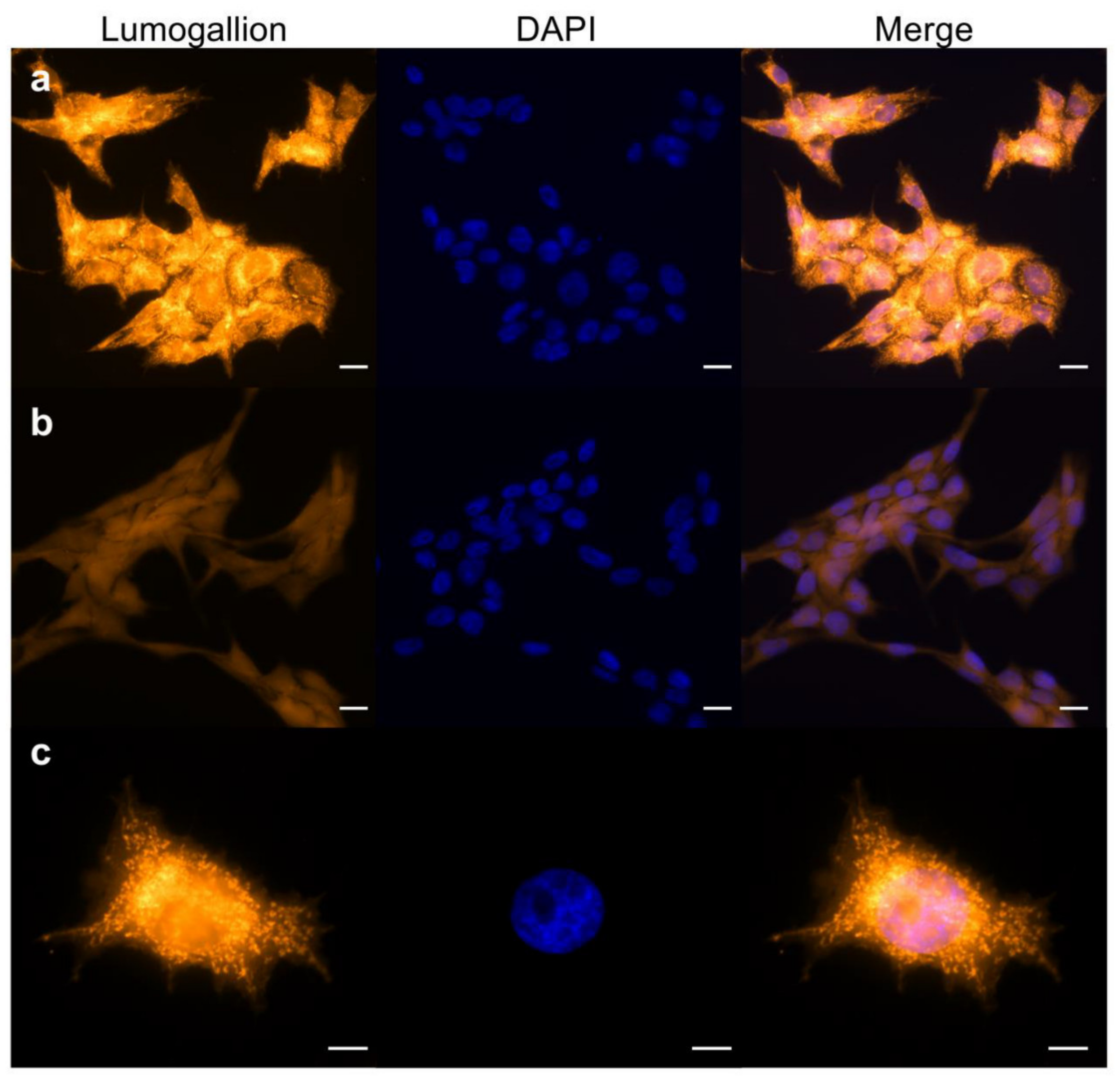
2.1. Aluminum Entry and Accumulation in V79 Cells
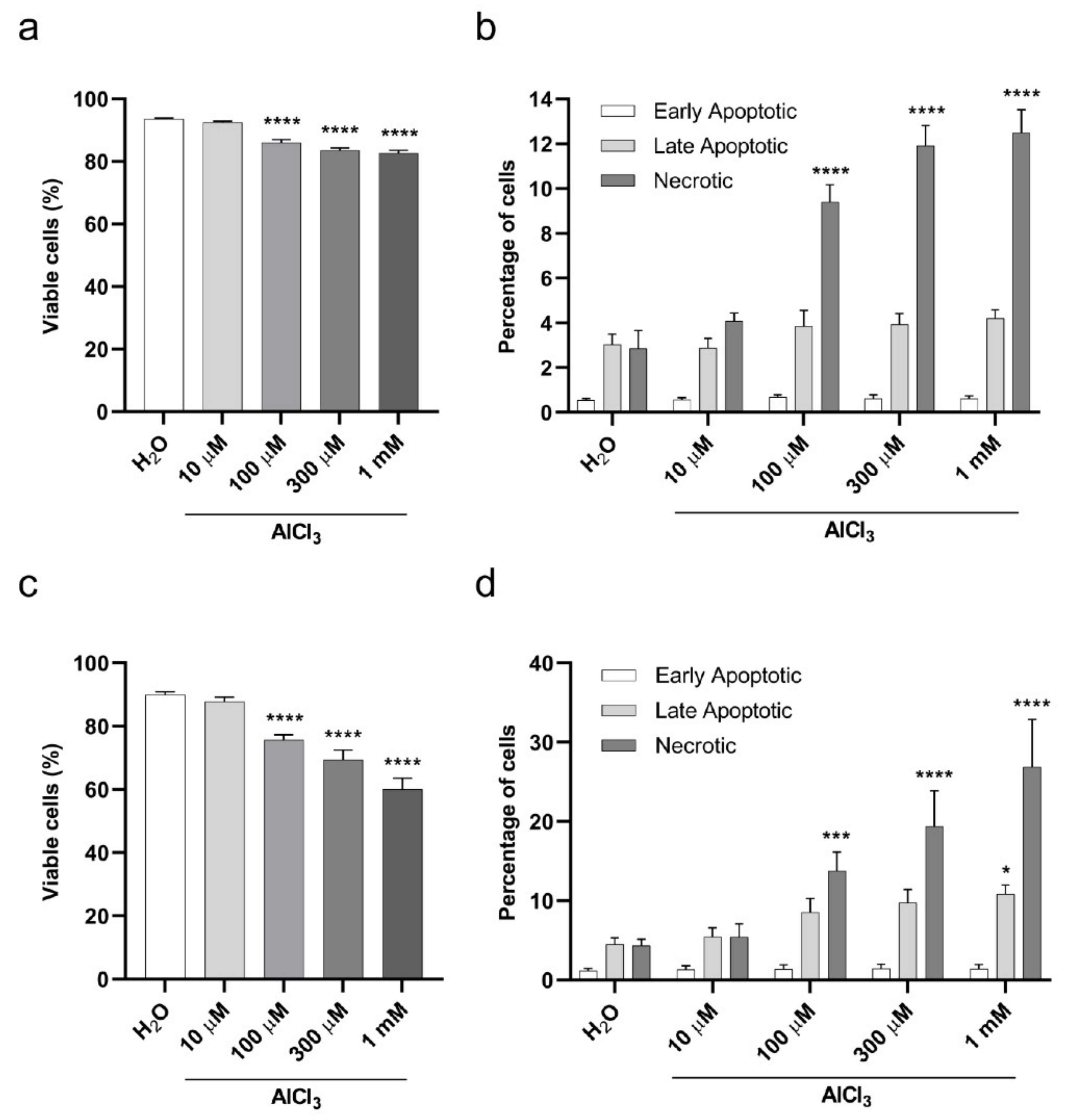
2.2. Viability of V79 Cells Exposed to Aluminum
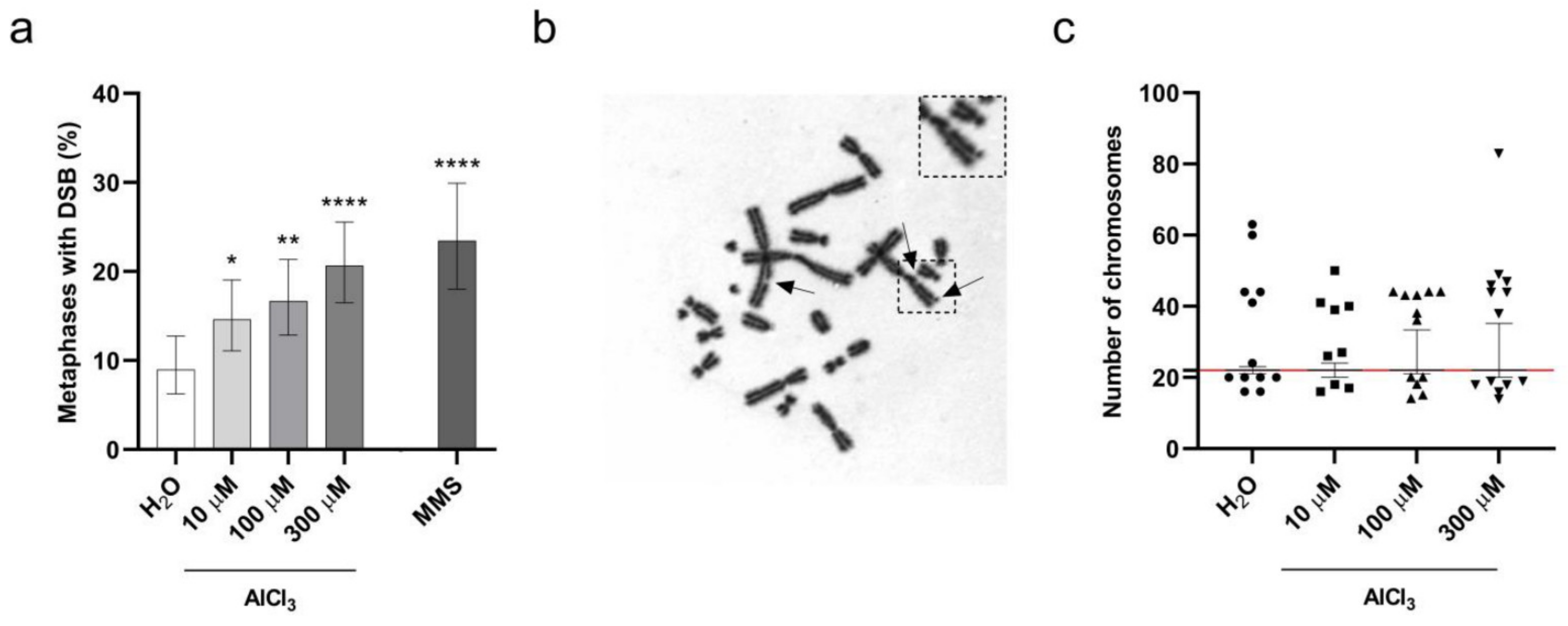
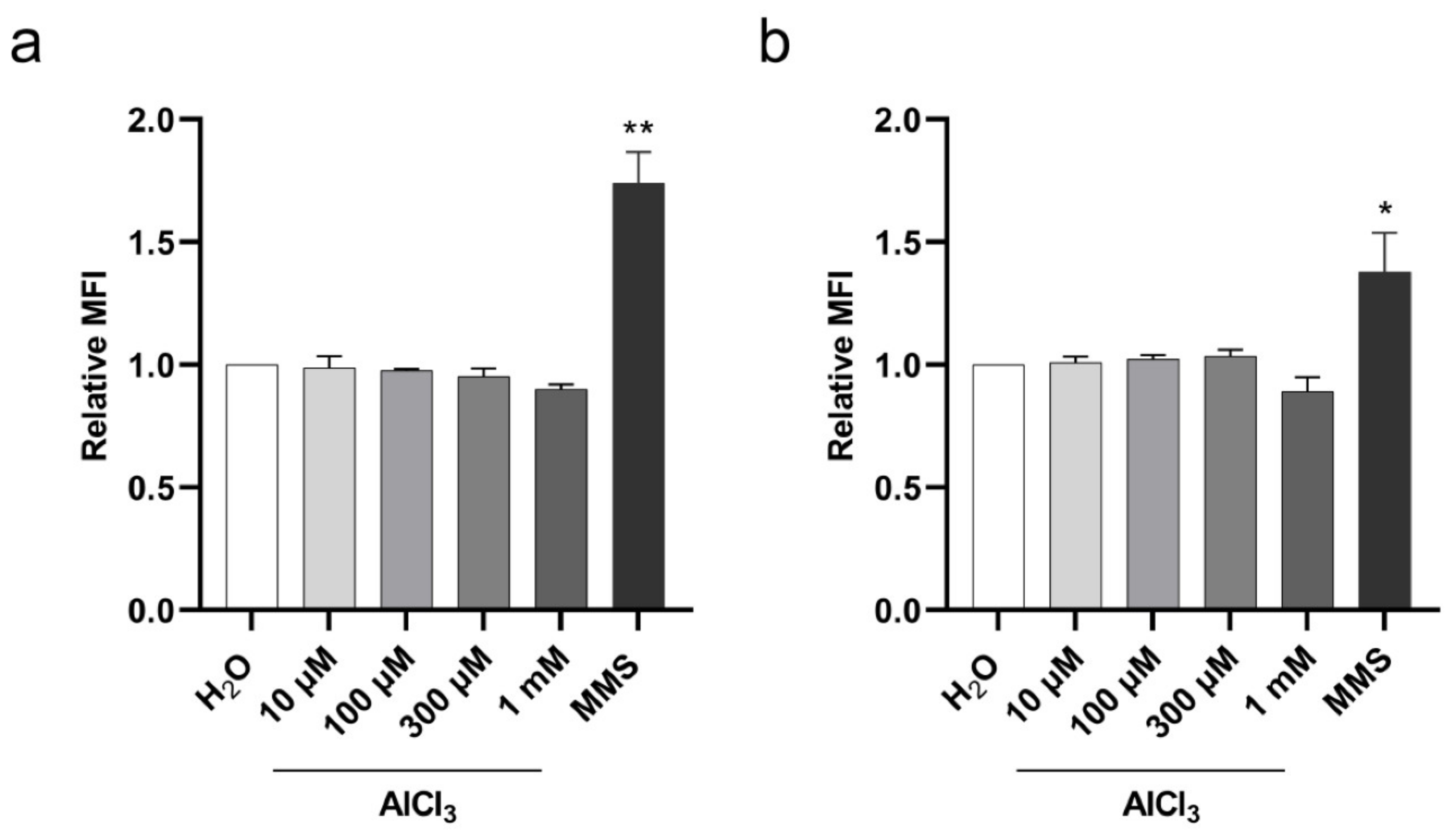
2.3. Aluminum Is a Clastogenic and Aneuploidy Inducing Agent in V79 Cells
2.4. V79 Cells Exposed to Aluminum Arrest in G2/M Phase of the Cell Cycle
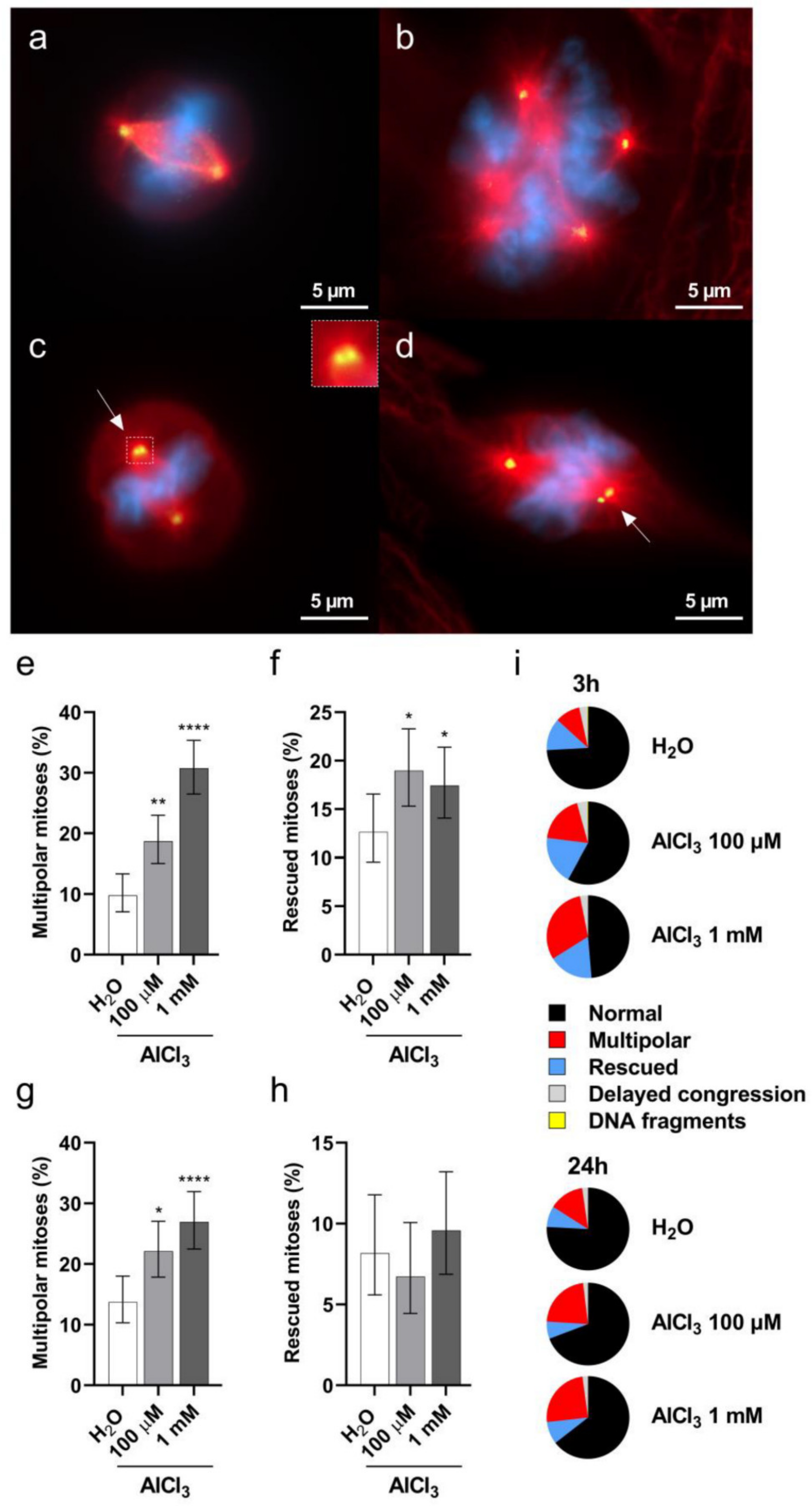
2.5. Aluminum Induces Multipolar Divisions in V79 Cells
3. Discussion
4. Materials and Methods
4.1. Reagents
4.2. Antibodies
4.3. Cell Culture
4.4. Lumogallion Staining
4.5. Cytotoxicity Assay by Annexin-V/PI Staining
4.6. Metaphase Spread
4.7. Cell Cycle Analysis
4.8. γ-H2AX Staining
4.9. Immunofluorescence
4.10. Image Acquisition
4.11. Statistical Analysis
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martinez-Finley, E.J.; Chakraborty, S.; Fretham, S.J.B.; Aschner, M. Cellular transport and homeostasis of essential and nonessential metals. Metallomics 2012, 4, 593–605. [Google Scholar] [CrossRef] [Green Version]
- Valko, M.; Morris, H.; Cronin, M.T.D. Metals, toxicity and oxidative stress. Curr. Med. Chem. 2005, 12, 1161–1208. [Google Scholar] [CrossRef] [Green Version]
- Frezza, M.; Hindo, S.; Chen, D.; Davenport, A.; Schmitt, S.; Tomco, D.; Ping Dou, Q. Novel Metals and Metal Complexes as Platforms for Cancer Therapy. Curr. Pharm. Des. 2010, 16, 1813–1825. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beyersmann, D.; Hartwig, A. Carcinogenic metal compounds: Recent insight into molecular and cellular mechanisms. Arch. Toxicol. 2008, 82, 493–512. [Google Scholar] [CrossRef] [PubMed]
- Jan, A.T.; Azam, M.; Siddiqui, K.; Ali, A.; Choi, I.; Haq, Q.M.R. Heavy metals and human health: Mechanistic insight into toxicity and counter defense system of antioxidants. Int. J. Mol. Sci. 2015, 16, 29592–29630. [Google Scholar] [CrossRef] [Green Version]
