The Efficacy of Black Chokeberry Fruits against Cardiovascular Diseases
Abstract
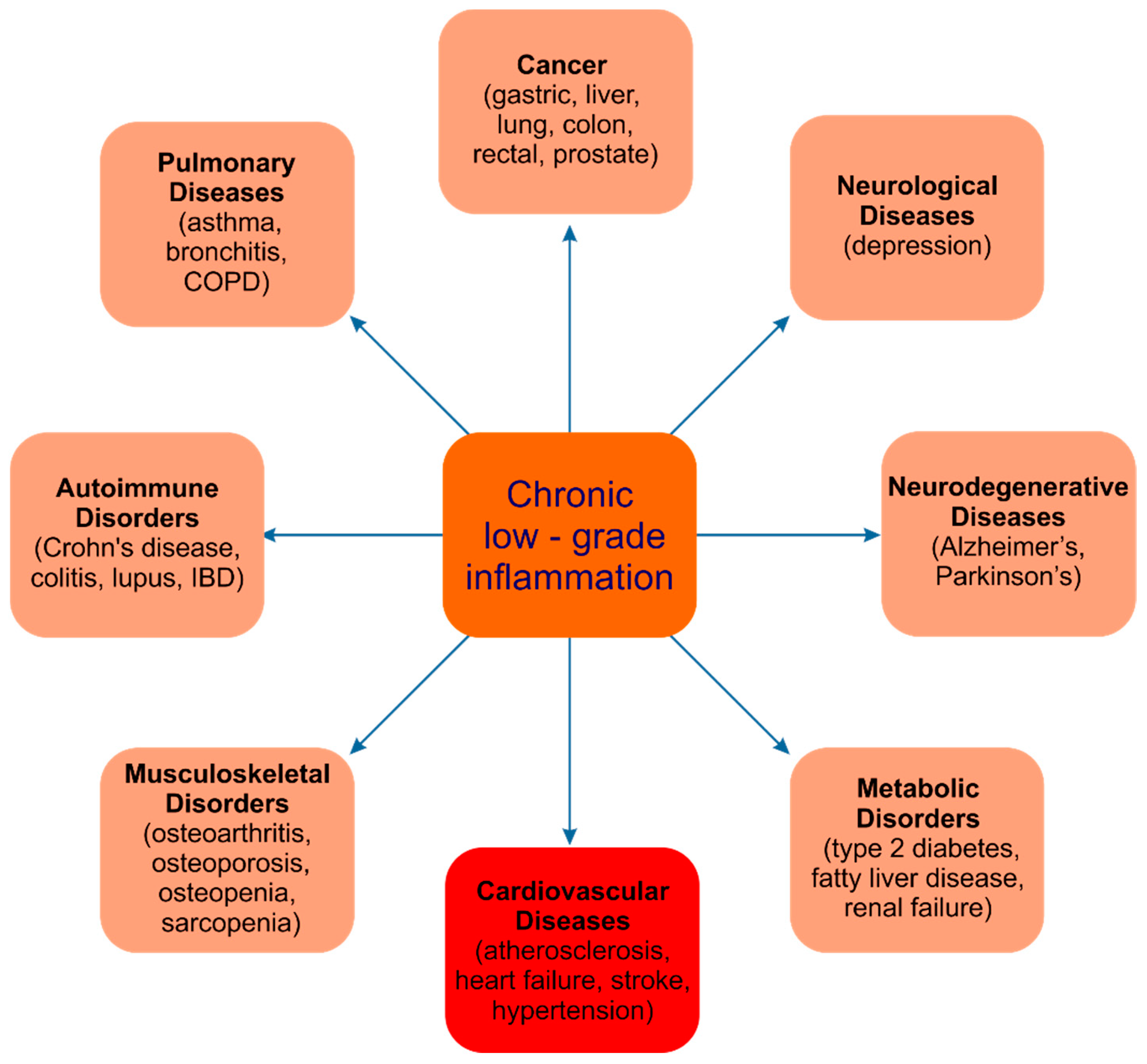
1. Introduction
2. Main Bioactive Compounds of Chokeberry
2.1. Polyphenolic Compounds—Introduction
2.2. Flavonoids
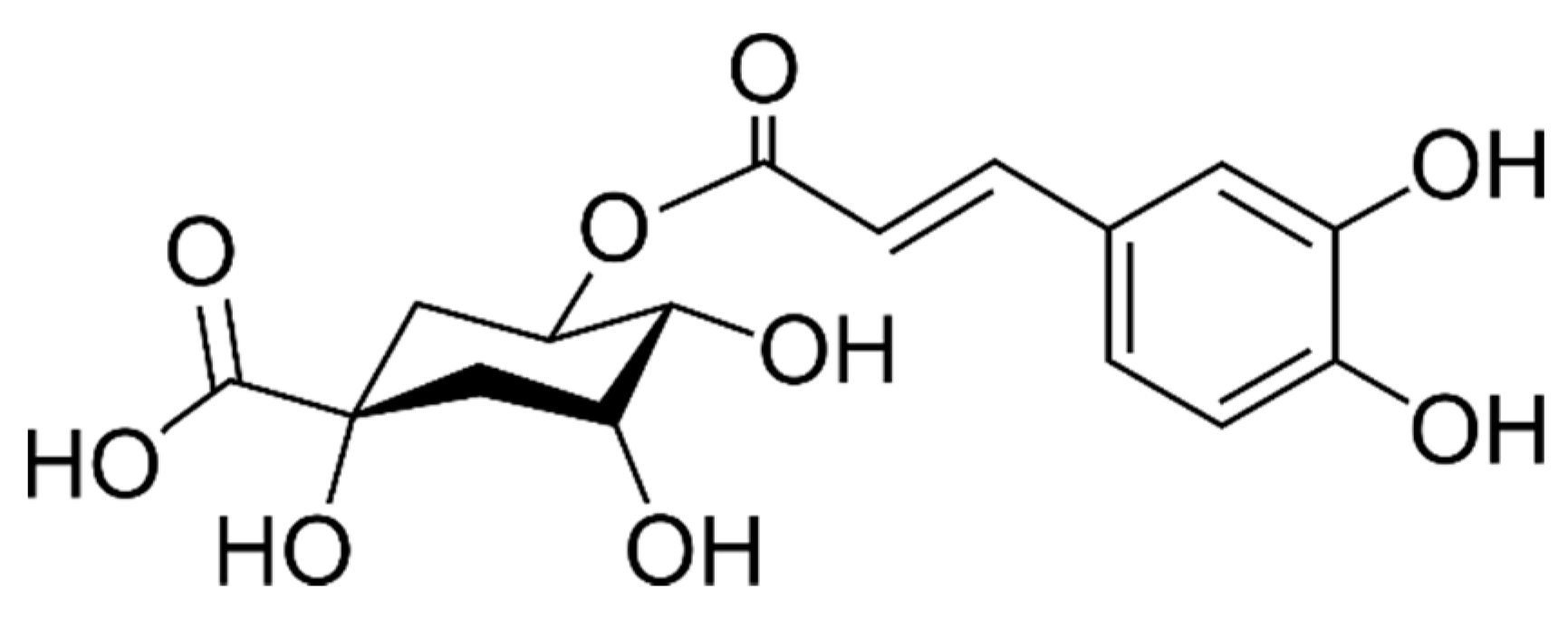
2.3. Phenolic Acids
2.4. Bioavailability of Chokeberry Polyphenols
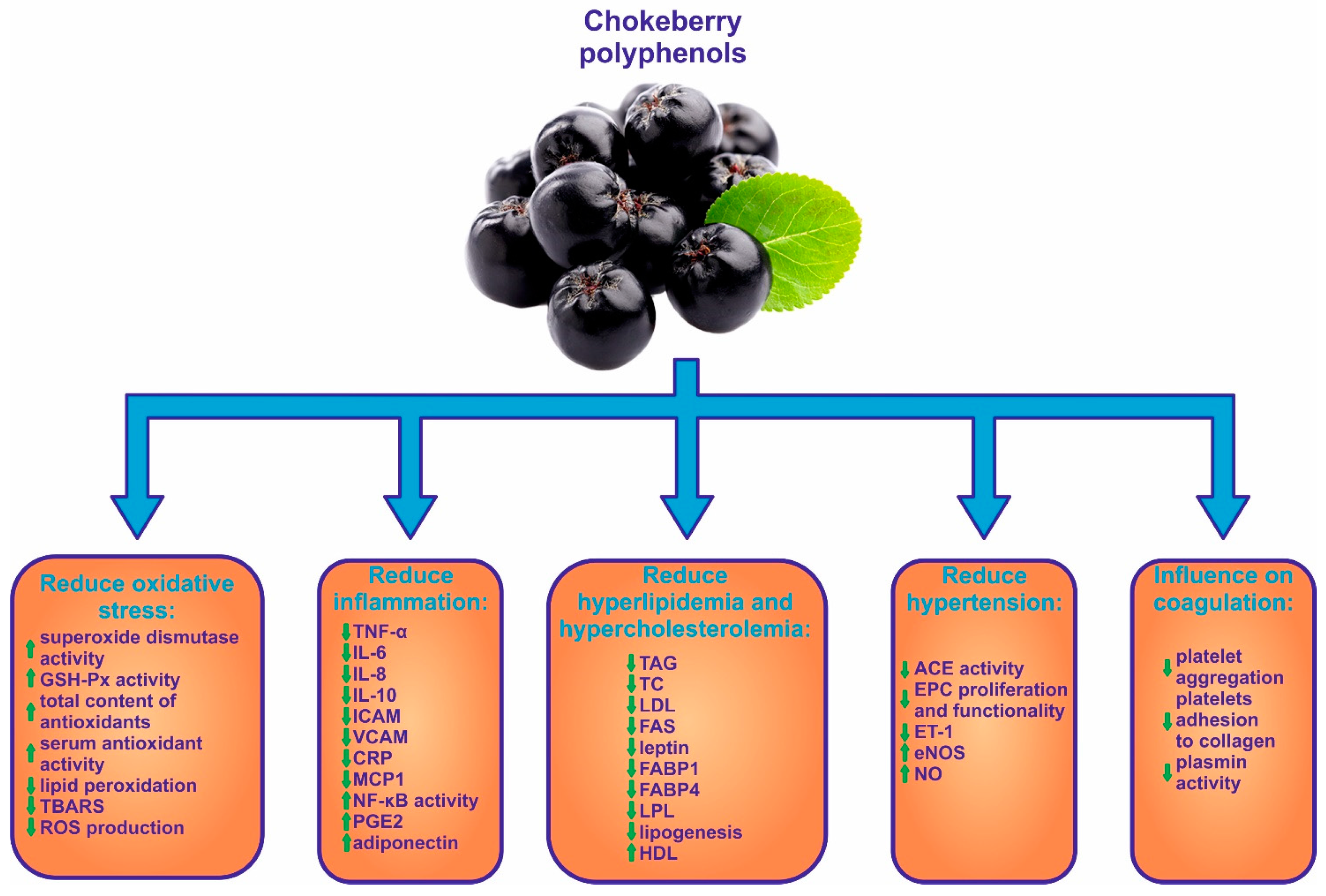
3. Antioxidant Activity of Chokeberry
4. Anti-Inflammatory Effect of Chokeberry Fruits
5. Role of Chokeberry in Hyperlipidemia and Hypercholesterolemia
6. Impact of Chokeberry on Platelet Function
7. Influence of Chokeberry on Hypertension
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Oniszczuk, A.; Oniszczuk, T.; Gancarz, M.; Szymańska, J. Role of Gut Microbiota, Probiotics and Prebiotics in the Cardiovascular Diseases. Molecules 2021, 26, 1172. [Google Scholar] [CrossRef] [PubMed]
- Kasprzak-Drozd, K.; Oniszczuk, T.; Stasiak, M.; Oniszczuk, A. Beneficial Effects of Phenolic Compounds on Gut Microbiota and Metabolic Syndrome. Int. J. Mol. Sci. 2021, 22, 3715. [Google Scholar] [CrossRef]
- Van de Velde, F.; Esposito, D.; Grace, M.H.; Pirovani, M.E.; Lila, M.A. Anti-Inflammatory and Wound Healing Properties of Polyphenolic Extracts from Strawberry and Blackberry Fruits. Food Res. Int. 2019, 121, 453–462. [Google Scholar] [CrossRef] [PubMed]
- Joseph, S.V.; Edirisinghe, I.; Burton-Freeman, B.M. Berries: Anti-Inflammatory Effects in Humans. J. Agric. Food Chem. 2014, 62, 3886–3903. [Google Scholar] [CrossRef] [PubMed]
- Sidor, A.; Drożdżyńska, A.; Gramza-Michałowska, A. Black Chokeberry (Aronia Melanocarpa) and Its Products as Potential Health-Promoting Factors—An Overview. Trends Food Sci. Technol. 2019, 89, 45–60. [Google Scholar] [CrossRef]
- Carneiro, N.V.Q.; Silva, H.B.F.D.; Silva, R.R.D.; Carneiro, T.C.B.; Costa, R.S.; Pires, A.O.; Marques, C.R.; Velozo, E.S.; Conceição, A.S.; Silva, T.M.S.D.; et al. Sambucus Australis Modulates Inflammatory Response via Inhibition of Nuclear Factor Kappa B (NF-KB) In Vitro. An. Acad. Bras. Ciênc. 2019, 91. [Google Scholar] [CrossRef]
- Esposito, D.; Overall, J.; Grace, M.H.; Komarnytsky, S.; Lila, M.A. Alaskan Berry Extracts Promote Dermal Wound Repair through Modulation of Bioenergetics and Integrin Signaling. Front. Pharmacol. 2019, 10. [Google Scholar] [CrossRef]
- Oniszczuk, T.; Widelska, G.; Oniszczuk, A.; Kasprzak, K.; Wójtowicz, A.; Olech, M.; Nowak, R.; Kulesza, K.W.; Jóźwiak, G.; Hajnos, M.W. Influence of Production Parameters on the Content of Polyphenolic Compounds in Extruded Porridge Enriched with Chokeberry Fruit (Aronia Melanocarpa (Michx.) Elliott). Open Chem. 2019, 17, 166–176. [Google Scholar] [CrossRef]
- Zielińska, M.; Gryglicka, H.; Hozyasz, K.K. Aronia czarnoowocowa—Kliniczne perspektywy. Pediatr. Pol. 2013, 88, 452–458. [Google Scholar] [CrossRef]
- Forbes-Hernandez, T.Y.; Gasparrini, M.; Afrin, S.; Bompadre, S.; Mezzetti, B.; Quiles, J.L.; Giampieri, F.; Battino, M. The Healthy Effects of Strawberry Polyphenols: Which Strategy behind Antioxidant Capacity? Crit. Rev. Food Sci. Nutr. 2016, 56 (Suppl. 1), S46–S59. [Google Scholar] [CrossRef]
- Muceniece, R.; Klavins, L.; Kviesis, J.; Jekabsons, K.; Rembergs, R.; Saleniece, K.; Dzirkale, Z.; Saulite, L.; Riekstina, U.; Klavins, M. Antioxidative, Hypoglycaemic and Hepatoprotective Properties of Five Vaccinium Spp. Berry Pomace Extracts. J. Berry Res. 2019, 9, 267–282. [Google Scholar] [CrossRef]
- Olech, M.; Kasprzak, K.; Wójtowicz, A.; Oniszczuk, T.; Nowak, R.; Waksmundzka-Hajnos, M.; Combrzyński, M.; Gancarz, M.; Kowalska, I.; Krajewska, A.; et al. Polyphenol Composition and Antioxidant Potential of Instant Gruels Enriched with Lycium Barbarum L. Fruit. Molecules 2020, 25, 4538. [Google Scholar] [CrossRef]
- Sikora, J.; Broncel, M.; Mikiciuk-Olasik, E. Aronia Melanocarpa Elliot Reduces the Activity of Angiotensin I-Converting Enzyme-in Vitro and Ex Vivo Studies. Oxid. Med. Cell. Longev. 2014, 2014, 739721. [Google Scholar] [CrossRef]
