The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease
Abstract
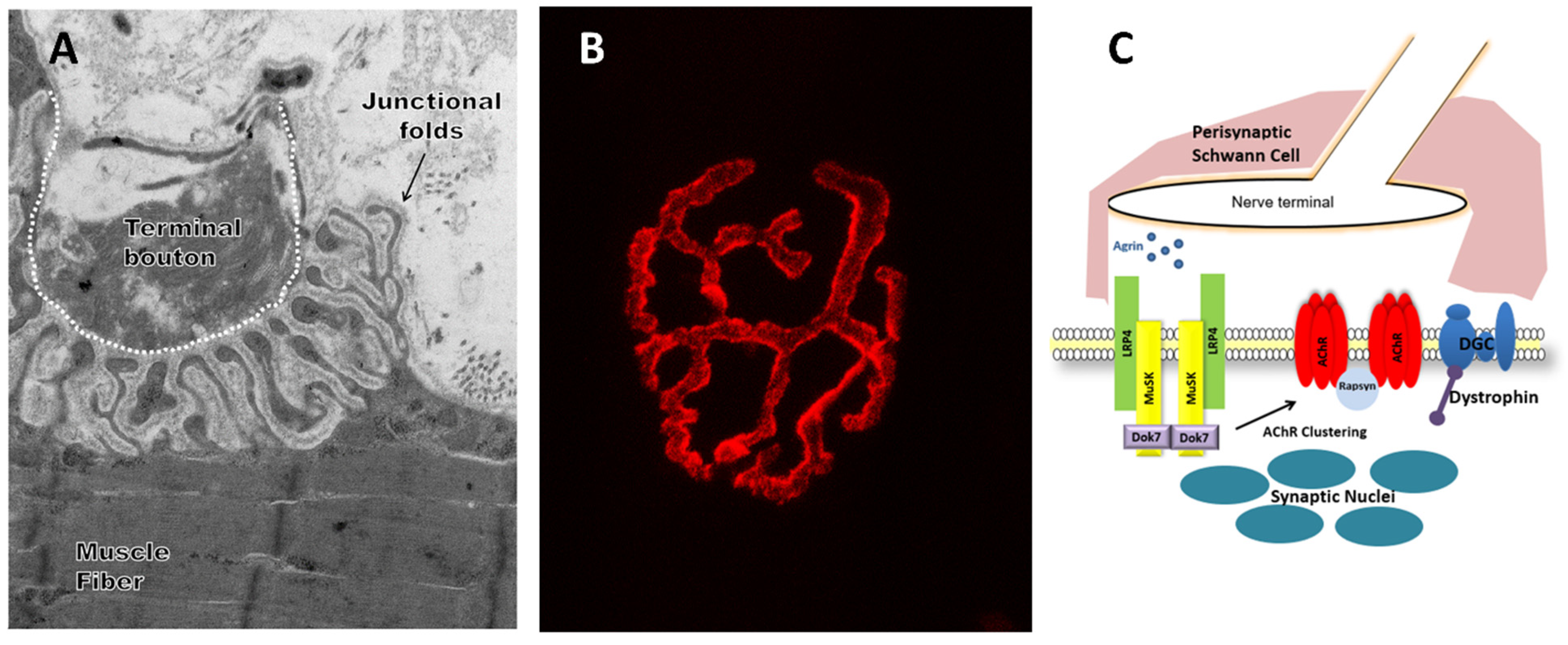
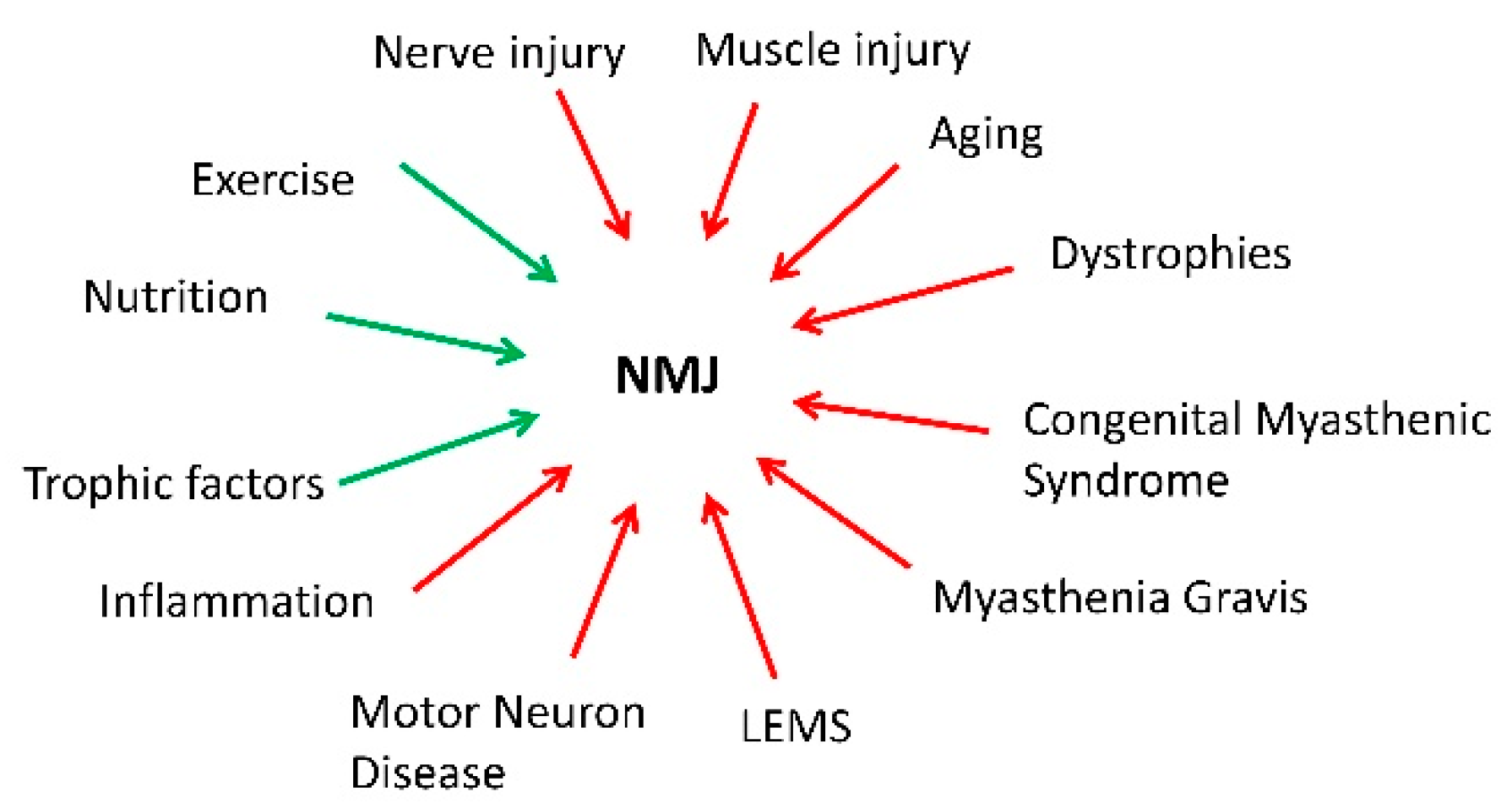
1. Introduction
2. The NMJ in Muscular Dystrophies
3. The NMJ in Aging Muscle
4. Neuromuscular Junction Disorders
5. NMJ in Motor Neuron Diseases
6. Therapeutic Approaches to NMJ Function
7. Exercise
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Wilson, M.H.; Deschenes, M.R. The neuromuscular junction: Anatomical features and adaptations to various forms of increased, or decreased neuromuscular activity 2. Int. J. Neurosci. 2005, 115, 803–828. [Google Scholar] [CrossRef] [PubMed]
- Astrow, S.H.; Qiang, H.; Ko, C.P. Perisynaptic Schwann cells at neuromuscular junctions revealed by a novel monoclonal antibody. J. Neurocytol. 1998, 27, 667–681. [Google Scholar] [CrossRef] [PubMed]
- Fambrough, D.M.; Devreotes, P.N.; Gardner, J.M.; Card, D.J. The life history of acetylcholine receptors 6. Prog. Brain Res. 1979, 49, 325–334. [Google Scholar]
- Fambrough, D.M. Control of acetylcholine receptors in skeletal muscle 7. Physiol. Rev. 1979, 59, 165–227. [Google Scholar] [CrossRef] [PubMed]
- Ferre, J.; Mayayo, E.; Brunet, R. Morphometric study of the neuromuscular synapses in the adult rat with special reference to the remodelling concept 1. Biol.Cell 1987, 60, 133–144. [Google Scholar] [CrossRef] [PubMed]
- Labovitz, S.S.; Robbins, N.; Fahim, M.A. Endplate topography of denervated and disused rat neuromuscular junctions: Comparison by scanning and light microscopy 1. Neuroscience 1984, 11, 963–971. [Google Scholar] [CrossRef]
- Xu, R.; Salpeter, M.M. Acetylcholine receptors in innervated muscles of dystrophic mdx mice degrade as after denervation 2. J. Neurosci. 1997, 17, 8194–8200. [Google Scholar] [CrossRef] [PubMed]
- Reddy, L.V.; Koirala, S.; Sugiura, Y.; Herrera, A.A.; Ko, C.P. Glial cells maintain synaptic structure and function and promote development of the neuromuscular junction in vivo. Neuron 2003, 40, 563–580. [Google Scholar] [CrossRef]
- Cardasis, C.A.; Padykula, H.A. Ultrastructural evidence indicating reorganization at the neuromuscular junction in the normal rat soleus muscle. Anat. Rec. 1981, 200, 41–59. [Google Scholar] [CrossRef]
- Kelly, A.M. Perisynaptic satellite cells in the developing and mature rat soleus muscle. Anat. Rec. 1978, 190, 891–903. [Google Scholar] [CrossRef]
- Fahim, M.A. Endurance exercise modulates neuromuscular junction of C57BL/6NNia aging mice 1. J. Appl. Physiol. 1997, 83, 59–66. [Google Scholar] [CrossRef]
- Prakash, Y.S.; Miyata, H.; Zhan, W.Z.; Sieck, G.C. Inactivity-induced remodeling of neuromuscular junctions in rat diaphragmatic muscle 1. Muscle Nerve 1999, 22, 307–319. [Google Scholar] [CrossRef]
- Prakash, Y.S.; Zhan, W.Z.; Miyata, H.; Sieck, G.C. Adaptations of diaphragm neuromuscular junction following inactivity 4. Acta Anat. 1995, 154, 147–161. [Google Scholar] [CrossRef]
- Sieck, D.C.; Zhan, W.Z.; Fang, Y.H.; Ermilov, L.G.; Sieck, G.C.; Mantilla, C.B. Structure-activity relationships in rodent diaphragm muscle fibers vs. neuromuscular junctions 1. Respir. Physiol. Neurobiol. 2012, 180, 88–96. [Google Scholar] [CrossRef]
- Elkerdany, M.K.; Fahim, M.A. Age changes in neuromuscular junctions of masseter muscle 9. Anat. Rec. 1993, 237, 291–295. [Google Scholar] [CrossRef] [PubMed]
- Jang, Y.C.; Van Remmen, H. Age-associated alterations of the neuromuscular junction 2. Exp. Gerontol. 2011, 46, 193–198. [Google Scholar] [CrossRef]
- Robbins, N. Compensatory plasticity of aging at the neuromuscular junction 10. Exp. Gerontol. 1992, 27, 75–81. [Google Scholar] [CrossRef]
- Santo, N.H.; Martins, A.J.; Minatel, E.; Marques, M.J. Axonal sprouting in mdx mice and its relevance to cell and gene mediated therapies for Duchenne muscular dystrophy 1. Neurosci. Lett. 2003, 343, 67–69. [Google Scholar]
- Kawabuchi, M.; Tan, H.; Wang, S. Age affects reciprocal cellular interactions in neuromuscular synapses following peripheral nerve injury 1. Ageing Res. Rev. 2011, 10, 43–53. [Google Scholar] [CrossRef] [PubMed]
- Sorensen, J.R.; Hoffman, D.B.; Corona, B.T.; Greising, S.M. Secondary denervation is a chronic pathophysiologic sequela of volumetric muscle loss. J. Appl. Physiol. 2021, 130, 1614–1625. [Google Scholar] [CrossRef] [PubMed]
- Adams, M.E.; Kramarcy, N.; Krall, S.P.; Rossi, S.G.; Rotundo, R.L.; Sealock, R.; Froehner, S.C. Absence of alpha-syntrophin leads to structurally aberrant neuromuscular synapses deficient in utrophin. J. Cell Biol. 2000, 150, 1385–1398. [Google Scholar] [CrossRef] [PubMed]
- Banks, G.B.; Chamberlain, J.S.; Froehner, S.C. Truncated dystrophins can influence neuromuscular synapse structure 1. Mol. Cell. Neurosci. 2009, 40, 433–441. [Google Scholar] [CrossRef]
- Chipman, P.H.; Franz, C.K.; Nelson, A.; Schachner, M.; Rafuse, V.F. Neural cell adhesion molecule is required for stability of reinnervated neuromuscular junctions 1. Eur. J. Neurosci. 2010, 31, 238–249. [Google Scholar] [CrossRef]
- Kulakowski, S.A.; Parker, S.D.; Personius, K.E. Reduced TrkB expression results in precocious age-like changes in neuromuscular structure, neurotransmission, and muscle function 1. J. Appl. Physiol. 2011, 111, 844–852. [Google Scholar] [CrossRef] [PubMed]
- Kong, J.; Anderson, J.E. Dystrophin is required for organizing large acetylcholine receptor aggregates 1. Brain Res. 1999, 839, 298–304. [Google Scholar] [CrossRef]
- Wood, S.J.; Slater, C.R. Safety factor at the neuromuscular junction. Prog. Neurobiol. 2001, 64, 393–429. [Google Scholar] [CrossRef]
- Personius, K.E.; Sawyer, R.P. Variability and failure of neurotransmission in the diaphragm of mdx mice. Neuromuscul. Disord. 2006, 16, 168–177. [Google Scholar] [CrossRef] [PubMed]
- Sanes, J.R.; Lichtman, J.W. Development of the vertebrate neuromuscular junction. Annu. Rev. Neurosci. 1999, 22, 389–442. [Google Scholar] [CrossRef] [PubMed]
