Enzymatic Decontamination of G-Type, V-Type and Novichok Nerve Agents
Abstract
:1. Introduction
2. Results and Discussion
2.1. Kinetic Parameters of Engineered Enzymes Towards Ethyl-Paraoxon and OPNA Surrogates
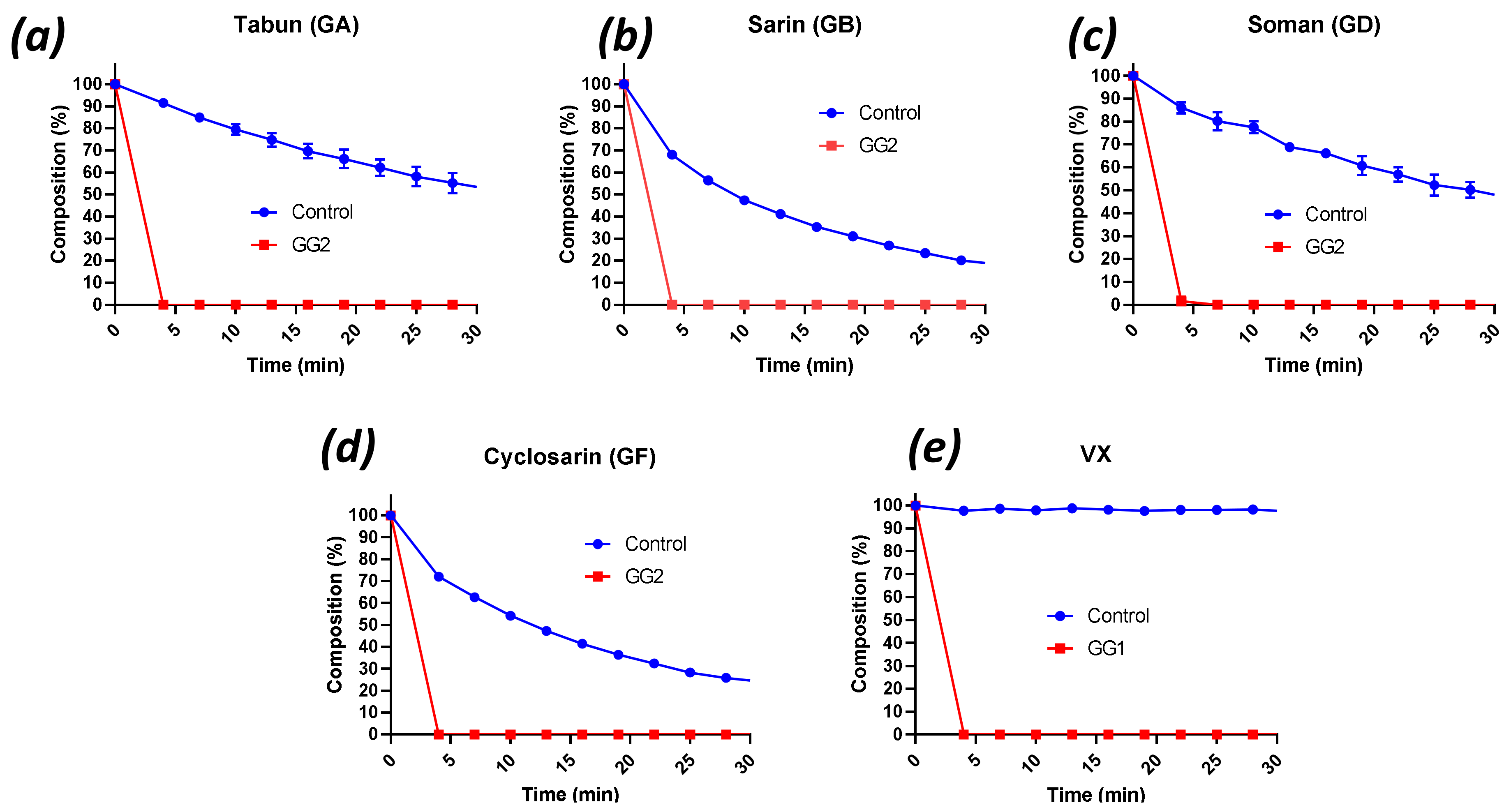
2.2. Decontamination of Live G- and V-Agents with Engineered Enzymes
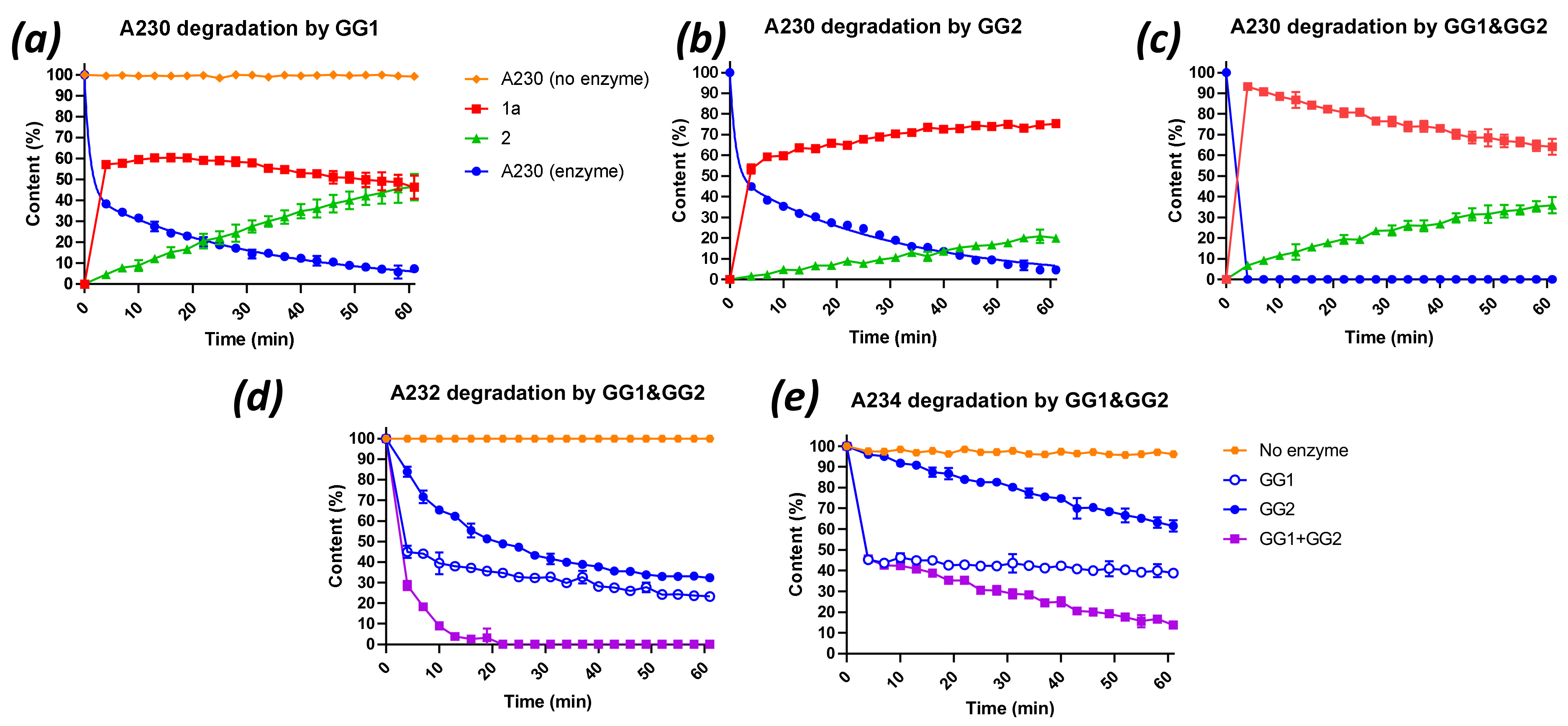
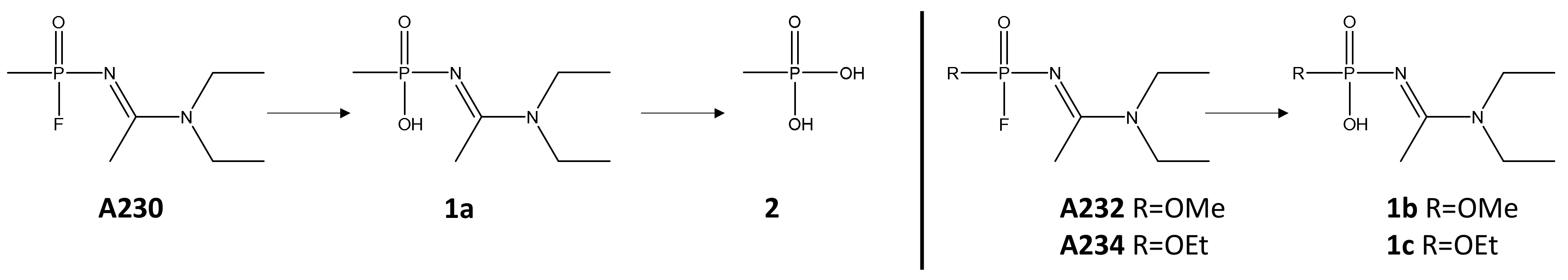
2.3. Degradation of Novichok Agents with Engineered Enzymes
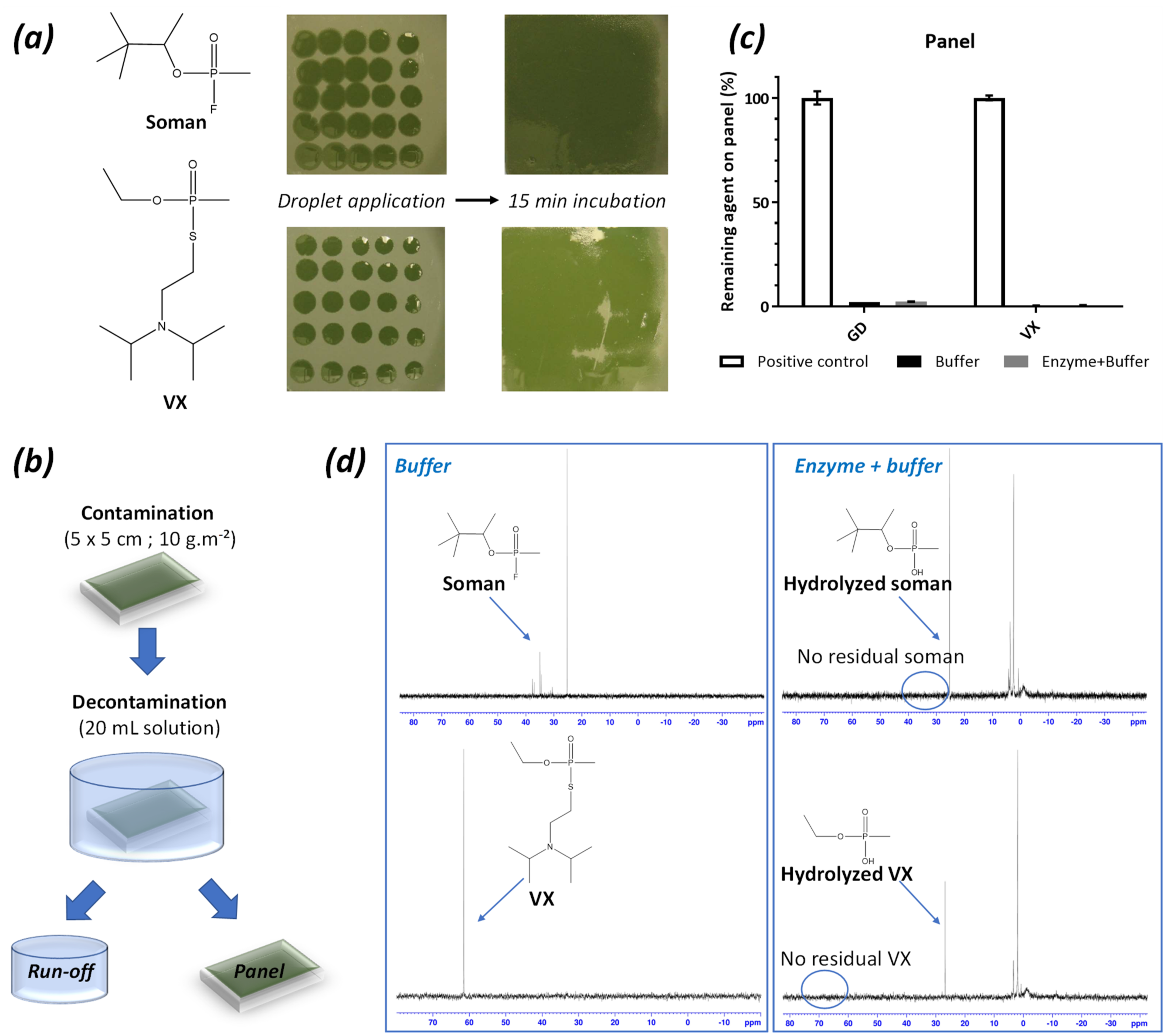
2.4. Decontamination of VX or GD-Contaminated Surfaces with the Enzyme-Solutions
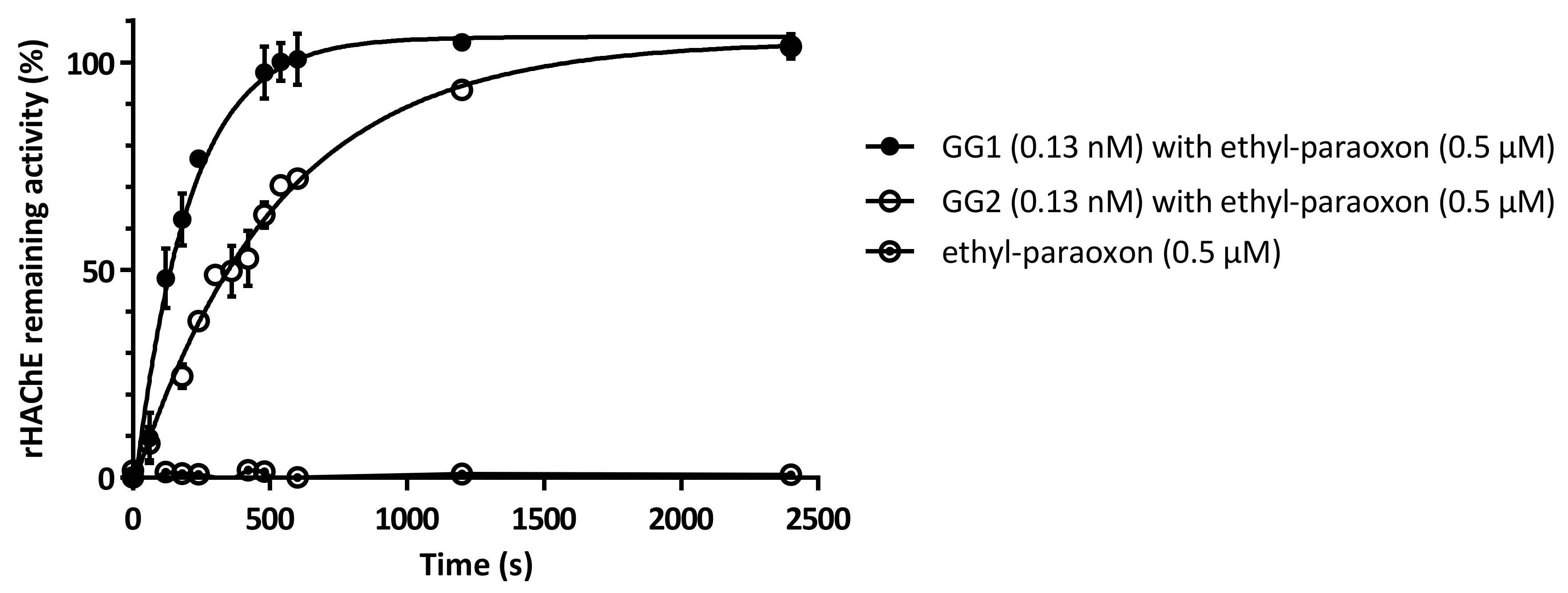
2.5. Enzymes Protect rHAChE from Inhibition by Ethyl-Paraoxon
3. Materials and Methods
3.1. Chemicals
3.2. Production and Purification of PTEs
3.3. Kinetic Parameters
3.4. Degradation Experiments with OPNAs
3.5. Liquid Chromatography Tandem Mass Spectrometry (LC-MS) Analyses
3.6. Production and Partial Purification of rHAChE
3.7. Surface Decontamination Experiments
3.8. rHAChE Inhibition Assay