- Zhao, C.-N.; Meng, X.; Li, Y.; Li, S.; Liu, Q.; Tang, G.-Y.; Li, H.-B. Fruits for Prevention and Treatment of Cardiovascular Diseases. Nutrients 2017, 9, 598. [Google Scholar] [CrossRef] [PubMed]
- Sidor, A.; Gramza-Michałowska, A. Black Chokeberry Aronia Melanocarpa L.—A Qualitative Composition, Phenolic Profile and Antioxidant Potential. Molecules 2019, 24, 3710. [Google Scholar] [CrossRef] [PubMed]
- Zdunić, G.; Aradski, A.A.; Gođevac, D.; Živković, J.; Laušević, S.D.; Milošević, D.K.; Šavikin, K. In Vitro Hypoglycemic, Antioxidant and Antineurodegenerative Activity of Chokeberry (Aronia Melanocarpa) Leaves. Ind. Crops Prod. 2020, 148, 112328. [Google Scholar] [CrossRef]
- Denev, P.; Číž, M.; Kratchanova, M.; Blazheva, D. Black Chokeberry (Aronia Melanocarpa) Polyphenols Reveal Different Antioxidant, Antimicrobial and Neutrophil-Modulating Activities. Food Chem. 2019, 284, 108–117. [Google Scholar] [CrossRef]
- Durazzo, A.; Lucarini, M.; Souto, E.B.; Cicala, C.; Caiazzo, E.; Izzo, A.A.; Novellino, E.; Santini, A. Polyphenols: A Concise Overview on the Chemistry, Occurrence, and Human Health. Phytother. Res. 2019, 33, 2221–2243. [Google Scholar] [CrossRef]
- Ku, Y.-S.; Ng, M.-S.; Cheng, S.-S.; Lo, A.W.-Y.; Xiao, Z.; Shin, T.-S.; Chung, G.; Lam, H.-M. Understanding the Composition, Biosynthesis, Accumulation and Transport of Flavonoids in Crops for the Promotion of Crops as Healthy Sources of Flavonoids for Human Consumption. Nutrients 2020, 12, 1717. [Google Scholar] [CrossRef] [PubMed]
- Tong, C.; Shi, F.; Tong, X.; Shi, S.; Ali, I.; Guo, Y. Shining Natural Flavonols in Sensing and Bioimaging. TrAC Trends Anal. Chem. 2021, 137, 116222. [Google Scholar] [CrossRef]
- Liang, Z.; Liang, H.; Guo, Y.; Yang, D. Cyanidin 3-O-Galactoside: A Natural Compound with Multiple Health Benefits. Int. J. Mol. Sci. 2021, 22, 2261. [Google Scholar] [CrossRef]
- Denev, P.N.; Kratchanov, C.G.; Ciz, M.; Lojek, A.; Kratchanova, M.G. Bioavailability and Antioxidant Activity of Black Chokeberry (Aronia Melanocarpa) Polyphenols: In Vitro and in Vivo Evidences and Possible Mechanisms of Action: A Review. Compr. Rev. Food Sci. Food Saf. 2012, 11, 471–489. [Google Scholar] [CrossRef]
- Oszmiański, J.; Wojdylo, A. Aronia Melanocarpa Phenolics and Their Antioxidant Activity. Eur. Food Res. Technol. 2005, 221, 809–813. [Google Scholar] [CrossRef]
- Jakobek, L.; Drenjančević, M.; Jukić, V.; Šeruga, M. Phenolic Acids, Flavonols, Anthocyanins and Antiradical Activity of “Nero”, “Viking”, “Galicianka” and Wild Chokeberries. Sci. Hortic. 2012, 147, 56–63. [Google Scholar] [CrossRef]
- Jurikova, T.; Mlcek, J.; Skrovankova, S.; Sumczynski, D.; Sochor, J.; Hlavacova, I.; Snopek, L.; Orsavova, J. Fruits of Black Chokeberry Aronia Melanocarpa in the Prevention of Chronic Diseases. Molecules 2017, 22, 944. [Google Scholar] [CrossRef]
- Deineka, V.I.; Tretyakov, M.Y.; Oleiniz, E.Y.; Pavlov, A.A.; Deineka, L.A.; Blinova, I.P.; Manokhina, L.A. Determination of Anthocyanins and Chlorogenic Acids in Fruits of Aronia Genus: The Experience of Chemosystematics. Russ. J. Bioorganic Chem. 2020, 46, 1390–1395. [Google Scholar] [CrossRef]
- Kim, B.; Ku, C.S.; Pham, T.X.; Park, Y.; Martin, D.A.; Xie, L.; Taheri, R.; Lee, J.; Bolling, B.W. Aronia Melanocarpa (Chokeberry) Polyphenol–Rich Extract Improves Antioxidant Function and Reduces Total Plasma Cholesterol in Apolipoprotein E Knockout Mice. Nutr. Res. 2013, 33, 406–413. [Google Scholar] [CrossRef]
- Veberic, R.; Slatnar, A.; Bizjak, J.; Stampar, F.; Mikulic-Petkovsek, M. Anthocyanin Composition of Different Wild and Cultivated Berry Species. LWT Food Sci. Technol. 2015, 60, 509–517. [Google Scholar] [CrossRef]
- Cavalcanti, R.N.; Santos, D.T.; Meireles, M.A.A. Non-Thermal Stabilization Mechanisms of Anthocyanins in Model and Food Systems—An Overview. Food Res. Int. 2011, 44, 499–509. [Google Scholar] [CrossRef]
- Wilkes, K.; Howard, L.R.; Brownmiller, C.; Prior, R.L. Changes in Chokeberry (Aronia Melanocarpa L.) Polyphenols during Juice Processing and Storage. J. Agric. Food Chem. 2014, 62, 4018–4025. [Google Scholar] [CrossRef] [PubMed]
