Possible Use of Phytochemicals for Recovery from COVID-19-Induced Anosmia and Ageusia
Abstract
1. Introduction
2. Post-Viral Anosmia
2.1. Diagnosis and Symptoms
2.1.1. Symptoms
2.1.2. Pathophysiology and Viruses
2.1.3. Diagnosis
2.2. Upper Respiratory Tract as a Major Gate for SARS-CoV-2
2.3. COVID-19-Induced Anosmia
3. Perception of Odors
3.1. Perceiving Odors
Olfactory Neuroscience
3.2. What Determines How the Odors Smell?
3.2.1. Environment, Experience and Epigenetic Influences on Olfactory Receptor Gene Expression
3.2.2. Modulation at the Olfactory Epithelium and at the Olfactory Bulb
3.2.3. Genetic Variation and Smell
4. Smell Training to Enhance the Recovery of Olfactory Sense
4.1. The History of Smell Training
4.2. Using Odorants for the Stimulation of Olfactory Neurogenesis
| Olfactory Exposure Sources | Method | References |
|---|---|---|
| Lavender, garlic, paprika, marjoram, curry, rosemary, nutmeg, thyme, basil leaves, cumin, cardamom, tarragon, whole cloves, chocolate, celery, anise, ginger, lemon, orange, banana. | Exposed daily for 24 h to different odors placed in a tea ball hanging from the filter cover of the cages. Exposure days: 20 days or 40 days [175], 42 days [191], 31 days or 63 days or 42 days of enrichment + 21 days of standard condition [179]. Using mice. | [175,179,191] |
| Twenty different odors: pepper, star anise, fennel, cinnamon, garlic, onion, ginger, juniper berries, clove, nutmeg, lemon, celery, cumin, chocolate, cardamom, thyme, tarragon, capsicum, lavender, orange. | Daily renewal condition: the same sequence of exposure continued 20 days. Odor source was placed in a tea ball hung from the cage lid for 24 h. Same odor condition: 20 odors were mixed and presented. Odor source was replaced every 2 days. Using mice. | [177] |
| Lemon oil, juniper oil, clove oil, mint oil, lavender, musk, rosemary oil, tangerine, orange, sandalwood oil, thyme oil, sage, eucalyptus oil, cinnamon, calamint. | Twice a day, odor containing swab in a tea strainer placed on the cover of the cage for 1 h. Odorants selected randomly daily. Using rats. | [176] |
| Acetophenone, amazonica, dill, balsamic vinegar, basil, cocoa, (+)-carvon, cedar, cheese, chives, cineol, cinnamon, cloves, coffee, cumin, curry, “deodorant granules envirofresh apple, floral, lemon, and peach”, garlic geraniol, geranium golden wattle, hexanol, “honey and lemon cream”, isoamylacetate, juniper berries, lavender oil, linalool, (−)-limonelle, (+)-limonelle, lyral, massale, menthe piperite, mix morocco tea, nutmeg, olive oil, onion, oregano, paprika, Provence herbs, rosa, shallots, soybean sauce, strawberry, tarragon, tandoori, tobacco, vanilla, yeast extract. | “Odor pot” placed on the cage floor for 3 weeks, daily for 24 h to 3 different aromatic fragrances. Using mice. | [170] |
4.3. Smell Training for the Suppression of Inflammation and Enhancing Regeneration
4.3.1. Inflammation in the Damaged Olfactory Epithelium
4.3.2. Smell Training for the Enhancement of Regeneration
5. Taste
5.1. Morphology of Taste Cells
5.1.1. Sweetness
5.1.2. Umami
5.1.3. Bitter
5.1.4. Sour
5.1.5. Saltiness
5.2. Turnover of Taste Cells and Regeneration
5.3. ACE2 Expression in the Oral Cavity and Ageusia
6. The Chemesthesis
7. Phytochemicals for COVID-19-Induced Anosmia and Ageusia
7.1. Phytochemicals with Anti-Inflammatory Effects to Enhance the Recovery of Olfactory Sense and Taste
7.1.1. CB2 Receptor
7.1.2. GABA and Sodium Channels
7.1.3. Potassium Channels
7.1.4. TRP Channels
7.1.5. Multiple Routes by a Single Chemical Compound
7.2. Phytochemicals to Enhance the Recovery of the Sense of Taste
7.2.1. Terpenes
7.2.2. Flavonoids and Others
7.3. Mechanisms of Anti-Inflammatory Effects
| Chemical Compounds | Type | Effects (Suppress, Enhance); Receptors/Channels and Pathways Other than Olfactory Receptors, If Known | Examples of Source Essential Oils or Plants | References |
|---|---|---|---|---|
| Andrographolide CID: 5318517 CAS: 5508-58-7 | Diterpene MW: 350.4 | Suppresses: in vitro, Suppress NO, PLCγ2/PKC and PI3K/AKT-MAPK signaling pathways inhibiting platelet aggregation Vapor pressure: 1E-14 mmHg (25 °C) | Green chiretta (Andrographis paniculata (Burm. f)) | [312] |
| Allicin CID: 65036 CAS: 539-86-6 | MW: 162.3 | Suppresses: in vivo, 50 mg/kgbw for 5 days in rabbits infected with Pasteurella multocida improved the inflammatory markers [313] | Garlic | [313,314] |
| Apigenin CID: 5280443 CAS: 520-36-5 | Flavonone MW: 270.24 | Suppresses: in vitro, NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome action | parsley, celery, chamomile flowers | [315] |
| ß-Asarone CID: 5281758 CAS: 5273-86-9 | Phen MW: 208.25 | Suppresses: in vivo, suppress inflammatory cytokines, IL-6, IL-1 ß, iNOS, COX-2; suppress dizocilpine induced cognitive impairment; in vitro, suppressed LPS induced NO release, iNOS, COX-2, IL-1 ß, IL-6, TNFα, p65, lkBα, JNK, p38 | Acorus tatarinowii, Acorus gramineus, Asarum, Guatteria | [316,317] |
| α-Bisabolol CID:1549992 CAS: 515-69-5 | Sesq MW: 222.37 | Suppresses: review, inhibition of leukotriene synthesis, leukotriene synthesis, 5-LOX; in vivo, in vitro | German chamomile (Matricaria chamomilla), Candeia (Eremanthus erythropappus), Smyrniopsis aucheri, Heartwood (Vanillosmopsis pohlii), sage (Salvia runcinate) | [276,318] |
| Bornyl acetate CID: 6448 CAS: 76-49-3 | Mono MW: 196.29 | Suppresses: IL-6, IL-8, MMP-1, MMP-13 Vapor pressure: 0.107 mmHg (25 °C) | Amomum villosum | [275,278] |
| (-)-Borneol CID: 64685 CAS: 10385-78-1 | Mono MW: 154.25 | Suppresses: IL-1ß, IL-6, TNF-α, CD16, CD206 expressions, IL-10, phosphorylation of NF-kB, IkBa, p38, JNK, TRPA1 mediated cationic currents Enhances: NO, iNOS enzymatic activity Vapor pressure: 5.02 × 10−2 mmHg (25 °C) | Sambong (Blumea balsamifera), Balkan heath, Dryobalanops aromatica (Borneol camphor), Erica spiculifolia; included in citrus peel oils, spices (nutmeg, ginger, thyme); skin irritation by contact; odor, such as camphor | [275,278,319] |
| Broussoflavonol B CID: 480828 CAS: 99217-70-6 | Polyphenol MW:452.5 | Activates: in vivo, in vitro, suppress pro-inflammatory responses by activating AMPK in 3T3-L1 adipocyte [320] | Paper mulberry (Broussonetia papyrifera) | [320] |
| Cafestol CID: 108052 CAS: 469-83-0 | Diter MW: 316.40 | Suppresses: review, PGE2, NO synthesis, COX2, iNOS, inhibit activation of inhibitor kB kinase Enhances: Nrf2/HO-1 pathway, increase the expression of HO-1 | Coffee beans | [321] |
| Camphene CID: 6616 CAS: 79-92-5 | Mono MW: 136.23 | Suppresses: reduced nociceptive behavior Vapor pressure: 2.5 mmHg (25 °C) | Cannabis sativa | [322] |
| (+/−)-Camphor CID: 2537 CAS: 76-22-2 | Mono MW: 152.23 | Suppresses: Receptors/Channels: TRPV1 Vapor pressure: 0.65 mmHg (25 °C) Hazardous warning | Camphor tree (Cinnamomum camphora) | [278,296] |
| Cannabigerol CID: 5315659 CAS: 25654-31-3 | Resorcinol MW:316.5 | Suppresses: IL-1ß, TNF-α, IFN-γ, PPARγ, nitrotyrosine, SOD1, iNOS levels Enhances: restored Nrf-2 level Receptors/Channels: TRPM8 (some reports say TRPM8 antagonist), CB2, TRPA1, TRPV2, PPARγ, GPR55; | Cannabis sativa, non-psychotropic; partial agonist of CB2 but the affinity depends on the assays. CB1 unclear [323]; non-psychoactive | [323,324] |
| Capsaicin CID: 1548943 CAS: 404-86-4 | MW: 305.4 | Suppresses: in vivo, rat, significantly suppressed experimentally induced oedema due to egg albumin in the sub-plantar of the paw by IP injection of capsaicin (2 mL/kgbw) | Chili pepper | [325] |
| Carvacrol CID: 10364 CAS: 499-75-2 | Mono MW: 150.22 | Suppresses: reduced the activation of TLR4/NF-kB signaling pathway, suppressed IL-1ß, IL-6, IL-18, triglyceride, TNFα, suppressed levels of IKK, NALP3, NF-kB, TLR4, reduced p-IRS-1 and p-InsR levels (reduce insulin signaling molecules) in high glucose-induced HUVEC (human umbilical vein endothelial cells) Receptor: GABAA | Sideritis, oregano, thyme, pepperwort, wild bergamot | [275,285,326] |
| L-Carveol CID: 7438 CAS: 99-48-9 | Mono MW: 152.23 | Suppresses: in vivo, in vitro, in silico, suppressed blood glucose in alloxan-induced diabetic rats [327]; in vitro, NF-kB activity, TNFα, IL-1 ß, IL-10 [328] | Orange peel, dill, seeds of caraway (Carum carvi) | [275,327,328] |
| L-Carvone CID: 7439 CAS: 99-49-0 | Mono MW: 150.22 | Suppresses: NF-kB activity, TNFα, IL-1 ß, IL-10 (S-carvone; [329], IL-13 (S-carvone), IgE (S-carvone) Enhances: IL-10 (R-carvone) [329], IFNγ Receptors/Channels: In vitro, activate hTRPA1 with EC50 value at 112.2 µM, 81% [297] Vapor pressure: 0.115 mmHg at 25 °C | Caraway (Carum carvi) (S-carvone), spearmint (Mentha spicata) (R-carvone) | [275,297,328,329] |
| ß-Caryophyllene CID: 5281515 CAS: 87-44-5 | Sesq MW: 204.35 | Suppresses: IL-1ß, TNF-α, PGE2, iNOS, NO, ROS biomarkers, NF-kB, COX-2, IkBα; MMP8, Casp8, Casp4, IL-6 (in vivo, [16]), TREM1 signaling, VCAM-1 Enhances: IL-6 (in review, [278]), IL-19 Arg-1, urea, GSH parameters Receptor: CB2, (in vivo, [16]) | Copaiba, lavender, rosemary, peppermint, common sage, clary sage, bushy lippia, Balkan heath | [16,278,319] |
| (+)-Catechin CID: 9064 CAS: 154-23-4 | Flavonol MW: 290.27 | Suppresses: in vitro, suppress LPS induced pro-inflammatory cytokines TNFα, IL-1 ß, IL-6. | Green tea, berries, grape seeds, kiwi, red wine, beer, cacao, etc. | [330] |
| Chamazulene CID: 10719 CAS: 529-05-5 | Sesq MW: 184.28 | Suppresses: TNFα, IL-6, MMP3, MMP9, p65 NF-kB, iNOS, COX2 | chamomile (Matricaria chamomilla), wormwood (Artemisia absinthium), yarrow (Achillea millefolium) | [276,331] |
| 1,8-Cineole CID: 2758 CAS: 470-82-6 | Mono MW: 154.25 | Suppresses: in vivo, LTB4, PGE2, TNF-α, IL-1ß, leukotriene B4, thromboxane B2, BALF, NO, IL-6, MMP-9, IL-4, IL-13, IL-17A in BALF, IL-5, MCP-1 in nasal lavage fluids, IFN-γ in lung tissues, NF-kB p65, JNK, TREM-1, NLRP3, p38, MKP-1 phosphatase, NLRP3 inflammasome activation, acetylcholinesterase activities Enhances: IL-10, IkBα; in vivo, wound healing by Croton adamantinus oil [332] Receptors/Channels: TRPM8 Vapor pressure: 1.90 mmHg at 25 °C | Eucalyptus, Alpinia calcarata(synonym: eucalyptol) | [275,278,296,300,301,302,303,332] |
| Cinnamaldehyde CID: 637511 CAS: 104-55-2 | Phenyl MW: 132.16 | Suppresses: VAM-1 and ICAM-1, NF-kB, NO, IL-1ß, IL-6, TNFα, iNOS, IRF3, COX2, PVGE2 (review [333]); in vivo, NO, IL-1ß, IL-18, TNF-α, IFN γ, HMGB-1 (high mobility group box 1 protein) [334] Vapor pressure: 2.89 × 10−2 mmHg at 25 °C | Cassia oil, cinnamon bark oil | [333,334] |
| (E)-Cinnamyl acetate CID: 5282110 CAS: 103-54-8 | MW: 176.21 | Suppresses: in vivo, iNOS, COX-2, NF-kB, IkBα, decreased paw edema after CARR administration Increased: activities of catalase, superoxide dismutase, glutathione peroxidase in paw tissue after Carr administration [335]; NO and PGE2 production (review [333]) | Cinnamon (Cinnamomum cassia, Cinnamomum osmophloeum) | [333,335] |
| trans-Citral CID: 638011 CAS: 141-27-5 | Mono MW: 152.23 | Suppresses: NF-kB activation, COX-2, TRPV1-3, TRPM8, TRPV4, TRPA1 [284]; TNF, IL-6, IL-1ß, NO, macrophage activation, NLRP3 inflammasome activation; Compared to neral (isomer of cis-citral, neral), lower inhibitory effect on IL-1ß, iNOS, COX-2, and NLRP-2, and different inhibitory effects on phosphorylation of ERK1/2, JNK1/3, p38 and IkB [336] Channels: inhibitory effects on KV1.3 channel in vivo and in vitro [290], activate TRPA1, CB2 [284] | Lemongrass (Cymbopogon citratus); bushy lippia, lemon myrtle, Litsea citrate, Litsea cubeba, lemon tea-tree, Ocimum gratissimum, Lindera citriodora(synonym: geranial); 31.3% in L. cubeba fruits essential oil | [275,278,284,290,336,337,338] |
| cis-Citral CID: 643779 CAS: 106-26-3 | Mono MW: 152.23 | Suppresses: Compared to neral (isomer of citral, Citral B), lower inhibitory effect on IL-1ß, iNOS, COX-2, and NLRP-2, and different inhibitory effects on phosphorylation of ERK1/2, JNK1/3, p38 and IkB [336] | Litsea cubeba, 37.6% in L. cubeba fruits essential oil(synonym: neral) | [336] |
| Citronellol CID: 8842 CAS:106-22-9 | Mono MW: 156.26 | Suppresses: in vitro, down-regulated expression of ACE2 and TMPRSS2 [291] Receptors/channels: inhibitory effects on KV1.3 channel (ß-citronellol [290]) Vapor pressure: 0.02 mmHg at 25 °C | Geranium oil | [275,278,290,291] |
| Curcumin CID: 969516 CAS: 458-37-7 | Polyp MW: 368.40 | Suppresses: in vitro, suppressed phosphorylation of IKK ß and NF-kB p65 and suppressed degradation of IkBα [339] Vapor pressure: 3.08 × 10−12 mmHg at 25 °C | Turmeric (Curcuma longa) | [339,340,341] |
| Cyclocurcumin CID: 69879809 CAS: 153127-42-5 | Polyp MW: 368.40 | Suppresses: in vitro, higher neuroprotection than curcumin [342] | Turmeric (Curcuma longa); curcumin derivative | [341,342] |
| p-Cymene CID: 7463 CAS: 99-87-6 | Mono MW: 134.22 | Suppresses: in vivo, NO, NF-kB activity, TNFα, IL-1α, IL-10, suppressed licking behavior after formalin-injection Vapor pressure: 1.50 mmHg at 25 °C | Black cumin, rosemary, clove, Spanish oregano, valerian | [322,328] |
| Dehydrocostus lactone CID: 73174 CAS: 477-43-0 | Sesq MW: 230.30 | Suppresses: in vivo, NF-kB, COX2, TNF-α, IL-1ß, MCP-1, MPO, SOD, IL-6, IL-17, IL-23, IL-6/STAT3 inflammatory signaling pathway | Elecampane (Inula helenium), costus (Saussurea lappa) | [276,343] |
| Embelin CID: 3218 CAS: 550-24-3 | benzoquinone MW: 294.4 | Suppresses: in vivo, IP injection suppressed paw edema produced by carrageenan in rats | False black pepper (Embella ribes) | [344] |
| Epigallocatechin-3-gallate CID: 65064 CAS: 989-51-5 | Polyphenol MW:458.4 | Suppresses: review, alter NF-kB pathway, JAK/STAT pathway, PI3K/Akt pathway and suppress inflammation, down-regulate pro-inflammatory cytokines, COX, and reduce translocation of NF-kB to nucleus | Tea plant (Camellia sinensis) | [345] |
| Eugenol CID: 3314 CAS: 97-53-0 | Mono MW: 164.20 | Suppresses: in vivo, TNFα, IL-1ß, IL-6, NF-kB p65, suppress oxidative stress, reduced caspase-3 and p38 MAPK expressions in rats with spinal cord injury, IFNγ, IL-2, IL-10 Enhances: activate TRPA1, TRPV1 Vapor pressure: 0.0221 mmHg at 25 °C | Clove (Eugenia caryophyllata), | [296,346,347,348] |
| Eugenyl acetate CID: 7136 CAS: 93-28-7 | Phenol MW: 206.24 | Suppresses: in vitro, IFNγ, IL-2, IL-10 | Clove (Eugenia caryophyllata), especially in bud, Laurus nobilis | [348] |
| Farnesol CID: 445070 CAS: 106-28-5 | Sesq MW: 222.37 | Suppresses: in vivo, slightly decreased IL-4, TNFα/IL-19 ratio Enhances: IL-10; contact allergen [349] Vapor pressure: 3.94 × 10−5 mmHg at 25 °C | Oils of lemongrass, chamomile, citronella | [276,349,350] |
| Ferruginol CID: 442027 CAS: 514-62-5 | Diterp MW: 286.50 | Suppresses: in vivo, TNFα, NF-kB, IL-1ß, COX2, MMP9, IL-6, iNOS in mice with ulcerative colitis | In needles of redwood (Sequoia sempervirens), heartwood of hinoki cypress (Chamaecyparis obtusa) | [351] |
| Geraniol CID: 637566 CAS: 106-24-1 | Mono MW: 154.25 | Suppresses: T cell proliferation, IL-2, TNF-α, IFN-γ; down-regulated expression of ACE2 and TMPRSS2 Receptors/channels: inhibitory to Kv1.3, in vivo, in vitro Vapor pressure: 3.0 × 10−2 mmHg at 25 °C | Geranium, Ylang-ylang, cinnamon, coriander, lemon grass, citronella grass, clary sage, roses | [275,278,290,291] |
| Geranyl acetate CID: 1549026 CAS: 105-87-3 | Mono MW: 196.29 | Suppresses: reduced nociceptive behavior Receptors/Channels: In vitro, activate hTRPA1 with EC50 value at 20.5 µM, 74% Vapor pressure: 3.3 × 10−2 mmHg at 25 °C | Citronella, lemongrass, neroli, geranium | [297,322] |
| 6-Gingerol CID: 442793 CAS: 23513-14-6 | Phenol MW:284.4 | Suppresses: review, inhibition of pro-inflammatory cytokines, decreasing inducible NO synthase and TNFα by suppression of lkBα phosphorylation, NF-kB nuclear activation, and PKC α translocation, suppress IL-6, IL-8, SAA1 [352]; review, suppress IL-1 ß, IL-6, TNFα, down-regulate NF-kB/MAPK signaling pathway, iNOS, COX-2, suppress astrocyte overactivation, inhibit the expression of GFAP and TNFα in rat brain Enhances: Intercellular ROS, NO, iNOS, improved cognitive ability, improved memory | Ginger (Zingiber officinale Roscoe) | [352,353,354] |
| 10-Gingerol CID: 168115 CAS: 23513-15-7 | Phenol MW: 350.50 | Suppresses: review, suppress IL-1 ß, IL-6, TNFα significantly; greatest anti-inflammatory and anti-oxidant effect compared to other gingerols [355]; review, suppress neuroinflammation [352] | Ginger (Zingiber officinale Roscoe) | [352,353,355] |
| 18 α-Glycyrrhizin CID: 158471 18 ß-Glycyrrhizin CID: 3495 18 ß-Glycyrrhetinic acid CID:14982 | Triterp | Anti-inflammatory: 18ß-Glycyrrhizin ,suppressed PGE2, ROS, TNFα, COX-2, iNOS, 18α-Glycyrrhizin, stronger anti-inflammatory effect than 18ß-Glycyrrhizin, 18ß-Glycyrrhetinic acid, anti-oxidant, decreased lipid peroxidation, suppressed NO, PGE2, ROS, iNOS, COX-2, LPS-induced TNFα, IL-6, IL-1ß [356]; 18ß-Glycyrrhetinic acid, suppressed LPS-induced iNOS, COX-2, TNFα, IL-6, IL-1ß [357] | Licorice Glycyrrhiza uralensis Fisch., G. inflata Bat., G. glabra L., roots and rhizomes | [356,357] |
| Herbacetin CID: 5280544 CAS: 527-95-7 | Flavonol MW: 302.23 | Suppresses: in vitro, RAW264.7 cells, reduced NO production, reduced the release of TNFα, IL-1ß, suppressed JNK kinase, NF-kB [358]; in vivo, in vitro, down-regulated MMP-9 and cathepsin K, significantly reduced LPS-induced inflammatory bone loss [359] | Ephedrae herba | [358,359] |
| Humulene CID: 5281520CAS: 6753-98-6 | Sesq MW: 204.35 | Suppresses: review, IL-5, CCL11, leukotriene B4 level, NF-kB and AP-1 activation (synonym: α-caryophyllene; α-humelene) | Aniba parviflora, cannabis, hop | [276,278,360] |
| Isoliquiritigenin CID: 638278CAS: 961-29-5 | ChalconeMW: 256.25 | Suppresses: in vitro, NO production, TNFα, IL-6, iNOS in IL-1ß treated cells | Licorice Glycyrrhiza uralensis Fisch., G. inflata Bat., G. glabra L., roots and rhizomes | [361] |
| (-)-Isopulegol CID: 170833 CAS: 89-79-2 | Mono MW: 154.25 | Suppresses: IL-1ß, TNFα, decreased albumin extravasation, leukocyte migration and myeloperoxidase (MPO) enzyme concentration Receptor: GABAA | Ironwort (Sideritis), chemical precursor to menthol, also found in lemongrass and geranium | [285,362] |
| Kahweol CID: 114778 CAS: 6894-43-5 | Diter MW: 314.40 | Suppresses: review, PGE2, NO synthesis, COX2, iNOS, inhibit activation of inhibitor kB kinase Enhances: Nrf2/HO-1 pathway, increase the expression of HO-1 | Coffee beans | [321] |
| Kaempferol CID: 5280863 CAS: 520-18-3 | Flavonol MW: 286.24 | Suppresses: in vitro, NOD-like receptor family pyrin domain containing 3 (NLRP3) inflammasome action Vapor pressure: 0.0 ± 1.5 mmHg (25 °C) | Grapes, tomatoes, broccoli | [315] |
| Kazinol J CID: 21637732 | Polyphenol MW:410.5 | Activates: in vivo, in vitro, suppress pro-inflammatory responses by activating AMPK in 3T3-L1 adipocyte | paper mulberry (Broussonetia papyrifera) | [320] |
| Kirenol CID: 15736732 | Diter MW: 338.50 | Suppresses: in vitro, in vivo, IL-6, IL-8, MMP-9, MAPK, p65, P50, JAK gene expression in RA-FLS (rheumatoid arthritis-associated synovial fibroblasts) | Siegesbeckiae Herba (S. pubescens Makino S. orientalis L., S. glabrescens Makino) | [363] |
| Licochalcone A CID: 5318998 Licochalcone B CID: 5318999 Licochalcone C CID: 9840805 Licochalcone D CID: 10473311 Licochalcone E CID: 46209991 | Flavonoid | Suppresses: key factors for biological activities, Licochalcone A, suppress NO, IL-6, PGE2 IL-4, IL-5, IL-13, Licochalcone C, suppressed iNOS, Licochalcone E suppressed PKC/JNK, ERK1/2, iNOS, COX-2, IL-6, IL-1ß, IL-12 p40, TNF- α, AKT, p38 MAPK | Licorice Glycyrrhiza uralensis Fisch., G. inflata Bat., G. glabra L. | [356] |
| D-Limonene CID: 440917 CAS: 5989-27-5 | Mono | Suppresses: TNF-α, IL-1ß, IL-6, NF-kB, COX-2, iNOS, NO levels, p38, JNK activation; lipoxygenase; in vitro, down-regulated expression of ACE2 and TMPRSS2 [291] Receptors/Channels: In vitro, activate hTRPA1 with EC50 value at 54.3 µM, 83% [297] Vapor pressure: 1.98 mmHg at 25 °C | Citrus aurantium (bitter orange, daidai) | [275,278,291,297] |
| Linalool CID: 6549 CAS: 78-70-6 | Mono MW: 154.25 | Suppresses: TNF-α, IL-6, NO, IL-1ß, PGE2, p38, MAPK, NOS2, COX2, IL-18, IFN-γ, HMGB-1, MLNs, Nrf2 markers, iNOS expression, NF-kB activation, JNK activation, phosphorylation of IkBα protein, p38, c-JNK, ERK; in vivo, NO, IL-1ß, IL-18, TNF-α, IFN γ, HMGB-1 (high mobility group box 1 protein) [334] Enhancees: Nuclear Nrf-2 protein translocation; anxiolytic effect through GABA [19]; enhance recovery after ischemia [21] Receptors, pathways involved: GABA, TRPA1, TRPM8; inhibitory effects on KV1.3 channel in vivo and in vitro [290]; In vitro, activate hTRPA1 with EC50 value at 167.7 µM, 89% [297] Vapor pressure: 0.159 mmHg at 23.5 °C | Mint, rosewood, lavender, laurel, sweet basil, Cinnamomum osmophloeum Kanehira | [19,21,275,278,290,296,297,334] |
| Linalyl acetate CID: 8294 CAS: 115-95-7 | Mono MW: 196.29 | Suppresses: In vitro, suppressed TNFα induced E-selection, P-selection, vascular cell adhesion molecule-1 (VCAM1), suppressed NF-kB activation [364] Receptors/Channels: In vitro, activate hTRPA1 with EC50 value at 30.2 µM, 69% [297] Vapor pressure: 0.111 mmHg at 25 °C | Bergamot, lavender; acetate ester of linalool | [275,297,364] |
| Luteolin CID: 5280445CAS: 491-70-3 | polyphenol MW: 286.24 | Suppresses: review, in vivo, in vitro, regulates cytokines by suppressing IL-1ß, IL-6, IL-2, IL-8, IL-12, IL-17, TNFα, which are pro-inflammatory cytokines, and enhancing IL-10, which is anti-inflammatory cytokine | apples, carrots, celery, olive oil, rosemary, thyme, oregano, chamomile and many others | [365] |