- Zhao, Q.; Wang, Y.; Cao, Y.; Chen, A.; Ren, M.; Ge, Y.; Yu, Z.; Wan, S.; Hu, A.; Bo, Q.; et al. Potential health risks of heavy metals in cultivated topsoil and grain, including correlations with human primary liver, lung and gastric cancer, in Anhui province, Eastern China. Sci. Total. Environ. 2014, 470–471, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Exley, C. Human exposure to aluminium. Environ. Sci. Process. Impacts 2013, 15, 1807–1816. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Igbokwe, I.O.; Igwenagu, E.; Igbokwe, N.A. Aluminium toxicosis: A review of toxic actions and effects. Interdiscip. Toxicol. 2020, 12, 45–70. [Google Scholar] [CrossRef] [Green Version]
- Darbre, P.D. Aluminium and the human breast. Morphologie 2016, 100, 65–74. [Google Scholar] [CrossRef]
- Alfrey, A.C.; LeGendre, G.R.; Kaehny, W.D. The dialysis encephalopathy syndrome. Possible aluminum intoxication. N. Engl. J. Med. 1976, 294, 184–188. [Google Scholar] [CrossRef]
- Ellis, H.A.; McCarthy, J.H.; Herrington, J. Bone aluminium in haemodialysed patients and in rats injected with aluminium chloride: Relationship to impaired bone mineralisation. J. Clin. Pathol. 1979, 32, 832–844. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hirsch, E.C.; Brandel, J.P.; Galle, P.; Javoy-Agid, F.; Agid, Y. Iron and aluminum increase in the substantia nigra of patients with Parkinson’s disease: An X-ray microanalysis. J. Neurochem. 1991, 56, 446–451. [Google Scholar] [CrossRef]
- Kawahara, M.; Kato-Negishi, M. Link between aluminum and the pathogenesis of Alzheimer’s disease: The integration of the aluminum and amyloid cascade hypotheses. Int. J. Alzheimers. Dis. 2011, 2011. [Google Scholar] [CrossRef] [Green Version]
- Yasui, M.; Kihira, T.; Ota, K. Calcium, magnesium and aluminum concentrations in Parkinson’s disease. Neurotoxicology 1992, 13, 593–600. [Google Scholar]
- Darbre, P.D. Underarm cosmetics are a cause of breast cancer. Eur. J. Cancer Prev. 2001, 10, 389–393. [Google Scholar] [CrossRef]
- McGrath, K.G. An earlier age of breast cancer diagnosis related to more frequent use of antiperspirants/deodorants and underarm shaving. Eur. J. Cancer Prev. 2003, 12, 479–485. [Google Scholar] [CrossRef]
