The Simple Method of Preparation of Highly Carboxylated Bacterial Cellulose with Ni- and Mg-Ferrite-Based Versatile Magnetic Carrier for Enzyme Immobilization
Abstract
1. Introduction
2. Results and Discussion
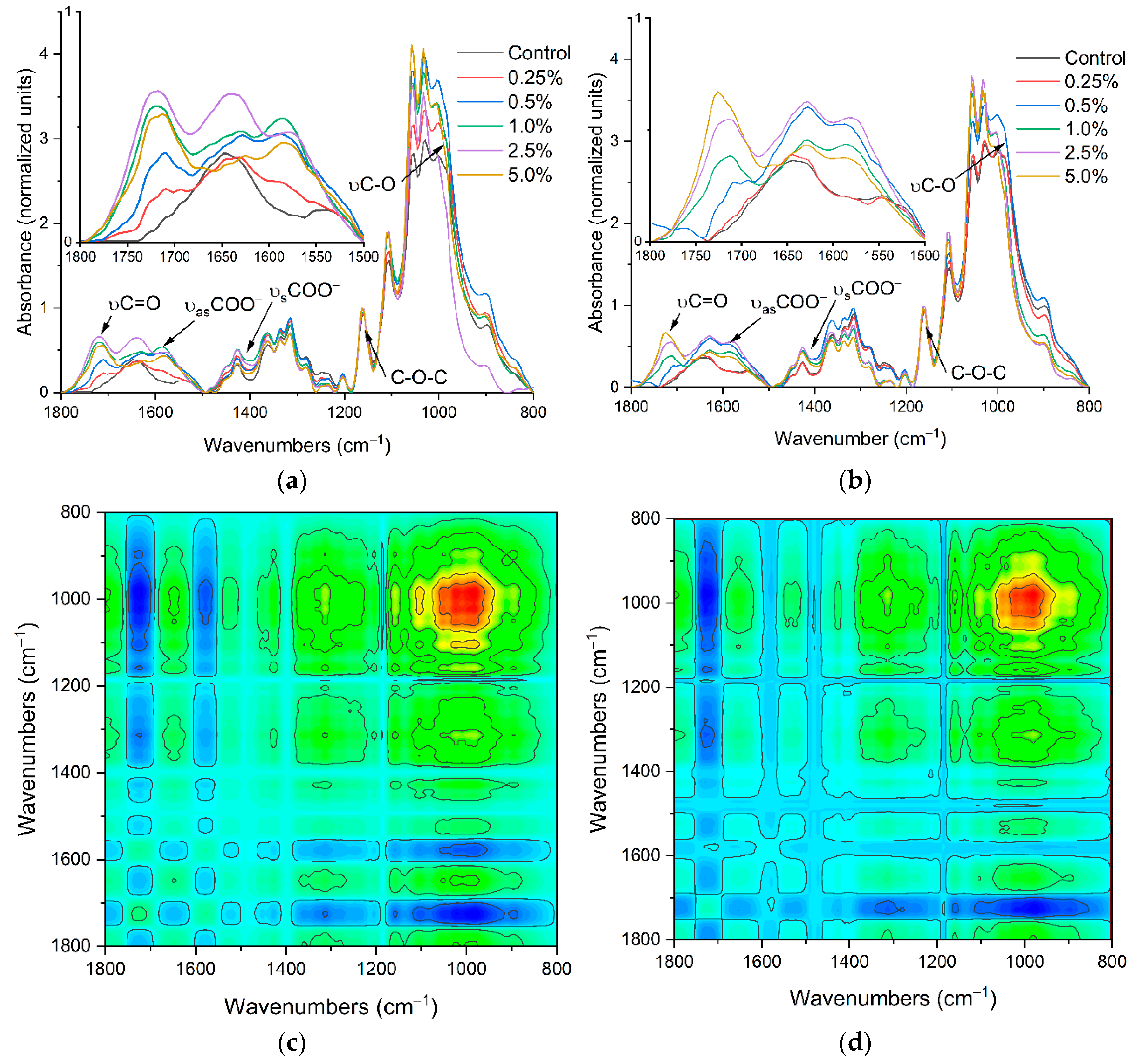
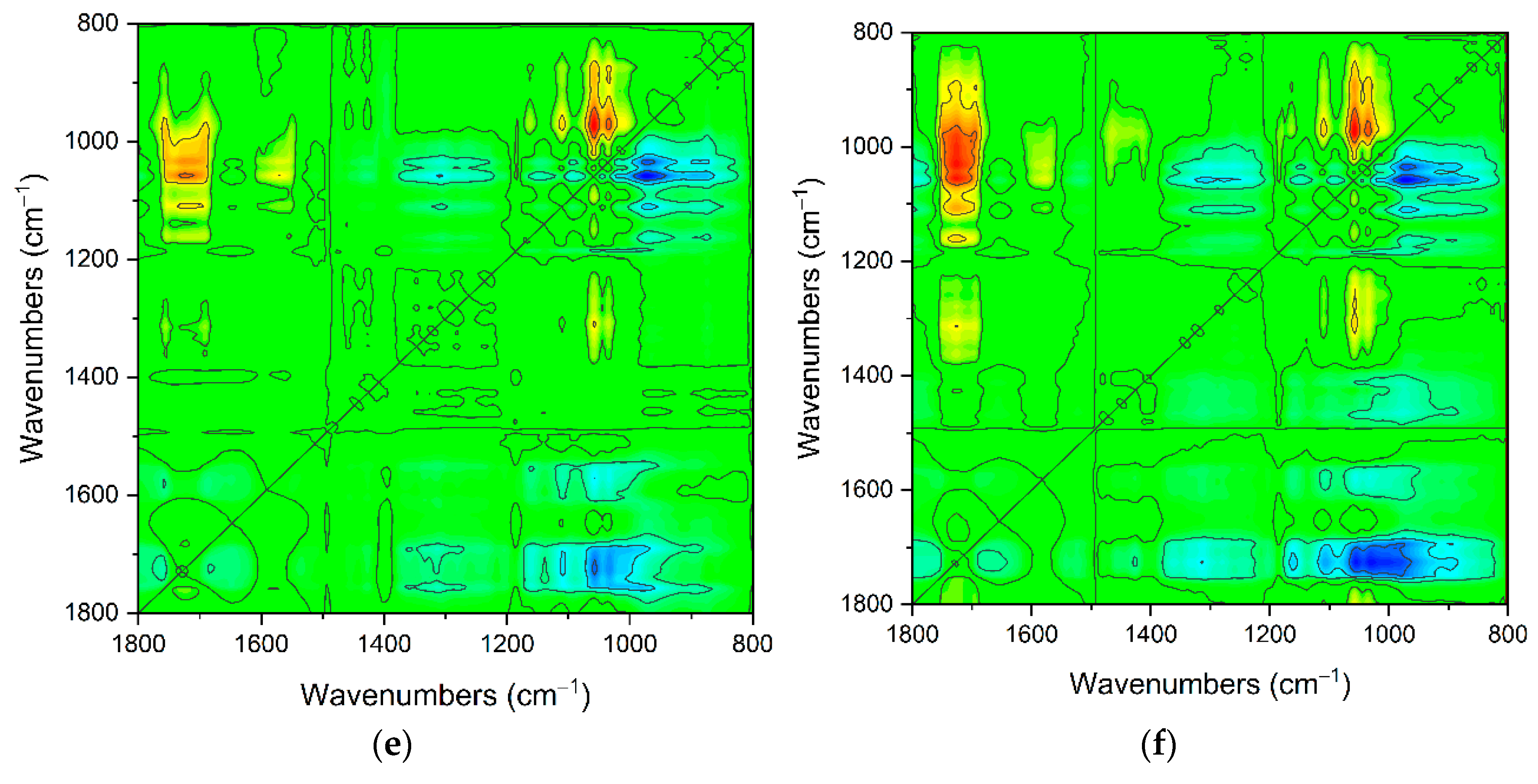
2.1. The Carriers Properties
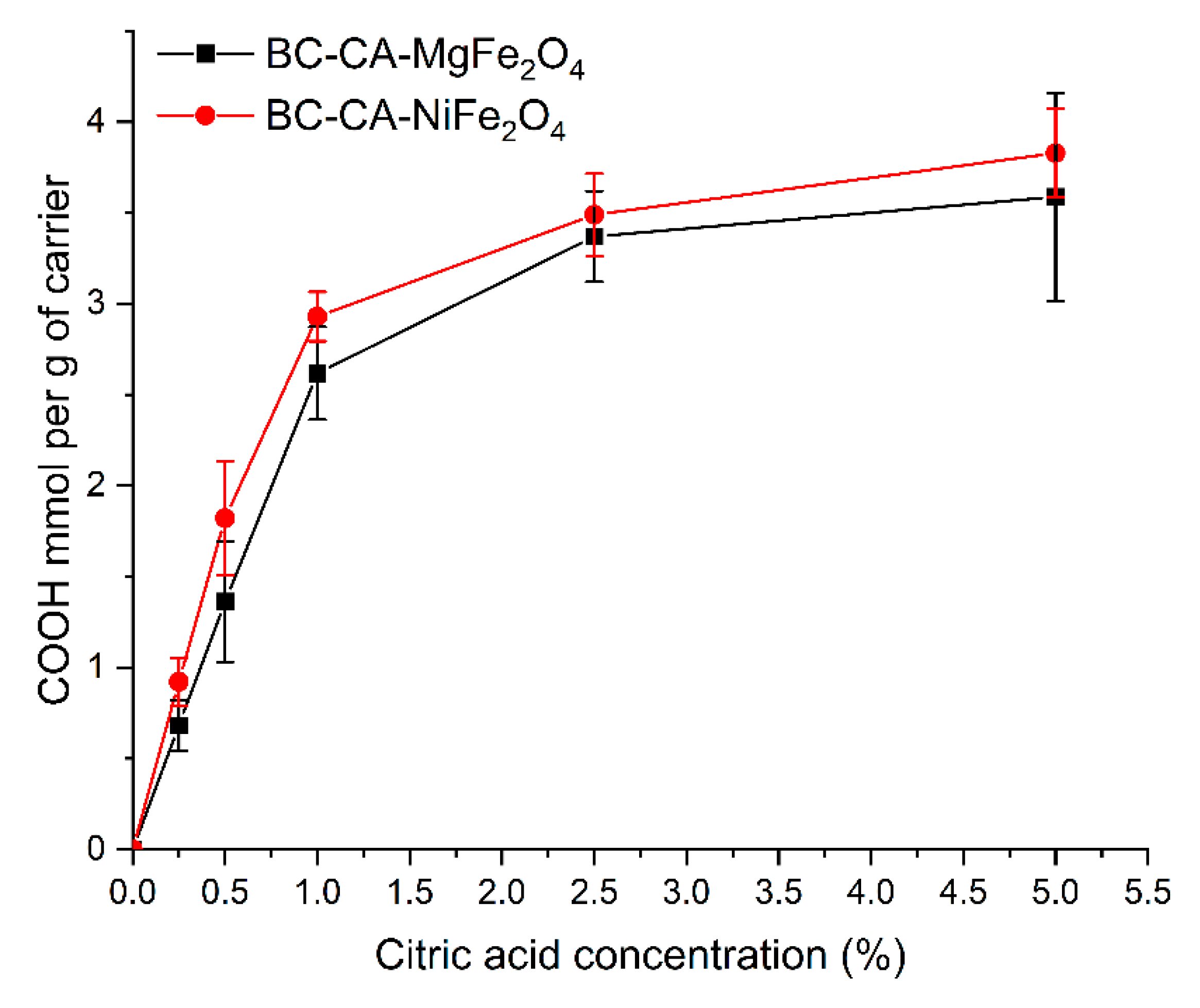
2.2. The Density of the Carboxyl Groups on the Carrier Surface
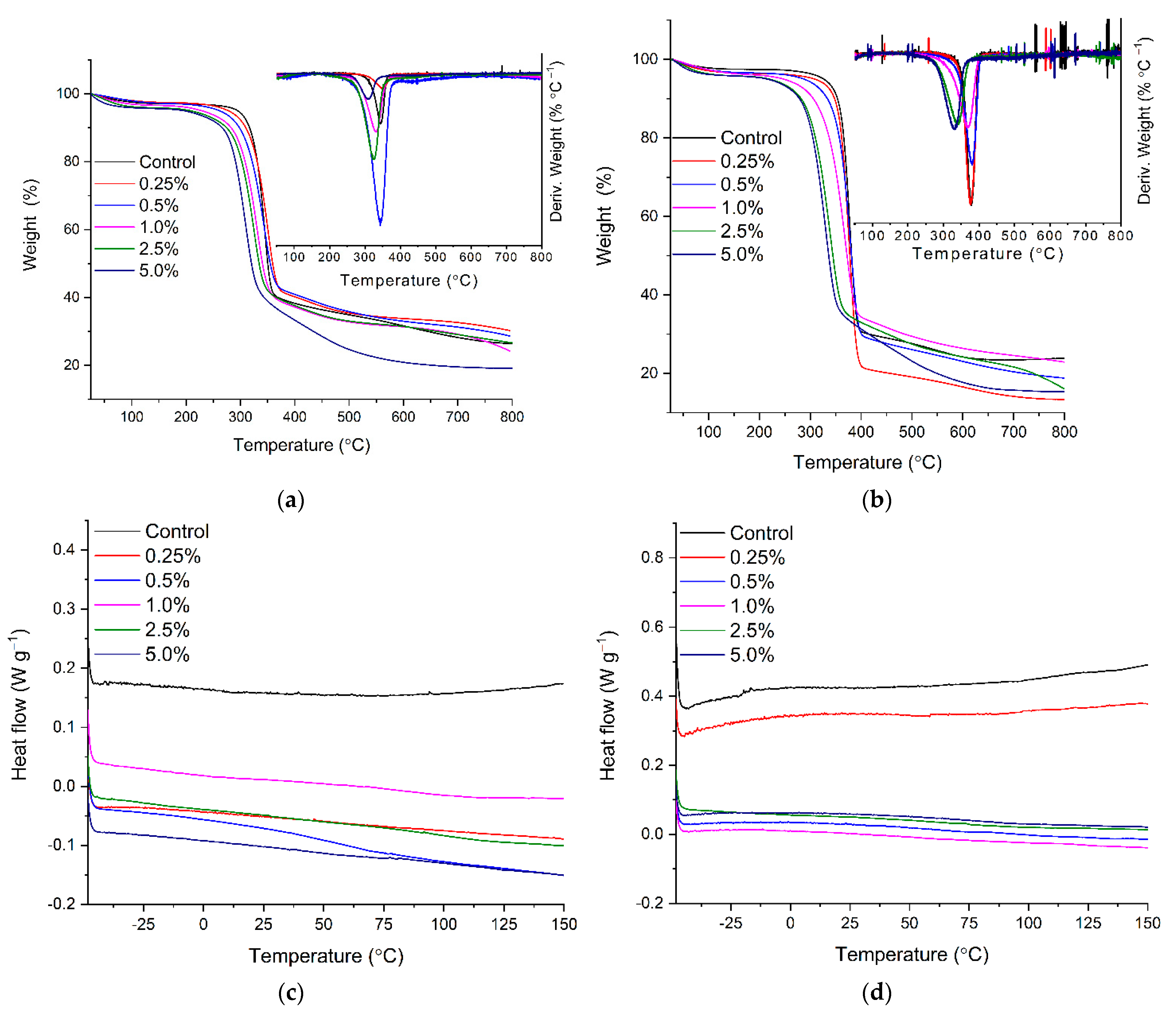
2.3. Thermal Stability of BC-CA-MgFe2O4 and BC-CA-NiFe2O4 Carriers by TGA and DSC
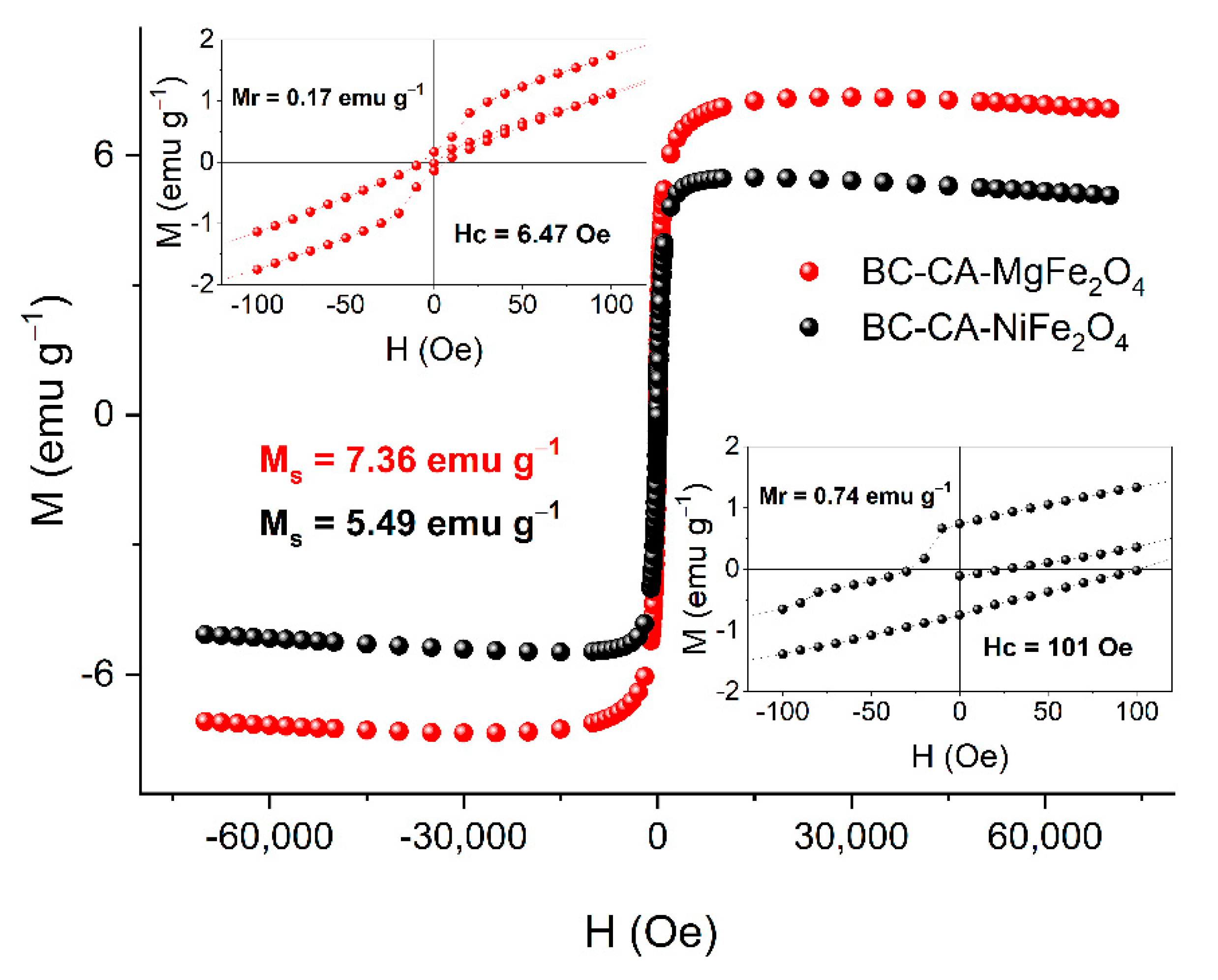
2.4. Magnetic Properties of BC-CA-Ni/Mg Magnetic Carriers
2.5. Characteristics of the BC-CA Magnetic Composites Morphology
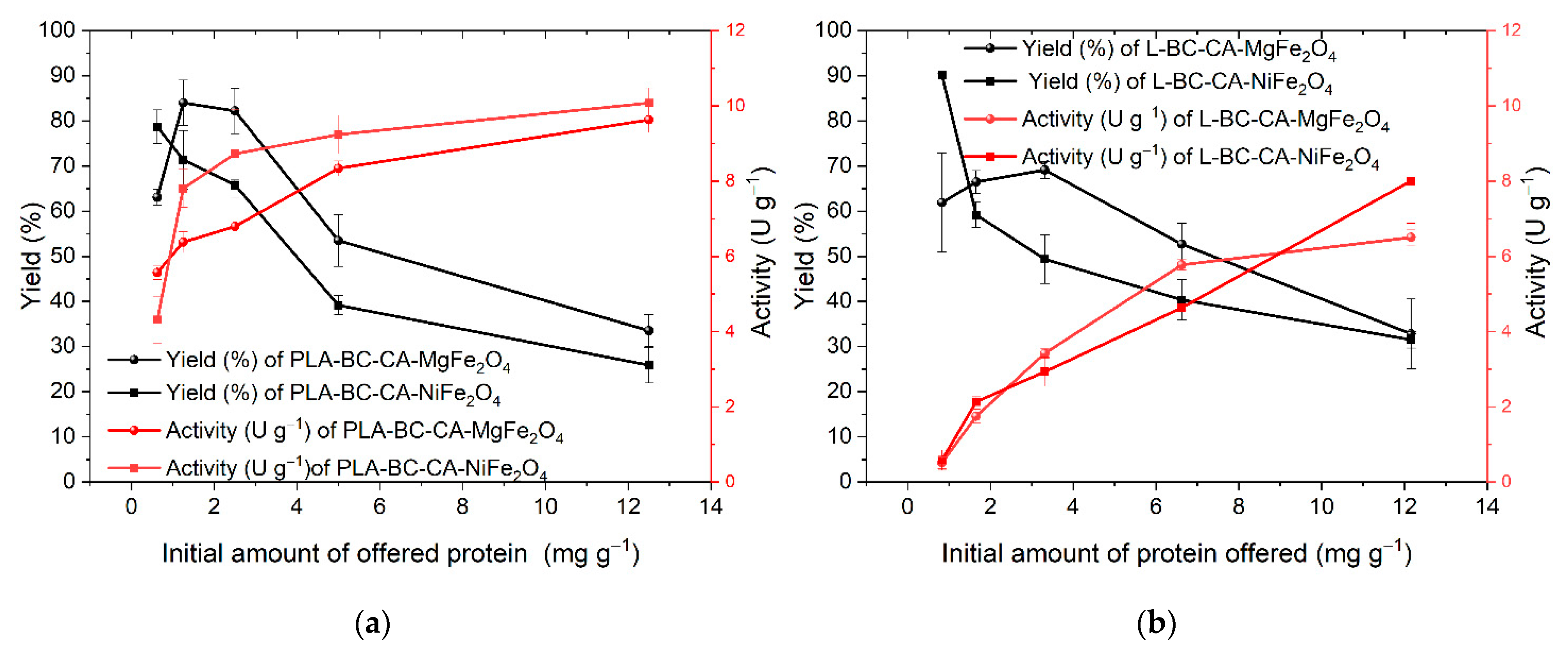
2.6. Efficiency of Enzymes Immobilization
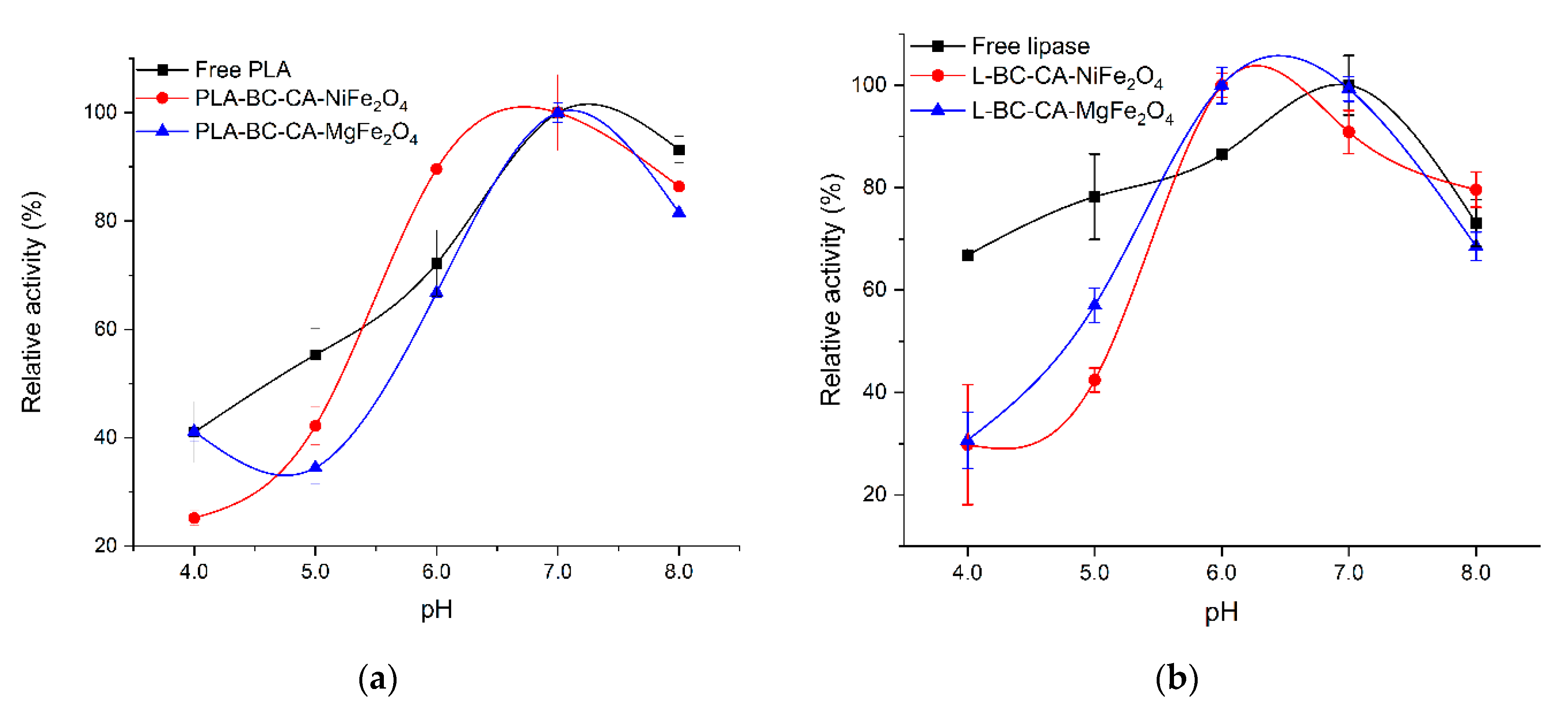
2.7. Effect of pH on the Activity of Immobilized Enzymes
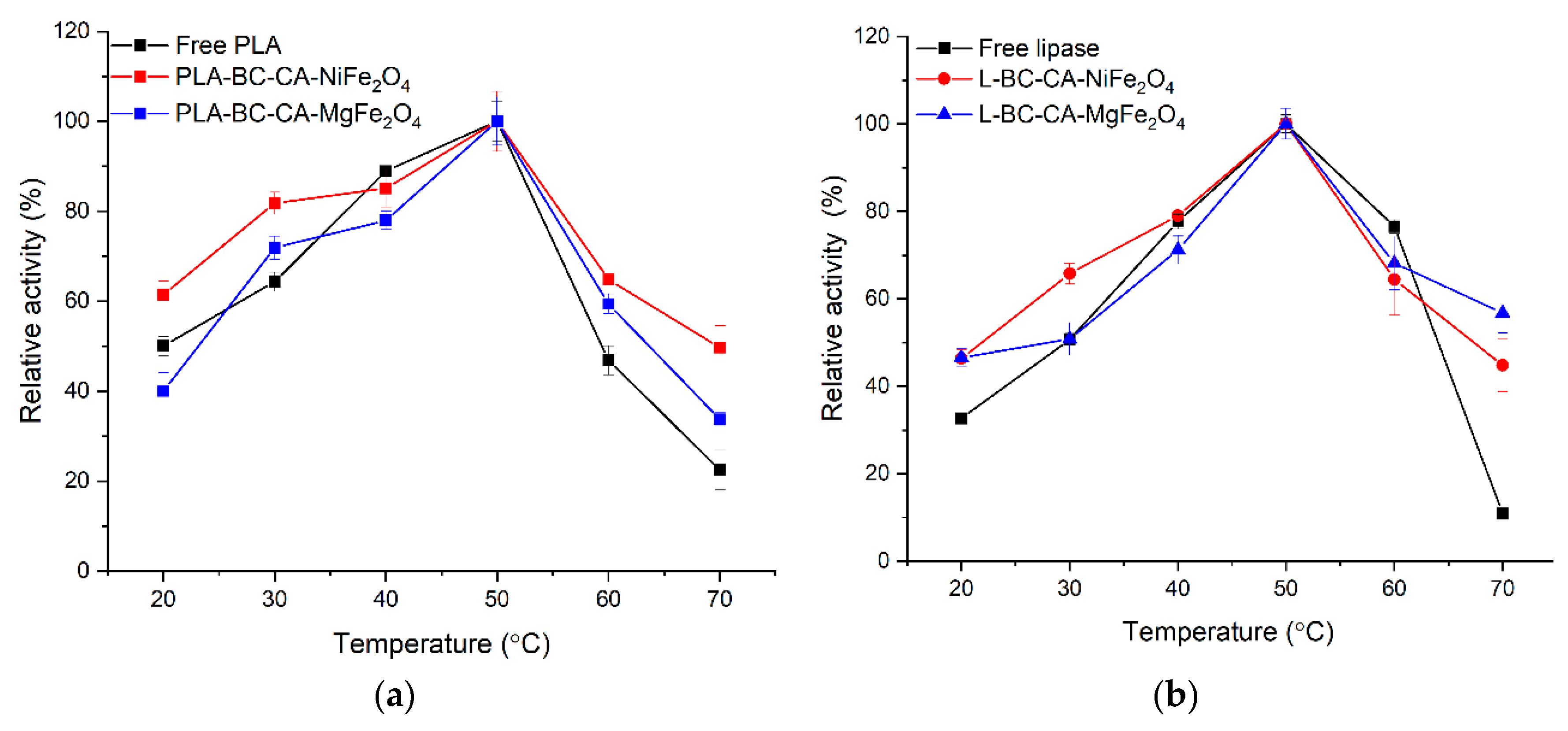
2.8. Effect of Temperature on Activity and Stability Immobilized Enzyme
2.9. Influence of Immobilization Process on Catalytic Constants KM and kcat
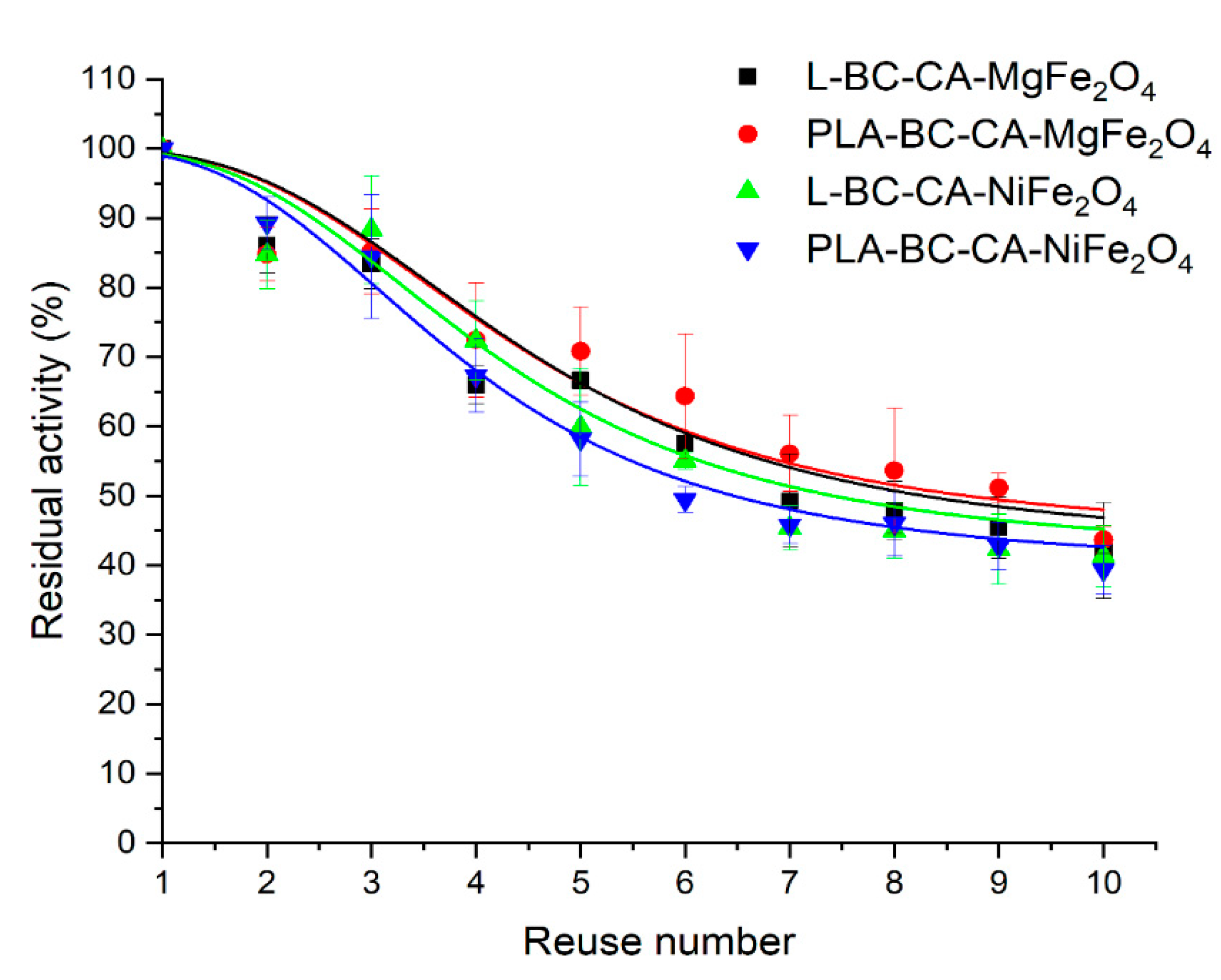
2.10. Reusability
3. Materials and Methods
3.1. Chemicals and Reagents
3.2. Bacterial Cellulose Preparation
3.3. The NiFe2O4 and MgFe2O4 Ferrites Particles Preparation
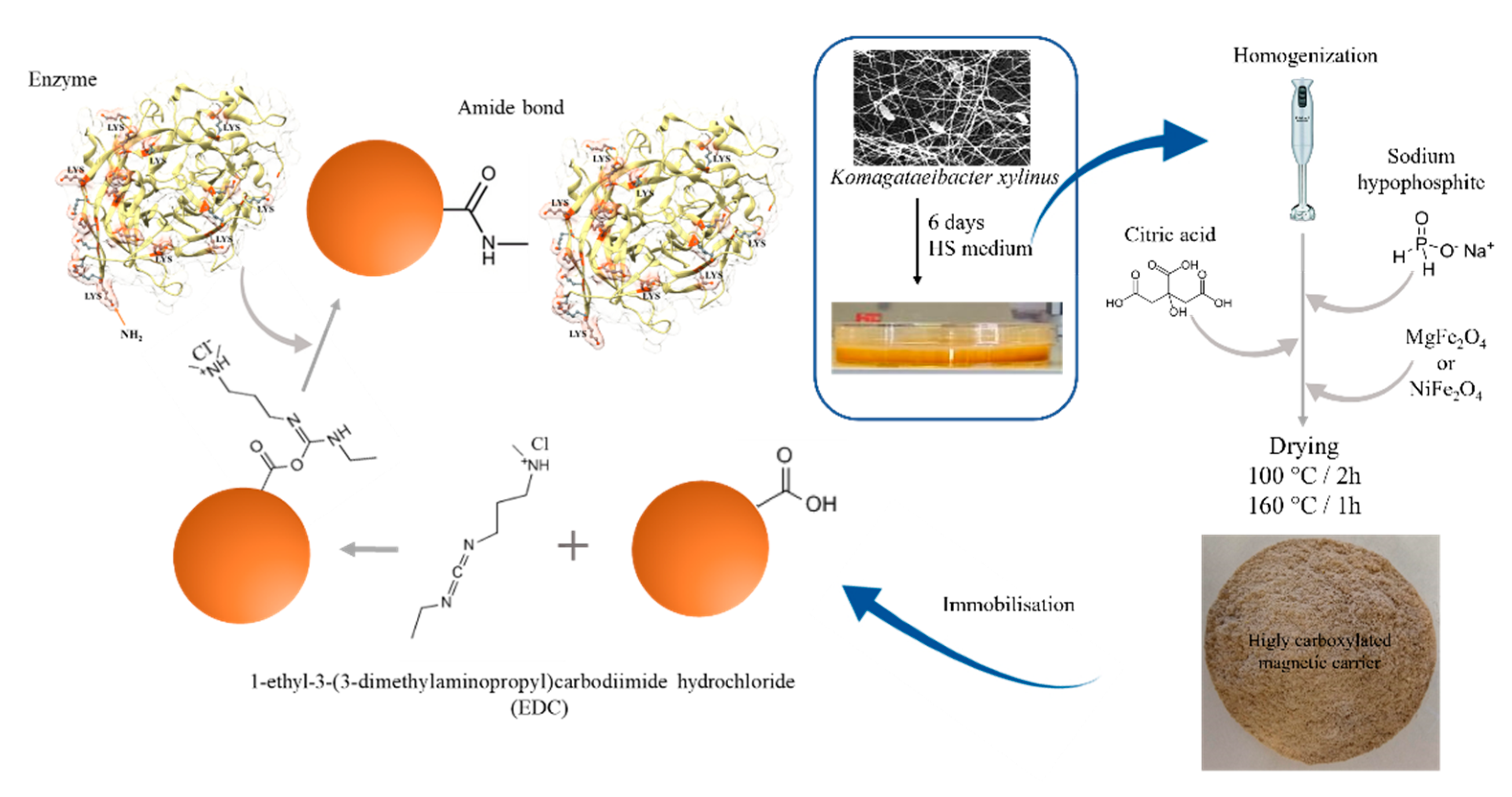
3.4. The Bacterial Cellulose Citric Acid-Modified Magnetic Composites Preparation
3.5. Carriers Properties Determination
3.5.1. The Carboxyl Group Content Assay at Carrier Surface
3.5.2. Scanning Electron Microscopy (SEM)
3.5.3. Attenuated Total Reflectance Fourier Transform Infrared Spectral Studies (ATR-FTIR)
3.5.4. Differential Scanning Calorimeter Analysis (DSC)
3.5.5. Thermogravimetric Analysis (TGA)
3.5.6. Static (Dc) Magnetic Susceptibility Measurements of BC-CA-Ni/Mg Ferrite Composites
3.6. Enzymes Immobilization
3.6.1. Lipase and Phospholipase a Assay of Hydrolytic Activity
3.6.2. Enzymes Immobilization
3.6.3. Protein Concentration Determination
3.6.4. Effect of Temperature and pH on Free and Immobilized Enzymes
3.6.5. Determination of Kinetic Parameters of Immobilized Enzymes
3.6.6. Reusability of Immobilized Enzymes
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Esa, F.; Tasirin, S.M.; Rahman, N.A. Overview of Bacterial Cellulose Production and Application. Agric. Agric. Sci. Procedia 2014, 2, 113–119. [Google Scholar] [CrossRef]
- Huang, Y.; Zhu, C.; Yang, J.; Nie, Y.; Chen, C.; Sun, D. Recent advances in bacterial cellulose. Cellulose 2014, 21, 1–30. [Google Scholar] [CrossRef]
