Generation of Electromagnetic Field by Microtubules
Abstract
:1. Introduction
2. Results
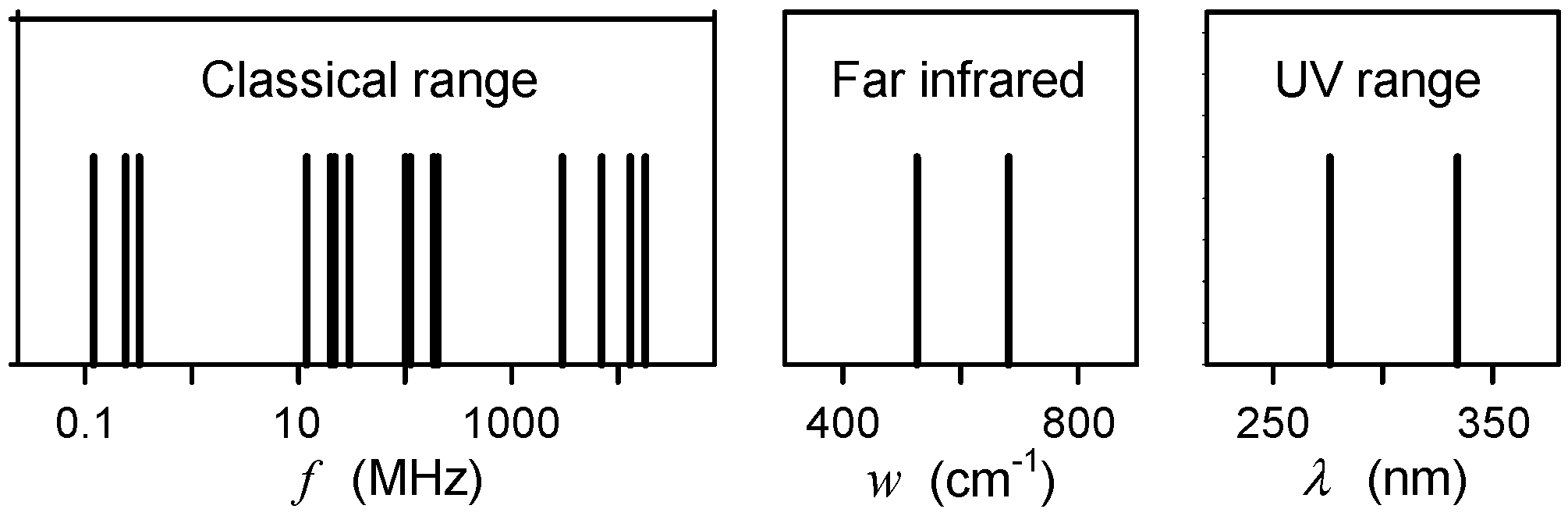
2.1. Available Experimental Data
2.2. Microtubule Structure
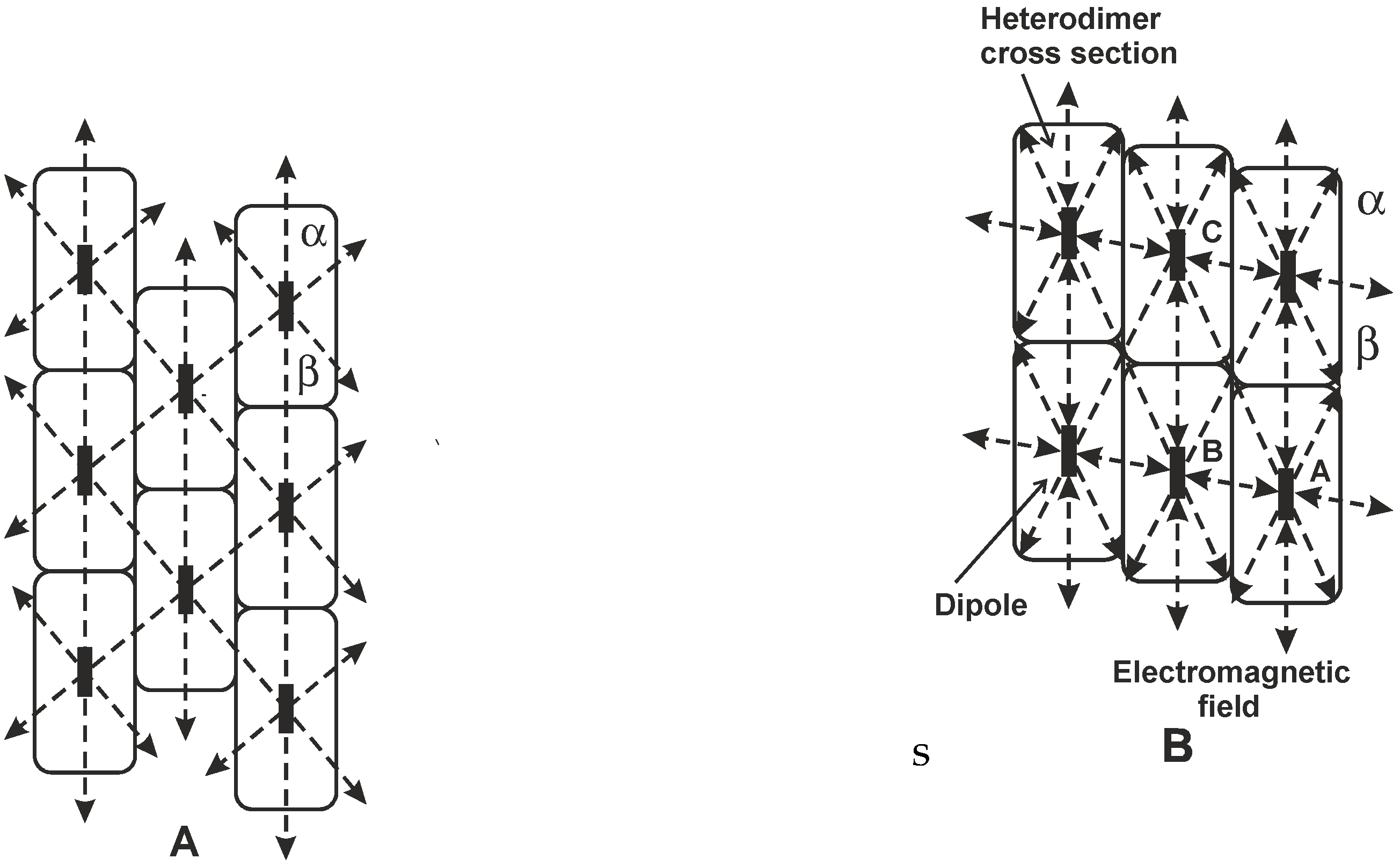
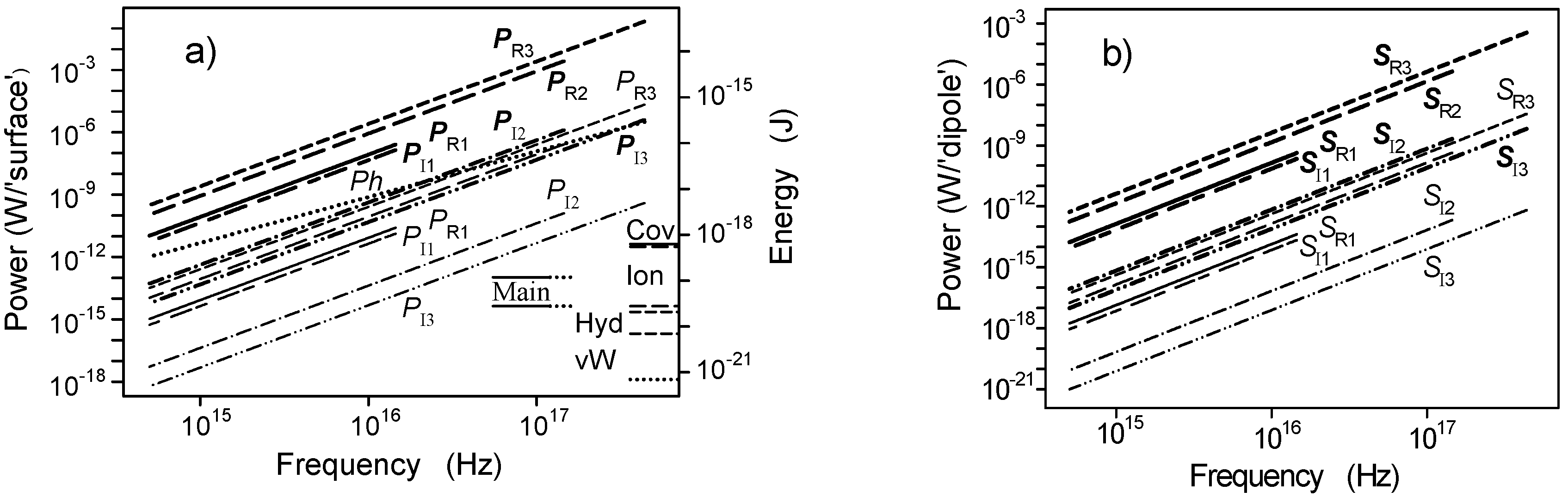
2.3. Microtubule Oscillations
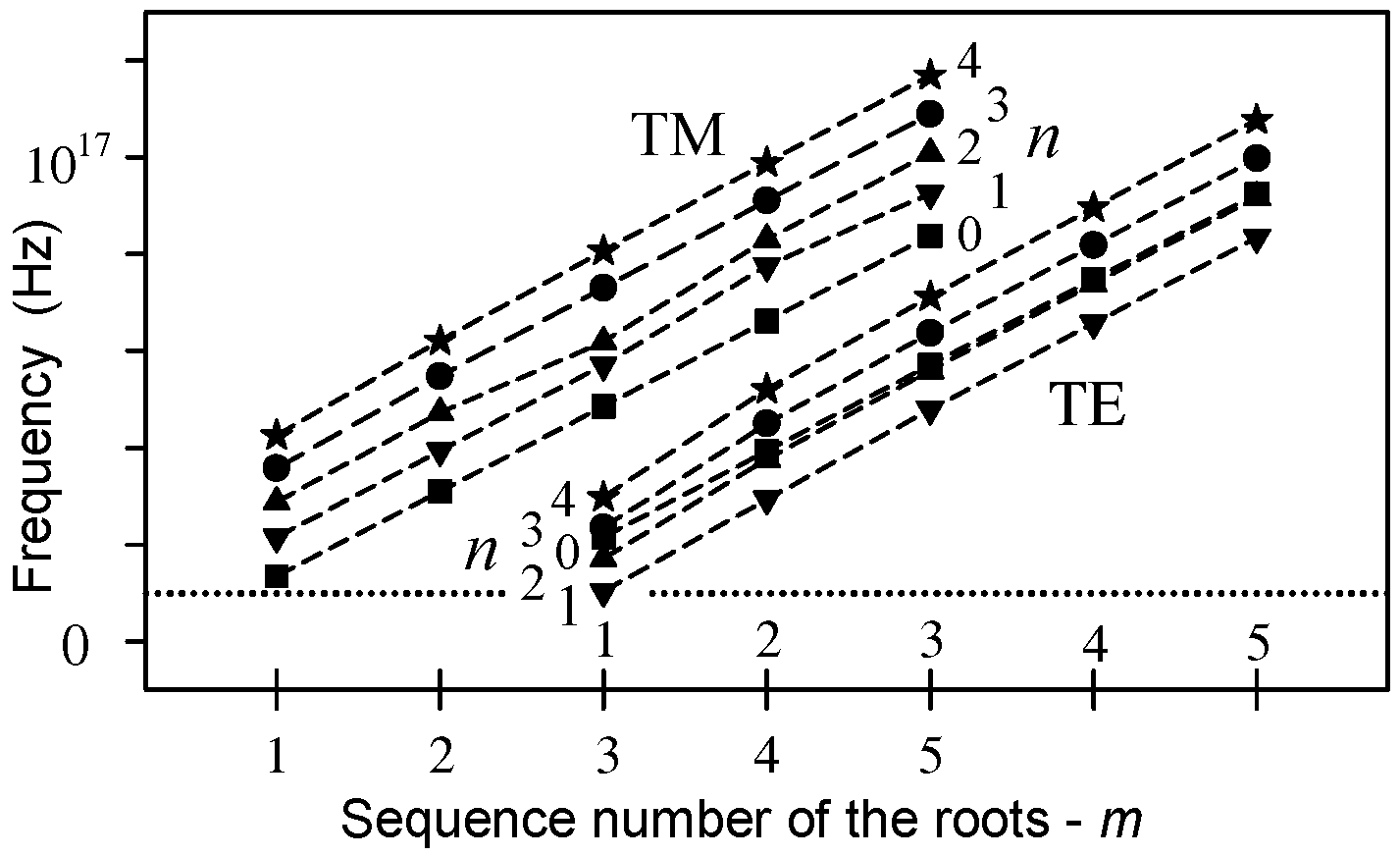
2.4. Oscillation in the Microtubule Cavity
2.5. Water around Microtubules
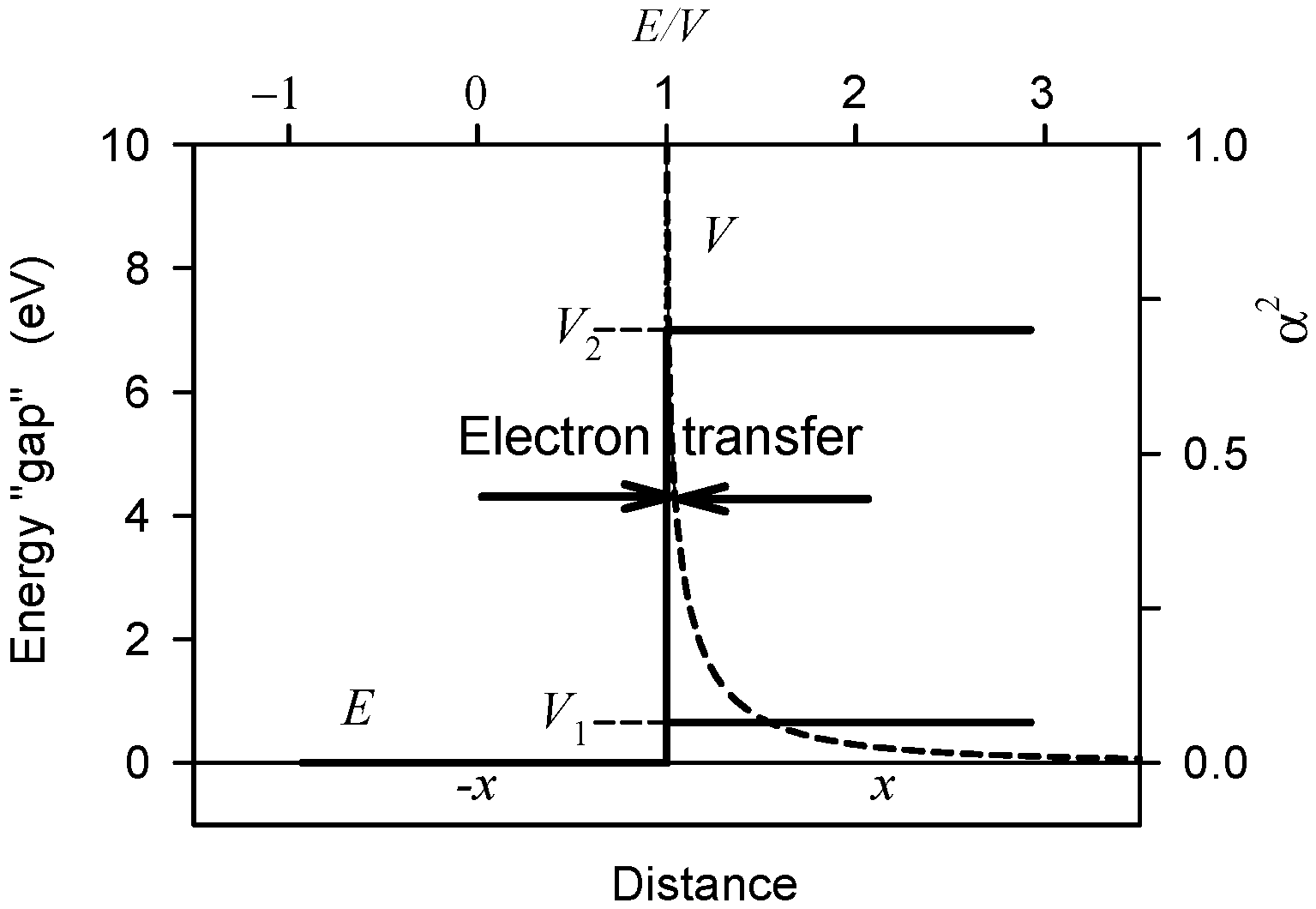
2.6. Interaction with Electrons on Molecular Orbitals
3. Discussion
4. Conclusions
Author Contributions
Funding
Conflicts of Interest
Appendix A
References
- Fröhlich, H. Bose condensation of strongly excited longitudinal electric modes. Phys. Lett. A 1968, 26, 402–403. [Google Scholar] [CrossRef]
- Fröhlich, H. Long-range coherence and energy storage in biological systems. Int. J. Quantum Chem. 1968, 2, 641–649. [Google Scholar] [CrossRef]
- Fröhlich, H. Quantum mechanical concepts in biology. In Theoretical Physics and Biology, Proceedings of The First International Conference on Theoretical Physics and Biology, Versailles, France, 26–30 June 1967; Marois, M., Ed.; North Holland Publishing Co.: North Holland, Amsterdam, 1969; pp. 13–22. [Google Scholar]
- Fröhlich, H. The biological effects of microwaves and related questions. In Advances in Electronics and Electron Physics; Marton, L., Marton, C., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: New York, NY, USA; London, UK; Toronto, ON, Canada; Sydney, Australia; San Francisco, CA, USA, 1980; Volume 53, pp. 85–152. [Google Scholar] [CrossRef]