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Young, R.A.; Watson, A. Chapter 8. Organophosphate nerve agents. In Handbook of Toxicology of Chemical Warfare Agents, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Boston, MA, USA, 2020; pp. 97–126. ISBN 978-0-12-819090-6. [Google Scholar]
- Gupta, R.C. Chapter Two. Neurotoxicity of organophosphate nerve agents. In Advances in Neurotoxicology; Aschner, M., Costa, L.G., Eds.; Neurotoxicity of Pesticides; Academic Press: Boston, MA, USA, 2020; Volume 4, pp. 79–112. [Google Scholar]
- Eddleston, M.; Buckley, N.A.; Eyer, P.; Dawson, A.H. Management of Acute Organophosphorus Pesticide Poisoning. Lancet 2008, 371, 597–607. [Google Scholar] [CrossRef] [Green Version]
- De Silva, H.J.; Samarawickrema, N.A.; Wickremasinghe, A.R. Toxicity Due to Organophosphorus Compounds: What about Chronic Exposure? Trans. R Soc. Trop. Med. Hyg. 2006, 100, 803–806. [Google Scholar] [CrossRef] [Green Version]
- Delfino, R.T.; Ribeiro, T.S.; Figueroa-Villar, J.D. Organophosphorus Compounds as Chemical Warfare Agents: A Review. J. Braz. Chem. Soc. 2009, 20, 407–428. [Google Scholar] [CrossRef]
- Jacquet, P.; Daudé, D.; Bzdrenga, J.; Masson, P.; Elias, M.; Chabrière, E. Current and Emerging Strategies for Organophosphate Decontamination: Special Focus on Hyperstable Enzymes. Environ. Sci. Pollut. Res. Int. 2016, 23, 8200–8218. [Google Scholar] [CrossRef]