- Akduman, B.; Uygun, M.; Coban, E.P.; Uygun, D.A.; Bıyık, H.; Akgöl, S. Reversible immobilization of urease by using bacterial cellulose nanofibers. Appl. Biochem. Biotechnol. 2013, 171, 2285–2294. [Google Scholar] [CrossRef]
- Mohite, B.V.; Patil, S.V. A novel biomaterial: Bacterial cellulose and its new era applications. Biotechnol. Appl. Biochem. 2014, 61, 101–110. [Google Scholar] [CrossRef]
- Liu, Y.; Chen, J.Y. Enzyme immobilization on cellulose matrixes. J. Bioact. Compat. Polym. 2016, 31, 553–567. [Google Scholar] [CrossRef]
- Estevinho, B.N.; Samaniego, N.; Talens-Perales, D.; Fabra, M.J.; López-Rubio, A.; Polaina, J.; Marín-Navarro, J. Development of enzymatically-active bacterial cellulose membranes through stable immobilization of an engineered β-galactosidase. Int. J. Biol. Macromol. 2018, 115, 476–482. [Google Scholar] [CrossRef]
- Bayazidi, P.; Almasi, H.; Asl, A.K. Immobilization of lysozyme on bacterial cellulose nanofibers: Characteristics, antimicrobial activity and morphological properties. Int. J. Biol. Macromol. 2018, 107, 2544–2551. [Google Scholar] [CrossRef] [PubMed]
- Chen, L.; Zou, M.; Hong, F.F. Evaluation of Fungal Laccase Immobilized on Natural Nanostructured Bacterial Cellulose. Front. Microbiol. 2015, 6, 1245. [Google Scholar] [CrossRef] [PubMed]
- Sampaio, L.M.P.; Padrão, J.; Faria, J.; Silva, J.P.; Silva, C.J.; Dourado, F.; Zille, A. Laccase immobilization on bacterial nanocellulose membranes: Antimicrobial, kinetic and stability properties. Carbohydr. Polym. 2016, 145, 1–12. [Google Scholar] [CrossRef]
- Wu, S.-C.; Wu, S.-M.; Su, F.-M. Novel process for immobilizing an enzyme on a bacterial cellulose membrane through repeated absorption. J. Chem. Technol. Biotechnol. 2017, 92, 109–114. [Google Scholar] [CrossRef]
- Drozd, R.; Rakoczy, R.; Wasak, A.; Junka, A.; Fijałkowski, K. The application of magnetically modified bacterial cellulose for immobilization of laccase. Int. J. Biol. Macromol. 2018, 108, 462–470. [Google Scholar] [CrossRef] [PubMed]
- Jesionowski, T.; Zdarta, J.; Krajewska, B. Enzyme immobilization by adsorption: A review. Adsorption 2014, 20, 801–821. [Google Scholar] [CrossRef]
- Pesaran, M.; Amoabediny, G.; Yazdian, F. Effect of Cultivation Time and Medium Condition in Production of Bacterial Cellulose Nanofiber for Urease Immobilization. Int. J. Polym. Sci. 2015, 2015, 1–8. [Google Scholar] [CrossRef]
- Ul-Islam, M.; Khan, S.; Ullah, M.W.; Park, J.K. Bacterial cellulose composites: Synthetic strategies and multiple applications in bio-medical and electro-conductive fields. Biotechnol. J. 2015, 10, 1847–1861. [Google Scholar] [CrossRef]