| 1-Menthol CID: 16666 CAS: 89-78-1 | Mono MW: 156.26 | Suppresses: in vivo, increased survival rates in mice with myocardial infarction. Suppressed TNFα, IL-1ß, IL-6, monocyte chemoattractant protein 1 (MCP-1) [366]; in vivo and in vitro, inhibits acid-induced inflammation, suppress TNFα, IL-1ß, IL-6 through regulating TRPV1 [367] Receptors/Channels: TRPM8, TRPV1 | Genus Mentha, Corn mint, peppermint | [275,300,337,366,367] |
| Menthone CID: 26447 CAS: 14073-97-3 | Mono MW: 154.25 | Suppresses: in vivo, alleviate depression symptoms, suppressed expression of pro-inflammatory cytokines, IL-1ß, IL-6, TNFα, NLRP3 inflammasome [368] | Genus Mentha | [275,368] |
| Methyl eugenol CID: 7127 CAS: 93-15-2 | Phenyl MW: 178.23 | Suppresses: in vitro, inhibited the release of ß-hexosaminidase, TNFα IL-4, PGE2, prostaglandin D2, leukotriene B4, leukotrience C4, Syk phosphorylation and expression ERK1/2, p38, JNK phosphorylation, cytosolic phospholipase A2, 5-lipoxygenase phosphorylation, COX2 expression, considered to inhibit allergic response by these suppressions Enhances: in vivo, wound healing by Croton adamantinus oil [332] Receptors/channels: Nav1.7, GABAA Vapor pressure: 0.012 mmHg at 25 °C | Croton adamantinus (major chemical compound methyl eugenol and 1,8-cineole) [332]; Anti-allergic, antinaphylactic, antinociceptive, anti-inflammatory effects | [18,287,332,369] |
| Myrcene CID: 31253 CAS: 123-35-3 | Mono MW: 136.23 | Suppresses: review, NO, iNOS, NF-kB, p38, JNK activation Vapor pressure: 2.09 mmHg at 25 °C | Dill, cinnamon, coriander, lemon grass, citronella grass, English lavender, bushy lippia, common sage, clary sage, myrcia, bay, rosemary, cannabis, ylang-ylang, wild thyme, parsley, cardamom, hops | [275,278,337] |
| Nootkatone CID: 1268142 CAS: 4674-50-4 | Sesq MW: 218.33 | Suppresses: in vivo, suppressed edema, inhibition of IL-1ß, TNFα production, inhibition of COX-2 activity, anti-H1 receptor [370]; in vitro, show synergistic effect of suppressing inflammation with schisandrin, a polyphenol included in Schisandra genus [371] Vapor pressure: 0.003 mmHg at 25 °C | In many species of Citrus, black cardamom (Alpinia oxyphylla) | [276,370,371] |
| (E)-ß-Ocimene CID: 5281553 CAS: 3779-61-1 | Mono MW: 136.23 | Suppresses: NO production inhibition; NO scavenging effect; inhibited inducible NO synthase expression | Basil (Ocimum basilicum), water hemlock (Ocenanthe crocata) common wormwood or absinthe (Artemisia absinthium) and many others | [17,337,372] |
| Oleanolic acid CID: 10494 CAS: 508-02-1 | Tri MW: 456.70 | Suppresses: IL-6 and TNF-α [373]; in vitro, inhibit NF-kB activation [374] Enhances: cell viability and release of lactate dehydrogenase | Olive (Oleaceae), grapes (Vitis vinifera) | [373,374] |
| Osthole CID: 10228 CAS: 484-12-8 | Coum MW: 224.28 | Suppresses: in vivo and in vitro, NO, PGE2, TNFα, IL-6, iNOS, COX-2, p38 MAPK, IkB [375] Receptors/Channels: In vitro, activate hTRPA1 with EC50 value at 6.0 µM, 92% [297] Vapor pressure: 6.9 × 10−6 mmHg at 25 °C | Cnidium (Cnidium monnieri), shishiudo or du huo (Angelica pubescens) | [297,375,376] |
| Parthenolide CID: 7251185 CAS: 20554-84-1 | Sesq MW: 248.32 | Suppresses: Binds directly to IkB kinase ß (IKKß) and inhibits its activity. IkB is an inhibitor of NF-kB and becomes phosphorylated by IkB kinase complex, IKK. There are two forms: IKKα and IKKß. (in vitro, [377]), thus parthenolide suppress NF-kB by targeting IkB kinase (in vitro, [378]); Parthenolide depleted feverfew still has anti-inflammatory effects (in vitro, [379]) Others: allergen (human subjects, [380]) | Feverfew (Tanacetum parthenium) | [337,377,378,379,380] |
| Perillyl alcohol CID: 10819 CAS: 536-59-4 | Mono MW: 152.23 | Suppresses: review, oxidative stress and lipid peroxidation, IL-1ß, TNFα, IL-6, COX-2, NOS-2, NF-kB Enhances: levels of glutathione, catalase, glutathione peroxidase, and glutathione reductase Others: D-limonene metabolite; strong candidate for cancer treatments; induce apoptosis to cancer cells; oral treatment cause intestinal side effects; tissue regeneration improved; blocked formalin-, capsaicin-, and glutamate-induced nociceptive behavior | Lavender, sage, peppermint, lemongrass, cannabis, hop | [275,360] |
| α-pinene CID: 2723720 | Mono MW: 136.23 | Suppresses: review, NF-kB, ERK, JNK; G2/M-phase cell cycle arrest miR-221 expression level Enhances: CDKN1B/p2-CDK1 and ATM-p52- Chk2 pathways activated | Pine, coniferous species, sagebrush, ironwort, sage, Cannabis, Humulus | [278,337] |
| (R)-(+)-Pulegone CID: 442495 CAS: 89-82-7 | Mono MW: 152.23 | Suppresses: in vivo, suppressed skin thickness and scratching, serum IgE level, IL-4, IFN-γ, IL-6, TNF-α, IL-1ß, phosphorylation of MAPK, inhibited IkBα degradation and NF-kB activation [381] | Mint species, for example, Mentha spicata Mentha pulegium, Mentha piperita, Hedeoma multiflorum, Minthostachys mollis, Satureja boliviana, Satureja odora, | [275,381,382] |
| Sabinene CID: 18818 CAS: 3387-41-5 | Mono MW: 136.23 | Suppresses: in vitro, using whole essential oil from hallabong flower which included 34.75% sabinene, suppressed NO, PGE2, COX-2, TNF-α, IL-6, IL-1 ß [383]; suppressed NO production in lipopolysaccharide and IFN-γ stimulated macrophages [372] | Oenanthe crocata, 34.75% in Hallabong flower (Citrus unshiu Marcov × Citrus sinensis Osbeck) × Citrus reticulata Blanco | [372,383] |
| Santamarine CID: 188297 CAS: 4290-13-5 | Sesq MW: 248.32 | Suppresses: Suppress NF-kB activation, induces oxidative stress in cancer cells [384] | Southern magnolia (Magnolia grandiflora), weakleaf bur ragweed (Ambrosia confertiflora) | [275,384] |
| 6-shogaol CID: 5281794 CAS: 555-66-8 | MW: 276.40 | Suppresses: review, inhibits direct binding between intercellular adhesion molecule, inhibits production of prostaglandin E2 and pro-inflammatory cytokines, together with 10-gingerol, suppressed NO, IL-1 ß, IL-6, TNF-α | Ginger (Zingiber officinale); Dehydration product of gingerol | [352] |
| Spathulenol CID: 92231 CAS: 6750-60-3 | Sesq MW: 220.35 | Suppresses: in vivo and in vitro. Using whole essential oil which contains 80% spathulenol. Inflammation measured by swelling in carrageenan-induced paw oedema | Brazilian guava (Psidium guineense Sw.) | [385] |
| α-terpinene CID: 7462 CAS: 99-86-5 | Mono MW: 136.23 | Suppresses: review, COX21 Enhances: review, Strong anti-oxidant; increased longevity of mice infected with Trypanosoma evansi | Tea tree (Melaleuca alternifolia), Litsea, cannabis, hops | [275,360] |
| γ-terpinene CID: 7461 CAS: 99-85-4 | Mono MW: 136.23 | Suppresses: review, TNF-α, IL-1ß, IL-6 Enhances: review, IL-10, COX-2, PGE2; In vivo, anti-nociceptive effect to formalin, capsaicin, glutamate-induced pain in rats, cholinergic and opioid systems were involved in anti-nociceptive effects [386] Vapor pressure: 1.09 mmHg | Narrow-leaved paperbark (Melaleuca alternifolia), thyme, savories (Satureja), cannabis, hops | [278,360,386] |
| Terpinen-4-ol CID: 11230CAS: 562-74-3 | MonoMW: 154.25 | Suppresses: NF-kB, NLRP3, IkBα, NF-kB p65; IL-1ß, IL-6, IL-10 Enhances: PPAR-γ, Route: GABAergic system Vapor pressure: 0.04 mmHg at 25 °C | Tea tree (Melaleuca alternifolia), lavender | [275,278] |
| α-terpineol CID: 17100 CAS: 98-55-5 | Mono MW: 154.25 | Suppresses: Nitrite production, NF-kB, IL-1ß, IL1R1; IL-1ß, IL-6, IL-10, IL-4, IL-17 TNF-α, COX-2, iNOS, Enhances: IL-10 Vapor pressure: 0.0423 mmHg at 24 °C | Melaleuca genus, eucalyptus, Balkan heath (Erica spiculifolia), cajuput, pine, orange juice | [275,278,319,387,388] |
| Terpinolene CID: 11463 CAS: 586-62-9 | Mono MW: 136.23 | Suppresses: IL-6, TNF-α, NO Vapor pressure: 0.74 mmHg at 25 °C | Melaleuca genus; Myrtle (Myrtus communis L., Myrtaceae) | [278] |
| Thymol CID: 6989 CAS: 89-83-8 | Mono MW: 150.22 | Suppresses: in vitro, IL-8, TNFα, reactive oxygen species (ROS) [389] Enhances: barrier function Vapor pressure: 0.016 mmHg at 25 °C | Lippia gracilis Schauer, oregano species, thyme species, Balkan heath (Erica spiculifolia) | [275,319,337,389] |
| Thymoquinone CID: 10281 CAS: 490-91-5 | Mono MW: 164.20 | Suppresses: in vivo and in vitro, suppressed NO, iNOS, TNFα, COX2, IL-6, IL-1ß in lipopolysaccharide-stimulated murine macrophage-like RAW264.7 cells, suppression of IRAK-linked AP-1/NF-kB pathways, suppressed hepatitis and gastritis symptoms in mouse models [390] | Black seed (Nigella sativa) | [275,390] |
| Tomentosin CID: 155173 CAS: 33649-15-9 | Sesq MW: 248.32 | Suppresses: in vitro, NF-kB, MAP, NO, PGE2, iNOS, COX-2, TNF-α, IL-6, p65 | Inula japonica, Inula viscosa (syn. Dittrichia viscosa Greuter), | [276,391] |
| Tussilagone CID: 13919185 CAS: 104012-37-5 | Sesq MW: 390.50 | Suppresses: in vitro, suppressed production of NO, TNF-α, PGE2, iNOS, COX2 [392]; in vivo, protective effect against dextran sulfate sodium-induced acute colitis in mice, TNF-α, IL-6, and myeloperoxidase activity reduced [393] Enhances: heme oxygenase-1 | Flower and buds of Tussilago farfara | [276,392,393] |
| Ursolic acid CID: 64945 CAS: 77-52-1 | Tri MW: 456.70 | Suppresses: in vitro, IL-6 and TNF-α [373]; in vitro, inhibit NF-kB activation [374] Enhances: cell viability and release of lactate dehydrogenase Vapor pressure: 3.49 × 10−14 mmHg at 25 °C | Various fruits and vegetables (apples, berries, peppermint, lavender, oregano, etc.) | [373,374,394] |
| Valencene CID: 9855795 CAS: 4630-07-3 | Sesq MW: 204.35 | Suppresses: in vivo and in vitro, IL-1ß, IL-6, IL-13, NF-kB, CXCL8, GM-CSF, I-CAM, reduced atopic dermatitis-like symptoms [395] Enhances: skin barrier protein, involucrin increased in murine skin [395] | Nut grass (Cyperus rotundus), Citrus; orange peel oil | [276,395] |
7.4. Terpenes and Other Volatile Phytochemicals with Anti-Viral Effects
| Chemical Compounds | Type | Targeted Virus, Parts of the Virus, and Effects | Essential Oils and Herbs with the Chemical Compoundsas Major Chemical Constituents | References |
|---|---|---|---|---|
| Acetoside CID: 5281800 CAS: 61276-17-3 | Phenol MW: 624.60 | Covalent docking with 3CLpro (6LU7) of COV2 and XP docking and covalent docking with spike RBD (6M0J) of S glycoprotein | Many plants, example, Ligustrum purpurascens, Rehmannia glutinosa | [412] |
| Andrographolide CAS: 5508-58-7 CID: 5318517 | Diter MW: 350.4 | Suppress: suppress the 3CLpro activities of SARS-CoV-2; | Green chiretta (Andrographis paniculate) | [312,341] |
| Anethole CID: 637563 CAS: 104-46-1 | Mono MW: 148.20 | COV2 by binding affinity to RBD of S glycoprotein | Star anise (Illicium verum), Apiaceae (fennel, celery, carrot, parsley), Myrtaceae (myrtle, bay rum tree, clove, guava) | [408] |
| Betulonic acid CID: 122844 CAS: 4481-62-3 | Triterp MW: 454.70 | COV1, HCPE at >10 µM 3CLpro, EC50 0.63 µM, CC50 112 µM, Selectivity 180 | Formosan juniper (Juniperus formosana) | [407] |
| Betulinic acid CID: 64971 CAS: 472-15-1 | Triterp MW: 456.70 | COV1, HCPE at >3.3 µM, EC50 >10 µM, CC50 150 µM, Selectivity <15 | Downy birch or white birch (Betula pubescens) | [407] |
| (-)-α-cadinol CID: 6431302 | Sesq MW: 222.37 | COV1, HCPE at >1 uM, EC50 4.44 µM, CC50 76.8 µM, Selectivity 17.3 | Hinoki cypress (Chamaecyparis obtuse var. formosana, or Chamaecyparis taiwanensis) | [407] |
| Cafestol CID: 108052 CAS: 469-83-0 | Diter MW: 316.40 | In silico, COV2, PLpro, Guanine-N7 methyl transferase (ExoN) | Coffee beans | [413] |
| Carvacrol CID: 10364 CAS: 499-75-2 | Mono MW: 150.22 | COV2 by binding affinity to RBD of S glycoprotein [408]; COV2 by binding affinity to Mpro [414] | Oregano (Origanum species), thyme (Thymus vulgaris), Spanish origanum (Thymus capitatus), pepperwort (Lepidium flavum), black cumin (Nigella sativa), summer savory (Satureja hortensis), winter savory (Satureja montana) | [408,414] |
| Cinnamaldehyde CID: 637511 CAS: 104-55-2 | Phenyl MW: 132.16 | COV2 by binding affinity to RBD of S glycoprotein | Cinnamon (Cinnamomum zeylanicum) | [408] |
| (E)-Cinnamyl acetate CID: 5282110 CAS: 103-54-8 | Ester MW: 176.21 | COV2 by binding affinity to RBD of S glycoprotein | Cinnamon (Cinnamomum zeylanicum) | [408] |
| Curcumin CID: 969516 CAS: 458-37-7 | Polyp MW: 368.40 | In silico, Covalent docking with 3CLpro (6LU7, 5R82) of COV2; in silico, COV2, 3CLpro | Turmeric (Curcuma longa) | [341,412,413] |
| Cyclocurcumin CID: 69879809 CAS: 153127-42-5 | Polyp MW: 368.4 | In silico, 3CLpro (5R82; G score -6.77) | Turmeric (Curcuma longa) | [341] |
| Dehydroabieta-7-one CID: 11289118 | Diterp MW: 284.40 | COV1, HCPE at >10 µM, EC50 4 µM, CC50 305.1 µM, Selectivity 76.3 | Hinoki cypress (Chamaecyparis obtuse var. formosana, or Chamaecyparis taiwanensis) | [407] |
| 6,7-dehydroroyleanone CID: 2751794 CAS: 6855-99-8 | Diterp MW: 314.40 | COV1, HCPE at >10 µM, EC50 5.55 µM, CC50 89.7 µM, Selectivity 16.2 | Hinoki cypress (Chamaecyparis obtuse var. formosana, or Chamaecyparis taiwanensis) | [407] |
| 3ß,12-diacetoxyabieta-6,8,11,13-tetraene Not in pubchem | Diterp | COV1, HCPE at >3.3 µM, EC50 1.57 µM, CC50 303.3 µM, Selectivity 193 | Formosan juniper (Juniperus formosana) | [407] |
| Embelin CID: 3218 CAS: 550-24-3 | Quinone MW: 294.40 | In silico, COV2, binding affinity to Nst7-Nsp8 complex [413]; with 3CLpro | False black pepper (Embella ribes), dotted loosestrife (Lysimachia punctata) | [413,415] |
| (-)-Epicatechin CID: 72276 CAS: 490-46-0 | Flavonol MW: 290.27 | In silico, COV2, binding affinity to PLpro, 3Lpro, hACE2, Nsp7-Nsp8, Guanine-N7 methyl transferase (ExoN), RdRp, Helicase, NendoU, M, NC | Tea plant (Camellia sinensis) | [413] |
| Epigallocatechin-3-gallate CID: 65064 CAS: 989-51-5 | Polyphenol MW: 458.4 | in vitro, COV2, suppress 3CLpro | Tea plant (Camellia sinensis) | [416] |
| Ferruginol CID: 442027 CAS: 514-62-5 | Diterp MW: 286.50 | COV1, HCPE at >3.3 µM, EC50 1.39 µM, CC50 80.4 µM, Selectivity 58 | Hinoki cypress (Chamaecyparis obtuse var. formosana, or Chamaecyparis taiwanensis) | [407] |
| Forskolin CID: 47936 CAS: 66428-89-5 | Diterp MW: 410.5 | COV1, HCPE at >3.3 µM, EC50 7.5 µM, CC50 674 µM, Selectivity 89.8 | Coleus barbatus | [407] |
| Geraniol CID: 637566 CAS: 106-24-1 | Mono MW: 154.25 | COV2 by binding affinity to RBD of S glycoprotein | Cymbopogon (palmarosa, citronella, lemongrass), Lavendula (lavender), rose | [408] |
| Gingerol CID: 442793 CAS: 23513-14-6 | Phenol MW: 284.4 | COV2 by binding affinity to 3CLpro (5R7Y), to RNA binding proteins 6W4B and 6VSB, and to S glycoprotein (6M3M) | Ginger | [409] |
| Glycyrrhizin (glycyrrhizic acid) CID: 14982 CAS: 1405-86-3 | Triterp MW: 822.9 | COV1, EC50 300 mg/L, CC50 >20,000 mg/L, Selectivity >67 if added during and after virus absorption | Liquorice (Glycyrrhiza glabra) | [417] |
| COV1, EC50 365 µM, CC50 >24,000 µM, Selectivity >65 | Liquorice (Glycyrrhiza glabra) | [418] | ||
| Herbacetin CID: 5280544 CAS: 527-95-7 | Flavonol MW: 302.23 | COV1 suppress 3CLpro | Ephedrae herba | [411,419] |
| ß-hydroxyabieta-9(11),13-dien-12-one Not in pubchem | Diterp | COV1, HCPE at >3.3 µM, EC50 1.47 µM, CC50 >750 µM, Selectivity >510 | Hinoki cypress (Chamaecyparis obtuse var. formosana, or Chamaecyparis taiwanensis) | [407] |
| 4-hydroxyderricin CID: 6438503 CAS: 55912-03-3 | Chalcone MW: 338.4 | COV1; suppress 3CLpro, PLpro | Ashitaba (Angelica keiskei) | [411] |
| 7ß-hydroxydeoxycryptojaponol Not in Pubchem | Diterp | COV1, HCPE at >10 µM, EC50 1.15 µM, CC50 127 µM Selectivity 111 | Japanese cedar or sugi (Cryptomeria japonica) | [407] |
| Kaempferol CID: 5280863 CAS: 520-18-3 | Flavonol MW: 286.24, | COV2, 3CLpro Vapor Pressure: 0.0 ± 1.5 mmHg at 25 °C | Grapes, tomatoes, broccoli; Green chiretta (Andrographis paniculata (Burm. f)) | [341] |
| Kazinol J CID: 21637732 | Polyphl MW:410.5 | In silico, COV2, binding affinity with 3CLpro | Paper mulberry (Broussonetia papyrifera) | [420] |
| Luteolin CID: 5280445CAS: 491-70-3 | Flavonone MW: 286.24 | COV2 by binding affinity to 3CLpro [421]; to 3pro and ACE2 [422] | Dyer’s weed (Reseda luteola) | [415,421,422] |
| 1-Menthol CID: 16666 CAS: 89-78-1 | Mono MW: 156.26 | COV2 by binding affinity to RBD of S glycoprotein | Peppermint oil | [408] |
| Murrayanine CID: 96942 CAS: 723-97-7 | Carbazole MW: 225.24 | In silico, COV2, inhibitory to Nsp10-Nsp16 complex | Curry tree (Murraya koenigii) | [413] |
| Murrayaquinone-A CID: 127481 CAS: 100108-66-5 | Carbazole MW: 211.22 | In silico, COV2, inhibitor of Nsp9, for H-bond with Val110 | [413] | |
| Oleanolic acid CID: 10494 CAS: 508-02-1 | Triterp MW: 456.70 | COV2 by binding affinity to 3CLpro | Olive (Oleaceae), grapes (Vitis vinifera) | [414] |
| Pectolinarin CID: 168849 CAS: 28978-02-1 | Flavone MW: 622.6 | COV1 suppress 3CLpro | [419] | |
| Pinusolidic acid CID: 25880646 | Diterp MW: 332.4 | COV1, HCPE at >10µM, EC50 4.71 µM, CC50 > 750 µM, Selectivity > 159 | Hinoki cypress (Chamaecyparis obtuse var. formosana, or Chamaecyparis taiwanensis) | [407] |
| (+)-Pulegone CID: 442495 CAS: 89-82-7 | Mono MW: 152.23 | COV2 by binding affinity to RBD of S glycoprotein | Creeping charlie (Glechoma hederacea or Nepeta Glechoma), catnip (Nepeta cataria), Pennyroyal (Mentha pulegium) | [408] |
| Quercetin CID: 5280343 CAS: 117-39-5 | Flavonole MW: 302.23 | COV1 suppress 3CLpro | [411,423] | |
| Rhoifolin | Flavone | COV1 suppress 3CLpro | [419] | |
| Rutin | Rutin | COV2 by binding affinity to RBD (6M0J) of S glycoprotein and to 3CLpro (6LU7) | [412,423] | |
| Solanine | alkaloid | COV2 by binding affinity to RBD (6M0J) of S glycoprotein and to 3CLpro (6LU7) | [412] | |
| 4-Terpineol | Mono | COV2 by binding affinity to RBD of S glycoprotein | Major chemical constituent of Tea tree oil, lavender, turpentine oils | [408] |
| Thymol CID: 6989 CAS: 89-83-8 | Mono MW: 150.22 | COV2 by binding affinity to RBD of S glycoprotein; HSV-1 | Isomeric with carvacrol; Thyme (Thymus vulgaris) | [408] (COV2); [424] (HSV) |
| Ursolic acid CID: 64945 CAS: 77-52-1 | Triterp MW: 456.70 | COV2 by binding affinity to 3CLpro | Peels of fruits | [414] |
| Xanthoangelol | COV1 suppress PLpro [425], 3CLpro [411] | Ashitaba (Angelica keiskei) | [425] |
| Oils, Plants, Fruits Type | Major Chemical Constituent | Confirmed Effects and Binding Affinities |
|---|---|---|
| Ashitaba (Angelica keiskei) | 4-hydroxyderricin (CID: 6438503; CAS: 55912-03-3) | Anti-viral: [411] |
| Xanthoangelol (CID: 643007; CAS: 62949-76-2) | Anti-viral: [411,425] | |
| Bitter orange (Citrus aurantium L. (Rutaceae)) | Limonene | Anti-inflammation [275,278,297] |
| Linalool | Anti-inflammation [19,20,21,22,275,278,290,296,297] | |
| β-Myrcene | Anti-inflammation [275,278,337] | |
| Kaempferol | Anti-inflammation [315] | |
| Quercetin | Anti-viral: [411,423] | |
| Luteolin | Anti-viral: COV2 by binding affinity to 3CLpro [421]; to 3pro and ACE2 [422] | |
| Cinnamon (Cinnamomum) | Camphor: 60% in root bark [426] | Anti-inflammation [278,296] |
| Cinnamaldehyde: 65–80% in bark [426] | Anti-viral: [408] | |
| Z-Cinnamyl acetate: 42–54% in fruit [426]; 11.85% in Cinnamomum verum oil [427] | Anti-viral: [408] | |
| E-Cinnamyl acetate: 41.98% in flowers of Ceylon cinnamon, Cinnamomum zeylanicum [426]; 11.78% in Cinnamomum verum oil [427] | Anti-viral: [408] | |
| Eugenol: 70~95% in leaves [428], Cinnamomum zeylanicum [426] | Anti-inflammation [296,346,347,348] | |
| Linalool: 16.85% in Cinnamomum verum oil [427] | Anti-inflammation [19,20,21,22,275,278,290,296,297] | |
| Citronella oil | See lemongrass | |
| Clove (Syzygium) | ß-Caryophyllene: 17.4% in leaf [429], 1.39% in bud [430], 14.84% and 12.79% in bud from Java and Manado, Indonesia, respectively [431]; 4.5% in oil [432] | Anti-inflammation [16,278,319] |
| Eugenol: 88.6% in bud [430], 74.65% and 55.65% in bud from Java and Manado, Indonesia, respectively [431], 76.8% in leaf [429], 69.4% in bud [433]; 85.7% in oil [432] | Anti-inflammation [296,346,347,348] | |
| Eugenyl acetate: 10.79% [433], 1.2% in leaf [429], 20.54% and 8.7% in bud from Java and Manado, Indonesia, respectively [431] | Anti-inflammation [348] | |
| α-Humulene: 2.1% in leaf [429], 2.75% and 1.53% in bud from Java and Manado, Indonesia, respectively [431] | Anti-inflammation [276,278,360] | |
| Copaiba (Copaifera) | ß-Caryophyllene: 24.9% in oil [434], 21.7% in oil [435] | Anti-inflammation [16,278,319] |
| α-Bergamotene: 20.5% in oil [435] | - | |
| ß-Bisabolene: 23.6% in oil [435] | - | |
| α-Humulene: 2.9% in oil [435] | Anti-inflammation [276,278,360] | |
| Caryophyllene oxide: 4.1% in oil [435] | Allergen | |
| Cypress (Hinoki) (Chamaecyparis obtusa) | α-cadinol: 10.9% in oil [436] | Anti-viral [407] |
| Borneol: 16.0% in oil [436] | Anti-inflammation [275,278,319] | |
| Dehydroabieta-7-one | Anti-viral [407] | |
| 6,7-Dehydroroyleanone | Anti-viral [407] | |
| Ferruginol: inclusion and % varies largely | Anti-inflammation [309] | |
| ß-Hydroxyabieta-9(11) | Anti-viral [407] | |
| α-Terpineol: 19.4% in oil [436] | Anti-inflammation [275,278,319,387,388] | |
| Elderberry (Sambucus nigra L.) | Quercetin | Anti-viral [411,423] |
| Rutin | Anti-viral [412,423] | |
| Kaempferol | Anti-inflammation [437] | |
| Caffeic acid | Anti-inflammation [437] | |
| 3,4-dihydroxyphenylacetic acid | Anti-inflammation [437] | |
| Eucalyptus (Eucalyptus) | Borneol: 5.5% in leaf and 5.5% in fruit [438] | Anti-inflammation [275,278,319] |
| 1,8-Cineole (eucalyptol): large percentage difference among species and countries of origin, 2.9% to 90.0% [439], species percentage differences in leaves from 49.07 to 83.59% [300], 14.1% in leaf and 34.5% in fruit [438] | Anti-inflammation [11,238,241,261,262,265,267,291,400] | |
| p-Cymene: 42.1% in leaf and 30.0% in fruit [438] | Anti-inflammation [322,328] | |