- Banach, M.; Wiloch, M.; Zawada, K.; Cyplik, W.; Kujawski, W. Evaluation of Antioxidant and Anti-Inflammatory Activity of Anthocyanin-Rich Water-Soluble Aronia Dry Extracts. Molecules 2020, 25, 4055. [Google Scholar] [CrossRef] [PubMed]
- Mayer-Miebach, E.; Adamiuk, M.; Behsnilian, D. Stability of Chokeberry Bioactive Polyphenols during Juice Processing and Stabilization of a Polyphenol-Rich Material from the By-Product. Agriculture 2012, 2, 244–258. [Google Scholar] [CrossRef]
- Kulling, S.E.; Rawel, H.M. Chokeberry (Aronia Melanocarpa)—A Review on the Characteristic Components and Potential Health Effects. Planta Med. 2008, 74, 1625–1634. [Google Scholar] [CrossRef]
- Dudonné, S.; Dubé, P.; Anhê, F.F.; Pilon, G.; Marette, A.; Lemire, M.; Harris, C.; Dewailly, E.; Desjardins, Y. Comprehensive Analysis of Phenolic Compounds and Abscisic Acid Profiles of Twelve Native Canadian Berries. J. Food Compos. Anal. 2015, 44, 214–224. [Google Scholar] [CrossRef]
- Mattila, P.; Hellström, J.; Törrönen, R. Phenolic Acids in Berries, Fruits, and Beverages. J. Agric. Food Chem. 2006, 54, 7193–7199. [Google Scholar] [CrossRef]
- Ochmian, I.; Oszmiański, J.; Skupień, K. Chemical Composition, Phenolics, and Firmness of Small Black Fruits. J. Appl. Bot. Food Qual. 2009, 83, 64–69. [Google Scholar]
- Häkkinen, S.; Heinonen, M.; Kärenlampi, S.; Mykkänen, H.; Ruuskanen, J.; Törrönen, R. Screening of Selected Flavonoids and Phenolic Acids in 19 Berries. Food Res. Int. 1999, 32, 345–353. [Google Scholar] [CrossRef]
- Bursać Kovačević, D.; Gajdoš Kljusurić, J.; Putnik, P.; Vukušić, T.; Herceg, Z.; Dragović-Uzelac, V. Stability of Polyphenols in Chokeberry Juice Treated with Gas Phase Plasma. Food Chem. 2016, 212, 323–331. [Google Scholar] [CrossRef] [PubMed]
- Yu, W.; Gao, J.; Hao, R.; Yang, J.; Wei, J. Effects of Simulated Digestion on Black Chokeberry (Aronia Melanocarpa (Michx.) Elliot) Anthocyanins and Intestinal Flora. J. Food Sci. Technol. 2020, 58. [Google Scholar] [CrossRef] [PubMed]
- Grgić, J.; Šelo, G.; Planinić, M.; Tišma, M.; Bucić-Kojić, A. Role of the Encapsulation in Bioavailability of Phenolic Compounds. Antioxidants 2020, 9, 923. [Google Scholar] [CrossRef] [PubMed]
- Mihaylova, D.; Desseva, I.; Stoyanova, M.; Petkova, N.; Terzyiska, M.; Lante, A. Impact of In Vitro Gastrointestinal Digestion on the Bioaccessibility of Phytochemical Compounds from Eight Fruit Juices. Molecules 2021, 26, 1187. [Google Scholar] [CrossRef]
- Lee, K.M.; Min, K.; Choi, O.; Kim, K.-Y.; Woo, H.M.; Kim, Y.; Han, S.O.; Um, Y. Electrochemical Detoxification of Phenolic Compounds in Lignocellulosic Hydrolysate for Clostridium Fermentation. Bioresour. Technol. 2015, 187, 228–234. [Google Scholar] [CrossRef]
- Felgines, C.; Texier, O.; Garcin, P.; Besson, C.; Lamaison, J.-L.; Scalbert, A. Tissue Distribution of Anthocyanins in Rats Fed a Blackberry Anthocyanin-Enriched Diet. Mol. Nutr. Food Res. 2009, 53, 1098–1103. [Google Scholar] [CrossRef]
- Kay, C.D.; Mazza, G.; Holub, B.J.; Wang, J. Anthocyanin Metabolites in Human Urine and Serum. Br. J. Nutr. 2004, 91, 933–942. [Google Scholar] [CrossRef] [PubMed]
- Kumar, N.; Goel, N. Phenolic Acids: Natural Versatile Molecules with Promising Therapeutic Applications. Biotechnol. Rep. 2019, 24, e00370. [Google Scholar] [CrossRef]
- Tomas-Barberan, F.; García-Villalba, R.; Quartieri, A.; Raimondi, S.; Amaretti, A.; Leonardi, A.; Rossi, M. In Vitro Transformation of Chlorogenic Acid by Human Gut Microbiota. Mol. Nutr. Food Res. 2014, 58, 1122–1131. [Google Scholar] [CrossRef] [PubMed]
- Raimondi, S.; Anighoro, A.; Quartieri, A.; Amaretti, A.; Tomás-Barberán, F.A.; Rastelli, G.; Rossi, M. Role of Bifidobacteria in the Hydrolysis of Chlorogenic Acid. MicrobiologyOpen 2015, 4, 41–52. [Google Scholar] [CrossRef] [PubMed]
- Desai, K.G.H.; Park, H.J. Recent Developments in Microencapsulation of Food Ingredients. Dry. Technol. 2005, 23, 1361–1394. [Google Scholar] [CrossRef]