- Yuan, H.; Chen, L.; Hong, F.F.; Zhu, M. Evaluation of nanocellulose carriers produced by four different bacterial strains for laccase immobilization. Carbohydr. Polym. 2018, 196, 457–464. [Google Scholar] [CrossRef]
- Shah, N.; Ul-Islam, M.; Khattak, W.A.; Park, J.K. Overview of bacterial cellulose composites: A multipurpose advanced material. Carbohydr. Polym. 2013, 98, 1585–1598. [Google Scholar] [CrossRef]
- Dahman, Y. Nanostructured biomaterials and biocomposites from bacterial cellulose nanofibers. J. Nanosci. Nanotechnol. 2009, 9, 5105–5122. [Google Scholar] [CrossRef]
- Reiniati, I.; Hrymak, A.N.; Margaritis, A. Recent developments in the production and applications of bacterial cellulose fibers and nanocrystals. Crit. Rev. Biotechnol. 2017, 37, 510–524. [Google Scholar] [CrossRef]
- Kim, J.H.; Park, S.; Kim, H.; Kim, H.J.; Yang, Y.-H.; Kim, Y.H.; Jung, S.-K.; Kan, E.; Lee, S.H. Alginate/bacterial cellulose nanocomposite beads prepared using Gluconacetobacter xylinus and their application in lipase immobilization. Carbohydr. Polym. 2017, 157, 137–145. [Google Scholar] [CrossRef] [PubMed]
- Cacicedo, M.L.; Islan, G.A.; Drachemberg, M.F.; Alvarez, V.A.; Bartel, L.C.; Bolzán, A.D.; Castro, G.R. Hybrid bacterial cellulose–pectin films for delivery of bioactive molecules. New J. Chem. 2018, 42, 7457–7467. [Google Scholar] [CrossRef]
- Kim, H.J.; Jin, J.N.; Kan, E.; Kim, K.J.; Lee, S.H. Bacterial cellulose-chitosan composite hydrogel beads for enzyme immobilization. Biotechnol. Bioprocess Eng. 2017, 22, 89–94. [Google Scholar] [CrossRef]
- Dayal, M.S.; Catchmark, J.M. Mechanical and structural property analysis of bacterial cellulose composites. Carbohydr. Polym. 2016, 144, 447–453. [Google Scholar] [CrossRef] [PubMed]
- Datta, S.; Christena, L.R.; Rajaram, Y.R.S. Enzyme immobilization: An overview on techniques and support materials. 3 Biotech 2013, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Zdarta, J.; Meyer, A.; Jesionowski, T.; Pinelo, M. A General Overview of Support Materials for Enzyme Immobilization: Characteristics, Properties, Practical Utility. Catalysts 2018, 8, 92. [Google Scholar] [CrossRef]
- Zhao, X.S.; Bao, X.Y.; Guo, W.; Lee, F.Y. Immobilizing catalysts on porous materials. Mater. Today 2006, 9, 32–39. [Google Scholar] [CrossRef]
- Neira, H.D.; Herr, A.E. Kinetic Analysis of Enzymes Immobilized in Porous Film Arrays. Anal. Chem. 2017, 89, 10311–10320. [Google Scholar] [CrossRef]