- Wood, S.J.; Slater, C.R. The contribution of postsynaptic folds to the safety factor for neuromuscular transmission in rat fast- and slow-twitch muscles. J. Physiol. 1997, 500 Pt 1, 165–176. [Google Scholar] [CrossRef]
- Mercuri, E.; Bonnemann, C.G.; Muntoni, F. Muscular dystrophies. Lancet 2019, 394, 2025–2038. [Google Scholar] [CrossRef]
- Belhasan, D.C.; Akaaboune, M. The role of the dystrophin glycoprotein complex on the neuromuscular system. Neurosci. Lett. 2020, 722, 134833. [Google Scholar] [CrossRef] [PubMed]
- Pilgram, G.S.; Potikanond, S.; Baines, R.A.; Fradkin, L.G.; Noordermeer, J.N. The roles of the dystrophin-associated glycoprotein complex at the synapse. Mol. Neurobiol. 2010, 41, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Van der Plas, M.C.; Pilgram, G.S.; Plomp, J.J.; de Jong, A.; Fradkin, L.G.; Noordermeer, J.N. Dystrophin is required for appropriate retrograde control of neurotransmitter release at the Drosophila neuromuscular junction. J. Neurosci. 2006, 26, 333–344. [Google Scholar] [CrossRef]
- Derry, K.L.; Venance, S.L.; Doherty, T.J. Decomposition-based quantitative electromyography in the evaluation of muscular dystrophy severity 1. Muscle Nerve 2012, 45, 507–513. [Google Scholar] [CrossRef]
- Frascarelli, M.; Rocchi, L.; Feola, I. EMG computerized analysis of localized fatigue in Duchenne muscular dystrophy 3. Muscle Nerve 1988, 11, 757–761. [Google Scholar] [CrossRef]
- Priez, A.; Duchene, J.; Goubel, F. Duchenne muscular dystrophy quantification: A multivariate analysis of surface EMG 1. Med. Biol. Eng. Comput. 1992, 30, 283–291. [Google Scholar] [CrossRef]
- Theroux, M.C.; Olivant, A.; Akins, R.E. C Histomorphology of neuromuscular junction in Duchenne muscular dystrophy. Paediatr. Anaesth. 2008, 18, 256–259. [Google Scholar] [CrossRef]
- Hui, T.; Jing, H.; Zhou, T.; Chen, P.; Liu, Z.; Dong, X.; Yan, M.; Ren, D.; Zou, S.; Wang, S.; et al. Increasing LRP4 diminishes neuromuscular deficits in a mouse model of Duchenne muscular dystrophy. Hum. Mol. Genet. 2021, 30, 135. [Google Scholar] [CrossRef]
- Pisani, C.; Strimpakos, G.; Gabanella, F.; Di Certo, M.G.; Onori, A.; Severini, C.; Luvisetto, S.; Farioli-Vecchioli, S.; Carrozzo, I.; Esposito, A.; et al. Utrophin up-regulation by artificial transcription factors induces muscle rescue and impacts the neuromuscular junction in mdx mice. BBA Mol. Basis Dis. 2018, 1864, 1172–1182. [Google Scholar] [CrossRef]
- Van der Pijl, E.M.; van Putten, M.; Niks, E.H.; Verschuuren, J.; Aartsma-Rus, A.; Plomp, J.J. Low dystrophin levels are insufficient to normalize the neuromuscular synaptic abnormalities of mdx mice. Neuromuscul. Disord. 2018, 28, 427–442. [Google Scholar] [CrossRef]
- Shiao, T.; Fond, A.; Deng, B.; Wehling-Henricks, M.; Adams, M.E.; Froehner, S.C.; Tidball, J.G. Defects in neuromuscular junction structure in dy Recovery of altered neuromuscular junction morphology and muscle function in mdx mice after injury strophic muscle are corrected by expression of a NOS transgene in dystrophin-deficient muscles, but not in muscles lacking alpha- and beta1-syntrophins 1. Hum. Mol. Genet. 2004, 13, 1873–1884. [Google Scholar]
- Pratt, S.J.; Shah, S.B.; Ward, C.W.; Inacio, M.P.; Stains, J.P.; Lovering, R.M. Effects of in vivo injury on the neuromuscular junction in healthy and dystrophic muscles 1. J. Physiol. 2013, 591, 559–570. [Google Scholar] [CrossRef] [PubMed]
- Pratt, S.J.; Shah, S.B.; Ward, C.W.; Kerr, J.P.; Stains, J.P.; Lovering, R.M. Recovery of altered neuromuscular junction morphology and muscle function in mdx mice after injury. Cell. Mol. Life Sci. 2015, 72, 153–164. [Google Scholar] [CrossRef]
- Pratt, S.J.; Valencia, A.P.; Le, G.K.; Shah, S.B.; Lovering, R.M. Pre- and postsynaptic changes in the neuromuscular junction in dystrophic mice. Front. Physiol. 2015, 6, 252. [Google Scholar] [CrossRef]
- Ririe, D.G.; Shapiro, F.; Sethna, N.F. The response of patients with Duchenne’s muscular dystrophy to neuromuscular blockade with vecuronium. Anesthesiology 1998, 88, 351–354. [Google Scholar] [CrossRef]
- Ihmsen, H.; Schmidt, J.; Schwilden, H.; Schmitt, H.J.; Muenster, T. Influence of disease progression on the neuromuscular blocking effect of mivacurium in children and adolescents with Duchenne muscular dystrophy. Anesthesiology 2009, 110, 1016–1019. [Google Scholar] [CrossRef] [PubMed]
- Personius, K.E.; Sawyer, R.P. Terminal Schwann cell structure is altered in diaphragm of mdx mice. Muscle Nerve 2005, 32, 656–663. [Google Scholar] [CrossRef] [PubMed]
- Grady, R.M.; Zhou, H.; Cunningham, J.M.; Henry, M.D.; Campbell, K.P.; Sanes, J.R. Maturation and maintenance of the neuromuscular synapse: Genetic evidence for roles of the dystrophin—Glycoprotein complex. Neuron 2000, 25, 279–293. [Google Scholar] [CrossRef]
- Gumerson, J.D.; Davis, C.S.; Kabaeva, Z.T.; Hayes, J.M.; Brooks, S.V.; Michele, D.E. Muscle-specific expression of LARGE restores neuromuscular transmission deficits in dystrophic LARGE(myd) mice. Hum. Mol. Genet. 2013, 22, 757–768. [Google Scholar] [CrossRef]
- Herbst, R.; Iskratsch, T.; Unger, E.; Bittner, R.E. Aberrant development of neuromuscular junctions in glycosylation-defective Large(myd) mice. Neuromuscul. Disord. 2009, 19, 366–378. [Google Scholar] [CrossRef][Green Version]
- Taniguchi, M.; Kurahashi, H.; Noguchi, S.; Fukudome, T.; Okinaga, T.; Tsukahara, T.; Tajima, Y.; Ozono, K.; Nishino, I.; Nonaka, I.; et al. Aberrant neuromuscular junctions and delayed terminal muscle fiber maturation in alpha-dystroglycanopathies. Hum. Mol. Genet. 2006, 15, 1279–1289. [Google Scholar] [CrossRef]
- Masaki, T.; Matsumura, K. Biological role of dystroglycan in Schwann cell function and its implications in peripheral nervous system diseases. J. Biomed. Biotechnol. 2010, 2010, 740403. [Google Scholar] [CrossRef]