- Picot, A.; Lacroix, C. Production of Multiphase Water-Insoluble Microcapsules for Cell Microencapsulation Using an Emulsification/Spray-Drying Technology. J. Food Sci. 2003, 68, 2693–2700. [Google Scholar] [CrossRef]
- Munin, A.; Edwards-Lévy, F. Encapsulation of Natural Polyphenolic Compounds; a Review. Pharmaceutics 2011, 3, 793–829. [Google Scholar] [CrossRef]
- Fang, Z.; Bhandari, B. Encapsulation of Polyphenols—A Review. Trends Food Sci. Technol. 2010, 21, 510–523. [Google Scholar] [CrossRef]
- Niki, E. Assessment of Antioxidant Capacity in Vitro and in Vivo. Free Radic. Biol. Med. 2010, 49, 503–515. [Google Scholar] [CrossRef]
- Apak, R.; Capanoglu, E.; Shahidi, F. Measurement of Antioxidant Activity and Capacity: Recent Trends and Applications; Wiley: Hoboken, NJ, USA, 2018; ISBN 978-1-119-13535-7. [Google Scholar]
- Bartosz, G. Druga Twarz Tlenu: Wolne Rodniki w Przyrodzie; Wydawnictwo Naukowe PWN: Warszawa, Poland, 2019; ISBN 978-83-01-13847-9. [Google Scholar]
- Apak, R. Current Issues in Antioxidant Measurement. J. Agric. Food Chem. 2019, 67, 9187–9202. [Google Scholar] [CrossRef] [PubMed]
- Apak, R.; Özyürek, M.; Güçlü, K.; Çapanoğlu, E. Antioxidant Activity/Capacity Measurement. 1. Classification, Physicochemical Principles, Mechanisms, and Electron Transfer (ET)-Based Assays. J. Agric. Food Chem. 2016, 64, 997–1027. [Google Scholar] [CrossRef] [PubMed]
- Kähkönen, M.P.; Hopia, A.I.; Vuorela, H.J.; Rauha, J.-P.; Pihlaja, K.; Kujala, T.S.; Heinonen, M. Antioxidant Activity of Plant Extracts Containing Phenolic Compounds. J. Agric. Food Chem. 1999, 47, 3954–3962. [Google Scholar] [CrossRef]
- Zheng, W.; Wang, S.Y. Oxygen Radical Absorbing Capacity of Phenolics in Blueberries, Cranberries, Chokeberries, and Lingonberries. J. Agric. Food Chem. 2003, 51, 502–509. [Google Scholar] [CrossRef] [PubMed]
- Tolić, M.-T.; Landeka Jurčević, I.; Panjkota Krbavčić, I.; Marković, K.; Vahčić, N. Phenolic Content, Antioxidant Capacity and Quality of Chokeberry (Aronia Melanocarpa) Products. Food Technol. Biotechnol. 2015, 53, 171–179. [Google Scholar] [CrossRef]
- Denev, P.; Ciz, M.; Ambrozova, G.; Lojek, A.; Yanakieva, I.; Kratchanova, M. Solid-Phase Extraction of Berries’ Anthocyanins and Evaluation of Their Antioxidative Properties. Food Chem. 2010, 123, 1055–1061. [Google Scholar] [CrossRef]
- Espín, J.C.; Soler-Rivas, C.; Wichers, H.J.; García-Viguera, C. Anthocyanin-Based Natural Colorants: A New Source of Antiradical Activity for Foodstuff. J. Agric. Food Chem. 2000, 48, 1588–1592. [Google Scholar] [CrossRef]
- Vinogradova, Y.; Vergun, O.; Grygorieva, O.; Ivanišová, E.; Brindza, J. Comparative Analysis of Antioxidant Activity and Phenolic Compounds in the Fruits of Aronia spp. Potravinarstvo 2020, 14, 393–401. [Google Scholar] [CrossRef]
- Wangensteen, H.; Bräunlich, M.; Nikolic, V.; Malterud, K.; Slimestad, R.; Barsett, H. Anthocyanins, Proanthocyanidins and Total Phenolics in Four Cultivars of Aronia: Antioxidant and Enzyme Inhibitory Effects. J. Funct. Foods 2014, 7. [Google Scholar] [CrossRef]
- Yang, H.; Kim, Y.-J.; Shin, Y. Influence of Ripening Stage and Cultivar on Physicochemical Properties and Antioxidant Compositions of Aronia Grown in South Korea. Foods 2019, 8, 598. [Google Scholar] [CrossRef]
- Gabrielska, J.; Oszmiański, J. Antioxidant Activity of Anthocyanin Glycoside Derivatives Evaluated by the Inhibition of Liposome Oxidation. Z. Für Naturforschung C 2005, 60, 399–407. [Google Scholar] [CrossRef] [PubMed]
- Kowalczyk, E.; Kopff, A.; Niedworok, J.; Kopff, M.; Jankowski, A. Anthocyanins—An Adjunct to Cardiovascular Therapy? Kardiol. Pol. 2002, 57, 332–334. [Google Scholar]
- Kedzierska, M.; Olas, B.; Wachowicz, B.; Glowacki, R.; Bald, E.; Czernek, U.; Szydłowska-Pazera, K.; Potemski, P.; Piekarski, J.; Jeziorski, A. Effects of the Commercial Extract of Aronia on Oxidative Stress in Blood Platelets Isolated from Breast Cancer Patients after the Surgery and Various Phases of the Chemotherapy. Fitoterapia 2012, 83, 310–317. [Google Scholar] [CrossRef]