- Jackson, E.; Correa, S.; Betancor, L. Cellulose-Based Nanosupports for Enzyme Immobilization. In Cellulose-Based Superabsorbent Hydrogels; Mondal, M.I.H., Ed.; Springer: Cham, Switzerland, 2019; pp. 1235–1253. ISBN 9783319778297. [Google Scholar]
- Wu, S.-C.; Lia, Y.-K. Application of bacterial cellulose pellets in enzyme immobilization. J. Mol. Catal. B Enzym. 2008, 54, 103–108. [Google Scholar] [CrossRef]
- Drozd, R.; Szymańska, M.; Rakoczy, R.; Junka, A.; Szymczyk, P.; Fijałkowski, K. Functionalized Magnetic Bacterial Cellulose Beads as Carrier for Lecitase® Ultra Immobilization. Appl. Biochem. Biotechnol. 2019, 187, 176–193. [Google Scholar] [CrossRef]
- Gennari, A.; Führ, A.J.; Volpato, G.; Volken de Souza, C.F. Magnetic cellulose: Versatile support for enzyme immobilization—A review. Carbohydr. Polym. 2020, 246, 116646. [Google Scholar] [CrossRef]
- Wang, D.; Yu, H.; Fan, X.; Gu, J.; Ye, S.; Yao, J.; Ni, Q. High Aspect Ratio Carboxylated Cellulose Nanofibers Cross-linked to Robust Aerogels for Superabsorption-Flocculants: Paving Way from Nanoscale to Macroscale. ACS Appl. Mater. Interfaces 2018, 10, 20755–20766. [Google Scholar] [CrossRef]
- Cui, X.; Honda, T.; Asoh, T.-A.; Uyama, H. Cellulose modified by citric acid reinforced polypropylene resin as fillers. Carbohydr. Polym. 2020, 230, 115662. [Google Scholar] [CrossRef] [PubMed]
- Beeran, P.T.Y.; Bobnar, V.; Gorgieva, S.; Grohens, Y.; Finšgar, M.; Thomas, S.; Kokol, V. Mechanically strong, flexible and thermally stable graphene oxide/nanocellulosic films with enhanced dielectric properties. RSC Adv. 2016, 6, 49138–49149. [Google Scholar] [CrossRef]
- Zhang, X.; Chen, L.; Yuan, L.; Liu, R.; Li, D.; Liu, X.; Ge, G. Conformation-Dependent Coordination of Carboxylic Acids with Fe3O4 Nanoparticles Studied by ATR-FTIR Spectral Deconvolution. Langmuir 2019, 35, 5770–5778. [Google Scholar] [CrossRef] [PubMed]
- Ul-Islam, M.; Shah, N.; Ha, J.H.; Park, J.K. Effect of chitosan penetration on physico-chemical and mechanical properties of bacterial cellulose. Korean J. Chem. Eng. 2011, 28, 1736–1743. [Google Scholar] [CrossRef]
- Szymanska-Chargot, M.; Zdunek, A. Use of FT-IR Spectra and PCA to the Bulk Characterization of Cell Wall Residues of Fruits and Vegetables Along a Fraction Process. Food Biophys. 2013, 8, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Fan, M.; Dai, D.; Huang, B. Fourier Transform Infrared Spectroscopy for Natural Fibres. In Fourier Transform—Materials Analysis; Salih, S., Ed.; InTech: Rijeka, Croatia, 2012; ISBN 9789535105947. [Google Scholar]