- Hack, A.A.; Cordier, L.; Shoturma, D.I.; Lam, M.Y.; Sweeney, H.L.; McNally, E.M. Muscle degeneration without mechanical injury in sarcoglycan deficiency. Proc. Natl. Acad. Sci. USA 1999, 96, 10723–10728. [Google Scholar] [CrossRef] [PubMed]
- Duclos, F.; Straub, V.; Moore, S.A.; Venzke, D.P.; Hrstka, R.F.; Crosbie, R.H.; Durbeej, M.; Lebakken, C.S.; Ettinger, A.J.; van der Meulen, J.; et al. Progressive muscular dystrophy in alpha-sarcoglycan-deficient mice. J. Cell Biol. 1998, 142, 1461–1471. [Google Scholar] [CrossRef]
- Hack, A.A.; Ly, C.T.; Jiang, F.; Clendenin, C.J.; Sigrist, K.S.; Wollmann, R.L.; McNally, E.M. Gamma-sarcoglycan deficiency leads to muscle membrane defects and apoptosis independent of dystrophin. J. Cell Biol. 1998, 142, 1279–1287. [Google Scholar] [CrossRef] [PubMed]
- Rudolf, R.; Khan, M.M.; Labeit, S.; Deschenes, M.R. Degeneration of neuromuscular junction in age and dystrophy. Front. Aging Neurosci. 2014, 6, 99. [Google Scholar] [CrossRef]
- Li, Y.; Lee, Y.; Thompson, W.J. Changes in aging mouse neuromuscular junctions are explained by degeneration and regeneration of muscle fiber segments at the synapse 1. J. Neurosci. 2011, 31, 14910–14919. [Google Scholar] [CrossRef] [PubMed]
- Ferraro, E.; Molinari, F.; Berghella, L. Molecular control of neuromuscular junction development. J. Cachexia Sarcopenia Muscle 2012, 3, 13–23. [Google Scholar] [CrossRef]
- Belotti, E.; Schaeffer, L. Regulation of gene expression at the neuromuscular junction. Neurosci. Lett. 2020, 735, 135163. [Google Scholar] [CrossRef]
- Grady, R.M.; Starr, D.A.; Ackerman, G.L.; Sanes, J.R.; Han, M. Syne proteins anchor muscle nuclei at the neuromuscular junction. Proc. Natl. Acad. Sci. USA 2005, 102, 4359–4364. [Google Scholar] [CrossRef]
- Lei, K.; Zhang, X.; Ding, X.; Guo, X.; Chen, M.; Zhu, B.; Xu, T.; Zhuang, Y.; Xu, R.; Han, M. SUN1 and SUN2 play critical but partially redundant roles in anchoring nuclei in skeletal muscle cells in mice. Proc. Natl. Acad. Sci. USA 2009, 106, 10207–10212. [Google Scholar] [CrossRef] [PubMed]
- Mejat, A.; Decostre, V.; Li, J.; Renou, L.; Kesari, A.; Hantai, D.; Stewart, C.L.; Xiao, X.; Hoffman, E.; Bonne, G.; et al. Lamin A/C-mediated neuromuscular junction defects in emery-dreifuss muscular dystrophy. J. Cell Biol. 2009, 184, 31–44. [Google Scholar] [CrossRef]
- Bonne, G.; Quijano-Roy, S. Emery-Dreifuss muscular dystrophy, laminopathies, and other nuclear envelopathies. Handb. Clin. Neurol. 2013, 113, 1367–1376. [Google Scholar] [CrossRef]
- Tang, L.; Yin, H.J.; Si, C.C.; Hu, X.Y.; Long, Z.H. Determination of benzyl glucosinolate in Lepidium meyenii from different regions by HPLC. China J. Chin. Mater. Med. 2015, 40, 4541–4544. [Google Scholar]
- De Sandre-Giovannoli, A.; Chaouch, M.; Kozlov, S.; Vallat, J.M.; Tazir, M.; Kassouri, N.; Szepetowski, P.; Hammadouche, T.; Vandenberghe, A.; Stewart, C.L.; et al. Homozygous defects in LMNA, encoding lamin A/C nuclear-envelope proteins, cause autosomal recessive axonal neuropathy in human (Charcot-Marie-Tooth disorder type 2) and mouse. Am. J. Hum. Genet. 2002, 70, 726–736. [Google Scholar] [CrossRef] [PubMed]
- Aspnes, L.E.; Lee, C.M.; Weindruch, R.; Chung, S.S.; Roecker, E.B.; Aiken, J.M. Caloric restriction reduces fiber loss and mitochondrial abnormalities in aged rat muscle. FASEB J. 1997, 11, 573–581. [Google Scholar] [CrossRef]
- Brooks, S.V.; Faulkner, J.A. Skeletal muscle weakness in old age: Underlying mechanisms. Med. Sci. Sports Exerc. 1994, 26, 432–439. [Google Scholar] [CrossRef]
- Faulkner, J.A.; Brooks, S.V.; Zerba, E. Muscle atrophy and weakness with aging: Contraction-induced injury as an underlying mechanism. J. Gerontol. A Biol. Sci. Med. Sci. 1995, 50, 124–129. [Google Scholar]
- Henry, M.; Baudry, S. Age-related changes in leg proprioception: Implications for postural control. J. Neurophysiol. 2019, 122, 525–538. [Google Scholar] [CrossRef]
- Johnson, M.E.; Mille, M.L.; Martinez, K.M.; Crombie, G.; Rogers, M.W. Age-related changes in hip abductor and adductor joint torques. Arch. Phys. Med. Rehabil. 2004, 85, 593–597. [Google Scholar] [CrossRef]
- Hilliard, M.J.; Martinez, K.M.; Janssen, I.; Edwards, B.; Mille, M.L.; Zhang, Y.; Rogers, M.W. Lateral balance factors predict future falls in community-living older adults. Arch. Phys. Med. Rehabil. 2008, 89, 1708–1713. [Google Scholar] [CrossRef]
- Alexander, B.H.; Rivara, F.P.; Wolf, M.E. The cost and frequency of hospitalization for fall-related injuries in older adults. Am. J. Public Health 1992, 82, 1020–1023. [Google Scholar] [CrossRef] [PubMed]
- Stevens, J.A.; Corso, P.S.; Finkelstein, E.A.; Miller, T.R. The costs of fatal and non-fatal falls among older adults. Inj. Prev. 2006, 12, 290–295. [Google Scholar] [CrossRef]
- Janssen, I.; Shepard, D.S.; Katzmarzyk, P.T.; Roubenoff, R. The healthcare costs of sarcopenia in the United States. J. Am. Geriatr. Soc. 2004, 52, 80–85. [Google Scholar] [CrossRef]
- Ling, S.M.; Conwit, R.A.; Ferrucci, L.; Metter, E.J. Age-associated changes in motor unit physiology: Observations from the Baltimore Longitudinal Study of Aging. Arch. Phys. Med. Rehabil. 2009, 90, 1237–1240. [Google Scholar] [CrossRef]
- Gao, N.; Zhao, K.; Cao, Y.; Ren, X.; Jing, H.; Xing, G.; Xiong, W.C.; Mei, L. A role of lamin A/C in preventing neuromuscular junction decline in mice. J. Neurosci. 2020, 40, 7203–7215. [Google Scholar] [CrossRef]
- Hughes, D.C.; Marcotte, G.R.; Marshall, A.G.; West, D.W.D.; Baehr, L.M.; Wallace, M.A.; Saleh, P.M.; Bodine, S.C.; Baar, K. Age-related differences in dystrophin: Impact on force transfer proteins, membrane integrity, and neuromuscular junction stability. J. Gerontol. A Biol. Sci. Med. Sci. 2017, 72, 640–648. [Google Scholar] [CrossRef]
- Ramaswamy, K.S.; Palmer, M.L.; van der Meulen, J.H.; Renoux, A.; Kostrominova, T.Y.; Michele, D.E.; Faulkner, J.A. Lateral transmission of force is impaired in skeletal muscles of dystrophic mice and very old rats 1. J. Physiol. 2011, 589, 1195–1208. [Google Scholar] [CrossRef] [PubMed]