| Limonene: 5.5% in leaf and none in fruit [438], 1.33% in E. lehmani leaves and 3.32% in E. sideroxylon leaves [300], from 0% to 28% depending on the species and location [439] | Anti-inflammation [275,278,297] | |
| α-Pheliandrene: from 0% to 20.1% depending on the species and location [439] | Anti-inflammation [278] | |
| α-Pinene: 12.7% in leaf and 9.0% in fruit [438], from 1.27% to 26.35% in 7 Eucalyptus species’ leaves with highest in E. lehmani [300], from 0% to 52.7% depending on the species and location [439] | Anti-inflammation [278,337] | |
| Spathulenol: 3.2% in leaf and none in fruit [438], 1.15% in E. astrengens leaves [300], from 0% to 41.5% depending on the species and location [439] | Anti-inflammation [387] | |
| γ-Terpinene: none in leaf and 5.1% in fruit [438], from 0% to 29.2% depending on the species and location [439] | Anti-inflammation [278,360,386] | |
| Geranium (Pelargonium) | ß-bourbonene: 2.7% in oil from Tajikistan [440] | - |
| Caryophyllene oxide: 3.7% in oil from Tajikistan [440] | Allergen [441] | |
| Citronellol: 37.5% in oil from Tajikistan [440] | Anti-inflammation [275,278,290,291] Anti-viral: Down-regulated expression of ACE2 and TMPRSS2, | |
| Geraniol: 6.0% in oil from Tajikistan [440] | Anti-inflammation [275,278,290,291] Anti-viral: [408] | |
| Geranyl formate: 2.0% in oil from Tajikistan [440] | ||
| Linalool: 3.0% in oil from Tajikistan [440] | Anti-inflammation [19,20,21,22,275,278,290,296,297] | |
| Ginger (Zingiber officinale Roscoe) | 6-Gingerol: (23–25%) [442] Other chemical constituents: 10-gengerol, 6-shogaol, α-Terpinene, α-Terpineol, 4-Terpineol, Terpinolene, γ-Terpinolene, Cineole, Nerol, Borneol, Citronellol, Geraniol, Linalool, Camphor, Neral | Anti-viral: [409] |
| Lavender (Lavandula) | Borneol: 0.3% to 22.4% [443] | Anti-inflammation [275,278,319] |
| Camphor: none to 11.76% [443] | Anti-inflammation [278,296] | |
| ß-Caryophyllene: none to 3.2% [443] | Anti-inflammation [16,278,319] | |
| 1,8-Cineole (Eucalyptol): 0.1% to 10.89% depending on location [443] | Anti-inflammation [275,278,296,297,300,302,303] | |
| Geraniol: none to 11.02% [443] | Anti-inflammation [275,278,290,291] Anti-viral: [408] | |
| Lavandulol: 3.7% in oil [444] | - | |
| Lavandulol acetate (lavandulyl acetate): 0.2% to 21.6% in oils depending on location [445]; 5.7% in oil [444]; 12.68% in oil [446], none to 10.78% [443] | - | |
| Linalool: 49.9% in oil [444]; 19.71% in oil [446], 4.91% to 57.48% [443] | Anti-inflammation [19,21,22,275,278,290,296,297] | |
| Linalyl acetate: 9.3% to 68.8% in oils depending on location [445]; 14.4% in oil [444]; 26.61% in oil [446], none to 35.39% [443] | Anti-inflammation [275,297,364] | |
| Myrcene: 1.4% in oil [444] | Anti-inflammation [275,278,337] | |
| Ocimene: 2.4% to 2.6% in oil [444] | Anti-inflammation [17,337,372] | |
| 4-Terpineol, terpinen-4-ol (isomer of terpineol): 0.1% to 5.8% in oils depending on location [445], none to 8.07% [443] | Anti-inflammation [275,278] Anti-viral: [408] | |
| Lemongrass (Cymbopogon citratus) | ß-Caryophyllene: 3.26% in oil [447], 1.09 in oil [338] | Anti-inflammation [16,278,319] |
| Citral A (Geranial, E-isomer of citral): 37.40% in oil from Guangxi, China [448], 26.1% in oil [447], 37.58 to 45.95% in oil [449], 40.16% in oil [338] | Anti-inflammation [275,278,284,290,337] | |
| Citral B (Neral, Z-isomer of citral): 31.97% in oil from Guangxi, China [448], 31.5% in oil [447], 29.44 to 31.13% in oil [449], 34.24% in oil [338] | Anti-inflammation: [336] | |
| Citronellol: 1.10% in oil from Guangxi, China [448], 2.95% in oil [447], 0.35% to 0.51% in oil [449] | Anti-inflammation [275,278,290,291] | |
| Geraniol: 1.55% in oil from Guangxi, China [448], 2.15% in oil [447], 5.11% in oil [338] | Anti-inflammation [275,278,290,291] Anti-viral: [408] | |
| Geranyl acetate: 1.06% to 2.16% in oil [449], 2.27% in oil [447], 2.89% in oil [338] | Anti-inflammation [297,332] | |
| Juniper camphor: 1.28 to 2.82% [449] | - | |
| Limonene: 0.65% in oil from Guangxi, China [448], 2.32% in oil [447], 0.33% in oil [338] | Anti-inflammation [275,278,297] | |
| Linalool: 1.12% in oil from Guangxi, China [448], 0.58% to 0.87% [449], 1.45% in oil [338] | Anti-inflammation [19,20,21,22,275,278,290,296,297]. | |
| Myrcene: 15.65% in oil from Guangxi, China [448] | Anti-inflammation [275,278,337] | |
| Licorice (Glycyrrhiza spp.) | Over 20 triterpenes and 300 flavonoids of natural active compounds and 73 bioactive compounds identified. “3 triterpenes, 18b-GC, 18a-GC and 18b-glycyrrhetinic acid (18b-GA), and 13 flavonoids, licochalcone A (LCA), licochalcone B (LCB), licochalcone C (LCC), licochalcone D (LCD), licochal- cone E (LCE), isoliquiritigenin (ISL), echinatin (EC), glabridin (GLD), isoangustone A (ISOA), licoricidin (LID), licorisoflavan A (LIA), dehydroglyasperin C (DGC) as well as dehydroglyas- perin D (DGD), all have been reported to possess anti-inflammatory activity.” [356]; contained in root twigs | Anti-inflammation [356,357] Anti-viral: [417,418] |
| Mint, mentha (spearmint, Mentha spicata) | Carvone: 40.8% in oil [450], 49.5% [451], 70.36% in oil [452] | Anti-inflammation [275,297,328,329] |
| ß-Caryophyllene: 1.2% in oil [450], 2.7% in oil [451], 1.1%. in oil [452] | Anti-inflammation [16,278,319] | |
| 1,8-Cineole (Eucalyptol): 17.0% in oil [450], 8.7% in oil [451], 2.24% in oil [452] | Anti-inflammation [275,278,296,297,300,301,302,303,332] | |
| Limonene: 20.8% in oil [450], 16.1% in oil [451], 6.6% in oil [452] | Anti-inflammation [275,278,297] | |
| ß-Pinene: 2.2% in oil [450], 1.1% in oil [451], 0.6% in oil [452] | - | |
| 4-Terpineol: 1.3% in oil [450], 1.5% in oil [451], 1.09% in oil [452] | Anti-inflammation [275,278] Anti-viral: [408] | |
| Oregano (Poliomintha longiflora) | Carvacrol: 12.6% in oil, 60.03% to 64.315 in fractions at 140C and undistilled oil, respectively [453] | Anti-inflammation [275,285,326,337] Anti-viral: [408] |
| P-Cymene: 11.5% to 35.7% in Oreganum vulgare in Nefza, Tunisia and different harvest years, 27.3% to 46.3% in Krib, Tunisia and different harvest years [454,455] | Anti-inflammation [322,328] | |
| o-Ocymene: 39.13% in oil, 47.96 to 53.97% in fractions at 82C and 100C, respectively [453] | - | |
| α-Terpinene: 5.57% in oil [453] | Anti-inflammation [275,360] | |
| γ-Terpinene: 22.34% in oil, 15.59%, 24.43%, 40.57% in fractions at 82C, 100C, 120C, respectively [453] | Anti-inflammation [278,360,386] | |
| Thymol: 1.71% in oil, 5.08% and 3.77% in fractions at 140C and undistilled oil, respectively [453] | Anti-inflammation [275,319,337,389] Anti-viral [408] | |
| Paper mulberry (Broussonetia papyrifera) | Bavachromene (CID: 5321800) | Anti-viral: [411] |
| 3′-(3-methylbut-2-enyl)-3′,4,7-trihydroxyflavane (CID: 129704069) | Anti-viral: [411] | |
| Broussoflavan A (CID: 44257078) | Anti-viral: [411] | |
| Kazinol A (CID: 442414; CAS: 99624-28-9) | Anti-viral: [411] | |
| Kazinol B (CID: 480869; CAS: 99624-27-8) | Anti-viral: [411] | |
| Kazinol J (CID: 21637732) | Anti-inflammation: [320] Anti-viral: [420] | |
| Broussonol E (CID: 10343070) | Anti-viral: [411] | |
| Broussoflavonol B (CID: 480828) | Anti-inflammation: [320] | |
| Others: polyphenols (broussochalcone A, papyriflavonol A, 3′-(3-methylbut-2-enyl)-3′,4′,7-trihydroxyflavane, kazinol F | Anti-inflammation: [456,457] Anti-viral: [420] | |
| Peppermint (hybrid mint, Mentha × piperita, or Mentha balsamea) | ß-Caryophyllene: 1.7% in oil [458] | Anti-inflammation [16,278,319] |
| 1,8-Cineole: 5.3% in oil [458]; 5.13% in oil [459]; 5.62% in oil [452] | Anti-inflammation [275,278,296,297,300,301,302,303,332] | |
| Limonene: 2.6% in oil [458]; 1.58% in oil [452] | Anti-inflammation [275,278,297] | |
| Menthol: 40% in oil [458]; 36.02% in oil [459]; 38.45% in oil [452] | Anti-inflammation [275,300,337,366,367] Anti-viral: [408] | |
| Menthone: 23.4% in oil [458]; 24.56% in oil [459]; 21.85% in oil [452] | Anti-inflammation [275,368] | |
| Rosemary (Rosmarinus officinalis) | Borneol: 3% in oil [432]; 4.08% to 8.17% depending on the location in Tunisia [460] | Anti-inflammation [275,278,319] |
| ß-Caryophyllene: 3.2% in oil [432] | Anti-inflammation [16,278,319] | |
| 1,8-Cineole (Eucalyptol): 43.1% in oil [432]; 33.08% to 37.75% depending on the location in Tunisia [460] | Anti-inflammation [275,278,296,297,300,301,302,303,332] | |
| Camphor: 11.3% in oil [432]; 13.55% to 18.13% depending on the location in Tunisia [460] | Anti-inflammation [278,296] | |
| α-Pinene: 11.4% in oil [432]; 8.58% to 9.32% depending on the location in Tunisia [460] | Anti-inflammation [278,337] | |
| Camphene: 5.0% in oil [432]; 3.58% to 5.07% depending on the location in Tunisia [460] | Anti-inflammation [322,328] | |
| Limonene: 2.6% in oil [432]; 2.99% to 3.19% depending on the location in Tunisia [460] | Anti-inflammation [275,278,297] | |
| Summer savory (Satureja hortensis) | Carvacrol: 11% to 67.0% depending on the location harvested [461]; 2.5% in oil [462] | Anti-inflammation [275,285,326,337] Anti-viral: [408] |
| P-Cymene: 3.4% to 11.7% in oil depending on the location harvested [461]; 6.30% [462] | Anti-inflammation [322,328] | |
| Γ-Terpinene: 15.30% to 38.7% depending on the location harvested [461]; 20.72% [462] | Anti-inflammation [278,360,386] | |
| ß-myrcene: 1.9% to 2.8% depending on the location harvested [461]; 1.98% [462] | Anti-inflammation [275,278,337] | |
| α-Terpinene: 1.29% to 4.9% depending on the location harvested [461]; 2.93% [462] | Anti-inflammation [275,360] | |
| 4-Terpineol: none to 1.6% depending on the location harvested [461]; 0.17% [462] | Anti-inflammation [275,278] Anti-viral: [408] | |
| Thymol: 23.12% [462]; none to 28.2% [461] | Anti-inflammation [275,319,337,389] Anti-viral: [408] | |
| Tea tree or Narrow-leaved paperbark (Melaleuca alternifolia) | ß-Caryophyllene: 2.2% in oil [432] | Anti-inflammation [16,278,319] |
| p-Cymene: 2.8% in oil [432]; 2.9% in oil [463] | Anti-inflammation [322,328] | |
| 1,8-Cineole (eucalyptol): 2.3% in oil [432]; 5.1% in oil [463] | Anti-inflammation [275,278,296,297,300,301,302,303,332] | |
| Limonene: 3.6% in oil [432]; 1.0% in oil [463] | Anti-inflammation [275,278,297] | |
| α-Pinene: 3.9% in oil [432] | Anti-inflammation [278,337] | |
| 4-Terpineol: 38.7% in oil [432]; 40.1% in oil [463] | Anti-inflammation [275,278] Anti-viral: [408] | |
| Γ-Terpenine: 16.3% in oil [432]; 23.0% in oil [463] | Anti-inflammation [241,318,344] | |
| α-Terpineol: 4.6% in oil [432]; 2.4% in oil [463] | Anti-inflammation [275,278,319,387] | |
| Tea plant (Camellia sinensis) | (+)-Catechin | Anti-inflammation [330] |
| (−)-Epicatechin | Anti-viral: [413] | |
| Epigallocatechin-3-gallate | Anti-inflammation [345] Anti-viral: [416] | |
| Thyme (Thymus vulgaris) | α-Phellandrene: 0.33% [464]; 0.3% [465]; 0.27% [466] | Anti-inflammation [278] |
| α-Pinene: 1.07% [464]; 0.8% [465]; 0.47% [466]; 1.6% in oil [432] | Anti-inflammation [278,337] | |
| α-Terpinene: 2.79% [466] | Anti-inflammation [275,360] | |
| γ-Terpinene: 30.9% [464]; 16.5% [465]; 29.12% [466]; 7.9% in oil [432] | Anti-inflammation [278,360,386] | |
| Thymol: 47.59% [464]; 44.7% [465]; 43.1% in oil [432] | Anti-inflammation [275,319,337,389] Anti-viral: [408] | |
| Turmeric (Curcuma longa) | Curcumin: Curcumin vs. demethoxycurcumin vs. bisdemothoxycurcumin is 80:15:5 but some reports suggest demethoxycurcumin has stronger bioactive potency [339] | Anti-inflammation: [339,340,341] Anti-viral: [341,412,413,467] |
| demethoxycurcumin | - | |
| Bisdemothoxycurcumin | - | |
| Cyclocurcumin | Anti-inflammation: [341,342] Anti-viral: [341] |
7.5. Phytochemicals for Neuropharmacological Effects
8. General Discussions
8.1. A New Smell Training Essential Oil Combination for COVID-19-Induced Anosmia/Dysnomia
| Essential Oil/Herbs/Plants/Fruits | Anti-Viral for SARS-CoV-1 or 2 | Anti-Inflammation | Comments |
|---|---|---|---|
| Ashitaba | 2 | - | |
| Bitter orange | 2 | 4 | |
| Cinnamon | 3 | 3 | |
| Clove | 0 | 4 | |
| Copaiba | 0 | 2 | Contains ß-caryophyllene, which is a CB2 ligand, at high %. Possibly good especially for tongue regeneration from the genes involved in turnover in the epidermis of skin and the pathways that ß-caryophyllene activates. |
| Cypress (hinoki) | 4 | 3 | |
| Elderberry | 2 | 3 | |
| Eucalyptus | 0 | 8 | |
| Geranium | 2 | 3 | |
| Ginger (fresh) | 1 | at least 2 | |
| Lavender | 2 | 10 | |
| Lemongrass | 1 | 9 | |
| Licorice | 2 | Many | |
| Mint | 1 | 5 | |
| Oregano | 2 | 5 | |
| Paper mulberry | Many | Many | |
| Peppermint | 1 | 5 | |
| Rosemary | 0 | 7 | |
| Summer savory | 3 | 7 | |
| Tea tree | 1 | 8 | |
| Tea plant | 2 | 1 | |
| Thyme | 2 | 9 | |
| Turmeric | 2 | Many |
8.2. A New Taste Training for COVID-19-Induced Ageusia/Dysgeusia
8.3. Possible Utilization of Phytochemicals as Supplementary Treatments in Clinical Settings
8.4. Possible Utilization of Phytochemicals for Prevention of Contracting SARS-CoV-2 and Treatment of COVID-19
8.5. Allergen Cautions
9. Concluding Remarks
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Oran, D.P.; Topol, E.J. Prevalence of Asymptomatic SARS-CoV-2 Infection. Ann. Intern. Med. 2020, 174, 286–287. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Gui, X.; Xiong, Y. Comparison of Clinical Characteristics of Patients with Asymptomatic vs Symptomatic Coronavirus Disease 2019 in Wuhan, China. JAMA Netw. Open. 2020, 3, e2010182. [Google Scholar] [CrossRef]
- Koyama, S.; Ueha, R.; Kondo, K. Loss of Smell and Taste in Patients with Suspected COVID-19: Analyses of Patients’ Reports on Social Media. J. Med. Internet Res. 2021, 23, e26459. [Google Scholar] [CrossRef]
- Lauring, A.S.; Hodcroft, E.B. Genetic Variants of SARS-CoV-2—What Do They Mean? JAMA 2020, 325, 529–531. [Google Scholar] [CrossRef]
- Plante, J.A.; Liu, Y.; Liu, J.; Xia, H.; Johnson, B.A.; Lokugamage, K.G.; Zhang, X.; Muruato, A.E.; Zou, J.; Fontes-Garfias, C.R.; et al. Spike Mutation D614G Alters SARS-CoV-2 Fitness. Nature 2020, 592, 116–121. [Google Scholar] [CrossRef]
- Tang, J.W.; Tambyah, P.A.; Hui, D.S. Emergence of a New SARS-CoV-2 Variant in the UK. J. Infect. 2021, 82, e27–e28. [Google Scholar] [CrossRef] [PubMed]
- Zhang, L.; Jackson, C.B.; Mou, H.; Ojha, A.; Peng, H.; Quinlan, B.D.; Rangarajan, E.S.; Pan, A.; Vanderheiden, A.; Suthar, M.S.; et al. SARS-CoV-2 Spike-Protein D614G Mutation Increases Virion Spike Density and Infectivity. Nat. Commun. 2020, 11, 6013. [Google Scholar] [CrossRef]
- Lechien, J.R.; Chiesa-Estomba, C.M.; De Siati, C.M.; Horoi, M.; Le Bon, S.D.; Rodriguez, A.; Dequanter, D.; Blecic, S.; Afia, F.E.; Distinguin, L.; et al. Olfactory and Gustatory Dysfunctions as a Clinical Presentation of Mild-to-moderate Forms of the Coronavirus Disease (COVID-19): A Multicenter European Study. Eur. Arch. Oto-Rhino-Laryngol. 2020, 277, 2251–2261. [Google Scholar] [CrossRef]
- Menni, C.; Valdes, A.; Freidin, M.B.; Ganesh, S.; Moustafa, J.E.-S.; Visconti, A.; Hysi, P.; Bowyer, R.C.E.; Mangino, M.; Falchi, M.; et al. Loss of Smell and Taste in Combination with Other Symptoms Is a Strong Predictor of COVID-19 Infection. medRxiv 2020. [Google Scholar] [CrossRef]
- Moein, S.T.; Hashemian, S.M.; Mansourafshar, B.; Khorram-Tousi, A.; Tabarsi, P.; Doty, R.L. Smell Dysfunction: A Biomarker for COVID-19. Int. Forum Allergy Rhinol. 2020, 10, 944–950. [Google Scholar] [CrossRef] [PubMed]
- Parma, V.; Ohla, K.; Veldhuizen, M.G.; Niv, M.Y.; Kelly, C.E.; Bakke, A.; Cooper, K.W.; Bouysset, C.; Pirastu, N.; Dibattista, M.; et al. More Than Smell-COVID-19 Is Associated With Severe Impairment of Smell, Taste, and Chemesthesis. Chem. Senses 2020, 45, 609–622. [Google Scholar] [CrossRef]
- Smith, B. Study Suggests 186,000 Hoosiers with COVID-19; Nearly Half without Symptoms; WFYI Public Media: Indianapolis, IN, USA, 2020. [Google Scholar]
- Hummel, T.; Rissom, K.; Reden, J.; Hahner, A.; Weidenbecher, M.; Huttenbrink, K.-B. Effects of Olfactory Training in Patients with Olfactory Loss. Laryngoscope 2009, 119, 496–499. [Google Scholar] [CrossRef]
- Gellrich, J.; Han, P.; Manesse, C.; Betz, A.; Junghanns, A.; Raue, C.; Schriever, V.A.; Hummel, T. Brain Volume Changes in Hyposmic Patients Before and After Olfactory Training. Laryngoscope 2018, 128, 1531–1536. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, R.; Han, P.; Reither, N.; Hummel, T. Effectiveness of Olfactory Training on Different Severities of Posttraumatic Loss of Smell. Laryngoscope 2019, 129, 1737–1743. [Google Scholar] [CrossRef]
- Koyama, S.; Purk, A.; Kaur, M.; Soini, H.A.; Novotny, M.V.; Davis, K.; Kao, C.C.; Matsunami, H.; Mescher, A. Beta-Caryophyllene Enhances Wound Healing through Multiple Routes. PLoS ONE 2019, 14, e0216104. [Google Scholar] [CrossRef]
- Koyama, S.; Heinbockel, T. The Effects of Essential Oils and Terpenes in Relation to Their Routes of Intake and Application. Int. J. Mol. Sci. 2020, 21, 1558. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-J.; Tabakoff, B.; Levinson, S.R.; Heinbockel, T. Inhibition of Nav1.7 Channels by Methyl Eugenol as a Mechanism Underlying Its Antinociceptive and Anesthetic Actions. Acta Pharmacol. Sin. 2015, 36, 791–799. [Google Scholar] [CrossRef] [PubMed]
- Harada, H.; Kashiwadani, H.; Kanmura, Y.; Kuwaki, T. Linalool Odor-Induced Anxiolytic Effects in Mice. Front. Behav. Neurosci. 2018, 12, 241. [Google Scholar] [CrossRef]
- Riera, C.; Menozzi-Smarrito, C.; Affolter, M.; Michlig, S.; Munari, C.; Robert, F.; Vogel, H.; Simon, S.A.; le Coutre, J. Compounds from Sichuan and Melegueta Peppers Activate, Covalently and Non-Covalently, TRPA1 and TRPV1 Channels. Br. J. Pharmacol. 2009, 157, 1398–1409. [Google Scholar] [CrossRef]
- Barrera-Sandoval, A.M.; Osorio, E.; Cardona-Gomez, G.P. Microglial-Targeting Induced by Intranasal Linalool during Neurological Protection Postischemia. Eur. J. Pharmacol. 2019, 857, 172420. [Google Scholar] [CrossRef] [PubMed]
- Behrendt, H.-J.; Germann, T.; Gillen, C.; Hatt, H.; Jostock, R. Characterization of the Mouse Cold-Menthol Receptor TRPM8 and Vanilloid Receptor Type-1 VR1 Using a Fluorometric Imaging Plate Reader (FLIPR) Assay: Characterization of TRPM8 and VR1. Br. J. Pharmacol. 2004, 141, 737–745. [Google Scholar] [CrossRef]
- Seiden, A.M. Postviral Olfactory Loss. Otolaryngol. Clin. N. Am. 2004, 37, 1159–1166. [Google Scholar] [CrossRef] [PubMed]
- Welge-Lussen, A.; Wolfensberger, M. Olfactory Disorders Following Upper Respiratory Tract Infections. Adv. Otorhinolaryngol. 2006, 63, 125–132. [Google Scholar] [CrossRef] [PubMed]
- Mori, J.; Aiba, T.; Sugiura, M.; Matsumoto, K.; Tomiyama, K.; Okuda, F.; Okigaki, S.; Nakai, Y. Clinical Study of Olfactory Disturbance. Acta Otolaryngol. Suppl. 1998, 538, 197–201. [Google Scholar] [PubMed]
- Deems, D.A.; Doty, R.L.; Settle, R.G.; Moore-Gillon, V.; Shaman, P.; Mester, A.F.; Kimmelman, C.P.; Brightman, V.J.; Snow, J.B., Jr. Smell and Taste Disorders, a Study of 750 Patients from the University of Pennsylvania Smell and Taste Center. Arch. Otolaryngol. Head Neck Surg. 1991, 117, 519–528. [Google Scholar] [CrossRef]
- Fark, T.; Hummel, T. Olfactory Disorders: Distribution According to Age and Gender in 3400 Patients. Eur. Arch. Otorhinolaryngol. 2013, 270, 777–779. [Google Scholar] [CrossRef] [PubMed]
- Sugiura, M.; Alba, T.; Mori, J.; Nakai, Y. An Epidemiological Study of Postviral Olfactory Disorder. Acta Otolaryngol. Suppl. 1998, 538, 191–196. [Google Scholar] [CrossRef] [PubMed]
- Reden, J.; Maroldt, H.; Fritz, A.; Zahnert, T.; Hummel, T. A Study on the Prognostic Significance of Qualitative Olfactory Dysfunction. Eur. Arch. Otorhinolaryngol. 2007, 264, 139–144. [Google Scholar] [CrossRef]
- Vaira, L.A.; Lechien, J.R.; Khalife, M.; Petrocelli, M.; Hans, S.; Distinguin, L.; Salzano, G.; Cucurullo, M.; Doneddu, P.; Salzano, F.A.; et al. Psychophysical Evaluation of the Olfactory Function: European Multicenter Study on 774 COVID-19 Patients. Pathogens 2021, 10, 62. [Google Scholar] [CrossRef]
- Liu, D.T.; Sabha, M.; Damm, M.; Philpott, C.; Oleszkiewicz, A.; Hahner, A.; Hummel, T. Parosmia Is Associated with Relevant Olfactory Recovery After Olfactory Training. Laryngoscope 2020, 131, 618–623. [Google Scholar] [CrossRef]
- Yamagishi, M.; Fujiwara, M.; Nakamura, H. Olfactory Mucosal Findings and Clinical Course in Patients with Olfactory Disorders Following Upper Respiratory Viral Infection. Rhinology 1994, 32, 113–118. [Google Scholar]
- Jafek, B.W.; Murrow, B.; Michaels, R.; Restrepo, D.; Linschoten, M. Biopsies of Human Olfactory Epithelium. Chem. Senses 2002, 27, 623–628. [Google Scholar] [CrossRef] [PubMed]
- Lane, A.P.; Turner, J.; May, L.; Reed, R. A Genetic Model of Chronic Rhinosinusitis-Associated Olfactory Inflammation Reveals Reversible Functional Impairment and Dramatic Neuroepithelial Reorganization. J. Neurosci. 2010, 30, 2324–2329. [Google Scholar] [CrossRef] [PubMed]
- Kanaya, K.; Kondo, K.; Suzukawa, K.; Sakamoto, T.; Kikuta, S.; Okada, K.; Yamasoba, T. Innate Immune Responses and Neuroepithelial Degeneration and Regeneration in the Mouse Olfactory Mucosa Induced by Intranasal Administration of Poly(I:C). Cell Tissue Res 2014, 357, 279–299. [Google Scholar] [CrossRef] [PubMed]
- Imamura, F.; Hasegawa-Ishii, S. Environmental Toxicants-Induced Immune Responses in the Olfactory Mucosa. Front. Immunol. 2016, 7, 475. [Google Scholar] [CrossRef]
- Suzuki, M.; Saito, K.; Min, W.-P.; Vladau, C.; Toida, K.; Itoh, H.; Murakami, S. Identification of Viruses in Patients With Postviral Olfactory Dysfunction. Laryngoscope 2007, 117, 272–277. [Google Scholar] [CrossRef]