- Pokorný, J.; Wu, T.-M. Biophysical Aspects of Coherence and Biological Order; Springer: Berlin/Heidelberg, Germany; New York, NY, USA; Academia: Prague, Czech Republic, 1998. [Google Scholar]
- Preparata, G. QED Coherence in Matter; Word Scientific: Singapore, 1995. [Google Scholar]
- Pokorný, J.; Pokorný, J.; Kobilková, J. Postulates on electromagnetic activity in biological systems and cancer. Integr. Biol. 2013, 5, 1439–1446. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Foletti, A.; Brizhik, L. Nonlinearity, coherence and complexity: Biophysical aspects related to health and disease. Electromagn. Biol. Med. 2017, 36, 315–324. [Google Scholar] [CrossRef]
- Kasas, S.; Ruggeri, F.S.; Benadiba, C.; Maillard, C.; Stupar, P.; Tournu, H.; Dietler, G.; Longo, G. Detecting nanoscale vibrations as signature of life. Proc. Natl. Acad. Sci. USA 2015, 112, 297–298. [Google Scholar] [CrossRef] [Green Version]
- Pokorný, J.; Hašek, J.; Jelínek, F.; Šaroch, J.; Palán, B. Electromagnetic activity of yeast cells in the M phase. Electro Magn. 2001, 20, 371–396. [Google Scholar] [CrossRef]
- Del Giudice, E.; Tedeschi, A. Water and autocatalysis in living matter. Electromagn. Biol. Med. 2009, 28, 46–52. [Google Scholar] [CrossRef]
- Pohl, H.A.; Braden, T.; Robinson, S.; Piclardi, J.; Pohl, D.G. Life cycle alterations of the micro-dielectrophoretic effects of cells. J. Biol. Phys. 1981, 9, 133–154. [Google Scholar] [CrossRef]