- Deschenes, M.R.; Roby, M.A.; Eason, M.K.; Harris, M.B. Remodeling of the neuromuscular junction precedes sarcopenia related alterations in myofibers. Exp. Gerontol. 2010, 45, 389–393. [Google Scholar] [CrossRef]
- Alchin, D.R. Sarcopenia: Describing rather than defining a condition. J. Cachexia Sarcopenia Muscle 2014, 5, 265–268. [Google Scholar] [CrossRef] [PubMed]
- Gonzalez-Freire, M.; de Cabo, R.; Studenski, S.A.; Ferrucci, L. The neuromuscular junction: Aging at the crossroad between nerves and muscle. Front. Aging Neurosci. 2014, 6, 208. [Google Scholar] [CrossRef]
- Garcia, M.L.; Lobsiger, C.S.; Shah, S.B.; Deerinck, T.J.; Crum, J.; Young, D.; Ward, C.M.; Crawford, T.O.; Gotow, T.; Uchiyama, Y.; et al. NF-M is an essential target for the myelin-directed “outside-in” signaling cascade that mediates radial axonal growth. J. Cell Biol. 2003, 163, 1011–1020. [Google Scholar] [CrossRef]
- Marszalek, J.R.; Williamson, T.L.; Lee, M.K.; Xu, Z.; Hoffman, P.N.; Becher, M.W.; Crawford, T.O.; Cleveland, D.W. Neurofilament subunit NF-H modulates axonal diameter by selectively slowing neurofilament transport. J. Cell Biol. 1996, 135, 711–724. [Google Scholar] [CrossRef]
- Punga, A.R.; Ruegg, M.A. Signaling and aging at the neuromuscular synapse: Lessons learnt from neuromuscular diseases 1. Curr. Opin. Pharmacol. 2012, 12, 340–346. [Google Scholar] [CrossRef]
- Shi, L.; Fu, A.K.; Ip, N.Y. Molecular mechanisms underlying maturation and maintenance of the vertebrate neuromuscular junction. Trends Neurosci. 2012, 35, 441–453. [Google Scholar] [CrossRef] [PubMed]
- Deschenes, M.R.; Roby, M.A.; Glass, E.K. Aging influences adaptations of the neuromuscular junction to endurance training. Neuroscience 2011, 190, 56–66. [Google Scholar] [CrossRef] [PubMed]
- Taetzsch, T.; Valdez, G. NMJ maintenance and repair in aging. Curr. Opin. Physiol. 2018, 4, 57–64. [Google Scholar] [CrossRef] [PubMed]
- Wokke, J.H.; Jennekens, F.G.; van den Oord, C.J.; Veldman, H.; Smit, L.M.; Leppink, G.J. Morphological changes in the human end plate with age. J. Neurol. Sci. 1990, 95, 291–310. [Google Scholar] [CrossRef]
- Padilla, C.J.; Harrigan, M.E.; Harris, H.; Schwab, J.M.; Rutkove, S.B.; Rich, M.M.; Clark, B.C.; Arnold, W.D. Profiling age-related muscle weakness and wasting: Neuromuscular junction transmission as a driver of age-related physical decline. Geroscience 2021, 43, 1265–1281. [Google Scholar] [CrossRef] [PubMed]
- Fahim, M.A.; Robbins, N. Ultrastructural studies of young and old mouse neuromuscular junctions. J. Neurocytol. 1982, 11, 641–656. [Google Scholar] [CrossRef]
- Smith, D.O.; Rosenheimer, J.L. Decreased sprouting and degeneration of nerve terminals of active muscles in aged rats. J. Neurophysiol. 1982, 48, 100–109. [Google Scholar] [CrossRef]
- Balice-Gordon, R.J. Age-related changes in neuromuscular innervation. Muscle Nerve Suppl. 1997, 5, 83–87. [Google Scholar] [CrossRef]
- Valdez, G.; Tapia, J.C.; Kang, H.; Clemenson, G.D., Jr.; Gage, F.H.; Lichtman, J.W.; Sanes, J.R. Attenuation of age-related changes in mouse neuromuscular synapses by caloric restriction and exercise. Proc. Natl. Acad. Sci. USA 2010, 107, 14863–14868. [Google Scholar] [CrossRef]
- Kurokawa, K.; Mimori, Y.; Tanaka, E.; Kohriyama, T.; Nakamura, S. Age-related change in peripheral nerve conduction: Compound muscle action potential duration and dispersion. Gerontology 1999, 45, 168–173. [Google Scholar] [CrossRef]
- Rosenheimer, J.L. Factors affecting denervation-like changes at the neuromuscular junction during aging. Int. J. Dev. Neurosci. 1990, 8, 643–654. [Google Scholar] [CrossRef]
- Courtney, J.; Steinbach, J.H. Age changes in neuromuscular junction morphology and acetylcholine receptor distribution on rat skeletal muscle fibres. J. Physiol. 1981, 320, 435–447. [Google Scholar] [CrossRef]
- Smith, D.O.; Chapman, M.R. Acetylcholine receptor binding properties at the rat neuromuscular junction during aging. J. Neurochem. 1987, 48, 1834–1841. [Google Scholar] [CrossRef] [PubMed]
- Kanda, K.; Hashizume, K. Changes in properties of the medial gastrocnemius motor units in aging rats. J. Neurophysiol. 1989, 61, 737–746. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.J.; Zhou, C.J.; Shi, Q.; Smith, N.; Li, T.F. Aging delays the regeneration process following sciatic nerve injury in rats. J. Neurotrauma 2007, 24, 885–894. [Google Scholar] [CrossRef]
- Ruff, R.L.; Lennon, V.A. How myasthenia gravis alters the safety factor for neuromuscular transmission. J. Neuroimmunol. 2008, 201–202, 13–20. [Google Scholar] [CrossRef]
- Maselli, R.A.; Ng, J.J.; Anderson, J.A.; Cagney, O.; Arredondo, J.; Williams, C.; Wessel, H.B.; Abdel-Hamid, H.; Wollmann, R.L. Mutations in LAMB2 causing a severe form of synaptic congenital myasthenic syndrome. J. Med. Genet. 2009, 46, 203–208. [Google Scholar] [CrossRef]
- Nicole, S.; Chaouch, A.; Torbergsen, T.; Bauche, S.; de Bruyckere, E.; Fontenille, M.J.; Horn, M.A.; van Ghelue, M.; Loseth, S.; Issop, Y.; et al. Agrin mutations lead to a congenital myasthenic syndrome with distal muscle weakness and atrophy. Brain 2014, 137, 2429–2443. [Google Scholar] [CrossRef] [PubMed]
- Webster, R.G. Animal models of the neuromuscular junction, vitally informative for understanding function and the molecular mechanisms of congenital myasthenic syndromes. Int. J. Mol. Sci. 2018, 19, 1326. [Google Scholar] [CrossRef] [PubMed]
- Arimura, S.; Okada, T.; Tezuka, T.; Chiyo, T.; Kasahara, Y.; Yoshimura, T.; Motomura, M.; Yoshida, N.; Beeson, D.; Takeda, S.; et al. Neuromuscular disease. DOK7 gene therapy benefits mouse models of diseases characterized by defects in the neuromuscular junction. Science 2014, 345, 1505–1508. [Google Scholar] [CrossRef]