- Liu, Y. Recent Progress in Fourier Transform Infrared (FTIR) Spectroscopy Study of Compositional, Structural and Physical Attributes of Developmental Cotton Fibers. Materials 2013, 6, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Maréchal, Y.; Chanzy, H. The hydrogen bond network in I β cellulose as observed by infrared spectrometry. J. Mol. Struct. 2000, 523, 183–196. [Google Scholar] [CrossRef]
- Cai, Z.; Ji, B.; Yan, K.; Zhu, Q. Investigation on Reaction Sequence and Group Site of Citric Acid with Cellulose Characterized by FTIR in Combination with Two-Dimensional Correlation Spectroscopy. Polymers 2019, 11, 2071. [Google Scholar] [CrossRef]
- Spinella, S.; Maiorana, A.; Qian, Q.; Dawson, N.J.; Hepworth, V.; McCallum, S.A.; Ganesh, M.; Singer, K.D.; Gross, R.A. Concurrent Cellulose Hydrolysis and Esterification to Prepare a Surface-Modified Cellulose Nanocrystal Decorated with Carboxylic Acid Moieties. ACS Sustain. Chem. Eng. 2016, 4, 1538–1550. [Google Scholar] [CrossRef]
- Drozd, R.; Rakoczy, R.; Konopacki, M.; Frąckowiak, A.; Fijałkowski, K. Evaluation of usefulness of 2DCorr technique in assessing physicochemical properties of bacterial cellulose. Carbohydr. Polym. 2017, 161, 208–218. [Google Scholar] [CrossRef]
- Zhu, J.; Zhang, J.; Lai, Q.; Jiang, B.; Gong, R. Covalent Immobilization of Lipase onto Citric Acid-Esterified Loofah Sponge. BioResources 2013, 8, 3289–3298. [Google Scholar] [CrossRef]
- Ye, P.; Wan, R.-B.; Wang, X.-P. Quantitative enzyme immobilization: Control of the carboxyl group density on support surface. J. Mol. Catal. B Enzym. 2009, 61, 296–302. [Google Scholar] [CrossRef]
- Hernandez, K.; Fernandez-Lafuente, R. Control of protein immobilization: Coupling immobilization and site-directed mutagenesis to improve biocatalyst or biosensor performance. Enzym. Microb. Technol. 2011, 48, 107–122. [Google Scholar] [CrossRef]
- Treesuppharat, W.; Rojanapanthu, P.; Siangsanoh, C.; Manuspiya, H.; Ummartyotin, S. Synthesis and characterization of bacterial cellulose and gelatin-based hydrogel composites for drug-delivery systems. Biotechnol. Rep. 2017, 15, 84–91. [Google Scholar] [CrossRef]
- Pa’e, N.; Salehudin, M.H.; Hassan, N.D.; Marsin, A.M.; Muhamad, I.I. Thermal Behavior of Bacterial Cellulose Based Hydrogels with Other Composites and Related Instrumental Analysis. In Cellulose-Based Superabsorbent Hydrogels; Mondal, M.I.H., Ed.; Springer International Publishing: Cham, Switzerland, 2020; pp. 1–25. ISBN 9783319765730. [Google Scholar]