- Albrecht-Buehler, G. Surface extensions of 3T3 cells towards distant infrared light sources. J. Cell. Biol. 1991, 114, 493–502. [Google Scholar] [CrossRef]
- Albrecht-Buehler, G. Rudimentary form of cellular “vision”. Proc. Natl. Acad. Sci. USA 1992, 89, 8288–8293. [Google Scholar] [CrossRef] [Green Version]
- Albrecht-Buehler, G. A long-range attraction between aggregating 3T3 cells mediated by near-infrared light scattering. Proc. Natl. Acad. Sci. USA 2005, 102, 5050–5055. [Google Scholar] [CrossRef] [Green Version]
- Hölzel, R. Electric activity of non-excitable biological cells at radio frequencies. Electro Magn. 2001, 20, 1–13. [Google Scholar] [CrossRef]
- Jelínek, F.; Cifra, M.; Pokorný, J.; Vaniš, J.; Šimša, J.; Hašek, J.; Frýdlová, I. Measurement of electrical oscillations and mechanical vibrations of yeast cells membrane around 1kHz. Electromagn. Biol. Med. 2009, 28, 223–232. [Google Scholar] [CrossRef] [PubMed]
- Craddock, T.J.A.; Tuszynski, J.A.; Hameroff, S. Cytoskeletal signaling: Is memory encoded in microtubule lattices by CaMKII phosphorylation? PLoS Comput. Biol. 2012, 8, e1002421. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tuszynski, J.A.; Friesen, D.; Freedman, H.; Sbitnef, V.I.; Kim, H.; Santelices, I.; Kalra, A.P.; Patel, S.D.; Shankar, K.; Chua, L.O. Microtubules as sub-cellular memristors. Sci. Rep. 2020, 10, 2108. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Amos, L.A.; Klug, A. Arrangement of subunits in flagellar microtubules. J. Cell Sci. 1974, 14, 523–549. [Google Scholar] [CrossRef] [PubMed]