- Croxen, R.; Young, C.; Slater, C.; Haslam, S.; Brydson, M.; Vincent, A.; Beeson, D. End-plate gamma- and epsilon-subunit mRNA levels in AChR deficiency syndrome due to epsilon-subunit null mutations. Brain 2001, 124, 1362–1372. [Google Scholar] [CrossRef] [PubMed]
- Cavalcante, P.; Le Panse, R.; Berrih-Aknin, S.; Maggi, L.; Antozzi, C.; Baggi, F.; Bernasconi, P.; Mantegazza, R. The thymus in myasthenia gravis: Site of innate autoimmunity? Muscle Nerve 2011, 44, 467–484. [Google Scholar] [CrossRef]
- Mantegazza, R.; Cordiglieri, C.; Consonni, A.; Baggi, F. Animal models of myasthenia gravis: Utility and limitations. Int. J. Gen. Med. 2016, 9, 53–64. [Google Scholar] [CrossRef] [PubMed]
- Kusner, L.L.; Losen, M.; Vincent, A.; Lindstrom, J.; Tzartos, S.; Lazaridis, K.; Martinez-Martinez, P. Guidelines for pre-clinical assessment of the acetylcholine receptor—Specific passive transfer myasthenia gravis model-Recommendations for methods and experimental designs. Exp. Neurol. 2015, 270, 3–10. [Google Scholar] [CrossRef]
- Grob, D.; Brunner, N.; Namba, T.; Pagala, M. Lifetime course of myasthenia gravis. Muscle Nerve 2008, 37, 141–149. [Google Scholar] [CrossRef]
- Ghazanfari, N.; Fernandez, K.J.; Murata, Y.; Morsch, M.; Ngo, S.T.; Reddel, S.W.; Noakes, P.G.; Phillips, W.D. Muscle specific kinase: Organiser of synaptic membrane domains 1. Int. J. Biochem. Cell Biol. 2011, 43, 295–298. [Google Scholar] [CrossRef]
- Burden, S.J. SnapShot: Neuromuscular junction 7. Cell 2011, 144, 826. [Google Scholar] [CrossRef]
- Dresser, L.; Wlodarski, R.; Rezania, K.; Soliven, B. Myasthenia gravis: Epidemiology, pathophysiology and clinical manifestations. J. Clin. Med. 2021, 10, 2235. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Li, M.; Wood, K.; Hettwer, S.; Muley, S.A.; Shi, F.D.; Liu, Q.; Ladha, S.S. Engineered agrin attenuates the severity of experimental autoimmune myasthenia gravis. Muscle Nerve 2018, 57, 814–820. [Google Scholar] [CrossRef] [PubMed]
- Morgan, B.P.; Chamberlain-Banoub, J.; Neal, J.W.; Song, W.; Mizuno, M.; Harris, C.L. The membrane attack pathway of complement drives pathology in passively induced experimental autoimmune myasthenia gravis in mice. Clin. Exp. Immunol. 2006, 146, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Guptill, J.T.; Sanders, D.B.; Evoli, A. Anti-MuSK antibody myasthenia gravis: Clinical findings and response to treatment in two large cohorts. Muscle Nerve 2011, 44, 36–40. [Google Scholar] [CrossRef] [PubMed]
- Gilhus, N.E.; Verschuuren, J.J. Myasthenia gravis: Subgroup classification and therapeutic strategies. Lancet Neurol. 2015, 14, 1023–1036. [Google Scholar] [CrossRef]
- Santa, T.; Engel, A.G.; Lambert, E.H. Histometric study of neuromuscular junction ultrastructure. I. Myasthenia gravis. Neurology 1972, 22, 71–82. [Google Scholar] [CrossRef] [PubMed]
- Kusner, L.L.; Sengupta, M.; Kaminski, H.J. Acetylcholine receptor antibody-mediated animal models of myasthenia gravis and the role of complement. Ann. N. Y. Acad. Sci. 2018, 1413, 136–142. [Google Scholar] [CrossRef]
- Titulaer, M.J.; Lang, B.; Verschuuren, J.J. Lambert-Eaton myasthenic syndrome: From clinical characteristics to therapeutic strategies. Lancet Neurol. 2011, 10, 1098–1107. [Google Scholar] [CrossRef]
- Lang, B.; Newsom-Davis, J.; Wray, D.; Vincent, A.; Murray, N. Autoimmune aetiology for myasthenic (Eaton-Lambert) syndrome. Lancet 1981, 2, 224–226. [Google Scholar] [CrossRef]
- Vincent, A. Antibodies and receptors: From neuromuscular junction to central nervous system. Neuroscience 2020, 439, 48–61. [Google Scholar] [CrossRef]
- Oh, S.J.; Claussen, G.G.; Hatanaka, Y.; Morgan, M.B. 3,4-Diaminopyridine is more effective than placebo in a randomized, double-blind, cross-over drug study in LEMS. Muscle Nerve 2009, 40, 795–800. [Google Scholar] [CrossRef]
- Zakharova, M. Modern approaches in gene therapy of motor neuron diseases. Med. Res. Rev. 2020, 40. [Google Scholar] [CrossRef]
- Campanari, M.L.; Bourefis, A.R.; Kabashi, E. Diagnostic challenge and neuromuscular junction contribution to ALS pathogenesis. Front. Neurol. 2019, 10, 68. [Google Scholar] [CrossRef] [PubMed]
- Gromova, A.; La Spada, A.R. Harmony lost: Cell-cell communication at the neuromuscular junction in motor neuron disease. Trends Neurosci. 2020, 43, 709–724. [Google Scholar] [CrossRef]
- Rosen, D.R.; Siddique, T.; Patterson, D.; Figlewicz, D.A.; Sapp, P.; Hentati, A.; Donaldson, D.; Goto, J.; O’Regan, J.P.; Deng, H.X.; et al. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 1993, 362, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Renton, A.E.; Majounie, E.; Waite, A.; Simon-Sanchez, J.; Rollinson, S.; Gibbs, J.R.; Schymick, J.C.; Laaksovirta, H.; van Swieten, J.C.; Myllykangas, L.; et al. A hexanucleotide repeat expansion in C9ORF72 is the cause of chromosome 9p21-linked ALS-FTD. Neuron 2011, 72, 257–268. [Google Scholar] [CrossRef]
- Sreedharan, J.; Blair, I.P.; Tripathi, V.B.; Hu, X.; Vance, C.; Rogelj, B.; Ackerley, S.; Durnall, J.C.; Williams, K.L.; Buratti, E.; et al. TDP-43 mutations in familial and sporadic amyotrophic lateral sclerosis. Science 2008, 319, 1668–1672. [Google Scholar] [CrossRef] [PubMed]
- Kwiatkowski, T.J., Jr.; Bosco, D.A.; Leclerc, A.L.; Tamrazian, E.; Vanderburg, C.R.; Russ, C.; Davis, A.; Gilchrist, J.; Kasarskis, E.J.; Munsat, T.; et al. Mutations in the FUS/TLS gene on chromosome 16 cause familial amyotrophic lateral sclerosis. Science 2009, 323, 1205–1208. [Google Scholar] [CrossRef]
- Eisen, A. The dying forward hypothesis of ALS: Tracing its history. Brain Sci. 2021, 11, 300. [Google Scholar] [CrossRef]
- Yaron, A.; Schuldiner, O. Common and divergent mechanisms in developmental neuronal remodeling and dying back neurodegeneration. Curr. Biol. 2016, 26, 628–639. [Google Scholar] [CrossRef]
- Clark, J.A.; Southam, K.A.; Blizzard, C.A.; King, A.E.; Dickson, T.C. Axonal degeneration, distal collateral branching and neuromuscular junction architecture alterations occur prior to symptom onset in the SOD1(G93A) mouse model of amyotrophic lateral sclerosis. J. Chem. Neuroanat. 2016, 76, 35–47. [Google Scholar] [CrossRef]