- Tian, J.; Pinto, J.M.; Li, L.; Zhang, S.; Sun, Z.; Wei, Y. Identification of Viruses in Patients With Postviral Olfactory Dysfunction by Multiplex Reverse-Transcription Polymerase Chain Reaction. Laryngoscope 2021, 131, 158–164. [Google Scholar] [CrossRef]
- Konstantinidis, I.; Haehner, A.; Frasnelli, J.; Reden, J.; Quante, G.; Damm, M.; Hummel, T. Post-Infectious Olfactory Dysfunction Exhibits a Seasonal Pattern. Rhinology 2006, 44, 135–139. [Google Scholar]
- Wang, J.H.; Kwon, H.J.; Jang, Y.J. Detection of Parainfluenza Virus 3 in Turbinate Epithelial Cells of Postviral Olfactory Dysfunction Patients. Laryngoscope 2007, 117, 1445–1449. [Google Scholar] [CrossRef] [PubMed]
- Potter, M.R.; Chen, J.H.; Lobban, N.-S.; Doty, R.L. Olfactory Dysfunction from Acute Upper Respiratory Infections: Relationship to Season of Onset. Int. Forum Allergy Rhinol. 2020, 10, 706–712. [Google Scholar] [CrossRef]
- Gamage, A.M.; Tan, K.S.; Chan, W.O.Y.; Liu, J.; Tan, C.W.; Ong, Y.K.; Thong, M.; Andiappan, A.K.; Anderson, D.E.; Wang, D.Y.; et al. Infection of Human Nasal Epithelial Cells with SARS-CoV-2 and a 382-Nt Deletion Isolate Lacking ORF8 Reveals Similar Viral Kinetics and Host Transcriptional Profiles. PLoS Pathog. 2020, 16, e1009130. [Google Scholar] [CrossRef] [PubMed]
- Lowe, J.S.; Anderson, P.G. Alimentary Tract. In Stevens Lowes Human Histology; Elsevier: Amsterdam, The Netherlands, 2015; pp. 186–224. ISBN 978-0-7234-3502-0. [Google Scholar]
- Stiblar-Martincic, D. Histology of Laryngeal Mucosa. Acta Otolaryngol. Suppl. 1997, 527, 138–141. [Google Scholar] [CrossRef]
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Müller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C.; et al. Virological Assessment of Hospitalized Patients with COVID-2019. Nature 2020, 581, 465–469. [Google Scholar] [CrossRef] [PubMed]
- Maltezou, H.C.; Raftopoulos, V.; Vorou, R.; Papadima, K.; Mellou, K.; Spanakis, N.; Kossyvakis, A.; Gioula, G.; Exindari, M.; Froukala, E.; et al. Association between Upper Respiratory Tract Viral Load, Comorbidities, Disease Severity and Outcome of Patients with SARS-CoV-2 Infection. J. Infect. Dis. 2021, 223, 1132–1138. [Google Scholar] [CrossRef] [PubMed]
- Yilmaz, A.; Marklund, E.; Andersson, M.; Nilsson, S.; Andersson, L.-M.; Lindh, M.; Gisslén, M. Upper Respiratory Tract Levels of Severe Acute Respiratory Syndrome Coronavirus 2 RNA and Duration of Viral RNA Shedding Do Not Differ Between Patients With Mild and Severe/Critical Coronavirus Disease 2019. J. Infect. Dis. 2021, 223, 15–18. [Google Scholar] [CrossRef] [PubMed]
- Cevik, M.; Tate, M.; Lloyd, O.; Maraolo, A.E.; Schafers, J.; Ho, A. SARS-CoV-2, SARS-CoV, and MERS-CoV Viral Load Dynamics, Duration of Viral Shedding, and Infectiousness: A Systematic Review and Meta-Analysis. Lancet Microbe 2021, 2, e13–e22. [Google Scholar] [CrossRef]
- Murphy, K. SARS CoV-2 Detection From Upper and Lower Respiratory Tract Specimens: Diagnostic and Infection Control Implications. Chest 2020, 158, 1804–1805. [Google Scholar] [CrossRef]
- Liu, L.; Wei, Q.; Alvarez, X.; Wang, H.; Du, Y.; Zhu, H.; Jiang, H.; Zhou, J.; Lam, P.; Zhang, L.; et al. Epithelial Cells Lining Salivary Gland Ducts Are Early Target Cells of Severe Acute Respiratory Syndrome Coronavirus Infection in the Upper Respiratory Tracts of Rhesus Macaques. J. Virol. 2011, 85, 4025–4030. [Google Scholar] [CrossRef]
- Bertram, S.; Heurich, A.; Lavender, H.; Gierer, S.; Danisch, S.; Perin, P.; Lucas, J.M.; Nelson, P.S.; Pöhlmann, S.; Soilleux, E.J. Influenza and SARS-Coronavirus Activating Proteases TMPRSS2 and HAT Are Expressed at Multiple Sites in Human Respiratory and Gastrointestinal Tracts. PLoS ONE 2012, 7, e35876. [Google Scholar] [CrossRef]
- Descamps, G.; Verset, L.; Trelcat, A.; Hopkins, C.; Lechien, J.R.; Journe, F.; Saussez, S. ACE2 Protein Landscape in the Head and Neck Region: The Conundrum of SARS-CoV-2 Infection. Biology 2020, 9, 235. [Google Scholar] [CrossRef]
- Sato, T.; Ueha, R.; Goto, T.; Yamauchi, A.; Kondo, K.; Yamasoba, T. Expression of ACE2 and TMPRSS2 Proteins in the Upper and Lower Aerodigestive Tracts of Rats. Laryngology 2021, 131, E932–E939. [Google Scholar]
- El-Anwar, M.W.; Elzayat, S.; Fouad, Y.A. ENT Manifestation in COVID-19 Patients. Auris Nasus Larynx 2020, 47, 559–564. [Google Scholar] [CrossRef]
- Lovato, A.; de Filippis, C. Clinical Presentation of COVID-19: A Systematic Review Focusing on Upper Airway Symptoms. Ear Nose Throat J. 2020, 99, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Asiaee, M.; Vahedian-azimi, A.; Atashi, S.S.; Keramatfar, A.; Nourbakhsh, M. Voice Quality Evaluation in Patients With COVID-19: An Acoustic Analysis. J. Voice 2020. [Google Scholar] [CrossRef] [PubMed]
- Cooper, K.W.; Brann, D.H.; Farruggia, M.C.; Bhutani, S.; Pellegrino, R.; Tsukahara, T.; Weinreb, C.; Joseph, P.V.; Larson, E.D.; Parma, V.; et al. COVID-19 and the Chemical Senses: Supporting Players Take Center Stage. Neuron 2020, 107, 219–233. [Google Scholar] [CrossRef] [PubMed]
- Zhou, P.; Yang, X.-L.; Wang, X.-G.; Hu, B.; Zhang, L.; Zhang, W.; Si, H.-R.; Zhu, Y.; Li, B.; Huang, C.-L.; et al. A Pneumonia Outbreak Associated with a New Coronavirus of Probable Bat Origin. Nature 2020, 579, 270–273. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, M.; Kleine-Weber, H.; Schroeder, S.; Krueger, N.; Herrier, T.; Erichsen, S.; Schiergens, T.S.; Herrier, G.; Wu, N.-H.; Nitsche, A.; et al. SARS-CoV-2 Cell Entry Depends on ACE2 and TMPRSS2 and Is Blocked by a Clinically Proven Protease Inhibitor. Cell 2020, 181, 271–280. [Google Scholar] [CrossRef]
- Bestle, D.; Heindl, M.R.; Limburg, H.; Lam van, T.V.; Pilgram, O.; Moulton, H.; Stein, D.A.; Hardes, K.; Eickmann, M.; Dolnik, O.; et al. TMPRSS2 and Furin Are Both Essential for Proteolytic Activation of SARS-CoV-2 in Human Airway Cells. Life Sci. Alliance 2020, 3, e202000786. [Google Scholar] [CrossRef] [PubMed]
- Brann, D.H.; Tsukahara, T.; Wenreb, C.; Lipovsek, M.; van den Berge, K.; Gong, B.; Chance, R.; Macaulay, I.C.; Chou, H.-J.; Fletcher, R.B.; et al. Non-Neuronal Expression of SARS-CoV-2 Entry Genes in the Olfactory System Suggests Mechanisms Underlying COVID-19-Associated Anosmia. Sci. Adv. 2020, 6, eabc5801. [Google Scholar] [CrossRef]
- Chen, M.; Shen, W.; Rowan, N.R.; Kulaga, H.; Hillel, A.; Ramanathan, M.; Lane, A.P. Elevated ACE2 Expression in the Olfactory Neuroepithelium: Implications for Anosmia and Upper Respiratory SARS-CoV-2 Entry and Replication. Eur. Respir. J. 2020, 56, 2001948. [Google Scholar] [CrossRef]
- Ueha, R.; Kondo, K.; Kagoya, R.; Shichino, S.; Ueha, S.; Yamasoba, T. ACE2, TMPRSS2, and Furin Expression in the Nose and Olfactory Bulb in Mice and Human. Rhinology 2021, 59, 105–109. [Google Scholar] [CrossRef] [PubMed]
- Fodoulian, L.; Tuberosa, J.; Rossier, D.; Boillat, M.; Kan, C.; Pauli, V.; Egervari, K.; Lobrinus, J.A.; Landis, B.N.; Carleton, A.; et al. SARS-CoV-2 Receptors and Entry Genes Are Expressed in the Human Olfactory Neuroepithelium and Brain. iScience 2020, 23, 101839. [Google Scholar] [CrossRef]
- Ye, Q.; Zhou, J.; Yang, G.; Li, R.-T.; He, Q.; Zhang, Y.; Wu, S.-J.; Chen, Q.; Shi, J.-H.; Zhang, R.-R.; et al. SARS-CoV-2 Infection Causes Transient Olfactory Dysfunction in Mice. bioRxiv 2020. [Google Scholar] [CrossRef]
- Eshraghi, A.A.; Mirsaeidi, M.; Davies, C.; Telischi, F.F.; Chaudhari, N.; Mittal, R. Potential Mechanisms for COVID-19 Induced Anosmia and Dysgeusia. Front. Physiol. 2020, 11, 1039. [Google Scholar] [CrossRef]
- Torabi, A.; Mohammadbagheri, E.; Dilmaghani, N.A.; Bayat, A.-H.; Fathi, M.; Vakili, K.; Alizadeh, R.; Rezaeimirghaed, O.; Hajiesmaeili, M.; Ramezani, M.; et al. Proinflammatory Cytokines in the Olfactory Mucosa Result in COVID-19 Induced Anosmia. ACS Chem. Neurosci. 2020, 11, 1909–1913. [Google Scholar] [CrossRef] [PubMed]
- De Melo, G.D.; Lazarini, F.; Levallois, S.; Hautefort, C.; Michel, V.; Larrous, F.; Verillaud, B.; Aparicio, C.; Wagner, S.; Gheusi, G.; et al. COVID-19-Associated Olfactory Dysfunction Reveals SARS-CoV-2 Neuroinvasion and Persistence in the Olfactory System. bioRxiv 2020. [Google Scholar] [CrossRef]
- Song, E.; Zhang, C.; Israelow, B.; Lu-Culligan, A.; Prado, A.V.; Skriabine, S.; Lu, P.; Weizman, O.-E.; Liu, F.; Dai, Y.; et al. Neuroinvasion of SARS-CoV-2 in Human and Mouse Brain. J. Exp. Med. 2021, 218, e2020135. [Google Scholar] [CrossRef]
- Thaker, S.K.; Ch’ng, J.; Christofk, H.R. Viral Hijacking of Cellular Metabolism. BMC Biol. 2019, 17, 59. [Google Scholar] [CrossRef] [PubMed]
- Keshavarz, M.; Solaymani-Mohammadi, F.; Namdari, H.; Arjeini, Y.; Mousavi, M.J.; Rezaei, F. Metabolic Host Response and Therapeutic Approaches to Influenza Infection. Cell Mol. Biol. Lett. 2020, 25, 15. [Google Scholar] [CrossRef]
- Moreno-Altamirano, M.M.B.; Kolstoe, S.E.; Sánchez-García, F.J. Virus Control of Cell Metabolism for Replication and Evasion of Host Immune Responses. Front. Cell. Infect. Microbiol. 2019, 9, 95. [Google Scholar] [CrossRef]
- Martín-Vicente, M.; González-Riaño, C.; Barbas, C.; Jiménez-Sousa, M.Á.; Brochado-Kith, O.; Resino, S.; Martínez, I. Metabolic Changes during Respiratory Syncytial Virus Infection of Epithelial Cells. PLoS ONE 2020, 15, e0230844. [Google Scholar] [CrossRef]
- Cheng, M.-L.; Chien, K.-Y.; Lai, C.-H.; Li, G.-J.; Lin, J.-F.; Ho, H.-Y. Metabolic Reprogramming of Host Cells in Response to Enteroviral Infection. Cells 2020, 9, 473. [Google Scholar] [CrossRef]
- Mullen, P.J.; Garcia, G., Jr.; Purkayastha, A.; Matulionis, N.; Schmid, E.W.; Momcilovic, M.; Sen, C.; Langerman, J.; Ramaiah, A.; Shackelford, D.B.; et al. SARS-CoV-2 Infection Rewires Host Cell Metabolism and Is Potentially Susceptible to MTORC1 Inhibition. Nat. Commun. 2021, 12, 1876. [Google Scholar] [CrossRef]
- Awasthi, M.; Gulati, S.; Sarkar, D.P.; Tiwari, S.; Kateriya, S.; Ranjan, P.; Verma, S.K. The Sialoside-Binding Pocket of SARS-CoV-2 Spike Glycoprotein Structurally Resembles MERS-CoV. Viruses 2020, 12, 909. [Google Scholar] [CrossRef]
- Campbell, F.; Archer, B.; Laurenson-Schafer, H.; Jinnai, Y.; Konings, F.; Batra, N.; Pavlin, B.; Vandemaele, K.; Van Kerkhove, M.D.; Jombart, T.; et al. Increased Transmissibility and Global Spread of SARS-CoV-2 Variants of Concern as at June 2021. Eurosurveillance 2021, 26, 2100509. [Google Scholar] [CrossRef]
- Han, Y.; Wang, Z.; Wei, Z.; Schapiro, I.; Li, J. Binding Affinity and Mechanisms of SARS-CoV-2 Variants. Comput. Struct. Biotechnol. J. 2021, 19, 4184–4191. [Google Scholar] [CrossRef] [PubMed]
- Kim, S.; Liu, Y.; Lei, Z.; Dicker, J.; Cao, Y.; Zhang, X.F.; Im, W. Differential Interactions Between Human ACE2 and Spike RBD of SARS-CoV-2 Variants of Concern. Biophysics 2021. [Google Scholar] [CrossRef]
- Bakhshandeh, B.; Sorboni, S.G.; Javanmard, A.-R.; Mottaghi, S.S.; Mehrabi, M.-R.; Sorouri, F.; Abbasi, A.; Jahanafrooz, Z. Variants in ACE2; Potential Influences on Virus Infection and COVID-19 Severity. Infect. Genet. Evol. 2021, 90, 104773. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Hu, G.; Wang, Y.; Ren, W.; Zhao, X.; Ji, F.; Zhu, Y.; Feng, F.; Gong, M.; Ju, X.; et al. Functional and Genetic Analysis of Viral Receptor ACE2 Orthologs Reveals a Broad Potential Host Range of SARS-CoV-2. Proc. Natl. Acad. Sci. USA 2021, 118, e2025373118. [Google Scholar] [CrossRef]
- Wang, R.; Simoneau, C.R.; Kulsuptrakul, J.; Bouhaddou, M.; Travisano, K.A.; Hayashi, J.M.; Carlson-Stevermer, J.; Zengel, J.R.; Richards, C.M.; Fozouni, P.; et al. Genetic Screens Identify Host Factors for SARS-CoV-2 and Common Cold Coronaviruses. Cell 2021, 184, 106–119.e14. [Google Scholar] [CrossRef] [PubMed]
- Cantuti-Castelvetri, L.; Ojha, R.; Pedro, L.D.; Djannatian, M.; Franz, J.; Kuivanen, S.; van der Meer, F.; Kallio, K.; Kaya, T.; Anastasina, M.; et al. Neuropilin-1 Facilitates SARS-CoV-2 Cell Entry and Infectivity. Science 2020, 370, 856–860. [Google Scholar] [CrossRef] [PubMed]
- Cuervo, N.Z.; Grandvaux, N. ACE2: Evidence of Role as Entry Receptor for SARS-CoV-2 and Implications in Comorbidities. eLife 2020, 9, e61390. [Google Scholar] [CrossRef] [PubMed]
- Sigrist, C.J.; Bridge, A.; Le Mercier, P. A Potential Role for Integrins in Host Cell Entry by SARS-CoV-2. Antivir. Res. 2020, 177, 104759. [Google Scholar] [CrossRef] [PubMed]
- Kliche, J.; Kuss, H.; Ali, M.; Ivarsson, Y. Cytoplasmic Short Linear Motifs in ACE2 and Integrin β3 Link SARS-CoV-2 Host Cell Receptors to Mediators of Endocytosis and Autophagy. Sci. Signal. 2021, 14, eabf1117. [Google Scholar] [CrossRef]
- Dakal, T.C. SARS-CoV-2 Attachment to Host Cells Is Possibly Mediated via RGD-Integrin Interaction in a Calcium-Dependent Manner and Suggests Pulmonary EDTA Chelation Therapy as a Novel Treatment for COVID 19. Immunobiology 2021, 226, 152021. [Google Scholar] [CrossRef] [PubMed]
- Nolte, M.A.; Hoen, E.N.M.N.; Margadant, C. Integrins Control Vesicular Trafficking; New Tricks for Old Dogs. Trends Biochem. Sci. 2021, 46, 124–137. [Google Scholar] [CrossRef]
- Hussein, H.A.M.; Walker, L.R.; Abdel-Raouf, U.M.; Desouky, S.A.; Montasser, A.K.M.; Akula, S.M. Beyond RGD: Virus Interactions with Integrins. Arch. Virol. 2015, 160, 2669–2681. [Google Scholar] [CrossRef]
- Yang, N.; Shen, H.-M. Targeting the Endocytic Pathway and Autophagy Process as a Novel Therapeutic Strategy in COVID-19. Int. J. Biol. Sci. 2020, 16, 1724–1731. [Google Scholar] [CrossRef]
- Morniroli, D.; Giannì, M.L.; Consales, A.; Pietrasanta, C.; Mosca, F. Human Sialome and Coronavirus Disease-2019 (COVID-19) Pandemic: An Understated Correlation? Front. Immunol. 2020, 11, 1480. [Google Scholar] [CrossRef]
- Qing, E.; Hantak, M.; Perlman, S.; Tom, G. Distinct Roles for Sialoside and Protein Receptors in Coronavirus Infection. mBio 2020, 11, e02764-19. [Google Scholar] [CrossRef]
- Wielgat, P.; Rogowski, K.; Godlewska, K.; Car, H. Coronaviruses: Is Sialic Acid a Gate to the Eye of Cytokine Storm? From the Entry to the Effects. Cells 2020, 9, 1963. [Google Scholar] [CrossRef] [PubMed]
- Seyran, M.; Takayama, K.; Uversky, V.N.; Lundstrom, K.; Palù, G.; Sherchan, S.P.; Attrish, D.; Rezaei, N.; Aljabali, A.A.A.; Ghosh, S.; et al. The Structural Basis of Accelerated Host Cell Entry by SARS-CoV-2. FEBS J. 2020. [Google Scholar] [CrossRef]
- Walther, T.; Karamanska, R.; Chan, R.W.Y.; Chan, M.C.W.; Jia, N.; Air, G.; Hopton, C.; Wong, M.P.; Dell, A.; Peiris, J.S.M.; et al. Glycomic Analysis of Human Respiratory Tract Tissues and Correlation with Influenza Virus Infection. PLoS Pathog. 2013, 9, e1003223. [Google Scholar] [CrossRef]
- Kuchipudi, S.V.; Nelli, R.K.; Gontu, A.; Satyakumar, R.; Nair, M.S.; Subbiah, M. Sialic Acid Receptors: The Key to Solving the Enigma of Zoonotic Virus Spillover. Viruses 2021, 13, 262. [Google Scholar] [CrossRef]
- Kumlin, U.; Olofsson, S.; Dimock, K.; Arnberg, N. Sialic Acid Tissue Distribution and Influenza Virus Tropism. Influenza Other Respir. Viruses 2008, 2, 147–154. [Google Scholar] [CrossRef]
- Bushdid, C.; Magnasco, M.O.; Vosshall, L.B.; Keller, A. Humans Can Discriminate More than 1 Trillion Olfactory Stimuli. Science 2014, 343, 1370–1372. [Google Scholar] [CrossRef]
- Sokpor, G.; Abbas, E.; Rosenbusch, J.; Staiger, J.F.; Tuoc, T. Transcriptional and Epigenetic Control of Mammalian Olfactory Epithelium Development. Mol. Neurobiol. 2018, 55, 8306–8327. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Suzukawa, K.; Sakamoto, T.; Watanabe, K.; Kanaya, K.; Ushio, M.; Yamaguchi, T.; Nibu, K.-I.; Kaga, K.; Yamasoba, T. Age-Related Changes in Cell Dynamics of the Postnatal Mouse Olfactory Neuroepithelium: Cell Proliferation, Neuronal Differentiation, and Cell Death. J. Comp. Neurol. 2010, 518, 1962–1975. [Google Scholar] [CrossRef]
- Ueha, R.; Mukherjee, S.; Ueha, S.; Nagata, D.E.A.; Sakamoto, T.; Kondo, K.; Yamasoba, T.; Lukacs, N.W.; Kunkel, S.L. Viral Disruption of Olfactory Progenitors Is Exacerbated in Allergic Mice. Int. Immunopharmacol. 2014, 22, 242–247. [Google Scholar] [CrossRef]
- Buck, L.; Axel, R. A Novel Multigene Family May Encode Odorant Receptors: A Molecular Basis for Odor Recognition. Cell 1991, 65, 175–187. [Google Scholar] [CrossRef]
- Buck, L.B. Unraveling the Sense of Smell (Nobel Lecture). Angew. Chem. Int. Ed. 2005, 44, 6128–6140. [Google Scholar] [CrossRef]
- Axel, R. Scents and Sensibility: A Molecular Logic of Olfactory Perception (Nobel Lecture). Angew. Chem. Int. Ed. 2005, 44, 6110–6127. [Google Scholar] [CrossRef]
- Young, J.M.; Friedman, C.; Williams, E.M.; Ross, J.A.; Tonnes-Priddy, L.; Trask, B.J. Different Evolutionary Processes Shaped the Mouse and Human Olfactory Receptor Gene Families. Hum. Mol. Genet. 2002, 11, 535–546. [Google Scholar] [CrossRef]
- Gilad, Y.; Lancet, D. Population Differences in the Human Functional Olfactory Repertoire. Mol. Biol. Evol. 2003, 20, 307–314. [Google Scholar] [CrossRef] [PubMed]
- Niimura, Y. Evolutionary Dynamics of Olfactory Receptor Genes in Chordates: Interaction between Environments and Genomic Contents. Hum. Genom. 2009, 4, 107–118. [Google Scholar] [CrossRef] [PubMed]
- Mainland, J.D.; Keller, A.; Li, Y.R.; Zhou, T.; Trimmer, C.; Snyder, L.L.; Moberly, A.H.; Adipietro, K.A.; Liu, W.L.L.; Zhuang, H.; et al. The Missense of Smell: Functional Variability in the Human Odorant Receptor Repertoire. Nat. Neurosci. 2014, 17, 114–120. [Google Scholar] [CrossRef]
- Hayden, S.; Teeling, E.C. The Molecular Biology of Vertebrate Olfaction: Molecular Biology Of Vertebrate Olfaction. Anat. Rec. 2014, 297, 2216–2226. [Google Scholar] [CrossRef]
- Serizawa, S.; Miyamichi, K.; Nakatani, H.; Suzuki, M.; Saito, M.; Yoshihara, Y.; Sakano, H. Negative Feedback Regulation Ensures the One Receptor-One Olfactory Neuron Rule in Mouse. Science 2003, 302, 2088–2094. [Google Scholar] [CrossRef] [PubMed]
- Ressler, K.J.; Sullivan, S.L.; Buck, L.B. A Zonal Organization of Odorant Receptor Gene Expression in the Olfactory Epithelium. Cell 1993, 73, 597–609. [Google Scholar] [CrossRef]
- Buck, L.B. Receptor Diversity and Spatial Patterning in the Mammalian Olfactory System. In Novartis Foundation Symposia; Chadwick, D., Marsh, J., Goode, J., Eds.; John Wiley & Sons, Ltd.: Chichester, UK, 2007; pp. 51–67. ISBN 978-0-470-51451-1. [Google Scholar]
- Sullivan, S.L.; Ressler, K.J.; Buck, L.B. Odorant Receptor Diversity and Patterned Gene Expression in the Mammalian Olfactory Epithelium. Prog. Clin. Biol. Res. 1994, 390, 75–84. [Google Scholar]
- Yoshihara, Y.; Kawasaki, M.; Tamada, A.; Fujita, H.; Hayashi, H.; Kagamiyama, H.; Mori, K. OCAM: A New Member of the Neural Cell Adhesion Molecule Family Related to Zone-to-Zone Projection of Olfactory and Vomeronasal Axons. J. Neurosci. 1997, 17, 5830–5842. [Google Scholar] [CrossRef] [PubMed]
- Mombaerts, P. Axonal Wiring in the Mouse Olfactory System. Annu. Rev. Cell Dev. Biol. 2006, 22, 713–737. [Google Scholar] [CrossRef] [PubMed]
- Klenoff, J.R.; Greer, C.A. Postnatal Development of Olfactory Receptor Cell Axonal Arbors. J. Comp. Neurol. 1998, 390, 256–267. [Google Scholar] [CrossRef]
- Sharp, F.R.; Kauer, J.S.; Shepherd, G.M. Local Sites of Activity-Related Glucose Metabolism in Rat Olfactory Bulb during Olfactory Stimulation. Brain Res. 1975, 98, 596–600. [Google Scholar] [CrossRef]
- Friedrich, R.W.; Korsching, S.I. Combinatorial and Chemotopic Odorant Coding in the Zebrafish Olfactory Bulb Visualized by Optical Imaging. Neuron 1997, 18, 737–752. [Google Scholar] [CrossRef]
- Buck, L.B. Information Coding in the Vertebrate Olfactory System. Annu. Rev. Neurosci. 1996, 19, 517–544. [Google Scholar] [CrossRef]
- Buonviso, N.; Chaput, M.A. Response Similarity to Odors in Olfactory Bulb Output Cells Presumed to Be Connected to the Same Glomerulus: Electrophysiological Study Using Simultaneous Single-Unit Recordings. J. Neurophysiol. 1990, 63, 447–454. [Google Scholar] [CrossRef]
- Mombaerts, P. Targeting Olfaction. Curr. Opin. Neurobiol. 1996, 6, 481–486. [Google Scholar] [CrossRef]
- McQuiston, A.R.; Katz, L.C. Electrophysiology of Interneurons in the Glomerular Layer of the Rat Olfactory Bulb. J. Neurophysiol. 2001, 86, 1899–1907. [Google Scholar] [CrossRef] [PubMed]
- Pinching, A.J.; Powell, T.P. The Neuropil of the Periglomerular Region of the Olfactory Bulb. J. Cell Sci. 1971, 9, 379–409. [Google Scholar] [CrossRef] [PubMed]
- Pinching, A.J.; Powell, T.P. The Neuron Types of the Glomerular Layer of the Olfactory Bulb. J. Cell Sci. 1971, 9, 305–345. [Google Scholar] [CrossRef]
- White, E.L. Synaptic Organization in the Olfactory Glomerulus of the Mouse. Brain Res. 1972, 37, 69–80. [Google Scholar] [CrossRef]
- White, E.L. Synaptic Organization of the Mammalian Olfactory Glomerulus: New Findings Including an Intraspecific Variation. Brain Res. 1973, 60, 299–313. [Google Scholar] [CrossRef]
- Shipley, M.T.; Ennis, M. Functional Organization of Olfactory System. J. Neurobiol. 1996, 30, 123–176. [Google Scholar] [CrossRef]
- Pinching, A.J.; Powell, T.P. The Neuropil of the Glomeruli of the Olfactory Bulb. J. Cell Sci. 1971, 9, 347–377. [Google Scholar] [CrossRef] [PubMed]
- Malnic, B.; Hirono, J.; Sato, T.; Buck, L.B. Combinatorial Receptor Codes for Odors. Cell 1999, 96, 713–723. [Google Scholar] [CrossRef]
- Shepherd, G.M.; Chen, W.R.; Greer, C.A. Olfactory Bulb. In Synaptic Organization of the Brain; Oxford University Press: New York, NY, USA, 2004; pp. 165–216. [Google Scholar]
- Ennis, M.; Hamilton, K.A.; Hayar, A. Neurochemistry of the Main Olfactory System. In Handbook of Neurochemistry and Molecular Neurobiology; Lajtha, A., Johnson, D.A., Eds.; Springer: Boston, MA, USA, 2007; pp. 137–204. ISBN 978-0-387-30349-9. [Google Scholar]
- Kiselycznyk, C.L.; Zhang, S.; Linster, C. Role of Centrifugal Projections to the Olfactory Bulb in Olfactory Processing. Learn. Mem. 2006, 13, 575–579. [Google Scholar] [CrossRef][Green Version]