- Amos, L.A. Structure of microtubules. In Microtubules; Roberts, K., Hyams, J.S., Eds.; Elsevier: Amsterdam, The Netherlands; Academic Press: New York, NY, USA; London, UK; Toronto, ON, Canada; Sydney, Australia; San Francisco, CA, USA, 1979; pp. 1–64. [Google Scholar]
- Satarić, M.; Tuszyński, J.A.; Žakula, R.B. Kinklike excitation as an energy transfer mechanism in microtubules. Phys. Rev. E 1993, 8, 589–597. [Google Scholar] [CrossRef]
- Tuszyński, J.A.; Hameroff, S.; Satarić, M.; Trpisová, B.; Nip, M.L.A. Ferroelectric behavior in microtubule dipole lattices: Implications for conformation processing, signalling and assembly/disassembly. J. Theor. Biol. 1995, 174, 371–380. [Google Scholar] [CrossRef]
- Sahu, S.; Ghosh, S.; Ghosh, B.; Aswani, K.; Hirata, K.; Fujita, D.; Bandyopadhyay, A. Atomic water channel controlling remarkable properties of a single brain microtubule: Correlating single protein to its supramolecular assembly. Biosens. Bioelectron. 2013, 47, 141–148. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sahu, S.; Ghosh, S.; Hirata, K.; Fujita, D.; Bandyopadhyay, A. Multi-level memory switching properties of a single brain microtubule. Appl. Phys. Lett. 2013, 102, 123701. [Google Scholar] [CrossRef]
- Sahu, S.; Ghosh, S.; Fujita, D.; Bandyopadhyay, A. Live visualizations of single isolated tubulin protein self-assembly via tunnelling current: Effect of electromagnetic pumping during spontaneous growth of microtubule. Sci. Rep. 2014, 4, 7303. [Google Scholar] [CrossRef] [Green Version]