- Chai, P.R.; Hayes, B.D.; Erickson, T.B.; Boyer, E.W. Novichok Agents: A Historical, Current, and Toxicological Perspective. Toxicol. Commun. 2018, 2, 45–48. [Google Scholar] [CrossRef] [Green Version]
- Dolgin, E. Syrian Gas Attack Reinforces Need for Better Anti-Sarin Drugs. Nat. Med. 2013, 19, 1194–1195. [Google Scholar] [CrossRef]
- John, H.; van der Schans, M.J.; Koller, M.; Spruit, H.E.T.; Worek, F.; Thiermann, H.; Noort, D. Fatal Sarin Poisoning in Syria 2013: Forensic Verification within an International Laboratory Network. Forensic Toxicol 2018, 36, 61–71. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, T.; Tu, A.T. Murders with VX: Aum Shinrikyo in Japan and the Assassination of Kim Jong-Nam in Malaysia. Forensic Toxicol. 2018, 36, 542–544. [Google Scholar] [CrossRef]
- Vale, J.A.; OBE, T.C.M.; CBE, R.L.M. Novichok: A Murderous Nerve Agent Attack in the UK. Clin. Toxicol. 2018, 56, 1093–1097. [Google Scholar] [CrossRef]
- Steindl, D.; Boehmerle, W.; Körner, R.; Praeger, D.; Haug, M.; Nee, J.; Schreiber, A.; Scheibe, F.; Demin, K.; Jacoby, P.; et al. Novichok Nerve Agent Poisoning. Lancet 2021, 397, 249–252. [Google Scholar] [CrossRef]