- Swanson, L.W. Brain maps: Structure of rat brain. In A Laboratory Guide with Printed and Electronic Templates for Data, Models and Schematics; Elsevier: Amsterdam, The Neterlands, 2004. [Google Scholar]
- Laaris, N.; Puche, A.; Ennis, M. Complementary Postsynaptic Activity Patterns Elicited in Olfactory Bulb by Stimulation of Mitral/Tufted and Centrifugal Fiber Inputs to Granule Cells. J. Neurophysiol. 2007, 97, 296–306. [Google Scholar] [CrossRef] [PubMed]
- Macrides, F.; Davis, B.J.; Youngs, W.M.; Nadi, N.S.; Margolis, F.L. Cholinergic and Catecholaminergic Afferents to the Olfactory Bulb in the Hamster: A Neuroanatomical, Biochemical, and Histochemical Investigation. J. Comp. Neurol. 1981, 203, 495–514. [Google Scholar] [CrossRef]
- Halasz, N. The Vertebrate Olfactory System; Akademia Kiado: Budapest, Hungary, 1990. [Google Scholar]
- Shipley, M.T. Olfactory System. In The Rat Nervous System; Academic Press: Sydnet, Autstralia, 1995; pp. 899–928. [Google Scholar]
- Cleland, T.A.; Linster, C. Central Olfactory Structures. In Handbook of Olfaction and Gustation; Marcel Dekker: New York, NY, USA, 2003; pp. 165–181. [Google Scholar]
- Matsutani, S.; Yamamoto, N. Centrifugal Innervation of the Mammalian Olfactory Bulb. Anat. Sci. Int. 2008, 83, 218–227. [Google Scholar] [CrossRef]
- Padmanabhan, K.; Osakada, F.; Tarabrina, A.; Kizer, E.; Callaway, E.M.; Gage, F.H.; Sejnowski, T.J. Centrifugal Inputs to the Main Olfactory Bulb Revealed Through Whole Brain Circuit-Mapping. Front. Neuroanat. 2019, 12, 115. [Google Scholar] [CrossRef] [PubMed]
- Haberly, L.B.; Price, J.L. Association and Commissural Fiber Systems of the Olfactory Cortex of the Rat. J. Comp. Neurol. 1978, 178, 711–740. [Google Scholar] [CrossRef]
- Mori, K.; von Campenhause, H.; Yoshihara, Y. Zonal Organization of the Mammalian Main and Accessory Olfactory Systems. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2000, 355, 1801–1812. [Google Scholar] [CrossRef] [PubMed]
- Mori, K.; Takahashi, Y.K.; Igarashi, K.M.; Yamaguchi, M. Maps of Odorant Molecular Features in the Mammalian Olfactory Bulb. Physiol. Rev. 2006, 86, 409–433. [Google Scholar] [CrossRef]
- Mori, K.; Sakano, H. How Is the Olfactory Map Formed and Interpreted in the Mammalian Brain? Annu. Rev. Neurosci. 2011, 34, 467–499. [Google Scholar] [CrossRef]
- Wysocki, C.J.; Beauchamp, G.K. Ability to Smell Androstenone Is Genetically Determined. Proc. Natl. Acad. Sci. USA 1984, 81, 4899–4902. [Google Scholar] [CrossRef]
- Vihani, A.; Hu, X.S.; Gundala, S.; Koyama, S.; Block, E.; Matsunami, H. Semiochemical Responsive Olfactory Sensory Neurons Are Sexually Dimorphic and Plastic. eLife 2020, 9, e54501. [Google Scholar] [CrossRef]
- Dey, S.; Chamero, P.; Pru, J.K.; Chien, M.-S.; Ibarra-Soria, X.; Spencer, K.R.; Logan, D.W.; Matsunami, H.; Peluso, J.J.; Stowers, L. Cyclic Regulation of Sensory Perception by a Female Hormone Alters Behavior. Cell 2015, 161, 1334–1344. [Google Scholar] [CrossRef] [PubMed]
- Stowers, L.; Liberles, S.D. State-Dependent Responses to Sex Pheromones in Mouse. Curr. Opin. Neurobiol. 2016, 38, 74–79. [Google Scholar] [CrossRef]
- van der Linden, C.J.; Gupta, P.; Bhuiya, A.I.; Riddick, K.R.; Hossain, K.; Santoro, S.W. Olfactory Stimulation Regulates the Birth of Neurons That Express Specific Odorant Receptors. Cell Rep. 2020, 33, 108210. [Google Scholar] [CrossRef] [PubMed]
- Jones, S.V.; Choi, D.C.; Davis, M.; Ressler, K.J. Learning-Dependent Structural Plasticity in the Adult Olfactory Pathway. J. Neurosci. 2008, 28, 13106–13111. [Google Scholar] [CrossRef] [PubMed]
- Dias, B.G.; Ressler, K.J. Parental Olfactory Experience Influences Behavior and Neural Structure in Subsequent Generations. Nat. Neurosci. 2014, 17, 89–96. [Google Scholar] [CrossRef]
- Dalton, P.; Doolittle, N.; Breslin, P.A.S. Gender-Specific Induction of Enhanced Sensitivity to Odors. Nat. Neurosci. 2002, 5, 199–200. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Li, W.; Voleti, V.; Zou, D.-J.; Hillman, E.M.C.; Firestein, S. Widespread Receptor-Driven Modulation in Peripheral Olfactory Coding. Science 2020, 368, eaaz5390. [Google Scholar] [CrossRef] [PubMed]
- Lin, D.Y.; Shea, S.D.; Katz, L.C. Representation of Natural Stimuli in the Rodent Main Olfactory Bulb. Neuron 2006, 50, 937–949. [Google Scholar] [CrossRef]
- Keller, A.; Zhuang, H.; Chi, Q.; Vosshall, L.B.; Matsunami, H. Genetic Variation in a Human Odorant Receptor Alters Odour Perception. Nature 2007, 449, 468–472. [Google Scholar] [CrossRef]
- Menashe, I.; Abaffy, T.; Hasin, Y.; Goshen, S.; Yahalom, V.; Luetje, C.W.; Lancet, D. Genetic Elucidation of Human Hyperosmia to Isovaleric Acid. PLoS Biol. 2007, 5, e284. [Google Scholar] [CrossRef]
- McRae, J.F.; Mainland, J.D.; Jaeger, S.R.; Adipietro, K.A.; Matsunami, H.; Newcomb, R.D. Genetic Variation in the Odorant Receptor OR2J3 Is Associated with the Ability to Detect the “Grassy” Smelling Odor, Cis-3-Hexen-1-Ol. Chem. Senses 2012, 37, 585–593. [Google Scholar] [CrossRef]
- Jaeger, S.R.; McRae, J.F.; Bava, C.M.; Beresford, M.K.; Hunter, D.; Jia, Y.; Chheang, S.L.; Jin, D.; Peng, M.; Gamble, J.C.; et al. A Mendelian Trait for Olfactory Sensitivity Affects Odor Experience and Food Selection. Curr. Biol. 2013, 23, 1601–1605. [Google Scholar] [CrossRef]
- Trimmer, C.; Keller, A.; Murphy, N.R.; Snyder, L.L.; Willer, J.R.; Nagai, M.H.; Katsanis, N.; Vosshall, L.B.; Matsunami, H.; Mainland, J.D. Genetic Variation across the Human Olfactory Receptor Repertoire Alters Odor Perception. Proc. Natl. Acad. Sci. USA 2019, 116, 9475–9480. [Google Scholar] [CrossRef]
- Hummel, T.; Whitcroft, K.L.; Andrews, P.; Altundag, A.; Cinghi, C.; Costanzo, R.M.; Damm, M.; Frasnelli, J.; Gudziol, H.; Gupta, N.; et al. Position Paper on Olfactory Dysfunction. Rhinol. Suppl. 2017, 54, 1–30. [Google Scholar] [CrossRef] [PubMed]
- Miwa, T.; Ikeda, K.; Ishibashi, T.; Kobayashi, M.; Kondo, K.; Matsuwaki, Y.; Ogawa, T.; Shiga, H.; Suzuki, M.; Tsuzuki, K.; et al. Clinical Practice Guidelines for the Management of Olfactory Dysfunction—Secondary Publication. Auris Nasus Larynx 2019, 46, 653–662. [Google Scholar] [CrossRef]
- Konstantinidis, I.; Tsakiropoulou, E.; Constantinidis, J. Long Term Effects of Olfactory Training in Patients with Post-Infectious Olfactory Loss. Rhinology 2016, 54, 170–175. [Google Scholar] [CrossRef]
- Damm, M.; Pikart, L.K.; Reimann, H.; Burkert, S.; Göktas, Ö.; Haxel, B.; Frey, S.; Charalampakis, I.; Beule, A.; Renner, B.; et al. Olfactory Training Is Helpful in Postinfectious Olfactory Loss: A Randomized, Controlled, Multicenter Study: Olfactory Training. Laryngoscope 2014, 124, 826–831. [Google Scholar] [CrossRef]
- Altundag, A.; Cayonu, M.; Kayabasoglu, G.; Salihoglu, M.; Tekeli, H.; Saglam, O.; Hummel, T. Modified Olfactory Training in Patients With Postinfectious Olfactory Loss. Laryngoscope 2015, 125, 1763–1766. [Google Scholar] [CrossRef]
- Kattar, N.; Do, T.M.; Unis, G.D.; Migneron, M.R.; Thomas, A.J.; McCoul, E.D. Olfactory Training for Postviral Olfactory Dysfunction: Systematic Review and Meta-Analysis. Otolaryngol. Head Neck Surg. 2021, 164, 244–254. [Google Scholar] [CrossRef] [PubMed]
- Pekala, K.; Chandra, R.K.; Turner, J.H. Efficacy of Olfactory Training in Patients with Olfactory Loss: A Systematic Review and Meta-Analysis: Systematic Review of Olfactory Training. Int. Forum Allergy Rhinol. 2016, 6, 299–307. [Google Scholar] [CrossRef]
- Díaz, D.; Gómez, C.; Muñoz-Castañeda, R.; Baltanás, F.; Alonso, J.R.; Weruaga, E. The Olfactory System as a Puzzle: Playing with Its Pieces. Anat. Rec. 2013, 296, 1383–1400. [Google Scholar] [CrossRef]
- Kempermann, G. Adult Neurogenesis, 2nd ed.; Oxford University Press: New York, NY, USA, 2011; ISBN 978-0-19-972969-2. [Google Scholar]
- Gritti, A.; Bonfanti, L.; Doetsch, F.; Caille, I.; Alvarez-Buylla, A.; Lim, D.A.; Galli, R.; Verdugo, J.M.G.; Herrera, D.G.; Vescovi, A.L. Multipotent Neural Stem Cells Reside into the Rostral Extension and Olfactory Bulb of Adult Rodents. J. Neurosci. 2002, 22, 437–445. [Google Scholar] [CrossRef] [PubMed]
- Alonso, M.; Ortega-Perez, I.; Grubb, M.S.; Bourgeois, J.-P.; Charneau, P.; Lledo, P.-M. Turning Astrocytes from the Rostral Migratory Stream into Neurons: A Role for the Olfactory Sensory Organ. J. Neurosci. 2008, 28, 11089–11102. [Google Scholar] [CrossRef]
- Brann, J.H.; Firestein, S.J. A Lifetime of Neurogenesis in the Olfactory System. Front. Neurosci. 2014, 8, 182. [Google Scholar] [CrossRef] [PubMed]
- Corotto, F.S.; Henegar, J.R.; Maruniak, J.A. Odor Deprivation Leads to Reduced Neurogenesis and Reduced Neuronal Survival in the Olfactory Bulb of the Adult Mouse. Neuroscience 1994, 61, 739–744. [Google Scholar] [CrossRef]
- Mandairon, N.; Jourdan, F.; Didier, A. Deprivation of Sensory Inputs to the Olfactory Bulb Up-Regulates Cell Death and Proliferation in the Subventricular Zone of Adult Mice. Neuroscience 2003, 119, 507–516. [Google Scholar] [CrossRef]
- Suzukawa, K.; Kondo, K.; Kanaya, K.; Sakamoto, T.; Watanabe, K.; Ushio, M.; Kaga, K.; Yamasoba, T. Age-Related Changes of the Regeneration Mode in the Mouse Peripheral Olfactory System Following Olfactotoxic Drug Methimazole-Induced Damage. J. Comp. Neurol. 2011, 519, 2154–2174. [Google Scholar] [CrossRef]
- Rochefort, C.; Gheusi, G.; Vincent, J.-D.; Lledo, P.-M. Enriched Odor Exposure Increases the Number of Newborn Neurons in the Adult Olfactory Bulb and Improves Odor Memory. J. Neurosci. 2002, 22, 2679–2689. [Google Scholar] [CrossRef] [PubMed]
- Martončíková, M.; Lievajová, K.; Orendáčová, J.; Blaško, J.; Račeková, E. Odor Enrichment Influences Neurogenesis in the Rostral Migratory Stream of Young Rats. Acta Histochem. 2011, 113, 326–332. [Google Scholar] [CrossRef]
- Veyrac, A.; Sacquet, J.; Nguyen, V.; Marien, M.; Jourdan, F.; Didier, A. Novelty Determines the Effects of Olfactory Enrichment on Memory and Neurogenesis Through Noradrenergic Mechanisms. Neuropsychopharmacology 2009, 34, 786–795. [Google Scholar] [CrossRef]
- Oboti, L.; Schellino, R.; Giachino, C.; Chamero, P.; Pyrski, M.; Leinders-Zufall, T.; Zufall, F.; Fasolo, A.; Peretto, P. Newborn Interneurons in the Accessory Olfactory Bulb Promote Mate Recognition in Female Mice. Front. Neurosci. 2011, 5, 113. [Google Scholar] [CrossRef]
- Bonzano, S.; Bovetti, S.; Fasolo, A.; Peretto, P.; De Marchis, S. Odour Enrichment Increases Adult-Born Dopaminergic Neurons in the Mouse Olfactory Bulb. Eur. J. Neurosci. 2014, 40, 3450–3457. [Google Scholar] [CrossRef]
- Mak, G.K.; Enwere, E.K.; Gregg, C.; Pakarainen, T.; Poutanen, M.; Huhtaniemi, I.; Weiss, S. Male Pheromone-Stimulated Neurogenesis in the Adult Female Brain: Possible Role in Mating Behavior. Nat. Neurosci. 2007, 10, 1003–1011. [Google Scholar] [CrossRef]
- Koyama, S.; Soini, H.A.; Foley, J.; Novotny, M.V.; Lai, C. Stimulation of Cell Proliferation in the Subventricular Zone by Synthetic Murine Pheromones. Front. Behav. Neurosci. 2013, 7, 101. [Google Scholar] [CrossRef] [PubMed]
- Oboti, L.; Savalli, G.; Giachino, C.; De Marchis, S.; Panzica, G.C.; Fasolo, A.; Peretto, P. Integration and Sensory Experience-Dependent Survival of Newly-Generated Neurons in the Accessory Olfactory Bulb of Female Mice. Eur. J. Neurosci. 2009, 29, 679–692. [Google Scholar] [CrossRef]
- Hoffman, E.; Pickavance, L.; Thippeswamy, T.; Beynon, R.J.; Hurst, J.L. The Male Sex Pheromone Darcin Stimulates Hippocampal Neurogenesis and Cell Proliferation in the Subventricular Zone in Female Mice. Front. Behav. Neurosci. 2015, 9, 106. [Google Scholar] [CrossRef]
- Watt, W.C.; Sakano, H.; Lee, Z.-Y.; Reusch, J.E.; Trinh, K.; Storm, D.R. Odorant Stimulation Enhances Survival of Olfactory Sensory Neurons via MAPK and CREB. Neuron 2004, 41, 955–967. [Google Scholar] [CrossRef]
- Santoro, S.W.; Dulac, C. The Activity-Dependent Histone Variant H2BE Modulates the Life Span of Olfactory Neurons. eLife 2012, 1, e00070. [Google Scholar] [CrossRef]
- François, A.; Laziz, I.; Rimbaud, S.; Grebert, D.; Durieux, D.; Pajot-Augy, E.; Meunier, N. Early Survival Factor Deprivation in the Olfactory Epithelium Enhances Activity-Driven Survival. Front. Cell. Neurosci. 2013, 7, 271. [Google Scholar] [CrossRef] [PubMed]
- Cadiou, H.; Aoudé, I.; Tazir, B.; Molinas, A.; Fenech, C.; Meunier, N.; Grosmaitre, X. Postnatal Odorant Exposure Induces Peripheral Olfactory Plasticity at the Cellular Level. J. Neurosci. 2014, 34, 4857–4870. [Google Scholar] [CrossRef]
- Ibarra-Soria, X.; Nakahara, T.S.; Lilue, J.; Jiang, Y.; Trimmer, C.; Souza, M.A.; Netto, P.H.; Ikegami, K.; Murphy, N.R.; Kusma, M.; et al. Variation in Olfactory Neuron Repertoires Is Genetically Controlled and Environmentally Modulated. eLife 2017, 6, e21476. [Google Scholar] [CrossRef]
- Marin, C.; Laxe, S.; Langdon, C.; Alobid, I.; Berenguer, J.; Fuentes, M.; Bernabeu, M.; Mullol, J. Olfactory Training Prevents Olfactory Dysfunction Induced by Bulbar Excitotoxic Lesions: Role of Neurogenesis and Dopaminergic Interneurons. Mol. Neurobiol. 2019, 56, 8063–8075. [Google Scholar] [CrossRef]
- Kikuta, S.; Sakamoto, T.; Nagayama, S.; Kanaya, K.; Kinoshita, M.; Kondo, K.; Tsunoda, K.; Mori, K.; Yamasoba, T. Sensory Deprivation Disrupts Homeostatic Regeneration of Newly Generated Olfactory Sensory Neurons after Injury in Adult Mice. J. Neurosci. 2015, 35, 2657–2673. [Google Scholar] [CrossRef]
- Bovetti, S.; Veyrac, A.; Peretto, P.; Fasolo, A.; De Marchis, S. Olfactory Enrichment Influences Adult Neurogenesis Modulating GAD67 and Plasticity-Related Molecules Expression in Newborn Cells of the Olfactory Bulb. PLoS ONE 2009, 4, e6359. [Google Scholar] [CrossRef] [PubMed]
- Raviv, J.R.; Kern, R.C. Chronic Rhinosinusitis and Olfactory Dysfunction. In Advances in Oto-Rhino-Laryngology; Hummel, T., Welge-Lüssen, A., Eds.; KARGER: Basel, Switzerland, 2006; Volume 63, pp. 108–124. ISBN 978-3-8055-8123-3. [Google Scholar]
- Litvack, J.R.; Fong, K.; Mace, J.; James, K.E.; Smith, T.L. Predictors of Olfactory Dysfunction in Patients with Chronic Rhinosinusitis. Laryngoscope 2008, 118, 2225–2230. [Google Scholar] [CrossRef] [PubMed]
- Brittebo, E.B. Metabolism-Dependent Toxicity of Methimazole in the Olfactory Nasal Mucosa. Pharm. Toxicol 1995, 76, 76–79. [Google Scholar] [CrossRef] [PubMed]
- Genter, M.B.; Deamer, N.J.; Blake, B.L.; Wesley, D.S.; Levi, P.E. Olfactory Toxicity of Methimazole: Dose-Response and Structure-Activity Studies and Characterization of Flavin-Containing Monooxygenase Activity in the Long-Evans Rat Olfactory Mucosa. Toxicol. Pathol. 1995, 23, 477–486. [Google Scholar] [CrossRef] [PubMed]
- Bergman, U.; Ostergren, A.; Gustafson, A.-L.; Brittebo, B. Differential Effects of Olfactory Toxicants on Olfactory Regeneration. Arch. Toxicol. 2002, 76, 104–112. [Google Scholar] [CrossRef]
- Mobley, A.S.; Rodriguez-Gil, D.J.; Imamura, F.; Greer, C.A. Aging in the Olfactory System. Trends Neurosci. 2014, 37, 77–84. [Google Scholar] [CrossRef]
- Kondo, K.; Kikuta, S.; Ueha, R.; Suzukawa, K.; Yamasoba, T. Age-Related Olfactory Dysfunction: Epidemiology, Pathophysiology, and Clinical Management. Front. Aging Neurosci. 2020, 12, 208. [Google Scholar] [CrossRef]
- Yamaguchi, M.; Mori, K. Critical Period for Sensory Experience-Dependent Survival of Newly Generated Granule Cells in the Adult Mouse Olfactory Bulb. Proc. Natl. Acad. Sci. USA 2005, 102, 9697–9702. [Google Scholar] [CrossRef]
- Nelson, G.M. Biology of Taste Buds and the Clinical Problem of Taste Loss. Anat. Rec. 1998, 253, 70–78. [Google Scholar] [CrossRef]
- Barlow, L.A. Progress and Renewal in Gustation: New Insights into Taste Bud Development. Development 2015, 142, 3620–3629. [Google Scholar] [CrossRef]
- Roper, S.D.; Chaudhari, N. Taste Buds: Cells, Signals and Synapses. Nat. Rev. Neurosci. 2017, 18, 485–497. [Google Scholar] [CrossRef]
- Iwasaki, S. Evolution of the Structure and Function of the Vertebrate Tongue. J. Anat. 2002, 201, 1–13. [Google Scholar] [CrossRef]
- Jung, H.-S.; Akita, K.; Kim, J.-Y. Spacing Patterns on Tongue Surface-Gustatory Papilla. Int. J. Dev. Biol. 2004, 48, 157–161. [Google Scholar] [CrossRef]
- Stone, L.M.; Finger, T.E.; Tam, P.P.; Tan, S.S. Taste Receptor Cells Arise from Local Epithelium, Not Neurogenic Ectoderm. Proc. Natl. Acad. Sci. USA 1995, 92, 1916–1920. [Google Scholar] [CrossRef] [PubMed]
- Okubo, T.; Clark, C.; Hogan, B.L.M. Cell Lineage Mapping of Taste Bud Cells and Keratinocytes in the Mouse Tongue and Soft Palate. Stem Cells 2009, 27, 442–450. [Google Scholar] [CrossRef]
- Thirumangalathu, S.; Harlow, D.E.; Driskell, A.L.; Krimm, R.F.; Barlow, L.A. Fate Mapping of Mammalian Embryonic Taste Bud Progenitors. Development 2009, 136, 1519–1528. [Google Scholar] [CrossRef] [PubMed]
- Gao, N.; Lu, M.; Echeverri, F.; Laita, B.; Kalabat, D.; Williams, M.E.; Hevezi, P.; Zlotnik, A.; Moyer, B.D. Voltage-Gated Sodium Channels in Taste Bud Cells. BMC Neurosci. 2009, 10, 20. [Google Scholar] [CrossRef]
- Kinnamon, S.C. Neurosensory Transmission without a Synapse: New Perspectives on Taste Signaling. BMC Biol. 2013, 11, 42. [Google Scholar] [CrossRef] [PubMed]
- Vandenbeuch, A.; Clapp, T.R.; Kinnamon, S.C. Amiloride-Sensitive Channels in Type I Fungiform Taste Cells in Mouse. BMC Neurosci. 2008, 9, 1. [Google Scholar] [CrossRef] [PubMed]
- Liman, E.R.; Zhang, Y.V.; Montell, C. Peripheral Coding of Taste. Neuron 2014, 81, 984–1000. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, N.; Roper, S.D. The Cell Biology of Taste. J. Cell Biol. 2010, 190, 285–296. [Google Scholar] [CrossRef]
- Roper, S.D. Signal Transduction and Information Processing in Mammalian Taste Buds. Pflug. Arch. Eur. J. Physiol. 2007, 454, 759–776. [Google Scholar] [CrossRef] [PubMed]
- Ariyasu, T.; Matsumoto, S.; Kyono, F.; Hanaya, T.; Arai, S.; Ikeda, M.; Kurimoto, M. Taste Receptor T1R3 Is an Essential Molecule for the Cellular Recognition of the Disaccharide Trehalose. Vitr. Cell Dev. Biol. Anim. 2003, 39, 80–88. [Google Scholar] [CrossRef]
- Temussi, P. The History of Sweet Taste: Not Exactly a Piece of Cake. J. Mol. Recognit. 2006, 19, 188–199. [Google Scholar] [CrossRef]
- Morini, G.; Bassoli, A.; Temussi, P.A. From Small Sweeteners to Sweet Proteins: Anatomy of the Binding Sites of the Human T1R2_T1R3 Receptor. J. Med. Chem. 2005, 48, 5520–5529. [Google Scholar] [CrossRef]
- Kurihara, K. Umami the Fifth Basic Taste: History of Studies on Receptor Mechanisms and Role as a Food Flavor. Biomed. Res. Int. 2015, 2015, 189402. [Google Scholar] [CrossRef]
- Chaudhari, N.; Landin, A.M.; Roper, S.D. A Metabotropic Glutamate Receptor Variant Functions as a Taste Receptor. Nat. Neurosci. 2000, 3, 113–119. [Google Scholar] [CrossRef]
- San Gabriel, A.; Maekawa, T.; Uneyama, H.; Torii, K. Metabotropic Glutamate Receptor Type 1 in Taste Tissue. Am. J. Clin. Nutr. 2009, 90, 743S–746S. [Google Scholar] [CrossRef]
- Niswender, C.M.; Conn, P.J. Metabotropic Glutamate Receptors: Physiology, Pharmacology, and Disease. Annu. Rev. Pharmacol. Toxicol. 2010, 50, 295–322. [Google Scholar] [CrossRef] [PubMed]
- Toyono, T.; Seta, Y.; Kataoka, S.; Kawano, S.; Shigemoto, R.; Toyoshima, K. Expression of Metabotropic Glutamate Receptor Group I in Rat Gustatory Papillae. Cell Tissue Res 2003, 313, 29–35. [Google Scholar] [CrossRef] [PubMed]
- Chaudhari, N.; Yang, H.; Lamp, C.; Delay, E.; Cartford, C.; Than, T.; Roper, S. The Taste of Monosodium Glutamate: Membrane Receptors in Taste Buds. J. Neurosci. 1996, 16, 3817–3826. [Google Scholar] [CrossRef]
- Toyono, T.; Kataoka, S.; Seta, Y.; Shigemoto, R.; Toyoshima, K. Expression of Group II Metabotropic Glutamate Receptors in Rat Gustatory Papillae. Cell Tissue Res. 2007, 328, 57–63. [Google Scholar] [CrossRef] [PubMed]
- Pal Choudhuri, S.; Delay, R.J.; Delay, E.R. Metabotropic Glutamate Receptors Are Involved in the Detection of IMP and L-Amino Acids by Mouse Taste Sensory Cells. Neuroscience 2016, 316, 94–108. [Google Scholar] [CrossRef] [PubMed]
- Meyerhof, W.; Batram, C.; Kuhn, C.; Brockhoff, A.; Chudoba, E.; Bufe, B.; Appendino, G.; Behrens, M. The Molecular Receptive Ranges of Human TAS2R Bitter Taste Receptors. Chem. Senses 2010, 35, 157–170. [Google Scholar] [CrossRef]
- Jaggupilli, A.; Howard, R.; Upadhyaya, J.D.; Bhullar, R.P.; Chelikani, P. Bitter Taste Receptors: Novel Insights into the Biochemistry and Pharmacology. Int. J. Biochem. Cell Biol. 2016, 77, 184–196. [Google Scholar] [CrossRef]
- Hass, N.; Schwarzenbacher, K.; Breer, H. T1R3 Is Expressed in Brush Cells and Ghrelin-Producing Cells of Murine Stomach. Cell Tissue Res 2010, 339, 493–504. [Google Scholar] [CrossRef]
- Wu, S.V.; Rozengurt, N.; Yang, M.; Young, S.H.; Sinnett-Smith, J.; Rozengurt, E. Expression of Bitter Taste Receptors of the T2R Family in the Gastrointestinal Tract and Enteroendocrine STC-1 Cells. Proc. Natl. Acad. Sci. USA 2002, 99, 2392–2397. [Google Scholar] [CrossRef] [PubMed]
- Liu, S.; Lu, S.; Xu, R.; Atzberger, A.; Günther, S.; Wettschureck, N.; Offermanns, S. Members of Bitter Taste Receptor Cluster Tas2r143/Tas2r135/Tas2r126 Are Expressed in the Epithelium of Murine Airways and Other Non-Gustatory Tissues. Front. Physiol. 2017, 8, 849. [Google Scholar] [CrossRef] [PubMed]
- Shah, A.S.; Ben-Shahar, Y.; Moninger, T.O.; Kline, J.N.; Welsh, M.J. Motile Cilia of Human Airway Epithelia Are Chemosensory. Science 2009, 325, 1131–1134. [Google Scholar] [CrossRef]
- Finger, T.E.; Kinnamon, S.C. Taste Isn’t Just for Taste Buds Anymore. F1000 Biol. Rep. 2011, 3, 20. [Google Scholar] [CrossRef]
- Nayak, A.P.; Shah, S.D.; Michael, J.V.; Deshpande, D.A. Bitter Taste Receptors for Asthma Therapeutics. Front. Physiol. 2019, 10, 884. [Google Scholar] [CrossRef] [PubMed]