- Cosic, I.; Lazar, K.; Cosic, D. Prediction of tubulin resonant frequencies using the resonant recognition model (RRM). IEEE Trans. Nanobiosci. 2015, 14, 491–496. [Google Scholar] [CrossRef]
- Priel, A.; Ramos, A.J.; Tuszyński, J.A.; Cantiello, H.F. A biopolymer transistor: Electric amplification by microtubules. Biophys. J. 2006, 90, 4639–4643. [Google Scholar] [CrossRef] [Green Version]
- Gurwitsch, A. Die Natur des spezifischen Erregers der Zellteilung. Arch. Mikrosk. Anat. Entw. Mech. 1923, 100, 11–40. [Google Scholar] [CrossRef]
- Volodyaev, I.; Beloussov, L.V. Revisiting the mitogenetic effect of ultra-weak photonemission. Front. Physiol. 2015, 6, 241. [Google Scholar] [CrossRef] [PubMed]
- Craddock, T.J.A.; Kurian, P.; Preto, J.; Sahu, K.; Hameroff, S.R.; Klobukowski, M.; Tuszynski, J.A. Anesthetic alterations of collective terahertz oscillations in tubulin correlate with clinical potency: Implications for anesthetic action and post-operative cognitive dysfunction. Sci. Rep. 2017, 29, 9877. [Google Scholar] [CrossRef] [Green Version]
- Duke, A.R.; Jenkins, M.W.; Lu, H.; McManus, J.M.; Chiel, H.J.; Jansen, E.D. Transient and selective suppression of neural activity with infrared light. Sci. Rep. 2013, 3, 2600. [Google Scholar] [CrossRef] [Green Version]
- Yoo, S.; Hong, S.; Choi, Y.; Park, J.-H.; Nam, Y. Photothermal inhibition of neural activity with near-infrared-sensitive nanotransducers. ACS Nano 2014, 8, 8040–8049. [Google Scholar] [CrossRef] [PubMed]