- Pilaczynska-Szczesniak, L.; Skarpanska-Steinborn, A.; Deskur, E.; Basta, P.; Horoszkiewicz-Hassan, M. The Influence of Chokeberry Juice Supplementation on the Reduction of Oxidative Stress Resulting from an Incremental Rowing Ergometer Exercise. Int. J. Sport Nutr. Exerc. Metab. 2005, 15, 48–58. [Google Scholar] [CrossRef]
- Kardum, N.; Konić-Ristić, A.; Savikin, K.; Spasić, S.; Stefanović, A.; Ivanišević, J.; Miljković, M. Effects of Polyphenol-Rich Chokeberry Juice on Antioxidant/pro-Oxidant Status in Healthy Subjects. J. Med. Food 2014, 17, 869–874. [Google Scholar] [CrossRef]
- Hussain, T.; Tan, B.; Yin, Y.; Blachier, F.; Tossou, M.C.B.; Rahu, N. Oxidative Stress and Inflammation: What Polyphenols Can Do for Us? Oxid. Med. Cell. Longev. 2016, 2016. [Google Scholar] [CrossRef] [PubMed]
- Gasparrini, M.; Forbes-Hernandez, T.Y.; Cianciosi, D.; Quiles, J.L.; Mezzetti, B.; Xiao, J.; Giampieri, F.; Battino, M. The Efficacy of Berries against Lipopolysaccharide-Induced Inflammation: A Review. Trends Food Sci. Technol. 2021. [Google Scholar] [CrossRef]
- Castro, A.M.; Macedo-de la Concha, L.E.; Pantoja-Meléndez, C.A. Low-Grade Inflammation and Its Relation to Obesity and Chronic Degenerative Diseases. Rev. Médica Hosp. Gen. México 2017, 80, 101–105. [Google Scholar] [CrossRef]
- Ho, G.T.T.; Bräunlich, M.; Austarheim, I.; Wangensteen, H.; Malterud, K.E.; Slimestad, R.; Barsett, H. Immunomodulating Activity of Aronia Melanocarpa Polyphenols. Int. J. Mol. Sci. 2014, 15, 11626–11636. [Google Scholar] [CrossRef] [PubMed]
- Appel, K.; Meiser, P.; Millán, E.; Collado, J.A.; Rose, T.; Gras, C.C.; Carle, R.; Muñoz, E. Chokeberry (Aronia Melanocarpa (Michx.) Elliot) Concentrate Inhibits NF-ΚB and Synergizes with Selenium to Inhibit the Release of pro-Inflammatory Mediators in Macrophages. Fitoterapia 2015, 105, 73–82. [Google Scholar] [CrossRef]
- Lee, K.P.; Choi, N.H.; Kim, H.-S.; Ahn, S.; Park, I.-S.; Lee, D.W. Anti-Neuroinflammatory Effects of Ethanolic Extract of Black Chokeberry (Aronia Melanocapa L.) in Lipopolysaccharide-Stimulated BV2 Cells and ICR Mice. Nutr. Res. Pract. 2018, 12, 13–19. [Google Scholar] [CrossRef] [PubMed]
- Borecki, K.; Żuchowski, M.; Siennicka, A.; Adler, G.; Jastrzębska, M. Polyphenol-Rich Extract of Aronia Melanocarpa Inhibits TNF-? Induced Apoptosis in H9c2 Cells. J. Med. Sci. 2016, 85, 185–191. [Google Scholar] [CrossRef]
- Naruszewicz, M.; Laniewska, I.; Millo, B.; Dłuzniewski, M. Combination Therapy of Statin with Flavonoids Rich Extract from Chokeberry Fruits Enhanced Reduction in Cardiovascular Risk Markers in Patients after Myocardial Infraction (MI). Atherosclerosis 2007, 194, e179–e184. [Google Scholar] [CrossRef] [PubMed]
- Qin, B.; Anderson, R.A. An Extract of Chokeberry Attenuates Weight Gain and Modulates Insulin, Adipogenic and Inflammatory Signalling Pathways in Epididymal Adipose Tissue of Rats Fed a Fructose-Rich Diet. Br. J. Nutr. 2012, 108, 581–587. [Google Scholar] [CrossRef]
- Cebova, M.; Klimentova, J.; Janega, P.; Pechanova, O. Effect of Bioactive Compound of Aronia Melanocarpa on Cardiovascular System in Experimental Hypertension. Oxid. Med. Cell. Longev. 2017, 2017. [Google Scholar] [CrossRef]
- Zapolska-Downar, D.; Bryk, D.; Małecki, M.; Hajdukiewicz, K.; Sitkiewicz, D. Aronia Melanocarpa Fruit Extract Exhibits Anti-Inflammatory Activity in Human Aortic Endothelial Cells. Eur. J. Nutr. 2012, 51, 563–572. [Google Scholar] [CrossRef]
- Krga, I.; Monfoulet, L.-E.; Konic-Ristic, A.; Mercier, S.; Glibetic, M.; Morand, C.; Milenkovic, D. Anthocyanins and Their Gut Metabolites Reduce the Adhesion of Monocyte to TNFα-Activated Endothelial Cells at Physiologically Relevant Concentrations. Arch. Biochem. Biophys. 2016, 599, 51–59. [Google Scholar] [CrossRef]
- Bararu, I.; Bǎdescu, L.; Bǎdulescu, O.; Ciocoiu, M.; Bǎdescu, M. Possibilities of Limiting the Inflammatory Syndrome Present in Experimental Diabetes Mellitus by Using Natural Poliphenols. Ann. Rom. Soc. Cell Biol. 2013, 18, 97–108. [Google Scholar]
- Lancrajan, I. Aronia Melanocarpa, a Potential Therapeutic Agent. Stud. Univ. “Vasile Goldiş” Ser. Ştiinţ. Vieţii 2012, 22, 6. [Google Scholar]