- Linhart, C.; Talasz, H.; Morandi, E.M.; Exley, C.; Lindner, H.H.; Taucher, S.; Egle, D.; Hubalek, M.; Concin, N.; Ulmer, H. Use of Underarm Cosmetic Products in Relation to Risk of Breast Cancer: A Case-Control Study. EBioMedicine 2017, 21, 79–85. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mandriota, S.J.; Tenan, M.; Ferrari, P.; Sappino, A.P. Aluminium chloride promotes tumorigenesis and metastasis in normal murine mammary gland epithelial cells. Int. J. Cancer 2016, 139, 2781–2790. [Google Scholar] [CrossRef]
- Mandriota, S.J.; Tenan, M.; Nicolle, A.; Jankowska, J.D.; Ferrari, P.; Tille, J.C.; Durin, M.A.; Green, C.M.; Tabruyn, S.; Moralli, D.; et al. Genomic instability is an early event in aluminium-induced tumorigenesis. Int. J. Mol. Sci. 2020, 21, 9332. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, S.P.; Vijayalaxmi, K.K.; Naik, P. Assessment of genotoxicity of aluminium acetate in bone marrow, male germ cells and fetal liver cells of Swiss albino mice. Mutat. Res.-Genet. Toxicol. Environ. Mutagen. 2014, 766, 16–22. [Google Scholar] [CrossRef] [PubMed]
- García-Alegría, A.M.; Gómez-Álvarez, A.; Anduro-Corona, I.; Burgos-Hernández, A.; Ruíz-Bustos, E.; Canett-Romero, R.; González-Ríos, H.; López-Cervantes, J.G.; Rodríguez-Martínez, K.L.; Astiazaran-Garcia, H. Genotoxic Effects of Aluminum Chloride and Their Relationship with N-Nitroso-N-Methylurea (NMU)-Induced Breast Cancer in Sprague Dawley Rats. Toxics 2020, 8, 31. [Google Scholar] [CrossRef] [PubMed]
- Lima, P.D.L.; Leite, D.S.; Vasconcellos, M.C.; Cavalcanti, B.C.; Santos, R.A.; Costa-Lotufo, L.V.; Pessoa, C.; Moraes, M.O.; Burbano, R.R. Genotoxic effects of aluminum chloride in cultured human lymphocytes treated in different phases of cell cycle. Food Chem. Toxicol. 2007, 45, 1154–1159. [Google Scholar] [CrossRef] [PubMed]
- Paz, L.N.F.; Moura, L.M.; Feio, D.C.A.; Cardoso, M.D.S.G.; Ximenes, W.L.O.; Montenegro, R.C.; Alves, A.P.N.; Burbano, R.R.; Lima, P.D.L. Evaluation of in vivo and in vitro toxicological and genotoxic potential of aluminum chloride. Chemosphere 2017, 175, 130–137. [Google Scholar] [CrossRef]