- Rafati, Y.; Cantu, J.C.; Sedelnikova, A.; Tolstykh, G.P.; Peralta, X.G.; Valdez, C.; Echchgadda, I. Effect of microtubule resonant frequencies on neuronal cells. In Optical Interactions with Tissue and Cells XXXI; SPIE: Bellingham, WA, USA, 2020; Volume 11238. [Google Scholar] [CrossRef]
- Singh, P.; Ghosh, S.; Sahoo, P.; Bandyopadhyay, A. Electrophysiology using coaxial atom probe array; Live imaging reveals hidden circuits of a hippocampal neural network. J. Neurophysiol. 2021. [Google Scholar] [CrossRef] [PubMed]
- Fröhlich, H. Coherent electric vibrations in biological systems and cancer problem. IEEE Trans. MTT 1978, 26, 613–617. [Google Scholar] [CrossRef]
- Pokorný, J.; Jelínek, F.; Trkal, V.; Lamprecht, I.; Hölzel, R. Vibrations in microtubules. J. Biol. Phys. 1997, 23, 171–179. [Google Scholar] [CrossRef]
- Böhm, K.J.; Mavromatos, N.E.; Michette, A.; Stracke, R.; Unger, E. Movement and alignment of microtubules in electric fields and electric-dipole-moment estimates. Electromagn. Biol. Med. 2005, 24, 319–330. [Google Scholar] [CrossRef]
- Schoutens, J.E. Dipole–dipole interactions in microtubules. J. Biol. Phys. 2005, 31, 35–55. [Google Scholar] [CrossRef] [Green Version]
- Sataric, M.V.; Tuszynski, J.A. Nonlinear dynamics of microtubules: Biophysical implications. J. Biol. Phys. 2005, 31, 487–500. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alberts, B.; Bray, D.; Lewis, J.; Raff, M.; Roberts, K.; Watson, J.D. Molecular Biology of the Cell, 3rd ed.; Garland Publishing, Inc.: New York, NY, USA; London, UK, 1994. [Google Scholar]