- Chand, K.K.; Lee, K.M.; Lee, J.D.; Qiu, H.; Willis, E.F.; Lavidis, N.A.; Hilliard, M.A.; Noakes, P.G. Defects in synaptic transmission at the neuromuscular junction precede motor deficits in a TDP-43(Q331K) transgenic mouse model of amyotrophic lateral sclerosis. FASEB J. 2018, 32, 2676–2689. [Google Scholar] [CrossRef]
- Picchiarelli, G.; Demestre, M.; Zuko, A.; Been, M.; Higelin, J.; Dieterle, S.; Goy, M.A.; Mallik, M.; Sellier, C.; Scekic-Zahirovic, J.; et al. FUS-mediated regulation of acetylcholine receptor transcription at neuromuscular junctions is compromised in amyotrophic lateral sclerosis. Nat. Neurosci. 2019, 22, 1793–1805. [Google Scholar] [CrossRef] [PubMed]
- Murray, L.M.; Comley, L.H.; Thomson, D.; Parkinson, N.; Talbot, K.; Gillingwater, T.H. Selective vulnerability of motor neurons and dissociation of pre- and post-synaptic pathology at the neuromuscular junction in mouse models of spinal muscular atrophy. Hum. Mol. Genet. 2008, 17, 949–962. [Google Scholar] [CrossRef]
- Abraham, S.; Ghonmode, W.N.; Saujanya, K.P.; Jaju, N.; Tambe, V.H.; Yawalikar, P.P. Effect of grape seed extracts on bond strength of bleached enamel using fifth and seventh generation bonding agents. J. Int. Oral Health 2013, 5, 101–107. [Google Scholar] [PubMed]
- Boido, M.; Vercelli, A. Neuromuscular junctions as key contributors and therapeutic targets in spinal muscular atrophy. Front. Neuroanat. 2016, 10, 6. [Google Scholar] [CrossRef] [PubMed]
- Ng, S.Y.; Ljubicic, V. Recent insights into neuromuscular junction biology in Duchenne muscular dystrophy: Impacts, challenges, and opportunities. EBioMedicine 2020, 61, 103032. [Google Scholar] [CrossRef] [PubMed]
- Hesser, B.A.; Henschel, O.; Witzemann, V. Synapse disassembly and formation of new synapses in postnatal muscle upon conditional inactivation of MuSK 1. Mol. Cell. Neurosci. 2006, 31, 470–480. [Google Scholar] [CrossRef] [PubMed]
- Trajanovska, S.; Ban, J.; Huang, J.; Gregorevic, P.; Morsch, M.; Allen, D.G.; Phillips, W.D. Muscle specific kinase protects dystrophic mdx mouse muscles from eccentric contraction-induced loss of force-producing capacity. J. Physiol. 2019, 597, 4831–4850. [Google Scholar] [CrossRef] [PubMed]
- Moll, J.; Barzaghi, P.; Lin, S.; Bezakova, G.; Lochmuller, H.; Engvall, E.; Muller, U.; Ruegg, M.A. An agrin minigene rescues dystrophic symptoms in a mouse model for congenital muscular dystrophy. Nature 2001, 413, 302–307. [Google Scholar] [CrossRef]
- Qiao, C.; Dai, Y.; Nikolova, V.D.; Jin, Q.; Li, J.; Xiao, B.; Li, J.; Moy, S.S.; Xiao, X. Amelioration of muscle and nerve pathology in LAMA2 muscular dystrophy by AAV9-mini-agrin. Mol. Ther. Methods Clin. Dev. 2018, 9, 47–56. [Google Scholar] [CrossRef] [PubMed]
- Qiao, C.; Li, J.; Zhu, T.; Draviam, R.; Watkins, S.; Ye, X.; Chen, C.; Li, J.; Xiao, X. Amelioration of laminin-alpha2-deficient congenital muscular dystrophy by somatic gene transfer of miniagrin. Proc. Natl. Acad. Sci. USA 2005, 102, 11999–12004. [Google Scholar] [CrossRef] [PubMed]
- Bergamin, E.; Hallock, P.T.; Burden, S.J.; Hubbard, S.R. The cytoplasmic adaptor protein Dok7 activates the receptor tyrosine kinase MuSK via dimerization. Mol. Cell 2010, 39, 100–109. [Google Scholar] [CrossRef] [PubMed]
- Zhao, K.; Shen, C.; Li, L.; Wu, H.; Xing, G.; Dong, Z.; Jing, H.; Chen, W.; Zhang, H.; Tan, Z.; et al. Sarcoglycan alpha mitigates neuromuscular junction decline in aged mice by stabilizing LRP4. J. Neurosci. 2018, 38, 8860–8873. [Google Scholar] [CrossRef] [PubMed]
- Ueta, R.; Sugita, S.; Minegishi, Y.; Shimotoyodome, A.; Ota, N.; Ogiso, N.; Eguchi, T.; Yamanashi, Y. DOK7 gene therapy enhances neuromuscular junction innervation and motor function in aged mice. iScience 2020, 23, 101385. [Google Scholar] [CrossRef]
- Hettwer, S.; Lin, S.; Kucsera, S.; Haubitz, M.; Oliveri, F.; Fariello, R.G.; Ruegg, M.A.; Vrijbloed, J.W. Injection of a soluble fragment of neural agrin (NT-1654) considerably improves the muscle pathology caused by the disassembly of the neuromuscular junction. PLoS ONE 2014, 9, e88739. [Google Scholar] [CrossRef]
- Cantor, S.; Zhang, W.; Delestree, N.; Remedio, L.; Mentis, G.Z.; Burden, S.J. Preserving neuromuscular synapses in ALS by stimulating MuSK with a therapeutic agonist antibody. eLife 2018, 7, e34375. [Google Scholar] [CrossRef]
- Boido, M.; De Amicis, E.; Valsecchi, V.; Trevisan, M.; Ala, U.; Ruegg, M.A.; Hettwer, S.; Vercelli, A. Increasing agrin function antagonizes muscle atrophy and motor impairment in spinal muscular atrophy. Front. Cell. Neurosci. 2018, 12, 17. [Google Scholar] [CrossRef]
- Sohal, R.S.; Forster, M.J. Caloric restriction and the aging process: A critique. Free Radic. Biol. Med. 2014, 73, 366–382. [Google Scholar] [CrossRef] [PubMed]
- McKiernan, S.H.; Bua, E.; McGorray, J.; Aiken, J. Early-onset calorie restriction conserves fiber number in aging rat skeletal muscle. FASEB J. 2004, 18, 580–581. [Google Scholar] [CrossRef]
- Lass, A.; Sohal, B.H.; Weindruch, R.; Forster, M.J.; Sohal, R.S. Caloric restriction prevents age-associated accrual of oxidative damage to mouse skeletal muscle mitochondria. Free Radic. Biol. Med. 1998, 25, 1089–1097. [Google Scholar] [CrossRef]
- Lopez-Lluch, G.; Navas, P. Calorie restriction as an intervention in ageing. J. Physiol. 2016, 594, 2043–2060. [Google Scholar] [CrossRef]
- Deschenes, M.R. Motor unit and neuromuscular junction remodeling with aging. Curr. Aging Sci. 2011, 4, 209–220. [Google Scholar] [CrossRef] [PubMed]
- Farmakidis, C.; Pasnoor, M.; Dimachkie, M.M.; Barohn, R.J. Treatment of myasthenia gravis. Neurol. Clin. 2018, 36, 311–337. [Google Scholar] [CrossRef]