- Eddleston, M.; Chowdhury, F.R. Organophosphorus Poisoning: The Wet Opioid Toxidrome. Lancet 2021, 397, 175–177. [Google Scholar] [CrossRef]
- Nepovimova, E.; Kuca, K. Chemical Warfare Agent NOVICHOK - Mini-Review of Available Data. Food Chem. Toxicol. 2018, 121, 343–350. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, L. After Salisbury Nerve Agents Revisited. Mol. Inform. 2019, 38, 1800106. [Google Scholar] [CrossRef] [Green Version]
- Kloske, M.; Witkiewicz, Z. Novichoks - The A Group of Organophosphorus Chemical Warfare Agents. Chemosphere 2019, 221, 672–682. [Google Scholar] [CrossRef] [PubMed]
- Franca, T.C.C.; Kitagawa, D.A.S.; Cavalcante, S.F.d.A.; da Silva, J.A.V.; Nepovimova, E.; Kuca, K. Novichoks: The Dangerous Fourth Generation of Chemical Weapons. Int. J. Mol. Sci. 2019, 20, 1222. [Google Scholar] [CrossRef] [Green Version]
- Lyagin, I.; Efremenko, E. Theoretical Evaluation of Suspected Enzymatic Hydrolysis of Novichok Agents. Catal. Commun. 2019, 120, 91–94. [Google Scholar] [CrossRef]