- Richter, T.A.; Caicedo, A.; Roper, S.D. Sour Taste Stimuli Evoke Ca 2+ and PH Responses in Mouse Taste Cells. J. Physiol. 2003, 547, 475–483. [Google Scholar] [CrossRef]
- Teng, B.; Wilson, C.E.; Tu, Y.-H.; Joshi, N.R.; Kinnamon, S.C.; Liman, E.R. Cellular and Neural Responses to Sour Stimuli Require the Proton Channel Otop1. Curr. Biol. 2019, 29, 3647–3656.e5. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Jin, H.; Zhang, W.; Ding, C.; O’Keeffe, S.; Ye, M.; Zuker, C.S. Sour Sensing from the Tongue to the Brain. Cell 2019, 179, 392–402.e15. [Google Scholar] [CrossRef]
- Lewandowski, B.C.; Sukumaran, S.K.; Margolskee, R.F.; Bachmanov, A.A. Amiloride-Insensitive Salt Taste Is Mediated by Two Populations of Type III Taste Cells with Distinct Transduction Mechanisms. J. Neurosci. 2016, 36, 1942–1953. [Google Scholar] [CrossRef]
- Roebber, J.K.; Roper, S.D.; Chaudhari, N. The Role of the Anion in Salt (NaCl) Detection by Mouse Taste Buds. J. Neurosci. 2019, 39, 6224–6232. [Google Scholar] [CrossRef]
- Perea-Martinez, I.; Nagai, T.; Chaudhari, N. Functional Cell Types in Taste Buds Have Distinct Longevities. PLoS ONE 2013, 8, e53399. [Google Scholar] [CrossRef] [PubMed]
- Barlow, L.A.; Klein, O.D. Developing and Regenerating a Sense of Taste. In Current Topics in Developmental Biology; Elsevier: Amsterdam, The Netherlands, 2015; Volume 111, pp. 401–419. ISBN 978-0-12-407759-1. [Google Scholar]
- Miura, H.; Scott, J.K.; Harada, S.; Barlow, L.A. Sonic Hedgehog-Expressing Basal Cells Are General Post-Mitotic Precursors of Functional Taste Receptor Cells. Dev. Dyn. 2014, 243, 1286–1297. [Google Scholar] [CrossRef]
- Castillo, D.; Seidel, K.; Salcedo, E.; Ahn, C.; de Sauvage, F.J.; Klein, O.D.; Barlow, L.A. Induction of Ectopic Taste Buds by SHH Reveals the Competency and Plasticity of Adult Lingual Epithelium. Development 2014, 141, 2993–3002. [Google Scholar] [CrossRef] [PubMed]
- Gaillard, D.; Xu, M.; Liu, F.; Millar, S.E.; Barlow, L.A. β-Catenin Signaling Biases Multipotent Lingual Epithelial Progenitors to Differentiate and Acquire Specific Taste Cell Fates. PLoS Genet. 2015, 11, e1005208. [Google Scholar] [CrossRef]
- Yee, K.K.; Li, Y.; Redding, K.M.; Iwatsuki, K.; Margolskee, R.F.; Jiang, P. Lgr5-EGFP Marks Taste Bud Stem/Progenitor Cells in Posterior Tongue. Stem Cells 2013, 31, 992–1000. [Google Scholar] [CrossRef] [PubMed]
- Feng, P.; Huang, L.; Wang, H. Taste Bud Homeostasis in Health, Disease, and Aging. Chem. Senses 2014, 39, 3–16. [Google Scholar] [CrossRef] [PubMed]
- Takeda, N.; Jain, R.; Li, D.; Li, L.; Lu, M.M.; Epstein, J.A. Lgr5 Identifies Progenitor Cells Capable of Taste Bud Regeneration after Injury. PLoS ONE 2013, 8, e66314. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez, A.C.d.O.; Costa, T.F.; Andrade, Z.d.A.; Medrado, A.R.A.P. Wound Healing—A Literature Review. An. Bras. Dermatol. 2016, 91, 614–620. [Google Scholar] [CrossRef] [PubMed]
- Landén, N.X.; Li, D.; Ståhle, M. Transition from Inflammation to Proliferation: A Critical Step during Wound Healing. Cell Mol. Life Sci. 2016, 73, 3861–3885. [Google Scholar] [CrossRef] [PubMed]
- Aragona, M.; Dekoninck, S.; Rulands, S.; Lenglez, S.; Mascré, G.; Simons, B.D.; Blanpain, C. Defining Stem Cell Dynamics and Migration during Wound Healing in Mouse Skin Epidermis. Nat. Commun. 2017, 8, 14684. [Google Scholar] [CrossRef]
- Blanpain, C.; Simons, B.D. Unravelling Stem Cell Dynamics by Lineage Tracing. Nat. Rev. Mol. Cell Biol. 2013, 14, 489–502. [Google Scholar] [CrossRef]
- Ito, M.; Liu, Y.; Yang, Z.; Nguyen, J.; Liang, F.; Morris, R.J.; Cotsarelis, G. Stem Cells in the Hair Follicle Bulge Contribute to Wound Repair but Not to Homeostasis of the Epidermis. Nat. Med. 2005, 11, 1351–1354. [Google Scholar] [CrossRef]
- Jaks, V.; Barker, N.; Kasper, M.; van Es, J.H.; Snippert, H.J.; Clevers, H.; Toftgård, R. Lgr5 Marks Cycling, yet Long-Lived, Hair Follicle Stem Cells. Nat. Genet. 2008, 40, 1291–1299. [Google Scholar] [CrossRef]
- Haegebarth, A.; Clevers, H. Wnt Signaling, Lgr5, and Stem Cells in the Intestine and Skin. Am. J. Pathol. 2009, 174, 715–721. [Google Scholar] [CrossRef]
- Barker, N.; Clevers, H. Leucine-Rich Repeat-Containing G-Protein-Coupled Receptors as Markers of Adult Stem Cells. Gastroenterology 2010, 138, 1681–1696. [Google Scholar] [CrossRef]
- Presland, R.B.; Dale, B.A. Epithelial Structural Proteins of the Skin and Oral Cavity: Function in Health and Disease. Crit. Rev. Oral Biol. Med. 2000, 11, 383–408. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Zhou, J.; Marshall, B.; Rekaya, R.; Ye, K.; Liu, H.-X. SARS-CoV-2 Receptor ACE2 Is Enriched in a Subpopulation of Mouse Tongue Epithelial Cells in Nongustatory Papillae but Not in Taste Buds or Embryonic Oral Epithelium. ACS Pharmacol. Transl. Sci. 2020, 3, 749–758. [Google Scholar] [CrossRef] [PubMed]
- Sakaguchi, W.; Kubota, N.; Shimizu, T.; Saruta, J.; Fuchida, S.; Kawata, A.; Yamamoto, Y.; Sugimoto, M.; Yakeishi, M.; Tsukinoki, K. Existence of SARS-CoV-2 Entry Molecules in the Oral Cavity. Int. J. Mol. Sci. 2020, 21, 6000. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, M.; Zunt, S.L.; Goldblatt, L.I. Oral Epithelial Expression of Angiotensin Converting Enzyme-2: Implications for COVID-19 Diagnosis and Prognosis. bioRxiv 2020. [Google Scholar] [CrossRef]
- Usami, Y.; Hirose, K.; Okumura, M.; Toyosawa, S.; Sakai, T. Brief Communication: Immunohistochemical Detection of ACE2 in Human Salivary Gland. Oral Sci. Int. 2020. [Google Scholar] [CrossRef]
- Aroke, E.; Powell-Roach, K.; Jaime-Lara, R.; Tesfaye, M.; Roy, A.; Jackson, P.; Joseph, P. Taste the Pain: The Role of TRP Channels in Pain and Taste Perception. Int. J. Mol. Sci. 2020, 21, 5929. [Google Scholar] [CrossRef]
- Rhyu, M.-R.; Kim, Y.; Lyall, V. Interactions between Chemesthesis and Taste: Role of TRPA1 and TRPV1. Int. J. Mol. Sci. 2021, 22, 3360. [Google Scholar] [CrossRef]
- Lin, W.; Margolskee, R.; Donnert, G.; Hell, S.W.; Restrepo, D. Olfactory Neurons Expressing Transient Receptor Potential Channel M5 (TRPM5) Are Involved in Sensing Semiochemicals. Proc. Natl. Acad. Sci. USA 2007, 104, 2471–2476. [Google Scholar] [CrossRef]
- Roper, S.D. TRPs in Taste and Chemesthesis. Handb. Exp. Pharm. 2014, 223, 827–871. [Google Scholar] [CrossRef]
- Simon, S.A.; Gutierrez, R. TRP Channels at the Periphery of the Taste and Trigeminal Systems. In Neurobiology of TRP Channels; Emir, T.L.R., Ed.; Frontiers in Neuroscience; CRC Press/Taylor & Francis: Boca Raton, FL, USA, 2017; ISBN 978-1-315-15283-7. [Google Scholar]
- Nakashimo, Y.; Takumida, M.; Fukuiri, T.; Anniko, M.; Hirakawa, K. Expression of Transient Receptor Potential Channel Vanilloid (TRPV) 1–4, Melastin (TRPM) 5 and 8, and Ankyrin (TRPA1) in the Normal and Methimazole-Treated Mouse Olfactory Epithelium. Acta Oto-Laryngol. 2010, 130, 1278–1286. [Google Scholar] [CrossRef] [PubMed]
- Doty, R.L.; Brugger, W.E.; Jurs, P.C.; Orndorff, M.A.; Snyder, P.J.; Lowry, L.D. Intranasal Trigeminal Stimulation from Odorous Volatiles: Psychometric Responses from Anosmic and Normal Humans. Physiol. Behav. 1978, 20, 175–185. [Google Scholar] [CrossRef]
- Frasnelli, J.; Hummel, T. Interactions between the Chemical Senses: Trigeminal Function in Patients with Olfactory Loss. Int. J. Psychophysiol. 2007, 65, 177–181. [Google Scholar] [CrossRef] [PubMed]
- Tremblay, C.; Frasnelli, J. Olfactory and Trigeminal Systems Interact in the Periphery. Chem. Senses 2018, 43, 611–616. [Google Scholar] [CrossRef]
- Gerkin, R.C.; Ohla, K.; Veldhuizen, M.G.; Joseph, P.V.; Kelly, C.E.; Bakke, A.J.; Steele, K.E.; Farruggia, M.C.; Pellegrino, R.; Pepino, M.Y.; et al. Recent Smell Loss Is the Best Predictor of COVID-19 Among Individuals With Recent Respiratory Symptoms. Chem. Senses 2021, 46, bjaa081. [Google Scholar] [CrossRef]
- Nguyen, M.Q.; Wu, Y.; Bonilla, L.S.; von Buchholtz, L.J.; Ryba, N.J.P. Diversity amongst Trigeminal Neurons Revealed by High Throughput Single Cell Sequencing. PLoS ONE 2017, 12, e0185543. [Google Scholar] [CrossRef]
- Nguyen, M.Q.; Le Pichon, C.E.; Ryba, N. Stereotyped Transcriptomic Transformation of Somatosensory Neurons in Response to Injury. eLife 2019, 8, e49679. [Google Scholar] [CrossRef] [PubMed]
- Shiers, S.; Ray, P.R.; Wangzhou, A.; Sankaranarayanan, I.; Tatsui, C.E.; Rhines, L.D.; Li, Y.; Uhelski, M.L.; Dougherty, P.M.; Price, T.J. ACE2 and SCARF Expression in Human Dorsal Root Ganglion Nociceptors: Implications for SARS-CoV-2 Virus Neurological Effects. Pain 2020, 161, 2494–2501. [Google Scholar] [CrossRef]
- Pellegrino, R.; Cooper, K.W.; Di Pizio, A.; Joseph, P.V.; Bhutani, S.; Parma, V. Corona Viruses and the Chemical Senses: Past, Present, and Future. Chem. Senses 2020, 45, 415–422. [Google Scholar] [CrossRef]
- Le Bon, S.-D.; Konopnicki, D.; Pisarski, N.; Prunier, L.; Lechien, J.R.; Horoi, M. Efficacy and Safety of Oral Corticosteroids and Olfactory Training in the Management of COVID-19-Related Loss of Smell. Eur. Arch. Otorhinolaryngol. 2021, 278, 3113–3117. [Google Scholar] [CrossRef]
- Henning, H. Der Geruch; Johann Ambrosius Barth: Leipzig, Germany, 1916. [Google Scholar]
- Sá, R.C.S.; Andrade, L.; de Sousa, D. A Review on Anti-Inflammatory Activity of Monoterpenes. Molecules 2013, 18, 1227–1254. [Google Scholar] [CrossRef]
- Sá, R.C.S.; Andrade, L.N.; de Sousa, D.P. Sesquiterpenes from Essential Oils and Anti-Inflammatory Activity. Nat. Prod. Commun. 2015, 10, 1767–1774. [Google Scholar]
- Cox-Georgian, D.; Ramadoss, N.; Dona, C.; Basu, C. Therapeutic and Medicinal Uses of Terpenes. In Medicinal Plants; Joshee, N., Dhekney, S.A., Parajuli, P., Eds.; Springer International Publishing: Cham, Switzerland, 2019; pp. 333–359. ISBN 978-3-030-31268-8. [Google Scholar]
- Kim, T.; Song, B.; Cho, K.S.; Lee, I.-S. Therapeutic Potential of Volatile Terpenes and Terpenoids from Forests for Inflammatory Diseases. Int. J. Mol. Sci. 2020, 21, 2187. [Google Scholar] [CrossRef] [PubMed]
- Gertsch, J.; Leonti, M.; Raduner, S.; Racz, I.; Chen, J.-Z.; Xie, X.-Q.; Altmann, K.-H.; Karsak, M.; Zimmer, A. Beta-Caryophyllene Is a Dietary Cannabinoid. Proc. Natl. Acad. Sci. USA 2008, 105, 9099–9104. [Google Scholar] [CrossRef]
- Gao, F.; Zhang, L.-H.; Su, T.-F.; Li, L.; Zhou, R.; Peng, M.; Wu, C.-H.; Yuan, X.-C.; Sun, N.; Meng, X.-F.; et al. Signaling Mechanism of Cannabinoid Receptor-2 Activation-Induced β-Endorphin Release. Mol. Neurobiol. 2016, 53, 3616–3625. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, M.M.; Porreca, F.; Lai, J.; Albrecht, P.J.; Rice, F.L.; Khodorova, A.; Davar, G.; Makriyannis, A.; Vanderah, T.W.; Mata, H.P.; et al. CB2 Cannabinoid Receptor Activation Produces Antinociception by Stimulating Peripheral Release of Endogenous Opioids. Proc. Natl. Acad. Sci. USA 2005, 102, 3093–3098. [Google Scholar] [CrossRef]
- Anand, P.; Whiteside, G.; Fowler, C.J.; Hohmann, A.G. Targeting CB2 Receptors and the Endocannabinoid System for the Treatment of Pain. Brain Res. Rev. 2009, 60, 255–266. [Google Scholar] [CrossRef]
- Klauke, A.-L.; Racz, I.; Pradier, B.; Markert, A.; Zimmer, A.M.; Gertsch, J.; Zimmer, A. The Cannabinoid CB2 Receptor-Selective Phytocannabinoid Beta-Caryophyllene Exerts Analgesic Effects in Mouse Models of Inflammatory and Neuropathic Pain. Eur. Neuropsychopharmacol. 2014, 24, 608–620. [Google Scholar] [CrossRef] [PubMed]
- Gonçalves, E.C.D.; Assis, P.M.; Junqueira, L.A.; Cola, M.; Santos, A.R.S.; Raposo, N.R.B.; Dutra, R.C. Citral Inhibits the Inflammatory Response and Hyperalgesia in Mice: The Role of TLR4, TLR2/Dectin-1, and CB2 Cannabinoid Receptor/ATP-Sensitive K+ Channel Pathways. J. Nat. Prod. 2020, 83, 1190–1200. [Google Scholar] [CrossRef]
- Kessler, A.; Sahin-Nadeem, H.; Lummis, S.C.R.; Weigel, I.; Pischetsrieder, M.; Buettner, A.; Villmann, C. GABA A Receptor Modulation by Terpenoids from Sideritis Extracts. Mol. Nutr. Food Res. 2014, 58, 851–862. [Google Scholar] [CrossRef]
- Tan, K.H.; Nishida, R. Methyl Eugenol: Its Occurrence, Distribution, and Role in Nature, Especially in Relation to Insect Behavior and Pollination. J. Insect Sci. 2012, 12, 1–60. [Google Scholar] [CrossRef]
- Moreira-Lobo, D.C.A.; Linhares-Siqueira, E.D.; Cruz, G.M.P.; Cruz, J.S.; Carvalho-de-Souza, J.L.; Lahlou, S.; Coelho-de-Souza, A.N.; Barbosa, R.; Magalhães, P.J.C.; Leal-Cardoso, J.H. Eugenol Modifies the Excitability of Rat Sciatic Nerve and Superior Cervical Ganglion Neurons. Neurosci. Lett. 2010, 472, 220–224. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.-J.; Heinbockel, T. Essential Oils and Their Constituents Targeting the GABAergic System and Sodium Channels as Treatment of Neurological Diseases. Molecules 2018, 23, 1061. [Google Scholar] [CrossRef] [PubMed]
- Zhu, M.; Zhu, H.; Tan, N.; Zeng, G.; Zeng, Z.; Chu, H.; Wang, H.; Xia, Z.; Wu, R. The Effects of Acorus Tatarinowii Schott on 5-HT Concentrations, TPH2 and 5-HT1B Expression in the Dorsal Raphe of Exercised Rats. J. Ethnopharmacol. 2014, 158 Pt A, 431–436. [Google Scholar] [CrossRef]
- Ye, C.-J.; Li, S.-A.; Zhang, Y.; Lee, W.-H. Geraniol Targets KV1.3 Ion Channel and Exhibits Anti-Inflammatory Activity in Vitro and in Vivo. Fitoterapia 2019, 139, 104394. [Google Scholar] [CrossRef] [PubMed]
- Kumar, K.J.S.; Vani, M.G.; Wang, C.-S.; Chen, C.-C.; Chen, Y.-C.; Lu, L.-P.; Huang, C.-H.; Lai, C.-S.; Wang, S.-Y. Geranium and Lemon Essential Oils and Their Active Compounds Downregulate Angiotensin-Converting Enzyme 2 (ACE2), a SARS-CoV-2 Spike Receptor-Binding Domain, in Epithelial Cells. Plants 2020, 9, 770. [Google Scholar] [CrossRef] [PubMed]
- Benigni, A.; Cassis, P.; Remuzzi, G. Angiotensin II Revisited: New Roles in Inflammation, Immunology and Aging. EMBO Mol. Med. 2010, 2, 247–257. [Google Scholar] [CrossRef]
- Samavati, L.; Uhal, B.D. ACE2, Much More Than Just a Receptor for SARS-COV-2. Front. Cell. Infect. Microbiol. 2020, 10, 00317. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Zhan, P.; Liu, X. Targeting the Entry Step of SARS-CoV-2: A Promising Therapeutic Approach. Signal Transduct. Target Ther. 2020, 5, 98. [Google Scholar] [CrossRef]
- Luan, B.; Huynh, T.; Cheng, X.; Lan, G.; Wang, H.-R. Targeting Proteases for Treating COVID-19. J. Proteome 2020, 19, 4316–4326. [Google Scholar] [CrossRef]
- Premkumar, L.S. Transient Receptor Potential Channels as Targets for Phytochemicals. ACS Chem. Neurosci. 2014, 5, 1117–1130. [Google Scholar] [CrossRef] [PubMed]
- Terada, Y.; Yamashita, R.; Ihara, N.; Yamazaki-Ito, T.; Takahashi, Y.; Masuda, H.; Sakuragawa, S.; Ito, S.; Ito, K.; Watanabe, T. Human TRPA1 Activation by Terpenes Derived from the Essential Oil of Daidai, Citrus Aurantium L. Var. Daidai Makino. Biosci. Biotechnol. Biochem. 2019, 83, 1721–1728. [Google Scholar] [CrossRef]
- Araujo, D.S.M.; Nassini, R.; Geppetti, P.; De Logu, F. TRPA1 as a Therapeutic Target for Nociceptive Pain. Expert Opin. Ther. Targets 2020, 24, 997–1008. [Google Scholar] [CrossRef] [PubMed]
- Khalil, M.; Alliger, K.; Weidinger, C.; Yerinde, C.; Wirtz, S.; Becker, C.; Engel, M.A. Functional Role of Transient Receptor Potential Channels in Immune Cells and Epithelia. Front. Immunol. 2018, 9, 174. [Google Scholar] [CrossRef]
- Sebei, K.; Sakouhi, F.; Herchi, W.; Khouja, M.; Boukhchina, S. Chemical Composition and Antibacterial Activities of Seven Eucalyptus Species Essential Oils Leaves. Biol. Res. 2015, 48, 7. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Lai, Y.; Wang, Y.; Liu, N.; Zhang, F.; Xu, P. 1, 8-Cineol Protect Against Influenza-Virus-Induced Pneumonia in Mice. Inflammation 2016, 39, 1582–1593. [Google Scholar] [CrossRef]
- Caceres, A.I.; Liu, B.; Jabba, S.V.; Achanta, S.; Morris, J.B.; Jordt, S.-E. Transient Receptor Potential Cation Channel Subfamily M Member 8 Channels Mediate the Anti-Inflammatory Effects of Eucalyptol. Br. J. Pharm. 2017, 174, 867–879. [Google Scholar] [CrossRef]
- Juergens, U.R.; Dethlefsen, U.; Steinkamp, G.; Gillissen, A.; Repges, R.; Vetter, H. Anti-Inflammatory Activity of 1.8-Cineol (Eucalyptol) in Bronchial Asthma: A Double-Blind Placebo-Controlled Trial. Respir. Med. 2003, 97, 250–256. [Google Scholar] [CrossRef]
- Singer, A.J.; Clark, R.A.F. Cutaneous Wound Healing. N. Engl. J. Med. 1999, 341, 738–746. [Google Scholar] [CrossRef]
- Busse, D.; Kudella, P.; Grüning, N.-M.; Gisselmann, G.; Ständer, S.; Luger, T.; Jacobsen, F.; Steinsträßer, L.; Paus, R.; Gkogkolou, P.; et al. A Synthetic Sandalwood Odorant Induces Wound-Healing Processes in Human Keratinocytes via the Olfactory Receptor OR2AT4. J. Investig. Derm. 2014, 134, 2823–2832. [Google Scholar] [CrossRef]
- Chéret, J.; Bertolini, M.; Ponce, L.; Lehmann, J.; Tsai, T.; Alam, M.; Hatt, H.; Paus, R. Olfactory Receptor OR2AT4 Regulates Human Hair Growth. Nat. Commun. 2018, 9, 3624. [Google Scholar] [CrossRef]
- Khan, S.; Shafiei, M.S.; Longoria, C.; Schoggins, J.; Savani, R.C.; Zaki, H. SARS-CoV-2 Spike Protein Induces Inflammation via TLR2-Dependent Activation of the NF-ΚB Pathway. bioRxiv 2021. [Google Scholar] [CrossRef]
- Chen, L.; Deng, H.; Cui, H.; Fang, J.; Zuo, Z.; Deng, J.; Li, Y.; Wang, X.; Zhao, L. Inflammatory Responses and Inflammation-Associated Diseases in Organs. Oncotarget 2018, 9, 7204–7218. [Google Scholar] [CrossRef] [PubMed]
- Cooke, J.P. Inflammation and Its Role in Regeneration and Repair: A Caution for Novel Anti-Inflammatory Therapies. Circ. Res. 2019, 124, 1166–1168. [Google Scholar] [CrossRef]
- Luo, Y.; Zheng, S.G. Hall of Fame among Pro-Inflammatory Cytokines: Interleukin-6 Gene and Its Transcriptional Regulation Mechanisms. Front. Immunol. 2016, 7, 604. [Google Scholar] [CrossRef]
- Ahmed, S.M.U.; Luo, L.; Namani, A.; Wang, X.J.; Tang, X. Nrf2 Signaling Pathway: Pivotal Roles in Inflammation. Biochim. Biophys. Acta (BBA) Mol. Basis Dis. 2017, 1863, 585–597. [Google Scholar] [CrossRef]
- Shi, T.-H.; Huang, Y.-L.; Chen, C.-C.; Pi, W.-C.; Hsu, Y.-L.; Lo, L.-C.; Chen, W.-Y.; Fu, S.-L.; Lin, C.-H. Andrographolide and Its Fluorescent Derivative Inhibit the Main Proteases of 2019-NCoV and SARS-CoV through Covalent Linkage. Biochem. Biophys. Res. Commun. 2020, 533, 467–473. [Google Scholar] [CrossRef]
- Alam, R.T.M.; Fawzi, E.M.; Alkhalf, M.I.; Alansari, W.S.; Aleya, L.; Abdel-Daim, M.M. Anti-Inflammatory, Immunomodulatory, and Antioxidant Activities of Allicin, Norfloxacin, or Their Combination against Pasteurella Multocida Infection in Male New Zealand Rabbits. Oxidative Med. Cell. Longev. 2018, 2018, 1780956. [Google Scholar] [CrossRef]
- Mikaili, P.; Maadirad, S.; Moloudizargari, M.; Aghajanshakeri, S.; Sarahroodi, S. Therapeutic Uses and Pharmacological Properties of Garlic, Shallot, and Their Biologically Active Compounds. Iran J. Basic Med. Sci. 2013, 16, 1031–1048. [Google Scholar]
- Lim, H.; Min, D.S.; Park, H.; Kim, H.P. Flavonoids Interfere with NLRP3 Inflammasome Activation. Toxicol. Appl. Pharmacol. 2018, 355, 93–102. [Google Scholar] [CrossRef] [PubMed]
- Xiao, X.; Xu, X.; Li, F.; Xie, G.; Zhang, T. Anti-Inflammatory Treatment with β-Asarone Improves Impairments in Social Interaction and Cognition in MK-801 Treated Mice. Brain Res. Bull. 2019, 150, 150–159. [Google Scholar] [CrossRef] [PubMed]
- Lim, H.-W.; Kumar, H.; Kim, B.-W.; More, S.V.; Kim, I.-W.; Park, J.-I.; Park, S.-Y.; Kim, S.-K.; Choi, D.-K. β-Asarone (Cis-2,4,5-Trimethoxy-1-Allyl Phenyl), Attenuates pro-Inflammatory Mediators by Inhibiting NF-ΚB Signaling and the JNK Pathway in LPS Activated BV-2 Microglia Cells. Food Chem. Toxicol. 2014, 72, 265–272. [Google Scholar] [CrossRef]
- Kamatou, G.P.P.; Viljoen, A.M. A Review of the Application and Pharmacological Properties of α-Bisabolol and α-Bisabolol-Rich Oils. J. Am. Oil Chem. Soc. 2010, 87, 1–7. [Google Scholar] [CrossRef]
- Mitic, V.D.; Ilic, M.D.; Stankov-Jovanovic, V.P.; Stojanovic, G.S.; Dimitrijevic, M.V. Essential Oil Composition of Erica Spiculifolia Salisb—First Report. Nat. Prod. Res. 2018, 32, 222–224. [Google Scholar] [CrossRef]
- Lee, J.M.; Choi, S.S.; Park, M.H.; Jang, H.; Lee, Y.H.; Khim, K.W.; Oh, S.R.; Park, J.; Ryu, H.W.; Choi, J.H. Broussonetia Papyrifera Root Bark Extract Exhibits Anti-Inflammatory Effects on Adipose Tissue and Improves Insulin Sensitivity Potentially Via AMPK Activation. Nutrients 2020, 12, 773. [Google Scholar] [CrossRef]
- Ren, Y.; Wang, C.; Xu, J.; Wang, S. Cafestol and Kahweol: A Review on Their Bioactivities and Pharmacological Properties. Int. J. Mol. Sci. 2019, 20, 4238. [Google Scholar] [CrossRef] [PubMed]
- Quintans-Júnior, L.; Moreira, J.C.F.; Pasquali, M.A.B.; Rabie, S.M.S.; Pires, A.S.; Schröder, R.; Rabelo, T.K.; Santos, J.P.A.; Lima, P.S.S.; Cavalcanti, S.C.H.; et al. Antinociceptive Activity and Redox Profile of the Monoterpenes (+)-Camphene, p-Cymene, and Geranyl Acetate in Experimental Models. ISRN Toxicol. 2013, 2013, 459530. [Google Scholar] [CrossRef] [PubMed]
- Navarro, G.; Varani, K.; Reyes-Resina, I.; Sánchez de Medina, V.; Rivas-Santisteban, R.; Sánchez-Carnerero Callado, C.; Vincenzi, F.; Casano, S.; Ferreiro-Vera, C.; Canela, E.I.; et al. Cannabigerol Action at Cannabinoid CB1 and CB2 Receptors and at CB1–CB2 Heteroreceptor Complexes. Front. Pharmacol. 2018, 9, 632. [Google Scholar] [CrossRef]
- Gugliandolo, A.; Pollastro, F.; Grassi, G.; Bramanti, P.; Mazzon, E. In Vitro Model of Neuroinflammation: Efficacy of Cannabigerol, a Non-Psychoactive Cannabinoid. Int. J. Mol. Sci. 2018, 19, 1992. [Google Scholar] [CrossRef]
- Jolayemi, A.; Ojewole, J. Comparative Anti-Inflammatory Properties of Capsaicin and EthylaAcetate Extract of Capsicum Frutescens Linn [Solanaceae] in Rats. Afr. H. Sci. 2013, 13, 357–361. [Google Scholar] [CrossRef]