- Lee, K.-Y.; Quero, F.; Blaker, J.J.; Hill, C.A.S.; Eichhorn, S.J.; Bismarck, A. Surface only modification of bacterial cellulose nanofibres with organic acids. Cellulose 2011, 18, 595–605. [Google Scholar] [CrossRef]
- Roca, M.; Liu, H.; Messer, B.; Warshel, A. On the relationship between thermal stability and catalytic power of enzymes. Biochemistry 2007, 46, 15076–15088. [Google Scholar] [CrossRef]
- Rigoldi, F.; Donini, S.; Redaelli, A.; Parisini, E.; Gautieri, A. Review: Engineering of thermostable enzymes for industrial applications. APL Bioeng. 2018, 2, 11501. [Google Scholar] [CrossRef]
- Zhang, H.; Liu, T.; Zhu, Y.; Hong, L.; Li, T.; Wang, X.; Fu, Y. Lipases immobilized on the modified polyporous magnetic cellulose support as an efficient and recyclable catalyst for biodiesel production from Yellow horn seed oil. Renew. Energy 2020, 145, 1246–1254. [Google Scholar] [CrossRef]
- Virgen-Ortíz, J.J.; dos Santos, J.C.; Ortiz, C.; Berenguer-Murcia, Á.; Barbosa, O.; Rodrigues, R.C.; Fernandez-Lafuente, R. Lecitase ultra: A phospholipase with great potential in biocatalysis. Mol. Catal. 2019, 473, 110405. [Google Scholar] [CrossRef]
- Singh, R.K.; Tiwari, M.K.; Singh, R.; Lee, J.-K. From protein engineering to immobilization: Promising strategies for the upgrade of industrial enzymes. Int. J. Mol. Sci. 2013, 14, 1232–1277. [Google Scholar] [CrossRef]
- da Silva, F.B.; Júnior, W.G.d.M.; da Silva, C.V.; Vieira, A.T.; Batista, A.C.F.; de Faria, A.M.; Assunção, R.M.N. Preparation and Characterization of Cellulose Triacetate as Support for Lecitase Ultra Immobilization. Molecules 2017, 22, 1930. [Google Scholar] [CrossRef]
- Huang, X.-J.; Chen, P.-C.; Huang, F.; Ou, Y.; Chen, M.-R.; Xu, Z.-K. Immobilization of Candida rugosa lipase on electrospun cellulose nanofiber membrane. J. Mol. Catal. B Enzym. 2011, 70, 95–100. [Google Scholar] [CrossRef]
- Harris, T.K.; Turner, G.J. Structural basis of perturbed pKa values of catalytic groups in enzyme active sites. IUBMB Life 2002, 53, 85–98. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Mukhopadhyay, M. Immobilization of Candida antarctica lipase onto cellulose acetate-coated Fe2O3 nanoparticles for glycerolysis of olive oil. Korean J. Chem. Eng. 2014, 31, 1225–1232. [Google Scholar] [CrossRef]
- Secundo, F. Conformational changes of enzymes upon immobilisation. Chem. Soc. Rev. 2013, 42, 6250–6261. [Google Scholar] [CrossRef]