- Bhakhoa, H.; Rhyman, L.; Ramasami, P. Theoretical Study of the Molecular Aspect of the Suspected Novichok Agent A234 of the Skripal Poisoning. R. Soc. Open Sci. 2019, 6, 181831. [Google Scholar] [CrossRef] [Green Version]
- Jeong, K.; Choi, J. Theoretical Study on the Toxicity of ‘Novichok’ Agent Candidates. R. Soc. Open Sci. 2019, 6, 190414. [Google Scholar] [CrossRef] [Green Version]
- Imrit, Y.A.; Bhakhoa, H.; Sergeieva, T.; Danés, S.; Savoo, N.; Elzagheid, M.I.; Rhyman, L.; Andrada, D.M.; Ramasami, P. A Theoretical Study of the Hydrolysis Mechanism of A-234; the Suspected Novichok Agent in the Skripal Attack. RSC Adv. 2020, 10, 27884–27893. [Google Scholar] [CrossRef]
- Harvey, S.P.; McMahon, L.R.; Berg, F.J. Hydrolysis and Enzymatic Degradation of Novichok Nerve Agents. Heliyon 2020, 6. [Google Scholar] [CrossRef]
- Organisation for the Prohibition of Chemical Weapons: OPCW Decision: Changes to Schedule 1 of the Annex on Chemicals to the Chemical Weapons Convention. 2019, C-24/DEC.5. Available online: https://www.opcw.org/sites/default/files/documents/2019/11/c24dec05%28e%29.pdf?fbclid=IwAR0Xn0wlqwxbtUWVWtMuepyGLvsd213k7sO2s3a_HD7buiVHjr3Q46K-wN0 (accessed on 27 December 2019).