- Lee, J.K.; Choi, Y.L.; Kwon, M.; Park, P.J. Mechanisms and Consequences of Cancer Genome Instability: Lessons from Genome Sequencing Studies. Annu. Rev. Pathol. Mech. Dis. 2016, 11, 283–312. [Google Scholar] [CrossRef]
- Kou, F.; Wu, L.; Ren, X.; Yang, L. Chromosome Abnormalities: New Insights into Their Clinical Significance in Cancer. Mol. Ther. Oncolytics 2020, 17, 562–570. [Google Scholar] [CrossRef]
- McGranahan, N.; Burrell, R.A.; Endesfelder, D.; Novelli, M.R.; Swanton, C. Cancer chromosomal instability: Therapeutic and diagnostic challenges. EMBO Rep. 2012, 13, 528–538. [Google Scholar] [CrossRef] [Green Version]
- Ganem, N.J.; Godinho, S.A.; Pellman, D. A mechanism linking extra centrosomes to chromosomal instability. Nature 2009, 460, 278–282. [Google Scholar] [CrossRef] [Green Version]
- Mirza, A.; King, A.; Troakes, C.; Exley, C. The Identification of Aluminum in Human Brain Tissue Using Lumogallion and Fluorescence Microscopy. J. Alzheimer’s Dis. 2016, 54, 1333–1338. [Google Scholar] [CrossRef] [Green Version]
- Mujika, J.I.; Lopez, X.; Rezabal, E.; Castillo, R.; Marti, S.; Moliner, V.; Ugalde, J.M. A QM/MM study of the complexes formed by aluminum and iron with serum transferrin at neutral and acidic pH. J. Inorg. Biochem. 2011, 105, 1446–1456. [Google Scholar] [CrossRef]
- OECD. Test No. 473: In Vitro Mammalian Chromosomal Aberration Test. In OECD Guidelines for the Testing of Chemicals, Section 4; OECD: Paris, France, 2016; ISBN 9789264264649. [Google Scholar]
- Fujikawa-Yamamoto, K.; Miyashita, N.; Odashima, S. Flow cytometric determination of cell cycle parameters of V79 cells by continuous labeling with bromodeoxyuridine. Cell Struct. Funct. 1986, 11, 115–124. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Paulovich, A.G.; Toczyski, D.P.; Hartwell, L.H. When checkpoints fail. Cell 1997, 88, 315–321. [Google Scholar] [CrossRef] [Green Version]
- Chaung, W.; Mi, L.J.; Boorstein, R.J. The p53 Status of Chinese Hamster V79 Cells Frequently Used for Studies on DNA Damage and DNA Repair. Nucleic Acids Res. 1997, 25, 992–994. [Google Scholar] [CrossRef] [PubMed]