- Nowak, D.; Grąbczewska, Z.; Gośliński, M.; Obońska, K.; Dąbrowska, A.; Kubica, J. Effect of Chokeberry Juice Consumption on Antioxidant Capacity, Lipids Profile and Endothelial Function in Healthy People: A Pilot Study. Czech J. Food Sci. 2016, 34, 39–46. [Google Scholar] [CrossRef]
- Kardum, N.; Petrović-Oggiano, G.; Takic, M.; Glibetić, N.; Zec, M.; Debeljak-Martacic, J.; Konić-Ristić, A. Effects of Glucomannan-Enriched, Aronia Juice-Based Supplement on Cellular Antioxidant Enzymes and Membrane Lipid Status in Subjects with Abdominal Obesity. Sci. World J. 2014, 2014, 869250. [Google Scholar] [CrossRef] [PubMed]
- Duchnowicz, P.; Nowicka, A.; Koter-Michalak, M.; Broncel, M. In Vivo Influence of Extract from Aronia Melanocarpa on the Erythrocyte Membranes in Patients with Hypercholesterolemia. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2012, 18, CR569–CR574. [Google Scholar] [CrossRef] [PubMed]
- Lipińska, P.; Jóźwik, A. Hepatoprotective, Hypoglycemic, and Hypolipidemic Effect of Chokeberry Pomace on Polish Merino Lambs. Anim. Biotechnol. 2018, 29, 136–141. [Google Scholar] [CrossRef]
- Park, H.; Liu, Y.; Kim, H.-S.; Shin, J.-H. Chokeberry Attenuates the Expression of Genes Related to de Novo Lipogenesis in the Hepatocytes of Mice with Nonalcoholic Fatty Liver Disease. Nutr. Res. 2016, 36, 57–64. [Google Scholar] [CrossRef]
- Daskalova, E.; Delchev, S.; Peeva, Y.; Vladimirova-Kitova, L.; Kratchanova, M.; Kratchanov, C.; Denev, P. Antiatherogenic and Cardioprotective Effects of Black Chokeberry (Aronia Melanocarpa) Juice in Aging Rats. Evid. Based Complement. Alternat. Med. 2015, 2015, e717439. [Google Scholar] [CrossRef]
- Yamane, T.; Kozuka, M.; Yamamoto, Y.; Nakano, Y.; Nakagaki, T.; Ohkubo, I.; Ariga, H. Effectiveness of Aronia Berries for Reduction of Mild Fibrosis and Gene Expression Analysis in Livers from Mice Fed a High-Fat Diet with Aronia Berries. Funct. Foods Health Dis. 2016, 6, 144–157. [Google Scholar] [CrossRef]
- Ciocoiu, M.; Miron, A.; Badescu, C.; Morosanu, A.; Badulescu, O.; Badescu, M.C. Biochemical and Morphofunctional Aspects of Aronia Melanocarpa Extract Intervention in Experimental Arterial Hypertension. Ann. Rom. Soc. Cell Biol. 2011, 16, 97–102. [Google Scholar]
- Kim, B.; Park, Y.; Wegner, C.J.; Bolling, B.W.; Lee, J. Polyphenol-Rich Black Chokeberry (Aronia Melanocarpa) Extract Regulates the Expression of Genes Critical for Intestinal Cholesterol Flux in Caco-2 Cells. J. Nutr. Biochem. 2013, 24, 1564–1570. [Google Scholar] [CrossRef]
- Tutusaus, A.; de Gregorio, E.; Cucarull, B.; Cristóbal, H.; Aresté, C.; Graupera, I.; Coll, M.; Colell, A.; Gausdal, G.; Lorens, J.B.; et al. A Functional Role of GAS6/TAM in Nonalcoholic Steatohepatitis Progression Implicates AXL as Therapeutic Target. Cell. Mol. Gastroenterol. Hepatol. 2020, 9, 349–368. [Google Scholar] [CrossRef]
- Broncel, M.; Kozirog, M.; Duchnowicz, P.; Koter-Michalak, M.; Sikora, J.; Chojnowska-Jezierska, J. Aronia Melanocarpa Extract Reduces Blood Pressure, Serum Endothelin, Lipid, and Oxidative Stress Marker Levels in Patients with Metabolic Syndrome. Med. Sci. Monit. Int. Med. J. Exp. Clin. Res. 2010, 16, CR28–CR34. [Google Scholar]
- Park, C.-H.; Kim, J.-H.; Lee, E.B.; Hur, W.; Kwon, O.-J.; Park, H.-J.; Yoon, S.K. Aronia Melanocarpa Extract Ameliorates Hepatic Lipid Metabolism through PPARγ2 Downregulation. PLoS ONE 2017, 12, e0169685. [Google Scholar] [CrossRef] [PubMed]
- Bijak, M.; Bobrowski, M.; Borowiecka, M.; Podsędek, A.; Golański, J.; Nowak, P. Anticoagulant Effect of Polyphenols-Rich Extracts from Black Chokeberry and Grape Seeds. Fitoterapia 2011, 82, 811–817. [Google Scholar] [CrossRef]
- Olas, B.; Wachowicz, B.; Nowak, P.; Kedzierska, M.; Tomczak, A.; Stochmal, A.; Oleszek, W.; Jeziorski, A.; Piekarski, J. Studies on Antioxidant Properties of Polyphenol-Rich Extract from Berries of Aronia Melanocarpa in Blood Platelets. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2008, 59, 823–835. [Google Scholar]