- Masson, P.; Lushchekina, S.V. Chapter 72. Catalytic bioscavengers: The second generation of bioscavenger-based medical countermeasures. In Handbook of Toxicology of Chemical Warfare Agents, 3rd ed.; Gupta, R.C., Ed.; Academic Press: Boston, MA, USA, 2020; pp. 1199–1229. ISBN 978-0-12-819090-6. [Google Scholar]
- Poirier, L.; Jacquet, P.; Elias, M.; Daudé, D.; Chabrière, E. Decontamination of organophosphorus compounds: Towards new alternatives. Ann. Pharm Fr. 2017. [Google Scholar] [CrossRef]
- Pulpea, D.; Bunea, M.; Rotariu, T.; Ginghina, R.; TOADER, G. Review of Materials and Technologies Used for Chemical and Radiological Decontamination. J. Mil. Technol. 2019, 2, 43–52. [Google Scholar] [CrossRef]
- Worek, F.; Thiermann, H.; Wille, T. Organophosphorus Compounds and Oximes: A Critical Review. Arch. Toxicol. 2020, 94, 2275–2292. [Google Scholar] [CrossRef] [PubMed]
- Masson, P. Evolution of and Perspectives on Therapeutic Approaches to Nerve Agent Poisoning. Toxicol. Lett. 2011, 206, 5–13. [Google Scholar] [CrossRef] [PubMed]
- Masson, P.; Nachon, F. Cholinesterase Reactivators and Bioscavengers for Pre- and Post-Exposure Treatments of Organophosphorus Poisoning. J. Neurochem. 2017, 142, 26–40. [Google Scholar] [CrossRef] [Green Version]
- Schwartz, M.D.; Hurst, C.G.; Kirk, M.A.; Reedy, S.J.D.; Braue, E.H. Reactpive Skin Decontamination Lotion (RSDL) for the Decontamination of Chemical Warfare Agent (CWA) Dermal Exposure. Curr. Pharm. Biotechnol. 2012, 13, 1971–1979. [Google Scholar] [CrossRef]
- Taysse, L.; Daulon, S.; Delamanche, S.; Bellier, B.; Breton, P. Skin Decontamination of Mustards and Organophosphates: Comparative Efficiency of RSDL and Fuller’s Earth in Domestic Swine. Hum. Exp. Toxicol. 2007, 26, 135–141. [Google Scholar] [CrossRef]
- Thors, L.; Lindberg, S.; Johansson, S.; Koch, B.; Koch, M.; Hägglund, L.; Bucht, A. RSDL Decontamination of Human Skin Contaminated with the Nerve Agent VX. Toxicol. Lett. 2017, 269, 47–54. [Google Scholar] [CrossRef]
- Renaud, P. New Decontamination Protocols in Response to a CBRN Attack; Ouvry—CBRN Protective System: Lyon, France, 2019. [Google Scholar]
- Poirier, L.; Jacquet, P.; Plener, L.; Masson, P.; Daudé, D.; Chabrière, E. Organophosphorus Poisoning in Animals and Enzymatic Antidotes. Environ. Sci. Pollut Res. 2018, 1–26. [Google Scholar] [CrossRef]
- Thakur, M.; Medintz, I.L.; Walper, S.A. Enzymatic Bioremediation of Organophosphate Compounds—Progress and Remaining Challenges. Front. Bioeng. Biotechnol. 2019, 7. [Google Scholar] [CrossRef] [Green Version]
- Goldsmith, M.; Ashani, Y. Catalytic Bioscavengers as Countermeasures against Organophosphate Nerve Agents. Chem. Biol. Interact. 2018, 292, 50–64. [Google Scholar] [CrossRef]
- Manco, G.; Porzio, E.; Suzumoto, Y. Enzymatic Detoxification: A Sustainable Means of Degrading Toxic Organophosphate Pesticides and Chemical Warfare Nerve Agents. J. Chem. Technol. Biotechnol. 2018, 93, 2064–2082. [Google Scholar] [CrossRef]
- Broomfield, C.; M Maxwell, D.; P Solana, R.; Castro, C.; Finger, A.; Lenz, D. Protection by Butyrylcholinesterase against Organophosphorus Poisoning in Nonhuman Primates. J. Pharmacol. Exp. Ther. 1991, 259, 633–638. [Google Scholar]
- Wang, X.; Wu, N.; Guo, J.; Chu, X.; Tian, J.; Yao, B.; Fan, Y. Phytodegradation of Organophosphorus Compounds by Transgenic Plants Expressing a Bacterial Organophosphorus Hydrolase. Biochem. Biophys. Res. Commun. 2008, 365, 453–458. [Google Scholar] [CrossRef]
- Bigley, A.N.; Mabanglo, M.F.; Harvey, S.P.; Raushel, F.M. Variants of Phosphotriesterase for the Enhanced Detoxification of the Chemical Warfare Agent VR. Biochemistry 2015, 54, 5502–5512. [Google Scholar] [CrossRef]
- Oudejans, L. Evaluation Report EPA 600/R-12/033: Enzymatic Decontamination of Chemical Warfare Agents; United States Environmental Protection Agency: Washington, DC, USA, 2013.