- Rogakou, E.P.; Pilch, D.R.; Orr, A.H.; Ivanova, V.S.; Bonner, W.M. DNA double-stranded breaks induce histone H2AX phosphorylation on serine 139. J. Biol. Chem. 1998, 273, 5858–5868. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ichijima, Y.; Sakasai, R.; Okita, N.; Asahina, K.; Mizutani, S.; Teraoka, H. Phosphorylation of histone H2AX at M phase in human cells without DNA damage response. Biochem. Biophys. Res. Commun. 2005, 336, 807–812. [Google Scholar] [CrossRef] [PubMed]
- Dodson, H.; Bourke, E.; Jeffers, L.J.; Vagnarelli, P.; Sonoda, E.; Takeda, S.; Earnshaw, W.C.; Merdes, A.; Morrison, C. Centrosome amplification induced by DNA damage occurs during a prolonged G2 phase and involves ATM. EMBO J. 2004, 23, 3864–3873. [Google Scholar] [CrossRef] [Green Version]
- Janssen, A.; Van Der Burg, M.; Szuhai, K.; Kops, G.J.P.L.; Medema, R.H. Chromosome segregation errors as a cause of DNA damage and structural chromosome aberrations. Science 2011, 333, 1895–1898. [Google Scholar] [CrossRef]
- Sappino, A.P.; Buser, R.; Lesne, L.; Gimelli, S.; Béna, F.; Belin, D.; Mandriota, S.J. Aluminium chloride promotes anchorage-independent growth in human mammary epithelial cells. J. Appl. Toxicol. 2012, 32, 233–243. [Google Scholar] [CrossRef] [Green Version]
- Nagasawa, K.; Ito, S.; Kakuda, T.; Nagai, K.; Tamai, I.; Tsuji, A.; Fujimoto, S. Transport mechanism for aluminum citrate at the blood-brain barrier: Kinetic evidence implies involvement of system Xc–in immortalized rat brain endothelial cells. Toxicol. Lett. 2005, 155, 289–296. [Google Scholar] [CrossRef]
- Mile, I.; Svensson, A.; Darabi, A.; Mold, M.; Siesjö, P.; Eriksson, H. Al adjuvants can be tracked in viable cells by lumogallion staining. J. Immunol. Methods 2015, 422, 87–94. [Google Scholar] [CrossRef]
- Exley, C.; Mold, M.J. The binding, transport and fate of aluminium in biological cells. J. Trace Elem. Med. Biol. 2015, 30, 90–95. [Google Scholar] [CrossRef]
- Chen, F.; Shi, X. Intracellular signal transduction of cells in response to carcinogenic metals. Crit. Rev. Oncol. Hematol. 2002, 42, 105–121. [Google Scholar] [CrossRef]