- Malinowska, J.; Babicz, K.; Olas, B.; Stochmal, A.; Oleszek, W. Aronia Melanocarpa Extract Suppresses the Biotoxicity of Homocysteine and Its Metabolite on the Hemostatic Activity of Fibrinogen and Plasma. Nutr. Burbank Los Angel. Cty. Calif. 2012, 28, 793–798. [Google Scholar] [CrossRef] [PubMed]
- Malinowska, J.; Oleszek, W.; Stochmal, A.; Olas, B. The Polyphenol-Rich Extracts from Black Chokeberry and Grape Seeds Impair Changes in the Platelet Adhesion and Aggregation Induced by a Model of Hyperhomocysteinemia. Eur. J. Nutr. 2013, 52, 1049–1057. [Google Scholar] [CrossRef] [PubMed]
- Ryszawa, N.; Kawczyńska-Drózdz, A.; Pryjma, J.; Czesnikiewicz-Guzik, M.; Adamek-Guzik, T.; Naruszewicz, M.; Korbut, R.; Guzik, T.J. Effects of Novel Plant Antioxidants on Platelet Superoxide Production and Aggregation in Atherosclerosis. J. Physiol. Pharmacol. Off. J. Pol. Physiol. Soc. 2006, 57, 611–626. [Google Scholar]
- Sikora, J.; Markowicz-Piasecka, M.; Broncel, M.; Mikiciuk-Olasik, E. Extract of Aronia Melanocarpa-Modified Hemostasis: In Vitro Studies. Eur. J. Nutr. 2014, 53, 1493–1502. [Google Scholar] [CrossRef]
- Siragy, H.M.; Carey, R.M. Role of the Intrarenal Renin-Angiotensin-Aldosterone System in Chronic Kidney Disease. Am. J. Nephrol. 2010, 31, 541–550. [Google Scholar] [CrossRef]
- Yamane, T.; Kozuka, M.; Imai, M.; Yamamoto, Y.; Ohkubo, I.; Sakamoto, T.; Nakagaki, T.; Nakano, Y. Reduction of Blood Pressure by Aronia Berries through Inhibition of Angiotensin-Converting Enzyme Activity in the Spontaneously Hypertensive Rat Kidney. Funct. Foods Health Dis. 2017, 7, 280–290. [Google Scholar] [CrossRef]
- Parzonko, A.; Oświt, A.; Bazylko, A.; Naruszewicz, M. Anthocyans-Rich Aronia Melanocarpa Extract Possesses Ability to Protect Endothelial Progenitor Cells against Angiotensin II Induced Dysfunction. Phytomed. Int. J. Phytother. Phytopharm. 2015, 22, 1238–1246. [Google Scholar] [CrossRef]
- Loo, B.-M.; Erlund, I.; Koli, R.; Puukka, P.; Hellström, J.; Wähälä, K.; Mattila, P.; Jula, A. Consumption of Chokeberry (Aronia Mitschurinii) Products Modestly Lowered Blood Pressure and Reduced Low-Grade Inflammation in Patients with Mildly Elevated Blood Pressure. Nutr. Res. 2016, 36, 1222–1230. [Google Scholar] [CrossRef]
- Kardum, N.; Milovanović, B.; Šavikin, K.; Zdunić, G.; Mutavdžin, S.; Gligorijević, T.; Spasić, S. Beneficial Effects of Polyphenol-Rich Chokeberry Juice Consumption on Blood Pressure Level and Lipid Status in Hypertensive Subjects. J. Med. Food 2015, 18, 1231–1238. [Google Scholar] [CrossRef]
- Ciocoiu, M.; Badescu, L.; Miron, A.; Badescu, M. The Involvement of a Polyphenol-Rich Extract of Black Chokeberry in Oxidative Stress on Experimental Arterial Hypertension. Evid. Based Complement. Altern. Med. ECAM 2013, 2013, 912769. [Google Scholar] [CrossRef] [PubMed]
- Hellström, J.K.; Shikov, A.N.; Makarova, M.N.; Pihlanto, A.M.; Pozharitskaya, O.N.; Ryhänen, E.-L.; Kivijärvi, P.; Makarov, V.G.; Mattila, P.H. Blood Pressure-Lowering Properties of Chokeberry (Aronia Mitchurinii, Var. Viking). J. Funct. Foods 2010, 2, 163–169. [Google Scholar] [CrossRef]
- Varela, C.E.; Fromentin, E.; Roller, M.; Villarreal, F.; Ramirez-Sanchez, I. Effects of a Natural Extract of Aronia Melanocarpa Berry on Endothelial Cell Nitric Oxide Production. J. Food Biochem. 2016, 40, 404–410. [Google Scholar] [CrossRef]
- Sikora, J.; Broncel, M.; Markowicz, M.; Chałubiński, M.; Wojdan, K.; Mikiciuk-Olasik, E. Short-Term Supplementation with Aronia Melanocarpa Extract Improves Platelet Aggregation, Clotting, and Fibrinolysis in Patients with Metabolic Syndrome. Eur. J. Nutr. 2012, 51, 549–556. [Google Scholar] [CrossRef] [PubMed]

| Black Chokeberry form and Dose | Study Model | Mechanism of Actions/Effects | Ref. |
|---|---|---|---|
| Alcoholic extract of chokeberry fruits (10%), 30 mL a day for 40 days | Human study: patients with hypercholesterolemia, arterial hypertension and deregulated protein metabolism | TC ↓, LDL ↓, TAG ↓, glucose ↓ | [83] |
| Organic chokeberry juice; 250 mL a day for 3 weeks | Human study: healthy, non-smoking subjects with correct BMI | TAG ↓, serum antioxidant capacity (DPPH) ↑, | [84] |