- Oudejans, L. Evaluation Report EPA 600/R-13/141: Enzymatic Decontamination of Chemical Warfare Agents Cyclosarin (GF); United States Environmental Protection Agency: Washington, DC, USA, 2013.
- Scott, C. Landguard (TM) A900. An Enzyme-Based Remediant for the Detoxification of Organophosphate Insecticides in Animal Dips. CSIRO Ecosyst. Sci. Canberra Aust. 2012. [Google Scholar] [CrossRef]
- Chabriere, E.; Daude, D.; Elias, M. Novel Mutated Pte Enzymes. Patent: WO 2019/016468 Al, 24 January 2019. [Google Scholar]
- Zueva, I.V.; Lushchekina, S.V.; Daudé, D.; Chabrière, E.; Masson, P. Steady-State Kinetics of Enzyme-Catalyzed Hydrolysis of Echothiophate, a P–S Bonded Organophosphorus as Monitored by Spectrofluorimetry. Molecules 2020, 25, 1371. [Google Scholar] [CrossRef] [Green Version]
- Briseno-Roa, L.; Timperley, C.M.; Griffiths, A.D.; Fersht, A.R. Phosphotriesterase Variants with High Methylphosphonatase Activity and Strong Negative Trade-off against Phosphotriesters. Protein Eng. Des. Sel. 2010, 24, 151–159. [Google Scholar] [CrossRef] [Green Version]
- Briseno-Roa, L.; Oliynyk, Z.; Timperley, C.M.; Griffiths, A.D.; Fersht, A.R. Highest Paraoxonase Turnover Rate Found in a Bacterial Phosphotriesterase Variant. Protein Eng. Des. Sel. 2010, 24, 209–211. [Google Scholar] [CrossRef] [Green Version]
- Bigley, A.N.; Xu, C.; Henderson, T.J.; Harvey, S.P.; Raushel, F.M. Enzymatic Neutralization of the Chemical Warfare Agent VX: Evolution of Phosphotriesterase for Phosphorothiolate Hydrolysis. J. Am. Chem. Soc. 2013, 135, 10426–10432. [Google Scholar] [CrossRef] [Green Version]
- Gäb, J.; John, H.; Melzer, M.; Blum, M.-M. Stable Adducts of Nerve Agents Sarin, Soman and Cyclosarin with TRIS, TES and Related Buffer Compounds—Characterization by LC-ESI-MS/MS and NMR and Implications for Analytical Chemistry. J. Chromatogr. B 2010, 878, 1382–1390. [Google Scholar] [CrossRef] [PubMed]
- Cherny, I.; Greisen, P.; Ashani, Y.; Khare, S.D.; Oberdorfer, G.; Leader, H.; Baker, D.; Tawfik, D.S. Engineering V-Type Nerve Agents Detoxifying Enzymes Using Computationally Focused Libraries. ACS Chem. Biol. 2013, 8, 2394–2403. [Google Scholar] [CrossRef] [PubMed]
- Goldsmith, M.; Ashani, Y.; Simo, Y.; Ben-David, M.; Leader, H.; Silman, I.; Sussman, J.L.; Tawfik, D.S. Evolved Stereoselective Hydrolases for Broad-Spectrum G-Type Nerve Agent Detoxification. Chem. Biol. 2012, 19, 456–466. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ellman, G.L.; Courtney, K.D.; Andres, V.; Featherstone, R.M. A New and Rapid Colorimetric Determination of Acetylcholinesterase Activity. Biochem. Pharmacol. 1961, 7, IN191–IN9095. [Google Scholar] [CrossRef]
| Substrates | PTE Variants | |||||||
|---|---|---|---|---|---|---|---|---|
| GG1 | GG2 | |||||||
| kcat (s−1) | KM (µM) | kcat/KM (s−1∙M−1) | kcat (s−1) | KM (µM) | kcat/KM (s−1∙M−1) | |||