- Smith, M.T.; Guyton, K.Z.; Gibbons, C.F.; Fritz, J.M.; Portier, C.J.; Rusyn, I.; DeMarini, D.M.; Caldwell, J.C.; Kavlock, R.J.; Lambert, P.F.; et al. Key characteristics of carcinogens as a basis for organizing data on mechanisms of carcinogenesis. Environ. Health Perspect. 2016, 124, 713–721. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maya, S.; Prakash, T.; Madhu, K.D.; Goli, D. Multifaceted effects of aluminium in neurodegenerative diseases: A review. Biomed. Pharmacother. 2016, 83, 746–754. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Gill, K.D. Oxidative stress and mitochondrial dysfunction in aluminium neurotoxicity and its amelioration: A review. Neurotoxicology 2014, 41, 154–166. [Google Scholar] [CrossRef]
- Willhite, C.C.; Karyakina, N.A.; Yokel, R.A.; Yenugadhati, N.; Wisniewski, T.M.; Arnold, I.M.F.; Momoli, F.; Krewski, D. Systematic review of potential health risks posed by pharmaceutical, occupational and consumer exposures to metallic and nanoscale aluminum, aluminum oxides, aluminum hydroxide and its soluble salts. Crit. Rev. Toxicol. 2014, 44, 1–80. [Google Scholar] [CrossRef] [PubMed]
- Kasparek, T.R.; Humphrey, T.C. DNA double-strand break repair pathways, chromosomal rearrangements and cancer. Semin. Cell Dev. Biol. 2011, 22, 886–897. [Google Scholar] [CrossRef]
- Wilhelm, T.; Olziersky, A.M.; Harry, D.; De Sousa, F.; Vassal, H.; Eskat, A.; Meraldi, P. Mild replication stress causes chromosome mis-segregation via premature centriole disengagement. Nat. Commun. 2019, 10, 1–14. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Godinho, S.A.; Kwon, M.; Pellman, D. Centrosomes and cancer: How cancer cells divide with too many centrosomes. Cancer Metastasis Rev. 2009, 28, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Dovgalyuk, A.; Kalynyak, T.; Blume, Y.B. Heavy metals have a different action from aluminium in disrupting microtubules in Allium cepa meristematic cells. Cell Biol. Int. 2003, 27, 193–195. [Google Scholar] [CrossRef]
- Macdonald, T.; Humphreys, W.; Martin, R. Promotion of tubulin assembly by aluminum ion in vitro. Science 1987, 236, 183–186. [Google Scholar] [CrossRef]