- Deschenes, M.R.; Maresh, C.M.; Crivello, J.F.; Armstrong, L.E.; Kraemer, W.J.; Covault, J. The effects of exercise training of different intensities on neuromuscular junction morphology. J. Neurocytol. 1993, 22, 603–615. [Google Scholar] [CrossRef] [PubMed]
- Andonian, M.H.; Fahim, M.A. Effects of endurance exercise on the morphology of mouse neuromuscular junctions during ageing. J. Neurocytol. 1987, 16, 589–599. [Google Scholar] [CrossRef]
- Dorlochter, M.; Irintchev, A.; Brinkers, M.; Wernig, A. Effects of enhanced activity on synaptic transmission in mouse extensor digitorum longus muscle. J. Physiol. 1991, 436, 283–292. [Google Scholar] [CrossRef]
- Porter, M.M.; Vandervoort, A.A.; Lexell, J. Aging of human muscle: Structure, function and adaptability 14. Scand. J. Med. Sci. Sports 1995, 5, 129–142. [Google Scholar] [CrossRef]
- Gillon, A.; Nielsen, K.; Steel, C.; Cornwall, J.; Sheard, P. Exercise attenuates age-associated changes in motoneuron number, nucleocytoplasmic transport proteins and neuromuscular health. Geroscience 2018, 40, 177–192. [Google Scholar] [CrossRef]
- Watt, K.I.; Turner, B.J.; Hagg, A.; Zhang, X.; Davey, J.R.; Qian, H.; Beyer, C.; Winbanks, C.E.; Harvey, K.F.; Gregorevic, P. The hippo pathway effector YAP is a critical regulator of skeletal muscle fibre size. Nat. Commun. 2015, 6, 6048. [Google Scholar] [CrossRef]
- Goodman, C.A.; Dietz, J.M.; Jacobs, B.L.; McNally, R.M.; You, J.S.; Hornberger, T.A. Yes-Associated Protein is up-regulated by mechanical overload and is sufficient to induce skeletal muscle hypertrophy. FEBS Lett. 2015, 589, 1491–1497. [Google Scholar] [CrossRef]
- Watt, K.I.; Goodman, C.A.; Hornberger, T.A.; Gregorevic, P. The hippo signaling pathway in the regulation of skeletal muscle mass and function. Exerc. Sport Sci. Rev. 2018, 46, 92–96. [Google Scholar] [CrossRef]
- Yang, Z.; Nakagawa, K.; Sarkar, A.; Maruyama, J.; Iwasa, H.; Bao, Y.; Ishigami-Yuasa, M.; Ito, S.; Kagechika, H.; Hata, S.; et al. Screening with a novel cell-based assay for TAZ activators identifies a compound that enhances myogenesis in C2C12 cells and facilitates muscle repair in a muscle injury model. Mol. Cell. Biol. 2014, 34, 1607–1621. [Google Scholar] [CrossRef]
- Watt, K.I.; Henstridge, D.C.; Ziemann, M.; Sim, C.B.; Montgomery, M.K.; Samocha-Bonet, D.; Parker, B.L.; Dodd, G.T.; Bond, S.T.; Salmi, T.M.; et al. Yap regulates skeletal muscle fatty acid oxidation and adiposity in metabolic disease. Nat. Commun. 2021, 12, 2887. [Google Scholar] [CrossRef]
- Zhao, K.; Shen, C.; Lu, Y.; Huang, Z.; Li, L.; Rand, C.D.; Pan, J.; Sun, X.D.; Tan, Z.; Wang, H.; et al. Muscle Yap is a regulator of neuromuscular junction formation and regeneration. J. Neurosci. 2017, 37, 3465–3477. [Google Scholar] [CrossRef] [PubMed]
- Judson, R.N.; Gray, S.R.; Walker, C.; Carroll, A.M.; Itzstein, C.; Lionikas, A.; Zammit, P.S.; De, B.C.; Wackerhage, H. Constitutive expression of Yes-associated protein (Yap) in adult skeletal muscle fibres induces muscle atrophy and myopathy. PLoS ONE 2013, 8, e59622. [Google Scholar] [CrossRef] [PubMed]
- Hulmi, J.J.; Oliveira, B.M.; Silvennoinen, M.; Hoogaars, W.M.; Ma, H.; Pierre, P.; Pasternack, A.; Kainulainen, H.; Ritvos, O. Muscle protein synthesis, mTORC1/MAPK/Hippo signaling, and capillary density are altered by blocking of myostatin and activins. Am. J. Physiol. Endocrinol. Metab. 2013, 304, 41–50. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.R.; Shah, S.B.; Ward, C.W.; Stains, J.P.; Spangenburg, E.E.; Folker, E.S.; Lovering, R.M. Differential YAP nuclear signaling in healthy and dystrophic skeletal muscle. Am. J. Physiol. Cell Physiol. 2019, 317, 48–57. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, N.; Endo, J.; Kinouchi, K.; Kitakata, H.; Moriyama, H.; Kataoka, M.; Yamamoto, T.; Shirakawa, K.; Morimoto, S.; Nishiyama, A.; et al. (Pro)renin receptor accelerates development of sarcopenia via activation of Wnt/YAP signaling axis. Aging Cell 2019, 18, 12991. [Google Scholar] [CrossRef] [PubMed]
- Iyer, S.R.; Hsia, R.C.; Folker, E.S.; Lovering, R.M. Age-dependent changes in nuclear-cytoplasmic signaling in skeletal muscle. Exp. Gerontol. 2021, 150, 111338. [Google Scholar] [CrossRef]
- Owens, D.J.; Messeant, J.; Moog, S.; Viggars, M.; Ferry, A.; Mamchaoui, K.; Lacene, E.; Romero, N.; Brull, A.; Bonne, G.; et al. Lamin-related congenital muscular dystrophy alters mechanical signaling and skeletal muscle growth. Int. J. Mol. Sci. 2020, 22, 306. [Google Scholar] [CrossRef] [PubMed]
- Owens, D.J.; Fischer, M.; Jabre, S.; Moog, S.; Mamchaoui, K.; Butler-Browne, G.; Coirault, C. Lamin mutations cause increased Yap nuclear entry in muscle stem cells. Cells 2020, 9, 816. [Google Scholar] [CrossRef] [PubMed]
- Bertrand, A.T.; Ziaei, S.; Ehret, C.; Duchemin, H.; Mamchaoui, K.; Bigot, A.; Mayer, M.; Quijano-Roy, S.; Desguerre, I.; Laine, J.; et al. Cellular microenvironments reveal defective mechanosensing responses and elevated Yap signaling in LMNA-mutated muscle precursors. J. Cell Sci. 2014, 127, 2873–2884. [Google Scholar] [CrossRef]
- Lavin, K.M.; Roberts, B.M.; Fry, C.S.; Moro, T.; Rasmussen, B.B.; Bamman, M.M. The importance of resistance exercise training to combat neuromuscular aging. Physiology 2019, 34, 112–122. [Google Scholar] [CrossRef]
- Grange, R.W.; Call, J.A. Recommendations to define exercise prescription for Duchenne muscular dystrophy 1. Exerc. Sport Sci. Rev. 2007, 35, 12–17. [Google Scholar] [CrossRef] [PubMed]
- Bell, C. Dopamine: Precursor or neurotransmitter in sympathetically innervated tissues? Blood Vessel. 1987, 24, 234–239. [Google Scholar] [CrossRef] [PubMed]
- Spaulding, H.R.; Selsby, J.T. Is exercise the right medicine for dystrophic muscle? Med. Sci. Sports Exerc. 2018, 50, 1723–1732. [Google Scholar] [CrossRef]
- Lott, D.J.; Taivassalo, T.; Cooke, K.D.; Park, H.; Moslemi, Z.; Batra, A.; Forbes, S.C.; Byrne, B.J.; Walter, G.A.; Vandenborne, K. Safety, feasibility, and efficacy of strengthening exercise in Duchenne muscular dystrophy. Muscle Nerve 2021, 63, 320–326. [Google Scholar] [CrossRef]