- Hoarau, M.; Badieyan, S.; Marsh, E.N.G. Immobilized enzymes: Understanding enzyme—Surface interactions at the molecular level. Org. Biomol. Chem. 2017, 15, 9539–9551. [Google Scholar] [CrossRef] [PubMed]
- Rodrigues, R.C.; Ortiz, C.; Berenguer-Murcia, Á.; Torres, R.; Fernández-Lafuente, R. Modifying enzyme activity and selectivity by immobilization. Chem. Soc. Rev. 2013, 42, 6290–6307. [Google Scholar] [CrossRef] [PubMed]
- Kazenwadel, F.; Wagner, H.; Rapp, B.E.; Franzreb, M. Optimization of enzyme immobilization on magnetic microparticles using 1-ethyl-3-(3-dimethylaminopropyl)carbodiimide (EDC) as a crosslinking agent. Anal. Methods 2015, 7, 10291–10298. [Google Scholar] [CrossRef]
- Khan, F.I.; Lan, D.; Durrani, R.; Huan, W.; Zhao, Z.; Wang, Y. The Lid Domain in Lipases: Structural and Functional Determinant of Enzymatic Properties. Front. Bioeng. Biotechnol. 2017, 5, 16. [Google Scholar] [CrossRef]
- Vanleeuw, E.; Winderickx, S.; Thevissen, K.; Lagrain, B.; Dusselier, M.; Cammue, B.P.A.; Sels, B.F. Substrate-Specificity of Candida rugosa Lipase and Its Industrial Application. ACS Sustain. Chem. Eng. 2019, 7, 15828–15844. [Google Scholar] [CrossRef]
- Cai, Q.; Hu, C.; Yang, N.; Wang, Q.; Wang, J.; Pan, H.; Hu, Y.; Ruan, C. Enhanced activity and stability of industrial lipases immobilized onto spherelike bacterial cellulose. Int. J. Biol. Macromol. 2018, 109, 1174–1181. [Google Scholar] [CrossRef] [PubMed]
- Yi, S.-S.; Noh, J.-M.; Lee, Y.-S. Amino acid modified chitosan beads: Improved polymer supports for immobilization of lipase from Candida rugosa. J. Mol. Catal. B Enzym. 2009, 57, 123–129. [Google Scholar] [CrossRef]
- Rödiger, S.; Ruhland, M.; Schmidt, C.; Schröder, C.; Grossmann, K.; Böhm, A.; Nitschke, J.; Berger, I.; Schimke, I.; Schierack, P. Fluorescence dye adsorption assay to quantify carboxyl groups on the surface of poly(methyl methacrylate) microbeads. Anal. Chem. 2011, 83, 3379–3385. [Google Scholar] [CrossRef] [PubMed]
- Sheldon, R.A.; van Pelt, S. Enzyme immobilisation in biocatalysis: Why, what and how. Chem. Soc. Rev. 2013, 42, 6223–6235. [Google Scholar] [CrossRef] [PubMed]
- Bradford, M.M. A rapid and sensitive method for the quantitation of microgram quantities of protein utilizing the principle of protein-dye binding. Anal. Biochem. 1976, 72, 248–254. [Google Scholar] [CrossRef]

| Initial CA Concentration (%) | BC-CA- NiFe2O4 | BC-CA- MgFe2O4 | ||||
|---|---|---|---|---|---|---|
| Tonset | Tmax | * Mass Loss (%) | Tonset | Tmax | * Mass Loss (%) | |
| (°C) | (°C) | |||||
| 0 | 335.1 | 355.5 | 59.6 | 348.7 | 392.8 | 66.7 |