- Asteriti, I.A.; Giubettini, M.; Lavia, P.; Guarguaglini, G. Aurora-A inactivation causes mitotic spindle pole fragmentation by unbalancing microtubule-generated forces. Mol. Cancer 2011, 10, 131. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bakhoum, S.F.; Thompson, S.L.; Manning, A.L.; Compton, D.A. Genome stability is ensured by temporal control of kinetochore-microtubule dynamics. Nat. Cell Biol. 2009, 11, 27–35. [Google Scholar] [CrossRef] [Green Version]
- Ly, P.; Brunner, S.F.; Shoshani, O.; Kim, D.H.; Lan, W.; Pyntikova, T.; Flanagan, A.M.; Behjati, S.; Page, D.C.; Campbell, P.J.; et al. Chromosome segregation errors generate a diverse spectrum of simple and complex genomic rearrangements. Nat. Genet. 2019, 51, 705–715. [Google Scholar] [CrossRef] [PubMed]
- Santaguida, S.; Richardson, A.; Iyer, D.R.; M’Saad, O.; Zasadil, L.; Knouse, K.A.; Wong, Y.L.; Rhind, N.; Desai, A.; Amon, A. Chromosome Mis-segregation Generates Cell-Cycle-Arrested Cells with Complex Karyotypes that Are Eliminated by the Immune System. Dev. Cell 2017, 41, 638–651.e5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mittal, K.; Kaur, J.; Jaczko, M.; Wei, G.; Toss, M.S.; Rakha, E.A.; Janssen, E.A.M.; Søiland, H.; Kucuk, O.; Reid, M.D.; et al. Centrosome amplification: A quantifiable cancer cell trait with prognostic value in solid malignancies. Cancer Metastasis Rev. 2021, 40, 319–339. [Google Scholar] [CrossRef]
- Bloomfield, M.; Duesberg, P. Inherent variability of cancer-specific aneuploidy generates metastases. Mol. Cytogenet. 2016, 9, 1–22. [Google Scholar] [CrossRef] [Green Version]
- Holland, A.J.; Cleveland, D.W. Losing balance: The origin and impact of aneuploidy in cancer. EMBO Rep. 2012, 13, 501–514. [Google Scholar] [CrossRef] [Green Version]
- Sansregret, L.; Swanton, C. The role of aneuploidy in cancer evolution. Cold Spring Harb. Perspect. Med. 2017, 7, 1–17. [Google Scholar] [CrossRef] [Green Version]