- Zhao, W.; Deng, C.; Han, Q.; Xu, H.; Chen, Y. Carvacrol May Alleviate Vascular Inflammation in Diabetic Db/Db Mice. Int. J. Mol. Med. 2020, 46, 977–988. [Google Scholar] [CrossRef] [PubMed]
- Ahmed, M.S.; Khan, A.; Kury, L.T.A.; Shah, F.A. Computational and Pharmacological Evaluation of Carveol for Antidiabetic Potential. Front. Pharmacol. 2020, 11, 919. [Google Scholar] [CrossRef]
- Marques, F.M.; Figueira, M.M.; Schmitt, E.F.P.; Kondratyuk, T.P.; Endringer, D.C.; Scherer, R.; Fronza, M. In Vitro Anti-Inflammatory Activity of Terpenes via Suppression of Superoxide and Nitric Oxide Generation and the NF-ΚB Signalling Pathway. Inflammopharmacology 2019, 27, 281–289. [Google Scholar] [CrossRef] [PubMed]
- Ribeiro-Filho, J.; da Silva Brandi, J.; Ferreira Costa, H.; Carla de Paula Medeiros, K.; Alves Leite, J.; Pergentino de Sousa, D.; Regina Piuvezam, M. Carvone Enantiomers Differentially Modulate IgE-Mediated Airway Inflammation in Mice. Int. J. Mol. Sci. 2020, 21, 9209. [Google Scholar] [CrossRef]
- Wang, F.; Han, Y.; Xi, S.; Lu, Y. Catechins Reduce Inflammation in Lipopolysaccharide-Stimulated Dental Pulp Cells by Inhibiting Activation of the NF-ΚB Pathway. Oral Dis. 2020, 26, 815–821. [Google Scholar] [CrossRef] [PubMed]
- Ma, D.; He, J.; He, D. Chamazulene Reverses Osteoarthritic Inflammation through Regulation of Matrix Metalloproteinases (MMPs) and NF-Kβ Pathway in in-Vitro and in-Vivo Models. Biosci. Biotechnol. Biochem. 2020, 84, 402–410. [Google Scholar] [CrossRef] [PubMed]
- Ximenes, R.M.; de Morais Nogueira, L.; Cassundé, N.M.R.; Jorge, R.J.B.; dos Santos, S.M.; Magalhães, L.P.M.; Silva, M.R.; de Barros Viana, G.S.; Araújo, R.M.; de Sena, K.X.; et al. Antinociceptive and Wound Healing Activities of Croton Adamantinus Müll. Arg. Essential Oil. J. Nat. Med. 2013, 67, 758–764. [Google Scholar] [CrossRef]
- Sá, R.C.S.; Andrade, L.; Oliveira, R.R.B.; Sousa, D. A Review on Anti-Inflammatory Activity of Phenylpropanoids Found in Essential Oils. Molecules 2014, 19, 1459–1480. [Google Scholar] [CrossRef]
- Lee, S.-C.; Wang, S.-Y.; Li, C.-C.; Liu, C.-T. Anti-Inflammatory Effect of Cinnamaldehyde and Linalool from the Leaf Essential Oil of Cinnamomum Osmophloeum Kanehira in Endotoxin-Induced Mice. J. Food Drug Anal. 2018, 26, 211–220. [Google Scholar] [CrossRef] [PubMed]
- Liao, J.-C.; Deng, J.-S.; Chiu, C.-S.; Hou, W.-C.; Huang, S.-S.; Shie, P.-H.; Huang, G.-J. Anti-Inflammatory Activities of Cinnamomum Cassia Constituents In Vitro and In Vivo. Evid. Based Complement. Altern. Med. 2012, 2012, 429320. [Google Scholar] [CrossRef]
- Liao, P.-C.; Yang, T.-S.; Chou, J.-C.; Chen, J.; Lee, S.-C.; Kuo, Y.-H.; Ho, C.-L.; Chao, L.K.-P. Anti-Inflammatory Activity of Neral and Geranial Isolated from Fruits of Litsea Cubeba Lour. J. Funct. Foods 2015, 19, 248–258. [Google Scholar] [CrossRef]
- Petrović, J.; Stojković, D.; Soković, M. Terpene core in selected aromatic and edible plants: Natural health improving agents. In Advances in Food and Nutrition Research; Elsevier: Amsterdam, The Netherlands, 2019; Volume 90, pp. 423–451. ISBN 978-0-12-816567-6. [Google Scholar]
- Mitoshi, M.; Kuriyama, I.; Nakayama, H.; Miyazato, H.; Sugimoto, K.; Kobayashi, Y.; Jippo, T.; Kuramochi, K.; Yoshida, H.; Mizushina, Y. Suppression of Allergic and Inflammatory Responses by Essential Oils Derived from Herbal Plants and Citrus Fruits. Int. J. Mol. Med. 2014, 33, 1643–1651. [Google Scholar] [CrossRef] [PubMed]
- Edwards, R.L.; Luis, P.B.; Nakashima, F.; Kunihiro, A.G.; Presley, S.-H.; Funk, J.L.; Schneider, C. Mechanistic Differences in the Inhibition of NF-ΚB by Turmeric and Its Curcuminoid Constituents. J. Agric. Food Chem. 2020, 68, 6154–6160. [Google Scholar] [CrossRef] [PubMed]
- Sandur, S.K.; Pandey, M.K.; Sung, B.; Ahn, K.S.; Murakami, A.; Sethi, G.; Limtrakul, P.; Badmaev, V.; Aggarwal, B.B. Curcumin, Demethoxycurcumin, Bisdemethoxycurcumin, Tetrahydrocurcumin and Turmerones Differentially Regulate Anti-Inflammatory and Anti-Proliferative Responses through a ROS-Independent Mechanism. Carcinogenesis 2007, 28, 1765–1773. [Google Scholar] [CrossRef] [PubMed]
- Rajagopal, K.; Varakumar, P.; Baliwada, A.; Byran, G. Activity of Phytochemical Constituents of Curcuma Longa (Turmeric) and Andrographis Paniculata against Coronavirus (COVID-19): An in Silico Approach. Future J. Pharm. Sci. 2020, 6, 104. [Google Scholar] [CrossRef]
- Chakraborty, S.; Karmenyan, A.; Tsai, J.-W.; Chiou, A. Inhibitory Effects of Curcumin and Cyclocurcumin in 1-Methyl-4-Phenylpyridinium (MPP+) Induced Neurotoxicity in Differentiated PC12 Cells. Sci. Rep. 2017, 7, 16977. [Google Scholar] [CrossRef]
- Zhou, Q.; Zhang, W.; He, Z.; Wu, B.; Shen, Z.; Shang, H.; Chen, T.; Wang, Q.; Chen, Y.; Han, S. The Possible Anti-Inflammatory Effect of Dehydrocostus Lactone on DSS-Induced Colitis in Mice. Evid. Based Complement. Altern. Med. 2020, 2020, 5659738. [Google Scholar] [CrossRef]
- Mahendran, S.; Badami, S.; Ravi, S.; Thippeswamy, B.S.; Veerapur, V.P. Synthesis and Evaluation of Analgesic and Anti-Inflammatory Activities of Most Active Free Radical Scavenging Derivatives of Embelin-A Structure-Activity Relationship. Chem. Pharm. Bull. 2011, 59, 913–919. [Google Scholar] [CrossRef][Green Version]
- Tasneem, S.; Liu, B.; Li, B.; Choudhary, M.I.; Wang, W. Molecular Pharmacology of Inflammation: Medicinal Plants as Anti-Inflammatory Agents. Pharmacol. Res. 2019, 139, 126–140. [Google Scholar] [CrossRef]
- Han, X.; Parker, T.L. Anti-Inflammatory Activity of Clove (Eugenia Caryophyllata) Essential Oil in Human Dermal Fibroblasts. Pharm. Biol. 2017, 55, 1619–1622. [Google Scholar] [CrossRef]
- Ma, L.; Mu, Y.; Zhang, Z.; Sun, Q. Eugenol Promotes Functional Recovery and Alleviates Inflammation, Oxidative Stress, and Neural Apoptosis in a Rat Model of Spinal Cord Injury. RNN 2018, 36, 659–668. [Google Scholar] [CrossRef]
- Saraphanchotiwitthaya, A.; Khorana, N.; Sripalakit, P. Comparative Anti-Inflammatory Activity of Eugenol and Eugenyl Acetate on the Murine Immune Response in Vitro. Songklanakarin J. Sci. Technol. 2019, 41, 641–648. [Google Scholar]
- Schnuch, A.; Uter, W.; Geier, J.; Lessmann, H.; Frosch, P.J. Contact Allergy to Farnesol in 2021 Consecutively Patch Tested Patients. Results of the IVDK*. Contact Dermat. 2004, 50, 117–121. [Google Scholar] [CrossRef]
- Ku, C.-M.; Lin, J.-Y. Farnesol, a Sesquiterpene Alcohol in Herbal Plants, Exerts Anti-Inflammatory and Antiallergic Effects on Ovalbumin-Sensitized and -Challenged Asthmatic Mice. Evid. Based Complement. Altern. Med. 2015, 2015, 387357. [Google Scholar] [CrossRef]
- Zhu, X.-Y.; Zhang, C.-L.; Lin, Y.; Dang, M.-Y. Ferruginol Alleviates Inflammation in Dextran Sulfate Sodium-Induced Colitis in Mice through Inhibiting COX-2, MMP-9 and NF-ΚB Signaling. Asian Pac. J. Trop. Biomed. 2020, 10, 308. [Google Scholar] [CrossRef]
- Semwal, R.B.; Semwal, D.K.; Combrinck, S.; Viljoen, A.M. Gingerols and Shogaols: Important Nutraceutical Principles from Ginger. Phytochemistry 2015, 117, 554–568. [Google Scholar] [CrossRef] [PubMed]
- Ma, R.-H.; Ni, Z.-J.; Zhu, Y.-Y.; Thakur, K.; Zhang, F.; Zhang, Y.-Y.; Hu, F.; Zhang, J.-G.; Wei, Z.-J. A Recent Update on the Multifaceted Health Benefits Associated with Ginger and Its Bioactive Components. Food Funct. 2021, 12, 519–542. [Google Scholar] [CrossRef] [PubMed]
- Mao, Q.-Q.; Xu, X.-Y.; Cao, S.-Y.; Gan, R.-Y.; Corke, H.; Beta, T.; Li, H.-B. Bioactive Compounds and Bioactivities of Ginger (Zingiber officinale Roscoe). Foods 2019, 8, 185. [Google Scholar] [CrossRef] [PubMed]
- Levita, J.; Syafitri, D.M.; Supu, R.D.; Mutakin, M.; Megantara, S.; Febrianti, M.; Diantini, A. Pharmacokinetics of 10-Gingerol and 6-Shogaol in the Plasma of Healthy Subjects Treated with Red Ginger (Zingiber Officinale Var. Rubrum) Suspension. Biomed. Rep. 2018, 9, 474–482. [Google Scholar] [CrossRef] [PubMed]
- Yang, R.; Yuan, B.-C.; Ma, Y.-S.; Zhou, S.; Liu, Y. The Anti-Inflammatory Activity of Licorice, a Widely Used Chinese Herb. Pharm. Biol. 2017, 55, 5–18. [Google Scholar] [CrossRef]
- Yu, J.-Y.; Ha, J.; Kim, K.-M.; Jung, Y.-S.; Jung, J.-C.; Oh, S. Anti-Inflammatory Activities of Licorice Extract and Its Active Compounds, Glycyrrhizic Acid, Liquiritin and Liquiritigenin, in BV2 Cells and Mice Liver. Molecules 2015, 20, 13041–13054. [Google Scholar] [CrossRef]
- Li, L.; Sapkota, M.; Kim, S.; Soh, Y. Herbacetin Inhibits Inducible Nitric Oxide Synthase via JNK and Nuclear Factor-ΚB in LPS-Stimulated RAW264.7 Cells. Eur. J. Pharm. 2015, 765, 115–123. [Google Scholar] [CrossRef]
- Li, L.; Sapkota, M.; Kim, S.; Soh, Y. Herbacetin Inhibits RANKL-Mediated Osteoclastogenesis in Vitro and Prevents Inflammatory Bone Loss in Vivo. Eur. J. Pharm. 2016, 777, 17–25. [Google Scholar] [CrossRef]
- Nuutinen, T. Medicinal Properties of Terpenes Found in Cannabis Sativa and Humulus Lupulus. Eur. J. Med. Chem. 2018, 157, 198–228. [Google Scholar] [CrossRef] [PubMed]
- Tanemoto, R.; Okuyama, T.; Matsuo, H.; Okumura, T.; Ikeya, Y.; Nishizawa, M. The Constituents of Licorice (Glycyrrhiza Uralensis) Differentially Suppress Nitric Oxide Production in Interleukin-1β-Treated Hepatocytes. Biochem. Biophys. Rep. 2015, 2, 153–159. [Google Scholar] [CrossRef]
- Ramos, A.G.B.; de Menezes, I.R.A.; da Silva, M.S.A.; Torres Pessoa, R.; de Lacerda Neto, L.J.; Rocha Santos Passos, F.; Melo Coutinho, H.D.; Iriti, M.; Quintans-Júnior, L.J. Antiedematogenic and Anti-Inflammatory Activity of the Monoterpene Isopulegol and Its β-Cyclodextrin (β-CD) Inclusion Complex in Animal Inflammation Models. Foods 2020, 9, 630. [Google Scholar] [CrossRef]
- Wu, J.; Li, Q.; Jin, L.; Qu, Y.; Liang, B.-B.; Zhu, X.-T.; Du, H.-Y.; Jie, L.-G.; Yu, Q.-H. Kirenol Inhibits the Function and Inflammation of Fibroblast-like Synoviocytes in Rheumatoid Arthritis in Vitro and in Vivo. Front. Immunol. 2019, 10, 1304. [Google Scholar] [CrossRef] [PubMed]
- Aoe, M.; Ueno-Iio, T.; Shibakura, M.; Shinohata, R.; Usui, S.; Arao, Y.; Ikeda, S.; Miyahara, N.; Tanimoto, M.; Kataoka, M. Lavender Essential Oil and Its Main Constituents Inhibit the Expression of TNF-α-Induced Cell Adhesion Molecules in Endothelial Cells. Acta Med. Okayama 2017, 71, 493–503. [Google Scholar] [CrossRef]
- Aziz, N.; Kim, M.-Y.; Cho, J.Y. Anti-Inflammatory Effects of Luteolin: A Review of in Vitro, in Vivo, and in Silico Studies. J. Ethnopharmacol. 2018, 225, 342–358. [Google Scholar] [CrossRef] [PubMed]
- Wang, Q.; Yang, Y.; Chen, K.; Li, D.; Tang, B.; Peng, K.; Wang, Z.; Yang, P.; Yang, D.; Yang, Y. Dietary Menthol Attenuates Inflammation and Cardiac Remodeling After Myocardial Infarction via the Transient Receptor Potential Melastatin 8. Am. J. Hypertens. 2020, 33, 223–233. [Google Scholar] [CrossRef]
- Zhang, Z.; Wu, X.; Zhang, L.; Mao, A.; Ma, X.; He, D. Menthol Relieves Acid Reflux Inflammation by Regulating TRPV1 in Esophageal Epithelial Cells. Biochem. Biophys. Res. Commun. 2020, 525, 113–120. [Google Scholar] [CrossRef]
- Xue, J.; Li, H.; Deng, X.; Ma, Z.; Fu, Q.; Ma, S. L-Menthone Confers Antidepressant-like Effects in an Unpredictable Chronic Mild Stress Mouse Model via NLRP3 Inflammasome-Mediated Inflammatory Cytokines and Central Neurotransmitters. Pharmacol. Biochem. Behav. 2015, 134, 42–48. [Google Scholar] [CrossRef]
- Tang, F.; Chen, F.; Ling, X.; Huang, Y.; Zheng, X.; Tang, Q.; Tan, X. Inhibitory Effect of Methyleugenol on IgE-Mediated Allergic Inflammation in RBL-2H3 Cells. Mediat. Inflamm. 2015, 2015, 463530. [Google Scholar] [CrossRef]
- Bezerra Rodrigues Dantas, L.; Silva, A.L.M.; da Silva Júnior, C.P.; Alcântara, I.S.; Correia de Oliveira, M.R.; Oliveira Brito Pereira Bezerra Martins, A.; Ribeiro-Filho, J.; Coutinho, H.D.M.; Rocha Santos Passos, F.; Quintans-Junior, L.J.; et al. Nootkatone Inhibits Acute and Chronic Inflammatory Responses in Mice. Molecules 2020, 25, 2181. [Google Scholar] [CrossRef] [PubMed]
- Qi, Y.; Cheng, X.; Gong, G.; Yan, T.; Du, Y.; Wu, B.; Bi, K.; Jia, Y. Synergistic Neuroprotective Effect of Schisandrin and Nootkatone on Regulating Inflammation, Apoptosis and Autophagy via the PI3K/AKT Pathway. Food Funct. 2020, 11, 2427–2438. [Google Scholar] [CrossRef]
- Valente, J.; Zuzarte, M.; Gonçalves, M.J.; Lopes, M.C.; Cavaleiro, C.; Salgueiro, L.; Cruz, M.T. Antifungal, Antioxidant and Anti-Inflammatory Activities of Oenanthe Crocata L. Essential Oil. Food Chem. Toxicol. 2013, 62, 349–354. [Google Scholar] [CrossRef] [PubMed]
- Tsai, S.-J.; Yin, M.-C. Antioxidative and Anti-Inflammatory Protection of Oleanolic Acid and Ursolic Acid in PC12 Cells. J. Food Sci. 2008, 73, H174–H178. [Google Scholar] [CrossRef]
- Takada, K.; Nakane, T.; Masuda, K.; Ishii, H. Ursolic Acid and Oleanolic Acid, Members of Pentacyclic Triterpenoid Acids, Suppress TNF-α-Induced E-Selectin Expression by Cultured Umbilical Vein Endothelial Cells. Phytomedicine 2010, 17, 1114–1119. [Google Scholar] [CrossRef]
- Fan, H.; Gao, Z.; Ji, K.; Li, X.; Wu, J.; Liu, Y.; Wang, X.; Liang, H.; Liu, Y.; Li, X.; et al. The in Vitro and in Vivo Anti-Inflammatory Effect of Osthole, the Major Natural Coumarin from Cnidium Monnieri (L.) Cuss, via the Blocking of the Activation of the NF-ΚB and MAPK/P38 Pathways. Phytomedicine 2019, 58, 152864. [Google Scholar] [CrossRef]
- Zhang, Z.-R.; Leung, W.N.; Cheung, H.Y.; Chan, C.W. Osthole: A Review on Its Bioactivities, Pharmacological Properties, and Potential as Alternative Medicine. Evid. Based Complement. Altern. Med. 2015, 2015, 919616. [Google Scholar] [CrossRef]
- Kwok, B.H.B.; Koh, B.; Ndubuisi, M.I.; Elofsson, M.; Crews, C.M. The Anti-Inflammatory Natural Product Parthenolide from the Medicinal Herb Feverfew Directly Binds to and Inhibits IκB Kinase. Chem. Biol. 2001, 8, 759–766. [Google Scholar] [CrossRef]
- Hehner, S.P.; Hofmann, T.G.; Dröge, W.; Schmitz, M.L. The Antiinflammatory Sesquiterpene Lactone Parthenolide Inhibits NF-Kappa B by Targeting the I Kappa B Kinase Complex. J. Immunol. 1999, 163, 5617–5623. [Google Scholar]
- Sur, R.; Martin, K.; Liebel, F.; Lyte, P.; Shapiro, S.; Southall, M. Anti-Inflammatory Activity of Parthenolide-Depleted Feverfew (Tanacetum Parthenium). Inflammopharmacology 2009, 17, 42–49. [Google Scholar] [CrossRef] [PubMed]
- Paulsen, E.; Christensen, L.P.; Andersen, K.E. Compositae Dermatitis from Airborne Parthenolide. Br. J. Derm. 2007, 156, 510–515. [Google Scholar] [CrossRef]
- Choi, Y.Y.; Kim, M.H.; Lee, H.; Jo, S.Y.; Yang, W.M. (R)-(+)-Pulegone Suppresses Allergic and Inflammation Responses on 2,4-Dinitrochlorobenzene-Induced Atopic Dermatitis in Mice Model. J. Dermatol. Sci. 2018, 91, 292–300. [Google Scholar] [CrossRef]
- Zaio, Y.; Mazzotta, M.; Sánchez, A.; Gomez, E.; Zunino, M.; Ponce, A. Effects of the Mint Monoterpene (R)-(+)-Pulegone Evaluated by Functional Observational Battery: A Potential Short Method. Pharmacogn. Res. 2019, 11, 31–36. [Google Scholar] [CrossRef]
- Kim, M.-J.; Yang, K.-W.; Kim, S.S.; Park, S.M.; Park, K.J.; Kim, K.S.; Choi, Y.H.; Cho, K.K.; Lee, N.H.; Hyun, C.-G. Chemical Composition and Anti-Inflammatory Effects of Essential Oil from Hallabong Flower. EXCLI J. 2013, 12, 933–942. [Google Scholar] [PubMed]
- Wu, X.; Zhu, H.; Yan, J.; Khan, M.; Yu, X. Santamarine Inhibits NF-κB Activation and Induces Mitochondrial Apoptosis in A549 Lung Adenocarcinoma Cells via Oxidative Stress. BioMed Res. Int. 2017, 2017, 4734127. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, K.F.; Moreira, F.M.F.; Santos, J.; Kassuya, C.A.L.; Croda, J.H.R.; Cardoso, C.A.L.; Vieira, M.D.C.; Góis Ruiz, A.L.T.; Ann Foglio, M.; de Carvalho, J.E.; et al. Antioxidant, Anti-Inflammatory, Antiproliferative and Antimycobacterial Activities of the Essential Oil of Psidium Guineense Sw. and Spathulenol. J. Ethnopharmacol. 2018, 210, 351–358. [Google Scholar] [CrossRef] [PubMed]
- Passos, F.F.d.B.; Lopes, E.M.; de Araújo, J.M.; de Sousa, D.P.; Veras, L.M.C.; Leite, J.R.S.A.; Almeida, F.R.d.C. Involvement of Cholinergic and Opioid System in γ-Terpinene-Mediated Antinociception. Evid. Based Complement. Altern. Med. 2015, 2015, 829414. [Google Scholar] [CrossRef]
- Held, S.; Schieberle, P.; Somoza, V. Characterization of α-Terpineol as an Anti-Inflammatory Component of Orange Juice by in Vitro Studies Using Oral Buccal Cells. J. Agric. Food Chem. 2007, 55, 8040–8046. [Google Scholar] [CrossRef]
- Li, A.; Wu, H.; Feng, Y.; Deng, S.; Hou, A.; Che, F.; Liu, Y.; Geng, Q.; Ni, H.; Wei, Y. A Strategy of Rapidly Screening out Herbicidal Chemicals from Eucalyptus Essential Oils. Pest. Manag. Sci. 2020, 76, 917–927. [Google Scholar] [CrossRef]
- Omonijo, F.A.; Liu, S.; Hui, Q.; Zhang, H.; Lahaye, L.; Bodin, J.-C.; Gong, J.; Nyachoti, M.; Yang, C. Thymol Improves Barrier Function and Attenuates Inflammatory Responses in Porcine Intestinal Epithelial Cells during Lipopolysaccharide (LPS)-Induced Inflammation. J. Agric. Food Chem. 2019, 67, 615–624. [Google Scholar] [CrossRef]
- Hossen, M.J.; Yang, W.S.; Kim, D.; Aravinthan, A.; Kim, J.-H.; Cho, J.Y. Thymoquinone: An IRAK1 Inhibitor with in Vivo and in Vitro Anti-Inflammatory Activities. Sci. Rep. 2017, 7, 42995. [Google Scholar] [CrossRef] [PubMed]
- Park, H.-H.; Kim, S.-G.; Kim, M.J.; Lee, J.; Choi, B.-K.; Jin, M.-H.; Lee, E. Suppressive Effect of Tomentosin on the Production of Inflammatory Mediators in RAW264.7 Cells. Biol. Pharm. Bull. 2014, 37, 1177–1183. [Google Scholar] [CrossRef]
- Hwangbo, C.; Lee, H.S.; Park, J.; Choe, J.; Lee, J.-H. The Anti-Inflammatory Effect of Tussilagone, from Tussilago Farfara, Is Mediated by the Induction of Heme Oxygenase-1 in Murine Macrophages. Int. Immunopharmacol. 2009, 9, 1578–1584. [Google Scholar] [CrossRef] [PubMed]
- Cheon, H.J.; Nam, S.-H.; Kim, J.-K. Tussilagone, a Major Active Component in Tussilago Farfara, Ameliorates Inflammatory Responses in Dextran Sulphate Sodium-Induced Murine Colitis. Chem. Biol. Interact. 2018, 294, 74–80. [Google Scholar] [CrossRef]
- Woźniak, Ł.; Skąpska, S.; Marszałek, K. Ursolic Acid—A Pentacyclic Triterpenoid with a Wide Spectrum of Pharmacological Activities. Molecules 2015, 20, 20614–20641. [Google Scholar] [CrossRef] [PubMed]
- Yang, I.J.; Lee, D.-U.; Shin, H.M. Inhibitory Effect of Valencene on the Development of Atopic Dermatitis-Like Skin Lesions in NC/Nga Mice. Evid. Based Complement. Altern. Med. 2016, 2016, 9370893. [Google Scholar] [CrossRef]
- Boukhatem, M.N.; Setzer, W.N. Aromatic Herbs, Medicinal Plant-Derived Essential Oils, and Phytochemical Extracts as Potential Therapies for Coronaviruses: Future Perspectives. Plants 2020, 9, 800. [Google Scholar] [CrossRef] [PubMed]
- Bhattacharya, R.; Dev, K.; Sourirajan, A. Antiviral Activity of Bioactive Phytocompounds against Coronavirus: An Update. J. Virol. Methods 2021, 290, 114070. [Google Scholar] [CrossRef]
- Khan, T.; Khan, M.A.; Mashwani, Z.-R.; Ullah, N.; Nadhman, A. Therapeutic Potential of Medicinal Plants against COVID-19: The Role of Antiviral Medicinal Metabolites. Biocatal. Agric. Biotechnol. 2021, 31, 101890. [Google Scholar] [CrossRef]
- Sagaya Jansi, R.; Khusro, A.; Agastian, P.; Alfarhan, A.; Al-Dhabi, N.A.; Arasu, M.V.; Rajagopal, R.; Barcelo, D.; Al-Tamimi, A. Emerging Paradigms of Viral Diseases and Paramount Role of Natural Resources as Antiviral Agents. Sci. Total Environ. 2021, 759, 143539. [Google Scholar] [CrossRef]
- Gilling, D.H.; Kitajima, M.; Torrey, J.R.; Bright, K.R. Antiviral Efficacy and Mechanisms of Action of Oregano Essential Oil and Its Primary Component Carvacrol against Murine Norovirus. J. Appl. Microbiol. 2014, 116, 1149–1163. [Google Scholar] [CrossRef]
- Astani, A.; Reichling, J.; Schnitzler, P. Comparative Study on the Antiviral Activity of Selected Monoterpenes Derived from Essential Oils: Antiviral activity of monoterpenes derived from essential oils. Phytother. Res. 2010, 24, 673–679. [Google Scholar] [CrossRef]
- Zhu, Z.; Lian, X.; Su, X.; Wu, W.; Marraro, G.A.; Zeng, Y. From SARS and MERS to COVID-19: A Brief Summary and Comparison of Severe Acute Respiratory Infections Caused by Three Highly Pathogenic Human Coronaviruses. Respir. Res. 2020, 21, 224. [Google Scholar] [CrossRef]
- Pilau, M.R.; Alves, S.H.; Weiblen, R.; Arenhart, S.; Cueto, A.P.; Lovato, L.T. Antiviral Activity of the Lippia Graveolens (Mexican Oregano) Essential Oil and Its Main Compound Carvacrol against Human and Animal Viruses. Braz. J. Microbiol. 2011, 42, 1616–1624. [Google Scholar] [CrossRef]
- Muramatsu, T.; Takemoto, C.; Kim, Y.-T.; Wang, H.; Nishii, W.; Terada, T.; Shirouzu, M.; Yokoyama, S. SARS-CoV 3CL Protease Cleaves Its C-Terminal Autoprocessing Site by Novel Subsite Cooperativity. Proc. Natl. Acad. Sci. USA 2016, 113, 12997–13002. [Google Scholar] [CrossRef]
- Zhang, L.; Lin, D.; Sun, X.; Curth, U.; Drosten, C.; Sauerhering, L.; Becker, S.; Rox, K.; Hilgenfeld, R. Crystal Structure of SARS-CoV-2 Main Protease Provides a Basis for Design of Improved α-Ketoamide Inhibitors. Science 2020, 368, 409–412. [Google Scholar] [CrossRef]
- Rathnayake, A.D.; Zheng, J.; Kim, Y.; Perera, K.D.; Mackin, S.; Meyerholz, D.K.; Kashipathy, M.M.; Battaile, K.P.; Lovell, S.; Perlman, S.; et al. 3C-like Protease Inhibitors Block Coronavirus Replication in Vitro and Improve Survival in MERS-CoV–Infected Mice. Sci. Transl. Med. 2020, 12, eabc5332. [Google Scholar] [CrossRef]
- Wen, C.-C.; Kuo, Y.-H.; Jan, J.-T.; Liang, P.-H.; Wang, S.-Y.; Liu, H.-G.; Lee, C.-K.; Chang, S.-T.; Kuo, C.-J.; Lee, S.-S.; et al. Specific Plant Terpenoids and Lignoids Possess Potent Antiviral Activities against Severe Acute Respiratory Syndrome Coronavirus. J. Med. Chem. 2007, 50, 4087–4095. [Google Scholar] [CrossRef]
- Kulkarni, S.A.; Nagarajan, S.K.; Ramesh, V.; Palaniyandi, V.; Selvam, S.P.; Madhavan, T. Computational Evaluation of Major Components from Plant Essential Oils as Potent Inhibitors of SARS-CoV-2 Spike Protein. J. Mol. Struct. 2020, 1221, 128823. [Google Scholar] [CrossRef]