| 0.25 | 317.9 | 365.1 | 61.1 | 356.2 | 390.7 | 68.4 |
| 0.5 | 305.8 | 360.7 | 60.1 | 351.2 | 393.7 | 70.9 |
| 1.0 | 295.7 | 349.1 | 59.9 | 327.2 | 386.1 | 66.4 |
| 2.5 | 289.0 | 340.5 | 59.8 | 297.0 | 358.6 | 66.5 |
| 5.0 | 278.1 | 327.7 | 60.3 | 294.5 | 349.4 | 66.2 |
| Temperature | ||
|---|---|---|
| 50 °C | 60 °C | |
| Relative Activity (%) | ||
| Free PLA * | 82.0 ± 4.0 | 12.4 ± 0.2 |
| Free Lipase | 45.2 ± 2.1 | 18.9 ± 1.6 |
| PLA-BC-CA-NiFe2O4 | 89.4 ± 3.7 | 56.5 ± 7.6 |
| PLA-BC-CA-MgFe2O4 | 77.4 ± 6.7 | 63.1 ± 2.2 |
| L-BC-CA-NiFe2O4 | 60.4 ± 3.2 | 50.5 ± 12.6 |
| L-BC-CA-MgFe2O4 | 87.4 ± 4.6 | 66.5 ± 2.1 |
| a KM (mmol L−1) | kcat (s−1) | kcat KM−1 (L mmol−1 s−1) | |
|---|---|---|---|
| Free PLA | 0.49 ± 0.10 | 57.6 ± 2.5 × 107 | 119.94 ± 22.34 × 107 |
| Free lipase | 0.16 ± 0.01 | 3.07 ± 0.24 × 107 | 19.56 ± 0.43 × 107 |
| PLA-BC-CA-NiFe2O4 | 0.43 ± 0.09 | 39.78 ± 3.74 | 92.40 ± 3.7 |
| PLA-BC-CA-MgFe2O4 | 1.49 ± 0.08 | 44.68 ± 1.56 | 39.78 ± 9.5 |
| L-BC-CA-NiFe2O4 | 0.19 ± 0.03 | 39.78 ± 3.74 | 147.05 ± 17.36 |
| L-BC-CA-MgFe2O4 | 0.29 ± 0.03 | 28.13 ± 1.21 | 30.07 ± 1.91 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Drozd, R.; Szymańska, M.; Przygrodzka, K.; Hoppe, J.; Leniec, G.; Kowalska, U. The Simple Method of Preparation of Highly Carboxylated Bacterial Cellulose with Ni- and Mg-Ferrite-Based Versatile Magnetic Carrier for Enzyme Immobilization. Int. J. Mol. Sci. 2021, 22, 8563. https://doi.org/10.3390/ijms22168563
Drozd R, Szymańska M, Przygrodzka K, Hoppe J, Leniec G, Kowalska U. The Simple Method of Preparation of Highly Carboxylated Bacterial Cellulose with Ni- and Mg-Ferrite-Based Versatile Magnetic Carrier for Enzyme Immobilization. International Journal of Molecular Sciences. 2021; 22(16):8563. https://doi.org/10.3390/ijms22168563
Chicago/Turabian StyleDrozd, Radosław, Magdalena Szymańska, Katarzyna Przygrodzka, Jakub Hoppe, Grzegorz Leniec, and Urszula Kowalska. 2021. "The Simple Method of Preparation of Highly Carboxylated Bacterial Cellulose with Ni- and Mg-Ferrite-Based Versatile Magnetic Carrier for Enzyme Immobilization" International Journal of Molecular Sciences 22, no. 16: 8563. https://doi.org/10.3390/ijms22168563
APA StyleDrozd, R., Szymańska, M., Przygrodzka, K., Hoppe, J., Leniec, G., & Kowalska, U. (2021). The Simple Method of Preparation of Highly Carboxylated Bacterial Cellulose with Ni- and Mg-Ferrite-Based Versatile Magnetic Carrier for Enzyme Immobilization. International Journal of Molecular Sciences, 22(16), 8563. https://doi.org/10.3390/ijms22168563