- Lindsay, A.; Larson, A.A.; Verma, M.; Ervasti, J.M.; Lowe, D.A. Isometric resistance training increases strength and alters histopathology of dystrophin-deficient mouse skeletal muscle. J. Appl. Physiol. 2019, 126, 363–375. [Google Scholar] [CrossRef]
- Petryk, A.; Polgreen, L.E.; Grames, M.; Lowe, D.A.; Hodges, J.S.; Karachunski, P. Feasibility and tolerability of whole-body, low-intensity vibration and its effects on muscle function and bone in patients with dystrophinopathies: A pilot study. Muscle Nerve 2017, 55, 875–883. [Google Scholar] [CrossRef][Green Version]
- Gordon, B.S.; Lowe, D.A.; Kostek, M.C. Exercise increases utrophin protein expression in the mdx mouse model of Duchenne muscular dystrophy. Muscle Nerve 2014, 49, 915–918. [Google Scholar] [CrossRef]
- Tinsley, J.; Deconinck, N.; Fisher, R.; Kahn, D.; Phelps, S.; Gillis, J.M.; Davies, K. Expression of full-length utrophin prevents muscular dystrophy in mdx mice. Nat. Med. 1998, 4, 1441–1444. [Google Scholar] [CrossRef] [PubMed]
- Lovering, R.M.; Iyer, S.R.; Edwards, B.; Davies, K.E. Alterations of neuromuscular junctions in Duchenne muscular dystrophy. Neurosci. Lett. 2020, 737, 135304. [Google Scholar] [CrossRef]
- Preisler, N.; Orngreen, M.C. Exercise in muscle disorders: What is our current state? Curr. Opin. Neurol. 2018, 31, 610–617. [Google Scholar] [CrossRef] [PubMed]
- Sveen, M.L.; Andersen, S.P.; Ingelsrud, L.H.; Blichter, S.; Olsen, N.E.; Jonck, S.; Krag, T.O.; Vissing, J. Resistance training in patients with limb-girdle and becker muscular dystrophies. Muscle Nerve 2013, 47, 163–169. [Google Scholar] [CrossRef]
- Sveen, M.L.; Jeppesen, T.D.; Hauerslev, S.; Krag, T.O.; Vissing, J. Endurance training: An effective and safe treatment for patients with LGMD2I. Neurology 2007, 68, 59–61. [Google Scholar] [CrossRef] [PubMed]
- Orngreen, M.C.; Olsen, D.B.; Vissing, J. Aerobic training in patients with myotonic dystrophy type 1. Ann. Neurol. 2005, 57, 754–757. [Google Scholar] [CrossRef] [PubMed]
- Andersen, G.; Heje, K.; Buch, A.E.; Vissing, J. High-intensity interval training in facioscapulohumeral muscular dystrophy type 1: A randomized clinical trial. J. Neurol. 2017, 264, 1099–1106. [Google Scholar] [CrossRef]
- Olsen, D.B.; Orngreen, M.C.; Vissing, J. Aerobic training improves exercise performance in facioscapulohumeral muscular dystrophy. Neurology 2005, 64, 1064–1066. [Google Scholar] [CrossRef] [PubMed]
- Porcelli, S.; Marzorati, M.; Morandi, L.; Grassi, B. Home-based aerobic exercise training improves skeletal muscle oxidative metabolism in patients with metabolic myopathies. J. Appl. Physiol. 2016, 121, 699–708. [Google Scholar] [CrossRef] [PubMed]
- O’Connor, L.; Westerberg, E.; Punga, A.R. Myasthenia gravis and physical exercise: A novel paradigm. Front. Neurol. 2020, 11, 675. [Google Scholar] [CrossRef]
- Westerberg, E.; Molin, C.J.; Lindblad, I.; Emtner, M.; Punga, A.R. Physical exercise in myasthenia gravis is safe and improves neuromuscular parameters and physical performance-based measures: A pilot study. Muscle Nerve 2017, 56, 207–214. [Google Scholar] [CrossRef]
- Westerberg, E.; Molin, C.J.; Sporndly Nees, S.; Widenfalk, J.; Punga, A.R. The impact of physical exercise on neuromuscular function in Myasthenia gravis patients: A single-subject design study. Medicine 2018, 97, 11510. [Google Scholar] [CrossRef] [PubMed]
- Wong, S.H.; Nitz, J.C.; Williams, K.; Brauer, S.G. Effects of balance strategy training in myasthenia gravis: A case study series. Muscle Nerve 2014, 49, 654–660. [Google Scholar] [CrossRef] [PubMed]
- Lohi, E.L.; Lindberg, C.; Andersen, O. Physical training effects in myasthenia gravis. Arch. Phys. Med. Rehabil. 1993, 74, 1178–1180. [Google Scholar] [PubMed]
- Lucia, A.; Mate-Munoz, J.L.; Perez, M.; Foster, C.; Gutierrez-Rivas, E.; Arenas, J. Double trouble (McArdle’s disease and myasthenia gravis): How can exercise help? Muscle Nerve 2007, 35, 125–128. [Google Scholar] [CrossRef]
- Deschenes, M.R.; Maresh, C.M.; Armstrong, L.E.; Covault, J.; Kraemer, W.J.; Crivello, J.F. Endurance and resistance exercise induce muscle fiber type specific responses in androgen binding capacity. J. Steroid Biochem. Mol. Biol. 1994, 50, 175–179. [Google Scholar] [CrossRef]
- Nishimune, H.; Stanford, J.A.; Mori, Y. Role of exercise in maintaining the integrity of the neuromuscular junction. Muscle Nerve 2014, 49, 315–324. [Google Scholar] [CrossRef]
- Ferraiuolo, L.; De Bono, J.P.; Heath, P.R.; Holden, H.; Kasher, P.; Channon, K.M.; Kirby, J.; Shaw, P.J. Transcriptional response of the neuromuscular system to exercise training and potential implications for ALS. J. Neurochem. 2009, 109, 1714–1724. [Google Scholar] [CrossRef]
- Deschenes, M.R.; Judelson, D.A.; Kraemer, W.J.; Meskaitis, V.J.; Volek, J.S.; Nindl, B.C.; Harman, F.S.; Deaver, D.R. Effects of resistance training on neuromuscular junction morphology. Muscle Nerve 2000, 23, 1576–1581. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Iyer, S.R.; Shah, S.B.; Lovering, R.M. The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease. Int. J. Mol. Sci. 2021, 22, 8058. https://doi.org/10.3390/ijms22158058
Iyer SR, Shah SB, Lovering RM. The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease. International Journal of Molecular Sciences. 2021; 22(15):8058. https://doi.org/10.3390/ijms22158058
Chicago/Turabian StyleIyer, Shama R., Sameer B. Shah, and Richard M. Lovering. 2021. "The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease" International Journal of Molecular Sciences 22, no. 15: 8058. https://doi.org/10.3390/ijms22158058
APA StyleIyer, S. R., Shah, S. B., & Lovering, R. M. (2021). The Neuromuscular Junction: Roles in Aging and Neuromuscular Disease. International Journal of Molecular Sciences, 22(15), 8058. https://doi.org/10.3390/ijms22158058