- Stebbings, H.; Hunt, C. The nature of the clear zone around microtubules. Cell. Tissue Res. 1982, 227, 609–617. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, J.-M.; Chin, W.-C.; Khijniak, E.; Khijniak, E., Jr.; Pollack, G.H. Surfaces and interfacial water: Evidence that hydrophilic surfaces have long-range impact. Adv. Colloid Interface Sci. 2006, 127, 19–27. [Google Scholar] [CrossRef] [PubMed]
- Modica-Napolitano, J.S.; Aprille, J.R. Basis for selective cytotoxicity of Rhodamine 123. Cancer Res. 1987, 47, 4361–4365. [Google Scholar]
- Warburg, O.; Posener, K.; Negelein, E. Über den Stoffwechsell der Carzinomzelle. Biochem. Z. 1924, 152, 309–344. [Google Scholar]
- Pokorný, J.; Pokorný, J.; Borodavka, F. Warburg effect–damping of electromagnetic oscillations. Electromagn. Biol. Med. 2017, 36, 270–278. [Google Scholar] [CrossRef]
- Dicke, R.H.; Wittke, J.P. Introduction to Quantum Mechanics; Addison–Wesley Publishing Co.: Massachusetts, MA, USA; London, UK, 1961. [Google Scholar]
- Šrobár, F. Radiating Fröhlich system as a model of cellular electromagnetism. Electromagn. Biol. Med. 2015, 34, 355–360. [Google Scholar] [CrossRef]
- Šrobár, F. Impact of mitochondrial electric field on modal occupancy in the Fröhlich model of cellular electromagnetism. Electromagn. Biol. Med. 2013, 32, 401–408. [Google Scholar] [CrossRef] [PubMed]
- Derjaguin, B.V. Die Formelastizität der dünnen Wasserschichten. Prog. Surf. Sci. 1933, 84, 657–670. [Google Scholar] [CrossRef]