- Howe, B.; Umrigar, A.; Tsien, F. Chromosome preparation from cultured cells. J. Vis. Exp. 2014, 3–7. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mendecki, L.; Granados-Focil, S.; Jendrlin, M.; Mold, M.; Radu, A. Self-plasticized, lumogallion-based fluorescent optical sensor for the determination of aluminium (III) with ultra-low detection limits. Anal. Chim. Acta 2020, 1101, 141–148. [Google Scholar] [CrossRef]
| G1 | S | G2/M | |
|---|---|---|---|
| H2O | 35.4 ± 1.3 | 44.5 ± 0.8 | 14.2 ± 0.3 |
| AlCl3 10 µM | 33.5 ± 1.3 | 46.6 ± 0.9 | 13.7 ± 0.4 |
| AlCl3 100 µM | 30.9 ± 1.1 c | 46.8 ± 0.7 | 16.6 ± 0.6 |
| AlCl3 300 µM | 28.9 ± 1.4 d | 46.5 ± 0.8 | 16.9 ± 0.7 |
| AlCl3 1 mM | 26.3 ± 1.4 d | 48.7 ± 0.6 b | 17.7 ± 0.5 a |
| H2O (+) | 32.3 ± 0.8 | 45.0 ± 0.5 | 16.5 ± 0.9 |
| MMS (+) | 18.3 ± 1.3 d | 48.3 ± 0.7 a | 23.8 ± 0.8 d |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tenan, M.R.; Nicolle, A.; Moralli, D.; Verbouwe, E.; Jankowska, J.D.; Durin, M.-A.; Green, C.M.; Mandriota, S.J.; Sappino, A.-P. Aluminum Enters Mammalian Cells and Destabilizes Chromosome Structure and Number. Int. J. Mol. Sci. 2021, 22, 9515. https://doi.org/10.3390/ijms22179515
Tenan MR, Nicolle A, Moralli D, Verbouwe E, Jankowska JD, Durin M-A, Green CM, Mandriota SJ, Sappino A-P. Aluminum Enters Mammalian Cells and Destabilizes Chromosome Structure and Number. International Journal of Molecular Sciences. 2021; 22(17):9515. https://doi.org/10.3390/ijms22179515
Chicago/Turabian StyleTenan, Mirna R., Adeline Nicolle, Daniela Moralli, Emeline Verbouwe, Julia D. Jankowska, Mary-Anne Durin, Catherine M. Green, Stefano J. Mandriota, and André-Pascal Sappino. 2021. "Aluminum Enters Mammalian Cells and Destabilizes Chromosome Structure and Number" International Journal of Molecular Sciences 22, no. 17: 9515. https://doi.org/10.3390/ijms22179515
APA StyleTenan, M. R., Nicolle, A., Moralli, D., Verbouwe, E., Jankowska, J. D., Durin, M.-A., Green, C. M., Mandriota, S. J., & Sappino, A.-P. (2021). Aluminum Enters Mammalian Cells and Destabilizes Chromosome Structure and Number. International Journal of Molecular Sciences, 22(17), 9515. https://doi.org/10.3390/ijms22179515