| Insecticides | Ethyl-paraoxon | 251 ± 2 | 13 ± 0.5 | 2.0 ± 0.4 × 107 | 3395 ± 55 | 148 ± 9 | 2.3 ± 0.7 × 107 | |
| Ethyl-parathion | 11 ± 0.2 | 375 ± 27 | 2.9 ± 0.1 × 104 | 29 ± 0.4 | 129 ± 3 | 2.3 ± 0.1 × 105 | ||
| Malathion | 65 ± 11 × 10−2 | 8 ± 2 × 103 | 8.0 ± 0.6 × 101 | 31 ± 8 × 10−2 | 11 ± 4 × 103 | 2.8 ± 0.3 × 101 | ||
| Chlorpyrifos | 16 ± 0.8 × 10−2 | 150 ± 12 | 1.1 ± 0.1 × 103 | 23 ± 3 × 10−2 | 524 ± 68 | 4.5 ± 0.2 × 102 | ||
| G-agent surrogates | CM Soman | Fast a | * | * | 3.1 ± 1.1 × 105 | * | * | 3.1 ± 0.8 × 105 |
| Slow a | * | * | 4.1 ± 4.0 × 104 | * | * | 1.3 ± 0.4 × 104 | ||
| CM Tabun | * | * | 2.4 ± 0.9 × 105 | * | * | 1.1 ± 0.2 × 105 | ||
| CM Sarin | * | * | 8.2 ± 2.4 × 106 | * | * | 3.5 ± 0.4 × 106 | ||
| CM Cyclosarin | * | * | 6.4 ± 2.6 × 105 | * | * | 2.0 ± 1.2 × 106 | ||
| V-agent surrogates | CM VX | * | * | 7.1 ± 1.3 × 106 | * | * | 5.9 ± 1.0 × 106 | |
| DEVX | 262 ± 14 | 4254 ± 215 | 6.2 ± 0.1 × 104 | n.d. | n.d. | n.d. | ||
| Enzyme | Agent (25 mM) | Enzyme Dilution a | Half-Life (min) b | kcat (s−1) c |
|---|---|---|---|---|
| GG1 | VX | 0 | <0.8 | 757 ± 56 |
| 50 | <0.8 | |||
| 100 | 1.6 ± 0.4 | |||
| 200 | 2.7 ± 0.2 | |||
| 400 | 5.1 ± 0.4 | |||
| GA | 200 | 7.0 ± 0.5 | 164 ± 22 | |
| GG2 | GD | 0 | <0.8 | 3014 ± 561 |
| 200 | ≈0.8 | |||
| 400 | 1.2 ± 0.2 | |||
| 1000 | 2.9 ± 0.2 | |||
| GF | 0 | <0.8 | 1633 ± 279 | |
| 1000 | 2.3 ± 0.4 | |||
| GB | 0 | <0.8 | 3778 ± 354 | |
| 1000 | <0.8 | |||
| 3000 | 3.4 ± 0.3 |
| Enzyme(s) | Agent (25 mM) | Half-Life (min) | kcat (s−1) |
|---|---|---|---|
| GG1/GG2 | A230 | <0.8 a | >5.2 |
| GG1/GG2 | A232 | 2.5 ± 0.0 b | 3.7 ± 0.1 |
| GG1/GG2 | A234 | t1/2 fast <0.8 a | >5.2 |
| A234 | t1/2 slow 76 ± 21 b | 0.1 ± 0.0 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jacquet, P.; Rémy, B.; Bross, R.P.T.; van Grol, M.; Gaucher, F.; Chabrière, E.; de Koning, M.C.; Daudé, D. Enzymatic Decontamination of G-Type, V-Type and Novichok Nerve Agents. Int. J. Mol. Sci. 2021, 22, 8152. https://doi.org/10.3390/ijms22158152
Jacquet P, Rémy B, Bross RPT, van Grol M, Gaucher F, Chabrière E, de Koning MC, Daudé D. Enzymatic Decontamination of G-Type, V-Type and Novichok Nerve Agents. International Journal of Molecular Sciences. 2021; 22(15):8152. https://doi.org/10.3390/ijms22158152
Chicago/Turabian StyleJacquet, Pauline, Benjamin Rémy, Rowdy P. T. Bross, Marco van Grol, Floriane Gaucher, Eric Chabrière, Martijn C. de Koning, and David Daudé. 2021. "Enzymatic Decontamination of G-Type, V-Type and Novichok Nerve Agents" International Journal of Molecular Sciences 22, no. 15: 8152. https://doi.org/10.3390/ijms22158152
APA StyleJacquet, P., Rémy, B., Bross, R. P. T., van Grol, M., Gaucher, F., Chabrière, E., de Koning, M. C., & Daudé, D. (2021). Enzymatic Decontamination of G-Type, V-Type and Novichok Nerve Agents. International Journal of Molecular Sciences, 22(15), 8152. https://doi.org/10.3390/ijms22158152