- Zheng, J.; Pollack, G.H. Long-range forces extending from polymer–gel surfaces. Phys. Rev. E 2003, 68. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Marais, A.; Adams, B.; Ringsmuth, A.K.; Ferretti, M.; Gruber, J.M.; Hendrikx, R.; Schuld, M.; Smith, S.L.; Sinayskiy, I.; Krüger, T.P.J.; et al. The future of quantum biology. J. Royal Soc. Interface 2018, 15. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pokorný, J.; Pokorný, J.; Jandová, A.; Kobilková, J.; Vrba, J.; Vrba, J., Jr. Energy parasites trigger oncogene mutation. Int. J. Rad. Biol. 2016, 92, 577–582. [Google Scholar] [CrossRef]
- Stratton, J.A. Electromagnetic Theory; McGraw–Hill Book Co. Inc.: New York, NY, USA, 1941; pp. 434–437, 492–497. [Google Scholar]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Pokorný, J.; Pokorný, J.; Vrba, J. Generation of Electromagnetic Field by Microtubules. Int. J. Mol. Sci. 2021, 22, 8215. https://doi.org/10.3390/ijms22158215
Pokorný J, Pokorný J, Vrba J. Generation of Electromagnetic Field by Microtubules. International Journal of Molecular Sciences. 2021; 22(15):8215. https://doi.org/10.3390/ijms22158215
Chicago/Turabian StylePokorný, Jan, Jiří Pokorný, and Jan Vrba. 2021. "Generation of Electromagnetic Field by Microtubules" International Journal of Molecular Sciences 22, no. 15: 8215. https://doi.org/10.3390/ijms22158215
APA StylePokorný, J., Pokorný, J., & Vrba, J. (2021). Generation of Electromagnetic Field by Microtubules. International Journal of Molecular Sciences, 22(15), 8215. https://doi.org/10.3390/ijms22158215