- Rathinavel, T.; Palanisamy, M.; Palanisamy, S.; Subramanian, A.; Thangaswamy, S. Phytochemical 6-Gingerol—A Promising Drug of Choice for COVID-19. IJASE 2020, 6, 1482–1489. [Google Scholar] [CrossRef]
- Dong, W.; Wei, X.; Zhang, F.; Hao, J.; Huang, F.; Zhang, C.; Liang, W. A Dual Character of Flavonoids in Influenza A Virus Replication and Spread through Modulating Cell-Autonomous Immunity by MAPK Signaling Pathways. Sci. Rep. 2015, 4, 7237. [Google Scholar] [CrossRef] [PubMed]
- Solnier, J.; Fladerer, J.-P. Flavonoids: A Complementary Approach to Conventional Therapy of COVID-19? Phytochem. Rev. 2020, 1–23. [Google Scholar] [CrossRef] [PubMed]
- Teli, D.M.; Shah, M.B.; Chhabria, M.T. In Silico Screening of Natural Compounds as Potential Inhibitors of SARS-CoV-2 Main Protease and Spike RBD: Targets for COVID-19. Front. Mol. Biosci. 2021, 7, 599079. [Google Scholar] [CrossRef] [PubMed]
- Gupta, S.; Singh, V.; Varadwaj, P.K.; Chakravartty, N.; Katta, A.V.S.K.M.; Lekkala, S.P.; Thomas, G.; Narasimhan, S.; Reddy, A.R.; Reddy Lachagari, V.B. Secondary Metabolites from Spice and Herbs as Potential Multitarget Inhibitors of SARS-CoV-2 Proteins. J. Biomol. Struct. Dyn. 2020, 1–20. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Choudhir, G.; Shukla, S.K.; Sharma, M.; Tyagi, P.; Bhushan, A.; Rathore, M. Identification of Phytochemical Inhibitors against Main Protease of COVID-19 Using Molecular Modeling Approaches. J. Biomol. Struct. Dyn. 2021, 39, 3760–3770. [Google Scholar] [CrossRef]
- Stalin, A.; Lin, D.; Senthamarai Kannan, B.; Feng, Y.; Wang, Y.; Zhao, W.; Ignacimuthu, S.; Wei, D.-Q.; Chen, Y. An in-Silico Approach to Identify the Potential Hot Spots in SARS-CoV-2 Spike RBD to Block the Interaction with ACE2 Receptor. J. Biomol. Struct. Dyn. 2021, 1–16. [Google Scholar] [CrossRef]
- Jang, M.; Park, R.; Park, Y.-I.; Cha, Y.-E.; Yamamoto, A.; Lee, J.I.; Park, J. EGCG, a Green Tea Polyphenol, Inhibits Human Coronavirus Replication in Vitro. Biochem. Biophys. Res. Commun. 2021, 547, 23–28. [Google Scholar] [CrossRef]
- Cinatl, J.; Morgenstern, B.; Bauer, G.; Chandra, P.; Rabenau, H.; Doerr, H. Glycyrrhizin, an Active Component of Liquorice Roots, and Replication of SARS-Associated Coronavirus. Lancet 2003, 361, 2045–2046. [Google Scholar] [CrossRef]
- Hoever, G.; Baltina, L.; Michaelis, M.; Kondratenko, R.; Baltina, L.; Tolstikov, G.A.; Doerr, H.W.; Cinatl, J. Antiviral Activity of Glycyrrhizic Acid Derivatives against SARS−Coronavirus. J. Med. Chem. 2005, 48, 1256–1259. [Google Scholar] [CrossRef] [PubMed]
- Jo, S.; Kim, S.; Shin, D.H.; Kim, M.-S. Inhibition of SARS-CoV 3CL Protease by Flavonoids. J. Enzym. Inhib. Med. Chem. 2020, 35, 145–151. [Google Scholar] [CrossRef] [PubMed]
- Ghosh, R.; Chakraborty, A.; Biswas, A.; Chowdhuri, S. Identification of Polyphenols from Broussonetia Papyrifera as SARS CoV-2 Main Protease Inhibitors Using in Silico Docking and Molecular Dynamics Simulation Approaches. J. Biomol. Struct. Dyn. 2020, 1–14. [Google Scholar] [CrossRef] [PubMed]
- Rakshit, G.; Dagur, P.; Satpathy, S.; Patra, A.; Jain, A.; Ghosh, M. Flavonoids as Potential Therapeutics against Novel Coronavirus Disease-2019 (NCOVID-19). J. Biomol. Struct. Dyn. 2021, 1–13. [Google Scholar] [CrossRef]
- Xia, S.; Zhong, Z.; Gao, B.; Vong, C.T.; Lin, X.; Cai, J.; Gao, H.; Chan, G.; Li, C. The Important Herbal Pair for the Treatment of COVID-19 and Its Possible Mechanisms. Chin. Med. 2021, 16, 25. [Google Scholar] [CrossRef]
- Ben-Shabat, S.; Yarmolinsky, L.; Porat, D.; Dahan, A. Antiviral Effect of Phytochemicals from Medicinal Plants: Applications and Drug Delivery Strategies. Drug Deliv. Transl. Res. 2020, 10, 354–367. [Google Scholar] [CrossRef]
- Sharifi-Rad, J.; Salehi, B.; Schnitzler, P.; Ayatollahi, S.A.; Kobarfard, F.; Fathi, M.; Eisazadeh, M.; Sharifi-Rad, M. Susceptibility of Herpes Simplex Virus Type 1 to Monoterpenes Thymol, Carvacrol, p-Cymene and Essential Oils of Sinapis Arvensis L., Lallemantia Royleana Benth. and Pulicaria Vulgaris Gaertn. Cell Mol. Biol. 2017, 63, 42–47. [Google Scholar] [CrossRef]
- Omrani, M.; Keshavarz, M.; Nejad Ebrahimi, S.; Mehrabi, M.; McGaw, L.J.; Ali Abdalla, M.; Mehrbod, P. Potential Natural Products Against Respiratory Viruses: A Perspective to Develop Anti-COVID-19 Medicines. Front. Pharm. 2020, 11, 586993. [Google Scholar] [CrossRef]
- Rao, P.V.; Gan, S.H. Cinnamon: A Multifaceted Medicinal Plant. Evid. Based Complement. Altern. Med. 2014, 2014, 642942. [Google Scholar] [CrossRef]
- Rao, B.R.R.; Rajput, D.K.; Bhattacharya, A.K. Essential Oil Composition of Petiole of Cinnamomum Verum Bercht. & Presl. J. Spices Aromat. Crop. 2007, 16, 38–41. [Google Scholar]
- Singh, G.; Maurya, S.; deLampasona, M.P.; Catalan, C.A.N. A Comparison of Chemical, Antioxidant and Antimicrobial Studies of Cinnamon Leaf and Bark Volatile Oils, Oleoresins and Their Constituents. Food Chem. Toxicol. 2007, 45, 1650–1661. [Google Scholar] [CrossRef]
- Jirovetz, L.; Buchbauer, G.; Stoilova, I.; Stoyanova, A.; Krastanov, A.; Schmidt, E. Chemical Composition and Antioxidant Properties of Clove Leaf Essential Oil. J. Agric. Food Chem. 2006, 54, 6303–6307. [Google Scholar] [CrossRef]
- Chaieb, K.; Hajlaoui, H.; Zmantar, T.; Kahla-Nakbi, A.B.; Rouabhia, M.; Mahdouani, K.; Bakhrouf, A. The Chemical Composition and Biological Activity of Clove Essential Oil, Eugenia Caryophyllata (Syzigium Aromaticum L. Myrtaceae): A Short Review. Phytother. Res. 2007, 21, 501–506. [Google Scholar] [CrossRef]
- Amelia, B.; Saepudin, E.; Cahyana, A.H.; Rahayu, D.U.; Sulistyoningrum, A.S.; Haib, J. GC-MS Analysis of Clove (Syzygium Aromaticum) Bud Essential Oil from Java and Manado; AIP Publishing LLC: Depok, Indonesia, 2017; p. 030082. [Google Scholar]
- Vasile, C.; Sivertsvik, M.; Miteluţ, A.; Brebu, M.; Stoleru, E.; Rosnes, J.; Tănase, E.; Khan, W.; Pamfil, D.; Cornea, C.; et al. Comparative Analysis of the Composition and Active Property Evaluation of Certain Essential Oils to Assess Their Potential Applications in Active Food Packaging. Materials 2017, 10, 45. [Google Scholar] [CrossRef]
- Uddin, A.; Shahinuzzaman, M.; Rana, S.; Yaakob, Z. Study of chemical composition and medicinal properties of volatile oil from clove buds (Eugenia caryophyllus). IJPSR 2017, 8, 895–899. [Google Scholar]
- Dias, D.; Fontes, L.; Crotti, A.; Aarestrup, B.; Aarestrup, F.; da Silva Filho, A.; Corrêa, J. Copaiba Oil Suppresses Inflammatory Cytokines in Splenocytes of C57Bl/6 Mice Induced with Experimental Autoimmune Encephalomyelitis (EAE). Molecules 2014, 19, 12814–12826. [Google Scholar] [CrossRef]
- Xavier-Junior, F.H.; Maciuk, A.; Rochelle do Vale Morais, A.; Alencar, E.d.N.; Garcia, V.L.; Tabosa do Egito, E.S.; Vauthier, C. Development of a Gas Chromatography Method for the Analysis of Copaiba Oil. J. Chromatogr. Sci. 2017, 55, 969–978. [Google Scholar] [CrossRef]
- Chien, T.-C.; Lo, S.-F.; Ho, C.-L. Chemical Composition and Anti-Inflammatory Activity of Chamaecyparis Obtusa f.Formosana Wood Essential Oil from Taiwan. Nat. Prod. Commun. 2014, 9, 723–726. [Google Scholar]
- Ho, G.T.T.; Wangensteen, H.; Barsett, H. Elderberry and Elderflower Extracts, Phenolic Compounds, and Metabolites and Their Effect on Complement, RAW 264.7 Macrophages and Dendritic Cells. Int. J. Mol. Sci. 2017, 18, 584. [Google Scholar] [CrossRef]
- Dogan, G.; Kara, N.; Bagci, E.; Gur, S. Chemical Composition and Biological Activities of Leaf and Fruit Essential Oils from Eucalyptus camaldulensis. Z. Naturforsch. C 2017, 72, 483–489. [Google Scholar] [CrossRef] [PubMed]
- Barbosa, L.C.A.; Filomeno, C.A.; Teixeira, R.R. Chemical Variability and Biological Activities of Eucalyptus Spp. Essential Oils. Molecules 2016, 21, 1671. [Google Scholar] [CrossRef]
- Sharopov, F.S.; Zhang, H.; Setzer, W.N. Composition of Geranium (Pelargonium graveolens) Essential Oil from Tajikistan. Am. J. Essent. Oils Nat. Prod. 2014, 2, 13–16. [Google Scholar]
- Sköld, M.; Karlberg, A.-T.; Matura, M.; Börje, A. The Fragrance Chemical Beta-Caryophyllene-Air Oxidation and Skin Sensitization. Food Chem. Toxicol. 2006, 44, 538–545. [Google Scholar] [CrossRef]
- Prasad, S.; Tyagi, A.K. Ginger and Its Constituents: Role in Prevention and Treatment of Gastrointestinal Cancer. Gastroenterol. Res. Pract. 2015, 2015, 142979. [Google Scholar] [CrossRef]
- Białoń, M.; Krzyśko-Łupicka, T.; Nowakowska-Bogdan, E.; Wieczorek, P.P. Chemical Composition of Two Different Lavender Essential Oils and Their Effect on Facial Skin Microbiota. Molecules 2019, 24, 3270. [Google Scholar] [CrossRef] [PubMed]
- Ozcan, M.M.; Starovic, M.; Aleksic, G.; Figueredo, G.; Juhaimi, F.A.; Chalchat, J.-C. Chemical Composition and Antifungal Activity of Lavender (Lavandula stoechas) Oil. Nat. Prod. Commun. 2018, 13, 895–898. [Google Scholar]
- Prusinowska, R.; Śmigielski, K.B. Composition, Biological Properties and Therapeutic Effects of Lavender (Lavandula Angustifolia L). A Review. Herba Pol. 2014, 60, 56–66. [Google Scholar] [CrossRef]
- Dong, G.; Bai, X.; Aimila, A.; Aisa, H.; Maiwulanjiang, M. Study on Lavender Essential Oil Chemical Compositions by GC-MS and Improved PGC. Molecules 2020, 25, 3166. [Google Scholar] [CrossRef]
- Brügger, B.P.; Martínez, L.C.; Plata-Rueda, A.; e Castro, B.M.d.C.; Soares, M.A.; Wilcken, C.F.; Carvalho, A.G.; Serrão, J.E.; Zanuncio, J.C. Bioactivity of the Cymbopogon citratus (Poaceae) Essential Oil and Its Terpenoid Constituents on the Predatory Bug, Podisus nigrispinus (Heteroptera: Pentatomidae). Sci. Rep. 2019, 9, 8358. [Google Scholar] [CrossRef]
- Li, M.; Liu, B.; Bernigaud, C.; Fischer, K.; Guillot, J.; Fang, F. Lemongrass (Cymbopogon citratus) Oil: A Promising Miticidal and Ovicidal Agent against Sarcoptes scabiei. PLoS Negl. Trop. Dis. 2020, 14, e0008225. [Google Scholar] [CrossRef]
- Tajidin, N. E Chemical Composition and Citral Content in Lemongrass (Cymbopogon Citratus) Essential Oil at Three Maturity Stages. Afr. J. Biotechnol. 2012, 11, 2685–2693. [Google Scholar] [CrossRef]
- Snoussi, M.; Noumi, E.; Trabelsi, N.; Flamini, G.; Papetti, A.; De Feo, V. Mentha spicata Essential Oil: Chemical Composition, Antioxidant and Antibacterial Activities against Planktonic and Biofilm Cultures of Vibrio spp. Strains. Molecules 2015, 20. [Google Scholar] [CrossRef]
- Bardaweel, S.K.; Bakchiche, B.; ALSalamat, H.A.; Rezzoug, M.; Gherib, A.; Flamini, G. Chemical Composition, Antioxidant, Antimicrobial and Antiproliferative Activities of Essential Oil of Mentha spicata L. (Lamiaceae) from Algerian Saharan Atlas. BMC Complement. Altern. Med. 2018, 18, 201. [Google Scholar] [CrossRef]
- Wu, Z.; Tan, B.; Liu, Y.; Dunn, J.; Martorell Guerola, P.; Tortajada, M.; Cao, Z.; Ji, P. Chemical Composition and Antioxidant Properties of Essential Oils from Peppermint, Native Spearmint and Scotch Spearmint. Molecules 2019, 24, 2825. [Google Scholar] [CrossRef]
- Rostro-Alanis, M.d.J.; Báez-González, J.; Torres-Alvarez, C.; Parra-Saldívar, R.; Rodriguez-Rodriguez, J.; Castillo, S. Chemical Composition and Biological Activities of Oregano Essential Oil and Its Fractions Obtained by Vacuum Distillation. Molecules 2019, 24, 1904. [Google Scholar] [CrossRef]
- Marchese, A.; Arciola, C.; Barbieri, R.; Silva, A.; Nabavi, S.; Tsetegho Sokeng, A.; Izadi, M.; Jafari, N.; Suntar, I.; Daglia, M.; et al. Update on Monoterpenes as Antimicrobial Agents: A Particular Focus on p-Cymene. Materials 2017, 10, 947. [Google Scholar] [CrossRef]
- Mechergui, K.; Jaouadi, W.; Coelho, J.P.; Khouja, M.L. Effect of Harvest Year on Production, Chemical Composition and Antioxidant Activities of Essential Oil of Oregano (Origanum vulgare subsp glandulosum (Desf.) Ietswaart) Growing in North Africa. Ind. Crop. Prod. 2016, 90, 32–37. [Google Scholar] [CrossRef]
- Ryu, H.W.; Park, M.H.; Kwon, O.-K.; Kim, D.-Y.; Hwang, J.-Y.; Jo, Y.H.; Ahn, K.-S.; Hwang, B.Y.; Oh, S.-R. Anti-Inflammatory Flavonoids from Root Bark of Broussonetia papyrifera in LPS-Stimulated RAW264.7 Cells. Bioorgan. Chem. 2019, 92, 103233. [Google Scholar] [CrossRef]
- Malaník, M.; Treml, J.; Leláková, V.; Nykodýmová, D.; Oravec, M.; Marek, J.; Šmejkal, K. Anti-Inflammatory and Antioxidant Properties of Chemical Constituents of Broussonetia papyrifera. Bioorgan. Chem. 2020, 104, 104298. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, E.; Bail, S.; Buchbauer, G.; Stoilova, I.; Atanasova, T.; Stoyanova, A.; Krastanov, A.; Jirovetz, L. Chemical Composition, Olfactory Evaluation and Antioxidant Effects of Essential Oil from Mentha × piperita. Nat. Prod. Commun. 2009, 4, 1107–1112. [Google Scholar] [CrossRef]
- Reddy, D.N.; Al-Rajab, A.J.; Sharma, M.; Mylabathula, M.M.; Gowkanapalli, R.R.; Albratty, M. Chemical Constituents, in Vitro Antibacterial and Antifungal Activity of Mentha × piperita L. (Peppermint) Essential Oils. J. King Saud Univ. Sci. 2019, 31, 528–533. [Google Scholar] [CrossRef]
- Hcini, K.; Sotomayor, J.A.; Jordan, M.J.; Bouzid, S. Chemical Composition of the Essential Oil of Rosemary (Rosmarinus officinalis L.) of Tunisian Origin. Asian J. Chem. 2013, 25, 2601. [Google Scholar] [CrossRef]
- Fierascu, I.; Dinu-Pirvu, C.E.; Fierascu, R.C.; Velescu, B.S.; Anuta, V.; Ortan, A.; Jinga, V. Phytochemical Profile and Biological Activities of Satureja Hortensis L.: A Review of the Last Decade. Molecules 2018, 23, 2458. [Google Scholar] [CrossRef]
- Hamidpour, R.; Hamidpour, S.; Hamidpour, M.; Shahlari, M.; Sohraby, M. Summer Savory: From the Selection of Traditional Applications to the Novel Effect in Relief, Prevention, and Treatment of a Number of Serious Illnesses Such as Diabetes, Cardiovascular Disease, Alzheimer’s Disease, and Cancer. J. Tradit. Complement. Med. 2014, 4, 140–144. [Google Scholar] [CrossRef]
- Carson, C.F.; Hammer, K.A.; Riley, T.V. Melaleuca alternifolia (Tea Tree) Oil: A Review of Antimicrobial and Other Medicinal Properties. Clin. Microbiol. Rev. 2006, 19, 50–62. [Google Scholar] [CrossRef]
- Borugă, O.; Jianu, C.; Mişcă, C.; Goleţ, I.; Gruia, A.T.; Horhat, F.G. Thymus Vulgaris Essential Oil: Chemical Composition and Antimicrobial Activity. J. Med. Life 2014, 7, 56–60. [Google Scholar]
- Porte, A.; Godoy, R. Chemical Composition of Thymus vulgaris L. (Thyme) Essential Oil from the Rio de Janeiro State, Brazil. J. Serb. Chem. Soc. 2008, 73, 307–310. [Google Scholar] [CrossRef]
- Alsaraf, S.; Hadi, Z.; Al-Lawati, W.M.; Al Lawati, A.A.; Khan, S.A. Chemical Composition, in Vitro Antibacterial and Antioxidant Potential of Omani Thyme Essential Oil along with in Silico Studies of Its Major Constituent. J. King Saud Univ.Sci. 2020, 32, 1021–1028. [Google Scholar] [CrossRef]
- Patel, B.; Sharma, S.; Nair, N.; Majeed, J.; Goyal, R.K.; Dhobi, M. Therapeutic Opportunities of Edible Antiviral Plants for COVID-19. Mol. Cell. Biochem. 2021, 476, 2345–2364. [Google Scholar] [CrossRef]
- McClintock, M. Menstrual Synchrony and Suppression. Nature 1971, 229, 244–245. [Google Scholar] [CrossRef]
- Stern, K.; McClintock, M.K. Regulation of Ovulation by Human Pheromones. Nature 1998, 392, 177–179. [Google Scholar] [CrossRef]
- Miller, S.L.; Maner, J.K. Scent of a Woman: Men’s Testosterone Responses to Olfactory Ovulation Cues. Psychol. Sci. 2010, 21, 276–283. [Google Scholar] [CrossRef]
- Tarumi, W.; Shinohara, K. Women’s Body Odour during the Ovulatory Phase Modulates Testosterone and Cortisol Levels in Men. PLoS ONE 2020, 15, e0230838. [Google Scholar] [CrossRef]
- Parma, V.; Tirindelli, R.; Bisazza, A.; Massaccesi, S.; Castiello, U. Subliminally Perceived Odours Modulate Female Intrasexual Competition: An Eye Movement Study. PLoS ONE 2012, 7, e30645. [Google Scholar] [CrossRef] [PubMed]
- Sobel, N.; Prabhakaran, V.; Hartley, C.A.; Desmond, J.E.; Glover, G.H.; Sullivan, E.V.; Gabrieli, J.D. Blind Smell: Brain Activation Induced by an Undetected Air-Borne Chemical. Brain 1999, 122, 209–217. [Google Scholar] [CrossRef] [PubMed]
- Savic, I.; Berglund, H.; Gulyas, B.; Roland, P. Smelling of Odorous Sex Hormone-like Compounds Causes Sex-Differentiated Hypothalamic Activations in Humans. Neuron 2001, 31, 661–668. [Google Scholar] [CrossRef]
- Gelstein, S.; Yeshurun, Y.; Rozenkrantz, L.; Shushan, S.; Frumin, I.; Roth, Y.; Sobel, N. Human Tears Contain a Chemosignal. Science 2011, 331, 226–230. [Google Scholar] [CrossRef]
- Preti, G.; Wysocki, C.J.; Barnhart, K.T.; Sondheimer, S.J.; Leyden, J.J. Male Axillary Extracts Contain Pheromones That Affect Pulsatile Secretion of Luteinizing Hormone and Mood in Women Recipients. Biol. Reprod. 2003, 68, 2107–2113. [Google Scholar] [CrossRef]
- Rozenkrantz, L.; Weissgross, R.; Weiss, T.; Ravreby, I.; Frumin, I.; Shushan, S.; Gorodisky, L.; Reshef, N.; Holzman, Y.; Pinchover, L.; et al. Unexplained Repeated Pregnancy Loss Is Associated with Altered Perceptual and Brain Responses to Men’s Body-Odor. eLife 2020, 9, e55305. [Google Scholar] [CrossRef]
- Schaal, B.; Saxton, T.K.; Loos, H.; Soussignan, R.; Durand, K. Olfaction Scaffolds the Developing Human from Neonate to Adolescent and Beyond. Philos. Trans. R. Soc. Lond. B Biol. Sci. 2020, 375, 20190261. [Google Scholar] [CrossRef] [PubMed]
- Tashiro, S.; Yamaguchi, R.; Ishikawa, S.; Sakurai, T.; Kajiya, K.; Kanmura, Y.; Kuwaki, T.; Kashiwadani, H. Odour-Induced Analgesia Mediated by Hypothalamic Orexin Neurons in Mice. Sci. Rep. 2016, 6, 37129. [Google Scholar] [CrossRef] [PubMed]
- Brand, G. Olfactory/Trigeminal Interactions in Nasal Chemoreception. Neurosci. Biobehav. Rev. 2006, 30, 908–917. [Google Scholar] [CrossRef] [PubMed]
- Fothergill, L.; Callaghan, B.; Rivera, L.; Lieu, T.; Poole, D.; Cho, H.-J.; Bravo, D.; Furness, J. Effects of Food Components That Activate TRPA1 Receptors on Mucosal Ion Transport in the Mouse Intestine. Nutrients 2016, 8, 623. [Google Scholar] [CrossRef]
- Kashiwadani, H.; Higa, Y.; Sugimura, M.; Kuwaki, T. Linalool Odor-induced Analgesia Is Triggered by TRPA1-Independent Pathway in Mice. Behav. Brain Funct. 2021, 17, 3. [Google Scholar] [CrossRef] [PubMed]
- Buchbauer, G.; Jirovetz, L.; Jäger, W.; Plank, C.; Dietrich, H. Fragrance Compounds and Essential Oils with Sedative Effects upon Inhalation. J. Pharm. Sci. 1993, 82, 660–664. [Google Scholar] [CrossRef]
- Aprotosoaie, A.C.; Hăncianu, M.; Costache, I.-I.; Miron, A. Linalool: A Review on a Key Odorant Molecule with Valuable Biological Properties: Linalool: A Key Odorant Molecule. Flavour Fragr. J. 2014, 29, 193–219. [Google Scholar] [CrossRef]
- Kuroda, K.; Inoue, N.; Ito, Y.; Kubota, K.; Sugimoto, A.; Kakuda, T.; Fushiki, T. Sedative Effects of the Jasmine Tea Odor and (R)-(-)-Linalool, One of Its Major Odor Components, on Autonomic Nerve Activity and Mood States. Eur. J. Appl. Physiol. 2005, 95, 107–114. [Google Scholar] [CrossRef] [PubMed]
- Sowndhararajan, K.; Kim, S. Influence of Fragrances on Human Psychophysiological Activity: With Special Reference to Human Electroencephalographic Response. Sci. Pharm. 2016, 84, 724–751. [Google Scholar] [CrossRef]
- Hopkins, C.; Alanin, M.; Philpott, C.; Harries, P.; Whitcroft, K.; Qureishi, A.; Anari, S.; Ramakrishnan, Y.; Sama, A.; Davies, E.; et al. Management of New Onset Loss of Sense of Smell during the COVID-19 Pandemic—BRS Consensus Guidelines. Clin. Otolaryngol. 2021, 46, 16–22. [Google Scholar] [CrossRef] [PubMed]
- Rudbäck, J.; Hagvall, L.; Börje, A.; Nilsson, U.; Karlberg, A.-T. Characterization of Skin Sensitizers from Autoxidized Citronellol—Impact of the Terpene Structure on the Autoxidation Process. Contact Dermat. 2014, 70, 329–339. [Google Scholar] [CrossRef] [PubMed]
- Sköld, M.; Börje, A.; Matura, M.; Karlberg, A.-T. Studies on the Autoxidation and Sensitizing Capacity of the Fragrance Chemical Linalool, Identifying a Linalool Hydroperoxide. Contact Dermat. 2002, 46, 267–272. [Google Scholar] [CrossRef] [PubMed]
- Hagvall, L.; Bäcktorp, C.; Svensson, S.; Nyman, G.; Börje, A.; Karlberg, A.-T. Fragrance Compound Geraniol Forms Contact Allergens on Air Exposure. Identification and Quantification of Oxidation Products and Effect on Skin Sensitization. Chem. Res. Toxicol. 2007, 20, 807–814. [Google Scholar] [CrossRef] [PubMed]
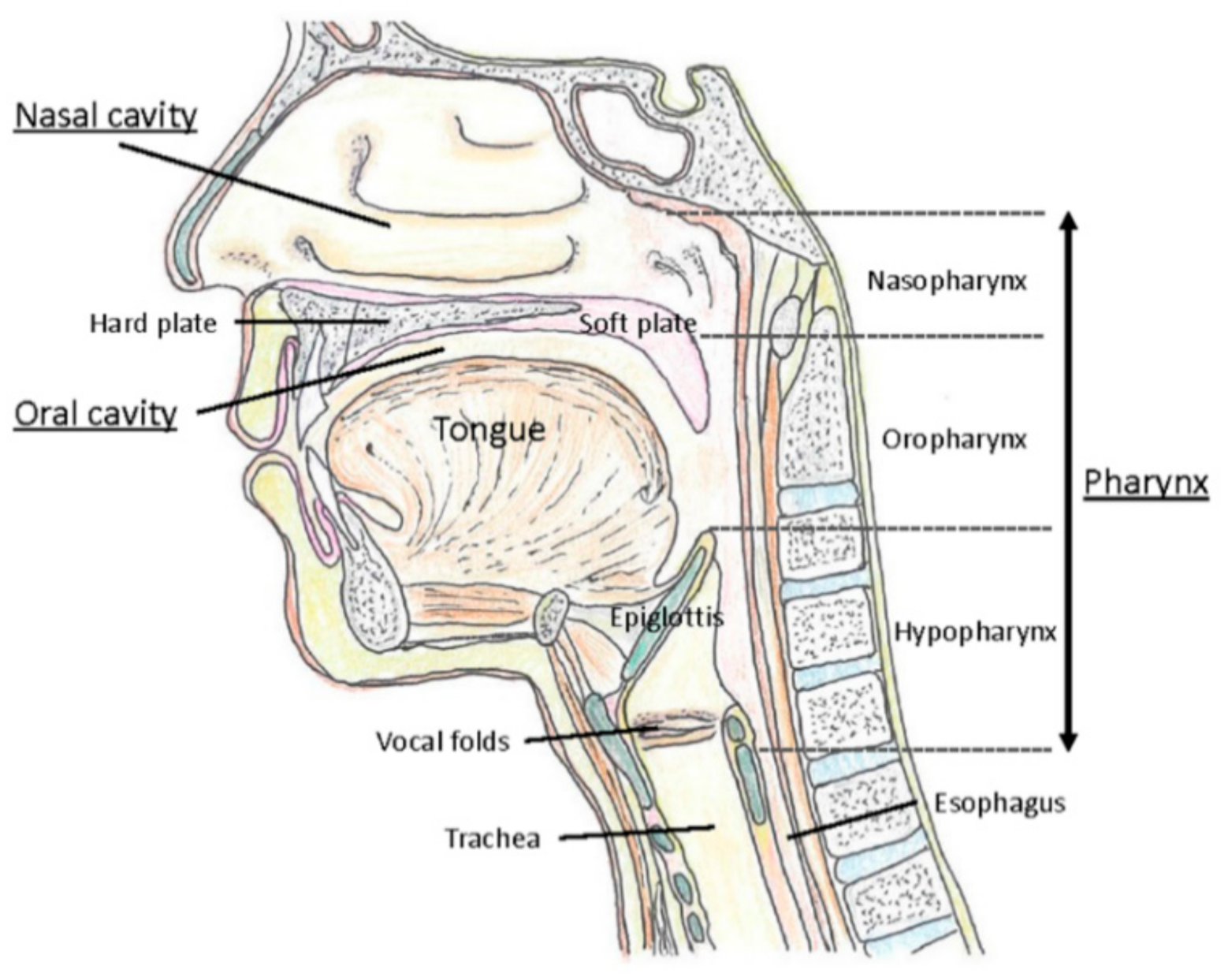
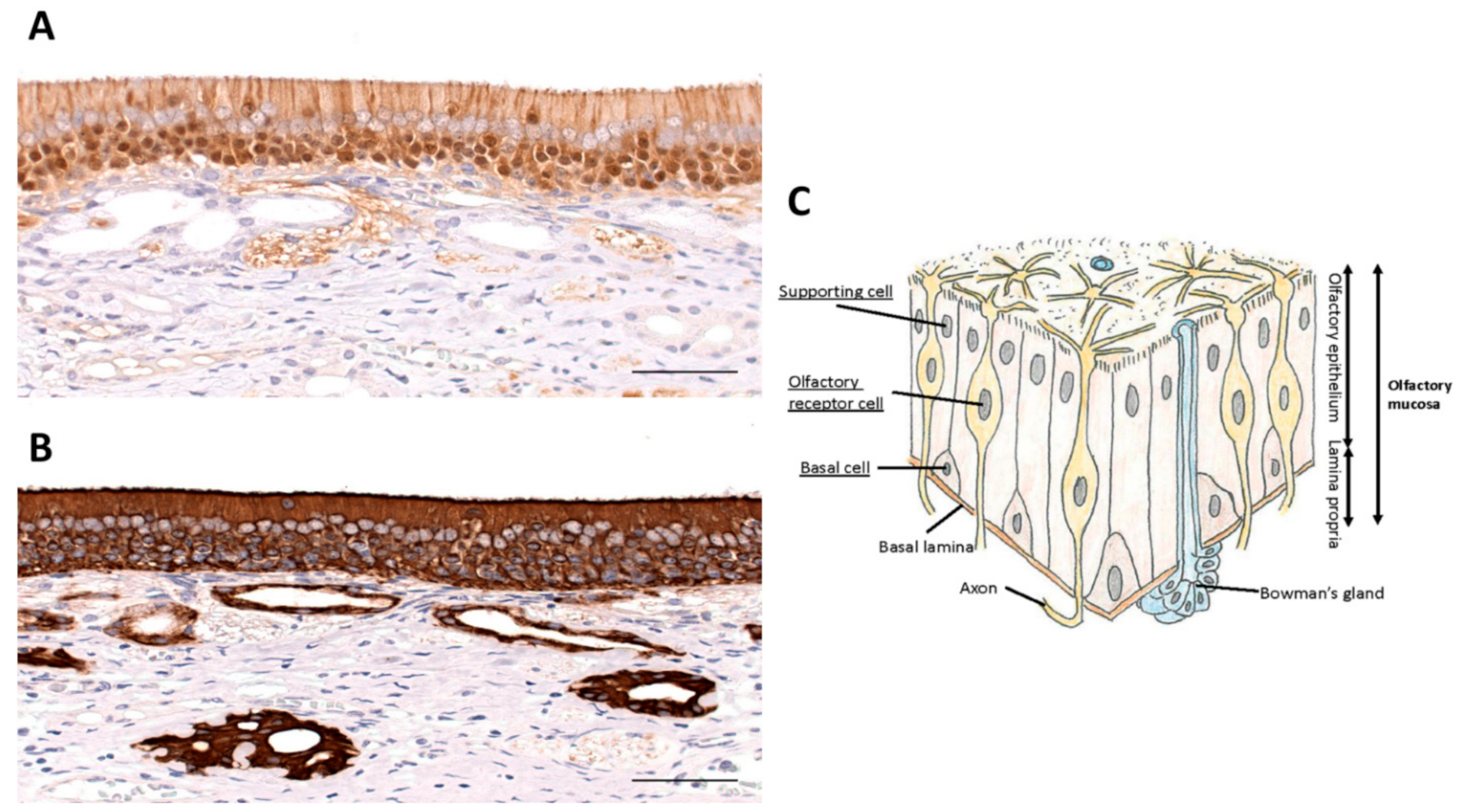
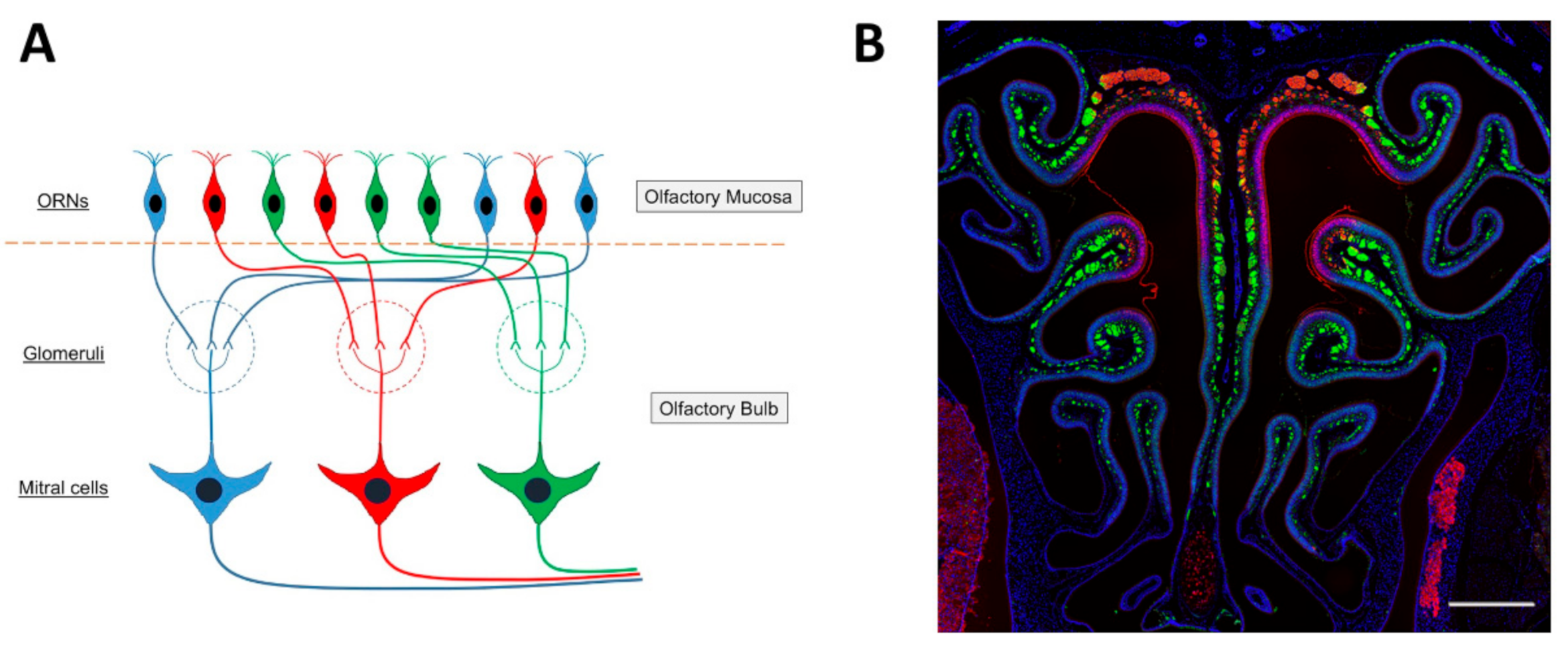
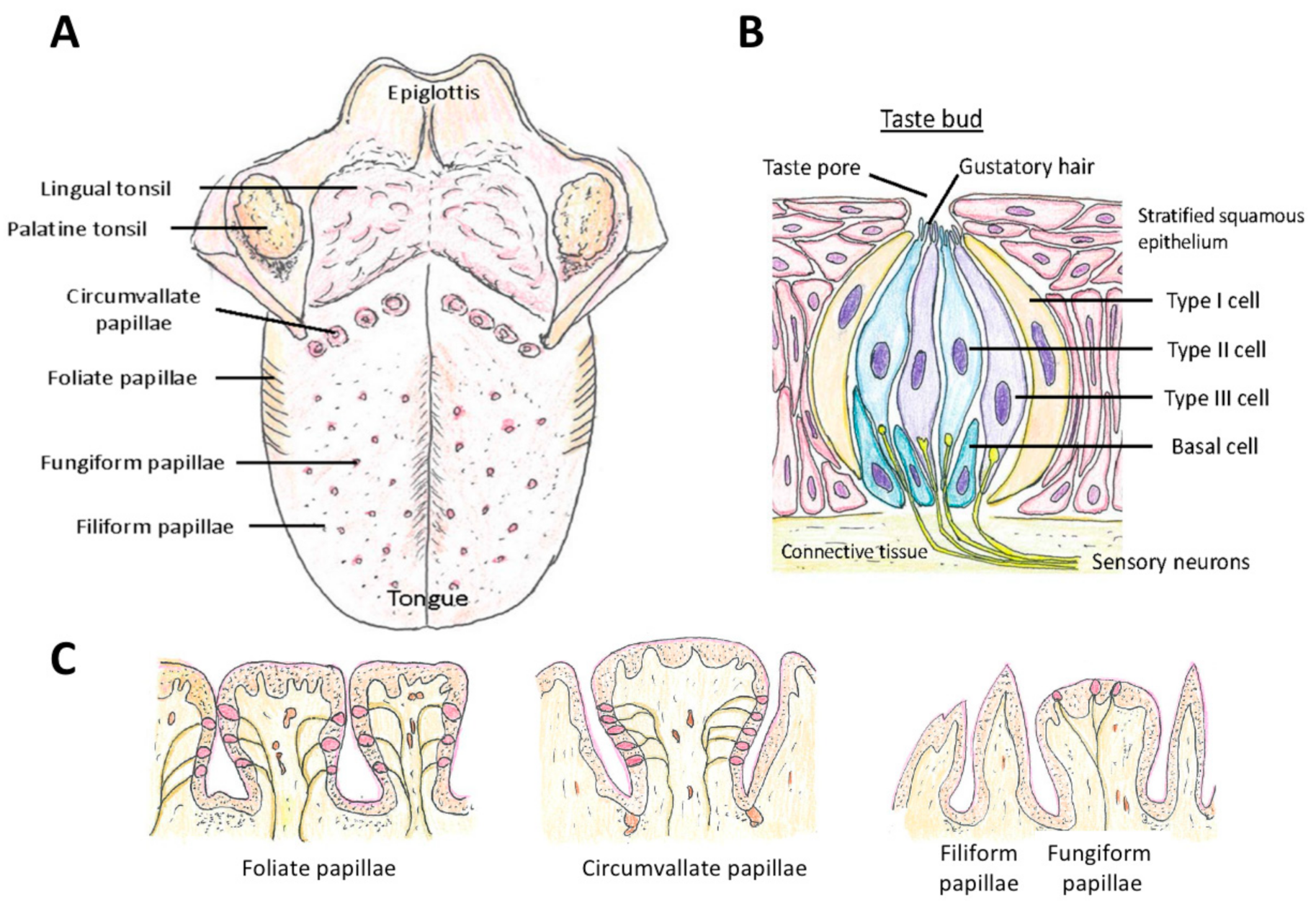
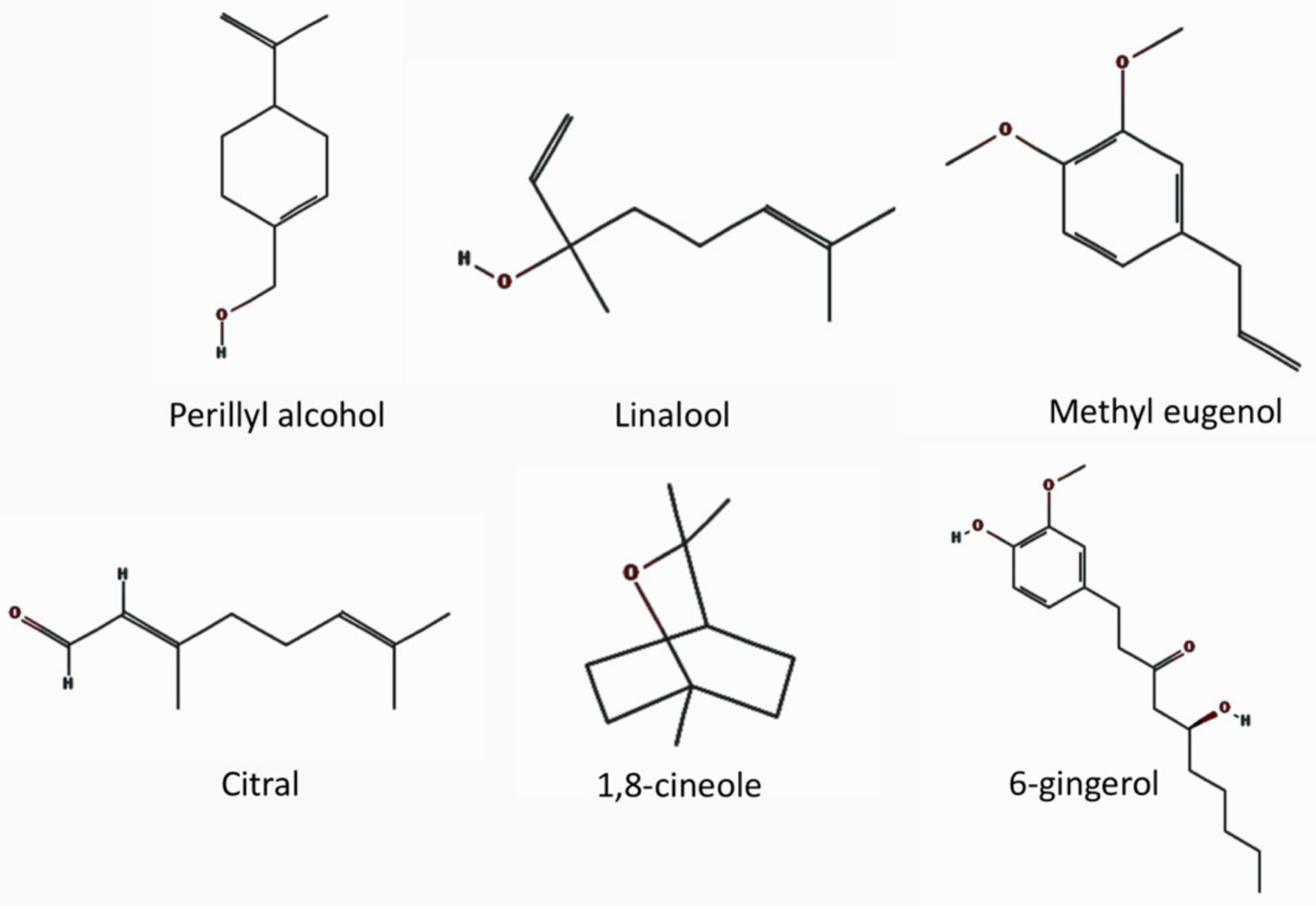
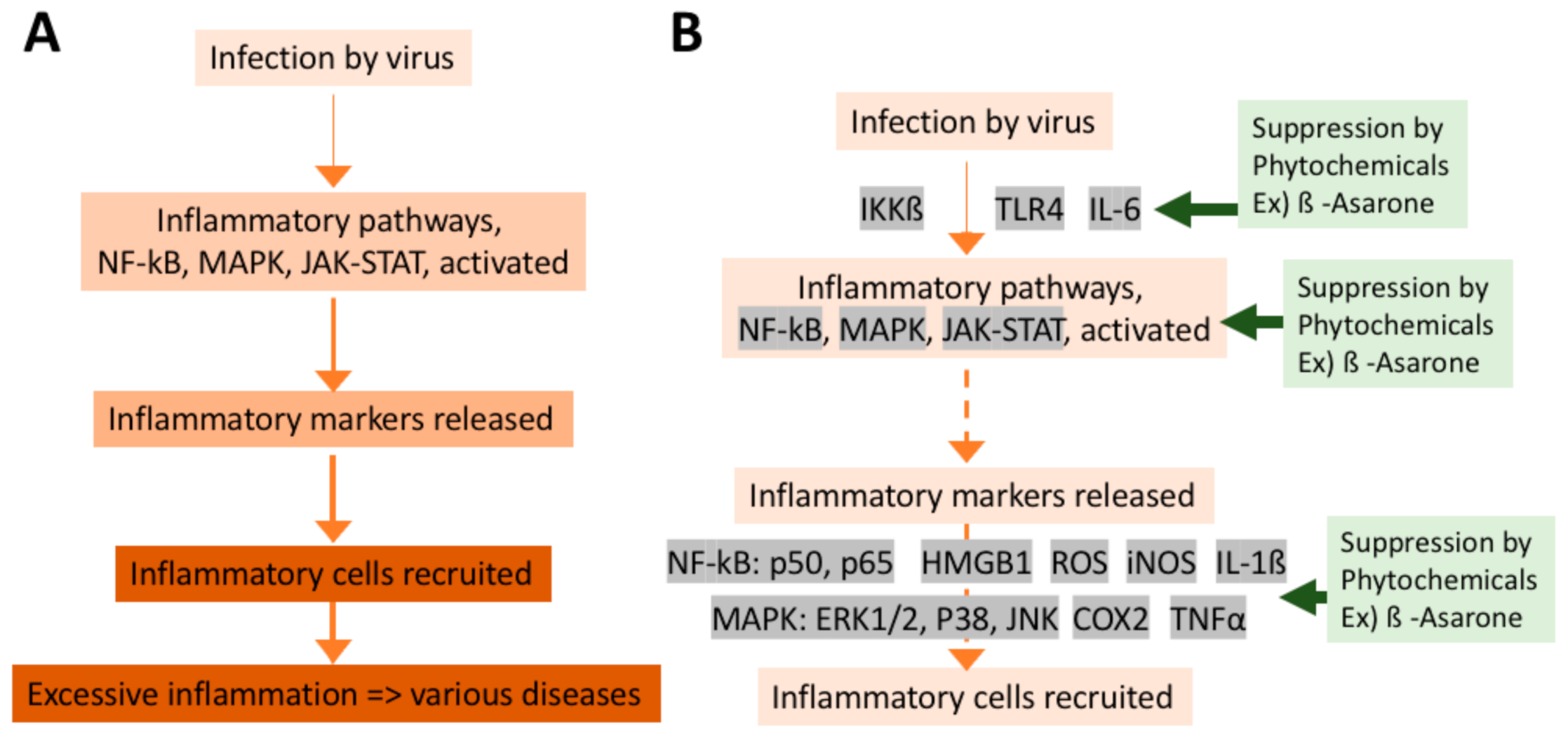
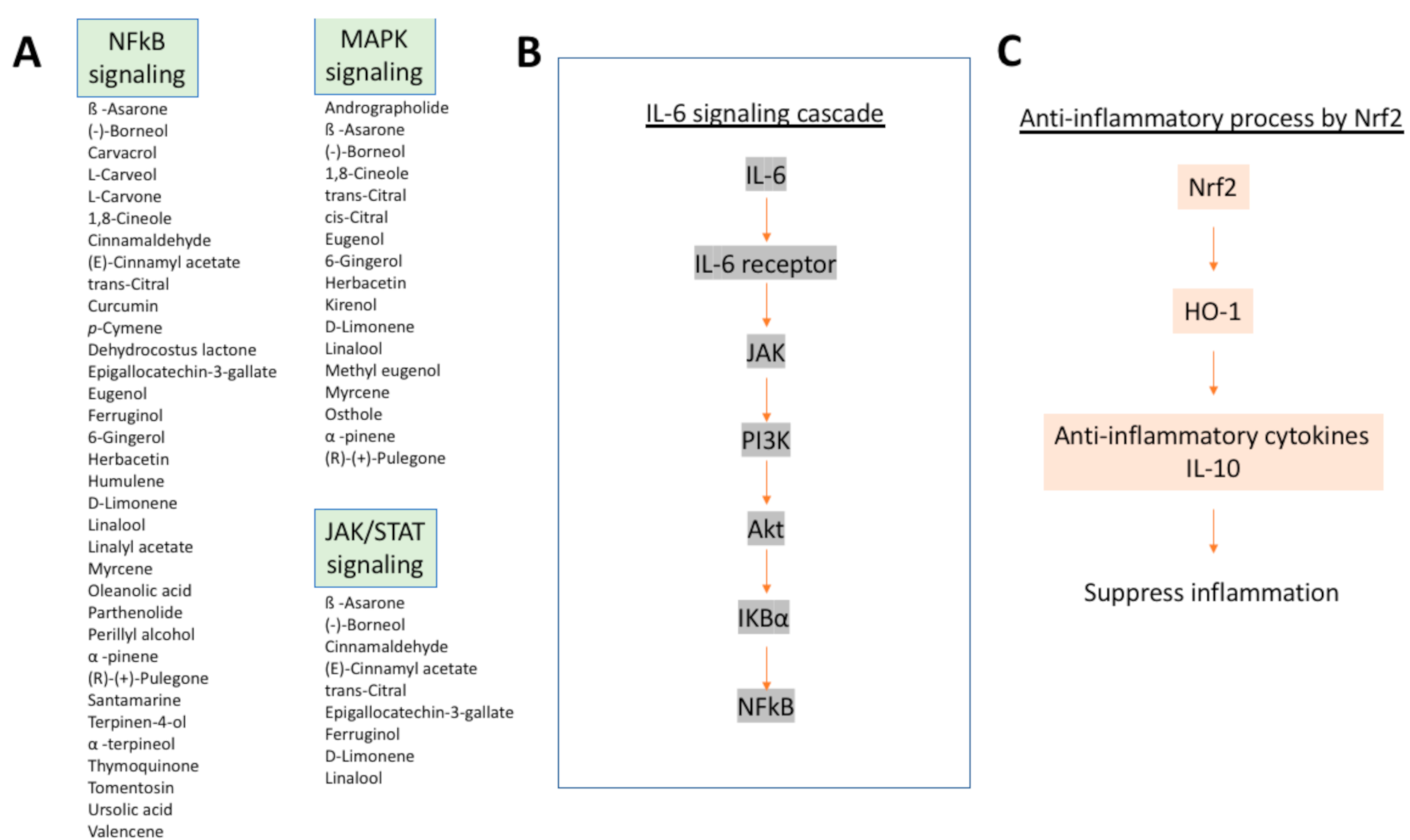
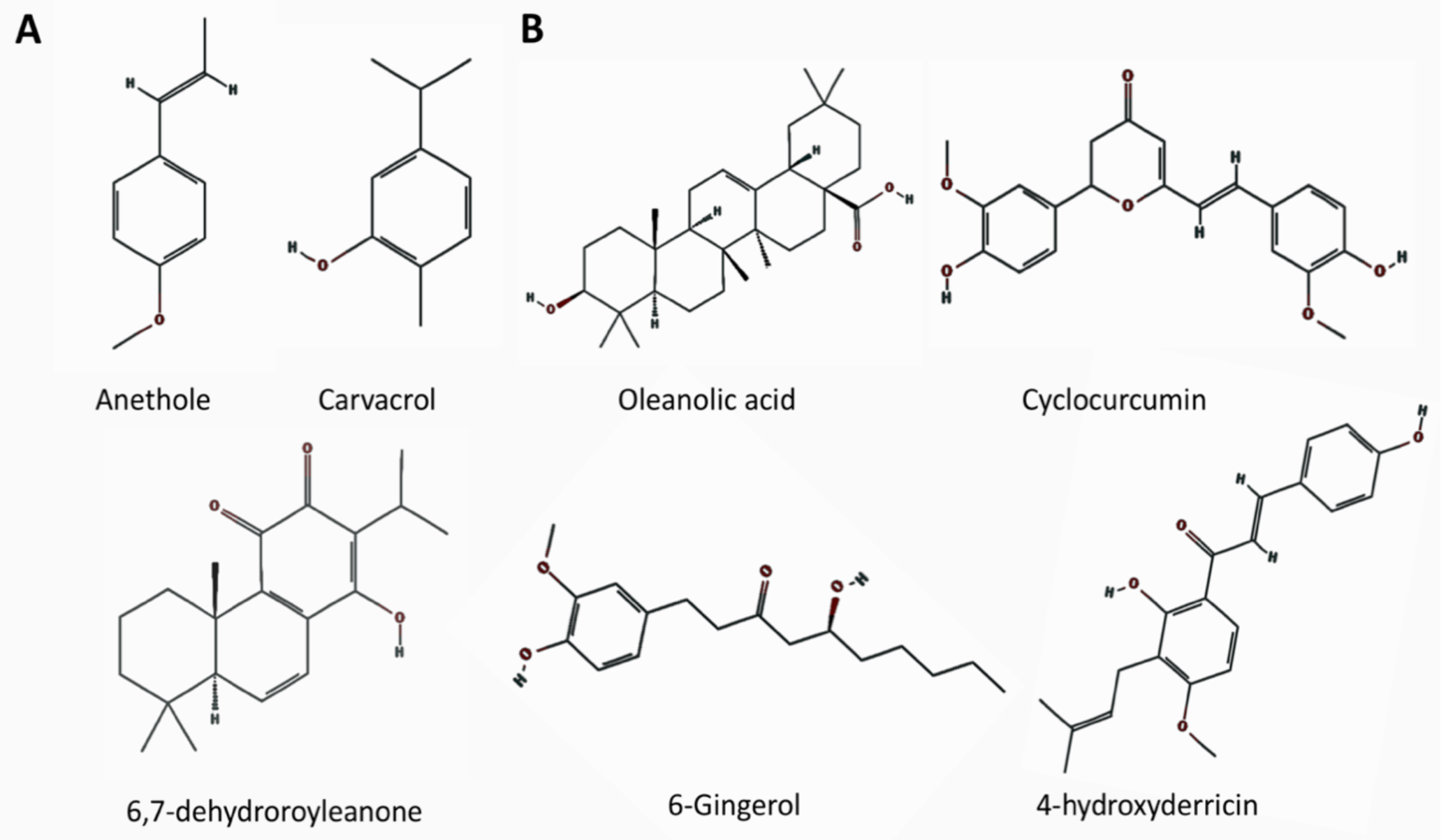
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Koyama, S.; Kondo, K.; Ueha, R.; Kashiwadani, H.; Heinbockel, T. Possible Use of Phytochemicals for Recovery from COVID-19-Induced Anosmia and Ageusia. Int. J. Mol. Sci. 2021, 22, 8912. https://doi.org/10.3390/ijms22168912
Koyama S, Kondo K, Ueha R, Kashiwadani H, Heinbockel T. Possible Use of Phytochemicals for Recovery from COVID-19-Induced Anosmia and Ageusia. International Journal of Molecular Sciences. 2021; 22(16):8912. https://doi.org/10.3390/ijms22168912
Chicago/Turabian StyleKoyama, Sachiko, Kenji Kondo, Rumi Ueha, Hideki Kashiwadani, and Thomas Heinbockel. 2021. "Possible Use of Phytochemicals for Recovery from COVID-19-Induced Anosmia and Ageusia" International Journal of Molecular Sciences 22, no. 16: 8912. https://doi.org/10.3390/ijms22168912
APA StyleKoyama, S., Kondo, K., Ueha, R., Kashiwadani, H., & Heinbockel, T. (2021). Possible Use of Phytochemicals for Recovery from COVID-19-Induced Anosmia and Ageusia. International Journal of Molecular Sciences, 22(16), 8912. https://doi.org/10.3390/ijms22168912