| Chokeberry supplement prepared from pure juice enriched with 2 g of stable glucomannan fibers; 100 mL a day for 4 weeks | Human study: postmenopausal women with abdominal obesity | BMI ↓, WC ↓, SBP ↓, HDL ↓, GSH-Px ↑; membrane fatty acid profile in erythrocytes: MUFA ↓, 18:1n-9 ↓, n-3 PUFA ↑, 22:6n-3 ↑, n-6/n-3 ↓, unsaturation index ↑ | [85] |
| Chokeberry extract; 100 mg 3 times a day for 2 months | Human study: patients with hypercholesterolemia without pharmacological treatment, healthy individuals as a control group. | erythrocytes: TC ↓, lipid peroxidation ↓, parameter S ↓; beneficial influence on rheological properties of erythrocytes | [86] |
| Chokeberry pomace; diets with the addition of 150/300 g of chokeberry pomace per each kg of the feed mixture. | Animal study: Polish Merino lambs | ALT ↓, AST ↓, CK ↓, GGT ↓, LDH ↓, TAG ↓, glucose ↓, HDL ↑ | [87] |
| Dried chokeberry powder; high fat diet with 0.5% or 1% chokeberry powder for 8 weeks. | Animal study: C57BL/6 J mice with NAFLD induced by high cholesterol, high fat and sucrose diet | TAG ↓, liver weight ↓, abdominal fat ↓, SREBP-1 ↓, ACC ↓, FAS ↓, | [88] |
| Sterilized chokeberry juice; 25 mL a day for 3 months | Animal study: male Wistar rats | BW ↑, BMI ↑, LDL ↓, retarded age-related changes in the aortic wall. | [89] |
| Freeze-dried chokeberry fruits; diet containing 10% freeze-dried fruits for 28 days. | Animal study: C57BL/6JmsSlc male mice fed a high-fat diet | reduced liver fibrosis; total lipids weight ↓; liver: TAG ↓, TC ↑, LDL ↓; serum: TG ↓, LDL ↓; FABP1 ↓, FABP4 ↓. | [90] |
| Black Chokeberry form and Dose | Study Model | Mechanism of Actions/Effects | Ref. |
|---|---|---|---|
| Cold-pressed chokeberry juice and convection oven dried chokeberry powder; 300 mL juice and 3 g powder a day for 8 weeks | Human study: subjects with mildly elevated blood pressure, no regular use of antihypertensive drugs | day DBP ↓, dU-potassium ↓; inflammation markers: IL10 ↓, TNFα ↓, | [105] |
| Organic chokeberry juice; 100 mL a day for 12 weeks | Human study: healthy women | 24 h SBP ↓, parameters oxidative status: TBARS ↓, PAB ↓, TAC ↓, DZOase ↑ | [69] |
| Organic chokeberry juice; 200 mL a day for 4 weeks | Human study: subjects with presence of high normal BP or grade I hypertension, no regular use of antihypertensive drugs | TAG ↓, 24 h SBP ↓, 24 h DBP ↓, awake SBP ↓, awake DBP ↓ | [106] |
| Freeze-dried chokeberry fruits; normal diet containing 10% chokeberry fruits for 28 days. | Animal study: SHR rats | SBP ↓, lung ACE ↑, kidney ACE ↓ | [103] |
| Powdered black chokeberry ethanol extract; 0.050 g/kg every 2 days, for 8 weeks. | Animal study: wistar white rats with induced arterial hypertension | GSH-Px ↑, GSH ↑, TAC ↑, SBP ↓, DBP ↓ | [107] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kasprzak-Drozd, K.; Oniszczuk, T.; Soja, J.; Gancarz, M.; Wojtunik-Kulesza, K.; Markut-Miotła, E.; Oniszczuk, A. The Efficacy of Black Chokeberry Fruits against Cardiovascular Diseases. Int. J. Mol. Sci. 2021, 22, 6541. https://doi.org/10.3390/ijms22126541
Kasprzak-Drozd K, Oniszczuk T, Soja J, Gancarz M, Wojtunik-Kulesza K, Markut-Miotła E, Oniszczuk A. The Efficacy of Black Chokeberry Fruits against Cardiovascular Diseases. International Journal of Molecular Sciences. 2021; 22(12):6541. https://doi.org/10.3390/ijms22126541
Chicago/Turabian StyleKasprzak-Drozd, Kamila, Tomasz Oniszczuk, Jakub Soja, Marek Gancarz, Karolina Wojtunik-Kulesza, Ewa Markut-Miotła, and Anna Oniszczuk. 2021. "The Efficacy of Black Chokeberry Fruits against Cardiovascular Diseases" International Journal of Molecular Sciences 22, no. 12: 6541. https://doi.org/10.3390/ijms22126541
APA StyleKasprzak-Drozd, K., Oniszczuk, T., Soja, J., Gancarz, M., Wojtunik-Kulesza, K., Markut-Miotła, E., & Oniszczuk, A. (2021). The Efficacy of Black Chokeberry Fruits against Cardiovascular Diseases. International Journal of Molecular Sciences, 22(12), 6541. https://doi.org/10.3390/ijms22126541









